Abstract
Purpose
To assess the prognostic and therapeutic significance of sentinel lymph node biopsy (SLNB) and completion lymph node dissection (CLND) in pediatric conventional melanoma (CM), while evaluating potential predictive factors for outcomes.
Methods
We conducted a retrospective analysis of medical records spanning 2009–2020, focusing on patients aged 18 or younger with localized cutaneous conventional melanoma.
Results
Among the 33 patients, SLNB detected metastasis in 57.6% of cases, with 52.6% undergoing CLND. Positive SLN patients had higher relapse risk (HR 5.92; 95% CI 1.27–27.7; P = 0.024) but similar overall survival (HR 3.19; 95% CI 0.31–33.1, P = 0.33).
No significant differences in disease-free survival (DFS) and OS were found between patients who underwent CLND and those who did not (HR 1.91; 95% CI 0.49–7.43, P = 0.35, and HR 0.52; 95% CI 0.03–8.32, P = 0.64, respectively). Univariate analysis showed age at diagnosis (P = 0.02) correlated with higher recurrence risk, with a 21% hazard increase per additional year of age.
Conclusions
Positive SLN status and age at diagnosis were associated with worse DFS in CM patients. Our study did not find any prognostic or therapeutic value in CLND for pediatric melanoma. Further multicenter trials are needed to confirm our single-institution experience.
Level of evidence
Level IV.
Keywords: Melanoma, Sentinel lymph mode biopsy, Spitz melanoma, Pediatric melanoma
Introduction
Although melanoma is a rare entity in childhood and adolescence, accounting for less than 1% of all pediatric malignancies [1–4], it is the deadliest pediatric skin malignancy [5]. Furthermore, it is the most common pediatric skin cancer, with an incidence that increases with age, rising to 4% of all malignancies in adolescents age 15–19 years [6, 7]. Three types of melanomas develop in the pediatric cohort: conventional melanoma (CM), melanoma that arises in a large/giant congenital melanocytic nevus, and Spitz melanoma (SM), the latter being the most common pediatric type [8, 9]. CM, also known as “adult-type melanoma,” shares the same causes and risk factors as in adults, and, therefore, treatment options are often based on adult protocols [8]. In contrast, SM exhibits different spreading patterns, and identification of prognostic factors remains challenging [8, 10, 11].
Like adult melanomas, most pediatric melanomas are localized, and their prognosis varies with age and stage of disease [12–14]. The more advanced the stage, the poorer the outcome [15, 16]. However, the prognostic significance of histologic risk factors identified for adult cases (e.g., vascular invasion, mitotic activity) has not been directly examined in pediatric tumors [17, 18]. More specifically, while traditional prognostic parameters for adult cutaneous melanoma, i.e., depth of the tumor (or Breslow thickness) and nodal metastases [19, 20] carry different predictive values in pediatric SM lesions, they have not been specifically evaluated in pediatric CM lesions [18, 21]. Efforts to identify reliable prognostic factors for pediatric melanoma were recently directed toward the evaluation of candidate immunohistochemical and molecular markers. This revealed the TERT (telomerase reverse transcriptase) oncogene promoter mutation in pediatric SM to be associated with a poorer prognosis [17, 21].
Uncertainty remains regarding the optimal management of regional lymph nodes in pediatric melanoma [2, 22]. Although sentinel lymph node (SLN) involvement has been validated as a prognostic marker in adult patients with melanoma [19, 23], its impact on overall survival (OS) in the pediatric population is unclear [9, 24, 25]. Due to its rarity in childhood and adolescence, pediatric melanoma is underrepresented in clinical trials, and pediatric oncologic teams have aligned their care strategies to guidelines developed in adults. Most melanoma studies in pediatric population had both CM and SM which are two entities that behave differently. In this study we aim to focus on patients with CM only.
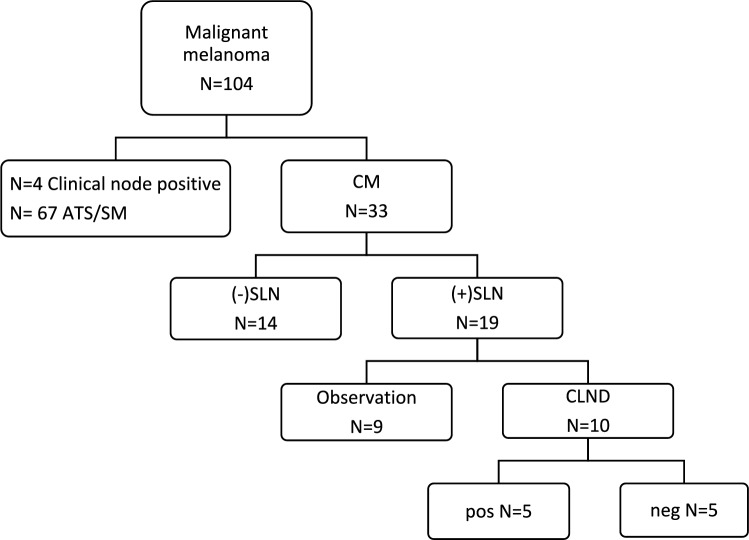
The aim of this study was two-fold. The primary objective was to provide a qualitative analysis of the prognostic role and impact of sentinel lymph node biopsy (SLNB) and completion lymph node dissection (CLND) on the outcome of pediatric conventional melanoma. Our secondary objective was to examine the different prognostic factors associated with survival and recurrence of disease (Fig. 1).
