Summary
Mast cells (MCs) are multifunctional immune cells that express a diverse repertoire of surface receptors and pre-stored bioactive mediators. They are traditionally recognized for their involvement in allergic and inflammatory responses, yet there is a growing body of literature highlighting their contributions to mounting adaptive immune responses. In particular, there is growing evidence that MCs can serve as antigen-presenting cells, owing to their often close proximity to T cells in both lymphoid organs and peripheral tissues. Recent studies have provided compelling support for this concept, by demonstrating the presence of antigen processing and presentation machinery in MCs and their ability to engage in classical and non-classical pathways of antigen presentation. However, there remain discrepancies and unresolved questions regarding the extent of the MC’s capabilities with respect to antigen presentation. In this review, we discuss our current understanding of the antigen presentation by MCs and its influence on adaptive immunity.
Keywords: mast cells, antigen presentation, DC, CD4 T cell, CD8 T cell
Graphical Abstract
Graphical Abstract.
Introduction
Mast cells (MCs) are granulated immune cells of the myeloid lineage that are dispersed throughout connective and mucosal tissues. A substantial proportion of MCs are located at the host-environment interfaces, including surrounding blood vessels and lymphatic vessels, and proximal to other tissue-resident immune cells [1]. Mature MCs are found only in tissues, not in the blood, and these tissue-resident MCs are heterogeneous in phenotype, appearing in mucosal tissues with an expression of chondroitin sulphate, a type of glycosaminoglycan (GAG) that is attached to the serglycin core protein, as the backbone of their granules, contrasting connective tissue MCs where the granule GAG is heparin [2]. The contents of immunomodulatory compounds entrapped in the granules also vary between different tissues, for example, they have differential inclusion of histamine and MC-specific proteases [2, 3]. With a wide range of receptors presented on its cell surface, a MC can be activated by many types of stimuli, including pathogens, IgE receptor crosslinking, complement components, and neuropeptides. Certain stimuli prompt MC degranulation, resulting in a release of variety of pro-inflammatory immune mediators, such as chemokines, cytokines, biogenic amines, and proteases [2], while others, such as toll-like receptor (TLR) stimulation (e.g. by TLR2, 4, 3, 7, or 9), appear to prompt transcriptional activation without degranulation [4–6]. MCs are most well-known for mediating harmful inflammatory responses, such as during asthma, allergy, or anaphylaxis; nevertheless, the significance of MCs extends beyond their involvement in pathological conditions, with mounting evidence pointing to their vital function in immunosurveillance. MCs play an indispensable role in recognizing and controlling certain infections, including those caused by parasites, bacteria, and viruses [1, 7–10].
The immediate MC-dependent pro-inflammatory responses that occur in tissues in response to insults and infections have an accelerated timeline compared to other immune cells, owing to the ability of MCs to respond within seconds to degranulating stimuli to release pre-formed mediators, contrasting the slower responses involving transcriptional changes. This response can prompt the recruitment of other immune cells from the circulation and lead to vasodilation and increased vascular permeability, improving tissue access by leukocytes [11]. Aside from innate immune activity promoting host defence, these same aspects of MC activation can also promote the development of adaptive immune responses [12–15]. It has been shown that functional adaptive immune responses are impaired in MC-deficient animals following infectious challenges and/or vaccination involving MC-activating stimuli. For example, mice immunized with antigen along with a potent MC activating compound showed increased antigen-specific IgG titres compared to MC-deficient mice (Kitw-sh/Kitw-sh; sash mice) or wild-type (WT) mice without MC stimulation [12]. Moreover, the resulting MC-induced humoral response provided protection against a lethal bacterial challenge [12]. Subsequently, additional studies have supported that MC-activating compounds can be potent vaccine adjuvants [13–15]. However, there are also data suggesting that MC-dependency of the humoral response and adjuvant activity is not always reproducible in other MC-depletion mouse models [12, 16]. Since mouse models of MC deficiency have inherent caveats for interpretation [17–19], the situations in which MCs are involved in humoral or adaptive immune responses remain under debate and investigation.
With respect to cellular immunity, there is also evidence that MCs enhance T cell responses. For example, FcRI-dependent MC activation enhanced T cell responses [20] and impaired T cell activation was observed in MC-deficient models of allergy [20] and infection [21]. Mechanistically, one way that MCs promote early adaptive immune responses is through orchestrating the trafficking of antigen-presenting cells (APCs), such as dendritic cells (DCs), which can be recruited to sites of inflammation dependent on MC-specific products like tumour necrosis factor (TNF), after which they are able to traffic to T cell zones of lymph nodes [22]. TNF has also been shown to promote lymph node hypertrophy, the process of lymphocyte retention in draining lymph nodes, which can increase the likelihood that rare antigen-specific lymphocytes will be present in lymph nodes during the initiation of adaptive immune responses [23, 24]. These studies outline several ways that MCs may influence functional adaptive immunity, including indirectly through the secretion of pro-inflammatory mediators.
Aside from indirect ways that MCs may promote adaptive immune activation, until recently, a debate has surrounded whether MCs can contribute directly to antigen presentation in vivo. In this review, we discuss the recent evidence that MCs function as conventional and non-conventional APCs, with subsequent effects on adaptive immunity and immune protection.
Professional antigen-presenting cells
Contrasting the sentinel function of MCs primarily localized in peripheral tissues or in the sinuses of lymph nodes [1, 3, 25], professional APCs migrate to or are resident in secondary lymphoid organs within the lymph node parenchyma [26]. Professional APCs such as DCs, macrophages, and B cells are characterized by their constitutive expression of major histocompatibility complex class II (MHC-II) and antigen processing machinery [27]. They take up exogenous antigens through multiple pathways including phagocytosis, micropinocytosis, and receptor-mediated endocytosis. These antigens are then trafficked into late-endosomal/lysosomal antigen processing compartments. These multivesicular bodies have a sufficiently low pH and are enriched in disulphate reductases, allowing the lysosomal enzymes (such as cathepsins S, H, and B) to process antigens efficiently, cleaving them into short peptides [27–29]. The invariant chain occupying the MHC-II molecule peptide binding groove is cleaved in the antigen processing compartment to generate class II associated invariant chain peptide (CLIP) [27]. MHC-II accessory molecules (such as HLA-DM and DO) then facilitate the removal of CLIP and the loading of antigenic peptides to the peptide binding groove [30, 31]. Following peptide binding, the peptide-MHC-II complex traffics to the plasma membrane, where antigens are presented to T cells. The presentation of antigen by APCs to T cells often occurs in draining lymph nodes or secondary lymphoid organs within T cell zones, where the physical communication of an antigen-bearing DC and a T cell specific for that antigen can last for hours as the cells form an ‘immunological synapse’ [26]. To become activated, a naïve CD4 T cell must not only recognize its cognate antigen presented on MHC-II but must also receive a secondary activating stimulus in the form of CD80 (B7.1) and/or CD86 (B7.2) expression on the APC surface, which binds to CD28 on T cells [32]. After being activated by DCs, T cells also interact with and license DCs through CD40L (on T cells) interaction with CD40 (on DCs), which results in the secretion of chemokines to recruit CD8 T cells [32]. This is followed by cytokine production from licensed DCs and CD4 T cells, which promotes CD8 T cell survival and their memory recall responses [33, 34]. In addition to providing T cell help to CD8 T cells, CD4 T cells also interact with B cells, as T follicular helper cells (TFH) cells, to promote antibody affinity maturation, resulting in the production of high-affinity antigen-specific antibodies [35]. Another important attribute of professional APCs is their ability to cross-present exogenous antigens via major histocompatibility complex class I (MHC-I) molecules, a process that contrasts normal MHC-I presentation which sources antigens endogenously from the cell cytosol and protein pool [36]. Cross-presentation is thought to be crucial for the elimination of intracellular pathogens and tumours [36]. An advantage of antigen cross-presentation is that APCs are able to initiate a CD8 T cell response without being infected themselves [36]. These interactions between conventional T cells and professional APCs take place mostly in secondary lymphoid organs, such as lymph nodes [26].
MCs may be relatively few in secondary lymphoid organs compared to connective tissue sites, such as skin but, increasingly, evidence supports their potential to perform certain functions of APCs including antigen processing and presentation and activation of T cells, in certain contexts. For example, MCs expressing human leukocyte antigen (HLA)-DR and costimulatory molecule CD80 were found to be present in close proximity to CD4 T cells in human tonsil tissue and were suggested to present antigens to T cells [37]. The unique aspects of MC gene expression and tissue distribution also may hint towards specialized roles in antigen presentation that warrant further investigation. While antigen presentation classically occurs in the draining lymph node T cell zones, there are many examples of antigen presentation events that occur at other sites, such as the presentation of antigen to patrolling T resident memory cells in tissues [38], or the presentation of antigen by non-classical antigen presentation machinery to various subsets of innate-like T cells, which happen to be enriched at peripheral sites [39–41].
