Abstract
This study aims to develop and validate a predictive nomogram for severe postoperative pleural effusion (SPOPE) in patients undergoing hepatectomy for liver cancer. A total of 536 liver cancer patients who underwent hepatectomy at the Department of Hepatobiliary Surgery I of the Affiliated Hospital of North Sichuan Medical College from January 1, 2018, to December 31, 2022, were enrolled in a retrospective observational study and comprised the training dataset. Lasso regression and logistic regression analyses were employed to construct a predictive nomogram. The nomogram was internally validated using Bootstrapping and externally validated with a dataset of 203 patients who underwent liver cancer resection at the Department of General Surgery III of the same hospital from January 1, 2020, to December 31, 2022. We evaluated the nomogram using the receiver operating characteristic curve, calibration curve, and decision curve analysis. Variables such as drinking history, postoperative serum albumin, postoperative total bilirubin, right hepatectomy, diaphragm incision, and intraoperative blood loss were observed to be associated with SPOPE. These factors were integrated into our nomogram. The C-index of the nomogram was 0.736 (95% CI: 0.692–0.781) in the training set and 0.916 (95% CI: 0.872–0.961) in the validation set. The nomogram was then evaluated using sensitivity, specificity, positive predictive value, negative predictive value, calibration curve, and decision curve analysis. The nomogram demonstrates good discriminative ability, calibration, and clinical utility.
Keywords: liver cancer, hepatectomy, pleural effusion, nomogram, prediction model
1. Introduction
Liver cancer is a prevalent malignant tumor disease, ranking sixth in global incidence and third in cancer-related deaths.[1] In China, liver cancer ranks fourth in incidence and second in cancer-related deaths,[2] with its incidence continuing to rise annually.[3] Currently, hepatectomy remains the primary treatment modality for primary liver cancer.[4] Pleural effusion, as a common postoperative complication following hepatectomy, presents with an incidence rate ranging from 18% to 71%.[5–8] Severe postoperative pleural effusion (SPOPE) can lead to symptoms such as dyspnea, chest tightness, palpitations, and respiratory distress.[8,9] As pleural effusion worsens, it can result in increased pulmonary shunting, severe ventilation/perfusion mismatch, and hypoxemia.[10,11]Additionally, it can impair cardiopulmonary and diaphragmatic function,[12] consequently affecting the patient postoperative recovery, prolonging hospital stay, and reducing the patient quality of life. Extensive research has been conducted worldwide to analyze the risk factors for postoperative pleural effusion in patients undergoing hepatectomy.[13–15] However, these studies have only conducted simple factor analyses. In a study conducted in Japan,[16] although a simple model was developed, the sample size was small. Furthermore, it not only analyzed the risk of postoperative pleural effusion in liver cancer patients but also examined the risk of postoperative pleural effusion in various diseases such as hepatic cysts, hepatic abscess, hepatic hemangioma, hepatocellular adenoma, and hepatic steatosis following liver resection. Currently, there is a lack of risk prediction models for SPOPE in patients undergoing hepatectomy for liver cancer, which can provide quantitative and objective risk stratification. This poses a significant challenge for clinicians in the management and prevention of SPOPE in liver cancer patients. In response to this situation, our study aims to establish a nomogram to predict the likelihood of developing SPOPE in liver cancer patients, providing a valuable predictive tool for the clinical management and intervention of SPOPE following liver cancer surgery.
2. Materials and methods
2.1. Data collection
This study is a retrospective study that recruited a total of 739 liver cancer patients who underwent hepatectomy at the Affiliated Hospital of North Sichuan Medical College. Among them, 536 patients who underwent hepatectomy in the Hepatobiliary Department 1 from January 1, 2018, to December 31, 2022, were included in the training dataset, while 203 patients who underwent hepatectomy in the General Surgery III from January 1, 2020, to December 31, 2022, were included in the validation dataset. The research involving human participants strictly adhered to the ethical guidelines set forth by the Institutional Ethics Committee of North Sichuan Medical College. Approval from the committee was obtained to conduct this retrospective analysis.
The following information was collected for the patients: gender, age, drinking history, diabetes, hepatocellular carcinoma: other, Hepatitis B virus, Hepatitis C virus, Hepatic Cirrhosis, tumor diameter, platelet count, prothrombin time(PT), preoperative serum albumin on the first day, postoperative serum albumin on the first day (POST-ALB), preoperative total bilirubin on the first day, postoperative total bilirubin on the first day (POST-TBIL), albumin-bilirubin grade, Child-Pugh, American Society of Anesthesiologists physical status classification, type of hepatectomy (including 1-segment resection, 2-segment resection, 3-segment resection or more), Resection containing S7, Resection containing S8, right hepatectomy, open or laparoscopic surgery, diaphragm incision, portal vein occlusion time, intraoperative blood loss, and duration of surgery.
Inclusion criteria were as follows: age ≥ 18 years; complete clinical and endoscopic data; Patients with confirmed postoperative pathology of primary liver cancer; absence of pleural effusion confirmed by ultrasound or CT examination before surgery. Exclusion criteria were as follows: incomplete clinical case data; inability to perform liver parenchymal resection or presence of incisions other than in the abdomen; concomitant pulmonary infection before surgery. A graphical representation of the study process is presented in Figure 1, illustrating the details of our research methodology.
Figure 1.
Flowchart of this study.
2.2. Positive patient
We defined patients with moderate to large pleural effusion (≥500 mL) confirmed by B-ultrasound between postoperative days 4 to 23 as positive cases.
