Abstract
In Ghana, most laboratory diagnoses of severe mycoses are based on histopathology findings due to inadequate availability of serology, culture, and molecular tests. The aim of this study was to evaluate the spectrum of mycoses diagnosed in Ghana. We retrospectively reviewed reports from 2012 to 2021 from three major pathology laboratories in Ghana to identify reports indicating the presence of fungal elements and diagnosis of a mycosis, then extracted demographic, clinical history, site of infection, stain(s), used and diagnosed mycosis details. Over the 10-year period, 107 cases were found. No apparent increasing and decreasing trend in the number of cases per year or in a period was observed. The age range of affected patients was from 4 to 86 years. Special stains for fungi were only used in 22 of 107 (20.6%) of cases. The most frequently affected site was the sino-nasal area (34%). Mycosis type was determined for 58 (54.2%) cases, comprising aspergillosis (21), candidiasis (14), dermatophytosis (6), mucormycosis (3), two cases each of chromoblastomycosis, histoplasmosis, eumycetoma, entomophthoromycosis, sporotrichosis, and Malassezia infection and a single case each of cryptococcosis and deep onychomycosis. Of the 53 (49.5%) cases with presumptive diagnosis data, only seven (13.2%) had a pre-biopsy suspicion of mycosis. There is a wide spectrum of mycoses in Ghana, including endemic mycoses not previously reported. Improving the use of special fungal stains could increase yield and mycoses identification. Laboratory diagnostic capacity needs enhancement to complement histopathology investigations with serology, culture, and molecular methods.
Keywords: epidemiology, Ghana, histopathology, mycosis, spectrum
Introduction
Mycoses have gained significant attention in recent times as a major cause of morbidity and mortality globally. The recent launch of the first-ever WHO Fungal Priority Pathogen List and the additions of some subcutaneous mycoses to the WHO Neglected Tropical Diseases list has been instrumental in championing efforts to improve the diagnosis of mycoses across the world.1, 2 Nonetheless, the epidemiology of mycoses in several countries in Africa has not been extensively described. In Ghana, the only attempt previously was a modelling survey that estimated about 4% of the Ghanaian populace is affected by major mycoses annually.3 The estimation limited its scope to the most common and severe mycoses comprising cryptococcal meningitis, Pneumocytis jirovecii pneumonia, disseminated histoplasmosis, invasive aspergillosis, chronic pulmonary aspergillosis, fungal asthma, invasive candidiasis, recurrent vulvovaginal candidiasis, oral and esophageal candidiasis, mucormycosis, tinea capitis, and fungal keratitis. Furthermore, there is no specific surveillance programme for any mycosis in Ghana. The diversity of the mycoses diagnosed in Ghana and their causative organisms is thus unclear. The present available data consist of case reports and small case series.3, 4
Furthermore, a diagnostic laboratory gap analysis study in Ghana reveals inadequate capacity and established that most laboratory diagnoses of mycoses are based on histopathology findings due to the absence of fungal culture and direct microscopy methods and contemporary antigen–antibody and molecular tests.3, 4 Moreover, histopathology is a critical requirement in confirming proven cases of mycoses. Although, histopathology cannot reliably identify all fungi to genus or species level, it can confidently classify some fungi such as Mucorales, Coccidioides, Paracoccidioides, Sporothrix, Histoplasma, and Blastomyces at least to family or genus level due to characteristic features.5 Analysing laboratory data is a common method of evaluating the epidemiology of infectious diseases and for mycoses, reviewing histopathology data is crucial, particularly in many African settings such as Ghana.3, 4, 6, 7 A review of histopathology reports has been previously used to evaluate the epidemiological parameters including prevalence and trends of single or spectrum of mycoses in some African countries such as Nigeria,8–10 Togo,11, 12 and recently Uganda.13 The aim of this study was to evaluate the spectrum of histologically diagnosed mycoses in Ghana.