Antigen uptake and processing by MCs
An important ability of professional APCs is that they can take up exogenous antigens. While MCs have also been shown to take up antigens from the extracellular environment, the type of antigen appears to influence the endocytic pathways used. For example, human skin-derived MCs were shown to take up cytomegalovirus (CMV) antigen via dynamin-dependent pathways, not through phagocytosis or micropinocytosis, whereas uptake of ovalbumin (OVA) was not restricted to a specific pathway [42, 43]. MCs were also shown to take up antigens by Fc receptor-mediated endocytosis, either by IgG- or IgE-mediated opsonization [42, 43]. Cytokine treatment could also affect antigen uptake, as IFNγ treatment was shown to enhance DEC-205 (C-type lectin) receptor expression, thus promoting DEC-205-mediated antigen internalization [42]. These studies suggest that there are multiple antibody-dependent and independent pathways through which MCs can potentially acquire antigens.
MHC class II and costimulatory molecules expression by MCs
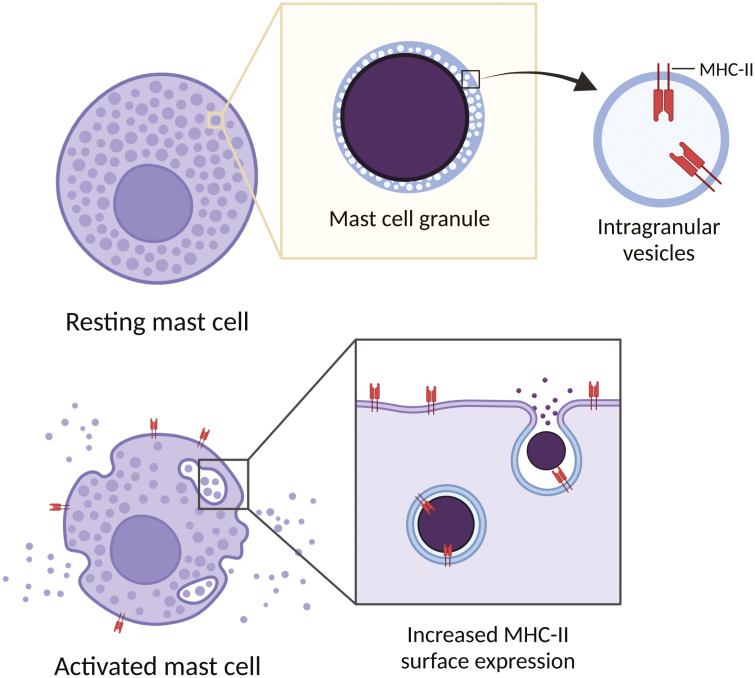
Unlike professional APCs (DCs, macrophages, B cells), MCs do not constitutively express MHC-II on the cell surface in their resting state. In mouse bone marrow-derived MCs (BMMCs), most of the MHC-II molecules are stored in the prelysosome/lysosome-related internal multivesicular membranes of MC granules [44]. Similarly, in humans, HLA-DR was observed to be colocalized with the secretary granules in skin-derived MCs [42]. Degranulation of MCs triggers the fusion of intracellular vesicles with the cell membrane and coincides with the increased expression of MHC-II on the MC surface [42, 44] (Fig. 1), suggesting that MC activation/degranulation could be a checkpoint for physiologically relevant levels of MC antigen presentation. Interestingly, MHC-II can also be induced by activation, independent of degranulation. Treatment of professional APCs with type-I or type-II interferons is known to modulate MHC-II expression [45–48] and, similarly, IFNγ treatment increased HLA-DR on human MCs without affecting degranulation [42]. Furthermore, treatment with anti-FcεRI in addition to IFNγ stimulation promotes longer HLA-DR persistence on the MC surface [42]. MHC-II surface induction was also shown in BMMCs and peritoneal MCs with activated Notch signalling [49]. These data support that MCs are capable of presenting MHC-II at the cell surface but that an activating stimulus may be required for substantial MHC-II presentation. This observation makes it likely that MCs are most consequential to antigen presentation when they are present in a pro-inflammatory microenvironment that induces their activation. Contrasting these observations of the type of reactions MCs are capable of in experimental systems, it should be noted that there are inconsistent findings regarding the expression of MHC-II on MCs in human patients under different conditions. For example, MC expression of MHC-II proteins HLA-DR and HLA-DQ were hardly detected in the skin of patients with various allergic and autoimmune conditions [50]. However, intraplaque MCs in atherosclerosis patients were stained positive for HLA-DR [51]. Nevertheless, the antigen presentation responses of human MCs during infections or during the presentation of non-endogenous antigens remain to be explored.
Figure 1:
MHC-II expression on MCs. MCs have low surface expression of MHC-II in the steady state, where most of the MHC-Il molecules are stored in the intra-granular vesicles. After activation of MCs, MHC-II surface expression increases when intracellular vesicles fuse with the plasma membrane. Diagram was created with Biorender.com.
Apart from the interaction between MHC-II–peptide complexes and T cell receptors (TCRs), the binding of co-stimulatory molecules such as B7 molecules (CD80 and CD86) on APCs with CD28 on T cells also plays a crucial role during the process of MHC-II-mediated antigen presentation [32]. CD80/86 expression on MCs can be influenced by cytokines and growth factors. For example, GM-CSF treatment was shown to increase CD80 and CD86 mRNA expression in BMMCs while IFNγ inhibited their expression [52]. However, there have been some discordant data that may be due to the species of MCs used, their tissue source, or culture conditions [42, 53]. For example, another study showed that IFNγ stimulates CD80 but not CD86 expression by human MCs [42], while a different study detected no effect of IFNγ on spleen-derived MCs [53]. Another co-stimulatory molecule, OX40L, which is commonly seen on APCs and interacts with OX40 on T cells, was also shown to be inducible on MCs, while the regulatory mechanism still remains to be elucidated [49, 54]. Thus, with respect to the ability of MCs to express the co-stimulatory signals required for naïve T cell activation, the context of the microenvironment appears key to whether they are capable of this function.
Peptide loading on MHC-II by MCs
Following the sequential steps in the process of antigen presentation, the next question is whether MCs, which are believed to generate antigenic peptides and express MHC-II molecules, possess the ability to effectively load these peptides onto MHC-II. Using antigens such as dye-quenched (DQ)-OVA or DQ-bovine serum albumin, one study suggested that antigens can be processed by human MCs as measured by proteolysis-mediated fluorescence [42]. YAe staining (which specifically recognizes MHC-II bound to Eα, a peptide derived from the α chain of the mouse MHC-II molecule I-E) was detected on 2,4,6-Trinitrophenyl hapten (TNP)-Eα-treated WT but not MHC-II-deficient mouse splenic MCs in an H2-DM (mouse homologue of HLA-DM in humans)-dependent manner, also suggesting that MCs were able to process and present protein antigens on MHC-II. Antigen uptake by Fc receptors did not enhance antigen processing or presenting ability, since TNP-Eα incorporated into MCs by TNP-specific IgG1 or IgE did not increase YAe staining [53]. However, another study showed that IgE/FcεRI-mediated Escherichia coli uptake protected the bacterial antigens from proteolysis, such that the bacteria remained intact and viable in MCs, and this was proposed to be associated with the particulate nature of the antigens [55]. Together, these studies suggest that while antigens can be loaded onto MHC-II in some contexts, internalization of antigen by MCs via Fc receptor-mediated endocytosis may divert the antigen to compartments that are distinct from antigen uptake pathways in conventional APCs.
