Abstract
Birt–Hogg–Dubé syndrome (BHD) is an inherited autosomal dominant condition caused by germline mutations in the FLCN gene, mapped to chromosome 17p11.2. Typical manifestations include pulmonary cysts, spontaneous pneumothorax, fibrofolliculomas, and kidney neoplasms. This report details the case of a 56‐year‐old female non‐smoker diagnosed with multiple pulmonary cysts, presenting with a history of recurrent spontaneous pneumothorax. A computed tomography (CT) scan of her daughter revealed similar pulmonary cysts, raising suspicion of BHD. Further abdominal enhanced CT revealed a left renal tumour and cutaneous fibrofolliculomas on her daughter's neck. Consequently, whole‐exome sequencing confirmed an FLCN germline mutation in the patient and three relatives, establishing a diagnosis of BHD. This case highlights the importance of familial pulmonary cysts as a clue for diagnosing BHD, providing crucial insights into comparable clinical presentations.
Keywords: Birt–Hogg–Dubé syndrome, familial pulmonary cysts, FLCN gene
Here, we present a case of a patient with BHD presenting with pulmonary cysts, repeated spontaneous pneumothorax, left renal tumour, and confirmed germline FLCN mutations. Noteworthy, her daughters also presented with concurrent pulmonary cysts; these findings served as a clue for genetic testing for FLCN mutations and the eventual diagnosis of BHD. We emphasize the importance of familial pulmonary cysts as a clue for diagnosing BHD, thus prompting early diagnosis and screening.

INTRODUCTION
Birt–Hogg–Dubé syndrome (BHD) is an inherited autosomal dominant condition characterized by pulmonary cysts, spontaneous pneumothorax, fibrofolliculomas and kidney neoplasms. 1 , 2 The tumour suppressor gene FLCN is the only confirmed pathogenic gene in BHD. A definitive diagnosis of BHD is established when a pathogenic germline heterozygous variant of FLCN is detected. As of 2017, the BHD Syndrome Foundation has reported more than 600 pedigree cases of BHD worldwide. In China, 287 patients from 143 families were identified. 3 Despite continuous reporting of BHD cases, missed diagnoses and misdiagnoses of BHD are common owing to the diversity of its clinical manifestations. Savatt et al. analysed the exomes of 136,000 individuals. They identified 35 patients with pathogenic or potentially pathogenic variants of the FLCN gene (1 in 3885). Of these, only four cases (11.4%) had a prior diagnosis of BHD. 4 Compared with Caucasian populations, Asians typically exhibit pulmonary cysts and/or recurrent spontaneous pneumothorax as the earliest and most frequently occurring symptoms of BHD. 5
Here, we present a case of a patient with BHD presenting with pulmonary cysts, repeated spontaneous pneumothorax, left renal tumour, and confirmed germline FLCN mutations. Noteworthy, her daughter also presented with concurrent pulmonary cysts; these findings served as a clue for genetic testing for FLCN mutations and the eventual diagnosis of BHD. We emphasize the importance of familial pulmonary cysts as a clue for diagnosing BHD, thus prompting early diagnosis and screening.
CASE REPORT
A 56‐year‐old female non‐smoker presented with dyspnea for 2 months. She was diagnosed with pulmonary cysts and pneumothorax at another hospital following a chest computed tomography (CT) examination (Figure 1A). Additionally, she had a 15‐year history of repeated spontaneous pneumothorax. The CT scan revealed multiple diffuse thin‐walled lung parenchyma. According to the algorithmic approach to cystic lung diseases, the differential diagnoses for multiple parenchymal cysts included BHD, lymphangioleiomyomatosis (LAM), pulmonary Langerhans cell histiocytosis (PLCH), lymphocytic interstitial pneumonia (LIP). These parenchymal cysts on this patient's CT scan varied in size, predominantly distributed in the peripheral and basal regions of the lung, with normal intervening parenchyma. Her daughter also showed her chest CT scan of annual health check‐ups, which indicated the presence of pulmonary cysts as well (Figure 1B). Taken together, BHD, a rare autosomal dominant disorder, was primarily considered. Furthermore, an abdominal enhanced CT scan of the patient revealed a left renal tumour (Figure 1C, red arrow) with a heterogeneous enhancement pattern. Cutaneous fibrofolliculomas were observed on the neck of the daughter (Figure 1D). Subsequently, we obtained a family history and acquired their chest CT scans and blood samples for whole‐exome sequencing. We noted that the patient had a frameshift mutation in exon 4 of the FLCN gene (c.57‐58del), three additional family members were also detected with the same mutation and were diagnosed with BHD (Figure 1E).
