Abstract
Purpose
Tubulointerstitial nephritis and uveitis (TINU) syndrome is an uncommon disease. We present a confirmed case of TINU syndrome, and a systematic review of epidemiological characteristics, clinical manifestations, management, and outcomes in Chinese patients.
Methods
A systematic search was carried out using defined terms and updated up to September 2022, in PubMed, Web of Science, Wanfang, CNKI, and VIP, to identify reported cases of TINU in China, according to PRISMA guidelines.
Results
An 18-year-old boy presented with elevated serum creatinine and 24-h urine protein level of > 2 g. Inspection result revealed acute tubulointerstitial nephritis, and bilateral uveitis. The patient was diagnosed with TINU syndrome and received treatment with methylprednisolone sodium succinate, which resulted in a significant decrease in creatinine and urinary protein levels. Systematic review identified 35 publications that met the inclusion criteria. A total of 71 cases were included in this article, of which 70 were from publications and 1 was from our hospital. The median age at onset was 42 years and was significantly lower in males than females (P < 0.05). The symptoms of uveitis often occurred after kidney injury (54%) and most uveitis was anterior (55%) and bilateral (75%). Among the 51 patients who were followed up for more than 6 months, 24 had recurrent ocular symptoms or progression to chronic uveitis. Twenty patients experienced chronic or progressive kidney disease.
Conclusion
TINU syndrome is prone to misdiagnosis because kidney damage may not occur simultaneously with uveitis. The incidence of kidney sequelae in children is lower than that in adults, and glucocorticoids are the preferred treatment.
INPLASY registration number
INPLASY202350050.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11255-023-03797-6.
Keywords: Tubulointerstitial nephritis, Uveitis, TINU syndrome, Glucocorticoid, Systematic review
Introduction
Tubulointerstitial nephritis (TINU) syndrome, triggered by external factors, including drugs and microbial pathogens, is an autoimmune disease involving multiple systems [1]. In the past 45 years, approximately 600 such cases have been reported worldwide. Approximately 2% of patients with uveitis are diagnosed with TINU syndrome [2]. The pathophysiology of TINU syndrome is currently unclear. In 1985, circulating immune complexes were first identified in patients with TINU syndrome [3]. Subsequently, researchers found that the concentration of C4 was decreased in patients [4–6]. Therefore, humoral and cellular immunity are involved in its occurrence and development [7].
The TINU syndrome is characterized by tubulointerstitial nephritis and uveitis. The main manifestation of kidney is acute renal injury, and some patients may develop permanent renal injuries [8]. Eye symptoms can manifest in different forms include redness, eye pain, blurred vision, etc. The most common being bilateral anterior uveitis and may develop to a chronic [9]. Approximately 80 cases of TINU have been reported in China. Most reports are of single cases, with few clinical studies with large samples, and this have biased understanding of the pathogenesis of TINU syndrome, diagnosis, and the clinical efficacy evaluation of treatment methods. We performed a systematic review of previous case reports and small-sample observational studies in China to further study the epidemiological characteristics, clinical manifestations, diagnosis and treatment methods, and clinical outcomes of TINU syndrome.
Methods
This review was performed in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses statements (PRISMA). Although there were two previous systematic reviews on TINU syndrome, involving western populations. We focus this systematic review on evaluation of the epidemiological characteristics, clinical manifestations, management, and outcomes of TINU syndrome reported in China. Improve awareness of TINU syndrome and reduce misdiagnosis rates.
Search strategy
This systematic literature review on TINU syndrome adhered to the PRISMA guidelines. Articles were located by searching the terms ‘tubulointerstitial nephritis and uveitis’ OR ‘tubulointerstitial nephritis and uveitis syndrome’ OR ‘TINU syndrome’ OR ‘tubulointerstitial nephritis with uveitis’ OR ‘Dobrin syndrome’, AND ‘China’, in the following databases from inception to September 2022: PubMed, Web of Science, Wanfang, CNKI, and VIP (Supplemental appendix A).
We included studies related to TINU syndrome that met the Mandeville diagnostic criteria. If the two reports described the same patient(s), we only considered the more detailed publication. If reports were duplicated, the most recent publication was retained. We excluded reviews and cases that lacked demographic data, or were not from China.
Data extraction
Two authors independently reviewed the titles, abstracts, and full texts of all retrieved articles and assessed whether the studies met the inclusion criteria. If there was a disagreement, a third reviewer was consulted.
All patient data included in the literature were classified and counted using an Excel table. The following data were collected: age, sex, pathogeny (infection, drug), prodrome (fever, fatigue, gastrointestinal symptoms, weight loss, and nocturia increase), eye symptoms (redness, pain, blurred vision, photophobia), type of uveitis, occurrence of the sequence of uveitis and TIN (uveitis occurs before renal injury, and both occur in parallel: the time between uveitis and renal injury is within 3 weeks; uveitis occurs after renal injury), treatment, follow-up time, and prognosis. In addition, the following laboratory parameters were collected: creatinine, urea, erythrocyte sedimentation rate (ESR), urinalysis, antinuclear antibodies (ANAs), C3, and C4.
We aimed to provide an overview of TINU symptoms, regardless of the risk of bias in the included studies. Therefore, the methodological quality of the included studies was not formally evaluated.
Statistics
SPS software (version 25.0; SPSS Inc., Chicago, IL) was used for the statistical analysis. The measurement data conforming to the normal distribution is expressed by mean ± standard deviation, and the comparison between groups was conducted using the student’s t-test. The non-normally distributed measurement data were expressed as median and quartile intervals, and the inter-group comparison was conducted using the rank sum test. The counting data were described by the rate. When the theoretical frequency was ≥ 5 and n > 40, the inter-group comparison used the Chi-squared (χ2) test; when the theoretical frequency was less than 5 or n < 40, Fisher's exact probability method was used. Statistical significance was set at P < 0.05.
Patient and public involvement
Not required.
Results
Case reports
The case report was conducted in accordance to the World Medical Association Declaration of Helsinki and approved by the Ethics Committee of Jiaxing First Hospital (2022-LY-411). The patient had signed informed consent forms.
An 18-year-old male patient was admitted to our hospital with a 1-month anorexia, accompanied by nausea, vomiting, and weight loss. The patient had been administered ceftriaxone and omeprazole for 15 days at a local hospital, without significant improvement.
