Abstract
Aim: Estimating the risk of developing ischemic stroke (IS) may assist health professionals in motivating individuals to modify their risk behavior.
Methods: A predictive model was derived from 178,186 participants from Fukushima Health Database, aged 40-74 years, who attended the health checkup in 2014 and completed at least one annual health checkup by 2018 (Cohort I). Cox proportional hazard regression model was used to build a 4-year prediction model, thus the risk scores were based on the regression coefficients. External validation for the risk scores was conducted in another cohort of 46,099 participants following between 2015 and 2019 (Cohort II).
Results: The 4-year cumulated incidence rate of IS was 179.80/100,000 person-years in Cohort I. The predictive model included age, sex, blood pressure, hypertension treatment, diabetes, low- and high-density lipoprotein cholesterol, smoking, walking pace, and body weight change of 3 kg within one year. Risk scores were interpreted based on the Cohort I predictive model function. The Harrell’sC-statistics of the discrimination ability of the risk score model (95% confidence interval) was 0.744 (0.729-0.759) in Cohort I and 0.770 (0.743-0.797) in Cohort II. The overall agreement of the risk score probability of IS incidence for the observed/expected case ratio and 95% CI was 0.98 (0.92-1.05) in Cohort I and 1.08 (0.95-1.22) in Cohort II.
Conclusions: The 4-year risk prediction model revealed a good performance for IS incidence, and risk scores could be used to estimate individual incidence risk of IS. Updated models with additional confirmed risk variables may be needed.
Keywords: Ischemic stroke, Incidence risk, Prediction model, External validation, Point-based score
Introduction
Given the high incidence of cardiovascular diseases (CVD) and high cost to society, reducing cardiovascular risk and preventing CVD occurrence in a population is crucial. Controlling and modifying lifestyle risks are effective in maintaining good health. The Great East Japan Earthquake in March 2011 affected the health status of residents in the disaster areas, and increased cardiometabolic risks of overweight/obesity, hypertension, diabetes mellitus, and dyslipidemia have been reported 1 - 6) . The prevalence of hypertension peaked one year after the disaster and showed a declining tendency 3) ; bodyweight/waist circumference increased with declining high-density lipoprotein cholesterol (HDL-C) level among relocated survivors, more than one year postdisaster 1 , 2 , 5) .
In epidemiologic studies in Japanese populations, cardiometabolic risk factors are consistently identified, and improvements in them have been incorporated into the primary CVD prevention strategies 7) . By synthesizing established factors into a single model, an individual risk assessment tool can be developed to serve as an effective approach to disseminating information on CVD prevention in the communities 7 , 8) .
The Framingham Heart Study initiated the coronary heart disease prediction model 9) . The Japanese Suita prediction scores were modified and developed in 2015 from the Framingham scores 10) . Fewer studies have been conducted on the absolute risk prediction of stroke onset than for heart disease. The most prominent studies on stroke incidence risk prediction in Japan were the JMS Cohort Study 11) , the NIPPON DATA80 12) , the JPHC Study 13 , 14) , the Suita Study 15 , 16) , and the Hisayama Study 17 - 19) , in which the Hisayama Study and the Suita Study had fewer participants but longer follow-up years relevantly; and the JMS Cohort Study and JPHC Study were used to derive the prediction model of ischemic stroke (IS) incidence risk. Recently, a study on stroke onset prediction in Chinese with two years of follow-up has been reported; however, the self-reported diagnosis of stroke failed to distinguish intracranial hemorrhage or IS 20) .
Japan initiated universal health insurance in the 1960s. Since 2008, the Japanese Ministry of Health, Labor, and Welfare has launched annual health screening and health promotion services, the “Specific Health Checkups” and “Specific Health Guidance,” as part of the national health insurance system 21) . The Fukushima prefectural health authority created the Fukushima Health Database (FDB) in 2017, which consists of individual health insurance data of Fukushima residents that allows for their health status monitoring and conducting of research.
Aim
Japanese people suffer a high burden of stroke, and IS is the primary type of stroke, while Fukushima Prefecture ranks sixth worst prefecture in mortality rates 22) . Studies assessing incidence disparity in different areas may provide novel information for facilitating community stroke control and prevention. However, no previous IS prediction studies exists that have used insurance data in Japan; such studies could enrich the community- and clinical-based research on stroke risk prediction. This study presents a 4-year predictive model for IS incidence risk, with a simplified score sheet derived and validated based on the FDB, to estimate the absolute individual risk of IS from the obtained lifestyle information.
Methods
Study Participants
In Japan, the universal health insurance coverage mainly includes three types of public health insurance: 1) employee health insurance provided by many companies to their employees and their dependents; 2) For people not covered by the employee health insurance (including the self-employed and those working in agriculture, forestry, fishery, or small businesses), the municipalities provide national health insurance for persons aged ≤ 74 years; and 3) the late elders’ insurance covers persons aged ≥ 75 18) .
The FDB includes the annual specific health checkups and national health insurance, late elders’ insurance, and employee health insurance claims (mainly for employees of small to medium-sized enterprises), which covers about 70% of residents of Fukushima Prefecture 23) . In this study, we used the national health insurance and employee health insurance data from the FDB. This study has been approved by the Institute’s Ethical Review Committee, at Fukushima Medical University (Generic 2021-169). Since this study used only information that does not identify specific individuals, the procedure for obtaining informed consent was waived.
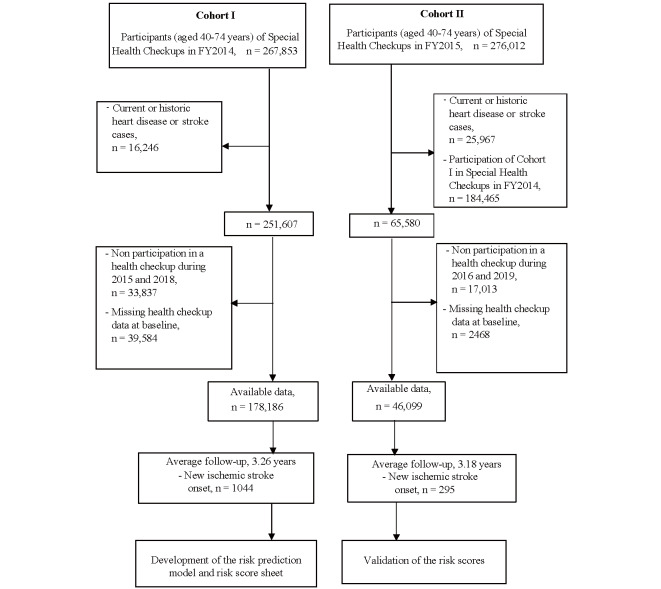
This study included participants without a history of ischemic heart disease or stroke reported during the health checkups in the fiscal year (FY) 2014 (between April 1, 2014, and March 31, 2015) were followed up till the end of March 31, 2018. Among 251,607 participants aged 40-74 years in 2014, we excluded those without health checkup data in subsequent years, leaving 178,186 participants for inclusion in the cohort for building the risk model of IS risk (Cohort I). In another cohort, 46,099 participants that underwent health checkups in 2015 at baseline, who did not attend the health checkups in 2014, but were followed up until the end of March 2019, were used to validate the prediction model ( Fig.1 ) .
Fig.1.
Data flow for the predictive models’ derivation and validation in the two cohorts, respectively, using the Fukushima Health Database data, 2014 -2019
Risk Factor Measurements
Demographic data (age, sex, and residential area) and risk factors (e.g., blood pressure [BP], body mass index [BMI]) were extracted from the annual health checkup records. The health checkups include a self-administered questionnaire with 22 items on medical history, smoking, and alcohol drinking status, physical activity, walking speed, body weight change of more than 3 kg in the past year, and dietary habits. In addition, serum specimens were collected during the health checkups to examine glucose, liver function, and cholesterol density.
