Abstract
Background:
Hyperuricemia is common in chronic kidney disease patients. This study aimed to investigate the prevalence and clinical characteristics of hyperuricemia in patients with chronic kidney disease undergoing haemodialysis.
Materials and methods:
This cross-sectional study was conducted among 104 patients with chronic kidney disease who visited the haemodialysis unit of a tertiary centre hospital from 11 September 2022, to 11 November 2022. A convenience sampling technique was used. The data were collected using a proforma form, entered into, and analyzed using the Statistical Package for Social Sciences version 21.
Results:
The mean age group of the study participants was 54.02 ± 14.03 years, and around two-thirds were male (n=70, 67.3%). Almost half of the participants (50, 48.07%) had hyperuricemia with a mean uric acid level of 6.76 ± 2.62 mg/dl. The majority of the participants had associated hypertension (n=100, 96.2%). More than three-fourths of the respondents (83, 79.8%) had haemoglobin levels below the target range.
Conclusion:
Nearly half of the study participants had hyperuricemia.
Keywords: chronic renal insufficiency, cross-sectional study, hyperuricemia, Nepal, renal dialysis
Introduction
Highlights
Chronic kidney disease (CKD) refers to abnormal kidney structure or function for 3 or more months.
There is decreased urinary excretion of uric acid in CKD patients.
Hyperuricemia is common in CKD patients.
Chronic kidney disease (CKD) is defined as the abnormalities of the kidney structure or function for greater than three months, with health implications1. Hyperuricemia is defined by the serum uric acid level greater than 7.0 mg/dl in males and greater than 6.0 mg/dl in females2. CKD results in electrolyte abnormalities including but not limited to hyperuricemia, hyperkalemia, hypocalcemia, hyperphosphatemia, and metabolic acidosis3,4. Hyperuricemia is common in CKD patients with its severity increasing with the progression of the chronic kidney disease.
Hyperuricemia has been associated with the development of several cardiovascular diseases and hypertension5,6. However, there are some controversies regarding hyperuricemia as a risk factor for CKD. Some studies have shown asymptomatic hyperuricemia to be a risk factor for its progression7,8. Untreated hyperuricemia is an independent risk factor for the onset of CKD9,10. Some studies suggest that hyperuricemia with crystalluria and not asymptomatic hyperuricemia alone contributes to the disease progression11.
Elevated serum uric acid is significantly associated with the risk of mortality; a 1 mg/dl increase in serum uric acid level correlates with an 8% increase in mortality12. Hence it is important to acknowledge hyperuricemia in CKD patients and manage it appropriately. Our study aimed to find the prevalence and clinical characteristics of hyperuricemia in patients with end-stage kidney disease (ESKD).
Methods
Study design and setting
This study was conducted in a haemodialysis unit of a tertiary care hospital where it receives a high volume of CKD patients representing different social and economic backgrounds visiting the haemodialysis unit for dialysis.
Study participants
A descriptive cross-sectional study was conducted among all the patients with chronic kidney disease who gave consent and underwent haemodialysis at the haemodialysis unit of a tertiary care hospital. The study took place from 11 September 2022 to 11 November 2022.
Sampling and sample size
With a convenience sampling approach, the study included patients with CKD who visited the haemodialysis unit of a tertiary care hospital during the research period and those who were receiving medical therapy. Patients aged more than 18 years who were diagnosed with CKD attending the haemodialysis, and willing to participate were included. Patients less than 18 years of age, known cases of hyperuricemia, and gout, and those who were unwilling to participate were excluded from the research.
Study instrument
A semi-structured pre-designed proforma prepared by the investigators themselves after studying other similar studies was used to collect the data. The proforma was pre-tested in 10% of the sample before being conducted, and necessary modifications were made.
Demographic data including age, sex, address, educational status, and duration of dialysis were gathered from the interviews of the patients. Data regarding co-morbidities like hypertension, diabetes, dyslipidemia, hyperuricemia, evidence of atherosclerosis, and others along with their durations were extracted from the patient’s hospital record book. A complete physical examination was performed including vital signs such as blood pressure, pulse rate, respiratory rate, and temperature, and pertinent systemic examinations.