Materials and methods
This was a multi-centre retrospective study conducted to review the histopathology reports of three major public histopathology laboratory services providers in Ghana, namely the Department of Pathology, Korle-Bu Teaching Hospital (KBTH); Cellular Pathology Division, Ghana Standard Authority (GSA) and J.M Wadhwani Department of Anatomical Pathology, 37 Military Hospital (37 MH) from 2012 to 2021 to identify reports indicating the presence of fungal elements (structures from fungi) or diagnosis of mycosis. These histopathology laboratories provide diagnostic services to the hospitals they are attached to as well as many hospitals from all regions across Ghana. KBTH is the premier and national referral hospital. The histopathology report records were manually reviewed at the KBTH and 37 MH. At the GSA, reports were electronically searched using the following keywords: fungi, fungal element(s), fungal bodies, hyphae, yeast, pseudohyphae, and spores. Secondarily, distinctive morphological appearance of fungal elements such as spherules, sclerotic/muriform/medlar/copper penny bodies and grains were looked for. All histology reports that indicate the presence of fungal elements and/or diagnosis of fungal infections were included irrespective of underlying condition or site of sample collection. Histology reports that mention fungal infection as a differential diagnosis and without reporting the presence of fungal elements were excluded. The age, gender, clinical history, site of infection, and clinical suspicion for mycosis were extracted from laboratory records. Additionally, the type of histological stains used, fungal elements or features seen, and mycoses diagnosed by examining pathologists were all extracted.
The study generally did not require full ethical clearance, as it only involved a review of laboratory records and no patients were involved, or no further analysis was conducted on archived tissue blocks. However, appropriate administrative permission was obtained from all three institutions involved in the study.
Data were statistically analyzed using the Statistical Products and Services Solutions (SPSS), version 25 (IBM Corp, Armonk, New York, USA). We summarized statistics of variables using frequencies, percentages, median and interquartile range where applicable. The distribution of cases according to specific years for each centre was analyzed.
Results
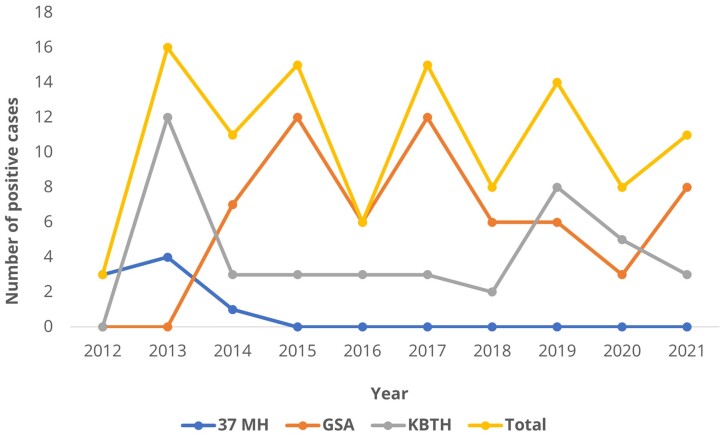
Over the 10-year study period from 2012 to 2021, we found 107 cases with histopathological reports revealing the presence of fungal elements or mycoses diagnosis (Supplementary Table 1). The cases comprised of 50 (46.7%) males and 57 (53.3%) females. Age ranged from 4 to 86 years with a median age of 41 years (interquartile range, 31–55). These comprised 91 deep mycoses (affecting deep-seated tissue and internal organs) and 16 superficial and muco-cutaneous mycoses. The number of cases per year among the three centres ranged from 3 to 16 with the highest number of cases recorded in 2013 (Fig. 1). There was no apparent increasing and decreasing trend in the number of cases observed over the 10-year period at all the three laboratories, except that no mycoses diagnosis was reported from 37 MH between 2015 and 2021. The majority (92.5%) of the cases were from the GSA and KBTH, accounting for 60 (56.1%) cases and 39 (36.4%) respectively.
Figure 1.
Mycoses diagnosed by histopathology from 2012 to 2021.