The estimated volume of pleural effusion (EV) was calculated using the formula: EV = (X + LDD) × 70, where EV represents the estimated volume of pleural effusion in milliliters (mL); X denotes the measurement of the range of pleural effusion on the posterior chest wall, with the patient in an upright or seated position, and the ultrasound probe oriented longitudinally; LDD represents the distance from the lung base to the midpoint of the diaphragm or the height of the effusion below the lung in centimeters (cm); and 70 is the empirical constant factor.[17]
2.3. Statistical methods
Statistical analysis was performed with SPSS 23.0 and R 4.3.0 software. The continuous variables were represented as mean ± standard deviation (X̅ ± S), and t-test was conducted for analysis. The categorical variables were presented as frequency and percentage (n/%) and analyzed using chi-square test or Fisher exact test. In the training dataset, Lasso regression was applied to identify potential risk factors, and the optimal parameter λ (corresponding to the minimum mean of the target parameter obtained among all λ values) was determined through ten-fold cross-validation. These potential risk factors were then included in logistic regression analysis to evaluate independent risk factors influencing the development of SPOPE following hepatectomy for liver cancer. Subsequently, the independent risk factors were incorporated into R software using the rms package to construct a predictive nomogram for the risk of SPOPE after hepatectomy for liver cancer. The predictive nomogram model was internally validated through 1000 repetitions of bootstrap sampling, using the validation set data for external validation. The discriminatory ability of the nomogram was evaluated using the ROC curve by the C-index. The calibration of the nomogram was assessed using the calibration curve (The Hosmer-Lemeshow test was used as a model fit indicator). The clinical utility of the nomogram was evaluated by determining the sensitivity, specificity, predictive values, and likelihood ratios at the optimal cutoff value (using the Youden index) and by analyzing net benefit at different threshold probabilities using the DCA curve. A P value < .05 was considered statistically significant.
3. Results
3.1. Patients’ clinical characteristics
Based on strict inclusion and exclusion criteria, we included 536 liver cancer patients who underwent hepatectomy in the training dataset. Among them, 186 patients developed SPOPE. In the validation dataset, we included 203 liver cancer patients who underwent hepatectomy, and among them, 42 patients experienced SPOPE. Table 1 displays the baseline clinical characteristics of patients in both the training and validation datasets. The majority of these factors had P values <.05, indicating the statistical differences between the training datasets and validation datasets. In Table 2, we further stratified the 2 datasets into SPOPE and Non-SPOPE to provide a clearer depiction of the baseline characteristics of the 2 groups.
Table 1.
A summary of the variables grouped by training and validation dataset in this study.
| Variables | Training dataset (n = 536) |
Validation dataset (n = 203) |
P | |
|---|---|---|---|---|
| Gender | Male: Female | 420:116 | 165:38 | .074 |
| Age | ≥60 = 1 | 240 (44.8%) | 99 (48.8%) | .163 |
| Drinking history | 202 (37.7%) | 77 (37.9%) | .963 | |
| Diabetes | 65 (12.1%) | 35 (17.2%) | .001 | |
| HCC:other* | HCC = 1 | 394 (73.5%) | 165 (81.3%) | .001 |
| HBV | 347 (64.7%) | 149 (73.4%) | .001 | |
| HCV | 7 (1.3%) | 0 | / | |
| Hepatic cirrhosis | 354 (66%) | 145 (71.4%) | .003 | |
| Tumor diameter | ≥5 cm = 1 | 196 (36.6%) | 84 (41.4%) | .036 |
| PC | <100 × 109/l = 1 | 107 (20%) | 49 (24.1%) | .016 |
| PT | >15 s = 1 | 54 (10.1%) | 16 (7.9%) | .066 |
| PRE-ALB | <35 g/L = 1 | 82 (15.3%) | 24 (11.8%) | .014 |
| POST-ALB | <35 g/L = 1 | 295 (55%) | 140 (69%) | .001 |
| PRE-TBIL | >17.1 µmol/L = 1 | 228 (42.5%) | 93 (45.8%) | .567 |
| POST-TBIL | >17.1 µmol/L = 1 | 408 (76.1%) | 157 (77.3%) | .481 |
| ALBI grade | ≥2 = 1 | 220 (41%) | 87 (42.9%) | .398 |
| Child-Pugh | B or C = 1 | 168 (31.3%) | 22 (10.8%) | .088 |
| ASA-PS | ≥3 = 1 | 173 (32.3%) | 62 (30.5) | .357 |
| Type of hepatectomy | ||||
| 1-segment resection | 227 (42.4%) | 118 (58.1%) |
.48 |
|
| 2-segment resection | 257 (47.9%) | 74 (36.5%) | ||
| 3-segment resection or more | 52 (9.7%) | 11 (5.4%) | ||
| Resection containing S7 | 77 (14.4%) | 5 (2.5%) | .001 | |
| Resection containing S8 | 59 (11%) | 9 (4.4%) | .001 | |
| Right hepatectomy | 349 (65.1%) | 98 (48.3%) | .001 | |
| Open or laparoscopic surgery | Laparoscopic surgery = 1 | 374 (69.8%) | 110 (54.2%) | .001 |
| Diaphragm incision | 32 (6%) | 6 (3%) | .001 | |
| PVOT | ≥15 min = 1 | 157 (29.3%) | 37 (18.2%) | .001 |
| Intraoperative blood loss | ≥500 mL = 1 | 219 (40.9%) | 95 (46.8%) | .024 |
| Duration of surgery | 4.061 ± 1.874 | 3.772 ± 1.614 | .031 | |
ALBI grade = albumin-bilirubin grade, ASA-PS = American Society of Anesthesiologists physical status classification, HBV = Hepatitis B virus, HCC = hepatocellular carcinoma, HCV = Hepatitis C virus, PC = platelet count, POST-ALB = postoperative serum albumin, POST-TBIL = postoperative total bilirubin, PRE-ALB = preoperative serum albumin, PRE-TBIL = preoperative total bilirubin, PT = prothrombin time, PVOT = portal vein occlusion time.
defined as the Intrahepatic cholangiocarcinoma (ICC)/mixed hepatocellular-cholangiocarcinoma.
Table 2.
