Abstract
HYPOTHESIS:
The middle ear epithelium transforms due to changed immunomodulation during infection.
INTRODUCTION:
The epithelial cells of the tympanic cavity represent the first line of defense in the context of otitis media (OM). They can convert from a typical mucosal site into a respiratory epithelium and vice versa. Our goal is to depict the specific immune response of epithelial cells following infection at the molecular level.
METHODS:
The investigations were carried out on healthy and inflamed middle ear tissue, removed during surgical interventions in mouse and human models, and in a human in vitro cell model in HMEEC (human middle ear epithelial cells). We determined the epithelial localization of the protein expression of Toll- and NOD-like immune receptors and their associated signaling molecules using immunohistochemistry. In addition, we examined growth behavior and gene expression due to direct stimulation and inhibition.
RESULTS:
We found clinically and immunobiologically confirmed transformation of the inflamed middle ear epithelium depending on their origin, as well as differences in the distribution of TLRs and NLR in the epithelial cell lining. Dysregulated gene and protein expression of the inflammatory and apoptotic genes could be modulated by stimulation and inhibition in the epithelial cells.
CONCLUSIONS:
The local middle ear mucosal tissue is believed to modulate downstream immune activity following pathogen invasion via intrinsic cellular mechanism. Using translation approaches to target these molecular pathways may offer more reliable clinical resolution of OM in the future.
INTRODUCTION
Otitis media (OM) is one of the most common diseases of the ear and represents challenging burden on healthcare system1. Resolution of uncomplicated OM within a week suggests healing based on the innate immune system. The pathogenesis of OM is yet not fully understood and it is assumed as a multifactorial process in which the innate immune response to pathogens plays a crucial role.
The middle ear (ME) mucosa consists of a monolayer of squamous epithelial cells with clusters of highly prismatic cells with cilia and can rapidly transform into respiratory-type epithelium2,3. Depending on the location, different types of epithelia are found in the ME and there is continuity amid the Eustachian tube and pharynx. Pseudostratified epithelium with cilia and secretory goblet cells is found in the anterior portion of the ME cavity surrounding the eardrum and Eustachian tube, while attic, ossicles, the posterior ME cavity with round and oval windows and the promontory have simple epithelium. The pseudostratified epithelium is based on endoderm and the simple epithelium on the neural crest4.
The epithelium forms the barrier against infection and toxic damage and houses the mechanisms of the innate immune system5. Omics profiling of the ME mucosa revealed that additionally to the barrier function, the epithelial cells implement genes characteristic of pathogen detection, defense response, immune-cell recruitment plus infection resolution6.
Bacterial molecules, so-called pathogen-associated molecular patterns (PAMP), are recognized by pattern recognition receptors (PRR) in the ME mucosa and lead to expression of cytokines, interferons, and antimicrobial peptides via intracellular signaling cascades7. The most important representatives of PRR are Toll-like receptors (TLR) and nucleotide-binding oligomerization domains (NOD)-like receptors (NLR).
TLR are transmembrane proteins for the control of the cell surface as well as the vesicular cell compartments for PAMP. Recognition of PAMP leads, depending on the respective TLR, to the activation of myeloid differentiation factor 88 (MyD88) or TIR domain-containing inducing interferon-β adapter (TRIF) and subsequently to the activation of nuclear factor κB (NFκB), the interferon-regulating factor and mitogen-activated protein kinase (MAPK). Responsible for the expression of crucial pro-inflammatory cytokines such as interleukin-1beta (IL-1β) and tumor necrosis factor-alpha (TNFα)8,9. The importance of TLR was demonstrated using TLR-negative mouse models, which consistently showed chronification of OM10,11 and impaired bacterial clearance12,13
NLRs are cytoplasm-localized proteins to recognize intracellular PAMP, consisting of a caspase recruitment domain (CARD), a nucleotide binding and oligomerization domain (NOD) as well as leucine-rich repeats, and via activation of the RIP2 adapter, also lead to the activation of NFκB and MAPK pathways with resulting expression of proinflammatory cytokines8. The best-known NLR are NOD-1 and NOD-2. While NOD-1 recognizes diaminopimelic acid (DAP), an amino acid in peptidoglycans of gram-negative bacteria, NOD-2 recognizes muramyl dipeptide (MDP), present in gram-positive and gram-negative bacteria. Furthermore, there are also interactions between the individual signal transduction pathways, e.g. activation of NOD-2 via RIP2 leads to down-regulation of TLR-214. NLR-negative mouse models showed delayed inflammation and mucosal hyperplasia15.
The connection between the MyD88/NFκB pathway, its resultant molecules TNFα, IL-6, and IL-8, and the changes associated with OM has been shown in several publications16,17. NFκB, consisting of the p50 and p65 subunits, exists in the cytoplasm in inactive form complexed with its inhibitor IkB18,19. Stimulation and subsequent degradation of the inhibitor lead to release of NFκB and activate the transcription of proteins such as TNFα, IL-6 and IL-816,17,20–22.
TNFα has a major role in apoptosis19. The induction of apoptosis is initiated by binding of TNFα to death receptors of the TNFα family, the death-induced signaling complex (DISC) and the activation of various caspases. This described extrinsic apoptosis pathway is supplemented by an intrinsic apoptosis pathway, which acts as a central component using BID and the mitochondria20. To avoid uncontrolled apoptosis, there is a negative feedback mechanism in which NFκB activation by TNFα causes an anti-apoptotic effect21.