FIGURE 1.
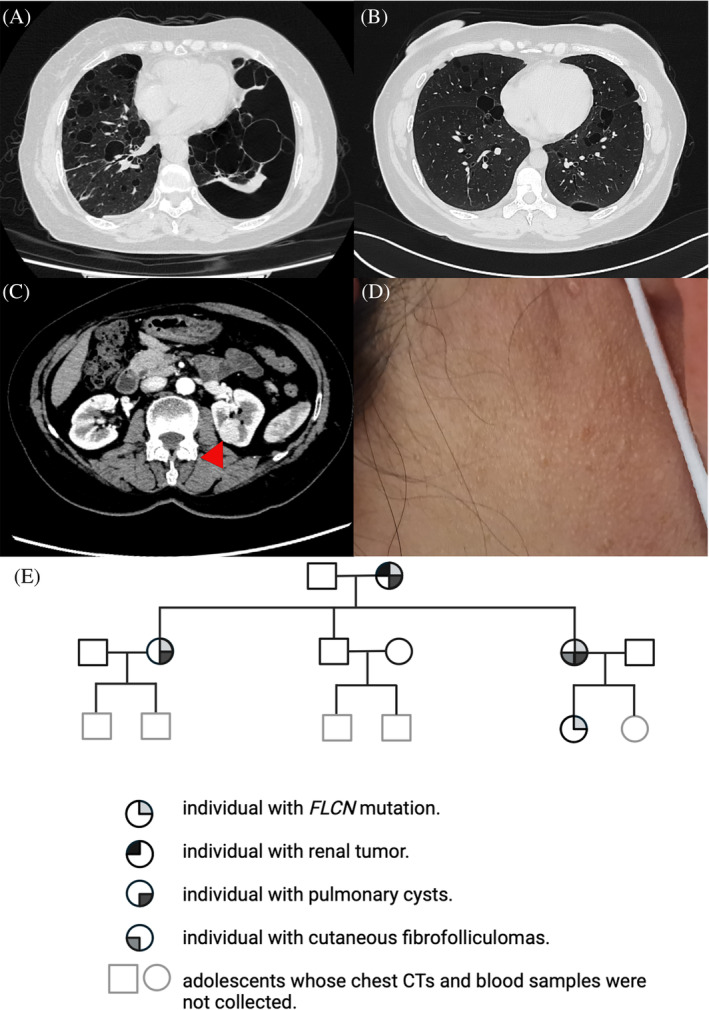
(A, B) Chest computed tomography (CT) scans of the patient and her daughter reveal multiple thin‐walled parenchymal cysts. (C) Enhanced abdominal CT of the patient indicates a left renal tumour, referred by a red arrow. (D) Shows cutaneous fibrofolliculomas observed in the daughter's neck. (E) Pedigree chart of the patient's family in three generations.
DISCUSSION
BHD was first described in 1977 and is caused by germline mutations in the tumour suppressor gene, FLCN. Patients with BHD are predisposed to develop multiple pulmonary cysts and/or spontaneous pneumothorax, fibrofolliculomas, and renal neoplasms. Among these, the respiratory system is the most frequently affected in China, with 92.4% of the cohort manifesting lung cysts, 6 consistent with prior reports in Japanese and Korean families. 7 A study by Furuya et al. investigated the clinicopathological features of 19 Asian families and reported that 89% had pulmonary cysts, 29% had fibrofolliculomas, and 20.0% had renal cell carcinomas. An analysis of 51 American families with BHD revealed that 90.2% of patients had fibrofolliculomas, and 88.2% had pulmonary cysts. A study involving 89 patients with diffuse cystic lung disease reported a 93% diagnosis of BHD by proficient radiologists based solely on imaging features on high‐resolution computed tomography (HRCT), indicating a better detection of BHD by HRCT. 8 The advent of HRCT has provided a significant adjunct for the differential diagnosis of diffused cystic lung diseases and distinguishing them from emphysema.