The baseline characteristics of patients in the training and validation datasets.
| Variables | Training dataset | Validation dataset | |||||
|---|---|---|---|---|---|---|---|
| SPOPE (n = 186) |
Non-SPOPE (n = 350) |
P | SPOPE (n = 42) |
Non-SPOPE (n = 161) |
P | ||
| Gender | Male: Female | 145:41 | 275:75 | .744 | 37:5 | 128:33 | .006 |
| Age | ≥60 = 1 | 93 (50%) | 147 (42%) | .028 | 17 (40.5%) | 82 (50.9%) | .018 |
| Drinking history | 92 (49.5%) | 110 (31.4%) | <.001 | 42 (100%) | 35 (21.7%) | <.001 | |
| Diabetes | 24 (12.9%) | 41 (11.7) | .425 | 4 (9.5%) | 31 (19.3%) | <.001 | |
| HCC:other* | HCC = 1 | 140 (75.3%) | 254 (72.6%) | .171 | 35 (83.3%) | 130 (80.7%) | .435 |
| HBV | 120 (64.5%) | 227 (64.9) | .876 | 34 (81%) | 115 (71.4%) | .006 | |
| HCV | 2 (1.1%) | 5 (2.7%) | .493 | 0 | 0 | / | |
| Hepatic cirrhosis | 136 (38.9%) | 218 (62.3%) | <.001 | 28 (66.7%) | 117 (72.7%) | .163 | |
| Tumor diameter | ≥5cm = 1 | 71 (38.2%) | 125 (35.7%) | .277 | 24 (57.1%) | 60 (37.3%) | .265 |
| PC | <100 × 109/l = 1 | 43 (23.1%) | 64 (18.3%) | .009 | 9 (21.4%) | 40 (28.2%) | .341 |
| PT | >15s = 1 | 24 (12.9%) | 30 (8.6%) | .002 | 4 (9.5%) | 12 (7.5%) | .384 |
| PRE-ALB | <35 g/L = 1 | 30 (16.1%) | 52 (14.9%) | .44 | 6 (14.3%) | 18 (11.2%) | .281 |
| POST-ALB | <35 g/L = 1 | 125 (67.2%) | 170 (48.6%) | <.001 | 35 (83.3%) | 105 (65.2%) | <.001 |
| PRE-TBIL | >17.1umol/L = 1 | 87 (46.8%) | 141 (40.3%) | .02 | 17 (40.5%) | 76 (47.2%) | .055 |
| POST-TBIL | >17.1umol/L = 1 | 157 (84.4%) | 251 (71.7%) | <.001 | 32 (76.2%) | 125 (77.6%) | .696 |
| ALBI grade | ≥2 = 1 | 80 (43%) | 140 (40%) | .207 | 18 (42.9%) | 69 (42.9%) | / |
| Child-Pugh | B or C = 1 | 19 (10.2%) | 28 (8%) | .087 | 5 (11.9%) | 17 (10.6%) | .623 |
| ASA-PS | ≥3 = 1 | 64 (34.4%) | 109 (31.1%) | .136 | 9 (21.4%) | 53 (32.9%) | .001 |
| Type of hepatectomy | |||||||
| One-segment resection | 76 (40.9%) | 151 (43.1%) |
.064 |
23 (54.8%) | 95 (59%) |
.343 |
|
| Two-segment resection | 83 (44.6%) | 174 (49.7%) | 18 (42.8%) | 56 (34.8%) | |||
| Three-segment resection or more | 27 (14.5%) | 25 (7.2%) | 1 (2.4%) | 10 (6.2%) | |||
| Resection containing S7 | 34 (18.3%) | 43 (12.3%) | <.001 | 2 (4.8%) | 3 (1.9%) | .034 | |
| Resection containing S8 | 31 (16.7%) | 28 (8%) | <.001 | 0 | 9 (5.6%) | .001 | |
| Right hepatectomy | 147 (79%) | 202 (57.7) | <.001 | 35 (83.3%) | 63 (39.1%) | <.001 | |
| Open or laparoscopic surgery | laparoscopic surgery = 1 | 117 (62.9%) | 257 (73.4%) | <.001 | 14 (33.3%) | 96 (59.6%) | .056 |
| Diaphragm incision | 24 (12.9%) | 8 (2.3%) | <.001 | 6 (14.3%) | 0 | <.001 | |
| PVOT | ≥15min = 1 | 68 (36.6%) | 89 (25.4%) | <.001 | 14 (33.3%) | 23 (14.3%) | <.001 |
| Intraoperative blood loss | ≥500ml = 1 | 100 (53.8%) | 119 (34%) | <.001 | 37 (88.1%) | 58 (36%) | <.001 |
| Duration of surgery | 4.125 ± 1.812 | 4.027 ± 1.908 | .614 | 4.762 ± 1.457 | 3.513 ± 1.556 | .326 | |
ALBI grade = albumin-bilirubin grade, ASA-PS = American Society of Anesthesiologists physical status classification, HBV = Hepatitis B virus, HCC = hepatocellular carcinoma, HCV = Hepatitis C virus, PC = platelet count, POST-ALB = postoperative serum albumin, POST-TBIL = postoperative total bilirubin, PRE-ALB = preoperative serum albumin, PRE-TBIL = preoperative total bilirubin, PT = prothrombin time, PVOT = portal vein occlusion time.
defined as the Intrahepatic cholangiocarcinoma (ICC)/mixed hepatocellular-cholangiocarcinoma.