Previous work showed that these innate immune system signaling pathways in the ME mucosa play a key role in pathogenesis of OM23,24,8,25. We hypothesize that OM can be considered as a disease of the ME epithelium since mucosal hyperplasia and proliferation is a characteristic feature. The study objective is to profile molecular changes in the ME epithelium after infection triggered by processes of the innate immune system using in vitro and in vivo OM models. Our study uncovered widespread immune gene regulation in ME epithelial cells
METHODS
MOUSE MODEL.
Wild-type mice studied were obtained from Jackson Laboratory (Bar Harbor, ME) and, following approval by Institutional Animal Care and Use Committee of the Veterans Affairs Medical Center in San Diego, CA, were tested under specific pathogen-free conditions according to National Institutes of Health guidelines conditions. Surgical sampling was carried out under general anesthesia. To induce OM, the Haemophilus influenzae strain 3655 (not typeable, biotype II; NTHi) was inoculated on both sides at a concentration of 105 / ml26.
HUMAN TISSUE SAMPLES
Samples (n=30) of healthy ME mucosa and ME inflammation were obtained with prior consent during ME surgery (tympanoplasty type I-III and cochlear implantation) at the ENT Clinic of the University Hospital Schleswig-Holstein. All studies were performed after approval by the Ethics Committee of the University of Lübeck (10–039, 20–434) and in accordance with the principles of the Declaration of Helsinki (1964). Samples collected were directly processed or temporarily preserved in liquid nitrogen and stored at −80°C27.
HISTOLOGY AND IMMUNOHISTOCHEMISTRY.
Micro surgically resected mouse middle ears and human tissue samples were stained with hematoxylin-eosin (HE) as paraffin sections. Detection of mucosal thickness and histological structure was carried out in specific locations. For immunohistochemistry, paraffin sections were first deparaffinized and rehydrated. Subsequently, after incubation with proteinase K (DAKO, Carpinteria, CA) for antigen generation, incubation with anti-NOD-1 (1:200, Imgenex IMG 5739), anti-NOD-2 (1:600, Santa Cruz, sc 56168) and anti-TLR-2 antibody (Abcam, Cambridge, MA, USA) was carried out. Finally, treatment was carried out using biotin sheep anti-rabbit secondary antibodies (DAKO) and AEC peroxidase substrate kit (Vector Laboratories) according to the manufacturer’s instructions. Detailed protocols have been published previously10,28.
CELL CULTURE.
Human middle ear epithelial cell line (HMEEC)29 was provided by David J. Lim (Department of Head and Neck Surgery, University of California Los Angeles, CA, USA) and cultivated according to protocol30. Immune modulation was performed using NTHi, TNFα, MDP, Tri-DAP, SB203580, and cycloheximide (CHX) alone or in combination with subsequent qPCR analysis after 6 and 24 hours. SB203580 as apoptosis inhibitor and CHX as apoptosis inducer served as negative and positive controls, respectively.
REAL-TIME CELL ANALYSIS ASSAY.
Using xCELLigence™ system (OLS, Bremen, Germany, formerly: Roche, Mannheim, Germany), real-time measurements of cell growth on HMEECs after inoculation with NTHi were performed. In comparison, the measurement was performed on an established cell line overexpressing receptors of the innate immune system. The real-time cell analysis assay was performed according to manufacturer’s instructions. Data were collected every minute for a total of 3 days. The assay was carried out in triplicate.
QUANTITATIVE REAL-TIME PCR (RT-PCR).
It was carried out analogously to the protocol of previously published work of our group31,28. Here, mRNA from HMEEC was obtained using RNeasy Mini Kits (Qiagen, Mississauga, ON, Canada). Using first-strand cDNA synthesis kits (Fermentas, St. Leon-Rot, Germany), 0.5 μg RNA was converted into cDNA, which was then subjected to real-time PCR analysis with Maxima SYBR Green QPCR Master Mix (Fermentas, St. Leon -Rot, Germany) using the gene-specific primers for TNFα, IL-1ß, −6 and −8 and caspase 3, caspase 7 and BID. Cycle conditions were as follows: 50°C for 2 minutes, 95°C for 10 minutes, followed by 40 cycles, each consisting of 95°C for 15 seconds, 60°C for 30 seconds, and 72°C for 30 seconds. Three independent experiments were performed for each target, each with three measurements.
CYTOKINE ANALYSIS.
IL-6 and IL-8 were measured in cell supernatants using Cytometric Bead Array Flex Set System (CBA; BD Bioscience, San Jose, Calif., USA) according to manufacturer’s protocol. This system combines a sandwich immunoassay with flow cytometry. A bead-based immunoassay that analyzes cell supernatants using specific antibody-coated, spectrally addressed polystyrene beads. The cytokines bind to polystyrene beads and a PE-coated detection antibody binds to the cytokines. Cytokine assay was performed using a FACSCanto instrument (BD Biosciences) and data were analyzed using FCAP array software (BD Biosciences).
STATISTICS.
Statistical analysis was performed using StatView and GraphPad Prism, differences at p < 0.05 considered significant. The two ears of each mouse were treated as independent samples, as they were found to be independent of each other. Data are presented as empirical mean with standard deviation. Normally distributed PCR values were processed by ANOVA with Bonferroni correction for multiple testing and an unpaired t-test.
