Abstract
Background:
Almost 75% of US adults are overweight or obese. Though intentional weight loss of as little as 3% improves physical functioning and reduces cardiometabolic risk, most adults are unsuccessful at long-term weight maintenance. Our hypothesis is that intermittent fasting (IF: short periods of intense energy restriction) will reduce weight regain. IF may combat obesity due to its effects on nutrient-sensing signaling pathways and circadian rhythm. The objective of this randomized clinical trial is to test the effectiveness of an intensive body weight management program with and without IF.
Methods:
In the Promotion of Successful Weight Management in Overweight and Obese Veterans (POWER-VET) trial (NCT04131647), 154 middle-aged and older adults (50–75 years) who are overweight and obese (BMI: 25–40 kg/m2) and seen at either a Baltimore, MD or San Antonio, TX Veterans Affairs Medical Center will be enrolled. Participants will undergo 12 weeks of weight loss (including a low-calorie heart healthy (HH) diet and exercise). Following weight loss, participants will be randomly assigned to one of two 24-week weight maintenance (WM) interventions: WM alone (continuation of HH diet and exercise) or WM + IF. The primary aim is to determine the effect of WM + IF compared to WM alone on body weight maintenance after intentional weight loss.
Discussion:
Determining effective, translatable strategies that minimize weight regain following successful weight loss holds public health relevance. This POWER-VET trial introduces an innovative practice of IF to prevent weight regain after clinically significant weight reduction and could provide evidence-based recommendations to promote this type of intervention in middle aged and older adults.
Keywords: Obesity, Weight management, Aging, Exercise, Intermittent fasting
1. Introduction
Caloric restriction to induce intentional weight loss (WL) of as little as 3–5%, especially in the abdominal region, reduces the risk for the development of type 2 diabetes mellitus (T2DM) and obesity-associated cardiovascular risk factors [1] and improves physical function [2]. Though performing aerobic and/or resistance exercise (EX) regularly during weight maintenance (WM; after successful weight loss) results in significantly less weight regain than non-exercisers [3,4], most adults are unsuccessful at long-term WM, regaining almost half the weight lost within the following two years with a return to baseline weight within the next 3–5 years [5-7].
Intermittent fasting (IF) has achieved popularity as a method to improve dietary adherence. This involves periods of reduced, very restricted energy intake (typically <20–25% of normal caloric intake for two consecutive or alternating days) interspersed with ad libitum energy intake. Strict adherence is only needed a few days of the week providing a potential for better compliance when implemented long-term. In addition to this potential behavior mechanisms, there also is evidence that IF can affect physiological systems [8], including stem cell differentiation, fatty acid oxidation, cellular autophagy, intestinal flora reorganization, hormone concentrations, nutrient-sensing signaling pathways, and circadian rhythm, all of which may have significant implications on body weight management. Though not universally observed [9], greater rates of weight loss are observed with IF as compared to traditional heart healthy diets [10]. Further, during WL, better outcomes (weight and abdominal fat loss) are observed with IF compared to continuous energy restriction [11,12]. IF is shown to reduce insulin levels, improve insulin sensitivity by oral glucose tolerance test, and reduce blood pressure even when there is no weight loss [13]. However, less is known about the effect of utilizing IF as a method to promote WM following successful WL, with results mixed [9,14]. Thus, more research is needed as to whether a more consistent IF protocol may be an advantageous method for maintaining the long-term weight benefits from initial WL (combined with exercise) in overweight and obese individuals.
The primary purpose of the Promotion of Successful Weight Management in Overweight and Obese Veterans (POWER-VET) trial is to compare the effect of WM alone (heart healthy diet and EX) with WM + IF (heart healthy diet and EX plus IF) on body weight and composition following successful weight loss (~5–10% induced by a low-calorie heart healthy diet and exercise). Our hypothesis is that IF will reduce weight regain. Our primary aim is to determine the effects of WM + IF compared to WM alone on 24-week changes in body weight and composition following 12 weeks of WL. We contend IF (combined with EX) during WM has the potential to confer widespread benefits for physical and cardiometabolic health among overweight and obese older adults that have recently undergone successful WL.
2. Methods
2.1. Overview
The POWER-VET trial is approved by both the University of Maryland Baltimore Institutional Review Board (IRB Number HP-00088304: Baltimore site) and University of Texas Health Science Center at San Antonio (IRB Number HSC20190792H: San Antonio site), their respective VA Research and Development Committees, and is registered on clinicaltrials.gov (NCT04131647). Prior to completing any data collection, all participants will be provided written informed consent and complete a HIPAA authorization form.