Laboratory investigations including complete blood count (CBC), serum calcium, serum phosphate, serum uric acid, serum sodium, serum potassium, total iron, serum ferritin, total reticulocyte count, mean corpuscular volume (MCV), mean corpuscular haemoglobin concentration (MCHC), and serology for HIV, hepatitis B, and hepatitis C were sent. Serum uric acid estimation was done by the uricase method. Different medical treatments including Erythropoietin supplements, allopurinol, non-steroidal anti-inflammatory drugs (NSAIDs), and thiazides along with dietary restrictions were collected from the personal record book.
Statistical analysis
Data were entered into and analyzed using Statistical Package for Social Sciences (SPSS) version 21. Descriptive statistics like frequency, percentages, mean ± SD, and/or median (minimum-maximum) were calculated wherever required and depicted accordingly in bar diagrams and pie-chart. The association of hyperuricemia with selected background characteristics was assessed using the chi-square test and binary logistic regression. A P value less than 0.05 was considered significant. The 95% CI of prevalence was estimated using OpenEpi version 3.
Ethical consideration
A letter of ethical approval was obtained from the Institutional Review Committee (IRC) of the institution (IRC Reg. No. 647). Written informed consent was taken from the participants. Anonymity and privacy of patient information were maintained.
The manuscript has been reported in line with the STROCSS criteria13.
Results
Among 104 patients visiting the haemodialysis unit on an outpatient basis, 50 (48.07%, 95% CI 38.72–57.57) were found to have hyperuricemia with a mean uric acid level of 6.76 ± 2.62 mg/dl [6.85 (0.4–13)].
Most of the people visiting the haemodialysis unit were of the age group 40–59 years (44, 42.3%) with a mean age of 54.02 ± 14.03 years. Around two-thirds of the participants were male (70, 67.3%). The most common associated comorbidity was hypertension (100, 96.2%). Most of the patients (45, 43.3%) had a history of haemodialysis started in the past 5 years, while half of the patients (54, 51.9%) had a history of erythropoietin use for anaemia in previous months. Complete blood counts and peripheral blood smears of the patients showed, mean haemoglobin of 9.19± 1.42 g/dl, MCV of 87.21± 15.87 fl, and MCHC of 32.90± 1.06 g/dl. Analysis of serum electrolytes profile revealed, Na+ of 136.64± 5.69 mEq/l, K+ of 5.10± 1.08 mEq/l, Ca2+ of 8.54± 1.52 mg/dl and PO43- of 5.95± 2.14 mg/dl (Table 1).
Table 1.
Background clinical characteristics of participants with ESKD attending haemodialysis on an outpatient basis
| Variables | Frequency, n (%) | |
|---|---|---|
| Age | ||
| 20–39 | 22 (21.2) | |
| 40–59 | 44 (42.3) | |
| ≥60 | 38 (36.5) | |
| Sex | ||
| Female | 34 (32.7) | |
| Male | 70 (67.3) | |
| Co-morbidities | ||
| Hypertension | 100 (96.2) | |
| Diabetes | 13 (12.5) | |
| Dyslipidemia | 6 (5.8) | |
| Othersa | 9 (8.7) | |
| Dialysis duration (years) | ||
| <5 | 45 (43.3) | |
| 5–10 | 26 (25.0) | |
| >10 | 33 (31.7) | |
| Erythropoietin useb | ||
| No | 50 (48.1) | |
| Yes | 54 (51.9) | |
| Normal values | Mean ± SD or [Median (Min–Max)] | |
| Age (years) | — | 54.02 ± 14.03 [55 (22–87)] |
| Dialysis duration (years) | — | 7.31 ± 5.68 [6 (1–20)] |
| SBPc, mm Hg | ≤130 | 148.94± 20.99 [140 (110–200)] |
| DBPc, mm Hg | ≤80 | 88.15± 10.48 [90 (70–120)] |
| Hemoglobinc, g/dl | 10–11.5 (KDIGO 2012) | 9.19± 1.42 [9 (6.20–13.80)] |
| MCV, fl | 80–100 | 87.21± 15.87 [90.40 (8.10–104)] |
| MCHC, g/dl | 33 ± 3 | 32.90± 1.06 [33.10 (30–35.1)] |
| Na+, mEq/l | 135–145 | 136.64± 5.69 [136 (113–158)] |
| K+, mEq/l | 3.5–5.2 | 5.10± 1.08 [5 (3.5–8.60)] |
| Ca2+, mg/dl | 8.4–10.2 | 8.54± 1.52 [8.43 (3.79–13)] |
| PO43-, mg/dl | 3–4.5 | 5.95± 2.14 [5.52 (2.37–12.08)] |
COPD, chronic obstructive pulmonary disease; DBP, Diastolic blood pressure; ESKD, end-stage kidney diseas; Max, maximum; MCHC, mean corpuscular haemoglobin concentration; MCV, mean corpuscular volume; Min, minimum; SBP, systolic blood pressure.