RESULTS
EPITHELIAL MIDDLE EAR RESPONSE.
We investigated the histopathological changes in the ME of wild-type mice after inoculation with NTHi. Before inoculation, we find a free ME cavity with thin flat single row epithelium without cilia and goblet cells in the epitympanum and pseudostratified columnar epithelium with cilia and secretory goblet cells in the area of mesotympanum, eardrum and Eustachian tube (Figure 1, 2A and 2C). After inoculation with NTHi, there is marked hyperplasia of the ME mucosa, altered ciliary covering, deposits of goblet cells, and an exudate of inflammatory cells in the ME cavity, most pronounced after 72 hours. (Figure 2B). Epithelial thickness increases from an average of 17μm to 40μm after inoculation with NTHi. Figure 2D shows the changes in the epithelial response to NTHi inoculation in wild-type mice with a high prismatic pseudostratified ciliated epithelium and goblet cells at day 3. Cell transformation could be confirmed, showing a 100x magnification gram staining with highly prismatic epithelial cells with kinocilia (Figure 2F), compared to a monolayer of cubic squamous epithelial cells of the ME (Figure 2E).
Figure 1.
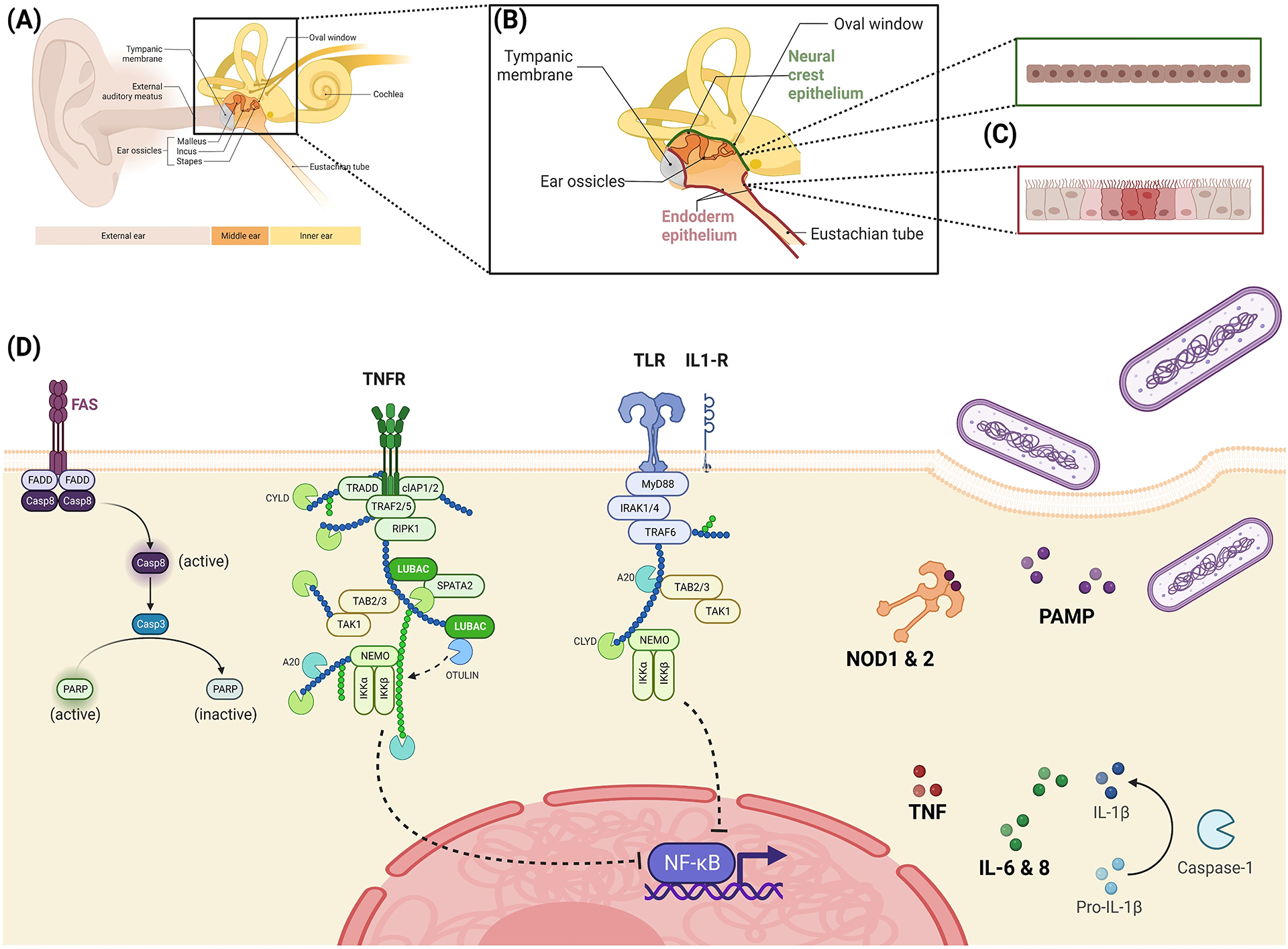
Representation of the signal cascades of the innate immune system of bacterial middle ear infections. Extracellular pathogen-associated molecular patterns (PAMP) via Toll-like receptors (TLRs) and cytoplasmic PAMP via NOD-like receptors (NLRs) lead to the activation of the nuclear factor of kappa-B (NF-κB), a key player in production inflammatory cytokines. Additionally, this production is stimulated by growth factors such as TNFα, which act via the mitogen-activated protein kinase (MAPK) pathway. NOD-2 also can induce PAMP autophagy. As a result of the inflammasome activation via NLRP3, the pro-forms of the cytokines are cleaved into their active form by caspase-1 and then released. In addition to these individual reactions to PAMP, there are also interactions between the various signaling pathways, so it is assumed that the immune response is modulated. [Figure created with BioRender]
Figure 2.

