Abstract
Background.
Little is known about how peripheral nerve injury affects human performance, behavior, and life. Hand use choices are important for rehabilitation after unilateral impairment, but rarely measured, and are not changed by the normal course of rehabilitation and daily life.
Objective.
To identify the relationship between hand use (L/R choices), motor performance, and patient-centered outcomes.
Methods.
Participants (n=48) with unilateral peripheral nerve injury were assessed for hand use via Block Building Task, Motor Activity Log, and Edinburgh Handedness Inventory; dexterity (separately for each hand) via Nine-Hole Peg Test, Jebsen Taylor Hand Function Test, and a precision drawing task; patient-centered outcomes via surveys of disability, activity participation, and health-related quality of life; and injury-related factors including injury cause and affected nerve. Factor Analysis of Mixed Data was used to explore relationships between these variables. The data were analyzed under two approaches: comparing dominant vs. non-dominant hand, or affected vs. unaffected hand.
Results.
The data were best explained by 5 dimensions. Good patient outcomes were associated with non-dominant hand performance, dominant hand performance (separately and secondarily to non-dominant hand performance), and preserved function and use of the affected hand; whereas poor patient outcomes were associated with preserved but unused function of the affected hand.
Conclusion.
After unilateral peripheral nerve injury, hand function, hand usage, and patient life arise from a complex interaction of many factors. To optimize rehabilitation after unilateral impairment, new rehabilitation methods are needed to promote performance and use with the non-dominant hand, as well as the injured hand.
Keywords: Peripheral nerve injury, lateralization, hand dominance, manual dexterity, quality of life
Introduction
Unilateral impairment is a common long-term consequence of stroke1–3 and peripheral nerve injury,4 but little is known about how human performance, behavior, and life change after unilateral impairment. Peripheral nerve injury provides a valuable model to characterize how the uninjured brain adapts to unilateral impairment, which may provide a “healthy brain baseline” to contextualize unilateral impairments after stroke. Moreover, peripheral nerve injuries are major source of chronic unilateral impairment: in the United States, approximately 55–66,000 patients per year suffer from long-term dissatisfaction with hand function, despite surgery and therapy.5–9 Nevertheless, few studies in any population have investigated how and whether unilateral impairments lead to changes in individual behavior and life.
Rehabilitation is the primary clinical tool for adapting patient behavior after impairment. Physiologically, patients with traumatic nerve injury often do not benefit from physical therapy,10, but success may be possible, since animal models have shown that exercise interventions can promote nerve and muscle regrowth11. Occupational therapy or related biopsychosocial interventions are recommended for rehabilitation after small fiber neuropathy12, and quality of life after peripheral nerve injury may depend on therapists’ ability to help patients engage in meaningful work and household roles4,13. However, little of this research has explored specific consequences of unilateral impairment, because many clinical assessments were not designed to consider which hand was impaired.
Most patient assessments are not designed to identify the functional consequences of lateralized injury because they omit or minimize assessment of actions that depend strongly on hand dominance. Many patient-reported outcome assessments focus on activities that do not require the dominant hand14 (e.g. DASH,15 QuickDASH16), and many physical assessments of upper limb function focus on hand actions with only modest left-right asymmetry17 (e.g. Fugl-Myer,18 Box & Blocks19) or provide insufficient detail for quantitative analysis (e.g. Action Research Arm Test20). To understand how unilateral impairment impacts patient lives, researchers and clinicians must identify – and deploy –assessments that are associated with patient behavior such as hand use.
Individual behavior after unilateral impairment can be quantified by measuring hand use: left/right choices over time in unconstrained situations.21,22 Hand choice and performance have overlapping neural mechanisms: in healthy right-handed adults, hand use is supported by interhemispheric parietal competition between left intraparietal sulcus (IPS) and right posterior parietal cortex (PPC);23 similarly, performance with the left non-dominant hand may involve interhemispheric connectivity between left IPS and right PPC.24 However, the functional relationship between hand use and performance remains unknown.
Here, the purpose of this study was to identify relationships between unilateral performance (and its inverse, impairment), hand use, and patient-centered factors (e.g. quality of life, participation) by exploring a dataset of 48 individuals with unilateral upper extremity peripheral nerve injury. We used factor analysis of mixed data (FAMD) to identify factors that co-varied with hand use and patient-centered variables, so that future studies can identify causal relationships between performance, hand use, and patient-centered variables after unilateral impairment.
Methods
Study Overview and Participants
This was a cross-sectional single-arm study involving a single laboratory visit. All participants gave informed consent, and all procedures were approved by the local Institutional Review Board. Data were stored and managed via the Research Electronic Data Capture system. Hypothesis-driven results from the current dataset have been previously published.21
A total of 48 individuals participated in the study; for recruitment flowchart, see21. Participant demographic and historical details are listed in Table 1.
Table 1:
Demographic & Historical Data
| Mean | SD | ||
|---|---|---|---|
| Age | 44.42 | 15.55 | |
| Sex (Female) | 28 (58%) | ||
| Race | Native Amer. | 3 (6%) | |
| White | 37 (77%) | ||
| Black/African American | 9 (19%) | ||
| Asian American/Pacific | 0 (0%) | ||
| Other | 2 (4%) | ||
| Education | < 9 years | 2 (4%) | |
| Some high school | 10 (21%) | ||
| High school or equivalent | 16 (44%) | ||
| Some college | 14 (29%) | ||
| College + | 6 (13%) | ||
| Dominant Hand = Right | 41 (85%) | ||
| Affected Hand = Dominant | 22 (46%) | ||
| Classification | Neurapraxia | 8 (17%) | |
| Axonotmesis | 18 (38%) | ||
| Neurotmesis | 22 (46%) | ||
| # Surgeries (for this injury) | 1.06 | 0.81 | |
| Months since last surgery | 5.56 | 8.79 | |
| Months since injury | 18.56 | 26.60 | |
| Affected Nerve | Ulnar | 27 (56%) | |
| Median | 28 (58%) | ||
| Radial | 22 (46%) | ||
| Cutaneous | 4 (8%) | ||
| Other | 8 (17%) | ||
| Injury Location | Brachial Plexus | 16 (44%) | |
| Upper Arm | 5 (10%) | ||
| Elbow | 7 (15%) | ||
| Forearm | 11 (23%) | ||
| Wrist | 17 (35%) | ||
| Hand | 5 (10%) | ||
| Injury Cause | Trauma | 29 (60%) | |
| Surgical Complication | 9 (19%) | ||
| Chronic Compression | 7 (15%) | ||
| Other | 3 (6%) | ||
| Nerve transfer surgery = Yes | 19 (40%) | ||
Means and SD for numerical data, counts and percentages for categorical data. No participants identified as Hispanic or Latino.
Inclusion criteria included: age of 18 years or older, English-speaking, unilateral upper extremity PNI (defined as mechanical origin rather than pathologic or tumor), and QuickDASH16 score ≥18, measured at the start of the study session. This threshold was chosen to select individuals whose life is affected by their impairment, at 1 minimum clinically important difference25 above 0. This threshold captures a wide range of patients with PNI because it also lies 1 SD below the mean of patients with upper extremity disorder.26
Exclusion criteria included cognitive disorders, uncorrected visual impairment, chronic pain diagnoses (unrelated to PNI), major mental health diagnosis (not including depression, anxiety, bipolar, or post-traumatic stress disorder), surgery within preceding 2 months, or motor function diagnosis that has affected the arm contralateral to their PNI in preceding 2 years. To examine effects of injury severity, injuries of all types and severity levels were recruited within the above criteria.
Data Collection
Assessments are summarized in Table 2 and detailed here. Note that *−1 transformations were used where needed to change the valence of specific measures, making positive scores indicative of better performance across all measures.
Table 2.
