Abstract
Hepatocellular carcinoma (HCC), a considerable leading cause of cancer-related deaths worldwide, is the most common primary liver cancer with poor prognosis and outcome. Many advances in prevention, screening, and new technologies in diagnostics and therapy have been achieved, but its incidence and mortality remain increasing. Co-infection of another viral disease in HCC patients with pregnancy might exacerbate the condition and double the mortality rate. The aim of this case report was to describe the co-infection of coronavirus disease 2019 (COVID-19) in an HCC patient during pregnancy. A 26-year-old woman with 16–17 weeks of gestation was admitted to Dr. Zainoel Abidin Hospital, Banda Aceh, Indonesia with shortness of breath. The patient also reported that the abdomen expanded rapidly in the last three weeks, followed by severe pain and collateral vein appearance. Laboratory findings revealed anemia, leukocytosis, HBsAg reactive, hypoalbuminemia, hyperbilirubinemia, elevated liver enzymes, increased alpha-fetoprotein (AFP), and cancer antigen 125 (CA-125). Ultrasonography indicated gestation with a single fetus, an enlarged liver with a 9.9 × 9.4 cm nodule, and massive ascites. The patient was also RT-PCR-confirmed COVID-19. On day 8 of hospitalization, the patient suddenly reported severe abdominal pain. Ultrasonography revealed fetal distress immediately followed by fetal death. Adequate management of cancer pain, continuous evacuation of ascites, and other supportive care could not save the patient who died on the day 17 of hospitalization. In this case, we found no proof that the patient experienced cirrhosis prior to HCC. Pregnancy through hormonal alteration is thought to be the aggravating factor that accelerates the progression of pre-existing liver disease into carcinoma and infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) worsened the outcome in this patient.
Keywords: Hepatocellular carcinoma, HCC, COVID-19, SARS-CoV-2, pregnancy
Introduction
Hepatocellular carcinoma (HCC) is the most common primary carcinoma of the liver that attributes to a poor prognosis and contributes significantly to the annual mortality rate worldwide [1]. Many achievements in terms of prevention, early detection, diagnostics, and therapy have been achieved but the HCC incidence increases over time. In 2020, Global Cancer Statistics (GLOBOCAN) recorded the liver cancer as the third most common cause of cancer death (8.3%), with its incidence was 905,000 cases and a total of 830,000 deaths [2]. The highest prevalence was reported in Asia and Africa with an incidence rate of >20 per 100,000 population [2]. A higher prevalence is most commonly found in hepatitis B endemic regions and HCC could be found in a younger population due to the fact that most hepatitis B infections are transmitted vertically during pregnancy and delivery [3]. In Indonesia, until 2020, at least 21,392 cases of HCC cases were documented, making HCC the fourth cancer with the highest rate of mortality [4]. The latest data from National Basic Health Research (Riset Kesehatan Dasar), the prevalence of hepatitis B in Indonesia until 2020 was 7.5% or approximately 17.5 million [5] Among individuals with hepatitis B infection patients, 20–30% will develop cirrhosis and/or HCC; therefore, it is estimated that 3.5–5.2 million Indonesians have the risk of having HCC in the future [5].
HCC during pregnancy is very uncommon. However, HCC in pregnancy worsens the prognosis of HCC. According to the available data, the mean age of pregnant women with HCC is 28.9±4.4 years and the majority of cases ended up in death [6].
Coronavirus disease 2019 (COVID-19) is an acute respiratory syndrome caused by infection of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) which has caused a devastating pandemic and pregnant women are among the populations that are more vulnerable to the severe disease and death [7]. Pregnancy altered the immune system response to infectious agents, including SARS-CoV-2, making this population becomes prone to advance severe COVID-19 [8,9]. Studies assessing the impact and outcome of COVID-19 in pregnant women found that women who are infected with SARS-CoV-2 during pregnancy have higher morbidity and mortality rates, particularly those who own pre-existing co-morbidities [8,10]. The morbidities caused by this condition include preeclampsia, premature birth, and stillbirth [8,10].
COVID-19 in HCC with pregnancy in very young age has not been ever reported previously. The aim of the case report was to describe the case of co-infection of COVID-19 in an HCC patient during pregnancy. There are many questions that have not been fully understood in such complicated conditions such as how could HCC occur at a very young age without any manifestations of clinical symptoms before pregnancy; how does the disease progression worsen rapidly when the patient is pregnant; and what is the role and impact of COVID-19 in pregnancy with HCC.
