Abstract
Green banana flour (GBF) is a novel ingredient that is high in resistant starch and could be a dietary fiber source in companion animal nutrition. In addition, with its light brown color and pectin content, GBF could potentially serve as a natural color additive and thickening agent in pet food manufacturing. The purpose of this research is to evaluate different sources of GBF, the effect of GBF on texture and color in canned foods, and its effect on apparent total tract digestibility (ATTD), fecal characteristics, and fecal fermentative end-products in healthy adult cats. Prior to the feline study, different sources of GBF were analyzed for chemical composition, manufacturing properties, true metabolizable energy, and fermentability. For the feline feeding trial, all treatment diets were formulated to meet or exceed the Association of American Feed Control Officials (Association of American Feed Control Officials (AAFCO) 2020. Official Publication. Champaign, IL.) guidelines for adult cat maintenance. There were five dietary treatments: rice control (4% rice flour), potato control (4% dehydrated potato flakes), 1% GBF (1% GBF and 3% rice flour), 2% GBF (2% GBF and 2% rice flour), and 4% GBF. All treatment diets were analyzed for texture and color. The animal study was conducted using a completely randomized design with 39 adult domestic cats. There was a 7-d diet adaptation period followed by a baseline fresh fecal collection to determine fecal score, pH, short-chain fatty acid, branched-chain fatty acid, phenol, indole, ammonia, and microbiota. The treatment period lasted for 21 d and a total fecal collection was performed during the last 4 d of this period to determine the ATTD. A fresh fecal sample was also collected during the total fecal collection to evaluate fecal score, pH, metabolites, and microbiota. The MIXED model procedures of SAS version 9.4 were used for statistical analysis. Treatment diets containing GBF had a lower hardness from the texture profile analysis (P < 0.05). For color analysis, the 4% GBF diet was darker in color compared with the rice diet (P < 0.05). There was no difference in food intake, fecal output, or ATTD of macronutrients among the treatment groups (P > 0.05). There was no interaction of treatment and time or main effects shown in fecal score, pH, metabolites, or microbiota diversity (P > 0.05). In conclusion, adding GBF to canned diets may affect the texture and color of the product, but GBF was comparable to traditional carbohydrate sources, rice, and potato, from a nutritional aspect.
Keywords: cats, color, green banana flour, nutrient digestibility, resistant starch, texture
Green banana flour can serve as a comparable starch source when compared with traditional starches in canned foods while acting as a functional ingredient in the manufacturing process for product texture and color.
Introduction
Dietary fiber has been gaining attention in companion animal nutrition throughout the years due to its effect on gut health (Dai and Chau, 2016). Resistant starch (RS) fits into the dietary fiber definition from Food and Drug Administration (2016): “non-digestible soluble and insoluble carbohydrates (with 3 or more monomeric units), and lignin that are intrinsic and intact in plants; isolated or synthetic non-digestible carbohydrates determined by the Food and Drug Administration to have physiological effects that are beneficial to human health.” As a type of soluble dietary fiber (SDF), RS possesses some health benefits, such as bowel health improvement, hypoglycemic effects, prebiotic effects, micronutrient absorption increase, and fat accumulation inhibition (Nugent, 2005; Fuentes-Zaragoza et al., 2010). RS is categorized into five types, RS1-RS5 (Englyst et al., 1992; Brown et al., 1995). Different sources contain different types and concentrations of RS. In companion animal nutrition, potato is a commonly used starch source that is rich in RS2. Green banana flour (GBF) is another ingredient that is high in RS2; however, there has been no study on the use of GBF in companion animal nutrition.
