Abstract
Human metapneumovirus (HMPV), recently identified in isolates from children hospitalized with acute respiratory tract illness, is associated with clinical diagnosis of pneumonia, asthma exacerbation, and acute bronchiolitis in young children. HMPV has been shown to cocirculate with respiratory syncytial virus (RSV) and mediate clinical disease features similarly to RSV. Little is known regarding the pathophysiology or immune response associated with HMPV infection; thus, animal models are needed to better understand the mechanisms of immunity and disease pathogenesis associated with infection. In this study, we examine features of the innate and adaptive immune response to HMPV infection in a BALB/c mouse model. Primary HMPV infection elicits weak innate and aberrant adaptive immune responses characterized by induction of a Th2-type cytokine response at later stages of infection that coincides with increased interleukin-10 expression and persistent virus replication in the lung. Examination of the cytotoxic T lymphocyte and antibody response to HMPV infection revealed a delayed response, but passive transfer of HMPV-specific antibodies provided considerable protection. These features are consistent with virus persistence and indicate that the immune response to HMPV is unique compared to the immune response to RSV.
Human metapneumovirus (HMPV) is a recently recognized human pathogen first identified in respiratory specimens from young children suffering with clinical respiratory syndromes ranging from mild to severe lower respiratory tract infection (47, 48). The disease burden associated with HMPV infection is not fully understood; however, serological studies suggest that HMPV has worldwide distribution and is acquired early in life, and by age of 5 years, approximately 70% of all children develop antibodies to HMPV (10, 12, 15, 19, 25, 29, 31). HMPV causes upper or lower respiratory tract illness in patients between ages 2 months and 87 years (7, 11, 19, 49, 50), may exacerbate asthma and wheezing in young children (29), and cocirculates with respiratory syncytial virus (RSV) (27, 36, 50) causing similar clinical disease (23, 50, 51). These findings underscore the need for a better understanding of the mechanisms of immunity and disease pathogenesis associated with HMPV infection to provide the foundation necessary for development of vaccines and treatment modalities.
HMPV is an enveloped, negative-strand RNA virus of approximately 13 kb and a member of the Pneumovirinae subfamily of paramyxoviruses whose genome consists of eight genes, namely, nucleocapsid (N), phosphoprotein (P), matrix (M), fusion (F), second matrix (M2), small hydrophobic (SH), attachment (G), and RNA-dependent RNA polymerase (L) in the order 3′-N-P-M-F-M2-SH-G-L-5′ (4, 5, 47). None of the predicted proteins have been completely biochemically identified and their functions have not been completely determined; however, recent data suggest that the F glycoprotein is an envelope protein that can be accessed by neutralizing antibodies and appears to be a major protective antigen (34, 37). There are two major groups of HMPV, strains A and B, as determined on the basis of sequence studies of the N, F, G, and L genes, and both strains cocirculate in the community (30, 31). Little is known about the immune response to HMPV; however, recent studies suggest that the evolution of the HMPV G glycoprotein may be driven by immune pressure directed at codon positions located mainly in the second hypervariable region of the ectodomain (30). On the basis of epidemiological and emerging disease burden studies, it appears HMPV has considerable impact on human health; thus, HMPV vaccine strategies are being considered (5).
To develop a better understanding of the pathophysiology associated with HMPV infection, our laboratory developed a BALB/c mice model of infection and showed that HMPV replicates in lung tissue with biphasic kinetics in which peak titers occur days 7 and 14 pi and infectious HMPV can be recovered from the lungs up to day 60 pi, and genomic RNA was detected in the lungs for ≥180 days pi by reverse transcription-PCR (2). In this mouse model, neither HMPV RNA nor infectious virus was detected in serum, spleen, kidneys, heart, or brain tissue, and in similarity to one trait characteristic of persistent virus infections (1, 14, 24, 32, 42), HMPV-infected mice exhibited splenomegaly that did not resolve until ≥day 60 pi. Interestingly, lung histopathology associated with HMPV infection was modest and characterized by mononuclear cell infiltration in the interstitium, beginning day 2 postinfection (pi) and peaking day 4 pi, which decreased by day 14 pi. There was evidence of airway remodeling and increased mucus production at day 2 pi that was concordant with bronchial and bronchiolar inflammation. In contrast, RSV infection in BALB/c mice results in peak lung virus titers occurring between days 5 and 6 pi, infectious virus is cleared between day 7 and 10 pi, and infection is associated with substantial lung histopathology and inflammatory response (39).
Thus, HMPV infection in BALB/c mice appears to be associated with a substantially lower pulmonary inflammatory response compared to RSV infection, a feature that may contribute to HMPV persistence.
In this study, we examined features of the humoral and cellular immune response to HMPV infection in a BALB/c mouse model. These studies provided important insights into the host response to HMPV infection and a fundamental understanding of some of the immune and pathophysiological process associated with HMPV infection required to provide the foundation for development of vaccines or treatment modalities.
MATERIALS AND METHODS
Animals.
Four- to 6-week-old, specific-pathogen-free female BALB/c mice were purchased from Harlan Sprague-Dawley Laboratories, Indianapolis, IN. The mice were housed in microisolator cages and fed sterilized water and food ad libitum.
Virus preparation and cell lines.
Vero E6 cells were maintained in tissue culture medium (TCM) consisting of minimal essential medium (MEM; Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum (HyClone, Logan, UT). HMPV stocks were prepared in Vero E6 cells. Infection of Vero E6 cells with strain A HMPV/CAN98-75, as previously described (2), does not require trypsin for propagation. Briefly, subconfluent Vero E6 cells in serum-free MEM were infected with plaque purified strain A HMPV/CAN98-75 (HMPV; accession no. AY297748) at a multiplicity of infection (MOI) of 0.1. The virus was allowed to adsorb for 1 h at 37°C, after which TCM was added. Infected cells were incubated for 72 h at 37°C or until >90% cytopathic effect was observed by light microscopy. Infected cells were harvested by removal of the medium and replacement with a minimal volume of serum-free MEM followed by two freeze-thaw cycles at −70°C and 4°C, respectively. The contents were collected and centrifuged at 4,000 × g for 20 min at 4°C, and the titer was determined by plaque assay as described below.
Infection, sampling, and virus titers.
