Abstract
The Seed Dispersal Syndrome Hypothesis (SDSH) posits that fruit traits predict the main dispersers interacting with plant species. Mammalian dispersers, relying heavily on olfactory cues, are expected to select dull-colored, scented, and larger fruits compared to birds. However, challenges like overabundant seed predators and context-dependency of frugivore-plant interactions complicate SDSH expectations. We studied the Iberian pear, Pyrus bourgaeana, an expected mammal-dispersed tree based on its fruit traits. Extensive camera-trapping data (over 35,000 records) from several tree populations and years revealed visits from seven frugivore groups, with ungulate fruit predators (59–97%) and carnivore seed dispersers (1–20%) most frequent, while birds, lagomorphs, and rodents were infrequent (0–10%). Red deer and wild boar were also the main fruit removers in all sites and years but acted as fruit and seed predators, and thus likely exert conflicting selection pressures to those exerted by seed dispersers. Although, as predicted by the SDSH, most Iberian pear fruits were consumed by large and medium-sized mammals, the traits of Iberian pear fruits likely reflect selection pressures from dispersal vectors in past times. Our results do not challenge the SDHS but do reveal the importance of considering frugivore functional roles for its adequate evaluation.
Subject terms: Community ecology, Ecology
Introduction
Fruit-frugivore interactions are critical for the dynamics, evolution, and conservation of plant communities worldwide1–4. Fruits provide frugivores with nutrients and water essential for their reproductive success and survival4. Plants benefit from the activity of certain frugivores by having their seeds dispersed which increases both local recruitment (i.e., short-distance dispersal) and also gene flow and the (re)colonization of vacant habitats (i.e., long-distance dispersal;5–8. Since fruit traits can influence foraging choices by mutualistic seed dispersers and, consequently, the probability of seed dispersal, specific combinations of fruit traits (e.g., color and size) that favor interactions with particular seed dispersers are known as seed dispersal syndromes9–12. Under the Seed Dispersal Syndrome Hypothesis (SDSH), fruit traits can predict the main type of dispersers with which plant species interact. For instance, avian seed dispersers are predicted to select brightly colored (red, blue and black) and small fleshy fruits because birds have acute color vision and commonly swallow fruits whole. Mammalian seed dispersers should select dull-colored (brown and dark green), scented and larger fruits, on average, compared to the bird syndrome, as mammals do not rely heavily on visual cues to find fruits and can eat fruits piecemeal10,11,13–15. Nonetheless, it is important to correctly differentiate between the set of traits that favor a particular dispersal mechanism (i.e., seed dispersal syndrome) and the vectors that move the seeds in a particular location and time (i.e., actual seed dispersal). Therefore, the presence of any seed dispersal syndrome does not preclude some seed dispersal by mechanisms other than the predicted one16.
Though the SDSH has regained attention and has been valuable in understanding plant-frugivore interactions12,16–18 some important drawbacks persist. Firstly, certain fruit traits (color, size) associated with seed dispersal syndromes are more influenced by shared evolutionary history among plant taxa than by direct selection pressures exerted by dispersers (i.e. phylogenetic constraints;19,20). Secondly, while such fruit traits may attract mutualistic seed dispersers, they may also attract antagonistic frugivores that act as seed predators or pulp feeders21. Antagonistic frugivores can exert selection pressures in opposite direction to that exerted by seed dispersers and thus, disrupt the predicted correlations between fruit traits and seed dispersers22–25. Thirdly, the variability and context-dependency of frugivore-plant interactions26,27 also pose significant challenges to the SDSH. Frugivore assemblages and their seed dispersal effectiveness can vary across different habitats, regions, and seasons even for a given disperser species6,27–29, potentially altering patterns of frugivory and thus the selective pressures expected from dispersal syndromes. Therefore, a comprehensive evaluation of the SDSH requires studies that account for the functional role of frugivores (e.g., seed dispersers vs. seed predators) and are replicated across sites and fruiting seasons.
Furthermore, during the last few decades some management practices such as those related to hunting can extremely alter frugivore assemblages. In many human-altered areas, limiting culling policies, food supplementation, and predator removal often led to overabundant ungulate populations30–33. These increased populations can alter ecosystem functioning in general and plant-seed disperser interactions in particular33–35. Ungulates feed intensively on ripe fruits and can act as either seed/fruit predators21 or seed dispersers36, depending on a combination of plant (e.g., fruit size and shape, seed toughness) and animal traits (e.g., foraging behavior, body size, gut characteristics;27,37). When acting as seed predators, overabundant ungulates have the potential to limit the number of fruits to dispersers and also to exert conflicting and overwhelming selection pressures on fruit traits to those exerted by seed dispersers23. Furthermore, by limiting the number of available fruits, ungulates could influence in the feeding decisions of other frugivores making them less choosy38,39. Surprisingly, despite the marked increase of ungulate populations in many ecosystems34,40, the implications of such novel selection pressures have been largely neglected in evaluations of the SDSH.
In this study, we illustrate the importance of accounting for the functional role of frugivores (e.g., seed dispersers vs. seed predators) in assessing the SDSH. To this end, we focus on the interaction between the Iberian pear, Pyrus bourgaeana Decne (Rosaceae), and its vertebrate fruit consumers in various Mediterranean ecosystems marked by overabundant ungulate populations in southern Spain. The fruits of this tree are notable for their size (2–4 cm in diameter), brownish hue, and potent aroma when ripe. As anticipated by the SDSH13,41,42, numerous mammal species consistently harvest a significant portion of the tree's fruit yield29,43,44. For instance, medium-sized carnivores typically consume the entire fruits, internally transporting the seeds and facilitating their dispersal, while wild ungulates generally ingest whole fruits, grinding most of the seeds they consume, acting thus as seed predators45. Despite prior research on the dispersal ecology of the Iberian pear, there still exists a knowledge gap concerning the consistency of the role of seed dispersers and seed predators over both space and time and the implications of this within the SDSH. Additionally, limited information is available regarding the contribution of smaller fruit consumers such as birds, lagomorphs, and rodents23. To address this gap in our understanding, we integrated a comprehensive dataset that includes both published and unpublished camera-trap observations of frugivore activity across various Iberian pear populations and years. Specifically, we aimed to (i) evaluate the spatial and temporal consistency in the relative importance of different functional groups of frugivores, and (ii) evaluate the prediction derived from the SDSH, that, in most locations and years, seed-disperser mammals (i.e. carnivores) rather than fruit and seed predators (i.e. ungulates) would dominate as the primary consumers of Iberian pear fruits.
