Abstract
Rheumatoid arthritis (RA) is a chronic inflammatory autoimmune disorder. Although the joints are typically the first area affected in RA, it can also involve extra-articular regions. This article provides an overview on rheumatoid arthritis-associated interstitial lung disease (RA-ILD), a component of the disease manifestations leading to significant morbidity and mortality. Managing these pulmonary symptoms in people with RA poses a number of difficulties for medical professionals. In this review article, we shed light on the prevalence of RA-ILD and the common pulmonary manifestations of RA, while focusing on the evolving pathogenesis concepts that link them to RA's autoimmune cascade. We also address the diagnostic challenges and the available screening modalities that aid in the early recognition and effective management of these pulmonary complications. Furthermore, glucocorticoids, disease-modifying antirheumatic medications, immunosuppressive medications, and biological agents are among the pharmacological approaches that have been explored in this review study.
Keywords: rheumatoid arthritis, interstitial lung disease in rheumatoid arthritis, rheumatoid arthritis lung complications, interstitial lung disease treatment, pulmonary involvement in rheumatoid arthritis, interstitial lung disease
Introduction and background
Rheumatoid arthritis (RA) is a chronic systemic inflammatory disease that can result in irreversible changes to the joints, causing critical disability and poor quality of life. Diagnosis is made by a combination of both clinical and laboratory features. Around 1.3 million people in the United States have RA, representing 0.6% to 1% of the population [1]. The precise cause of RA is unknown; in any case, it appears to result from the interaction between genetic liabilities and environmental factors [2]. Females, smokers, and those with a family history of the illness are most frequently affected [3]. Extra-articular manifestations of the disease can occur in roughly 50% of the affected patients, with the most common site of involvement being the lung [4]. Whereas any part of the lung can be affected, rheumatoid arthritis-associated interstitial lung disease (RA-ILD) is the driving cause of death in patients with RA, leading to significant morbidity and mortality [5]. While it can be the initial presenting manifestation in 10% to 20% of patients, most RA-ILD occurs within the first five years after diagnosis [6]. RA-ILD can present clinically with a wide range of symptoms, the most common of which are dyspnea and cough [7]. Histopathological analysis is the most valuable in classifying and differentiating the different types of interstitial lung disease (ILD), which can be challenging to differentiate [8]. Additionally, high-resolution CT (HRCT) is the gold standard tool for classifying ILD [9]. Current studies highlight that RA-ILD patients may have up to a three-fold chance of mortality compared to those without ILD. Even though the overall mortality from RA is diminishing, mortality from RA-ILD is rising, especially in women and in the elderly population [10,11]. They are raising concerns regarding early diagnosis and effective management of RA-ILD. In this review article, we shed light on the classifications, pathophysiology, and manifestations of RA-ILD, vocalize the possibilities and advantages of screening and early diagnosis, as well as discuss clinical and therapeutic approaches, including immunomodulating and anti-inflammatory drugs, such as biological and disease-modifying antirheumatic drugs (DMARDs).
Review
RA may affect any portion of the lung; it can involve the parenchyma showing as ILD, or it can affect the pleura, leading to pleural inflammation and effusions, small or large airways manifesting as cricoarytenoiditis, constrictive or follicular bronchiolitis, and bronchiectasis. Lastly, it can involve the lung vessels causing vasculitis or pulmonary hypertension [5].
Interstitial lung disease
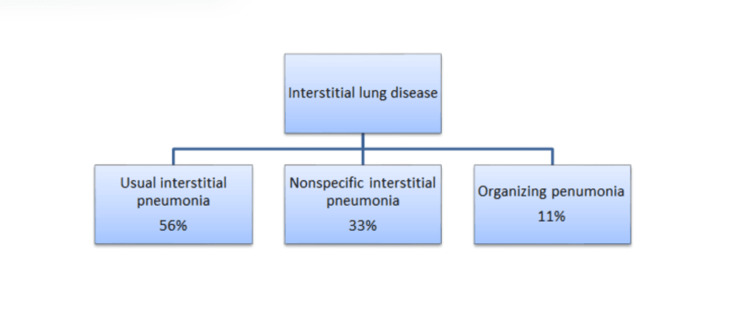
Estimates of prevalence and other general features of RA-ILD vary widely. Several histopathological patterns of RA-ILD have been described. Usual interstitial pneumonia (UIP) is the most frequent one, followed by nonspecific interstitial pneumonia (NSIP); other patterns are seen less frequently [12]. The frequency of RA-ILD forms is summarized in Figure 1. The consensus classification for idiopathic interstitial pneumonias (IIPs) has been used to define RA-ILD, as there is no specific categorization for the condition [13]. Acute interstitial pneumonia, diffuse alveolar damage, organizing pneumonia (OP), desquamative interstitial pneumonia, lymphocytic interstitial pneumonia, and others have also been identified in patients with RA [14,15]. On HRCT, UIP is characterized by honeycombing that appears as clustered cystic airspaces, which are usually 3-10 mm in diameter but can infrequently reach 2.5 cm. It has clearly defined walls and is typically subpleural [16]. According to biopsy findings, NSIP is characterized by diffuse alveolar septal thickening with lymphoplasmacytic inflammation and accentuation around bronchioles.
Figure 1. Common classification of interstitial lung disease in rheumatoid arthritis.
Data retrieved from [9].
Pathophysiology and risk factors
The pathophysiology of RA-ILD still needs to be better understood. Genetics and environmental factors are critical in how ILD develops in RA patients [17]. Some significant human leukocyte antigen (HLA) variations that can contribute to the emergence of ILDs in RA patients include HLA-DRB1, HLA-DR4, and HLA-B40 [18]. Smoking has been shown to interact with HLA-DR shared epitope (SE) genes and play a significant role in initiating the immune response to citrulline-modified proteins [19]. Lung parenchymal and airway damage due to exposure to environmental factors can increase protein citrullination in lung cells [20]. In a genetically susceptible person, the pathological process starts with an inflammatory process that activates cytokines, chemokines, and growth factors such as tumor necrosis factor (TNF), interleukins (IL), and vascular endothelial growth factor (VEGF) [21]. Matrix metalloproteinases (MMP) become hyperactive, and extracellular matrix (ECM) is deposited more readily as a result of fibroblast proliferation and differentiation, which leads to the onset of pulmonary fibrosis and ILD [5]. Recent research revealed that the role of IL-17 in the development of pulmonary fibrosis in mice with RA-ILD and idiopathic pulmonary fibrosis (IPF) may have clinical implications for treatment strategies targeting pulmonary fibrosis in both conditions. Transforming growth factor-1 and other TH17 cytokines, like IL-17A, stimulate fibroblast proliferation and ECM production, contributing to fibrosis development [22,23]. There are significant variances between IPF and RA-ILD even though both conditions share immunological pathways. Patients with RA-ILD have more inducible bronchial-associated lymphoid tissue in their lung tissue than do those with IPF, indicating that immunological dysregulation may affect RA-ILD more so than IPF [24].
