Abstract
Chronic rhinosinusitis with nasal polyps (CRSwNP) is an inflammatory condition of the nasal and paranasal tissues, characterized by the presence of bilateral nasal polyps. While etiology and pathogenetic mechanisms are heterogeneous and complex, in most patients, disease is mediated predominantly through type 2 inflammatory processes. Clinical management is challenging, and a multidisciplinary approach is preferred. Principal treatment approaches are the use of local/systemic corticosteroids and sinonasal surgery, although outcomes can be unsatisfactory. Recent availability of biological therapies targeting underlying inflammatory processes can offer effective treatment options in uncontrolled disease. Specialist guidelines greatly assist clinical decision-making, although as these are chiefly written from a global/international perspective, they may not wholly accommodate disease patterns and clinical practice at a regional level. An expert panel of specialists from Latin America was convened to develop regional guidance on the management of CRSwNP through a consensus approach. The present article presents the chief observations and recommendations which can provide guidance for clinicians in the Latin American region.
Keywords: Biologics, Chronic rhinosinusitis with nasal polyposis, CRSwNP, Type 2 inflammation
Introduction
Chronic rhinosinusitis (CRS) is common, affecting more than 10% of the general population worldwide, and globally, poses a substantial clinical burden.1,2 When evaluated using standardized criteria, the global prevalence of symptomatic CRS ranges from 5.5% in Brazil to as high as 28% in Iran.1,2 These values may overestimate the prevalence; others report that the true prevalence may be less than 5%.3 Two broad CRS phenotypes exist, characterized on the basis of the presence or absence of nasal polyps; patients with chronic rhinosinusitis with nasal polyps (CRSwNP) account for up to 20% of all CRS.4
Clinical management of CRSwNP can be complex. Conventional treatment strategies include topical intranasal (INS) or oral corticosteroids (OCS)1,2 with surgical intervention in patients with persistent or uncontrolled disease.5,6 The availability of biologics now provides a far greater scope of therapeutic options for those patients not responding to conventional medical therapy. Clinical decision-making has been greatly assisted by the development of comprehensive evidence-based specialist guidelines, which continue to evolve and update in response to the latest evidence. These include the updated European Position Paper on Rhinosinusitis and Nasal Polyps (EPOS 2020),1,2 the International Consensus Statement on Allergy and Rhinology: Rhinosinusitis (ICAR-RS-2021),7 and consensus-based recommendations from the European Forum for Research and Education in Allergy and Airway Diseases (EUFOREA).8,9 Although some differences exist, chiefly related to different methodological approaches, these guidelines present broadly complementary guidance for CRSwNP management.10 While such guidelines provide valuable insight from an international/global perspective, they may not wholly account for differences in clinical disease patterns, available clinical resources and therapeutic options and strategies that exist at a regional level. In addition, there may be some uncertainty in how such guidelines would apply at a more local level. Development of regional guidance can inform an approach to CRSwNP management that accommodates any such regional differences, and can provide perspective on the validity of such guidance in the regional setting. At present such guidance is lacking for the Latin America (LATAM) region. Furthermore, our experience is that most CRSwNP cases are still managed principally by primary care physicians, who are unlikely to be aware of evidence-based guidelines such as EPOS or EUFOREA, and so guidance tailored towards our colleagues within primary care can have value. With this aim, an expert panel was convened to evaluate the utility of existing guidelines to clinical practice within the region, identify gaps, and develop guidance for the management of CRSwNP in adults that may better reflect physician experience and patient needs within LATAM.
Methodology
A multidisciplinary expert panel was formed comprising specialists in respiratory, allergy, and otorhinolaryngology from Argentina, Brazil, Colombia, and Mexico. Panelists were selected based on their expertise in treating CRSwNP, publication record, and involvement in similar activities.
Two online meetings were held in October and November 2021. The first encompassed a general discussion on CRSwNP disease and management within the LATAM region. The second meeting focused on the international guidance for adult CRSwNP, with discussion on existing gaps or possible modifications to address the loco-regional needs. Based on these discussions and recommendations within EPOS 2020 guidelines,1,2 a series of discussion points on the principles of CRSwNP diagnosis and treatment were developed and reviewed between January 31 and February 14, 2022 using an interactive online platform (Within3; https://www.within3.com). Each panelist either agreed, disagreed, or proposed an alternate to each discussion point. The aim was not to generate formal recommendation statements or to report on consensus agreement values. The discussion points, feedback, and cross-panel dialog were collated and presented as a series of explanatory text and a narrative review of clinical management, which formed the basis of the present manuscript. All panelists reviewed and commented on manuscript drafts and this is considered as agreement on the proposals for management of CRSwNP. The results of this process are outlined in the following sections.
Disease definitions and diagnostic approach
Current guidelines provide clear diagnostic criteria for CRS, based upon clinical symptoms and evidence of sinonasal mucosal disease.1,2,7, 8, 9, 10 Diagnosis of rhinosinusitis requires the presence of ≥2 symptoms, one of which should be either nasal blockage/obstruction/congestion, or discharge, and either facial pain/pressure or headache and/or reduction or loss of smell.1,2 In addition, clinical confirmation of mucosal disease (presence of polyps, mucopurulent discharge and/or edema, or mucosal obstruction within the ostiomeatal complex or sinuses) is required via endoscopy or computed tomography (CT).1,2 The cardinal symptoms of CRS are similar to acute disease.1,2,11,12 In the panel's experience, overt facial pain with CRSwNP is uncommon in the LATAM region, whereas headache or reported symptoms of pressure or fullness may be more common. CRS diagnosis requires rhinosinusitis lasting for ≥12 weeks (without interruption),1,2 and assumes that the acute episode has been managed appropriately with nasal irrigation and INS. Although acute rhinosinusitis may be diagnosed on clinical grounds alone in clinical practice, the importance of endoscopic/CT confirmation of CRS must be emphasized, as findings will determine disease classification and subsequent treatment.
