Abstract
Rationale and objectives
To develop a prognostic nomogram using mammography data and AJCC staging to predict breast cancer survival.
Materials and methods
A prognostic nomogram was created using data from 1000 women diagnosed with breast cancer at a medical cancer center in Taiwan between 2011 and 2015. The variables included age at diagnosis (≤60 or > 60 years), mammography purpose (screening or diagnostic), mammography modality (digital mammogram or digital breast tomosynthesis), and the 7th American Joint Committee on Cancer (AJCC) stage. The outcome predicted was breast cancer-related mortality. The nomogram utilized Kaplan-Meier analysis for all subsets and Cox proportional hazards regression analysis for prediction. The nomogram's accuracy was internally validated using the concordance index and receiver operating characteristic (ROC) curve analysis, focusing on 3-year and 5-year survival predictions.
Results
Participants' mean age at breast cancer diagnosis was 54 years (SD = 11.2 years). The 1-year, 3-year, and 5-year overall survival (OS) rates were found to be 99.7%, 95.3%, and 91.4%, respectively. The bootstrap-corrected concordance indices indicated the following: nomogram, 0.807 and AJCC, 0.759. A significant difference was observed between the nomogram's area under the curve (AUC) and the AJCC stage in predicting the probability of 5-year survival (p = 0.005). A nomogram, constructed based on mammography and AJCC, demonstrated excellent calibration through internal validation using bootstrapping.
Conclusion
The utilization of a nomogram that incorporates mammography data and the AJCC registry data has been demonstrated to be a reliable predictor of breast cancer survival.
Keywords: Breast cancer, Breast cancer survival, Mammography, AJCC staging, Nomogram
Highlights
-
•
Nomogram predicts breast cancer survival using mammography data, clinical features, AJCC stage.
-
•
Accurate 3- & 5-yr breast cancer survival predictions through nomogram validated by concordance index & ROC analysis.
-
•
Nomogram more reliable than AJCC staging alone in predicting breast cancer survival.
1. Introduction
Breast cancer is a prevalent malignancy and the leading cause of cancer-related mortality globally, accounting for 25% of all cancer cases and 15% of all cancer deaths among women. In Taiwan, specific ethnic and age groups have been found to be disproportionately affected, contributing to over 2000 deaths annually, a number that has been rising in recent years according to local health records [1]. Several clinical and pathological features, as detailed in the nomogram, have been associated with the long-term prognosis of breast cancer [2,3]. Although the AJCC staging system is widely used, individual patient survival can still vary significantly due to other factors [4], such as the poor prognosis for young women under 40 and elderly women over 80 with axillary lymph node negative breast cancer [2]. To address the limitations of the AJCC staging system in capturing individual variations in breast cancer prognosis, solutions could include personalized risk models [5,6], liquid biopsies [7], and age- and subgroup-specific guidelines [8]. Screening mammography has been shown to reduce breast cancer mortality by 41% in Sweden [1], and a study in Taiwan found a similar association with population-based screening mammography [9]. Additionally, very low mammographic breast density has been identified as an independent prognostic feature associated with higher tumor grade and worse survival in newly diagnosed breast cancer patients [10]. Castellano et al. reported a decrease in breast cancer mortality in areas where digital breast tomosynthesis (DBT) screening was implemented compared to conventional digital mammography (DM) [11].
A nomogram has been developed as a statistical tool for estimating a patient's 10-year survival and relapse probability based on factors such as age and comorbidity [12]. However, the potential of mammography data as a predictor of survival has not been extensively researched [13]. However, the use of mammography data as a predictor of survival has been less studied in Asia.
We present a novel nomogram in this study, designed to combine detailed mammography data with existing AJCC staging, enhancing the accuracy of breast cancer survival predictions for patients.
2. Material and methods
The Institutional Review Board of Kaohsiung Veterans General Hospital approved this retrospective study and waived individual informed consent. (IRB number: KSVGH22-CT11-01, Clinical Trials: NCT05600257).