Case
A 26-year-old woman was referred from Simeuleu General Hospital, Aceh to Dr. Zainoel Abidin Hospital, a provincial referral hospital in Banda Aceh, Indonesia with the main complaint of dyspnea for one month which had been worsening within the last two weeks. The patient had orthopnea, dyspnea and non-productive cough with no history of paroxysmal nocturnal dyspnea. The patient had the third pregnancy at 16–18 weeks of gestation. The patient easily became tired since the early pregnancy and was getting worse rapidly. The jaundice was observed in the last two weeks before hospital admission. Prior history of jaundice was denied. There was no history of blood transfusion or usage of certain drugs.
The abdomen distended rapidly in the last three weeks followed by bilateral pretibial edema. In the beginning, the distended abdomen was painless but the pain started as the distension progressed and the intensity of the pain increased gradually. Nausea and vomiting, followed by appetite loss were also reported from one week before the admission. The patient noticed that the urine was darker, but the daily volume did not change.
The pregnancy and delivery of the first child was normal but the second child born premature with gestational age 6.5 months. The patient used to take hormonal contraception (injection and implants) between the three pregnancies. The patient was a housewife, denied of having multiple sex partners, and had no history of hepatitis B vaccination. The patient was the third child of four. The three siblings did not have a history of the same disease as the patient. The patient’s father passed away due to serious injuries, while the mother died at the age of 50’s due to breast cancer. The patient’s maternal grandmother also died from uterine cancer. Her paternal uncle had a history of hepatitis B and suffered from an enlarged abdomen and jaundice before died.
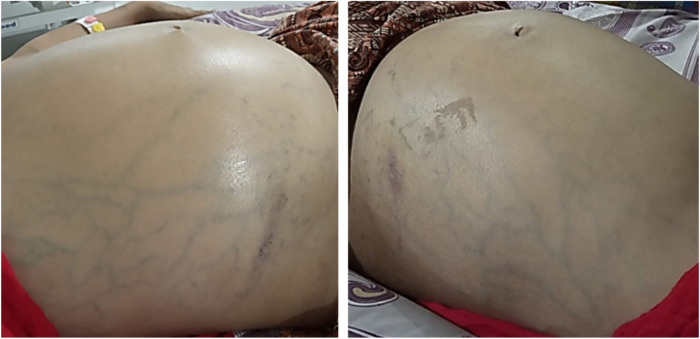
At the admission, the vital signs were in the normal range, except the respiratory rate was 30 times per minute with a numerical rating scale (NRS) of pain scale was 6 (moderate pain). Physical examination showed pale inferior palpebral conjunctiva and icteric sclera; distended and tender abdomen with obvious collateral veins (Figure 1). The tenderness was mainly felt in the epigastric region and extended to the right hypochondria and umbilicus region. Leopold’s examination of gynecology could not be assessed. Bilateral pretibial pitting edema was found.
Figure 1. Abdominal distension with overt collateral vein was seen in the patient in this case.
Laboratory examination revealed anemia (hemoglobin (Hb) 7.5 g/dL), leukocytosis (12.15x103/mm3, segment neutrophils 83%), normal platelets (439x103/mm3), normal coagulation function (international normalized ratio (INR) 1.18 seconds and activated partial thromboplastin time (APTT) 1.12 seconds), reactive HBsAg, inversed albumin-globulin ratio (albumin 2.6 g/dL and globulin 3.7 g/dL), hyperbilirubinemia (direct bilirubin 5.56 and indirect bilirubin 4.3), elevated transaminases enzyme (aspartic aminotransferase (AST) 456 U/L, alanine aminotransferase (ALT) 136 U/L), normal renal function test, and normal electrolytes. There was a significant increase of alpha-fetoprotein (AFP) (10,000 U/ml) and cancer antigen 125 (CA-125) (431 ng/mL).