All experiments were performed at least three times using three to four mice/time point/experiment except for antibody assays which used four to six mice/experiment. Bronchoalveolar lavage (BAL) cells were pooled for each time point/experiment to provide sufficient cells for analysis. Mice were anesthetized by intraperitoneal administration of 2,2,2-tribromoethanol (Avertin) and infected intranasally (i.n.) with 106 PFU of HMPV. Prior to removal of lungs or tissues on days 0, 3, 4, 5, 7, 10, 14, and 28 pi, anesthetized mice were exsanguinated by severing the right caudal artery, and the blood was collected for serum antibody analysis. Lung tissue was collected in 1.0 ml Dulbecco's phosphate-buffered saline (D-PBS; Invitrogen) and stored at −70°C for simultaneous analysis of virus titers. Since HMPV does not form readily detectable plaques in Vero E6 cells, HMPV titers from homogenized lungs were determined by plaque assay using immunostaining to detect HMPV N protein as previously described (3). Briefly, 1.0 g of HMPV-infected and uninfected lungs was homogenized in 1 ml of PBS using a hand-held Tissuemiser homogenizer (Fisher Scientific, Pittsburgh, PA). Cleared lung lysates were diluted 10-fold in serum-free MEM (Invitrogen), added to 95% confluent Vero E6 cells cultured in serum-free MEM in 24-well plates (BD Falcon, San Jose, CA), and incubated for 1 h at 37°C, followed by 2% methylcellulose semisolid overlay. At 72 h pi, the medium was removed from the cells on the 24-well plates, the wells were carefully washed with PBS, and the cells were fixed with acetone:methanol (60:40). After air drying, the cells were immunostained with affinity-purified hyperimmune sera reactive to a conserved metapneumovirus N protein (amino acid sequence DLSYKHAILKESQYTIKRDV) as previously described (3). The anti-N protein antibody was appropriately diluted in PBS containing blocking agents (Blotto; Bio-Rad, Hercules, CA) and detected using alkaline phosphatase-conjugated goat anti-rabbit immunoglobulin G (IgG) (Sigma, The Woodlands, TX), and the plaques were enumerated with 3′,3′, diaminobenzidine (Vector Laboratories, Burlingame, CA). Bronchoalveolar leukocytes (BAL) were collected from individual lungs by lavage using three successive 1.0 ml washes with D-PBS containing 1% bovine serum albumin (BSA) (Sigma).
Flow cytometry.
Single-cell suspensions of BAL cells were blocked with 1% BSA in D-PBS and then stained with the appropriate combination of fluorescein isothiocyanate- or phycoerythrin-labeled anti-CD4 (LM4-4), anti-CD8 (53-6.7), anti-CD45R/B220 (RA3-6B2), anti-pan NK cell (DX5), anti-neutrophil (RB6-8C5), or anti-a CD11b (M1/70), anti-CD44 (IM7), anti- CD62L (MEL-14), or mouse isotype antibody control (all from BD Biosciences, San Diego, CA). A lymphocyte gate was used to select 10,000 events for CD4+, CD8+, B220+, CD44+, and CD62L lymphocytes, and 10,000 ungated events were used for DX5+, RB6-8C5+, and CD11b+ cells. The distribution of cell surface markers was determined in two-color mode on a FACScan apparatus with CellQuest software (Becton Dickinson, San Jose, CA).
The procedure used for intracellular cytokine staining was modified for microculture as previously described (40). Briefly, cells were washed in D-PBS, stained with an appropriate dilution of fluorescein isothiocyanate anti-CD4 (LM4-4) or anti-CD8 (53-6.7) monoclonal antibody for 30 min on ice, washed in 1% BSA-D-PBS, and resuspended in Cytofix/Cytoperm (BD Biosciences) for 15 min on ice. Subsequently, the cells were washed in Cytofix/Cytoperm and resuspended in an appropriate dilution of phycoerythrin-labeled anti-interleukin-2 (IL-2) (JES6-5H4), anti-IL-4 (BVD4-1D11), anti-IL-5 (TRFK5), anti-IL-6 (MP5-20F3), anti-IL-10 (1B1.3a), anti-gamma interferon (IFN-γ) (XMG1.2), or anti-tumor necrosis factor alpha (TNF-α) (MP6-XT22) antibody (all from BD Biosciences) diluted in 1% BSA-D-PBS. The cells were stained on ice for 30 min, washed in D-PBS, and resuspended in D-PBS containing 1% BSA for flow cytometric analysis using a FACScan apparatus with CellQuest software (Becton Dickinson).
HMPV ELISA.
Uninfected and HMPV-infected Vero E6 cell lysates (MOI = 1.0) were prepared as described above and assayed for protein concentration by BCA protein analysis (Pierce Biotechnology, Inc., Rockford, IL). Protein concentrations were adjusted by dilution in carbonate-bicarbonate buffer (pH 9.6). A total of 6 μg of HMPV-infected cell lysate in 100 μl buffer was added to appropriate wells of an enzyme-linked immunosorbent assay (ELISA) plate (Immulon-2; Dynex Technologies, Inc., Chantilly, VA), and the plates were incubated at 4°C overnight. Subsequently, the supernatants were removed, and the wells were washed 3× with PBS-0.05% Tween (PBS-T) and blocked using 150 μl/well of 5% dry milk-phosphate-buffered saline (blocking buffer). Pooled sera (n = 3 to 5 mice/time point) collected at various time points post-HMPV infection were appropriately diluted in blocking buffer, 100 μl/dilution was added in triplicate to wells of an ELISA plate, and the plate was incubated for 1 h at 37°C. The wells were washed 3× with PBS-T, and 100 μl/well of alkaline phosphatase-conjugated goat anti-mouse IgG (Sigma) diluted in blocking buffer was added for 1 h at 37°C. Subsequently, the wells were decanted and washed 3× with PBS-T, and the reactions were developed using pNpp substrate tablets (Sigma). Absorbance was measured at 415- and 630-nm wavelengths. Antibody endpoint titers were determined according to the method of Reed and Muench (33) and are reported as the mean reciprocal log2 ± standard deviation (SD).
Passive protection studies.
For protection studies, 4- to 6-week-old, specific-pathogen-free female BALB/c mice were purchased from Harlan Sprague-Dawley Laboratories, Indianapolis, IN, and immunized on days 0, 14, 28, and 56 with 106 PFU of HMPV. The animals were bled after the last immunization, and the HMPV-specific antibody titers were determined by ELISA and neutralization assays as previously described (2). HMPV-specific IgG was purified from hyperimmune sera over a protein A column (Pierce Biotechnology, Inc., Rockford, IL) according to the manufacturer's protocol. Antibody concentrations were assayed for protein concentration by BCA protein analysis (Pierce Biotechnology, Inc., Rockford, IL). To determine the protective efficacy of HMPV-immune sera, 150 μg of normal mouse sera, hyperimmune HMPV-specific mouse sera, or affinity-purified IgG from hyperimmune HMPV-specific mouse sera was passively transferred to naïve BALB/c mice, and the mice were intranasally challenged with 106 PFU of HMPV. At day 7 pi, virus titers in the lungs were assayed as previously described (2).
Cytotoxic T lymphocyte (CTL) assay.