Fig. 1.
Flow Diagram of children diagnosed with cutaneous melanoma treated at our hospital. ATS/SM atypical spitzoid melanoma/spitzoid melanoma, CM conventional melanoma, SD standard deviation, mm millimeter, SLN sentinel lymph node, (+) positive, (−) negative
Methods
Study design and data source
This retrospective single-center cohort study and related protocol were approved by the Institutional Review Board at St. Jude Children’s Research Hospital. We included pediatric patients aged 18 years or younger with a diagnosis of localized cutaneous melanoma of the conventional subtype without clinically apparent regional node involvement, seen at our institution between 2009 and 2020. We reanalyzed all pathologic data and slides for patients referred to our center who had their primary surgery and/or SLNB at another institution. We excluded patients with non-CM subtype, clinically detectable regional nodal and/or distant metastases. Demographic data (i.e., age at the time of diagnosis, sex, race), primary tumor characteristics (tumor location, type of melanoma, Breslow thickness, presence of ulceration, mitotic index, resection margin), nodal status, regional lymph node management, recurrence rate, and survival were collected from patients’ medical records. Diseases were staged according to the American Joint Committee on Cancer (AJCC) staging system [20].
Statistical analysis
First, the pediatric patients who underwent SLNB were categorized based on the results of the procedure, specifically the SLN status. Then, the subgroup of patients identified with a positive SLN were classified based on whether they underwent nodal observation or CLND. Descriptive statistics included evaluating central tendency using mean ± standard deviation (SD) and median with interquartile ranges (IQR). Continuous variables were compared using the two-tailed Student’s t test or Wilcoxon rank sum test, while categorical variables were compared using the chi-square or Fisher exact test.
The primary endpoints of our analysis were OS, which was defined by time from diagnosis of CM to death from any causes, and disease-free survival (DFS) defined by time from complete resection to recurrence. OS and DFS were analyzed using Kaplan–Meier curves, and they were compared using a log-rank test. Prognostic factors for both OS and DFS were examined utilizing the log-rank test and the Cox Proportional Hazards (Cox-PH) model in univariate analysis. Results are reported as a P value or hazard ratio (HR) with 95% confidence interval (CI). Statistical analysis was performed using (R version 3.6.2). A P value < 0.05 was considered significant.
Results
Study population and tumor characteristics
Among the 100 patients seen at our institution who underwent SLNB for a melanocytic lesion, 67 had ATS/SM subtype and were thus excluded from the study. The final cohort included 33 patients with conventional melanoma. Among the 19 (57.6%) patients with positive SLN, 10 (52.6%) underwent CLND.
There were 14 (42%) female patients, with a median age at diagnosis of 13 years (IQR = 2–21). All the patients were white. Fifteen (45%) patients had primary lesions on the truncal area. Nodular subtype was the most common (n = 8) histologic subtype. The mean tumor depth was 3.47 mm (SD = 2.54); 19 (57%) of the patients had a tumor depth exceeding 2 mm. The primary tumor of 30 (91%) patients had a mitotic rate of at least 1/mm2 (Table 1).
Table 1.
Patient and tumor characteristics of patients with pediatric CM cutaneous melanoma
| Characteristic | No. (%)a |
|---|---|
| Age at diagnosis (years) | |
| Mean (SD) | 12.55 (4.02) |
| Median (range) | 13 (2, 21) |
| Sex | |
| Female | 14 (42%) |
| Male | 19 (58%) |
| Race | |
| White | 33 (100%) |
| Histology subtypes | |
| Acral lentiginous | 1 (3%) |
| Folliculocentric | 1 (3%) |
| Nevoid | 4 (12%) |
| Nodular | 8 (24%) |
| Pigment synthesizing | 1 (3%) |
| Superficial spreading | 5 (15%) |
| Unknown | 13 (40%) |
| Primary tumor location | |
| Extremity | 9 (27%) |
| Head or neck | 9 (27%) |
| Trunk | 15 (45%) |
| Tumor depth | |
| T1 | 3 (9%) |
| T2 (1.01–2.00 mm) | 11 (33%) |
| T3 (2.01–4.00 mm) | 9 (27%) |
| T4 (> 4.00 mm) | 10 (30%) |
| Depth (mm) | |
| Mean (SD) | 3.47 (2.54) |
| Median (range) | 2.36 (0.42, 9.4) |
| Ulceration | |
| Yes | 14 (42%) |
| No | 19 (58%) |
| Mitotic counts | |
| Mean | 6.4 (6.49) |
| Median | 4 (0,26) |
| Mitosis | |
| Absence | 3 (9%) |
| Presence | 30 (91%) |
CM conventional melanoma, mm millimeter, N/A not available, SD standard deviation
aValues indicate the number of patients (%), unless otherwise indicated
Sentinel lymph node evaluation
Of the 33 patients, 19 had lymph node metastasis (Table 2). The median follow-up was 5.16 years (95% IQR 2.92–9.16) in the negative SLN group and 4.74 years (95% IQR 2.07–5.77) in the positive SLN group. For all patients with extremity lesions (nine patients), the SLN, as localized with a nuclear medicine scan, was consistently in the ipsilateral groin for lower extremities and in the ipsilateral axilla for the upper extremities. None of the patients with a T1 lesion or a tumor depth of less than 1 mm had a positive SLN.