GBF was introduced to the human nutrition industry as a gluten-free substitute for wheat flour. With the banana plant being highly produced (Food and Agriculture Organization of the United Nations, 2018), the supply of raw fruit is abundant to manufacture GBF. Processing bananas into the dried powder form of GBF could also help with decreasing the high loss due to handling problems, commercial criteria (such as size, shape, and appearance), and spoilage of ripe bananas (Zhang et al., 2005). Unripe banana is rich in RS2 (40% to 50% dry matter [DM] basis) and low in digestible starch and free sugars (Menezes et al., 2010; Moongngarm et al., 2014; Khoozani et al., 2019). Attention has been brought to GBF because of its high RS content and studies have been done to test its nutritional functionality. Studies have shown GBF has the potential in protecting gastric mucus, preventing complications of type 1 diabetes, and helping with glycemic control in rats (Dunjic et al., 1993; Dan et al., 2015; Eleazu and Okafor, 2015; da Silva et al., 2016). In humans, GBF has been found effective in laxation for functional constipation, decreasing ileal digestibility of energy, modulating glycemic response, increasing satiety after meals, increasing insulin sensitivity, and maintaining body weight (Langkilde et al., 2002; Ble-Castillo et al., 2010; Sardá et al., 2016; Falcomer et al., 2019). These benefits may also be seen in companion animals but there is no scientific literature available evaluating the functional properties of GBF in companion animals.
In addition to a high RS2 content, green banana also contains 2.5% pectin in its pulp on a DM basis (Chiang et al., 1987). Pectin in GBF could function as a potential thickening agent during pet food processing, especially in wet pet food. The natural light brown color of GBF also could serve as a natural coloring aid as natural food is a growing segment in the pet food industry. GBF is a natural ingredient that has several beneficial attributes related to food manufacturing and nutraceutical properties. A pilot study was first conducted to better understand the chemical composition, manufacturing functionalities, and fiber characteristics of different sources of GBF. Then, one source of GBF was selected for the feline study to test the effect of GBF on texture and color in canned foods, as well as its effect on apparent total tract digestibility (ATTD) of macronutrients and fecal metabolites in cats. It was hypothesized that the addition of GBF would serve as a natural coloring and thickening in wet pet food, and would favor saccharolytic fermentation of cats fed GBF-containing diets in contrast with cats fed the rice flour-based diet.
Materials and Methods
All animal-related procedures were performed in accordance with the United States Public Health Service Policy on Humane Care and Use of Laboratory Animals.
Study 1
Experimental design.
Four types of green banana flour (GBF1: Banana Multi-purpose Flour, Pereg, Clifton, NJ; GBF2: Farinha de Banana Verde, Duraci, Lençóis Paulista, Brazil; GBF3: Let’s Do Organic GBF, Edward & Sons, Carpinteria, CA; GBF4: Dried Banana, The Peterson Company, Kalamazoo, MI) were analyzed to determine their nutritional compositions. Canned foods were made at Simmons Pet Food, Inc. (Siloam Springs, AR) laboratories, each containing one of the four sources of GBF in different amounts, to test the effects of GBF on the texture and/or color of the products. These retorted foods were sorted into three categories: loaf test, water binding test, and gravy test. Formulations of the test canned foods were based on commercial standards. The loaf test canned foods were formulated to be complete and balanced according to the Association of American Feed Control Officials (AAFCO) nutrient profile for adult dogs at maintenance with different amounts of one of the four GBF sources (Table 1) and were tested for the texture. The water binding test canned foods were composed of mechanically separated chicken, water, and either a commercial egg-based binder (Ovabind, Nuvin, St. Louis, MO, USA) or GBF (Table 2). The gravy in the gravy test canned foods was composed of water and thickeners, including guar gum, corn starch, and/or GBF (Table 3). Both water binding test and Gravy Test foods were used to examine the effect of GBF on color. Differences in inclusion rates were aimed at examining the possibility of GBF being a partial replacer of commonly used gums. Raw ingredients were mixed together with KitchenAid (Benton Harbor, MI) and 100 g of the raw mixture was filled into each of the 89.5 mm diameter trays. Canned foods were processed in 1 batch for approximately 1 h at 121.7 °C in an Allpax Gentle Motion Retort (Allpax, Covington, LA) and the temperature was monitored throughout the process. The diets were cooled to 32.2 °C in the retort system and then taken out to cool to room temperature. These diets were made at a small scale as a pilot study to obtain descriptive data as a means to better understand the characteristics of the different sources of GBF.
