CASE PRESENTATION
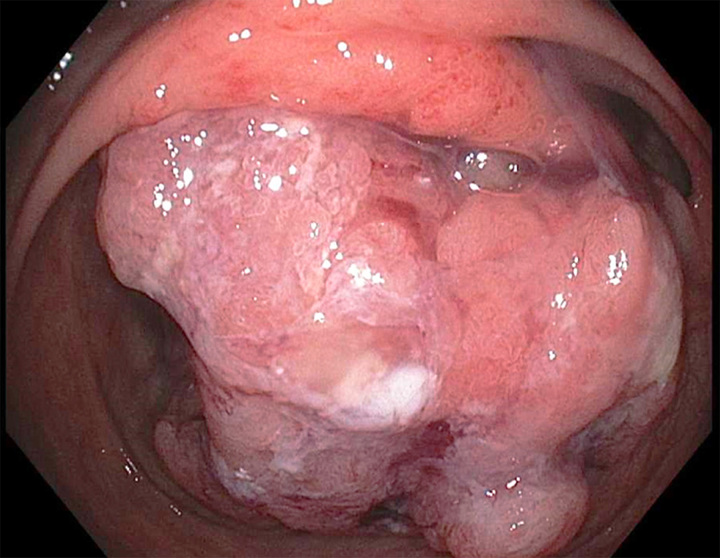
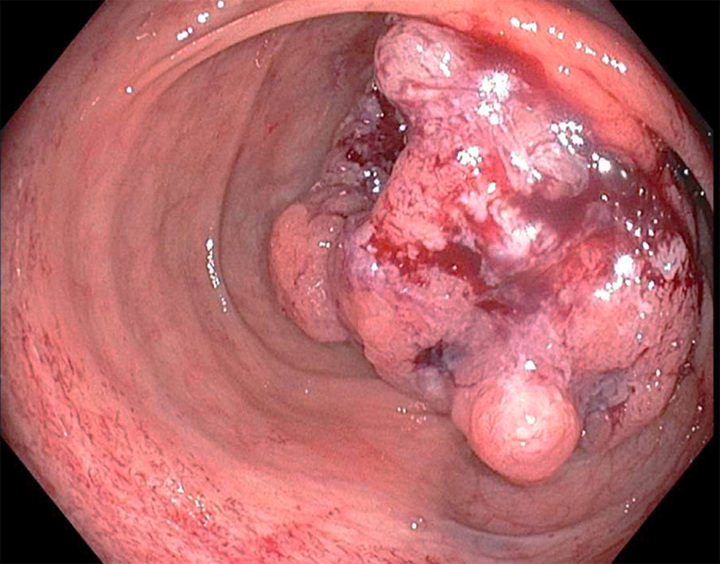
A 47-year-old man with longstanding ulcerative colitis and primary sclerosing cholangitis (PSC) presents with progressive iron deficiency and jaundice. The patient has been lost to follow-up for the past several years and has not had regular medical care. He has had pan-colonic ulcerative colitis for more than 20 years with intermittently active disease. The patient was diagnosed with PSC 7 years ago and has not had ascites, variceal bleeding, or HE, although he has become more cholestatic over the past 2 years. There are no other medical conditions. Colonoscopy shows a 4 cm, friable cecal mass and biopsy confirms adenocarcinoma of the colon (Figures 1 and 2). Imaging reveals no regional or distant metastatic disease and no ascites. Upper endoscopy shows large esophageal varices without stigmata of recent bleeding or high-risk marks. Laboratory assessment shows total bilirubin of 7.5 mg/dL (128 µmol/L), alanine aminotransferase 167 IU/L, serum creatinine 0.9 mg/dL (80 µmol/L), international normalized ratio (INR) 1.7, serum sodium 139 mmol/L, albumin 3.0 g/dL (30 g/L), and platelet count 97×109/L. The patient’s calculated Model for End-Stage Liver Disease (MELD) score is 20 and he has Child-Turcotte-Pugh (CTP) B(9) cirrhosis.