2.1. Study patients and data collection
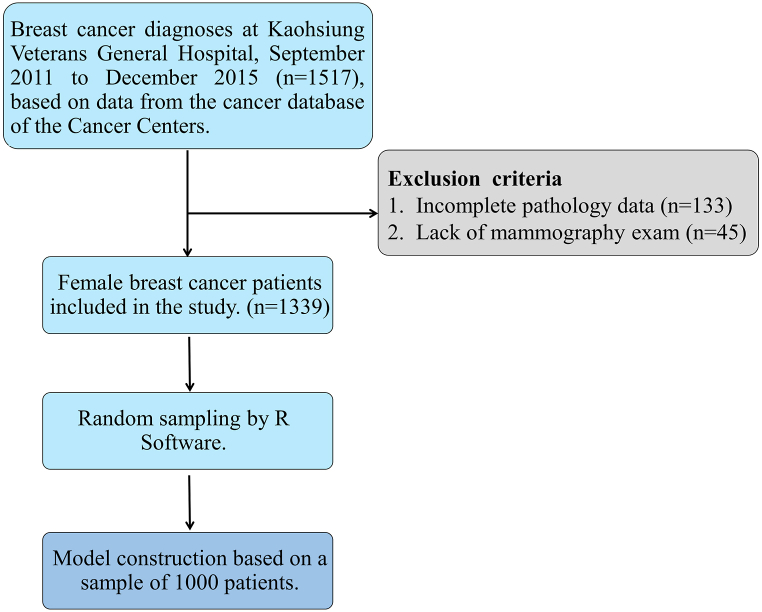
This retrospective study accumulated data from a Cancer Registry Database provided by a Cancer Center of a recognized healthcare institution, as well as the mammography examination registry from the radiology department at a medical center situated in the southern region of an Asian country. A dedicated research assistant was responsible for collecting and collating the data, ensuring that each case from the Cancer Registry Database was accurately matched with corresponding records from the mammography examination registry. The collected data were then meticulously organized and managed in an Excel file, facilitating systematic analysis and data integrity. The analysis included 1517 breast cancer cases diagnosed between September 2011 and December 2015, with 178 cases excluded due to incomplete pathology data for staging in the breast cancer registry (n = 133) or a lack of preoperative mammography examinations (n = 45) (Fig. 1). The final cohort for survival analysis consisted of 1339 patients, with 1000 patients randomly selected for nomogram construction. This research compares two prediction techniques using repeated measurements within a single group of breast cancer patients, eliminating the need for a separate control group.
Fig. 1.
Flowchart of the model selection process.
2.2. Study variables
The following patient clinical information was collected: age at diagnosis (categorized as ≤ 60 or > 60 years), vital status (alive or dead), American Joint Committee on Cancer 7th edition stage (stages 0-IV), mammography purpose (screening or diagnostic), mammography modality (DM or DBT), and BI-RADS category of breast density as determined by mammography using the 5th edition of the American College of Radiology (ACR). The study outcomes were the 3-year and 5-year survival rates of breast cancer patients. The data on breast cancer survival was updated in August 2021, providing up-to-date information on breast cancer outcomes with a maximum 9-year follow-up period.
2.3. Nomogram build-up
A breast cancer survival nomogram was developed using R software version 4.2.2 and RStudio 2022.07.1 build 554. Using univariate and multivariate Cox proportional hazard regression models, variables were examined. The building of the nomogram involved analyzing the association between various predictor variables and time-to-event outcomes in survival analysis [14]. The nomogram aimed to predict the 3-year or 5-year survival rate of patients under two distinct scenarios: scenario 1 relied solely on the 7th AJCC staging system, whereas scenario 2 incorporated all variables under consideration. By comparing the predictions from both scenarios, researchers can better understand the potential impact of additional variables on the accuracy and utility of breast cancer survival predictions, ultimately providing a more comprehensive and informed approach to patient prognosis and management.
2.4. Internal validation
The calibration curves were used to examine the agreement between predicted and observed survival, and the nomogram's 3-year and 5-year overall survival (OS) were internally validated using the concordance index (C-index) and receiver operating characteristic (ROC) curves. The differences in the accuracy of 3-year and 5-year survival predictions between the nomogram using all variables and the nomogram using only the AJCC staging variable were also evaluated. The calibration plot of the nomogram by bootstrapping with one thousand resamples, with the 45° line indicating excellent predictions [15]. The C-index of 1.0 indicates a perfect prediction model, whereas 0.5 indicates just a 50% possibility of accurate prediction [15]. Validation was performed with 1000 bootstraps to correct the C-index and demonstrate variation resulting from over-optimism. The sensitivity and specificity of two scenarios using the nomogram and AJCC system for OS prediction were compared using ROC curves and the area under the receiver operating characteristic curve (AUC) values [16].
2.5. Statistical analysis
Continuous variables were shown as means ± standard deviation (SD). The Kaplan-Meier method, accompanied by the log-rank test, was utilized to generate survival curves. Nomogram development was performed using the “rms” package in R software. A multivariable Cox regression model was developed using clinical and radiological data to predict survival outcomes [14]. To evaluate the predictive performance of a nomogram for survival outcomes in breast cancer patients, the AUC was compared. An AUC value of 0.7 or higher was considered good. Statistical tools for data analysis and hypothesis testing included SPSS and R packages “survival”, “rms”, “ggplot 2”, and “timeROC”. All statistical tests conducted were two-sided, and a significance level of p < 0.05 was used.
3. Result
3.1. Clinicopathological characteristics
The study cohort included 1000 women, of whom 41.3% (n = 413) were diagnosed with breast cancer through screening mammography, while 58.7% (n = 587) underwent diagnostic mammography for symptomatic breast lesions or pre-surgery evaluation (Table 1). The mean age at breast cancer diagnosis was 54 years (SD = 11.2 years). Among the patients, 63.8% (n = 638) received diagnostic mammography before surgery, while 36.2% (n = 362) underwent DBT.
Table 1.