HMPV-specific CTL cytotoxicity was determined by modification of a previously described method (46). Briefly, uninfected and HMPV-infected (106 PFU) mice were anesthetized and sacrificed, and the spleens from three mice/treatment were removed at day 7 or 28 pi. Individual spleens were dissociated in DMEM using a glass pestle (Sigma) and 100-μm nylon cell strainer (Falcon; Becton Dickinson Labware, Franklin Lakes, NJ) and washed in DMEM. The spleen cells were resuspended in TCM to 2 × 106 cells/ml, and 2 ml/well was added to 24-well flat-bottom plates (Costar). Spleen cells were in vitro stimulated for 7 days at 37°C following addition of 4 × 106 HMPV-infected (MOI = 10) syngeneic spleen cells per well. P815 cells (ATCC TIB 64), a mastocytoma cell line, were maintained in TCM and prepared for CTL targets by washing and resuspending 106 cells in 1.0 ml of serum-free D-MEM containing 107 PFU/ml HMPV/75 (MOI = 10), 107 PFU/ml RSV strain A2 (MOI = 10), 10 μg HMPV N peptide (DLSYKHAILKESQYTIKRDV) (3), 10 μg RSV M2-peptide (SYIGSINNI) (17), or an equivalent dilution of uninfected Vero E6 cell control lysate for 18 h at 37°C. Subsequently, an additional 1.0 ml of TCM containing 200 μCi of 51Cr (Na2CrO4; Amersham Biosciences, Piscataway, NJ) was added to the target cells and incubated for 2 h 37°C, and the cells were washed in TCM.
Cytotoxicity was determined using a standard 6-h 51Cr release assay. Briefly, secondary in vitro-stimulated spleen cells were washed in D-MEM and resuspended in TCM to achieve effector-to-target cell ratios of 40:1, 10:1, and 2:1. All experiments were performed using triplicate cultures/target cell population, and the experiment was repeated three separate times. Percent specific lysis was determined from the mean 51Cr release of experimental − mean 51Cr release of control over the mean total 51Cr release − mean of control × 100.
Statistical analysis.
All experiments were performed at least three times using three to four mice/time point/experiment except for antibody assays which used four to six mice/experiment. The data presented represent the mean values ± SD for three separate assays. Statistical significance was determined using a Student's t test where a P value of < 0.05 was considered statistically significant.
RESULTS
HMPV replication in the lungs is linked to weight loss.
To determine whether the level of virus replication in the lungs is associated with illness, BALB/c mice were intranasally (i.n.) infected with 106 PFU HMPV and body weight loss and lung virus titers were determined at days 0, 3, 5, 7, 10, 14, and 28 pi (Table 1). Loss in body weight associated with RSV infection in mice can be used to gauge illness (13); in addition, HMPV-infected mice were visually examined for overt signs of illness, including ruffled coat, huddling, and heavy breathing. Mice infected with HMPV displayed signs of illness after infection that coincided with body weight loss from day 0 to day 7 pi; however, no overt signs of illness were evident by day 10 pi, and the body weights returned to normal by day 28 pi (Table 1). Decrease in body weight was associated with increased virus titers in the lungs (Table 1). As expected based on previous studies (2), HMPV replicated with biphasic growth kinetics peaking at day 7 (108.1 PFU/g lung tissue) and day 14 pi (107.5 PFU/g lung tissue), and replication declined to 104.1 PFU/g lung tissue by day 28 pi, but virus was not cleared (Table 1).
TABLE 1.
HMPV titers and weight loss in BALB/c mice
| Days postinfectiona | Mean virus titer (log10PFU/g ± SD)b | Weight (g ± SD) |
|---|---|---|
| 0 | 0.0 ± 0.00 | 22.80 ± 1.770 |
| 3 | 7.1 ± 0.10 | 23.01 ± 0.934 |
| 5 | 7.2 ± 0.05 | 18.03 ± 0.610 |
| 7 | 8.1 ± 0.10 | 17.46 ± 0.437 |
| 10 | 5.8 ± 0.05 | 19.86 ± 0.377 |
| 14 | 7.5 ± 0.04 | 19.17 ± 0.633 |
| 28 | 4.1 ± 0.02 | 22.13 ± 1.212 |
BALB/c mice were i.n. infected with 106 PFU of HMPV/75.
Five animals from each group were weighed and euthanized at day 0, 3, 5, 7, 10, 14, or 28 pi and lungs were harvested to determine virus titers.
Bronchoalveolar leukocyte (BAL) response to infection.
To provide an indication of the pulmonary immune response to HMPV replication in the lung, the total number of BAL cells at days 0, 3, 5, 7, 10, 14, and 28 pi was determined. BAL cell numbers increased from a range of 0.5 × 105 to 1 × 105 BAL cells/lung at day 0 to 1.7 × 105 to 2 × 105 cells/lung between days 3 to 14 pi, before declining to 0.7 × 105 to 1.3 ×105 cells/lung at day 28 pi, suggesting that primary HMPV infection does not engender an emphatic BAL cell response as previously described (2). To determine the leukocyte populations responding to HMPV infection, flow cytometry was used to examine CD4+ or CD8+ T cells, B220+ B cells, DX5+ NK cells, RB6-8C5+ polymorphonuclear leukocyte cells, and CD11b+ cells.
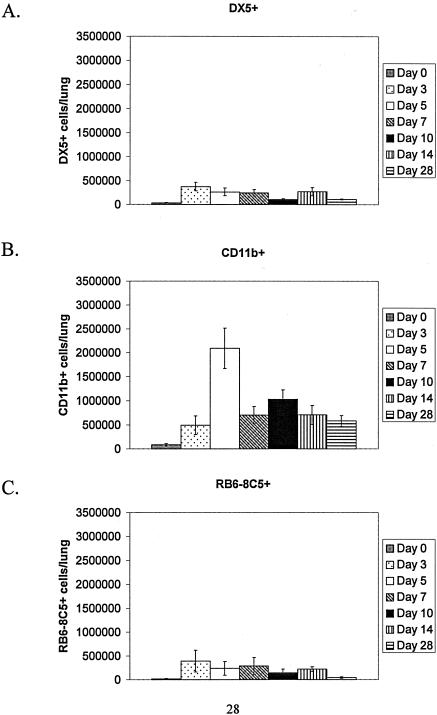
The innate response to HMPV infection was minimal, as few DX5+ cells and RB6-8C5+ cells were detected at any time point examined (0.37 × 105 to 3.7 × 105 cells/lung); however, a higher percentage of CD11b+ cells was detected at day 5 pi (2.1 × 106 cells/lung) (Fig. 1). The percentage of CD11b+ cells was substantially decreased by day 7 pi (7 × 105 cells/lung) and remained low out to day 28 pi (5.8 × 105 cells/lung) (Fig. 1). CD11b is an integrin molecule expressed on granular cells (neutrophils, eosinophils, and basophils) and macrophages (22). Since gating for flow cytometry analysis of CD11b+ cells precluded macrophages, and few RB6-8C5+ neutrophils were detected in the BAL, it is likely these cells represented eosinophils and/or basophils. These results are markedly different than those observed following similar RSV infection in BALB/c mice, where BAL cell numbers may reach 3 × 105 to 5 × 105 cells/lung between day 3 to 5 pi and substantially higher percentages of DX5+, RB6-8C5+, and CD11b+ cells may be found (39, 45).