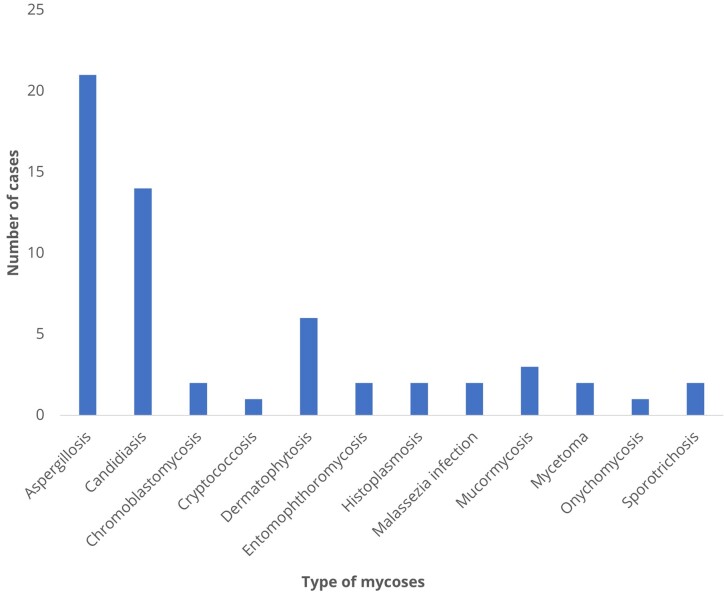
The determination of the type of mycosis was made for 58 (54.2%) cases (Fig. 2). The spectrum of mycosis was relatively broad and included aspergillosis (21 cases), candidiasis (14 cases), dermatophytosis (six cases), mucormycosis (three cases), and chromoblastomycosis, histoplasmosis, eumycetoma, entomophthoromycosis, sporotrichosis, and Malassezia infection (two cases each). The remaining mycoses implicated were cryptococcosis and onychomycosis, a single case each.
Figure 2.
Number of cases of different types of mycoses diagnosed.
Clinical information was available for 53 cases, but only seven (14.6%) had a clinical suspicion for mycosis showing that many of the cases were probably an unexpected diagnosis. Regarding the sites involved, the nose or nasal regions were the commonest with 34 cases mostly affected by aspergillosis (18 cases) and mucormycosis (three cases), followed by the gastrointestinal tract (19 cases), limbs (17 cases), and the skin (seven cases); other body sites had less than five cases (Supplementary Table 1).
A major observation made in this study was that special fungal stains were rarely used. Special stains used included periodic acid-Schiff (PAS) (16 cases), Grocott-Gomori methenamine silver (GMS) (three cases), and an unspecified special stain (three cases) with the remaining 85 cases diagnosed on haematoxylin and eosin (H&E) alone. There were both regular fungal morphologies (yeasts and hyphae) and special fungal structures such as sclerotic bodies (Figs. 3 and 4). Generally, the majority (87 cases) of the fungal structures detected was not adequately described with several scanty descriptions such as fungal bodies, fungal cysts, fungal elements, hyphae, and yeast. This observation was consistent among the three centres.
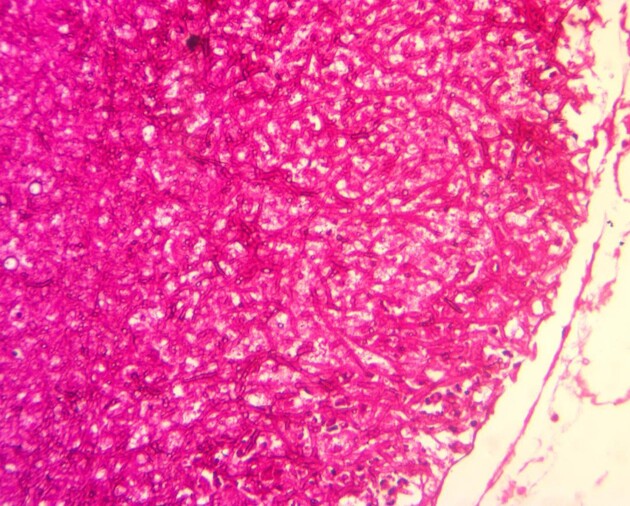
Figure 3.