Demographic characteristics of the 1000 patients diagnosed with breast cancer.
| Characteristics | Number N (%) |
|---|---|
| Mean age ± SD, year | 54.0 ± 11.2 |
| Mean survival month ± SD, month | 73.71 ± 21.47 |
| Age group | |
| 60 | 706 (70.2) |
| > 60 | 298 (29.8) |
| Purpose of mammography | |
| Screening | 413 (41.3) |
| Diagnosis | 587 (58.7) |
| Mammographic modality | |
| DM | 638 (63.8) |
| DBT | 362 (36.2) |
| Breast Density (BI-RADS 5th) | |
| Almost entirely fatty | 58 (5.8) |
| Scattered fibroglandular | 126 (12.6) |
| Heterogeneously dense | 700 (70.0) |
| Extremely dense | 116 (11.6) |
| Staging of diagnosis (AJCC 7th) | |
| 0 | 222 (22.2) |
| I | 317 (31.7) |
| II | 329 (32.9) |
| III | 102 (10.2) |
| IV | 30 (3.0) |
Note. - Numbers in parentheses are the percentage.
DM, Digital mammography; DBT, Digital breast tomosynthesis; AJCC, American Joint Committee on Cancer; BI-RADS, Breast Imaging-Reporting and Data System; SD, standard derivation; CI, confidence interval.
The most common breast density observed was heterogeneous density, accounting for 70.0% of cases, followed by scattered fibroglandular density at 12.6%. Regarding cancer stages, stage II was the most frequent at 32.9% (n = 329), followed by stage I at 31.7%, stage 0 at 22.2%, stage III at 10.2%, and stage IV at 3.0%. During the study period, 119 patients died from breast cancer, with the maximum overall survival follow-up time reaching 111 months (mean = 73.3 months). Survival analyses were conducted to further evaluate the relationship between various factors, such as age at diagnosis, breast density, cancer stage, and imaging modality, and their impact on patient outcomes.
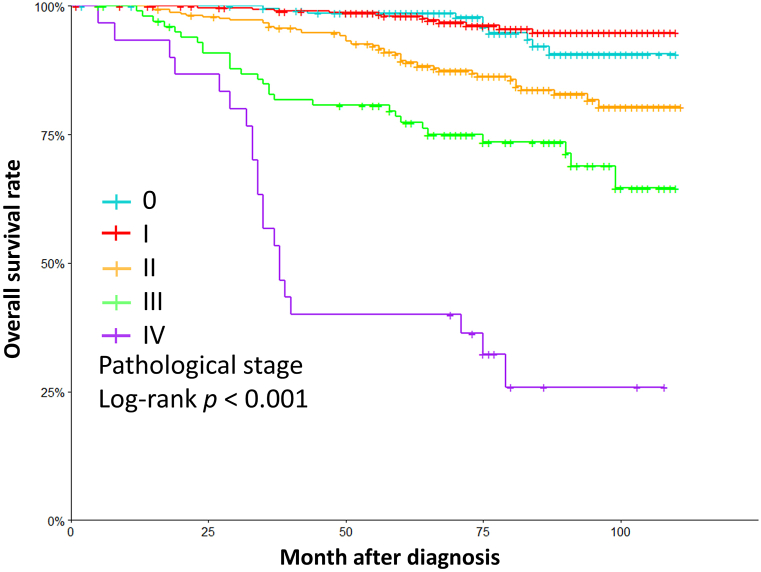
The OS rates for 1-year, 3-year, and 5-year were 99.7% (95% confidence interval [CI] = [99.4%–100%]), 95.3% (95% CI = [94.0%–96.7%]), and 91.4% (95% CI = [89.7%–93.2%]), respectively. The Kaplan-Meier curve analysis of OS in the study population revealed significant differences based on the AJCC pathological stage (stage 0–4, p < 0.001), as seen in Fig. 2. However, no association was found between OS and breast density categories (BI-RADS A-D, p = 0.394).
Fig. 2.
The figure displays Kaplan-Meier (K–M) curves that demonstrate the overall survival (OS) rates for various pathological stages according to the 7th edition of the American Joint Committee on Cancer (AJCC) staging system.
3.2. Prognostic variables
A univariate and multivariate analysis was conducted to identify factors influencing the OS of breast cancer patients (Table 2). The univariate analysis indicated that both screening mammography (hazard ratio [HR] = 0.25, 95% CI = [0.15–0.41], p < 0.001) and DBT (HR = 0.59, 95% CI = [0.39–0.90], p = 0.015) were significantly associated with increased OS. In contrast, age at diagnosis over 60 (HR = 2.80, 95% CI = [1.88–4.17], p < 0.001) and AJCC stages II, III, and IV showed a significant correlation with decreased OS (e.g., stage II vs. stage 0, HR = 3.19, 95% CI = [1.62–6.32], p = 0.001).
Table 2.
