Abstract
Trauma exposure, whether experienced during childhood or adulthood, is prevalent among women. While experiences of trauma are well known to impact mental health, emerging research also links them to women’s physical health. The Study of Women’s Health Across the Nation (SWAN) and the MsHeart/MsBrain studies, two separate studies devoted to studying midlife women’s health, have contributed importantly to the understanding of the implications of trauma to women’s health at midlife and beyond. Specifically, findings from these studies have revealed that both childhood and adult trauma exposure are associated with poorer cardiovascular and cerebrovascular health in women, including greater subclinical cardiovascular disease, indicators of cerebral small-vessel disease, and increased risk for clinical cardiovascular disease events. When considering trauma types, findings have pointed to the particular importance of sexual and interpersonal violence, such as childhood sexual abuse, intimate-partner violence, sexual harassment, and sexual assault to women’s vasculatures. Further, using a range of measures of menopausal vasomotor symptoms, the SWAN and the MsHeart/MsBrain studies have also shown that women with greater trauma exposure have more objectively assessed and self-reported vasomotor symptoms. Finally, although links between trauma exposure and health are not typically explained by post-traumatic stress disorder, work also points to the additional importance of post-traumatic stress disorder to women’s cardiovascular and brain health. Collectively, these studies have underscored the importance of trauma to the occurrence of menopausal symptoms, to cardiovascular health, and to women’s brain health at midlife and beyond. Future directions and implications for prevention and intervention are discussed.
Keywords: trauma, abuse, post-traumatic stress disorder, menopause, cardiovascular disease, brain aging
Introduction
Most women will experience at least one traumatic event in their lifetime. At least a quarter of women will experience maltreatment in childhood (e.g., childhood physical abuse, emotional abuse, sexual abuse, neglect) (1, 2), and up to 69% of women will experience a traumatic event in adulthood (3). Among these traumatic experiences, sexual and interpersonal trauma, such as intimate partner violence (IPV), sexual assault, workplace sexual harassment, and childhood sexual abuse are particularly prevalent among women. For example, over a quarter of women endorse a history of intimate partner violence (4), and 44% of women a history of sexual assault (5). These traumatic experiences are well-established to negatively impact mental health. However, an emerging body of research also underscores their importance to women’s physical health.
The menopause transition is a particularly relevant time to consider the implications of trauma for women’s health. The menopause is a critical time for women’s cardiovascular health, as it is accompanied by accelerated increases in carotid atherosclerosis, vascular stiffening, adiposity, adverse lipid changes, and metabolic syndrome occurrence beyond the effects of aging alone (6). The menopause is also a time of occurrence of symptoms such as vasomotor symptoms (VMS), sleep problems, and mood changes that may further exacerbate the adverse cardiovascular and mental health effects of trauma exposure (7). Thus, the menopause can be conceptualized as a time of vulnerability to both poor mental and cardiovascular health, when these systems may be particularly susceptible to stress-related insults such as trauma or abuse.
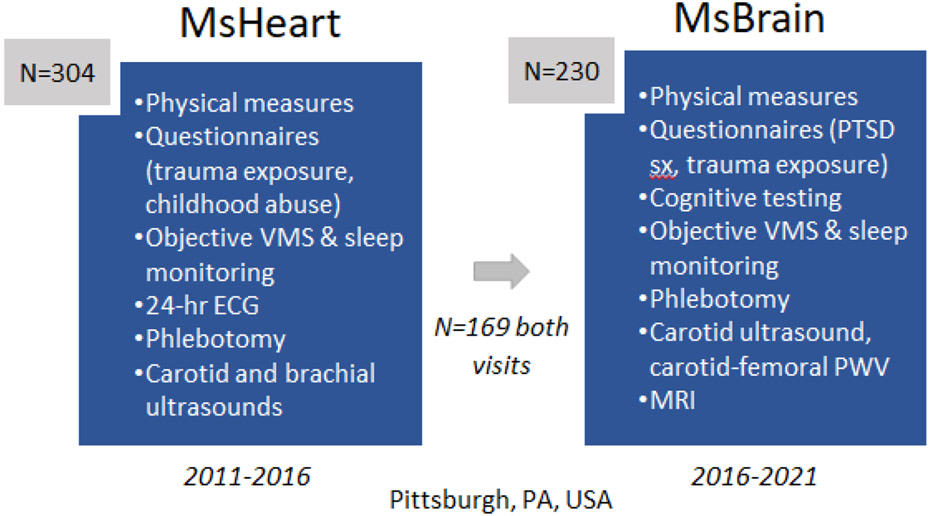
The Study of Women’s Health Across the Nation (SWAN) and the MsHeart/MsBrain studies are two studies that have provided critical insights about the importance of trauma exposure, experienced in either childhood or adulthood, for midlife women’s health (see Figures 1 and 2 for graphical overviews). SWAN is a longitudinal epidemiologic cohort study conducted at seven sites around the United States. SWAN was designed to characterize the natural history of the menopause transition and its implications for women’s health (8). SWAN enrolled 3302 participants between the ages of 42 and 52 and assessed them for up 16 study visits over 27 years; at each visit, participants