Abstract
Background
Suboptimal clinical trial recruitment contributes to research waste. Evidence suggests there may be gender-based differences in willingness to participate in clinical research. Identifying gender-based differences impacting the willingness of trial participation may assist trial recruitment.
Objectives
To examine factors that influence the willingness of men and women to participate in clinical trials and to identify modifiable factors that may be targeted to optimise trial participation.
Material and methods
Electronic databases were searched with key words relating to ‘gender’, ‘willingness to participate’ and ‘trial’. Included studies were English language and reported gender-based differences in willingness to participate in clinical trials, or factors that influence a single gender to participate in clinical trials. Studies were excluded if they described the demographic factors of trial participants or if the majority of participants were pregnant. Extracted data were coded, categorized, analysed thematically and interpreted using Arksey and O'Malley's framework.
Results
Sixty-three studies were included. Two main themes were identified: trial characteristics and participant characteristics. A number of gender-based differences moderating willingness to participate were observed although only one, ‘concern for self’ was found to influence actual trial participation rates between genders.
Conclusion
The relationship between factors influencing willingness to participate in clinical trials is complex. The influence of gender on willingness to participate, while important, may be moderated by other factors including socioeconomic status, ethnicity and health condition. Exploring factors that influence willingness to participate specific to a study cohort likely offers the most promise to optimise trial recruitment of that cohort.
Keywords: Willingness, Trial, Participation, Recruitment, Gender, Scoping review
1. Introduction
Clinical trials are the gold-standard for evaluating the effectiveness of interventions [1]. However, conducting a successful clinical trial can be challenging [2]. A key contributor to clinical trial failure is low participant recruitment rates [3,4]; up to 35% of clinical trials are discontinued because of difficulties recruiting participants [5] and only 17% of randomised clinical trials in surgery reach target sample sizes within the planned timeframe [4]. Additionally, one third of completed clinical trials are at risk of being underpowered as pre-specified recruitment targets are not attained, jeopardising the trustworthiness of trial findings [4,6].
Strategies to overcome low recruitment rates can include extending trial timelines and incorporating additional recruitment sites [4,6]. However, these strategies often incur a rise in trial costs. In some instances, the cost of overestimating trial recruitment rates can exceed the planned budget by 260% [3]. Moreover, suboptimal clinical trial recruitment and the associated delay or discontinuation raises ethical concerns. Discontinued, delayed or underpowered trials may not meaningfully answer the research question [7], unnecessarily exposing participants to trial interventions without a worthwhile contribution to knowledge. This compromises the implicit ethical contract between researchers and participants [3,6]. Consequently, strategies optimising participant recruitment may reduce clinical trial waste and improve adherence to ethical standards [3].
Many factors influence an individual's willingness to participate (WTP) in a clinical trial, with previous reviews investigating factors among diverse cultures and ethnicities [[8], [9], [10], [11], [12], [13]], and health conditions [[13], [14], [15], [16], [17], [18], [19]]. Despite the breadth of factors investigated in previous reviews, the influence of gender on WTP in clinical trials has not been explicitly explored. Further, while strategies to enhance recruitment to clinical trials have been explored, there is a notable gap in addressing the potential influence of gender on WTP [20]. Men and women do not participate in randomised clinical medical trials at the same rate [21], with a recent systematic review of 300 randomised controlled trials reporting the overall mean enrolment rate of women was 41% of the total number of participants enrolled [22]. Further, men and women differ with regard to health information; women more actively seek health-related information, are more attentive to the health effects of their purchases and receive more informal health-related information from their family members [23]. It follows that men and women may approach the decision to participate in clinical research differently or be influenced by different trial features. However, little is known about the factors influencing the WTP from the perspective of gender. Determining the factors influencing the WTP of both men and women in clinical trials may inform strategies to increase the WTP of both genders, potentially boosting trial recruitment rates. Therefore, the aim of this scoping review was to identify gender-specific differences relating to WTP in clinical trials. With the goal of reducing research waste, a further objective was to summarize any pertinent findings and present them in an accessible format that could function as ‘a checklist’ for researchers. This format is designed to address the main drivers of willingness to participate for both genders. In this scoping review, ‘gender’ refers to personal identification while ‘sex’ refers to biological sex.
2. Material and methods
2.1. Methodological framework
A methodological framework for scoping reviews developed by Arksey and O'Malley [24] was used to guide this review and present a narrative account of the existing literature. The PRISMA-ScR reporting criteria were adhered to Ref. [25].
2.2. Search strategy
A search strategy relating to the terms ‘gender’, ‘willingness to participate’ and ‘trial’ was developed and tested on six electronic medical databases. The search strategy was then refined by one researcher (LH) in consultation with a senior librarian and researchers SB and PO (Appendix 1).
Electronic databases Medline, EMBASE and PsychINFO were searched in July 2022 and uploaded to the bibliographic management software Endnote X7. All primary research was included. Preliminary trials of the search strategy conducted on the CINAHL, PUBMed and Cochrane databases determined these databases did not yield further articles. References of key articles and Google Scholar were also reviewed. Search results were imported into the web-based software platform Covidence [26]. Duplicate references were removed.
2.3. Selecting studies and charting the data
Included studies were English language, peer-reviewed, primary research, qualitative, quantitative or mixed methods studies involving adults that reported gender-based differences in WTP in clinical trials, or factors that influence a single gender to participate in medical clinical trials. Medical clinical trials encompassed studies conducted in healthcare settings, evaluating specific interventions such as medical products, medical devices, lifestyle modifications, or surgeries. These trials compared the effectiveness of the intervention against a comparator, which could be a placebo, no intervention (i.e., standard practice), or another form of intervention [19,20]. Studies exploring WTP in both actual and hypothetical trials were included. Quantitative studies using surveys and questionnaires were included to identify factors influencing WTP in clinical trials and qualitative studies were included to further explore the context of these factors. The review examined responses reported directly by trial participants. Studies were excluded if a majority of participants were pregnant or if the study only described the demographics of those who did or did not participate.
Four researchers (SB,EN, PO [women]; LH [man]) independently assessed study titles and abstracts against inclusion and exclusion criteria in two teams of two, ensuring double checking was adhered to. Full-text articles were retrieved for studies meeting the inclusion criteria and for those requiring further examination to determine eligibility. A final decision on study eligibility was made by consensus.
Data were synthesised using a framework analysis approach adapted for the context of a scoping review. This approach is designed to sift, chart and organise data in accordance with key issues or themes [27]. This method involved familiarization and data extraction, developing a thematic framework, and synthesis and interpretation [27]. Three researchers read the full texts (LH, EN, PO) and extracted relevant content using a standardised data extraction pro rata spreadsheet. Two researchers (LH, EN) used open coding [28] to identify key concepts relating to WTP in a clinical trial, in all papers. Codes were grouped into categories from which a thematic framework was developed. Manuscripts were re-read by two researchers (LH, EN) to refine categories and ensure the thematic framework captured all key concepts outlined in included studies. Themes and domains that emerged from the key concepts and categories were presented to all researchers with final consensus reached by discussion. For single-gender studies, themes were developed separately and then compared and contrasted between the genders. Two researchers (LH, EN) summarised the main facilitators and barriers of WTP for both genders into a table. All authors then reviewed the table, provided comments, and the table was subsequently updated through consensus.
3. Results
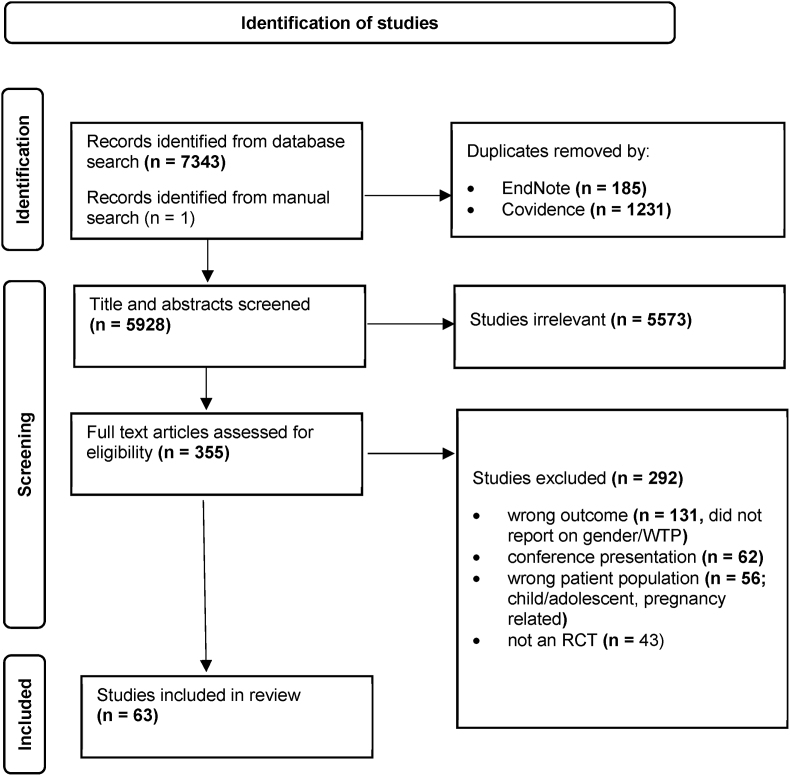
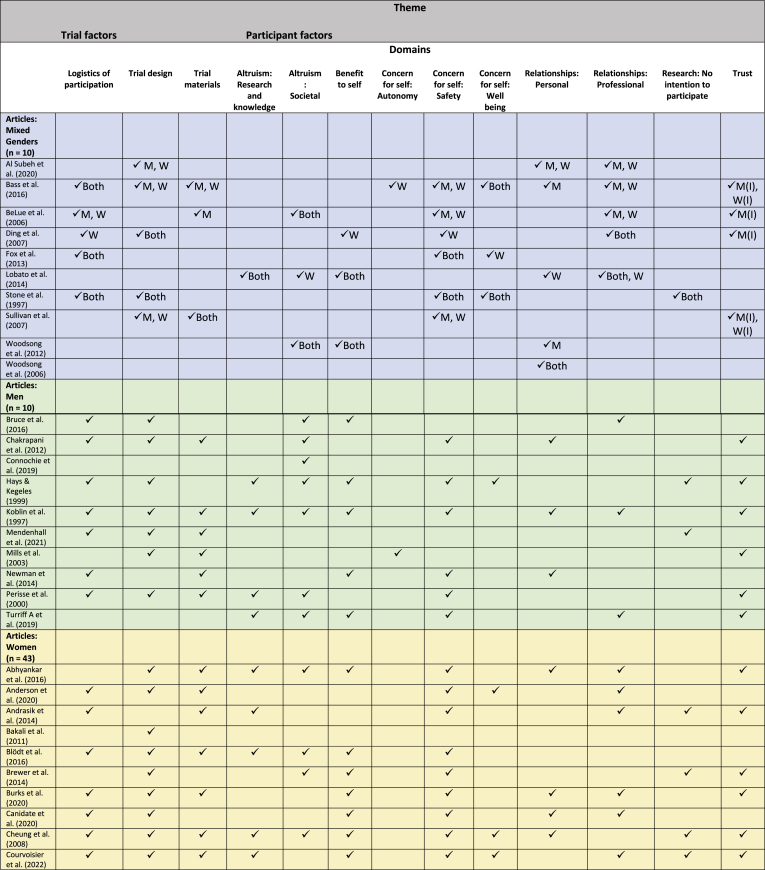
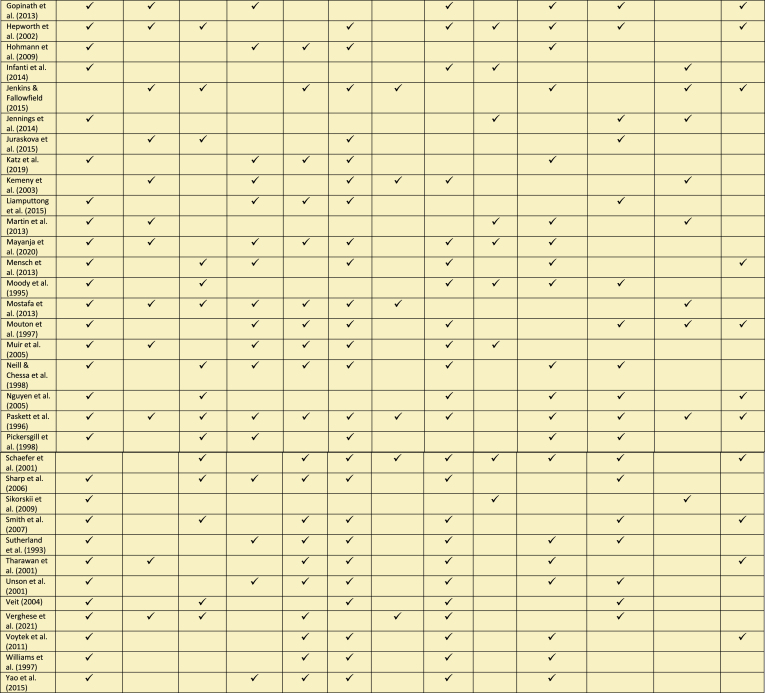
Sixty-three studies were included (Fig. 1), comprising a total of 20,414 participants (10,710 women; 9704 men) across 62 (98%) studies. Notably, one study did not report the number of participants included [29]. Study characteristics are summarised in Table 1 and themes are presented in a narrative form below. Appendix 2 provides a summary of the two main themes influencing WTP: trial factors and participant factors. The breakdown covers the 12 identified domains, further categorized into three gender groups: mixed-genders, men, and women.