Evidence for MC-dependent T cell activation
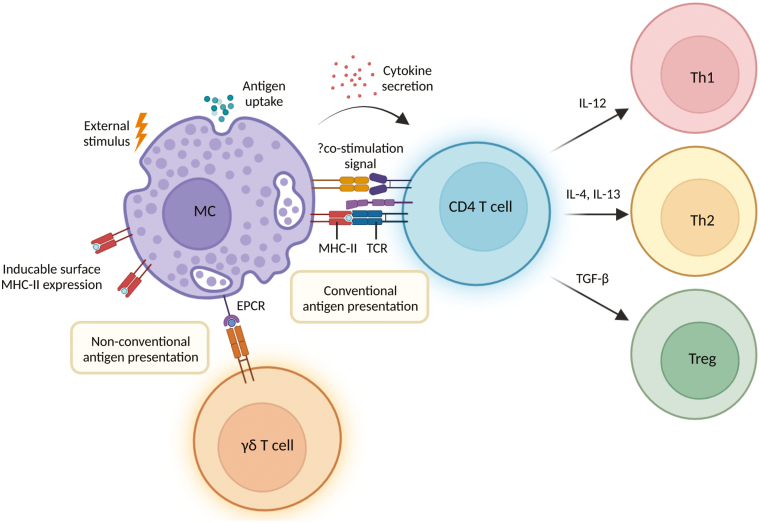
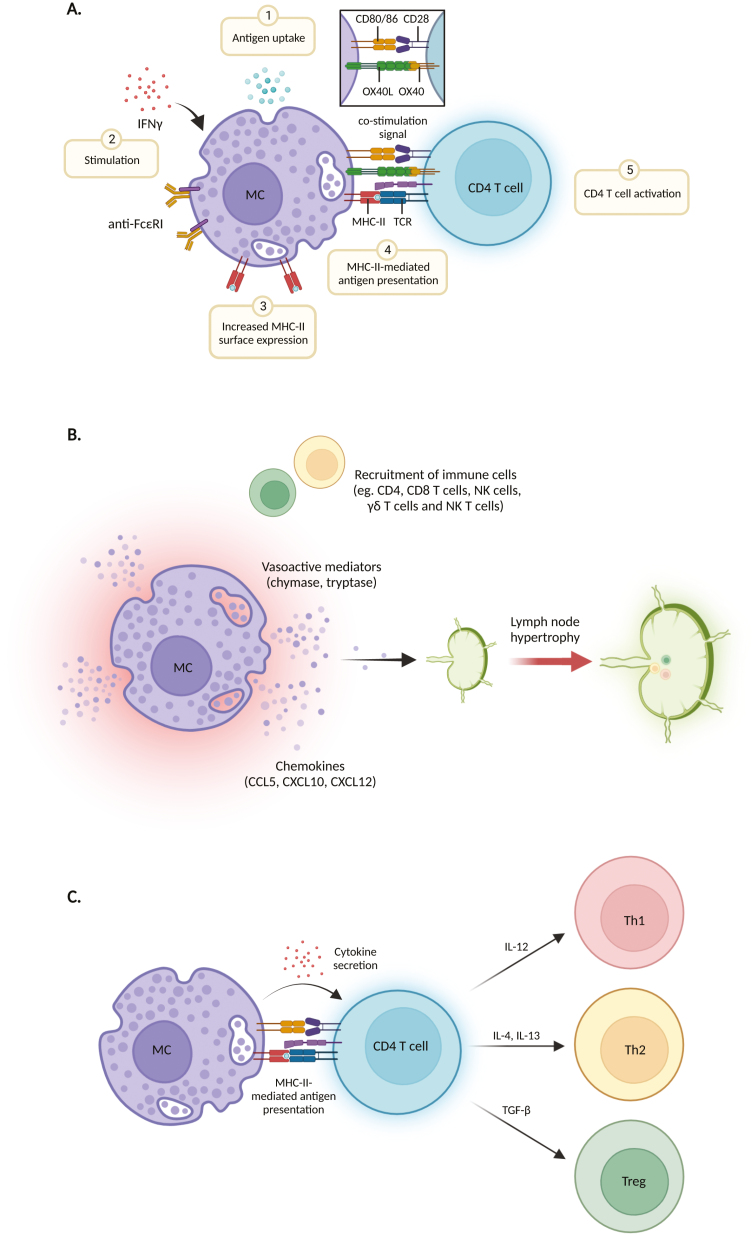
In vitro studies have supported that human and mouse MCs can promote T cell activation in cell culture. Activation of T cells was observed when they were co-cultured with OVA peptide-pulsed BMMCs that had been stimulated by FcɛRI crosslinking [49]. In that experiment, activation of the Notch signalling pathway in BMMCs also promoted a significant increase in T cell activation [49]. Another study co-cultured IgE-pre-sensitized human cord blood-derived MCs isolated from healthy donors with T cells and observed significant activation of CD4 T cells [54]. Moreover, IL-4-treated BMMCs or exosomes derived from them were shown to induce T cell proliferation in vitro [56, 57]. Co-stimulatory signalling provided by APCs through B7-CD28 ligation is generally thought to be crucial for CD4 T cell activation, yet interestingly, it appears that MC-mediated co-stimulation could occur independently of these interactions [58]. Specifically, activation of CD4 T cells via anti-CD3 treatment was enhanced in the presence of human peripheral blood-derived MCs, and no reduction was seen when CD28 was blocked by CTLA4-Ig [58]. These reports suggest that MCs may be able to present antigens to T cells (Fig. 2a) but also that they might have alternate mechanisms for promoting T cell activation or co-stimulation aside from those used by conventional APCs.
Figure 2:
MC-dependent T cell activation and differentiation. (A) MCs can take up and process exogenous antigens. Stimulation of MCs by degranulating stimuli boosts MHC-Il-mediated Ag presentation, where processed antigens can then be loaded on to MHC-lI molecules, presented to CD4 T cells and activate them. Co-stimulatory interactions similar to those used by APCs and some that are yet to be defined also likely contribute to naïve T cell activation. (B) Tissue-resident MCs have been shown to secrete vasoactive mediators and chemokines to promote extravasation and recruitment of immune cells, including T cells to the site of inflammation. MCs also promote LN swelling or hypertrophy, which can promote efficient adaptive immune responses. (C) CD4 T cell polarization can be MC-dependent, and the skewing of different CD4 subsets varies between studies. Diagrams were created with Biorender.com.
Studies that have aimed to confirm MCs can present antigen to T cells in vivo have done so in mice, but, importantly, there are MC-dependent effects on T cells that have been observed in mice that may precede or augment opportunities for antigen presentation by MCs themselves or by conventional APCs. Multiple studies have demonstrated the coordination between MCs and T cells during inflammatory responses. For example, MCs promote CD8 T cell recruitment and activation during Newcastle disease virus and lymphocytic choriomeningitis virus (LCMV) infections, respectively [59, 60], and the recruitment and activation of natural killer (NK) T cells, CD4 T cells, CD8 T cells, and γδT cells were also shown to be MC-dependent during dengue virus infection in vivo in mice [7, 21]. On the other hand, in addition to releasing vasoactive mediators to promote leukocyte extravasation, MCs release chemokines such as CCL5, CXCL10, CXCL12, and CX3CL1 during viral infections, which are known to attract various subsets of T cells [7, 59, 61] (Fig. 2b). During parasitic infections, such as Leishmania major skin infection, recruitment of DCs, macrophages, and neutrophils was shown to be MC-dependent, and the loss of MCs resulted in markedly larger skin lesions and delayed antigen-specific T cell priming [62]. Aside from pro-inflammatory responses, IL-10 production by MCs likely plays a role in the suppression of CD4 T cell responses [63], or contraction of a pro-inflammatory T cell-mediated responses [64], for example, during Schistosoma mansoni or E. coli infections, respectively. These results show that MCs can regulate T cell functions and trafficking in tissues during both the acute and resolution phases of infection/inflammation, in ways independent of antigen presentation (Fig. 2b).
In addition to infections, MCs have been suggested to play a role in solid tumours as well, as MC infiltration was observed in multiple types of cancer and has been correlated with tumour progression [65]. MCs are thought to shape the tumour microenvironment, exemplified by their ability to promote angiogenesis by releasing vascular endothelial growth factor or by their facilitation of metastasis by degrading extracellular matrix with matrix metallopeptidase 9 (MMP-9) [66, 67]. In addition to these functions in tumours that maybe antigen independent, mounting evidence suggests the interaction between MCs and T cells within the tumour microenvironment. Tumour-infiltrating MCs recruited by tumour-secreted stem cell factor, produced cytokines that favoured Treg differentiation in a hepatocarcinoma model [68]. Moreover, in that system, adenosine released by MCs supressed T cell and NK cell proliferation [68]. A recent investigation of the transcriptomic alterations in head and neck squamous cell carcinoma found that MC gene signatures could be used to predict tumour prognosis and that patients rated low-risk by MC gene signature scoring were the most likely to benefit from immune checkpoint inhibitor-related treatments such as anti-PD-1 or CTLA4 [69]. This was further supported by another study where tumour-infiltrating MCs were associated with anti-PD1 resistance in a humanized mouse melanoma model [70]. There, co-localization of MCs and Tregs was also observed post-anti-PD1 treatment [70], suggesting possible interactions between MCs and Tregs. This interaction was shown to be associated with down-regulation of HLA-I expression in tumours and, therefore, it was proposed that the depletion of MCs might improve the efficacy of anti-PD1 therapy [70]. Although these data were supportive of MC/T cell interactions having functional consequences in the tumour microenvironment, this observation was made in a humanized mouse model of tumour progression and it remains unclear if this benefit of MC-depletion pertains to certain types or stages of tumours and if it is consistent in humans. Interestingly, a study targeting gastric cancer saw that intra-tumour MCs express higher levels of PD-L1 and that this is induced by tumour-secreted TNF-α [71]. These findings indicate a potential T cell regulatory role by MCs, possibly dependent on contact and/or utilizing antigen presentation machinery.