Follow-Up and Case Ascertainment
Information on disease occurrence, including medical, diagnostic procedure combination (DPC), and dispensing receipts, was identified from health claims in the database 24) . About 80% of hospitals in Fukushima joined the DPC system. To identify IS occurrence, we considered in-hospital receipts confirmed based on the Inter-national Classification of Diseases 10th Revision (ICD-10 code) I630-I639 in combination with any prescriptions of pharmaceutical classification 212 (arrhythmia), 213 (diuretic), 214 (antihypertensive), 217 (vasodilator), 333 (blood coagulation inhibitor), and 339 (other blood and body fluid drugs), as IS 24 , 25) .
Statistical Analysis
We checked the normality distribution of clinical measurements. Variables that were normally distributed were expressed as the mean and standard deviation (SD), whereas those that were not normally distributed were expressed as the median and interquartile range (IQR) and were further categorized by cut-off values. We tested the differences between the risk proportions using the Chi-square test and the differences in clinical measurements using parametric (F test) and nonparametric (Kruskal–Wallis test) analyses of variance across risk groups.
Person-years of follow-up were determined from the date of the baseline health checkup until the date of the first IS occurrence or the last health checkup date in participants without any stroke incidence.
The absolute 4-year risk of stroke was predicted with a Cox hazard regression model derived from Cohort I, where P=1−S(t)exp(f[x,M]) and f[x,M]=β1(x1−M1)+β2(x2−M2)+...+βj ( xj − Mj ). β1...βj are the regression coefficients, x1... xj represent individual risk factors, M1... Mj are the mean values of the risk factors in the datasets (the proportion of each category), and S(t) is the survival rate at the mean values of the risk factors at time t (t=4-years in this study). Based on the age and sex-adjusted regression analysis for potential risk factors available in the health checkups (categorized variables), including clinical measurements (BMI, BP, glucose, HDL-C and low-density lipoprotein cholesterol [LDL-C], and triglycerides) and Specific Health Checkup questionnaires (on smoking status, alcohol drinking frequency, physical exercise, walking speed, sleeping quality, body weight change, current diabetes mellitus, treatment of hypertension, diabetes mellitus, and dyslipidemia), all significant variables were pooled into a single multivariable analysis, then a backward selection of variables was performed, leaving the significant variables in the final prediction model (at P<0.05 to be retained in the model). The proportional hazards assumption of the variables were confirmed. Internal validations with 20 bootstrapping were conducted using Cohort I, to examine the fitness of models and shrinkages of the coefficients for candidate variables 26) . When we added an interaction term between two variables (i.e., sex and each covariate, age group and BP, BP and diabetes, smoke and alcohol drinking, and BMI and BP, respectively), none revealed a significant contribution to the main effect model.
The final prediction model included age, sex, BP levels with/(without antihypertensive medication), smoking status, current diabetes, HDL-C, LDL-C, walking pace, and body weight change of 3 kg within one year. Age was categorized into 5-yearly intervals (40-44, 45-49, 50-54, 55-59, 60-64, 65-70, and 70-74 years). Based on the adult BP guideline 27) , we classified blood pressure into normal (systolic BP [SBP] <120 mmHg and diastolic BP [DBP] <80 mmHg [without antihypertensive medication]); high-normal (SBP <130 mmHg and DBP <80 mmHg [without antihypertensive medication]); elevated (SBP <140 mmHg or DBP <90 mmHg [without antihypertensive medication]); and hypertension (SBP ≥ 140 mmHg or DBP ≥ 90 mmHg [or using antihypertensive medication]) HDL-C was grouped into three levels (<40, 40-59, and ≥ 60 mg/dL), and LDL-C was grouped into three levels (<130, 130-159, and ≥ 160 mg/dl). Smoking habits, diabetes, antihypertensive medication, and bodyweight change of 3 kg within one year were categorized into two levels (yes or no). Walking pace was categorized as fast or normal/slow.
A simple-point risk score sheet for IS was developed based on the β coefficients of the prediction model, wherein points were assigned to each risk factor category by rounding the quotients of its β coefficient divided by the lowest coefficient among all variables categories 28) . The risk score of each participant (range -4 to 35) was the sum of the points of each available risk factor.
We used Harrell’s C-statistics and the 95% confidence interval (95% CI) to assess the discrimination ability of the predictive model, to separate those who experienced an event from those who did not 29) .
The calibration of the models’ performance was plotted using the observed and predicted probability of IS events 30) . Participants were divided into ten deciles of individual risk, and in each decile, the expected number of events was the sum of individual predicted risk 30) . The ratio of observed and expected (O/E) events was applied to examine the overall difference between the observed and estimated risk groups in deciles 31 - 33) . The 95% CI for the O/E ratio was calculated as ×exp . The prediction model underestimates the outcome risk if the O/E ratio is greater than 1, while it overestimates if the O/E ratio is less than 1.
All data analysis was conducted using SAS version 9.4 (SAS Institute, Cary NC).
Results
The median (IQR) number of years of follow-up and the number of newly diagnosed IS cases in Cohort I were 3.9 (2.2, 4.0) and 1044 (0.058%), respectively, while in Cohort II, these were 3.9 (2.1, 4.0) and 295 (0.064%), respectively. The observed 4-year accumulated incidence rate of IS was 270.30 in men and 127.36 in women per 100,000 person-year in Cohort I, and 254.31 in men and 108.57 in women per 100,000 person-year in Cohort II standardized by the age groups in Cohort I.
The characteristics of the Cohort I participants are shown in Table 1 . The participants’ mean (SD) age was 59.9 (9.6) years, and 48.7% were men. Overall, 29% of the participants were overweight, 22% were smokers, and 28.6% drank alcohol daily. Ten percent of the participants had diabetes, and 34.9% had hypertension or were undergoing antihypertensive therapy, with 12.4% having high LDL-C, 5.2% having low HDL-C, and 20.1% having high triglycerides. In addition, the proportions of participants who were physically active, walked fast, and had weight change of approximately 3 kg were 30%, 44.1%, and 22.1%, respectively. As age increased, DBP and proportions of participants with overweight, high triglyceride, current smoking, or body weight changes tended to decline. However, with SBP and hypertension, diabetes, using antihyperglycemic medication, and walking in fast pace, the proportions tended to increase.