Fig. 1.
PRISMA flow chart of study identification.
Table 1.
Characteristics of the included studies.
| Author and year | Country | Study cohort | Age | Actual or Hypothetical Trial* | Data Collection |
|---|---|---|---|---|---|
| Mixed gender studies | |||||
| Al Subeh & Alzoubi 2020 [91] | Jordan | University students in Jordan (n = 1265; 256 M, 1,009W) | <18 yrs = 4 (0.3%); 18–25 = 1246 (98.5%); >25 yrs = 15 (1.2%) | Hypothetical | Survey |
| Bass et al., 2015 [76] | USA | HIV positive patients with urban minority ethnic backgrounds (n = 50; 22 M, 28W) | 47 (28–63) | Hypothetical | Survey |
| BeLue et al., 2006 [71] | USA | African Americans from diverse educational and socioeconomic backgrounds (n = 67; 32 M; 35W) |
Men: mean age 32.3, range 19–60; Women: mean age 42.7, range 19–65 | Hypothetical | Focus group |
| Ding et al., 2007 [89] | USA | People attending internal medicine and cardiology clinics (n = 783; 383 M, 400W) | Men: mean age 55.2 (SD15.6); Women: 52.8 (SD15.6) | Hypothetical | Survey |
| Fox et al., 2021 [90] | UK | Adults with cancer (n = 93; 56 M, 37W) |
Median age category was 60–69 years |
Hypothetical | Survey |
| Lobato et al., 2014 [83] | Brazil | Adults residing in an area of Brazil endemic for intestinal helminth infections (n = 143; 48 M, 95W) |
Mean age 34 years | Actual | Questionnaire |
| Stone et al., 1997 [67] | USA | People receiving primary care for HIV (n = 202; Gender demographics are for the entire study (n = 260; 186 M, 74W)) |
Age distribution: age 21–34 35.4% age 35–49 57.7% 50 and above 6.9% Age demographic are for entire study (n = 260) while reasons for nonparticipation were collected from n = 202 |
Actual | Survey |
| Sullivan et al., 2007 [69] | USA | Adults with HIV or AIDS (n = 6892; 5010 M, 1882W) *Only people who had not participated in a clinical trial were asked about reasons for non-participation (83% M, 85%W) |
Age distribution: Both genders: 18-29: 11% 30-39: 36% ≥40: 53% |
Actual | Interview |
| Woodsong et al., 2006 [29] | India, Malawi, South Africa, Tanzania, USA, Zambia, Zimbabwe | Standard of Care Study (Study 1): Mostly female research participants who were past or potential research participants, selected among those who had participated in previous HIV prevention studies and/or were non-study consumers of HIV prevention and treatment services (n = not reported; approximately 10 individual interviews or 2 focus groups were conducted with potential participants at each of the 9 research sites) Measures of Condom Use (Study 2): Women and men who had recently completed participation in a condom promotion study (n = 80) |
Study 1 and Study 2: ≥18 yrs |
Hypothetical | Interview and focus group |
| Woodsong et al., 2012 [88] | Malawi, Zimbabwe | Women who participated in a previous RCT of two candidate microbicide gels and their male partners (n = 106; 40 M, 66W) | Not reported | Actual | Interview |
| Men only studies | |||||
| Bruce et al., 2016 [70] | USA | Injured urban black men admitted to trauma centre (n = 83) |
Mean age 38.2 (16.2) | Actual | Interview |
| Chakrapani et al., 2012 [81] | India | Men who have sex with men in Chennai and Mumbai, India (n = 68) |
Mean age 28, age range 20–46 | Hypothetical | Focus group |
| Connochie et al., 2019 [86] | USA | Young men who have sex with men at high risk of HIV infection (n = 137) |
Mean age 21.7 (1.83) | Hypothetical | Survey |
| Hays & Kegeles 1999 [66] | USA | Young gay and bisexual men (n = 390) |
Age range 18–29 | Hypothetical | Survey |
| Koblin et al., 1997 [82] | USA | Gay and bisexual men who were HIV-1 antibody negative enrolled in an HIV-1 vaccine preparedness study (n = 698) | Mean age 31.5 years | Hypothetical | Questionnaire |
| Mendenhall et al., 2021 [74] | USA | Men aged ≥50 years (n = 1046) | Age distribution: <60 years 386 (37%); 60+ years 628 (60%); Missing 32 (3%) |
Hypothetical | Survey |
| Mills et al., 2003 [72] | UK | Men with localized prostate cancer (n = 21) | Age range: 50–69 years | Actual | Interview |
| Newman et al., 2014 [68] | India | Men who have sex with men in Chennai and Mumbai, India (n = 400) | Median age 25 years; Age distribution: ≤25 years 51.75%; >25 years 48.28% |
Hypothetical | Survey |
| Perisse et al., 2000 [78] | Brazil | HIV seronegative homosexual and bisexual men in Rio de Janeiro, Brazil (n = 815) |
Age range 18–50 years | Hypothetical | Questionnaire |
| Turriff et al., 2019 [73] | USA | Men with X-linked retinoschisis (n = 13) |
Median age = 47 (range 23–72) | Actual | Interview |
| Women only studies | |||||
| Abhyankar et al., 2016 [85] | UK | Women receiving cancer treatments (n = 21) |
57, 29–81 yrs | Actual | Interview |
| Anderson et al., 2020 [30] | USA | Adolescents and young women with cancer (n = 1264) |
Age at diagnosis 15–39 years. Age at survey not reported. | Actual | Survey |
| Andrasik et al., 2014 [31] | USA | Transgender women (n = 42) |
18–24 yrs 16.7%, 25–29 yrs 19.0%, 30–39 yrs 28.6%, 40–49 yrs 21.4%, >50 yrs 14.3% | Hypothetical | Focus group |
| Bakali et al., 2011 [79] | UK & USA | Women attending urogynecology clinics (n = 363) | Not reported | Hypothetical | Questionnaire |
| Blödt et al., 2016 [32] | Germany | Women participating in a trial evaluating app-administered self-care acupressure for menstrual pain (n = 25) | Incentive group: mean age 26.4, range 24–33; Nonincentive group: mean 22.7, range 21–25 | Actual | Interview |
| Brewer et al. (2014) [84] | USA | African American women attending The Links, Incorporated 2012 National Assembly (n = 381) | Mean age 58 ± 10, range 20–87 | Hypothetical | Survey |
| Burks et al., 2020 [33] | USA | Women enrolled in a phase 2 clinical trial using IORT for early stage breast cancer (n = 20) | Mean age 67.8 (6.99), range 51–79 | Actual | Interview |
| Canidate et al., 2020 [34] | USA | Women with HIV and hazardous drinking (n = 20) | Mean 49.3 | Actual | Interview |
| Cheung et al., 2008 [35] | Canada | Postmenopausal women who had been contacted for potential participation in the RUTH trial (n = 270) | Mean 68.6 (8.0) | Actual | Survey |
| Courvoisier et al., 2022 [36] | Switzerland | Women living with HIV (n = 20) | Mean 48 | Hypothetical | Interview |
| Gopinath et al., 2013 [37] | UK | Women who had declined participation in a surgical RCT (n = 23) | Mean 49, range 34–75 | Actual | Interview |
| Hepworth et al., 2002 [38] | Australia | Post-menopausal women (n = 21) | Mean 57, range 50–69 | Hypothetical | Focus group |
| Hohmann et al., 2009 [39] | USA | Women presenting for screening appointments for either barrier or hormonal contraceptive efficacy trials (n = 108) | Age range 18–40; Barrier subjects: 28 (18–40); Hormonal subjects: 23.5 (18–36) | Actual | Survey |
| Infanti et al., 2014 [40] | Ireland | Women with a recent history of GDM who declined participation in a RCT (n = 156) | Not reported | Actual | Narrative statement |
| Jenkins & Fallowfield 2015 [75] | UK | Women with breast cancer (n = 152) | Age range 29–87 years; the majority of women were in the age group 51–69 years (65%) | Actual | Questionnaire |
| Jennings et al., 2014 [41] | USA | Women who survived rectal or anal cancer (n = 94) | Consenting women (56 years (11.2)) younger than those who declined (69 years (12.7)) | Actual | Interview |
| Juraskova et al., 2015 [77] | Australia & New Zealand | Initial pilot: members of Breast Cancer Network Australia (n = 25); Main pilot: women identified as eligible for the SNAC-2 trial (breast cancer surgical trial) (n = 20) |
Initial Pilot: mean age 52.8 years, (SD 10.32); Main Pilot: mean age 52.7 years, (SD 12.78) | Initial pilot: Hypothetical; Main pilot: Actual |
Questionnaire and interview |
| Katz et al., 2019 [42] | South Africa, Uganda, Zimbabwe | Women who were former VOICE trial participants (n = 171 of which n = 106 also completed questionnaire) | Mean age 28.9 years, Age range 20–41 | Actual | Interview and/or focus group; questionnaire |
| Kemeny et al., 2003 [80] | USA | Women with breast cancer eligible for open treatment trial (n = 154; 77 matched pairs of younger (<65 years old) and older (≥65 years old) women) | Mean age: Younger group 48 years; Older group: 74 years | Actual | Questionnaire (narrative data) |
| Liamputtong et al., 2015 [43] | Thailand | Thai women living with HIV/AIDS (n = 26) | Age ranges 20–30: 15.5% 31-40: 65.5% >40: 19% |
Actual | Interview |
| Martin et al., 2013 [44] | USA | Black and Latina postpartum women who declined participation in a RCT to assess the impact of a behavioural educational intervention aimed at preventing postpartum depression (n = 128) | Mean age 29 (SD 6) | Actual | Interview |
| Mayanja et al., 2020 [45] | Uganda | HIV negative sex workers in Uganda (n = 311; 219 cases, 92 controls) | Median age 27 years (IQR: 23–32) | Hypothetical | Questionnaire |
| Mensch et al., 2013 [46] | India | Female sex workers in southern India (n = 730 surveyed for hypothetical WTP; of these who were eligible, n = 267 enrolled in actual trial, n = 99 eligible but did not enrol in trial) | Mean age 30.8 years; approximately 18% aged under age 25; 13% 40 aged years old or over (those surveyed for WTP) | Hypothetical and Actual | Survey |
| Moody et al., 1995 [47] | USA | Post-menopausal African American women approached for participation in a trial for osteoporosis (n = 8) | Not reported for WTP sample. Mean age of 66.9 years (SD 10.95) for sample recruited to substudy (N = 21). | Actual | Not specified, likely interview |
| Mostafa et al., 2013 [48] | UK | Women with stress urinary incontinence eligible for a surgical trial (n = 166) | Age distribution: 26–44 years: 34.1% accepted, 51.6% refused; 45–64 years: 54.1% accepted, 38.7% refused; ≥65 years: 11.8% accepted, 9.7% refused |
Actual | Questionnaire |
| Mouton et al., 1997 [49] | USA | Nonrespondent women initially invited to participate in the Women's Health Initiative (n = 80) | Mean age 62 years (range 37–86) | Actual | Survey |
| Muir et al., 2005 [50] | Australia | Australian women at high risk of breast cancer (n = 35) | Mean age 37.7 years; age range: 30-45 | Hypothetical | Interview |
| Neill & Chessa 1998 [51] | USA | Women in nontherapeutic drug clinical trials (n = 13) | Mean age 26.75 years; age range 21–46 | Actual | Focus group |
| Nguyen et al., 2005 [52] | USA | Asian-American women with cancer (n = 19) | Age range 33–71 years | Hypothetical | Interview |