Besides the broader functional role of MCs in modulating T cell responses during infection and in the tumour microenvironment, the effect of MCs on antigen-specific T cell responses has also been studied. MC activation by OVA-specific IgE sensitization and crosslinking promotes antigen-specific T cell activation in a system combining 2W1S peptide (variant of chain in I-E molecule) immunization and TLR4 agonist treatment in vivo [72]. This observation comparing the T cell responses in MC-deficient and -sufficient animals implicated MCs in the antigen-specific responses to T cells. MCs were also shown to potentiate CD4 T cell activation, in the absence of antigen presentation [73, 74]. Finally, it was shown that MC-derived nicotinamide adenine dinucleotide, an important co-enzyme in re-dox reactions [75] can promote T cell differentiation and cytokine production in vivo and in vitro in a manner independent of conventional APCs [73, 74].
Potential of conventional antigen presentation by MCs to CD4 T cells
Whether MCs can present antigen to CD4 T cells, and do so to an appreciable extent in vivo, is suggested by many studies but still remains a question that is not fully resolved. For example, splenic MCs expressing MHC-II-loaded OVA peptides failed to activate naïve T cells isolated from mice expressing TCRs specific for OVA (OT-II mice), despite their treatment with lipopolysaccharide (LPS) and IFNγ [53]. The absence of MC-mediated CD4 T cell activation was supported by another study where CMV peptide-pulsed, human skin-derived MCs isolated from CMV seronegative donors failed to activate autologous CD4 T cells ex vivo [42]. However, contradicting results were shown by a group co-culturing human MCs with cells expressing an adenovirus-specific T cell clone. There, they observed elevated cytokine-producing CD4 T cells post-peptide pulsing, which was abolished using HLA-DR blocking antibodies [37]. Similarly, another study showed that BMMCs could stimulate OVA-specific T cells after their treatment with either OVA peptide or IgE-TNP-OVA complexes [43], also supporting the ability of BMMCs to both present peptides and process and present complex antigens. Finally, BMMCs exposed to dengue virus also caused CD4 T cell activation ex vivo in co-culture experiments [21]. These studies suggest that naïve T cells can be activated by MCs, potentially when complex pathogens or degranulating stimuli are provided as antigens rather than cytokines alone.
In contrast to naïve CD4 T cell activation, antigen-specific T cell memory recall by MCs appears to require only cytokines. For example, OVA-pulsed MCs isolated from WT but not MHC-II-deficient mice were able to restimulate previously activated OT-II T cells with the addition of LPS/IFNγ, suggesting that this recall response was dependent on MHC-II-mediated antigen presentation [53]. OVA-pulsed peritoneal cell-derived MCs, primed with IFNγ/IL-4, induced activation of antigen-specific effector T cells but not their naïve counterparts [76]. Similarly, human skin-derived MCs isolated from CMV seropositive patients induced autologous CD4 T cell proliferation ex vivo [42]. Likewise, human MCs cultured from CD133+ peripheral blood precursors were shown to form functional immune synapses with memory CD4 T cells after priming with IFNγ [77]. These consistent findings support the role of MCs as APCs in initiating memory recall responses.
Skewing of T cell responses may also be influenced by MCs. MCs are known to produce a variety of cytokines and chemokines, which have the potential to influence T cell polarization and activation [3]. IL-4 and IL-13, for example, are known to polarize CD4 T cells towards the Th2 subset [78], and TGF-β is understood to promote T cell differentiation into iTreg (induced Treg) cells [79]. Although MCs responses to allergic diseases are primarily Th2-driven [80, 81], MCs can promote the polarization of immune responses towards a Th1 profile during virus exposure, through the production of IL-12 [82], and through memory recall of Th1-polarized CD4 T cells [42]. Contrasting the CMV-specific T cell activation by human MCs that showed Th1 skewing [42], preferential activation of Treg cells was shown after co-culturing influenza peptide-pulsed LPS/IFN-γ-stimulated MCs with T cells isolated from mice expressing endogenous influenza haemagglutinin (HA) protein [53]. These studies indicate that the skewing of T cell subsets by MCs could be model- or antigen-dependent, as well as affected by the exogenous or endogenous expression of antigens. Nonetheless, these studies support that MCs are capable of influencing T cell polarization, either through a contact-dependent or -independent fashion (Fig. 2c).
Cross-presentation of antigens by MCs to CD8 T cells
Cross-presentation, a process defined as the presentation of exogenous antigens to T cells on MHC-I molecules, allows the initiation of CD8 T cell responses without the APCs being infected. Although various types of APCs have been shown to cross-present antigens, DCs are the main cross-presenting APC [36]. One study that investigated whether MC are capable of cross-presenting antigens observed that adoptive transfer of antigen (OVA)-pulsed BMMCs into WT mice induced MHC class I-dependent, antigen-specific primary CD8+ T cell proliferation in vivo [83]. Consistent with this, using an experimental autoimmune encephalomyelitis model, with myelin oligodendrocyte glycoprotein (MOG) peptide-immunized MC-deficient mice, only MOG-specific CD8 T cells showed decreased proliferation compared to WT mice, while no effect was seen in the CD4 T cell subset [83], also supporting the ability of MCs to promote cross-presentation (yet suggesting again that CD4 T cell responses to endogenous antigens are not enhanced by MCs). Despite the need of more supporting studies, these studies provide new insights into the MCs’ functional resemblance to professional APCs.
Non-conventional antigen presentation by MCs
In contrast to conventional antigen presentation that requires peptides binding to the groove of classical MHC proteins and the relatively slower initiation of adaptive immune responses, non-classical antigen presentation involves MHC-like molecules (e.g. CD1, MR1, endothelial protein C receptor (EPCR), HLA-E, G, and F) [84] that present a wider range of antigens, such as lipids, metabolites and modified peptides, and stimulate innate immune-like rapid effector responses [85, 86]. For example, in a dengue virus infection mouse model, activation and proliferation of γδT cells were shown to be MC-dependent, and further investigation revealed that MCs activate γδT cells via direct contact between the non-classical antigen presentation molecule EPCR and γδTCR [21]. MCs can also express CD1d, and mouse primary MCs have been found to promote invariant NKT cell proliferation and CD1d-dependent release of IFN-γ, IL-13, and IL-4. Co-stimulatory molecules including CD48, CD137L, CD252, CD274, and CD275 were shown to promote these activation read-outs in T cells based on monoclonal antibody-blocking experiments [87]. In vivo, in an MC-deficient mouse model of atherosclerosis with and without experimental reconstitution of MCs with CD1d-deficient BMMCs, interaction between MCs and T cells was shown to reduce plaque sizes, while augmenting intraplaque CD4 T cell numbers and cytokine production [88]. HLA-G is a non-classical antigen presentation molecule, primarily associated with inducing tolerance, which has been shown to be expressed on human MCs, and produced by MCs in the livers of patients with liver diseases, including those caused by hepatitis C virus [89, 90]. For these patients, the number of HLA-G positive MCs was correlated with disease progression [89, 90]. Ligands for HLA-G are not only expressed by T cells but also expressed in various immune cells, including NK cells and DCs [91–94], so the effects of HLA-G might not be confined to T cells alone. MCs have also been shown to present superantigens to T cells. For instance, BMMCs presented exogenous superantigen (staphylococcal enterotoxin B) to antigen-specific T cells [52]. A similar phenomenon was observed in humans, as human skin-derived MCs treated with superantigens such as staphylococcal enterotoxin E were able to induce Jurkat (lymphocyte T cell line) cell activation with IFNγ stimulation [42]. This did not occur with toxic shock syndrome toxin from Staphylococcus aureus, possibly suggesting antigen specificity [42]. Together these studies indicate that non-classical antigen presentation by MCs or the use of non-classical antigen presentation molecules may have important effects on T cell-mediated functions from infection clearance to inflammation resolution to tissue remodelling (Table 1).