underwent comprehensive assessments, including of cardiovascular health and trauma. The MsHeart (N=304) and MsBrain (N=239) studies are two clinical studies that together form a longitudinal study (N=169 participated in both visits) conducted in Pittsburgh, Pennsylvania, United States. The MsHeart/MsBrain studies were originally designed to investigate the implications of menopausal symptoms for women’s cardiovascular and brain health and leveraged objective assessments of sleep and VMS, measures of trauma and post-traumatic stress disorder (PTSD) symptoms, a 24-hour electrocardiogram, and vascular and neuroimaging to deeply phenotype women’s health (9, 10). While distinct studies, SWAN, an epidemiologic cohort study, and MsHeart/MsBrain, clinical studies providing in-depth measurements, together provide a synergistic perspective on trauma and women’s cardiovascular health at midlife.
Figure 1.
Graphical overview of the Study of Women’s Health Across the Nation
Figure 2.
Graphical overview of the MsHeart/MsBrain studies
Methods
This review summarizes results from SWAN and the MsHeart/MsBrain studies on trauma exposure and women’s cardiovascular health during midlife and beyond. Relevant manuscripts were identified by searching PubMed [All fields] and the SWAN study database for manuscripts published or in press by December 15, 2023 using terms: “abuse”, “trauma”, “violence”, “intimate partner violence”, “harassment,” “cardiovascular disease”, “cardiovascular”, “vascular”, “atherosclerosis”, “subclinical cardiovascular disease”, “heart disease”, “intima media thickness”, “inflammation”, “heart rate variability”, “autonomic nervous system”, "SWAN", "Study of Women's Health Across the Nation", "MsHeart", and "MsBrain". Select manuscripts were also identified based upon a manual search of reference lists.
Subclinical CVD
Childhood Maltreatment.
Subclinical cardiovascular disease (CVD) measures use imaging modalities to index the health of the vasculature before clinical disease is present and are particularly useful for indexing the vascular health of midlife women, who rarely present with clinical disease. Both SWAN and MsHeart implemented subclinical CVD measures. Among 1402 SWAN participants, we considered the relationship between childhood or adult physical or sexual abuse (each assessed via single questions) in relation to carotid intima media thickness (IMT) (11), a well-validated ultrasound measure of subclinical atherosclerosis predictive of future clinical CVD (12). Notably, 16% of the SWAN cohort endorsed a history of childhood sexual abuse, with the highest rates among the African American participants (20%). SWAN participants with a history of childhood sexual abuse had higher IMT than women without this history, adjusting for demographic factors, CVD risk factors, and negative mood (11).
In MsHeart, we also considered associations between childhood abuse or neglect (collectively termed maltreatment) and carotid IMT as well as carotid plaque (a subclinical CVD indicator particularly related to future clinical CVD in women (13)). In MsHeart, 45% of the cohort scored positive for a history of childhood maltreatment on a validated multidimensional assessment of childhood abuse (14). Childhood maltreatment, including physical abuse, emotional abuse, and emotional neglect, was related to higher carotid IMT and plaque. These associations were not explained by demographics, CVD risk factors, mood, nor by factors such as sleep or VMS, which we previously linked to subclinical CVD (9, 15). Although menopausal symptoms did not explain relationships between childhood maltreatment and IMT/plaque, a synergy between trauma exposure and objectively-assessed menopausal symptoms emerged, such that women with a history of childhood abuse and sleep VMS or short sleep (<6 hours) had the highest IMT or plaque (14). Conversely, women with a history of childhood maltreatment who were sleeping adequately or did not have sleep VMS had a comparable IMT to their non-abused counterparts. We subsequently observed similar links between childhood abuse and vascular stiffness (16). Thus, a history of childhood maltreatment was associated with more subclinical CVD at midlife, and the adverse vascular sequelae of childhood abuse appeared most pronounced among women experiencing sleep VMS or short sleep.