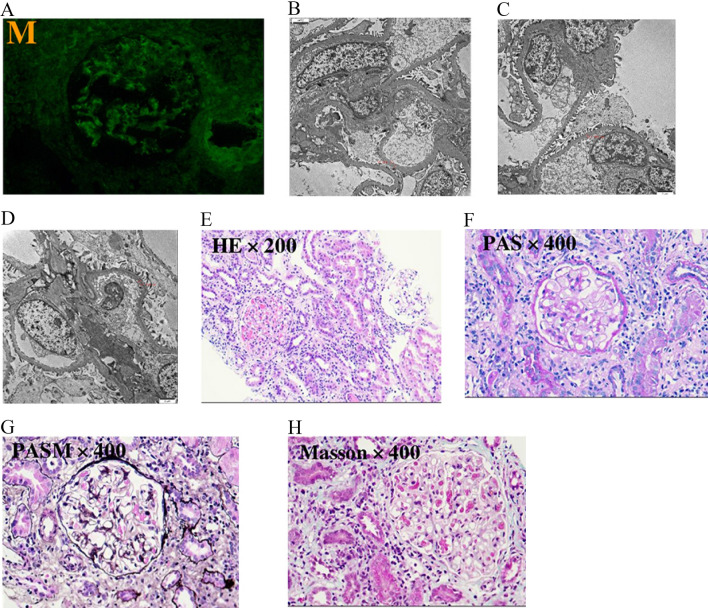
Serum creatinine and urine protein levels increased within 3 months. Therefore, this patient was diagnosed with acute kidney disease. A detailed list of the paraclinical investigations is presented in Supplementary Appendix B. The kidney biopsy showed acute tubulointerstitial nephritis (Fig. 1).
Fig. 1.
Results of renal biopsy. A Immunofluorescence: no immune complex deposit. B–D The electron microscope showed that the podocytes of the glomerulus were segmental fused, and there was no exact deposition of electronic dense matter in various parts of the glomerulus. (E) HE staining (×200). (F) PAS staining (×400). (G) PASM staining (×400). (H) Masson staining
The patient had no recent history of exposure to chemicals, drugs or foods that might cause acute renal injury. The patient reported redness and discomfort in his eyes during his preceding admission to a local hospital, which improved after 1 day. Accordingly, we consulted ophthalmologists and conducted slit-lamp examinations, which identified bilateral iridocyclitis (Fig. 2).
Fig. 2.
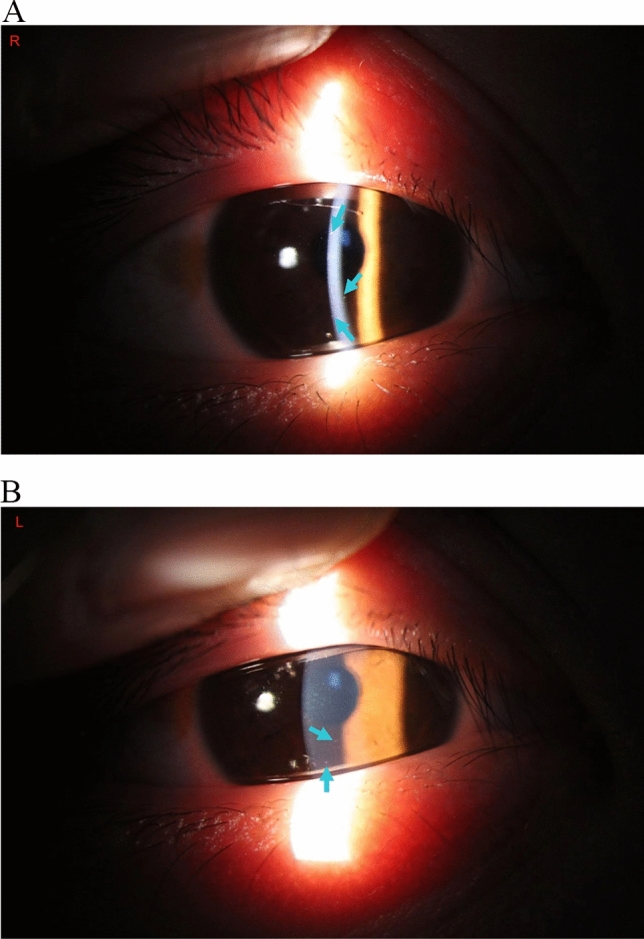
Slit lamp inspection results. (A) Right eye. (B) Left eye. The green arrow indicates keratic precipitates (KP)
TINU syndrome was diagnosed, based on the renal pathology and the ocular lesions. Methylprednisolone (80 mg) was administered intravenously once a day for four consecutive days, and methylprednisolone tablets (48 mg) were administered once a day. All indicators improved after the treatment (Supplement Appendix B). During a follow-up period of > 5 months, the patient’s steroid therapy was discontinued, and there was no recurrence of renal damage or uveitis.
Systematic review search results
A total of 115 relevant documents were retrieved; 57 duplicate documents were excluded, three documents were excluded because they are not related to the theme of this article after review of titles and abstracts, and 20 documents were excluded according to the exclusion criteria after visiting the full text (Fig. 3). Thirty-five publications met the inclusion criteria [10–44], comprising 34 case reports and one case series, totaling 70 cases. The characteristics of the included publications are summarized in Table 1.
Fig. 3.
PRISMA study selection flowchart for systematic literature Review
Table 1.