Pulmonary cysts are seen in a broad spectrum of disorders. Usually, when a patient presents with pulmonary cysts, differential diagnosis based on chest CT scans involves four steps, referring to their location (sub‐pleural or parenchymal), number (solitary or diffuse), and associated CT findings (nodules or ground glass opacities). 9 In brief, sub‐pleural cysts are usually caused by bullae and paraseptal emphysema; solitary parenchymal cysts could be incidental cysts, pneumatoceles, or bronchogenic cysts; multifocal or diffuse lung parenchymal cysts are differentiated among BHD, LAM, PLCH; Cysts associated with discrete lung nodules may be seen with LIP, amyloidosis, or light chain deposition disease (LCDD) and PLCH. Cysts associated with diffuse ground glass are often caused by desquamative interstitial pneumonia (DIP) and pneumocystis jirovecii pneumonia (PJP). In addition, cystic metastasis should be suspected in patients presenting with multiple pulmonary cysts with a known malignancy, especially head and neck primary squamous carcinoma. 9 , 10
This patient reported here presented with multifocal or diffuse cysts detected in lung parenchyma on her chest CT scan, which need to be differentiated among BHD, LAM, PLCH, and LIP. Radiologic characteristics, including the cystic wall, distribution and size, and associated findings, help narrow down the differential diagnosis. 9 , 10 LAM appears with thin‐walled cysts distributed evenly without lobar predominance, the size is usually 2–10 mm in diameter. Nodules are seen in the early stages of PLCH and then cavitate to thin‐ or thick‐walled cysts, featuring irregular cystic spaces of varying sizes (2 mm or larger than 2 cm), relatively sparing in bases and costophrenic angles of the lung. LIP also presents with randomly distributed thin‐walled cysts, but may also be associated with ground‐glass attenuation and nodules. 11 Cysts caused by BHD are thin‐walled, distributed in peripheral and lower lung zones and along the mediastinum, with variable shape and size. 12 The typically characterized lung parenchymal cysts, incorporating repeated spontaneous pneumothorax, familial pulmonary cysts, and non‐smoking history, lead to BHD as our primary diagnosis.
Approximately 25%–35% of patients with BHD develop renal tumours, typically characterized by slow, bilateral, and multifocal growth. The median age for tumour diagnosis ranges from 46 to 50 years. 1 , 13 The risk of bilateral and multifocal tumours is elevated up to sevenfold in individuals with BHD. 14 The most prevalent histological types include mixed chromophobe oncocytic tumours (50%), chromophobe carcinomas (34%), and oncocytomas (9%). Less common variants include clear cell carcinoma and papillary renal cancer. 14 , 15 , 16 Renal cancer surveillance commencing at the age of 20 years is advisable for individuals with BHD to preserve their renal function, with lifelong follow‐up every 3–4 years. 1 Magnetic resonance imaging (MRI) is the preferred screening modality for minimizing radiation exposure. 11 No routine surveillance is commended for lung cysts and fibrofolliculoma, but a full skin examination could be conducted every 6–12 months for the potential risk of melanoma. Moreover, patients with LAM may also be affected by renal angiomyolipomas, around 30%–40% of women with sporadic LAM and almost 90% of women with LAM and underlying tuberous sclerosis complex. 17 , 18
FLCN is currently recognized as the sole pathogenic gene responsible for BHD. Despite racial differences in mutation spots between Asians and Caucasians, the FLCN hotspot mutation c.1285dup/del in exon 11 remains consistent across ethnicities. 19 A retrospective study comprising 221 cases from 120 families with BHD in the Chinese population reported c.1285dup/del in exon 11 as a mutation hotspot, accounting for 17.4% of 144 cases reported in detail, consistent with that reported in Caucasian populations. 6 , 20 The FLCN mutation detected in the patient reported here was c.57‐58del in exon 4, which is a rare mutation and was previously reported only in two Asian families and a few Caucasian families. 3 , 14 , 21 , 22 As for the autosomal dominant inherited manner of BHD, when a pathogenic mutation is detected in an index patient, FLCN‐mutation surveillance is recommended in at‐risk family members including parents, sibs, and offspring of a proband, other family members depend on whether proband's parents are affected. Usually, genetic counselling commences at age 20. 23 Consequently, clinical information was not collected from the paediatric individuals in the BHD‐affected family reported here. 1
In conclusion, BHD should be considered when patients with familial cystic lung disease, especially in non‐smokers. FLCN mutation should be evaluated. Renal cancer surveillance is also recommended in patients with BHD beginning at the age of 20 years.