Univariate and multivariate cox regression analyses of overall survival.
| Characteristics | N | Univariable Analysis | Multivariable Analysis | ||
|---|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | ||
| Age at diagnosis | |||||
| 60 | 723 | Reference | |||
| > 60 | 277 | 2.60 (1.81–3.74) | <0.001 | 2.80 (1.88–4.17) | <0.001 |
| Mammography purpose | |||||
| Diagnosis | 587 | Reference | |||
| Screening | 413 | 0.25 (0.15–0.41) | <0.001 | 0.48 (0.29–0.81) | 0.006 |
| Mammographic modality | |||||
| DM | 638 | Reference | |||
| DBT | 362 | 0.59 (0.39–0.90) | 0.015 | 0.76 (0.49–1.18) | 0.220 |
| Breast Density (BI-RADS 5th) | |||||
| Almost entirely fatty | 58 | Reference | |||
| Scattered fibroglandular | 126 | 1.07 (0.45–2.56) | 0.881 | 1.36 (0.56–3.28) | 0.499 |
| Heterogeneously dense | 700 | 0.93 (0.43–2.00) | 0.845 | 1.36 (0.61–3.02) | 0.447 |
| Extremely dense | 116 | 0.55 (0.20–1.45) | 0.229 | 1.21 (0.43–3.42) | 0.716 |
| Staging (AJCC 7th) | |||||
| 0 | 222 | Reference | |||
| I | 317 | 0.80 (0.35–1.85) | 0.604 | 0.70 (0.30–1.62) | 0.400 |
| II | 329 | 3.19 (1.62–6.32) | 0.001 | 2.53 (1.27–5.02) | 0.008 |
| III | 102 | 6.49 (3.15–13.36) | <0.001 | 5.30 (2.56–10.98) | <0.001 |
| IV | 30 | 25.83 (12.14–54.96) | <0.001 | 19.93 (9.14–43.49) | <0.001 |
Note. - All variables in univariable analysis were included in multivariable analysis.
DM, Digital mammography; DBT, Digital breast tomosynthesis; AJCC, American Joint Committee on Cancer; BI-RADS, Breast Imaging-Reporting and Data System; HR, Hazard ratio; CI, confidence interval.
The multivariate analysis demonstrated that DBT might influence OS, although the effect was not statistically significant (HR = 0.76, 95% CI = [0.49–1.18], p = 0.220). Other variables, such as age over 60, screening mammography, and AJCC stages II, III, and IV, maintained their significant associations with OS in the multivariate analysis (all p < 0.05). These results indicate that the AJCC stage continued to have a significant impact on determining the outcomes for patients.
3.3. Build nomogram
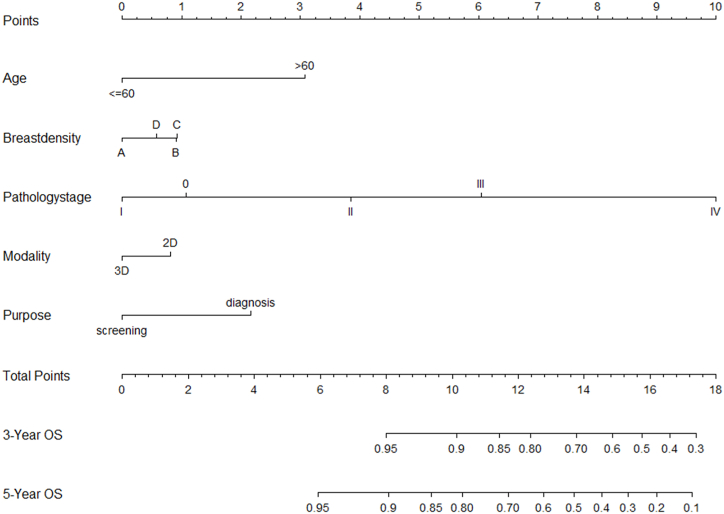
A nomogram was developed to facilitate the prediction of 3-year and 5-year OS for breast cancer patients by assigning scores to each subtype within the identified variables on a continuous scale. The assignment of scores was informed by the strength of the evidence linking each variable to the OS outcome. The weighted total score for each patient was then calculated by combining the scores for all relevant variables. This cumulative score enabled the generation of a predicted probability of 3-year and 5-year OS for individual patients (Fig. 3).
Fig. 3.
A nomogram predicts 3- and 5-year survival rates for breast cancer patients, utilizing vertical lines from each factor to the corresponding point score.
3.4. Nomogram validation
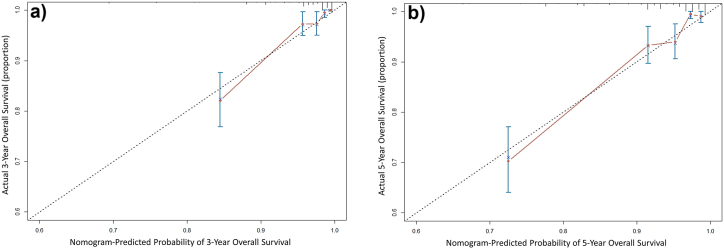
The calibration plot, which compared the predicted OS probabilities to actual observations, demonstrated a high degree of agreement between the nomogram's predictions and real-world outcomes in both 3-year (Fig. 4A) and 5-year (Fig. 4B) analyses. To evaluate the internal validation, the C-index was calculated, resulting in a value of 0.807 (95% CI = [0.787–0.827]). This indicated strong suitability for predicting OS in this patient population. The calibration plots for both 3-year and 5-year OS predictions were well-aligned, further supporting the nomogram's accuracy and reliability as a prognostic tool in clinical practice for breast cancer patients (Fig. 5A).
