Abstract
Background:
With the exponential growth of publications in the field of investigator-initiated research/trials (IIRs/IITs), it has become necessary to employ text mining and bibliometric analysis as tools for gaining deeper insights into this area of study. By using these methods, researchers can effectively identify and analyze research topics within the field.
Methods:
This study retrieved relevant publications from the Web of Science Core Collection and conducted bioinformatics analysis. The latent Dirichlet allocation model, which is based on machine learning, was utilized to identify subfield research topics.
Results:
A total of 4315 articles related to IIRs/IITs were obtained from the Web of Science Core Collection. After excluding duplicates and articles with missing abstracts, a final dataset of 3333 articles was included for bibliometric analysis. The number of publications showed a steady increase over time, particularly since 2000. The United States, Germany, the United Kingdom, the Netherlands, Canada, Denmark, Japan, Switzerland, and France emerged as the most productive countries in terms of IIRs/IITs. The citation analysis revealed intriguing trends, with certain highly cited articles showing a significant increase in citation frequency in recent years. A model with 45 topics was deemed the best fit for characterizing the extensively researched fields within IIRs/IITs. Our analysis revealed 10 top topics that have garnered significant attention, spanning domains such as community health, cancer treatment, brain development and disease mechanisms, nursing research, and stem cell therapy. These top topics offer researchers valuable directions for further investigation and innovation. Additionally, we identified 12 hot topics, which represent the most cutting-edge and highly regarded research areas within the field.
Conclusion:
This study contributes to a comprehensive understanding of the current research landscape and provides valuable insights for researchers working in this domain.
Keywords: bibliometric, investigator-initiated trials, latent Dirichlet allocation, Web of Science
1. Introduction
Clinical research can be broadly classified into investigator-initiated research/trials (IIRs/IITs) and industry-sponsored trials (ISTs), each serving distinct purposes based on their initiators. IST primarily focuses on obtaining drug approval from regulatory authorities, whereas IIRs/IITs aim to explore scientific value and address specific clinical questions,[1] including rare diseases, comparative diagnostics or treatments, novel uses for approved drugs, new indications, or innovative administration methods.[2,3] Clinical studies can employ noninterventional (prospective or retrospective) or interventional research methods. One key advantage of IIRs/IITs is its origin from real clinical problems encountered in daily practice, allowing investigators to have greater autonomy and a closer connection to practical aspects of medicine. This aspect enhances the acceptability of IIRs/IITs among participants compared to IST. However, it is important to acknowledge certain drawbacks associated with IIRs/IITs, such as potentially less rigorous study designs and less stringent regulatory review processes and supervision. Additionally, participating centers may bear a greater burden of research costs.[4–6]
In recent years, there has been a notable shift among researchers from passive participation in industry-sponsored clinical trials to taking on the role of independent initiators of clinical research. This transition has resulted in significant contributions toward optimizing diagnostic and therapeutic decision-making and promoting innovation in clinical practices. As a result, IIRs/IITs have attracted considerable attention and discussion within the medical community. This trend has led to a rapid growth in the number of investigator-initiated trials, aiming to explore fundamental medical principles and accumulate knowledge in healthcare.[7] Several crucial factors contribute to the flourishing development of IIRs/IITs and create excellent opportunities for physicians to engage in clinical research. First, IIRs/IITs play a crucial role in addressing significant clinical questions and bridging knowledge gaps. Through their exploration of scientific inquiries, numerous important discoveries have been made. Notably, studies such as the Heyland study[8] and Professor Kistler’s CAPLA randomized clinical trial[9] exemplify the impact of IIRs/IITs, being published in prestigious journals such as Lancet and Nature Medicine, respectively. Second, recent reforms within regulatory agencies have allowed for the approval of indications based on certain IIRs/IITs, particularly in real-world clinical investigations.[10] Traditional clinical trials often have specific inclusion criteria, limiting their applicability to a narrow patient population. In contrast, IIRs/IITs research accommodates a more diverse patient population, validating treatment effectiveness in real-world scenarios.[11] Lastly, when existing treatment options prove ineffective or limited, IIRs/IITs research has the potential to explore new treatment strategies and uncover additional therapeutic possibilities through investigator-led trial designs.[12] Additionally, IIRs/IITs research also promotes collaboration between academic institutions, medical centers, and industry partners,[13] fostering interdisciplinary approaches to treatment development. Compared to the traditional drug development model, IIRs/IITs offer a faster and more efficient process for drug evaluation and clinical trials.[14] In light of these factors, the prominence of IIRs/IITs research continues to grow.
Given the exponential growth in the volume of publications regarding IIRs/IITs, text mining and bibliometric analysis have emerged as feasible methods for gaining deeper insights into this field. Consequently, the aim of this study is to outline the research topics associated with IIRs/IITs, enabling a more nuanced understanding of each topic. The results of this study not only provide a trajectory of development within the field but also elucidate understandable subdomains and thematic connections. Furthermore, the study demonstrates how each research topic branches out and integrates multiple disciplines. By considering these trends and factors, researchers can better understand the shifting dynamics within their respective research areas and make informed decisions about their research directions. This knowledge can help them navigate resource constraints and identify overlooked research opportunities, leading to advancements and groundbreaking discoveries.
2. Research method
2.1. Data collection
The research utilized the Web of Science Core Collection to search for IIRs/IITs articles until July 25, 2023. The search terms included “Investigator Initiated Research,” “Investigator Initiated Trial,” “Investigator Initiated Clinical Trial,” “Investigator Initiated Research,” and “Investigator Initiated Clinical Trials.” One investigator (L.H.) was selected for the study to conduct the literature search, and 2 investigators (L.H. and F.S.) assessed the eligibility of the studies included in the analysis, excluding duplicates, retractions, and articles without abstracts. Relevant information including the title, author, publication, publication year, abstract, and citation frequency of the included articles was downloaded and stored as an Excel file for further bibliometric analysis.