The assessment of risk factors is crucial for RA-ILD due to the disease's relevant prevalence as well as the effect that the diagnosis has on mortality rates and treatment options [25]. A cross-sectional study was done by Juge et al. on patients who met either the American College of Rheumatology (ACR) 1987 classification criteria for RA or the 2010 ACR/European Alliance of Associations for Rheumatology (EULAR) classification criteria for RA. Results have identified independent risk factors for RA‐ILD; compared with patients with RA who do not have ILD, those with RA‐ILD more frequently carried the MUC5B rs35705950 T risk allele, were more frequently men, were older at the onset of RA, and had higher DAS28-ESR (Disease Activity Score in 28 joints using the erythrocyte sedimentation rate) scores. Patients with RA-ILD also exhibited higher mean C-reactive protein levels during follow-up, higher body mass index (BMI), and longer duration of tobacco use (pack years) [26]. Another prospective cohort study conducted in Boston, Massachusetts, including 1419 RA patients who started enrolling subjects in 2003, concluded that the level of disease activity was directly associated with an increased risk of developing RA-ILD, Accordingly, reducing systemic inflammation is hypothesized to alter the course of RA-ILD development [27].
Diagnosis and screening
A multidisciplinary approach, typically comprising pulmonologists, radiologists, and pathologists, is necessary for the differential diagnosis of ILDs [28]. A summary of the 2023 ACR guidelines for the screening of ILD in people with systemic autoimmune rheumatic diseases (SARDs) is included in Table 1. The gold standard method for detecting ILD in RA patients was found to be HRCT, considering its extreme sensitivity [29]. A subpleural, basal predominance, reticular abnormality with honeycombing, traction bronchiectasis, a relative lack of ground-glass opacities, and air trapping on exhale are the hallmarks of the UIP [30]. HRCT can be used to predict the increasing fibrosis of ILD associated with RA in some cases. A retrospective study published in 2019 concluded that a wide distribution of subpleural reticular pattern (RP) and/or interlobular septal thickening and peribronchovascular interstitium (PBVI) thickening on HRCT shows predictivity of progressive fibrosis in RA-ILD [31]. In 2014, a prospective study was done in Switzerland on 205 systemic sclerosis (SSc) patients, suggesting that a specific, reduced HRCT procedure with nine slices distributed according to a basal-apical gradient is a reliable and accurate way to identify ILD in SSc patients. This study proves that it can be incorporated into daily clinical routine for early diagnosis and screening of ILD, with the significant benefit of a low radiation dosage compared with the standard whole-chest HRCT [32]. In clinical practice, multiple pulmonary function tests (PFTs) are often considered when assessing the presence of lung restriction as in RA-ILD; however, it was found that when depending merely on PFTs, there is a considerable chance of missing major SSc-related ILD [33]. There is some research on using transthoracic lung ultrasonography for checking impending pulmonary structural alterations in RA patients. In a study done by Moazedi-Fuerst et al. on 64 consecutive patients with RA and 40 healthy volunteers, 28% of RA patients showed pleural nodules or B-line phenomena. In these patients, CT scans showed signs of incipient ILD [34]. As for monitoring for ILD progression, the 2023 ACR guidelines recommend using HRCT chest and/or PFTs. Using both HRCT chest and PFTs is preferred over using PFTs alone. For people with RA-ILD, it is suggested to use PFTs for monitoring every three to 12 months rather than shorter or longer intervals, for the first year, then less frequently once stable [35]. A summary of studies showing some of the risk factors and diagnosis modalities in RA-ILD is included in Table 2.
Table 1. The 2023 American College of Rheumatology (ACR) guidelines for the screening of ILD in people with systemic autoimmune rheumatic diseases.
PFTs: pulmonary function tests; SARDs: systemic autoimmune rheumatic diseases; ILD: interstitial lung disease; HRCT: high-resolution computed tomography; 6MWD: 6-minute walk test distance.
Data retrieved from [35].
| Screening modality | ACR recommendations |
| Pulmonary function test | Conditionally recommend screening with PFTs for people with SARDs at increased risk of developing ILD. |
| High-resolution CT scan | Conditionally recommend screening with HRCT of the chest for people with SARDs at increased risk of developing ILD. |
| 6-minute walk test distance | Conditionally recommend against screening with 6MWD chest for people with SARDs at increased risk of developing ILD. |
| Chest radiography | Conditionally recommend against screening with chest radiography for people with SARDs at increased risk of developing ILD. |
| Ambulatory desaturation testing | Conditionally recommend against screening with ambulatory desaturation testing for people with SARDs at increased risk of developing ILD. |
| Bronchoscopy | Conditionally recommend against screening with bronchoscopy for people with SARDs at increased risk of developing ILD. |
| Surgical lung biopsy | Strongly recommend against screening with surgical lung biopsy for people with SARDs at increased risk of developing ILD. |
Table 2. Overview of the included studies.