Results
Relative importance of frugivore functional groups
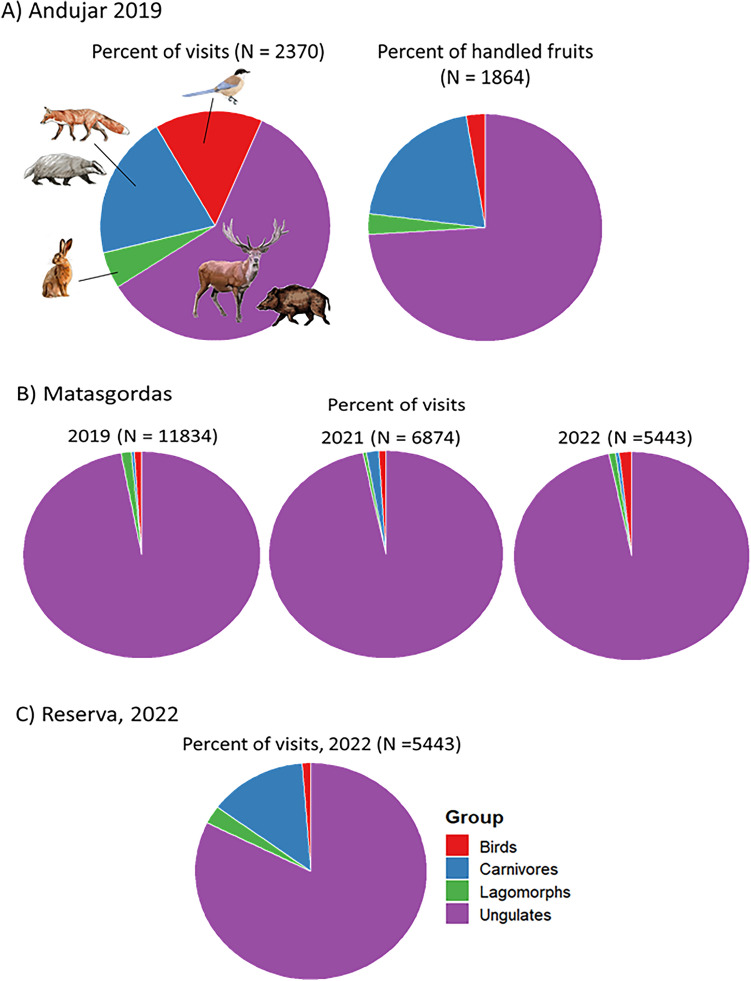
Overall, fruiting trees were visited by the seven functional groups comprising 32 vertebrate species (Table 1). The most frequent visitor were always ungulates (59.2–97.1%) and, in particular, red deer (56.5–83.4%) and wild boar (1.9–30%; Fig. 1). Furthermore, ungulates were very reliable visitors, with either red deer (Andujar) or red deer plus wild boar (Matasgordas and Reserva) visiting all target fruiting trees.
Table 1.
Frequency of visits and number of visited tree by different frugivore species in the three study sites within southern Spain.
| Andujar | Matasgordas | Reserva | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 2019 | 2019 | 2021 | 2022 | 2022 | |||||||
| % visits | # visited trees | % visits | # visited trees | % visits | # visited trees | % visits | # visited trees | % visits | # visited trees | ||
| Role | (n = 2370)a | (n = 30) | (n = 11,834) | (n = 12) | (n = 6873) | (n = 12) | (n = 5443) | (n = 9) | (n = 8829) | (n = 11) | |
| Ungulates | 59.2 | 30 | 97.1 | 12 | 96.5 | 12 | 96.6 | 9 | 82.3 | 11 | |
| Red deer | SP | 56.5 | 30 | 83.4 | 12 | 68.2 | 12 | 66.3 | 9 | 58.3 | 11 |
| Fallow deer | SP | 0.7 | 6 | 0.1 | 4 | 0.1 | 4 | 0.2 | 4 | 0.3 | 4 |
| Wild boar | SP/LD | 1.9 | 21 | 13.6 | 12 | 28.2 | 12 | 30.0 | 9 | 23.8 | 11 |
| Pyrenean ibex | SP | < 0.1 | 1 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| Livestock | 0.0 | 0.0 | 0.1 | 0.1 | 1 | 2.7 | 9 | ||||
| Cow | SP/LD | 0.0 | 0.0 | 0.0 | 0.0 | 0.8 | 9 | ||||
| Horse | SP/LD | 0.0 | 0.0 | 0.1 | 0.1 | 1 | 1.9 | 7 | |||
| Carnivores | 20.2 | 29 | 0.4 | 8 | 1.7 | 11 | 0.5 | 7 | 13.7 | 11 | |
| Red fox | LD | 17.3 | 29 | 0.1 | 5 | 0.5 | 8 | < 0.1 | 2 | 12.9 | 11 |
| Eurasian badger | LD | 2.0 | 12 | 0.3 | 8 | 1.1 | 11 | 0.5 | 5 | 0.5 | 10 |
| Beech marten | LD | 0.6 | 4 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| Genet | LD | 0.0 | 0.0 | 0.0 | 0.0 | 0.4 | 4 | ||||
| Egyptian mongoose | LD | 0.3 | 3 | 0.0 | 0.1 | 4 | < 0.1 | 2 | < 0.1 | 2 | |
| Lagomorphs | 5.1 | 12 | 1.4 | 6 | 0.5 | 8 | 0.9 | 4 | 0.0 | 1 | |
| European rabbit | PF | 0.8 | 7 | 1.4 | 6 | 0.5 | 8 | 0.9 | 4 | < 0.1 | 1 |
| Granada hare | PF | 4.3 | 6 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| Rodents | 0.1 | 3 | 0.0 | 0 | 0.0 | 0 | 0.1 | 1 | 0.0 | ||
| Wood mouse | SP | < 0.1 | 3 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| Rattus spp. | SP | 0.0 | 0.0 | 0.0 | 0.1 | 1 | 0.0 | ||||
| Garden doormouse | SP | < 0.1 | 1 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| Mediterranean pine vole | SP | < 0.1 | 1 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| Corvids | 9.7 | 28 | 0.9 | 10 | 0.9 | 8 | 1.5 | 5 | 0.9 | 8 | |
| Iberian magpie | PF/LD | 9.7 | 28 | 0.8 | 10 | 0.8 | 8 | 1.3 | 5 | 0.0 | |
| Eurasian magpie | PF/LD | 0.0 | 0.1 | 1 | 0.1 | 2 | 0.1 | 2 | 0.9 | 8 | |
| Jaybird | PF/LD | < 0.1 | 1 | 0.0 | 0 | 0.0 | 0.0 | 0.0 | |||
| Other birds | 5.4 | 29 | 0.1 | 5 | 0.2 | 5 | 0.2 | 5 | 0.3 | 10 | |
| European starling | PF/LD | 0.3 | 7 | 0.1 | 3 | 0.1 | 1 | 0.0 | 0.3 | 4 | |
| Woodchat shrike | PF/LD | 0.0 | < 0.1 | 1 | 0.0 | 0.0 | 0.0 | ||||
| Mistle thrush | PF/LD | 1.4 | 15 | < 0.1 | 1 | 0.0 | 0.0 | < 0.1 | 1 | ||
| Song thrush | PF/LD | 0.6 | 7 | 0.0 | 0.1 | 0.0 | 0.0 | ||||
| Wood pigeon | PF/LD | < 0.1 | 1 | 0.0 | 0.0 | 3 | < 0.1 | 1 | 0.0 | ||
| Eurasian collared dove | PF/LD | 0.1 | 1 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| Black bird | PF/LD | 2.8 | 10 | 0.0 | 0.0 | < 0.1 | 2 | < 0.1 | 1 | ||
| Red-legged partridge | PF | 0.2 | 3 | < 0.1 | 1 | < 0.1 | 1 | 0.2 | 5 | < 0.1 | 2 |
| European robin | PF | 0.0 | < 0.1 | 1 | < 0.1 | 1 | < 0.1 | 1 | < 0.1 | 1 | |
| Common chaffinch | PF | 0.0 | 0.0 | 0.0 | < 0.1 | 1 | 0.0 | ||||
| Great tit | PF | 0.0 | 0.0 | 0.0 | 0.0 | < 0.1 | 1 | ||||
| Black redstart | PF | 0.0 | 0.0 | 0.0 | 0.0 | < 0.1 | 2 | ||||
| Bluethroat | PF | 0.0 | 0.0 | 0.0 | 0.0 | < 0.1 | 2 | ||||
| European stonechat | PF | 0.0 | 0.0 | 0.0 | 0.0 | < 0.1 | 1 | ||||
SP = Seed Predator; LP = Legitimate Disperser; PF = Pulp Feeder.
aIt includes also three visits by non-frugivore vertebrates and one unidentified frugivore.