2.2. Topic modeling
Furthermore, topic modeling, a statistical tool for analyzing unstructured textual data, was employed in this study (the detailed procedure of the latent Dirichlet allocation [LDA] method is shown in Supplementary 1, Supplemental Digital Content, http://links.lww.com/MD/L831). The widely used algorithm, LDA, was utilized. LDA assigns a probability distribution of words to each topic and determines the probability that a document belongs to a particular topic.[15] The data underwent several preprocessing steps for text mining, including tokenization to separate the text into simple words, converting all words to lowercase letters, and removing special characters and punctuation marks. Nonsense and stop words (such as “a,” “an,” “is,” “the,” “of,” and “for” etc) were excluded from the analysis. Word stemming was also performed to obtain the word stem.[16] After all these preprocessing steps, the final corpus was produced with the remaining words, and LDA analysis was performed on this data set.
In topic modeling, it was crucial to determine the appropriate number of topics. Choosing too few topics resulted in broad and less informative categories, while choosing too many topics led to extremely specific but semantically insignificant categories. Therefore, finding a balance was important. Perplexity, a common tool in natural language processing models, measured how well probabilistic models predicted samples. In the context of topic modeling, lower perplexity indicated a better fit of the model to the training set data.[17] Consequently, perplexity determines the optimal number of topics K for this study. After determining the best-performing model, 2 reviewers (L.H. and F.S.) independently generated topic concepts for each topic by reviewing the top 20 keywords clustered for each topic. In cases of doubt or disagreement, the final decision on the topic concepts was made by the third most senior investigator (D.K.). Additionally, the degree of concern for each topic, referred to as topic intensity, was calculated by counting the number of documents assigned to each topic.
The weights of each topic were plotted over time to identify trends in topic impact. A linear regression model was applied to each topic, with the topic weights in corresponding years as the dependent variable and time as the independent variable. Slopes were calculated at various time points to quantitatively assess relative importance. Topics were ranked based on their change in slope, from the greatest increase to the greatest decrease. The coefficient β was particularly important in determining whether a topic was considered hot or cold, based on its significance and direction.[18] Hot topics indicate the most cutting-edge and highly regarded research directions, and selecting research directions related to them is beneficial in acquiring resources and driving research progress. On the other hand, cold topics represent research areas with hidden potential, and exploring these areas can lead to critical breakthroughs and new discoveries for independent researchers. Furthermore, to explore more complex nonlinear relationships between the research variables and time, generalized additive models were employed. generalized additive model is a nonparametric regression model that enables us to better understand potential nonlinear patterns between variables and time. To further analyze and manage the topics, they were categorized into several clusters. All statistical analyses were performed using R version 4.3.0 software.
3. Results
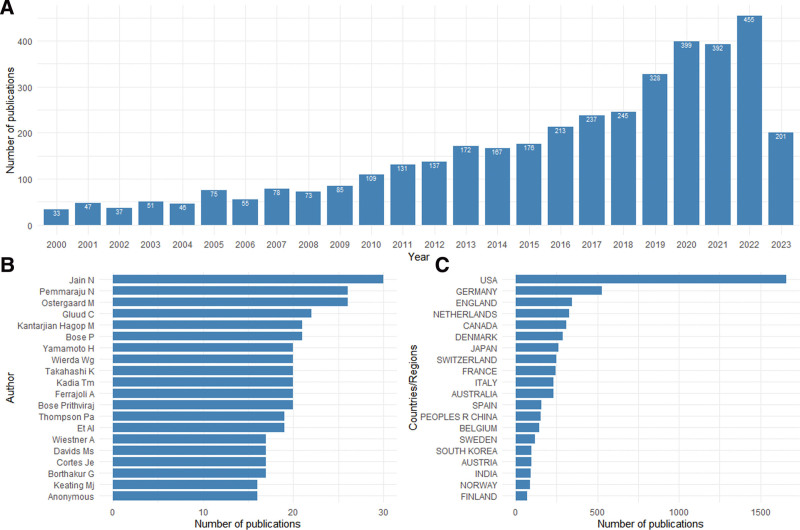
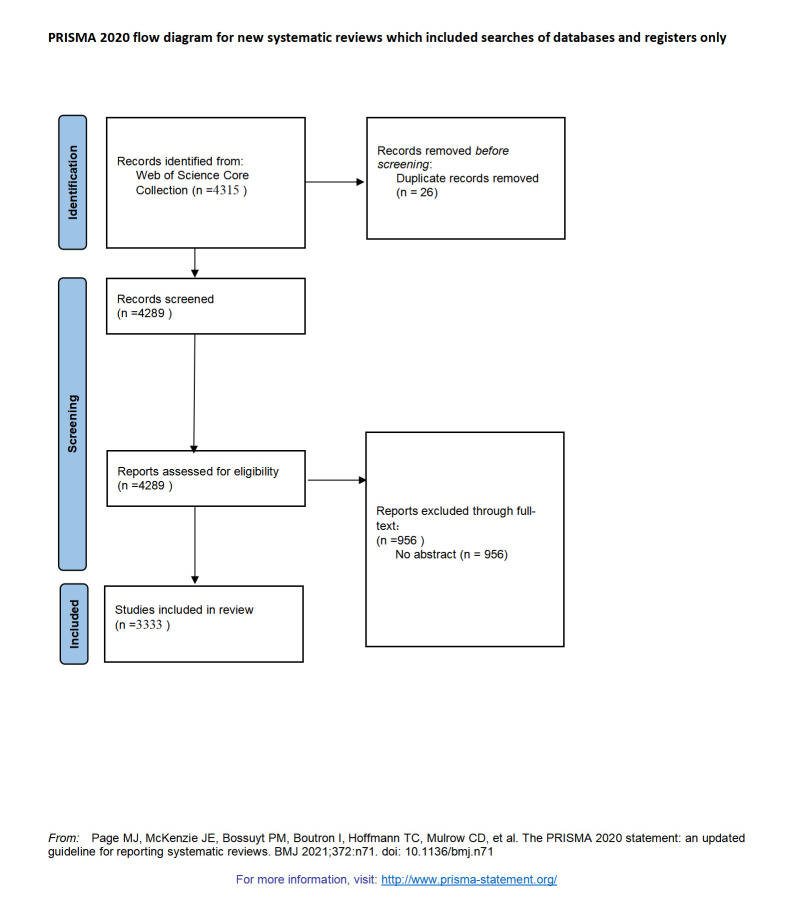
A total of 4315 articles related to IIRs/IITs were retrieved from the Web of Science Core Collection, from July 25, 2020. After excluding duplicate articles and articles with missing abstracts, a total of 3333 articles were included for bibliometric analysis (Figure S1, Supplemental Digital Content, http://links.lww.com/MD/L836). A steady increase in annual publications was observed (Fig. 1A). Additionally, we found that Jain N was the highest publishing author (Fig. 1B), and the most productive countries in terms of IIRs/IITs were the United States, Germany, the United Kingdom, the Netherlands, Canada, Denmark, Japan, Switzerland, and France (Fig. 1C). Furthermore, we analyzed the citation trends of the top 20 cited articles since 2000. The study revealed an increasing citation frequency of highly cited articles over the years. Specifically, several studies on randomized controlled clinical trials showed a sharp increase in citation frequency in recent years (Figure S2, Supplemental Digital Content, http://links.lww.com/MD/L829).