FIGURE 1.
Colon mass in the cecum of the case presentation patient.
FIGURE 2.
Colon mass after biopsy.
DISCUSSION
A strong association between PSC and inflammatory bowel disease (IBD) exists, with the overall prevalence of IBD in patients with PSC at ~70%. Various studies have demonstrated the 10-year cumulative risk for the development of colonic dysplasia and colorectal cancer in PSC-IBD of ~11%, while the risk of development of dysplasia or CRC for IBD alone is ~2%.1 The mean time interval between diagnosis of IBD and development of CRC in PSC-IBD ranges between 12–20 years. Dysplasia or CRC in PSC-IBD frequently develops in the proximal colon, with reported rates varying between 29% and 100%. For IBD alone, the development of adenocarcinoma in the rectosigmoid colon is more common in ulcerative colitis, whereas Crohn’s disease shows a more equal distribution between right-sided and left-sided colonic malignancy.1 Increasing time intervals since IBD diagnosis, extensive involvement of the colon, and advancing age are risk factors for colorectal cancer in patients with ulcerative colitis.2
Cirrhosis and the risk of surgery
It is well-established that patients with cirrhosis have a higher risk of morbidity and mortality when undergoing nonhepatic surgeries compared to the general population. The CTP score was frequently used in the past to classify the degree of liver disease, and CTP class correlates with postoperative 30-day mortality, with class A having a 10% risk, class B having a 30% risk, and class C with up to an 80% risk of death.3 The use of the CTP score in determining surgical risk has been criticized because some aspects of the scoring system are subjective, the scoring system does not take into account the specific surgical procedure, and the lack of granularity of the system leads to the overestimation of risk for many.4 The MELD score overcomes some of the shortcomings of the CTP system and has been studied in various populations and surgical scenarios, including colorectal surgery.5–7 In the first report of surgical survival correlating with MELD score, the 30-day mortality roughly increased 1% per MELD point to a MELD of 22 and afterward increased an additional 2% per MELD point.5 It should be noted that the original MELD score,8 not Model for End-Stage Liver Disease Sodium (MELD-Na) or MELD 3.0, was used. This study was limited by sample size and limited sub-analysis of various types of surgery. Later, a larger series using a similar methodology found a comparable correlation7 using more factors in addition to the MELD score in the calculation (the Mayo Mortality Risk Score). A weakness of this model was the requirement of the American Society of Anesthesiologists (ASA) class 4 (“severe systemic disease that is a constant threat to life”) to be given to all patients with decompensated cirrhosis which clearly does not universally hold true.
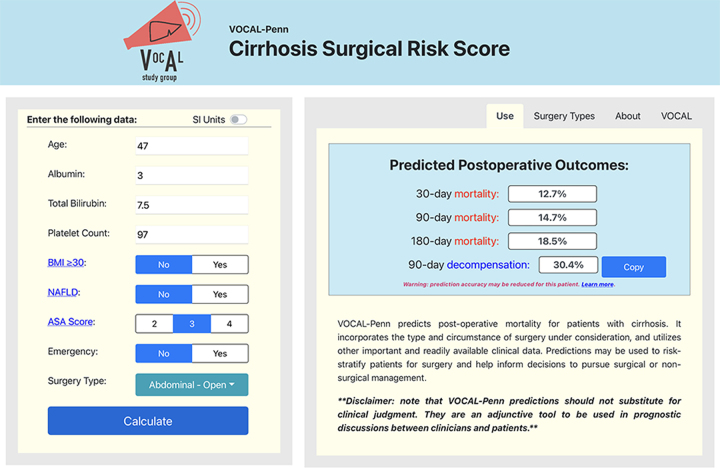
The VOCAL-PENN score is a novel tool used to predict mortality in patients with cirrhosis undergoing specific nonhepatic surgical procedures that was developed and validated in a large cohort of patients.9 It has been shown to predict mortality outcomes better than existing clinical standards, such as MELD or CTP score, and has a readily accessible online calculator for use at the bedside or in the clinics (https://www.vocalpennscore.com). Despite the large sample size and external validation,10 the VOCAL-PENN score fails to account for the stability of the patient’s liver disease over time and the level of portal hypertension. However, true ASA class is used in this model versus the Mayo Risk Score, and there are pertinent sub-analyses based on specific surgical procedures. The VOCAL-PENN score for the patient from the case presentation is shown in Figure 3.
