Abstract
Background
Patients with paraneoplastic syndromes (PNS) are excluded from clinical trials involving immune checkpoint inhibitors (ICIs) due to safety concerns. Moreover, real-world data on efficacy and safety is scarce.
Methods
In this retrospective study, data were collected on patients with PNS and solid tumors receiving ICI between 2015 and 2022 at nine institutions. Patients were classified into: Cohort 1 (pre-existing PNS before ICI initiation), cohort 2 (PNS during ICI treatment), and cohort 3 (PNS after ICI discontinuation). Patients with metastatic non-small cell lung cancer (NSCLC) (mNSCLC) from cohort 1 were matched to patients who were PNS-free at each institution up to a 1:3 ratio for age, sex, type of ICI, use of concurrent chemotherapy, and number of lines of systemic therapy prior to ICI initiation. Kaplan-Meier method was used to assess overall survival (OS) and time-to-next treatment (TTNT).
Results
Among 109 patients with PNS treated with ICIs, median age at ICI initiation was 67 years (IQR: 58–74). The most represented cancer type was NSCLC (n=39, 36%). In cohort 1 (n=55), PNS exacerbations occurred in 16 (29%) patients with median time to exacerbation after ICI of 1.1 months (IQR: 0.7–3.3). Exacerbation or de novo PNS prompted temporary/permanent interruption of ICIs in 14 (13%) patients. For cohort 2 (n=16), median time between ICI initiation and de novo PNS was 1.2 months (IQR: 0.4–3.5). Treatment-related adverse events (trAEs) occurred in 43 (39%) patients. Grade ≥3 trAEs occurred in 18 (17%) patients. PNS-directed immunosuppressive therapy was required in 55 (50%) patients. We matched 18 patients with mNSCLC and PNS (cohort 1) to 40 without PNS, treated with ICIs. There was no significant difference in OS or TTNT between patients with mNSCLC with and without PNS, although a trend was seen towards worse outcomes in patients with PNS. TrAEs occurred in 6/18 (33%) and 14/40 (35%), respectively. Grade ≥3 trAEs occurred in 4 (22%) patients with PNS and 7 (18%) patients without PNS.
Conclusions
Exacerbations of pre-existing PNS occurred in 29% of patients treated with ICIs and both exacerbations and de novo PNS occur early in the ICI course. TrAE from ICIs were similar between patients with and without PNS. Our data suggest that pre-existing PNS should not preclude consideration of ICI therapy although patients may not derive the same clinical benefit compared with patients without PNS.
Keywords: PARANEOPLASTIC SYNDROME, Immune Checkpoint Inhibitors, Non-Small Cell Lung Cancer
WHAT IS ALREADY KNOWN ON THIS TOPIC
Patients with pre-existing paraneoplastic syndromes (PNS) have traditionally been excluded from clinical trials using immune checkpoint inhibitors (ICIs). Data on ICI safety and efficacy in this unique population is scarce.
WHAT THIS STUDY ADDS
The exacerbation of pre-existing PNS and the development of de novo PNS typically occur early on during ICI treatment. Patients with PNS and non-small cell lung cancer have similar adverse event profiles compared with patients without PNS.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
The data provided in this study will aid clinicians in counseling patients treated with ICIs on their safety, efficacy, and impact on pre-existing PNS. It will also provide further evidence to support the inclusion of patients with certain PNS in ICI clinical trials.
Introduction
Paraneoplastic syndromes (PNS) arise due to the activity of ectopic hormones produced by cancer cells1 or autoimmune-mediated death of normal cells that share common epitopes with cancer cells.2 PNS impact up to 8% of patients with cancer3 and can affect different organ systems such as the nervous system (eg, limbic encephalitis), skin (eg, paraneoplastic pemphigus), and musculoskeletal system (eg, dermatomyositis), among others.4 The most common solid tumor types that are associated with PNS are lung cancer, breast cancer, and gynecologic tumors.3 Survival outcomes are usually related to the underlying cancer, but PNS may lead to severe disability and are even fatal at times with profound effects on the patients’ quality of life and survival.5
Two treatment approaches for PNS have been suggested including tumor-directed therapy and suppression of the immune response (eg, steroids, plasma exchange, intravenous immunoglobulin).1 For most PNS, the former approach is the only effective treatment while the latter can be leveraged for immune-mediated processes.
Among tumor-directed therapies, immune checkpoint inhibitors (ICIs) have transformed the treatment paradigm for patients with cancer6–13; however it is not clear how these advances translate among patients with PNS. The presence of an aberrant immune system is a hallmark of PNS.14 Abnormal expression of shared antigens between tumor and normal cells, accompanied by potentiated T-cell responses after ICI treatment may contribute to PNS worsening.15 In theory, ICIs may induce a robust immune response and tilt an underlying immune-mediated PNS beyond a critical threshold, which in turn, can lead to serious clinical manifestations. As such, it is important to evaluate the impact of ICIs on them, and whether the presence of PNS may or may not hinder therapeutic opportunities for patients with cancer.
Unfortunately, patients with pre-existing autoimmune diseases, including PNS, have traditionally been excluded from clinical trials involving ICIs due to concerns of exacerbation of the underlying autoimmune component.16 In a multi-institutional effort, we herein assemble an international retrospective cohort to study the safety and clinical outcomes of ICIs in patients with solid tumors and PNS.