Histology of a middle ear of a wild-type mouse (C57/BL6) and pathological changes by NTHi-induced otitis media. Brackets represent the epithelial thickness. (A) HE staining on non-infected middle ear mucosa (MM) of the middle ear cavity (MEC) shows the tympanic membrane (TM), Eustachian tube (ET), and external auditory canal skin (EAS) (4×). (B) Morphological assessment by HE stain of pathophysiological changes in the middle ear on the 3rd day after NTHi inoculation (4×). (C) The region of interest (ROI) from (A) displays a thin, superficial epithelial layer of the middle ear cavity (40×) (D) Pseudostratified ciliated epithelium and goblet cells on 3rd day after NTHi inoculation in ROI from (B) (40×). (E) Gram staining of the thin, simple epithelial layer of the middle ear from (C) on the 3rd day after NTHi(100×). (F) Gram-stained pseudostratified ciliated epithelium and goblet cells on the 3rd day after NTHi inoculation in ROI from (D) (100×). Arrow points to the kinocilium.
EPITHELIAL EXPRESSION OF INNATE IMMUNE RECEPTORS.
Translational histology of human ME tissue depends on its localization and subsequent immunohistochemical display of pattern recognition receptor expression in OM (Figure 3). Regarding our previous results in mice and human immune patterns in OM31, we focused on specific immune expression in relation to the different epithelia of different origins. Expression of NOD-1, NOD-2, and TLR-2 was successfully detected with different distribution patterns. We see low specific staining for NOD-1 in both localizations, with minimal expression of NOD-1 in the simple cuboid epithelium. NOD-2 and TLR-2 protein expression seem more pronounced in both regions of the epithelium compared to NOD-1 expression. Whereas NOD-2 is predominantly found in the prismatic epithelium of the mesotympanic area, TLR-2 displays stable staining in the epithelium of both origins. (Figure 3).
Figure 3.

Histology of the human middle ear tissue depending on its localization and subsequent immunohistochemical display of the expression of pattern recognition receptors in otitis media. (A) Epitympanum with simple epithelium and mesotympanum with prismatic epithelium with cilia and goblet cells, magnification 20× and 40×, HE section. (B) Immunohistochemical representation of the epithelial expression of innate immune receptors (NOD-1, NOD-2, and TLR-2) in epi- and mesotympanum. Low specific staining for NOD-1 in both localizations. NOD-2 and TLR-2 are found to be stable expressed in the mesotympanic region. Arrows point to the stained target molecules
REAL-TIME CELL ANALYSIS ASSAY OF THE HUMAN MIDDLE EAR EPITHELIAL CELL LINE (HMEEC).
For a better understanding of the functional cell behavior of ME epithelial cells, we performed real-time cell growth measurements on HMEEC in vitro (Figure 4). HMEEC were compared to an already established cell line to investigate the innate immune system after triggering with NTHi. Commercially available HT29 cell line is derived from colonic adenocarcinoma and has variants overexpressing RIP2, NOD-1 and NOD-2. In order to obtain meaningful growth curves, experiments were carried out with cell counts of 30,000 (Figure 4A) and 60,000 (Figure 4B). In both series of experiments, we see continuous almost linear growth of HMEEC cell line until 50 hours, within 60,000 cells (Figure 4A) and 60 hours, within 30,000 cells (Figure 4B). Afterward the epithelial growing curve flattens down and reaches almost the level of a plateau at 70 hours, which is consistent with our data of greatest ME response to NTHi at around 3 days in mice (Figure 2).
Figure 4.

The real-time proliferation of HMEEC after triggering with NTHi. The proliferation of HMEEC in real-time is compared with HT29-WT (human colorectal adenocarcinoma cell line) and its variants overexpressing RIP2 (HT29-RIP2), NOD-1 (HT29-NOD-1), and NOD-2 (HT29-NOD-2) initially seeded at 30 000 (A) and 60 000 (B) cells/mL. While the proliferation rate of HT29 start to decrease after around 25 hours, HMEEC constantly proliferates in the time window of 50–60 hours and flattens down until 70 hours (nearly 3 days).
IMMUNOMODULATION IN THE HUMAN MIDDLE EAR EPITHELIAL CELL LINE (HMEEC).
Due to our previously published role of innate immune system in vivo models (mouse and human tissue) in OM31, investigations of additional effector molecules, apoptosis genes, and protein expression in the in-vitro model of HMEEC were carried out, with focusing on epithelial behavior (Figure 5). Evaluations of cytokines, IL-8, and IL-6 after stimulation of HMEEC with Tri-DAP, MDP, and SB 203580 also in presence of NTHi at 6 or 24 hours in culture are shown in Figure 5A. These data demonstrate that, in addition to the variation in gene expression of TNFα and IL-1β (already partially published in our previous work31), these cytokines have different reaction patterns. We found expression increased in IL-1β, IL-6, IL-8, and TNFα both in sole stimulation and in presence of NTHi. A significant response is seen using the NOD-1 ligand (Tri-DAP), at 6 hours and is most pronounced with expression of TNFα and IL-8. Looking at the NOD-2 signaling pathway, we see significant involvement in epithelial response across all molecules after stimulation with MDP, especially at 24 hours (Figure 5A). Stimulation with map kinase inhibitor (SB203580) showed no remarkable effect at 6 or 24 hours, compared to their specific controls.