FIG. 1.
Innate immune cell trafficking to the lung following HMPV infection. Mice were i.n. infected with 106 PFU HMPV/75 and BAL cells recovered at the time points indicated for cell surface staining of A) DX5+ cells; B) CD11b+ cells; and C) RB6-8C5+ cells. Each bar represents the mean ± SD from three separate experiments.
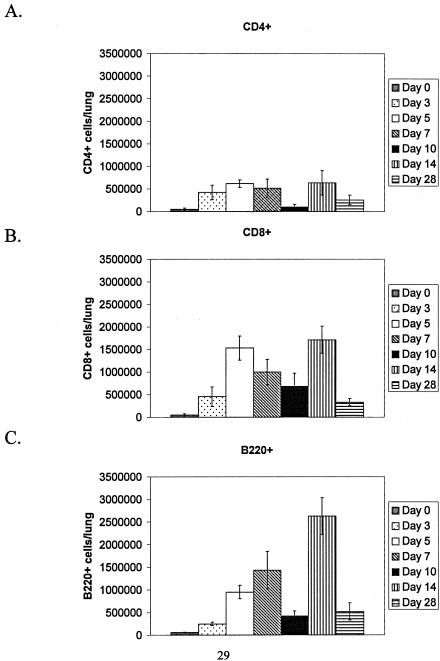
Flow cytometry and lymphocyte gating of BAL cells was used to determine the percentages of positive CD4+, CD8+ or B220+ lymphocytes responding to HMPV infection at days 0, 3, 5, 7, 10, 14, and 28 pi (Fig. 2). In similarity to the innate response, the percentage of CD4+ T cells was low at all time points examined (1.5 × 105 to 6.3 × 105 cells/lung); however, a considerable increase in the percentage of CD8+ T cells was detected at day 5 pi (1.2 × 106 to 1.8 × 106 cells/lung) and day 14 pi (1.4 × 106 to 2.1 × 106 cells/lung). Peak CD8+ T-cell numbers were generally associated with peak virus titers in the lung (Table 1), suggesting that these cell types may be responding to virus replication. The percentage of CD8+ T cells declined to lower levels at day 28 pi (2.5 × 105 to 4.2 × 105 cells/lung) despite the presence of infectious HMPV in the lung (Table 1). Similarly, the percentage of B220+ B cells increased at days 5, 7, and 14 pi, with peak numbers occurring at day 14 pi (2.2 × 106 to 3.1 × 106 cells/lung) and fewer B220+ cells detected at day 28 pi (3.3 × 105 cells/lung). The decline in the percentages of CD8+ T cells and B220+ B cells at day 28 pi despite the presence of infectious virus in the lungs may reflect the contraction phase associated with the generation of a virus-specific memory response (40) and/or the consequence of potential immune evasion mechanisms employed by HMPV to establish persistent infection.
FIG. 2.
Lymphocyte trafficking to the lung following HMPV infection. Mice were i.n. infected with 106 PFU HMPV/75 and BAL cells recovered at the time points indicated for cell surface staining of A) CD4+ cells; B) CD8+ cells; and C) B220+ cells. Each bar represents the mean ± SD from three separate experiments.
Expression of activation and adhesion molecules on leukocytes was examined, as the expression pattern of these molecules contributes to cell trafficking and provides a qualitative method for discriminating between “naïve” (CD44LO CD62LHI) and “antigen-experienced” (CD44HI/CD62LLO) immune cells (40). The percentages of positive CD4+/CD44HI, CD8+/CD44HI, CD4+/CD62LLO, or CD8+/CD62LLO T cells in the BAL were determined at days 0, 3, 5, 7, 10, 14, and 28 pi (data not shown). No significant differences in CD44 or CD62L expression by CD4+ or CD8+ were detected at any time point examined, except for a slight increase in CD44HI expression by CD4+ and CD8+ T cells at day 5 pi. These results are consistent with the minimal pulmonary BAL cell response associated with HMPV infection and the hypothesis that primary HMPV infection does not engender a prominent immune response, an effect that may contribute to persistence.
Intracellular Th1/Th2 cytokine expression by CD4+ and CD8+ T cells.
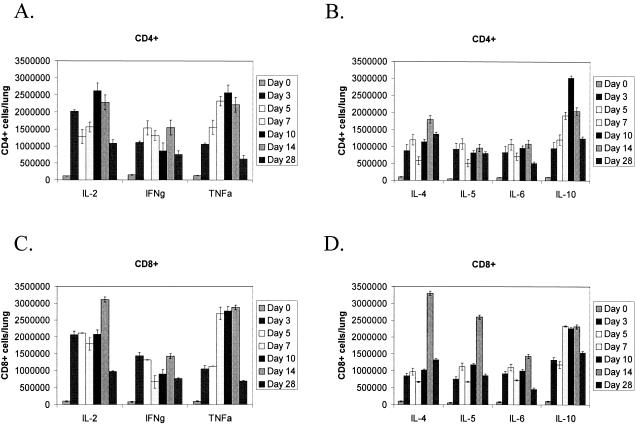
The pattern of Th1- or Th2-type cytokine expression affects the course of the immune response to virus infection. For example, IFN-γ produced by Th1-type cells stimulates Th1 development and inhibits Th2 development, whereas IL-10 produced by Th2-type cells has the opposite effect (40). To determine the pattern of Th1- or Th2-type cytokine expression by BAL cells, flow cytometry was used to analyze intracellular Th1-type (IL-2, IFN-γ, TNF-α) or Th2-type (IL-4, IL-5, IL-6 IL-10) cytokine expression by CD4+- or CD8+-gated T cells at days 0, 3, 5, 7, 10, 14, and 28 pi (Fig. 3). Early in the response to HMPV infection, i.e., between days 3 and 10 pi, the magnitude of IL-2, IFN-γ, and TNF-α expression by CD4+ T cells was generally higher compared to IL-4, IL-5, IL-6 or IL-10 expression; however, between days 7 and 28 pi, high IL-10 expression was evident. These results suggest that the early CD4+ T-cell response to HMPV infection is predominately Th1-like, but between days 7 to 28 pi, a substantial Th2-type IL-10 response occurs (Fig. 2). Similarly, the magnitude of IL-2 expression by CD8+ T cells was substantially higher than IL-4, IL-5, IL-6, or IL-10 expression between days 3 and 10 pi, and the magnitude of TNF-α expression was higher between days 7 and 10 pi; however, CD8+ T cells expressed high levels of IL-10 between days 7 and 28 pi (Fig. 3). IFN-γ expression by CD4+ and CD8+ T cells was generally low at all time points examined (7.6 × 105 to 15.4 × 105 cells/lung), and IFN-γ expression by CD8+ T cells was significantly lower at day 7 pi (P < 0.01) compared to day 3, 5, 14, or 28 pi. The low IFN-γ expression by CD8+ T cells and increased IL-10 expression by CD4+ and CD8+ T cells at day 7 pi coincided with peak virus replication in the lungs (Table 1), suggesting that the pattern and magnitude of expression of these cytokines may be related to increased HMPV replication and/or development of persistence.