FIGURE 3.
VOCAL-PENN preoperative risk assessment score for the patient in the case presentation. Printed with permission from https://www.vocalpennscore.com. Abbreviations: ASA, American Society of Anesthesiologists; BMI, body mass index.
Cirrhosis and general preoperative optimization
Patients with cirrhosis undergoing colorectal surgery should have a detailed preoperative evaluation, including a thorough clinical exam and laboratory tests.11 Preoperative evaluation should include standard laboratory assessments, clinical and radiographic assessment of liver disease and portal hypertension, endoscopic evaluation, cardiopulmonary evaluation, and nutritional risk stratification. A complete discussion of perioperative management of patients with liver disease is covered elsewhere.12,13 Surgical risk in cirrhosis should not be considered as an absolute yes/no or a mere “clearance” decision as risk is continuous in nature and requires an in-depth informed consent discussion between the patient, the surgical team, and the involved subspecialists. Potentially lifesaving procedures, as in the case presentation, should be presented as a balance of risks, not as an absolute statement of “clearance,” even with advanced liver disease. Patients with cirrhosis should meet with a hepatologist preoperatively to ensure there is optimal control of the liver disease and any complications of portal hypertension. For instance, these patients should be evaluated for varices, ascites, and encephalopathy. They may require preoperative endoscopy, diuretic management, and optimization of portal hypertension. Despite the perceived intuitive attraction of prophylactic preoperative TIPS in patients with cirrhosis, this practice should not be routine as it increases rates of postoperative HE and has not shown a decrease in surgical mortality.14 All scheduled surgeries in patients with advanced liver disease should be performed at centers with hepatology and surgery staff experienced in the care of these patients. Consideration of preoperative evaluation for liver transplantation, if feasible, is indicated in case of severe hepatic decompensation. Specifically in relation to colorectal surgery, if an ostomy is required during surgery, establishing care with a stoma nurse before surgery allows appropriate stoma management education and allows for optimal placement of the stoma.15
Cirrhosis and colorectal surgery
Surgical resection of colorectal cancer can be done using an open or minimally invasive approach. A retrospective case series demonstrated reduced estimated blood loss and lower Clavien-Dindo grade II complications compared to open surgery, although there was no difference in overall and disease-free survival with minimally invasive colorectal cancer surgery in patients with cirrhosis.16 Using data from the American College of Surgeons National Surgical Quality Improvement Program, Davis et al, identified 44,540 elective colorectal resection cases from 2012 to 2014 and stratified patients based on the MELD-Na17 scores—specifically MELD-Na 10–14, 15–19, and > 20. In all elective colorectal procedures, for every 1-point increase in MELD-Na score, anastomotic leak, mortality, and overall complications increased. Patients with a MELD-Na score > 20 had approximately an 11% anastomotic leak rate during partial resections. Given the elevated leak rate, these patients may benefit from a diverting loop ileostomy to mitigate the potentially devastating consequences of an anastomotic leak.12
CASE PRESENTATION
After consultation with hepatology and the liver transplant team, the patient underwent esophageal band ligation, and nonselective beta-blockers were instituted. The patient underwent total proctocolectomy with diverting ileostomy with minor surgical bleeding requiring 3 units of packed red cell transfusion during the case but recovered otherwise uneventfully (Table 1).
TABLE 1.