Table 1. Baseline characteristics of participants by age group (years) in Cohort I, Fukushima Health Database.
| All, n (%) | All | 40-44 | 45-49 | 50-54 | 55-59 | 60-64 | 65-69 | 70-74 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 178186 | (100) | 17214 | (9.7) | 16109 | (9.0) | 18918 | (10.6) | 22242 | (12.5) | 32457 | (18.2) | 39945 | (22.4) | 31301 | (17.6) | P-value | |
| Men, n (%) | 86703 | (48.7) | 9937 | (57.7) | 8706 | (54.0) | 9965 | (52.7) | 11321 | (50.9) | 14754 | (45.5) | 18256 | (45.7) | 13764 | (44.0) | <0.0001 |
| BMI, mean (SD), kg/m2 | 23.4 | (3.5) | 23.5 | (4.2) | 23.5 | (4.0) | 23.4 | (3.7) | 23.4 | (3.6) | 23.3 | (3.4) | 23.4 | (3.3) | 23.3 | (3.2) | <0.0001 |
| BMI ≥ 25 kg/m2, n (%) | 51747 | (29.0) | 5276 | (30.6) | 4970 | (30.9) | 5715 | (30.2) | 6554 | (29.5) | 9308 | (28.7) | 11275 | (28.2) | 8649 | (27.6) | <0.0001 |
| SBP, mean (SD), mmHg | 127 | (15.9) | 119 | (15.3) | 122 | (16.1) | 125 | (16.5) | 127 | (16.2) | 128 | (15.8) | 129 | (15.1) | 130 | (14.6) | <0.0001 |
| DBP, mean (SD), mmHg | 76.6 | (10.8) | 74.7 | (12.1) | 76.6 | (12.3) | 78.1 | (12.0) | 78.2 | (11.3) | 77.3 | (10.5) | 76.4 | (9.8) | 75.2 | (9.5) | <0.0001 |
| Hypertension or antihypertensive medication, n (%) | 62149 | (34.9) | 1832 | (10.6) | 2669 | (16.6) | 4668 | (24.7) | 6959 | (31.3) | 12089 | (37.2) | 17845 | (44.7) | 16087 | (51.4) | <0.0001 |
| Diabetes, n (%) | 18287 | (10.3) | 639 | (3.7) | 845 | (5.2) | 1406 | (7.4) | 2256 | (10.1) | 3715 | (11.4) | 5295 | (13.3) | 4131 | (13.2) | <0.0001 |
| LDL-C , median (IQR), mg/dL | 123 | (103, 144) | 118 | (98, 141) | 121 | (101, 143) | 126 | (106, 148) | 127 | 107, 148) | 125 | (105, 145) | 123 | (103, 142) | 119 | (101, 138) | <0.0001 |
| LDL-C ≥ 160, n (%) | 22031 | (12.4) | 1990 | (11.6) | 2086 | (12.9) | 2864 | (15.1) | 3390 | (15.2) | 4382 | (13.5) | 4590 | (11.5) | 2729 | (8.7) | <0.0001 |
| HDL-C, median (IQR), mg/dL | 60 | (50, 71) | 58 | (48, 70) | 59 | (49, 71) | 60 | (50, 72) | 61 | (51, 72) | 60 | (51, 71) | 59 | (50, 70) | 59 | (50, 70) | <0.0001 |
| HDL-C <40, n (%) | 9350 | (5.2) | 7871 | (45.7) | 7145 | (44.4) | 8149 | (43.1) | 9509 | (42.8) | 14218 | (43.8) | 18038 | (45.2) | 14342 | (45.8) | <0.0001 |
| Triglyceride, median (IQR), mg/dL | 95 | (68, 136) | 87 | (59, 136) | 90 | (60 138) | 95 | (66, 142) | 96 | (69, 140) | 96 | (70, 137) | 97 | (71, 135) | 95 | (70, 130) | <0.0001 |
| Triglyceride ≥ 150, n (%) | 35862 | (20.1) | 3590 | (20.9) | 3515 | (21.8) | 4288 | (22.7) | 4826 | (21.7) | 6598 | (20.3) | 7654 | (19.2) | 5391 | (17.2) | <0.0001 |
| Antihyperglycemic medication, n (%) | 32225 | (18.1) | 449 | (2.6) | 790 | (4.9) | 1669 | (8.8) | 3170 | (14.3) | 6571 | (20.2) | 10276 | (25.7) | 9300 | (29.7) | <0.0001 |
| Smoke, n (%) | 39137 | (22.0) | 6949 | (40.4) | 5869 | (36.4) | 5839 | (30.9) | 6153 | (27.7) | 6050 | (18.6) | 5355 | (13.4) | 2922 | (9.3) | <0.0001 |
| Alcohol drink everyday, n (%) | 50965 | (28.6) | 4949 | (28.7) | 4803 | (29.8) | 5913 | (31.3) | 7214 | (32.4) | 9507 | (29.3) | 10992 | (27.5) | 7587 | (24.2) | <0.0001 |
| Physical exercise, n (%) | 53481 | (30.0) | 2664 | (15.5) | 2568 | (15.9) | 3215 | (17.0) | 4339 | (19.5) | 9293 | (28.6) | 16219 | (40.6) | 15183 | (48.5) | <0.0001 |
| Fast walking pace, n (%) | 78502 | (44.1) | 6674 | (38.8) | 6745 | (41.9) | 8107 | (42.9) | 9540 | (42.9) | 14242 | (43.9) | 18420 | (46.1) | 14774 | (47.2) | <0.0001 |
| Body weight change ±3 kg/year, n (%) | 39430 | (22.1) | 5738 | (33.3) | 4629 | (28.7) | 4596 | (24.3) | 4990 | (22.4) | 6729 | (20.7) | 7698 | (19.3) | 5050 | (16.1) | <0.0001 |
Abbreviation: BMI, body mass index; SD, standard deviation; SBP, systolic blood pressure; DBP, diastolic blood pressure; LDL-C, low-density lipoprotein cholesterol; IQR, interquartile range; HDL-C, high-density lipoprotein cholesterol.
Incidence rates of IS by risk factors are shown in Supplementary Table 1 . Similar risk profiles occurred in participants from the two cohorts; for example, with increasing age, BMI, and BP, the IS incidence rates increased. Lower incidence rates were seen in women, persons without diabetes, and those without antihypertensive or antihyperglycemic medications, with higher HDL levels, lower LDL levels, and in those who drank alcohol sometimes, non-smokers, walked fast, or did not experience a 3 kg change in body weight within one year. However, the Cohort II participants tended to have a higher risk in most factor categories than the Cohort I participants, except in those aged 40-44 years, or with BMI <18.5 kg/m2, elevated BP, HDL-C <40 mg/dL, or without diabetes, or smokers, everyday drinkers, those with a fast walking pace, or with body weight change.
Supplementary Table 1. Cases, person-time, and incidence rates of ischemic stroke in variable classes of Cohort I and II.