Lifetime Trauma Exposure.
In addition to childhood abuse, we considered lifetime or adult trauma exposure in relation to subclinical CVD. We administered a 9-item trauma checklist which included queries about experiences such as sexual assault, workplace sexual harassment, a serious accident, natural disaster, muggings, and the death of a child. Over 60% of the women endorsed at least one traumatic experience, and 18% experienced three or more traumas (17). Exposure to more traumatic events, particularly three or more, was associated with poorer endothelial function as assessed by brachial artery flow mediated dilation, a validated measure of endothelial function (17, 18). These associations were not explained by standard CVD risk factors or by anxiety. Similar to IMT, there was indication that the relationship between trauma and endothelial function was particularly pronounced when accompanied by short sleep.
Sexual Violence.
Sexual violence is a prevalent, intimate, and invasive form of trauma and thereby a particularly potent trauma exposure. In one of the first studies to consider workplace sexual harassment in relation to measured blood pressure, we found that among MsHeart participants, women with a history of workplace sexual harassment (19%) had an over doubling of the odds of hypertension (19). We also tested associations between sexual assault and vascular health among the MsHeart/MsBrain participants (N=169) who participated in both visits and thereby underwent a carotid ultrasound twice over five years. Notably, 28% of participants endorsed a sexual assault history. A sexual assault history was associated with over 4-fold odds of elevated carotid plaque at baseline, over 3-fold odds of elevated carotid plaque at follow-up, and accelerated plaque progression between over 5 years, adjusting for confounders and CVD risk factors (20). Neither depression nor post-traumatic symptoms were related to plaque, suggesting that these associations are independent of ongoing psychological symptoms. Collectively, these findings show that women who have experienced sexual violence have poorer vascular health at midlife as well as accelerated degradations in vascular health as they age.
Cerebrovascular Health
We considered whether associations of trauma exposure to women’s peripheral vascular health extended to women’s cerebrovasculature, testing these associations among 145 MsBrain participants who underwent brain magnetic resonance imaging (MRI) to characterize brain white matter hyperintensities (WMHs), which are markers of small vessel disease in the brain linked to increased risk for stroke, dementia, and mortality later in life (21). Findings indicated that women with a lifetime trauma history had greater brain WMHs (22). These associations were not explained by CVD risk factors, mood, PTSD symptoms, or by a history of childhood abuse. Notably, the single trauma most strongly associated with WMHs was sexual assault, further underscoring the potency of sexual trauma to women’s vascular health.
Cardiovascular Disease (CVD) Events
As women age, they progress from subclinical atherosclerosis to clinical CVD events, including heart attacks and strokes. As SWAN participants have been followed into their 70’s, they have entered a stage in the lifecourse in which they are beginning to experience CVD events. Therefore, among SWAN participants (N=2201), we considered the relationship of childhood abuse, adult abuse, and IPV to risk of CVD events, a combined outcome of hearts attacks, strokes, revascularization, and CVD mortality, over 22 years of follow up. Women with a history of childhood abuse had a 1.65 increased risk of a later CVD event, after adjusting for CVD risk factors and depression (23). Childhood sexual abuse was particularly deleterious, associated with a doubling of the risk of a CVD event later in life. We also considered IPV. Over 26% of women endorsed IPV, which was principally emotional IPV. Women with an IPV history had a doubling of the risk of a CVD event relative to women without this history. Notably, the associations between IPV and CVD were mediated through elevated systolic blood pressure among IPV-exposed women. Thus, childhood and adult trauma, particularly sexual and interpersonal violence, were associated with increased risk for CVD events later in life, and in the case of IPV, blood pressure played an important role in these relationships.
Select Mechanisms
The mechanisms that may link trauma exposure to CVD risk are multiple, and include social and structural pathways, psychological and affective factors, health behaviors, as well as direct stress-sensitive biologic pathways (24). We have examined select mechanisms, with a focus on the immune and autonomic nervous systems which may be potential physiologic pathways linking psychosocial stress to cardiovascular health.
Inflammation.