Fig. 4.
Calibration curves for the nomogram model predict survival rates in breast cancer patients at 3 years (A) and 5 years (B). The nomogram-predicted probability of overall survival is plotted on the x-axis, and the actual overall survival estimated by the Kaplan-Meier method is plotted on the y-axis. The closer the points are to the 45-degree line, the higher the prediction accuracy.
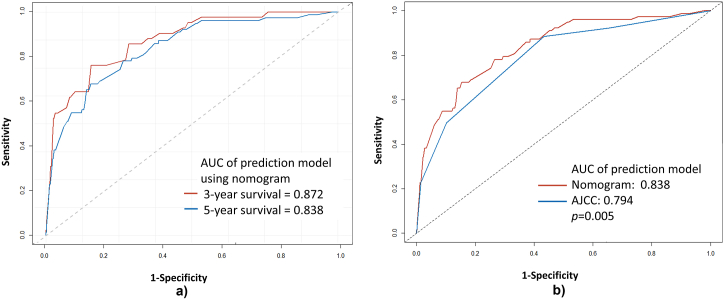
Fig. 5.
Comparisons of prediction performance using receiver operating characteristic (ROC) curves. (A) Model accuracy in predicting 3-year and 5-year survival rates using a nomogram. (B) Model accuracy in predicting 5-year survival rates using a nomogram and the 7th edition of the American Joint Committee on Cancer (AJCC) staging system. The x-axis represents 1-Specificity, while the y-axis represents Sensitivity.
The C-index utilizing only AJCC stage data was 0.759, lower than the nomogram's C-index of 0.807. The ROC curves for 3-year and 5-year OS demonstrated that the nomogram's AUC values (3-year: 0.872, 95% CI = [0.818–0.925]; 5-year: 0.838, 95% CI = [0.790–0.885]) were larger than those of the AJCC stage (3-year: 0.851, 95% CI = [0.789–0.911]; 5-year: 0.794, 95% CI = [0.741–0.845]) (Fig. 5B). This comparison reveals the nomogram's significantly higher AUC in predicting the probability of 5-year survival (p = 0.005) compared to the AJCC stage. However, no significant difference was found between the two predictive methods for 3-year survival (p = 0.227).
4. Discussion
The study focused on developing a prognostic nomogram for breast cancer by analyzing data from 1000 randomly selected cases, obtained from a Cancer Registry and correlated with mammography data from a medical center in Asia. The nomogram, which incorporated variables such as age, purpose and modality of mammography, and AJCC stage, was specifically designed to predict breast cancer-related mortality. Statistical analysis identified key factors-age over 60, AJCC stage, mammographic modality, and exam purpose - as significantly influencing 5-year OS rates in breast cancer patients. Consequently, a new nomogram was constructed using these determinants, and its predictive accuracy for 5-year OS was confirmed through calibration plots and ROC curves, demonstrating its efficacy.
Previous research has established that biomarkers with AUC values ranging from 0.8 to 0.9 are considered to demonstrate good performance [16]. In this study, the nomogram developed for predicting 3- and 5-year survival yielded AUCs of 0.872 and 0.838, respectively, suggesting moderate accuracy [16]. The nomogram serves as a valuable tool for clinicians, enabling them to better comprehend the influence of each variable on breast cancer patients' prognosis. Furthermore, this nomogram may facilitate the identification of patients at higher risk of mortality, warranting closer monitoring and more aggressive treatment strategies to improve patient outcomes. The integration of such predictive models in clinical practice could significantly enhance the overall management of breast cancer patients and contribute to personalized treatment approaches.
Mammography data has been primarily used to predict malignancy risk, with fewer studies focusing on survival outcomes [17]. One study developed a nomogram to predict malignancy risk in microcalcifications detected on mammography, considering four variables such as menopausal status, maximum diameter, distribution, and morphology of malignant microcalcifications, with an AUC of 0.839 [18]. Another group of researchers made a nomogram to predict the risk of malignant upgrade in women diagnosed with atypical ductal hyperplasia (ADH) on core-needle biopsy, taking into account mammographic parenchymal density, the presence of a mass on ultrasound, and the number of ADH foci, with a C-statistic of 0.81, allowing low-risk women to avoid surgical excision [19]. The current study applied straightforward algorithm and clinical history data from mammography exams and pathological stages to create an accessible and easily understood nomogram for use in clinical practice, offering valuable information to both healthcare providers and cancer patients.
For the past three decades, mammography has been the major tool implemented in breast cancer screening programs, as it is the only method proven to reduce breast cancer mortality rates [20]. Research indicates that mammography can decrease breast cancer mortality by 20–35% in women over 50 years of age [20]. In Taiwan, a nationwide biennial mammography screening program for women aged 40–69 was introduced in July 2004, leading to a decrease in breast cancer mortality [9].