Figure 5.

Immunomodulation in the Human Middle Ear Epithelial Cell Line (HMEEC). (A) Gene expression of IL-1ß, IL-6, IL-8, and TNFα after 6 and 24 hours of consecutive stimulation by NTHi, Tri-DAP, MDP, and SB203580 or double stimulation by qPCR. Expression increase in IL-1ß, IL-6, IL-8, and TNFα both in the sole stimulation and occasionally in the presence of NTHi (B) Gene expression of apoptotic genes (caspase 3, caspase 7, BID) after 6 hours and 24 hours of consecutive stimulation with TNFα, Tri-DAP, MDP, SB203580, CHX and in double stimulation with additional NTHi using RT-PCR. CHX served here as a positive control. No significant increase in gene expression, even decreases after 24 hours. (C) Protein expression of IL-6 and IL-8 after 6 hours and 24 hours consecutive stimulation with NTHi, TNFα, Tri-DAP, MDP, and SB203580 and in double stimulation using the Cytometric Bead Array Flex Set System. Significant increase by NOD-1 and NOD-2 pathways.
Examining the apoptosis genes (Figure 5B), the epithelial cells did not react significantly at 6 hours, either after immune stimulation with TNFα, NOD-1 or NOD-2 (SB203580) with or without presence of NTHi at 24 hours. For caspase 3, a reduction in expression level is observed after NOD-receptor immunostimulation alone. However, caspase 7 and BID respond with decreased gene expression after NTHi and/or double stimulation with TNFα and in case of caspase 7 in dual stimulation with the NOD ligands.
We confirmed gene expression using Cytometric Bead Array Flex Set System and exemplified IL-6 and IL-8 at 6 hours and 24 hours (Figure 5C). There is significant increase in IL-6 and expression after stimulation via NTHi, TNFα, as well as the NOD-1 and NOD-2 pathways, presence of NTHi further enhanced this effect. The addition of the p38 MAP kinase inhibitor showed a reduction in IL-6 expression and in presence of NTHi only a significantly delayed expression could be measured.
DISCUSSION
The ME epithelium is the first line of defense against pathogens, a functioning innate immune system is responsible for resolution of OM32 and here rapid reaction of the epithelium is decisive. In addition to pathogen detection and response, epithelial cells also appear to play a role in the secretion of antibacterial peptides and leukocyte migration6. The development of homeostasis of pro- and anti-inflammatory signals is necessary for normal ME mucosa33.
OM leads to increased gene expression of PRR and effector molecules34. Furthermore, it has been shown in numerous animal models that disorders of innate immune system, e.g., absence of PRRs such as TLR-2 or NLR or disruptions in the signal transduction chains via MyD88 or RIP2, lead to bacterial persistence and the resulting chronification of OM26,35,10,15,36.
On histological examination, we see the ME cavity lined by two different types of epithelia. The attic and posterior ME contain simple epithelium, whereas the ventral part and around the eustachian tube consist of prismatic epithelium with cilia and goblet cells. This coincides with studies by Thompson and Tucker4, who were able to demonstrate a different developmental origin in further analyses. The simple epithelium of the attic originates from neural crest and the ciliated epithelium around and beyond the eustachian tube from endoderm24.
As a result of the inoculation with NTHi, we see hyperplasia of ME mucosa and leukocyte infiltration in our well-established mouse model, which peaks at 72 hours and focuses on the different epithelial characteristics.. We find altered expression of innate immune receptors via immunohistochemical staining for the PRRs TLR-2, NOD-1, and NOD-2 in the ME epithelium, supporting the hypothesis of different features of the different epithelial layers due to their origin. Thereby, TLR-2 and NOD-2 could be easily detected in the epithelial layers, showing distinct distribution pattern, while NOD-1 was only slightly expressed overall. These results are in line with results of previous studies, which support the influence of PRR in pathogenesis of OM37,38.
Based on our findings, it can be assumed that expression of TLR-2 and NOD-2 is mainly found in the ventral ME, the eardrum, and the eustachian tube, i.e. the epithelium of endodermal origin. Increased expression of PRR in the area of pseudostratified epithelium suggests that this may be a crucial point in treatment of OM and may thus provide an explanation for observed differences in inflammatory activity and mucosal thickness during ME surgery and different success rates in treatment of OM. It is likely that the simple epithelium, consisting of a monolayer of squamous cells with no occupancy of cilia, therefore has reduced ability to clear bacteria and thus severe courses of OM may occur with the spread of infection into areas of the simple epithelium.
Considering the hypothesis that the epithelium of endodermal origin develops from the upper pharyngeal pouch and thus represents a continuation of the epithelium of the upper airway into the ME, therapeutic approaches, such as biologicals modulating the inflammatory cycle, from this area could also be applicable in treatment of acute and chronic OM. Further indications of similarity to respiratory epithelium are the detection of expression of SOX-2, keratin 5, and Wnt signaling in the mesotympanic area24. However, it must be noted that the hypothesis of different origins of the epithelium is based only on mouse models and so far there are no adequate studies on human ME to support this hypothesis.