Others co-morbidities: thyroid dysfunction, COPD, asthma.
Erythropoietin use in previous months.
Optimal targets defined as in this study.
Distribution pattern of haemoglobin and red blood cell indices
79.8% of the total patients had haemoglobin levels below the target range. However, most of them had normal MCV (85.6%) and all the patients had normal MCHC (Fig. 1).
Figure 1.
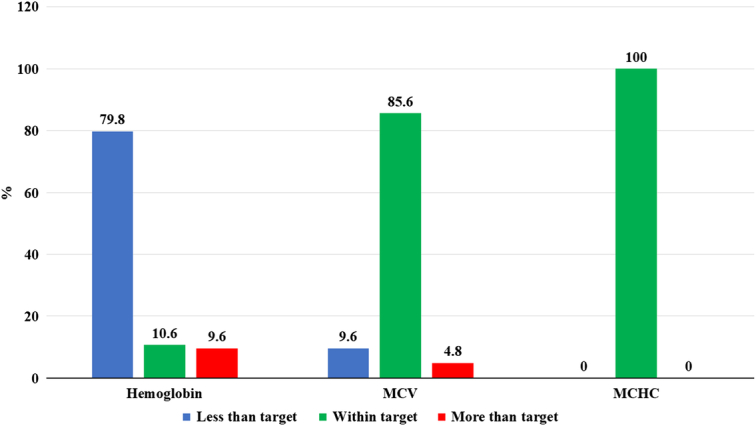
Distribution pattern of haemoglobin and red blood cell indices. MCHC, mean corpuscular haemoglobin concentration; MCV, mean corpuscular volume.
Distribution pattern of electrolytes
Most of the patients had normal sodium (52.9%) and potassium (61.5%) levels, lower calcium levels (48.1%), and higher phosphate level (74%) (Fig. 2).
Figure 2.

Distribution pattern of electrolytes.
Serum uric acid profile
Among all those patients visiting the haemodialysis unit on an outpatient basis, 48.1% of the patients had increased uric acid levels (Fig. 3).
Figure 3.
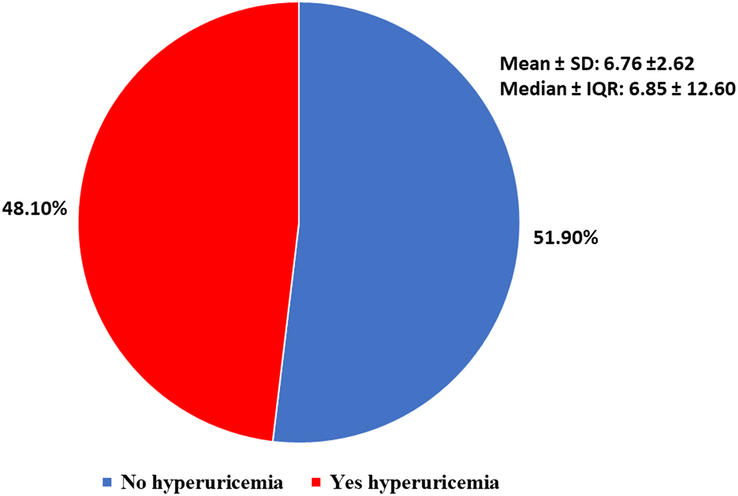
Serum uric acid profile of the participants. IQR, interquartile range.
Effects of different variables on serum uric acid level
We used the χ2 test and binary logistic regression to analyze the effect of different variables on serum uric acid levels. As shown in Table 2, there were statistically non-significant lower odds of hyperuricemia in those aged 40–59 years [odds ratio (OR) 0.76, 95% CI 0.27–2.12) and aged older than or equal to 60 (OR 0.67, 95% CI 0.23–1.94) than those aged 20–29 years, those with dialysis 5–10 years (OR 0.42, 95% CI 0.16–1.15) and older than 10 years (OR 0.75, 95% CI 0.30–1.85) than those for younger than 5 years and non-erythropoietin use in previous months (OR 0.62, 95% CI 0.29–1.36) than erythropoietin users. The male participants had non-significant higher odds of hyperuricemia (OR 1.27, 95% CI 0.56–2.89) than female participants.