Characteristics of the included studies
| Author | Age (years) | Gender | Eye symptoms | Urinalysis | Renal biopsy | Uveitis | Uveitis onset | Treatment | follow-up time(month) | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|
| Wu Hua et al. | 18 | Male | Eye redness |
Proteinuria Glycosuria Microscopic hematuria |
Interstitial nephritis | Bilateral | After |
Hormone Immunosuppressant |
6 | Recurrence of kidney disease or CKD |
| Liu Guangren et al. | 14 | Female | NA | NA | Acute interstitial nephritis | NA | Before | Hormone | 36 | Recurrence of kidney disease or CKD |
| Zhao Minghui et al. | 74 | Female | NA |
Proteinuria Glycosuria |
Acute interstitial nephritis | NA | After | Hormone | 12 |
Recurrence of kidney disease or CKD Recurrence of uveitis or Chronic uveitis |
| Ma Yingchun et al. | 28 | Male | NA |
Proteinuria Glycosuria Microscopic hematuria |
Acute interstitial nephritis | Bilateral | After | Hormone | 1 | No recurrence |
| Zhang Minfang et al. | 43 | Female |
Eye redness Blurred vision |
Proteinuria | Acute interstitial nephritis | Bilateral | Before | Hormone | 2 | No recurrence |
| Li Yayu et al. | 75 | Female |
Eye pain Blurred vision Photophobia |
Glycosuria | NA | Bilateral | After | Hormone | 0.5 | No recurrence |
| Cheng Hongxin et al. | 41 | Female | Eye redness |
Proteinuria Glycosuria |
Interstitial nephritis | Unilateral | After |
Hormone Immunosuppressant |
NA | No recurrence |
| Zhang Wen et al. | 56 | Female |
Eye redness Eye pain Photophobia |
Proteinuria | Acute interstitial nephritis | Bilateral | Before | Hormone | 1 | No recurrence |
| Yao Yong et al. | 10 | Male |
Eye redness Eye pain Photophobia |
Proteinuria Glycosuria |
Acute interstitial nephritis | Bilateral | After | Hormone | 15 | Recurrence of uveitis or Chronic uveitis |
| 15 | Male |
Eye pain Photophobia |
Proteinuria Glycosuria |
Acute interstitial nephritis | Bilateral | After | Hormone | 19 | Recurrence of uveitis or Chronic uveitis | |
| Hu Junhua et al. | 17 | Male |
Eye redness Blurred vision Photophobia |
Proteinuria Glycosuria Microscopic hematuria |
Interstitial nephritis | Unilateral | After | Hormone | 2 | No recurrence |
| 30 | Female | Eye redness |
Proteinuria Glycosuria Microscopic hematuria |
Acute interstitial nephritis | Bilateral | Concurrent | Hormone | 2 | No recurrence | |
| Chow,KM.et al. | 30 | Male | NA | Proteinuria | Acute interstitial nephritis | Bilateral | After | Hormone | NA | Recurrence of uveitis or Chronic uveitis |
| Liu Wenhui et al. | 36 | Male |
Eye redness Eye pain |
Proteinuria | Acute interstitial nephritis | Unilateral | After | Hormone | 5 | No recurrence |
| Zhou Letian et al. | 15 | Female |
Eye redness Blurred vision |
Proteinuria Glycosuria |
Interstitial nephritis | Unilateral | Concurrent | Hormone | 2 | No recurrence |
| Tang Lin et al. | 60 | Female |
Eye redness Eye pain Blurred vision |
Proteinuria | Acute interstitial nephritis | Bilateral | Concurrent | Hormone | 4 | No recurrence |
| Hong Fuyuan et al. | 52 | Female |
Eye redness Blurred vision |
Proteinuria Glycosuria |
Acute interstitial nephritis | Bilateral | Before | Hormone | 2 | No recurrence |
| Zhou, Letian et al. | 15 | Female |
Eye redness Photophobia |
Proteinuria Glycosuria |
Acute interstitial nephritis | unilateral | Concurrent | Hormone | 60 | No recurrence |
| Yang Xiaochun et al. | 52 | Female |
Eye redness Blurred vision |
Proteinuria Glycosuria |
Acute interstitial nephritis | Bilateral | Concurrent |
Hormone Immunosuppressant |
18 | Recurrence of kidney disease or CKD |
| Ma Dong et al. | 40 | Female | Blurred vision | NA | NA | Unilateral | Concurrent | Hormone | 0.5 | No recurrence |
| Sun Dezhen et al. | 41 | Female |
Eye redness Eye pain Photophobia |
Proteinuria Glycosuria |
NA | Bilateral | Before | Hormone | 36 | No recurrence |
| 37 | Female |
Eye redness Eye pain Photophobia |
Proteinuria Glycosuria |
Interstitial nephritis | Bilateral | Before | Hormone | 36 | No recurrence | |
| 65 | Female |
Eye redness Eye pain Photophobia |
Proteinuria Glycosuria |
Interstitial nephritis | Unilateral | Concurrent | Hormone | 24 | No recurrence | |
| Zhou Limei et al. | 44 | Male |
Eye redness Blurred vision |
Proteinuria | Interstitial nephritis | Bilateral | After | Hormone | 0.5 | No recurrence |
| Lei Wenhui et al. | 60 | Male | NA |
Proteinuria Glycosuria Microscopic hematuria |
Interstitial nephritis | Bilateral | Before | Hormone | 6 | No recurrence |
| Chen Min et al. | 42 | Female |
Eye redness Eye pain Blurred vision Photophobia |
Proteinuria Glycosuria |
Interstitial nephritis | Bilateral | Concurrent | Hormone | 0.5 | No recurrence |
| Zhang Shu et al. | 56 | Female |
Eye redness Photophobia |
Proteinuria | Acute interstitial nephritis | Bilateral | Before | Hormone | 7 | No recurrence |
| Ren Wei et al. | 16 | Male |
Eye pain Blurred vision |
Proteinuria | Interstitial nephritis | Bilateral | Concurrent | Hormone | 6 | Recurrence of uveitis or Chronic uveitis |
| Zhou Yucong et al. | 48 | Male |
Eye redness Eye pain Blurred vision |
Proteinuria Glycosuria |
Acute interstitial nephritis | Unilateral | Concurrent | Hormone | 2 | No recurrence |
| Lei Lei et al. | 55 | Female |
Eye redness Blurred vision |
Proteinuria | NA | Bilateral | After | Hormone | 24 | No recurrence |
| Wu Yao et al. | 11 | Male |
Eye redness Eye pain Blurred vision |
Glycosuria | Acute interstitial nephritis | Unilateral | After | Hormone | 24 | No recurrence |
| Guo Jingxiao et al. | 11 | Female | Eye pain |
Proteinuria Glycosuria |
Interstitial nephritis | Unilateral | After | Hormone | 2 | No recurrence |
| Kong Weiying et al. | 37 | Female | None |
Proteinuria Glycosuria |
Interstitial nephritis | Unilateral | After | Hormone | 0.75 | No recurrence |
| Wang Li et al. | 63 | Female | Eye redness |
Proteinuria Glycosuria Microscopic hematuria |
Interstitial nephritis | Bilateral | After renal involvement |
Hormone Traditional Chinese Medicine |