The results obtained are all subject to certain limitations. There is no genetic homology between animal and human models and special conditions of animal models do not reflect the real complexity of human OM. Therefore, we need studies with the humanized animal model39. HMEEC lacks phenotypic markers such as ciliary coverage and cannot fully reflect the complexity in-vivo, but shows significant immunomodulatory response to specific immune stimulation.
Based on our knowledge of PRR expression in human tissue and mouse model of OM, we extended our investigations to HMEEC. Here we focused on three aspects of the ME epithelial cells, growth behavior, expression of downstream effector molecules IL-1β, IL-6, IL-8 and TNFα, and expression of apoptosis pathway via caspase-3, caspase 7, and BID.
We analyzed real-time cell growth behavior of HMEEC, compared to an established cellular model system (HT29) that overexpresses relevant genes of innate immune regulation. HMEEC proliferation cell rate corresponds to the response to an infectious stimulus and is consistent with clinical observation of ME epithelial hyperplasia associated with OM with a peak around the 3rd day. Increased expression of IL-1ß and TNFα seen in HMEEC, in response to TLR-2 and NOD activating stimuli, indicates that rapid upregulation of TLR-2 and NOD expression as an innate immune response may prevent increased proliferation of ear epithelial cells during infection. Thus, the mentioned receptors or their downstream molecules could be identified as potential drug targets.
TNFα and the interleukins IL-1β, IL-6, and IL-8 represent the epithelial response to NTHi stimulation. Their rapid increase in expression after 6 hours reflects the rapid response of innate immune system. Stimulation in presence of NTHi, the classic otitis pathogen, is more potent than single stimulation, taking into account the increased protein expression of IL-6 and IL-8 possibly due to the simultaneous response of multiple signaling cascades. But regarding TNFα, it is also a double sword, since dual stimulation at 24 hours with NTHi and either TNFα, Tri-Dap or MDP results in decreased TNFα gene expression, supporting a negative feedback protective mechanism to strong immune reaction/stimulation31,40.
These results confirm the complex role of PRR and their signaling in direct epithelial immune responses. Considering the importance of PRR and the different distribution patterns in the epithelium of the ME due to its different developmental origin, we see epithelial differentiation as a possible future therapeutic approach.
Recovery from OM occurs via apoptosis of the damaged epithelium. In our investigation, we see that, compared to effector molecules, changes in apoptosis genes only occur after 24 hours with tendency to decrease after double stimulation. While caspase 3 displays a reduction in gene expression after NOD-receptor immunostimulation alone, caspase 7 and BID respond with decreased gene expression after NTHi and/or double stimulation with TNFα and/or NOD ligands. One possible explanation could be the time of measurement, meaning that there might be no significant changes in apoptosis at the early state of a ME inflammatory reaction. In summary, dysregulated gene and protein expression of inflammatory and apoptotic genes could be modulated differently by stimulation and inhibition in our presented epithelial cell models, giving us one mosaic more in a complex regulated network.
CONCLUSION
In this work, we were able to present the changes in the epithelium and the following signaling pathways of innate immune system. The transformation of the epithelium and activation of epithelial innate immune system occurs as a result of pathogen contact. Furthermore, we could compare the activity of innate immune system with the hypothesis of the different epithelial types. These results may make it possible to develop new therapeutic approaches to treat OM.
Acknowledgments.
We thank Sylvia Grammerstorf, (Department of Otorhinolaryngology, Lübeck, Germany) for excellent technical support and Jan Rupp (Department of Infectious Diseases and Microbiology, University of Lübeck, Germany) for supporting the bacterial and cell experiments; as well as the late Dr. David J. Lim (University of California, Los Angeles (UCLA)) for providing the human middle ear epithelial cell line (HMEEC).
Funding.
This work was supported by Grant E37-2010 (Anke Leichtle) from the University of Lübeck, the NIH Grants DC000129 and the VA Research Service-Grant (Allen F. Ryan), and NIH Grant DC014801 (Arwa Kurabi).
ABBREVIATIONS
- BID
BH3 interacting-domain death agonist
- CHX
Cycloheximide
- HE
Hematoxylin-eosin stain
- HMEEC
Human Middle Ear Epithelial Cell
- IL-1β
Interleukin 1β
- IL-6
Interleukin 6
- IL-8
Interleukin 8
- MDP
Muramyl dipeptide
- NLR
NOD-like receptor
- NOD-1/2
Nucleotide-binding oligomerization domain receptor1/2
- ME
Middle ear
- MyD88
Myeloid differentiation primary response 88
- NTHi
Not typable Haemophilus influenza
- OM
Otitis Media
- PAMPS
Pathogen-associated molecular patterns
- PRR
Pattern Recognition Receptors
- RIP2
Receptor Interacting Serine/Threonine Kinase 2
- SB203580
Pyridinyl imidazole and p38 MAP Kinase Inhibitor
- TLR
Toll-like receptor
- TNFα
Tumor necrosis factor α
- Tri-DAP
L-Ala-γ-D-Glu-mDAP
Footnotes
Conflict of Interest. The authors declare that they have no other competing interests, commercial or financial relationships.