Table 2.
Effect of selected clinical variables on serum uric acid level of participants with ESKD attending haemodialysis on an outpatient basis
| Hyperuricemia (≥7 mg/dl) | Binary logistic regression | |||
|---|---|---|---|---|
| Variables | No, n (%) | Yes, n (%) | OR (95% CI) | P a |
| Age (years) | 0.541 | |||
| 20–39 | 10 (45.5) | 12 (54.5) | 1 (Ref.) | |
| 40–59 | 23 (52.3) | 21 (47.7) | 0.76 (0.27–2.12) | |
| ≥60 | 21 (55.3) | 17 (44.7) | 0.67 (0.23–1.94) | |
| Sex | 0.314 | |||
| Female | 19 (55.9) | 15 (44.1) | 1 (Ref.) | |
| Male | 35 (50.0) | 35 (50.0) | 1.27 (0.56–2.89) | |
| Duration of haemodialysis | 0.235 | |||
| <5 | 20 (44.4) | 25 (55.6) | 1 (Ref.) | |
| 5–10 | 17 (65.4) | 9 (34.6) | 0.42 (0.16–1.15) | |
| >10 | 17 (51.5) | 16 (48.5) | 0.75 (0.30–1.85) | |
| Erythropoietin use (prior month) | 0.233 | |||
| Yes | 25 (46.3) | 29 (53.7) | 1 (Ref.) | |
| No | 29 (58.0) | 21 (42.0) | 0.62 (0.29–1.36) | |
ESKD, end-stage kidney diseas; OR, odds ratio.
P value from the χ2 test.
Discussion
Our cross-sectional study included 104 patients with chronic kidney disease stage 5 under haemodialysis. In our study, more than three-fourths of the study participants (82, 78.8%) were aged 40 years and above, with a mean age of 54.02 ± 14.03 years and the majority being male (70, 67.3%). The demographic findings in our study were similar to other studies from Nepal. A study conducted among CKD V visiting the haemodialysis unit of a teaching hospital in Nepal showed the mean age of the participants was 46.9 ± 17.9 years, with male predominance (57, 57%)14. Similarly, another observational study from a tertiary hospital in Nepal showed the mean age of end-stage kidney disease patients on maintenance haemodialysis was 52.2 ± 15.6 years, with male predominance (96, 61.5%)15. A nationwide population-based cross-sectional study from Nepal showed that CKD was independently associated with older age, with the odds of having CKD being 1.4 (95% CI 1.0–1.8) and 2.6 (95% CI 1.9–3.6) times higher for 40–59 years and older than or equal to 60 years compared to 20–39 years people16. Likewise, a study from Pakistan also supports the demographic findings of our study, which showed the mean age of CKD patients undergoing maintenance haemodialysis was 50.3 ± 12.1 years with a higher prevalence in males (47, 64.4%)17.
CKD and hypertension are closely related conditions, the latter can worsen kidney function while the former can, in turn, make it more difficult to control blood pressure18. Hypertension in CKD could be due to various factors such as reduced nephron mass, endothelial dysfunction, sympathetic overactivity, activation of the renin-angiotensin-aldosterone system, and increased sodium and water retention18. Longer haemodialysis sessions were associated with a highly statistically significant decrease in mean systolic and diastolic blood pressure (p value < 0.001)19. Our study showed that hypertension (100, 96.2%) was the most commonly associated comorbidity followed by diabetes (13, 12.5%). A nationwide population-based representative study from Nepal showed that there are higher odds of occurrence of CKD among patients with diabetes and hypertension, 3.2 (95% CI 2.5–4.1) and 2.4 (95% CI 2.0–3.0) times higher than normal participants, respectively16. A study among CKD patients under haemodialysis from Pakistan and Nepal showed similar results, the former study showed hypertension (38, 52.1%) and diabetes (30, 41.1%)20 to be the most commonly associated comorbidity, and similar results in the latter study with 54% of hypertension and 18% of diabetes patients14. In contrast to our study, other studies from Nepal showed that 42 (30.4%)21 and 68 (43.6%)15 patients undergoing maintenance haemodialysis had diabetes mellitus as a leading cause of ESKD.