36 |
Recurrence of kidney disease or CKD Recurrence of uveitis or Chronic uveitis |
| Liu Jianliang et al. | 38 | Female |
Eye redness Eye pain Blurred vision |
Proteinuria Glycosuria |
Interstitial nephritis | Unilateral | After | Hormone | 1 | No recurrence |
| Zhao | 37 | Male | NA |
Proteinuria Glycosuria |
Interstitial nephritis | Bilateral | NA | Hormone | 24 |
Recurrence of kidney disease or CKD Recurrence of uveitis or Chronic uveitis |
| Xu Xialian et al. | 64 | Male | NA | None | Interstitial nephritis | NA | Before | Hormone | 6 | No recurrence |
| Zhang Kang et al. | 49 | Female |
Eye redness Eye pain |
Proteinuria Glycosuria |
Acute interstitial nephritis | unilateral | Concurrent |
Hormone Traditional Chinese Medicine |
24 | No recurrence |
| Yang Menglu et al. | Mean = 41 | 13 males and 19 females |
28 Eye pain 28 Blurred vision and 16 Photophobia |
NA | Interstitial nephritis | 2 unilateral and 30 Bilateral | 2 Before; 10 Concurrent and 20 After |
13 Hormone therapy alone and 19 Hormone + Immunosuppressant |
Mean = 37.8 | 18 Recurrence of uveitis or Chronic uveitis and 14 Recurrence of kidney disease or CKD |
Demographic information
A total of 71 cases were included in this article, of which 70 were from publications and 1 was from our hospital. 38 were from Beijing, but there was no evidence of regional ethnic distribution differences in TINU syndrome in China. In 39 cases mentioning the triggering factors of TINU syndrome was: unclear (69%), infections (28%), drugs (3%). 11 patients had infectious triggers before acute kidney injury, of which nine patients had upper respiratory tract infection, one had cholecystitis, and one had urinary tract infection. Only one patients used herbal medicine which was not elaborated in the case report for 2 weeks before his serum creatinine found to be elevated. Twenty-eight patients were male and 43 were female, with a male-to-female ratio of 1:1.5 (Table 2). For those aged ≥ 18 years, 67% were female, while males accounted for 59% of those under 18 years of age. TINU syndrome tends to occur in young men and middle-aged women in China.
Table 2.
Data of patients with TINU syndrome
| Characteristics | N | Values |
|---|---|---|
| Gender (male:female), n (%) | 71 | 28 (39):43 (61) |
| Age (years), median (IQR) | 71 | 42 (18–55) |
| Pathogeny (s), n (%) | 39 | |
| None | 27 (69) | |
| Infection | 11 (28) | |
| Drugs | 1 (3) | |
| Prodromal symptoms, n (%) | 71 | |
| Fever | 48 (68) | |
| Fatigue | 54 (76) | |
| Gastrointestinal symptoms | 40 (56) | |
| Increased nocturia | 11 (15) | |
| Eye symptoms, n (%) | 71 | |
| Redness | 33 | 26 (79) |
| Eye pain | 44 (62) | |
| Blurred vision | 43 (61) | |
| Photophobia | 27 (38) | |
| Uveitis, n (%) | 71 | |
| Anterior uveitis | 39 (55) | |
| Intermediate uveitis | 9 (13) | |
| Panuveitis | 7 (10) | |
| None | 16 (23) | |
| Uveitis (unilateral:bilateral), n (%) | 71 | 15 (21):53 (75) |
| Uveitis onset, n (%) | 70 | |
| Before renal involvement | 11 (16) | |
| Concurrent renal involvement | 21 (30) | |
| After renal involvement | 38 (54) | |
| Urinary abnormalities, n (%) | ||
| Proteinuria | 37 | 34 (92) |
| Glycosuria | 33 | 27 (82) |
| Microscopic hematuria | 32 | 7 (22) |
| Treatment | 71 | |
| Glucocorticoid alone | 47 (66) | |
| Glucocorticoid + immunosuppressant | 22 (31) | |
| Other | 2 (3) | |
| The data of follow-up > 6 months | 51 | |
| Months, median (IQR) | 24 (12–36) | |
| Recurrence of uveitis or Chronic uveitis, n (%) | 24 (47) | |
| Recurrence of kidney disease or Chronic kidney disease, n (%) | 42 | 20 (48) |
Clinical and laboratory data
The patients had various symptoms, including fatigue, fever, and gastrointestinal manifestations such as nausea, vomiting, loss of appetite, and weight loss. Eleven patients urinate at least 2 times per night (Table 2). We conducted subgroup analysis according to sex and age. There were no statistically significant differences in clinical symptoms between men and women or in terms of age (Tables 3, 4).
Table 3.
Comparison between male subgroup and female subgroup
| Characteristics | Male | Female | P value |
|---|---|---|---|
| n | 28 | 43 | |
| Age (years), median (IQR) | 29 (16–47) | 50 (38–56) | < 0.05 |
| Pathogeny (s), n (%) | 15 | 24 | |
| Unknown | 11 (73) | 16 (67) | 0.734 |
| Infection | 3 (20) | 8 (33) | 0.477 |
| Drugs | 1 (7) | 0 (0) | 0.385 |
| Prodromal symptoms, n (%) | 15 | 24 | |
| Fever | 11 (73) | 10 (42) | 0.098 |
| Fatigue | 12 (80) | 12 (50) | 0.093 |
| Gastrointestinal symptoms | 7 (47) | 11 (46) | 1 |
| Increased nocturia | 2 (13) | 7 (29) | 0.437 |
| Weight loss | 3 (20) | 3 (13) | 0.658 |
| Eye symptoms, n (%) | 11 | 22 | |
| Redness | 8 (73) | 18 (82) | 0.661 |
| Eye pain | 6 (55) | 10 (46) | 0.721 |
| Blurred vision | 5 (46) | 10 (46) | 1 |
| Photophobia | 3 (27) | 8 (36) | 0.709 |
| Uveitis, n (%) | 15 | 24 | |
| Anterior uveitis | 6 (40) | 14 (58.3) | 0.333 |
| Intermediate uveitis | 1 (7) | 1 (4.2) | 1 |
| Panuveitis | 0 (0) | 1 (4.2) | 1 |
| Unknown | 8 (53) | 8 (33.3) | 0.318 |
| Uveitis (unilateral:bilateral), n (%) | 5 (18):22 (79) | 10 (23):31 (72) | |
| Uveitis onset, n (%) | 27 | 43 | |
| Before renal involvement | 3 (11) | 8 (19) | 0.616 |
| Concurrent renal involvement | 7 (26) | 14 (33) | 0.556 |
| After renal involvement | 17 (63) | 21 (49) | 0.248 |
| Urinary abnormalities, n (%) | |||
| Proteinuria | 13 (87) | 21 (96) | 0.554 |
| Glycosuria | 10 (71) | 17 (90) | 0.363 |
| Microscopic hematuria | 5 (39) | 2 (11) | 0.091 |
| Treatment | 28 | 43 | |
| Glucocorticoid alone | 21 (75) | 26 (61) | 0.206 |
| Glucocorticoid + immunosuppressant | 7 (25) | 15 (35) | 0.379 |
| The data of follow-up > 6 months | 21 | 30 | |
| Months, median (IQR) | 24 (9–24) | 30 (12–51) | |
| Recurrence of uveitis or chronic uveitis, n (%) | 10 (48) | 14 (47) | 0.947 |
| Recurrence of kidney disease or chronic kidney disease, n (%) | 7 (39) | 13 (54) | 0.327 |
Table 4.