| Paskett et al., 1996 [53] | USA | Women with breast cancer (n = 82) | Age distribution: <65 years 55%; ≥65 years 45% |
Actual and hypothetical | Interview |
| Pickersgill et al., 1998 [54] | UK | Women who had volunteered to participate in contraceptive pill trials (n = 126) | Mean age 30.5 years; Age range 18–40 | Actual | Questionnaire |
| Schaefer et al., 2001 [87] | USA | Women who considered participating in the breast cancer prevention trial (BCPT) (n = 26) | Mean age 53 years; Age range 44–67 | Actual | Interview |
| Sharp et al., 2006 [55] | UK | Women with abnormal cervical screening results who enrolled in or declined participation in the TOMBOLA trial (n = 629; n = 492 agreed to participate; n = 137 declined to participate) | Age range 20–59 years | Actual | Questionnaire |
| Sikorskii et al., 2011 [56] | USA | Women with breast cancer (n = 123) | Age for people who declined participation and provided reasons for unwillingness to participate was not recorded. Mean age for participants who dropped out early: 58.12 (SD 12.66) Mean age for participants who completed baseline interview: 57.34 (SD 11.36) |
Actual | Interview |
| Smith et al., 2007 [57] | USA | African American woman (n = 31) | Age range 30–60 | Hypothetical | Focus group |
| Sutherland et al., 1993 [58] | Canada | Healthy women enrolled in randomised controlled trials of cancer prevention (n = 66) | Mean age not reported; at least 30 years | Actual | Questionnaire |
| Tharawan et al., 2001 [59] | Thailand | Thai women from an area of higher HIV infection rate (n = 370) | Mean age = 26.9 years (SD 6.7) | Hypothetical | Questionnaire |
| Unson et al., 2001 [60] | USA | Older African American women invited to participate in a clinical trial on osteoporosis (n = 16) | Mean age = 75; age range 67–86 | Actual | Focus group |
| Veit 2004 [61] | USA | Post-menopausal women (n = 180) | Age distribution: <70 years 70–80 years |
Hypothetical | Questionnaire |
| Verghese et al., 2021 [62] | UK | Postmenopausal women with symptomatic Pelvic Organ Prolapse eligible to participate in the LOTUS study (n = 32) | Age distribution: <65 68% ≥65 14% Age range 52–76 years |
Actual | Interview and focus group |
| Voytek et al., 2011 [63] | USA | African-American women at high risk of HIV infection (n = 17) | Mean age = 36.4 years | Actual | Interview |
| Williams et al., 1997 [64] | USA | HIV positive women (n = 116) | Age range 23–55 years | Actual | Survey |
| Yao et al., 2015 [65] | China | Female sex workers in China (n = 404) | Median age 24 (IQR 20–30) | Hypothetical | Interview |
Abbreviation Definitions: LOTUS: Local Oestrogen Treatment in Postmenopausal Women Undergoing Pelvic Organ Prolapse Surgery; IORT: Intraoperative Radiation Therapy; RUTH: Raloxifene Use for the Heart; RCT: Randomised controlled trial; GDM: Gestational diabetes mellitus; SNAC-2: Sentinel Node Biopsy versus Axillary Clearance 2; VOICE: Vaginal and Oral Interventions to Control the Epidemic trial (MTN-003); IQR: Interquartile range.
Two main themes influencing gender-based WTP in clinical trials were identified: factors relating to the trial (trial characteristics) and factors relating to the individual (participant characteristics). A further twelve domains relating to the two main themes were identified (Appendix 3). Themes and domains are described narratively. Illustrative quotes from qualitative studies are included for context (Table 2). In the discussion, domains were further summarised and presented as strategies that may enhance WTP in a clinical trial for both genders.
Table 2.
Illustrative quotes.
| Logistics of participation | (I am) so busy and have many family and social issues, so cannot commit to any further follow-up (Woman; [48]) |
| I think that women are busy with children and have no time to do that (be in a study) (Woman; [36]). | |
| When I went there … I was thinking about the tokens, money in my pocket to eat with … (Woman; [63]) | |
| Trial design | I realise blind studies are necessary to compare and contrast the effects of new drugs, but it would really piss me off if I |
| discovered I was in the placebo group (Man; [66]) | |
| … I think, well, one of the three (study groups) is going to be better than the other two for me (Man; [72]) | |
| Trial information and materials | I thought the screening was really good because you actually had a doctor there and you could ask any question under the |
| sun about what was going to happen with the drugs and the interactions that might occur and any side effects that might | |
| happen (Woman; [51]) | |
| Altruism: research and knowledge | I would feel privileged to know that, if my participation helped lead to a vaccine, I had played some small roll in its creation |
| (Man; [66]) | |
| Altruism: societal | If you tell me that researching this will help save millions of lives then I'm more apt to do it than if it's some rare thing and |
| it might one day help somebody. So if you can make a closer link to how my participating can actually help, I'll be more | |
| likely to want to get involved (Gender not specified; [71]) | |
| If more attention were being paid to the African American community, and dealing with diseases in the African American | |
| community, dealing with the health issues, I would be willing to offer my time or whatever I needed to help out research in | |
| those particular situations (Gender not specified; [71]) | |
| I am brave and ready to volunteer for the study in order to prevent people like me from infection. I take this stance like I | |
| am deputing myself to the military (Man; [81]) | |
| I would like to improve the level of provided healthcare for future patients (Woman; [48]) | |
| I am happy to take part in any trial that may benefit not only me, but others who may need to have treatment for cancer | |
| (Woman; [75]) | |
| 44 | |
| I feel that it is my duty as a gay man to undergo a little inconvenience in order to put an end to the AIDS epidemic (Man; | |
| [66]) | |
| Benefit to self | Honestly, I hope that there's benefit to myself personally (Man; [73]) |
| I am very pleased to be asked to take part in these trials as I think they will be beneficial to myself e.g. having extra scans, | |
| blood tests etc. and to help other people with my illness on the outcome of the trials (Woman; [75]) | |
| My main intention was to receive ya tan (antiretroviral drugs). It was free then and the doctor said that it was free | |
| because they wanted patients to look and feel better (Woman; [43]) | |
| I decided to go into a trial because if I did not, I might die quickly like my husband. When I started to take part in a trial, | |
| too many people I knew were dying (Woman; [43]) | |
| Concern for self:safety | If for some reason I felt I was risking my own health by participating, I would not do it (Man; [66]) |
| I don't want to be anyone's guinea pig (Man; [66]) | |
| My understanding was they were going to shoot me with AIDS or HIV and see if your body can counteract it like a mouse | |
| to a scientist (Transwoman; [31]) | |
| If you go in for a vaccine trial and want to be a part of it you are admitting to be at high risk … you are admitting to be one | |
| of the marginalised populations (Man; [81]) | |
| I have a whole lot of (trans) girlfriends that want to know about the vaccine … but they too scared to go into certain places | |
| to learn about it because they don't want no one to think that they have it [HIV], which they don't have it (Transwoman; | |
| [31]) | |
| Concern for self:autonomy | When you get my age, you should make your own decisions anyhow, you know, because you know it is your body, and you |
| know what you want to do with it.” (Woman; [60]) | |
| Relationships:professional | I contacted the research team and spoke to the trial team when I went home, I felt this was a brilliant service (Woman; |
| [62]) | |
| Oh, I felt [the physician] and the people that work with her were very good at explaining the trial to come and what the | |
| procedure entailed, and they it, and I just felt very well informed and secure in their talents (Woman; [33]) | |
| A lot of it is the attitude of the person conducting the research … if they have a negative attitude that makes me feel | |
| uncomfortable (Woman; [71]) | |
| (I participated) because you helped me so much the first time. It was very thoughtful (Man; [70]) | |
| 45 | |
| If they had a representative from our community that was hired to work for them and advocate to us on their behalf, then | |
| we could actually have some kind of relationship (Transwoman; [31]) | |
| That is why I will never do something without asking my doctor (Woman; [36]) | |
| Relationships:personal | When you sit in a clinic environment you definitely feel on the spot and though there were a number a questions going |
| through my mind, in clinic I did not feel I could make a decision. But when I went home spoke to my husband and then | |
| contacted the trial team I felt more confident (Woman; [62]) | |
| [We are] … affected by our peers … I talked with [my friend] and.., immediately the fears go away (Woman; [51]) | |
| Because the gel will prevent her from contracting HIV as well as pregnancy. When I considered all these, I decided to | |
| encourage her to continue using the gel (Man; [88]) | |
| Trust | I'm saying it's a guise to suggest that all clinical research is for the purpose of making sure that people survive. Some |
| clinical research is for the purpose of making sure people can make money. We should call it that (Man; [71]) | |
| I trust the doctors here, and I trust the results they've gotten from A to Z (Man; [73]) |
4. Theme 1 trial characteristics
4.1. Logistics of participation
The perceived burden of trial involvement encompassing factors such as effort, commitment and disruption (e.g. attending appointments, access to transportation, travel difficulty), emerged as a primary factor contributing to a decreased WTP across all studies. Notably, this was more pronounced in women [[30], [31], [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42], [43], [44], [45], [46], [47], [48], [49], [50], [51], [52], [53], [54], [55], [56], [57], [58], [59], [60], [61], [62], [63], [64], [65]] and was consistently observed across different ethnicities, geographical locations and health conditions.