| Variable | Category | Cohort I, 2014-2018 | Cohort II, 2015-2019 | ||||
|---|---|---|---|---|---|---|---|
| Person-time (year) | Case (n) |
Incidence rate (1/100,000 person-year) |
Person-time (year) | Case (n) |
Incidence rate (1/100,000 person-year) |
||
| All | 580,636 | 1044 | 179.80 | 146,614 | 295 | 201.21 | |
| Age group, years | 40-44 | 57,887 | 20 | 34.55 | 25,296 | 6 | 23.72 |
| 45-49 | 54,606 | 37 | 67.76 | 14,305 | 10 | 69.91 | |
| 50-54 | 64,554 | 46 | 71.26 | 14,504 | 15 | 103.42 | |
| 55-59 | 72,016 | 87 | 120.81 | 16,633 | 22 | 132.27 | |
| 60-64 | 109,933 | 190 | 172.83 | 29,001 | 58 | 199.99 | |
| 65-69 | 141,428 | 377 | 266.57 | 33,816 | 111 | 328.25 | |
| 70-74 | 80,212 | 287 | 357.80 | 13,059 | 73 | 559.00 | |
| Sex | Women | 299,614 | 346 | 115.48 | 71,554 | 91 | 127.18 |
| Men | 281,022 | 698 | 248.38 | 75,060 | 204 | 271.78 | |
| BMI, kg/m2 | <18.5 | 32,198 | 37 | 114.91 | 8,446 | 7 | 82.88 |
| 18.5-<25.0 | 380,899 | 626 | 164.35 | 93,946 | 167 | 177.76 | |
| ≥ 25 | 167,539 | 381 | 227.41 | 44,221 | 121 | 273.63 | |
| Blood pressure, mmHg§ | Normal | 145,304 | 92 | 62.32 | 39,427 | 29 | 73.55 |
| High-normal | 162,268 | 207 | 127.57 | 40,130 | 52 | 129.58 | |
| Elevated | 75,380 | 147 | 195.01 | 19,470 | 37 | 190.03 | |
| Hypertension | 197,684 | 598 | 302.50 | 47,586 | 177 | 371.96 | |
| Diabetes | No | 522,953 | 804 | 153.74 | 131,408 | 201 | 152.96 |
| Yes | 57,683 | 240 | 416.07 | 15,205 | 94 | 618.22 | |
| HDL-C, mg/dL | <40 | 29,756 | 98 | 329.34 | 8,579 | 28 | 326.38 |
| 40-59 | 257,572 | 530 | 205.77 | 65,499 | 154 | 235.12 | |
| ≥ 60 | 293,307 | 416 | 141.83 | 72,535 | 113 | 155.79 | |
| LDL-C, mg/dL | <130 | 340,550 | 610 | 179.12 | 85,468 | 179 | 209.44 |
| 130-159 | 167,921 | 304 | 181.04 | 41,613 | 79 | 189.84 | |
| ≥ 160 | 72,165 | 130 | 180.14 | 19,532 | 37 | 189.43 | |
| Triglyceride, mg/dL | <150 | 464,431 | 777 | 167.30 | 115,994 | 216 | 186.22 |
| ≥ 150 | 116,205 | 267 | 229.77 | 30,619 | 79 | 258.01 | |
| Antihyperglycemic medication | No | 476,884 | 826 | 173.21 | 129,450 | 232 | 179.22 |
| Yes | 103,752 | 218 | 210.12 | 17,163 | 63 | 367.07 | |
| Smoking | No smoke | 453,923 | 748 | 164.79 | 108,489 | 207 | 190.80 |
| Smoke | 126,713 | 296 | 233.60 | 38,125 | 88 | 230.82 | |
| Alcohol drinking | Almost no | 266,908 | 429 | 160.73 | 63,208 | 129 | 204.09 |
| Sometimes | 147,593 | 228 | 154.48 | 39,615 | 66 | 166.60 | |
| Everyday | 166,135 | 387 | 232.94 | 43,791 | 100 | 228.36 | |
| Sports exercise | No | 408,624 | 679 | 166.17 | 109,461 | 210 | 191.85 |
| Yes | 172,012 | 365 | 212.19 | 37,152 | 85 | 228.79 | |
| Walking pace | Normal/slow | 324,879 | 635 | 195.46 | 85,201 | 203 | 238.26 |
| Fast | 255,757 | 409 | 159.92 | 61,413 | 92 | 149.81 | |
| Bodyweight change ±3 kg/year | No | 452,506 | 786 | 173.70 | 107,135 | 217 | 202.55 |
| Yes | 128,130 | 258 | 201.36 | 39,479 | 78 | 197.57 | |
Abbreviation: DBP, diastolic blood pressure; SBP, systolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol. §Normal (SBP <120 and DBP <80 mmHg [without antihypertensive medication]); High-normal (SBP <130 and DBP <80 mmHg [without antihypertensive medication]); Elevated (SBP <140 or DBP <90 mmHg [without antihypertensive medication]);Hypertension (SBP ≥ 140 or DBP ≥ 90 mmHg [or using antihypertensive medication]).
The β coefficients and hazard ratios (HRs) for IS risk factors obtained from the multivariate Cox regression analysis in Cohort I are shown in Table 2 . In the discriminatory analysis in Cohort I, the Harrell’s C-statistics (95% CI) was 0.752 (0.737-0.767) for IS risk, showing a moderately good ability to distinguish cases from non-cases.
Table 2. Coefficients, hazard ratio, and 95% confidence intervals of variables included in the final regression model for Cohort I (2014-2018).
| β | P-value | HR | 95% CI | ||
|---|---|---|---|---|---|
| Age, years | 40-44 | Ref. | |||
| 45-49 | 0.6179 | 0.0263 | 1.86 | (1.08– 3.20) | |
| 50-54 | 0.6784 | 0.0116 | 1.97 | (1.16 – 3.34) | |
| 55-59 | 1.2034 | <0.0001 | 3.33 | (2.04 – 5.44) | |
| 60-64 | 1.6252 | <0.0001 | 5.08 | (3.19 – 8.10) | |
| 65-69 | 2.0380 | <0.0001 | 7.68 | (4.86 – 12.13) | |
| 70-74 | 2.5434 | <0.0001 | 12.72 | (7.81 – 19.72) | |
| Sex | Women | Ref. | |||
| Men | 0.5988 | <0.0001 | 1.82 | (1.58 – 2.10) | |
| Blood pressure, mmHg§ | Normal | Ref. | |||
| High-normal | 0.4883 | 0.0001 | 1.63 | (1.27 – 2.09) | |
| Elevated | 0.8680 | <0.0001 | 2.38 | (1.83 – 3.10) | |
| Hypertension | 1.0425 | <0.0001 | 2.84 | (2.27 – 3.55) | |
| Diabetes | No | Ref. | |||
| Yes | 0.5754 | <0.0001 | 1.78 | (1.53 – 2.06) | |
| HDL-C, mg/dL | <40 | Ref. | |||
| 40-59 | -0.2841 | 0.0108 | 0.75 | (0.61 – 0.94) | |
| ≥ 60 | -0.3797 | 0.0011 | 0.68 | (0.55 – 0.86) | |
| LDL-C, mg/dL | <130 | Ref. | |||
| 130-159 | 0.1469 | 0.0385 | 1.16 | (1.01 – 1.33) | |
| ≥ 160 | 0.2721 | 0.0058 | 1.31 | (1.08 – 1.59) | |
| Smoking | No | Ref. | |||
| Smoke | 0.4843 | <0.0001 | 1.62 | (1.40 – 1.88) | |
| Walking pace | Normal/slow | Ref. | |||
| Fast | -0.1853 | 0.004 | 0.83 | (0.73 – 0.94) | |
| Bodyweight change ±3 kg/year | No | Ref. | |||
| Yes | 0.2164 | 0.0029 | 1.24 | (1.08 – 1.43) | |
| Baseline survival function at 4-year, st (4) = 0.99639 |
Abbreviation: DBP, diastolic blood pressure; SBP, systolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol. §Normal (SBP <120 and DBP <80 mmHg [without antihypertensive medication]); High-normal (SBP <130 and DBP <80 mmHg [without antihypertensive medication]); Elevated (SBP <140 or DBP <90 mmHg [without antihypertensive medication]); Hypertension (SBP ≥ 140 or DBP ≥ 90 mmHg [or using antihypertensive medication]).
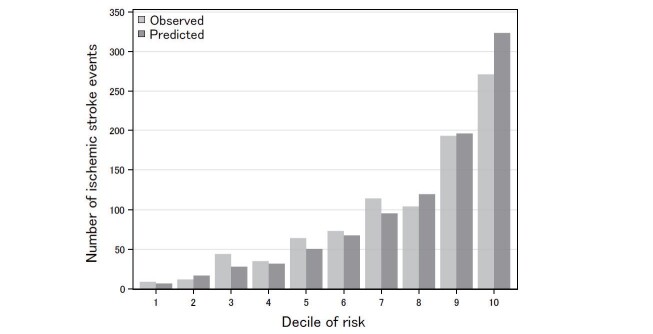
Based on Cohort I derived prediction model, a simple-point score sheet was developed. The mean (SD) score at baseline was 15.0 (7.35), whereas the HR (95% CI) of 4-year IS incidence associated with the score (continuous variable) was 1.16 (1.15-1.17). The Harrell’s C-statistics (95% CI) for the 4-year IS risk was 0.744 (0.729-0.759). In the calibration analysis, the correlations between (O/E) absolute risk numbers based on the 10 risk decile groups are shown in Fig.2 . The calibration plots show that the agreements between the predicted and observed events were good in most risk groups, except in the third and tenth groups. The overall O/E ratio (95% CI) was 0.98 (0.92-1.05), P=0.592.