The 27 analyzed factors (31 variables, due to: § = separate variables for each hand). Dex = dexterity. Three participants had other injury causes (1 tumor, 1 infection, 1 unknown). All variables have been rescaled so positive values represent better outcomes.
| Type | Abbreviation | Name |
|---|---|---|
| Performance | Dex-Finger§ | Nine-Hole Peg Test (9HPT) |
| Dex-Hand§ | Jebsen-Taylor Hand Function Test (JHFT) | |
| Draw-Smooth§ | Precision Drawing Task, velocity smoothness | |
| Draw-SAT§ | Precision Drawing Task, speed/error rate tradeoff (speed * (100%-error%)) | |
| Hand Use | Lego | Block Building Task (high value = use of DH or AH; low = NH or UH) |
| Brick | Brick Stacking Task (high value = use of DH or AH; low = NH or UH) | |
| Edin-Preserve | Edinburgh Handedness (−1 * absolute value of change pre/post injury) | |
| MAL-Amount | Motor Activity Log, amount scale (use of affected hand) | |
| Patient-centered | ACS-Retained | Activity Card Sort, retained activity level (%) |
| ACS-Current | Activity Card Sort, current activity level (# activities) | |
| Satisfaction | Activity Card Sort, satisfaction with level of participation in Top 5 favorite activities | |
| Ability | QuickDASH disability * −1 | |
| HRQoL | PROMIS-29, health-related quality of life | |
| Injury-elated | MonthsSince | Months since injury |
| PainLow | Recent injury related pain (0–10) * −1 | |
| Transfer | Did patient have nerve injury surgery (y/n) | |
| Classification | Nerve injury severity (Seddon classification, high = less severe) | |
| DomInjured | Was the dominant hand the injured hand? (y/n) | |
| Inj-Trauma | Injury cause: trauma (y/n) | |
| Inj-Surgery | Injury cause: surgical complication (y/n) | |
| Inj-Chronic | Injury cause: chronic compression (y/n) | |
| Loc-Plexus | Lesion location: brachial plexus or thoracic outlet (y/n) | |
| Loc-UpperArm | Lesion location: upper arm (y/n) | |
| Loc-Elbow | Lesion location: elbow (y/n) | |
| Loc-Forearm | Lesion location: forearm (y/n) | |
| Loc-Wrist | Lesion location: wrist (y/n) | |
| Loc-Hand | Lesion location: hand (y/n) |
Assessments: performance
Three assessments of hand performance were used, with a total of eight performance variables. A fourth assessment was attempted and discarded, as described below. For all performance assessments, outcome measures were unilateral measures calculated for each hand.
The Nine-Hole Peg test (9HPT)27–29 is an assessment from the NIH Toolbox30 to measure finger dexterity related to pinching movements.31 In brief, participants move 9 identical pegs from a bowl into holes, and then return the pegs to the bowl, moving one peg at a time. The outcome measure was completion time (ceiling minus actual, in seconds).
The Jebsen-Taylor Hand Function Test (JHFT)32 is a standard clinical test of hand function. Four subtests were used, based on factor analysis indicating that only these four measure unique constructs.31,33 Participants completed each subtest with each hand, then moved on to the next subtest. Two experimenters recorded the time for each subtest, and performance was videotaped. The outcome measure was average time across subtests (ceiling minus actual, in seconds).
The Precision Drawing Task is a preexisting assessment of pen control34,35 designed to capture the sensorimotor demands of handwriting. In brief, participants use a pen stylus to draw a line through hollow shapes, as shown in Figure 1. Participants are instructed to avoid making errors (going outside the shape boundaries), and secondarily to move as quickly as possible. Participants draw directly on a Cintiq 13HD Touch digitizing tablet (Wacom Co., Otome Japan), which captures endpoint position at 30 Hz. Participants performed 24 trials, reflecting 12 shapes at each of two widths (4 mm or 5 mm tolerance). The outcome measures were movement smoothness (−1 * number of velocity peaks per 45 mm segment), and speed-accuracy tradeoff (speed in cm/sec, divided by # errors per segment).
Figure 1:
Precision Drawing Task. Five sample stimuli shown, out of 45 possible.
Two-Point Orientation Discrimination36 is a test of tactile sensory acuity on the fingertip, designed as a variant of the standard Moving Two-Point Discrimination.37,38 A 2-up 1-down staircase procedure was used to identify the tactile sensory threshold on the index and small fingertips of each hand, to 0.5 mm precision. However, our Two-Point Orientation Discrimination results were unreliable, as described in Results. To avoid introducing noise into our FAMD, the Two-Point Orientation Discrimination results were omitted from our main analyses.
Assessments: hand use
Four assessments of hand usage were used, with one outcome measure each. For all four, the outcome was a single variable reflecting relative use of one hand versus the other.
The Block Building Task21,22,39 directly measured hand use during precision reach-to-grasp movements in an unconstrained environment. For details, see21, but in brief: Participants sat in front of a table with 40 Lego blocks (The Lego Group, Billund, Denmark): 4 copies each of 10 block types, in standardized locations. Participants were presented with a “model,” an abstract construction containing 10 blocks (one of each type). Participants were instructed to build the model as quickly and accurately as possible; participants received no cues about how to use their hands. Performance was video-recorded, and each video was reviewed by two raters using BORIS event-logging software40 to assess the fraction of grasps made with either hand. The outcome measure was the number of grasps made with a given hand.
The Brick Stacking Task was created as a novel variant of the Block Building Task, to measure hand use during reach-to-grasp of large heavy objects. The procedure was identical, except the table contained 20 painted half-bricks (each approximately 9 × 8 × 4 cm, weight 750 g), comprising 4 instances each of 5 colors. The four “target objects” differed from each other in the pattern of brick colors only. The primary outcome was the number of grasps made with a given hand.
The Edinburgh Handedness Inventory41 was used to confirm self-reported hand choices, and changes therein. The primary outcome was “preserved hand use”, −1 * the absolute change in score between pre-injury and current.
The Motor Activity Log (MAL)42 is a standard structured interview for hand usage at home, designed for stroke patients. Unlike our other measures of hand usage, the MAL measures usage of the affected hand alone, rather than as part of a between-hands relationship. The outcome measure was the participant’s average report of how much they used their affected upper extremity for each of 30 standard activities (on a scale from “not at all” to “same as pre-injury”).
Assessments: patient-centered variables
Five assessments were used to measure patient-centered factors, with a total of six outcome variables.
Disability (here, “ability,” because converted to positive scores = better) was assessed via the QuickDASH questionnaire.16 The QuickDASH was chosen instead of the full DASH because our recruitment sources collected QuickDASH measurements for all potential participants. The primary outcome is ability score (0–100).
The Activity Card Sort (ACS)43–45 questionnaire was used to assess participants’ ability to participate in 87 meaningful activities across four categories (22 leisure, 19 domestic, 24 active/fitness, and 22 social, plus the opportunity to fill in extra activities). The ACS has previously been used to identify changes in activity participation in individuals with peripheral nerve injury.46 Here, the ACS was implemented in the form of a checklist.45 For each activity, participants checked one of two options for “prior to injury” (either “never did this” or “I did it before my injury”); if they ever participated in the activity, they also selected one of three options for “current activity” (“I do it now,” “I do it less than before,” or “I no longer do it”). For quantitative measurements, “less than before” was counted as 0.5 activities. The primary outcomes were “retained activity level,”44 calculated as (# now / # before); and current activity level (“ACS Current”), measured as # activities currently performed.
The ACS also had a second part (“Top 5”), in which participants provided detailed answers on their five favorite activities (not necessarily from the list in the first part of the ACS). For each of these five activities, participants completed three visual analog scales: “level of participation before injury,” “current level of participation,” and “satisfaction with current performance.” All values were represented as a 0–5 scale. If a participant did not provide a pre-injury participation amount, the ceiling value of 5 was used. Two outcome measures were calculated from the second part of the ACS: “Top 5 Amount” (level of participation: current / before injury) and “Top 5 Satisfaction” (taken directly from “satisfaction with current performance” scale). These two variables were highly correlated (r = 0.88), so only “Satisfaction” was used for analysis.
The Patient Reported Outcome Measurement Information System 29 (PROMIS-29) questionnaire47 was used to assess health outcomes and health-related quality of life. This standard survey measures physical functioning, anxiety, depression, fatigue, sleep disturbance, social functioning, and pain. The outcome measure was health-related quality of life (HRQOL) measured as PROMIS-29 total score (0–100).