Diagnosis of CRSwNP requires the presence of bilateral, endoscopically visualized polyps in the middle meatus.1,2 Where polyps are absent, patients are considered to have chronic rhinosinusitis without nasal polyps (CRSsNP). If only unilateral disease is found on endoscopy/CT, then alternative diagnoses must be considered (eg, tumors, encephaloceles).1,2 Other forms of CRS (eg, allergic fungal rhinosinusitis), may present with focal unilateral disease.2,13,14
Etiology and pathogenesis of CRS
Pathophysiologic mechanisms in CRS involve an often complex interaction of host and environmental factors, each contributing to an individual disease profile.15 Such host-environment interactions are described as “internal milieu” and “exposome”, respectively. The exposome includes the environment and social aspects including personal or family medical history (eg, excess antibiotics use in childhood, atopy in close family members). Host-environmental interactions influence disease profiles in terms of both endotype and phenotype, which are particularly important when considering biological therapies. Endotypes broadly reflect the underlying biology and pathogenetic mechanisms (such as type-2 inflammation) whereas phenotypes reflect the patient's clinical profile (eg, comorbidities).16,17
Ultimately, the patient's profile results from both endotype and phenotype, and the subtle interplay between both. For example, while both CRSwNP and CRSsNP present with broadly similar primary clinical features, differences exist in the underlying pathophysiology. From a phenotypic perspective, although asthma is more prevalent in patients with CRS compared with the general population, co-existing asthma is strongly associated with CRSwNP (30–70% of patients) and asthma severity is greater in patients with coexisting CRSwNP.4,18, 19, 20 A wide range of comorbidities such as nonsteroidal anti-inflammatory drug (NSAID)-exacerbated respiratory disease (N-ERD) and aspirin-exacerbated respiratory disease (AERD), as well as atopic dermatitis, allergic rhinitis, and food allergy, are also more prevalent in CRSwNP versus CRSsNP.21 Such associations reflect common underlying type 2 inflammatory processes, with eosinophilic tissue inflammation mediated by type 2 cytokines, eg, interleukin (IL)-4, IL-5, and IL-13 and local/circulating IgE, evident in the majority of patients with CRSwNP.16
Greater understanding of such processes has led to the distinction of CRS on the basis of the predominant underlying inflammatory pattern (or endotype) in current classifications such as EPOS 2020.1,2 This is relevant, given the development of novel biologics targeting underlying type-2 inflammation in CRSwNP22,23 and their subsequent introduction in current recommendations.1,2,7, 8, 9, 10 Dupilumab, human monoclonal IgG4 antibody, binds the IL-4Rα subunit present on the IL-4 and IL-13 receptor complexes and inhibits both IL-4 and IL-13 signaling (key type-2 inflammatory pathway), leading to reduced IgE production and eosinophil recruitment.23, 24, 25 Omalizumab, human monoclonal IgG1 antibody, binds the Fc region of circulating IgE and blocks IgE interaction with mast cells, basophils, and B-cells carrying the high-affinity (Fc-ε-RI) IgE receptor, with subsequent downregulation of receptor expression by these cells.25 Mepolizumab, human monoclonal IgG1 antibody, binds circulating IL-5, thereby blocking its interaction with the alpha-chain on the IL-5 receptor on eosinophils, inhibiting downstream effects on eosinophil maturation, recruitment and survival.24,25
CRS classification
EPOS 2020 classifies CRS on the basis of: disease distribution ie, localized (unilateral) and diffuse (bilateral), dominant endotype (eg, the presence/absence of type-2 inflammation), and clinical phenotype.1,2 In this approach, diffuse disease with a predominantly type-2 inflammatory endotype includes CRSwNP.
EPOS 2020 includes laboratory criteria for type-2 inflammation in CRS.1,2 Clinical and laboratory criteria suggestive of type-2 inflammation in CRS have also been recently reported from Brazil,24 and the panel considered these criteria useful in identifying patients with type-2 inflammation. Accordingly, type-2 inflammation is suggested by the presence of at least 3 out of the following 5 clinical criteria; age of onset between 30 and 50 years, improvement in smell with OCS, co-existing adult-onset asthma, aspirin or NSAID intolerance, and confirmed bilateral nasal polyps and thick nasal mucus (allergic/eosinophilic mucin); and at least 1 of 3 inflammatory biomarkers; tissue eosinophilia ≥10 cells/high-power field, or serum eosinophilia ≥250 cells/μL, or total serum IgE ≥100 IU/mL.24
Approximately 80% of CRSwNP patients in the western hemisphere present with a type-2 inflammatory profile. In the remaining 20%, the inflammatory profile is considered non-type 2, either type-1 or type-3. Type-1 inflammatory profiles (characterized by IFN-gamma and IL-12 expression within the sinonasal tissues) are more commonly associated with CRSwNP in Asian populations; type-3 inflammatory profiles are less common, characterized by IL-17 and IL-22 expression.26 Furthermore, some patients exhibit a mixed inflammatory profile (ie, type-2 and non-type-2). Other forms of CRS with predominantly type-2 inflammatory pathways include allergic fungal rhinosinusitis (AFRS) and central compartment atopic disease (CCAD), a relatively recently described CRS variant, with a strong association with inhalant allergen exposure.1 While both AFRS and CCAD are characterized by the presence of polyps, these conditions are classified separately from classical CRSwNP.1,2 AFRS is recognized by the presence of eosinophilic mucin with non-invasive fungal hyphae within the sinonasal mucosa, and often a type I hypersensitivity to fungi.2,13,14 In CCAD, the disease pattern is that of polypoid mucosal changes in the superior nasal septum, middle and/or superior turbinates and meatuses.27 While polyps associated with CRSwNP and polypoidal degeneration may seem similar on CT, these can be differentiated during nasal endoscopy and surgery.
Diagnostic and clinical assessment of CRSwNP
Clinical and laboratory evaluation of CRSwNP follows well-established guidance.1,2,7, 8, 9, 10 Most clinical and laboratory investigations are performed in the specialist setting, although some (eg, complete blood count and serum total IgE levels) can be done within primary care. Patients with CRSwNP often have altered lower airway function (frequently unrecognized) at presentation and may show lower FEV1 and FEV1/FVC ratio. Thus, baseline spirometry and Asthma Control Test (ACT) at initial specialist evaluation is useful for all patients with CRSwNP (and throughout follow-up). Nasal airflow and patency can be evaluated via peak nasal inspiratory flow (PNIF) or other modalities such as rhinomanometry (RM) and acoustic rhinometry (AR).28
Nasal endoscopy and CT are essential for confirmatory diagnosis of CRSwNP. Both modalities have high diagnostic accuracy,2,29 although choice may differ depending on physician specialty and available resources. While large nasal polyps may be visualized using anterior rhinoscopy alone, endoscopy is the optimal modality and could be considered the "standard of care", and even mandatory by otorhinolaryngologists (ORL). However, endoscopy may not be available in all settings, where possible, referral to centers providing endoscopy is recommended. CT scans offer an alternative for initial diagnosis, especially for physicians in other specialties, and is useful in excluding differential diagnoses. CT scans are often preferred for surgical evaluations.
For each modality the extent of sinus disease can be assessed using simple, widely used measures.1,8,9 The endoscopic nasal polyp score (NPS) assesses polyp size, with scores from 0 to 4 on each side; no polyps (0), polyps confined to the middle meatus (1), multiple polyps occupying the middle meatus (2), polyps extending beyond middle meatus and/or olfactory cleft (3), and polyps completely obstructing the nasal cavity (4).30 Scores are aggregated to generate a total NPS ranging from 0 to 8, with higher scores indicating more extensive disease. For CT evaluations, the Lund-Mackay scoring system (LMS) is long-established and a principal outcome in most clinical studies.31 This grades each of the paranasal sinuses (frontal, maxillary, sphenoid sinuses, anterior and posterior ethmoidal cells and the ostiomeatal complex) on the basis of none, partial, or complete opacification (scored as 0, 1 or 2, respectively) to generate a total LMS score ranging from 0–24.31
Laboratory investigations include assessment of eosinophils and allergy testing, typically involving evaluation of serum allergen-specific IgE levels and skin prick tests (SPT). Total serum IgE and serum eosinophils are important biomarkers to assess endotype and confirm the presence of type 2 inflammation; while tissue eosinophilia is also indicative, this is usually performed on surgical specimens (and is not part of the routine initial work-up).