In both mixed and single-gender studies, women reported that interruption to child-care, caring and family responsibilities were barriers, although the percentage of women reporting trial logistics as a barrier was moderated by other factors like age. For example, in a study investigating WTP in a diabetes prevention trial among women with a history of gestational diabetes mellitus, younger women were more concerned with travel associated with trial involvement [40]. Additionally, a study of Indian female sex workers reported that those who were more willing to be apart from their family had increased WTP [46]. In contrast, inconvenience as a barrier to trial participation was reported to concern 9% of men in one men-only trial [66].
Trials that offered flexibility (e.g. in appointment times) were reported to increase WTP by a number of women-only studies [44,47]. Further, access to car parking, public transport options or transport provided by the trial facility positively influenced women's WTP [35,36,67]. Long travel distances to trial sites reduced WTP for both genders [68,69]. Additionally, some women-only studies reported WTP increased when the burden of participation was reduced, e.g. the intervention arm had fewer medical appointments than standard treatment [33] or follow-up took place over the phone [37].
The concept of cost neutral trial participation was considered important [39,65,70] and substantially increased WTP for both genders. In a study involving healthy fertile women, it was found 77% of participants would have declined trial participation if ‘reasonable expenses’ had not been provided [54]. Financial incentives were viewed as appropriate to cover the costs of participation (e.g. childcare, transportation, parking) [32,39] and acknowledgement of participant's time [61].
While remuneration increased the WTP of both genders, the underlying reason why sometimes differed. Men considered remuneration important to ensure their family would be ‘taken care of’ should trial participation expose them to harm. In both mixed and women-only studies, compensation for the time required to participate and the gesture it represented towards valuing their effort was considered more important by women [61,71].
4.2. Trial design
The domain of trial design encompassed the acceptance of entrenched research concepts and the impact of trial structure on WTP. The inclusion of the research concepts of randomisation, placebo, and equipoise reduced WTP in both men and women [37,48,66,[72], [73], [74]]. A study involving men with prostate cancer reported the concept that a computer ‘decides’ who receives the intervention was deemed unacceptable by participants [72]. In a study examining the barriers and drivers affecting recruitment to breast cancer trials among women, it was found 39% of non-participants expressed concerned about the process of randomisation [75].
Similarly, the concept of ‘placebo’ was often misunderstood by both genders and reduced WTP. In one mixed-gender exploring WTP among urban minority HIV patients, women expressed concern about being in a placebo group, where they would not receive HIV medicine, fearing potential instability in their health condition [76]. In contrast, men were more likely to conflate the concept of placebo with a distrust of the medical system [76]. In another men-only study investigating WTP in a preventive HIV vaccine trial, participants reported the concept of placebo as unethical and associated it with receiving an ineffective intervention [66]. One women-only study exploring the motivation and experiences of Thai women living with HIV/AIDS participating in clinical trials involving HIV drugs, found WTP was reduced in studies that compared a placebo with a proven intervention. However, WTP increased when the placebo option was removed, and the trial involved the comparison of two established interventions [43].
Both men and women were concerned by the concept of equipoise. They expressed scepticism researchers genuinely felt the options being investigated were equal in terms of patient outcomes [37,66,72]. In a study involving men with prostate cancer, it was reported that 91% of non-participants found the concept of equipoise unacceptable [72].
Studies of both genders hypothesised that a clear explanation of placebo, randomisation and equipoise, detailing why these concepts are included in clinical trials and how these concepts impact an individual's participation, was important to alleviate concern and increase WTP [48,52,66,72,73,[75], [76], [77], [78]]. In a HIV vaccine efficacy trial conducted among HIV negative female sex workers in Uganda, it was reported actual WTP increased when research concepts were understood [45].
Further, trial structure was noted to moderate the WTP for both genders, but was reported more often in women-only trials. For example, in a study assessing the willingness of postmenopausal women to participate in a long-term hormone replacement therapy clinical trial, participants indicated that a five-year study timeframe was more acceptable than a ten-year commitment [38].
Additionally, trial structure relating to the type of comparator arms used moderated WTP for both genders. In a study evaluating decision making regarding prostate cancer treatment and research participation, WTP increased 21% when trial interventions were perceived to produce similar outcomes, compared to trials where the outcomes of the two interventions were perceived to differ significantly [74]. Further, one women-only study reported WTP was higher for trial designs evaluating an existing surgical procedure with a new procedure than when trials evaluated two new surgical procedures [79]. Additionally, WTP reduced when a trial intervention design was perceived to be burdensome for example, when trial participation required diary keeping [58] or regular blood tests [63,65].
4.3. Trial information and materials
The domain of trial information and materials included whether trial information was presented to potential participants, as well as the clarity of the information. Providing adequate trial information was equally important to both men and women. One mixed gender study reported 75% of men and 75% of women who declined participation indicated lack of information was critical to their decision [69]. Further, a number of mixed and male and female single-gender studies indicated the main reason for non-participation in clinical research was lack of information about the trial or not being offered enrolment [30,31,67,69,80].
Trial materials (e.g. handouts or advertisements) that were easy to understand, free from medical jargon and included clearly stated goals [32,36,38,53,55,62,78,81] increased WTP. One women-only study found individuals who had a good understanding of research where four times more likely to participate [53]. Both genders valued honest information describing the risks and benefits of clinical trial participation [36,76,82]. Trial information available from multiple sources (e.g. written format and face-to-face sessions with researchers) and the availability of staff to answer questions about the trial in a timely manner were noted by a number of women-only trials as increasing WTP [51,77].
5. Theme 2 participant factors
5.1. Altruism
Altruistic motivations emerged as an important enabler in WTP in clinical trials for both genders [71,83]. Two types of altruism were noted: Altruism relating to ‘research and knowledge’ described a willingness to be part of a process that specifically advanced knowledge or solved a problem; societal altruism described a WTP motivated by a desire to improve the well-being of others. One mixed-gender study noted both research and societal altruism were equally important to both genders [71] while another mixed-gender study noted that, altruism significantly influenced women [83]. Additionally, research and societal altruism were noted as important moderators of WTP in both men and women-only studies, although there were exceptions. While two women-only studies, including women with pelvic organ prolapse and Thai women at risk of HIV, noted societal altruism was the most important factor influencing WTP [59,62], two other women-only studies (women with breast cancer and women on hormone replacement therapy) noted altruism had minimal impact on WTP [38,75]. Societal altruism in particular appeared important to both men and women from minority communities, including ethnic or sexual orientation, when there was a perception that trial outcomes would specifically benefit that minority [84], particularly immediate family members, or future generations of that minority [35,57,60,70,71].
Improving the health outcome of people with the same health condition also influenced WTP and was observed in both men and women. This was particularly evident in health conditions with outcomes perceived as life-threatening, including cancer and HIV/AIDS [43,53,59,63,64,66,68,75,76,80,81,[85], [86], [87], [88]]. Conversely, when a minority perceived they would not benefit from research, WTP among that minority diminished. One study noted the prevailing consensus was transwomen would be last to benefit from HIV vaccine research. Consequently, transwomen perceived minimal benefit to their community, which reduced their WTP in research [31].
5.2. Benefit to self
The concept of ‘benefit to self’ included access to perceived new or superior health management (e.g. new medication or procedure), improved or more timely access to medical services (e.g. bypassing the waitlist), increased exposure (e.g. more consultations) to people perceived as having greater expertise in managing a particular health condition, accessing health services, medication or health equipment at a reduced cost, and an opportunity to better understand their health condition.
Benefit to self (when an individual perceived trial participation would improve their health outcomes) was described by many studies as a powerful factor influencing WTP among both genders [[32], [33], [34], [35], [36], [37], [38],42,46,49,50,55,64,65,71,74,75,85,88]. In particular, both men and women were motivated by the hope that trial participation would improve their health. For example, all but one participant in a study of men with X-linked retinoschisis participated in a trial in the hope of therapeutic benefit [73].
Benefit to self in the form of improved access to health services appeared to be a particularly strong motivator for trials involving both men and women from lower socioeconomic areas and developing countries where access to basic health services was perceived as limited [42,43,59,64,68,83,88].
Improving health outcomes was also a strong motivator for men and women facing life-threatening outcomes i.e. HIV and cancer, with participation perceived as a means to prolong life [33,54,85]. One study of women with cancer noted that engaging with research was perceived as an active pursuit of treatment and the only option available [85].
5.3. Concern for self
Concern for self was the most reported concept influencing WTP. Three different types of concern for self were noted: safety, autonomy and appropriateness.
Safety concerns that trial participation may be detrimental to health outcomes, in particular, cause undesirable side-effects, pain or a worsening of their health condition, reduced the WTP of both genders. However, it was in this domain that the only quantifiable gender-based difference in WTP was reported. One mixed study of cardiovascular patients determined women were 15% less willing to participate in hypothetical cardiovascular trials than men [89]. This result was attributed to gender-based differences in the perception of benefits and harms, with women perceiving more harm. Another study conducted during the COVID-19 pandemic observed that, while women had higher levels of anxiety and concern than men, no significant difference in trial participation rates was observed between the genders [90]. Willingness to participate was observed to increase when strategies to mitigate concerns were apparent, for example, a women-only study noted WTP increased when an anti-dote for potential side-effects was included [60].
The fear of being ‘experimented on’ was another major barrier to participation and was related to the idea that researchers viewed participants as human guinea pigs [67]. This was observed in both men and women. Further, one women-only gynaecology study noted WTP reduced when there was a concern the intervention under investigation (a new surgical procedure) may not be delivered proficiently [37].
Safety concerns also included potential psychological and societal harm when trial participation was perceived to threaten a person's reputation or position in society. This was observed in two male-only trials in developing countries [68,81], one trial of transwomen [31] and a trial of female sex-workers in China [65]. Closeted gay men in India expressed concern they would be exposed to stigma, discrimination and loss of social status should their participation in a HIV/AIDS vaccine prevention trial reveal their hidden sexuality to their immediate community [68,81]. Similarly, transwomen perceived their participation in a preventative HIV vaccine trial would exacerbate their already marginalised reality as they were concerned others would erroneously think they were HIV positive [31].
Concern about autonomy was observed only in women and involved the perception trial participation may result in relinquishing control over their health management to others, often strangers [36,60,76]. One mixed-gender study of people with HIV reported some women feared trial participation may compromise the stability their current medication regime provided [76].