RA: rheumatoid arthritis; RA-ILD: rheumatoid arthritis-associated interstitial lung disease; HRCT: high-resolution computed tomography; SSc: systemic sclerosis; SSc-ILD: systemic sclerosis-associated interstitial lung disease; DAS28-ESR: Disease Activity Score in 28 joints using the erythrocyte sedimentation rate.
| References | Publication year | Type of the study | Population | Conclusion |
| Juge et al. [26] | 2022 | Cross-sectional study | The discovery population incorporated 163 patients with RA, and the replication population incorporated 89 patients with RA. | Findings suggest that patients with RA‐ILD more frequently (1) carried the MUC5B rs35705950 T risk allele; (2) were more frequently men; (3) were older at the onset of RA; (4) had higher DAS28-ESR and higher mean C-reactive protein levels; (4) had a higher body mass index; (5) had a longer duration of tobacco use (pack years). |
| Sparks et al. [27] | 2019 | Prospective cohort study | 1,419 participants at Brigham and Women’s Hospital in Boston; all subjects have RA according to the treating physician and accepted criteria. | Active RA is a risk factor for the emergence of RA-ILD. In contrast to remission/low disease activity, high/moderate disease activity was linked to a two-fold increased risk of RA-ILD. |
| Li et al. [31] | 2019 | Retrospective study | 1096 RA patients, 213 of whom had a diagnosis of RA-ILD underwent serial chest HRCT. | Increasing fibrosis in RA-ILD appears to be more likely predicted by HRCT abnormalities than by other criteria. |
| Frauenfelder et al. [32] | 2014 | Prospective study | A total of 205 consecutive patients with a diagnosis of SSc and annual follow-up in the Department of Rheumatology, University Hospital Zurich. | Reduced chest HRCT procedure has the advantage of a significantly lower radiation dose compared to traditional whole-chest HRCT, and it reliably detects even moderate SSc-ILD in clinical practice. |
| Moazedi-Fuerst et al. [34] | 2014 | Prospective study | 64 consecutive patients with rheumatoid arthritis and 40 healthy volunteers. | Transthoracic ultrasound of the lung is an affordable and secure method for screening RA patients for developing pulmonary structural abnormalities. |
Treatment
Immunosuppressants
The effect of immunosuppressants on typical interstitial pneumonia (UIP) in RA or connective tissue disease (CTD) is unclear. However, some retrospective research has revealed that immunosuppressants perform better in ILD types other than UIP. Thus, immunosuppressive medication might be more effective on RA-ILD with NSIP or OP patterns than on the UIP pattern [13,36]. However, a lung biopsy can also reveal varied histologic abnormalities in different specimens, and some individuals have unclassifiable or mixed patterns on HRCT. Unfortunately, not much data exists that explains how well the medication was performed and how the condition progressed in these patients over time [37].
Given their effectiveness in CTD-ILD rather than RA-ILD, glucocorticoids are frequently included in the first therapy regimen for clinically significant RA-ILD [38]. NSIP and OP ILD patterns are more likely to respond to glucocorticoids than UIP [38]. In a retrospective case series published in 2018 on 26 ILD patients with underlying CTD diagnoses, prednisone and oral tacrolimus were administered after two courses of pulse-dose methylprednisolone therapy, which seemed to be well tolerated and to have multifaceted efficacy [39]; on the contrary, there is no evidence to support a role for steroids as monotherapy in IPF. Mayo Clinic investigators presented a retrospective intent-to-treat study of survival for 157 IPF patients receiving no medication, 54 patients receiving maintenance doses of prednisolone alone, 167 patients receiving colchicine alone, and 71 patients receiving both colchicine and prednisolone. There was no statistically significant difference in survival between individuals receiving prednisolone medication and those not receiving any treatment after adjusting for age, sex, and lung function [40]. Corticosteroids raised the risk of life-threatening infections in RA-ILD patients. Despite the use of DMARDs, It was discovered that a higher frequency of infections was associated with a mean daily dose of prednisone greater than 10 mg [41]. As such, their best use is in the early management of acute exacerbations or their treatment until new medicines with better long-term safety profiles are introduced.
Other immunomodulatory treatments such as mycophenolate mofetil (MMF), cyclophosphamide, azathioprine, cyclosporine, and tacrolimus may also be used to treat RA-ILD, but their effects are still unclear. MMF was well tolerated in a large, heterogeneous cohort of 125 CTD-ILD patients (18 with RA-ILD), and it had a low discontinuation rate. Over a median follow-up of 2.5 years, MMF therapy was linked to either stable or better pulmonary physiology [42].
Despite the lack of controlled clinical trials for cyclophosphamide in RA-ILD and a lack of efficacy data, it continues to be used in clinical practice [43], especially in cases of ILD that are highly progressive. In 2019, a retrospective study examined the variables linked to progression and survival in 266 RA-ILD patients, finding that those receiving cyclophosphamide treatment had a better prognosis [44]. In RA-ILD, azathioprine is frequently used as an alternative to methotrexate. According to a single-center retrospective cohort analysis of CTD-ILD, azathioprine patients experienced comparable clinical events and longitudinal PFTs compared to MMF patients (n = 97, 24% RA-ILD) [45]. Although these additional immunomodulatory treatments (such as MMF and azathioprine, for example) may successfully treat ILD, healthcare professionals must consider the possibility of increased side effects and less favorable effects on articular disease [46,47].
Conventional Disease-Modifying Antirheumatic Drugs (cDMARDs)
Only 0.3% to 0.4% of RA patients using methotrexate develop pneumonitis [48]. Moreover, methotrexate does not increase the risk of RA-ILD. Results from prospective early RA inception cohorts indicated a tendency for RA patients on methotrexate to have a lower likelihood of developing ILD (odds ratio: 0.54; 95% CI: 0.28-1.06) [49]. A study done by Rojas-Serrano et al. published in 2017 including 78 patients observed a prolonged survival (HR: 0.13, 95% CI: 0.02-0.64) in RA-ILD patients receiving MTX compared to patients receiving other cDMARDs after adjusting for confounding variables [50]. The avoidance of methotrexate in patients with or at risk for RA-ILD may be partly responsible for the higher rate of ILD observed with leflunomide treatment [51]. Leflunomide should be either avoided or used with caution in patients with prior methotrexate pneumonitis and in patients with pre-existing ILD [52]. They indicate that in these situations, it should not be the substitute for methotrexate. Pneumonitis has also been documented with sulfasalazine use [53]. Data on the safety of hydroxychloroquine in RA-ILD are limited.