3.2. Risk factors of SPOPE
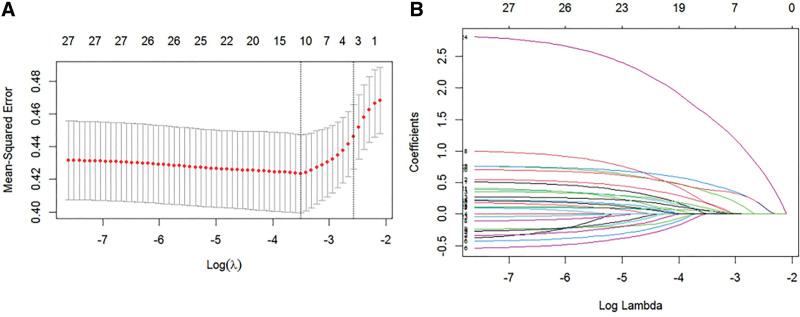
In the training dataset, a lasso regression analysis was conducted with SPOEO OR Non-SPOPE as the dependent variable and the potential risk factors from Table 2 as independent variables. The regression results showed that the Lambda.min was 0.03013, and a total of ten risk factors were identified, including Age, Drinking History, Hepatic Cirrhosis, PT, POST-ALB, POST-TBIL, Right Hepatectomy, Diaphragm Incision, Intraoperative Blood loss, duration of surgery (Fig. 2).
Figure 2.
Variable selection by the LASSO regression. (A) LASSO lambda graph, it is used to describe the relationship between coefficients and regularization parameters in Lasso regression. (B) lambda.pdf, 10 variables with nonzero coefficients were selected by deriving the optimal lambda.
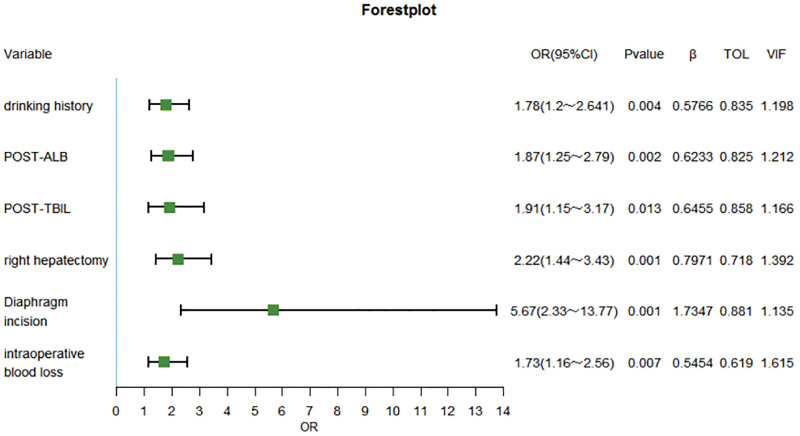
Furthermore, the ten selected risk factors from lasso regression were further subjected to binary logistic regression analysis. In this analysis, 6 independent risk factors were identified, including Drinking History, POST-ALB, POST-TBIL, Right Hepatectomy, Diaphragm Incision, and Intraoperative Blood Loss (Fig. 3).
Figure 3.
Multivariate logistic regression analysis and forest plot based on training dateSet.
Finally, collinearity diagnostics were performed on these 6 independent risk factors, and the results showed that the tolerance (Tol) values were all >0.1 and the variance inflation factor values were all <10, indicating the absence of collinearity (Fig. 3).
3.3. Development and validation of the nomogram
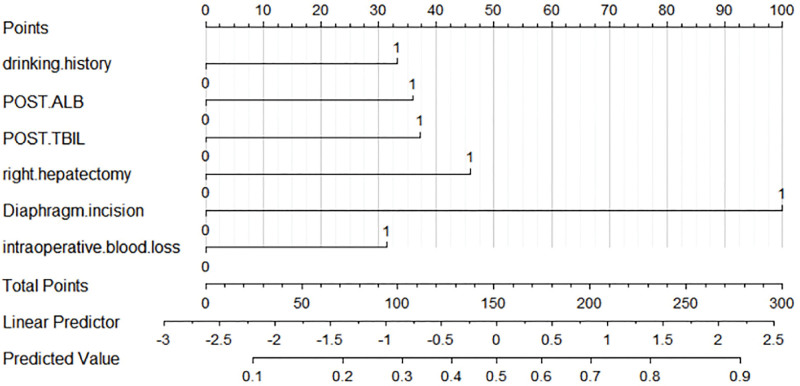
3.3.1. Development of the nomogram.
Based on the logistic regression analysis, a predictive nomogram for SPOPE risk after liver cancer resection was constructed using the 6 independent risk factors. Each independent risk factor corresponds to a score on the scoring scale above (points), and the total score for each patient (Total Points) is calculated as the sum of scores for each independent risk factor. The corresponding risk of SPOPE after liver cancer resection can be determined by locating the total score on the “Predicted Value” risk axis. A higher total score indicates a higher risk (Fig. 4).
Figure 4.
Nomogram to estimate the probability of severe postoperative pleural effusion. POST-ALB = postoperative serum albumin, POST-TBIL = postoperative total bilirubin.
3.3.2. Validation of the nomogram.
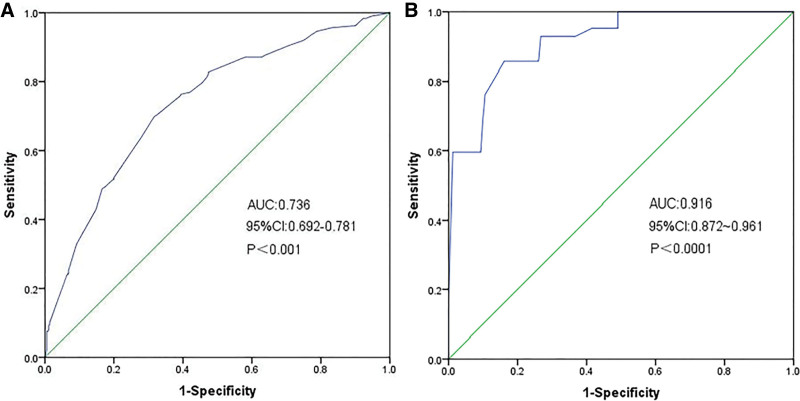
By performing 1000 repeated Bootstrap resampling, the nomogram was internally validated, and the validation dataset was used for external validation. In the training dataset, the nomogram demonstrated good discrimination and calibration in predicting the risk of SPOPE after hepatectomy, with a C-index (area under the ROC curve, AUC) of 0.736 (95% CI: 0.692–0.781), indicating good discriminative ability (Fig. 5A). The calibration curve showed good consistency between the predicted risk of SPOPE and the actual risk as estimated by the nomogram (Fig. 6A). Furthermore, the Hosmer-Lemeshow test was conducted as a measure of model fit, and the result showed a P value >.05, indicating good model fit (Fig. 6A).