Fig.2. Calibration plots of the risk scores and observed 4-year risk probability of ischemic stroke incidence in Cohort I.
The ratio (95% confidence interval) of the observed and predicted event was 0.98 (0.92-1.05), P=0.592.
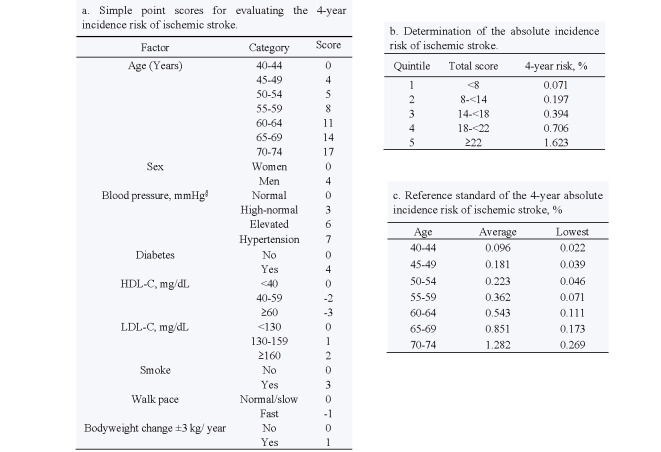
Fig.3 shows the score points for evaluating the 4-year risk of IS incidence by the identified variable and the predicted probability of the total score. The score sheets also provided a comparison of the4-yearly average and low-risk of IS for the age groups.
Fig.3. Four-year incidence risk scores for developing ischemic stroke, using the Fukushima Health Database data, 2014-2019.
Abbreviation: DBP, diastolic blood pressure; SBP, systolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol.
§Normal (SBP <120 and DBP <80 mmHg [without antihypertensive medication]); High-normal (SBP <130 and DBP <80 mmHg [without antihypertensive medication]); Elevated (SBP <140 or DBP <90 mmHg [without antihypertensive medication]); Hypertension (SBP ≥140 or DBP ≥ 90 mmHg [or using antihypertensive medication]).
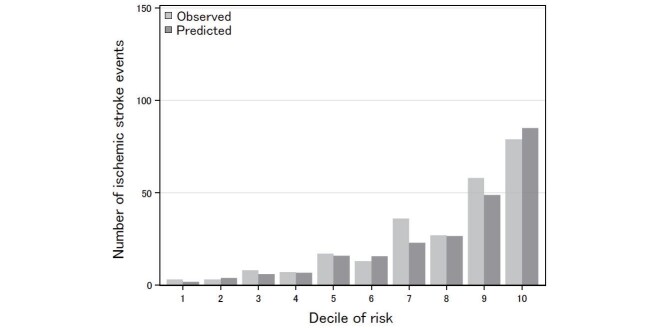
By computing the risk scores based on the Cohort I function, the mean (SD) score at baseline was 14.2 (7.94), and the HR (95% CI) of 4-year IS incidence associated with the score (continuous variable) was 1.17 (1.15-1.19) in Cohort II participants. Fig.4 shows the calibration of the prediction scores in Cohort II, where the Harrell’s C-statistics (95% CI) was 0.770 (0.743-0.797). The overall O/E ratio (95% CI) was 1.08 (0.95 - 1.22), P=0.222, and no significant difference was found between observed and predictive cases in each decile, except in the seventh group. In Cohort II, the variable-specific O/E ratios were insignificant among the categories, except for an underestimation in those with normal BP and those walked normally/slowly ( Supplementary Table 2 ) .
Fig.4. Calibration plots based on risk scores and observed 4-year risk probability of ischemic stroke incidence in Cohort II.
The ratio (95% confidence interval) of the observed and predicted event was 1.08 (0.95-1.22), P=0.222.
Supplementary Table 2. The 4-year observed and expected ischemic stroke events, ratio, and confidence intervals in Cohort II participants (2015-2019).
| Observed | Expected | O/E ratio | 95% confidence interval | |||
|---|---|---|---|---|---|---|
| All | 251 | 233 | 1.08 | 0.95 | 1.22 | |
| Age, years | 40-44 | 4 | 6 | 0.69 | 0.26 | 1.84 |
| 45-49 | 7 | 6 | 1.07 | 0.51 | 2.24 | |
| 50-54 | 11 | 9 | 1.33 | 0.74 | 2.40 | |
| 55-59 | 18 | 16 | 1.12 | 0.71 | 1.78 | |
| 60-64 | 51 | 43 | 1.20 | 0.91 | 1.57 | |
| 65-69 | 95 | 80 | 1.19 | 1.97 | 1.45 | |
| 70-74 | 65 | 74 | 0.88 | 0.69 | 1.13 | |
| Sex | Women | 84 | 68 | 1.24 | 0.99 | 1.53 |
| Men | 167 | 165 | 1.01 | 0.87 | 1.18 | |
| Blood pressure, mmHg§ | Normal | 26 | 18 | 1.47 | 1.00 | 2.16 |
| High-normal | 43 | 40 | 1.06 | 0.79 | 1.43 | |
| Elevated | 34 | 34 | 1.00 | 0.72 | 1.41 | |
| Hypertension | 148 | 141 | 1.05 | 0.89 | 1.23 | |
| Diabetes | No | 178 | 169 | 1.05 | 0.91 | 1.22 |
| Yes | 73 | 64 | 1.15 | 0.91 | 1.44 | |
| HDL-C, mg/dL | <40 | 21 | 26 | 0.81 | 0.53 | 1.25 |
| 40-59 | 132 | 122 | 1.08 | 0.91 | 1.28 | |
| ≥ 60 | 98 | 85 | 1.16 | 0.95 | 1.41 | |
| LDL-C, mg/dL | <130 | 154 | 137 | 1.12 | 0.96 | 1.31 |
| 130-159 | 65 | 64 | 1.01 | 0.79 | 1.29 | |
| ≥ 160 | 32 | 32 | 1.02 | 0.72 | 1.44 | |
| Smoking | No | 179 | 163 | 1.10 | 0.95 | 1.27 |
| Smoke | 72 | 70 | 1.02 | 0.81 | 1.29 | |
| Walking pace | Normal/slow | 173 | 145 | 1.19 | 1.03 | 1.38 |
| Fast | 78 | 88 | 0.89 | 0.71 | 1.11 | |
| Bodyweight change ±3 kg/year | No | 183 | 168 | 1.09 | 0.94 | 1.26 |
| Yes | 68 | 65 | 1.05 | 0.83 | 1.23 | |
Abbreviation: DSP, diastolic blood pressure; SBP, systolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol. §Normal (SBP <120 and DBP <80 [without antihypertensive medication]); High-normal (SBP <130 and DBP <80 mmHg [without antihypertensive medication]); Elevated (SBP <140 or DBP <90 mmHg [without antihypertensive medication]); Hypertension (SBP ≥ 140 or DBP ≥ 90 mmHg [or using antihypertensive medication]).
Discussion
By employing an extensive health insurance database, we developed a 4-year risk prediction model by presenting a simple-point score sheet for the first occurrence of IS, with established risk factors including age, sex, BP, diabetes, smoking, dyslipidemia (low HDL-C and/or high LDL-C), antihypertensive medication, daily walking pace, and body weight change of 3 kg within one year for middle-aged Japanese. The prediction model has a good performance, and was validated in an external cohort both. This shows that IS risk increased by about 16% in 4 years when the total risk score increased by one point from the baseline.