Patient self-evaluation of olfactory function is highly subjective and correlates poorly with objective measures. Olfactory function can be objectively assessed using a variety of tests. In the 40-item University of Pennsylvania Smell Identification Test (UPSIT), patients identify correct smell from 4 possible answers, across 40 distinct odorant strips. The final score (ranging from 0 to 40, higher score indicates better olfaction) can be compared with normative age- and sex-specific thresholds. Olfactory function can then be graded as normal (typically ≥35), mild (31–34), moderate (26–30) or severe (19–25) microsmia or with complete loss/anosmia (<18). Although widely used in clinical studies, challenges with UPSIT include cultural biases.2 UPSIT has been validated for Brazil,32 but may not necessarily be practical for routine olfactory assessment in the LATAM region. A short form (12-item) version (the Brief-Smell Identification Test, B-SIT) is also available.2 Another widely used olfactory function tool is the Connecticut Chemosensory Clinical Research Center (CCCRC) test, which assesses both the odor threshold component and the odor identification component, allowing for both quantitative and qualitative evaluations.33 The CCCRC test has been adapted and validated for Brazil.34 Other objective tests include the “Sniffin’ Sticks” test, a 3-step test (evaluating 16 odorants) spanning olfactory threshold, discrimination and identification domains to generate a Threshold, Discrimination, Identification (TDI) global olfactory score (ranging from 0 to 48). Higher scores indicate better olfaction, with outcomes graded as normal, hyposmia, and anosmia (thresholds are based upon normative values). This has been validated for Brazil.35 However, objective olfactory tests are still not widely available in all LATAM regions, and a standard approach for smell assessment in CRSwNP has yet to be determined.
CRS disease severity is based upon assessment of disease impact on quality of life (QoL).2 The Sino-Nasal Outcome Test-22 (SNOT-22) is the most widely used tool, and cross-cultural translated versions have been validated for Brazil, Chile and for Spanish-speaking patients.36, 37, 38, 39 SNOT-22 is a 22-item patient questionnaire which examines severity of physical symptoms (12 questions) and impact on health-related QoL (10 questions). Each question asks the patient to rate severity/impact from none through very mild, mild, moderate, severe, and very severe (scored as 0 to 5, respectively). From this a total SNOT-22 score is generated (range 0–110). Scores >50 are generally considered to reflect severe disease impact.40,41 A reduction of 8–9 points in the SNOT-22 score is considered as the minimum clinically important difference (MCID).42,43
Visual analog scale (VAS) can also be used to quantify severity of symptoms and evaluate the impact on patient QoL. On a 10-cm scale, with 0 indicating absence of symptoms and 10 indicating the most severe symptoms, patients mark on a horizontal line that they feel represents the intensity/severity of their symptoms. VAS scores for total and individual sinonasal symptoms can easily be quantified, where a higher score indicates greater severity. Scores of <2, >2 to ≤5 and > 5 cm represent well-controlled, partially controlled and uncontrolled symptoms, respectively.1,44 VAS scores for total nasal symptoms and individual symptoms correlate with SNOT-22, suggesting its utility in assessing disease severity, monitoring disease course, and treatment decisions.44
While the diagnostic tests and patient-centered outcomes described above are considered important in the routine work-up, some may seem impractical and/or beyond the scope of routine clinical assessment. However, they remain the most valid tools for complete assessment and their use during initial diagnostic and prognostic work-up is to be encouraged.
Treatment approach and care pathways
The primary goal of CRS treatment is to achieve and maintain clinical control, in which the patient is symptom-free or where symptoms are not impacting QoL. Definitions of control differ slightly in different guidelines. EPOS 2020 consider CRS as controlled, partly-controlled or uncontrolled based on the presence/absence of a range of clinical signs and symptoms. These include nasal blockage, mucopurulent rhinorrhea/postnasal drip, facial pain/pressure, smell impairment, sleep disturbance or fatigue, presence/absence of mucosal disease on endoscopy, and use or need for rescue medication i.e., short-term OCS or antibiotics.1,2 For routine clinical practice the panel favored a more simplified approach to assessment of CRSwNP control (as either controlled or uncontrolled), in which CRSwNP may be considered as uncontrolled if one or more of the above signs or symptoms persist despite appropriate treatment. Physician assessment of control is chiefly based upon clinical findings and endoscopic examination, while patient-reported symptom severity, disturbance in smell and impact on QoL, and sleep or fatigue can be measured via VAS or the SNOT-22.
Patient education is a crucial element of care at all stages of management. The impact of recognized irritants and the need to avoid exposure should be emphasized, as should the potential benefits of treatment on comorbidities such as asthma. Practical advice on self-care (ie, correct nasal irrigation technique) is fundamental. Information on the benefits of different therapies (and potential adverse effects) is essential for patient engagement.
First-line treatment options are nasal saline irrigation and use of topical INS, either as sprays or drops.45 A wide range of INS preparations are available, and while data from clinical studies would not seem to favor any particular agent, some reports suggest that drops or high volume irrigations may be more effective.21,45, 46, 47 INS are well tolerated with no significant adverse effects.1,2 There are limited data for benefit of antibiotics in the treatment of CRS or CRSwNP.1,2 If response is poor, OCS can be used, with the caveat that short-term use (eg, for up to 2 weeks) is recommended due to risks with prolonged OCS use.1,2,48 There is only some evidence for short-term improvement in symptoms (eg, smell and nasal blockage) with OCS,1,2,49 and use of short-term OCS doses (eg, 1–2 courses per year) can be beneficial in patients with poor response or uncontrolled disease.1,2 While there are no specific dosing recommendations, tapering down from an initial 25–50 mg equivalent of prednisone (or 1 mg/kg/day [max 80 mg]) is a common strategy in clinical practice.
Multidisciplinary specialist management involving ORLs and allergists/immunologists/pulmonologists provide the best approach for ensuring timely and appropriate evaluation, and use of the most appropriate therapeutic strategies to achieve optimal outcomes.50,51 When considering referral and care pathways for CRSwNP, immediate referral is essential if any red-flag alarm signs or symptoms exist (eg, visual impairment, severe headache, neurological signs) or with persistent unilateral signs/symptoms which may indicate an important alternative etiology.1,2 In other cases, the panel advises early referral from primary care for all patients with suspected CRSwNP (ideally within 6–12 weeks if unresponsive to topical treatments).1,2 This facilitates the necessary endoscopic and/or CT assessments to confirm CRSwNP and exclude other diagnoses, and a more complete clinical and laboratory evaluation to identify the inflammatory endotype.