Methods
Patient cohort
In a retrospective multicenter database, clinical information was gathered from nine participating institutions in the USA and Europe (online supplemental table 1) and data was analyzed at Dana-Farber Cancer Institute (DFCI).
jitc-2023-008724supp001.xlsx (2MB, xlsx)
Patients with solid tumors had to meet all the following criteria for inclusion: (1) Diagnosed with at least 1 of 22 predefined PNS: Cushing’s syndrome, hyperparathyroidism, secondary polycythemia from malignancy, acquired ichthyosis, acquired hypertrichosis lanuginosa, dermatopolymyositis, paraneoplastic pemphigus, acrodermatitis continua, nephritic or nephrotic syndrome, hypertrophic pulmonary osteoarthropathy, polymyalgia rheumatica, synovitis/tenosynovitis, encephalitis/encephalomyelitis, Lambert Eaton myasthenic syndrome, or myasthenia gravis. PNS were diagnosed by specialists (example neurologist diagnosing neurologic PNS). Each case of PNS was adjudicated by an expert (2) had a biopsy-proved solid tumor. (3) Received at least one dose of ICI therapy, defined as anti-programmed cell death protein 1/ligand 1 (PD-1/L-1) and/or anti-cytotoxic T-lymphocyte-associated antigen-4 alone or in combination with chemotherapy or tyrosine kinase inhibitors between 2015 and 2022.
Patients with PNS were classified into one of three observational cohorts. Cohort 1 consisted of patients diagnosed with a PNS prior to the initiation of ICI. Cohort 2 comprised patients diagnosed with a PNS during ICI treatment. Cohort 3 included patients with PNS diagnosed after cessation of ICI.
Clinical outcomes and toxicity profiles
The primary endpoint of this study was safety of ICI therapy in terms of (1) treatment-related adverse events (trAEs): graded per the Common Terminology Criteria for Adverse Events (CTCAE) V.5.0 and (2) worsening of pre-existing PNS for patients in cohort 1. The secondary endpoints were (1) time-to-next treatment (TTNT): the time from ICI initiation to next line of systemic therapy, death, or censored on the date of last follow-up (2) objective response rates (ORRs) measured either by the clinical investigator or whenever possible per the Response Evaluation Criteria in Solid Tumors (RECIST) V.1.1 criteria for solid tumors. In addition, the time interval between the initiation of ICI treatment and the exacerbation (cohort 1) or appearance (cohort 2) of a PNS, the frequency of PNS exacerbation (cohort 1), and the treatment regimens for the management of PNS were reported. Overall survival (OS) was considered an exploratory endpoint and was defined as the date of ICI initiation to death or censored at the date of last follow-up.
Matched cohort of non-small cell lung cancer with metastatic non-small cell lung cancer
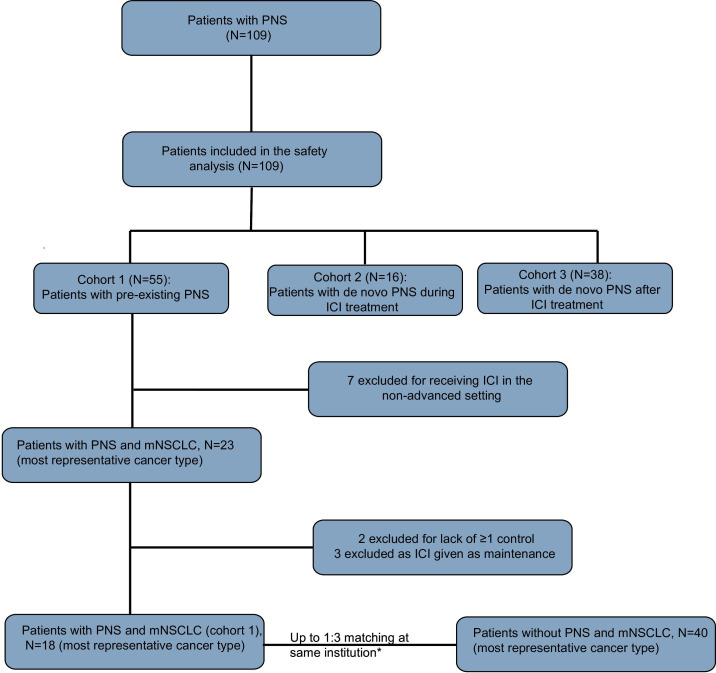
To evaluate the impact of the presence of a PNS on clinical outcomes and safety profiles, we assembled a matched cohort. We matched patients with metastatic non-small cell lung cancer (mNSCLC), the most common cancer type in this cohort of patients, and PNS from cohort 1 to a group of patients with mNSCLC without any history of PNS. PNS and non-PNS mNSCLC were matched at each participating institution up to a 1:3 ratio based on the available number of matches. Matching was performed for all of the following variables: (1) 10-year age groups (41–50, 51–60, 61–70, 71–80), (2) sex, (3) class of ICI, (4) use of concurrent chemotherapy (yes/no), and (5) number of lines of systemic therapy prior to ICI initiation. The matched cohort included a subset of 18 patients with mNSCLC and PNS from cohorts 1 and 40 matched mNSCLC without PNS across six institutions. A flowchart of the study design is represented in figure 1.
Figure 1.
Consolidated Standards of Reporting Trials diagram. *Matched variables are: sex, age group, ICI class, use of chemotherapy, and prior lines of systemic therapy. ICI, immune checkpoint inhibitor; mNSCLC, metastatic non-small cell lung cancer; PNS, paraneoplastic syndromes.
Statistical analysis
Descriptive statistics were used to summarize patient characteristics and clinical outcomes. TrAEs were reported and graded as per the CTCAE V.5.0. OS and TTNT were estimated using the Kaplan-Meier method and log-rank tests were performed to compare them among categorical subcohorts. For ORR, responders were defined as patients with partial response (PR) or complete response (CR) at any time after the initiation of ICI and prior to starting another line of therapy. Statistical analyses were performed using SAS V.9.4.