Figure 5.
Receiver operating characteristic (ROC) curves of nomograms in the training dataset and validation dataset, respectively. (A) The AUC value of the training dataset is 0.736, 95% CI: 0.692–0.781 (P < .05). (B) The AUC value of the validation dataset is 0.916, 95% CI: 0.872–0.961 (P < .05).
Figure 6.
The calibration curve of the nomogram for predicting severe postoperative pleural effusion in the training dataset (A) and validation dataset (B), respectively.
The nomogram also demonstrated strong discriminative and calibration performance in the validation dataset for predicting the risk of SPOPE after hepatectomy, as evidenced by a C-index of 0.916 (95% CI: 0.872–0.961) (Fig. 5B). The predicted probabilities from the nomogram showed good consistency with the observed probabilities (Fig. 6B). The Hosmer-Lemeshow test also indicated a good model fit.
3.3.3. Clinical utility analysis of the nomogram.
The optimal cutoff value for the total score on the nomogram, determined by the Youden index, was calculated to be 115.65 points. Patients with a total score ≥ 115.65 were classified as the high-risk group, while those with a total score < 115.65 were classified as the low-risk group. In the training dataset, the sensitivity (SEN), specificity (SPE), positive predictive value (PPV), negative predictive value (NPV), positive likelihood ratio (LR+), and negative likelihood ratio (−LR) at the cutoff value were 69.9% (95% CI: 60%–78.5%), 68.3% (95% CI: 58.1%–77%), 68.8% (95% CI: 58.7%–77.4%), 69.4% (95% CI: 59.2%–78%), 2.21, and 0.44, respectively. In the validation dataset, these values were 85.7% (95% CI: 76.9%–91.6%), 83.9% (95% CI: 74.9%–90.2%), 84.2% (95% CI: 75.3%–90.4%), 85.4% (95% CI: 76.5%–91.5%), 5.32, and 0.17, respectively (Table 3).
Table 3.
Clinical utility analysis of the nomogram.
| Indicators | Training dataset (95%CI) | Validation dataset (95%CI) |
|---|---|---|
| C-index | 0.736 (0.692–0.781) | 0.916 (0.872–0.961) |
| Optimal cutoff value | 115.65 | 115.65 |
| Sensitivity, SEN | 69.9% (60%–78.5%) | 85.7% (76.9%–91.6%) |
| Specificity, SPE | 68.3% (58.1%–77%) | 83.9% (74.9%–90.2%) |
| positive predictive value, PPV | 68.8% (58.7%–77.4%) | 84.2% (75.3%–90.4%) |
| Negative predictive value, NPV | 69.4% (59.2%–78%) | 85.4% (76.5%–91.5%) |
| Positive likelihood ratio, LR+ | 2.21 (1.61–3.02) | 5.32 (3.38–8.39) |
| Negative likelihood ratio, −LR | 0.44 (0.30–0.6) | 0.17 (0.11–0.28) |
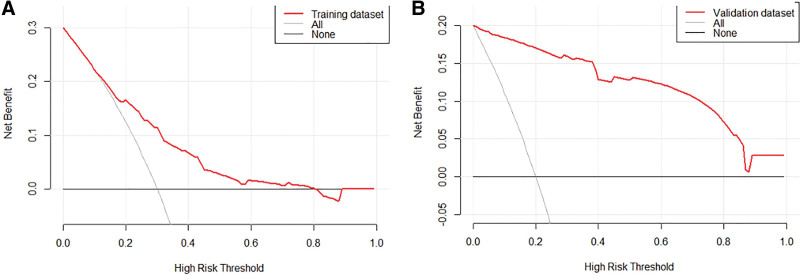
3.3.4. Clinical decision curve analysis of the nomogram.
To assess the clinical utility of the nomogram, we conducted decision curve analysis, which evaluates the net benefit across various threshold probabilities. The results showed that in the training dataset, the model had a greater advantage in identifying high-risk patients when the threshold probability ranged from 0.1 to 0.8. In the validation dataset, the model performed well across the entire range of threshold probabilities. Therefore, it can be concluded that the nomogram has clinical value in predicting the risk of SPOPE in patients with liver cancer, and its application can benefit patients (Fig. 7).
Figure 7.
Decision curve analysis for the severe postoperative pleural effusion predictive nomogram. (A) Training dataset. (B) Validation dataset.