H&E-stained section of a left antral mass from a 55-year-old female with epistaxis and chronic headache showing regular acute branching dichotomous septate hyphae typical of Aspergillus species.
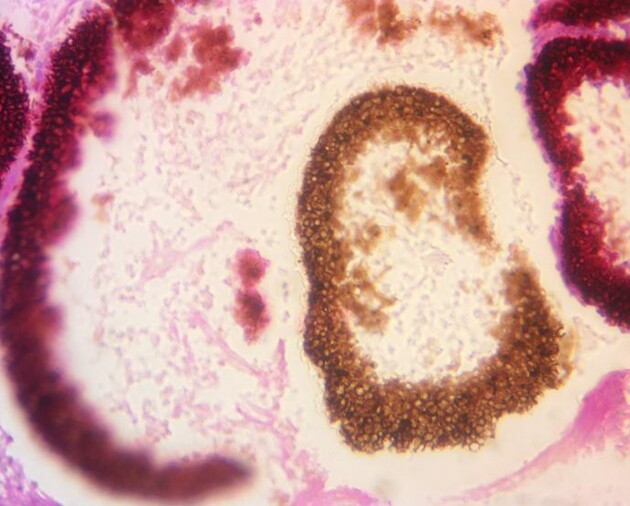
Figure 4.
PAS-stained section of a left foot infected ganglion from a 49-year-old male showing a ring of aggregates of pigment-producing and double-contoured spherical structures known as sclerotic bodies which are consistent with chromoblastomycosis.
Discussion
The study highlights the spectrum of mycoses diagnosed by histopathology in Ghana. Over the 10-year study period, from 2012 to 2021, there were 107 histopathology-diagnosed mycoses recorded, including 91 deep mycoses among the three histopathology laboratories. There were diverse mycoses reported and this finding offers a remarkable narrative about the epidemiology of mycoses in Ghana, particularly deep and endemic mycoses. This study reveals a broader spectrum of mycoses in Ghana including some uncommonly reported and endemic mycoses.3 These include mostly subcutaneous mycoses such as eumycetoma, chromoblastomycosis, sporotrichosis, and entomophthoromycosis. In a review of mycetoma in West Africa, there was no indigenous case reported from Ghana.14 Only two studies have previously reported entomophthoromycosis, specifically, basidiobolomycosis, in Ghana.15, 16 Enhanced immigration and international travel are likely to further increase the diversity of mycoses in the future.7, 14 The nasal region was the site frequently diagnosed with deep mycosis, probably because samples from these areas are relatively easy to collect in comparison to sampling internal organs such as lungs where most severe mycoses originate or occur.
The fact that most of the cases were seen at the KBTH and GSA is probably due to long-serving pathology consultants with at least 30 years of experience supporting those centres. Additionally, these centres process samples from several private and public hospitals within and outside the Greater Accra region and from all regions of the country, unlike the 37 MH which seldom receives outside samples.
The use of fungal stains is strongly recommended by guidelines and experts to improve the detection of fungal elements or structures in tissues during histopathological analysis.5, 17 In the present study, we observed that major fungal stains, that is, PAS and GMS were rarely used. Other special fungal stains such as Alcian blue (commonly for Cryptococcus spp.), mucicarmine (commonly for Cryptococcus spp.,15Blastomyces spp. and Rhinosporidium spp.), and Fontana-Masson (mainly for dematiaceous fungi) were not used at all.18 Attempting to diagnose mycoses with only H&E as noted in 79% cases of the study, is generally difficult and frequently associated with reduced sensitivity. Although fungi can be seen with conventional H&E staining, special stains enhance the detection of fungi in tissues. These stains also allow for the narrowing identification of possible aetiologies and aid in preliminary diagnosis. The diverse ways of describing and reporting fungal structures including the use of broad descriptions such as yeasts, hyphae, fungal bodies, and fungal elements offer limited diagnostic benefits to enable appropriate clinical decisions. Otherwise, a detailed description of fungal structures or elements could provide hints to clinicians on likely aetiologies which could play an important role in making treatment decisions.