Biological Disease-Modifying Antirheumatic Drugs (bDMARDs )
Anti-tumor necrosis factor (anti-TNF) agents have demonstrated excellent effectiveness in slowing the advancement of articular disease and symptoms. However, warnings about possible pulmonary toxicity have come up as a result of its growing use [54]. All anti-TNF drugs approved for RA have been correlated with new-onset or worsening of existing ILD: infliximab [55], etanercept [56], and adalimumab [57]; also, some of the newer agents, including certolizumab [58] and golimumab [59]. Other investigations disproved the lung toxicity of TNF inhibitors (TNFi) and demonstrated that these agents have the potential for stabilizing or possibly improving pulmonary interstitial disease [60-62]. The British national prospective observational trial of 367 individuals with pre-existing RA-ILD found that treatment with TNFi did not increase mortality in patients with RA-ILD when compared to cDMARDs; however, the percentage of RA-ILD-related mortality was higher (34%) among patients receiving TNFi therapy [63]. In a longitudinal observational trial involving 263 patients with RA-ILD, it appears that abatacept (ABA) is similarly beneficial in stabilizing dyspnea, lung function, and radiological deterioration in both UIP and NSIP patterns of the disease. Regardless of the radiological pattern, early ABA administration may stop the progression of RA-ILD [64].
Researchers have found that 10% of patients experienced ILD following the introduction of anti-TNF agents in a case series of 226 patients taking anti-TNF drugs (of whom 83% had RA) [65]. Interestingly, anti-TNF-induced ILD has a significant mortality rate of one-third of patients, which rises to two-thirds in patients who already have ILD [65,66]. Anti-TNF therapy should be used cautiously in patients who already have RA-ILD. Other factors that increase the possibility of death include advanced age (>65 years), a later diagnosis of ILD, and increased immunosuppression [66].
Rituximab (RTX) is a monoclonal antibody targeting the B-cell marker CD20 and has been approved for treating RA in anti-TNF nonresponders. Follicular B-cell hyperplasia and interstitial plasma cell infiltrates were found in RA-ILD patients. This raised the possibility that B cells were involved in the disease's etiology and aroused interest in using RTX for the treatment of RA-ILD [67]. In a retrospective multicenter cohort study done in Portugal, RTX seems to be a promising treatment for CTD-ILD patients (61.2% of them were RA patients). Impressive efficacy outcomes were observed in patients with non-specific interstitial pneumonia patterns [68]. On the other hand, a meta-analysis of biological treatments for CTD showed that RTX use is linked to an increase in non-infectious parenchymal lung disease [69].
IL-6 inhibitors such as tocilizumab demonstrated that the profibrotic effects of the proinflammatory cytokine IL-6 are countered by IL-6R inhibition [70], indicating a possible advantage of this therapeutic strategy in pulmonary fibrosis caused by RA. In a case series of four RA patients, it has been observed that tocilizumab (TCZ) as monotherapy maintains or potentially improves ILD [71]. In contrast, some have reported ILD incidence or progression after using tocilizumab [59].
In clinical trials, Janus kinase inhibitors (JAKi), particularly tofacitinib (TOF), have demonstrated encouraging outcomes. According to a multicenter retrospective study, JAKi therapy may be a safe therapeutic option for patients with RA-ILD, leading to significant stability of ILD at HRCT and potentially preventing PFT deterioration [72]. Another prospective study done in 2023 with a total of 28,559 patients with RA found that out of all the patients who received bDMARDs, those on tofacitinib had the lowest incidence of ILD [73].
The anti-fibrotic agents might prevent the disease progression of RA-ILD. At the current time, pirfenidone and nintedanib are the two anti-fibrotics that are being studied for the treatment of IPF. The INBUILD research compared nintedanib to a placebo in patients with progressive, fibrotic lung disease (13% RA-ILD), in 15 different countries. The annual rate of reduction in the forced expiratory volume (FVC) was considerably lower in patients receiving nintedanib than in patients receiving a placebo in those with progressive fibrosing ILD. One prevalent adverse reaction was diarrhea [74]. Pirfenidone appeared to be effective in the unclassifiable ILD study, a phase 2 randomized controlled trial that enrolled 253 patients with progressive fibrosing unclassifiable ILD; FVC decreased by 87.7 mL in the treatment arm compared to 157.1 mL with placebo [75]. A summary of the treatment is included in Table 3.
Table 3. Overview of the included studies.
MTX: methotrexate; RA-ILD: rheumatoid arthritis-associated interstitial lung disease; ILD: interstitial lung disease; CTD-ILD: connective tissue disease-interstitial lung disease; DMARDs: disease-modifying anti-rheumatic drugs; anti-TNF: anti-tumor necrosis factor; JAKi: Janus kinases inhibitors.
| Reference | Publication year | Study design | Conclusion summary |
| Atienza-Mateo et al. [64] | 2024 | Observational longitudinal study | Early administration of abatacept may prevent RA-ILD progression. |
| Baker et al. [73] | 2023 | Retrospective study | Patients treated with tofacitinib had the lowest incidence of ILD compared with patients treated with biological DMARDs. |
| Venerito et al. [72] | 2023 | Retrospective study | JAKi therapy might be a safe therapeutic option for patients with RA-ILD. |
| Maher et al. [75] | 2022 | Placebo-controlled phase 2 trial | Patients with progressive fibrosing unclassifiable ILD could benefit from pirfenidone treatment. |
| Flaherty et al. [74] | 2019 | Placebo-controlled phase 3 trial | The annual rate of reduction in the forced expiratory volume was considerably lower in patients receiving nintedanib. |
| Kiely et al. [49] | 2019 | Prospective cohort | Treatment with MTX was not linked to a higher incidence of RA-ILD. |
| Fu et al. [44] | 2019 | Retrospective cohort | Treatment with cyclophosphamide contributes to improving the prognosis of RA-ILD. |
| Manfredi et al. [71] | 2018 | Case series | Tocilizumab (TCZ) as a monotherapy was reported to stabilize or even improve ILD. |
| Yamano et al. [39] | 2018 | Retrospective case series | Two courses of pulse-dose methylprednisolone therapy followed by prednisone and oral tacrolimus therapy seemed to be well tolerated and effective. |
| Rojas-Serrano et al. [50] | 2017 | - | Treatment with methotrexate was strongly associated with increased survival in RA-ILD patients. |
| Oldham et al. [45] | 2016 | Retrospective cohort | Azathioprine use in CTD-ILD was associated with stability in pulmonary function. |
| Fischer et al. [42] | 2013 | Retrospective study | Treatment with mycophenolate was associated with either stable or improved pulmonary physiology in CTD-ILD patients. |
| Dixon et al. [63] | 2010 | Prospective observational study | When compared to conventional DMARDs, anti-TNF medication does not result in a higher mortality rate in RA-ILD patients. |
| Douglas et al. [40] | 2000 | Retrospective study | Patients using colchicine or prednisone did not significantly vary in survival from those receiving no treatment. |
Lung transplantation should be considered for patients with severe and progressive RA-ILD who do not have extrapulmonary contraindications and who have not responded to appropriate treatment [76].