4. Discussion
Severe pleural effusion (SPOPE) is defined as the accumulation of more than 500 milliliters of fluid in the pleural cavity following surgery. It often presents with symptoms such as dyspnea, chest tightness, palpitations, and respiratory distress, which can Significantly affecting patients’ respiratory and circulatory functions, it can cause abnormalities in gas exchange, respiratory mechanics, diaphragm function, and hemodynamics, resulting in delayed postoperative recovery, decreased quality of life, and ultimately impacting patient prognosis.[12,18] According to previous literature, the incidence of pleural effusion after hepatectomy ranges from 18% to 71%.[5–8] Furthermore, the probability of developing severe pleural effusion is 30.1%.[5] In our study, the incidence of SPOPE was as high as 34.7%. In a previous study, Jaclyn Moore et al conducted a 7-day intervention trial on prehabilitation programs for patients undergoing major abdominal surgery.[19] The results demonstrated a significant improvement in pulmonary complications, suggesting that early intervention during the perioperative period can enhance patient outcomes. However, at present, due to the lack of a risk prediction tool for post-hepatectomy severe pleural effusion (SPOPE), clinical practitioners often rely on clinical judgment and subjective assessment to predict and manage SPOPE. The role of conventional ultrasound and CT in the detection of pleural effusion after hepatectomy is certain; however, their utilization often relies on the subjective feelings of the patient and the clinical judgment of healthcare professionals, potentially leading to missed early-stage conditions in certain cases. Furthermore, disparities between different physicians and healthcare institutions can affect the execution and interpretation of these examinations. Despite their high sensitivity, B-ultrasound and CT may still be constrained by factors such as equipment availability, operator skill levels, and patient cooperation. These widespread circumstances pose challenges in implementing targeted prehabilitation programs. Our study fills this gap by providing a predictive nomogram that overcomes these limitations and enables clinicians to better assess the risk of severe pleural effusion in liver cancer patients undergoing hepatectomy. This empowers early intervention for improving patient outcomes.
The nomogram model includes 6 risk factors: Drinking History, POST-ALB, POST-TBIL, Right Hepatectomy, Diaphragm Incision, and Intraoperative Blood Loss. These factors are easily obtainable in clinical practice, and the nomogram demonstrates good discrimination, calibration, and clinical utility analysis. This indicates that our predictive nomogram provides a valuable assessment tool, and implementing it in clinical practice can aid in the early identification of high-risk individuals and facilitate proactive interventions. Ultimately, the application of this nomogram can potentially contribute to a reduction in the occurrence of SPOPE and lead to improved patient outcomes.
In our study, drinking history was identified as an independent risk factor for the development of severe pleural effusion in patients undergoing hepatectomy for liver cancer. We found that including drinking history as a factor in the predictive model for postoperative pleural effusion improved the model performance. Through further data analysis, we discovered a significant association between drinking history and postoperative pleural effusion occurrence, even after excluding the collinearity effects of liver cirrhosis and liver dysfunction. This suggests that drinking history itself may represent broader influences. Firstly, alcohol consumption can exacerbate liver inflammation and fibrosis,[20–24] Inflammation leads to liver tissue damage and destruction, affecting normal liver function. Inflammatory responses also activate the release of inflammatory mediators, further increasing vascular permeability and impairing lymphatic circulation, thereby promoting pleural effusion formation. Additionally, liver fibrosis is a key pathological change in the progression of liver disease, characterized by the proliferation and deposition of fibrous tissue in the liver. The development of liver fibrosis results in abnormal liver structure and function, affecting blood circulation and lymphatic drainage, thereby increasing the risk of pleural effusion. Secondly, alcohol consumption can suppress the function of the immune system,[25] reducing the body ability to resist infection and inflammation, thereby increasing the risk of postoperative pleural effusion. Overall, these findings suggest that a history of alcohol consumption can have detrimental effects on liver function, inflammation, immune response, and fibrosis, which contribute to an increased risk of postoperative pleural effusion.
Previous studies have consistently shown that POST-ALB[8,26] and POST-TBIL[27] are closely associated with the development of SPOPE. These findings are consistent with the results of our study. The potential mechanisms underlying the development of pleural effusion in hypoalbuminemia are as follows: As POST-ALB levels decrease, there is a reduction in its ability to maintain plasma colloid osmotic pressure and clear free radicals, which play a role in preserving lymphatic vessel function during inflammation and preventing edema formation. Consequently, this leads to an increase in capillary filtration and accumulation of pleural effusion.[8] On the other hand, Lower levels of serum albumin and higher levels of total bilirubin are associated with liver dysfunction and metabolic disorders,[28] which are closely related to the occurrence of pleural effusion. Impaired liver function and metabolic disorders can lead to changes in the intrahepatic and extrahepatic environment, such as hepatic vasodilation and lymphatic circulation alterations, which can affect the drainage and circulation of pleural effusion.
Meanwhile, Right hepatectomy is often associated with a higher risk of pleural effusion.[7,13,29] Tumors in the right liver are often larger and softer, making the resection more challenging. There is a higher risk of tumor rupture, bleeding, and parenchymal disruption, which further increases the risk of postoperative pleural effusion. Furthermore, intraoperative blood loss has a proportional relationship with pleural effusion.[14,28,29] In our study, we identified a significant correlation between intraoperative blood loss exceeding 500 ml and a heightened incidence of SPOPE. This relationship can be attributed to the intricate interplay of various factors during hepatectomy. Firstly, extensive blood loss during surgery leads to an increased degree of surgical trauma. This heightened trauma can activate systemic inflammatory responses, resulting in the release of proinflammatory cytokines. These cytokines contribute to changes in vascular permeability, thereby facilitating the accumulation of fluid in the pleural cavity. The combination of vascular alterations and enhanced fluid retention predisposes patients to pleural effusion. Furthermore, substantial blood loss can have systemic repercussions, leading to the disruption of lymphatic circulation and microcirculatory disturbances. These factors further exacerbate the formation of pleural effusion in the postoperative period. Moreover, it crucial to recognize that increased intraoperative blood loss may lead to hypoalbuminemia, which subsequently reduces intravascular osmotic pressure. This reduction in intravascular osmotic pressure enhances transudation, thereby increasing the amount of pleural fluid formed.