We considered the relationship between childhood maltreatment and inflammation. Among SWAN participants at the Pittsburgh site (N=326), a history of childhood abuse or neglect, experienced by 37% of the women, was associated with higher high-sensitivity C-reactive protein (hsCRP) over 7 years, adjusting for covariates. The types of maltreatment associated with hsCRP were sexual abuse, emotional neglect, and physical neglect. Body mass index (BMI) was a significant mediator, such that several of these relationships were explained by the higher BMI of the maltreated women (25). We also considered the relationship between childhood maltreatment and inflammation in MsHeart (N=304), finding that women with a history of emotional abuse (experienced by 24% of the women) had higher interleukin-6 levels than women without this history (26). These associations were robust to adjustment of covariates including BMI.
Heart Rate Variability.
Another potential mechanism that may link trauma to cardiovascular disease risk is via the autonomic nervous system, with reduced parasympathetic nervous system influence over the heart considered a putative pathway by which psychosocial stress may increase cardiovascular risk (27-29). High frequency heart rate variability (HF-HRV) measured via electrocardiogram is an index of parasympathetic influence over heart rate. Findings indicated that a greater number of lifetime trauma exposures were associated with reduced HF-HRV during wake and sleep over 24 hours, with results particularly pronounced for HF-HRV during sleep (30). Further, when considering sexual abuse, a history of childhood sexual abuse was associated with pronounced reductions in HF-HRV, and largely during sleep. Finally, women with both a history of lifetime trauma exposure and of childhood sexual abuse had the lowest sleep HF-HRV, suggesting a compounding effect of child and adult traumatic experiences. Notably, none of these effects were explained by sleep characteristics measured via actigraphy. Together, these findings indicated that women with trauma exposure had reduced parasympathetic influence over the heart, with these effects most pronounced during sleep, yet not explained by sleep itself.
Menopausal Symptoms
Vasomotor Symptoms (VMS).
VMS, or hot flashes and night sweats, are the hallmark symptom of menopause and are experienced by most midlife women.(7) VMS are associated with pronounced impairment in quality of life, and we and others have found VMS associated with elevated CVD risk (31). In the Pittsburgh SWAN cohort, women with a history of childhood abuse or neglect had a 1.55-1.73 increased odds of reporting hot flashes and night sweats, respectively, over 8 years of midlife, after controlling for confounding factors (32). There were also racial/ethnic differences. Relative to the White women, the Black women were particularly affected, such that among the Black women, childhood abuse associated with an over doubling of odds of VMS. We also considered childhood abuse in relation to VMS in MsHeart, a study notable for its implementation of physiological assessments of VMS. Among women with VMS, those with a history of childhood physical or sexual abuse had 1.5-2 fold more physiologically-documented VMS during sleep than women without this history; these associations were not explained by factors such as BMI, mood, or sleep (33). Thus, women with an abuse history had not only increased reporting of VMS, but also increased physiologic occurrence of VMS, particularly nocturnally.
Sleep.
Sleep problems are common during the menopause transition. In MsHeart, a sexual assault or sexual harassment history were each associated with an approximate doubling of the odds of sleep symptoms consistent with clinical insomnia (19). Further, in emerging longitudinal findings from the MsHeart/MsBrain studies, women with a history of childhood abuse or neglect had a doubling of the odds of persistently elevated actigraphy-assessed waking during the night than women without this history; further, women with lifetime trauma history had a doubling of the odds of persistently poor subjective sleep quality (34). These findings were not explained by psychological symptoms.
Collectively, these data indicate that women with trauma histories have a more symptomatic menopause transition. As VMS and poor sleep are linked to poorer cardiovascular health during the menopause transition (15, 31), these symptoms may further amplify the adverse cardiovascular implications of a trauma history (30, 35-37).
The Role of Post-Traumatic Stress Disorder (PTSD)
PTSD is a major mental health outcome of trauma, with clinical PTSD experienced by approximately 12% of women in their lifetime (3). Although PTSD and other psychiatric symptoms do not typically account for relationships between trauma exposure and women’s cardiovascular health in these studies, PTSD does have implications for women’s health. In MsBrain, we found that women with greater PTSD symptoms had higher carotid IMT than women with lower levels of these symptoms; associations were not explained by demographic factors, CVD risk factors, or depressive symptoms (38). In addition, among carriers of the apolipoprotein epsilon 4 (APOEε4) genotype, a key risk factor for dementia, PTSD symptoms were associated with greater brain white matter hyperintensities and poorer cognitive performance (38). Importantly, the PTSD symptoms endorsed in MsBrain were largely in the subclinical range: Only 19% of the sample had clinically elevated symptoms, indicating the clinical importance of even subclinical levels of PTSD symptoms for women’s heart and brain health. Given the availability of effective treatments for PTSD, this work is particularly relevant to identifying points of potential intervention.