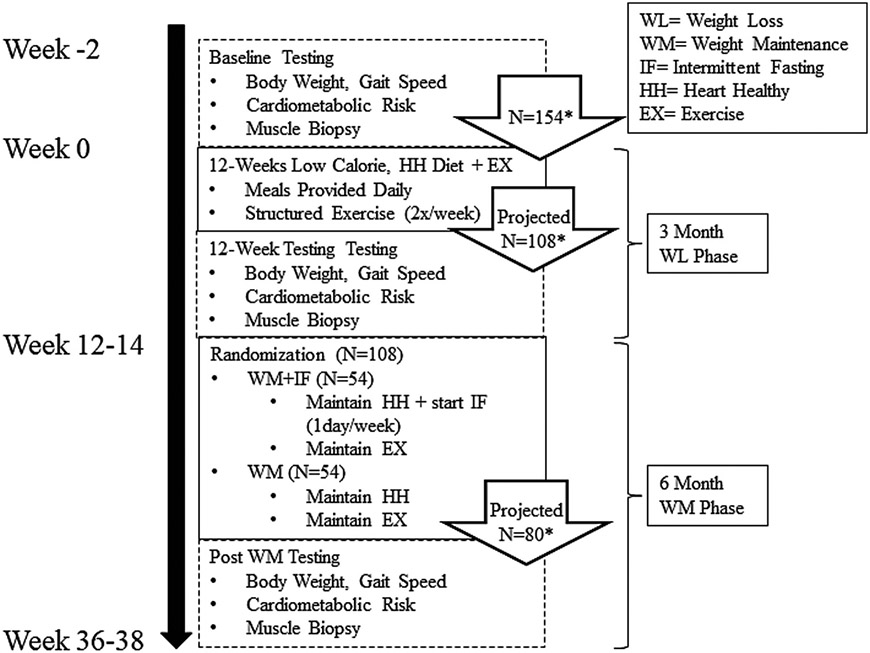
This randomized clinical trial involves six phases (Fig. 1), completed over 11–12 months per person: Phase 1: Recruitment, Screening, Enrollment; Phase 2: Baseline Testing; Phase 3: Hypocaloric Weight Loss Intervention; Phase 4: Post Weight Loss Intervention Testing; Phase 5: Weight Maintenance Intervention; Phase 6: Post Weight Maintenance Intervention Testing.
Fig. 1. Study design.
* Number of Veterans split equally between Baltimore and San Antonio VA sites
2.2. Recruitment, screening, enrollment (Phase 1)
2.2.1. Participant characteristics and eligibility
The POWER-VET trial will recruit 154 male and female Veterans (77/site) who are older (50–75 years) and overweight or obese (body mass index [BMI]: 25–40 kg/m2). Specific participant inclusion and exclusion criteria are displayed in Table 1 and were selected to identify individuals likely to benefit from the intervention and identify those that may be at risk for experiencing adverse events from participation.
Table 1.
Participant inclusion and exclusion criteria of the POWER-VET study.
| Criterion | Assessment |
|---|---|
| Inclusion Criteria | |
| Veteran | Self-report |
| 50–75 years of age | Self-report |
| BMI: 25–40 kg/m2 | Measured on scale |
| Postmenopausal status for women | Self-report |
| Exclusion Criteria | |
| Uncontrolled diabetes (HbA1c >10% or the current use of insulin) | Blood work (HbA1c) and self-report (medications) |
| Neurologic, musculoskeletal, or other condition that limits subject’s ability to complete study physical assessments | Physical exam, medical history, and self-report |
| Active inflammatory, COVID-19, autoimmune, infectious, hepatic (LFTs >2.5 x WNL), renal (eGFR<60), gastrointestinal, malignant, and psychiatric disease | Blood work, medical history, and self-report |
| Weight change within the past month of >5 kg | Self-reported |
| Alcohol or drug abuse | Self-reported |
2.2.2. Participant recruitment and screening
Veterans will be enrolled from the VA Maryland and South Texas Veterans Healthcare Systems. Recruitment methods may include electronic medical record queries, posting study fliers in medical offices or senior centers, community engagement activities, and/or newspaper or web-based advertisements including social media. With the approval of the IRB and VA Research and Development Committees, following a partial HIPAA waiver, we will perform a medical record review to prescreen potential Veteran participants who live close to either Medical Center. Potential participants will be mailed an IRB approved recruitment letter. Postage will be provided on the opt in/out post-card, which will allow for potential subjects to indicate whether they would like to be contacted in the future related to the study. If no response is received within two weeks, we will follow up with a phone call to gauge interest. Those who qualify preliminarily will be scheduled for a full in-clinic evaluation and will be provided with an advance copy of the consent form.
Telephone pre-screening prior to the clinic screening visit will be employed to ensure potential candidates will meet inclusion and exclusion criteria for enrollment. The participants will complete a standard screening visit at the respective Maryland or South Texas Veterans Healthcare System, which includes a medical and medication history and physical exam with a research clinician, blood sampling, and electrocardiogram (ECG).