FIG. 3.
Intracellular cytokine expression following HMPV infection. Mice were i.n. infected with 106 PFU HMPV/75 and BAL cells recovered at the time points indicated. CD4+- and CD8+-gated T cells were examined for intracellular Th1 or Th2 cytokine expression: A) CD4+ T cells/Th1 cytokines; B) CD4+ T cells/Th2 cytokines; C) CD8+ T cells/Th1 cytokines; and D) CD8+ T cells/Th2 cytokines. Each bar represents the mean ± SD from three separate experiments.
HMPV replicates in the presence of a HMPV-specific antibody response.
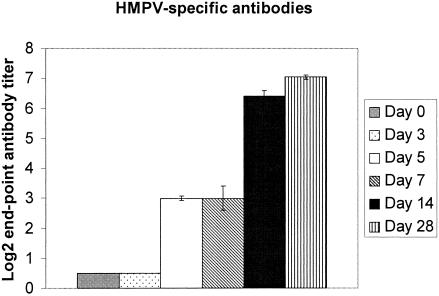
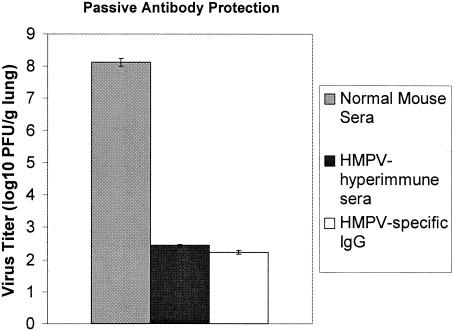
Previous studies by our laboratory showed that infectious HMPV occurs in the lungs of BALB/c mice up to day 60 pi (101 PFU/g lung tissue) despite the presence of neutralizing antibodies which can be detected by day 14 pi and which reach neutralizing titers ≥ 6.46 log2 by day 28 pi (2). In this study, the serum levels of HMPV-specific IgG were determined at days 0, 3, 5, 7, 10, 14, and 28 pi (Fig. 4). The mean endpoint titers from individual HMPV-infected mice (n ≥ 6) were similar to the mean endpoint antibody titer in pooled sera from HMPV-infected mice (n = four to six), a finding consistent with our previous study and findings reported for HMPV-infected hamsters (34). Serum antibody titers to HMPV were undetectable until day 14 pi and peaked at day 28 pi. These results are consistent with the decrease in HMPV lung titers that occurs between day 14 pi (107 PFU/g lung tissue) and day 28 pi (104 PFU/g lung tissue) and are suggestive that the rise in serum HMPV-specific IgG may contribute to regulation of virus replication. To determine the protective efficacy of HMPV-immune sera, 150 μg of normal mouse sera, hyperimmune HMPV-specific mouse sera, or affinity-purified IgG from hyperimmune HMPV-specific mouse sera (neutralizing endpoint titers ≥ 7.04 log2; data not shown) was passively transferred to naïve BALB/c mice and the mice were intranasally challenged with 106 PFU of HMPV. At day 7 pi, virus titers in the lungs of mice treated with normal mouse sera were higher (8.12 PFU/g lung) compared to those of mice treated with HMPV-specific hyperimmune sera (2.45 PFU/g lung) or affinity-purified IgG from hyperimmune HMPV-specific mouse sera (2.235 PFU/g lung) (Fig. 5). These results indicate that HMPV-specific antibodies provide a level of protection from HMPV challenge when passively transferred to naïve mice. However, our recent findings showing that HMPV persists in the lungs despite the presence of a neutralizing antibody response (2) suggest that persistence likely occurs before a substantial neutralizing antibody response develops.
FIG. 4.
HMPV-specific antibody response associated with infection. HMPV-specific antibodies in sera were quantitated by ELISA at the time points indicated following i.n. infection of BALB/c mice. Each bar represents the mean ± SD from three separate experiments.
FIG. 5.
HMPV hyperimmune sera protects from HMPV challenge. Naïve mice were intraperitoneally administered 150 μg of normal mouse sera, hyperimmune HMPV-specific mouse sera, or affinity-purified IgG from hyperimmune HMPV-specific mouse sera (neutralizing endpoint titers ≥ 7.04 log2; data not shown), rested, and subsequently intranasally challenged with 106 PFU of HMPV. At day 7 pi, the virus titers in the lungs of treated mice were determined. Each point represents the mean ± SD from three separate experiments.
The CTL response to HMPV is delayed.
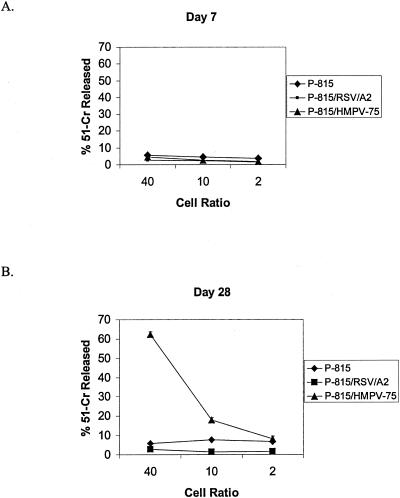
During primary RSV or influenza virus infection in mice, the peak of CTL activity (days 7 to 10 pi) is generally associated with peak resistance to respiratory virus replication (8, 9, 16, 17, 41). To determine whether the anti-HMPV CTL response was functional at day 7 pi, a peak time point of HMPV replication in the lung (107 PFU/g lung tissue), or at day 28 pi, CTL activity toward syngeneic H-2d-restricted P815 target cells infected with either HMPV or RSV (MOI = 10), peptide pulsed with HMPV N peptide or RSV M2 peptide, or uninfected P815 cells was determined (Fig. 6). No CTL activity toward any target was detected at day 7 pi; however, a substantial anti-HMPV CTL response was evident at day 28 pi. Not surprisingly, there was no CTL response toward uninfected P815 cells, P815 cells pulsed with RSV M2 peptide, or P815 cells pulsed with a 20-mer metapneumovirus N peptide (data not shown) previously shown to contain an antibody epitope (3). These results suggest that the CTL response to HMPV is delayed compared to the CTL response to primary infection by RSV or influenza virus (8, 9, 16, 17, 41); however, the decreased HMPV lung titers at day 28 pi compared to other time points examined (Table 1) are suggestive that the CTL response may contribute to control of HMPV replication. These results are consistent with our previous findings showing that antibody depletion of CD3+ T cells is associated with increased HMPV titers in the lung (2).