Figure 1.
Publications of studies on the investigator-initiated research/trials. (A) The number of IIT-related studies published annually since 2000. (B) Top 20 authors with most publications of investigator-initiated research/trials. (C) Top 20 countries with most publications of investigator-initiated research/trials. IIT = investigator-initiated trials.
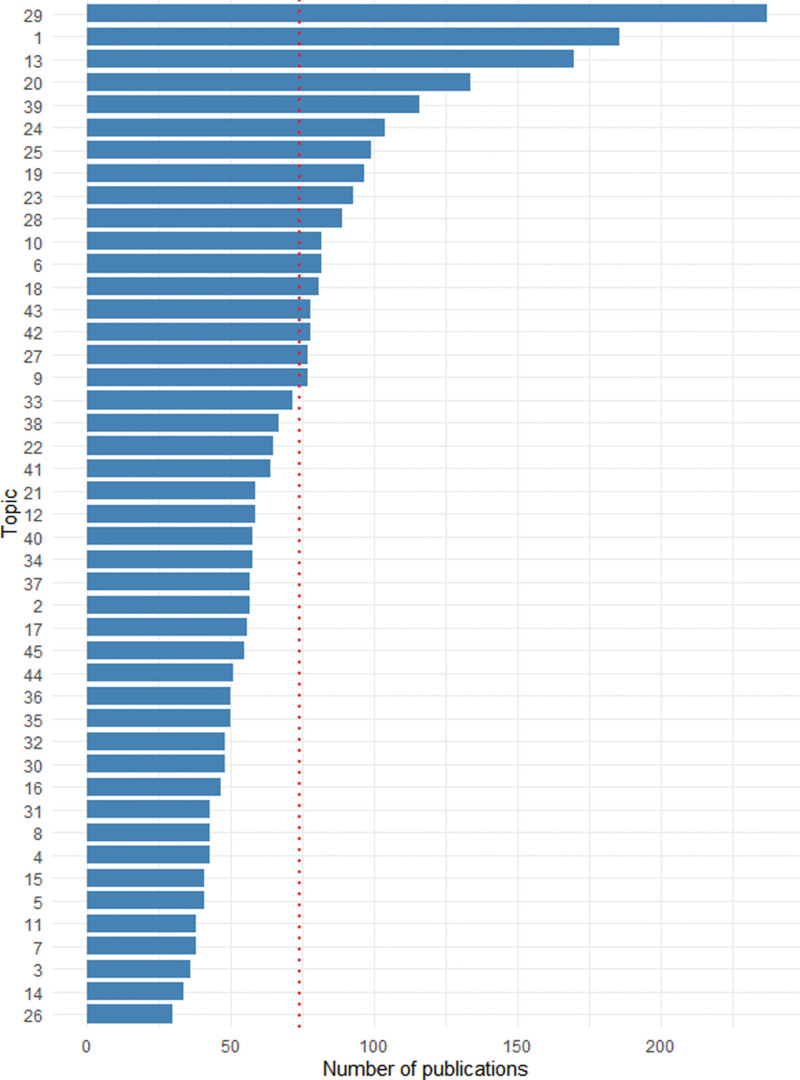
To comprehensively characterize the extensively researched fields and enhance the precision of research categorization within IIRs/IITs, we employed the LDA algorithm. Text-mining analysis was conducted using abstracts from all 3333 publications: through analysis, under the condition that other parameters are fixed, the correspondence between the perplexity degree of the LDA topic model constructed in this paper and the number of topics is shown in Figure S3, Supplemental Digital Content, http://links.lww.com/MD/L830. So, we identified the model with 45 topics as the best fit. Table 1 presents all 45 topics along with their most relevant words, while Figure 2 displays publication attribution for all topics. Our analysis revealed that, on average, each topic covered 74 publications. Notably, 18 topics had a prominence exceeding the average of 74 publications. The top 5 topics with the highest intensity were topic 29, topic 1, topic 13, topic 20, and topic 39, indicating substantial attention in the areas of cancer drug development, the impact of research and development programs on community health, survival, and therapeutic response in cancer trials, improving healthcare interventions through nursing research, informatics, and education, and randomized trials on coronary artery disease, respectively.
Table 1.