2.3. Baseline testing (Phase 2)
Data for primary (body weight) and secondary (gait speed, abdominal fat area, systemic and skeletal muscle lipoprotein lipase activity) outcomes will be collected using standardized protocols and trained staff. All baseline testing will be conducted at the respective Maryland or South Texas Veterans Healthcare System over 4–6 visits during a 2–4 week period.
2.4. Hypocaloric weight loss (Phase 3)
2.4.1. Low calorie diet
All participants then will begin a similar weight loss treatment. Participants will receive instructions for changing their dietary composition to follow recommendations of a heart healthy diet composed of 50–55% carbohydrate, <30% fat, and 15–20% protein. Participants will be provided with food and dietary counseling to provide 1200–1300 kcal/d (women) and 1500–1600 kcal/d (men). The provided food will provide ~65–70% of total caloric needs and will be a mixture of both freshly prepared and pre-packaged foods, which will be consumed daily across the 12-week weight loss intervention. Participants will also meet in in-person or remote, hour-long group behavior modification weight loss sessions once per week for 12 weeks with the RD or trained study staff. During these sessions, participants will learn how to adjust their eating habits by following recommendations for reducing dietary fat and substituting complex carbohydrates and reducing energy intake to induce gradual weight loss while maintaining proper dietary composition. Participants receive a notebook of course content and will weigh in prior to each session. They will be given detailed written and verbal instructions. Those who do not demonstrate adequate weight loss will meet individually with the RD to re-evaluate their caloric intake and compliance with the meals provided. The goal is for each person to have a 5–10% reduction in body weight across the 12-week Hypocaloric Weight Loss Phase.
2.4.2. Exercise
All participants will take part in 2×/week exercise sessions in-person at their respective Veterans Healthcare System, engaging in supervised training for 12 weeks. All facility-based sessions will be supervised by trained research staff. The training sessions will consist of aerobic exercise, performed initially for 15–20 min at 50% heart rate reserve (HRR; maximal HR-resting HR). Intensity will increase 5–10% weekly as tolerated. The end-target intensity will be 40 min at 70–85% of HRR, which we will aim to reach during week 3 of the intervention. HR will be recorded during each session using a chest-strap heart rate monitor. Veterans also will undergo whole body resistance training for 11 different exercises (knee extension, knee flexion, hip abduction, hip adduction, shoulder abduction, shoulder adduction, shoulder extension, shoulder flexion, triceps kickback, bicep curl, and chest press) using resistance bands. In general, participants will undergo two sets of 15 repetitions for each exercise. Bands with greater resistance will be provided as participants are able to progress to 20 repetitions on the second set for each exercise. Participants will be asked to perform both aerobic and resistance band exercise on their own at home a third day of the week. Participants will be asked about his/her home exercise at each supervised exercise session. Participants will be given a Fitbit (San Francisco, CA) Inspire fitness tracker with heart rate capabilities to aid in home exercise.
2.5. Post weight loss (Phase 4) and weight maintenance (Phase 6) intervention testing
All tests as described below in the assessment sections (Sections 2.8.-2.10.) will be repeated after the weight loss and weight maintenance phases using similar scheduling. During the last week of Phase 4, participants will also meet with the RD to learn the weight maintenance dietary interventions.
2.6. Weight maintenance (Phase 5)
2.6.1. Randomization
Participants will be randomized following 12-week (Phase 4) testing using a computer based pseudo-random number generator to one of two groups: weight maintenance alone (continuation of a heart healthy diet and exercise) or WM + IF.
2.6.2. Weight maintenance intervention
During the weight maintenance phase, all Veterans will be instructed to continue at home their heart healthy diet and exercise routine, but without the emphasis on WL. All participants will be encouraged to walk at home 10,000 steps per day and perform three days of resistance band exercises. Both groups (WM and WM + IF) will be called weekly by the study coordinator during the first month and bi-weekly thereafter to ask about compliance with their meal plans, assess body weight (body weight scales provided), and asked for the prior day activity recorded by the Fitbit.
2.6.3. Intermittent fasting
Participants randomized to IF also will be provided with sample menus (Table 2) and instructed by the RD to consume small meals per day (~500 kcal/d for women and ~ 600 kcal/d for men) one day per week. While they will be encouraged to consume 3–4 small meals per day, they will be given the liberty of consuming these calories whenever they desire throughout the day. These small meals will be based upon dietary exchanges [15] (e.g., milk, fruit, starch, lean meat, fat, and vegetables) (Table 3). No additional diet or exercise instructions are provided for the non-IF days beyond those outlined in section 2.6.2. If participants report difficulties with following the meal plan, the RD will call the participant to assist with meal plan compliance. Adherence to IF will be assessed during the first month and bi-weekly thereafter during the weekly study coordinator calls.
Table 2.