Table 2.
Patients and tumor characteristics of those with positive vs negative sentinel lymph nodes
| Characteristic | Positive SLN (n = 19) n (%)a |
Negative SLN (n = 14) n (%)a |
P value |
|---|---|---|---|
| Age at diagnosis | 0.21 | ||
| Mean (SD) | 13.16 (3.95) | 11.71 (4.1) | |
| Median (range) | 14 (2, 21) | 11.5 (4, 18) | |
| Age at diagnosis | > 0.99 | ||
| < 10 years | 3 (16%) | 3 (21%) | |
| ≥ 10 years | 16 (84%) | 11 (79%) | |
| Sex | 0.45 | ||
| Female | 7 (37%) | 7 (50%) | |
| Male | 12 (63%) | 7 (50%) | |
| Primary tumor location | 0.062 | ||
| Extremity | 3 (16%) | 6 (43%) | |
| Head or neck | 4 (21%) | 5 (36%) | |
| Trunk | 12 (63%) | 3 (21%) | |
| Depth (mm) | 0.25 | ||
| Mean (SD) | 3.75 (2.43) | 3.1 (2.72) | |
| Median (range) | 3.3 (1.3, 9.4) | 2.02 (0.42–9.20) | |
| Tumor depth | 0.28 | ||
| T1 | – | 3 (21%) | |
| T2 (1.01–2.00 mm) | 7 (37%) | 4 (29%) | |
| T3 (2.01–4.00 mm) | 6 (32%) | 3 (21%) | |
| T4 (> 4.00 mm) | 6 (32%) | 4 (29%) | |
| N/A | |||
| Ulceration | 0.17 | ||
| Yes | 10 (53%) | 4 (29%) | |
| No | 9 (47%) | 10 (71%) | |
| Mitosis | 0.56 | ||
| Absence | 1 (5%) | 2 (14%) | |
| Presence | 18 (95%) | 12 (86%) | |
| Mitotic counts | 0.37 | ||
| Mean | 6.79 (6.38) | 5.87 (6.84) | |
| Median | 5 (0, 26) | 3 (0, 21) | |
| Margin statusb | 0.016 | ||
| 1–5 mm | 7 (37%) | 2 (14%) | |
| 5–10 mm | 2 (11%) | 4 (29%) | |
| > 10 mm | 4 (21%) | 8 (57%) | |
| Free | 6 (32%) | – | |
| Adjuvant treatmentc | < 0.001 | ||
| Yes | 17 (89%) | 2 (14%) | |
| No | 2 (11%) | 12 (86%) | |
| Median number (range) of SLN identified per patient | 3 (1,7) | 2 (1,13) | 0.37 |
CM conventional melanoma, SD standard deviation, mm millimeter, SLN sentinel lymph node
aValues indicate the number of patients (%), unless otherwise indicated
bMargin status indicates the size of the resection margin
cAdjuvant treatment included interferon alpha or immunotherapy
The hazard of relapse in the positive SLN group was 5.92 times higher than the risk in the negative SLN group (HR 5.92; 95% CI 1.27–27.7; P = 0.02) (Table 3).
Table 3.
Univariate hazard ratios for melanoma recurrence and death of overall 33 patients
| Prognostic factors | Recurrence factor | Survival factor | ||
|---|---|---|---|---|
| HR (95% CI) | P value | HR (95% CI) | P value | |
| SLN (pos vs neg) | 5.92 (1.27–27.7) | 0.024 | 3.19 (0.31–33.1) | 0.33 |
| Age at diagnosis | ||||
| (≥ 10 years vs < 10 years) | 3 (0.38–23.4) | 0.29 | –a | |
| Age (range) | 1.21 (1.02–4.44) | 0.028 | –a | |
| Primary tumor location | ||||
| Trunk vs extremity | 7.74 (0.9–66.7) | 0.062 | –a | |
| Head or neck vs extremity | 6.98 (0.69–70.4) | 0.1 | –a | |
| Sex | ||||
| (Male vs female) | 0.87 (0.29–2.62) | 0.81 | 0.82 (0.11–5.93) | 0.84 |
| Ulceration | ||||
| (Yes vs no) | 1.93 (0.64–5.77) | 0.24 | 0.37 (0.04–3.64) | 0.36 |
| Median tumor depth (range) | 1.15 (0.96–1.4) | 0.14 | 0.79 (0.48–1.3) | 0.36 |
| Margin statusb (vs 10 mm) | ||||
| < 5 mm | 1.31(0.33–5.25) | 0.71 | –a | |
| ≥ 5 mm to < 10 mm | 0.87(0.16–4.81) | 0.88 | –a | |
| Mitotic counts (presence vs absence) | 1.6 (0.21–12.4) | 0.65 | –a | |
CI confidence interval, CLND complete lymph node dissection, CM conventional melanoma, HR hazard ratio, neg negative, pos positive, Q quartile, SD standard deviation, SLN sentinel lymph node
aThe samples were too small to analyze HR
bMargin status indicates the size of the resection margin
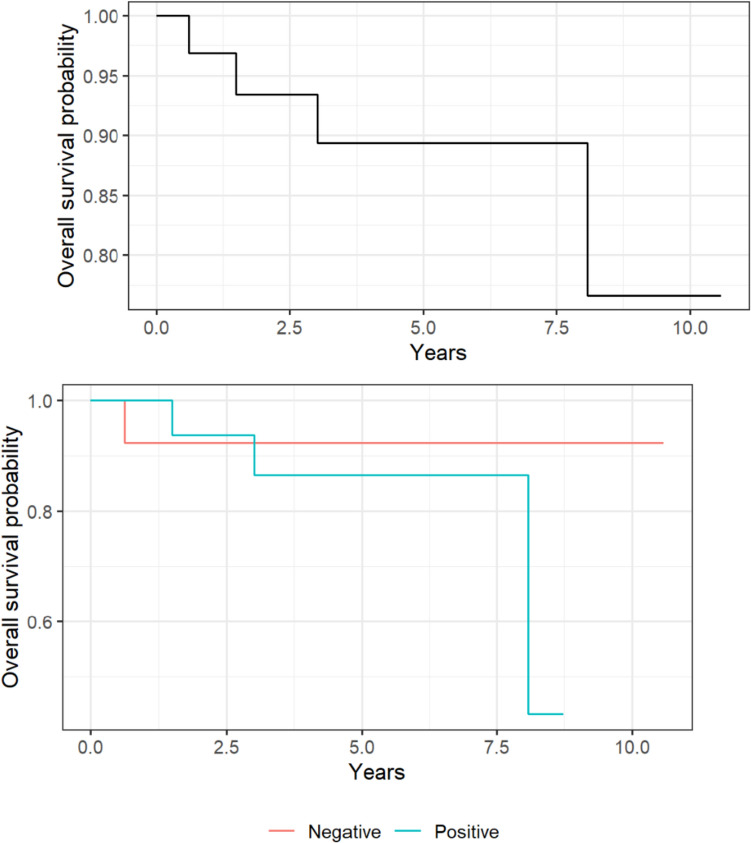