Table 1.
Ingredient composition of loaf test treatments containing green banana flour
| Treatments1 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ingredient2, % as-is | LCO | LT1 | LT2 | LT3 | LT4 | LT5 | LT6 | LT7 | LT8 | LT9 | LT10 |
| Chicken heart and liver | 1.60 | 1.59 | 1.57 | 1.59 | 1.57 | 1.59 | 1.57 | 1.60 | 1.60 | 1.60 | 1.60 |
| Chicken mix frozen | 16.37 | 16.21 | 16.04 | 16.21 | 16.04 | 16.21 | 16.04 | 16.37 | 16.37 | 16.37 | 16.37 |
| Chicken mechanically deboned low ash | 53.56 | 53.03 | 52.49 | 53.03 | 52.49 | 53.03 | 52.49 | 53.56 | 53.56 | 53.56 | 53.56 |
| Dog vitamin premix | 0.06 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.06 | 0.06 | 0.06 | 0.06 |
| Dog chelated mineral premix | 0.11 | 0.11 | 0.11 | 0.11 | 0.11 | 0.11 | 0.11 | 0.11 | 0.11 | 0.11 | 0.11 |
| Choline chloride 70% | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 |
| Salt | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 |
| Potassium chloride | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 |
| Chicory inulin | 0.06 | 0.06 | 0.06 | 0.06 | 0.06 | 0.06 | 0.06 | 0.06 | 0.06 | 0.06 | 0.06 |
| Cassia gum | 0.35 | 0.35 | 0.34 | 0.35 | 0.34 | 0.35 | 0.34 | 0.18 | 0.00 | 0.35 | 0.35 |
| Guar gum | 0.20 | 0.20 | 0.20 | 0.20 | 0.20 | 0.20 | 0.20 | 0.10 | 0.20 | 0.00 | 0.00 |
| Kappa carrageenan | 0.35 | 0.35 | 0.34 | 0.35 | 0.34 | 0.35 | 0.34 | 0.13 | 0.35 | 0.35 | 0.00 |
| Steam | 11.51 | 11.40 | 11.28 | 11.40 | 11.28 | 11.40 | 11.28 | 11.51 | 11.51 | 11.51 | 11.51 |
| Water | 15.23 | 15.08 | 14.92 | 15.08 | 14.92 | 15.08 | 14.92 | 15.23 | 15.23 | 15.23 | 15.23 |
| GBF1 | 0.00 | 1.00 | 2.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| GBF2 | 0.00 | 0.00 | 0.00 | 1.00 | 2.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| GBF3 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 1.00 | 2.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| GBF4 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.50 | 0.35 | 0.20 | 0.35 |
1LCO, loaf test control; LT, loaf test.
2GBF1, Pereg Banana Multi-purpose Flour, Clifton, NJ; GBF2, Duraci Farinha de Banana Verde, Lençóis Paulista, Brazil; GBF3, Edward & Sons Let’s Do Organic Green Banana Flour, Carpinteria, CA; GBF4, Peterson Dried Banana, Kalamazoo, MI.
The bolded numbers are the main difference among treatments which was the green banana flour inclusion rate.
Table 2.
Ingredient composition of water binding test treatments containing green banana flour
| Treatments1 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ingredient2, % as-is | −WCO | +WCO | WT1 | WT2 | WT3 | WT4 | WT5 | WT6 | WT7 | WT8 |
| Mechanically deboned chicken | 90.00 | 82.00 | 82.00 | 82.00 | 82.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Water | 10.00 | 10.00 | 10.00 | 10.00 | 10.00 | 66.67 | 75.00 | 80.00 | 83.33 | 85.71 |
| Ovabind | 0.00 | 8.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| GBF1 | 0.00 | 0.00 | 8.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| GBF2 | 0.00 | 0.00 | 0.00 | 8.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| GBF3 | 0.00 | 0.00 | 0.00 | 0.00 | 8.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| GBF4 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 33.33 | 25.00 | 20.00 | 16.67 | 14.29 |
1−WCO, water binding test negative control; +WCO, water binding test positive control; WT, water binding test.