Care in the specialist setting and surgical considerations
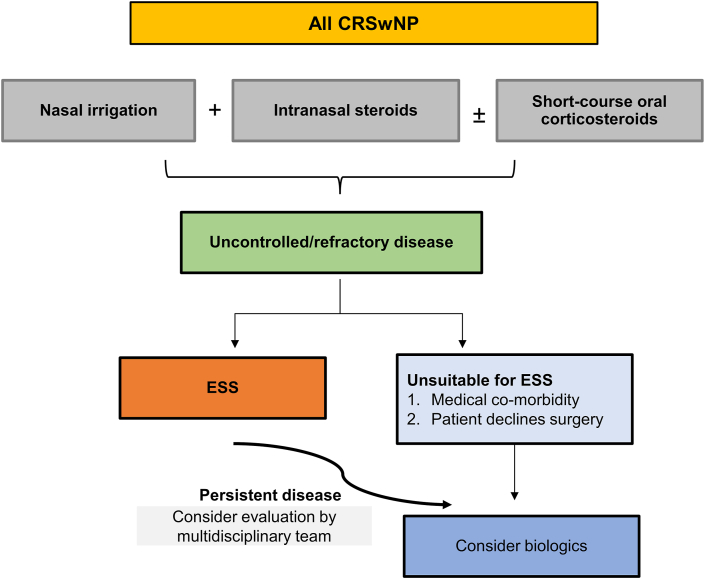
Following complete assessment (including endoscopy and/or CT evaluation), the initial treatment approach in the specialist setting remains focused on delivering and evaluating response to adequate medical treatment (topical irrigation, INS and periodic OCS)1,2 (Fig. 1). It should be emphasized that such therapy remains the foundation of care throughout management.1,2,7, 8, 9, 10,51 The majority of patients with uncontrolled disease despite optimal medical therapies are then considered for endoscopic sinus surgery (ESS),5,6,52 although (as discussed below) for some patients use of biologics can be considered at this point.51 A prolonged delay between diagnosis and surgery is associated with worse outcomes, thus a timely intervention is highly relevant in disease management.1
Fig. 1.
Treatment approach in CRSwNP.
A step-wise approach is recommended. Abbreviations: CRSwNP, chronic rhinosinusitis with nasal polyps; ESS, endoscopic sinus surgery
The aims of ESS are to improve anatomical function within the paranasal sinus and nasal cavities and remove diseased tissue.52 As well as restoring patency, remodeling facilitates mucociliary clearance and optimizes access for greater distribution of subsequent INS.51,52 While much of the supportive evidence for the benefits of ESS are drawn from non-randomised or observational studies, a recent randomized controlled study has shown that ESS combined with routine first-line medical therapy results in improved patient reported outcomes (reduction in SNOT-22 scores) compared with routine medical therapy alone.53 While criteria for timing and choice of specific surgical options vary, a useful starting point is the criteria developed by an international expert panel to reduce unnecessary surgery.5 Using the RAND/UCLA appropriateness methodology, they agreed that ESS should be considered in patients with LMS ≥1, persistent symptomatic disease after ≥8 weeks of medical therapies (INS and short-course OCS) and post-treatment total SNOT-22 score ≥20.5 These are the minimal threshold criteria for ESS, also described in EPOS 2020, with the qualification that not all patients meeting these criteria should necessarily have surgery.1,2 Ideally patients should be referred to a Center of Excellence for ESS. While the extent of ESS procedures can vary (from partial to complete), most guidelines recommend a complete ESS procedure rather than focal procedures,1,2,7 which also reflects the present panel's views. Extensive descriptions of surgical procedures are well-reviewed elsewhere.52 Most involve complete sinus opening including anterior and posterior ethmoidectomy, with middle meatal antrostomies, sphenoidotomy and frontal opening (a Draf IIa procedure), although more extensive procedures beyond the confines of sinuses (eg, Draf III) may be used on occasion.
Disease recurrence following ESS is high, and many patients require subsequent revision procedures. A meta-analysis reported an overall revision rate of 18.6%; another reported a revision rate of 24.9% within 6 months.54,55 Revision rates are higher in patients with prior incomplete ESS, prior revision surgery, those with CRSwNP, and higher still in CRSwNP with co-existing asthma.54,56,57
Use of biologics in CRSwNP
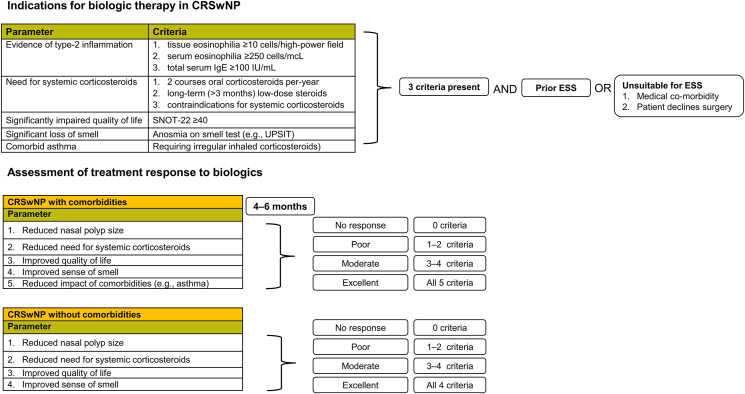
The emergence of effective biologics targeting the underlying type-2 inflammation now provides additional options for patients with uncontrolled CRSwNP.25,58 Their use is now established in current guidelines, although with slight differences in specific criteria and thresholds.1,2,7, 8, 9, 10 The panel considered that criteria reported in EPOS 20201,2 represent a pragmatic approach in clinical decision-making. Accordingly, biologics can be considered based on the presence of at least three of the following five features; (1) evidence of type-2 inflammation (tissue eosinophilia ≥10 cells/high-power field, or serum eosinophilia ≥250 cells/μL, or total serum IgE ≥100 IU/mL); (2) need for systemic corticosteroids (≥2 courses OCS per-year OR long-term [>3 months] low-dose steroids) or contraindications for systemic corticosteroids; (3) significantly impaired QoL (eg, with SNOT-22 ≥ 40); (4) significant loss of smell (anosmia on smell test); (5) with comorbid asthma (requiring regular inhaled corticosteroids).1,2 (Fig. 2). Other factors and treatment options may be considered before considering biologics; for example, for those patients with N-ERD/AERD, aspirin desensitization can be effective although safety and tolerability may affect its usefulness in clinical practice.21,50
Fig. 2.
Criteria for use of biologics and assessment of treatment response.
Abbreviations: CRSwNP, chronic rhinosinusitis with nasal polyps; ESS, endoscopic sinus surgery; SNOT-22: Sino-nasal outcome test-22; UPSIT: University of Pennsylvania smell identification test
The positioning of biologics in the treatment of uncontrolled CRSwNP continues to evolve. At present, EPOS 2020 recommends biologics for those patients with CRSwNP refractory to routine medical therapy, and where disease remains uncontrolled despite prior (and often repeated) ESS. The panel agreed with this general approach, as when adequately performed with appropriate patient selection, ESS remains an effective treatment. However, EPOS 2020 also indicates that biologics can be considered in those patients where surgery may be contraindicated (eg, unfit for surgery to medical comorbidities).1,2 Biologics may also be considered for patients with CRSwNP and asthma, as these patients have a higher rate of disease recurrence even post-surgery. In addition, in some cases, patients may prefer not to have surgery. Again, the panel agreed with this framework and recognized that informed shared-decision making is an important consideration in treatment choices.51
In line with established guidance from EPOS 20201,2 and EUFOREA,8,9 the panel does not advocate for use of specific agents, although most clinical experience to date in CRSwNP is with dupilumab. Ultimately choice of any specific biologic will be governed by patient profile, physician experience and agent availability, ideally within a shared decision-making framework. In this, patient education as to the benefits and risks of biologics is essential. Given their relatively recent introduction, inform patients of the favorable safety profile observed in clinical studies, and discuss potential adverse effects (eg, nasopharyngitis, headache, injection-site erythema).