In the era of growing antifungal resistance and expansion of rare species of moulds and yeasts as causative agents of mycoses, accurate identification of aetiological agents of infections has become more important now than ever. Identifying the right cause of infection allows for the right choice of antifungal medications to ensure the right treatment for better clinical outcomes. In view of this, in the absence of routine fungal culture, proceeding with further molecular investigations on tissue blocks in which histopathology analysis demonstrated the presence of fungal elements will go a long way to influence drug options while providing data on emerging or rare species causing human infections.19, 20 This is extremely important in the Ghanaian clinical setting particularly considering the critical role of histopathology in aiding diagnosis and in most instances the commonly available means of diagnosis.4 Despite the common use of molecular methods on formalin-fixed-paraffin-embedded to aid species-level identification in many clinical mycology laboratories, the methodology or procedures of available assays are not extensively standardized and not available in many African settings.21–25
Overall, the study retrospectively evaluated histopathologically diagnosed mycoses in Ghana. The findings highlight a possible underdiagnosis, low index of clinical suspicion and diverse mycoses including a few not previously reported. There is a need to improve awareness among clinicians, sensitize pathologists, support histopathology analysis with molecular assays for accurate identification of fungal pathogens and explore antigen–antibody tests and cultures. Finally, prospective epidemiological studies are recommended to better appreciate the burden of the major mycoses outlined particularly fungal rhinosinusitis.
Limitations
A drawback of the study is that patients’ medical records could not be accessed to obtain relevant symptoms, detailed clinical history and examination findings, findings from other laboratory and medical imaging investigations, final diagnosis, management approach, and outcomes to confirm if the presence of fungi in histological samples truly indicated infection and not colonization or contamination. Additionally, the tissue blocks and sections were not further examined to confirm the identity of the genus or species of fungi reported during histological examination.
Supplementary Material
Acknowledgement
The authors are grateful to the Heads of the three Pathology Laboratories involved in the study: Dr Afua Abrahams, Prof. Agyeman Badu Akosa, and Lt Col (Dr) Frederick Hobenu. We are extremely appreciative of the support of the technical staff of the Histopathology Laboratories especially Andrew Hooper, Joseph Nyadia, and Emmanuel Ametepe (Korle-Bu Teaching Hospital), Felix Manu (Ghana Standards Authority), and Irene Padi (37 Military Hospital) for their varied roles in supporting the identification of positive cases. The contributions of Benjamin Otoo (University of Wisconsin, Madison, USA) and Pious Appiah (University of Ghana Medical School) in assisting with the manual review of laboratory reports at the KBTH are duly acknowledged.
Contributor Information
Bright Ocansey, Division of Evolution, Infection and Genomics, Faculty of Biology, Medicine and Health, University of Manchester, Manchester Academic Health Science Centre, Manchester, M13 9NT, UK.
Isaac Erskine, Department of Pathology, Korle-Bu Teaching Hospital and University of Ghana Medical School, Korle-Bu, GA-221-1570, Ghana.
Leonard Okine, Cellular Pathology Division, Ghana Standard Authority, Accra, GA-288-5605, Ghana.
Daniel Potakey, Department of Pathology, Korle-Bu Teaching Hospital and University of Ghana Medical School, Korle-Bu, GA-221-1570, Ghana.
Prince Pappoe-Ashong, Department of Medical Microbiology, University of Ghana Medical School, Korle-Bu, GA-270-4330, Ghana.
Isaac Sraku, Department of Medical Microbiology, University of Ghana Medical School, Korle-Bu, GA-270-4330, Ghana.