Figure 1.
Frequency of visits and number of visited Pyrus bourgaeana trees by the main four functional groups in the three study sites (Andujar, Matasgordas, Reserva) within southern Spain. The Andujar and Reserva populations were sampled during the fruiting seasons of 2019 and 2022, respectively. The Matasgordas populations was sampled during 2019, 2021 and 2022 fruiting seasons. The percent of handled fruits by the main frugivore functional groups in Andujar are also shown (A).
Despite the pervasive importance of ungulates as visitors, we found significant differences in the proportion of visits by different frugivore groups between study sites pairs (Chi-squared tests, χ2 ≥ 20.3, d.f. = 3, P < 0.001; Fig. 1). Such differences were even larger when we only compared pairs of study sites monitored during the same fruiting season (Andujar-Matasgordas 2019: χ2 = 3661.7, d.f. = 3, P < 0.001; Matasgordas-Reserva 2022: χ2 = 806.1, d.f. = 3, P < 0.001). Carnivores most frequently visited fruiting trees in Andujar (20.2%) and Reserva (15.1%) as compared to Matasgordas (0.4–1.7%; Table 1). Birds (especially corvids) were much more frequent visitors in Andujar (15.1%) than in Matasgordas and Reserva (always < 1.7%). Whereas corvids were relatively reliable visitors, other bird species visited only small subsets of target fruiting trees (Table 1). Lagomorphs were more frequent in Andujar (5.1%) than in Matasgordas and Reserva (0.0–1.4%). Rodents were very infrequent visitors in all study sites (≤ 0.1%; Table 1). Interannual differences only could be tested in Matasgordas. Though the relative frequency of visitation by different groups of frugivores varied annually (χ2 = 143.5, d.f. = 3, P < 0.001), ungulates in Matasgordas accounted for the vast majority of frugivore visits in the three sampled fruiting seasons (≥ 96.5%; Fig. 1B).
Fruit removal and pulp feeding
In Andujar, frugivores were recorded handling about 70% of offered fruits (1887 out of 2700). Ungulates (mostly red deer) were by far the main fruit consumers, removing 1380 out of 1887 removed fruits (73.1%). Although they removed fruits in only 16.6% (ntotal = 1404 ungulate visits) of their visits, those visits in which ungulates consumed the fruits they removed a large number of fruits (5.9 ± 7.9; mean ± 1SD). Carnivores were the second most important fruit removers in Andujar, accounting for 20.1% of total removed fruits (380 out of 1887; Fig. 1A). Most fruits removed by carnivores were due to red foxes (90.8%), followed by Eurasian badgers (8.2%) and then by beech martens (1.0%). Almost a third (142 out of 479) of carnivore visits resulted on fruit removal. On average, carnivores removed 2.7 ± 2.8 fruits per visit. Birds (mostly Iberian magpie and black bird) accounted for only 3.8% of handled fruits (i.e. 72 out of 1887). Such a low number of handled fruits was the result of a small fraction (10.6%) of visits leading to fruit handling (only 38 out of 360) and also of a low number of fruits handled per visit (1.3 ± 0.5). Furthermore, almost a third (23 out of 72) of fruits handled by birds consisted on fruit picking, not removal. Lagomorphs (mostly Iberian hare, but also European rabbit) only harvested about 3% (55 out of 1887) of total removed fruits (Fig. 1A). No rodent fruit removal was recorded.
Though fruit removal was not systematically quantified in Matasgordas and Reserva, our casual camera-trap observations indicated that ungulates were also the main fruit removers in these two tree populations. Specifically, we recorded 83 and 15 instances of fruit removal by red deer in Matasgordas and Reserva, respectively. Fruit removal by wild boar was recorded in five and three instances in Matasgordas and Reserva, respectively. Red foxes in Matasgordas were recorded removing two fruits. In Reserva red foxes were recorded 54 times interacting with fruits, 12 times removing fruit plus 42 times climbing fruiting pear trees. Further, genets in Reserva were photographed 14 times climbing fruiting trees probably in search of fruit. Badger fruit removal was recorded only once in Matasgordas. Iberian magpies in Matasgordas were recorded in 43 instances harvesting fruit, whereas in Reserva Eurasian magpies and European starling were recorded in only seven and one instances, respectively. Furthermore, a large fraction (39.2%, n = 51) of fruit harvesting by birds in Matasgordas and Reserva corresponded to fruit picking rather than fruit removal. No fruit removal by lagomorphs or rodents was recorded in these two populations.
Discussion
Understanding the functional diversity of frugivores and the temporal and spatial variations in the strength of their interactions with fruiting plants is crucial for assessing their potential impact on plant dynamics, potential selection pressures on fruit traits, and also to assess seed dispersal syndromes. To this end, we conducted an ample monitoring of a frugivore assemblage over several years and Iberian pear populations. As predicted by the SDSH, the majority of fruits were consumed by mammals. Intriguingly, however, most fruits were removed by red deer and wild boar that grind most ingested seeds acting thus as fruit and seed predators. These antagonistic fruit predators are likely limiting the availability of fruits to seed dispersers and potentially imposing selection pressures on fruit traits that run counter to those exerted by mutualistic seed dispersers. For example, Fedriani and Delibes23 showed throughout field experiments that both seed dispersers (i.e. carnivores) and fruit predators (i.e., ungulates) preferred large fruits and thus, that they exerted selection pressures on fruit size in opposite directions. Therefore, we propose that ungulate populations, which often act as seed predators and have become overabundant in recent decades31,33,46, are likely disrupting the selection scenarios exerted by seed dispersers in many fleshy-fruited plant communities.
Antagonistic overabundant ungulates as main fruit consumers
Though ungulates were by far the most frequent frugivore visitors in all study sites and years (Fig. 1), their proportion of visits was larger in the two Doñana populations than in Andujar. These differences could relate to differences between study sites in ungulate and/or other frugivore densities43. Another non-exclusive possibility is that ungulates in Andujar are less prone to visit some fruiting pear trees due to their behavioral responses to hunting47 and/or predation risk by Iberian lynx48. Importantly, by lessening tree fitness and limiting the number of seeds that would be dispersed by carnivores, ungulates likely reduced the chance for selection by these mutualistic agents. Besides this, although ungulates visited most individual trees, the rates of fruit predation by ungulates varied enormously among trees. If this variation was based on heritable traits, predation pressure exerted by ungulates on populations of Iberian pear would affect the relative representation of different genotypes in future generations, having thus an additional impact on the selective processes among carnivores and the Iberian pear. Further research is certainly needed to evaluate the potential role of ungulates as selective agents.