Sample 500 kcal exchange menu and meal plan.
| Carb (g) |
Pro (g) |
Fat (g) |
Calories (kcal) |
Meal Plan | |
|---|---|---|---|---|---|
| Starch servings (1) | 15 | 3 | 0.5 | 80 | 1 slice wheat toast |
| Very Lean/Lean | – | 14 | 3 | 90 | 2 oz. 95% fat free lunch meat |
| Meat Serving (3) | – | 7 | 1.5 | 45 | 1 hot dog |
| Vegetable Serving (3) | 5 | 2 | – | 25 | 1 cup raw carrots |
| 5 | 2 | – | 25 | ½ cup cook spinach | |
| 5 | 2 | – | 25 | spinach ½ cup 100% vegetable juice | |
| Fruit Serving (1) | 15 | – | – | 60 | 1 raw apple |
| Skim or Low Fat Milk Serving (1.5) | 6 | 4 | 1 | 50 | ½ cup skim milk |
| 12 | 8 | 1.5 | 100 | 8 oz. sugar-free nonfat yogurt | |
| Fat Serving (1) | – | – | 5 | 45 | 1 T mayo |
| Total | 63 | 45 | 12.5 | 520 |
Table 3.
Intermittent fasting meal patterns during weight maintenance.
| Women: ~500 cal | Men: ~600 cal |
|---|---|
| Skim or Low Fat Milk Serving – 1.5 | Skim or Low Fat Milk Serving – 2 |
| Fruit Serving - 1 | Fruit Serving - 1 |
| Starch Serving- 1 | Starch Serving- 1 |
| Very Lean/Lean Meat Serving - 3 | Very Lean/Lean Meat Serving - 4 |
| Fat Serving- 1 | Fat Serving- 1 |
| Vegetable Serving 3++ | Vegetable Serving 3++ |
2.7. Study assessments timeline
Study assessments will be collected at baseline (Phase 2), following the 12 week intensive weight loss intervention (Phase 4), and following the 24 week weight maintenance intervention (Phase 6). The assessments include a series of cardiometabolic outcomes (anthropometrics, imaging, and systemic T2DM and cardiovascular risk), questionnaire, physical examinations, and physical performance tests. Adverse events and changes in health status and medications will be assessed and recorded by asking participants to complete a health status questionnaire during assessment visits and at each in-person intervention session.
2.8. Primary outcome measure
The POWER-VET trial is powered to detect group difference in our primary outcome variable of 36 week change in total body weight measured on a calibrated, digital scale with the participant dressed in light clothing without shoes at baseline, 12 weeks, and 36 weeks with the primary contrast looking at changes over 36 weeks. Body weight collected at the 12-week time point allows for further examination of body composition following the intensive WL phase.
2.9. Secondary outcome measures
2.9.1. Gait speed
A 6-min walk distance test will be conducted where participants are instructed to “cover as much distance as they can” over a flat 100 ft walking surface demarcated by traffic cones while timed by a stop watch.
2.9.2. Abdominal fat area
Participants will undergo spiral computed tomography (CT) scans (Siemens Somatom Sensation 64 Scanner, Fairfield, CT). The slice at L4-L5 region will be used to determine visceral and subcutaneous abdominal adipose tissue area, and analyzed using Medical Image Processing, Analysis and Visualization, version 7.0.0 (NIH Center for Information Technology, Bethesda, MD). Visceral fat tissue will be determined by tracing along the fascial plane defining the internal abdominal wall to distinguish between subcutaneous abdominal fat. Visceral fat will be measured from the whole-body scan in a five cm wide region across the entire abdomen proximal of the iliac crest at a level approximately corresponding to L4. Each scan will be acquired with a tube voltage of 120 kV. Quality assurance procedures of the CT system will be performed regularly according to the manufacturer’s guidelines and a cross calibration phantom will be circulated to assess differences across sites.
2.9.3. Skeletal muscle lipoprotein lipase (LPL) activity
A vastus lateralis muscle biopsy will be taken from a subset of participants (n = 40) under local anesthesia for the measurement of enzyme activities important to energy metabolism (lipoprotein lipase and citrate synthase). Muscle samples will be frozen immediately in clamps cooled in liquid nitrogen and stored at −80 °C until assay.
2.10. Additional outcome measures
2.10.1. Anthropometrics
Maximal waist circumference will be measured at the greatest anterior extension of the abdomen, usually at the level of the umbilicus. Minimal waist circumference will be measured at the narrowest part of the torso (above the umbilicus and below the xiphoid process). Hip circumferences will be measured maximal circumference of the buttocks, typically in line with the pubic bone. All measured will be collected using a Gulick spring loaded tape measure. Triplicate measures at each site will be collected and retested if measurements are not within 0.5 cm.