Allen Ryan is a co-founder of Otonomy Inc., serves as a member of the Scientific Advisory Board, and holds an equity position in the company. The UCSD Committee on Conflict of Interest has approved this relationship. Otonomy, Inc. played no part in the research reported here.
References
- 1.Monasta L, Ronfani L, Marchetti F, et al. Burden of disease caused by otitis media: systematic review and global estimates. PLoS One. 2012;7(4):e36226. doi: 10.1371/journal.pone.0036226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Arnold W Reaktionsformen der Mittelohrschleimhaut. Arch Otorhinolaryngol. 1977;216(1):369–473. [PubMed] [Google Scholar]
- 3.Lang J Klinische Anatomie des Ohres. Springer Wien; 1992. [Google Scholar]
- 4.Thompson H, Tucker AS. Dual origin of the epithelium of the mammalian middle ear. Science. 2013;339(6126):1453–1456. doi: 10.1126/science.1232862 [DOI] [PubMed] [Google Scholar]
- 5.Mulay A, Akram KM, Williams D, et al. An in vitro model of murine middle ear epithelium. Dis Model Mech. 2016;9(11):1405–1417. doi: 10.1242/dmm.026658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ryan AF, Nasamran CA, Pak K, et al. Single-Cell Transcriptomes Reveal a Complex Cellular Landscape in the Middle Ear and Differential Capacities for Acute Response to Infection. Front Genet. 2020;11:358. doi: 10.3389/fgene.2020.00358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Takeuchi O, Akira S. Pattern recognition receptors and inflammation. Cell. 2010;140(6):805–820. doi: 10.1016/j.cell.2010.01.022 [DOI] [PubMed] [Google Scholar]
- 8.Kurabi A, Pak K, Ryan AF, Wasserman SI. Innate Immunity: Orchestrating Inflammation and Resolution of Otitis Media. Curr Allergy Asthma Rep. 2016;16(1):6. doi: 10.1007/s11882-015-0585-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kawai T, Akira S. The roles of TLRs, RLRs and NLRs in pathogen recognition. Int Immunol. 2009;21(4):317–337. doi: 10.1093/intimm/dxp017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Leichtle A, Hernandez M, Pak K, et al. TLR4-mediated induction of TLR2 signaling is critical in the pathogenesis and resolution of otitis media. Innate Immun. 2009;15(4):205–215. doi: 10.1177/1753425909103170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Leichtle A, Hernandez M, Ebmeyer J, et al. CC chemokine ligand 3 overcomes the bacteriocidal and phagocytic defect of macrophages and hastens recovery from experimental otitis media in TNF−/− mice. J Immunol. 2010;184(6):3087–3097. doi: 10.4049/jimmunol.0901167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ngo CC, Massa HM, Thornton RB, Cripps AW. Predominant Bacteria Detected from the Middle Ear Fluid of Children Experiencing Otitis Media: A Systematic Review. PLoS One. 2016;11(3):e0150949. doi: 10.1371/journal.pone.0150949 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Huang Y, Wang Z, Jin C, et al. TLR2 promotes macrophage recruitment and Streptococcus pneumoniae clearance during mouse otitis media. Pediatr Res. 2016;80(6):886–893. doi: 10.1038/pr.2016.154 [DOI] [PubMed] [Google Scholar]
- 14.Strober W, Murray PJ, Kitani A, Watanabe T. Signalling pathways and molecular interactions of NOD1 and NOD2. Nat Rev Immunol. 2006;6(1):9–20. doi: 10.1038/nri1747 [DOI] [PubMed] [Google Scholar]
- 15.Lee J, Leichtle A, Zuckerman E, et al. NOD1/NOD2-mediated recognition of non-typeable Haemophilus influenzae activates innate immunity during otitis media. Innate Immun. 2019;25(8):503–512. doi: 10.1177/1753425919872266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Johnson M, Leonard G, Kreutzer DL. Murine model of interleukin-8-induced otitis media. Laryngoscope. 1997;107(10):1405–1408. doi: 10.1097/00005537-199710000-00020 [DOI] [PubMed] [Google Scholar]
- 17.Nassif PS, Simpson SQ, Izzo AA, Nicklaus PJ. Interleukin-8 concentration predicts the neutrophil count in middle ear effusion. Laryngoscope. 1997;107(9):1223–1227. doi: 10.1097/00005537-199709000-00012 [DOI] [PubMed] [Google Scholar]
- 18.Baltimore D, Beg AA. DNA-binding proteins. A butterfly flutters by. Nature. 1995;373(6512):287–288. doi: 10.1038/373287a0 [DOI] [PubMed] [Google Scholar]
- 19.Kieran M, Blank V, Logeat F, et al. The DNA binding subunit of NF-κB is identical to factor KBF1 and homologous to the rel oncogene product. Cell. 1990;62(5):1007–1018. doi: 10.1016/0092-8674(90)90275-j [DOI] [PubMed] [Google Scholar]
- 20.Fujisawa K, Aono H, Hasunuma T, Yamamoto K, Mita S, Nishioka K. Activation of transcription factor NF-kappa B in human synovial cells in response to tumor necrosis factor alpha. Arthritis Rheum. 1996;39(2):197–203. doi: 10.1002/art.1780390205 [DOI] [PubMed] [Google Scholar]
- 21.Fakler CR, Wu B, McMicken HW, Geske RS, Welty SE. Molecular mechanisms of lipopolysaccharide induced ICAM-1 expression in A549 cells. Inflamm Res. 2000;49(2):63–72. doi: 10.1007/s000110050560 [DOI] [PubMed] [Google Scholar]