Comparison between adolescent subgroup and adult subgroup
| Characteristics | < 18 years | ≥ 18 years | P value |
|---|---|---|---|
| n | 17 | 54 | |
| Gender (male:female), n (%) | 10(59):7(41) | 18(33):36(67) | |
| Pathogeny (s), n (%) | 9 | 30 | |
| Unknown | 5 (56) | 22 (73) | 0.416 |
| Infection | 4 (44) | 7 (23) | 0.238 |
| Drugs | 0 (0) | 1 (3.3) | 1 |
| Prodromal symptoms, n (%) | 9 | 30 | |
| Fever | 5 (56) | 16 (53) | 1 |
| Fatigue | 8 (89) | 16 (53) | 0.115 |
| Gastrointestinal symptoms | 2 (22) | 16 (53) | 0.139 |
| Increased nocturia | 2 (22) | 7 (23) | 1 |
| Weight loss | 3 (33) | 3 (10) | 0.123 |
| Eye symptoms, n (%) | 8 | 25 | |
| Redness | 5 (63) | 21 (84) | 0.32 |
| Eye pain | 5 (63) | 11 (44) | 0.438 |
| Blurred vision | 4 (50) | 11 (44) | 1 |
| Photophobia | 4 (50) | 7 (28) | 0.391 |
| Uveitis, n (%) | 9 | 30 | |
| Anterior uveitis | 6 (67) | 14 (47) | 0.451 |
| Intermediate uveitis | 0 (0) | 2 (7) | 1 |
| Panuveitis | 0 (0) | 1 (3) | 1 |
| Unknown | 3 (33) | 13 (43) | 0.711 |
| Uveitis (unilateral:bilateral), n (%) | 5 (29):11 (65) | 10 (19):42 (78) | |
| Uveitis onset, n (%) | 17 | 53 | |
| Before renal involvement | 1 (6) | 10 (19) | 0.37 |
| Concurrent renal involvement | 3 (18) | 18 (34) | 0.201 |
| After renal involvement | 13 (77) | 25 (47) | 0.035 |
| Urinary abnormalities, n (%) | |||
| Proteinuria | 7 (87) | 27 (93) | 0.53 |
| Glycosuria | 7 (88) | 20 (80) | 1 |
| Microscopic hematuria | 1 (13) | 6 (25) | 0.646 |
| Treatment | 17 | 54 | |
| Glucocorticoid alone | 17 (100) | 30 (56) | < 0.01 |
| Glucocorticoid + immunosuppressant | 0 (0) | 22 (41) | < 0.01 |
| The data of follow-up > 6 months | 14 | 37 | |
| Months, median (IQR) | 24 (14.25–36) | 24 (12–48) | |
| Recurrence of uveitis or chronic uveitis, n (%) | 6 (42) | 18 (49) | 0.712 |
| Recurrence of kidney disease or chronic kidney disease, n (%) | 2 (18) | 18 (58) | 0.023 |
Among the 35 patients with renal function data, all showed non-oliguric acute renal injury; average values of serum creatinine, urea nitrogen and 24-h urine protein were was 317.8 μmol/L, 13.7 mmol/L, and 1.14 ± 0.7 g/24 h, respectively. Increased ESR was reported in 26/27 patients. Proteinuria was present in 34/37 (92%) patients and Urinary glucose abnormalities with normal blood glucose levels developed in 27/33 (82%) patients. The renal biopsy results in 67 patients were consistent with acute tubulointerstitial nephritis (ATIN). Four patients who did not undergo renal biopsy had abnormal renal function, abnormal urine test results, and systemic disease lasting for 2 weeks.
Uveitis
In the 71 cases, anterior uveitis was found in 55%, intermediate uveitis in 13%, and panuveitis 10%. Bilateral uveitis was present in 53/71 cases. The patients exhibited various ocular symptoms, including redness, eye pain, blurred vision, and photophobia. The relationship between uveitis and ATIN remains unclear; 54% of the 70 patients had uveitis after ATIN, 30% had uveitis at the same time as ATIN, and 16% had uveitis before ATIN (Table 2). Subgroup analysis found that uveitis occurred after ATIN in 77% of those aged under 18 years, compared to 47% of adult patients (P < 0.05). There were not statistically significant differences in ocular symptoms between men and women (Tables 3, 4).
Treatments and outcomes
Glucocorticoid therapy was the first-line treatment for TINU in China. 47 patients were treated with glucocorticoids alone and 22 cases were treated with glucocorticoids combined with immunosuppressive agents, including mycophenolate mofetil (n = 10), cyclophosphamide (n = 8), azathioprine (n = 3) and unspecified (n = 1). All patients under 18 years received glucocorticoid therapy alone.
The outcomes of 51 patients with a follow-up period of > 6 months were analyzed. The median follow-up period was 24 months (IQR 12–36). Recurrent uveitis means that the eye symptoms of patients disappear after treatment, and then the eye symptoms reappear 3 months later. Recurrent uveitis or the course over 3 months defined chronic uveitis was present in 24/51 (47%) patients, comprising 10 males and 14 females, 6 aged less than 18 years and 18 adults. Forty-two out of 51 patients with a follow-up period of > 6 months for whom there was follow-up data on renal function, of which twenty patients experienced kidney disease recurrence or progression to CKD, including seven males and thirteen females. The incidence was higher in adults (58%) than in young adults (18%) (P < 0.05).