The observed predominance of ungulates as Iberian pear fruit consumers likely occurs also in other regions within the tree distribution range (southern Iberian Peninsula and northern Morocco;49), where ungulate population densities are unnaturally high. Such high ungulate densities relate to recent extinctions and population declines of natural predators (i.e., the gray wolf;50,51) and human-managed factors related to hunting practices (e.g., supplemental food, introductions, etc.;30,31,52). Additionally, it is probable that overabundant ungulates play a significant role as fruit and seed predator for many other fleshy fruited36,53,54 and non-fleshy fruited species55–57 across the Iberian Peninsula and elsewhere. Thus, given the global trend towards increasing ungulate populations31,33,46, further research is essential to assess ungulate roles on plant dynamics (herbivory, seed predation, seed dispersal, physical and chemical engineering;36,58).
The contrasting roles of carnivores and smaller frugivores
As reported elsewhere for other large-fruited species59–62, our study unequivocally confirms the importance of medium-sized carnivores, particularly red foxes and badgers (much less than foxes), as seed dispersers of the Iberian pear. This pattern was most evident in Andujar and Reserva populations, where foxes removed significant proportions of the available fruits. In Matasgordas, however, fox occurred in much lower frequencies likely attributable to a sharp decline in their population due to human activities (e.g., illegal poaching, road accidents, habitat use changes;63) and perhaps also to interspecific killing by the locally abundant Iberian lynx29. Although badgers in Matasgordas may partly compensate for the lack of fox seed dispersal, they are unlikely to fully replace them due to interspecific carnivore differences in patterns of habitat use and fecal marking behavior63. Seed dispersal limitation in Matasgordas likely contributes to the low local Iberian pear recruitment and adult aggregated pattern observed8. Indeed, field studies have indicated that this population exhibits very limited reproductive and regenerative capacity, featuring a skewed demographic structure with the majority of individuals in older age classes, few juveniles, and even fewer seedlings and saplings64. Nonetheless, other factors, such as the overabundance of seedling predators (i.e., ungulates) and summer droughts, likely also contribute to this situation8.
Our results also indicate that corvids may serve as alternative seed dispersers44 in areas where carnivores are scarce, such as Matasgordas (Fig. 1B). Corvids can act as seed dispersers either by consuming small pears entirely or, if they cannot swallow them whole, by carrying them in their beaks to preferred perches, where they may land and peck at them or drop them in flight (i.e. stomatochory;65). Further, corvids can consume both fallen fruits from the ground and also can feed on fruits directly from trees before falling (Authors pers. obs.), which could increase somewhat their importance as seed dispersers. However, corvids generally disperse seeds over much shorter distances66–68 than do carnivores8,59. Other smaller bird species visited fruiting trees, although they primarily acted as pulp feeders and seldom removed any fruits. Rabbits in Doñana act mostly as pulp feeders23, whereas hares in Andujar appeared to behave as seed predators (i.e., no remnants of fruits handled by hares, other than pedicels, were observed in 53 recorded instances). Pulp feeding by small birds and rabbits has been proven to increase seedling emergence (via disinhibition effect) and survival and thus, local tree recruitment69. Although our camera-traps provided limited information on the role of rodents, previous seed predation experiments conducted in Doñana70 and Andujar71 indicated that they primarily act as seed predators. However, rodents have also been reported to hoard Iberian pear fruits63, thereby playing a dual role as both predators and short-distance dispersers of Iberian pear seeds. Thus, it is unlikely that birds, lagomorphs or rodents will significantly compensate for the loss of seed dispersal services provided by carnivores.
Concluding remarks
Ungulate fruit predators, rather than carnivore seed dispersers, overwhelmingly dominate as the primary consumers of Iberian pear fruits. Thus, the fruits of this species, characterized by dull colors and scented aroma, and large size, could not be explained based on current dominant selection pressures. Instead, these traits are likely vestiges selected by extant and extinct frugivorous fauna in past times where ungulates fruit predators were not as abundant as nowadays33. These results do not challenge the SDHS as it refers for the presence of evolutionary plant traits rather than actual ecological processes. Our results, however, do emphasize the importance of accounting for the functional role of frugivores when assessing the SDSH. Indeed, these plant traits could have evolved before the arrival of the Pyrus genus in the Iberian Peninsula, originating in Central Asia, as proposed for the ancestors of many present-day plants, including the sweet apple Malus pumila72–74. Specifically, large fruits in the Rosaceae appear to have evolved by hybridization and then by extinct megafauna seed dispersers74. Thus, current ecological conditions are likely significantly different from those under which the tree evolved in the western Mediterranean basin75. For instance, although ungulates (e.g. red deer, wild boar) have inhabited in the Doñana area, at least, for thousands of years75,76, their densities during the last few decades has increased dramatically77. Also, during the last millennium, at least two potential seed dispersers, the wolf Canis lupus78 and the brown bear Ursus arctos79, became extinct in Doñana, and further extinctions occurred during the Pleistocene (e.g., the Barbary macaque Macaca sylvanis;80). Such decline in seed disperser populations has been aggravated over the last few decades by a marked rise in of ungulate fruit predators. To conclude, our results indicates that the composition of the frugivore assemblage of the Iberian pear has dramatically changed in recent times due to global change drivers (e.g., defaunation, overfaunation) which likely alter the selective pressures acting on fruit traits. We highlight the importance of discriminating between seed dispersal syndromes (i.e. sets of traits that favor a particular mechanism) and the current assemblages of seed dispersers and predators in humanized landscapes. A rigorous evaluation of SDSH requires to account for not only the size and sensorial abilities of frugivore species or groups, but also for their functional roles (seed dispersers, seed predators, pulp feeders, etc.).
Study sites and methods
Study sites
Our study was conducted at three sites located in Southern Spain, one within Sierra de Andújar Natural Park (hereafter, Andujar; 38° 14′ N, 4° 4′ W, ~ 740 km2; altitude, 400–800 m) and two within the Doñana National Park (hereafter Matasgordas and Reserva; 37° 9′ N, 6° 26′ W, ~ 543 km2, altitude 0–40 m). All three sites have typical Mediterranean climates with high annual average temperatures (~ 18 °C) and limited rainfalls (500–700 mm): (i) Andujar is located within extensive private lands which vegetation is dominated by Mediterranean scrubland and holm oaks Quercus ilex. Some of the most common fleshy-fruited species are the strawberry tree Arbutus unedo, the mastic Pistacea lentiscus and the Iberian pear, (ii) Matasgordas is located in the northern portion of the Doñana. The most representative habitats in this site include a Mediterranean scrubland dominated by P. lentiscus, Halimium halimifolium, Chamaerops humilis and small clusters of trees species such as Quercus suber, Iberian pear, and Fraxinus angustifolia, and (iii) Reserva is located at the Doñana core and its main habitats comprise a scrubland dominated by H. halimifolium and Stauracanthus genistoides, with several fleshy-fruited species including Rubus ulmifolius, C. humilis, and Iberian pear. Quercus suber trees are scattered across the area.