Assessments: Injury
Fourteen injury-related variables were also analyzed: months since injury, pain, surgery type (transfer y/n), injury classification, dominant hand injury (y/n), plus three variables for injury cause and six for lesion location. Pain was rated as “recent injury-related pain” (0–10), multiplied by −1 to produce a “PainLow” measure. One participant did not report their pain, so their value was imputed as equal to the pain question on the PROMIS-29 survey. Other time-related variables (e.g. time since surgery, patient age) were omitted from analysis due to covariance with months since surgery. Injury classification was an ordinal variable with three levels (neuropraxia, axonotmesis, neurotmesis). For injury cause, the three variables (all categorical y/n) were: trauma, surgical complication, and chronic compression; three participants had other injury causes (1 tumor, 1 infection, 1 unknown), but “other” injury type was omitted from the model to serve as a baseline. Lesion location was modeled as six separate y/n categorical variables (brachial plexus/thoracic outlet, upper arm, elbow, forearm, wrist, hand) because 50% of participants had injuries across multiple locations.
Data Analysis
All analysis were conducted with R 4.1.248,49 with different sets of data (“approaches”). First, the data were analuzed according to hand dominance: this “DH/NH” approach used separate scores for all variables for the dominant (DH) and non-dominant hand (NH). Second, the data were analyzed according to which hand was impaired: this “AH/UH” approach used separate scores for all variables for the affected hand (AH) and the unaffected hand (UH). In both sets of data, identical analyses were performed.
First, a factor analysis with mixed data (FAMD)50,51 was conducted because our variable set included both continuous (e.g., speed and accuracy) and categorical factors (e.g., lesion location and injury cause). Conceptually, FAMD is a dimension reduction technique that decomposes the multidimensional datasets into the smallest number of underlying (and orthogonal) dimensions that explain the most variance. FAMD combines a principal components analysis of the continuous variables with multiple correspondence analysis for the categorical variables, and each variable can then be assigned a % contribution to underlying dimensions. Continuous data were normalized using a z-transformation and categorical variables were transformed into a series of binomial variables capturing all possible levels of a categorical factor prior to the FAMD. This scaling ensure a balanced influence of both continuous and categorical variables when expressed as a % contribution to each underlying dimension. To determine the number of underlying dimensions to interpret in each analysis (out of 31 possible dimensions), the constraint was imposed that informative dimensions should explain about twice as much variance as any one variable. Therefore, a threshold was imposed that a dimension must have an eigenvalue ≥2.0, equivalent to explaining ≥6.5% of the data to be retained (2*(1/31)=0.065). This led to 5 dimensions being retained as the most informative low-dimensional representation of the data in both the D/ND and A/UA approaches.
Within each dimension, variables were reported if their contribution to the dimension exceeded 3.22%, which is the expected contribution of a random variable (1/31, for 31 variables).
Note that because the final FAMD solution expresses the loading of each variable as a % of the variance explained in each dimension, these values are unsigned. To determine the direction of the relationships between variables, we also extracted the signed loadings from the principal components analysis of the continuous variables and the multiple correspondence analysis for the categorical variables, separately.
Finally, cluster analyses were conducted to explore possible subgroups of participants in both the DH/NH and AH/UH variable spaces. K-means cluster analysis was used to iteratively test different numbers of clusters (1 to 10) and evaluate the usefulness of these clusters with three different statistics the variance explained by cluster membership, the residual variance within each cluster, and the gap statistic.52
Results
Dominant vs. non-dominant hand: five dimensions
FAMD analysis of DH/NH data revealed five dimensions with eigenvalues above 2.0, with possible elbows (change in slope) after dimensions 2, 3, or 5, as shown in Figure 2A. Each of these dimensions accounted for >8.1% of variance, and together accounted for 56% of total variance. In the following section we detail all five dimensions.
Figure 2:
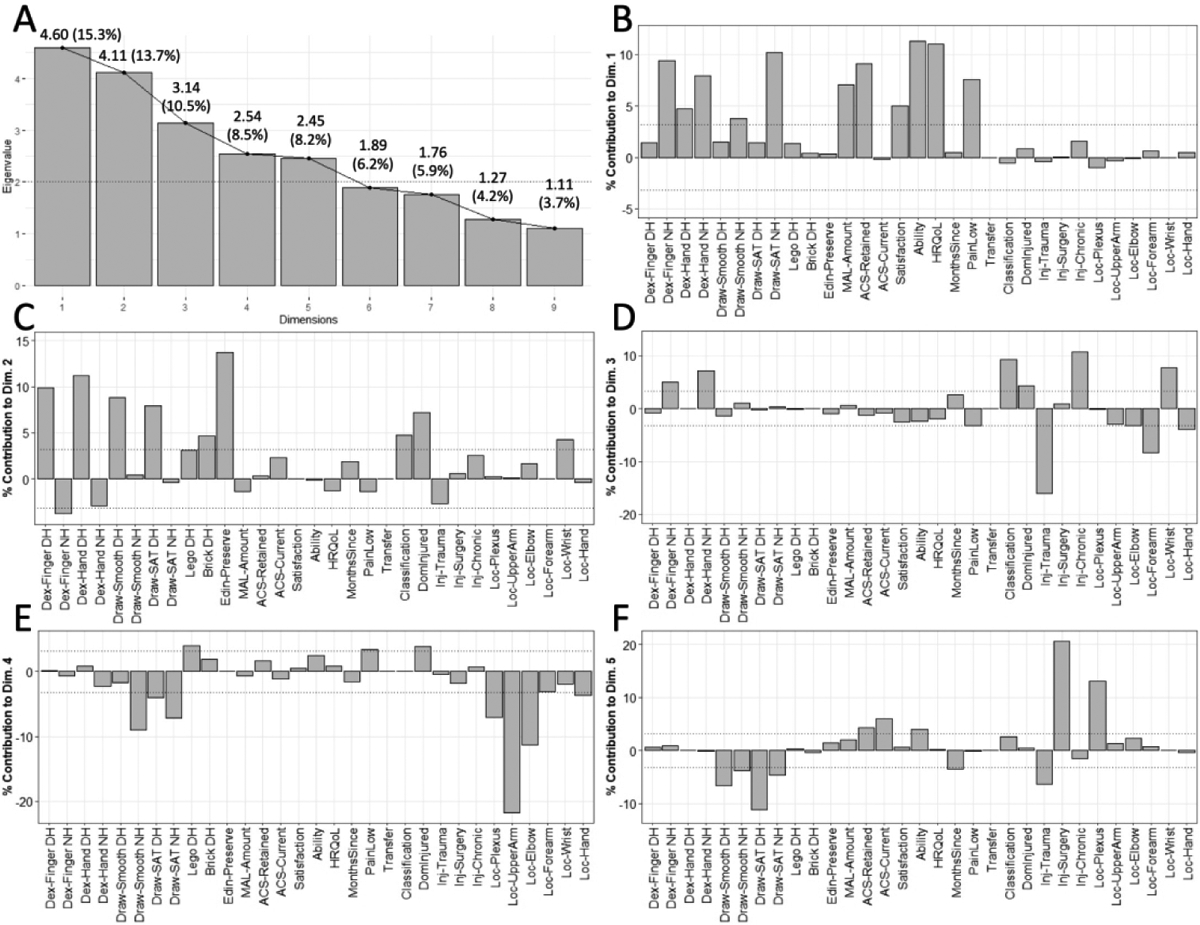
Factor Analysis of Mixed Data results from DH/NH approach. A: Scree plot showing all dimensions with eigenvalues ≥ 1.0 (9/30 dimensions). Numbers are: eigenvalue (% variance explained). B-F: factor loadings for the five dimensions with eigenvalue ≥ 2.0.
Dominant vs. non-dominant hand dimensions: preserved dominant hand, non-dominant advantage, using the uninjured dominant hand, and spared activities.
The first dimension (DN1; Figure 2B) was associated with NH performance variables: Draw-SAT NH+, Dex-Finger NH+, Dex-Hand bilaterally (NH+ > DH+), and Draw-Smooth NH+. This component had a weak contribution of hand usage, wherein only MAL-Amount+ (continued use of injured hand) contributed, and no other hand usage variables contributed. Three of the five patient-centered variables contributed to this dimension: Ability+, HRQoL+, and Satisfaction+. Among injury-related variables, only PainLow+ contributed. We interpret this dimension as “NH motor performance, associated with good patient outcomes.”
The second dimension (DN2) was associated with DH function (Figure 2C). Performance variables included Dex-Hand DH+, Dex-Finger DH+, Dex-Finger NH−, and Draw-SAT DH+. Paralleling this association with DH performance, this dimension was associated with DH usage: Brick DH+ and Edinburgh Preserve+ contributed. This component was not associated with patient-centered variables, but it was associated with two injury variables: a Dominant Injury−, Classification+, and Location (wrist+). We interpret this dimension as “preserved DH after NH injury, unrelated to patient outcomes.”