Discussion
To the best of our knowledge, approximately 600 TINU cases have been reported worldwide. TINU syndrome has highly variable clinical features and a lack of awareness and recognition probably contributes to an underestimation of its incidence rate. The etiology and pathogenesis of TINU syndrome may be related to infection, chemicals, drugs (antibiotics and non-steroidal anti-inflammatory drugs), and genetic factors, such as human leukocyte antigen (HLA) [45]. There has been two systematic reviews about TINU syndrome before, involving western populations [46, 47]. Currently, there is no systematic review of TINU syndrome in China.
In the sample described here, the proportion of men and women with TINU syndrome in China was approximately 1:1.5, which is inconsistent with published articles [48, 49]. Nevertheless, this gender effect seems to be weakening in recent years. Regusci et al. reported that the male-to-female ratio was approximately 1:1.9, and in Japan, the proportion was approximately 1:2.3 [48, 50–52]. TINU syndrome is speculated to be a multisystem autoimmune disorder. The reason why women have an advantage may be related to their high levels of estrogen and immunoglobulin. Estrogen plays an important role in regulating innate immunity and is an enhancer of humoral immunity [53, 54]. And high levels of immunoglobulin will increase immune reactivity [55].
TINU syndrome has a variety of characteristic clinical manifestations, including fatigue, nausea and vomiting, fever, weight loss, and some patients have increased nocturia. Laboratory examination revealed non-oliguric acute renal injury with elevated serum creatinine and urea nitrogen levels. Urinalysis revealed mild-to-moderate proteinuria, urinary glucose abnormalities with normal blood glucose levels and renal tubular dysfunction. Renal injury and uveitis usually occur at different times, and uveitis can be diagnosed 2 months before or 12 months after interstitial nephritis (TIN) [51]. In our study, uveitis was diagnosed after TIN in 54% of cases, and most were anterior (55%) and bilateral (75%). This was very similar to the results of the systematic review in which reported that anterior accounts for 65% and bilateral accounts for 88% [46]. Consistently, Mandeville et al. reviewed the world's medical literature on TINU syndrome in 2001 and found bilateral at presentation in 77% of cases [51]. Uveitis in children was usually diagnosed after TIN (P < 0.05). These findings are consistent with data reported by Regusci et al. [46]. This is because children usually seek medical attention for nonspecific symptoms, and during this period, renal dysfunction is detected and diagnosed as TIN through renal biopsy. Subsequently, eye examinations were performed. In addition, the description of eye discomfort in children may be unclear and not been taken seriously by parents.
There are no standardized guidelines for the treatment of patients with TINU syndrome due to the lack of evidence; therefore, a prospective randomized controlled trial of glucocorticoids, placebos, and glucocorticoids combined with immunosuppressants is warranted. Glucocorticoid therapy is the first-line of treatment in China. Interestingly, subgroup analysis revealed that all children were treated with glucocorticoids alone, and the outcomes were good. It is recognized that the immune response of adolescents is not fully developed; therefore, the renal immune response damage caused by TINU syndrome is minimal in adolescents. This can also be used to explain the use of glucocorticoids alone can benefit children.
Previous studies have reported that TINU syndrome has a good renal outcome; however, our data show that approximately the half patient will progress to CKD [56, 57]. Among the forty-two patients with follow-up data on renal function, twenty individual experienced recurrence or progression to CKD. A study by Su et al. found that 63% of patients had estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2 after 3 years of follow-up [58]. The difference between the data in the previous literature and the newer results may be related to the length of follow-up. Our subgroup analysis showed that children underwent renal recovery more frequently than adults (P < 0.05). This is consistent with the results of previous studies [48–50]. A study suggest age at onset was associated with an increased risk of CKD development [46].
Our systematic review analyzed patients with TINU syndrome reported in the Chinese literature and we analyzed the renal and ocular outcomes of TINU syndrome, increasing people's understanding of TINU syndrome. Some of the included publications did not fully describe the clinical data, which may have led to bias in the results. The duration of follow-up varied, which may have affected the reported outcomes. This systematic review is a preliminary exploration of the TINU syndrome in China and needs to be further confirmed in a well-designed prospective study.
Conclusion
We present a case of acute renal tubulointerstitial nephritis and bilateral uveitis, with a diagnosis of TINU syndrome. TINU syndrome affects predominantly adults in China. The etiology, clinical manifestations, and duration of uveitis and kidney injury are unclear, which increases the risk of missed diagnosis of the disease. When a patient's examination indicates acute renal injury with tubule dysfunction and excludes other diseases, it is necessary to consider the possibility of TINU syndrome. Simultaneously, doctors must carefully inquire about the eye symptoms and conduct an ophthalmic examination. Once confirmed, appropriate treatment strategies should be selected based on the patient's condition. Glucocorticoid therapy is currently the first-line treatment in China.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
This research was supported by The First Hospital of Jiaxing, First Affiliated Hospital of Jiaxing University.
Author contributions
Conception and design: JS, HW. Data collection: JS, SX. Analysis and interpretation of data: JS, SX. Writing, review, and/or revision of the manuscript: JS, SX, JC. Study supervision: JS, HW.
Funding
This project is supported in kind by the Jiaxing Key Discipline of Medcine-Nephrology (2023-ZC-011).
Data availability
All data relevant to the study are included in the article or uploaded as supplementary information.
Declarations
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Jiazhen Shi and Shaoyan Xu contributed equally to this work.
Contributor Information
Jianxiang Chen, Email: chenjianxiang@163.com.
Henglan Wu, Email: whl525487@sina.com.