2.10.2. Dual energy X-ray absorptiometry (DXA) derived measures
Total body, right hip, and lumbar spine scans will be conducted using DXA at each site within the respective Radiology Department (Baltimore site: iDXA, LUNAR Radiation Corp., Madison, WI and San Antonio site: Hologic Discovery A, Hologic, Inc., Marlborough, MA). Scans will be used to determine total body and regional (i.e., arms, legs, total appendicular) fat mass and lean mass. Total body, total hip, femoral neck, and lumbar spine bone mineral density (BMD) will be determined. All scans will be performed and analyzed by certified radiology technicians. Daily quality control scans will be performed utilizing a manufacture-provided a calibration phantom and a set of cross calibration phantoms will be circulated to assess differences across sites.
2.10.3. CT muscle composition
The same spiral CT scans will be used as above to quantify regional muscle area, intramuscular fat area (low-density lean tissue), and subcutaneous fat via a 64-slice CT scanner. Each scan will be acquired with a tube voltage of 120 kV. Quality assurance procedures of the CT system will be performed regularly according to the manufacturer’s guidelines.
2.10.4. Cardiovascular disease risk factors
Following 12-h of fasting by the participants, we will collect blood for the measurement of complete blood count, lipid panel, thyroid stimulating hormone, hemoglobin A1c, and comprehensive metabolic panel measured via the respective VA clinical laboratory. Blood also will be collected for measurement of metabolic and obesity-related factors . Participants will be requested to refrain from physical activity for at least 24 h before providing the sample. After centrifugation for 15 min at 4 °C (plasma) and 10 min at room temperate (serum), aliquots of plasma and serum will be stored at −70 °C locally until shipment of samples to Baltimore and batch analyses at a Baltimore VA research laboratory at study completion.
2.10.5. T2DM risk
T2DM risk will be assessed via an Oral Glucose Tolerance Test (OGTT) in those without known T2DM at baseline. An antecubital 20-gauge polyethylene catheter is inserted by percutaneous venipuncture for blood sampling at 10 min, 5 min, and immediately before oral ingestion of 75 g of glucose. For the determination of changes in glucose tolerance and insulin responses, blood samples will be drawn at 30, 60, 90, and 120 min after the glucose ingestion.
2.10.6. Fitness and physical function
VO2peak will be used as the primary measure of aerobic capacity. Participants will be asked to exercise to voluntary exhaustion. Mode of testing will be consistent at each time point for each participant. Oxygen (O2) consumption, carbon dioxide (CO2) production, and minute ventilation will be measured breath-by-breath using a metabolic cart (Baltimore: Quark, Cosmed USA, Chicago, IL and San Antonio: TrueOne 2400, Parvo Medics, Salt Lake City, UT). Physical function will be assessed via 1) modified physical performance test (MPPT), consisting of balance testing, timed 4 m walk, timed repeated chair stands, putting on and removing a jacket, placing a book on a shelf above shoulder level, picking a penny up off the flood, turning in a 360 degree circle, walking 50 ft, and climbing a flight of stairs; 2) 6-min walk test as above; 3) maximal isometric hand grip strength of both hands measured with the elbow flexed to 90 degrees and the forearm in a neutral position using a handheld dynamometer with the mean value of two trials included in analyses; and 4) 1-repeitition maximum strength (1RM) will be determined within 4–6 trials with rest periods of 90 s between trial for the chest press and knee extension.
2.10.7. Daily physical activity
Participants will be given a GT3X ActiGraph monitor (ActiGraph, Pensacola, FL) to be worn on the waist for seven 24-h periods. They will be distributed sometime during the final two weeks of baseline assessment and during the final two weeks of both the WL (Phase 4) and WM (Phase 6) phases. Time spent in sedentary behavior, and in light, moderate and vigorous activity will be measured using ActiLife software (ActiGraph, Pensacola, FL). The participant will keep a daily sleep/activity log.
2.10.8. Questionnaires/subjective assessments
The following questionnaires will be administered: 1) Fatigue Severity Scale [16] to assess fatigue; 2) Short Form Survey Instrument (SF-36) [17] to assess health-related quality of life; 3) Patient-Reported Outcomes Measurement Information System −57 (PROMIS-57) [18] to assess depression, anxiety, physical function, pain interference, fatigue, sleep disturbance, and satisfaction with participation in social roles; 5) International Physical Activity Questionnaire (IPAQ) [19] to assess current level of physical activity. A Registered Dietitian (RD) also will assist in collect a 24-h dietary recall from the participant on at least one week day using the USDA Automated Multiple-Pass Method [20].
2.10.9. Sample size and statistical power
We base our power calculations on a previous study utilizing IF during 1-years of WM following 12 weeks of WL (averaging ~10% of initial body weight) [14]. This study showed that IF during WM resulted in a mean increase in body weight of only 1.75 ± 7.7 kg, compared to 8.75 ± 8.1 kg following a traditional heart healthy WM intervention. Our proposed sample size of 80 subjects (40/group) will provide 90% power to detect a difference of at least 7 ± 8 kg between the WM + IF and WM groups.