Study species
The Iberian pear is a small tree endemic to the Iberian Peninsula and North Africa49. Its distribution is highly fragmented, with trees occupying patches of Mediterranean scrubland at low densities (usually < 1 individual ha−1), with occupied patches isolated from each other by natural and human barriers81. The reproduction and regeneration of this tree is very limited due to factors such as high mortality of seeds and seedlings, and seed dispersal limitation8. Each tree produces yearly between ~ 200–1500 fruits. Fruits are globose, fleshy pomes (~ 9.5 g and 2—4 cm in diameter;43), with strong aroma when ripe. Each fruit includes 1–5 viable seeds (46–91 mg each;45) with easily breakable coats. After ripening, fruits drop to the ground from September to December and are harvested by a diverse coterie of mammalian and avian frugivores29,43,44.
The most common frugivores of the Iberian pear are: (i) wild ungulates such as red deer Cervus elaphus and wild boar Sus scrofa occur in high densities in both areas because lack of natural predators (i.e. the gray wolf Canis lupus) and, in Andujar, also because supplemental food by hunters; (ii) medium-sized carnivores such as the red fox Vulpes Vulpes, Eurasian badger Meles meles, and the common genet Genetta genetta are common in both areas, whereas the stone marten Martes foina occurs only in Andujar; (iii) corvids (magpie Pica pica, Iberian magpie Cyanopica cooki and, only in Andujar, the Eurasian jay Garrulus glandarius) and several passerine species; (iv) lagomorphs (Iberian hare Lepus granatensis and European rabbit Oryctolagus cunniculus); and (v) rodents (e.g., Apodemus sylvaticus, Mus spretus, Rattus spp.). Fedriani and Delibes45 provided quantitative data indicating that, in Doñana, carnivores act as seed dispersers of the Iberian pear whereas wild ungulates as fruit and seed predators. Also, rodents have been shown to act mostly act as seed predators both in Doñana70 and Andujar71. Very little information is available, however, concerning the role of lagomorphs and birds as either pulp feeders, seed predators or seed dispersers of the Iberian pear.
Methods
To estimate the importance of different frugivores for the Iberian pear in Andujar, we used published data from Burgos et. al.29 as well as our own unpublished data. We considered 30 fruiting Iberian pear trees during the dispersal season of 2019 (from October 15th to October 30th) and placed fruit depots beneath each tree. Each fruit depot was comprised of 30 ripe fruits within a 1 m side squared plot. Fruits were set regularly in six lines (five fruits per line) about 10 cm apart. Overall, 2,700 Iberian pear fruits were offered in the 30 studied trees. This fruit arrangement allowed to systematically quantify the number of fruits consumed by each frugivore visitor by comparing every image with the previous one to count the number of fruits left. Also, we recorded frugivore visitation as the presence in the pictures of any vertebrate frugivore within or in the immediacy (i.e. ≤ 2 m) of our experimental fruit depots (i.e. irrespective of whether they feed on fruits). A camera trap was placed (Scoutguard SG562-C; white led) on each target pear tree trunk at the height of 60 cm with a slope of 45 degrees. We programmed the cameras to record three images per second when movement was detected, with a minimum time delay (0 s) between consecutive records to maximize the number of images taken per visit. Only visits separated by a period greater than 30 min or clearly identified as different individuals were considered. Fruit removal was recorded during 15 consecutive days. Camera traps were visited every 5 days to refill the fruit depots. In overall, cameras were working 437 trap-days (see29 for further details).
To estimate the importance of different frugivores in Matasgordas, we collected data for 12, 12 and 9 fruiting Iberian pears in 2019 (from September 19th to November 24th), 2021 (from September 22nd to November 24th) and 2022 (from September 26th to December 8th), respectively (44, Authors unpublished data). Also, in Reserva we monitored 11 fruiting trees during 2022 (from September 21st to December 13th; Authors unpublished data). In both study sites, camera traps (LTL ACORN 5310A, detection range = 18 m) were installed to collect data regarding to frugivore species that visited ripe fruiting trees, but we did not supplement any additional fruit beneath target trees. This method allowed an effective record of frugivore visitation, defined as the presence of any vertebrate frugivore underneath the target fruiting tree (i.e. irrespective of whether they feed on fruits). In contrast with Andujar, fruit removal in Matasgordas and Reserva could not be systematically quantified, although we noted all fruit removal events that were occasionally recorded by our camera-traps. Cameras were placed from 3 to 5 m distance from the focal trees and were automatically activated any time a movement occurred, taking a three-photo sequence. We set a trigger delay of 1 min after every three-photo sequence. Frugivore activity was monitored from the start of fruit ripening (mid-September) until all or most fruits have fallen (usually, from late November to early December). For a given camera and frugivore species, we considered successive visits separated by more than 5 min between them. Although 5 min might lead to recording the same individuals several times, we were interested in quantifying successive visits by the same individuals since such accumulated number of visits is likely to lead to higher fruit removal44. In overall, cameras in Matasgordas were working 558, 609, and 694 trap-days in 2019, 2021 and 2022, respectively. In Reserva cameras were working for a total of 420 trap-days in 2022.
For the sake of simplicity, in all three study sites recorded frugivore species were grouped into seven functional groups: ungulates, livestock, carnivores, lagomorphs, rodents, corvids, and ‘other birds’. Because the time interval for considering successive visits by a given frugivore species differed between Andujar (30 min) and Matasgordas/Reserva (5 min), across study site comparisons of the number of frugivore visits would be misleading. However, we here are particularly interested in comparing the proportion of visits by different functional groups of frugivores (e.g., ungulates vs. carnivores vs. birds) both within and between study sites, which is not subject of such limitation. Proportions were compared with Chi-squared tests using the R stats package82.
Methodological approaches for recording frugivore visits varied among study sites. In Andújar, Iberian pear fruits (n = 30) were provided beneath each target tree, while in Matasgordas and Reserva, no fruits were added. This difference likely contributed to increased frugivore visitation in Andújar compared to the other sites. Moreover, the camera-trap detection range in Andújar was narrower, focusing mainly on fruit depots, while in Matasgordas and Reserva, it covered a larger area beneath each target tree. This discrepancy might have led to fewer frugivore records in Andújar. These variations in monitoring techniques prevented a direct comparison of frugivore visit numbers across sites, prompting focus on the percentages of visits by frugivore groups. Additionally, camera-trapping duration in Matasgordas spanned three years, whereas only one year was monitored in the other sites, potentially confounding spatial and temporal patterns of frugivore visitation. Thus, when comparing the proportion of visits by different frugivore groups, initially, all sites and sampling years were considered, followed by a more conservative approach of analyzing pairs of study sites monitored in the same year. Results from both approaches were highly consistent.
Acknowledgements
This study was partially supported by Spanish Minister of Science and Innovation and Universities (ref. PID2022-142067NB-I00, CGL2017- 84633- P, FPU17/04375). Authors thank ICTS-RBD facilities that provided logistical support during the fieldwork at Doñana. The Sierra de Andújar Natural Park and the Territorial Delegation of Environment of Jaén provided the corresponding authorizations and the Foundation Amigos del Lince Ibérico y del Águila Imperial for supported this study on private lands.
Author contributions
All authors conceived the idea and designed the study; T.B., P.J.G. and B.M. collected the data; J.M.F. analyzed the data and led the writing of the manuscript. All authors contributed significantly to the drafts and gave final approval for publication.