Obtained topics from the LDA.
| Name | Top 10 words | Slope | P value | Type | |
|---|---|---|---|---|---|
| Topic 1 | The Impact of Research and Development Programs on Community Health | research, develop, program, health, investig, project, collabor, communiti, support, science | −0.159 | .000 | Cold topic |
| Topic 2 | Ethical Considerations in Clinical Trials | ethic, trial, approv, dissemin, committe, randomis, analysi, outcom, peer review, include | 0.089 | .000 | Hot topic |
| Topic 3 | Comparing Oocyte Therapies for Different Disease Cycles | oocyt, therapi, respons, compar, differ, number, diseas, cycl, system, phase | −0.005 | .762 | |
| Topic 4 | Enrollment and Data Reporting in Clinical Trials | clinic, trial, enrol, data, report, particip, random, compar, investig, includ | 0.014 | .411 | |
| Topic 5 | Comparing the Effects of Pregnancy and Birth on Women and Men | women, pregnanc, differ, birth, men, compar, contrast, effect, random, outcom | 0.004 | .838 | |
| Topic 6 | Medical Data Collection and Analysis Methods | data, research, inform, investig, system, clinic, provid, method, databas, includ | −0.093 | .000 | Cold topic |
| Topic 7 | Investigating HIV Load and ART Participation Over Time | HIV, load, art, particip, time, oral, test, health, viral, investig | −0.058 | .001 | Cold topic |
| Topic 8 | Improving Atopic Dermatitis Treatment | improv, atop, subject, assess, dermat, easi, skin, investig, signific, achiev | 0.015 | .391 | |
| Topic 9 | Randomized Trials on Blood Pressure and Hypertension | random, pressur, blood, hypertens, trial, ablat, outcom, effect, compar, cathet | −0.030 | .083 | |
| Topic 10 | Exploring Stroke Outcomes | Stroke, outcom, trial, random, ischem, scale, clinic, onset, time, therapi | 0.049 | .005 | Hot topic |
| Topic 11 | Animal Models and Clinical Research Methods | system, data, investig, time, anim, standard, model, laser, research, safeti | −0.068 | .000 | Cold topic |
| Topic 12 | Clinical Trial Registry and Randomized Controlled Trials | trial, RCTs, registri, clinic, investig, exercis, data, includ, effect, random | −0.002 | .920 | |
| Topic 13 | Survival and Therapeutic Response in Cancer Trials | surviv, cancer, respons, trial, chemotherapi, phase, overal, therapi, combin, grade | 0.121 | .000 | Hot topic |
| Topic 14 | Studies and Predictive Analyses of Fractures and Osteoporosis | Fractur, model, system, investig, delay, age, predict, respons, effect, bone | −0.015 | .387 | |
| Topic 15 | HCV Genotypes and Sustained Virological Response Rates | HCV, hepat, trial, genotyp, SVR, respons, effect, data, signific, investig | −0.021 | .220 | |
| Topic 16 | Comparative Nutritional Studies | Nutrit, risk, compar, outcom, associ, mortal, clinic, hospit, signific, adjust | 0.074 | .000 | Hot topic |
| Topic 17 | Early Arthritis Treatment With Methotrexate | diseas, arthriti, joint, activ, methotrex, score, clinic, MTX, earli, therapi | 0.028 | .112 | |
| Topic 18 | Insulin Effects in Type 2 Diabetes | insulin, diabet, type, glucos, signific, effect, chang, weight, HbAc, compar | −0.003 | .858 | |
| Topic 19 | Investigating Brain Development and Mechanisms of Disease | diseas, investig, brain, develop, research, sever, gene, factor, system, mechan | −0.202 | .000 | Cold topic |
| Topic 20 | Improving Healthcare Interventions Through Nursing Research, Informatics, and Education | care, health, intervent, particip, nurs, research, inform, interview, educ, practic | −0.084 | .000 | Cold topic |
| Topic 21 | Antibiotic Therapy Trials in COPD and Sepsis | trial, antibiot, clinic, random, therapi, outcom, sepsi, CPAP, infect, COPD | 0.033 | .057 | |
| Topic 22 | COVID-19 Trials and Randomized Outcomes | trial, outcom, includ, covid, randomis, risk, effect, data, particip, review | 0.027 | .121 | |
| Topic 23 | Stem Cell Therapies in Cancer Treatment | cell, tumor, cancer, clinic, stem, therapi, trial, immun, combin, respons | −0.019 | .263 | |
| Topic 24 | Pain and Symptom Improvement Scores | score, improv, pain, symptom, effect, scale, signific, assess, chang, trial | 0.037 | .031 | Hot topic |
| Topic 25 | Research Grants and Funding Programs | Research, grant, fund, institut, program, applic, health, NIH, investig, nation | −0.059 | .001 | Cold topic |
| Topic 26 | Analysis of Drug-Gene Interactions and Their Effects | drug, gene, effect, express, data, ABUS, evalu, chang, test, includ | −0.029 | .098 | |
| Topic 27 | Site Protocol and Process in Clinical Research | research, protocol, site, process, investig, clinic, review, particip, institut, data | −0.002 | .891 | |
| Topic 28 | Studies of the Effect of Placebo in Clinical Trials | placebo, event, advers, receiv, dose, safeti, assign, plus, random, efficaci | 0.099 | .000 | Hot topic |
| Topic 29 | Cancer Drug Development | trial, clinic, drug, research, conduct, investig, develop, investigator-initi, phase, cancer | −0.056 | .001 | Cold topic |
| Topic 30 | Effect of Cognitive Therapy on Pediatric Disorders | therapi, trial, disord, effect, schizophrenia, pediatr, cognit, includ, clinic, investig | −0.019 | .269 | |
| Topic 31 | Preventive Intervention and Risk Assessment for Depression | includ, risk, depress, therapi, intervent, prevent, review, assess, light, compar | −0.009 | .624 | |