Risk prediction is highly relevant for decision making 34) . The multifactorial problem involves various factors in the development of clinical manifestations. Similarly, in community settings, an early disease risk estimation can help to effectively deliver individualized interventions 13) . Nevertheless, when building and examining a prediction model’s performance, internal and external validations are firmly required 26) . In this study, following the TRIPOD guideline 35) , we conducted internal validations in Cohort I for the identified variables to check the model performance, and implemented an external validation for the risk scores derived.
A few studies have reported individual risk prediction models using multiple risk factors for stroke incidence risk in a relatively short follow-up term. Because of the large population with sufficient case numbers, we could develop the prediction model in this regard. Identical to other studies on stroke risk prediction 12 , 13 , 15 - 20) , in this study, age, sex, BP, smoking, dyslipidemia (either high LDL-C or low HDL-C), and diabetes were the main factors for predicting IS risk. In the Japanese population, serum cholesterol is an independent risk factor for IS incidence, including the inverse associations of HDL-C in the Oyabe Study 36) and the CIRC Study 37) , and the positive associations of LDL-C in the Hisayama Study 38) and non-HDL-C in the Jichi Study 39) and the JPHC Study 40) . However, among stroke incidence prediction studies, only the JPHC Study separately derived the IS risk prediction model by incorporating HDL-C and/or non-HDL-C 14) . It was noted that diabetes mellitus was maintained in the updated prediction model in the Suita Study 16) but not in the Hisayama Study 19) . In the Suita Study (2022), replaced by atrial fibrillation, dyslipidemia was not included in the model 16) . Both the Suita Study 15 , 16) and the Hisayama Study 19) had incorporated chronic kidney disease as a predictor in the lasted publications. In the FDB, BMI, alcohol drinking (frequency and amount information), physical activity, and history of kidney disease (only 0.25% at baseline) were established risk factors 13) , and they were significantly associated with IS risk in the age-sex adjusted regression but not in the multivariate regression analysis. These factors might not be stronger predictors in this population. For diabetes mellitus risk, the Japan Atherosclerosis Society utilized glucose tolerance in the prediction tool for the atherosclerosis disease incidence tool 41) ; however, we did not have the information in the FDB. In addition, we could not confirm the risk of damaged kidney function or urine protein in participants when building the multivariable regression models due to the unavailability of the FDB.
Except for the same risk factors of age, hypertension, diabetes mellitus, and smoking as ours, a Chinese study on a two-year risk prediction model via a national longitudinal survey added heart disease 20) and a Korean study on a ten-year prediction model using a massive insurance data included total cholesterol, physical activity, BMI, and alcohol intake 42) . However, these two studies did not separate the types of stroke in building models. A recent UK Biobank study reported that a slow walking pace is associated with a higher risk of stroke in people over 64 years of age 43) . Our study identified that walking pace and body weight gain or loss of 3 kg within one year were significant predictors of IS risk, which may improve the predictive ability of our model. In the general public health practice, recommendations for weight gain or loss might be necessary depending on individuals’ current body weight, to reduce stroke risk.
The major limitation of this study was that the present outcome was defined using receipts and not by confirming the clinical symptoms or by computed tomography. A validation study from a claims database reported the positive predictive value of IS was 41.6% based on the diagnosis and drug codes, and medical procedures data such as rehabilitation increased the plausibility of identifying IS events 25) . The prediction model could overestimate the IS incidence risk because we included cases that were reported within two years of follow-up in building the model, thus caution is required when applying the prediction tool during individual consultations. Therefore, it is necessary to confirm the present results in the future using ongoing data collection from the stroke incidence registry; and the WHO anatomical therapeutic chemical codes may be applied instead of Japanese drug classifications. Second, due to unavailable information on participants in the FDB, we could not consider other clinical (e.g., atrial fibrillation 12 , 13) , estimated glomerular filtration rate 12) ) , dietary 21) , psychosocial 21) , and genetic factors 35 , 44) for building the risk prediction model. In the future, efforts need to be made to include additional variables incorporating interaction effects 45) , to improve the predictive power. The third limitation is that the short-term follow-up led to less statistical power for deriving the predictive models for other types of stroke, e.g., intracerebral hemorrhage and subarachnoid hemorrhage; therefore, the total stroke risk could be predictive 13 , 42 , 46) . Fourth, we conducted an external validation for the predictive model in Fukushima residents, the generalization of the prediction model may need to be tested in other populations 26 , 35) . Fifth, due to the different categories of predictors for building the prediction model, the results may not be comparable with those of other studies directly 11 , 13 , 20) . Also, we only considered the lifestyle factors - including subjective measurements such as walking speed and body weight changes in the past year - at baseline without monitoring them during the follow-up time. Further, we did not have data on changes in insurance type in study participants, especially for those at retirement ages. Therefore, we could not elucidate the risk in the elderly, such as those over 74 years, and the predictive probability tended to be underestimated in this study.
Conclusion
We developed a 4-year risk prediction model with main factors obtained through health checkups from an extensive insurance database, with good discriminatory power and acceptable calibration ability for IS risk prediction. The simple-point scores may be used by public health professionals, clinicians, or individuals to estimate IS incident risk and advocate lifestyle modifications or seek related medical service. The results of this study may also be applied to update the IS control strategy at the community level. Studies with additional number of factors in a long-term follow-up and with further validations in Japanese or other populations, are necessary to improve the IS risk prediction.
Acknowledgements
This study was supported by a consignment fee for the Health Promotion Center of the Fukushima Medical University from the Fukushima Prefectural Government. The authors are grateful to Mr. Takashi Saito for his administrative support in data preparation and liaison with the Fukushima Government.
Conflict of Interest
All authors declare no conflict of interest.