FIG. 6.
HMPV-specific CTL activity associated with infection. The CTL response to HMPV infection was examined in restimulated cultures of spleen cells isolated from HMPV-infected mice at days 7 and 28 pi using a 51Cr-release assay. Each point represents the mean ± SD from three separate experiments.
DISCUSSION
HMPV is a newly recognized human respiratory pathogen, and similarly to RSV, may cause a spectrum of respiratory illnesses ranging from asymptomatic infection to severe bronchiolitis (6, 7, 11, 19, 49-51). Little is known regarding the mechanisms of immunity and disease pathogenesis associated with HMPV infection; however, a recent prospective study showed that HMPV infection in previously naïve infants elicited a significantly lower inflammatory cytokine response than did RSV infection (18). These results suggest that HMPV and RSV may cause disease by different mechanisms or share a common mechanism that is distinct from factors which contribute to or elicit the inflammatory cytokine response. In this study, we examined aspects of the innate and adaptive immune response associated with HMPV infection in a BALB/c mouse model and showed that primary HMPV infection is associated with an indolent inflammatory response characterized by minimal innate immune and CD4+ T-cell trafficking to the lung, low-level IFN-γ expression, induction of Th2-type IL-10 expression at later stages of infection, and delayed CTL activity that coincides with persistent virus replication in the lung. These findings are consistent with the attenuated inflammatory response in HMPV-infected infants (18) and our previous studies showing that HMPV replicates in the lungs with biphasic growth kinetics in which infectious HMPV can be recovered from lungs up to day 60 pi and that genomic HMPV RNA can be detected in the lungs for ≥180 days pi by reverse transcription-PCR (2).
Viruses employ a variety of strategies to weaken or evade the immune response, including replication in immune-privileged sites, down-regulation of immune molecules, antigenic variation, and molecular mimicry (38, 40). RSV appears to employ a variety of strategies to promote virus replication. For example, the central conserved cysteine noose region of the RSV G protein has been shown to contain a CX3C chemokine motif at amino acid positions 182 to 186 capable of interacting with the CX3C chemokine receptor, CX3CR1, inhibiting fractalkine-mediated responses and altering trafficking of CX3CR1+ cells, which include T lymphocytes, NK cells, monocytes, and macrophages (44). The putative HMPV G protein does not contain a CX3C chemokine motif; thus, it is unlikely to similarly antagonize fractalkine-mediated responses. However, based on the distribution of hydrophobic and hydrophilic regions along the predicted amino acid sequence (30), the HMPV G protein appears similar to RSV G protein, which consists of a type II mucin-like glycoprotein (21). RSV G protein expression has been associated with altered Th1-type cytokine (45) and CC and CXC chemokine mRNA expression (43) by BAL cells responding to infection. The mechanism(s) associated with RSV G protein expression and altered cytokine or chemokine mRNA expression is not known; thus, it is possible that potential structural similarities between RSV and HMPV G protein may allow for similar effects. In addition, the RSV genome contains genes which encode nonstructural proteins NS1 and NS2 that suppress the induction of alpha, beta, and gamma interferons in RSV-infected human epithelial cells and by macrophages (35); however, these genes are absent in the HMPV genome (47).
An association between virus persistence and disease chronicity has been indicated for RSV infection of young children and asthma exacerbation (20, 26, 28). Several studies suggest that RSV may become latent or persist in vivo. For example, RSV latency or persistence has been reported in mice, and RSV protein and genomic RNA have been detected in guinea pig alveolar macrophages for at least 60 days after infection and similarly in B lymphocytes following infection with bovine RSV (38). The results in this study, and from our previous study (2), show that HMPV persists in the lungs of BALB/c mice, suggesting that HMPV persistence may also affect disease pathogenesis and disease chronicity. Consistent with this hypothesis, HMPV has been shown to exacerbate asthma and wheezing in young children (29), cocirculate with RSV (27, 36, 50), and cause similar clinical disease as RSV (52). Notably, HMPV persists in the lungs despite the presence of an HMPV-specific IgG response and neutralizing antibody response (2), and depletion of T cells or NK cells results in increased HMPV replication (2). These results suggest that both innate and adaptive arms of the immune response may be important in controlling HMPV replication and persistence.
In summary, BALB/c mice provide a highly reproducible small animal model to investigate the mechanisms of immunity and disease pathogenesis associated with HMPV infection. These results from this study suggest that aspects of the immune response to HMPV infection are unique compared to RSV results, a finding consistent with the attenuated inflammatory response in children infected with HMPV compared to RSV (18). The availability of a mouse model to investigate parameters of immunity and disease pathogenesis associated with HMPV infection will help to contribute to our understanding of the pathophysiology associated with infection and to the development of vaccine and treatment modalities to combat HMPV infection.
Acknowledgments
We thank Larry J. Anderson (Center for Disease Control and Prevention, Atlanta), who supported preliminary studies, and the Georgia Research Alliance, who provided additional support for R. Alvarez and these studies.