Limitations
RA is a complex disease. The frequency and intensity of pulmonary symptoms may differ depending on the severity, duration, coexisting conditions, and the form of lung involvement. This study is primarily focused on the common forms of RA-ILD, such as UIP and NSIP, and did not include enough information about the rare forms.
Conclusions
Pulmonary involvement in RA can affect any area of the lung, including the pleura, airways, lung parenchyma, and vessels, with varying degrees and frequency. Lung injury mechanisms have been linked to a combination of autoimmune, environmental, and hereditary causes. HRCT is considered the primary diagnostic modality. The improvement of prognosis and quality of life for patients with lung involvement of RA depends on early detection, multidisciplinary cooperation, and tailored therapy approaches. Steroids are typically the first line of treatment, according to the abovementioned studies. After carefully evaluating the patient's prior medical history, further immunosuppressive medications and biologics may be added to the regimen. In the end, additional research is suggested for the future, specifically controlled clinical trials with more patient populations, to develop new and secure treatment alternatives.
The authors have declared that no competing interests exist.
Author Contributions
Concept and design: Shahad Al-Baldawi
Acquisition, analysis, or interpretation of data: Shahad Al-Baldawi, Gabriel Zúñiga Salazar, Diego Zúñiga, Sneha Balasubramanian, Khawar Tariq Mehmood
Drafting of the manuscript: Shahad Al-Baldawi, Gabriel Zúñiga Salazar, Sneha Balasubramanian, Khawar Tariq Mehmood
Critical review of the manuscript for important intellectual content: Shahad Al-Baldawi, Diego Zúñiga
References
- 1.Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Helmick CG, Felson DT, Lawrence RC, et al. Arthritis Rheum. 2008;58:15–25. doi: 10.1002/art.23177. [DOI] [PubMed] [Google Scholar]
- 2.Epidemiology, pathophysiology, and diagnosis of rheumatoid arthritis: a synopsis. Gibofsky A. https://www.ajmc.com/view/ace017_may14_ra-ce_gibofsky1_s128. Am J Manag Care. 2014;20:128–135. [PubMed] [Google Scholar]
- 3.Diagnosis and management of rheumatoid arthritis. Wasserman AM. https://www.aafp.org/pubs/afp/issues/2011/1201/p1245.html. Am Fam Physician. 2011;84:1245–1252. [PubMed] [Google Scholar]
- 4.Occurrence of extraarticular disease manifestations is associated with excess mortality in a community based cohort of patients with rheumatoid arthritis. Turesson C, O'Fallon WM, Crowson CS, Gabriel SE, Matteson EL. https://www.jrheum.org/content/29/1/62.long. J Rheumatol. 2002;29:62–67. [PubMed] [Google Scholar]
- 5.Rheumatoid arthritis-interstitial lung disease: manifestations and current concepts in pathogenesis and management. Kadura S, Raghu G. Eur Respir Rev. 2021;30:210011. doi: 10.1183/16000617.0011-2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rheumatoid lung disease. Brown KK. Proc Am Thorac Soc. 2007;4:443–448. doi: 10.1513/pats.200703-045MS. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Patterns of interstitial lung disease and mortality in rheumatoid arthritis. Zamora-Legoff JA, Krause ML, Crowson CS, Ryu JH, Matteson EL. Rheumatology (Oxford) 2017;56:344–350. doi: 10.1093/rheumatology/kew391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.An official American Thoracic Society/European Respiratory Society statement: update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Travis WD, Costabel U, Hansell DM, et al. Am J Respir Crit Care Med. 2013;188:733–748. doi: 10.1164/rccm.201308-1483ST. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Histopathologic pattern and clinical features of rheumatoid arthritis-associated interstitial lung disease. Lee HK, Kim DS, Yoo B, Seo JB, Rho JY, Colby TV, Kitaichi M. Chest. 2005;127:2019–2027. doi: 10.1378/chest.127.6.2019. [DOI] [PubMed] [Google Scholar]
- 10.Rheumatoid arthritis-interstitial lung disease-associated mortality. Olson AL, Swigris JJ, Sprunger DB, et al. Am J Respir Crit Care Med. 2011;183:372–378. doi: 10.1164/rccm.201004-0622OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Incidence and mortality of interstitial lung disease in rheumatoid arthritis: a population-based study. Bongartz T, Nannini C, Medina-Velasquez YF, et al. Arthritis Rheum. 2010;62:1583–1591. doi: 10.1002/art.27405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.The multifaceted aspects of interstitial lung disease in rheumatoid arthritis. Cavagna L, Monti S, Grosso V, Boffini N, Scorletti E, Crepaldi G, Caporali R. Biomed Res Int. 2013;2013:759760. doi: 10.1155/2013/759760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lung diseases directly associated with rheumatoid arthritis and their relationship to outcome. Tsuchiya Y, Takayanagi N, Sugiura H, Miyahara Y, Tokunaga D, Kawabata Y, Sugita Y. Eur Respir J. 2011;37:1411–1417. doi: 10.1183/09031936.00019210. [DOI] [PubMed] [Google Scholar]
- 14.Rheumatoid lung disease: prognostic analysis of 54 biopsy-proven cases. Nakamura Y, Suda T, Kaida Y, et al. Respir Med. 2012;106:1164–1169. doi: 10.1016/j.rmed.2012.04.004. [DOI] [PubMed] [Google Scholar]
- 15.Usual interstitial pneumonia in rheumatoid arthritis-associated interstitial lung disease. Kim EJ, Elicker BM, Maldonado F, et al. Eur Respir J. 2010;35:1322–1328. doi: 10.1183/09031936.00092309. [DOI] [PubMed] [Google Scholar]