The third dimension (DN3; Figure 2D) was associated primarily with injury-related variables. Performance variables included only Dex-Hand NH+ and Dex-Finger NH+, while no hand usage or patient-centered variables contributed to the dimension. For injury-related variables, this dimension included Injury Type (trauma− > chronic+), Classification+, Location (forearm− > wrist+ > hand− > elbow−), and PainLow−. We interpret this dimension as “NH advantage after DH injury, unrelated to patient outcomes.”
The fourth dimension (DN4; Figure 2E) was driven primarily by injury location variables. The performance variables were Draw-Smooth NH+ and Draw-SAT bilateral (NH− > DH−). The only hand usage variable was Lego DH+. No patient-centered variables contributed. The largest contribution came from injury-related variables, especially Location (upper arm− >> elbow− > plexus− > hand−), Dominant Injury−, and PainLow+. We interpret this dimension as “use of an uninjured DH in people with a low-performance NH.”
The fifth and final dimension (DN5; Figure 2F) was associated mostly with injury and performance characteristics. Contributing performance variables included Draw-SAT bilateral (DH− > NH−) and Draw-Smooth bilateral (DH− > NH−). No hand usage variables contributed. Patient-centered variables included ACS-Current+, ACS-Retained+, and Ability+. Injury-related variables included Injury Type (surgery+ >> trauma−), Location (Plexus+), and Months Since−. We interpret this component as “preserved activities despite reduced performance after proximal injury.”
Full dimension contribution values are reported in Supplementary Table 1. Performance and injury variables contributed to all five dimensions, but hand usage variables contributed to only three (DN1, DN2, DN4) and patient-centered variables to only two (DN1, DN5). Notably, the variable “Dominant Injury” was only included in the dimensions without any patient-centered factors. Overall, these five dimensions suggest that patients achieve better outcomes with better motor performance (especially in the NH), separable from any characteristics of the injury.
Affected vs. unaffected hand: five dimensions
FAMD analysis of AH/UH data revealed five dimensions with eigenvalues above 2.0, with possible elbows (change in slope) after components 1, 2, or 4, as shown in Figure 3A. Each of these dimensions accounted for >7.5% of variance, and together accounted for 54% of total variance. In the following section we detail all five dimensions.
Figure 3:
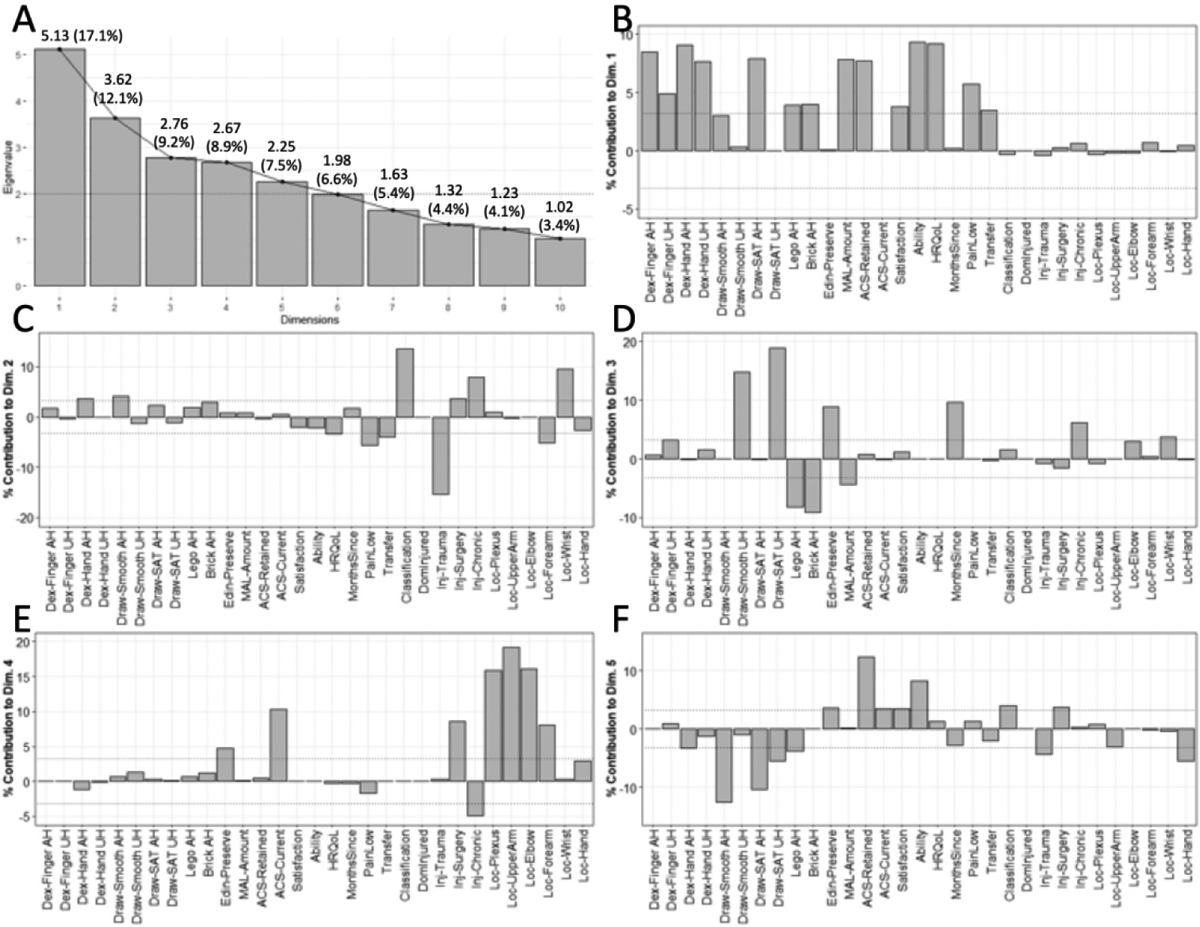
Factor Analysis of Mixed Data results from AH/UH approach. A: Scree plot showing all dimensions with eigenvalues ≥ 1.0 (10/30 dimensions). Numbers are: eigenvalue (% variance explained). B-F: factor loadings for the five dimensions with eigenvalue ≥ 2.0.
Affected vs. unaffected hand: components for preserved function, unaffected hand performance, preserved life, and activity-specific use of the affected hand.
The first dimension (“AU1”) was associated with patient-relevant outcomes and performance with the affected hand (Figure 3B). Contributing performance variables included Dex-Hand bilateral (AH+ > UH+), Dex-Finger bilateral (AH+ > UH+), and Draw-SAT AH+. Contributing hand usage variables included MAL+, Brick AH+, and Lego AH+, their uniform direction indicated preserved use of the AH. Many patient-centered variables also contributed to this dimension: Ability+, HRQoL+, ACS-Retained+, and Satisfaction+. For injury-related variables, only PainLow+ and Transfer Surgery+ contributed. We interpret this component as “preserved function and use of the AH, associated with good patient outcomes.”
The second dimension (“AU2”) was associated primarily with injury characteristics (Figure 3C). The only contributing performance variables were Draw-Smooth AH+ and Dex-Hand AH+. No hand usage variables contributed to this dimension. Among life-relevant variables, only HRQoL– contributed. Many injury-related variables contributed, primarily Injury Type (trauma− > chronic+ > surgery+), Classification−, Location (Wrist+ > Forearm−), PainLow−, and Transfer Surgery−. We interpret this dimension as “preserved function of the AH, but not use of the AH, is accompanied by low quality of life.”
The third AH/UH dimension (“AU3”) was dominated by UH function (Figure 3D). Contributing performance variables include Draw-SAT UH+, Draw-Smooth UH+, and Dex-Finger UH+. All four hand usage variables contributed: Brick AH−, Edinburgh Preserve+, Lego AH−, and MAL−; their directions indicates high use of the AH, especially when it’s the DH. No patient-centered variables contributed. Among injury-related characteristics, contributors included MonthSince+, Injury Type (chronic+), and Location (wrist+). We interpret this dimension as “function and use of the UH, unrelated to patient outcomes.”