While biologics such as dupilumab show evidence of improvement within 4–8 weeks,59 assessment at 6 months is generally considered an appropriate time-point for evaluation of treatment response, although assessment at 4 months can be made.1,2,7, 8, 9, 10 Building on criteria developed by EUFOREA,8 EPOS 2020 reports specific criteria for assessment of treatment response.1,2 In these, 5 objective criteria are considered; reduced nasal polyp size; reduced need for systemic corticosteroids; improved QoL; improved sense of smell; and reduced impact of comorbidities. Response is then graded based on the number of these parameters being met; 0 (no response); 1–2 (poor); 3–4 (moderate); and 5 (excellent). While the panel considered these to be generally useful for the LATAM setting, one limitation would seem to be the requirement for improvement in comorbidities for rating the response to biologics as excellent; and consequently, for patients without comorbidities (eg, asthma) an excellent response may not be achievable. To accommodate those patients without comorbidities, the panel's view was that comorbidity impact should not apply to assessment of such patients (ie, reducing these criteria to a 4-item set for patients without comorbidities (Fig. 2). A holistic view on clinical benefits is essential; while reductions in NPS, reduced OCS need, and improved asthma control, are clearly of real clinical value, other outcomes such as improvements in smell and specific symptoms such as nasal congestion may be more important from a patient perspective. Furthermore, a broader chronological assessment, rather than assessment at specific time-points, may be more suitable in some settings.
These criteria can be used to evaluate benefits of a chosen biologic, and to assess whether discontinuation and/or switching to an alternative biologic is indicated. At present, there are limited data to support specific recommendations. However, some general proposals can be made. Firstly, continuing with conventional medical treatment (topical irrigation and use of INS) during biologic therapy is essential, and patients must be educated on the importance of this. Where there is some evidence of improvement early on (eg, after 4–6 months of biologics), then treatment should be continued (and re-evaluated after a further 6 months). Where there is limited or no response at 6 months, then we would suggest switching to an alternative agent. However, such patients should also be revaluated to confirm evidence of type-2 inflammation, and their suitability for biologics. In addition, surgical options should be reconsidered. Aspirin desensitization should be considered in patients with N-ERD/AERD.21,50 This emphasizes the importance of a multi-disciplinary approach throughout the management of patients with severe/uncontrolled CRSwNP.
Additional observations and considerations
The emergence of biologics has the potential to transform the current approach to CRSwNP management. However, some additional points are worth making. Firstly, there remains a need to better characterize the burden of CRSwNP in the region. Epidemiological data are lacking; limited data are available for Brazil, with the prevalence of CRS estimated as 5.5% based on population questionnaires.60 However, these data were reported in 2012, and more complete data for CRS, and in particular for CRSwNP in other countries in the LATAM region are lacking. Similarly, laboratory assessment of immune-phenotypes is not yet routinely performed within the LATAM region, and such regional data are limited. More robust regional data on epidemiology and the impact of CRSwNP, may help in cost-effectiveness evaluations to support broader, appropriate use of biologics in the region. Furthermore, at present, there is relatively limited real-world data for the use of biologics in CRSwNP. Studies reporting biologic use within the LATAM region are to be encouraged. Finally, while the value of a multidisciplinary approach is emphasized, access to specialist services may be limited in rural areas. Educational initiatives and direct support of clinicians and patients are essential to provide equitable access to the most appropriate care.
The principles of management of CRSwNP we present are subject to a number of limitations. As a starting point we chose to focus chiefly on guidance presented in EPOS 20,1,2 which we believe provides a practical and pragmatic approach to CRSwNP patient care. In this we discuss core aspects of these guidelines in the context of how these apply to our colleagues in the LATAM region. While the principles and specific details for diagnosis and treatment of CRSwNP we describe are drawn from evidence-based guidance (chiefly EPOS 20), and represent the consensus view from a multidisciplinary expert panel, our aim was not to make specific formal recommendations or to systematically evaluate the underlying evidence base. As such, the guidance we present is our expert opinion, and should be considered on these terms. In addition, while we have made some proposals as to use of medical agents (topical steroids and biologics) we have remained relatively neutral in respect to specific agents, and their relative merits in CRSwNP. In part this limitation was deliberate as agent availability and licensed indications may vary across the region. Subsequent to our initial workshops, a number of evidence-based clinical guidelines have been developed, including one from the American Academy of Allergy, Asthma & Immunology (AAAAI)/American College of Allergy, Asthma, and Immunology (ACAAI).61 This guideline, drawing from a number of quality systematic reviews and network meta-analyses of clinical studies,62,63 reports on the use of and relative merits of different INS formulations, aspirin desensitization, and different biologics in the medical management of CRSwNP.61 These articles can serve as a valuable resource for clinicians when considering appropriate biologic agents.
Conclusions
Here, we present key aspects of the current understanding of CRSwNP and provide a practical approach and guidance on the clinical management of patients within the LATAM region. Our consensus is that most patients will benefit from a multidisciplinary approach to evaluation and clinical management. Many patients respond to medical treatment (principally focused on topical treatment directed towards inflamed nasal mucosa and adjunctive short-term use of systemic corticosteroids). However, those with more severe/uncontrolled disease require surgery. In this, patient selection is critical and for some patients, treatment with biologics could be considered at this stage. For those patients with persistent, uncontrolled symptoms despite surgery, biologics should be considered. When used, treatment response should be assessed using established criteria (ideally after 4–6 months) and consider alternative treatments if response is poor.
Abbreviations
AR, acoustic rhinometry; ACT, Asthma control test; AFRS, Allergic fungal rhinosinusitis; B-SIT, Brief-smell identification test; CCCRC, Connecticut Chemosensory Clinical Research Center; CRS, Chronic rhinosinusitis; CRSsNP, Chronic rhinosinusitis without nasal polyps; CRSwNP, Chronic rhinosinusitis with nasal polyps (CRSwNP); CCAD, Central compartment atopic disease (CCAD); EPOS, European position paper on rhinosinusitis and nasal polyps; ESS, Endoscopic sinus surgery; EUFOREA, European forum for research and education in allergy and airway diseases; ICAR-RS, International consensus statement on allergy and rhinology-rhinosinusitis; INS, Intranasal steroids; LATAM, Latin America; LMS, Lund-Mackay scoring system; MCID, Minimum clinically important difference; N-ERD/AERD, NSAID/aspirin-exacerbated respiratory disease; OCS, Oral corticosteroids; PNIF, peak nasal inspiratory flow; QoL, Quality of life; SNOT-22, Sino-nasal outcome test-22; SPT, skin prick tests; TDI, Threshold, discrimination, identification; UPSIT, University of Pennsylvania smell identification test; VAS, Visual analog scale.