Results
Cohort characteristics
Among 109 patients with PNS treated with ICIs, the median age was 67 years (IQR: 58–73). The most represented cancer types were NSCLC (n=39, 36%), renal cell carcinoma (RCC, n=20, 18%) and small cell lung cancer (SCLC, n=17, 16%, table 1). The majority were white (n=100, 92%, table 1). The median follow-up time was 39 months (95% CI: 26 to 44). ICI treatment was administered in the stage IV setting for 78% (n=85) of patients, while the remainder received ICIs either in the extensive-stage SCLC (n=17, 16%) or stage III setting (n=6, 6.1%) (table 1, online supplemental table 2). ICIs were administered in the second line setting or beyond in 53 (49%) patients. The most common ICI regimens were single agent anti-PD-1 or anti-PD-L1, n=64, 59%), chemoimmunotherapy (n=23, 21%), and dual anti-PD-1+ anti-CTLA-4 (n=14, 13%). Of 96 patients who discontinued treatment, the main reasons were progression or death in 55 of 96 (57%), treatment-related toxicity in 22 (20%), and PNS exacerbation in 14 (13%). Baseline demographics and clinical characteristics of the overall cohort are shown in table 1.
Table 1.
Baseline characteristics of patients with paraneoplastic syndrome (PNS)
| Total (N=109) | Patients with pre-existing PNS (cohort 1, N=55) | Patients with de-novo PNS during ICI treatment (cohort 2, N=16) | Patients with de novo PNS after ICI discontinuation (cohort 3, N=38) | |
| N (%) | N (%) | N (%) | N (%) | |
| Age at ICI initiation | ||||
| Median (IQR) | 67 (58–74) | 66 (58–75) | 69 (63–79) | 67 (56–71) |
| Sex | ||||
| Females | 46 (42) | 21 (38) | 4 (25) | 21 (55) |
| Males | 63 (58) | 34 (62) | 12 (75) | 17 (45) |
| Race | ||||
| White | 100 (92) | 49 (89) | 14 (93) | 37 (97) |
| Other* | 8 (7.4) | 6 (11) | 1 (6.7) | 1 (2.6) |
| Not reported† | 1 | 0 | 1 | 0 |
| Ethnicity | ||||
| Hispanic/Latinx | 5 (4.6) | 1 (1.9) | 3 (19) | 1 (2.6) |
| Non-Hispanic/non-Latinx | 103 (95) | 53 (98) | 13 (81) | 37 (97) |
| Not reported† | 1 | 1 | 0 | 0 |
| Region | ||||
| North America | 104 (95) | 50 (91) | 16 (100) | 38 (100) |
| Europe | 5 (4.6) | 5 (9.1) | 0 | 0 |
| Smoking | ||||
| Current/former | 76 (70) | 39 (72) | 11 (69) | 26 (68) |
| Never | 32 (30) | 15 (28) | 5 (31) | 12 (32) |
| Not reported† | 1 | 1 | 0 | 0 |
| Type of malignancy | ||||
| NSCLC | 39 (36) | 23 (42) | 8 (50) | 8 (21) |
| RCC | 20 (18) | 6 (11) | 3 (19) | 11 (29) |
| SCLC | 17 (16) | 8 (15) | 3 (19) | 6 (16) |
| HNSCC | 5 (4.6) | 3 (5.5) | 0 (0.0) | 2 (5.3) |
| Melanoma | 5 (4.6) | 3 (5.5) | 1 (6.3) | 1 (2.6) |
| UC | 3 (2.8) | 1 (1.8) | 0 (0.0) | 2 (5.3) |
| Non-melanoma skin cancer | 3 (2.8) | 3 (5.5) | 0 (0.0) | 0 (0.0) |
| Other | 17 (16) | 8 (15) | 1 (6.3) | 8 (21) |
| No. of systemic therapy lines prior to ICI initiation | ||||
| 0 | 56 (51) | 28 (51) | 14 (88) | 14 (37) |
| 1 | 37 (34) | 20 (36) | 1 (6.3) | 16 (42) |
| ≥2 | 16 (15) | 7 (13) | 1 (6.3) | 8 (21) |
| ICI regimen used | ||||
| Anti-PD-1/anti-PD-L1 monotherapy | 64 (59) | 33 (60) | 7 (44) | 24 (63) |
| Anti-PD-1/anti-PD-L1 + chemotherapy | 23 (21) | 12 (20) | 5 (31) | 6 (16) |
| Anti-PD-1 + anti-CTLA-4 | 13 (12) | 5 (9.1) | 3 (19) | 5 (13) |
| Anti-PD-1/anti-PD-L1 + targeted agents | 8 (7.3) | 4 (7.3) | 1 (6.3) | 3 (7.9) |
| Anti-PD-1 + anti-CTLA-4 + targeted agent | 1 (0.9) | 1 (1.8) | 0 (0.0) | 0 (0.0) |
| ECOG at ICI initiation | ||||
| 0 | 26 (24) | 9 (17) | 3 (20) | 14 (37) |
| 1 | 47 (44) | 25 (46) | 10 (67) | 12 (32) |
| ≥2 | 34 (32) | 20 (37) | 2 (13) | 12 (32) |
| Not reported† | 2 | 1 | 1 | 0 |
| Stage at ICI initiation | ||||
| III | 7 (6.4) | 5 (9.1) | 1 (6.3) | 1 (2.6) |
| IV | 85 (78) | 42 (76) | 15 (94) | 37 (93) |
| Extensive-stage SCLC | 17 (16) | 8 (15) | 3 (19) | 6 (16) |
*This group included patients who self-identified as Asian (n=4), black (n=2), or other unspecified race (n=2).
†Missing values were not included in the denominator for calculation of % in subgroups.
CTLA-4, cytotoxic T-lymphocyte-associated antigen-4; ECOG, Eastern Cooperative Oncology Group; HNSCC, head and neck squamous cell carcinoma; ICI, immune checkpoint inhibitor; NSCLC, non-small cell lung cancer; PD-1, programmed cell death protein 1; PD-L1, programmed cell death ligand 1; PNS, paraneoplastic syndrome; RCC, renal cell carcinoma; SCLC, small cell lung cancer; UC, urothelial carcinoma.
Types of PNS and their manifestations
Neurologic PNS were the most common (n=32, 29%, table 2), followed by endocrine PNS (n=27, 24%) and rheumatologic PNS (n=20, 18%). The most common individual PNS subtype was hypercalcemia of malignancy, of which all 22 patients had elevated parathyroid hormone-related protein (PTHrP). Using PNS-specific panels, neuronal antibodies were detected in 20/32 (63%, (online supplemental table 2) patients.