References
- 1).Nagai M, Ohira T, Takahashi H, Nakano H, Sakai A, Hashimoto S, Yasumura S, Abe M and Fukushima Health Management S: Impact of evacuation onstrends in the prevalence, treatment, and control of hypertension before and after a disaster. J Hypertens, 2018; 36: 924-932 [DOI] [PubMed] [Google Scholar]
- 2).Ohira T, Nakano H, Nagai M, Yumiya Y, Zhang W, Uemura M, Sakai A, Hashimoto S and Fukushima Health Management Survey G: Changes in Cardiovascular Risk Factors After the Great East Japan Earthquake. Asia Pac J Public Health, 2017; 29: 47S-55S [DOI] [PubMed] [Google Scholar]
- 3).Takahashi S, Nakamura M, Yonekura Y, Tanno K, Sakata K, Ogawa A and Kobayashi S: Association between relocation and changes in cardiometabolic risk factors: a longitudinal study in tsunami survivors of the 2011 Great East Japan Earthquake. BMJ Open, 2016; 6: e011291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4).Ohira T, Hosoya M, Yasumura S, Satoh H, Suzuki H, Sakai A, Ohtsuru A, Kawasaki Y, Takahashi A, Ozasa K, Kobashi G, Hashimoto S, Kamiya K, Yamashita S and Abe M: Evacuation and Risk of Hypertension After the Great East Japan Earthquake: The Fukushima Health Management Survey. Hypertension, 2016; 68: 558-564 [DOI] [PubMed] [Google Scholar]
- 5).Ohira T, Hosoya M, Yasumura S, Satoh H, Suzuki H, Sakai A, Ohtsuru A, Kawasaki Y, Takahashi A, Ozasa K, Kobashi G, Kamiya K, Yamashita S and Abe M: Effect of Evacuation on Body Weight After the Great East Japan Earthquake. Am J Prev Med, 2016; 50: 553-560 [DOI] [PubMed] [Google Scholar]
- 6).Takahashi A, Ohira T, Okazaki K, Yasumura S, Sakai A, Maeda M, Yabe H, Hosoya M, Ohtsuru A, Kawasaki Y, Shimabukuro M, Kazama J, Hashimoto S, Watanabe K, Nakano H, Hayashi F, Ohto H, Kamiya K and Ohira H: Effects of Psychological and Lifestyle Factors on Metabolic Syndrome Following the Fukushima Daiichi Nuclear Power Plant Accident: The Fukushima Health Management Survey. J Atheroscler Thromb, 2020; 27: 1010-1018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7).Kinoshita M, Yokote K, Arai H, Iida M, Ishigaki Y, Ishibashi S, Umemoto S, Egusa G, Ohmura H, Okamura T, Kihara S, Koba S, Saito I, Shoji T, Daida H, Tsukamoto K, Deguchi J, Dohi S, Dobashi K, Hamaguchi H, Hara M, Hiro T, Biro S, Fujioka Y, Maruyama C, Miyamoto Y, Murakami Y, Yokode M, Yoshida H, Rakugi H, Wakatsuki A, Yamashita S, Committee for E and Clinical Management of A: Japan Atherosclerosis Society (JAS) Guidelines for Prevention of Atherosclerotic Cardiovascular Diseases 2017. J Atheroscler Thromb, 2018; 25: 846-984 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8).Kannel WB and McGee DL: Composite scoring--methods and predictive validity: insights from the Framingham Study. Health Serv Res, 1987; 22: 499-535 [PMC free article] [PubMed] [Google Scholar]
- 9).D’Agostino RB, Sr., Vasan RS, Pencina MJ, Wolf PA, Cobain M, Massaro JM and Kannel WB: General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation, 2008; 117: 743-753 [DOI] [PubMed] [Google Scholar]
- 10).Nishimura K, Okamura T, Watanabe M, Nakai M, Takegami M, Higashiyama A, Kokubo Y, Okayama A and Miyamoto Y: Predicting coronary heart disease using risk factor categories for a Japanese urban population, and comparison with the framingham risk score: the suita study. J Atheroscler Thromb, 2014; 21: 784-798 [DOI] [PubMed] [Google Scholar]
- 11).Ishikawa S, Matsumoto M, Kayaba K, Gotoh T, Nago N, Tsutsumi A, Kajii E and Jichi Medical School Cohort Study G: Risk charts illustrating the 10-year risk of stroke among residents of Japanese rural communities: the JMS Cohort Study. J Epidemiol, 2009; 19: 101-106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12).Harada A, Ueshima H, Kinoshita Y, Miura K, Ohkubo T, Asayama K and Ohashi Y: Absolute risk score for stroke, myocardial infarction, and all cardiovascular disease: Japan Arteriosclerosis Longitudinal Study. Hypertens Res, 2019; 42: 567-579 [DOI] [PubMed] [Google Scholar]
- 13).Yatsuya H, Iso H, Yamagishi K, Kokubo Y, Saito I, Suzuki K, Sawada N, Inoue M and Tsugane S: Development of a point-based prediction model for the incidence of total stroke: Japan public health center study. Stroke, 2013; 44: 1295-1302 [DOI] [PubMed] [Google Scholar]
- 14).Yatsuya H, Iso H, Li Y, Yamagishi K, Kokubo Y, Saito I, Sawada N, Inoue M and Tsugane S: Development of a Risk Equation for the Incidence of Coronary Artery Disease and Ischemic Stroke for Middle-Aged Japanese- Japan Public Health Center-Based Prospective Study. Circ J, 2016; 80: 1386-1395 [DOI] [PubMed] [Google Scholar]
- 15).Nakai M, Watanabe M, Kokubo Y, Nishimura K, Higashiyama A, Takegami M, Nakao YM, Okamura T and Miyamoto Y: Development of a Cardiovascular Disease Risk Prediction Model Using the Suita Study, a Population-Based Prospective Cohort Study in Japan. J Atheroscler Thromb, 2020; 27: 1160-1175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16).Arafa A, Kokubo Y, Sheerah HA, Sakai Y, Watanabe E, Li J, Honda-Kohmo K, Teramoto M, Kashima R, Nakao YM and Koga M: Developing a Stroke Risk Prediction Model Using Cardiovascular Risk Factors: The Suita Study. Cerebrovasc Dis, 2022; 51: 323-330 [DOI] [PubMed] [Google Scholar]
- 17).Arima H, Yonemoto K, Doi Y, Ninomiya T, Hata J, Tanizaki Y, Fukuhara M, Matsumura K, Iida M and Kiyohara Y: Development and validation of a cardiovascular risk prediction model for Japanese: the Hisayama study. Hypertens Res, 2009; 32: 1119-1122 [DOI] [PubMed] [Google Scholar]
- 18).Honda T, Yoshida D, Hata J, Hirakawa Y, Ishida Y, Shibata M, Sakata S, Kitazono T and Ninomiya T: Development and validation of modified risk prediction models for cardiovascular disease and its subtypes: The Hisayama Study. Atherosclerosis, 2018; 279: 38-44 [DOI] [PubMed] [Google Scholar]
- 19).Honda T, Chen S, Hata J, Yoshida D, Hirakawa Y, Furuta Y, Shibata M, Sakata S, Kitazono T and Ninomiya T: Development and Validation of a Risk Prediction Model for Atherosclerotic Cardiovascular Disease in Japanese Adults: The Hisayama Study. J Atheroscler Thromb, 2022; 29: 345-361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20).Yao Q, Zhang J, Yan K, Zheng Q, Li Y, Zhang L, Wu C, Yang Y, Zhou M and Zhu C: Development and validation of a 2-year new-onset stroke risk prediction model for people over age 45 in China. Medicine (Baltimore), 2020; 99: e22680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21).O’Donnell MJ, Chin SL, Rangarajan S, Xavier D, Liu L, Zhang H, Rao-Melacini P, Zhang X, Pais P, Agapay S, Lopez-Jaramillo P, Damasceno A, Langhorne P, McQueen MJ, Rosengren A, Dehghan M, Hankey GJ, Dans AL, Elsayed A, Avezum A, Mondo C, Diener HC, Ryglewicz D, Czlonkowska A, Pogosova N, Weimar C, Iqbal R, Diaz R, Yusoff K, Yusufali A, Oguz A, Wang X, Penaherrera E, Lanas F, Ogah OS, Ogunniyi A, Iversen HK, Malaga G, Rumboldt Z, Oveisgharan S, Al Hussain F, Magazi D, Nilanont Y, Ferguson J, Pare G, Yusuf S and investigators I: Global and regional effects of potentially modifiable risk factors associated with acute stroke in 32 countries (INTERSTROKE): a case-control study. Lancet, 2016; 388: 761-775 [DOI] [PubMed] [Google Scholar]