In our study, intraoperative diaphragm incision exhibited a remarkably high odds ratio (OR) of 5.67 (95% CI: 2.33–13.77). By excluding methodological biases and individual differences and conducting collinearity diagnostics to account for interaction effects among factors, we identified the unusually strong association between intraoperative diaphragm incision and pleural effusion as a subject worthy of further investigation. Previous research has highlighted several factors that may contribute to the development of pleural effusion during diaphragm incision. Firstly, during diaphragm incision, the diaphragm muscle inevitably incurs a partial or complete injury, and even after closure, small gaps may form, resulting in altered pressure differentials between the intrathoracic and extrathoracic compartments. This change increases the tendency for fluid accumulation within the pleural cavity, often leading to the occurrence of pleural effusion. This phenomenon has been demonstrated by Mouroux et al,[30] where patients who did not undergo diaphragm closure experienced pleural effusion.[31] Secondly, diaphragm incision may trigger local inflammatory responses and tissue trauma,[32] subsequently affecting fluid balance within the pleural cavity. Surgical trauma and inflammatory reactions can cause local vascular dilation, increased permeability, and infiltration of inflammatory cells. These changes may contribute to the formation and accumulation of pleural effusion. However, further extensive studies are needed to validate the exceptional association between intraoperative diaphragm incision and pleural effusion. Future research can employ larger sample sizes, multicenter designs, and combine clinical observations with laboratory analyses to gain a deeper understanding of the underlying mechanisms linking diaphragm incision to pleural effusion.
The predictive nomogram can serve as a valuable communication tool among multidisciplinary healthcare teams, presenting data-driven SPOPE risk in a clear and concise manner. Physicians, nurses, and other healthcare professionals can collectively analyze the data to assess the need for interventions or adjustments to treatment plans, and develop personalized treatment strategies tailored to each patient condition. Furthermore, through visualizing the data, healthcare providers and nurses can effectively communicate with patients and their families, facilitate shared decision-making, advance the implementation of prehabilitation, and improve patient satisfaction.
However, our study has several limitations. Firstly, the data were collected from a single center, and all patients included were of Chinese ethnicity. Therefore, the generalizability of the findings to other populations may be limited. In future investigations, it would be valuable to include data from multiple centers to enhance the robustness and external validity of the findings. Additionally, the retrospective design of the study introduces the possibility of selection bias and confounding factors. Further validation of our findings is warranted through prospective studies with larger sample sizes. Furthermore, the predictive nomogram was developed based on clinical and demographic variables, and the incorporation of molecular or genetic markers may further improve its accuracy and predictive power.
5. Conclusion
The predictive nomogram developed in this study, incorporating factors such as drinking history, POST-ALB and POST-TBIL, right hepatectomy, diaphragm incision, and intraoperative blood loss, holds significant potential for application in the clinical management and nursing care of severe pleural effusion after hepatocellular carcinoma hepatectomy. By facilitating risk assessment, guiding treatment decisions, and enhancing patient monitoring, this predictive nomogram can contribute to improved patient outcomes, individualized care plans, and optimized postoperative management strategies.
Acknowledgments
The authors would like to thank All authors made substantial contributions to the article.
Author contributions
Conceptualization: Xu-Jian Huang.
Data curation: Hang-Yu Lin, Cai-Fang Gong.
Formal analysis: Meng-Yi Xie.
Methodology: Hong Zhang.
Writing – original draft: Jun-Yu Zhao.
Writing – review & editing: Chuan You.
Abbreviations:
- POST-ALB
- postoperative serum albumin on the first day
- POST-TBIL
- postoperative total bilirubin on the first day
- PT
- prothrombin time
- SPOPE
- severe postoperative pleural effusion
The study was supported by the China Medical Foundation (No. 2022HX011).
The research involving human participants strictly adhered to the ethical guidelines set forth by the Medical Ethics Committee of Affiliated Hospital of North Sichuan Medical College. Approval from the committee was obtained to conduct this retrospective analysis (approval no. 2023ER250-1).
Patient consent was waived by the Medical Ethics Committee of Affiliated Hospital of North Sichuan Medical College due to the retrospective character of the study and participants’ anonymity.
The datasets generated during and/or analyzed during the current study are not publicly available, but are available from the corresponding author on reasonable request.
The authors have no conflicts of interest to disclose.
How to cite this article: Zhao J-Y, Lin H-Y, Gong C-F, Zhang H, Huang X-J, Xie M-Y, You C. Establishment and validation of a predictive nomogram for severe pleural effusion in liver cancer patients after hepatectomy. Medicine 2024;103:10(e36556).
Contributor Information
Jun-Yu Zhao, Email: zhaojy12123@outlook.com.
Hang-Yu Lin, Email: 1946295638@qq.com.
Cai-Fang Gong, Email: 2909700082@qq.com.
Hong Zhang, Email: 224133218@qq.com.
Xu-Jian Huang, Email: 1373689225@qq.com.
Meng-Yi Xie, Email: xiemy@nsmc.edu.cn.