Table 2.
Clinical characteristics of paraneoplastic syndromes (PNS)
| Total: 109 patients n (%) |
55 patients with pre-existing PNS (cohort 1), n (%) | 16 patients with de novo PNS during ICI treatment (cohort 2), n (%) | 38 patients with de novo PNS after ICI discontinuation (cohort 3), n (%) | |
| Neurologic PNS, n patients (%) | 32 (29.4) | 14 (25.5) | 4 (25) | 14 (37) |
| Cerebellar degeneration | 2 (1.8) | 1 (1.8) | 0 (0.0) | 1 (2.6) |
| Chronic inflammatory demyelinating polyneuropathy | 2 (1.8) | 0 (0.0) | 1 (6.3) | 1 (2.6) |
| Encephalitis/encephalomyelitis | 10 (9.2) | 4 (7.3) | 2 (13) | 4 (11) |
| Neuropathy | 8 (7.3) | 5 (9.1) | 1 (6.3) | 2 (5.3) |
| Myasthenia gravis | 4 (3.7) | 2 (3.6) | 0 (0.0) | 2 (5.3) |
| Other | 6 (5.5) | 2 (3.6) | 0 (0.0) | 4 (11) |
| Endocrine, n patients (%) | 27 (24.8) | 8 (14.5) | 5 (31) | 14 (37) |
| Cushing syndrome | 5 (4.6) | 2 (3.6) | 1 (6.3) | 2 (5.3) |
| Hypercalcemia | 22 (20) | 6 (10.9) | 4 (25) | 12 (32) |
| Dermatologic, n patients (%) | 15 (13.8) | 11 (20.0) | 1 (6.3) | 3 (7.9) |
| Amyopathic dermatopolymyositis | 3 (2.8) | 2 (3.6) | 1 (6.3) | 0 (0.0) |
| Dermatomyositis | 7 (6.4) | 5 (9.1) | 0 (0.0) | 2 (5.3) |
| Other | 5 (4.6) | 4 (7.3) | 1 (6.3) | 0 (0.0) |
| Rheumatologic, n patients (%) | 20 (18.3) | 16 (29.1) | 0 (0.0) | 4 (11) |
| Polymyalgia rheumatica | 8 (7.3) | 7 (12.7) | 0 (0.0) | 1 (2.6) |
| Hypertrophic pulmonary osteoarthropathy | 6 (5.5) | 4 (7.3) | 0 (0.0) | 2 (5.3) |
| Synovitis/tenosynovitis | 3 (2.8) | 2 (3.6) | 0 (0.0) | 1 (2.6) |
| Other | 3 (2.8) | 3 (5.4) | 0 (0.0) | 0 (0.0) |
| Hematologic, n patients (%) | 6 (5.5) | 1 (1.8) | 1 (6.3) | 4 (11) |
| Malignancy-induced erythrocytosis | 4 (3.7) | 0 (0.0) | 1 (6.3) | 3 (1.9) |
| Other | 2 (1.8) | 1 (1.8) | 0 (0.0) | 1 (2.6) |
| Gastrointestinal, n patients (%) | 2 (1.8) | 1 (1.8) | 1 (6.3) | 0 (0.0) |
| Pseudoachalasia | 1 (0.9) | 0 (0.0) | 1 (6.3) | 0 (0.0) |
| Stauffer syndrome | 1 (0.9) | 1 (1.8) | 0 (0.0) | 0 (0.0) |
| Nephrologic, n patients (%) | 6 (5.5) | 4 (7.3) | 2 (13) | 0 (0.0) |
| Nephritic/nephrotic syndrome | 6 (5.5) | 4 (7.3) | 2 (13) | 0 (0.0) |
| CTCAE V.5 grade of PNS | ||||
| 1 | 8 (9.6) | 3 (7.7) | 1 (6.3) | 4 (11) |
| 2 | 19 (23) | 12 (31) | 1 (6.3) | 6 (16) |
| 3 | 43 (52) | 16 (41) | 12 (75) | 15 (39) |
| 4 | 12 (14) | 7 (18) | 1 (6.3) | 4 (11) |
| 5 | 1 (1.2) | 1 (2.6) | 0 (0.0) | 0 (0.0) |
| Not reported* | 26 | 16 | 1 | 9 |
*Missing values were not included in the denominator for calculation of % in subgroups.
CTCAE, Common Terminology Criteria for Adverse Events; ICI, immune checkpoint inhibitor.
Management of PNS
Of 83 patients with available PNS CTCAE grade, 55 (66%) were grade 3 or 4. One patient died from grade 5 PNS (encephalitis). 55 patients required PNS-directed systemic immunosuppressive therapy which included steroids (n=46, 42%), intravenous immunoglobulin (n=13, 12%), rituximab (n=5, 5%), and plasmapheresis (n=4, 4%) among other immunosuppressants.