In response to our previous experience with similar WL trials showing >85% compliance and 20% attrition, we will account for attrition rates. We will employ a 30% and 25% attrition rate in the proposed study during the WL and WM phases, respectively. Thus, 154 individuals (77/site) will enter the study.
2.11. Statistical methods
We will use intention-to-treat principles in full accordance with CONSORT guidelines; however, we will only perform primary analyses on those achieving at least 3% weight loss during the initial WL phase (Phase 4) to isolate the effects of the WM and WM + IF interventions on the potential for body weight regain. All data will undergo range checks at the time of data entry and will be examined monthly by histograms and bivariate scatterplots to check for inconsistencies, unusual data needing further verification, and outliers. Plots of longitudinal observations will be used to inspect for unusual changes requiring verification against source documentation. Regression diagnostics and exploratory analyses will be performed to find appropriate transformations of variables if needed. Order of priority in choosing a transformation will be to satisfy: 1) linearity, 2) homogeneity, and 3) normality assumptions. We only include unadjusted analyses for our primary aim, as suggested in CONSORT guidelines [21]. Randomization will give us rough balance across the study groups, and we can control for additional confounders, such as age and site, in subsequent analyses beyond our primary outcome.
Repeated-measures analyses for serial primary and secondary outcome data will be performed with a means model via the SAS MIXED Procedure using SAS (SAS Institute, Cary, NC), providing separate estimates of the means by time on study (baseline, after 12-week WL, and after 36-week WM or WM + IF). The model will include three predictors (treatment arm, time on study and the statistical interaction between treatment arm and time on study). A compound-symmetric variance-covariance form in repeated measurements will be assumed for each outcome and robust estimates of the standard errors of parameters will be used to perform statistical tests and construct 95% confidence intervals. The model-based means are unbiased with unbalanced and missing data, so long as the missing data are non-informative (missing at random). A P-value <0.05 will be considered statistically significant for the main effects (treatment and time on study) and for the treatment by time on study interaction effect from the repeated measures analysis. The statistical test for interaction between time on study and treatment will be the primary overall hypothesis test to determine whether body weight (primary outcome) and the secondary endpoints in the two study groups changed in significantly different ways during WM (i.e., different temporal patterns from 12 to 36 weeks). If the mean outcome in the treatment groups is consistently different or similar over time (i.e., no statistical interaction), then the main effect test for treatment will be used as the primary test. If a significant interaction is detected, then t-tests will be used to compare the differences between the model-based intervention means at each time point and to compare differences over time within each treatment arm. Specific statistical tests will be done within the framework of the mixed effects linear model. All statistical tests will be 2-sided and unadjusted for multiple comparisons.
3. Discussion
The purpose of the POWER-VET trial is to understand the effects of IF as a method to promote long-term successful WL in older adults who are obese or overweight. We hypothesize that despite similar WL between groups (~5–10%), IF will provide the stimulus for prevention of weight regain at 36 weeks and will improve cardiometabolic and functional health factors. By reducing caloric intake to ~500 kcal/d, one day/week during WM, this could prevent weight regain of almost 0.75 kg/month.
We [22,23] and others [24-26] have previously show that the addition of EX to WL may optimize intentional WL cardiometabolic and functional outcomes in middle-aged and older adults. Despite this notion, there are challenges in translating EX as an effective strategy for long-term WM mainly due to non-compliance. Further, weight regain, even in those that EX, is common in the years following successful WL. The POWER-VET study is designed to address whether the addition of dietary modification to EX during WM will address this concern. The use of IF has been investigated for WL and yet, little is known in how it could be utilized to maintain weight reduction/prevent weight regain. This study could provide evidence-based recommendations to include modest IF (one day/week) in WM programs.
3.1. Strengths and limitations
The POWER-VET trial provides a novel, powered comparison between WM alone and WM + IF, which may lead to non-pharmacologic strategies to optimize body weight. Our conduct in two urban locations will allow for assessment in two distinct and ethnic diversity populations (Baltimore, MD (62% Non-Hispanic Black) and San Antonio, TX (57% Hispanic White). Further, the majority of trials of IF to date have enrolled women. In a recent systematic review, it was reported that only 10 men were included in the entire analysis [27]. Further, there are a lack of studies examining the effects of IF in older adults. Our inclusion of Veterans, 91% of which are male and with a high prevalence of comorbidities, will help guide the treatment of overweight and obesity in aging men. Overall obesity contributes to the development of mobility limitations and major mobility disability in older adults. Increased body weight and obesity may lead to physical limitations and difficulty in ambulation. There is a current gap in the literature as to the effects of weight regain on changes in mobility status. We will enroll high risk sedentary, overweight and obese, older adults; thus, targeting those with the greatest need.
