Abstract
Purpose:
Frailty is not commonly assessed on intake to cardiac rehabilitation (CR), but screening could enable targeted interventions and potentially reduce secondary complications. This study aimed to develop and retrospectively examine the feasibility of utilizing a CR-specific algorithm based on the Clinical Frailty Scale (CFS). Our CFS-CR algorithm endeavoured to screen for frailty in older adults (> 65 y) entering CR following cardiac surgery/procedure.
Method:
The charts of 30 former patients (mean age: 74.0 ± 6.9 y) were examined by a clinician working in CR.
Results:
The clinician was unable to score any of the patients based on their medical charts using the CFS-CR due to insufficient data. Documentation was typically limited in the areas of instrumental and basic activities of daily living whereas exercise data were readily available.
Conclusions:
Current intake documentation in CR limited the ability to retrospectively screen for frailty. This finding suggests a need for a frailty-specific tool to support routine clinical screening. Prospective evaluation of the CFS-CR is warranted to further examine the clinical utility of the algorithm during CR intake assessments.
Key Words: adult, algorithms, cardiac rehabilitation, frailty, geriatric assessment
Résumé
Objectif:
la fragilité est peu évaluée à l’admission en réadaptation cardiaque (RC), mais le dépistage pourrait permettre de cibler des interventions et peutêtre de réduire les complications secondaires. La présente étude visait à créer un algorithme de RC d’après l’échelle de fragilité clinique (ÉFC) et à procéder à une analyse rétrospective pour déterminer la faisabilité de l’utiliser. L’algorithme ÉFC-RC était conçu pour dépister la fragilité chez les personnes âgées (de 65 ans ou plus) qui arrivaient en RC après une opération ou une intervention cardiaque.
Méthodologie :
une clinicienne qui travaillait en RC a examiné les dossiers de 30 anciens patients (âge moyen de 74,0 ± 6,9 ans).
Résultats :
la clinicienne n’a pu mesurer les résultats d’aucun patient d’après leur dossier médical au moyen de l’ÉFC-RC en raison de données insuffisantes. Les éléments du dossier se limitaient généralement aux activités déterminantes et courantes de la vie quotidienne, tandis que les données sur les exercices étaient facilement accessibles.
Conclusions :
l’information contenue dans les dossiers d’admission actuels en RC limitait la possibilité de procéder à l’analyse rétrospective de la fragilité. Cette observation laisse croire à la nécessité de concevoir un outil axé sur la fragilité pour contribuer au dépistage clinique systématique. Une évaluation prospective de l’ÉFC-RC s’impose pour mieux analyser l’utilité clinique de l’algorithme lors des évaluations à l’admission en RC.
Mots-clés : adulte, algorithmes, évaluation gériatrique, fragilité, réadaptation cardiaque
Frailty, considered a geriatric syndrome, is a state of reduced physiological reserve and increased vulnerability after a stressor event. It has been associated with an increased risk of adverse outcomes such as falls, delirium, and disability.1–4 Several methods of frailty measurement have been suggested, one of the most common is Fried’s criteria for identifying frailty, which includes five phenotypic characteristics: unintended weight loss, exhaustion, weakness, slow gait speed, and low physical activity.1 Individuals who are frail are more likely than non-frail patients to have comorbidities such as diabetes, chronic obstructive pulmonary disease (COPD), cardiovascular disease (CVD), and present with an increased dependence on the health care system.5
Frailty is reported in up to 50% of older adults with CVD and those undergoing cardiac procedures, including surgery.4,6 Not only do individuals with CVD often have multiple comorbidities, they experience reduced exercise capacity and skeletal muscle dysfunction, which contribute to the development of frailty.4,7 Studies suggest a bidirectional link between CVD and frailty,6,8 as they share common pathophysiological mechanisms related to chronic inflammation.8
In addition, frailty is associated with increased adverse outcomes in the cardiac surgery population including mortality,5,9,10 institutionalization,5 poor quality of life (QOL),9 and increased length of hospital stays.4,11 Functionally, frailty in older adults is correlated with disabilities in activities of daily living (ADLs), such as dressing, bathing, and eating. It is also correlated with instrumental activities of daily living (IADLs), such as cooking, shopping, and doing housework.1,12 A systematic review provided strong evidence that following a cardiac procedure, frailty predicted mortality at six months or later, functional decline, and poor QOL.9 Therefore, measuring frailty along the spectrum of care after cardiac surgery/ procedure could be important in identifying those at risk for poor outcomes.