Temporal interconnections between PNS and ICI
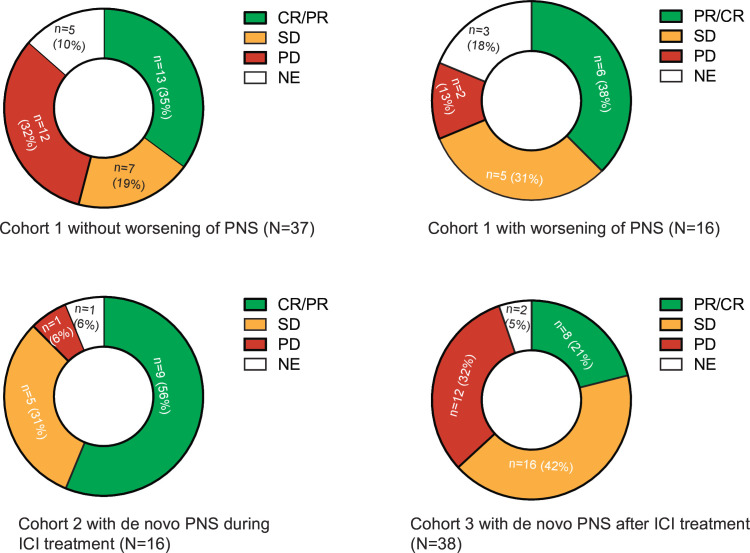
In cohort 1 (n=55 patients), 16 (29%) patients experienced exacerbation of their PNS after ICI initiation and 11 had evaluable ORR data. Among these, one patient with NSCLC was responding at the time of worsening of PNS. Five patients were progressing and five had stable disease (figure 2). The median time between the start of immunotherapy and the PNS exacerbation was 1.1 months (IQR: 0.7–3.3) following ICI initiation. Among patients in cohort 1, most PNS flare-ups or worsening of PNS occurred among patients with neurologic (50%), rheumatologic (31%), endocrine (25%), or nephrologic (25%) PNS (online supplemental table 3). Of six patients with pre-existing PNS who received dual ICI (nivolumab+ipilimumab), one patient (17%) experienced a flare-up 0.66 months after starting ICI whereas the other patients did not. In comparison, 15/47 (32%) patients treated with ICI monotherapy had flare-ups. Median time to flare-up was not reached in both groups and was not significantly different (p=0.38). Furthermore, 7 out of 14 patients with pre-existing auto-antibodies developed a flare-up of their PNS. Of the 14 patients with positive antibodies, 5 had extensive-stage SCLC, and 4 out of 5 had neurologic PNS that flared-up on treatment with ICI.
Figure 2.
Objective responses by clinical cohort. Shown are the best responses for patients in cohort 1 who developed worsening of paraneoplastic syndrome (PNS), patients in cohort 1 without worsening of PNS, patients in cohort 2 who were diagnosed with de novo PNS during ICI treatment, and patients in cohort 3 who were diagnosed with de novo PNS after ICI discontinuation. CR, complete response; ICI, immune checkpoint inhibitors; NE, not evaluable; PD, progressive disease; PR, partial response; SD, stable disease.
For cohort 2 (n=16), the median time between ICI initiation and onset of new PNS was 1.2 months (IQR: 0.4–3.5). Among evaluable patients in cohort 2, the ORR=60% (9/15). In this cohort, only one patient had progressive disease (PD) as best response on ICI treatment although eventually 14 of 16 patients had progression of disease. The median TTNT was 7.9 (95% CI: 3.8 to 11.3) months, whereas the median time for development of PNS was 1.2 months (IQR: 0.4–3.5). For cohort 3 (n=38), the median time between ICI discontinuation and onset of new PNS was 3 months (IQR: 0.7–8.6). Of 8 patients in cohort 3 who prior to development of a PNS had discontinued ICI due to trAE, 3 (38%) eventually developed grade 3 or 4 PNS versus 16 out of 21 (76%) patients who discontinued ICIs due to non-trAE etiologies and had grade 3 or 4 PNS (p=0.08). At the time of development of PNS in cohort 3, 21 out of 36 (58%) patients who had evaluable ORR data were progressing on imaging scans. Across the entire cohort, the exacerbation or new diagnosis of PNS prompted temporary interruption of ICI treatment in 1% (n=1) or permanent discontinuation in 12% of patients (n=13).
Survival outcomes
Among 102 patients with advanced solid tumors (stage IV or extensive-stage SCLC) and PNS treated with ICIs, the median OS was 15.6 (95% CI: 11.3 to 18.3) months, whereas the median TTNT was 8.4 (95% CI: 5.7 to 11.9) months. Of 95 patients with evaluable response data, ORR occurred in 37 patients (39%); 7 patients with a CR and 30 with a PR. For mNSCLC, median OS and TTNT were 12.8 months (95% CI: 7.4 to 25.6) and 8.1 months (95% CI: 5.0 to 14.2), respectively. Among other common cancer types, median OS for patients with extensive SCLC and RCC was 8.6 months (95% CI: 3.0 to 14.8) and 18.3 months (95% CI: 13 to NR), respectively. Median TTNT for patients with extensive-stage SCLC and RCC was 8.6 months (95% CI: 3.0 to 13.5) and 13.3 months (95% CI: 3.7 to 22.0), respectively. The outcomes for the 55 patients with an exacerbated pre-existing PNS (cohort 1) or 54 with a newly diagnosed PNS (cohorts 2 and 3) are shown in figure 2. Of patients in cohort 1, patients with PNS exacerbation did not have significantly different OS and TTNT compared with patients without PNS exacerbation.
Toxicity profiles of ICI-based regimens
TrAEs of any grade occurred in 39% (n=43) of patients (table 3). Grade ≥3 trAEs were reported in 17% (n=18) patients. The most common trAEs were thyroid dysfunction (n=10, 9.2%), diarrhea/colitis (n=9, 8.3%), and pneumonitis (n=8, 7.3%). Of the 38 patients with trAEs, 17 (45%) required hospitalization and 23 (61%) received steroids. Of these, 6 patients received low-dose steroids (<1 mg/kg prednisone equivalent) and 16 received high doses of steroids (≥1 mg/kg prednisone equivalent). The steroid dose was not known for 1 patient. 23 patients required additional immunosuppression (2 with infliximab).
Table 3.