The fourth dimension (“AU4”) was associated primarily with injury location (Figure 3E). No performance or variables contributed to this dimension; among hand usage variables, the only contributor was Edinburgh Preserve+. Among patient-centered variables, only ACS-Current+ contributed. Most contributors were injury-specific variables: Location (upper arm+ > elbow+ > plexus+ > forearm−) and Injury Type (surgery+ > chronic−). We interpret this dimension as “engagement in activities despite proximal injury, regardless of function.”
The fifth and final dimension (“AU5”) was associated with numerous variables, as shown in Figure 3F. Contributing performance variables included Draw-Smooth AH−, Draw-SAT bilateral (AH− > UH−), and Dex-Hand AH−. Contributing hand usage variables included Lego AH− and Edinburgh Preserve+ (together suggesting use of the UH, especially when it’s the DH). Contributing patient-centered variables included Ability+, Satisfaction+, and ACS-Current+. Among injury-related factors, contributing variables were Location (hand−), Injury Type (trauma− > surgery+), and Classification+. We interpret this dimension as “good patient outcomes despite reduced AH performance and use.”
Full dimension contribution values are reported in Supplementary Table 2. Injury variables contributed to all five dimensions, while the other categories contributed to four dimensions each: performance variables (AU1, 2, 3, 5), hand usage variables (AU1, 3, 4, 5) and patient-centered variables (AU1, 2, 4, 5). Overall, these components suggest that patients achieve better outcomes when they have preserved function with their AH.
Correlation analyses
To provide a simple illustration of the linear relationships between variables (i.e. raw data), we show correlograms (Spearman’s rho) for both approaches in Figure 4. For visual clarity we reordered the variables to show positive correlations between nearby variables.
Figure 4:
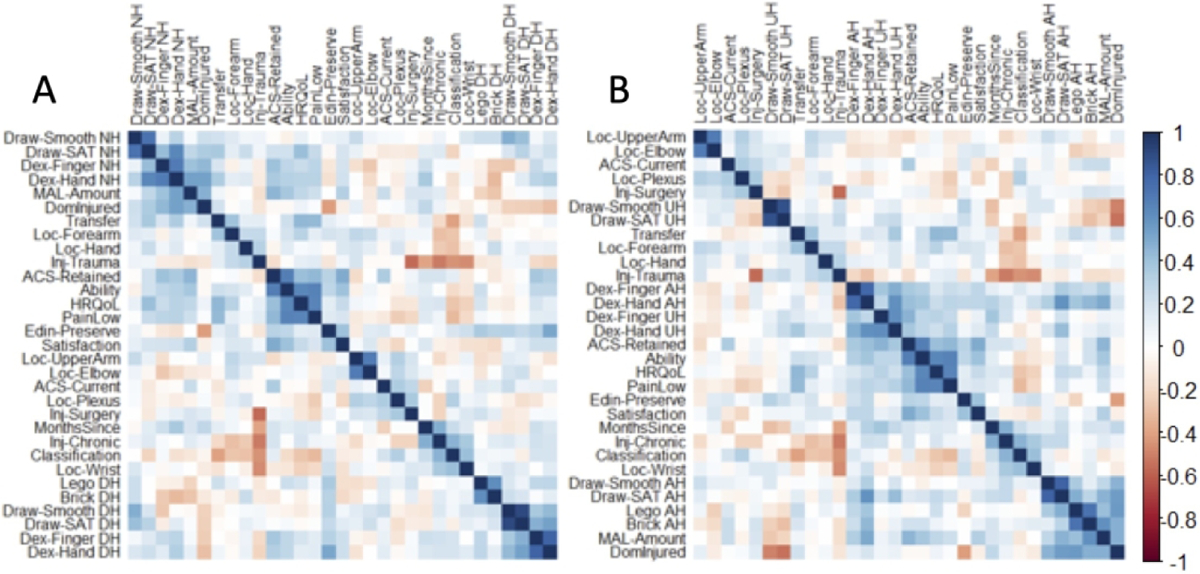
Correlograms of linear relationships between all variables. For visual clarity, variables are ordered to show positive correlations between nearby variables. A: DH/NH approach. B: AH/UH approach.
Cluster analyses
Although the FAMD tells us about the underlying dimensions in both variable spaces, it does not tell us about how participants cluster together in those spaces. For example, intuitively one might expect separate clusters (i.e. subgroups) for people whose AH was the DH, vs. people whose AH was their NH. However, our k-means cluster analysis revealed no good evidence for clusters.
As measured by the gap statistic, the optimal number of clusters was 1 under either the DH/NH or AH/UH approach. In addition, neither explained variance nor residual variance offered a clear break point (or “elbow”) that would indicate a reliable number of clusters, as shown in Supplementary Figure 1. Therefore, any clusters in our data were too subtle to reliably detect with the current sample size.
Tactile sensation measures were not reliable
We assessed tactile sensory thresholds in the fingertips via two-point orientation discrimination. We were able to collect data from the injured hand in 42 participants. We defined “sensation loss” as (threshold of worst finger on affected hand) – (threshold of same finger on contralateral hand). We found negative sensation loss (i.e. better sensation in the injured hand than the uninjured hand) in 14/42 participants (33%). Therefore, we considered the data unreliable, and excluded them from analyses.
Potentially, this finding could arise from confounds in our sample’s diverse nerve injuries (e.g. if different patient types had different changes in sensation). To test this hypothesis, we performed a general linear regression to determine whether sensation (threshold in the most-affected finger) depended on any injury characteristics (injury location, lesion classification, time since injury, surgery type). This did not produce a statistically significant model (adjusted r2 = 0.02, p = 0.42), and no individual factor had a significant impact on sensation (p > 0.05). Therefore, evidence does not support the idea that our tactile sensation results are an artifact of the diversity in our patient sample.
Discussion
After unilateral peripheral nerve injury, hand function, hand usage, and patient life arise from a complex interaction of many factors. We found that good patient outcomes were associated with non-dominant hand performance (dimension DN1), and secondarily and separately with dominant hand performance (DN5). Good patient outcomes were also associated with preserved function and use in the affected hand (AU1), but preserved function without use led to poor patient outcomes (AU2), and some patients could achieve good outcomes despite impaired performance in their affected hand (AU5).
Whether we analyzed our data through the approach of an dominant/non-dominant hand distinction (DH/NH) or an affected/unaffected hand distinction (AH/UH), we found five components that met our threshold of eigenvalue ≥ 2.0 (i.e. twice as useful as a single variable); those five together accounted for only 54–55% of our data, which suggests that these five components tell the majority of the story, but much still remains unknown about the underlying factors that support patient-centered outcomes after unilateral peripheral nerve injury.
Dominant vs. Non-Dominant Hand: patient outcomes associated with non-dominant hand function
When we analyzed our data through the approach of an DH/NH distinction, the biggest component (DN1, 15.3% of variance) comprised high NH performance and good outcomes in multiple patient-centered variables. This suggests that the ability to use either hand effectively leads to better outcomes after injury, even though this factor showed little influence of actual hand choices. (It was associated with self-reported use of the injured hand via the Motor Activity Log, but not with actual hand use in the Lego or Brick tasks.)
The only other component involving patient-centered variables was component DN5 (8.2% of variance), which comprised low performance (bilateral but especially DH) and good outcomes in multiple patient-centered variables, especially under certain kinds of PNI – particularly, brachial plexus injuries caused by surgical complication. This suggests that, in some patients, physical impairment is not necessarily an obstacle to continued participation in life activities, and a life without disability.
In other components, DH function was associated with preserved DH use (DN2, 13.7%), non-traumatic injuries were associated with NH function (DN3, 10.5%), and patients used their uninjured DH more when they had an injured, low-performance NH (DN4, 8.5%). However, none of those components involved a significant contribution from patient-relevant outcomes, which is particularly notable for DN2, which had strong contributions from DH performance variables but no evidence that these were related to patient outcomes.
Interestingly, the variable “dominant hand injury (y/n)” was never associated with patient outcomes; it contributed only to DN2, DN3, and DN4. This may be because many current measures of patient outcome and performance are insensitive to hand dominance,14 as detailed further in a separate section below (“Assessment specificity for unilateral impairment”). Future studies should develop and validate patient-centered assessments (e.g. of quality of life or activity participation) that are sensitive to how lateralization and handedness impact a subset of human activities.
Together, these findings suggest NH function can be an important factor in patient outcomes after PNI, but some patients are able to achieve good outcomes despite poor bilateral function.