Funding
The logistics of the panel selection, meeting facilitation and subsequent discussions and manuscript development were supported by an unrestricted grant from Sanofi. The sponsor had no influence or involvement in the recommendations developed from the discussions or on the content and viewpoints expressed in this manuscript.
Author contributions
JFM chaired the panel discussions and wrote the initial draft. All authors reviewed the article for content accuracy and provided input into subsequent drafts, and provided approval of the final draft.
Ethics statement
This project and the underlying process was conducted through review of publicly available literature, and did not involve specific human participants or any identifiable data. Institutional review board approval and informed consent were not required.
Consent to publish
All authors agree to publication of this manuscript in World Allergy Organization Journal.
Availability of data and materials
The data that support the findings of this paper are available on request from the corresponding author.
Declaration of competing interest
JFM has acted as a speaker, advisory board member, and/or investigator for Sanofi, AstraZeneca, Novartis, Inmunito, Uriach, GSK, Menarini and MSD. DB has attended advisory board and received sponsorship from Sanofi. AS has acted as an advisor for Sanofi. WA-L, MAJC, JO, RR and RSR have no direct or indirect conflict of interests to declare.
Acknowledgements
The authors would like to thank Irena Mandic and Stephen McGrath (IntraMed Communications) for their support in facilitating the Expert Panel meetings and subsequent activities; Iain O'Neill (independent medical writer) and Tejinder Kaur (IntraMed Communications) provided support in manuscript development and editorial assistance.
Footnotes
Full list of author information is available at the end of the article
References
- 1.Fokkens W.J., Lund V.J., Hopkins C., et al. European position paper on rhinosinusitis and nasal polyps 2020. Rhinology. 2020;58(Suppl S29):1–464. doi: 10.4193/Rhin20.600. [DOI] [PubMed] [Google Scholar]
- 2.Fokkens W.J., Lund V.J., Hopkins C., et al. Executive summary of EPOS 2020 including integrated care pathways. Rhinology. 2020;58(2):82–111. doi: 10.4193/Rhin20.601. [DOI] [PubMed] [Google Scholar]
- 3.Sedaghat A.R., Kuan E.C., Scadding G.K. Epidemiology of chronic rhinosinusitis: prevalence and risk factors. J Allergy Clin Immunol Pract. 2022;10(6):1395–1403. doi: 10.1016/j.jaip.2022.01.016. [DOI] [PubMed] [Google Scholar]
- 4.Laidlaw T.M., Mullol J., Woessner K.M., et al. Chronic rhinosinusitis with nasal polyps and asthma. J Allergy Clin Immunol Pract. 2021;9(3):1133–1141. doi: 10.1016/j.jaip.2020.09.063. [DOI] [PubMed] [Google Scholar]
- 5.Rudmik L., Soler Z.M., Hopkins C., et al. Defining appropriateness criteria for endoscopic sinus surgery during management of uncomplicated adult chronic rhinosinusitis: a RAND/UCLA appropriateness study. Rhinology. 2016;54(2):117–128. doi: 10.4193/Rhino16.023. [DOI] [PubMed] [Google Scholar]
- 6.Beswick D.M., Mace J.C., Soler Z.M., et al. Appropriateness criteria predict outcomes for sinus surgery and may aid in future patient selection. Laryngoscope. 2018;128(11):2448–2454. doi: 10.1002/lary.27227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Orlandi R.R., Kingdom T.T., Smith T.L., et al. International consensus statement on allergy and rhinology: rhinosinusitis 2021. Int Forum Allergy Rhinol. 2021;11(3):213–739. doi: 10.1002/alr.22741. [DOI] [PubMed] [Google Scholar]
- 8.Fokkens W.J., Lund V., Bachert C., et al. EUFOREA consensus on biologics for CRSwNP with or without asthma. Allergy. 2019;74(12):2312–2319. doi: 10.1111/all.13875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bachert C., Han J.K., Wagenmann M., et al. EUFOREA expert board meeting on uncontrolled severe chronic rhinosinusitis with nasal polyps (CRSwNP) and biologics: definitions and management. J Allergy Clin Immunol. 2021;147(1):29–36. doi: 10.1016/j.jaci.2020.11.013. [DOI] [PubMed] [Google Scholar]
- 10.Fokkens W.J., Lund V., Luong A.U., et al. A comparison of international guidelines for rhinosinusitis. J Allergy Clin Immunol Pract. 2022;10(6):1418–1422. doi: 10.1016/j.jaip.2022.01.013. [DOI] [PubMed] [Google Scholar]
- 11.Dietz de Loos D.A., Hopkins C., Fokkens W.J. Symptoms in chronic rhinosinusitis with and without nasal polyps. Laryngoscope. 2013;123(1):57–63. doi: 10.1002/lary.23671. [DOI] [PubMed] [Google Scholar]
- 12.Passali D., Cingi C., Cambi J., et al. A survey on chronic rhinosinusitis: opinions from experts of 50 countries. Eur Arch Oto-Rhino-Laryngol. 2016;273(8):2097–2109. doi: 10.1007/s00405-015-3880-6. [DOI] [PubMed] [Google Scholar]
- 13.Laury A.M., Wise S.K. Chapter 7: allergic fungal rhinosinusitis. Am J Rhinol Allergy. 2013;27(Suppl 1):S26–S27. doi: 10.2500/ajra.2013.27.3891. [DOI] [PubMed] [Google Scholar]
- 14.Bulkhi A.A., Mirza A.A., Aburiziza A.J., et al. Dupilumab: an emerging therapy in allergic fungal rhinosinusitis. World Allergy Organ J. 2022;15(3) doi: 10.1016/j.waojou.2022.100638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schleimer R.P., Berdnikovs S. Etiology of epithelial barrier dysfunction in patients with type 2 inflammatory diseases. J Allergy Clin Immunol. 2017;139(6):1752–1761. doi: 10.1016/j.jaci.2017.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bachert C., Marple B., Hosemann W., et al. Endotypes of chronic rhinosinusitis with nasal polyps: pathology and possible therapeutic implications. J Allergy Clin Immunol Pract. 2020;8(5):1514–1519. doi: 10.1016/j.jaip.2020.03.007. [DOI] [PubMed] [Google Scholar]
- 17.Cho S.H., Hamilos D.L., Han D.H., et al. Phenotypes of chronic rhinosinusitis. J Allergy Clin Immunol Pract. 2020;8(5):1505–1511. doi: 10.1016/j.jaip.2019.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jarvis D., Newson R., Lotvall J., et al. Asthma in adults and its association with chronic rhinosinusitis: the GA2LEN survey in Europe. Allergy. 2012;67(1):91–98. doi: 10.1111/j.1398-9995.2011.02709.x. [DOI] [PubMed] [Google Scholar]