Following cardiac surgery/procedure, referral to cardiac rehabilitation (CR) is the standard of practice for secondary prevention.13 CR is a multidisciplinary program that aims to reduce CVD risk with individualized, multi-component exercise interventions, nutrition and health coaching, education, and behaviour/lifestyle modifica-tion.14 CR has been shown to significantly improve aerobic capacity15 and QOL,15 and reduce hospitalizations and cardiovascular mortality.16,17 Similarly, the benefits of exercise for individuals with frailty include increased aerobic capacity,18,19 muscular strength,20 power, and functional status.20 As well, exercise reduces frailty levels as measured with various frailty tools such as the Short Physical Performance Battery, Fried’s criteria, Cardiovascular Health Study criteria, the Frailty Index, and the Clinical Frailty Scale (CFS).21 A recent study found that individuals with CVD improved in frailty scores upon completing CR.22 Importantly, individuals with greater frailty at baseline may derive the greatest benefit from CR, making it useful to identify frailty on entry to CR.22
Despite the link between CVD and frailty, frailty is not routinely assessed or characterized in CR programming or at intake to CR.23 A recent study highlighted the importance of evaluating the feasibility of adding frailty assessments to CR intake as well as testing their prognostic ability.23 Easy-to-use, rapid, standardized tools, such as the CFS, have been recommended as an instrument to identify and screen for frailty in older adults.2,23,24
In 2005, the Canadian Study of Health and Aging developed a 7-point CFS to assess frailty in older adults. This scale ranged in categories from Very Fit (1) to Severely Frail (7). Individuals with higher scores generally had more comorbidities and impaired mobility.2 The CFS was later expanded to include two clinically distinct groups: Very Severely Frail (8) and Terminally Ill (9). A score of 5 (Mildly Frail) and greater is considered frail.2,25 The CFS shows good interrater reliability (interclass correlation coefficient 0.97, p < 0.001), good criterion (area under the receiving operator characteristic curve 0.70 for death and 0.75 for entry into an institution), and construct validity (Pearson coefficient 0.80, p < 0.01), and each of the category increments were related to increased risk of death and entry into institutional care.2 This category-based scale with a brief description and visual depiction of each score aims to support clinical judgment of frailty level in older adults with a broad range of health care needs.2 The CFS is a promising tool as it requires no additional training or staff, little time, and no specialized equipment to complete.2 It has been widely used in multiple settings with older adults, particularly with in-patient geriatric medicine and cardiology, and has also been used to predict outcomes such as mortality, comorbidity, and functional decline.26
The CFS requires some clinical judgment, and most studies have included trained assessors or geriatricians utilizing the tool to determine frailty levels.27,28 Several studies have attempted to further simplify the application of the CFS by operationalizing the scale;29 modifying it into four combined categories;30 creating an algorithm for use with geriatric in-patients;28 and creating a step-by-step telephone interview tool accompanied by a visual decision tree.31 Most of these modified CFS tools and algorithms were developed for use in acute-care settings with specialized populations, making it difficult to extrapolate findings to an outpatient post-cardiac surgery/procedure population. No algorithm has emerged as the clinical standard, and to our knowledge none have been developed for post-cardiac surgery/procedure populations entering CR.
The purpose of this study was to address this gap by developing an algorithm based on the CFS; namely, the Clinical Frailty Scale-Cardiac Rehabilitation (CFS-CR). The algorithm could support clinical judgment in screening for frailty in the post-cardiac surgery/procedure population that is 65 years of age and older, and entering an outpatient CR program. Early frailty screening in this population may allow for the further individualization of exercise programs to target specific components contributing to frailty, decreasing the incidence of frailty-related health care complications, and improving QOL.32,33 The secondary objective was to retrospectively examine the feasibility of applying the CFS-CR to the participant data collected during CR intake assessment.
Methods
Development of the CFS-CR
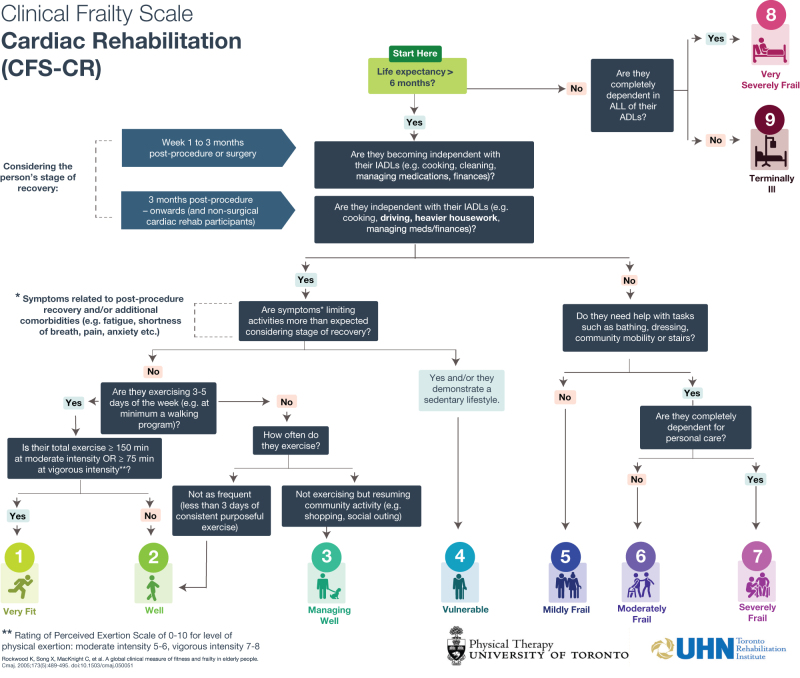
We aimed to develop an algorithm that places the established CFS descriptors of levels of frailty into the context of CR participants. Our CFS-CR algorithmic tool would inform clinical judgment when administering the CFS in a CR setting, inclusive of post-surgical and post-procedure patients (valve repair/replacement, coronary artery bypass graft [CABG], transcatheter aortic valve implantation [TAVI], percutaneous coronary intervention [PCI], across the spectrum of recovery.) (See Figure 1.) The process of developing a scale consisted consists of three phases: item development, scale revision, and scale evaluation.34 The CFS-CR aims to adapt the established scale, the CFS, into a condition-specific algorithm for use in CR. The guiding principle during algorithm development was to keep the wording and differentiation of frailty categories in line with the original CFS, adding only further definitions that would help apply the CFS to assessments in a CR setting.