Affected vs. Unaffected Hand: patient outcomes associated with many factors, including preserved function and use of the affected hand
When we analyzed our data through the approach of an AH/UH distinction, the biggest component (AU1, 17.1% of variance) comprised high AH performance and use, along with good outcomes in multiple patient-centered variables. This suggests, intuitively, that better outcomes after PNI are associated with broad preservation of the AH.
In the AH/UH distinction, more components were associated with patient-centered outcomes. Component AU2 (12.1%) comprised high AH performance but low health-related quality of life (HRQoL), particularly in patients with non-traumatic injuries and less severe Seddon classification, for reasons unclear. Component AU4 (8.9%) comprised high current participation in activities, and preserved use of the dominant hand, particularly in patients with proximal injuries from surgical complications. Both of these patient-centered outcome measures (HRQoL and current participation) also appeared in other components, suggesting that these two components reflect additional/independent ways to drive patient outcomes, rather than primary explanations for those patient outcome measures.
Here, only one component (AU3, 9.2%) was not associated with patient-centered outcomes. Specifically, UH performance and use showed no association with good patient outcomes, especially in patients with chronic injury (many months since surgery and/or a chronic compression injury). In other words, for long-term patients, it matters little whether or not they use their UH.
Finally, the fifth component (AU5, 7.5%) included a complex combination of all four variable types, which we interpret primarily as good patient outcomes despite AH impairment and disuse. This component was dominated by performance and patient-centered factors, but injury-related factors suggest an association with non-hand injuries, surgical complications over trauma, and severe Seddon classification. Ultimately, this component suggests that seriously impaired patients may nevertheless have a path to achieve good outcomes, independently of preserved AH function (AU1, AU2) or UH function (AU3).
Hand usage: the affected hand is associated with patient outcomes, but the dominant hand is not
Previous studies from our laboratory (using the same participants) have shown that, after unilateral PNI, patients continue to use their DH even when a DH injury has made it their less-dexterous hand,21 even after normal life and typical rehabilitation. Our current study also found continued use of the DH after its injury, in that the variable “dominant hand injured” was positively correlated with AH use, and negatively correlated with DH use.
Our current analysis provides three new insights into the value and possibility of hand usage after PNI. First, good outcomes are possible with use of the AH (AU1) or without (AU5), but patient-centered outcomes are more strongly associated with AH use than disuse (AU1 explains more data than AU5, and has larger contributions from patient-centered variables). Second, AH usage is associated with bilateral dexterity (AU1), which suggests a possible role for effector-nonspecific factors (e.g. patient confidence in motor performance, or a neural mechanism of hand choice that overlaps with motor execution networks). Third, increased use of the DH is not necessarily adaptive: high DH usage was not associated with any change in patient-centered outcomes (DN4). Therefore, patients are unlikely to be harmed by interventions to reduce their natural overuse of the unaffected DH. Together, our results suggest that AH usage should be promoted after unilateral PNI, possibly via both hand-specific and hand-nonspecific rehabilitation.
Measurement challenges in patients with peripheral nerve injury, and other limitations
Our exploratory analysis revealed a number of measurement concerns. First and foremost, while changes in tactile sensation are a hallmark symptom of peripheral nerve injury,53,54 and plays a major role in patient life,55,56 we were unable to collect a reliable measurement of tactile sensation in the injured hand. We used two-point orientation discrimination due to its numerous theoretical advantages,36 but administration proved unreliable in practice. This aligns with multiple reviews that found two-point discrimination to have low responsiveness57 and inconsistent results58 in patients with PNI. Future studies should explore existing methods of tactile assessment that provide precise, reliable measurement in these patients.57,58
Our methods did not include assessment of proprioception in the hand or upper limb. Sensory re-innervation after peripheral nerve injury, including re-innervation errors (cross-innervation of targets other than the original target), can play a role in restoration of function59. We hope that future studies can identify how proprioception fits into the other factors that drive patient outcomes after peripheral nerve injury.
Quantitative assessment of activity participation is an ongoing challenge in rehabilitation research.60–62 We used three measures from the Activity Card Sort questionnaire:43–45 current activities (#), retained activities (% of pre-injury) and satisfaction (subjective satisfaction with the individual’s top 5 activities). Of these, “current activities” did not load alongside with other patient-centered variables, suggesting that it may not matter to patients how many activities they engage in. This is a reasonable conclusion given that the ACS does not assess how much time a person spends in various activities: a person with 10 activities could be more active than a person with 20. Instead, what matters to patients is their change in activities since their injury, whether measured as a number of activities (Retained) or within their few favorite activities (Satisfaction). Future studies should identify which kinds of activities are most often affected by peripheral nerve injuries and the rehabilitation thereof.
In the current study, we performed tests in a consistent order, so that we would have the most important results if a participant withdrew due to exhaustion. However, it is possible that this introduced some between-tests biases, which is a limitation of our current study.
We found no evidence for clusters of participants, even though our sample included left-handed and right-handed people with injuries to either the DH or NH. We attempted to address this by including “dominant hand injured?” as a factor in the model, but we presume that larger samples might have allowed us to detect behavioral clustering of these groups. This illustrates the challenges of working with a condition that has complex, highly individual effects on patient performance, hand usage, and life.
Our FAMD analysis only addressed the variance-covariance structure of the data, not mean differences between hands. We did not explore mean differences because we believe they would not be informative in our current analysis structure; e.g. differences between DH and NH are expected and trivial. Mean differences could be scientifically interesting in a subgroup analysis (e.g. DH-affected vs. NH-affected), but our small sample size limited our ability to test hypotheses about subgroup differences. Future work should investigate how the key variables here relate to clinically meaningful phenotypes (e.g., using discriminant function analysis to predict dominant-affected versus non-dominant-affected individuals).
Most upper extremity peripheral nerve conditions are not isolated unilateral impairments such as the ones we studied here. For example, the prevalence of upper limb sensorimotor neuropathy is over 30% among individuals with diabetes,63 indicating a substantial population of peripheral nerve patients with major comorbidities and systemic (i.e. bilateral) underlying causes. Small fiber neuropathies, another common form of peripheral nerve disorder, are complex conditions that are difficult to diagnose64 or treat (pharmacologically or via rehabilitation).65 Our current findings may or may not extend to individuals with more complex peripheral nerve conditions, but we hope this study provides a framework to improve understanding of the lives of patients with unilateral upper extremity impairment, regardless of the underlying cause.
Assessment specificity for unilateral impairment
Our study found no association between patient outcomes and whether the dominant hand was the injured hand (as discussed above in “Dominant vs. Non-Dominant Hand”); given the intuition , which is likely a measurement artifact that arises because most measures of patient outcome and performance are insensitive to hand dominance.14 For example, the Disabilities of the Arm, Shoulder, and Hand (DASH) questionnaire15 asks about 21 activities, of which 2 depend on the dominant hand;14 the QuickDASH (used here to assess ability/disability) omits such activities entirely;16 and the ACS involves a list of activities of which 90% do not require fine motor skills.66 Many major physical assessments are also ill-designed for the purpose of unilateral assessment: the Fugl-Myer18 and Box & Blocks19 tests measure hand function via grasp, which has only modest left/right asymmetry;17 and the Action Research Arm Test20 provides 0–3 scores for each activity, which provides insufficient detail for quantitative analysis.
Some of the assessments in the current work avoid this problem. Quantitative unilateral measures of precision hand performance (9HPT, JHTF, precision drawing) allow separate assessment for each hand, and the Block Building and Brick Stacking tasks provide a quantitative comparison between hands. Future research should identify or develop occupational assessments that can assess altered participation in activities that require the dominant hand, and clinicians should consider assessments that can identify the distinct consequences of dominant hand impairment.
Conclusions
We sought to identify how hand usage choices and patient-centered outcomes interact with hand motor performance after unilateral peripheral nerve injury to the upper extremity. Patient outcomes were associated with many factors, the strongest of which being non-dominant hand performance, and preserved function and use in the affected hand. Therefore, to help patients with chronic impairment of the dominant hand, future research and rehabilitation should focus on facilitating patient non-dominant hand function, and use of the affected hand; and on developing lateralization-specific measures of patients’ participation in their life activities.
This exploratory study provides and explores a dataset with broad applications for future studies of unilateral impairment. Asymmetric impairment can arise from numerous conditions including stroke and traumatic brain injury, but unilateral peripheral nerve injury provides a unique opportunity in its ability to reveal a “healthy brain baseline” to contrast against changes following central neurological injury.