An observational study reported a significant increase in breast cancer detection rates, from 4.0 to 5.4 per 1000 screenings, following the introduction of breast tomosynthesis into clinical screening practice [21]. Previous research has also demonstrated the long-term benefits of DBT screening compared to DM [11]. In the present study, the purpose and modality of mammography emerged as important predictors for determining long-term outcomes, highlighting the importance of effective breast cancer screening methods in improving patient prognosis.
The 8th AJCC staging system introduced a dual staging approach, incorporating both anatomical and prognostic staging systems for breast cancer patients [22]. The prognostic staging system considers various biological biomarkers, including estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2) status, and nuclear grade, and recommends the routine use of the Oncotype multi-gene assay for patients with T1-2, N0, M0, ER+, and HER2-breast cancer [22]. The performance of these staging systems, assessed using the C-index, yielded values of 0.767 for the anatomical staging system (7th AJCC) and 0.814 for the prognostic staging system (7th edition AJCC with prognostic factors) [22].
Considering this, we launched a study using the 7th AJCC staging system, combined with clinical and mammography data, to enhance the prediction of survival outcomes for breast cancer patients. In our study, the performance of these staging systems, assessed using the C-index, yielded values of 0.759 for the 7th edition AJCC and 0.807 for the nomogram, indicating that incorporating prognostic factors like mammography data into the staging system can also improve its predictive capacity.
The study's theoretical strength is rooted in its innovative integration of traditional AJCC staging with detailed clinical and mammography data, creating a more comprehensive approach to improve breast cancer survival predictions and offering a nuanced assessment of prognosis. The study's methodology is characterized by its rigorous analysis of a large, carefully compiled dataset from a Cancer Registry and mammography records, employing advanced statistical tools such as calibration plots and ROC curves for comprehensive validation. The C-index showed enhanced predictive capacity, with values of 0.759 for the 7th edition AJCC and 0.807 for the nomogram, indicating the benefits of incorporating additional prognostic factors in patient management and treatment strategies.
Incorporating mammography imaging and clinical characteristics into a nomogram for prognosis prediction offers advantages, such as increased accuracy and a comprehensive understanding of a patient's health [23,24]. However, it also presents disadvantages, including increased complexity for interpretation, potential challenges in applying the nomogram in practice, and variability or discrepancies in data quality, which could affect overall reliability.
There are limitations to using mammography imaging and clinical characteristics for prognosis prediction in a nomogram. Mammography may not be available or feasible for all patients, and its quality and interpretation accuracy can vary [25]. Including clinical characteristics may introduce bias due to varying patient information availability. Despite these limitations, mammography is widely used in breast cancer screening and evaluating pre-surgery conditions, with information easily accessible through patient records [26]. Future research can explore imaging patterns from mammography and integrate other imaging techniques, such as contrast-enhanced mammography, MRI, and ultrasound, to enhance the accuracy of breast cancer survival predictions.
5. Limitation
This retrospective study has potential limitations. Firstly, the 9-year study data came from a single medical center, possibly affecting the generalizability of the findings. However, our hospital has a well-established breast cancer registry, with dedicated screening program using DBT mammography since 2011 and regular patient follow-up, enhancing data quality and reliability. Secondly, the retrospective design of the study introduced inherent biases, such as incomplete or imprecise pathology data, resulting in the exclusion of some participants. Despite these limitations and a sample of 1000 patients, the study provides valuable insights into breast cancer survival prediction using a nomogram incorporating the AJCC staging system and additional variables. Thirdly, this study relied on the 7th edition of AJCC due to data available period (2011–2015). The more recent 8th AJCC includes additional prognostically relevant tumor features and molecular biomarkers. Subsequent research integrating mammography data with the 8th edition of AJCC staging system could better assess the nomogram's value. Future studies in different healthcare settings and groups of people could confirm the nomogram's accuracy and usefulness. These studies would combine mammograms with newer tumor details and markers to refine the nomogram and personalize breast cancer treatment.
6. Conclusion
Participants diagnosed with breast cancer showed a 5-year overall survival rate of 91.4%; the prognostic nomogram proved more accurate than the AJCC in predicting 5-year survival for breast cancer survival, evidenced by a significant difference in the AUC.
Data availability statement
Data sharing is restricted by the Kaohsiung Veterans General Hospital IRB due to strict regulations for ethical issues. Access to the data is contingent upon a data sharing agreement approved by the Kaohsiung Veterans General Hospital IRB. (IRB number: KSVGH22-CT11-01, Clinical Trials: NCT05600257).
Funding sources
This work was supported by grants from Kaohsiung Veterans Hospital Research Found (VGHKS110-121).
CRediT authorship contribution statement
Zi-Han Yu: Writing – original draft. Yun Lin: Writing – original draft. Pei-Shan Wu: Data curation. Chao-Hsien Lee: Data curation. Chen-Pin Chou: Writing – review & editing, Writing – original draft.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This work was supported by grants from Kaohsiung Veterans General Hospital Research Found (KSVGH111-109).