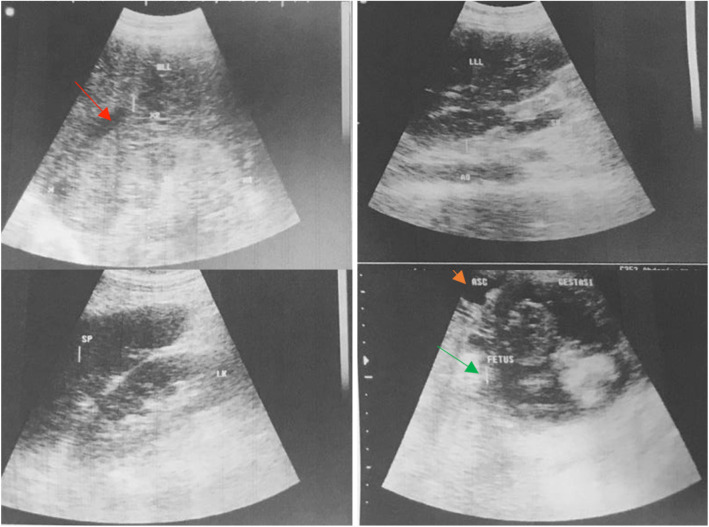
Ultrasonography (USG) showed a pregnancy with a single fetus at 16–18 weeks of gestation, an enlarged liver with increased parenchymal echogenicity, irregular edges, and dilated intrahepatic bile duct (IHBD), extrahepatic bile duct (EHBD), portal vein, and hepatic vein (Figure 2). A single nodule in the liver was also seen with a size of 9.9 × 9.4 cm and massive free fluid in the abdominal cavity (ascites). A three-phase abdominal computed tomography (CT) scan could not be performed because of the inability of the patient to lie down in a supine position and also the presence of the pregnancy.
Figure 2. Ultrasonography showed a 9.9 × 9.4 cm liver nodule (red arrow), ascites (yellow arrow), and a living fetus (green arrow).
We diagnosed the patient with HCC, chronic hepatitis B, moderate hypochromic microcytic anemia, moderate hypoalbuminemia, and G3P2A0 gravida of 16–18 weeks of gestation. We found it hard to assess the HCC based on the Barcelona Clinic Liver Cancer (BCLC) staging system due to the lack of supporting data due to such circumstance.
During the hospitalization, the fetus and mother’s conditions were closely monitored and adequate pain management was provided. After two days of being hospitalized, the patient was also diagnosed with RT-PCR confirmed COVID-19. The shortness of breath was getting worse and the abdomen distended more rapidly. Venous thromboembolism prophylactic therapy was initiated with fondaparinux injection because the D-dimer increased significantly (>4000 U). Paracentesis was performed to evacuate the ascites fluid and 3000 cc of hemorrhagic ascitic fluid was drained initially. Since aggressive production of ascitic fluid, a pigtail catheter was inserted at the left inside to permit the evacuation of the ascitic fluid regularly. At least, one liter of hemorrhagic fluid was drained daily.
The fetus was closely monitored by the obstetrician and gynecologist. In the first week, the fetus was healthy and well-developed. Although the mother suffered from dyspnea, there were no signs of fetal distress. However, on the 13th day of hospitalization, the patient suddenly reported severe abdominal pain; fetal distress was confirmed and rapidly followed by spontaneous abortion.
After that unexpected event, the patient suffered from deep sorrow and the provided maximal supportive cares did not significantly improve the clinical conditions in terms of pain, refractory of ascites, and shortness of breath. The family decided to get the patient back home. Right after the patient arrived in the hometown, the family informed that the patient deteriorated rapidly. The shortness of breath and jaundice got worse. The patient was then admitted to the local hospital and died immediately just after arrived in the hospital.
Discussion
HCC is the most common liver malignancy and is one of the main causes of cancer-related deaths worldwide. Cirrhosis of the liver is a risk factor that plays a remarkable role in the progression to HCC. Hepatitis B virus (HBV) and hepatitis C virus (HCV) are most common viruses that cause liver cirrhosis are independent risk factors for the development of HCC [11,12]. HBV, a circular DNA virus with 8 genotypes (A-H), is transmitted horizontally through contact with contaminated blood, needles, or sexual contact but vertical transmission from mother to fetus during pregnancy or childbirth remains the main cause of HBV infection worldwide [13]. It is estimated that at least 5% of the world population has been infected with HBV [14]. In our case, the patient was known to have a family history of chronic hepatitis B; therefore, vertical transmission from the mother was the most likely cause of hepatitis B in this patient.