There was no difference in OS between the patients with negative SLN and those with positive SLN (HR 3.19; 95% CI 0.31–33.1, P = 0.33), with a 5-year OS rate of 92.3% (95% CI 78.9–100%), and 86% (95% CI 70.7–100%), respectively; Fig. 2. Of note, one patient had anaphylaxis secondary to methylene blue. This patient completely recovered after management with adrenaline and steroids. DFS changed significantly with age at diagnosis HR (95% CI) = 1.2 (1.02–1.44), P = 0.03, and with a 5-year DFS of 85.1% (95% CI: 68–100%) and of 39.1% (95% CI: 21.1–72.3%) in the negative SLN and positive subgroup, respectively; Fig. 3.
Fig. 2.
The Kaplan–Meier survival curves illustrate the overall survival probability for the entire cohort (upper panel) and as a function of sentinel lymph node status (lower panel)
Fig. 3.
Disease-free survival probability for the entire cohort (upper panel) and as a function of sentinel lymph node status (lower panel)
Complete lymph node dissection vs observation for sentinel-node metastasis
Table 4 shows the tumor characteristics and demographics of patients who underwent CLND vs observation for a positive SLN status. Of the 19 patients who had a positive SLN, 10 individuals (53%) underwent complete CLND, while the remaining 9 patients (47%) were managed through observation-alone. The decision to perform a CLND was influenced by the presence of high-risk clinical features. Notably, patients who underwent CLND were slightly older (P = 0.043) and had more ulceration (P = 0.023) compared to those who received conservative treatment (Table 5). As a result of CLND, 5 (50%) patients were found to have further nodal metastasis. Median follow-up was 5.45 years (IQR 4.87–6) in the CLND group and 2.6 years (IQR 1.42–3.66) in the observation group.
Table 4.
Patient and tumor characteristics of those who underwent CLND vs no CLND
| Characteristic | CLND (n = 10) n (%)a |
Non CLND (n = 9) n (%)a |
P value |
|---|---|---|---|
| Age at diagnosis (years) | 0.043 | ||
| Mean (SD) | 15 (2.83) | 11.11 (4.14) | |
| Median (range) | 14 (11, 21) | 13 (2, 15) | |
| Sex | > 0.99 | ||
| Female | 4 (40%) | 3 (33%) | |
| Male | 6 (60%) | 6 (67%) | |
| Primary tumor location | 0.18 | ||
| Extremity | – | 3 (33%) | |
| Head or neck | 3 (30%) | 1 (11%) | |
| Trunk | 7 (70%) | 5 (56%) | |
| Depth (mm) | 0.97 | ||
| Mean (SD) | 4.08 (3.04) | 3.38 (1.63) | |
| Median (range) | 2.63 (1.3, 9.4) | 3.5 (1.55, 7) | |
| Tumor depth | 0.62 | ||
| T1 | – | – | |
| T2 (1.01–2.00 mm) | 4 (40%) | 3 (33%) | |
| T3 (2.01–4.00 mm) | 2 (20%) | 4 (44%) | |
| T4 (> 4.00 mm) | 4 (40%) | 2 (22%) | |
| Ulceration | 0.023 | ||
| Yes | 8 (80%) | 2 (22%) | |
| No | 2 (20%) | 7 (78%) | |
| Mitosis | 0.47 | ||
| Absence | – | 1 (11%) | |
| Presence | 10 (100%) | 8 (89%) | |
| Mitotic counts | 0.15 | ||
| Mean | 8.7 (7.56) | 4.67 (4.21) | |
| Median | 6.5 (2, 26) | 3 (0, 13) | |
| Margin statusb | 0.78 | ||
| 1–5 mm | 4 (40%) | 3 (33%) | |
| 5–10 mm | 1 (10%) | 1 (11%) | |
| > 10 mm | 3 (30%) | 1 (11%) | |
| Free | 2 (20%) | 4 (44%) | |
| Adjuvant treatmentc | 0.21 | ||
| Yes | 10 (100%) | 7 (78%) | |
| No | – | 2 (22%) | |
| Median number (range) of SLN identified per patient | 3 (1, 7) | 3 (1, 4) | 0.79 |
CM conventional melanoma, SD standard deviation, mm millimeter, SLN sentinel lymph node
aValues indicate the number of patients (%), unless otherwise indicated
bMargin status indicates the size of the resection margin
cAdjuvant treatment included interferon alpha or immunotherapy
Table 5.