Solomon Quayson, Department of Pathology, Korle-Bu Teaching Hospital and University of Ghana Medical School, Korle-Bu, GA-221-1570, Ghana.
Japheth Opintan, Department of Medical Microbiology, University of Ghana Medical School, Korle-Bu, GA-270-4330, Ghana.
Chris Kosmidis, Division of Evolution, Infection and Genomics, Faculty of Biology, Medicine and Health, University of Manchester, Manchester Academic Health Science Centre, Manchester, M13 9NT, UK; National Aspergillosis Centre, Manchester University NHS Foundation Trust, Manchester, M23 9LT, UK.
David Denning, Division of Evolution, Infection and Genomics, Faculty of Biology, Medicine and Health, University of Manchester, Manchester Academic Health Science Centre, Manchester, M13 9NT, UK.
Author contributions
Bright Ocansey (Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Visualization, Writing – original draft), Isaac Erskine (Data curation, Investigation, Methodology, Validation, Visualization, Writing – review & editing), Leonard Okine (Data curation, Formal analysis, Investigation, Methodology, Resources, Writing – review & editing), Daniel Potakey (Data curation, Investigation, Methodology, Resources, Writing – review & editing), Prince Pappoe-Ashong (Data curation, Investigation, Methodology, Resources, Writing – review & editing), Isaac Sraku (Data curation, Investigation, Methodology, Writing – review & editing), Solomon Quayson (Data curation, Investigation, Methodology, Resources, Writing – review & editing), Japheth Opintan (Investigation, Methodology, Project administration, Supervision, Writing – review & editing), Chris Kosmidis (Conceptualization, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – review & editing), and David Denning (Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Supervision, Validation, Writing – review & editing)
Funding
This study was supported by the CARIGEST SA as part of the ‘Epidemiology of Invasive Fungal Infections in Ghana/West Africa’ research award to D.D. which includes a studentship award to B.O. CARIGEST SA did not play any role in the design, implementation, and analysis of the study.
Declaration of interest
The authors declare no conflict of interest.
References
- 1. Hay R, Denning DW, Bonifaz A et al. The diagnosis of fungal neglected tropical diseases (Fungal NTDs) and the role of investigation and laboratory tests: an expert consensus report. Trop Med Infect Dis. 2019;4: 122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. WHO. WHO fungal priority pathogens list to guide research, development and public health action. 2022. Accessed December 30, 2022, https://www.who.int/publications-detail-redirect/9789240060241. [Google Scholar]
- 3. Ocansey BK, Pesewu GA, Codjoe FS, Osei-Djarbeng S, Feglo PK, Denning DW. Estimated burden of serious fungal infections in Ghana. J Fungi. 2019; 5: 38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ocansey BK, Dadzie EA, Eduful SK et al. Improving awareness, diagnosis and management of invasive fungal infections in Ghana: establishment of the Ghana Medical Mycology Society. Med Mycol. 2022; 60: myac069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Donnelly JP, Chen SC, Kauffman CA et al. Revision and update of the consensus definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin Infect Dis. 2020; 71: 1367–1376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Driemeyer C, Falci DR, Oladele RO et al. The current state of clinical mycology in Africa: a European Confederation of Medical Mycology and International Society for Human and Animal Mycology survey. Lancet Microbe. 2022; 3(6): 464–470. 10.1016/S2666-5247(21)00190-7. [DOI] [PubMed] [Google Scholar]
- 7. Africa diagnostic reports—Gaffi . Gaffi—Global action for fungal infections. November 22, 2022. Accessed November 27, 2022. https://gaffi.org/africa-diagnostic-reports/.