Table 1:
Non-classical MHC molecules expressed by MCs
| Non-classical MHC molecule | Species where shown | MC-related function | Citation |
|---|---|---|---|
| CD1d | Mouse |
|
[87, 88] |
| HLA-G | Human (liver-resident MCs) |
|
[89, 90] |
| EPCR | Mouse |
|
[21] |
| HLA-DM (H2-DM in mice) | Human (skin-derived MCs) and mouse |
|
[42, 53] |
| MICA, MICB | Human (MC leukemic cell lines) |
|
[95] |
MC partnerships with conventional APCs to promote antigen presentation
Although a field less explored, several independent groups have shown evidence of antigen transferring between MCs and other immune cells. For example, mouse perivascular DCs can take up blood-borne allergens, and after their allergen-carrying micro-vesicles are transferred to MCs, activation and degranulation of MCs occurs, leading to anaphylaxis in vivo [96]. In another study, dermal DCs were shown to transfer MHC-II complexes to surrounding MCs during dinitrofluorobenzene (DNFB)-induced skin inflammation. This DC–MC interaction was thought to contribute to antigen presentation by MCs to T cells ex vivo and T cell-driven skin inflammation in vivo [97]. On the other hand, activation of perivascular MCs through FcɛRI crosslinking has been shown to trigger cell–cell interaction with DCs [98]. This contact facilitates the transport of MC-endocytosed antigens to DCs, which can further be processed and presented to T cells [98]. Another group showed that BMMCs that had taken up OVA (via interactions between anti-OVA IgE and FcɛRI) enhanced CD4 T cell activation, and this was not dependent on MHC-II expression by MCs [99]. Instead, the captured antigens were taken up by DCs after FcɛRI-mediated MC apoptosis, thus promoting T cell activation [99]. These studies suggest that in addition to secreting immune mediators, MCs may also promote antigen presentation of conventional APCs by sampling and transferring antigens.
Conclusion
MCs are present in the lymphoid tissues supporting the idea that they may have an evolutionary conserved role in these tissues. The growing body of literature has established that MCs are consequential for cellular and humoral adaptive immune responses that are evoked after antigen exposure. In addition to their ability to secrete mediators to act on immune cells, MCs are capable of antigen uptake and presentation to B and T cells, which influences the downstream functions of these lymphocytes. However, there are discrepancies in the functions of MCs as APCs and effectors of adaptive immune responses that have emerged from discordant data from mouse models of MC deficiency as well as differences noted between disease models. These may not necessarily be in contradiction but support the idea that MCs responses may be disease- or antigen-specific. Despite these inconsistent results from different studies, there are some consistencies between groups, such as the inducible expression of MHC-II by MCs, their ability to uptake and process exogenous antigens, and their ability to reactivate memory T cells.
There are still major gaps in our understanding of antigen processing by MCs and whether MCs uniquely contribute to antigen processing compared to other conventional APCs. It should also be noted that MCs could utilize non-conventional mechanisms to activate or co-stimulate T cells. Future research should consider these aspects when designing experiments towards addressing the role of MCs as APCs. Nonetheless, the increasing evidence of antigen uptake, processing, and naïve T cell activation, or memory T cell recall by MCs suggests that they are capable of antigen presentation, adding to their well-described roles in allergy and inflammation.
Glossary
Abbreviations
- APCs
antigen-presenting cells
- BMMCs
bone marrow-derived MCs
- BSA
bovine serum albumin
- CLIP
class II associated invariant chain peptide
- CMV
cytomegalovirus
- DCs
dendritic cells
- DNFB
dinitrofluorobenzene
- DQ
dye-quenched
- EAE
experimental autoimmune encephalomyelitis
- EPCR
endothelial protein C receptor
- GAG
glycosaminoglycan
- HA
haemagglutinin
- HLA
human leukocyte antigen
- iTreg
induced Treg
- LCMV
lymphocytic choriomeningitis virus
- LPS
lipopolysaccharide
- MCs
mast cells
- MHC-I
major histocompatibility complex class I
- MHC-II
major histocompatibility complex class II
- MMP-9
matrix metallopeptidase 9
- MOG
myelin oligodendrocyte glycoprotein
- NAD
nicotinamide adenine dinucleotide
- NK
natural killer
- OVA
ovalbumin
- SCF
stem cell factor
- TCR
T cell receptor
- TFH
T follicular helper cells
- TLR
toll-like receptor
- TNF
tumour necrosis factor
- TNP
2,4,6-trinitrophenyl hapten
- Treg
T regulatory cells
- VEGF
vascular endothelial growth factor
- WT
wild-type.
Contributor Information
Chi-Ching Tung, Program in Emerging Infectious Diseases, Duke-National University of Singapore Medical School, Singapore, Singapore.
Abhay P S Rathore, Department of Pathology, Duke University Medical Center, Durham, NC, USA.
Ashley L St. John, Program in Emerging Infectious Diseases, Duke-National University of Singapore Medical School, Singapore, Singapore; Department of Pathology, Duke University Medical Center, Durham, NC, USA; Department of Microbiology and Immunology, Yong Loo Lin School of Medicine, National University of Singapore, Singapore, Singapore; SingHealth Duke-NUS Global Health Institute, Singapore, Singapore.
Funding
The authors acknowledge funding from the Singapore Ministry of Education (T2EP30222-0017).
Conflicts of interest
None declared.
Author contributions
C.C.T., A.L.S., and A.P.S.R. planned the scope of the review. C.C.T. prepared the first draft of the manuscript and designed the figures and tables. All authors contributed to the writing upon revision.
Ethics approval
None declared.
Data availability
None declared.
References
- 1.Abraham SN, St John AL.. Mast cell-orchestrated immunity to pathogens. Nat Rev Immunol 2010, 10, 440–52. doi: 10.1038/nri2782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wernersson S, Pejler G.. Mast cell secretory granules: armed for battle. Nat Rev Immunol 2014, 14, 478–94. doi: 10.1038/nri3690. [DOI] [PubMed] [Google Scholar]
- 3.St John AL, Rathore APS, Ginhoux F.. New perspectives on the origins and heterogeneity of mast cells. Nat Rev Immunol 2023, 23, 55–68. doi: 10.1038/s41577-022-00731-2. [DOI] [PubMed] [Google Scholar]
- 4.Wierzbicki M, Brzezińska-Błaszczyk E.. Diverse effects of bacterial cell wall components on mast cell degranulation, cysteinyl leukotriene generation and migration. Microbiol Immunol 2009, 53, 694–703. doi: 10.1111/j.1348-0421.2009.00174.x. [DOI] [PubMed] [Google Scholar]
- 5.Matsushima H, Yamada N, Matsue H, Shimada S.. TLR3-, TLR7-, and TLR9-mediated production of proinflammatory cytokines and chemokines from murine connective tissue type skin-derived mast cells but not from bone marrow-derived mast cells. J Immunol 2004, 173, 531–41. doi: 10.4049/jimmunol.173.1.531. [DOI] [PubMed] [Google Scholar]
- 6.Qiao H, Andrade MV, Lisboa FA, Morgan K, Beaven MA.. FcepsilonR1 and toll-like receptors mediate synergistic signals to markedly augment production of inflammatory cytokines in murine mast cells. Blood 2006, 107, 610–8. doi: 10.1182/blood-2005-06-2271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.St John AL, Rathore APS, Yap H, Ng M-L, Metcalfe DD, Vasudevan SG, et al. Immune surveillance by mast cells during dengue infection promotes natural killer (NK) and NKT-cell recruitment and viral clearance. Proc Natl Acad Sci USA 2011, 108, 9190–5. doi: 10.1073/pnas.1105079108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rathore AP, St John AL.. Protective and pathogenic roles for mast cells during viral infections. Curr Opin Immunol 2020, 66, 74–81. doi: 10.1016/j.coi.2020.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Woodbury RG, Miller HR, Huntley JF, Newlands GF, Palliser AC, Wakelin D.. Mucosal mast cells are functionally active during spontaneous expulsion of intestinal nematode infections in rat. Nature 1984, 312, 450–2. doi: 10.1038/312450a0. [DOI] [PubMed] [Google Scholar]