- 22).Nomura S, Sakamoto H, Glenn S, Tsugawa Y, Abe SK, Rahman MM, Brown JC, Ezoe S, Fitzmaurice C, Inokuchi T, Kassebaum NJ, Kawakami N, Kita Y, Kondo N, Lim SS, Maruyama S, Miyata H, Mooney MD, Naghavi M, Onoda T, Ota E, Otake Y, Roth GA, Saito E, Tabuchi T, Takasaki Y, Tanimura T, Uechi M, Vos T, Wang H, Inoue M, Murray CJL and Shibuya K: Population health and regional variations of disease burden in Japan, 1990-2015: a systematic subnational analysis for the Global Burden of Disease Study 2015. Lancet, 2017; 390: 1521-1538 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23).Fukushima Health Database (2021). 2022: http: //www.pref.fukushima.lg.jp/sec/21045a/hukushimakennbannkennkoude-tabe-su.html (Assecced 2022 May 15) [Google Scholar]
- 24).Sobue T: Monitoring time trends of disease incidence and mortality on Fukushima and other areas in Japan. 2017; 5-1: [Google Scholar]
- 25).Shima D, Ii Y, Higa S, Kohro T, Hoshide S, Kono K, Fujimoto S, Niijima S, Tomitani N and Kario K: Validation of novel identification algorithms for major adverse cardiovascular events in a Japanese claims database. J Clin Hypertens (Greenwich), 2021; 23: 646-655 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26).Steyerberg EW and Harrell FE, Jr.: Prediction models need appropriate internal, internal-external, and external validation. J Clin Epidemiol, 2016; 69: 245-247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27).Umemura S, Arima H, Arima S, Asayama K, Dohi Y, Hirooka Y, Horio T, Hoshide S, Ikeda S, Ishimitsu T, Ito M, Ito S, Iwashima Y, Kai H, Kamide K, Kanno Y, Kashihara N, Kawano Y, Kikuchi T, Kitamura K, Kitazono T, Kohara K, Kudo M, Kumagai H, Matsumura K, Matsuura H, Miura K, Mukoyama M, Nakamura S, Ohkubo T, Ohya Y, Okura T, Rakugi H, Saitoh S, Shibata H, Shimosawa T, Suzuki H, Takahashi S, Tamura K, Tomiyama H, Tsuchihashi T, Ueda S, Uehara Y, Urata H and Hirawa N: The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2019). Hypertens Res, 2019; 42: 1235-1481 [DOI] [PubMed] [Google Scholar]
- 28).Nishimoto M, Tagawa M, Matsui M, Eriguchi M, Samejima KI, Iseki K, Iseki C, Asahi K, Yamagata K, Konta T, Fujimoto S, Narita I, Kasahara M, Shibagaki Y, Moriyama T, Kondo M, Watanabe T and Tsuruya K: A Prediction Model with Lifestyle in Addition to Previously Known Risk Factors Improves Its Predictive Ability for Cardiovascular Death. Sci Rep, 2019; 9: 12953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29).Harrell FE, Jr., Lee KL and Mark DB: Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med, 1996; 15: 361-387 [DOI] [PubMed] [Google Scholar]
- 30).Ramspek CL, Jager KJ, Dekker FW, Zoccali C and van Diepen M: External validation of prognostic models: what, why, how, when and where? Clin Kidney J, 2021; 14: 49-58 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31).D’Agostino RB, Nam BH: Evaluation of the performance of survival analysis models: discrimination and calibration measures. Handbook of Statistics, 23, 2004; 23 [Google Scholar]
- 32).Park Y, Freedman AN, Gail MH, Pee D, Hollenbeck A, Schatzkin A and Pfeiffer RM: Validation of a colorectal cancer risk prediction model among white patients age 50 years and older. J Clin Oncol, 2009; 27: 694-698 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33).Crowson CS, Atkinson EJ and Therneau TM: Assessing calibration of prognostic risk scores. Stat Methods Med Res, 2016; 25: 1692-1706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34).Van Calster B, Steyerberg EW and Harrell FH: Risk Prediction for Individuals. JAMA, 2015; 314: 1875 [DOI] [PubMed] [Google Scholar]
- 35).Debray TP, Vergouwe Y, Koffijberg H, Nieboer D, Steyerberg EW and Moons KG: A new framework to enhance the interpretation of external validation studies of clinical prediction models. J Clin Epidemiol, 2015; 68: 279-289 [DOI] [PubMed] [Google Scholar]
- 36).Soyama Y, Miura K, Morikawa Y, Nishijo M, Nakanishi Y, Naruse Y, Kagamimori S, Nakagawa H and Oyabe S: High-density lipoprotein cholesterol and risk of stroke in Japanese men and women: the Oyabe Study. Stroke, 2003; 34: 863-868 [DOI] [PubMed] [Google Scholar]
- 37).Chei CL, Yamagishi K, Kitamura A, Kiyama M, Imano H, Ohira T, Cui R, Tanigawa T, Sankai T, Ishikawa Y, Sato S, Hitsumoto S, Iso H and Investigators C: High-density lipoprotein subclasses and risk of stroke and its subtypes in Japanese population: the Circulatory Risk in Communities Study. Stroke, 2013; 44: 327-333 [DOI] [PubMed] [Google Scholar]
- 38).Imamura T, Doi Y, Arima H, Yonemoto K, Hata J, Kubo M, Tanizaki Y, Ibayashi S, Iida M and Kiyohara Y: LDL cholesterol and the development of stroke subtypes and coronary heart disease in a general Japanese population: the Hisayama study. Stroke, 2009; 40: 382-388 [DOI] [PubMed] [Google Scholar]
- 39).Kakehi E, Kotani K, Ishikawa S, Gotoh T, Kayaba K, Nakamura Y and Kajii E: Serum non-high-density lipoprotein cholesterol levels and the incidence of ischemic stroke in a Japanese population: the Jichi Medical School cohort study. Asia Pac J Public Health, 2015; 27: NP535-543 [DOI] [PubMed] [Google Scholar]
- 40).Saito I, Yamagishi K, Kokubo Y, Yatsuya H, Iso H, Sawada N, Inoue M and Tsugane S: Non-High-Density Lipoprotein Cholesterol and Risk of Stroke Subtypes and Coronary Heart Disease: The Japan Public Health Center-Based Prospective (JPHC) Study. J Atheroscler Thromb, 2020; 27: 363-374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41).Incidence prediction tool for atherosclerosis disease. 2022: [Google Scholar]
- 42).Jee SH, Park JW, Lee SY, Nam BH, Ryu HG, Kim SY, Kim YN, Lee JK, Choi SM and Yun JE: Stroke risk prediction model: a risk profile from the Korean study. Atherosclerosis, 2008; 197: 318-325 [DOI] [PubMed] [Google Scholar]
- 43).Hayes S, Forbes JF, Celis-Morales C, Anderson J, Ferguson L, Gill JMR, Gray S, Hastie C, Iliodromoti S, Lyall D, Pellicori P, Sattar N, Welsh CE and Pell J: Association Between Walking Pace and Stroke Incidence: Findings From the UK Biobank Prospective Cohort Study. Stroke, 2020; 51: 1388-1395 [DOI] [PubMed] [Google Scholar]
- 44).Boehme AK, Esenwa C and Elkind MS: Stroke Risk Factors, Genetics, and Prevention. Circ Res, 2017; 120: 472-495 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45).Li Y, Yatsuya H, Tanaka S, Iso H, Okayama A, Tsuji I, Sakata K, Miyamoto Y, Ueshima H, Miura K, Murakami Y and Okamura T: Estimation of 10-Year Risk of Death from Coronary Heart Disease, Stroke, and Cardiovascular Disease in a Pooled Analysis of Japanese Cohorts: EPOCH-JAPAN. J Atheroscler Thromb, 2021; 28: 816-825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46).Ishikawa S, Kazuomi K, Kayaba K, Gotoh T, Nago N, Nakamura Y, Tsutsumi A and Kajii E: Linear relationship between blood pressure and stroke: the Jichi Medical School Cohort Study. J Clin Hypertens (Greenwich), 2007; 9: 677-683 [DOI] [PMC free article] [PubMed] [Google Scholar]