Epidemiological studies have proven the hepatocarcinogenicity of the hepatitis viruses [11,15,16]. Hepatitis B carriers have a 10–25% risk of developing HCC over their lifetime [17]. Unlike other causes of chronic hepatitis, HBV is special for its ability to trigger HCC development without passing the cirrhosis phase. Genotype C infection is associated with a higher risk of triggering progression to HCC than genotypes A, B, and D [18]. Active infection in HBV carrier patients is an independent risk factor for HCC, where HBV DNA >105/mL copy is associated with 2.5–3 times increased risk of developing HCC in the next 8–10 years [18]. HBsAg is not the only biomarker that significantly predicts the risk of progression to HCC. Patients who are anti-HBc positive but HBsAg negative are also at risk of developing HCC. The hepatocarcinogenicity of HBV can be significantly reduced with the use of hepatitis B antivirals. Suppression of viral replication played a significant role in reducing the risk of progression to HCC in a 5-year prospective evaluation from 13.7% (control) to 3.7% [19,20].
In our patient, there were no previous signs and symptoms of liver cirrhosis. The patient reported no complaint related to signs and symptoms of liver failure or portal hypertension, including jaundice, ascites, enlarged abdomen, shortness of breath, swollen feet, gastrointestinal bleeding, or hemorrhoids. The patient was very healthy and had gone through two pregnancies and deliveries without any health problems. However, the patient had no history of previous hepatitis B vaccination. Therefore, it was very possible that this patient developed HCC without passing the cirrhosis phase.
HCC in pregnancy also has a worse prognosis than HCC in women without pregnancy. According to an existing epidemiological study, the mean age of pregnant women with HCC is 28.9±4.4 years [21]. The risk of developing HCC increases with parity in those with risk factors [22]. However, the incidence of HCC during pregnancy is very rare because HCC generally occurs in women of non-productive age. Pregnancy could trigger detrimental effects in the progression of HCC due to the effect of hormonal alteration. Estrogen is thought to trigger hepatocyte mitosis, hypervascularization, free radical formation, reactivate HBV, and reduce humoral immunity [6,23]. HCC is generally asymptomatic in early pregnancy, but right upper quadrant pain, palpable mass, weight loss, and hepatomegaly are reported as common complaints in reported cases [24,25]. In this case, shortness of breath due to compression of the thoracic cavity and abdominal pain were the main clinical symptoms complained.
The elevation of AFP titer can be used as an additional diagnostic modality [26]. However, maternal AFP can also be used to screen for Down syndrome and neural tube defects. An increase in AFP in pregnant women with normal fetal conditions may indicate disease in the mother, unexcepted HCC [6,26]. In this case, there was a significant elevation in AFP and CA-125 with normal fetus development. CA-125 was reported to have a much higher sensitivity (92%) to HCC than AFP (58.8%) with a cutoff value of 200 ng/mL [27]. CA 125 also has a higher negative predictive value (NPV) (84.6%) than AFP (69.2%) but when it comes to combination, these two markers have an NPV that reaches 91.7% [27, 28]. However, overall AFP is more helpful than CA 125 to establish the diagnosis of HCC. A positive AFP result is a strong indication of HCC, but a negative result does not necessarily rule out HCC [26,28,29].
COVID-19 in pregnancy requires a special approach and consideration in terms of therapy with many considerations to fetal well-being and the condition of the mother. The safety of anti-COVID-19 drugs in general is not established because long-term prospective studies to ensure the level of safety of these drugs are not available. Molnupiravir, ritonavir, nirmatrelvir, favipiravir, and remdesevir in some literature have no evidence of safety for use in pregnant women [30]. However, in conditions that are life-threatening to the mother, the use of these antivirals can be considered with close monitoring of the potential for toxicity that may arise, particularly impaired liver function [31,32]. In a systematic review involving 113 pregnant women from 13 studies found that administration of remdesevir improved the clinical condition of pregnant patients with COVID-19, particularly those with better initial conditions and received remdesevir earlier [33]. In our case, antiviral was not given because of considering the condition of the fetus. The advices from the Department of Pulmonology and Department of Obstetrics and Gynecology did not recommend administration of those antivirals to the patient due to their unclear safety and teratogenic effects that cannot be fully ruled out. However, the patient received supportive therapy of vitamin C, vitamin D, zinc, ferrous sulfate, and folic acid. The patient also received fondaparinux injections for venous thromboembolism prophylactic therapy.