Univariate hazard ratios for melanoma recurrence and death of positive SLN patients (N = 19)
| Prognostic factors | Recurrence factor | Survival factor | ||
|---|---|---|---|---|
| HR (95% CI) | P value | HR (95% CI) | P value | |
| CLND (yes vs no) | 1.98 (0.28–14.2) | 0.35 | 0.52 (0.03–8.32) | 0.64 |
| Age at diagnosis | ||||
| (≥ 10 years vs < 10 years) | 0.76 (0.09–6.5) | 0.8 | –a | |
| Primary tumor location | ||||
| Trunk vs extremity | 0.51 (0.06–4.63) | 0.55 | –a | |
| Head or neck vs extremity | 0.9 (0.09–9.5) | 0.93 | –a | |
| Sex | ||||
| (Male vs female) | 1.88 (0.48–7.38) | 0.37 | 1.45 (0.13–16.3) | 0.76 |
| Ulceration | ||||
| (Yes vs no) | 1.63 (0.46–5.81) | 0.45 | 0.47 (0.04–5.24) | 0.54 |
| Median tumor depth (range) | 1.12 (0.86–1.47) | 0.38 | –a | |
| Margin statusc (vs 10 mm) | ||||
| < 5 mm | 0.37 (0.03–4.13) | 0.42 | –a | |
| ≥ 5 mm to < 10 mm | – | – | ||
| Mitotic counts | ||||
| (presence vs absence) | 0.37 (0.04–3.17) | 0.36 | –a | |
aThe samples were too small to analyze HR
bMargin status indicates the size of the resection margin
CI confidence interval, CLND complete lymph node dissection, CM conventional melanoma, HR hazard ratio, neg negative, pos positive, Q quartile, SD standard deviation, SLN sentinel lymph node
There was no difference in DFS and OS between the patients who underwent CLND and those who did not undergo CLND. The hazard ratios for DFS and OS were (HR 1.91 (0.49–7.43), P = 0.35) and (HR 0.52; 95% CI 0.03–8.32, P = 0.64), respectively. Two (20%) patients had complications related to the CLND that included a seroma and a wound infection (n = 1 each).
Adjuvant therapy
Adjuvant therapy was administered to 19 patients. Within this group, 17 patients had positive SLN, while 2 individuals presented with negative SLN. In the positive SLN subgroup, 8 patients opted for immunotherapy and 11 underwent treatment with interferon. Fatalities were observed for three patients who received interferon. In the negative SLN subgroup, two patients received interferon.
Among the 14 individuals who did not undergo adjuvant treatment, 2 had positive SLN, resulting in 1 case of recurrence at stage T3aN0. The remaining 12 patients in this subset exhibited negative SLN, and 1 patient in this category succumbed to stage T4N0M1.
Predictor analyses
Four patients died of melanoma. All these patients experienced a recurrence of their tumor and 3 had a positive SLNB (Table 6). Univariate analysis was performed to identify prognostic factors associated with mortality and recurrence (Table 3). The recurrence of trunk or head and neck lesions tended to be greater than that of lesions on the extremities (HR = 7.74; 95% CI 0.9–66.7; P = 0.062) and HR 6.98 (95% CI 0.69–70.4; P = 0.1) for the truncal lesion and head/neck lesion, respectively. None of the factors were correlated with survival overall.
Table 6.
Characteristics of the four patients with pediatric melanoma who died
| # | Characteristic | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sex, age at Dx (years) | Histology | Primary tumor location | Tumor stage | SLNB (status) | CLND (result) | Margin (mm) | Mitototic counta | Genomics | Time to deathb (months) | |
| 1 | M, 13 | Nodular | Truncal | T2a | Pos (1/2), subcapsular | N | Free | 3 | n/a | 37 |
| 2 | F, 16 | Melanoma | Truncal | T3a | Pos (2/5), subcapsular | Y (neg) | 0.5 | 3 | n/a | 96 |
| 3 | M, 14 | Superficial spreading | Truncal | T2b | Pos (1/1), extracapsular | Y (neg) | 7 | 5 | BRAF | 17 |
| 4 | F, 13 | Folliculocentric | Head/neck | T4a | neg, 0/5 | N | 8 | > 20 | BRAF and P16 deletion | 6 |
aMitotic counts indicate number of mitosis per mm2
bTime to death indicates the period from diagnosis to mortality
CLND complete lymph node dissection, Dx diagnosis, F female, M male, pos positive, N no, neg negative, Pt No. patient number, SLNB sentinel lymph node biopsy
Discussion
This study revealed that CM pediatric patients with a positive SLN were significantly more likely to experience melanoma recurrence than those with a negative SLN. It also appears that, for patients with positive SLNB, CLND did not change the outcome; however, the small sample size and the limited statistical power are acknowledged.