- 10.Malaviya R, Gao Z, Thankavel K, van der Merwe PA, Abraham SN.. The mast cell tumor necrosis factor alpha response to FimH-expressing Escherichia coli is mediated by the glycosylphosphatidylinositol-anchored molecule CD48. Proc Natl Acad Sci USA 1999, 96, 8110–5. doi: 10.1073/pnas.96.14.8110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kunder CA, St John AL, Abraham SN.. Mast cell modulation of the vascular and lymphatic endothelium. Blood 2011, 118, 5383–93. doi: 10.1182/blood-2011-07-358432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.McLachlan JB, Shelburne CP, Hart JP, Pizzo SV, Goyal R, Brooking-Dixon R, et al. Mast cell activators: a new class of highly effective vaccine adjuvants. Nat Med 2008, 14, 536–41. doi: 10.1038/nm1757. [DOI] [PubMed] [Google Scholar]
- 13.St John AL, Choi HW, Walker QD, Blough B, Kuhn CM, Abraham SN, et al. Novel mucosal adjuvant, mastoparan-7, improves cocaine vaccine efficacy. NPJ Vaccines 2020, 5. doi: 10.1038/s41541-020-0161-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Johnson-Weaver BT, Choi HW, Yang H, Granek JA, Chan C, Abraham SN, et al. Nasal immunization with small molecule mast cell activators enhance immunity to co-administered subunit immunogens. Front Immunol 2021, 12, 730346. doi: 10.3389/fimmu.2021.730346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ontiveros-Padilla L, Batty CJ, Hendy DA, Pena ES, Roque JA, Stiepel RT, et al. Development of a broadly active influenza intranasal vaccine adjuvanted with self-assembled particles composed of mastoparan-7 and CpG. Front Immunol 2023, 14, 1103765. doi: 10.3389/fimmu.2023.1103765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Schubert N, Lisenko K, Auerbach C, Weitzmann A, Ghouse SM, Muhandes L, et al. Unimpaired responses to vaccination with protein antigen plus adjuvant in mice with kit-independent mast cell deficiency. Front Immunol 2018, 9, 1870. doi: 10.3389/fimmu.2018.01870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Grimbaldeston MA, Chen C-C, Piliponsky AM, Tsai M, Tam S-Y, Galli SJ.. Mast cell-deficient W-sash c-kit mutant Kit W-sh/W-sh mice as a model for investigating mast cell biology in vivo. Am J Pathol 2005, 167, 835–48. doi: 10.1016/S0002-9440(10)62055-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Feyerabend TB, Weiser A, Tietz A, Stassen M, Harris N, Kopf M, et al. Cre-mediated cell ablation contests mast cell contribution in models of antibody- and T cell-mediated autoimmunity. Immunity 2011, 35, 832–44. doi: 10.1016/j.immuni.2011.09.015. [DOI] [PubMed] [Google Scholar]
- 19.Dudeck A, Dudeck J, Scholten J, Petzold A, Surianarayanan S, Köhler A, et al. Mast cells are key promoters of contact allergy that mediate the adjuvant effects of haptens. Immunity 2011, 34, 973–84. doi: 10.1016/j.immuni.2011.03.028. [DOI] [PubMed] [Google Scholar]
- 20.Nakae S, Suto H, Kakurai M, Sedgwick JD, Tsai M, Galli SJ.. Mast cells enhance T cell activation: importance of mast cell-derived TNF. Proc Natl Acad Sci USA 2005, 102, 6467–72. doi: 10.1073/pnas.0501912102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mantri CK, St John AL.. Immune synapses between mast cells and γδ T cells limit viral infection. J Clin Invest 2019, 129, 1094–108. doi: 10.1172/JCI122530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.deLuca LS, Gommerman JL.. Fine-tuning of dendritic cell biology by the TNF superfamily. Nat Rev Immunol 2012, 12, 339–51. doi: 10.1038/nri3193. [DOI] [PubMed] [Google Scholar]
- 23.Jawdat DM, Rowden G, Marshall JS.. Mast cells have a pivotal role in TNF-independent lymph node hypertrophy and the mobilization of Langerhans cells in response to bacterial peptidoglycan. J Immunol 2006, 177, 1755–62. doi: 10.4049/jimmunol.177.3.1755. [DOI] [PubMed] [Google Scholar]
- 24.Kunder CA, St John AL, Li G, Leong KW, Berwin B, Staats HF, et al. Mast cell-derived particles deliver peripheral signals to remote lymph nodes. J Exp Med 2009, 206, 2455–67. doi: 10.1084/jem.20090805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tomita M, Matsuzaki Y, Edagawa M, Shimizu T, Hara M, Onitsuka T.. Distribution of mast cells in mediastinal lymph nodes from lung cancer patients. World J Surg Oncol 2003, 1, 25. doi: 10.1186/1477-7819-1-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Eisenbarth SC. Dendritic cell subsets in T cell programming: location dictates function. Nat Rev Immunol 2019, 19, 89–103. doi: 10.1038/s41577-018-0088-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Roche PA, Furuta K.. The ins and outs of MHC class II-mediated antigen processing and presentation. Nat Rev Immunol 2015, 15, 203–16. doi: 10.1038/nri3818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Plüger EB, Boes M, Alfonso C, Schröter CJ, Kalbacher H, Ploegh HL, et al. Specific role for cathepsin S in the generation of antigenic peptides in vivo. Eur J Immunol 2002, 32, 467–76. doi:. [DOI] [PubMed] [Google Scholar]
- 29.Chapman HA. Endosomal proteases in antigen presentation. Curr Opin Immunol 2006, 18, 78–84. doi: 10.1016/j.coi.2005.11.011. [DOI] [PubMed] [Google Scholar]
- 30.Schulze MS, Wucherpfennig KW.. The mechanism of HLA-DM induced peptide exchange in the MHC class II antigen presentation pathway. Curr Opin Immunol 2012, 24, 105–11. doi: 10.1016/j.coi.2011.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Welsh RA, Sadegh-Nasseri S.. The love and hate relationship of HLA-DM/DO in the selection of immunodominant epitopes. Curr Opin Immunol 2020, 64, 117–23. doi: 10.1016/j.coi.2020.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Swain SL, McKinstry KK, Strutt TM.. Expanding roles for CD4+ T cells in immunity to viruses. Nat Rev Immunol 2012, 12, 136–48. doi: 10.1038/nri3152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schoenberger SP, Toes RE, van der Voort EI, Offringa R, Melief CJ.. T-cell help for cytotoxic T lymphocytes is mediated by CD40-CD40L interactions. Nature 1998, 393, 480–3. doi: 10.1038/31002. [DOI] [PubMed] [Google Scholar]
- 34.Bennett SR, Carbone FR, Karamalis F, Miller JF, Heath WR.. Induction of a CD8+ cytotoxic T lymphocyte response by cross-priming requires cognate CD4+ T cell help. J Exp Med 1997, 186, 65–70. doi: 10.1084/jem.186.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Crotty S. Follicular helper CD4 T cells (TFH). Annu Rev Immunol 2011, 29, 621–63. doi: 10.1146/annurev-immunol-031210-101400. [DOI] [PubMed] [Google Scholar]
- 36.Joffre OP, Segura E, Savina A, Amigorena S.. Cross-presentation by dendritic cells. Nat Rev Immunol 2012, 12, 557–69. doi: 10.1038/nri3254. [DOI] [PubMed] [Google Scholar]
- 37.Suurmond J, van Heemst J, van Heiningen J, Dorjée AL, Schilham MW, van der Beek FB, et al. Communication between human mast cells and CD4(+) T cells through antigen-dependent interactions. Eur J Immunol 2013, 43, 1758–68. doi: 10.1002/eji.201243058. [DOI] [PubMed] [Google Scholar]
- 38.Masopust D, Choo D, Vezys V, Wherry EJ, Duraiswamy J, Akondy R, et al. Dynamic T cell migration program provides resident memory within intestinal epithelium. J Exp Med 2010, 207, 553–64. doi: 10.1084/jem.20090858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Le Bourhis L, Martin E, Péguillet I, Guihot A, Froux N, Coré M, et al. Antimicrobial activity of mucosal-associated invariant T cells. Nat Immunol 2010, 11, 701–8. doi: 10.1038/ni.1890. [DOI] [PubMed] [Google Scholar]