Supplementary Material
Acknowledgments
This work was funded by NINDS R01 NS114046 to BAP. We thank Claudia Gonzalez and Kayla Stone for their help implementing the Block Building Task; and Jackie Hardy, Rachel Graves, Sarah Sherman, and Zuying Gao for their help with data collection and coding.
Footnotes
Declaration of Conflicting Interests
Dr. Philip and Washington University in St. Louis have a licensing agreement with PlatformSTL to commercialize the precision drawing task used in this study.
References
- 1.Foerch C, Misselwitz B, Sitzer M, Berger K, Steinmetz H, Neumann-Haefelin T. Difference in recognition of right and left hemispheric stroke. The Lancet. 2005;366(9483):392–393. [DOI] [PubMed] [Google Scholar]
- 2.Mani S, Mutha PK, Przybyla A, Haaland KY, Good DC, Sainburg RL. Contralesional motor deficits after unilateral stroke reflect hemisphere-specific control mechanisms. Brain. 2013;136(4):1288–1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hedna VS, Bodhit AN, Ansari S, et al. Hemispheric differences in ischemic stroke: is left-hemisphere stroke more common? Journal of Clinical Neurology. 2013;9(2):97–102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stonner MM, Mackinnon SE, Kaskutas V. Predictors of Disability and Quality of Life After Nerve Injury. American Journal of Occupational Therapy. 2017;71(1):7101190050p1–7101190050p8. [DOI] [PubMed] [Google Scholar]
- 5.Brattain K Analysis of the Peripheral Nerve Repair Market in the United States. 2013.
- 6.Philip BA, Kaskutas V, Mackinnon SE. Handedness has a Narrow Impact on Disability After Unilateral Peripheral Nerve Disorder. presented at: American Society for Neurorehabilitation; 2017; Baltimore, MD. [Google Scholar]
- 7.Dyck PJ, Boes CJ, Mulder D, et al. History of standard scoring, notation, and summation of neuromuscular signs. A current survey and recommendation. Journal of the Peripheral Nervous System. 2005;10(2):158–173. [DOI] [PubMed] [Google Scholar]
- 8.Council MR. Aids to the investigation of the peripheral nervous system. Her Majesty’s Stationary Office; 1943. [Google Scholar]
- 9.He B, Zhu Z, Zhu Q, et al. Factors predicting sensory and motor recovery after the repair of upper limb peripheral nerve injuries. Research and Report. Neural Regeneration Research. 15th March 1, 2014. 2014;9(6):661–672. doi: 10.4103/1673-5374.130094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Adiguzel E, Yasar E, Tecer D, et al. Peripheral nerve injuries: Long term follow-up results of rehabilitation. J Back Musculoskelet Rehabil. Apr 27 2016;29(2):367–371. doi: 10.3233/BMR-160681 [DOI] [PubMed] [Google Scholar]
- 11.Chiaramonte R, Pavone V, Testa G, et al. The Role of Physical Exercise and Rehabilitative Implications in the Process of Nerve Repair in Peripheral Neuropathies: A Systematic Review. Diagnostics (Basel). Jan 18 2023;13(3)doi: 10.3390/diagnostics13030364 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Geerts M, Hoeijmakers JGJ, Gorissen-Brouwers CML, Faber CG, Merkies ISJ. Small Fiber Neuropathy: A Clinical and Practical Approach. The Journal for Nurse Practitioners. 2023;19(4)doi: 10.1016/j.nurpra.2023.104547 [DOI] [Google Scholar]
- 13.Stonner MM, Mackinnon SE, Kaskutas V. Predictors of functional outcome after peripheral nerve injury and compression. J Hand Ther. Jul–Sep 2021;34(3):369–375. doi: 10.1016/j.jht.2020.03.008 [DOI] [PubMed] [Google Scholar]
- 14.Philip BA, Kaskutas V, Mackinnon SE. Impact of handedness on disability after unilateral upper extremity peripheral nerve disorder. HAND. 2020;15(3):327–334. doi: 10.1177/1558944718810880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Beaton DE, Davis AM, Hudak P, McConnell S. The DASH (Disabilities of the Arm, Shoulder and Hand) outcome measure: what do we know about it now? The British Journal of Hand Therapy. 2001;6(4):109–118. [Google Scholar]
- 16.Beaton DE, Wright JG, Katz JN, Group UEC. Development of the QuickDASH: comparison of three item-reduction approaches. JBJS. 2005;87(5):1038–1046. [DOI] [PubMed] [Google Scholar]
- 17.Tretriluxana J, Gordon J, Winstein CJ. Manual asymmetries in grasp pre-shaping and transport–grasp coordination. Experimental Brain Research. 2008;188(2):305–315. [DOI] [PubMed] [Google Scholar]
- 18.Fugl-Meyer AR, Jaasko L, Leyman I, Olsson S, Steglind S. The post-stroke hemiplegic patient. 1. 1975:7. [PubMed] [Google Scholar]
- 19.Mathiowetz V, Volland G, Kashman N, Weber K. Adult norms for the Box and Block Test of manual dexterity. American Journal of Occupational Therapy. 1985;39(6):386–391. [DOI] [PubMed] [Google Scholar]
- 20.Lyle RC. A performance test for assessment of upper limb function in physical rehabilitation treatment and research. International journal of rehabilitation research Internationale Zeitschrift fur Rehabilitationsforschung Revue internationale de recherches de readaptation. 1981;4(4):483–492. [DOI] [PubMed] [Google Scholar]
- 21.Philip BA, Thompson MR, Baune NA, Hyde M, Mackinnon SE. Failure to Compensate: Patients With Nerve Injury Use Their Injured Dominant Hand, Even When Their Nondominant Is More Dexterous. Arch Phys Med Rehabil. Oct 30 2022;103(5):899–907. doi: 10.1016/j.apmr.2021.10.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stone KD, Bryant DC, Gonzalez CL. Hand use for grasping in a bimanual task: evidence for different roles? Exp Brain Res. Feb 2013;224(3):455–67. doi: 10.1007/s00221-012-3325-z [DOI] [PubMed] [Google Scholar]
- 23.Fitzpatrick AM, Dundon NM, Valyear KF. The neural basis of hand choice: An fMRI investigation of the Posterior Parietal Interhemispheric Competition model. Neuroimage. Jan 15 2019;185:208–221. doi: 10.1016/j.neuroimage.2018.10.039 [DOI] [PubMed] [Google Scholar]
- 24.Philip BA, McAvoy MP, Frey SH. Interhemispheric Parietal-Frontal Connectivity Predicts the Ability to Acquire a Nondominant Hand Skill. Brain Connectivity. May 10, 2021. 2021;11(4)doi: 10.1089/brain.2020.0916 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Franchignoni F, Vercelli S, Giordano A, Sartorio F, Bravini E, Ferriero G. Minimal clinically important difference of the disabilities of the arm, shoulder and hand outcome measure (DASH) and its shortened version (QuickDASH). journal of orthopaedic & sports physical therapy. 2014;44(1):30–39. [DOI] [PubMed] [Google Scholar]
- 26.Gummesson C, Ward MM, Atroshi I. The shortened disabilities of the arm, shoulder and hand questionnaire (QuickDASH): validity and reliability based on responses within the full-length DASH. BMC Musculoskelet Disord. May 18 2006;7:44. doi: 10.1186/1471-2474-7-44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang Y-C, Magasi SR, Bohannon RW, et al. Assessing dexterity function: a comparison of two alternatives for the NIH Toolbox. Journal of Hand Therapy. 2011;24(4):313–321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kellor M, Frost J, Silberberg N, Iversen I, Cummings R. Hand strength and dexterity. The American journal of occupational therapy: official publication of the American Occupational Therapy Association. 1971;25(2):77–83. [PubMed] [Google Scholar]
- 29.Grice KO, Vogel KA, Le V, Mitchell A, Muniz S, Vollmer MA. Adult norms for a commercially available Nine Hole Peg Test for finger dexterity. American Journal of Occupational Therapy. 2003;57(5):570–573. [DOI] [PubMed] [Google Scholar]
- 30.Gershon RC, Wagster MV, Hendrie HC, Fox NA, Cook KF, Nowinski CJ. NIH toolbox for assessment of neurological and behavioral function. Neurology. 2013;80(11 Supplement 3):S2–S6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.O*NET. U.S. Department of Labor. Accessed March 6, 2017. https://www.onetonline.org