References
- 1.Duffy S.W., Tabar L., Yen A.M., Dean P.B., Smith R.A., Jonsson H., Tornberg S., Chen S.L., Chiu S.Y., Fann J.C., Ku M.M., Wu W.Y., Hsu C.Y., Chen Y.C., Svane G., Azavedo E., Grundstrom H., Sunden P., Leifland K., Frodis E., Ramos J., Epstein B., Akerlund A., Sundbom A., Bordas P., Wallin H., Starck L., Bjorkgren A., Carlson S., Fredriksson I., Ahlgren J., Ohman D., Holmberg L., Chen T.H. Mammography screening reduces rates of advanced and fatal breast cancers: results in 549,091 women. Cancer. 2020;126(13):2971–2979. doi: 10.1002/cncr.32859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brandt J., Garne J.P., Tengrup I., Manjer J. Age at diagnosis in relation to survival following breast cancer: a cohort study. World J. Surg. Oncol. 2015;13:33. doi: 10.1186/s12957-014-0429-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wang Z., Xing L., Luo X., Ren G. A nomogram for survival prediction in 275,812 U.S. patients with breast cancer: a population-based cohort study based on the SEER database. Gland Surg. 2022;11(7):1166–1179. doi: 10.21037/gs-22-321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wu S.G., Wang J., Lian C.L., Lei J., Hua L., Lin Q., Chen Y.X., He Z.Y. Evaluation of the 8th edition of the American joint committee on cancer's pathological staging system in prognosis assessment and treatment decision making for stage T1-2N1 breast cancer after mastectomy. Breast. 2020;51:2–10. doi: 10.1016/j.breast.2020.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chavez‐MacGregor M., Mittendorf E.A., Clarke C.A., Lichtensztajn D.Y., Hunt K.K., Giordano S.H. Incorporating tumor characteristics to the American Joint Committee on Cancer breast cancer staging system. Oncol. 2017;22(11):1292–1300. doi: 10.1634/theoncologist.2017-0116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kantor O., Niu J., Zhao H., Giordano S.H., Hunt K.K., King T.A., Mittendorf E.A., Chavez-MacGregor M. Comparative analysis of proposed strategies for incorporating biologic factors into breast cancer staging. Ann. Surg Oncol. 2020;27:2229–2237. doi: 10.1245/s10434-019-08169-y. [DOI] [PubMed] [Google Scholar]
- 7.Cayrefourcq L., Alix-Panabieres C. Clinical relevance of liquid biopsy in breast cancer: update in 2020. Expert Rev. Mol. Diagn. 2020;20(9):913–919. doi: 10.1080/14737159.2020.1816828. [DOI] [PubMed] [Google Scholar]
- 8.Shieh Y., Eklund M., Madlensky L., Sawyer S.D., Thompson C.K., Stover Fiscalini A., Ziv E., van’t Veer L.J., Esserman L.J., Tice J.A. Breast cancer screening in the precision medicine era: risk-based screening in a population-based trial. J. Natl. Cancer Inst. 2017;109(5):djw290. doi: 10.1093/jnci/djw290. [DOI] [PubMed] [Google Scholar]
- 9.Yen A.M., Tsau H.S., Fann J.C., Chen S.L., Chiu S.Y., Lee Y.C., Pan S.L., Chiu H.M., Kuo W.H., Chang K.J., Wu Y.Y., Chuang S.L., Hsu C.Y., Chang D.C., Koong S.L., Wu C.Y., Chia S.L., Chen M.J., Chen H.H., Chiou S.T. Population-based breast cancer screening with risk-based and universal mammography screening compared with clinical breast examination: a propensity score analysis of 1 429 890 Taiwanese women. JAMA Oncol. 2016;2(7):915–921. doi: 10.1001/jamaoncol.2016.0447. [DOI] [PubMed] [Google Scholar]
- 10.Masarwah A., Auvinen P., Sudah M., Rautiainen S., Sutela A., Pelkonen O., Oikari S., Kosma V.M., Vanninen R. Very low mammographic breast density predicts poorer outcome in patients with invasive breast cancer. Eur. Radiol. 2015;25(7):1875–1882. doi: 10.1007/s00330-015-3626-2. [DOI] [PubMed] [Google Scholar]
- 11.Castellano C.R., Aguilar Angulo P.M., Hernandez L.C., Gonzalez-Carrato P.S., Gonzalez R.G., Alvarez J., Chacon J.I., Ruiz J., Fuentes Guillen M.A., Gutierrez Avila G. Breast cancer mortality after eight years of an improved screening program using digital breast tomosynthesis. J. Med. Screen. 2021;28(4):456–463. doi: 10.1177/09691413211002556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Balachandran V.P., Gonen M., Smith J.J., DeMatteo R.P. Nomograms in oncology: more than meets the eye. Lancet Oncol. 2015;16(4):e173–e180. doi: 10.1016/S1470-2045(14)71116-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gill P.G., Farshid G., Luke C.G., Roder D.M. Detection by screening mammography is a powerful independent predictor of survival in women diagnosed with breast cancer. Breast. 2004;13(1):15–22. doi: 10.1016/S0960-9776(03)00169-3. [DOI] [PubMed] [Google Scholar]