- 16.An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Raghu G, Collard HR, Egan JJ, et al. Am J Respir Crit Care Med. 2011;183:788–824. doi: 10.1164/rccm.2009-040GL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mechanisms of lung disease development in rheumatoid arthritis. Wang D, Zhang J, Lau J, Wang S, Taneja V, Matteson EL, Vassallo R. Nat Rev Rheumatol. 2019;15:581–596. doi: 10.1038/s41584-019-0275-x. [DOI] [PubMed] [Google Scholar]
- 18.The pathogenesis of rheumatoid arthritis. McInnes IB, Schett G. N Engl J Med. 2011;365:2205–2219. doi: 10.1056/NEJMra1004965. [DOI] [PubMed] [Google Scholar]
- 19.A new model for an etiology of rheumatoid arthritis: smoking may trigger HLA-DR (shared epitope)-restricted immune reactions to autoantigens modified by citrullination. Klareskog L, Stolt P, Lundberg K, et al. Arthritis Rheum. 2006;54:38–46. doi: 10.1002/art.21575. [DOI] [PubMed] [Google Scholar]
- 20.Citrullination and autoimmunity. Valesini G, Gerardi MC, Iannuccelli C, Pacucci VA, Pendolino M, Shoenfeld Y. Autoimmun Rev. 2015;14:490–497. doi: 10.1016/j.autrev.2015.01.013. [DOI] [PubMed] [Google Scholar]
- 21.Antibodies against cyclic citrullinated peptide and IgA rheumatoid factor predict the development of rheumatoid arthritis. Rantapää-Dahlqvist S, de Jong BA, Berglin E, et al. Arthritis Rheum. 2003;48:2741–2749. doi: 10.1002/art.11223. [DOI] [PubMed] [Google Scholar]
- 22.Profibrotic effect of IL-17A and elevated IL-17RA in idiopathic pulmonary fibrosis and rheumatoid arthritis-associated lung disease support a direct role for IL-17A/IL-17RA in human fibrotic interstitial lung disease. Zhang J, Wang D, Wang L, et al. Am J Physiol Lung Cell Mol Physiol. 2019;316:0–97. doi: 10.1152/ajplung.00301.2018. [DOI] [PubMed] [Google Scholar]
- 23.Transforming growth factor beta 1 is present at sites of extracellular matrix gene expression in human pulmonary fibrosis. Broekelmann TJ, Limper AH, Colby TV, McDonald JA. Proc Natl Acad Sci U S A. 1991;88:6642–6646. doi: 10.1073/pnas.88.15.6642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Inducible bronchus-associated lymphoid tissue (iBALT) in patients with pulmonary complications of rheumatoid arthritis. Rangel-Moreno J, Hartson L, Navarro C, Gaxiola M, Selman M, Randall TD. J Clin Invest. 2006;116:3183–3194. doi: 10.1172/JCI28756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pulmonary involvement in early rheumatoid arthritis patients. Habib HM, Eisa AA, Arafat WR, Marie MA. Clin Rheumatol. 2011;30:217–221. doi: 10.1007/s10067-010-1492-5. [DOI] [PubMed] [Google Scholar]
- 26.A risk score to detect subclinical rheumatoid arthritis-associated interstitial lung disease. Juge PA, Granger B, Debray MP, et al. Arthritis Rheumatol. 2022;74:1755–1765. doi: 10.1002/art.42162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rheumatoid arthritis disease activity predicting incident clinically apparent rheumatoid arthritis-associated interstitial lung disease: a prospective cohort study. Sparks JA, He X, Huang J, et al. Arthritis Rheumatol. 2019;71:1472–1482. doi: 10.1002/art.40904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Diagnostic ability of a dynamic multidisciplinary discussion in interstitial lung diseases: a retrospective observational study of 938 cases. De Sadeleer LJ, Meert C, Yserbyt J, et al. Chest. 2018;153:1416–1423. doi: 10.1016/j.chest.2018.03.026. [DOI] [PubMed] [Google Scholar]
- 29.Screening of interstitial lung disease in patients with rheumatoid arthritis: a systematic review. Garrote-Corral S, Silva-Fernández L, Seoane-Mato D, et al. Reumatol Clin (Engl Ed) 2022;18:587–596. doi: 10.1016/j.reumae.2021.07.002. [DOI] [PubMed] [Google Scholar]
- 30.Diagnosis of idiopathic pulmonary fibrosis. An official ATS/ERS/JRS/ALAT clinical practice guideline. Raghu G, Remy-Jardin M, Myers JL, et al. Am J Respir Crit Care Med. 2018;198:0–68. doi: 10.1164/rccm.201807-1255ST. [DOI] [PubMed] [Google Scholar]
- 31.A preliminary study of lung abnormalities on HRCT in patients of rheumatoid arthritis-associated interstitial lung disease with progressive fibrosis. Li L, Gao S, Fu Q, et al. Clin Rheumatol. 2019;38:3169–3178. doi: 10.1007/s10067-019-04673-4. [DOI] [PubMed] [Google Scholar]
- 32.Screening for interstitial lung disease in systemic sclerosis: performance of high-resolution CT with limited number of slices: a prospective study. Frauenfelder T, Winklehner A, Nguyen TD, Dobrota R, Baumueller S, Maurer B, Distler O. Ann Rheum Dis. 2014;73:2069–2073. doi: 10.1136/annrheumdis-2014-205637. [DOI] [PubMed] [Google Scholar]
- 33.Brief report: pulmonary function tests: high rate of false-negative results in the early detection and screening of scleroderma-related interstitial lung disease. Suliman YA, Dobrota R, Huscher D, et al. Arthritis Rheumatol. 2015;67:3256–3261. doi: 10.1002/art.39405. [DOI] [PubMed] [Google Scholar]
- 34.Ultrasound screening for interstitial lung disease in rheumatoid arthritis. Moazedi-Fuerst FC, Kielhauser SM, Scheidl S, et al. https://www.clinexprheumatol.org/abstract.asp?a=7164. Clin Exp Rheumatol. 2014;32:199–203. [PubMed] [Google Scholar]
- 35.American College of Rheumatology. Interstitial lung disease guideline. [ Jan; 2024 ]. 2023. https://rheumatology.org/interstitial-lung-disease-guideline https://rheumatology.org/interstitial-lung-disease-guideline