Figure 1.
The Clinical Frailty Scale-Cardiac Rehabilitation (CFS-CR) Algorithm.
On examining the scale, we determined that defining the level of frailty based on life expectancy was an appropriate starting point for the CFS-CR algorithm. Information regarding life expectancy placed individuals with a prognosis of less than six months in these CFS categories:
8 = Very Severely Frail, and 9 = Terminally Ill. Commonly, individuals entering CR have a prognosis of greater than six months and therefore, this directs clinicians specifically to categories 1 to 7.4
We then reviewed the CFS definition of ADLs and IADLs that direct decision-making regarding categories 5 to 7.2,35,36 it is important to consider that many individuals entering CR have activity restrictions after cardiac surgery involving median sternotomy and graft donor sites that would limit their ability to perform IADLs. These restrictions include no heavy lifting, no driving, and no heavy housework; they are usually followed for four to eight weeks or longer, depending on the procedure and cardiac surgeon cardiac surgeon guidelines.37 To account for these restrictions, the second level of the algorithm proposes that the clinician consider the individual’s stage of recovery and activity restrictions. The CFS-CR proposes assessment of participation in IADLs based on the length of time after surgery; namely, “1 week to 3 months post-surgery/procedure” or “3 months post-surgery/procedure and after.” This takes activity restrictions into account and allows for frailty screening of individuals entering CR at different points in their recovery.
Next, we further defined the descriptor in category 4 “symptoms limit activities” as those related to postoperative recovery and/or additional comorbidities; that is, experiencing fatigue, shortness of breath, and/or anxiety. These represent common activity-limiting symptoms in individuals with a cardiac history.38 Aligned with the CFS, those with symptoms limiting their activity would be scored in category 4 (Vulnerable), and those not limited would be scored in category 3 (Managing Well) or above in category 1 (Very Fit).
To differentiate between categories 1, 2, and 3, the frequency and duration of exercise were expanded in the context of CR definitions of “exercise” and “activity.” Exercising “regularly” in category 1 (Very Fit) and exercising “occasionally, e.g., seasonally” in category 2 (Well) were the original descriptors in the CFS.2 To differentiate between categories 1 and 2, we utilized the American College of Sports Medicine (ACSM) guidelines for aerobic exercise in older adults, which are the accepted guidelines used at our CR site (exercising 3–5 d/wk, with total exercise time3 150 min of moderate intensity exercise or3 75 min of vigorous intensity).39,40 We used the Perceived Exertion Scale rating of 0 to 10 for level of physical exertion, with moderate intensity rated as 5 or 6, and vigorous intensity as 7 or 8.39,40 Those who met ACSM guidelines would be scored in category 1 (Very Fit). Those who did not meet the requirements but were generally active and participating in less than three days of consistent purposeful exercise would be classified as category 2 (Well). Category 3 (Managing Well) in the CFS-CR includes those who were not exercising but were resuming general community activities (e.g., shopping, social outings); this aligns with the original CFS descriptor of individuals “not regularly active beyond routine walking.”2 All decisions regarding the development of the algorithm and delineation of categories in the CFS-CR were made through team discussions and approved by the senior author (TJFC). The study team consisted of research scientists and experienced clinicians practising in a large urban CR programme in Toronto, Canada with expertise in nursing and rehabilitation.
Study design
A previous study explored the application of the CFS retrospectively by reviewing electronic medical records and charts.27 Accordingly, we completed a retrospective chart review using patient data collected during intake assessments on entry to CR to examine the feasibility of applying the CFS-CR. Specifically, we aimed to explore if the CFS-CR could be used to screen for frailty based on the patient information that was currently collected during CR intake assessments of patients after cardiac procedures. Thirty participant charts for individuals entering the programme between 2019 and 2020 who met inclusion criteria were included in this study. Charts from participants who met the following inclusion criteria were reviewed: ≥65 years old or older and who had undergone a cardiac surgical procedure, either a valve repair/replacement, CABG, TAVI, or PCI. These patients were included as they have been shown to have a higher incidence of frailty in CR populations.5,25,41
This study was approved by the University Health Network/Toronto Rehabilitation Research Ethics Board (REB#19-6098).