Anaemia is a very common complication associated with reduced quality of life and cardiovascular abnormalities in chronic kidney disease patients14. Looking at the haematological parameters in our study, 79.8% of patients had haemoglobin levels below the target range. However, most of them had normal MCV (85.6%) and all the patients had normal MCHC (100%). Our findings were supported by other studies from Nepal which showed that anaemia was present among 85 (85%)14 and 127 (92%)21 CKD patients on haemodialysis respectively. Likewise, a cross-sectional study from India conducted among chronic kidney disease patients under haemodialysis also showed that 124 (96.8%) CKD patients were anaemic, with normocytic normochromic anaemia (113, 91.1%) being the most common type22.
Lack of erythropoietin is one of the several mechanisms for the development of anaemia in CKD patients14. Half of our study population (54, 51.9%) also used erythropoietin, similar to a cross-sectional study from Nepal where 78 (43.5%) end-stage kidney disease patients under haemodialysis were regularly using erythropoietin analogues21. The clinical use of erythropoietin in the correction of anaemia among CKD patients has been successful in reducing related morbidity and mortality, improving functioning, exercise tolerance, cognitive function, and overall quality of life, through its anti-apoptotic, anti-inflammatory, angiogenetic, and cytoprotective effects in kidneys, cardiovascular system, brain and retina23. However, higher dose requirement may exert toxic effects and is an independent mortality predictor in haemodialysis patients, and adjuvant therapies should be considered for poor responders to erythropoietin23,24.
In our study, almost half of the CKD patients undergoing haemodialysis (50, 48.07%) were found to have hyperuricemia, which is higher compared to another study from Nepal (43, 31.2%), conducted among end renal stage disease patients undergoing haemodialysis21. However, the prevalence of hyperuricemia in our study was lower compared to the study from Pakistan that showed nearly three-fourths of patients (56, 76.7%) who were under thrice weekly haemodialysis had hyperuricemia17. In our study, hyperuricemia was more prevalent in males (35, 70%). The higher prevalence of hyperuricemia in males could be due to their habit of smoking and alcohol consumption tendency than females25.
In various studies, evidence has shown hyperuricemia is not only a marker of renal dysfunction but also plays role in the progression and pathogenesis of CKD17,26,27. Hyperuricemia is associated with an increased risk of all causes of mortality (especially cardiovascular) among patients with CKD and on haemodialysis12,26–28. A study done by Selllamyr et al showed asymptomatic hyperuricemia does not particularly affect CKD progression unless uric acid crystallizes in the kidney which contributes to CKD progression29. However, urate-lowering therapies have shown evidence to slow the progression. A systematic review and meta-analysis of 12 randomized controlled trials reported that the treatment group (uric acid lowering therapy) had a reduced risk of worsening kidney function or ESKD or mortality than the control group (RR 0.39, 95% CI 0.28–0.52, P<0.01)30.
Malnutrition might adversely affect the outcome in haemodialysis patients 15; however, nutritional status and body mass index (BMI) of the participants were not accounted for in our study, which is a limitation of the study. Likewise, dietary regimens that might influence serum uric acid, alcohol consumption habits, and medication that might alter the serum uric acid level weren’t considered during the study. This study being a single centred cross-sectional study, the findings mightn’t be generalized, highlighting the need for multi-centric studies in the future.
Conclusion
Our study showed that nearly half of the chronic kidney disease patients under haemodialysis had hyperuricemia. Around three-fourths of the study participants with hyperuricemia were aged 40 years and above, the majority being male. The most commonly associated comorbidity was hypertension and normocytic normochromic anaemia was common in the study participants.
Ethical approval
The study was approved by the Institutional Review Committee (IRC) of the institution (Reference no. 647) on July 2022.
Consent
Written informed consent was obtained from the participants for publication and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Source of funding
This study did not receive any grant from any funding agencies in public or organizations.