These results were consistent with the MSLT (Multicenter Selective Lymphadenectomy Trial-I) and MSLT-II studies in the adult population [23, 26]. According to the MSLT-I, there was no significant treatment-related difference in 10-year melanoma-specific survival (MSS) among the overall patient population. However, DFS and MSS were significantly improved in the SLNB group with intermediate lesions, which were defined as 1.20- to 3.50-mm depth. In our study, none of the patients with lesions less than 1.01 mm had a positive SLN.
This study also revealed that SLNB status is a prognostic indicator for melanoma recurrence and death of patients with lesions greater than or equal to 1.2-mm depth. Nevertheless, CLND did not increase MSS among patients with melanoma and SLN metastases [6, 15]. A previous report on pediatric melanomas from Kim et al. (2016) revealed that SLNB does not confer survival benefit to children with melanoma [2]. A study of the Surveillance, Epidemiology, and End Results database (SEER) showed that a positive SLN was associated with a poorer melanoma-specific free survival (89% vs 100% at 84 months, P = 0.04) [2]. However, it is unclear if a CLND should be performed in all SLN-positive patients, especially when considering the high risk of lymphedema [26] and other associated postoperative complications. Conservative clinical management of positive lymph nodes in pediatric melanoma is supported by the MSLT-II study, which found that observation of SLN-positive patients is safe in the low-risk group, which includes patients who do not have any extracapsular spread/extension, any concomitant microsatellites of the primary tumor, more than three involved nodes, or any involvement of more than two nodal basins in patients with immunosuppression [19, 26].
Currently, the mainstay of treatment of pediatric melanoma is to perform a wide local excision, potentially including a SLN [22]. The controversy is whether these patients need to undergo SLNB or CLND, per the adult guidelines [2, 3, 22]. SLNB is not recommended for patients who have lesions less than 0.8 mm, without ulceration, and categorized as T1a, because the incidence of positive SLN is less than 5% [23]. Pediatric SM has a very different behavior and a better prognosis than CM or melanoma in the adult population [11, 17, 18, 21]. Thus, the staging criteria being used in adults with SM, such as Breslow thickness or nodal metastasis, do not have the same prognostic value in pediatric patients.
Although SLNB does not improve MSS in the pediatric population, it can be used as a prognostic indicator of poorer outcomes [5]. This is strengthened by a study of 310 pediatric patients (< 20 years old) with melanoma whose lesion had a Breslow depth exceeding 0.75 mm that revealed an impact of SLNB positivity on MSS (100% for the group with negative SLNB vs 89% for the group with positive SLNB) [2].
Based on previous publications and our findings, it is still recommended to perform a SLNB in lesions that are 0.8 mm deep with ulceration or deeper than 0.8 mm with or without ulceration [19]. However, we do not recommend CLND in low-risk CM without risk factors associated with extranodal metastasis (i.e., age ≥ 10 years, presence of ulceration, mitotic activity > 5/mm2 [21]) or TERT promotor mutation [11, 17]. Our institutional protocol for patients with low-risk disease but a positive SLN includes clinical examination every 4 months during the first 2 years, every 6 months during years 3 through 5, and annually thereafter. Ultrasonographic assessment of the sentinel-node basin is performed at each visit for the first 5 years. Abnormal ultrasonographic findings include a lymph node length-to-depth ratio of less than 2 cm, a hypoechoic center, an absence of hilar vessels, or focal nodularity with increased vascularity [26].
We found, on our univariate analysis, that a positive SLNB and age at diagnosis, as a continuous data, were associated with higher recurrence in patients with CM. The limited sample of patients prevented multivariable analysis. According to the previous studies the prognosis of childhood melanoma is associated with lesion thickness, stage of the disease, truncal location, and presence of ulceration [2, 5, 22, 27]. Controversy persists in terms of how wide the resection should be in the pediatric population. According to the adult recommendations, the margin is determined by the depth of the lesion. However, we do not usually perform local excision wider than 1 cm due to the limitation of body surface area in children. We found that recurrence and mortality rates were higher in patients whose excision margins were < 5 mm, however, the difference was not statistically significant.
Conclusion
Positive SLN status and age at diagnosis were associated with worse DFS in CM patients. Our study did not find any prognostic or therapeutic value in CLND for pediatric melanoma. However, further multicenter trials are needed to confirm our single-institution experience. In extremity cutaneous melanoma, the location of the SLN is always predictable, being the axilla for the upper-extremity melanoma and the groin for the lower-extremity melanoma.
Acknowledgements
Thanks to Angela McArthur for editing the manuscript.