| Topic 32 | Quality Assessment and Effectiveness Trials in Psoriasis | qualiti, assess, effect, trial, psoriasi, therapi, evid, includ, outcom, particip | 0.009 | .599 | |
| Topic 33 | Ventilatory Care and Oxygen Therapy in ICU | ventil, care, ICU, oxygen, ill, critic, mechan, outcom, respiratori, intens | 0.016 | .354 | |
| Topic 34 | Research Equipment, Clinical Funding and Scientific Research Development Support | research, devic, clinic, fund, nation, institut, investig, support, translat, lung | −0.029 | .099 | |
| Topic 35 | Comparing the Effects of Inhibitors and Baseline Measurements on Statin Therapy | effect, trial, inhibitor, chang, random, baselin, clinic, compar, statin, measur | 0.030 | .086 | |
| Topic 36 | Vision Improvements Following Implant and ICD in Wound and Acuity Cases | implant, ICD, eye, visual, wound, acuiti, receiv, effect, clinic, trial | 0.002 | .914 | |
| Topic 37 | Exploring Gene Identification Data to Enhance Treatment Strategies for Infants | clinic, diseas, therapi, trial, data, investig, testosteron, infant, advanc, increas | −0.035 | .042 | Cold topic |
| Topic 38 | Randomized Valve Trials and Balloon Angioplasty | trial, primari, random, valv, clinic, balloon, outcom, stenosi, patenc, compar | 0.079 | .000 | Hot topic |
| Topic 39 | Randomized Trials on Coronary Artery Disease | trial, random, coronari, primari, bleed, risk, event, outcom, myocardi, stroke | 0.074 | .000 | Hot topic |
| Topic 40 | Cardiac Arrest and Randomized Trials | trial, cardiac, outcom, arrest, random, primari, includ, clinic, mortal, improv | 0.024 | .171 | |
| Topic 41 | Cancer Survival Rates and Responsiveness to Sorafenib | cancer, surviv, respons, diseas, carcinoma, breast, sorafenib, progress, mutat, overall | 0.057 | .001 | Hot topic |
| Topic 42 | Comparing Postoperative Surgical Outcomes in Kidney Transplants | surgeri, postop, kidney, trial, transplant, random, complic, function, compar, outcom | 0.049 | .005 | Hot topic |
| Topic 43 | Randomized Trials on Coronary Stents | stent, trial, coronari, lesion, clinic, myocardi, infarct, random, failur, cardiac | 0.077 | .000 | Hot topic |
| Topic 44 | Comparing Randomized Trials on Blood Transfusions | trial, clinic, random, bleed, transfus, primari, outcom, blood, vitamin, compar | 0.048 | .006 | Hot topic |
| Topic 45 | Vaccine Safety Investigations | vaccin, advers, safeti, event, seizur, includ, report, investig, dose, random | −0.003 | .842 |
The documentation for 2023 was excluded from the study of temporal trends due to incomplete publications for that year.
ART = antiretroviral therapy, COPD = chronic obstructive lung disease, COVID-19 = corona virus disease 2019, HCV = hepatitis C virus, HIV = human immunodeficiency virus, ICD = implantable cardioverter defibrillator, ICU = intensive care unit, LDA = latent Dirichlet allocation, NIH = National Institutes of Health.
Figure 2.
Expected topic distribution.
The field of IIRs/IITs exhibited dynamic topic evolution, with the majority of topics maintaining stability. Topics showing positive trends were referred to as “hot topics,” while those with declining trends were considered “cold topics.” Table 1 presents the trends of hot topics along with their top relevant words. Notably, out of the 12 hot topics, the following 4 topics have remained hot for years: survival and therapeutic response in cancer trials, studies of the effect of placebo in clinical trials, ethical considerations in clinical trials, and randomized trials on coronary artery disease. However, 9 topics (topic 1, topic 19, topic 6, topic 20, topic 11, topic 25, topic 7, topic 29, and topic 37) demonstrated a downward trend. Among them, topic 1, topic 19, topic 20, topic 25, and topic 29 accounted for a large proportion of all topics but showed a decline in prominence. The additive model also revealed the same dynamic pattern of thematic evolution (Figure S4, Supplemental Digital Content, http://links.lww.com/MD/L837).
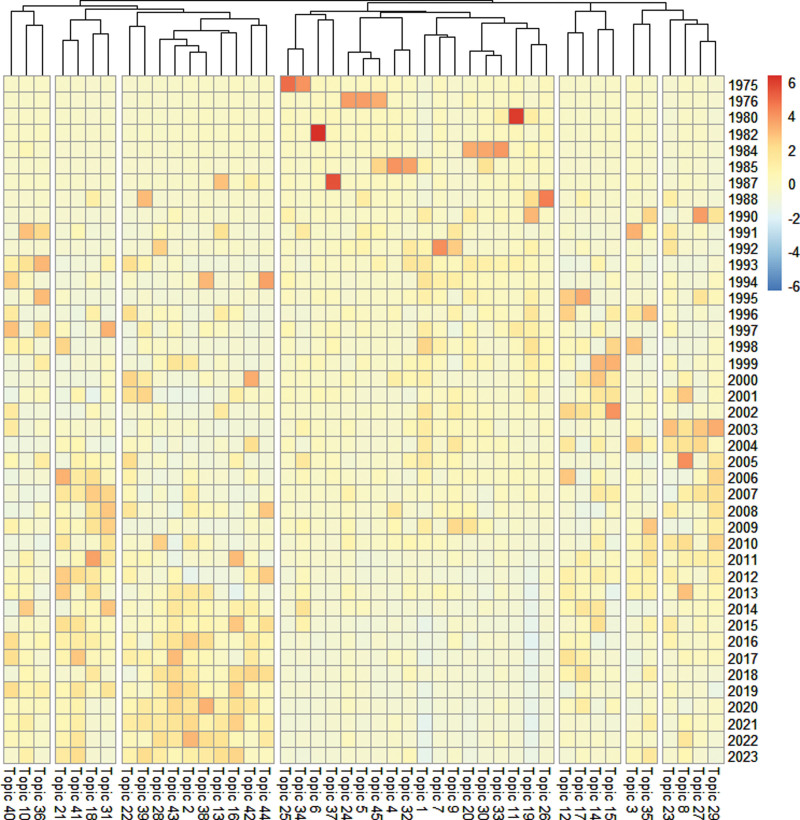
To further categorize the 45 topics, they have been analyzed and grouped into 7 clusters for analysis and management (Fig. 3). The clusters identified are as follows: cluster 1: researching the effectiveness of cardiovascular disease treatments, cluster 2: evaluation of clinical trials and treatment methods for infection, cancer, diabetes, and depression, cluster 3: evaluation of clinical trials and treatment methods for corona virus disease 2019, coronary artery disease, cancer, and kidney transplantation, cluster 4: clinical trial data analysis and interpretation, cluster 5: comparative studies of clinical trials and treatment strategies in disease management, cluster 6: Researching cutting-edge applications and evaluating their effects, cluster 7: data management and optimization of clinical trial processes. Further clustering analysis reveals that despite the broad range of topics, the primary focus remains on clinical trials for key diseases (clusters 1–6), clinical trial data analysis and interpretation (cluster 4), as well as data management and optimization of clinical trial processes (cluster 7).