AUTHOR CONTRIBUTIONS
GH: Conceptualization; resources; supervision; validation; writing – review & editing. JM: Writing – original draft; writing – review & editing. QG: Writing – original draft; writing – review & editing. ZW: Writing – original draft; writing – review & editing.
FUNDING INFORMATION
This research was supported by National High Level Hospital Clinical Research Funding (2022‐NHLHCRF‐LX‐01), Nonprofit Central Research Institute Fund of the Chinese Academy of Medical Sciences (No. 2020‐PT‐320‐001) and the Elite Medical Professionals Project of China‐Japan Friendship Hospital (No. ZRJY2021‐BJ08).
CONFLICT OF INTEREST STATEMENT
None declared.
ETHICS STATEMENT
The authors declare that appropriate written informed consent was obtained for the publication of this manuscript and accompanying images.
Miao J, Gao Q, Wang Z, Hou G. Familial pulmonary cysts: A clue to diagnose Birt–Hogg–Dubé syndrome: A case report and literature review. Respirology Case Reports. 2024;12(3):e01319. 10.1002/rcr2.1319
Jinrui Miao, Qian Gao, and Zilin Wang have contributed equally.
Associate Editor: Philip Eng
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- 1. Menko FH, van Steensel MA, Giraud S, et al. Birt‐Hogg‐Dubé syndrome: diagnosis and management. Lancet Oncol. 2009;10(12):1199–1206. [DOI] [PubMed] [Google Scholar]
- 2. Skolnik K, Tsai WH, Dornan K, Perrier R, Burrowes PW, Davidson WJ. Birt‐Hogg‐Dubé syndrome: a large single family cohort. Respir Res. 2016;17:22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Zhou W, Liu K, Xu KF, Liu Y, Tian X. Clinical and genetic comparison of Birt‐Hogg‐Dubé syndrome (Hornstein‐Knickenberg syndrome) in Chinese: a systemic review of reported cases. Int J Gen Med. 2022;15:5111–5121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Savatt JM, Shimelis H, Moreno‐De‐Luca A, et al. Frequency of truncating FLCN variants and Birt‐Hogg‐Dubé–associated phenotypes in a health care system population. Genet Med. 2022;24(9):1857–1866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Guo T, Shen Q, Ouyang R, Song M, Zong D, Shi Z, et al. The clinical characteristics of east Asian patients with Birt‐Hogg‐Dubé syndrome. Ann Transl Med. 2020;8(21):1436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Hu X, Zhang G, Chen X, Xu KF. Birt‐Hogg‐Dubé syndrome in Chinese patients: a literature review of 120 families. Orphanet J Rare Dis. 2021;16(1):223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lee JH, Jeon MJ, Song JS, Chae EJ, Choi JH, Kim GH, et al. Birt‐Hogg‐Dubé syndrome in Korean: clinicoradiologic features and long term follow‐up. Korean J Intern Med. 2019;34(4):830–840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Gupta N, Langenderfer D, McCormack FX, et al. Chest computed tomographic image screening for cystic lung diseases in patients with spontaneous pneumothorax is cost effective. Ann Am Thorac Soc. 2017;14(1):17–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Raoof S, Bondalapati P, Vydyula R, Ryu JH, Gupta N, Raoof S, et al. Cystic lung diseases: algorithmic approach. Chest. 2016;150(4):945–965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Lee KC, Kang EY, Yong HS, Kim C, Lee KY, Hwang SH, et al. A stepwise diagnostic approach to cystic lung diseases for radiologists. Korean J Radiol. 2019;20(9):1368–1380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Expert consensus on the diagnosis and management of Birt‐Hogg‐Dubé syndrome. Zhonghua Jie He He Hu Xi Za Zhi. 2023;46(9):897–908. [DOI] [PubMed] [Google Scholar]