- 22.Melotti P, Nicolis E, Tamanini A, Rolfini R, Pavirani A, Cabrini G. Activation of NF-kB mediates ICAM-1 induction in respiratory cells exposed to an adenovirus-derived vector. Gene Ther. 2001;8(18):1436–1442. doi: 10.1038/sj.gt.3301533 [DOI] [PubMed] [Google Scholar]
- 23.Tateossian H, Morse S, Parker A, et al. Otitis media in the Tgif knockout mouse implicates TGFβ signalling in chronic middle ear inflammatory disease. Hum Mol Genet. 2013;22(13):2553–2565. doi: 10.1093/hmg/ddt103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tucker AS, Dyer CJ, Fons Romero JM, Teshima THN, Fuchs JC, Thompson H. Mapping the distribution of stem/progenitor cells across the mouse middle ear during homeostasis and inflammation. Development. 2018;145(1). doi: 10.1242/dev.154393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Juhn SK, Jung M-K, Hoffman MD, et al. The role of inflammatory mediators in the pathogenesis of otitis media and sequelae. Clin Exp Otorhinolaryngol. 2008;1(3):117–138. doi: 10.3342/ceo.2008.1.3.117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hernandez M, Leichtle A, Pak K, et al. Myeloid differentiation primary response gene 88 is required for the resolution of otitis media. J Infect Dis. 2008;198(12):1862–1869. doi: 10.1086/593213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Leichtle A, Leffers D, David CS, Ryan AF, Bruchhage K-L. Immunmodulation im Cholesteatom. In: Deutsche Gesellschaft für Hals-Nasen-Ohren-Heilkunde, Kopf- und Hals-Chirurgie e.V., Bonn. Georg Thieme Verlag KG; 2021. [Google Scholar]
- 28.Leichtle A, Klenke C, Ebmeyer J, et al. NOD-Like Receptor Signaling in Cholesteatoma. Biomed Res Int. 2015;2015:408169. doi: 10.1155/2015/408169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chun Y-M, Moon S-K, Lee H-Y, et al. Immortalization of normal adult human middle ear epithelial cells using a retrovirus containing the E6/E7 genes of human papillomavirus type 16. Ann Otol Rhinol Laryngol. 2002;111(6):507–517. doi: 10.1177/000348940211100606 [DOI] [PubMed] [Google Scholar]
- 30.Woo J-I, Oh S, Webster P, Lee YJ, Lim DJ, Moon SK. NOD2/RICK-dependent β-defensin 2 regulation is protective for nontypeable Haemophilus influenzae-induced middle ear infection. PLoS One. 2014;9(3):e90933. doi: 10.1371/journal.pone.0090933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Leichtle A, Kurabi A, Leffers D, et al. Immunomodulation as a Protective Strategy in Chronic Otitis Media. Front Cell Infect Microbiol. 2022;12:826192. doi: 10.3389/fcimb.2022.826192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Underwood M, Bakaletz L. Innate immunity and the role of defensins in otitis media. Curr Allergy Asthma Rep. 2011;11(6):499–507. doi: 10.1007/s11882-011-0223-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hernandez M, Leichtle A, Pak K, Webster NJ, Wasserman SI, Ryan AF. The transcriptome of a complete episode of acute otitis media. BMC Genomics. 2015;16:259. doi: 10.1186/s12864-015-1475-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Leichtle A, Lai Y, Wollenberg B, Wasserman SI, Ryan AF. Innate signaling in otitis media: pathogenesis and recovery. Curr Allergy Asthma Rep. 2011;11(1):78–84. doi: 10.1007/s11882-010-0158-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ebmeyer J, Leichtle A, Hernandez M, et al. TNFA deletion alters apoptosis as well as caspase 3 and 4 expression during otitis media. BMC Immunol. 2011;12:12. doi: 10.1186/1471-2172-12-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kurabi A, Lee J, Pak K, Leichtle A, Ryan AF. Essential Role of the Innate Immune Adaptor RIP2 in the Response to Otitis Media. Front Genet. 2022;13:893085. doi: 10.3389/fgene.2022.893085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Granath A, Cardell L-O, Uddman R, Harder H. Altered Toll- and Nod-like receptor expression in human middle ear mucosa from patients with chronic middle ear disease. J Infect. 2011;63(2):174–176. doi: 10.1016/j.jinf.2011.06.006 [DOI] [PubMed] [Google Scholar]
- 38.Hirai H, Kariya S, Okano M, et al. Expression of toll-like receptors in chronic otitis media and cholesteatoma. Int J Pediatr Otorhinolaryngol. 2013;77(5):674–676. doi: 10.1016/j.ijporl.2013.01.010 [DOI] [PubMed] [Google Scholar]
- 39.Son YL, Pak K, Muradagha N, Heo KW, Leichtle A, Kurabi A. Resolution of otitis media in a humanized mouse model. Front Genet. 2022;13:958540. doi: 10.3389/fgene.2022.958540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hruz P, Eckmann L. Caspase recruitment domain-containing sensors and adaptors in intestinal innate immunity. Curr Opin Gastroenterol. 2008;24(2):108–114. doi: 10.1097/MOG.0b013e3282f50fdf [DOI] [PMC free article] [PubMed] [Google Scholar]