Distribution of trAEs in patients with paraneoplastic syndromes
| All cohorts (n=109) | Cohort 1 (n=55) | Cohort 2 (n=16) | Cohort 3 (n=38) | |||||
| Any grade (n, %) | Grade ≥3 (n, %) | Any grade (n, %) | Grade ≥3 (n, %) | Any grade (n, %) | Grade ≥3 (n, %) | Any grade (n, %) | Grade ≥3 (n, %) | |
| Any trAE* | 43 (39) | 18 (17) | 17 (30.9) | 10 (18) | 7 (44) | 2 (13) | 19 (50) | 6 (16) |
| Diarrhea/colitis | 9 (8.3) | 5 (4.6) | 6 (10.9) | 4 (7.3) | 0 (0.0) | 0 (0.0) | 3 (7.9) | 1 (2.6) |
| Thyroid | 10 (9.2) | 1 (0.9) | 4 (7.3) | 1 (1.8) | 2 (13) | 0 (0.0) | 4 (11) | 0 (0.0) |
| Pneumonitis | 8 (7.3) | 4 (3.7) | 4 (7.3) | 3 (5.5) | 2 (13) | 0 (0.0) | 2 (5.3) | 1 (2.6) |
| Skin | 9 (8.3) | 1 (0.9) | 5 (9.1) | 1 (1.8) | 0 (0.0) | 0 (0.0) | 4 (11) | 0 (0.0) |
| Hepatitis | 4 (3.7) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (6.3) | 0 (0.0) | 3 (7.5) | 0 (0.0) |
| Hypophysitis | 2 (1.8) | 1 (0.9) | 1 (1.8) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (2.6) | 1 (2.6) |
| Myocarditis | 2 (1.8) | 1 (0.9) | 1 (1.8) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (2.6) | 1 (2.6) |
| Myositis | 3 (2.8) | 2 (1.8) | 0 (0.0) | 0 (0.0) | 1 (6.3) | 1 (6.3) | 2 (5.3) | 1 (2.6) |
| Other | 13 (12) | 3 (2.8) | 5 (9.1) | 2 (3.6) | 2 (13) | 0 (0.0) | 6 (16) | 1 (2.6) |
| Steroids used, n (%) | 27 (25) | 12 (22) | 4 (25) | 11 (29) | ||||
| Discontinued due to toxicity | 22 (20) | 9 (16) | 2 (13) | 11 (29) | ||||
*Individual trAE are greater than the sum as a subset of patients had more than one trAE
trAE, treatment-related adverse events .
Clinical outcomes and toxicity profiles: matched cohort of patients with NSCLC
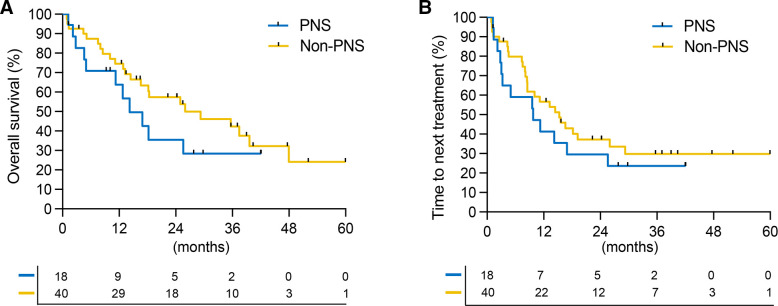
We next turned to a matched cohort of patients with mNSCLC+/–PNS (online supplemental table 4 and 5). NSCLC was selected as it was the most represented cancer type. We matched 18 patients with mNSCLC and pre-existing PNS (cohort 1) to 40 mNSCLC without PNS, all treated with ICIs. Patients with PNS and mNSCLC had a trend towards inferior OS compared with patients without PNS (median overall survival (mOS) PNS: 14.2 months, mOS control: 26 months, HR=1.6, (95% CI: 0.7 to 3.5), p=0.09, log-rank test) on univariable analysis (figure 3). Patients with PNS had a trend towards inferior TTNT outcomes compared with patients without PNS (HR=1.4, (95% CI: 0.7 to 2.8), p=0.1, figure 3). The prevalence of trAEs was similar between matched mNSCLC with and without PNS, where any grade trAEs occurred in 6/18 (33%) versus 14/40 (35%), respectively. Moreover, grade ≥3 trAEs occurred in 4 (22%) patients with PNS versus 7 (18%) patients without PNS. Systemic steroids were used in 22% (4/18) and 15% (6/40) of patients with and without PNS, respectively (online supplemental table 6).
Figure 3.
Survival outcomes of matched cohort of patients with metastatic non-small cell lung cancer (NSCLC) with and without paraneoplastic syndrome (PNS). Shown are Kaplan-Meier curves for (A) Overall survival and (B) Time-to-next treatment between patients with metastatic NSCLC with and without PNS. Log-rank test was used to compute the p values. Tick marks indicate censored observations.
Discussion
Herein, we assembled a multi-institutional cohort and found that in patients with cancer undergoing treatment with ICI, PNS exacerbations and de novo PNS generally occur early in the ICI treatment course. We then leveraged a matched cohort of patients with mNSCLC+/–PNS and showed similar trAE profiles between both groups. There was shorter median OS among patients with PNS compared with patients without PNS, although this was not statistically significant, likely due to small sample size.
With increasing awareness and improvement of the diagnostic accuracy of PNS on one hand, alongside more widespread use of ICI globally, we need a better understanding of how to use ICI in patients with underlying PNS, and how to treat these complex syndromes. Moreover, patients in clinical practice are prone to having undiagnosed PNS at the time of systemic treatment initiation and as such, it is important to understand the clinical ramifications of introducing systemic therapies as it relates to subclinical PNS. In our cohort, 29% patients with pre-existing PNS developed worsening of their PNS after the initiation of ICI. A case series found that for 16 patients with pre-existing PNS, 8 had worsening of their PNS after initiation of ICI,17 while another study of 7 patients with thymoma and 1 patient with thymic carcinoma showed that 4 patients with pre-existing muscle acetylcholine receptor (mAchR) antibodies had an increased risk of developing myositis after challenging with avelumab relative to patients without mAchR antibodies.18 Although worsening of pre-existing PNS can occur in the setting of progression of disease instead of ICI-mediated effect, most of the patients that developed worsening were responding at the time of ICI.