Data availability
The datasets generated and/or analysed during the current study are available in the Figshare repository, 10.6084/m9.figshare.25205729.v1.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Levey, D. J., Silva, W. R., & Galetti, M. (Eds.). Seed Dispersal and Frugivory: Ecology, Evolution, and Conservation (CABI, 2002).
- 2.Bascompte J, Jordano P. Mutualistic Networks. Princeton University Press; 2013. [Google Scholar]
- 3.Fleming TH, Kress WJ. The Ornaments of Life: Coevolution and Conservation in the Tropics. University of Chicago Press; 2019. [Google Scholar]
- 4.Corlett RT. Frugivory and seed dispersal. In: Del-Claro K, Torezan-Silingardi HM, editors. Plant-Animal Interactions. Cham: Springer; 2021. [Google Scholar]
- 5.Nathan R, Schurr FM, Spiegel O, Steinitz O, Trakhtenbrot A, Tsoar A. Mechanisms of long-distance seed dispersal. Trends Ecol. Evol. 2008;23(11):638–647. doi: 10.1016/j.tree.2008.08.003. [DOI] [PubMed] [Google Scholar]
- 6.Escribano-Avila G, Calviño-Cancela M, Pías B, Virgos E, Valladares F, Escudero A. Diverse guilds provide complementary dispersal services in a woodland expansion process after land abandonment. J. Appl. Ecol. 2014;51(6):1701–1711. doi: 10.1111/1365-2664.12340. [DOI] [Google Scholar]
- 7.González-Varo JP, Rumeu B, Albrecht J, Arroyo JM, Bueno RS, Burgos T, Traveset A. Limited potential for bird migration to disperse plants to cooler latitudes. Nature. 2021;595(7865):75–79. doi: 10.1038/s41586-021-03665-2. [DOI] [PubMed] [Google Scholar]
- 8.Fedriani JM, Wiegand T, Garrote PJ, Leiva MJ, Ayllón D. Seed dispersal effectiveness in fragmented and defaunated landscapes. Ecosphere. 2023;14(9):e4658. doi: 10.1002/ecs2.4658. [DOI] [Google Scholar]
- 9.Janson CH. Adaptation of fruit morphology to dispersal agents in a neotropical forest. Science. 1983;219(4581):187–189. doi: 10.1126/science.219.4581.187. [DOI] [PubMed] [Google Scholar]
- 10.Lomáscolo SB, Levey DJ, Kimball RT, Bolker BM, Alborn HT. Dispersers shape fruit diversity in Ficus (Moraceae) Proc. Natl. Acad. Sci. 2010;107(33):14668–14672. doi: 10.1073/pnas.1008773107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rojas TN, Zampini IC, Isla MI, Blendinger PG. Fleshy fruit traits and seed dispersers: which traits define syndromes? Ann. Bot. 2022;129(7):831–838. doi: 10.1093/aob/mcab150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Valenta K, Nevo O. The illusiveness of seed dispersal syndromes. A commentary on: Fleshy fruit traits and seed dispersers: Which traits define syndromes? Ann. Bot. 2022;129(7):6–7. doi: 10.1093/aob/mcac009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Janzen DH, Martin PS. Neotropical anachronisms: The fruits the gomphotheres ate. Science. 1982;215(4528):19–27. doi: 10.1126/science.215.4528.19. [DOI] [PubMed] [Google Scholar]
- 14.Hughes L, Dunlop M, French K, Leishman MR, Rice B, Rodgerson L, Westoby M. Predicting dispersal spectra: A minimal set of hypotheses based on plant attributes. J. Ecol. 1994;1:933–950. doi: 10.2307/2261456. [DOI] [Google Scholar]
- 15.Casorso JG, DePasquale AN, Romero Morales S, Cheves Hernandez S, Lopez Navarro R, Hockings KJ, Melin AD. Seed dispersal syndrome predicts ethanol concentration of fruits in a tropical dry forest. Proc. R. Soc. B. 2023;290:20230804. doi: 10.1098/rspb.2023.0804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vargas P, Heleno R, Costa JM. EuDiS-A comprehensive database of the seed dispersal syndromes of the European flora. Biodivers. Data J. 2023;11:1. doi: 10.3897/BDJ.11.e104079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Valenta K, Nevo O. The dispersal syndrome hypothesis: How animals shaped fruit traits, and how they did not. Funct. Ecol. 2020;34(6):1158–1169. doi: 10.1111/1365-2435.13564. [DOI] [Google Scholar]
- 18.Green, A. J., Baltzinger, C., & Lovas‐Kiss, Á. (2022). Plant dispersal syndromes are unreliable, especially for predicting zoochory and long‐distance dispersal. Oikos, 2022(2).
- 19.Herrera CM. Frugivores and Seed Dispersal. Springer; 1986. Vertebrate-dispersed plants: why they don’t behave the way they should; pp. 5–18. [Google Scholar]
- 20.Jordano P. Angiosperm fleshy fruits and seed dispersers: A comparative analysis of adaptation and constraints in plant-animal interactions. Am. Nat. 1995;145(2):163–191. doi: 10.1086/285735. [DOI] [Google Scholar]
- 21.Schaefer HM, Ruxton GD. Plant–Animal Communication. Oxford: OUP; 2011. [DOI] [PubMed] [Google Scholar]
- 22.Alcántara JM, Rey PJ. Conflicting selection pressures on seed size: Evolutionary ecology of fruit size in a bird-dispersed tree, Olea europaea. J. Evol. Biol. 2003;16(6):1168–1176. doi: 10.1046/j.1420-9101.2003.00618.x. [DOI] [PubMed] [Google Scholar]
- 23.Fedriani JM, Delibes M. Pulp feeders alter plant interactions with subsequent animal associates. J. Ecol. 2013;101(6):1581–1588. doi: 10.1111/1365-2745.12146. [DOI] [Google Scholar]
- 24.Traveset A, Escribano-Avila G, Gómez JM, Valido A. Conflicting selection on Cneorum tricoccon (Rutaceae) seed size caused by native and alien seed dispersers. Evolution. 2019;73(11):2204–2215. doi: 10.1111/evo.13852. [DOI] [PubMed] [Google Scholar]
- 25.Cui, J., Zhang, Y., Guo, J., Wu, N., & Zhou, Y. Conflicting selection pressures on seed size and germination caused by carnivorous seed dispersers. Integrative Zoology (2023). [DOI] [PubMed]
- 26.Schupp EW, Jordano P, Gómez JM. Seed dispersal effectiveness revisited: A conceptual review. New Phytol. 2010;188(2):333–353. doi: 10.1111/j.1469-8137.2010.03402.x. [DOI] [PubMed] [Google Scholar]
- 27.Perea R, Delibes M, Polko M, Suárez-Esteban A, Fedriani JM. Context-dependent fruit–frugivore interactions: Partner identities and spatio-temporal variations. Oikos. 2013;122(6):943–951. doi: 10.1111/j.1600-0706.2012.20940.x. [DOI] [Google Scholar]
- 28.McConkey KR, Drake DR. Flying foxes cease to function as seed dispersers long before they become rare. Ecology. 2006;87(2):271–276. doi: 10.1890/05-0386. [DOI] [PubMed] [Google Scholar]
- 29.Burgos T, Fedriani JM, Escribano-Ávila G, Seoane J, Hernández-Hernández J, Virgós E. Predation risk can modify the foraging behaviour of frugivorous carnivores: Implications of rewilding apex predators for plant–animal mutualisms. J. Animal Ecol. 2022;91(5):1024–1035. doi: 10.1111/1365-2656.13682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Côté SD, Rooney TP, Tremblay JP, Dussault C, Waller DM. Ecological impacts of deer overabundance. Annu. Rev. Ecol. Evol. Syst. 2004;35:113–147. doi: 10.1146/annurev.ecolsys.35.021103.105725. [DOI] [Google Scholar]