- 19.Castagnoli R., Licari A., Brambilla I., et al. An update on the role of chronic rhinosinusitis with nasal polyps as a co-morbidity in severe asthma. Expet Rev Respir Med. 2020;14(12):1197–1205. doi: 10.1080/17476348.2020.1812388. [DOI] [PubMed] [Google Scholar]
- 20.Philpott C.M., Erskine S., Hopkins C., et al. Prevalence of asthma, aspirin sensitivity and allergy in chronic rhinosinusitis: data from the UK National Chronic Rhinosinusitis Epidemiology Study. Respir Res. 2018;19(1):129. doi: 10.1186/s12931-018-0823-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kowalski M.L., Agache I., Bavbek S., et al. Diagnosis and management of NSAID-Exacerbated Respiratory Disease (N-ERD)-a EAACI position paper. Allergy. 2019;74(1):28–39. doi: 10.1111/all.13599. [DOI] [PubMed] [Google Scholar]
- 22.Agache I., Song Y., Alonso-Coello P., et al. Efficacy and safety of treatment with biologicals for severe chronic rhinosinusitis with nasal polyps: a systematic review for the EAACI guidelines. Allergy. 2021;76(8):2337–2353. doi: 10.1111/all.14809. [DOI] [PubMed] [Google Scholar]
- 23.Fokkens W., Van Der Lans R., Reitsma S. Dupilumab for the treatment of chronic rhinosinusitis with nasal polyposis. Expet Opin Biol Ther. 2021;21(5):575–585. doi: 10.1080/14712598.2021.1901881. [DOI] [PubMed] [Google Scholar]
- 24.Anselmo-Lima W.T., Tamashiro E., Romano F.R., et al. Guideline for the use of immunobiologicals in chronic rhinosinusitis with nasal polyps (CRSwNP) in Brazil. Braz J Otorhinolaryngol. 2021 doi: 10.1016/j.bjorl.2021.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bachert C., Zhang N., Cavaliere C., et al. Biologics for chronic rhinosinusitis with nasal polyps. J Allergy Clin Immunol. 2020;145(3):725–739. doi: 10.1016/j.jaci.2020.01.020. [DOI] [PubMed] [Google Scholar]
- 26.Staudacher A.G., Peters A.T., Kato A., et al. Use of endotypes, phenotypes, and inflammatory markers to guide treatment decisions in chronic rhinosinusitis. Ann Allergy Asthma Immunol. 2020;124(4):318–325. doi: 10.1016/j.anai.2020.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Steehler A.J., Vuncannon J.R., Wise S.K., et al. Central compartment atopic disease: outcomes compared with other subtypes of chronic rhinosinusitis with nasal polyps. Int Forum Allergy Rhinol. 2021;11(11):1549–1556. doi: 10.1002/alr.22819. [DOI] [PubMed] [Google Scholar]
- 28.Rimmer J., Hellings P., Lund V.J., et al. European position paper on diagnostic tools in rhinology. Rhinology. 2019;57(Suppl S28):1–41. doi: 10.4193/Rhin19.410. [DOI] [PubMed] [Google Scholar]
- 29.Kim D.H., Seo Y., Kim K.M., et al. Usefulness of nasal endoscopy for diagnosing patients with chronic rhinosinusitis: a meta-analysis. Am J Rhinol Allergy. 2020;34(2):306–314. doi: 10.1177/1945892419892157. [DOI] [PubMed] [Google Scholar]
- 30.Gevaert P., Calus L., Van Zele T., et al. Omalizumab is effective in allergic and nonallergic patients with nasal polyps and asthma. J Allergy Clin Immunol. 2013;131(1):110–116 e1. doi: 10.1016/j.jaci.2012.07.047. [DOI] [PubMed] [Google Scholar]
- 31.Hopkins C., Browne J.P., Slack R., et al. The Lund-Mackay staging system for chronic rhinosinusitis: how is it used and what does it predict? Otolaryngol Head Neck Surg. 2007;137(4):555–561. doi: 10.1016/j.otohns.2007.02.004. [DOI] [PubMed] [Google Scholar]
- 32.Fornazieri M.A., dos Santos C.A., Bezerra T.F., et al. Development of normative data for the Brazilian adaptation of the university of Pennsylvania smell identification test. Chem Senses. 2015;40(2):141–149. doi: 10.1093/chemse/bju068. [DOI] [PubMed] [Google Scholar]
- 33.Veyseller B., Ozucer B., Karaaltin A.B., et al. Connecticut (CCCRC) olfactory test: normative values in 426 healthy volunteers. Indian J Otolaryngol Head Neck Surg. 2014;66(1):31–34. doi: 10.1007/s12070-013-0632-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fenolio G.H.M., Anselmo-Lima W.T., Tomazini G.C., et al. Validation of the Connecticut olfactory test (CCCRC) adapted to Brazil. Braz J Otorhinolaryngol. 2022;88(5):725–732. doi: 10.1016/j.bjorl.2020.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Silveira-Moriyama L., Carvalho Mde J., Katzenschlager R., et al. The use of smell identification tests in the diagnosis of Parkinson's disease in Brazil. Mov Disord. 2008;23(16):2328–2334. doi: 10.1002/mds.22241. [DOI] [PubMed] [Google Scholar]
- 36.Kosugi E.M., Chen V.G., Fonseca V.M., et al. Translation, cross-cultural adaptation and validation of SinoNasal outcome test (SNOT): 22 to Brazilian Portuguese. Braz J Otorhinolaryngol. 2011;77(5):663–669. doi: 10.1590/s1808-86942011000500021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mascarenhas J.G., da Fonseca V.M., Chen V.G., et al. Long-term outcomes of endoscopic sinus surgery for chronic rhinosinusitis with and without nasal polyps. Braz J Otorhinolaryngol. 2013;79(3):306–311. doi: 10.5935/1808-8694.20130055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.de los Santos G., Reyes P., del Castillo R., et al. Cross-cultural adaptation and validation of the sino-nasal outcome test (SNOT-22) for Spanish-speaking patients. Eur Arch Oto-Rhino-Laryngol. 2015;272(11):3335–3340. doi: 10.1007/s00405-014-3437-0. [DOI] [PubMed] [Google Scholar]
- 39.Valdés P.C., Muñoz M.T., Barría E.T., et al. Validación de encuesta de calidad de vida SNOT-22 en población chilena. Revista de otorrinolaringología y cirugía de cabeza y cuello. 2020;80:425–433. [Google Scholar]
- 40.Hopkins C., Gillett S., Slack R., et al. Psychometric validity of the 22-item sinonasal outcome test. Clin Otolaryngol. 2009;34(5):447–454. doi: 10.1111/j.1749-4486.2009.01995.x. [DOI] [PubMed] [Google Scholar]
- 41.Toma S., Hopkins C. Stratification of SNOT-22 scores into mild, moderate or severe and relationship with other subjective instruments. Rhinology. 2016;54(2):129–133. doi: 10.4193/Rhino15.072. [DOI] [PubMed] [Google Scholar]