- 8. Khalil M, Ekanem IO, Gugnani HC, Attah EB. Some deep mycoses diagnosed by histopathology in South Eastern Nigeria. Rev Iberoam Micol. 1999; 16: 221–224. [PubMed] [Google Scholar]
- 9. Ngwu BAF, Oluwasola AO, Iyare FE, Ogunbiyi JO, Akang EE. Epidemiology of histopathologically diagnosed mycoses: the Ibadan 37 years experience. J Adv Med Medical Res. 2015; 10: 1–9. 10.9734/BJMMR/2015/19301. [DOI] [Google Scholar]
- 10. Onuigbo WI, Gugnani HC. Deep mycoses prevalent in the igbos of Nigeria. Int J Dermatol. 1976; 15: 432–437. [DOI] [PubMed] [Google Scholar]
- 11. Darré T, Saka B, Mouhari-Toure A et al. Mycetoma in the Togolese: an update from a single-center experience. Mycopathologia. 2018; 183: 961–965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Darré T, Saka B, Mouhari-Touré A et al. Histoplasmosis by histoplasma capsulatum var. Duboisii observed at the Laboratory of Pathological Anatomy of Lomé in Togo. J Pathogens. 2017; 2017: 2323412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Kwizera R, Bongomin F, Lukande R. Deep fungal infections diagnosed by histology in Uganda: a 70-year retrospective study. Med Mycol. 2020; 58: 1044–1052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Oladele RO, Ly F, Sow D et al. Mycetoma in West Africa. Trans R Soc Trop Med Hyg. 2021; 115: 328–336. [DOI] [PubMed] [Google Scholar]
- 15. Sackey A, Ghartey N, Gyasi R. Subcutaneous basidiobolomycosis: a case report. Ghana Med J. 2017; 51: 43–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Rodrigues O, Commey JOO. Basidiobolomycosis in Ghanaian children. Tropical Doctor. 1994; 24(4): 170–171. [DOI] [PubMed] [Google Scholar]
- 17. Denning DW, Kibbler CC, Barnes. RA “British Society for Medical Mycology proposed standards of care for patients with invasive fungal infections. Lancet Infect Dis. 2003;3: 230–240. [DOI] [PubMed] [Google Scholar]
- 18. Walsh TJ, Hayden RT, Larone DH. Larone's Medically Important Fungi: A Guide to Identification. John Wiley & Sons, 2018 Jun 1. [Google Scholar]
- 19. Valero C, Martín-Gómez MT, Buitrago MJ. Molecular diagnosis of endemic mycoses. J Fungi (Basel). 2022;9: 59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Sparks R, Halliday C, Chen SCA. Panfungal PCR on formalin fixed paraffin embedded tissue – to proceed or not proceed?. Pathology (Phila). 2023; 55: S46. [DOI] [PubMed] [Google Scholar]
- 21. Valero C, de la Cruz-Villar L, Zaragoza Ó, Buitrago MJ. New panfungal real-time PCR assay for diagnosis of invasive fungal infections. J Clin Microbiol. 2016; 54: 2910–2918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Buitrago M, Bernal-Martínez L, Castelli M, Rodriguez-Tudela J, Cuenca-Estrella M. Performance of panfungal-and specific-PCR-based procedures for etiological diagnosis of invasive fungal diseases on tissue biopsy specimens with proven infection: a 7-year retrospective analysis from a reference laboratory. J Clin Microbiol. 2014; 52(5): 1737–1740. 10.1128/JCM.00328-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Buitrago MJ, Aguado JM, Ballen A et al. Efficacy of DNA amplification in tissue biopsy samples to improve the detection of invasive fungal disease. Clin Microbiol Infect. 2013; 19: E271–277. [DOI] [PubMed] [Google Scholar]
- 24. Larkin PMK, Lawson KL, Contreras DA et al. Amplicon-based next-generation sequencing for detection of fungi in Formalin-fixed, paraffin-embedded tissues: correlation with histopathology and clinical applications. J Mol Diagn. 2020; 22:1287–1293. [DOI] [PubMed] [Google Scholar]
- 25. Lysen C, Silva-Flannery L, Zaki SR, Gary JM, Lockhart SR. Performance evaluation of fungal DNA PCR amplification from formalin-fixed paraffin-embedded tissue for diagnosis: experience of a tertiary reference laboratory. Mycoses. 2021; 64: 603–611. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.