Author contribution
A.M.B.: literature review, conceptualization, methodology, data collection, writing—original draft, review and edit. S.G.: literature review, conceptualization, methodology, data collection, formal analysis, writing—original draft, review and edit. A.A.: literature review, conceptualization, methodology, data collection, writing—original draft, review and edit. S.K.: literature review, conceptualization, methodology, data collection, writing—original draft, review and edit. B.D.P.: literature review, conceptualization, methodology, data collection, writing—original draft, review and edit. B.R.: literature review, methodology, data interpretation, writing—original draft, review and edit. M.K.: literature review, methodology, data interpretation, writing—original draft, review and edit. G.K.Y.: literature review, methodology, data interpretation, writing—original draft, review and edit. S.B.: literature review, methodology, data interpretation, writing—original draft, review and edit. All the authors approved the final version of the manuscript.
Conflicts of interest disclosure
The authors declare that they have no conflicts of interest.
Research registration unique identifying number (UIN)
Name of the registry: research registry.
Unique Identifying number or registration ID: researchregistry9269.
Hyperlink to your specific registration (must be publicly accessible and will be checked): researchregistry.
Guarantor
Dr. Ayush Mohan Bhattarai.
Data availability statement
The datasets generated during and/or analyzed during the current study are publicly available.
Provenance and peer review
Not commissioned, externally peer-reviewed.
Footnotes
Sponsorships or competing interests that may be relevant to content are disclosed at the end of this article
Published online 15 January 2024
Contributor Information
Ayush Mohan Bhattarai, Email: ayushbhattarai77@gmail.com.
Shekhar Gurung, Email: drshekhargurung@gmail.com.
Ayush Adhikari, Email: ayush_adhikari@ymail.com.
Saurab Karki, Email: saurabkarki1010@gmail.com.
Bishnu Deep Pathak, Email: bishnupathak433@gmail.com.
Bishal Regmi, Email: bishal.regmi06@naihs.edu.np.
Manoj Khadka, Email: khadkamanoj432@gmail.com.
Gopal Kumar Yadav, Email: gopalbpkihs@gmail.com.
Somika Basnet, Email: basnetsomika@gmail.com.
References
- 1.KDIGO 2012Clinical practice guideline for the evaluation and management of chronic kidney disease. Off J Int Soc Nephrol, 3x. [DOI] [PubMed] [Google Scholar]
- 2.Chuang SY, Chen JH, Yeh WT, et al. Hyperuricemia and increased risk of ischemic heart disease in a large Chinese cohort. Int J Cardiol 2012;154:316–321. [DOI] [PubMed] [Google Scholar]
- 3.Ohno I. Relationship between hyperuricemia and chronic kidney disease. Nucleosides Nucleotides Nucleic Acids 2011;30:1039–1044. [DOI] [PubMed] [Google Scholar]
- 4.Barbour SJ, Er L, Djurdjev O, et al. The prevalence of hematologic and metabolic abnormalities during chronic kidney disease stages in different ethnic groups. Kidney Int 2008;74:108–114. [DOI] [PubMed] [Google Scholar]
- 5.Stack AG, Hanley A, Casserly LF, et al. Independent and conjoint associations of gout and hyperuricaemia with total and cardiovascular mortality. QJM 2013;106:647–658. [DOI] [PubMed] [Google Scholar]
- 6.Gagliardi ACM, Miname MH, Santos RD. Uric acid: a marker of increased cardiovascular risk. Atherosclerosis 2009;202:11–17. [DOI] [PubMed] [Google Scholar]
- 7.Momoki K, Kataoka H, Moriyama T, et al. Hyperuricemia as a predictive marker for progression of nephrosclerosis: clinical assessment of prognostic factors in biopsy-proven arterial/arteriolar nephrosclerosis. J Atheroscler Thromb 2017;24:630–642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Petreski T, Ekart R, Hojs R, et al. Asymptomatic hyperuricemia and cardiovascular mortality in patients with chronic kidney disease who progress to hemodialysis. Int Urol Nephrol 2019;51:1013–1018. [DOI] [PubMed] [Google Scholar]
- 9.Weiner DE, Tighiouart H, Elsayed EF, et al. Uric acid and incident kidney disease in the community. J Am Soc Nephrol 2008;19:1204–1211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chonchol M, Shlipak MG, Katz R, et al. Relationship of uric acid with progression of kidney disease. Am J Kidney Dis 2007;50:239–247. [DOI] [PubMed] [Google Scholar]