- 36.Therapeutic options for the treatment of interstitial lung disease related to connective tissue diseases. a narrative review. Vacchi C, Sebastiani M, Cassone G, Cerri S, Della Casa G, Salvarani C, Manfredi A. J Clin Med. 2020;9:407. doi: 10.3390/jcm9020407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Histopathologic variability in usual and nonspecific interstitial pneumonias. Flaherty KR, Travis WD, Colby TV, et al. Am J Respir Crit Care Med. 2001;164:1722–1727. doi: 10.1164/ajrccm.164.9.2103074. [DOI] [PubMed] [Google Scholar]
- 38.Interstitial lung disease guideline: the British Thoracic Society in collaboration with the Thoracic Society of Australia and New Zealand and the Irish Thoracic Society. Bradley B, Branley HM, Egan JJ, et al. Thorax. 2008;63:0–58. doi: 10.1136/thx.2008.101691. [DOI] [PubMed] [Google Scholar]
- 39.Multidimensional improvement in connective tissue disease-associated interstitial lung disease: two courses of pulse dose methylprednisolone followed by low-dose prednisone and tacrolimus. Yamano Y, Taniguchi H, Kondoh Y, et al. Respirology. 2018;23:1041–1048. doi: 10.1111/resp.13365. [DOI] [PubMed] [Google Scholar]
- 40.Idiopathic pulmonary fibrosis: impact of oxygen and colchicine, prednisone, or no therapy on survival. Douglas WW, Ryu JH, Schroeder DR. Am J Respir Crit Care Med. 2000;161:1172–1178. doi: 10.1164/ajrccm.161.4.9907002. [DOI] [PubMed] [Google Scholar]
- 41.Risk of serious infection in patients with rheumatoid arthritis-associated interstitial lung disease. Zamora-Legoff JA, Krause ML, Crowson CS, Ryu JH, Matteson EL. Clin Rheumatol. 2016;35:2585–2589. doi: 10.1007/s10067-016-3357-z. [DOI] [PubMed] [Google Scholar]
- 42.Mycophenolate mofetil improves lung function in connective tissue disease-associated interstitial lung disease. Fischer A, Brown KK, Du Bois RM, et al. J Rheumatol. 2013;40:640–646. doi: 10.3899/jrheum.121043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Usefulness of cyclophosphamide pulse therapy in interstitial lung diseases. Schupp JC, Köhler T, Müller-Quernheim J. Respiration. 2016;91:296–301. doi: 10.1159/000445031. [DOI] [PubMed] [Google Scholar]
- 44.Risk factors for progression and prognosis of rheumatoid arthritis-associated interstitial lung disease: single center study with a large sample of Chinese population. Fu Q, Wang L, Li L, Li Y, Liu R, Zheng Y. Clin Rheumatol. 2019;38:1109–1116. doi: 10.1007/s10067-018-4382-x. [DOI] [PubMed] [Google Scholar]
- 45.Azathioprine response in patients with fibrotic connective tissue disease-associated interstitial lung disease. Oldham JM, Lee C, Valenzi E, et al. Respir Med. 2016;121:117–122. doi: 10.1016/j.rmed.2016.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Methotrexate versus azathioprine in the treatment of rheumatoid arthritis. A forty-eight-week randomized, double-blind trial. Jeurissen ME, Boerbooms AM, van de Putte LB, et al. Arthritis Rheum. 1991;34:961–972. doi: 10.1002/art.1780340805. [DOI] [PubMed] [Google Scholar]
- 47.Mycophenolate mofetil in the treatment of adults with advanced rheumatoid arthritis: three 24-week, randomized, double-blind, placebo- or ciclosporin-controlled trials. Schiff M, Beaulieu A, Scott DL, Rashford M. Clin Drug Investig. 2010;30:613–624. doi: 10.2165/11537460-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 48.Methotrexate and lung disease in rheumatoid arthritis: a meta-analysis of randomized controlled trials. Conway R, Low C, Coughlan RJ, O'Donnell MJ, Carey JJ. Arthritis Rheumatol. 2014;66:803–812. doi: 10.1002/art.38322. [DOI] [PubMed] [Google Scholar]
- 49.Is incident rheumatoid arthritis interstitial lung disease associated with methotrexate treatment? Results from a multivariate analysis in the ERAS and ERAN inception cohorts. Kiely P, Busby AD, Nikiphorou E, et al. BMJ Open. 2019;9:0. doi: 10.1136/bmjopen-2018-028466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rheumatoid arthritis-related interstitial lung disease (RA-ILD): methotrexate and the severity of lung disease are associated to prognosis. Rojas-Serrano J, Herrera-Bringas D, Pérez-Román DI, Pérez-Dorame R, Mateos-Toledo H, Mejía M. Clin Rheumatol. 2017;36:1493–1500. doi: 10.1007/s10067-017-3707-5. [DOI] [PubMed] [Google Scholar]
- 51.Leflunomide use and the risk of interstitial lung disease in rheumatoid arthritis. Suissa S, Hudson M, Ernst P. Arthritis Rheum. 2006;54:1435–1439. doi: 10.1002/art.21806. [DOI] [PubMed] [Google Scholar]
- 52.Clinical expression of leflunomide-induced pneumonitis. Chikura B, Lane S, Dawson JK. Rheumatology (Oxford) 2009;48:1065–1068. doi: 10.1093/rheumatology/kep050. [DOI] [PubMed] [Google Scholar]
- 53.Sulphasalazine and lung toxicity. Parry SD, Barbatzas C, Peel ET, Barton JR. Eur Respir J. 2002;19:756–764. doi: 10.1183/09031936.02.00267402. [DOI] [PubMed] [Google Scholar]
- 54.Treatment of rheumatoid arthritis-associated interstitial lung disease: a perspective review. Iqbal K, Kelly C. Ther Adv Musculoskelet Dis. 2015;7:247–267. doi: 10.1177/1759720X15612250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Development of interstitial pneumonia in a rheumatoid arthritis patient treated with infliximab, an anti-tumor necrosis factor alpha-neutralizing antibody. Mori S, Imamura F, Kiyofuji C, Sugimoto M. Mod Rheumatol. 2006;16:251–255. doi: 10.1007/s10165-006-0491-5. [DOI] [PubMed] [Google Scholar]
- 56.Acute progression of interstitial lung disease: a complication of etanercept particularly in the presence of rheumatoid lung and methotrexate treatment. Lindsay K, Melsom R, Jacob BK, Mestry N. Rheumatology (Oxford) 2006;45:1048–1049. doi: 10.1093/rheumatology/kel090. [DOI] [PubMed] [Google Scholar]