HCC in pregnancy has a poor prognosis. Delivery by cesarean section was carried out in 44.8% and live births were reported in only 58.7% of all recorded cases [34]. Complications of COVID-19 for the fetus during pregnancy can be in the form of abortion (2%), intrauterine growth restriction (IUGR, 10%), and preterm labor (39%) [9,10]. In our patient, the stillbirth that occurred in the second trimester might have been triggered by the presence of a neoplasm that inhibited fetal growth and development; and was exacerbated by SARS-CoV-2 infection.
COVID-19 in HCC and pregnancy had never been reported, making this case was the first case of pregnancy with HCC and COVID-19 infection occurring with the young age that had been recorded. There are still many aspects that cannot be understood clearly, such as how HCC can occur at such a young age without showing real clinical symptoms before pregnancy and how could the deterioration have occurred rapidly when the patient had the pregnancy
Conclusion
We reported a 26-year-old pregnant woman with HCC that also was infected with COVID-19. Chronic hepatitis B which was considered as the cause of HCC in this patient did not pass the liver cirrhosis phase. The history of cancer in the patient’s family members was thought as one of the contributing factors that accelerated the progression of chronic hepatitis B to HCC. Pregnancy was also believed to accelerate the progression of this disease through the influence of hormonal alteration during pregnancy. SARS-CoV-2 infection in this patient increased the maternal and fetal mortality. This case highlights that COVID-19 in pregnancy requires a special approach and consideration in terms of therapy with attention to fetal well-being and the condition of the mother.
Acknowledgements
We would like to express our gratitude to all participants in this study.
Ethical approval
The family of the patient provided the consent to include the case for scientific purpose.
Competing interests
The authors declare that there is no conflict of interest.
Funding
This study received no external funding.
Underlying data
All data underlying the results are available as part of the article and no additional source data are required.
How to cite
Abubakar Z, Yusuf F, Firdausa S, et al. Co-incidence of COVID-19 and hepatocellular carcinoma during pregnancy: Double punches to disease severity and mortality? Narra J 2023; 3 (3): e264 - http://doi.org/10.52225/narra.v3i3.264.
References
- 1.Kotsari M, Dimopoulou V, Koskinas J, et al. Immune system and hepatocellular carcinoma (HCC): New insights into HCC progression. Int J Mol Sci 2023;24(14):11471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021;71(3):209–249. [DOI] [PubMed] [Google Scholar]
- 3.McGlynn KA, Petrick JL, El-Serag HB. Epidemiology of hepatocellular carcinoma. Hepatology 2021;73 Suppl 1:4–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021;71(3):209–249. [DOI] [PubMed] [Google Scholar]
- 5.Parewangi ML, Akil F, Nu’man AS, et al. Perubahan paradigma pada strategi penanganan karsinoma hepatoseluler: systematic review. UMI Med J 2022;7(1):59–71. [Google Scholar]
- 6.Norouzi A, Tabei MN, Tavassoli S, et al. Hepatocellular carcinoma in pregnancy with unusual presentations. Middle East J Dig Dis 2012;4(4):228. [PMC free article] [PubMed] [Google Scholar]
- 7.Narang K, Enninga EAL, Gunaratne MDSK, et al. SARS-CoV-2 infection and COVID-19 during pregnancy: A multidisciplinary review. Mayo Clinic Proceedings 95. Elsevier; 2020:1750–1765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bangsawan CC, Rodiani R.. COVID-19 dan dampak yang ditimbulkan pada kehamilan. J Kesmas Jambi 2022;6(1):15–22. [Google Scholar]
- 9.Wenling Y, Junchao Q, Xiao Z, et al. Pregnancy and COVID-19: Management and challenges. Rev Inst Med Trop Sao Paulo 2020;62:e62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Agolli A, Agolli O, Velazco DFS, et al. Fetal complications in COVID-19 infected pregnant woman: A systematic review and meta-analysis. Avicenna J Med 2021;11(04):200–209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Russo FP, Zanetto A, Pinto E, et al. Hepatocellular carcinoma in chronic viral hepatitis: where do we stand? Int J Mol Sci 2022;23(1):500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sharma B, John S.. Hepatic cirrhosis. In: StatPearls. StatPearls Publishing; 2022. [PubMed] [Google Scholar]