- 11.Sellmayr M, Petzsche MRH, Ma Q, et al. Only hyperuricemia with crystalluria, but not asymptomatic hyperuricemia, drives progression of chronic kidney disease. J Am Soc Nephrol 2020;31:2773–2792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Xia X, Luo Q, Li B, et al. Serum uric acid and mortality in chronic kidney disease: a systematic review and meta-analysis. Metabolism 2016;65:1326–1341. [DOI] [PubMed] [Google Scholar]
- 13.Mathew G, Agha R, for the STROCSS Group . STROCSS 2021: Strengthening the Reporting of cohort, cross-sectional and case-control studies in Surgery. Int J Surg 2021;96:106165. [DOI] [PubMed] [Google Scholar]
- 14.Chhetri PK, Manandhar DN, Bhattarai SP, et al. Chronic kidney disease 5 on hemodialysis in Nepal Medical College Teaching Hospital. Nep Med Col J 2008;10:8–10. [PubMed] [Google Scholar]
- 15.Ghimire M, Vaidya S, Upadhyay HP. Clinicodemographic profile and outcome of maintenance hemodialysis patients in a tertiary hospital of central nepal, chitwan. Kathmandu Univ Med J 2020;18:9–14. [PubMed] [Google Scholar]
- 16.Poudyal A, Karki KB, Shrestha N, et al. Prevalence and risk factors associated with chronic kidney disease in Nepal: evidence from a nationally representative population-based cross-sectional study. BMJ Open 2022;12:e057509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Moon F, Alam S, Yaqoob MY, et al. Prevalence of hyperuricemia in thrice weekly hemodialysis patients. Pakistan J Kidney Dis 2022;6:10–14. [Google Scholar]
- 18.Ku E, Lee BJ, Wei J, et al. Hypertension in CKD: Core Curriculum 2019. Am J Kidney Dis 2019;74:120–131. [DOI] [PubMed] [Google Scholar]
- 19.Teama NM, Soliman HS, Elsharkawy M, et al. Impact of hemodialysis time prolongation on blood pressure control. Egypt J Hosp Med 2021;83:949–956. [Google Scholar]
- 20.Moon F, Alam S, Yaqoob M Yousuf, et al. Prevalence of hyperuricemia in thrice weekly hemodialysis patients. Pakistan J Kidney Dis 2022;6:10–14. [Google Scholar]
- 21.Nepal R, Sapkota K, Paudel M, et al. Clinical profile of end stage renal disease patients undergoing hemodialysis in Chitwan. Nepal, J Nepal Health Res Counc 2021;19:467–473. [DOI] [PubMed] [Google Scholar]
- 22.Khadayate R, Sahu P, Sahu S, et al. Study of hematological profile in chronic renal failure patients on hemodialysis in a tertiary care hospital. Int J Heal Sci Res 2020;10:12. [Google Scholar]
- 23.Provatopoulou ST, Ziroyiannis PN. Clinical use of erythropoietin in chronic kidney disease: outcomes and future prospects. Hippokratia 2011;15:109. [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang Y, Thamer M, Stefanik K, et al. Epoetin requirements predict mortality in hemodialysis patients. Am J Kidney Dis 2004;44:866–876. [PubMed] [Google Scholar]
- 25.Mufti NUD, Khan RM, Haseeb A, et al. Hyperuricemia in patients with chronic renal failure: a single center study, Pakistan. Pakistan J Med Heal Sci 2022;16:634–634. [Google Scholar]
- 26.Gouri A, Dekaken A, Bentorki AA, et al. Serum uric acid level and cardiovascular risks in hemodialysis patients: an Algerian cohort study. Clin Lab 2014;60:751–758. [DOI] [PubMed] [Google Scholar]
- 27.Sugano N, Maruyama Y, Kidoguchi S, et al. Effect of hyperuricemia and treatment for hyperuricemia in Japanese hemodialysis patients: a cohort study 2019PLoS One, 14:e0217859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Luo Q, Xia X, Li B, et al. Serum uric acid and cardiovascular mortality in chronic kidney disease: a meta-analysis. BMC Nephrol 2019;20:1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sellmayr M, Hernandez Petzsche MR, Ma Q, et al. Only hyperuricemia with crystalluria, but not asymptomatic hyperuricemia, drives progression of chronic kidney disease. J Am Soc Nephrol 2020;31:2773–2792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu X, Zhai T, Ma R, et al. Effects of uric acid-lowering therapy on the progression of chronic kidney disease: a systematic review and meta-analysis. Ren Fail 2018;40:289–297. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are publicly available.