- 57.Pulmonary fibrosis in a patient with rheumatoid arthritis treated with adalimumab. Schoe A, van der Laan-Baalbergen NE, Huizinga TW, Breedveld FC, van Laar JM. Arthritis Rheum. 2006;55:157–159. doi: 10.1002/art.21716. [DOI] [PubMed] [Google Scholar]
- 58.Severe interstitial lung disease following treatment with certolizumab pegol: a case report. Lager J, Hilberg O, Løkke A, Bendstrup E. Eur Respir Rev. 2013;22:414–416. doi: 10.1183/09059180.00002013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Non-infectious pulmonary complications of newer biological agents for rheumatic diseases--a systematic literature review. Hadjinicolaou AV, Nisar MK, Bhagat S, Parfrey H, Chilvers ER, Ostör AJ. Rheumatology (Oxford) 2011;50:2297–2305. doi: 10.1093/rheumatology/ker289. [DOI] [PubMed] [Google Scholar]
- 60.Interstitial lung diseases induced or exacerbated by DMARDs and biologic agents in rheumatoid arthritis: a systematic literature review. Roubille C, Haraoui B. Semin Arthritis Rheum. 2014;43:613–626. doi: 10.1016/j.semarthrit.2013.09.005. [DOI] [PubMed] [Google Scholar]
- 61.Association between anti-TNF-α therapy and interstitial lung disease. Herrinton LJ, Harrold LR, Liu L, et al. Pharmacoepidemiol Drug Saf. 2013;22:394–402. doi: 10.1002/pds.3409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Clinical response of rheumatoid arthritis-associated pulmonary fibrosis to tumor necrosis factor-alpha inhibition. Vassallo R, Matteson E, Thomas CF Jr. Chest. 2002;122:1093–1096. doi: 10.1378/chest.122.3.1093. [DOI] [PubMed] [Google Scholar]
- 63.Influence of anti-TNF therapy on mortality in patients with rheumatoid arthritis-associated interstitial lung disease: results from the British Society for Rheumatology Biologics Register. Dixon WG, Hyrich KL, Watson KD, Lunt M, Symmons DP. Ann Rheum Dis. 2010;69:1086–1091. doi: 10.1136/ard.2009.120626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Abatacept in usual and in non-specific interstitial pneumonia associated with rheumatoid arthritis. Atienza-Mateo B, Fernández-Díaz C, Vicente-Rabaneda EF, et al. Eur J Intern Med. 2024;119:118–124. doi: 10.1016/j.ejim.2023.08.025. [DOI] [PubMed] [Google Scholar]
- 65.Autoimmune diseases induced by TNF-targeted therapies: analysis of 233 cases. Ramos-Casals M, Brito-Zerón P, Muñoz S, et al. Medicine (Baltimore) 2007;86:242–251. doi: 10.1097/MD.0b013e3181441a68. [DOI] [PubMed] [Google Scholar]
- 66.Interstitial lung disease induced or exacerbated by TNF-targeted therapies: analysis of 122 cases. Perez-Alvarez R, Perez-de-Lis M, Diaz-Lagares C, et al. Semin Arthritis Rheum. 2011;41:256–264. doi: 10.1016/j.semarthrit.2010.11.002. [DOI] [PubMed] [Google Scholar]
- 67.Morphologic and quantitative assessment of CD20+ B cell infiltrates in rheumatoid arthritis-associated nonspecific interstitial pneumonia and usual interstitial pneumonia. Atkins SR, Turesson C, Myers JL, Tazelaar HD, Ryu JH, Matteson EL, Bongartz T. Arthritis Rheum. 2006;54:635–641. doi: 10.1002/art.21758. [DOI] [PubMed] [Google Scholar]
- 68.Rituximab in connective tissue disease-associated interstitial lung disease. Duarte AC, Cordeiro A, Fernandes BM, et al. Clin Rheumatol. 2019;38:2001–2009. doi: 10.1007/s10067-019-04557-7. [DOI] [PubMed] [Google Scholar]
- 69.Non-infectious pulmonary toxicity of rituximab: a systematic review. Hadjinicolaou AV, Nisar MK, Parfrey H, Chilvers ER, Ostör AJ. Rheumatology (Oxford) 2012;51:653–662. doi: 10.1093/rheumatology/ker290. [DOI] [PubMed] [Google Scholar]
- 70.Interleukin-6 receptor superantagonist Sant7 inhibits TGF-beta-induced proliferation of human lung fibroblasts. Gallelli L, Falcone D, Pelaia G, et al. Cell Prolif. 2008;41:393–407. doi: 10.1111/j.1365-2184.2008.00538.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Tocilizumab for the treatment of patients with rheumatoid arthritis and interstitial lung diseases: a case series. Manfredi A, Sebastiani M, Cassone G, Colaci M, Sandri G, Ferri C. https://www.clinexprheumatol.org/abstract.asp?a=12161. Clin Exp Rheumatol. 2018;36:342. [PubMed] [Google Scholar]
- 72.Evolution of rheumatoid-arthritis-associated interstitial lung disease in patients treated with JAK inhibitors: a retrospective exploratory study. Venerito V, Manfredi A, Carletto A, et al. J Clin Med. 2023;12:957. doi: 10.3390/jcm12030957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Incidence of interstitial lung disease in patients with rheumatoid arthritis treated with biologic and targeted synthetic disease-modifying antirheumatic drugs. Baker MC, Liu Y, Lu R, Lin J, Melehani J, Robinson WH. JAMA Netw Open. 2023;6:0. doi: 10.1001/jamanetworkopen.2023.3640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Nintedanib in progressive fibrosing interstitial lung diseases. Flaherty KR, Wells AU, Cottin V, et al. N Engl J Med. 2019;381:1718–1727. doi: 10.1056/NEJMoa1908681. [DOI] [PubMed] [Google Scholar]
- 75.Pirfenidone in patients with unclassifiable progressive fibrosing interstitial lung disease: a double-blind, randomised, placebo-controlled, phase 2 trial. Maher TM, Corte TJ, Fischer A, et al. Lancet Respir Med. 2020;8:147. doi: 10.1016/S2213-2600(19)30341-8. [DOI] [PubMed] [Google Scholar]
- 76.Consensus document for the selection of lung transplant candidates: an update from the International Society for Heart and Lung Transplantation. Leard LE, Holm AM, Valapour M, et al. J Heart Lung Transplant. 2021;40:1349–1379. doi: 10.1016/j.healun.2021.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]