Figure 3.
Heat map of the topics by year.
4. Discussion
The number of publications has increased gradually since 2000. This trend indicates that more researchers, medical institutions, and organizations have dedicated their efforts to IIRs/IITs, aiming to address the clinical demands across various fields and patient groups. The continuous advancement and application of IIRs/IITs have been facilitated by this ongoing trend. In this study, we utilized the widely-accepted topic modeling method, namely the LDA model, to extract a total of 45 pertinent topics. Through the examination of the significance and temporal variations of these topics, we have drawn the following conclusions:
Ten top topics currently hold a prominent position in IIRs/IITs. These topics span multiple domains, such as community health, cancer treatment, brain development, nursing research, and stem cell therapy. They have garnered significant attention and extensive investigation in scholarly literature. Researchers who choose research directions related to these top topics can build upon existing research findings and make headway into cutting-edge issues. For example, within the domain of community health, researchers can explore various dimensions influencing community health and propose innovative solutions to improve the well-being of community residents.[19,20] In the field of cancer treatment, researchers can expand upon existing research to explore novel treatment methods, targeted drugs, and immunotherapies that enhance patient survival rates and overall quality of life.[21] Moreover, by enhancing healthcare practices, researchers can bolster the efficacy and quality of medical interventions. In nursing research, for instance, researchers can focus on innovation and improvement in nursing techniques to elevate the quality of care, reduce complication rates, and provide guidance for clinical practice.[22] Furthermore, patient informatics and education play pivotal roles in enhancing the quality of healthcare services.[23] In summary, the top topics identified in our analysis hold significant influence within current research. Selecting research directions aligned with these topics can leverage existing research and drive cutting-edge progress. Through interdisciplinary collaborations and specific research efforts, researchers can foster the growth of their respective disciplines and enhance healthcare interventions, ultimately advancing the quality of healthcare services.
Twelve topics have exhibited a significant increase in attention, highlighting the most notable issues in the current research field. These topics encompass ethical considerations, stroke research, comparative nutritional studies, cancer survival rates, and response to sorafenib, as well as randomized trials on coronary artery stenting, among others. Opting for research directions related to these hot topics can streamline researchers’ access to funding and resource support, enabling faster application of research findings in practical settings. Notably, 4 of these hot topics fall within the top 10 key topics. The first key topic focuses on “survival and therapeutic response in cancer trials,” as cancer is a critical public health concern that receives substantial global attention and research efforts,[24] examining survival rates and therapeutic responses in cancer trials contributes to the development of clinical practice guidelines and the improvement of treatment outcomes. The second key topic delves into “pain and symptom improvement scores,” alleviating pain and improving symptoms are crucial aspects in the treatment of various diseases. Given the significant impact of pain relief and symptom improvement on patients’ well-being, assessing treatment outcomes and enhancing their overall quality of life is of utmost importance.[25,26] The third key topic, “studies of the effect of placebo in clinical trials,” aims to enhance understanding of the effects and responses to placebos, thereby aiding in the scientific foundation for designing clinical trials. Adapting clinical trial design to evolving ethical requirements is essential, given the increasing attention and ongoing discussions surrounding the use of placebos in trials.[27] Lastly, the 4th key topic, as mentioned previously, focuses on “randomized trials on coronary artery heart disease,” coronary artery disease is a prevalent global health issue, making relevant randomized trials imperative. Evaluating primary outcomes, bleeding risks, and stroke events in coronary artery heart disease trials holds paramount significance in improving treatment outcomes and enhancing the quality of life for patients.[28,29] By prioritizing research directions aligned with these key topics, researchers can contribute to advancements in these critical areas of study, improve patient outcomes, and enhance overall healthcare practice. Additionally, there are other rapidly developing topics such as “ethical considerations in clinical trials,” “comparative nutritional studies,” and “comparing randomized trials on blood transfusions.” Although these topics have smaller volumes, their high growth rates suggest their emergence as important areas for future research.
Among the top 10 topics, 5 exhibit a noticeable declining trend. This suggests that as research delves deeper and progresses, attention may gradually shift from broad issues to more specific and exact ones. For example, in the study of brain development and disease mechanisms, as progress is made in researching genetic factors and severe cases, the focus may start to shift toward other specific issues related to brain development and diseases. Moreover, the level of attention given to certain topics can be influenced by factors such as resource allocation and policy decisions. For instance, if funding from organizations like the National Institutes of Health or other funding agencies is reduced for national health surveys, or if there are adjustments in research funding allocation strategies, researchers may choose to redirect their focus to other research areas that offer greater financial support.[30,31] Additionally, inadequate resource allocation and limited recognition of the importance of nursing research and practice in certain regions or institutions may lead researchers to prioritize other research areas that are easier to obtain support and recognition.[32,33] Lastly, we identified a “cold” topic that encompasses topics such as medical data collection and analysis methods, human immunodeficiency virus load and participation in antiretroviral therapy, animal models, and gene identification data to enhance treatment strategies for infants. For independent researchers, these cold topics present opportunities to achieve breakthroughs and make new discoveries by exploring the untapped potential in these fields.