Retrospective chart review
A nurse clinician who participates in intake assessments and uses the electronic and paper chart system performed the chart review. The clinician did not receive any additional training in applying the CFS-CR and rated the patient’s level of frailty using information contained in the initial CR intake assessment. Additionally, we developed a checklist of the information required to complete the CFS-CR to determine what information was available in the patient intake assessment to inform the completion of the algorithm. The clinician reviewed the paper and electronic charts of 30 patients enrolled in CR using the CFS-CR and the checklist. The clinician conducting the review documented comorbidities after reviewing each patient’s past medical history and then added the total number. The reviewer completed the checklist and provided a frailty score for each patient or indicated the level of the algorithm at which they were unable to continue due to a lack of information.
Statistical analysis
We performed descriptive statistics by expressing continuous variables as mean ± standard deviation (e.g., age, d from procedure to intake). Categorical variables were expressed as frequencies and percentages (e.g., sex, diagnosis, procedure, comorbidities). We reported the availability of relevant data in the patient charts to inform the CFS-CR as a percentage of charts reviewed. Analysis was conducted using IBM SPSS Statistics, version 26.0 (IBM Corporation, Amonk, NY).
Results
Figure 1 depicts the CFS-CR algorithm we developed to address aim 1, which entailed creating an algorithm based on the CFS to support screening for frailty. The CFS-CR is proposed to screen for frailty in patients (≥ 65 y) after a cardiac surgery/procedure. Aim 2 was to examine the feasibility of applying the CFS-CR to current CR intake assessments retrospectively. Thirty patient charts were reviewed with equal representation of males and females, and a mean age of 74.0 ± 6.9 years. Patients completed CR intake an average of 107.9 ± 61.2 days after the surgery/ procedure (min-max 31-236 days). The most common diagnoses were coronary artery disease (CAD) (n = 10; 33.3%) and valvular heart disease (VHD) (n = 8; 26.7%) (Table 1).
Table 1.
Baseline Characteristics of Participants in the Chart Review
| Mean | Standard deviation | Range | |
|---|---|---|---|
| Age (y) | 74.0 | 6.9 | 65-88 |
| Time from procedure to intake (d) | 107.9 | 61.2 | 31-236 |
| Categories | Percentage (%) | ||
| Sex | Male | 50 | |
| Female | 50 | ||
| Diagnosis | CAD | 33.3 | |
| VHD | 26.7 | ||
| CAD + Other | 16.7 | ||
| VHD + Other | 13.3 | ||
| CAD + VHD | 6.7 | ||
| Other | 3.3 | ||
| Procedure | CABG | 50.0 | |
| 1 CABG | 13.3 | ||
| 2 CABGs | 16.7 | ||
| 3 CABGs | 13.3 | ||
| 4 CABGs | 6.7 | ||
| Transcatheter aortic valve implantation | 16.7 | ||
| Aortic valve replacement | 16.7 | ||
| Mitral valve replacement | 6.7 | ||
| CABG + Other | 10.0 | ||
| Comorbidities | 0 | 10.0 | |
| 1 | 13.3 | ||
| 2 | 30.0 | ||
| 3 | 33.3 | ||
| 4 or more | 13.3 | ||
CAD = coronary artery disease; VHD = valvular heart disease; CABG = coronary artery bypass graft.
Procedures consisted of aortic valve replacement (n = 5; 16.7%), TAVI (n = 5; 16.7%) mitral valve replacement (n = 2; 6.7%) and those with multiple procedures (n = 3; 10.0%), with CABG being the most common procedure (n = 15; 50.0%). Comorbidities included hyperlipidemia, hypertension, diabetes, obstructive sleep apnea, previous stroke, rheumatic heart disease, COPD, atrial fibrillation, previous cardiac procedures, CAD, fibromyalgia, asthma, anxiety, and depression. With regard to comor-bidities, three charts recorded none, four charts recorded one, nine charts recorded two, 10 charts recorded three, and four charts recorded four or more. This fits the demographic profile of those likely to be at risk for frailty. The most common comorbidities were hyperlipidemia (n = 20; 66.7%) and hypertension (n = 19; 63.3%).
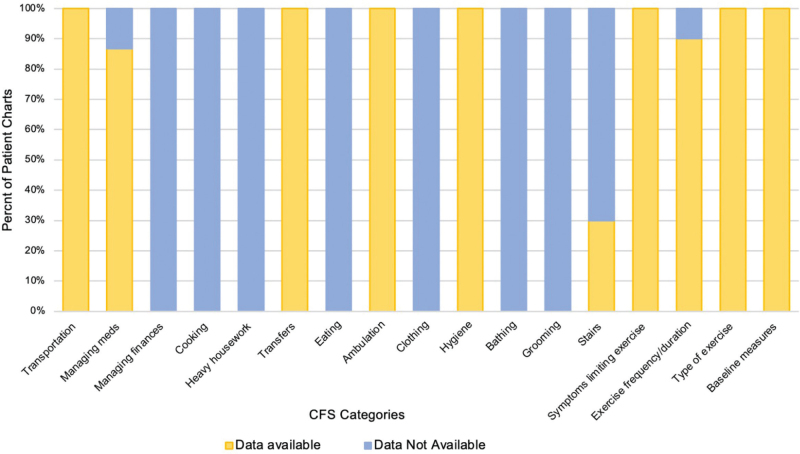
The CFS-CR scores could not be determined retrospectively for any of the 30 charts due to insufficient data. This limited the clinician rater from progressing beyond the second level of the algorithm, which involved the ability to perform certain IADLs. Of the five IADLs (heavy housework, cooking, managing finances, managing medications and transportation), information about transportation was available in all 30 charts, and information about medication management was available in 26 charts (86.7%). There was no information about managing finances, cooking, and heavy housework in any of the 30 charts (0%). Information regarding ambulation was available in all 30 charts, and the ability to navigate stairs was available in nine charts (30%) (Figure 2). Information regarding these IADLs was necessary to complete this step and progress beyond the second level of the algorithm.