- 42.Hopkins C., Rudmik L., Lund V.J. The predictive value of the preoperative Sinonasal Outcome Test-22 score in patients undergoing endoscopic sinus surgery for chronic rhinosinusitis. Laryngoscope. 2015;125(8):1779–1784. doi: 10.1002/lary.25318. [DOI] [PubMed] [Google Scholar]
- 43.Chowdhury N.I., Mace J.C., Bodner T.E., et al. Does medical therapy improve SinoNasal outcomes test-22 domain scores? An analysis of clinically important differences. Laryngoscope. 2019;129(1):31–36. doi: 10.1002/lary.27470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Doulaptsi M., Prokopakis E., Seys S., et al. Visual analogue scale for sino-nasal symptoms severity correlates with sino-nasal outcome test 22: paving the way for a simple outcome tool of CRS burden. Clin Transl Allergy. 2018;8:32. doi: 10.1186/s13601-018-0219-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Grayson J.W., Harvey R.J. Topical corticosteroid irrigations in chronic rhinosinusitis. Int Forum Allergy Rhinol. 2019;9(S1):S9–S15. doi: 10.1002/alr.22331. [DOI] [PubMed] [Google Scholar]
- 46.Bachert C., Zhang L., Gevaert P. Current and future treatment options for adult chronic rhinosinusitis: focus on nasal polyposis. J Allergy Clin Immunol. 2015;136(6):1431–1440. doi: 10.1016/j.jaci.2015.10.010. [DOI] [PubMed] [Google Scholar]
- 47.Neubauer P.D., Schwam Z.G., Manes R.P. Comparison of intranasal fluticasone spray, budesonide atomizer, and budesonide respules in patients with chronic rhinosinusitis with polyposis after endoscopic sinus surgery. Int Forum Allergy Rhinol. 2016;6(3):233–237. doi: 10.1002/alr.21688. [DOI] [PubMed] [Google Scholar]
- 48.Hox V., Lourijsen E., Jordens A., et al. Benefits and harm of systemic steroids for short- and long-term use in rhinitis and rhinosinusitis: an EAACI position paper. Clin Transl Allergy. 2020;10:1. doi: 10.1186/s13601-019-0303-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Head K., Chong L.Y., Hopkins C., et al. Short-course oral steroids as an adjunct therapy for chronic rhinosinusitis. Cochrane Database Syst Rev. 2016;4:CD011992. doi: 10.1002/14651858.CD011992.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lal D., Borish L., Detwiller K.Y., et al. The rationale for multidisciplinary management of chronic rhinosinusitis with nasal polyposis. J Allergy Clin Immunol Pract. 2020;8(5):1565–1566. doi: 10.1016/j.jaip.2020.03.001. [DOI] [PubMed] [Google Scholar]
- 51.Han J.K., Bosso J.V., Cho S.H., et al. Multidisciplinary consensus on a stepwise treatment algorithm for management of chronic rhinosinusitis with nasal polyps. Int Forum Allergy Rhinol. 2021;11(10):1407–1416. doi: 10.1002/alr.22851. [DOI] [PubMed] [Google Scholar]
- 52.Weber R.K., Hosemann W. Comprehensive review on endonasal endoscopic sinus surgery. GMS Curr Top Otorhinolaryngol, Head Neck Surg. 2015;14:Doc08. doi: 10.3205/cto000123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lourijsen E.S., Reitsma S., Vleming M., et al. Endoscopic sinus surgery with medical therapy versus medical therapy for chronic rhinosinusitis with nasal polyps: a multicentre, randomised, controlled trial. Lancet Respir Med. 2022;10(4):337–346. doi: 10.1016/S2213-2600(21)00457-4. [DOI] [PubMed] [Google Scholar]
- 54.Loftus C.A., Soler Z.M., Koochakzadeh S., et al. Revision surgery rates in chronic rhinosinusitis with nasal polyps: meta-analysis of risk factors. Int Forum Allergy Rhinol. 2020;10(2):199–207. doi: 10.1002/alr.22487. [DOI] [PubMed] [Google Scholar]
- 55.Loftus C.A., Soler Z.M., Desiato V.M., et al. Factors impacting revision surgery in patients with chronic rhinosinusitis with nasal polyposis. Int Forum Allergy Rhinol. 2020;10(3):289–302. doi: 10.1002/alr.22505. [DOI] [PubMed] [Google Scholar]
- 56.Gill A.S., Smith K.A., Meeks H., et al. Asthma increases long-term revision rates of endoscopic sinus surgery in chronic rhinosinusitis with and without nasal polyposis. Int Forum Allergy Rhinol. 2021;11(8):1197–1206. doi: 10.1002/alr.22779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Laidlaw T.M., Buchheit K.M. Biologics in chronic rhinosinusitis with nasal polyposis. Ann Allergy Asthma Immunol. 2020;124(4):326–332. doi: 10.1016/j.anai.2019.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Mullol J., Azar A., Buchheit K.M., et al. Chronic rhinosinusitis with nasal polyps: quality of life in the biologics era. J Allergy Clin Immunol Pract. 2022 doi: 10.1016/j.jaip.2022.03.002. [DOI] [PubMed] [Google Scholar]
- 59.Bachert C., Han J.K., Desrosiers M., et al. Efficacy and safety of dupilumab in patients with severe chronic rhinosinusitis with nasal polyps (LIBERTY NP SINUS-24 and LIBERTY NP SINUS-52): results from two multicentre, randomised, double-blind, placebo-controlled, parallel-group phase 3 trials. Lancet. 2019;394(10209):1638–1650. doi: 10.1016/S0140-6736(19)31881-1. [DOI] [PubMed] [Google Scholar]
- 60.Pilan R.R., Pinna F.R., Bezerra T.F., et al. Prevalence of chronic rhinosinusitis in sao Paulo. Rhinology. 2012;50(2):129–138. doi: 10.4193/Rhino11.256. [DOI] [PubMed] [Google Scholar]
- 61.Rank M.A., Chu D.K., Bognanni A., et al. The Joint Task Force on Practice Parameters GRADE guidelines for the medical management of chronic rhinosinusitis with nasal polyposis. J Allergy Clin Immunol. 2023;151(2):386–398. doi: 10.1016/j.jaci.2022.10.026. [DOI] [PubMed] [Google Scholar]
- 62.Bognanni A., Chu D.K., Rank M.A., et al. Topical corticosteroids for chronic rhinosinusitis with nasal polyposis: GRADE systematic review and network meta-analysis. J Allergy Clin Immunol. 2022;150(6):1447–1459. doi: 10.1016/j.jaci.2022.07.023. [DOI] [PubMed] [Google Scholar]
- 63.Oykhman P., Paramo F.A., Bousquet J., et al. Comparative efficacy and safety of monoclonal antibodies and aspirin desensitization for chronic rhinosinusitis with nasal polyposis: a systematic review and network meta-analysis. J Allergy Clin Immunol. 2022;149(4):1286–1295. doi: 10.1016/j.jaci.2021.09.009. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this paper are available on request from the corresponding author.