Summary, Next Steps, and Clinical Implications
SWAN and the MsHeart/MsBrain studies have underscored the importance of childhood maltreatment, lifetime trauma exposure, and PTSD for women’s cardiovascular health at midlife. Midlife women with trauma histories had greater subclinical CVD, poorer cerebrovascular health, as well as greater risk for clinical CVD events such as heart attacks, strokes, and CVD mortality as they aged. These associations persisted after accounting for potential explanatory factors, such as CVD risk factors, psychological symptoms, and sleep. When specific traumas were considered, sexual violence, including sexual assault, sexual harassment, or childhood sexual abuse, were particularly toxic to the vasculature. Greater systemic inflammation and reduced parasympathetic control over the heart, particularly during sleep, may be key pathways linking trauma exposure to cardiovascular health. In certain cases, elevated BMI and/or blood pressure were pathways. Another notable finding is that women with a trauma history had more menopausal symptoms, including VMS and poor sleep. In fact, these menopausal symptoms may synergize with trauma exposure, such that women with both a trauma history and VMS or short sleep may be at greatest cardiovascular risk. Finally, although links between trauma exposure and women’s health are typically not explained by PTSD, PTSD symptoms, even at subclinical levels, were associated with cardiovascular and neurocognitive risk in our work. Collectively, these data point to the importance of trauma to the experience of the menopause transition as well as women’s cardiovascular and cerebrovascular health at midlife and beyond.
There are important next steps of this work. Sexual violence was a particularly potent exposure; future work should consider more in-depth assessment of sexual violence, including the timing and chronicity of exposures, non-contact forms of sexual violence such as stalking and harassment (39), as well as any treatments received. Further, other features of the menopause experience, such as menopause timing or type (40) and a wider range of menopausal symptoms (41) should be considered. As both the rates of exposure to trauma and CVD can vary markedly by race/ethnicity, further considering racial/ethnic differences in the impact of trauma exposure on health is critical. Moreover, identifying modifiable factors that can build resilience to the adverse effects of earlier life trauma is imperative to providing women with agency to mitigate the adverse impacts of early life trauma. Initial work identifies adequate sleep as a potential buffer to protect the vasculature of trauma-exposed women, but further investigation is warranted. Finally, it is not yet known whether treating trauma-related symptoms may improve cardiovascular health. Tests of a PTSD intervention to improve cardiovascular health suggest modest yet potential benefits to the autonomic nervous system (42); further examination of trauma-related interventions specifically addressing the needs of midlife women is warranted.
The clinical implications of this work are multiple. First, clinicians working with trauma-exposed populations should familiarize themselves with trauma-informed principles of care, which emphasize safety, trustworthiness, transparency, peer support, collaboration, empowerment, and choice, as well as cultural and gender awareness (43). An understanding of an individual’s trauma history, such childhood abuse or neglect, and major traumatic experiences in adulthood including sexual violence, can help healthcare providers identify individuals most at risk for adverse cardiovascular outcomes. Women with trauma histories, particularly multiple traumas, may warrant focused CVD risk reduction efforts to prevent CVD later in life. However, although trauma screening may be warranted, disclosure of traumatic experiences requires trust, rapport, and assurance of confidentiality in a safe patient-provider relationship. Therefore, guidance for addressing traumas such as IPV in medical settings often recommends moving beyond a simple screening approach to instead provide universal education and resources (44), an approach that does not require disclosure and has the additional advantages of promoting education, awareness, and peer-to-peer education. Screening for common mental health sequelae of trauma, such as PTSD, depression, and substance use disorders should be routine; if these conditions are detected, women merit referral to and connection with behavioral health providers who are trained in trauma-focused therapies. Finally, women with childhood trauma histories are more likely to suffer from menopausal symptoms that can further degrade mental health and potentially cardiovascular health during the menopause transition; these symptoms warrant management.
Thus, a history of trauma, particularly if experienced multiply, may have pleotropic effects across multiple bodily systems, increasing the likelihood that trauma-exposed women will have a more symptomatic menopause transition as well as greater CVD risk as they age. These findings underscore the persistent impact of trauma exposure: even trauma exposure experienced quite early in life can have implications over the life course. Focused attention and intervention for trauma-exposed populations and public health interventions aimed at trauma prevention may be critical to protecting women’s mental and physical health at midlife and beyond.