Some components of the evaluation of a patient with cirrhosis undergoing major nonhepatic surgery
| Preoperative medical |
| • Mortality and decompensation risk assessment using validated tools • Assessment of level of portal hypertension with imaging, laboratory tests, and endoscopy • Medical and/or procedural optimization of portal hypertension as indicated • Cardiopulmonary risk stratification • Discussion of the timing of elective procedures based on level of liver disease • Optimize nutritional status of the patient if time allows • Consideration of liver transplant evaluation before high-risk procedures if appropriate • Multidisciplinary discussion and planning with the involved consultants and surgical teams at a center experienced in the care of advanced liver disease patients |
| Perioperative |
| • Discussion of surgical approach and minimally invasive options as feasible • Consultation with anesthesiology and consideration of regional anesthesia approaches as applicable • Minimization of volume overload before, during, and after the surgery • Evidence-based assessment of bleeding risk and interventions to affect that risk • Develop postoperative pain control plan that minimizes the risk of HE exacerbation |
| Postoperative |
| • Consultation with hepatology and other needed specialists during the recovery period • Minimize volume overload and optimize diuretics management • Careful attention to renal and liver function laboratory assessments on a daily basis if indicated • Avoid protocol transfusions based on laboratory assessments unless the bleeding is ongoing • Adhere to an aggressive bowel function regimen to help with ileus and bowel function restoration • Minimize sedating medications as much as feasible to avoid flares in hepatic encephalopathy • Avoid hepatoxic medications, NSAIDs, and combination medications including acetaminophen • Arrange follow-up appointments in appropriate time windows based on the patient’s recovery and disease severity |
KEY TEACHING POINTS
Patients with colorectal cancer in the setting of PSC-IBD should be managed in a multidisciplinary fashion with input from hepatologists, anesthesiologists, surgeons, and stoma nurses.
Conditions related to liver disease and portal hypertension, such as ascites and varices, should be optimally managed by an experienced hepatologist both before surgery and in the postoperative recovery phase.
Preoperative surgical considerations should include a minimally invasive approach and the possibility of diverting loop ileostomy if a partial colectomy is performed.
Preoperative risk stratification tools are available as an adjunct to the informed consent discussion with the patient.
QUESTIONS:
- Which of the below is the most accurate statement related to the patient's underlying liver disease and risk for colon cancer surgery?
- This patient should not be offered potentially curative surgery as his risk for liver decompensation is too high
- A cancer surgery should be performed because the patient's risk for liver decompensation is less than 5% and liver transplant can be performed if needed
- Surgery in this patient is minimal risk because his platelet count and INR can be corrected with transfusions at the time of surgery
- The patient’s predicted 30-day mortality rate from this surgery is in the range of 4–13% and decompensation rate is about 30%
- Preoperative transvenous intrahepatic portosystemic shunt (TIPS) has been shown to improve survival and decrease complications in patients with portal hypertension
- To guarantee a good outcome, a patient with cirrhosis undergoing a surgical procedure should be "cleared" for the surgery by a hepatologist prior to the procedure.
- True
- False
Acknowledgments
CONFLICTS OF INTEREST
The authors have no conflicts to report.
EARN CME FOR THIS ARTICLE
Footnotes
Abbreviations: ASA, American Society of Anesthesiologists; CTP, Child-Turcotte-Pugh; IBD, Inflammatory bowel disease; INR, international normalized ratio; MELD, Model for End-Stage Liver Disease; MELD-Na, Model for End-Stage Liver Disease Sodium; PSC, Primary sclerosing cholangitis.
Contributor Information
Alejandro Torres-Hernandez, Email: Alejandro.Torres-Hernandez@nyulangone.org.
Patrick G. Northup, Email: Patrick.northup@nyulangone.org.