- 13.Kanda T, Goto T, Hirotsu Y, et al. Molecular mechanisms driving progression of liver cirrhosis towards hepatocellular carcinoma in chronic hepatitis B and C infections: a review. Int J Mol Sci 2019;20(6):1358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Balogh J, David Victor I, Asham EH, et al. Hepatocellular carcinoma: A review. J Hepatocell Carcinoma 2016;3:41–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maepa MB, Ely A, Kramvis A, et al. Hepatitis B virus research in South Africa. Viruses 2022;14(9):1939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Alqahtani SA, Colombo M.. Treatment for viral hepatitis as secondary prevention for hepatocellular carcinoma. Cells 2021;10(11):3091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ng J, Wu J.. Hepatitis B- and Hepatitis C-related hepatocellular carcinomas in the United States: Similarities and differences. Hepat Mon 2012;12(10 HCC). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yang HI, Yeh SH, Chen PJ, et al. Associations between hepatitis B virus genotype and mutants and the risk of hepatocellular carcinoma. J Natl Cancer Inst 2008;100(16):1134–143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chayanupatkul M, Omino R, Mittal S, et al. Hepatocellular carcinoma in the absence of cirrhosis in patients with chronic hepatitis B virus infection. J Hepatol 2017;66(2):355–362. [DOI] [PubMed] [Google Scholar]
- 20.Wong DKH, Cheng SCY, Mak LLY, et al. Among patients with undetectable hepatitis B surface antigen and hepatocellular carcinoma, a high proportion has integration of HBV DNA into hepatocyte DNA and no cirrhosis. Clin Gastroenterol Hepatol 2020;18(2):449–456. [DOI] [PubMed] [Google Scholar]
- 21.Norouzi A, Naeimi Tabei M, Tavassoli S, et al. Hepatocellular carcinoma in pregnancy with unusual presentations. Middle East J Dig Dis 2012;4(4):228–31. [PMC free article] [PubMed] [Google Scholar]
- 22.Fwu CW, Chien YC, Kirk GD, et al. Hepatitis B virus infection and hepatocellular carcinoma among parous Taiwanese women: nationwide cohort study. J Natl Cancer Inst 2009;101(14):1019–1027. [DOI] [PubMed] [Google Scholar]
- 23.Maeda T, Imai D, Wang H, et al. Hepatic resection for recurrent hepatocellular carcinoma during pregnancy: A case report. Surg Case Rep 2020;6(1):1–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Garko SB, David OS, Mohammed T, et al. Hepatocellular carcinoma in pregnancy. Ann Afr Med 2009;8(4):284. [DOI] [PubMed] [Google Scholar]
- 25.Alvarez de la Rosa M, Nicolás-Pérez D, Muñiz-Montes JR, et al. Evolution and management of a hepatocellular carcinoma during pregnancy. J Obstet Gynaecol Res 2006;32(4):437–439. [DOI] [PubMed] [Google Scholar]
- 26.El-Serag HB, Kanwal F.. α-Fetoprotein in hepatocellular carcinoma surveillance: Mend it but do not end it. Clin Gastroenterol Hepatol 2013;11(4):441–443. [DOI] [PubMed] [Google Scholar]
- 27.Lopez JB, Balasegaram M, Thambyrajah V.. Serum CA 125 as a marker of hepatocellular carcinoma. Int J Biol Markers 1996;11(3):178–182. [DOI] [PubMed] [Google Scholar]
- 28.Zhao YJ, Ju Q, Li GC. Tumor markers for hepatocellular carcinoma. Mol Clin Oncol 2013;1(4):593–598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.El-Serag HB, Kanwal F, Davila JA, et al. A new laboratory-based algorithm to predict development of hepatocellular carcinoma in patients with hepatitis C and cirrhosis. Gastroenterology 2014;146(5):1249-1255.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sharun K, Tiwari R, Yatoo MI, et al. A comprehensive review on pharmacologic agents, immunotherapies and supportive therapeutics for COVID-19. Narra J 2022;2(3):e92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Burhan E, Dwi Susanto A, Isbaniah F, et al. Pedoman tatalaksana COVID-19 Edisi 4. Jakarta. [Google Scholar]
- 32.Siahaan SCPT. Kehamilan dengan COVID–19. Prosiding FK UC 2020;1(1):28. [Google Scholar]
- 33.Budi DS, Pratama NR, Wafa IA, et al. Remdesivir for pregnancy: A systematic review of antiviral therapy for COVID-19. Heliyon 2022;8(1):e08835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lau WY, Leung WT, Ho S, et al. Hepatocellular carcinoma during pregnancy and its comparison with other pregnancy-associated malignancies. Cancer 1995;75(11):2669–2676. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data underlying the results are available as part of the article and no additional source data are required.