- 32.Jebsen RH, Taylor N, Trieschmann RB, Trotter MJ, Howard LA. An objective and standardized test of hand function. Arch Phys Med Rehabil. Jul 1969;50(6):311–319. [PubMed] [Google Scholar]
- 33.Jarus T, Poremba R. Hand function evaluation: a factor analysis study. American Journal of Occupational Therapy. 1993;47(5):439–443. [DOI] [PubMed] [Google Scholar]
- 34.Philip BA, Frey SH. Compensatory Changes Accompanying Chronic Forced Use of the Nondominant Hand by Unilateral Amputees. J Neurosci. Apr 05 2014;34(10):3622–3631. doi: 10.1523/JNEUROSCI.3770-13.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Philip BA, Frey SH. Increased functional connectivity between cortical hand areas and praxis network associated with training-related improvements in non-dominant hand precision drawing. Neuropsychologia. Jul 1 2016;87:157–68. doi: 10.1016/j.neuropsychologia.2016.05.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tong J, Mao O, Goldreich D. Two-point orientation discrimination versus the traditional two-point test for tactile spatial acuity assessment. Front Hum Neurosci. 2013;7:579. doi: 10.3389/fnhum.2013.00579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Callahan A Sensibility assessment: prerequisites and techniques for nerve lesions in continuity and nerve lacerations. In: Hunter J, Mackin E, Callahan A, eds. Rehabilitation of the hand: surgery and therapy. Mosby; 1995:129–152:chap 10. [Google Scholar]
- 38.Moberg E Two-point discrimination test. A valuable part of hand surgical rehabilitation, eg in tetraplegia. Scandinavian journal of rehabilitation medicine. 1989;22(3):127–134. [PubMed] [Google Scholar]
- 39.Gonzalez CLR, Ganel T, Goodale MA. Hemispheric specialization for the visual control of action is independent of handedness. J Neurophysiol. Jul 01 2006;95(6):3496–3501. doi: 10.1152/jn.01187.2005 [DOI] [PubMed] [Google Scholar]
- 40.Friard O, Gamba M. BORIS: a free, versatile open-source event-logging software for video/audio coding and live observations. Methods in Ecology and Evolution. 2016;7(11):1325–1330. [Google Scholar]
- 41.Oldfield RC. The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia. 1971;9(1):97–113. doi: 10.1016/0028-3932(71)90067-4 [DOI] [PubMed] [Google Scholar]
- 42.Uswatte G, Taub E, Morris D, Light K, Thompson P. The Motor Activity Log-28 assessing daily use of the hemiparetic arm after stroke. Neurology. 2006;67(7):1189–1194. [DOI] [PubMed] [Google Scholar]
- 43.Baum CM, Edwards D. ACS: Activity Card Sort. Washington University School of Medicine; 2001. [Google Scholar]
- 44.Katz N, Karpin H, Lak A, Furman T, Hartman-Maeir A. Participation in occupational performance: Reliability and validity of the Activity Card Sort. OTJR: Occupation, Participation and Health. 2003;23(1):10–17. [Google Scholar]
- 45.Everard KM, Lach HW, Fisher EB, Baum MC. Relationship of activity and social support to the functional health of older adults. The Journals of Gerontology Series B: Psychological Sciences and Social Sciences. 2000;55(4):S208–S212. [DOI] [PubMed] [Google Scholar]
- 46.Bailey R, Kaskutas V, Fox I, Baum CM, Mackinnon SE. Effect of upper extremity nerve damage on activity participation, pain, depression, and quality of life. J Hand Surg Am. Nov 2009;34(9):1682–8. doi: 10.1016/j.jhsa.2009.07.002 [DOI] [PubMed] [Google Scholar]
- 47.Cella D, Yount S, Rothrock N, et al. The Patient-Reported Outcomes Measurement Information System (PROMIS): progress of an NIH Roadmap cooperative group during its first two years. Medical care. 2007;45(5 Suppl 1):S3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.RStudio: Integrated Development Environment for R. RStucio, PBC; 2022. http://www.rstudio.com [Google Scholar]
- 49.R: A language and environment for statistical computing. Foundation for Statistical Coputing; 2021. https://www.R-project.org [Google Scholar]
- 50.Lê S, Josse J, Husson F. FactoMineR: an R package for multivariate analysis. Journal of statistical software. 2008;25:1–18. [Google Scholar]
- 51.Kassambara A, Mundt F. Extract and visualize the results of multivariate data analyses [R package factoextra version 1.0. 7]. Comprehensive R Archive Network (CRAN). 2020; [Google Scholar]
- 52.James G, Witten D, Hastie T, Tibshirani R. An introduction to statistical learning. vol 112. Springer; 2013. [Google Scholar]
- 53.Seddon H Peripheral nerve injuries. Glasgow Medical Journal. 1943;139(3):61. [PMC free article] [PubMed] [Google Scholar]
- 54.McAllister R, Calder J. Paradoxical clinical consequences of peripheral nerve injury: a review of anatomical, neurophysiological and psychological mechanisms. British journal of plastic surgery. 1995;48(6):384–395. [DOI] [PubMed] [Google Scholar]
- 55.Ring D. Symptoms and disability after major peripheral nerve injury. Hand Clinics. 2013;29(3):421–425. [DOI] [PubMed] [Google Scholar]
- 56.Ewald SG, Beckmann-Fries V. Rehabilitation following peripheral nerve injury. Modern Concepts of Peripheral Nerve Repair. Springer; 2017:109–125. [Google Scholar]
- 57.Jerosch-Herold C Assessment of sensibility after nerve injury and repair: a systematic review of evidence for validity, reliability and responsiveness of tests. Journal of hand surgery. 2005;30(3):252–264. [DOI] [PubMed] [Google Scholar]
- 58.Chen L, Ogalo E, Haldane C, Bristol SG, Berger MJ. Relationship Between Sensibility Tests and Functional Outcomes in Patients With Traumatic Upper Limb Nerve Injuries: A Systematic Review. Archives of Rehabilitation Research and Clinical Translation. 2021;3(4):100159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Adidharma W, Khouri AN, Lee JC, et al. Sensory nerve regeneration and reinnervation in muscle following peripheral nerve injury. Muscle Nerve. Oct 2022;66(4):384–396. doi: 10.1002/mus.27661 [DOI] [PubMed] [Google Scholar]
- 60.Romli MH, Wan Yunus F, Mackenzie L. Overview of reviews of standardised occupation-based instruments for use in occupational therapy practice. Australian occupational therapy journal. 2019;66(4):428–445. [DOI] [PubMed] [Google Scholar]
- 61.Boone AE, Wolf TJ, Baum CM. Development and Initial Testing of the Electronic Activity Card Sort (ACS3) Among Community-Dwelling Adults. The American Journal of Occupational Therapy. 2022;76(3) [DOI] [PubMed] [Google Scholar]
- 62.Martins JC, Aguiar LT, Nadeau S, Scianni AA, Teixeira-Salmela LF, Faria C. Measurement properties of self-report physical activity assessment tools for patients with stroke: a systematic review. Braz J Phys Ther. Nov–Dec 2019;23(6):476–490. doi: 10.1016/j.bjpt.2019.02.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kender Z, Groener JB, Jende JM, et al. Diabetic neuropathy is a generalized phenomenon with impact on hand functional performance and quality of life. European Journal of Neurology. 2022;29(10):3081–3091. [DOI] [PubMed] [Google Scholar]
- 64.Chiaramonte R, Romano M, Vecchio M. A Systematic Review of the Diagnostic Methods of Small Fiber Neuropathies in Rehabilitation. Diagnostics (Basel). Aug 20 2020;10(9)doi: 10.3390/diagnostics10090613 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Vecchio M, Chiaramonte R, Romano M, Pavone P, Musumeci G, Mauro GL. A Systematic Review of Pharmacologic and Rehabilitative Treatment of Small Fiber Neuropathies. Diagnostics (Basel). Nov 28 2020;10(12)doi: 10.3390/diagnostics10121022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Anthony M, Hattori R, Nicholas ML, Randolph S, Baum CM, Connor LT. Social Support Mediates the Association between Abilities and Participation after Stroke. Unpublished work; under review. [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