Abbreviations
- AJCC
American Joint Committee on Cancer
- AST
Atypical spitzoid tumor
- CLND
Complete lymph node dissection
- CM
Conventional melanoma
- DFS
Disease-free survival
- HR
Hazard ratio
- IQR
Interquartile range
- M
Median
- MSLT
Multicenter Selective Lymphadenectomy Trial
- MSS
Melanoma-specific survival
- OS
Overall survival
- SLN
Sentinel lymph node
- SLNB
Sentinel lymph node biopsy
- SM
Spitz melanoma
Author contributions
All authors took part in writing the manuscript, reviewing it, and revising its intellectual and technical content. All authors also assume responsibility and accountability for the results.
Funding
This work was supported by the American Lebanese Syrian Associated Charities (ALSAC). The authors have no financial relationships relevant to this work to disclose.
Data availability
The data supporting this study's findings are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request. Data are located in controlled access data storage at St Jude Children's Research Hospital.
Declarations
Conflict of interest
The authors have no conflicts to declare.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Hill SJ, Delman KA (2012) Pediatric melanomas and the atypical spitzoid melanocytic neoplasms. Am J Surg 203:761–767. 10.1016/j.amjsurg.2011.04.008 [DOI] [PubMed] [Google Scholar]
- 2.Kim J, Sun Z, Gulack BC, Adam MA, Mosca PJ, Rice HE, Tracy ET (2016) Sentinel lymph node biopsy is a prognostic measure in pediatric melanoma. J Pediatr Surg 51:986–990. 10.1016/j.jpedsurg.2016.02.067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tracy ET, Aldrink JH (2016) Pediatric melanoma. Semin Pediatr Surg 25:290–298. 10.1053/j.sempedsurg.2016.09.010 [DOI] [PubMed] [Google Scholar]
- 4.Neier M, Pappo A, Navid F (2012) Management of melanomas in children and young adults. J Pediatr Hematol Oncol 34(Suppl 2):S51-54. 10.1097/MPH.0b013e31824e3852 [DOI] [PubMed] [Google Scholar]
- 5.Saiyed FK, Hamilton EC, Austin MT (2017) Pediatric melanoma: incidence, treatment, and prognosis. Pediatric Health Med Ther 8:39–45. 10.2147/PHMT.S115534 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Childhood Cancer by the Iccc. In: Howlader N, Noone Am, Krapcho M et al (eds) Seer cancer statistics review (Csr) 1975–2014. National Cancer Institute, Section 29. https://seer.cancer.gov/archive/csr/1975_2014/. Accessed 05 Oct 2020
- 7.Bleyer A, O’leary M, Barr R et al (eds) Cancer epidemiology in older adolescents and young adults 15 to 29 years of age, including seer incidence and survival: 1975–2000. National Cancer Institute, 2006. NIH Pub. No. 06–5767. https://seer.cancer.gov/archive/publications/aya/index.html. Accessed 12 Jan 2021
- 8.Merkel EA, Mohan LS, Shi K, Panah E, Zhang B, Gerami P (2019) Paediatric melanoma: clinical update, genetic basis, and advances in diagnosis. Lancet Child Adolesc Health 3:646–654. 10.1016/S2352-4642(19)30116-6 [DOI] [PubMed] [Google Scholar]
- 9.Jen M, Murphy M, Grant-Kels JM (2009) Childhood melanoma. Clin Dermatol 27:529–536. 10.1016/j.clindermatol.2008.09.011 [DOI] [PubMed] [Google Scholar]
- 10.Pappo AS (2003) Melanoma in children and adolescents. Eur J Cancer 39:2651–2661. 10.1016/j.ejca.2003.06.001 [DOI] [PubMed] [Google Scholar]
- 11.Duncan LM (2014) Atypical Spitz tumours and sentinel lymph nodes. Lancet Oncol 15:377–378. 10.1016/S1470-2045(13)70397-8 [DOI] [PubMed] [Google Scholar]
- 12.Wong JR, Harris JK, Rodriguez-Galindo C, Johnson KJ (2013) Incidence of childhood and adolescent melanoma in the United States: 1973–2009. Pediatrics 131:846–854. 10.1542/peds.2012-2520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Strouse JJ, Fears TR, Tucker MA, Wayne AS (2005) Pediatric melanoma: risk factor and survival analysis of the surveillance, epidemiology and end results database. J Clin Oncol 23:4735–4741. 10.1200/JCO.2005.02.899 [DOI] [PubMed] [Google Scholar]
- 14.Brecht IB, Garbe C, Gefeller O, Pfahlberg A, Bauer J, Eigentler TK, Offenmueller S, Schneider DT, Leiter U (2015) 443 paediatric cases of malignant melanoma registered with the German Central Malignant Melanoma Registry between 1983 and 2011. Eur J Cancer 51:861–868. 10.1016/j.ejca.2015.02.014 [DOI] [PubMed] [Google Scholar]
- 15.Lange JR, Palis BE, Chang DC, Soong SJ, Balch CM (2007) Melanoma in children and teenagers: an analysis of patients from the National Cancer Data Base. J Clin Oncol 25:1363–1368. 10.1200/JCO.2006.08.8310 [DOI] [PubMed] [Google Scholar]