- 31.Carpio AJ, Apollonio M, Acevedo P. Wild ungulate overabundance in Europe: Contexts, causes, monitoring and management recommendations. Mammal Review. 2021;51(1):95–108. doi: 10.1111/mam.12221. [DOI] [Google Scholar]
- 32.Leal AI, Bugalho MN, Palmeirim JM. Effects of ungulates on oak regeneration in Mediterranean woodlands: A meta-analysis. Forest Ecology and Management. 2022;509:120077. doi: 10.1016/j.foreco.2022.120077. [DOI] [Google Scholar]
- 33.Pires MM, Galetti M. Beyond the “empty forest”: The defaunation syndromes of Neotropical forests in the Anthropocene. Glob. Ecol. Conserv. 2023;1:e02362. [Google Scholar]
- 34.Lecomte X, Fedriani JM, Caldeira MC, Clemente AS, Olmi A, Bugalho MN. Too many is too bad: Long-term net negative effects of overabundant ungulates on a dominant Mediterranean shrub. Plos One. 2016 doi: 10.1371/journal.pone.0158139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Muñoz-Gallego R, Wiegand T, Traveset A, Fedriani JM. From seed dispersal service to reproductive collapse: Density-dependent outcome of a palm–mammal interaction. Oikos. 2023;1:e10002. doi: 10.1111/oik.10002. [DOI] [Google Scholar]
- 36.Delibes M, Castañeda I, Fedriani JM. Spitting seeds from the cud: A review of an endozoochory exclusive to ruminants. Front. Ecol. Evol. 2019;7:265. doi: 10.3389/fevo.2019.00265. [DOI] [Google Scholar]
- 37.Albert A, Mårell A, Picard M, Baltzinger C. Using basic plant traits to predict ungulate seed dispersal potential. Ecography. 2015;38:440–449. doi: 10.1111/ecog.00709. [DOI] [Google Scholar]
- 38.Schaefer HM, Schaefer V. The fruits of selectivity: How birds forage on Goupia glabra fruits of different ripeness. J. Ornithol. 2006;147(4):638–643. doi: 10.1007/s10336-006-0089-x. [DOI] [Google Scholar]
- 39.Encinas-Viso F, Revilla TA, van Velzen E, Etienne RS. Frugivores and cheap fruits make fruiting fruitful. J. Evol. Biol. 2014;27:313–324. doi: 10.1111/jeb.12301. [DOI] [PubMed] [Google Scholar]
- 40.Perea R, Girardello M, San Miguel A. Big game or big loss? High deer densities are threatening woody plant diversity and vegetation dynamics. Biodivers. Conserv. 2014;23:1303–1318. doi: 10.1007/s10531-014-0666-x. [DOI] [Google Scholar]
- 41.Guimaraes PR, Jr, Galetti M, Jordano P. Seed dispersal anachronisms: Rethinking the fruits extinct megafauna ate. PloS One. 2008;3(3):e1745. doi: 10.1371/journal.pone.0001745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kistler L, Newsom LA, Ryan TM, Clarke AC, Smith BD, Perry GH. Gourds and squashes (Cucurbita spp.) adapted to megafaunal extinction and ecological anachronism through domestication. Proc. Natl. Acad. Sci. 2015;112(49):15107–15112. doi: 10.1073/pnas.1516109112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Garrote PJ, Calvo G, Żywiec M, Delibes M, Suárez-Esteban A, Fedriani JM. Strong among population variation in frugivory strength by functional diverse frugivores: A ‘reciprocal translocation’ experiment. Oecologia. 2018;187:143–154. doi: 10.1007/s00442-018-4102-x. [DOI] [PubMed] [Google Scholar]
- 44.Selwyn M, Garrote PJ, Castilla AR, Fedriani JM. Interspecific interactions among functionally diverse frugivores and their outcomes for plant reproduction: A new approach based on camera-trap data and tailored null models. PloS One. 2020;15(10):e0240614. doi: 10.1371/journal.pone.0240614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fedriani JM, Delibes M. Seed dispersal in the Iberian pear, Pyrus bourgaeana: A role for infrequent mutualists. Ecoscience. 2009;16(3):311–321. doi: 10.2980/16-3-3253. [DOI] [Google Scholar]
- 46.San MA, Perea R, García-Olalla M. Wild ungulates vs extensive livestock. Looking back to face the future. Options Meditérranneenes. 2010;92:27–34. [Google Scholar]
- 47.Lone K, Loe LE, Meisingset EL, Stamnes I, Mysterud A. An adaptive behavioural response to hunting: Surviving male red deer shift habitat at the onset of the hunting season. Anim. Behav. 2015;102:127–138. doi: 10.1016/j.anbehav.2015.01.012. [DOI] [Google Scholar]
- 48.Delibes M. Feeding ecology of the Spanish lynx in the Coto Dońana. Acta Theriologica. 1980;25(24):309–324. doi: 10.4098/AT.arch.80-28. [DOI] [Google Scholar]
- 49.Aldasoro JJ, Aedo C, Garmendia FM. The genus Pyrus L.(Rosaceae) in south-west Europe and north Africa. Bot. J. Linnean Soc. 1996;121(2):143–158. [Google Scholar]
- 50.Dufresnes C, Miquel C, Remollino N, Biollaz F, Salamin N, Taberlet P, Fumagalli L. Howling from the past: Historical phylogeography and diversity losses in European grey wolves. Proc. R. Soc. B. 2018;285:20181148. doi: 10.1098/rspb.2018.1148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Clavero M, García-Reyes A, Fernández-Gil A, Revilla E, Fernández N. Where wolves were: Setting historical baselines for wolf recovery in Spain. Anim. Conserv. 2023;26(2):239–249. doi: 10.1111/acv.12814. [DOI] [Google Scholar]
- 52.Pringle RM. Wolves, otters, ungulates, and a promising path for ecology. Proc. Natl. Acad. Sci. 2023;120(7):e2221817120. doi: 10.1073/pnas.2221817120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Castañeda I, Fedriani JM, Delibes M. Potential of red deer (Cervus elaphus) to disperse viable seeds by spitting them from the cud. Mammalian Biol. 2018;90(8):9–91. [Google Scholar]
- 54.Muñoz-Gallego R, Fedriani JM, Serra PE, Traveset A. Nonadditive effects of two contrasting introduced herbivores on the reproduction of a pollination-specialized palm. Ecology. 2022;103(11):e3797. doi: 10.1002/ecy.3797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Furedi and McGraw White-tailed deer: Dispersers or predators of American ginseng seeds? Am. Midland Nat. 2004;152:268–276. doi: 10.1674/0003-0031(2004)152[0268:WDDOPO]2.0.CO;2. [DOI] [Google Scholar]
- 56.Gómez JM, Schupp EW, Jordano P. Synzoochory: The ecological and evolutionary relevance of a dual interaction. Biol. Rev. 2019;94(3):874–902. doi: 10.1111/brv.12481. [DOI] [PubMed] [Google Scholar]
- 57.Morán-López T, Sánchez-Dávila J, Torre I, Navarro-Castilla A, Barja I, Díaz M. Ungulate presence and predation risks reduce acorn predation by mice in dehesas. Plos one. 2022;17(8):e026041958. doi: 10.1371/journal.pone.0260419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Baltzinger, C., & Mårell, A. Plant assemblages and ecosystem functioning, a legacy of long‐term interactions with large herbivores. In: Historical Ecology: Learning from the Past to Understand the Present and Forecast the Future of Ecosystems 163–176 (2022).