References
- 1.Dobrin RS, Vernier RL, Fish AL. Acute eosinophilic interstitial nephritis and renal failure with bone marrow-lymph node granulomas and anterior uveitis. A new syndrome. Am J Med. 1975;59:325–333. doi: 10.1016/0002-9343(75)90390-3. [DOI] [PubMed] [Google Scholar]
- 2.Amaro D, Carreno E, Steeples LR, et al. Tubulointerstitial nephritis and uveitis (TINU) syndrome: a review. Br J Ophthalmol. 2020;104:742–747. doi: 10.1136/bjophthalmol-2019-314926. [DOI] [PubMed] [Google Scholar]
- 3.Vanhaesebrouck P, Carton D, De Bel C, et al. Acute tubulo-interstitial nephritis and uveitis syndrome (TINU syndrome) Nephron. 1985;40:418–422. doi: 10.1159/000183511. [DOI] [PubMed] [Google Scholar]
- 4.Conz PA, Milan M, Bragantini L, et al. TINU syndrome associated with reduced complement levels. Nephron. 2001;89:340–341. doi: 10.1159/000046096. [DOI] [PubMed] [Google Scholar]
- 5.Abed L, Merouani A, Haddad E, et al. Presence of autoantibodies against tubular and uveal cells in a patient with tubulointerstitial nephritis and uveitis (TINU) syndrome. Nephrol Dialysis Transplant. 2008;23:1452–1455. doi: 10.1093/ndt/gfm890. [DOI] [PubMed] [Google Scholar]
- 6.Li C, Su T, Chu R, et al. Tubulointerstitial nephritis with uveitis in Chinese adults. Clin J Am Soc Nephrol. 2014;9:21–28. doi: 10.2215/CJN.02540313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kobayashi Y, Honda M, Yoshikawa N, et al. Immunohistological study in sixteen children with acute tubulointerstitial nephritis. Clin Nephrol. 1998;50:14–20. [PubMed] [Google Scholar]
- 8.Amaro D, Carreno E, Steeples LR, Oliveira-Ramos F, Marques-Neves C, Leal I. Tubulointerstitial nephritis and uveitis (TINU) syndrome: a review. Br J Ophthalmol. 2020;104:742–747. doi: 10.1136/bjophthalmol-2019-314926. [DOI] [PubMed] [Google Scholar]
- 9.Clive DM, Vanguri VK. The syndrome of tubulointerstitial nephritis with uveitis (TINU) Am J Kidney Dis. 2018;72:118–128. doi: 10.1053/j.ajkd.2017.11.013. [DOI] [PubMed] [Google Scholar]
- 10.Wu H, Xu Z. Idiopathic tubulointerstitial nephritis uveitis syndrome: a case report. Chin J Pract Internal Med. 1995;25:363. [Google Scholar]
- 11.Liu G, Ma H. Hyperthyroidism is a new feature of tubulointerstitial nephritis and uveitis syndrome. Int J Pediatr. 2000;20:165–166. [Google Scholar]
- 12.Zhao M, Liu Y, Zou W, et al. The 10th case of fever, renal insufficiency and red eye disease. Natl Med J China. 2001;81:1216. [Google Scholar]
- 13.Ma Y. A case report of TINU syndrome. Hebei Med J. 2003;25:673. [Google Scholar]
- 14.Li Y. A case report of TINU syndrome. J Hangzhou Teachers Coll. 2004;25:193–193. [Google Scholar]
- 15.Zhang M, Qian J. A case of tubulointerstitial nephritis uveitis syndrome. Chin J Nephrol. 2004;20:272. [Google Scholar]
- 16.Cheng H, Yang X. A case of atypical renal tubulointerstitial nephritis retinitis syndrome. J Nongken Med. 2006;28:319–320. [Google Scholar]
- 17.Zhang W, Tao X, Cheng J, et al. A case of TINU syndrome and literature review. Chin J Integrat Tradit Western Nephrol. 2006;7:411–412. [Google Scholar]
- 18.Hu J, Wang Y. Two cases of TINU syndrome and literature review. J Pract Med. 2007;23:3075–3076. [Google Scholar]
- 19.Yao Y, Yang J. Two cases of tubular interstitial nephritis uveitis syndrome in children. Chin J Pediatr. 2007;45:310–311. [PubMed] [Google Scholar]
- 20.Chow KM, Lai FM, Szeto CC, et al. Quiz page March 2008: fever, anorexia, and renal failure. TINU syndrome. Am J Kidney Dis. 2008;51:A39–40. doi: 10.1053/j.ajkd.2007.07.030. [DOI] [PubMed] [Google Scholar]
- 21.Liu W, Shi G. A case report of TINU syndrome. Chin J Misdiagnostics. 2008;8:1259. [Google Scholar]
- 22.Zhou L, Li Y, Liu Y, et al. A case report of tubulointerstitial nephritis uveitis syndrome. Chin J Nephrol. 2008;24:857. [Google Scholar]
- 23.Hong F, Liu J, Xu P, et al. A case report of TINU syndrome. J Fujian Med Univ. 2010;44:70. [Google Scholar]
- 24.Tang L, Zhang X, Zhao Z, et al. A case of elderly tubulointerstitial nephritis uveitis syndrome. Chin J Nephrol. 2010;26:486. [Google Scholar]
- 25.Zhou L, Li Y, Liu F, et al. Excellent outcome of tubulointerstitial nephritis and uveitis: case report and five-year follow-up. Renal Fail. 2012;34:934–936. doi: 10.3109/0886022X.2012.690924. [DOI] [PubMed] [Google Scholar]
- 26.Ma D, Guo C, Liu L. A case report of tubulointerstitial nephritis uveitis syndrome (TINU syndrome) China J Chin Ophthalmol. 2012;22:214. [Google Scholar]
- 27.Yang X, Wu R, Li G, et al. A case of TINU syndrome with thyroiditis and dynamic evolution of HLA-B27. Chin J Nephrol. 2012;28:664. [Google Scholar]
- 28.Sun D, Zhao J. Misdiagnosis of three cases of tubulointerstitial nephritis and uveitis syndrome. Chin J Clin Electron Ed. 2013;7:7314–7315. [Google Scholar]
- 29.Zhou M, Bao X, Jin L (2014) A case report of TINU syndrome. In 2014 Zhejiang Provincial renal disease academic annual conference, Hangzhou, Zhejiang, China, p 313
- 30.Lei W-H, Xin J, Yu X-P, et al. Tubulointerstitial nephritis and uveitis syndrome in an elderly man case report and literature review. Medicine. 2015;94:25. doi: 10.1097/MD.0000000000002101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chen M, Zhao J, Tang F, et al. A case report of tubulointerstitial nephritis uveitis syndrome. Chin J Nephrol. 2015;31:60–61. [Google Scholar]
- 32.Zhang S, Lan P, Xie L, et al. A case report of tubulointerstitial nephritis uveitis syndrome. Shanxi Med J. 2017;46:242–243. [Google Scholar]