Despite these strengths, the trial contains potential limitations. While our preliminary data suggest that poor adherence to the interventions are unlikely, it is possible that this could occur. It is possible that not all participants will be motivated to complete both forms of EX (aerobic and resistance) when performed unsupervised and in the home setting. Different forms of exercise can affect body composition differently, nevertheless, the prescribed protocol reflects current recommendations. Further, it is possible that not all participants randomized to the IF group will adhere to this restricted eating each week during WM. Phone call reminders and logs to document the Fitbit activity will aid in assessing adherence. Such variations in adherence to IF may allow for exploration analyses to determine the dose-response between IF and changes in body weight.
4. Conclusion
If successful, IF is a relatively low-cost and low-risk intervention, which may simultaneously reduce disability among aging adults with comorbidity associate healthcare-related costs. Long-term goals would be to collaborate with implementation scientists to integrate this model of weight maintenance into clinical care. This clinical trial will examine whether the successful promotion of WL is best maintained by inclusion of IF to a WM program.
Acknowledgements
The POWER-VET Project Team includes the following collaborative authors: Kristina K. Marcus, MS; Mason Cervantes, MS; Kelly A. Ort, RD; Tim Calderon, MS; Jose Renero, BS; Ronna Robbins, PhD; Rozmin Jiwani, PhD.
Source of funding
Research reported in this publication was supported by the United States (U.S.) Department of Veterans Affairs (Merit Review Award I01 CX001965 (MPI: Ryan/Ortmeyer/Serra), a Senior Research Career Scientist Award IK6 RX003977 from the United States Department of Veterans Affairs Rehabilitation R&D (Rehab RD) Service (PI: A.S.R.), the Baltimore and San Antonio GRECCs, as well as the University of Maryland Claude D. Pepper Older Americans Independence Center (P30 AG028747).
Footnotes
CRediT authorship contribution statement
Monica C. Serra: Conceptualization, Data curation, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Writing – original draft, Writing – review & editing. Heidi K. Ortmeyer: Conceptualization, Funding acquisition. Alice S. Ryan: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Writing – original draft, Writing – review & editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability
No data was used for the research described in the article.
References
- [1].Jensen MDRD, Donato KA, Apovian CM, Ard JD, Comuzzie AG, Hu FB, Hubbard VS, Jakicic JM, Kushner RF, Loria CM, Millen BE, Nonas CA, Pi-Sunyer FX, Stevens J, Stevens VJ, Wadden TA, Wolfe BM, Yanovski SZ, Guidelines (2013) for managing overweight and obesity in adults, Obesity 22 (S2) (2014) S1–S140. [Google Scholar]
- [2].Villareal DT, Chode S, Parimi N, Sinacore DR, Hilton T, Armamento-Villareal R, et al. , Weight loss, exercise, or both and physical function in obese older adults, N. Engl. J. Med 364 (13) (2011) 1218–1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Wadden TA, Vogt RA, Foster GD, Anderson DA, Exercise and the maintenance of weight loss: 1-year follow-up of a controlled clinical trial, J. Consult. Clin. Psychol 66 (2) (1998) 429–433. [DOI] [PubMed] [Google Scholar]
- [4].Fogelholm M, Kukkonen-Harjula K, Nenonen A, Pasanen M, Effects of walking training on weight maintenance after a very-low-energy diet in premenopausal obese women: a randomized controlled trial, Arch. Intern. Med 160 (14) (2000) 2177–2184. [DOI] [PubMed] [Google Scholar]
- [5].Byrne S, Cooper Z, Fairburn C, Weight maintenance and relapse in obesity: a qualitative study, Int. J. Obes. Relat. Metab. Disord 27 (8) (2003) 955–962. [DOI] [PubMed] [Google Scholar]
- [6].Visram S, Crosland A, Cording H, Triggers for weight gain and loss among participants in a primary care-based intervention, Br. J. Communit. Nurs 14 (11) (2009) 495–501. [DOI] [PubMed] [Google Scholar]
- [7].Phelan S, Hill JO, Lang W, Dibello JR, Wing RR, Recovery from relapse among successful weight maintainers, Am. J. Clin. Nutr 78 (6) (2003) 1079–1084. [DOI] [PubMed] [Google Scholar]
- [8].Liu S, Zeng M, Wan W, Huang M, Li X, Xie Z, et al. , The health-promoting effects and the mechanism of intermittent fasting, J. Diabetes Res 2023 (2023) 4038546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Trepanowski JF, Kroeger CM, Barnosky A, Klempel MC, Bhutani S, Hoddy KK, et al. , Effect of alternate-day fasting on weight loss, weight maintenance, and Cardioprotection among metabolically healthy obese adults: a randomized clinical trial, JAMA Intern. Med 177 (7) (2017) 930–938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Ganesan K, Habboush Y, Sultan S, Intermittent fasting: the choice for a healthier lifestyle, Cureus 10 (7) (2018), e2947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Barnosky AR, Hoddy KK, Unterman TG, Varady KA, Intermittent fasting vs daily calorie restriction for type 2 diabetes prevention: a review of human findings, Transl. Res 164 (4) (2014) 302–311. [DOI] [PubMed] [Google Scholar]
- [12].Klempel MC, Kroeger CM, Bhutani S, Trepanowski JF, Varady KA, Intermittent fasting combined with calorie restriction is effective for weight loss and cardio-protection in obese women, Nutr. J 11 (2012) 98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Sutton EF, Beyl R, Early KS, Cefalu WT, Ravussin E, Peterson CM, Early time-restricted feeding improves insulin sensitivity, blood pressure, and oxidative stress even without weight loss in men with prediabetes, Cell Metab. 27 (6) (2018) 1212–21 e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Zuo L, He F, Tinsley GM, Pannell BK, Ward E, Arciero PJ, Comparison of high-protein, intermittent fasting low-calorie diet and heart healthy diet for vascular health of the obese, Front. Physiol 7 (2016) 350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].National Heart Lung and Blood Institute, Food Exchange Lists, Available from: URL, https://www.nhlbi.nih.gov/health/educational/lose_wt/eat/fd_exch.htm, 2023.