REFERENCES
- 1.Alfieri, C., F. Ghibu, and J. H. Joncas. 1984. Lytic, nontransforming Epstein-Barr virus (EBV) from a patient with chronic active EBV infection. Can. Med. Assoc. J. 131:1249-1252. [PMC free article] [PubMed] [Google Scholar]
- 2.Alvarez, R., K. S. Harrod, W. J. Shieh, S. Zaki, and R. A. Tripp. 2004. Human metapneumovirus persists in BALB/c mice despite the presence of neutralizing antibodies. J. Virol. 78:14003-14011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Alvarez, R., L. P. Jones, B. S. Seal, D. R. Kapczynski, and R. A. Tripp. 2004. Serological cross-reactivity of members of the Metapneumovirus genus. Virus Res. 105:67-73. [DOI] [PubMed] [Google Scholar]
- 4.Biacchesi, S., M. H. Skiadopoulos, G. Boivin, C. T. Hanson, B. R. Murphy, P. L. Collins, and U. J. Buchholz. 2003. Genetic diversity between human metapneumovirus subgroups. Virology 315:1-9. [DOI] [PubMed] [Google Scholar]
- 5.Biacchesi, S., M. H. Skiadopoulos, K. C. Tran, B. R. Murphy, P. L. Collins, and U. J. Buchholz. 2004. Recovery of human metapneumovirus from cDNA: optimization of growth in vitro and expression of additional genes. Virology 321:247-259. [DOI] [PubMed] [Google Scholar]
- 6.Boivin, G., Y. Abed, G. Pelletier, L. Ruel, D. Moisan, S. Cote, T. C. Peret, D. D. Erdman, and L. J. Anderson. 2002. Virological features and clinical manifestations associated with human metapneumovirus: a new paramyxovirus responsible for acute respiratory-tract infections in all age groups. J. Infect. Dis. 186:1330-1334. [DOI] [PubMed] [Google Scholar]
- 7.Boivin, G., G. De Serres, S. Cote, R. Gilca, Y. Abed, L. Rochette, M. G. Bergeron, and P. Dery. 2003. Human metapneumovirus infections in hospitalized children. Emerg. Infect. Dis. 9:634-640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Doherty, P. C., S. Hou, and R. A. Tripp. 1994. CD8+ T-cell memory to viruses. Curr. Opin. Immunol. 6:545-552. [DOI] [PubMed] [Google Scholar]
- 9.Doherty, P. C., D. J. Topham, R. A. Tripp, R. D. Cardin, J. W. Brooks, and P. G. Stevenson. 1997. Effector CD4+ and CD8+ T-cell mechanisms in the control of respiratory virus infections. Immunol. Rev. 159:105-117. [DOI] [PubMed] [Google Scholar]
- 10.Ebihara, T., R. Endo, H. Kikuta, N. Ishiguro, M. Yoshioka, X. Ma, and K. Kobayashi. 2003. Seroprevalence of human metapneumovirus in Japan. J. Med. Virol. 70:281-283. [DOI] [PubMed] [Google Scholar]
- 11.Falsey, A. R., D. Erdman, L. J. Anderson, and E. E. Walsh. 2003. Human metapneumovirus infections in young and elderly adults. J. Infect. Dis. 187:785-790. [DOI] [PubMed] [Google Scholar]
- 12.Freymouth, F., A. Vabret, L. Legrand, N. Eterradossi, F. Lafay-Delaire, J. Brouard, and B. Guillois. 2003. Presence of the new human metapneumovirus in French children with bronchiolitis. Pediatr. Infect. Dis. J. 22:92-94. [DOI] [PubMed] [Google Scholar]
- 13.Graham, B. S., M. D. Perkins, P. F. Wright, and D. T. Karzon. 1988. Primary respiratory syncytial virus infection in mice. J. Med. Virol. 26:153-162. [DOI] [PubMed] [Google Scholar]
- 14.Hasselblom, S., A. Linde, and B. Ridell. 2004. Hodgkin's lymphoma, Epstein-Barr virus reactivation and fatal haemophagocytic syndrome. J. Intern. Med. 255:289-295. [DOI] [PubMed] [Google Scholar]
- 15.Jartti, T., B. van den Hoogen, R. P. Garofalo, A. D. Osterhaus, and O. Ruuskanen. 2002. Metapneumovirus and acute wheezing in children. Lancet 360:1393-1394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kulkarni, A. B., M. Connors, C. Y. Firestone, H. C. Morse III, and B. R. Murphy. 1993. The cytolytic activity of pulmonary CD8+ lymphocytes, induced by infection with a vaccinia virus recombinant expressing the M2 protein of respiratory syncytial virus (RSV), correlates with resistance to RSV infection in mice. J. Virol. 67:1044-1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kulkarni, A. B., H. C. Morse III, J. R. Bennink, J. W. Yewdell, and B. R. Murphy. 1993. Immunization of mice with vaccinia virus-M2 recombinant induces epitope-specific and cross-reactive Kd-restricted CD8+ cytotoxic T cells. J. Virol. 67:4086-4092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Laham, F. R., V. Israele, J. M. Casellas, A. M. Garcia, C. M. Lac Prugent, S. J. Hoffman, D. Hauer, B. Thumar, M. I. Name, A. Pascual, N. Taratutto, M. T. Ishida, M. Balduzzi, M. Maccarone, S. Jackli, R. Passarino, R. A. Gaivironsky, R. A. Karron, N. R. Polack, and F. P. Polack. 2004. Differential production of inflammatory cytokines in primary infection with human metapneumovirus and with other common respiratory viruses of infancy. J. Infect. Dis. 189:2047-2056. [DOI] [PubMed] [Google Scholar]
- 19.Maggi, F., M. Pifferi, M. Vatteroni, C. Fornai, E. Tempestini, S. Anzilotti, L. Lanini, E. Andreoli, V. Ragazzo, M. Pistello, S. Specter, and M. Bendinelli. 2003. Human metapneumovirus associated with respiratory tract infections in a 3-year study of nasal swabs from infants in Italy. J. Clin. Microbiol. 41:2987-2991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Martinez, F. D. 2003. Respiratory syncytial virus bronchiolitis and the pathogenesis of childhood asthma. Pediatr. Infect. Dis. J. 22:S76-S82. [DOI] [PubMed] [Google Scholar]
- 21.Martinez, I., J. Dopazo, and J. A. Melero. 1997. Antigenic structure of the human respiratory syncytial virus G glycoprotein and relevance of hypermutation events for the generation of antigenic variants. J. Gen. Virol. 78(Pt. 10):2419-2429. [DOI] [PubMed] [Google Scholar]
- 22.Mazzone, A., and G. Ricevuti. 1995. Leukocyte CD11/CD18 integrins: biological and clinical relevance. Haematologica 80:161-175. [PubMed] [Google Scholar]
- 23.Mullins, J. A., D. D. Erdman, G. A. Weinberg, K. Edwards, C. B. Hall, F. J. Walker, M. Iwane, and L. J. Anderson. 2004. Human metapneumovirus infection among children hospitalized with acute respiratory illness. Emerg. Infect. Dis. 10:700-705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nash, A. A., B. M. Dutia, J. P. Stewart, and A. J. Davison. 2001. Natural history of murine gamma-herpesvirus infection. Philos. Trans. R. Soc. Lond. B Biol. Sci. 356:569-579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nissen, M. D., D. J. Siebert, I. M. Mackay, T. P. Sloots, and S. J. Withers. 2002. Evidence of human metapneumovirus in Australian children. Med. J. Aust. 176:188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Openshaw, P. J., G. S. Dean, and F. J. Culley. 2003. Links between respiratory syncytial virus bronchiolitis and childhood asthma: clinical and research approaches. Pediatr. Infect. Dis. J. 22:S58-S64; discussion S64-S65. [DOI] [PubMed] [Google Scholar]