Concern about one's own appropriateness to participate in research influenced WTP [87]. People who perceived their health condition was too far progressed, or that other health comorbidities were too substantial, demonstrated reduced WTP [35,38,67]. Similarly, people who perceived their health was too good (i.e. they had a mild form of the health condition being investigated) also had reduced WTP. This concept was observed mainly in women [35,37,40,41,47,62] but was also observed in men in two mixed-gender trials [67,90].
5.4. Relationships
The relationships with significant others both professional (e.g. members of the research team, medical professionals) and personal (e.g. family, friends) influenced WTP, although mixed findings were observed. Two mixed-gender studies reported the concept of relationships influenced the WTP of women more than men [71,83]. However, another mixed-gender study observed the influence of relationships on WTP was equal among women and men [76], and was also observed in two men-only studies [81,82].
The intentional fostering of personal relationships by the research team was reported more frequently as highly valued by women [38,62,71]. This included when researchers were perceived to take a genuine interest in the individual, were responsive to the individual's needs and concerns, listened to participants, answered questions, were respectful of privacy, and were understanding of the difficulties encountered when participating in research [31,38,49,62,63]. Positive interactions with the research team increased women's confidence to participate [62,87].
A substantial proportion of women from minority communities were observed to be more willing to participate when a member of the community was represented in the research team [5,31,57,84]. Transwomen indicated that having representation on the research team would increase researcher's understanding of that minority, which would lead to relationship building and ultimately increase transwomen's WTP in research [31].
The approval or recommendation of a trusted doctor or health professional substantially increased WTP of both men and women [30,31,33,37,51,76,81] although this was particularly noted among women from minority communities. One women-only study of Asian American women reported increased rates of research participation among women with Asian oncologists, possibly due to a language and cultural congruence [52]. Similarly, having a personal doctor or health professional involved in the study, or the perception the facility had expertise in the area, increased WTP of both genders. This was related to a decreased risk of harm associated with prestigious organisations [33,51,71,81].
The opinion of family and friends substantially affected the WTP of both genders. Both men and women were noted to consult extensively with family and friends prior to committing to participate in clinical trials [33,34,42,46,52,54,76,81,83]. Researchers that encouraged and acknowledged input from significant family members or friends were perceived to provide an opportunity to emphasize the safety of trial involvement [45]. Conversely, WTP reduced when it was perceived family members were not supportive of trial participation [46,47,53,75].
The influence of personal relationships on WTP was observed to be particularly powerful in some cultures and geographical locations. For example, in Jordan and parts of Africa, a large proportion of both men and women valued and expected a significant family member, usually male (e.g. male spouse, father or brother), to endorse a woman's participation in a clinical trial [29,88,91]. Women from India were observed to consult their extended family more broadly for approval to participate [29,46] compared to women from Jordan and Africa. In contrast, both men and women in the USA held the belief that women should make a decision regarding trial participation independent of men [29].
5.5. Trust
The concept of trust was observed to moderate the WTP in men more than women and was reported in a higher percentage of mixed and men-only studies. Two mixed studies reported men were more likely to be suspicious of, or to hold conspiratorial views about, research [71,76].
Higher levels of research mistrust were also observed in women from minorities [49,52,57]. For example, one study of African American women found there was a general perception that research was biased to benefit white people [57]. Transwomen were also observed to have high levels of research distrust [31].
5.6. Research disinterest
Several studies, encompassing mixed-gender, men-only, and women-only cohorts, concluded that pre-established negative attitudes toward research was a major factor influencing WTP. Notably, certain studies reported over 40% of eligible people were not interested in participating in clinical research [41,56,74]. In contrast, research disinterest was as low as 3% in other studies [40]. The concept of research disinterest was predominately reported in women-only studies.
6. Discussion
A scoping review of 63 studies explored gender-based differences influencing WTP in clinical trials. Two themes emerged that influenced the WTP in clinical trials of both genders: trial characteristics and participant factors. Consistent with these themes, strategies to enhance the WTP for both men and women were developed.
Differences between the genders moderating WTP in clinical trials were identified: social impact of participation, risk perception and mistrust. The social impact of trial participation appeared to influence women more than men. While the WTP of both genders was moderated by the perceived burden of participation, women more commonly related this to social impact. In particular, the impact of trial participation on women's ability to care for their family or dependents was important. Consequently, factors that eased burden of trial participation or increased the efficiency of trial participation (e.g. access to transport and parking, telephone follow-up) increased WTP in women. This review included articles from different socioeconomic, ethnic and geographical backgrounds, including some studies from more ‘traditional’ cultures, which may explain this finding. Additionally, women placed a higher value on the personal connection with the research team. Women's WTP in trials was observed to increase when it was perceived the researchers were intentional about building rapport and demonstrated empathy and understanding. Women valued an environment where they were able to tell their story, and felt heard, respected and valued. Being treated as a ‘number’ substantially reduced women's WTP.
Women were more concerned about potential health risks from trial participation than men. While both genders were observed to overestimate the health risks associated with trial involvement, one hypothetical mixed-gender study reported this concern reduced women's WTP by 15% [89]. The authors speculated that these results may be explained by sociocultural norms promoting coping styles in women that are less effective at reducing anxieties provoked by external threats [92]. However, another explanation may be the higher risk-taking behaviour observed in men. Men take health promotion messages less seriously and are more likely to refrain from consulting medical professionals [93,94]. There is speculation these behaviours are related to the societal pressure to act ‘masculine’ [95,96]. Paradoxically however, while men's risk-taking behaviours may be detrimental at the level of the individual, men's tendency to accept risk may mean men have fewer concerns regarding clinical trial participation.
On the other hand, men were also observed to be more suspicious, distrustful and conspiratorial of the research process than women, [71,76,89]. Why men demonstrate higher levels of conspiratorial thinking is not well understood [89,90,97], however, similar findings were observed in relation to COVID-19, with more men subscribing to conspiratorial views about the pandemic [97]. Although higher levels of research distrust were observed in men, one hypothetical study of people with cardiovascular disease [89] found this did not translate into an observable decrease in WTP in clinical trials. Trials embracing transparency of research goals, conflicts of interest and research proficiency may further increase the WTP of men in clinical trials.
Although gender-based differences in WTP in clinical trials were observed, substantial areas of similarity between the genders were noted, including in the areas of remuneration; non-acceptance of the research concepts of placebo, randomisation and equipoise; the importance of trial information; altruism; recommendations of trusted health professionals and the consulting with trusted individuals. However, the reasons why a concept was important sometimes differed between the genders. For example, remuneration for trial participation was observed to increase the WTP of both genders. For women, remuneration was viewed as an acknowledgement of their participation. In contrast, men viewed remuneration as providing financial stability in the event trial participation resulted in an adverse health outcome. Trials offering remuneration will likely increase the WTP of both genders although the reasons underpinning why remuneration is a moderator of willingness differs between genders. Consequently, how remuneration (cash incentive) is presented in the trial design when recruiting for clinical trials may be important in increasing the WTP of men compared to women. For instance, for women, acknowledgement of their participation holds greater significance as a form of remuneration, whereas for men, remuneration is perceived as a more transactional process.
What underpins the decision to participate in a clinical research trial is complex. Gender appears to be an important factor that moderates the decision making process, however the influence of gender is also moderated significantly by other factors including socioeconomic status, geographical location, ethnic and cultural background, and the health condition being investigated. This dynamic is observed to be fluid; changes in one factor change the weight of impact of another factor [61]. For example, when the perceived risk of an intervention increased, the amount of remuneration required to maintain WTP also increased, however, the amount of remuneration increase required to maintain WTP was contingent on the individual's age and ethnicity [61]. The more barriers reduce WTP, proportionally greater incentives are required to maintain WTP. It therefore stands to reason that, to increase overall WTP, trial designs mitigate as many barriers to participation as possible while amplifying as many factors that increase WTP as possible.
Consequently, based on these review findings, strategies that may enhance WTP among both men and women are summarised and presented in Appendix 4. While it is acknowledged that some drivers may hold more significance for one gender over the other, a comprehensive checklist covering all aspects of WTP for both genders is deemed more effective in reducing overall research waste. In some instances, factors other than gender, such as cultural background, the type of health condition being studies, or whether participants are from a minority, may play a more crucial role in WTP. Therefore, a checklist that encompasses both genders is deemed more appropriate in these situations. Addressing these strategies in trial design and execution may offer opportunities to increase the WTP in clinical trials of both genders. However, given the complexity of interactions influencing WTP in clinical trials, exploring WTP specific to a particular study cohort is recommended to optimise recruitment.
6.1. Design considerations and limitations
This review included studies from different geographical locations, cultures, ethnicities, minorities, marginalised groups, health conditions and interventions. Consequently, emergent strategies to influence WTP in clinical trials may be translational across contexts. However, this scoping review has a number of limitations. Only ten mixed-gender studies explicitly compared differences in WTP between genders. A larger number of mixed-gender studies may be required for gender-differences to be fully explored. Additionally, this review may have been constrained by the design of included studies; a number of studies used questionnaires or surveys where participants chose between predetermined options set by the researcher to indicate reasons for WTP. This method is known to suffer from several drawbacks relating to validity and biases, where this inflexible design may not capture the depth of participant responses [98,99]. Also, studies exploring hypothetical WTP were included. Care must be taken when interpreting these study findings as behavioural intention does not always translate to actual behaviour [6]. Further, in line with scoping review protocols [24], risk of bias and quality of included studies was not assessed, potentially reducing the accuracy of our findings. However, the aim of this scoping review was to understand the landscape of the influence of gender on WTP in clinical trials, therefore studies were not excluded based on quality, and a robust search strategy appropriate for scoping reviews was adhered to Ref. [24]. Further, this review reported the recurring themes from a large number of studies and generated pragmatic strategies to increase the WTP of both genders.
7. Conclusions
A number of important gender-differences moderating WTP in clinical trials were observed in the areas of social impact of trial participation, risk perception and distrust. However, the reasons that fully determine WTP is complex. Exploring factors that influence WTP specific to a study cohort likely offers the most promise to optimise trial recruitment of that cohort.
Funding details
This project was supported by a University of Melbourne, Department of Surgery Translational Grant. The funding source had no role in the study design and did not have any role during its conduct, collections, analyses, and interpretation of data or in the writing of reports and decision to submit for publication.
Institutional review board or ethics committee approval
This review was based on published literature and did not involve the participation of study subjects. Therefore, no IRB or ethics committee approval was required.
CRediT authorship contribution statement
Lyndon J. Hawke: Data curation, Formal analysis, Investigation, Methodology, Validation, Visualization, Writing – original draft, Writing – review & editing. Elizabeth Nelson: Data curation, Formal analysis, Investigation, Methodology, Validation, Visualization, Writing – original draft, Writing – review & editing. Penny O'Brien: Formal analysis, Investigation, Methodology, Writing – review & editing. Kay M. Crossley: Writing – review & editing. Peter F. Choong: Conceptualization, Funding acquisition, Methodology, Resources, Writing – review & editing. Samantha Bunzli: Conceptualization, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – review & editing. Michelle M. Dowsey: Conceptualization, Funding acquisition, Methodology, Project administration, Resources, Supervision, Writing – review & editing.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
The authors thank and acknowledge librarian Mr Patrick Condron for assisting with the search strategies.