- 40.Treiner E, Duban L, Bahram S, Radosavljevic M, Wanner V, Tilloy F, et al. Selection of evolutionarily conserved mucosal-associated invariant T cells by MR1. Nature 2003, 422, 164–9. doi: 10.1038/nature01433. [DOI] [PubMed] [Google Scholar]
- 41.Iwasaki A, Medzhitov R.. Control of adaptive immunity by the innate immune system. Nat Immunol 2015, 16, 343–53. doi: 10.1038/ni.3123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lotfi-Emran S, Ward BR, Le QT, Pozez AL, Manjili MH, Woodfolk JA, et al. Human mast cells present antigen to autologous CD4(+) T cells. J Allergy Clin Immunol 2018, 141, 311–321.e10. doi: 10.1016/j.jaci.2017.02.048. [DOI] [PubMed] [Google Scholar]
- 43.Gong J, Yang N-S, Croft M, Weng I-C, Sun L, Liu F-T, et al. The antigen presentation function of bone marrow-derived mast cells is spatiotemporally restricted to a subset expressing high levels of cell surface FcepsilonRI and MHC II. BMC Immunol 2010, 11, 34. doi: 10.1186/1471-2172-11-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Raposo G, Tenza D, Mecheri S, Peronet R, Bonnerot C, Desaymard C.. Accumulation of major histocompatibility complex class II molecules in mast cell secretory granules and their release upon degranulation. Mol Biol Cell 1997, 8, 2631–45. doi: 10.1091/mbc.8.12.2631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hershman MJ, Appel SH, Wellhausen SR, Sonnenfeld G, Polk HC Jr. Interferon-gamma treatment increases HLA-DR expression on monocytes in severely injured patients. Clin Exp Immunol 1989, 77, 67–70. [PMC free article] [PubMed] [Google Scholar]
- 46.Pan J, Zhang M, Wang J, Wang Q, Xia D, Sun W, et al. Interferon-gamma is an autocrine mediator for dendritic cell maturation. Immunol Lett 2004, 94, 141–51. doi: 10.1016/j.imlet.2004.05.003. [DOI] [PubMed] [Google Scholar]
- 47.Hou B, Reizis B, DeFranco AL.. Toll-like receptors activate innate and adaptive immunity by using dendritic cell-intrinsic and -extrinsic mechanisms. Immunity 2008, 29, 272–82. doi: 10.1016/j.immuni.2008.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kastenmüller K, Wille-Reece U, Lindsay RWB, Trager LR, Darrah PA, Flynn BJ, et al. Protective T cell immunity in mice following protein-TLR7/8 agonist-conjugate immunization requires aggregation, type I IFN, and multiple DC subsets. J Clin Invest 2011, 121, 1782–96. doi: 10.1172/JCI45416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Nakano N, Nishiyama C, Yagita H, Koyanagi A, Akiba H, Chiba S, et al. Notch signaling confers antigen-presenting cell functions on mast cells. J Allergy Clin Immunol 2009, 123, 74–81.e1. doi: 10.1016/j.jaci.2008.10.040. [DOI] [PubMed] [Google Scholar]
- 50.Lipski S, Grabbe J, Henz BM.. Absence of MHC class II antigen on mast cells at sites of inflammation in human skin. Exp Dermatol 1996, 5, 120–4. doi: 10.1111/j.1600-0625.1996.tb00105.x. [DOI] [PubMed] [Google Scholar]
- 51.Kritikou E, van der Heijden T, Swart M, van Duijn J, Slütter B, Wezel A, et al. Hypercholesterolemia induces a mast cell–CD4+ T cell interaction in atherosclerosis. J Immunol 2019, 202, 1531–9. doi: 10.4049/jimmunol.1800648. [DOI] [PubMed] [Google Scholar]
- 52.Frandji P, Tkaczyk C, Oskeritzian C, David B, Desaymard C, Mécheri S.. Exogenous and endogenous antigens are differentially presented by mast cells to CD4+ T lymphocytes. Eur J Immunol 1996, 26, 2517–28. doi: 10.1002/eji.1830261036. [DOI] [PubMed] [Google Scholar]
- 53.Kambayashi T, Allenspach EJ, Chang JT, Zou T, Shoag JE, Reiner SL, et al. Inducible MHC class II expression by mast cells supports effector and regulatory T cell activation. J Immunol 2009, 182, 4686–95. doi: 10.4049/jimmunol.0803180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Fujita T, Kambe N, Uchiyama T, Hori T.. Type I interferons attenuate T cell activating functions of human mast cells by decreasing TNF-alpha production and OX40 ligand expression while increasing IL-10 production. J Clin Immunol 2006, 26, 512–8. doi: 10.1007/s10875-006-9043-1. [DOI] [PubMed] [Google Scholar]
- 55.Shin JS, Shelburne CP, Jin C, LeFurgey EA, Abraham SN.. Harboring of particulate allergens within secretory compartments by mast cells following IgE/FcepsilonRI-lipid raft-mediated phagocytosis. J Immunol 2006, 177, 5791–800. doi: 10.4049/jimmunol.177.9.5791. [DOI] [PubMed] [Google Scholar]
- 56.Skokos D, Botros HG, Demeure C, Morin J, Peronet R, Birkenmeier G, et al. Mast cell-derived exosomes induce phenotypic and functional maturation of dendritic cells and elicit specific immune responses in vivo. J Immunol 2003, 170, 3037–45. doi: 10.4049/jimmunol.170.6.3037. [DOI] [PubMed] [Google Scholar]
- 57.Skokos D, Le Panse S, Villa I, Rousselle JC, Peronet R, David B, et al. Mast cell-dependent B and T lymphocyte activation is mediated by the secretion of immunologically active exosomes. J Immunol 2001, 166, 868–76. doi: 10.4049/jimmunol.166.2.868. [DOI] [PubMed] [Google Scholar]
- 58.Suurmond J, Dorjée AL, Huizinga TW, Toes RE.. Human mast cells costimulate T cells through a CD28-independent interaction. Eur J Immunol 2016, 46, 1132–41. doi: 10.1002/eji.201545914. [DOI] [PubMed] [Google Scholar]
- 59.Orinska Z, Bulanova E, Budagian V, Metz M, Maurer M, Bulfone-Paus S.. TLR3-induced activation of mast cells modulates CD8+ T-cell recruitment. Blood 2005, 106, 978–87. doi: 10.1182/blood-2004-07-2656. [DOI] [PubMed] [Google Scholar]
- 60.Hackler Y, Siebenhaar F, Löhning M, Maurer M, Muñoz M.. Mast cells modulate antigen-specific CD8(+) T cell activation during LCMV infection. Front Immunol 2021, 12, 688347. doi: 10.3389/fimmu.2021.688347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ebert S, Becker M, Lemmermann NAW, Büttner JK, Michel A, Taube C, et al. Mast cells expedite control of pulmonary murine cytomegalovirus infection by enhancing the recruitment of protective CD8 T cells to the lungs. PLoS Pathog 2014, 10, e1004100. doi: 10.1371/journal.ppat.1004100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Maurer M, Kostka SL, Siebenhaar F, Moelle K, Metz M, Knop J, et al. Skin mast cells control T cell-dependent host defense in Leishmania major infections. FASEB J 2006, 20, 2460–7. doi: 10.1096/fj.06-5860com. [DOI] [PubMed] [Google Scholar]
- 63.Prendergast CT, Sanin DE, Mountford AP.. CD4 T-cell hyporesponsiveness induced by schistosome larvae is not dependent upon eosinophils but may involve connective tissue mast cells. Parasite Immunol 2016, 38, 81–92. doi: 10.1111/pim.12300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Chan CY, St John AL, Abraham SN.. Mast cell interleukin-10 drives localized tolerance in chronic bladder infection. Immunity 2013, 38, 349–59. doi: 10.1016/j.immuni.2012.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Khazaie K, Blatner NR, Khan MW, Gounari F, Gounaris E, Dennis K, et al. The significant role of mast cells in cancer. Cancer Metastasis Rev 2011, 30, 45–60. doi: 10.1007/s10555-011-9286-z. [DOI] [PubMed] [Google Scholar]
- 66.Ribatti D, Crivellato E.. Mast cells, angiogenesis, and tumour growth. Biochim Biophys Acta 2012, 1822, 2–8. doi: 10.1016/j.bbadis.2010.11.010. [DOI] [PubMed] [Google Scholar]
- 67.Hempel HA, Cuka NS, Kulac I, Barber JR, Cornish TC, Platz EA, et al. Low intratumoral mast cells are associated with a higher risk of prostate cancer recurrence. Prostate 2017, 77, 412–24. doi: 10.1002/pros.23280. [DOI] [PubMed] [Google Scholar]