Figure 2.
Availability of Data in Patient Charts to Assign a Score Using the CFS-CR Algorithm.
The CFS-CR categories reflect important data that raters require when determining the level of frailty. The percentage of charts in which “Data Available” in the intake assessment of the chart is reported with yellow bars. The percentage of charts in which “Data is Not Available” is indicated with blue bars.
Data related to many ADLs, such as eating, clothing, bathing, and grooming, were unavailable, with the exception of hygiene (toileting). In almost all of the 30 charts, information was available regarding transfers; exercise (frequency, duration, type); symptoms limiting exercise; and routine baseline measures.
Discussion
We developed the CFS-CR algorithm, which is based on the CFS, to support clinical judgment when screening for frailty in the post-cardiac surgical population entering CR, fulfilling this study’s primary objective. The CFS-CR can be used by various health care professionals that perform intake assessments of patients in the CR setting without requiring training as it provides the rater with CR specific definitions to inform clinical judgment. To our knowledge, this is the only study that has adapted the CFS to examine frailty in a post-cardiac surgery/procedure population entering CR. Our secondary objective was to examine the feasibility of utilizing the CFS-CR to retrospectively assess frailty using participants’ medical charts. Applying the CFS-CR to CR intake assessment documentation revealed insufficient information in patient charts to assign a frailty score to patients using the CFS-CR. This finding suggests that a tool, such as the CFS-CR, may serve to guide the assessment of patient health and function to assist screening for frailty at entry into CR; however, this requires a prospective study design to explore.
The CFS is an established clinical tool for frailty screening across a broad range of patient populations.26 The development of the CFS-CR followed similar methodologies as previously developed the CFS tools and algorithms that aimed to support clinical judgment in a specific patient group. For example, the CFS algorithm (CFS-A) was developed for use in a hospitalized geriatric population with a mean age of 89.4 ± 4.6 years.28 Specific to this patient population, the CFS-A delineated between categories 7 to 9 by adding descriptors of worsening health and including “unplanned hospital admissions and rapidly worsening symptoms of chronic disease.”28
In comparison, the CFS-CR clearly differentiated between regular and occasional exercise to describe categories 1 to 4, specifically aligning the definitions with that of current definitions of participant activity levels commonly used in CR.40,42 Furthermore, the CFS-CR provides guidance regarding specific ADL and IADL tasks to be assessed based on level of independence in patients after cardiac surgery/ procedure who present to CR at different stages of recovery. The development of patient-specific CFS algorithms to guide clinical judgment is the first step in increasing screening for frailty in programmes such as CR.
Investigating the feasibility of utilizing the CFS-CR through a retrospective review of patient charts in a CR programme revealed findings that are important to assessing frailty in the CR setting. Retrospectively, the clinician was consistently unable to progress beyond the second level in the algorithm related to the patient’s ability to perform IADLs. Our findings identified suggest that only two IADLs, transportation and managing medications, were commonly reported. Additionally, only three of the eight ADLs (transfers, ambulation, hygiene/toileting) were consistently reported. Information regarding mobility and exercise were readily available. The CFS has been applied retrospectively, using electronic medical records and chart reviews, in an outpatient geriatric population demonstrating the ability for its use when data is available.27,43 Our findings suggest that the CFS-CR may assist in further guiding assessments at CR intake to assist in clinical judgment and screening of the patients’ level of frailty.
It is important to note that the CR intake documentation reviewed in this study aligns with the goals of CR programming (i.e., exercise-based therapy, nutritional, psychosocial counselling, etc.) and is combined with physiological assessments to further individualize a patient’s exercise programme. ADLs and IADLs are typically discussed on a case-by-case basis and could be documented at subsequent sessions causing these to be missed in our review of initial intake assessment reports. Importantly, the lack of details regarding ADLs and IADLs are not likely to impact exercise prescription related to increasing endurance and the behavioural self-management of exercise - the foundational goals of CR.15 However, this lack of information may limit the ability to detect frailty or those at risk of developing frailty, and the opportunity to expand treatment plans to directly address areas of function, in addition to fitness, that may positively impact the participant’s frailty profile. Including a tool such as the CFS-CR as part of the intake assessment may assist CR programmes to better monitor and address characteristics related to the spectrum of frailty.44 Exercise-based CR programmes are well situated to have beneficial effects on the short- and long-term outcomes associated with frailty and/or the development of frailty in the post-cardiac surgery/procedure populations.22,45,46
Our study has limitations. The sample size was small (n = 30), affecting the generalizability of our results. It is expected, however, that by increasing the sample size, the results would be similar because all of the data were missing in some CFS-related categories. Additionally, this was a single-site study, which limits the ability to extrapolate our results to other CR programmes, as the data collected in other programmes may vary. Although, this study included only one reviewer, multiple reviewers would not have likely changed the results. For future research, multiple reviewers are recommended to assess the tool’s reliability. While the study team took a multidisciplinary approach to developing the CFS-CR, future research that addresses the use of the algorithm should be expanded to include all members of the CR team (i.e., cardiac technologists, kinesiologists, nurses, physicians) involved in intake assessments. Lastly, a prospective investigation of the application of the CFS-CR during CR intake to screen for frailty is required to more comprehensively explore the tool’s utility.