SCLC is associated with a myriad of neurologic PNS. In our cohort, four out of five patients with SCLC who had detectable onconeural antibodies developed a flare-up of their pre-existing neurological PNS and had to discontinue ICI. This suggests that these patients are at high risk of PNS complications if treated with ICIs. Hence, we believe that patients with SCLC who present with pre-existing neurological PNS should be screened for onconeural antibodies. If onconeural antibodies are detected, we advocate for in-depth discussions about the risk-benefit ratio of starting ICIs in this population given the elevated risk for PNS flare-ups.
Overall, the timing of PNS worsening or appearance relative to ICI initiation was short with a median of 1.1 and 1.2 months (cohorts 1 and 2, respectively). Moreover, the burden of malignant disease in most patients in cohort 2 (14/15, 93%) was stable or regressing indicating that PNS development was less likely associated with tumor progression. The results from cohorts 1 and 2 suggest that treating physicians should be cognizant of PNS flare-ups and should remain vigilant for the development of new PNS diagnoses early in the clinical course of patients on ICI. For cohort 3, there was no significant difference in the rate of grade ≥3 PNS between patients who previously discontinued ICIs for trAE versus non-trAE causes. In fact, there was a trend towards higher grade PNS among patients who had discontinued ICIs for non-trAE reasons. This suggests that developing a trAE that prompts ICI treatment discontinuation is not associated with a higher likelihood of developing a severe PNS. Future translational work is warranted to predict patients at risk of developing worsening of PNS or overt PNS on ICIs.
In our robustly matched cohort of patients with mNSCLC+/−PNS, we showed that there was a trend towards worse OS among patients with PNS although this did not reach statistical significance. Nonetheless, the mOS was almost twofold longer among patients with mNSCLC that did not have PNS compared with patients with PNS. Prior work in SCLC had suggested favorable clinical outcomes for patients with neurologic PNS compared with patients without PNS. However, this work was mostly done before ICI therapy was widely used and was based on the fact that in general, patients with PNS tend to have localized disease, likely due to increased CD4+ and CD8+ T-cell infiltration of their tumors. The increased presence of T cells in the tumor microenvironment makes the use of ICI risky for these patients. We additionally demonstrated comparable rates of trAEs among patients with and without PNS. Despite that, future studies should center around matched analyses of other tumor types and additionally analyze clinical outcomes by PNS systems.
Our work was hampered by several limitations. First, there was under-representation of non-white populations (8% non-white) and questions could therefore be raised regarding the generalizability of the findings. Second, some patients did not have PD-L1 and tumor mutational status reported to enable matching in the mNSCLC cohort. Third, response evaluations were a mix of objective response assessments by RECIST V.1.1 and investigator-based evaluations. Fourth, the incidence of trAEs is lower than expected based on publicly reported trAE rates. This may be driven by the fact that patients were treated outside of clinical trials and thus subject to suboptimal capture of events. However, the rate of higher grade trAEs (grade ≥3) is less likely burdened by under-reporting. Fifth, despite obtaining data from nine centers, we still had a relatively low sample size and may have been underpowered for the matched cohort analyses. Hence, it is hard to make definitive conclusions about survival outcomes in the matched cohort without conducting further larger studies. However, this is one of the largest studies of PNS related to ICI use and we had granular clinical data available across many centers. Another limitation is the overlap between PNS and conventional trAE in cohort 2 and cohort 3 which makes it challenging to distinguish both. To circumvent this, we limited to a list of prespecified PNS and manually reviewed each. While all cases were adjudicated to be due to PNS, it is possible that some may have been due to immune-related adverse events from ICI treatment cannot be ruled out.
Conclusions
In conclusion, de novo PNS and exacerbations in pre-existing ones typically occur early in ICI treatment. Patients with PNS and NSCLC have similar trAE profiles compared with patients without PNS. Further studies are needed to determine whether patients with PNS have worse survival outcomes compared with patients without PNS.
Acknowledgments
We are grateful to all the participating patients with PNS and their families whose contributions have made this study possible. We thank the investigators and staff members at the participating centers who have contributed to our work.
Footnotes
Twitter: @AminNassarMD, @thenasheffect, @ShrutiGKidney, @DrChoueiri, @sonpavde
Contributors: Conception and design: AHN, TEZ, ABK, GS. Administrative support: DF, GS, AHN. Provision of study materials or patients: GS, AHN. Collection and assembly of data: All authors. Data analysis and interpretation: CZ, WX, TEZ, AHN. Manuscript writing: All authors. Final approval of manuscript: All authors. Accountable for all aspects of the work: All authors. AHN is acting as the guarantor of this work.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: AHN receives honoraria from OncLive, TEMPUS, and Korean Society for Medical Oncology. Consulting fees: Guidepoint Global. RRM: Consulting/Advisory Board – Aveo, AstraZeneca, Bayer, Bristol Myers Squibb, Blue Earth Diagnostics, Calithera, Caris, Denderon, Exelixis, Janssen, Merck, Myovant, Pfizer, Sanofi, SeaGen, Sorrento Therapeutics, Tempus. Institutional Research Funding – AstraZeneca, BMS, Exelixis, Artera, Oncternal, Bayer, Tempus. JAS is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases (grant numbers R01 AR080659, R01 AR077607, P30 AR070253, and P30 AR072577), the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award funded by the Gordon and Llura Gund Foundation. JAS has received research support from Bristol Myers Squibb and performed consultancy for AbbVie, Amgen, Boehringer Ingelheim, Bristol Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, Pfizer, ReCor, and Sobi unrelated to this work. The funders had no role in the decision to publish or preparation of this manuscript. The content is solely the responsibility of the authors and does not necessarily represent the official views of Harvard University, its affiliated academic health care centers, or the National Institutes of Health. AC received grants for consultancies/advisory boards: MSD, OncoC4, IQVIA, AstraZeneca, Access Infinity, Ardelis Health, Alpha Sight. Speaker fees: AstraZeneca, Eisai, Pierre-Fabre, MSD. Writing/Editorial activity: BMS. Travel support: Sanofi and MSD. ARN reports Funding to Institution for Trials he is PI on:Loxo@Lilly, Surface Oncology, ADC Therapeutics, IGM Biosciences, EMD Serono, Aravive, Nikang Therapeutics, Inspirna, Exelixis, Revolution Medicine, Jacobio, Pionyr, Jazz Pharmaceuticals, NGM Biopharmaceuticals. ARN receives Consultant Editor Compensation: JCO Precision Oncology. Consulting/Advisory Board: Foundation Med. ARN reports Travel Compensation from: SITC/ AACR/ Conquer Cancer Foundation, Jazz Pharmaceuticals, Binay Tara Foundation, Foundation Med.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
All data relevant to the study are included in the article or uploaded as supplementary information.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
This retrospective study was approved by the institutional review board (IRB) at DFCI (Protocol #21-329) and local IRBs at participating sites, in accordance with the Declaration of Helsinki. Participants gave informed consent to participate in the study before taking part.