REFERENCES
- 1.de Vries AB, Janse M, Blokzijl H, Weersma RK. Distinctive inflammatory bowel disease phenotype in primary sclerosing cholangitis. World J Gastroenterol. 2015;21:1956–1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ekbom A, Helmick C, Zack M, Adami H-O. Ulcerative colitis and colorectal cancer. N Engl J Med. 1990;323:1228–1233. [DOI] [PubMed] [Google Scholar]
- 3.Ziser A, Plevak David J, Wiesner Russell H, Rakela J, Offord Kenneth P, Brown David L. Morbidity and mortality in cirrhotic patients undergoing anesthesia and surgery. Anesthesiology. 1999;90:42–53. [DOI] [PubMed] [Google Scholar]
- 4.Nicoll A. Surgical risk in patients with cirrhosis. J Gastroenterol Hepatol. 2012;27:1569–1575. [DOI] [PubMed] [Google Scholar]
- 5.Northup PG, Wanamaker RC, Lee VD, Adams RB, Berg CL. Model for End-Stage Liver Disease (MELD) predicts nontransplant surgical mortality in patients with cirrhosis. Ann Surg. 2005;242:244–251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hedrick TL, Swenson BR, Friel CM. Model for End-stage Liver Disease (MELD) in predicting postoperative mortality of patients undergoing colorectal surgery. Am Surg. 2013;79:347–352. [PubMed] [Google Scholar]
- 7.Teh SH, Nagorney DM, Stevens SR, Offord KP, Therneau TM, Plevak DJ, et al. Risk factors for mortality after surgery in patients with cirrhosis. Gastroenterology. 2007;132:1261–1269. [DOI] [PubMed] [Google Scholar]
- 8.Kamath PS, Wiesner RH, Malinchoc M, Kremers W, Therneau TM, Kosberg CL, et al. A model to predict survival in patients with end-stage liver disease. Hepatology. 2001;33:464–470. [DOI] [PubMed] [Google Scholar]
- 9.Mahmud N, Fricker Z, Hubbard RA, Ioannou GN, Lewis JD, Taddei TH, et al. Risk Prediction models for post-operative mortality in patients with cirrhosis. Hepatology. 2021;73:204–218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mahmud N, Fricker Z, Panchal S, Lewis JD, Goldberg DS, Kaplan DE. External validation of the VOCAL-Penn Cirrhosis Surgical Risk Score in 2 large, independent health systems. Liver Transpl. 2021;27:961–970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jadaun SS, Saigal S. Surgical risk assessment in patients with chronic liver diseases. J Clin Exp Hepatol. 2022;12:1175–1183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Coakley KM, Sarasani S, Prasad T, Steele SR, Paquette I, Heniford BT, et al. MELD-Na Score as a predictor of anastomotic leak in elective colorectal surgery. J Surg Res. 2018;232:43–48. [DOI] [PubMed] [Google Scholar]
- 13.de Goede B, Klitsie PJ, Lange JF, Metselaar HJ, Kazemier G. Morbidity and mortality related to non-hepatic surgery in patients with liver cirrhosis: A systematic review. Best Practice & Research Clinical Gastroenterology. 2012;26:47–59. [DOI] [PubMed] [Google Scholar]
- 14.Gil A, Martínez-Regueira F, Hernández-Lizoain JL, Pardo F, Olea JM, Bastarrika G, et al. The role of transjugular intrahepatic portosystemic shunt prior to abdominal tumoral surgery in cirrhotic patients with portal hypertension. Eur J Surg Oncol (EJSO). 2004;30:46–52. [DOI] [PubMed] [Google Scholar]
- 15.Forsmo HM, Pfeffer F, Rasdal A, Sintonen H, Korner H, Erichsen C. Pre- and postoperative stoma education and guidance within an enhanced recovery after surgery (ERAS) programme reduces length of hospital stay in colorectal surgery. Int J Surg. 2016;36:121–126. [DOI] [PubMed] [Google Scholar]
- 16.Zhou S, Zhu H, Li Z, Ying X, Xu M. Safety of laparoscopic resection for colorectal cancer in patients with liver cirrhosis: A retrospective cohort study. Int J Surg. 2018;55:110–116. [DOI] [PubMed] [Google Scholar]
- 17.Biggins SW, Kim WR, Terrault NA, Saab S, Balan V, Schiano T, et al. Evidence-based incorporation of serum sodium concentration into MELD. Gastroenterology. 2006;130:1652–1660. [DOI] [PubMed] [Google Scholar]