- 68.Huang B, Lei Z, Zhang G-M, Li D, Song C, Li B, et al. SCF-mediated mast cell infiltration and activation exacerbate the inflammation and immunosuppression in tumor microenvironment. Blood 2008, 112, 1269–79. doi: 10.1182/blood-2008-03-147033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Cai Z, Tang B, Chen L, Lei W.. Mast cell marker gene signature in head and neck squamous cell carcinoma. BMC Cancer 2022, 22, 1–577. doi: 10.1186/s12885-022-09673-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Somasundaram R, Connelly T, Choi R, Choi H, Samarkina A, Li L, et al. Tumor-infiltrating mast cells are associated with resistance to anti-PD-1 therapy. Nat Commun 2021, 12, 346. doi: 10.1038/s41467-020-20600-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lv Y, Zhao Y, Wang X, Chen N, Mao F, Teng Y, et al. Increased intratumoral mast cells foster immune suppression and gastric cancer progression through TNF-α-PD-L1 pathway. J ImmunoTher Cancer 2019, 7, 1–54. doi: 10.1186/s40425-019-0530-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Phong BL, D'Souza SJ, Baudier RL, Wu E, Immethun VE, Bauer DL, et al. IgE-activated mast cells enhance TLR4-mediated antigen-specific CD4(+) T cell responses. Sci Rep 2021, 11, 9686. doi: 10.1038/s41598-021-88956-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Tullius SG, Biefer HRC, Li S, Trachtenberg AJ, Edtinger K, Quante M, et al. NAD+ protects against EAE by regulating CD4+ T-cell differentiation. Nat Commun 2014, 5, 5101. doi: 10.1038/ncomms6101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Rodriguez Cetina Biefer H, Heinbokel T, Uehara H, Camacho V, Minami K, Nian Y, et al. Mast cells regulate CD4(+) T-cell differentiation in the absence of antigen presentation. J Allergy Clin Immunol 2018, 142, 1894–1908.e7. doi: 10.1016/j.jaci.2018.01.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Li W, Sauve AA.. NAD+ content and its role in mitochondria. Methods Mol Biol 2015, 1241, 39–48. doi: 10.1007/978-1-4939-1875-1_4. [DOI] [PubMed] [Google Scholar]
- 76.Gaudenzio N, Espagnolle N, Mars LT, Liblau R, Valitutti S, Espinosa E.. Cell-cell cooperation at the T helper cell/mast cell immunological synapse. Blood 2009, 114, 4979–88. doi: 10.1182/blood-2009-02-202648. [DOI] [PubMed] [Google Scholar]
- 77.Gaudenzio N, Laurent C, Valitutti S, Espinosa E.. Human mast cells drive memory CD4+ T cells toward an inflammatory IL-22+ phenotype. J Allergy Clin Immunol 2013, 131, 1400–7.e11. doi: 10.1016/j.jaci.2013.01.029. [DOI] [PubMed] [Google Scholar]
- 78.Walker JA, McKenzie ANJ.. T(H)2 cell development and function. Nat Rev Immunol 2018, 18, 121–33. doi: 10.1038/nri.2017.118. [DOI] [PubMed] [Google Scholar]
- 79.Li MO, Flavell RA.. TGF-beta: a master of all T cell trades. Cell 2008, 134, 392–404. doi: 10.1016/j.cell.2008.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Amin K. The role of mast cells in allergic inflammation. Respir Med 2012, 106, 9–14. doi: 10.1016/j.rmed.2011.09.007. [DOI] [PubMed] [Google Scholar]
- 81.Msallam R, Balla J, Rathore APS, Kared H, Malleret B, Saron WAA, et al. Fetal mast cells mediate postnatal allergic responses dependent on maternal IgE. Science 2020, 370, 941–50. doi: 10.1126/science.aba0864. [DOI] [PubMed] [Google Scholar]
- 82.Syenina A, Saron WAA, Jagaraj CJ, Bibi S, Arock M, Gubler DJet al. Th1-polarized, dengue virus-activated human mast cells induce endothelial transcriptional activation and permeability. Viruses 2020, 12, 1379. doi: 10.3390/v12121379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Stelekati E, Bahri R, D'Orlando O, Orinska Z, Mittrücker H-W, Langenhaun R, et al. Mast cell-mediated antigen presentation regulates CD8+ T cell effector functions. Immunity 2009, 31, 665–76. doi: 10.1016/j.immuni.2009.08.022. [DOI] [PubMed] [Google Scholar]
- 84.Halenius A, Gerke C, Hengel H.. Classical and non-classical MHC I molecule manipulation by human cytomegalovirus: so many targets—but how many arrows in the quiver? Cell Mol Immunol 2015, 12, 139–53. doi: 10.1038/cmi.2014.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.D’Souza MP, Adams E, Altman JD, Birnbaum ME, Boggiano C, Casorati G, et al. Casting a wider net: Immunosurveillance by nonclassical MHC molecules. PLoS Pathog 2019, 15, e1007567. doi: 10.1371/journal.ppat.1007567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hansen TH, Huang S, Arnold PL, Fremont DH.. Patterns of nonclassical MHC antigen presentation. Nat Immunol 2007, 8, 563–8. doi: 10.1038/ni1475. [DOI] [PubMed] [Google Scholar]
- 87.Roldan NG, Orinska Z, Ewers H, Bulfone-Paus S. CD252 regulates mast cell mediated, CD1d-restricted NKT-cell activation in mice. Eur J Immunol 2016, 46, 432–9. doi: 10.1002/eji.201545879. [DOI] [PubMed] [Google Scholar]
- 88.Kritikou E, van Duijn J, Nahon JE, van der Heijden T, Bouwman M, Groeneveldt C, et al. Disruption of a CD1d-mediated interaction between mast cells and NKT cells aggravates atherosclerosis. Atherosclerosis 2019, 280, 132–9. doi: 10.1016/j.atherosclerosis.2018.11.027. [DOI] [PubMed] [Google Scholar]
- 89.Amiot L, Vu N, Rauch M, L'Helgoualc'h A, Chalmel F, Gascan H, et al. Expression of HLA-G by mast cells is associated with hepatitis C virus-induced liver fibrosis. J Hepatol 2014, 60, 245–52. doi: 10.1016/j.jhep.2013.09.006. [DOI] [PubMed] [Google Scholar]
- 90.Amiot L, Vu N, Samson M.. Biology of the immunomodulatory molecule HLA-G in human liver diseases. J Hepatol 2015, 62, 1430–7. doi: 10.1016/j.jhep.2015.03.007. [DOI] [PubMed] [Google Scholar]
- 91.Loustau M, Anna F, Dréan R, Lecomte M, Langlade-Demoyen P, Caumartin J.. HLA-G neo-expression on tumors. Front Immunol 2020, 11, 1685. doi: 10.3389/fimmu.2020.01685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Gros F, Cabillic F, Toutirais O, Le Maux A, Sebti Y, Amiot L.. Soluble HLA-G molecules impair natural killer/dendritic cell crosstalk via inhibition of dendritic cells. Eur J Immunol 2008, 38, 742–9. doi: 10.1002/eji.200736918. [DOI] [PubMed] [Google Scholar]
- 93.Rouas-Freiss N, Marchal RE, Kirszenbaum M, Dausset J, Carosella ED.. The alpha1 domain of HLA-G1 and HLA-G2 inhibits cytotoxicity induced by natural killer cells: is HLA-G the public ligand for natural killer cell inhibitory receptors? Proc Natl Acad Sci USA 1997, 94, 5249–54. doi: 10.1073/pnas.94.10.5249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Le Gal FA, Riteau B, Sedlik C, Khalil-Daher I, Menier C, Dausset J, et al. HLA-G-mediated inhibition of antigen-specific cytotoxic T lymphocytes. Int Immunol 1999, 11, 1351–6. doi: 10.1093/intimm/11.8.1351. [DOI] [PubMed] [Google Scholar]
- 95.Aponte-López A, Enciso J, Muñoz-Cruz S, Fuentes-Pananá EM.. An in vitro model of mast cell recruitment and activation by breast cancer cells supports anti-tumoral responses. Int J Mol Sci 2020, 21, 5293. doi: 10.3390/ijms21155293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Choi HW, Suwanpradid J, Kim IH, Staats HF, Haniffa M, MacLeod AS, et al. Perivascular dendritic cells elicit anaphylaxis by relaying allergens to mast cells via microvesicles. Science 2018, 362, eaao0666. doi: 10.1126/science.aao0666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Dudeck J, Medyukhina A, Fröbel J, Svensson C-M, Kotrba J, Gerlach M, et al. Mast cells acquire MHCII from dendritic cells during skin inflammation. J Exp Med 2017, 214, 3791–811. doi: 10.1084/jem.20160783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Carroll-Portillo A, Cannon JL, te Riet J, Holmes A, Kawakami Y, Kawakami T, et al. Mast cells and dendritic cells form synapses that facilitate antigen transfer for T cell activation. J Cell Biol 2015, 210, 851–64. doi: 10.1083/jcb.201412074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Kambayashi T, Baranski JD, Baker RG, Zou T, Allenspach EJ, Shoag JE, et al. Indirect involvement of allergen-captured mast cells in antigen presentation. Blood 2008, 111, 1489–96. doi: 10.1182/blood-2007-07-102111. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
None declared.