References
- 1.Darnell RB, Posner JB. Paraneoplastic syndromes involving the nervous system. N Engl J Med 2003;349:1543–54. 10.1056/NEJMra023009 [DOI] [PubMed] [Google Scholar]
- 2.Gandhi L, Johnson BE. Paraneoplastic syndromes associated with small cell lung cancer. J Natl Compr Canc Netw 2006;4:631–8. 10.6004/jnccn.2006.0052 [DOI] [PubMed] [Google Scholar]
- 3.Pelosof LC, Gerber DE. Paraneoplastic syndromes: an approach to diagnosis and treatment. Mayo Clin Proc 2010;85:838–54. 10.4065/mcp.2010.0099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Geng G, Yu X, Jiang J, et al. Aetiology and pathogenesis of Paraneoplastic autoimmune disorders. Autoimmun Rev 2020;19. 10.1016/j.autrev.2019.102422 [DOI] [PubMed] [Google Scholar]
- 5.Giometto B, Grisold W, Vitaliani R, et al. Paraneoplastic neurologic syndrome in the PNS Euronetwork database: a European study from 20 centers. Arch Neurol 2010;67:330–5. 10.1001/archneurol.2009.341 [DOI] [PubMed] [Google Scholar]
- 6.Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with Ipilimumab in patients with metastatic Melanoma. N Engl J Med 2010;363:711–23. 10.1056/NEJMoa1003466 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Robert C, Ribas A, Schachter J, et al. Pembrolizumab versus Ipilimumab in advanced Melanoma (KEYNOTE-006): post-hoc 5-year results from an open-label, Multicentre, randomised, controlled, phase 3 study. Lancet Oncol 2019;20:1239–51. 10.1016/S1470-2045(19)30388-2 [DOI] [PubMed] [Google Scholar]
- 8.Marabelle A, Le DT, Ascierto PA, et al. Efficacy of Pembrolizumab in patients with Noncolorectal high Microsatellite instability/mismatch repair-deficient cancer: results from the phase II KEYNOTE-158 study. J Clin Oncol 2020;38:1–10. 10.1200/JCO.19.02105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.André T, Shiu K-K, Kim TW, et al. Pembrolizumab in Microsatellite-instability-high advanced colorectal cancer. N Engl J Med 2020;383:2207–18. 10.1056/NEJMoa2017699 [DOI] [PubMed] [Google Scholar]
- 10.Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Combined Nivolumab and Ipilimumab or monotherapy in untreated Melanoma. N Engl J Med 2015;373:23–34. 10.1056/NEJMoa1504030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Burtness B, Harrington KJ, Greil R, et al. Pembrolizumab alone or with chemotherapy versus Cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): a randomised, open-label, phase 3 study. Lancet 2019;394:1915–28. 10.1016/S0140-6736(19)32591-7 [DOI] [PubMed] [Google Scholar]
- 12.Gandhi L, Rodríguez-Abreu D, Gadgeel S, et al. Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N Engl J Med 2018;378:2078–92. 10.1056/NEJMoa1801005 [DOI] [PubMed] [Google Scholar]
- 13.Morris VK, Salem ME, Nimeiri H, et al. Nivolumab for previously treated Unresectable metastatic Anal cancer (Nci9673): a Multicentre, single-arm, phase 2 study. Lancet Oncol 2017;18:446–53. 10.1016/S1470-2045(17)30104-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Graus F, Dalmau J. Paraneoplastic neurological syndromes in the era of immune-Checkpoint inhibitors. Nat Rev Clin Oncol 2019;16:535–48. 10.1038/s41571-019-0194-4 [DOI] [PubMed] [Google Scholar]
- 15.Valencia-Sanchez C, Zekeridou A. Paraneoplastic neurological syndromes and beyond emerging with the introduction of immune Checkpoint inhibitor cancer Immunotherapy. Front Neurol 2021;12. 10.3389/fneur.2021.642800 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Haanen J, Ernstoff MS, Wang Y, et al. Autoimmune diseases and immune-Checkpoint inhibitors for cancer therapy: review of the literature and personalized risk-based prevention strategy. Ann Oncol 2020;31:724–44. 10.1016/j.annonc.2020.03.285 [DOI] [PubMed] [Google Scholar]
- 17.Manson G, Maria ATJ, Poizeau F, et al. Worsening and newly diagnosed Paraneoplastic syndromes following anti-PD-1 or anti-PD-L1 Immunotherapies, a descriptive study. J Immunother Cancer 2019;7:337. 10.1186/s40425-019-0821-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mammen AL, Rajan A, Pak K, et al. Pre-existing Antiacetylcholine receptor Autoantibodies and B cell Lymphopaenia are associated with the development of Myositis in patients with Thymoma treated with Avelumab, an immune Checkpoint inhibitor targeting programmed death-ligand 1. Ann Rheum Dis 2019;78:150–2. 10.1136/annrheumdis-2018-213777 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jitc-2023-008724supp001.xlsx (2MB, xlsx)
Data Availability Statement
All data relevant to the study are included in the article or uploaded as supplementary information.