Highlights.
A history of childhood and/or adult trauma is common among midlife women.
Trauma is linked to poorer cardiovascular and cerebrovascular health at midlife.
Midlife women with a history of childhood abuse have more vasomotor symptoms.
Sexual and interpersonal violence may be particularly toxic for the vasculature.
Acknowledgements
The Study of Women’s Health Across the Nation (SWAN) has grant support from the National Institutes of Health (NIH), DHHS, through the National Institute on Aging (NIA), the National Institute of Nursing Research (NINR) and the NIH Office of Research on Women’s Health (ORWH) (U01NR004061, U01AG012505, U01AG012535, U01AG012531, U01AG012539, U01AG012546, U01AG012553, U01AG012554, U01AG012495, and U19AG063720). This research was also supported by the NIH, NIA (RF1AG053504 and R01AG53504 to Thurston and Maki; NIH National Heart Lung and Blood Institute (R01HL105647 to Thurston and K24HL123565 to Thurston). This work was also supported by the University of Pittsburgh Clinical and Translational Science Institute (NIH Grant UL1T R000005); University of Pittsburgh Small Molecule Biomarker Core (NIH Grant S10RR023461).
Funding
No funding from an external source was received for the preparation of this review.
Abbreviations
- APOEε4
apolipoprotein epsilon 4
- BMI
body mass index
- CVD
cardiovascular disease
- HF-HRV
high-frequency heart rate variability
- IMT
intima media thickness
- IPV
intimate partner violence
- MRI
magnetic resonance imaging
- PTSD
post-traumatic stress disorder
- SWAN
Study of Women’s Health Across the Nation
- VMS
vasomotor symptoms
- WMH
white matter hyperintensities
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Declaration of competing interest
Dr. Thurston is a consultant/advisor for Astellas, Bayer, and Hello Therapeutics.
References
- 1.Wildeman C, Emanuel N, Leventhal JM, Putnam-Hornstein E, Waldfogel J, Lee H. The prevalence of confirmed maltreatment among US children, 2004 to 2011. JAMA Pediatr. 2014;168(8):706–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kim H, Wildeman C, Jonson-Reid M, Drake B. Lifetime Prevalence of Investigating Child Maltreatment Among US Children. Am J Public Health. 2017;107(2):274–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Resnick HS, Kilpatrick DG, Dansky BS, Saunders BE, Best CL. Prevalence of civilian trauma and posttraumatic stress disorder in a representative national sample of women. J Consult Clin Psychol. 1993;61(6):984–91. [DOI] [PubMed] [Google Scholar]
- 4.Black MC, Basile KC, Breiding MJ, Smith SG, Walters ML, Merrick MT, Chen J, & Stevens MR The National Intimate Partner and Sexual Violence Survey (NISVS): 2010 Summary Report. Atlanta, GA: National Center for Injury Prevention and Control; 2011. [Google Scholar]
- 5.Smith SG, Zhang X, Basile KC, Merrick MT, Wang J, Kresnow M, Chen J The National Intimate Partner and Sexual Violence Survey : 2015 data brief – updated release. Report. Atlanta, GA; 2018. [Google Scholar]
- 6.El Khoudary SR, Aggarwal B, Beckie TM, Hodis HN, Johnson AE, Langer RD, et al. Menopause Transition and Cardiovascular Disease Risk: Implications for Timing of Early Prevention: A Scientific Statement From the American Heart Association. Circulation. 2020;142(25):e506–e32. [DOI] [PubMed] [Google Scholar]
- 7.Gold EB, Colvin A, Avis N, Bromberger J, Greendale GA, Powell L, et al. Longitudinal analysis of the association between vasomotor symptoms and race/ethnicity across the menopausal transition: study of women's health across the nation. Am J Public Health. 2006;96(7):1226–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sowers M, Crawford S, Sternfeld B, Morganstein D, Gold E, Greendale G, et al. SWAN: A Multicenter, Multiethnic, Community-Based Cohort Study of Women and the Menopausal Transition. In: Lobo RA, Kelsey J, R. M, editors. Menopause: Biology and Pathobiology. San Diego: Academic Press; 2000. p. 175–88. [Google Scholar]