References
- [1].Yang A, Zhang P, Sun Z, et al. Lysionotin induces apoptosis of hepatocellular carcinoma cells via caspase-3 mediated mitochondrial pathway. Chem Biol Interact. 2021;344:109500. [DOI] [PubMed] [Google Scholar]
- [2].Department of Medical Administration, National Health and Health Commission of the People’s Republic of China. [Guidelines for diagnosis and treatment of primary liver cancer in China (2019 edition)]. Zhonghua Gan Zang Bing Za Zhi. 2020;28:112–8. Chinese. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Chen H, Jia W. Progress in hepatectomy for hepatocellular carcinoma and peri-operation management. Genes Dis. 2020;7:320–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Colasanti M, Berardi G, Mariano G, et al. Laparoscopic left hepatectomy for hepatocellular carcinoma recurrence following liver transplantation. Ann Surg Oncol. 2022;29:2984. [DOI] [PubMed] [Google Scholar]
- [5].Fuks D, Cauchy F, Fteriche S, et al. Laparoscopy decreases pulmonary complications in patients undergoing major liver resection: a propensity score analysis. Ann Surg. 2016;263:353–61. [DOI] [PubMed] [Google Scholar]
- [6].Nobili C, Marzano E, Oussoultzoglou E, et al. Multivariate analysis of risk factors for pulmonary complications after hepatic resection. Ann Surg. 2012;255:540–50. [DOI] [PubMed] [Google Scholar]
- [7].Sun Z, Yu Z, Yu S, et al. Post-operative complications in living liver donors: a single-center experience in China. PLoS One. 2015;10:e135557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Jeong HW, Kim JW, Shin WJ, et al. Early postoperative hypoalbuminaemia is associated with pleural effusion after donor hepatectomy: a propensity score analysis of 2316 donors. Sci Rep. 2019;9:2790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Dondero F, Taille C, Mal H, et al. Respiratory complications: a major concern after right hepatectomy in living liver donors. Transplantation. 2006;81:181–6. [DOI] [PubMed] [Google Scholar]
- [10].Agusti AG, Cardus J, Roca J, et al. Ventilation-perfusion mismatch in patients with pleural effusion: effects of thoracentesis. Am J Respir Crit Care Med. 1997;156(4 Pt 1):1205–09. [DOI] [PubMed] [Google Scholar]
- [11].Razazi K, Boissier F, Neuville M, et al. Pleural effusion during weaning from mechanical ventilation: a prospective observational multicenter study. Ann Intensive Care. 2018;8:103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Thomas R, Jenkins S, Eastwood PR, et al. Physiology of breathlessness associated with pleural effusions. Curr Opin Pulm Med. 2015;21:338–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Tsai KY, Chen HA, Wang WY, et al. Risk factors analysis of postoperative pleural effusion after liver resection. Dig Surg. 2019;36:514–21. [DOI] [PubMed] [Google Scholar]
- [14].Uchiyama H, Harimoto N, Itoh S, et al. Pleural effusion after hepatectomy for hepatocellular carcinoma: risk factor analyses and its impact on oncological outcomes. World J Surg. 2017;41:1089–99. [DOI] [PubMed] [Google Scholar]
- [15].Park J, Kwon D, Choi GS, et al. Safety and risk factors of pure laparoscopic living donor right hepatectomy: comparison to open technique in propensity score-matched analysis. Transplantation. 2019;103:e308–16. [DOI] [PubMed] [Google Scholar]
- [16].Nitta H, Mitsuura C, Shiraishi Y, et al. Predictive model for postoperative pleural effusion after hepatectomy. Ann Gastroenterol Surg. 2021;5:373–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Ibitoye BO, Idowu BM, Ogunrombi AB, et al. Ultrasonographic quantification of pleural effusion: comparison of four formulae. Ultrasonography. 2018;37:254–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Banini BA, Alwatari Y, Stovall M, et al. Multidisciplinary management of hepatic hydrothorax in 2020: an evidence-based review and guidance. Hepatology. 2020;72:1851–63. [DOI] [PubMed] [Google Scholar]
- [19].Moore J, Scoggins CR, Philips P, et al. Implementation of prehabilitation for major abdominal surgery and head and neck surgery: a simplified seven-day protocol. J Gastrointest Surg. 2021;25:2076–82. [DOI] [PubMed] [Google Scholar]
- [20].Kashiwagi K, Yamaguchi A, Shiba S, et al. Moderate alcohol consumption is not associated with subclinical cardiovascular damage but with hepatic fibrosis in non-alcoholic fatty liver disease. Alcohol. 2020;89:1–7. [DOI] [PubMed] [Google Scholar]
- [21].Vatsalya V, Song M, Schwandt ML, et al. Effects of sex, drinking history, and omega-3 and omega-6 fatty acids dysregulation on the onset of liver injury in very heavy drinking alcohol-dependent patients. Alcohol Clin Exp Res. 2016;40:2085–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Rice BA, Naimi TS, Long MT. Nonheavy alcohol use associates with liver fibrosis and nonalcoholic steatohepatitis in the framingham heart study. Clin Gastroenterol Hepatol. 2022;21:2854–2863.e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Kawaratani H, Moriya K, Namisaki T, et al. Therapeutic strategies for alcoholic liver disease: focusing on inflammation and fibrosis (Review). Int J Mol Med. 2017;40:263–70. [DOI] [PubMed] [Google Scholar]
- [24].Dukic M, Radonjic T, Jovanovic I, et al. Alcohol, inflammation, and microbiota in alcoholic liver disease. Int J Mol Sci. 2023;24:3735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Malherbe DC, Messaoudi I. Transcriptional and epigenetic regulation of monocyte and macrophage dysfunction by chronic alcohol consumption. Front Immunol. 2022;13:911951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Smeets HJ, Kievit J, Dulfer FT, et al. Analysis of post-operative hypalbuminaemia: a clinical study. Int Surg. 1994;79:152–7. [PubMed] [Google Scholar]
- [27].Ou YQ, Zhang S, Cheng QB, et al. Dynamic change of total bilirubin after portal vein embolization is predictive of major complications and posthepatectomy mortality in patients with hilar cholangiocarcinoma. J Gastrointest Surg. 2016;20:960–9. [DOI] [PubMed] [Google Scholar]
- [28].Wang S, Zhang J, Zhu L, et al. Association between liver function and metabolic syndrome in Chinese men and women. Sci Rep. 2017;7:44844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Chen HW, Deng FW, Wang FJ, et al. Laparoscopic right hepatectomy via an anterior approach for hepatocellular carcinoma. JSLS. 2018;22:e2017.00084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Mouroux J, Perrin C, Venissac N, et al. Management of pleural effusion of cirrhotic origin. Chest. 1996;109:1093–6. [DOI] [PubMed] [Google Scholar]
- [31].Matsuo Y, Nomi T, Hokuto D, et al. Pulmonary complications after laparoscopic liver resection. Surg Endosc. 2021;35:1659–66. [DOI] [PubMed] [Google Scholar]
- [32].Breuer T, Bruells CS, Horst K, et al. Effect of long-term polytrauma on ventilator-induced diaphragmatic dysfunction in a piglet model. Shock. 2019;52:443–8. [DOI] [PubMed] [Google Scholar]