Conclusion
Identifying frailty during CR intake may provide clinicians with a more comprehensive clinical picture that will help direct individualized, exercise-based treatment strategies. Future research is needed to validate the CFS-CR and examine the interrater reliability of its use. Further studies are also recommended to explore the administration of the CFS-CR in the CR setting.
The CFS-CR algorithm is an important first step in providing a CR-specific frailty screening tool that can be implemented on intake to CR. Providing targeted interventions based on frailty screening may reduce the incidence, severity, and adverse effects of frailty experienced by individuals enrolled in CR.22 From a systems perspective, the long-term improvements in overall physical fitness and QOL could also reduce burgeoning health care costs.24,32,33
Key Messages
What is already known on this topic
Despite the link between frailty and cardiac disease, frailty screening at intake to CR is not commonly performed. Various studies have shown the beneficial effects of exercise and CR in those with frailty. There are many existing frailty screening tools; however, none have emerged as the gold standard, and there is limited evidence for their application in a CR outpatient setting.
What this study adds
To our knowledge, this is the first study to develop an algorithm (the CFS-CR), building from the established CFS, to screen for frailty in older adults entering a CR programme. However, the feasibility of using the CFS-CR could not be determined by a retrospective chart review due to the lack of frailty-specific clinical documentation. This highlights the importance of investigating the prospective application of the CFS-CR to identify frailty in the CR setting.
References
- 1.Fried LP Tangen CM, Walston J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56(3):M146–57. 10.1093/gerona/56.3.m146. Medline: [DOI] [PubMed] [Google Scholar]
- 2.Rockwood K, Song X, MacKnight C, et al. A global clinical measure of fitness and frailty in elderly people. CMAJ. 2005;173(5):489–95. 10.1503/cmaj.050051. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Clegg A, Young J, Iliffe S, et al. Frailty in elderly people. Lancet. 2013;381(9868):752–62. 10.1016/s0140-6736(12)62167-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lytwyn J, Stammers AN, Kehler DS, et al. The impact of frailty on functional survival in patients 1 year after cardiac surgery. J Thorac Cardiovasc Surg. 2017;154(6):1990–9. 10.1016/j.jtcvs.2017.06.040. Medline: [DOI] [PubMed] [Google Scholar]
- 5.Lee DH, Buth KJ, Martin BJ, et al. Frail patients are at increased risk for mortality and prolonged institutional care after cardiac surgery. Circulation. 2010;121(8):973–8. 10.1161/circulationaha.108.841437. Medline: [DOI] [PubMed] [Google Scholar]
- 6.Afilalo J, Karunananthan S, Eisenberg MJ, et al. Role of frailty in patients with cardiovascular disease. Am J Cardiol. 2009;103 (11): 1616–21. 10.1016/j.amjcard.2009.01.375. Medline: [DOI] [PubMed] [Google Scholar]
- 7.Singh M, Stewart R, White H. Importance of frailty in patients with cardiovascular disease. Eur Heart J. 2014;35(26):1726–31. 10.1093/eurheartj/ehu197. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gary R. Evaluation of frailty in older adults with cardiovascular disease: incorporating physical performance measures. J Cardiovasc Nurs. 2012;27(2):120–31. 10.1097/jcn.0b013e318239f4a4. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim DH, Kim CA, Placide S, et al. Preoperative frailty assessment and outcomes at 6 months or later in older adults undergoing cardiac surgical procedures: a systematic review. Ann Intern Med. 2016;165(9): 650–60. 10.7326/m16-0652. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tran DTT, Tu JV Dupuis JY, et al. Association of frailty and long-term survival in patients undergoing coronary artery bypass grafting. J Am Heart Assoc. 2018;7(15):e009882. 10.1161/jaha.118.009882. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Goldfarb M, Bendayan M, Rudski LG, et al. Cost of cardiac surgery in frail compared with nonfrail older adults. Can J Cardiol. 2017;33(8):1020–6. 10.1016/jxjca.2017.03.019. Medline: [DOI] [PubMed] [Google Scholar]