Appendix 1. Search strategy example (Medline)
| 1 | Searches sex factors/ |
|---|---|
| 2 | (women* or men* or female* or male*).ti,kf. |
| 3 | (women* or men* or female* or male*).ab. |
| 4 | female/or male/ |
| 5 | gender.mp. [mp = title, book title, abstract, original title, name of substance word, subject heading word, floating sub-heading word, keyword heading word, organism supplementary concept word, protocol supplementary concept word, rare disease supplementary concept word, unique identifier, synonyms] |
| 6 | (gender difference* or gender preference* or gender participat* or gender role*).mp. [mp = title, book title, abstract, original title, name of substance word, subject heading word, floating sub-heading word, keyword heading word, organism supplementary concept word, protocol supplementary concept word, rare disease supplementary concept word, unique identifier, synonyms] |
| 7 | 1 or 2 or 3 or 4 or 5 or 6 |
| 8 | Patient Selection/ |
| 9 | (recruitment or patient selection or sign up or recruiting or involvement or involved).ab. |
| 10 | (recruitment or patient selection or sign up or recruiting or involvement or involved).ti,kf,ab. |
| 11 | (recruit or recruitment or participat* or willing* or involv* or consent or difference* or different or barrier* or incentive* or enrol* or accru*).mp. [mp = title, book title, abstract, original title, name of substance word, subject heading word, floating sub-heading word, keyword heading word, organism supplementary concept word, protocol supplementary concept word, rare disease supplementary concept word, unique identifier, synonyms] |
| 12 | 9 or 10 or 11 |
| 13 | clinical studies as topic/or exp clinical trials as topic/or observational studies as topic/or qualitative studies as topic/or survey as topic/ |
| 14 | (clinical trial or clinical trials or controlled trial or controlled trials or medical research or clinical research or clinical study or clinical studies).ti,kf. |
| 15 | (clinical trial or clinical trials or controlled trial or controlled trials or medical research or clinical research or clinical study or clinical studies).ab. |
| 16 | 13 or 14 or 15 |
| 17 | 7 and 12 and 16 |
| 18 | ((women* or men* or female* or male* or gender*) adj6 (recruit or recruitment or participat* or willing* or involv* or consent or difference* or different or barrier* or incentive* or enrol* or accru*)).ab,kf,ti. |
| 19 | 17 and 18 |
| 20 | (women* or men* or female* or male* or gender*).ti. |
| 21 | 19 and 20 |
| 22 | (recruit or recruitment or participat* or willing* or involv* or consent or difference* or different or barrier* or incentive* or enrol* or accru*).ti. |
| 23 | 21 and 22 |
Appendix 2. Summary of themes and domains relating to willingness to participate for each study
Appendix 3. Themes and domains for the main factors affecting willingness to participate for both genders
| Theme | Domains | Description |
|---|---|---|
| Trial characteristics | Logistics of participation |
|
| Trial design |
|
|
| Trial materials |
|
|
| Participant factors | Altruism: Research and knowledge |
|
| Altruism: Societal |
|
|
| Benefit to self |
|
|
| Concern for self: Autonomy |
|
|
| Concern for self: Safety |
|
|
| Concern for self: Appropriateness |
|
|
| Relationships: Personal |
|
|
| Relationships: Professional |
|
|
| Trust |
|
|
| Research: Disinterest |
|
Appendix 4. Identified strategies to enhance willingness to participate in a clinical trial for both genders
| Domain | Sub-theme | Strategy to increase willingness to participate |
|---|---|---|
| Logistics of participation | Accessibility |
|
|
||
|
||
|
||
|
||
|
||
|
||
|
||
| Minimise disruption |
|
|
|
||
|
||
|
||
|
||
|
||
| Reduce financial burden |
|
|
|
||
|
||
| Trial design | What is research? |
|
| Address key research components |
|
|
|
||
|
||
| Relevance |
|
|
| Trial materials | Keep it simple |
|
|
||
|
||
|
||
| Transparency |
|
|
|
||
|
||
| Distribute information to everyone |
|
|
|
||
|
||
|
||
|
||
| Altruism: Research and | Gaining knowledge |
|
| knowledge |
|
|
|
||
| Altruism: Societal | Helping others |
|
|
||
|
||
|
||
| Benefit to self | Helping myself |
|
|
||
| Concern for Self: Autonomy | Maintaining control |
|
| Concern for self: Safety | Participating in research is unsafe |
|
|
||
|
||
|
||
| Concern for self: Appropriateness | Too unwell or not sick enough |
|
| Relationships: Personal | What other people think matters |
|
|
||
|
||
|
||
| Relationships: | Rapport is everything |
|
| Professional |
|
|
|
||
|
||
|
||
| Cultural appropriateness |
|
|
|
||
|
||
| My doctor knows best |
|
|
|
||
| Trust | Research is not exploitation |
|
|
||
|
||
|
||
| Track record and reputation |
|
|
|
||
|
||
|
||
|
References
- 1.Hariton E., Locascio J.J. Randomised controlled trials - the gold standard for effectiveness research: study design: randomised controlled trials. BJOG. 2018;125(13):1716. doi: 10.1111/1471-0528.15199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bondemark L., Ruf S. Randomized controlled trial: the gold standard or an unobtainable fallacy? Eur. J. Orthod. 2015;37(5):457–461. doi: 10.1093/ejo/cjv046. [DOI] [PubMed] [Google Scholar]
- 3.Schilling C., Tew M., Bunzli S., Shadbolt C., Lohmander L.S., Balogh Z.J., et al. An economic model for estimating trial costs with an application to placebo surgery trials. Appl. Health Econ. Health Pol. 2023;21(2):263–273. doi: 10.1007/s40258-022-00775-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shadbolt C., Naufal E., Bunzli S., Price V., Rele S., Schilling C., et al. Analysis of rates of completion, delays, and participant recruitment in randomized clinical trials in surgery. JAMA Netw. Open. 2023;6(1) doi: 10.1001/jamanetworkopen.2022.50996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mouw T.J., Hong S.W., Sarwar S., Fondaw A.E., Walling A.D., Al-Kasspooles M., et al. Discontinuation of surgical versus nonsurgical clinical trials: an analysis of 88,498 trials. J. Surg. Res. 2018;227:151–157. doi: 10.1016/j.jss.2018.02.039. [DOI] [PubMed] [Google Scholar]
- 6.Bunzli S., Choong E., Shadbolt C., Wall L., Nelson E., Schilling C., et al. Placebo surgery controlled trials: do they achieve what they set out to do? a systematic review. Ann. Surg. 2021;273(6):1102–1107. doi: 10.1097/SLA.0000000000004719. [DOI] [PubMed] [Google Scholar]
- 7.Fogel D.B. Factors associated with clinical trials that fail and opportunities for improving the likelihood of success: a review. Contemp. Clin. Trials Commun. 2018;11:156–164. doi: 10.1016/j.conctc.2018.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Aabdien M., Abdallah I., Bougmiza M.I., Siepmann T., Illigens B. Perceptions and attitudes toward participation in clinical research in the eastern mediterranean region: a systematic review. Medicine (Baltim.) 2022;101(33) doi: 10.1097/MD.0000000000029959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Browne J.L., Rees C.O., van Delden Jjm, Agyepong I., Grobbee D.E., Edwin A., et al. The willingness to participate in biomedical research involving human beings in low-and middle-income countries: a systematic review. Trop. Med. Int. Health. 2019;24(3):264–279. doi: 10.1111/tmi.13195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.George S., Duran N., Norris K. A systematic review of barriers and facilitators to minority research participation among African Americans, Latinos, Asian Americans, and Pacific Islanders. Am. J. Publ. Health. 2014;104(2):e16–e31. doi: 10.2105/AJPH.2013.301706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Limkakeng A., Phadtare A., Shah J., Vaghasia M., Wei D.Y., Shah A., et al. Willingness to participate in clinical trials among patients of Chinese heritage: a meta-synthesis. PLoS One. 2013;8(1) doi: 10.1371/journal.pone.0051328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pariera K.L., Murphy S.T., Meng J., McLaughlin M.L. Exploring willingness to participate in clinical trials by ethnicity. J. Racial Ethn. Health Disparit. 2017;4(4):763–769. doi: 10.1007/s40615-016-0280-6. [DOI] [PubMed] [Google Scholar]
- 13.Rivers D., August E.M., Sehovic I., Lee Green B., Quinn G.P. A systematic review of the factors influencing African Americans' participation in cancer clinical trials. Contemp. Clin. Trials. 2013;35(2):13–32. doi: 10.1016/j.cct.2013.03.007. [DOI] [PubMed] [Google Scholar]
- 14.Ford J.G., Howerton M.W., Lai G.Y., Gary T.L., Bolen S., Gibbons M.C., et al. Barriers to recruiting underrepresented populations to cancer clinical trials: a systematic review. Cancer. 2008;112(2):228–242. doi: 10.1002/cncr.23157. [DOI] [PubMed] [Google Scholar]
- 15.Mills E., Wilson K., Rachlis B., Griffith L., Wu P., Guyatt G., et al. Barriers to participation in HIV drug trials: a systematic review. Lancet Infect. Dis. 2006;6(1):32–38. doi: 10.1016/S1473-3099(05)70324-8. [DOI] [PubMed] [Google Scholar]
- 16.Nielsen Z.E., Berthelsen C.B. Cancer patients' perceptions of factors influencing their decisions on participation in clinical drug trials: a qualitative meta-synthesis. J. Clin. Nurs. 2019;28(13–14):2443–2461. doi: 10.1111/jocn.14785. [DOI] [PubMed] [Google Scholar]
- 17.Salman A., Nguyen C., Lee Y.H., Cooksey-James T. A review of barriers to minorities' participation in cancer clinical trials: implications for future cancer research. J. Immigr. Minority Health. 2016;18(2):447–453. doi: 10.1007/s10903-015-0198-9. [DOI] [PubMed] [Google Scholar]
- 18.Unger J.M., Hershman D.L., Till C., Minasian L.M., Osarogiagbon R.U., Fleury M.E., et al. "When offered to participate": a systematic review and meta-analysis of patient agreement to participate in cancer clinical trials. J. Natl. Cancer Inst. 2021;113(3):244–257. doi: 10.1093/jnci/djaa155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Houghton C., Dowling M., Meskell P., Hunter A., Gardner H., Conway A., et al. Factors that impact on recruitment to randomised trials in health care: a qualitative evidence synthesis. Cochrane Database Syst. Rev. 2020;(10) doi: 10.1002/14651858.MR000045.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Treweek S., Pitkethly M., Cook J., Fraser C., Mitchell E., Sullivan F., et al. Strategies to improve recruitment to randomised trials. Cochrane Database Syst. Rev. 2018;2(2) doi: 10.1002/14651858.MR000013.pub6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wizeman T.M. National Academies Press; 2012. Sex-specific Reporting of Scientific Research: a Workshop Summary. [PubMed] [Google Scholar]