- 14.Iasonos A., Schrag D., Raj G.V., Panageas K.S. How to build and interpret a nomogram for cancer prognosis. J. Clin. Oncol. : official journal of the American Society of Clinical Oncology. 2008;26(8):1364–1370. doi: 10.1200/JCO.2007.12.9791. [DOI] [PubMed] [Google Scholar]
- 15.Pan X., Yang W., Chen Y., Tong L., Li C., Li H. Nomogram for predicting the overall survival of patients with inflammatory breast cancer: a SEER-based study. Breast. 2019;47:56–61. doi: 10.1016/j.breast.2019.05.015. [DOI] [PubMed] [Google Scholar]
- 16.Xia J., Broadhurst D.I., Wilson M., Wishart D.S. Translational biomarker discovery in clinical metabolomics: an introductory tutorial. Metabolomics : Official journal of the Metabolomic Society. 2013;9(2):280–299. doi: 10.1007/s11306-012-0482-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Youk J.H., Gweon H.M., Son E.J., Eun N.L., Choi E.J., Kim J.A. Scoring system to stratify malignancy risks for mammographic microcalcifications based on breast imaging reporting and data system 5th edition descriptors. Korean J. Radiol. 2019;20(12):1646–1652. doi: 10.3348/kjr.2019.0262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chen L., Duan H.Y., Tang X.M., Ma C.C., Yang L., Xie Z.Y., Gao Z.Z., Chen J.F. A mammography-based nomogram for prediction of malignancy in breast suspicious calcification. Acad. Radiol. 2022;29(7):1022–1028. doi: 10.1016/j.acra.2021.09.003. [DOI] [PubMed] [Google Scholar]
- 19.Bong T.S.H., Tan J.K.T., Ho J.T.S., Tan P.H., Lau W.S., Tan T.M., Wong J.S.L., Tan V.K.M., Tan B.K.T., Madhukumar P., Yong W.S., Lim S.Z., Wong C.Y., Ong K.W., Sim Y. Atypical ductal hyperplasia of the breast on core needle biopsy: risk of malignant upgrade on surgical excision. Journal of breast cancer. 2022;25(1):37–48. doi: 10.4048/jbc.2022.25.e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fletcher S.W., Elmore J.G. Clinical practice. Mammographic screening for breast cancer. N. Engl. J. Med. 2003;348(17):1672–1680. doi: 10.1056/NEJMcp021804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rose S.L., Tidwell A.L., Bujnoch L.J., Kushwaha A.C., Nordmann A.S., Sexton R., Jr. Implementation of breast tomosynthesis in a routine screening practice: an observational study. AJR. American journal of roentgenology. 2013;200(6):1401–1408. doi: 10.2214/AJR.12.9672. [DOI] [PubMed] [Google Scholar]
- 22.Abdel-Rahman O. Validation of the 8th AJCC prognostic staging system for breast cancer in a population-based setting. Breast Cancer Res. Treat. 2018;168(1):269–275. doi: 10.1007/s10549-017-4577-x. [DOI] [PubMed] [Google Scholar]
- 23.Evans D.G.R., Harkness E.F., Brentnall A.R., van Veen E.M., Astley S.M., Byers H., Sampson S., Southworth J., Stavrinos P., Howell S.J., Maxwell A.J., Howell A., Newman W.G., Cuzick J. Breast cancer pathology and stage are better predicted by risk stratification models that include mammographic density and common genetic variants. Breast Cancer Res. Treat. 2019;176(1):141–148. doi: 10.1007/s10549-019-05210-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Momenimovahed Z., Tiznobaik A., Taheri S., Hassanipour S., Salehiniya H. A review of barriers and facilitators to mammography in Asian women. Ecancermedicalscience. 2020;14:1146. doi: 10.3332/ecancer.2020.1146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Esserman L., Cowley H., Eberle C., Kirkpatrick A., Chang S., Berbaum K., Gale A. Improving the accuracy of mammography: volume and outcome relationships. J. Natl. Cancer Inst. 2002;94(5):369–375. doi: 10.1093/jnci/94.5.369. [DOI] [PubMed] [Google Scholar]
- 26.Mariscotti G., Houssami N., Durando M., Bergamasco L., Campanino P.P., Ruggieri C., Regini E., Luparia A., Bussone R., Sapino A., Fonio P., Gandini G. Accuracy of mammography, digital breast tomosynthesis, ultrasound and MR imaging in preoperative assessment of breast cancer. Anticancer Res. 2014;34(3):1219–1225. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is restricted by the Kaohsiung Veterans General Hospital IRB due to strict regulations for ethical issues. Access to the data is contingent upon a data sharing agreement approved by the Kaohsiung Veterans General Hospital IRB. (IRB number: KSVGH22-CT11-01, Clinical Trials: NCT05600257).