- 12.Kim DH, Glynn RJ, Avorn J, et al. Validation of a claims-based frailty index against physical performance and adverse health outcomes in the health and retirement study. J Gerontol A Biol Sci Med Sci. 2019; 74(8):1271–6. 10.1093/gerona/gly197. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Thomas RJ, Beatty AL, Beckie TM, et al. Home-based cardiac rehabilitation: a scientific statement from the American Association of Cardiovascular and Pulmonary Rehabilitation, the American Heart Association, and the American College of Cardiology. J Am Coll Cardiol. 2019;74(1):133–53. 10.1016/j.jacc.2019.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Balady GJ, Williams MA, Ades PA, et al. Core components of cardiac rehabilitation/secondary prevention programs: 2007 update: a scientific statement from the American Heart Association Exercise, Cardiac Rehabilitation, and Prevention Committee, the Council on Clinical Cardiology; the Councils on Cardiovascular Nursing, Epidemiology and Prevention, and Nutrition, Physical Activity, and Metabolism; and the American Association of Cardiovascular and Pulmonary Rehabilitation. Circulation. 2007;115(20):2675–82. 10.1161/circulationaha.106.180945. Medline: [DOI] [PubMed] [Google Scholar]
- 15.Bjarnason-Wehrens B, Nebel R, Jensen K, et al. Exercise-based cardiac rehabilitation in patients with reduced left ventricular ejection fraction: the Cardiac Rehabilitation Outcome Study in Heart Failure (CROS-HF): a systematic review and metaanalysis. Eur J Prev Cardiol. 2020;27(9):929–952. 10.1177/2047487319854140. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Anderson L, Oldridge N, Thompson DR, et al. Exercise-based cardiac rehabilitation for coronary heart disease Cochrane systematic review and meta-analysis. J Am Coll Cardiol. 2016;67(1):1–12. 10.1016/j.jacc.2015.10.044. [DOI] [PubMed] [Google Scholar]
- 17.O’Connor CM, Whellan DJ, Lee KL, et al. Efficacy and safety of exercise training in patients with chronic heart failure HF-ACTION randomized controlled trial. J Am Med Assoc. 2009;301(14):1439–50. 10.1001/jama.2009.454. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ehsani AA, Spina RJ, Peterson LR, et al. Attenuation of cardiovascular adaptations to exercise in frail octogenarians. J Appl Physiol. 2003;95(5):1781–8. 10.1152/japplphysiol.00194.2003. Medline: [DOI] [PubMed] [Google Scholar]
- 19.Vaitkevicius PV Ebersold C, Shah MS, et al. Effects of aerobic exercise training in community-based subjects aged 80 and older: a pilot study. J Am Geriatr Soc. 2002;50(12):2009–13. 10.1046/j.1532-5415.2002.50613.x. Medline: [DOI] [PubMed] [Google Scholar]
- 20.Lopez P, Pinto RS, Radaelli R, et al. Benefits of resistance training in physically frail elderly: a systematic review. Aging Clin Exp Res. 2018;30(8):889–99. 10.1007/s40520-017-0863-z. Medline: [DOI] [PubMed] [Google Scholar]
- 21.Liu X, Ng DHM, Seah JWT, et al. Update on interventions to prevent or reduce frailty in community-dwelling older adults: a scoping review and community translation. Curr Geriatr Reports. 2019;8(2):72–86. 10.1007/s13670-019-0277-1. [DOI] [Google Scholar]
- 22.Kehler DS, Giacomantonio N, Firth W, et al. Association between cardiac rehabilitation and frailty. Can J Cardiol. 2020;36(4):482–9. 10.1016/j.cjca.2019.08.032. Medline: [DOI] [PubMed] [Google Scholar]
- 23.Vigorito C, Abreu A, Ambrosetti M, et al. Frailty and cardiac rehabilitation: a call to action from the EAPC cardiac rehabilitation section. Eur J Prev Cardiol. 2017;24(6):577–90. 10.1177/2047487316682579. Medline: [DOI] [PubMed] [Google Scholar]
- 24.Morley JE, Vellas B, van Kan GA, et al. Frailty consensus: a call to action. J Am Med Dir Assoc. 2013;14(6):392–7. 10.1016/j.jamda.2013.03.022. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Afilalo J, Lauck S, Kim DH, et al. Frailty in older adults undergoing aortic valve replacement: the FRAILTY-AVR study. J Am Coll Cardiol. 2017;70(6):689–700. 10.1016/j.jacc.2017.06.024. [DOI] [PubMed] [Google Scholar]
- 26.Church S, Rogers E, Rockwood K, et al. A scoping review of the Clinical Frailty Scale. BMC Geriatr. 2020;20(1):1–18. 10.1186/s12877-020-01801-7. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Davies J, Whitlock J, Gutmanis I, et al. Inter-rater reliability of the retrospectively assigned Clinical Frailty Scale score in a geriatric outreach population. Can Geriatr J. 2018;21(1):1–5. 10.5770/cgj.21.263. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chong E, Chia JQ, Law F, et al. Validating a standardised approach in administration of the Clinical Frailty Scale in hospitalised older adults. Ann Acad Med Singapore. 2019;48(4):115–24. [PubMed] [Google Scholar]