- 59.González-Varo JP, López-Bao JV, Guitián J. Functional diversity among seed dispersal kernels generated by carnivorous mammals. J. Animal Ecol. 2013;82(3):562–571. doi: 10.1111/1365-2656.12024. [DOI] [PubMed] [Google Scholar]
- 60.Escribano-Avila G. Non-specialized frugivores as key seed dispersers in dry disturbed environments: An example with a generalist neotropical mesocarnivore. J. Arid Environ. 2019;167:18–25. doi: 10.1016/j.jaridenv.2019.04.015. [DOI] [Google Scholar]
- 61.Draper JP, Young JK, Schupp EW, Beckman NG, Atwood TB. Frugivory and seed dispersal by carnivorans. Front. Ecol. Evol. 2022;10:864864. doi: 10.3389/fevo.2022.864864. [DOI] [Google Scholar]
- 62.Nakashima, Y., & Do Linh San, E. Seed dispersal by mesocarnivores: Importance and functional uniqueness in a changing world. Small Carnivores: Evolution, Ecology, Behaviour, and Conservation 347–391 (2022).
- 63.Fedriani JM, Ayllón D, Wiegand T, Grimm V. Intertwined effects of defaunation, increased tree mortality and density compensation on seed dispersal. Ecography. 2020;43(9):1352–1363. doi: 10.1111/ecog.05047. [DOI] [Google Scholar]
- 64.Fedriani JM, Garrote PJ, Calvo G, Delibes M, Castilla AR, Żywiec M. Combined effects of seed provenance, plant facilitation and restoration site on revegetation success. J. Appl. Ecol. 2019;56(4):996–1006. doi: 10.1111/1365-2664.13343. [DOI] [Google Scholar]
- 65.McConkey, K. R., Sushma, H. S., & Sengupta, A. (2024). Seed dispersal by frugivores without seed swallowing: Evaluating the contributions of stomatochoric seed dispersers. Funct. Ecol.
- 66.Gómez JM. Spatial patterns in long-distance dispersal of Quercus ilex acorns by jays in a heterogeneous landscape. Ecography. 2003;26(5):573–584. doi: 10.1034/j.1600-0587.2003.03586.x. [DOI] [Google Scholar]
- 67.Pesendorfer MB, Sillett TS, Morrison SA, Kamil AC. Context-dependent seed dispersal by a scatter-hoarding corvid. J. Animal Ecol. 2016;85(3):798–805. doi: 10.1111/1365-2656.12501. [DOI] [PubMed] [Google Scholar]
- 68.Martínez-Baroja L, Pérez-Camacho L, Villar-Salvador P, Rebollo S, Quiles P, Gómez-Sánchez D, Rey-Benayas JM. Massive and effective acorn dispersal into agroforestry systems by an overlooked vector, the Eurasian magpie (Pica pica) Ecosphere. 2019;10(12):e02989. doi: 10.1002/ecs2.2989. [DOI] [Google Scholar]
- 69.Fedriani JM, Zywiec M, Delibes M. Thieves or mutualists? Pulp feeders enhance local endozoochore recruitment. Ecology. 2012;93:575–587. doi: 10.1890/11-0429.1. [DOI] [PubMed] [Google Scholar]
- 70.Garrote PJ, Calvo G, Żywiec M, Delibes M, Suárez-Esteban A, Castilla AR, Fedriani JM. Extrinsic factors rather than seed traits mediate strong spatial variation in seed predation. Perspect. Plant Ecol. Evol. Syst. 2019;38:39–47. doi: 10.1016/j.ppees.2019.02.005. [DOI] [Google Scholar]
- 71.Burgos, T. Trophic cascades and plant-disperser mutualisms: The effects of a top predator on seed dispersal of Mediterranean plants. PhD Thesis. Universidad Rey Juan Carlos, Madrid, Spain (2023).
- 72.Juniper, B. E., & Mabberley, D. J. (2006). The story of the apple Imber Press. Portland, Oregon, USA.
- 73.Juniper BE. The mysterious origin of the sweet apple: On its way to a grocery counter near you, this delicious fruit traversed continents and mastered coevolution. Am. Sci. 2007;95(1):44–51. doi: 10.1511/2007.63.44. [DOI] [Google Scholar]
- 74.Spengler RN. Origins of the apple: The role of megafaunal mutualism in the domestication of Malus and rosaceous trees. Front. Plant Sci. 2019;10:617. doi: 10.3389/fpls.2019.00617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Flannery, T. Europe: A Natural History. (Text Publishing, 2018).
- 76.de Carvalho CN, Muñiz F, Galán JM, Rodríguez-Vidal J, Gómez P, Toscano A, Finlayson C. First vertebrate tracks and palaeoenvironment in a MIS-5 context in the Doñana National Park (Huelva, SW Spain) Quaternary Science Reviews. 2020;243:106508. doi: 10.1016/j.quascirev.2020.106508. [DOI] [Google Scholar]
- 77.Gortázar C, Torres MJ, Vicente J, Acevedo P, Reglero M, de la Fuente J, Aznar-Martín J. Bovine tuberculosis in Donana Biosphere Reserve: The role of wild ungulates as disease reservoirs in the last Iberian lynx strongholds. PLoS One. 2008;3(7):e2776. doi: 10.1371/journal.pone.0002776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Valverde, J. A. (1967). Estructura de una comunidad mediterránea de vertebrados terrestres. Van der Pijl, L. (1982). Principles of dispersal in higher plants (Vol. 214) (Springer, Berlin, 1967).
- 79.Swenson JE, Gerstl N, Dahle B, Zedrosser A. Action plan for the conservation of the brown bear (Ursus arctos) in Europe. Nat. Environ. 2000;114:1–69. [Google Scholar]
- 80.Herrera, C. M. Frugivory and seed dispersal by carnivorous mammals, and associated fruit characteristics. In undisturbed Mediterranean habitats. Oikos, 250–262 (1989).
- 81.Fedriani JM, Wiegand T, Delibes M. Spatial pattern of adult trees and the mammal-generated seed rain in the Iberian pear. Ecography. 2010;33(3):545–555. doi: 10.1111/j.1600-0587.2009.06052.x. [DOI] [Google Scholar]
- 82.Team, R. C., Team, M. R. C., Suggests, M. A. S. S., & Matrix, S. (2018). Package stats. The R Stats Package.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated and/or analysed during the current study are available in the Figshare repository, 10.6084/m9.figshare.25205729.v1.