- 16.Ferrari A, Brecht IB, Gatta G, Schneider DT, Orbach D, Cecchetto G, Godzinski J, Reguerre Y, Bien E, Stachowicz-Stencel T, Ost M, Magni C, Kearns P, Vassal G, Massimino M, Biondi A, Bisogno G, Trama A (2019) Defining and listing very rare cancers of paediatric age: consensus of the Joint Action on Rare Cancers in cooperation with the European Cooperative Study Group for Pediatric Rare Tumors. Eur J Cancer 110:120–126. 10.1016/j.ejca.2018.12.031 [DOI] [PubMed] [Google Scholar]
- 17.Lee S, Barnhill RL, Dummer R, Dalton J, Wu J, Pappo A, Bahrami A (2015) TERT promoter mutations are predictive of aggressive clinical behavior in patients with spitzoid melanocytic neoplasms. Sci Rep 5:11200. 10.1038/srep11200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cerrato F, Wallins JS, Webb ML, McCarty ER, Schmidt BA, Labow BI (2012) Outcomes in pediatric atypical spitz tumors treated without sentinel lymph node biopsy. Pediatr Dermatol 29:448–453. 10.1111/j.1525-1470.2011.01699.x [DOI] [PubMed] [Google Scholar]
- 19.Wong SL, Faries MB, Kennedy EB, Agarwala SS, Akhurst TJ, Ariyan C, Balch CM, Berman BS, Cochran A, Delman KA, Gorman M, Kirkwood JM, Moncrieff MD, Zager JS, Lyman GH (2018) Sentinel lymph node biopsy and management of regional lymph nodes in melanoma: American Society of Clinical Oncology and Society of Surgical Oncology clinical practice guideline update. J Clin Oncol 36:399–413. 10.1200/JCO.2017.75.7724 [DOI] [PubMed] [Google Scholar]
- 20.Crompton JG, Gilbert E, Brady MS (2019) Clinical implications of the eighth edition of the American Joint Committee on Cancer melanoma staging. J Surg Oncol 119:168–174. 10.1002/jso.25343 [DOI] [PubMed] [Google Scholar]
- 21.Bahrami A, Barnhill RL (2018) Pathology and genomics of pediatric melanoma: a critical reexamination and new insights. Pediatr Blood Cancer. 10.1002/pbc.26792.10.1002/pbc.26792 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Aldrink JH, Polites S, Lautz TB, Malek MM, Rhee D, Bruny J, Christison-Lagay ER, Tracy ET, Abdessalam S, Ehrlich PF, Dasgupta R, Austin MT (2020) What’s new in pediatric melanoma: An update from the APSA cancer committee. J Pediatr Surg 55:1714–1721. 10.1016/j.jpedsurg.2019.09.036 [DOI] [PubMed] [Google Scholar]
- 23.Morton DL, Thompson JF, Cochran AJ, Mozzillo N, Nieweg OE, Roses DF, Hoekstra HJ, Karakousis CP, Puleo CA, Coventry BJ, Kashani-Sabet M, Smithers BM, Paul E, Kraybill WG, McKinnon JG, Wang HJ, Elashoff R, Faries MB, Group M (2014) Final trial report of sentinel-node biopsy versus nodal observation in melanoma. N Engl J Med 370:599–609. 10.1056/NEJMoa1310460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Murali R, Thompson JF, Scolyer RA (2008) Sentinel lymph node biopsy for melanoma: aspects of pathologic assessment. Future Oncol 4:535–551. 10.2217/14796694.4.4.535 [DOI] [PubMed] [Google Scholar]
- 25.Lorimer PD, White RL, Walsh K, Han Y, Kirks RC, Symanowski J, Forster MR, Sarantou T, Salo JC, Hill JS (2016) Pediatric and adolescent melanoma: a national cancer data base update. Ann Surg Oncol 23:4058–4066. 10.1245/s10434-016-5349-2 [DOI] [PubMed] [Google Scholar]
- 26.Faries MB, Thompson JF, Cochran AJ, Andtbacka RH, Mozzillo N, Zager JS, Jahkola T, Bowles TL, Testori A, Beitsch PD, Hoekstra HJ, Moncrieff M, Ingvar C, Wouters M, Sabel MS, Levine EA, Agnese D, Henderson M, Dummer R, Rossi CR, Neves RI, Trocha SD, Wright F, Byrd DR, Matter M, Hsueh E, MacKenzie-Ross A, Johnson DB, Terheyden P, Berger AC, Huston TL, Wayne JD, Smithers BM, Neuman HB, Schneebaum S, Gershenwald JE, Ariyan CE, Desai DC, Jacobs L, McMasters KM, Gesierich A, Hersey P, Bines SD, Kane JM, Barth RJ, McKinnon G, Farma JM, Schultz E, Vidal-Sicart S, Hoefer RA, Lewis JM, Scheri R, Kelley MC, Nieweg OE, Noyes RD, Hoon DSB, Wang HJ, Elashoff DA, Elashoff RM (2017) Completion dissection or observation for sentinel-node metastasis in melanoma. N Engl J Med 376:2211–2222. 10.1056/NEJMoa1613210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Paradela S, Fonseca E, Pita S, Kantrow SM, Goncharuk VN, Diwan H, Prieto VG (2009) Spitzoid melanoma in children: clinicopathological study and application of immunohistochemistry as an adjunct diagnostic tool. J Cutan Pathol 36:740–752. 10.1111/j.1600-0560.2008.01153.x [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data supporting this study's findings are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request. Data are located in controlled access data storage at St Jude Children's Research Hospital.