- 9.Thurston RC, Chang Y, Barinas-Mitchell E, Jennings JR, Landsittel DP, Santoro N, et al. Menopausal hot flashes and carotid intima media thickness among midlife women. Stroke. 2016;47(12):2910–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Thurston RC, Wu M, Chang YF, Aizenstein HJ, Derby CA, Barinas-Mitchell EA, Maki P. Menopausal Vasomotor Symptoms and White Matter Hyperintensities in Midlife Women. Neurology. 2023;100(2):e133–e41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Thurston RC, Chang Y, Derby CA, Bromberger JT, Harlow SD, Janssen I, Matthews KA. Abuse and subclinical cardiovascular disease among midlife women: the study of women's health across the nation. Stroke. 2014;45(8):2246–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stein JH, Korcarz CE, Hurst RT, Lonn E, Kendall CB, Mohler ER, et al. Use of carotid ultrasound to identify subclinical vascular disease and evaluate cardiovascular disease risk: a consensus statement from the American Society of Echocardiography Carotid Intima-Media Thickness Task Force. Endorsed by the Society for Vascular Medicine. J Am Soc Echocardiogr. 2008;21(2):93–111; quiz 89-90. [DOI] [PubMed] [Google Scholar]
- 13.Iemolo F, Martiniuk A, Steinman DA, Spence JD. Sex differences in carotid plaque and stenosis. Stroke. 2004;35(2):477–81. [DOI] [PubMed] [Google Scholar]
- 14.Thurston RC, Barinas-Mitchell E, Jennings JR, Santoro N, von Känel R, Matthews KA. Childhood abuse and subclinical cardiovascular disease among midlife women: modifying role of sleep duration (abstract). Psychosom Med. 2016;78(3):A97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Thurston RC, Chang Y, von Kanel R, Barinas-Mitchell E, Jennings JR, Hall MH, et al. Sleep characteristics and carotid atherosclerosis among midlife women. Sleep. 2017;40(2). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Carson MY, Barinas-Mitchell E, Maki PM, Thurston RC. Childhood Maltreatment and Arterial Stiffness Among Midlife Women. J Am Heart Assoc. 2022;11(21):e026081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Thurston RC, Barinas-Mitchell E, von Kanel R, Chang Y, Koenen KC, Matthews KA. Trauma exposure and endothelial function among midlife women. Menopause. 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yeboah J, Crouse JR, Hsu FC, Burke GL, Herrington DM. Brachial flow-mediated dilation predicts incident cardiovascular events in older adults: the Cardiovascular Health Study. Circulation. 2007;115(18):2390–7. [DOI] [PubMed] [Google Scholar]
- 19.Thurston RC, Chang Y, Matthews KA, von Känel R, Koenen K. Association of sexual harassment and sexual assault with midlife women’s mental and physical health. JAMA Intern Med. 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Thurston RC, Jakubowski K, Chang Y, Koenen K, Maki PM, Barinas Mitchell E. Sexual Assault and Carotid Plaque Among Midlife Women. J Am Heart Assoc. 2021;10(5):e017629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Debette S, Markus HS. The clinical importance of white matter hyperintensities on brain magnetic resonance imaging: systematic review and meta-analysis. BMJ. 2010;341:c3666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Thurston RC, Jakubowski KP, Wu M, Aizenstein HJ, Chang Y, Derby CA, et al. Sexual assault and white matter hyperintensities among midlife women. Brain Imaging Behav. 2022;16(2):773–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Thurston RC, Chang Y, Matthews KA, Harlow S, El Khoudary SR, Janssen I, Derby C. Interpersonal Trauma and Risk of Incident Cardiovascular Disease Events Among Women. J Am Heart Assoc. 2022;11(7):e024724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Suglia SF, Koenen KC, Boynton-Jarrett R, Chan PS, Clark CJ, Danese A, et al. Childhood and Adolescent Adversity and Cardiometabolic Outcomes: A Scientific Statement From the American Heart Association. Circulation. 2018;137(5):e15–e28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Matthews KA, Chang YF, Thurston RC, Bromberger JT. Child abuse is related to inflammation in mid-life women: role of obesity. Brain Behav Immun. 2014;36:29–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nguyen JK, Thurston RC. Association of Childhood Trauma Exposure with Inflammatory Biomarkers Among Midlife Women. J Womens Health (Larchmt). 2020;29(12):1540–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hillebrand S, Gast KB, de Mutsert R, Swenne CA, Jukema JW, Middeldorp S, et al. Heart rate variability and first cardiovascular event in populations without known cardiovascular disease: meta-analysis and dose-response meta-regression. Europace. 2013;15(5):742–9. [DOI] [PubMed] [Google Scholar]