- [16].Krupp LB, LaRocca NG, Muir-Nash J, Steinberg AD, The fatigue severity scale. application to patients with multiple sclerosis and systemic lupus erythematosus, Arch. Neurol 46 (10) (1989) 1121–1123. [DOI] [PubMed] [Google Scholar]
- [17].Brazier JE, Harper R, Jones NM, O’Cathain A, Thomas KJ, Usherwood T, et al. , Validating the SF-36 health survey questionnaire: new outcome measure for primary care, Bmj 305 (6846) (1992) 160–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Cella D, Riley W, Stone A, Rothrock N, Reeve B, Yount S, et al. , The patient-reported outcomes measurement information system (PROMIS) developed and tested its first wave of adult self-reported health outcome item banks: 2005-2008, J. Clin. Epidemiol 63 (11) (2010) 1179–1194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Hagströmer M, Oja P, Sjöström M, The international physical activity questionnaire (IPAQ): a study of concurrent and construct validity, Public Health Nutr. 9 (6) (2006) 755–762. [DOI] [PubMed] [Google Scholar]
- [20].Blanton CA, Moshfegh AJ, Baer DJ, Kretsch MJ, The USDA automated multiple-pass method accurately estimates group total energy and nutrient intake, J. Nutr 136 (10) (2006) 2594–2599. [DOI] [PubMed] [Google Scholar]
- [21].Butcher NJ, Monsour A, Mew EJ, Chan AW, Moher D, Mayo-Wilson E, et al. , Guidelines for reporting outcomes in trial reports: the CONSORT-outcomes 2022 extension, JAMA 328 (22) (2022) 2252–2264. [DOI] [PubMed] [Google Scholar]
- [22].Ryan AS, Ge S, Blumenthal JB, Serra MC, Prior SJ, Goldberg AP, Aerobic exercise and weight loss reduce vascular markers of inflammation and improve insulin sensitivity in obese women, J. Am. Geriatr. Soc 62 (4) (2014) 607–614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Ryan AS, Serra MC, Goldberg AP, Metabolic benefits of prior weight loss with and without exercise on subsequent 6-month weight regain, Obesity (Silver Spring) 26 (1) (2018) 37–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].You T, Murphy KM, Lyles MF, Demons JL, Lenchik L, Nicklas BJ, Addition of aerobic exercise to dietary weight loss preferentially reduces abdominal adipocyte size, Int. J. Obes 30 (8) (2006) 1211–1216. [DOI] [PubMed] [Google Scholar]
- [25].Nicklas BJ, Rogus EM, Goldberg AP, Exercise blunts declines in lipolysis and fat oxidation after dietary-induced weight loss in obese older women, Am. J. Phys 273 (1 Pt 1) (1997) E149–E155. [DOI] [PubMed] [Google Scholar]
- [26].You T, Berman DM, Ryan AS, Nicklas BJ, Effects of hypocaloric diet and exercise training on inflammation and adipocyte lipolysis in obese postmenopausal women, J. Clin. Endocrinol. Metab 89 (4) (2004) 1739–1746. [DOI] [PubMed] [Google Scholar]
- [27].Harris L, Hamilton S, Azevedo LB, Olajide J, De Bran C, Waller G, et al. , Intermittent fasting interventions for treatment of overweight and obesity in adults: a systematic review and meta-analysis, JBI Database System Rev. Implement. Rep 16 (2) (2018) 507–547. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data was used for the research described in the article.