- 27.Osterhaus, A., and R. Fouchier. 2003. Human metapneumovirus in the community. Lancet 361:890-891. [DOI] [PubMed] [Google Scholar]
- 28.Peebles, R. S., Jr. 2004. Viral infections, atopy, and asthma: is there a causal relationship? J. Allergy Clin. Immunol. 113:S15-S18. [DOI] [PubMed] [Google Scholar]
- 29.Peiris, J. S., W. H. Tang, K. H. Chan, P. L. Khong, Y. Guan, Y. L. Lau, and S. S. Chiu. 2003. Children with respiratory disease associated with metapneumovirus in Hong Kong. Emerg. Infect. Dis. 9:628-633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Peret, T. C., Y. Abed, L. J. Anderson, D. D. Erdman, and G. Boivin. 2004. Sequence polymorphism of the predicted human metapneumovirus G glycoprotein. J. Gen. Virol. 85:679-686. [DOI] [PubMed] [Google Scholar]
- 31.Peret, T. C., G. Boivin, Y. Li, M. Couillard, C. Humphrey, A. D. Osterhaus, D. D. Erdman, and L. J. Anderson. 2002. Characterization of human metapneumoviruses isolated from patients in North America. J. Infect. Dis. 185:1660-1663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pichler, R., J. Berg, A. Hengstschlager, W. Maschek, J. Wiesinger, and H. Schon. 2001. Recurrent infectious mononucleosis caused by Epstein-Barr virus with persistent splenomegaly. Mil. Med. 166:733-734. [PubMed] [Google Scholar]
- 33.Reed, L. J., and H. Muench. 1938. A simple method of estimating 50 percent end-points. Am. J. Hyg. 27:493-497. [Google Scholar]
- 34.Skiadopoulos, M. H., S. Biacchesi, U. J. Buchholz, J. M. Riggs, S. R. Surman, E. Amaro-Carambot, J. M. McAuliffe, W. R. Elkins, M. St Claire, P. L. Collins, and B. R. Murphy. 2004. The two major human metapneumovirus genetic lineages are highly related antigenically, and the fusion (F) protein is a major contributor to this antigenic relatedness. J. Virol. 78:6927-6937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Spann, K. M., K. C. Tran, B. Chi, R. L. Rabin, and P. L. Collins. 2004. Suppression of the induction of alpha, beta, and gamma interferons by the NS1 and NS2 proteins of human respiratory syncytial virus in human epithelial cells and macrophages. J. Virol. 78:4363-4369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Stockton, J., I. Stephenson, D. Fleming, and M. Zambon. 2002. Human metapneumovirus as a cause of community-acquired respiratory illness. Emerg. Infect. Dis. 8:897-901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tang, R. S., J. H. Schickli, M. MacPhail, F. Fernandes, L. Bicha, J. Spaete, R. A. Fouchier, A. D. Osterhaus, R. Spaete, and A. A. Haller. 2003. Effects of human metapneumovirus and respiratory syncytial virus antigen insertion in two 3′ proximal genome positions of bovine/human parainfluenza virus type 3 on virus replication and immunogenicity. J. Virol. 77:10819-10828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tripp, R. A. 2004. The brume surrounding respiratory syncytial virus persistence. Am. J. Respir. Crit. Care Med. 169:778-779. [DOI] [PubMed] [Google Scholar]
- 39.Tripp, R. A. 2004. Pathogenesis of respiratory syncytial virus infection. Viral Immunol. 17:165-181. [DOI] [PubMed] [Google Scholar]
- 40.Tripp, R. A. 2003. Role of cytokines in the development and maintenance of memory T cells during respiratory viral infection. Curr. Pharm. Des. 9:51-59. [DOI] [PubMed] [Google Scholar]
- 41.Tripp, R. A., and L. J. Anderson. 1998. Cytotoxic T-lymphocyte precursor frequencies in BALB/c mice after acute respiratory syncytial virus (RSV) infection or immunization with a formalin-inactivated RSV vaccine. J. Virol. 72:8971-8975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tripp, R. A., A. M. Hamilton-Easton, R. D. Cardin, P. Nguyen, F. G. Behm, D. L. Woodland, P. C. Doherty, and M. A. Blackman. 1997. Pathogenesis of an infectious mononucleosis-like disease induced by a murine gamma-herpesvirus: role for a viral superantigen? J. Exp. Med. 185:1641-1650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tripp, R. A., L. Jones, and L. J. Anderson. 2000. Respiratory syncytial virus G and/or SH glycoproteins modify CC and CXC chemokine mRNA expression in the BALB/c mouse. J. Virol. 74:6227-6229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tripp, R. A., L. P. Jones, L. M. Haynes, H. Zheng, P. M. Murphy, and L. J. Anderson. 2001. CX3C chemokine mimicry by respiratory syncytial virus G glycoprotein. Nat. Immunol. 2:732-738. [DOI] [PubMed] [Google Scholar]
- 45.Tripp, R. A., D. Moore, L. Jones, W. Sullender, J. Winter, and L. J. Anderson. 1999. Respiratory syncytial virus G and/or SH protein alters Th1 cytokines, natural killer cells, and neutrophils responding to pulmonary infection in BALB/c mice. J. Virol. 73:7099-7107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tripp, R. A., S. R. Sarawar, and P. C. Doherty. 1995. Characteristics of the influenza virus-specific CD8+ T cell response in mice homozygous for disruption of the H-2lAb gene. J. Immunol. 155:2955-2959. [PubMed] [Google Scholar]
- 47.van den Hoogen, B. G., T. M. Bestebroer, A. D. Osterhaus, and R. A. Fouchier. 2002. Analysis of the genomic sequence of a human metapneumovirus. Virology 295:119-132. [DOI] [PubMed] [Google Scholar]
- 48.van den Hoogen, B. G., J. C. de Jong, J. Groen, T. Kuiken, R. de Groot, R. A. Fouchier, and A. D. Osterhaus. 2001. A newly discovered human pneumovirus isolated from young children with respiratory tract disease. Nat. Med. 7:719-724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Viazov, S., F. Ratjen, R. Scheidhauer, M. Fiedler, and M. Roggendorf. 2003. High prevalence of human metapneumovirus infection in young children and genetic heterogeneity of the viral isolates. J. Clin. Microbiol. 41:3043-3045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Vicente, D., G. Cilla, M. Montes, and E. Perez-Trallero. 2003. Human metapneumovirus and community-acquired respiratory illness in children. Emerg. Infect. Dis. 9:602-603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Vicente, D., M. Montes, G. Cilla, E. G. Perez-Yarza, and E. Perez-Trallero. 2003. Hospitalization for respiratory syncytial virus in the paediatric population in Spain. Epidemiol. Infect. 131:867-872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Xepapadaki, P., S. Psarras, A. Bossios, M. Tsolia, D. Gourgiotis, G. Liapi-Adamidou, A. G. Constantopoulos, D. Kafetzis, and N. G. Papadopoulos. 2004. Human metapneumovirus as a causative agent of acute bronchiolitis in infants. J. Clin. Virol. 30:267-270. [DOI] [PMC free article] [PubMed] [Google Scholar]