We summarize several key factors that have been identified as contributing to changes in the trends of these topics over time. First, the level of public concern greatly influences the trajectory of these topics. These hot topics encompass significant issues that capture the interest of the general public, such as cancer, cardiovascular diseases, and pain. Given their direct impact on personal well-being and quality of life, they have garnered widespread attention. Second, scientific progress plays a pivotal role in shaping the evolution of these topics. In the past few decades, the medical and scientific communities have made remarkable strides, making research in these areas more feasible and meaningful. The emergence of novel technologies and methodologies has empowered researchers with superior tools to explore these domains and achieve groundbreaking discoveries. Lastly, policy and financial support are crucial factors in determining the significance of these topics. Governments and organizations recognize the importance of these issues, leading to increased funding and support for relevant research. As a result, researchers can conduct more comprehensive and in-depth investigations, accelerating scientific progress and the practical application of research findings. Further, after conducting further cluster analysis of these 45 topics, it became evident that they cover a wide range of areas, predominantly focusing on clinical trials, statistical analysis, data management, and trial processes. IIRs/IITs play a crucial role in evaluating the safety and efficacy of new drugs, treatments, or vaccines. Statistical analysis facilitates researchers in drawing accurate conclusions from collected data by assessing treatment effects and identifying disease-associated factors. Additionally, the proper execution of research procedures ensures the reliability and scientific validity of the obtained results.
However, our study does have certain limitations that should be acknowledged. First, while the LDA method enables the identification of topic trends and popularity using data-driven approaches, it does not directly provide explanations or reveal the underlying reasons and trends behind these topics. Therefore, further research is needed to explore the factors driving the emergence and growth of these topics. Second, our decision to use journal abstracts instead of full-text articles limits the size of our corpus. Access to the full text of original research articles is not always available, which may impact the comprehensiveness of our analysis. Future studies could consider incorporating more comprehensive datasets to obtain a more holistic understanding of the research landscape. Lastly, our study relies on human interpretation of symbols to identify topic models and names. While this approach allows for a deeper understanding of the topics, it is subjective and potentially prone to bias.
To sum up, by discussing various topics, we draw the following insights for current IIRs/IITs: top topics represent areas that have received widespread attention, and researchers can continue to explore and deepen their understanding within these fields. The analysis of hot and cold topics provides valuable insights into the current research landscape. Researchers should consider aligning their research directions with these top topics and emerging areas to contribute to advancements, improve patient outcomes, and enhance healthcare practice.
5. Conclusion
In conclusion, the utilization of text mining and bibliometric analysis in this study has provided valuable insights into the research topics within the field of IIRs/IITs. This comprehensive analysis not only presents an overview of these topics but also reveals their interconnections and how they have evolved over time. Researchers can leverage this knowledge to navigate the complexities of IIRs/IITs, identify emerging trends, and make informed decisions about their research directions. By doing so, they can contribute to the advancement of the field and drive meaningful progress in IIRs/IITs. Furthermore, these findings have practical implications for both the academic and practical domains, providing guidance on how to apply the research discoveries in real-world settings. Finally, based on the analysis conducted, recommendations for future research directions can be made in order to address the remaining gaps and further enhance our understanding of the field.
Author contributions
Conceptualization: Litao Huang, Deying Kang.
Data curation: Litao Huang.
Formal analysis: Litao Huang.
Funding acquisition: Deying Kang.
Investigation: Dan Hu.
Methodology: Litao Huang, Fanfan Shi.
Software: Fanfan Shi.
Validation: Litao Huang, Dan Hu.
Visualization: Fanfan Shi.
Writing—original draft: Litao Huang, Deying Kang.
Writing—review & editing: Deying Kang.
Supplementary Material


Abbreviations:
- ART
- antiretroviral therapy
- CHD
- coronary artery heart disease
- COVID-19
- corona virus disease 2019
- GAM
- generalized additive models
- HIV
- human immunodeficiency virus
- IIRs/IITs
- investigator-initiated researchs/trials
- IST
- industry-sponsored trials
- LDA
- the latent Dirichlet allocation
- NIH
- National Institutes of Health
The data that supports the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Digital Content is available for this article.
The authors have no conflicts of interest to disclose.
All the work was developed using published/preprint data.
This research did not involve any direct participation from patients or the public.
This work was supported by the National Clinical Research Center for Geriatrics, West China Hospital, Sichuan University (Z20192005); 1.3.5 project for disciplines of excellence, West China Hospital, Sichuan University (ZYGD23002).
How to cite this article: Huang L, Shi F, Hu D, Kang D. Analysis of research topics and trends in investigator-initiated research/trials (IIRs/IITs): A topic modeling study. Medicine 2024;103:10(e37375).
Contributor Information
Litao Huang, Email: huanglitao@wchscu.cn.
Fanfan Shi, Email: shifanfan0706@163.com.