- 12. Toro JR, Pautler SE, Stewart L, Glenn GM, Weinreich M, Toure O, et al. Lung cysts, spontaneous pneumothorax, and genetic associations in 89 families with Birt‐Hogg‐Dubé syndrome. Am J Respir Crit Care Med. 2007;175(10):1044–1053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Toro JR, Wei MH, Glenn GM, Weinreich M, Toure O, Vocke C, et al. BHD mutations, clinical and molecular genetic investigations of Birt‐Hogg‐Dubé syndrome: a new series of 50 families and a review of published reports. J Med Genet. 2008;45(6):321–331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Benusiglio PR, Giraud S, Deveaux S, Méjean A, Correas JM, Joly D, et al. Renal cell tumour characteristics in patients with the Birt‐Hogg‐Dubé cancer susceptibility syndrome: a retrospective, multicentre study. Orphanet J Rare Dis. 2014;9:163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Pavlovich CP, Walther MM, Eyler RA, et al. Renal tumors in the Birt‐Hogg‐Dubé syndrome. Am J Surg Pathol. 2002;26(12):1542–1552. [DOI] [PubMed] [Google Scholar]
- 16. Gupta S, Kang HC, Ganeshan D, Morani A, Gautam R, Choyke PL, et al. The ABCs of BHD: an in‐depth review of Birt‐Hogg‐Dubé syndrome. Am J Roentgenol. 2017;209(6):1291–1296. [DOI] [PubMed] [Google Scholar]
- 17. McCarthy C, Gupta N, Johnson SR, Yu JJ, McCormack F. Lymphangioleiomyomatosis: pathogenesis, clinical features, diagnosis, and management. Lancet Respir Med. 2021;9(11):1313–1327. [DOI] [PubMed] [Google Scholar]
- 18. Ryu JH, Moss J, Beck GJ, Lee JC, Brown KK, Chapman JT, et al. The NHLBI lymphangioleiomyomatosis registry: characteristics of 230 patients at enrollment. Am J Respir Crit Care Med. 2006;173(1):105–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Liu Y, Xu Z, Feng R, Zhan Y, Wang J, Li G, et al. Clinical and genetic characteristics of chinese patients with Birt‐Hogg‐Dubé syndrome. Orphanet J Rare Dis. 2017;12(1):104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Sattler EC, Syunyaeva Z, Mansmann U, Steinlein OK. Genetic risk factors for spontaneous pneumothorax in Birt‐Hogg‐Dubé syndrome. Chest. 2020;157(5):1199–1206. [DOI] [PubMed] [Google Scholar]
- 21. Furuya M, Yao M, Tanaka R, Nagashima Y, Kuroda N, Hasumi H, et al. Genetic, epidemiologic and clinicopathologic studies of Japanese Asian patients with Birt‐Hogg‐Dubé syndrome. Clin Genet. 2016;90(5):403–412. [DOI] [PubMed] [Google Scholar]
- 22. Rossing M, Albrechtsen A, Skytte AB, Jensen UB, Ousager LB, Gerdes AM, et al. Genetic screening of the FLCN gene identify six novel variants and a Danish founder mutation. J Hum Genet. 2017;62(2):151–157. [DOI] [PubMed] [Google Scholar]
- 23. Sattler EC, Feldman J, Mirzaa GM, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2024. https://www.ncbi.nlm.nih.gov/books/NBK1522/
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