- 28.Berntson GG, Bigger JT Jr., Eckberg DL, Grossman P, Kaufmann PG, Malik M, et al. Heart rate variability: origins, methods, and interpretive caveats. Psychophysiology. 1997;34(6):623–48. [DOI] [PubMed] [Google Scholar]
- 29.Hillebrand S, Gast KB, de Mutsert R, Swenne CA, Jukema JW, Middeldorp S, et al. Heart rate variability and first cardiovascular event in populations without known cardiovascular disease: meta-analysis and dose-response meta-regression. EP Europace. 2013;15(5):742–9. [DOI] [PubMed] [Google Scholar]
- 30.Thurston RC, Carson MY, Koenen KC, Chang Y, Matthews KA, von Kanel R, Jennings JR. The relationship of trauma exposure to heart rate variability during wake and sleep in midlife women. Psychophysiology. 2019:e13514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Carson MY, Thurston RC. Vasomotor symptoms and their links to cardiovascular disease risk. Curr Opin Endocr Metab Res. 2023;30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Thurston RC, Bromberger J, Chang Y, Goldbacher E, Brown C, Cyranowski JM, Matthews KA. Childhood abuse or neglect is associated with increased vasomotor symptom reporting among midlife women. Menopause. 2008;15(1):16–22. [PMC free article] [PubMed] [Google Scholar]
- 33.Carson MY, Thurston RC. Childhood abuse and vasomotor symptoms among midlife women. Menopause. 2019;26(10):1093–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jakubowski KP, Maki P, Chang Y, Thurston RC. Trauma and sleep problems over midlife in women. Menopause. 2020;27(12):1466. [Google Scholar]
- 35.Thurston RC, Chang Y, Barinas-Mitchell E, von Känel R, Jennings JR, Santoro N, et al. Child Abuse and Neglect and Subclinical Cardiovascular Disease Among Midlife Women. Psychosom Med. 2017;79(4):441–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Thurston RC, Barinas-Mitchell E, von Känel R, Chang Y, Koenen KC, Matthews KA. Trauma exposure and endothelial function among midlife women. Menopause. 2018;25(4):368–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cyranowski JM, Schott LL, Kravitz HM, Brown C, Thurston RC, Joffe H, et al. Psychosocial features associated with lifetime comorbidity of major depression and anxiety disorders among a community sample of mid-life women: the SWAN mental health study. Depress Anxiety. 2012;29(12):1050–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Thurston RC, Jakubowski K, Chang Y, Wu M, Barinas Mitchell E, Aizenstein H, et al. Posttraumatic Stress Disorder Symptoms and Cardiovascular and Brain Health in Women. JAMA Netw Open. 2023;6(11):e2341388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lawn RB, Nishimi KM, Sumner JA, Chibnik LB, Roberts AL, Kubzansky LD, et al. Sexual Violence and Risk of Hypertension in Women in the Nurses' Health Study II: A 7-Year Prospective Analysis. Journal of the American Heart Association. 2022;11(5):e023015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nishimi K, Thurston RC, Chibnik LB, Roberts AL, Sumner JA, Lawn RB, et al. Posttraumatic stress disorder symptoms and timing of menopause and gynecological surgery in the Nurses' Health Study II. J Psychosom Res. 2022;159:110947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Gibson CJ, Huang AJ, McCaw B, Subak LL, Thom DH, Van Den Eeden SK. Associations of Intimate Partner Violence, Sexual Assault, and Posttraumatic Stress Disorder With Menopause Symptoms Among Midlife and Older Women. JAMA Intern Med. 2019;179(1):80–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Watkins LL, LoSavio ST, Calhoun P, Resick PA, Sherwood A, Coffman CJ, et al. Effect of cognitive processing therapy on markers of cardiovascular risk in posttraumatic stress disorder patients: A randomized clinical trial. J Psychosom Res. 2023;170:111351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Centers for Disease Control and Prevention OoRaR. Infographic: 6 Guiding Principles to a Trauma-Informed Approach 2020. [Available from: https://www.cdc.gov/orr/infographics/6_principles_trauma_info.htm].
- 44.Miller E, Beach SR, Thurston RC. Addressing intimate partner violence and abuse of older or vulnerable adults in the health care setting—beyond screening. JAMA Intern Med. 2018. [DOI] [PubMed] [Google Scholar]