- 22.Daitch V., Turjeman A., Poran I., Tau N., Ayalon-Dangur I., Nashashibi J., et al. Underrepresentation of women in randomized controlled trials: a systematic review and meta-analysis. Trials. 2022;23(1):1038. doi: 10.1186/s13063-022-07004-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ek S. Gender differences in health information behaviour: a Finnish population-based survey. Health Promot. Int. 2015;30(3):736–745. doi: 10.1093/heapro/dat063. [DOI] [PubMed] [Google Scholar]
- 24.Arksey H., O'Malley L. Scoping studies: towards a methodological framework. Int. J. Soc. Res. Methodol. 2005;8(1):19–32. [Google Scholar]
- 25.Tricco A.C., Lillie E., Zarin W., O'Brien K.K., Colquhoun H., Levac D., et al. PRISMA extension for scoping reviews (PRISMA-ScR): checklist and explanation. Ann. Intern. Med. 2018;169(7):467–473. doi: 10.7326/M18-0850. [DOI] [PubMed] [Google Scholar]
- 26.Babineau J. Product review: covidence (systematic review software) J. Can. Health Libr. Assoc. 2014;35(2) [Google Scholar]
- 27.Ritchie J., Spencer L. In: Analyzing Qualitative Data. Bryman A., Burgess R., editors. Routledge; London: 1994. Qualitative data analysis for applied policy research; pp. 305–329. [Google Scholar]
- 28.Gale N.K.H.G., Cameron E., Rashid S., Redwood S. Using the framework method for the analysis of qualitative data in multi-disciplinary health research. BMC Med. Res. Methodol. 2013;13:117. doi: 10.1186/1471-2288-13-117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Woodsong C., MacQueen K., Namey E., Seema S., Morrar N., Mlingo M., et al. Women's autonomy and informed consent in microbicides clinical trials. J. Empir.Res. Hum. Res. Ethics. 2006;1(3):11–26. doi: 10.1525/jer.2006.1.3.11. [DOI] [PubMed] [Google Scholar]
- 30.Anderson C., Smitherman A.B., Meernik C., Edwards T.P., Deal A.M., Cannizzaro N., et al. Patient/provider discussions about clinical trial participation and reasons for nonparticipation among adolescent and young adult women with cancer. J. Adolesc. Young Adult Oncol. 2020;9(1):41–46. doi: 10.1089/jayao.2019.0078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Andrasik M.P., Yoon R., Mooney J., Broder G., Bolton M., Votto T., et al. Exploring barriers and facilitators to participation of male-to-female transgender persons in preventive HIV vaccine clinical trials. Prev. Sci. 2014;15(3):268–276. doi: 10.1007/s11121-013-0371-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Blodt S., Witt C.M., Holmberg C. Women's reasons for participation in a clinical trial for menstrual pain: a qualitative study. BMJ Open. 2016;6(12) doi: 10.1136/bmjopen-2016-012592. (no pagination) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Burks A.C., Doede A., Showalter S.L., Keim-Malpass J. Perceptions of clinical trial participation among women of varying health literacy levels. Oncol. Nurs. Forum. 2020;47(3):273–280. doi: 10.1188/20.ONF.273-280. [DOI] [PubMed] [Google Scholar]
- 34.Canidate S.S., Cook C.L., Varma D., Carnaby G.D., Ennis N., Stetten N.E., et al. Recruitment, experience, and retention among women with HIV and hazardous drinking participating in a clinical trial. BMC Publ. Health. 2020;20(1):1169. doi: 10.1186/s12889-020-09233-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cheung A.M., Lee Y., Kapral M., Scher J., Ho I., Lui-Yee D., et al. Barriers and motivations for women to participate in cardiovascular trials. J. Obstet. Gynaecol. Can. 2008;30(4):332–337. doi: 10.1016/S1701-2163(16)32802-X. [DOI] [PubMed] [Google Scholar]
- 36.Courvoisier N., Storari C., Lesage S., Vittoz L., Barbieux C., Peytremann-Bridevaux I., et al. Facilitators and barriers of women's participation in HIV clinical research in Switzerland: a qualitative study. HIV Med. 2022;23(4):441–447. doi: 10.1111/hiv.13259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gopinath D., Smith A.R.B., Holland C., Reid F.M. Why don't women participate? A qualitative study on non-participation in a surgical randomised controlled trial. Int. UrogynEcol. J. Pelvic Floor Dysfunct. 2013;24(6):969–975. doi: 10.1007/s00192-012-1967-9. [DOI] [PubMed] [Google Scholar]
- 38.Hepworth J., Paine B., Miles H., Marley J., MacLennan A. The willingness of women to participate in a long-term trial of hormone replacement therapy: a qualitative study using focus groups. Psychol. Health Med. 2002;7(4):469–476. [Google Scholar]
- 39.Hohmann H., Reid L., Creinin M.D. Women's motivation to participate in contraceptive efficacy trials. Contraception. 2009;80(3):270–275. doi: 10.1016/j.contraception.2009.03.015. [DOI] [PubMed] [Google Scholar]
- 40.Infanti J.J., O'Dea A., Gibson I., McGuire B.E., Newell J., Glynn L.G., et al. Reasons for participation and non-participation in a diabetes prevention trial among women with prior gestational diabetes mellitus (GDM) BMC Med. Res. Methodol. 2014;14:13. doi: 10.1186/1471-2288-14-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jennings S., Philip E.J., Nelson C., Schuler T., Starr T., Jandorf L., et al. Barriers to recruitment in psycho-oncology: unique challenges in conducting research focusing on sexual health in female survivorship. Psycho Oncol. 2014;23(10):1192–1195. doi: 10.1002/pon.3520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Katz A.W.K., Mensch B.S., Woeber K., Musara P., Etima J., Van Der Straten A. Understanding women's motivations to participate in MTN-003/VOICE, a phase 2b HIV prevention trial with low adherence. BMC Wom. Health. 2019;19(1) doi: 10.1186/s12905-019-0713-6. (no pagination) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Liamputtong P., Haritavorn N., Kiatying-Angsulee N. Participating in HIV clinical trials: reasons and experiences among women living with HIV in Thailand. J. HIV AIDS Soc. Serv. 2015;14(2):239–256. [Google Scholar]
- 44.Martin A., Negron R., Balbierz A., Bickell N., Howell E.A. Recruitment of black and Latina women to a randomized controlled trial. J. Health Care Poor Underserved. 2013;24(3):1102–1114. doi: 10.1353/hpu.2013.0125. [DOI] [PubMed] [Google Scholar]
- 45.Mayanja Y., Abaasa A., Namale G., Price M.A., Kamali A. Willingness of female sex workers in Kampala, Uganda to participate in future HIV vaccine trials: a case control study. BMC Publ. Health. 2020;20(1):1789. doi: 10.1186/s12889-020-09932-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Mensch B.S., Friedland B.A., Abbott S.A., Katzen L.L., Tun W., Kelly C.A., et al. Characteristics of female sex workers in southern India willing and unwilling to participate in a placebo gel trial. AIDS Behav. 2013;17(2):585–597. doi: 10.1007/s10461-012-0259-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Moody L.E., Gregory S.J., Bocanegra T., Vasey F. Factors influencing post-menopausal African-American women's participation in a clinical trial. J. Am. Acad. Nurse Pract. 1995;7(10):483–488. doi: 10.1111/j.1745-7599.1995.tb01116.x. [DOI] [PubMed] [Google Scholar]
- 48.Mostafa A., N'Dow J., Abdel-Fattah M. Factors influencing women's decision to participate or not in a surgical randomised controlled trial for surgical treatment of female stress urinary incontinence. BioMed Res. Int. 2013;2013 doi: 10.1155/2013/139813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Mouton C.P., Harris S., Rovi S., Solorzano P., Johnson M.S. Barriers to black women's participation in cancer clinical trials. J. Natl. Med. Assoc. 1997;89(11):721–727. [PMC free article] [PubMed] [Google Scholar]
- 50.Muir A., Meiser B., Tucker M., Andrews L., Tucker K., Friedlander M. The feasibility of women at high risk for breast cancer participating in chemoprevention trials: an attitudinal study. J. Psychosoc. Oncol. 2005;22(4):31–45. [Google Scholar]
- 51.Neill K.M., Chessa F. Recruitment and retention of women in nontherapeutic clinical trials. Appl. Nurs. Res. 1998;11(3):148–151. doi: 10.1016/s0897-1897(98)80152-3. [DOI] [PubMed] [Google Scholar]
- 52.Nguyen T.T., Somkin C.P., Ma Y., Fung L.C., Nguyen T. Participation of Asian-American women in cancer treatment research: a pilot study. J. Natl. Cancer Inst. 2005;(35):102–105. doi: 10.1093/jncimonographs/lgi046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Paskett E.D., Muss H.B., Case L.D., Cooper M.R. Participation in clinical treatment trials: factors affecting participation for women with breast cancer. J. Womens Health. 1996;5(6):585–592. [Google Scholar]
- 54.Pickersgill A., Furniss H., Seif M.W. Why do women volunteer to participate in clinical studies? J. Obstet. Gynaecol. 1998;18(1):68–71. doi: 10.1080/01443619868334. [DOI] [PubMed] [Google Scholar]
- 55.Sharp L., Cotton S.C., Alexander L., Williams E., Gray N.M., Reid J.M. Reasons for participation and non-participation in a randomized controlled trial: postal questionnaire surveys of women eligible for TOMBOLA (Trial of Management of Borderline and Other Low-Grade Abnormal smears) Clin. Trials. 2006;3(5):431–442. doi: 10.1177/1740774506070812. [DOI] [PubMed] [Google Scholar]
- 56.Sikorskii A., Wyatt G.K., Siddiqi A.E.A., Tamkus D. Recruitment and early retention of women with advanced breast cancer in a complementary and alternative medicine trial. Evid. Based Complement Alternat. Med. 2011;2011 doi: 10.1093/ecam/nep051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Smith Y.R., Johnson A.M., Newman L.A., Greene A., Johnson T.R.B., Rogers J.L. Perceptions of clinical research participation among African American women. J. Womens Health. 2007;16(3):423–428. doi: 10.1089/jwh.2006.0124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Sutherland H.J., Carlin K., Harper W., Martin L.J., Greenberg C.V., Till J.E., et al. A study of diet and breast cancer prevention in Canada: why healthy women participate in controlled trials. Cancer Causes Control. 1993;4(6):521–528. doi: 10.1007/BF00052427. [DOI] [PubMed] [Google Scholar]
- 59.Tharawan K., Manopaiboon C., Ellertson C., Limpakarnjanarat K., Chaikummao S., Kilmarx P.H., et al. Women's willingness to participate in microbicide trials in Northern Thailand. J. Acquir. Immune Defic. Syndr. 2001;28(2):180–186. doi: 10.1097/00126334-200110010-00011. [DOI] [PubMed] [Google Scholar]
- 60.Unson C.G., Dunbar N., Curry L., Kenyon L., Prestwood K. The effects of knowledge, attitudes, and significant others on decisions to enroll in a clinical trial on osteoporosis: implications for recruitment of older African-American women. J. Natl. Med. Assoc. 2001;93(10):392–401. ; discussion 2. [PMC free article] [PubMed] [Google Scholar]
- 61.Veit C.T. A single mathematical model predicts physicians' recommendations and postmenopausal women's decisions to participate in a clinical trial to prevent breast cancer or coronary heart disease. Med. Decis. Making. 2004;24(4):330–350. doi: 10.1177/0272989X04267007. [DOI] [PubMed] [Google Scholar]