- 29.Theou O, Brothers TD, Mitnitski A, et al. Operationalization of frailty using eight commonly used scales and comparison of their ability to predict all-cause mortality. J Am Geriatr Soc. 2013;61(9):1537–51. 10.1111/jgs.12420. Medline: [DOI] [PubMed] [Google Scholar]
- 30.Ge L, Yap CW, Heng BH. Prevalence of frailty and its association with depressive symptoms among older adults in Singapore. Aging Ment Heal. 2019;23(3):319–24. 10.1080/13607863.2017.1416332. Medline: [DOI] [PubMed] [Google Scholar]
- 31.Chan DC, Tsou HH, Chen CY, et al. Validation of the Chinese-Canadian study of health and aging clinical frailty scale (CSHA-CFS) telephone version. Arch Gerontol Geriatr. 2010;50(3):74–80. 10.1016/j.archger.2009.06.004. Medline: [DOI] [PubMed] [Google Scholar]
- 32.Buckinx F, Rolland Y, Reginster JY, et al. Burden of frailty in the elderly population: perspectives for a public health challenge. Arch Public Heal. 2015;73(1):1–7. 10.1186/s13690-015-0068-x. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Eamer GJ, Clement F, Holroyd-Leduc J, et al. Frailty predicts increased costs in emergent general surgery patients: a prospective cohort cost analysis. Surgery. 2019;166(1):82–7. 10.1016/j.surg.2019.01.033. Medline: [DOI] [PubMed] [Google Scholar]
- 34.Boateng GO, Neilands TB, Frongillo EA, et al. Best practices for developing and validating scales for health, social, and behavioral research: a primer. Front Public Heal. 2018;6:1–18. 10.3389/fpubh.2018.00149. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Katz S. Assessing self-maintenance: activities of daily living, mobility, and instrumental activities. J Am Geriatr Soc. 1983; 31 (12): 721–727. 10.1111/j.1532-5415.1983.tb03391.x. Medline: [DOI] [PubMed] [Google Scholar]
- 36.Lawton MP, Brody EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist. 1969;9(3):179–86. 10.1093/geront/9.3_part_1.179. [DOI] [PubMed] [Google Scholar]
- 37.Cahalin LP, LaPier TK, Shaw DK. Sternal precautions: is it time for change? Precautions versus restrictions: a review of literature and recommendations for revision. Cardiopulm Phys Ther J. 2011;22(1):5–15. 10.1097/01823246-201122010-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nonoyama ML, Marzolini S, Brooks D, et al. Comparison of cardiac rehabilitation outcomes in individuals with respiratory, cardiac or no comorbidities: a retrospective review. Can J Respir Ther. 2016;52(2):43–9. [PMC free article] [PubMed] [Google Scholar]
- 39.American College of Sports Medicine. ACSM’s guidelines for exercise testing and prescription. 9th ed. Philadelphia, PA: Lippincott Williams &Wilkins; 2014 [Google Scholar]
- 40.Nelson ME, Rejeski WJ, Blair SN, et al. Physical activity and public health in older adults: recommendation from the American College of Sports Medicine and the American Heart Association. Circulation. 2007;116(9):1094–105. 10.1161/circulationaha.107.185650. Medline: [DOI] [PubMed] [Google Scholar]
- 41.Abdullahi YS, Athanasopoulos LV, Casula RP, et al. Systematic review on the predictive ability of frailty assessment measures in cardiac surgery/procedure. Interact Cardiovasc Thorac Surg. 2017;24(4):619–24. 10.1093/icvts/ivw374. Medline: [DOI] [PubMed] [Google Scholar]
- 42.Tremblay MS, Warburton DER, Janssen I, et al. New Canadian physical activity guidelines. Appl Physiol Nutr Metab. 2011;36(1): 36–46. 10.1139/h11-009. Medline: [DOI] [PubMed] [Google Scholar]
- 43.Marincowitz C, Turner V, Allgar V, et al. Can patient frailty be estimated from inpatient records? A prospective cohort study. Adv Geriatr Med Res. 2019;2(1):1–12. 10.20900/agmr20200004. [DOI] [Google Scholar]
- 44.Flint KM, Stevens-Lapsley J, Forman DE. Cardiac rehabilitation in frail older adults with cardiovascular disease: a new diagnostic and treatment paradigm. J Cardiopulm Rehabil Prev. 2020;40 (2):72–8. 10.1097/hcr.0000000000000492. Medline: [DOI] [PubMed] [Google Scholar]
- 45.Tamuleviciute-Prasciene E, Drulyte K, Jurenaite G, et al. Frailty and exercise training: how to provide best care after cardiac surgery/procedure or intervention for elder patients with valvular heart disease. Biomed Res Int. 2018;2018. 10.1155/2018/9849475. Medline: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Eichler S, Salzwedel A, Reibis R, et al. Multicomponent cardiac rehabilitation in patients after transcatheter aortic valve implantation: predictors of functional and psychocognitive recovery. Eur J Prev Cardiol. 2017;24(3):257–64. https://doi.org/10.1177/2047487316679527. Medline:27852810 [DOI] [PubMed] [Google Scholar]