- 33.Zhou Y, Yu L, Gou R, et al. A case of male tubulointerstitial nephritis uveitis syndrome. Clin Med. 2017;37:127–128. [Google Scholar]
- 34.Ren W, Li W, Zhuo L. A case of tubulointerstitial nephritis uveitis syndrome and literature review. Chin J Difficult Compl Cases. 2017;16:947–948. [Google Scholar]
- 35.Guo J, Zhang D. A child with tubulointerstitial nephritis and uveitis syndrome. Chin J Difficult Compl Cases. 2018;17:1387–1388. [Google Scholar]
- 36.Kong W, Hu Z. A case of TINU syndrome with acute abdominal pain as the initial symptom. J Clin Nephrol. 2018;18:717–718. [Google Scholar]
- 37.Lei L, Li F, Wang D, et al. A case report of tubulointerstitial nephritis uveitis syndrome. Chongqing Med. 2018;47:716–717. [Google Scholar]
- 38.Wu Y, Liu X. Nursing care of a child with tubular interstitial nephritis and uveitis syndrome: a one case report. J Nurs Sci. 2018;33:31–32. [Google Scholar]
- 39.Yang M, Chi Y, Guo C, et al. Clinical profile, ultra-wide-field fluorescence angiography findings, and long-term prognosis of uveitis in tubulointerstitial nephritis and uveitis syndrome at one tertiary medical institute in China. Ocul Immunol Inflamm. 2019;27:371–379. doi: 10.1080/09273948.2017.1394469. [DOI] [PubMed] [Google Scholar]
- 40.Liu J, Li X. A case of TINU syndrome and literature review. J Med Theory Pract. 2019;32:103–104. [Google Scholar]
- 41.Wang L, Zhang G, Rao X. Treatment of acute renal injury in tubulointerstitial nephritis uveitis syndrome with traditional Chinese medicine: a case report and literature review. Chin J Integrat Tradit Western Nephrol. 2019;20:77–79. [Google Scholar]
- 42.Zhao Y, Huang J, Su T, et al. Acute kidney injury relevant to tubulointerstitial nephritis with late-onset uveitis superimposed by thrombotic microangiopathy: a case report and review of the literature. Kidney Dis (Basel, Switzerland) 2020;6:414–421. doi: 10.1159/000507668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Xu X, Jin S, Lin P, et al. A case of TINU syndrome. Shanghai Med J. 2020;43:621–624. [Google Scholar]
- 44.Zhang K, Cai L, Li Q, et al. Yue Pi Tang for the treatment of a case of tubulointerstitial nephritis uveitis syndrome. J Emerg Tradit Chin Med. 2022;31:1501–1504. [Google Scholar]
- 45.Reddy AK, Hwang YS, Mandelcorn ED, et al. HLA-DR, DQ class II DNA typing in pediatric panuveitis and tubulointerstitial nephritis and uveitis. Am J Ophthalmol. 2014;157(678–686):e671–672. doi: 10.1016/j.ajo.2013.12.006. [DOI] [PubMed] [Google Scholar]
- 46.Regusci A, Lava SAG, Milani GP, Bianchetti MG, Simonetti GD, Vanoni F. Tubulointerstitial nephritis and uveitis syndrome: a systematic review. Nephrol Dialysis Transplant. 2022;37:876–886. doi: 10.1093/ndt/gfab030. [DOI] [PubMed] [Google Scholar]
- 47.Southgate G, Clarke P, Harmer MJ. Renal outcomes in tubulointerstitial nephritis and uveitis (TINU) syndrome: a systematic review and meta-analysis. J Nephrol. 2023;36:507–519. doi: 10.1007/s40620-022-01478-8. [DOI] [PubMed] [Google Scholar]
- 48.Regusci A, Lava SAG, Milani GP, et al. Tubulointerstitial nephritis and uveitis syndrome: a systematic review. Nephrol Dialysis Transplant. 2022;37:876–886. doi: 10.1093/ndt/gfab030. [DOI] [PubMed] [Google Scholar]
- 49.Sinnamon KT, Courtney AE, Harron C, et al. Tubulointerstitial nephritis and uveitis (TINU) syndrome: epidemiology, diagnosis and management. NDT Plus. 2008;1:112–116. doi: 10.1093/ndtplus/sfn006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mackensen F, Smith JR, Rosenbaum JT. Enhanced recognition, treatment, and prognosis of tubulointerstitial nephritis and uveitis syndrome. Ophthalmology. 2007;114:995–999. doi: 10.1016/j.ophtha.2007.01.002. [DOI] [PubMed] [Google Scholar]
- 51.Mandeville JT, Levinson RD, Holland GN. The tubulointerstitial nephritis and uveitis syndrome. Surv Ophthalmol. 2001;46:195–208. doi: 10.1016/S0039-6257(01)00261-2. [DOI] [PubMed] [Google Scholar]
- 52.Zandman-Goddard G, Peeva E, Shoenfeld Y. Gender and autoimmunity. Autoimmun Rev. 2007;6:366–372. doi: 10.1016/j.autrev.2006.10.001. [DOI] [PubMed] [Google Scholar]
- 53.Moulton VR. Sex hormones in acquired immunity and autoimmune disease. Front Immunol. 2018;9:2279. doi: 10.3389/fimmu.2018.02279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Trenti A, Tedesco S, Boscaro C, Trevisi L, Bolego C, Cignarella A. Estrogen, angiogenesis, immunity and cell metabolism: solving the puzzle. Int J Mol Sci. 2018;19:25. doi: 10.3390/ijms19030859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Virdi V, Juarez P, Boudolf V, Depicker A. Recombinant IgA production for mucosal passive immunization, advancing beyond the hurdles. Cell Mol Life Sci. 2016;73:535–545. doi: 10.1007/s00018-015-2074-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Gorrono-Echebarria MB. The tubulointerstitial nephritis and uveitis syndrome. Surv Ophthalmol. 2002;47:293. doi: 10.1016/S0039-6257(02)00282-5. [DOI] [PubMed] [Google Scholar]
- 57.Goda C, Kotake S, Ichiishi A, et al. Clinical features in tubulointerstitial nephritis and uveitis (TINU) syndrome. Am J Ophthalmol. 2005;140:637–641. doi: 10.1016/j.ajo.2005.04.019. [DOI] [PubMed] [Google Scholar]
- 58.Su T, Gu Y, Sun P, et al. Etiology and renal outcomes of acute tubulointerstitial nephritis: a single-center prospective cohort study in China. Nephrol Dial Transplant. 2018;33:1180–1188. doi: 10.1093/ndt/gfx247. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data relevant to the study are included in the article or uploaded as supplementary information.