References
- [1].Suvarna V. Investigator initiated trials (IITs). Perspect Clin Res. 2012;3:119–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Stephenson D, Ollivier C, Brinton R, et al. Can innovative trial designs in orphan diseases drive advancement of treatments for common neurological diseases? Clin Pharmacol Ther. 2022;111:799–806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Tanemura N, Sasaki T, Sato J, et al. Study factors associated with the incompletion of clinical trials that include pediatric patients: a retrospective analysis of the European Clinical Trials Database and a lesson from the European region. Trials. 2021;22:204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Nury E, Bischoff K, Wollmann K, et al. Impact of investigator initiated trials and industry sponsored trials on medical practice (IMPACT): rationale and study design. BMC Med Res Methodol. 2020;20:246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Heravi AT, Henn A, Deuster S, et al. Investigational medicinal products, related costs and hospital pharmacy services for investigator-initiated trials: a mixed-methods study. PLoS One. 2022;17:e0264427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Herfarth HH, Jackson S, Schliebe BG, et al. Investigator-initiated IBD trials in the United States: facts, obstacles, and answers. Inflamm Bowel Dis. 2017;23:14–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Yildirim O, Gottwald M, Schueler P, et al. Opportunities and challenges for drug development: public-private partnerships, adaptive designs and big data. Front Pharmacol. 2016;7:461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Heyland DK, Patel J, Compher C, et al.; EFFORT Protein Trial Team. The effect of higher protein dosing in critically ill patients with high nutritional risk (EFFORT Protein): an international, multicentre, pragmatic, registry-based randomised trial. Lancet. 2023;401:568–76. [DOI] [PubMed] [Google Scholar]
- [9].Kistler PM, Chieng D, Sugumar H, et al. Effect of catheter ablation using pulmonary vein isolation with vs without posterior left atrial wall isolation on atrial arrhythmia recurrence in patients with persistent atrial fibrillation: the CAPLA randomized clinical trial. JAMA. 2023;329:127–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Feinberg BA, Gajra A, Zettler ME, et al. Use of real-world evidence to support FDA approval of oncology drugs. Value Health. 2020;23:1358–65. [DOI] [PubMed] [Google Scholar]
- [11].Gray DM, II, Nolan TS, Gregory J, et al. Equity in digestive health diversity in clinical trials: an opportunity and imperative for community engagement comment. Lancet Gastroenterol Hepatol. 2021;6:605–7. [DOI] [PubMed] [Google Scholar]
- [12].Istaiti M, Revel-Vilk S, Becker-Cohen M, et al. Upgrading the evidence for the use of ambroxol in Gaucher disease and GBA related Parkinson: investigator initiated registry based on real life data. Am J Hematol. 2021;96:545–51. [DOI] [PubMed] [Google Scholar]
- [13].McDonald HG, Cassim EB, Harper MM, et al. The development of investigator-initiated clinical trials in surgical oncology. Surg Oncol Clin N Am. 2023;32:13–25. [DOI] [PubMed] [Google Scholar]
- [14].Mudaranthakam DP, Phadnis MA, Krebill R, et al. Improving the efficiency of clinical trials by standardizing processes for investigator initiated trials. Contemp Clin Trials Commun. 2020;18:100579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Jelodar H, Wang Y, Yuan C, et al. Latent Dirichlet allocation (LDA) and topic modeling: models, applications, a survey. Multimed Tools Appl. 2019;78:15169–211. [Google Scholar]
- [16].Yan J. Text mining with R: a tidy approach. Nat Lang Eng. 2022;28:137–9. [Google Scholar]
- [17].Zhao W, Chen JJ, Perkins R, et al. A heuristic approach to determine an appropriate number of topics in topic modeling. BMC Bioinf. 2015;16:S8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Jung YJ, Kim Y. Research trends of sustainability and marketing research, 2010-2020: topic modeling analysis. Heliyon. 2023;9:e14208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Kandiah J, Lo T, Jin D, et al. A community-based pancreatic cancer screening study in high-risk individuals: preliminary efficacy and safety results. Clin Transl Gastroenterol. 2022;13:e00516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Poulsen CH, Egmose CH, Ebersbach BK, et al. A community-based peer-support group intervention “Paths to EvERyday life” (PEER) added to service as usual for adults with vulnerability to mental health difficulties—a study protocol for a randomized controlled trial. Trials. 2022;23:727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Saesen R, Lacombe D, Huys I. Real-world data in oncology: a questionnaire-based analysis of the academic research landscape examining the policies and experiences of the cancer cooperative groups. ESMO Open. 2023;8:100878–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Backhouse A, Ogunlayi F. Quality improvement into practice. BMJ. 2020;368:m865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Otokiti A. Using informatics to improve healthcare quality. Int J Health Care Qual Assur. 2019;32:425–30. [DOI] [PubMed] [Google Scholar]
- [24].Etemadi A, Safiri S, Sepanlou SG, et al. The global, regional, and national burden of stomach cancer in 195 countries, 1990-2017: a systematic analysis for the Global Burden of Disease study 2017. Lancet Gastroenterol Hepatol. 2020;5:42–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Fink R. Pain assessment: the cornerstone to optimal pain management. Proc (Bayl Univ Med Cent). 2000;13:236–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Mori N, Hosomi K, Nishi A, et al. Difference in analgesic effects of repetitive transcranial magnetic stimulation according to the site of pain. Front Hum Neurosci. 2021;15:786225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Enck P, Klosterhalfen S. Placebos and the placebo effect in drug trials. Handb Exp Pharmacol. 2019;260:399–431. [DOI] [PubMed] [Google Scholar]
- [28].Ralapanawa U, Sivakanesan R. Epidemiology and the magnitude of coronary artery disease and acute coronary syndrome: a narrative review. J Epidemiol Glob Health. 2021;11:169–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Dezsi CA, Dezsi BB, Dezsi DA. Antithrombotic treatment in anticoagulated atrial fibrillation patients undergoing percutaneous coronary intervention. Eur J Intern Med. 2017;40:1–7. [DOI] [PubMed] [Google Scholar]
- [30].Teufel-Shone NI, Schwartz AL, Hardy LJ, et al. Supporting new community-based participatory research partnerships. Int J Environ Res Public Health. 2019;16:44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Devaiah A, Murchison C. Characteristics of NIH- and industry-sponsored head and neck cancer clinical trials. Laryngoscope. 2016;126:E300–3. [DOI] [PubMed] [Google Scholar]
- [32].Mills A. Health care systems in low- and middle-income countries. N Engl J Med. 2014;370:552–7. [DOI] [PubMed] [Google Scholar]
- [33].Hatfield LA, Kutney-Lee A, Hallowell SG, et al. Fostering clinical nurse research in a hospital context. J Nurs Adm. 2016;46:245–9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.