- 62.Verghese T.S., Merriel A., Leighton L., Latthe P. Willingness of postmenopausal women to participate in a study involving local vaginal oestrogen treatment as an adjunct to pelvic organ prolapse surgery: a qualitative study. Int. UrogynEcol. J. Pelvic Floor Dysfunct. 2021;32(2):413–421. doi: 10.1007/s00192-020-04480-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Voytek C.D., Jones K.T., Metzger D.S. Selectively willing and conditionally able: HIV vaccine trial participation among women at " high risk" of HIV infection. Vaccine. 2011;29(36):6130–6135. doi: 10.1016/j.vaccine.2011.06.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Williams A.B., Singh M.P., Dos Santos K., Winfrey J., Mezger J. Report from the field: participation of HIV-positive women in clinical research. AIDS Publ. Pol. J. 1997;12(1):46–52. [PubMed] [Google Scholar]
- 65.Yao N., Zeng Q., Zhong N.X., Li D.X., Huang L.A., Shao M.Y., et al. Use of partial least squares path modelling to assess the willingness of Chinese female sex workers to participate in a microbicide trial. Publ. Health. 2015;129(9):1187–1193. doi: 10.1016/j.puhe.2015.06.001. [DOI] [PubMed] [Google Scholar]
- 66.Hays R.B., Kegeles S.M. Factors related to the willingness of young gay men to participate in preventive HIV vaccine trials. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 1999;20(2):164–171. doi: 10.1097/00042560-199902010-00009. [DOI] [PubMed] [Google Scholar]
- 67.Stone V.E., Mauch M.Y., Steger K., Janas S.F., Craven D.E. Race, gender, drug use, and participation in AIDS clinical trials: lessons from a municipal hospital cohort. J. Gen. Intern. Med. 1997;12(3):150–157. doi: 10.1007/s11606-006-5022-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Newman P.A., Chakrapani V., Weaver J., Shunmugam M., Rubincam C. Willingness to participate in HIV vaccine trials among men who have sex with men in Chennai and Mumbai, India. Vaccine. 2014;32(44):5854–5861. doi: 10.1016/j.vaccine.2014.08.043. [DOI] [PubMed] [Google Scholar]
- 69.Sullivan P.S., McNaghten A.D., Begley E., Hutchinson A., Cargill V.A. Enrollment of racial/ethnic minorities and women with HIV in clinical research studies of HIV medicines. J. Natl. Med. Assoc. 2007;99(3):242–250. [PMC free article] [PubMed] [Google Scholar]
- 70.Bruce M.M., Ulrich C.M., Kassam-Adams N., Richmond T.S. Seriously injured urban black men's perceptions of clinical research participation. J. Racial Ethn. Health Disparit. 2016;3(4):724–730. doi: 10.1007/s40615-015-0191-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.BeLue R., Taylor-Richardson K.D., Lin J., Rivera A.T., Grandison D. African Americans and participation in clinical trials: differences in beliefs and attitudes by gender. Contemp. Clin. Trials. 2006;27(6):498–505. doi: 10.1016/j.cct.2006.08.001. [DOI] [PubMed] [Google Scholar]
- 72.Mills N., Donovan J.L., Smith M., Jacoby A., Neal D.E., Hamdy F.C. Perceptions of equipoise are crucial to trial participation: a qualitative study of men in the ProtecT study. Contr. Clin. Trials. 2003;24(3):272–282. doi: 10.1016/s0197-2456(03)00020-5. [DOI] [PubMed] [Google Scholar]
- 73.Turriff A., Blain D., Similuk M., Biesecker B., Wiley H., Cukras C., et al. Motivations and decision making processes of men with x-linked retinoschisis considering participation in an ocular gene therapy trial. Am. J. Ophthalmol. 2019;204:90–96. doi: 10.1016/j.ajo.2019.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Mendenhall N.P., Rausch Osian S.M., Bryant C.M., Hoppe B.S., Morris C.G. What men want: results from a national survey on decision making for prostate cancer treatment and research participation. Clin. Transl. Sc. 2021;14(6):2314–2326. doi: 10.1111/cts.13090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Jenkins V.A., Fallowfield L.J. For the benefit of others: reasons why women with breast cancer participate in RCTs. Breast Care. 2015;10(2):88–93. doi: 10.1159/000376563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Bass S.B., Wolak C., Greener J., Tedaldi E., Nanavati A., Ruppert K., et al. Using perceptual mapping methods to understand gender differences in perceived barriers and benefits of clinical research participation in urban minority HIV+ patients. AIDS Care. 2015;28(4):528–536. doi: 10.1080/09540121.2015.1112352. [DOI] [PubMed] [Google Scholar]
- 77.Juraskova I., Butow P., Fisher A., Bonner C., Anderson C., Bu S., et al. Development and piloting of a decision aid for women considering participation in the sentinel node biopsy versus axillary clearance 2 breast cancer trial. Clin. Trials. 2015;12(4):409–417. doi: 10.1177/1740774515586404. [DOI] [PubMed] [Google Scholar]
- 78.Perisse A.R., Schechter M., Moreira R.I., do Lago R.F., Santoro-Lopes G., Harrison L.H. Willingness to participate in HIV vaccine trials among men who have sex with men in Rio de Janeiro, Brazil. Projeto praca onze study group. J. Acquir. Immune Defic. Syndr. 2000;25(5):459–463. doi: 10.1097/00126334-200012150-00012. [DOI] [PubMed] [Google Scholar]
- 79.Bakali E., Gafni-Kane A., Botros S., Tincello D.G. Factors influencing women's participation in urogynecology research in the UK and USA. Neurourol. Urodyn. 2011;30(8):1462–1466. doi: 10.1002/nau.21167. [DOI] [PubMed] [Google Scholar]
- 80.Kemeny M.M., Peterson B.L., Kornblith A.B., Muss H.B., Wheeler J., Levine E., et al. Barriers to clinical trial participation by older women with breast cancer. J. Clin. Oncol. 2003;21(12):2268–2275. doi: 10.1200/JCO.2003.09.124. [DOI] [PubMed] [Google Scholar]
- 81.Chakrapani V., Newman P.A., Singhal N., Jerajani J., Shunmugam M. Willingness to participate in HIV vaccine trials among men who have sex with men in Chennai and Mumbai, India: a social ecological approach. PLoS One. 2012;7(12) doi: 10.1371/journal.pone.0051080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Koblin B.A., Avrett S., Taylor P.E., Stevens C.E. Willingness to participate in HIV-1 vaccine efficacy trials and the effect of media events among gay and bisexual men in New York City: project ACHIEVE. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 1997;15(2):165–171. doi: 10.1097/00042560-199706010-00011. [DOI] [PubMed] [Google Scholar]
- 83.Lobato L., Bethony J.M., Pereira F.B., Grahek S.L., Diemert D., Gazzinelli M.F. Impact of gender on the decision to participate in a clinical trial: a cross-sectional study. BMC Publ. Health. 2014;14:1156. doi: 10.1186/1471-2458-14-1156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Brewer L.C., Hayes S.N., Parker M.W., Balls-Berry J.E., Halyard M.Y., Pinn V.W., et al. African American women's perceptions and attitudes regarding participation in medical research: the Mayo Clinic/The Links, Incorporated partnership. J. Womens Health. 2014;23(8):681–687. doi: 10.1089/jwh.2014.4751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Abhyankar P., Velikova G., Summers B., Bekker H.L. Identifying components in consent information needed to support informed decision making about trial participation: an interview study with women managing cancer. Soc. Sci. Med. 2016;161:83–91. doi: 10.1016/j.socscimed.2016.05.040. [DOI] [PubMed] [Google Scholar]
- 86.Connochie D., Tingler R.C., Bauermeister J.A. Young men who have sex with men's awareness, acceptability, and willingness to participate in HIV vaccine trials: results from a nationwide online pilot study. Vaccine. 2019;37(43):6494–6499. doi: 10.1016/j.vaccine.2019.08.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Schaefer K.M., Ladd E., Gergits M.A., Gyauch L. Backing and forthing: the process of decision making by women considering participation in a breast cancer prevention trial. Oncol. Nurs. Forum. 2001;28(4):703–709. [PubMed] [Google Scholar]
- 88.Woodsong C., Alleman P., Musara P., Chandipwisa A., Chirenje M., Martinson F., et al. Preventive misconception as a motivation for participation and adherence in microbicide trials: evidence from female participants and male partners in Malawi and Zimbabwe. AIDS Behav. 2012;16(3):785–790. doi: 10.1007/s10461-011-0027-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ding E.L., Powe N.R., Manson J.E., Sherber N.S., Braunstein J.B. Sex differences in perceived risks, distrust, and willingness to participate in clinical trials: a randomized study of cardiovascular prevention trials. Arch. Intern. Med. 2007;167(9):905–912. doi: 10.1001/archinte.167.9.905. [DOI] [PubMed] [Google Scholar]
- 90.Fox L., Wylie H., Cahill F., Haire A., Green S., Kibaru J., et al. Gender differences in concerns about participating in cancer research during the COVID-19 pandemic. Cancer Control. 2021;28 doi: 10.1177/1073274821989315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Al Subeh Z.Y., Alzoubi K.H. Cultural and religious barriers influencing the participation of women in research: a study from Jordan. Gend. Issues. 2020;37(2):173–186. doi: 10.1007/s12147-019-09235-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.McLean C.P., Anderson E.R. Brave men and timid women? A review of the gender differences in fear and anxiety. Clin. Psychol. Rev. 2009;29(6):496–505. doi: 10.1016/j.cpr.2009.05.003. [DOI] [PubMed] [Google Scholar]
- 93.Mursa R., Patterson C., Halcomb E. Men's help-seeking and engagement with general practice: an integrative review. J. Adv. Nurs. 2022;78(7):1938–1953. doi: 10.1111/jan.15240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Sharp P., Bottorff J.L., Hunt K., Oliffe J.L., Johnson S.T., Dudley L., et al. Men's perspectives of a gender-sensitized health promotion program targeting healthy eating, active living, and social connectedness. Am. J. Men's Health. 2018;12(6):2157–2166. doi: 10.1177/1557988318799159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Doyal L. Sex, gender, and health: the need for a new approach. BMJ. 2001;323(7320):1061–1063. doi: 10.1136/bmj.323.7320.1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Griffiths S. Men's Health. 1996;312(7023):69–70. doi: 10.1136/bmj.312.7023.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Cassese E.C., Farhart C.E., Miller J.M. Gender differences in COVID-19 conspiracy theory beliefs. Polit. Gend. 2020;16(4):1009–1018. [Google Scholar]
- 98.Affleck W., Glass K., Macdonald M.E. The limitations of language: male participants, stoicism, and the qualitative research interview. Am. J. Men's Health. 2013;7(2):155–162. doi: 10.1177/1557988312464038. [DOI] [PubMed] [Google Scholar]
- 99.Ogden J., Lo J. How meaningful are data from Likert scales?: an evaluation of how ratings are made and the role of the response shift in the socially disadvantaged. J. Health Psychol. 2012;17(3):350–361. doi: 10.1177/1359105311417192. [DOI] [PubMed] [Google Scholar]