2Ovabind, IsoNova, St. Louis, MO; GBF1, Pereg Banana Multi-purpose Flour, Clifton, NJ; GBF2, Duraci Farinha de Banana Verde, Lençóis Paulista, Brazil; GBF3, Edward & Sons Let’s Do Organic Green Banana Flour, Carpinteria, CA; GBF4, Peterson Dried Banana, Kalamazoo, MI.
The bolded numbers are the main difference among treatments which was the green banana flour inclusion rate.
Table 3.
Ingredient composition of gravy test treatments containing green banana flour
| Treatments1 | ||||||||
|---|---|---|---|---|---|---|---|---|
| Ingredient2, % as-is | GCO | GT1 | GT2 | GT3 | GT4 | GT5 | GT6 | GT7 |
| Water | 97.80 | 97.30 | 97.30 | 97.30 | 97.80 | 97.80 | 97.80 | 97.80 |
| Guar gum | 0.70 | 0.70 | 0.70 | 0.70 | 0.35 | 0.00 | 0.70 | 0.00 |
| Clear gel (waxy corn starch) | 0.50 | 0.00 | 0.00 | 0.00 | 0.25 | 0.50 | 0.00 | 0.00 |
| Palatability enhancer | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| GBF1 | 0.00 | 1.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| GBF2 | 0.00 | 0.00 | 1.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| GBF3 | 0.00 | 0.00 | 0.00 | 1.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| GBF4 | 0.00 | 0.00 | 0.00 | 0.00 | 0.60 | 0.70 | 0.50 | 1.20 |
1GCO, gravy test control; GT, gravy test.
2GBF1, Pereg Banana Multi-purpose Flour, Clifton, NJ; GBF2, Duraci Farinha de Banana Verde, Lençóis Paulista, Brazil; GBF3, Edward & Sons Let’s Do Organic Green Banana Flour, Carpinteria, CA; GBF4, Peterson Dried Banana, Kalamazoo, MI.
The bolded numbers are the main difference among treatments which was the green banana flour inclusion rate.
Chemical analyses.
GBF ingredients were analyzed in duplicate for the determination of DM, ash, organic matter (OM), acid hydrolyzed fat (AHF), crude protein (CP), total dietary fiber (TDF), SDF, insoluble dietary fiber (IDF), RS, gross energy (GE), and sugar profiles. DM and ash were analyzed according to AOAC (2006; methods 934.01 and 942.05). CP was calculated from the nitrogen values from Leco (TruMac N, Leco Corporation, St. Joseph, MI) according to AOAC (2006; method 992.15). AHF was analyzed according to AACC (1983) and Budde (1952). GE was analyzed with bomb calorimetry (Model 6,200, Parr Instruments Co., Moline, IL, USA). TDF, SDF, and IDF were analyzed according to the study by Prosky et al. (1992). Free sugar profile analyses were according to the methods from Churms (1982) and Kakehi and Honda (1989). Hydrolyzed sugar profiles were determined based on the methods from Oxley et al. (2004), Hudson et al. (1982), Kiho et al. (1986), and Fox et al. (1989). RS and total starch were analyzed based on AOAC method 2002.02 (McCleary and Monaghan, 2002) and AACC (2010); method 32-40.01).
Precision-fed rooster assay.
The true metabolizable energy corrected to zero nitrogen retention (TMEn) of the four GBF ingredients was determined by a precision-fed rooster assay (Parsons, 1985). Sixteen single-comb White Leghorn roosters were included in the assay with four roosters per treatment or ingredient. The roosters were housed individually in a temperature-controlled room in cages with wire floors. The light-to-dark cycle was set at 16:8. After 26 h of fasting, the roosters were tube-fed with a mixture of 15 g of GBF and 50 mL of water into their crops. The excreta were collected 48 h after feeding and were freeze-dried and ground for analysis. The TMEn was calculated according to the following equation (Parsons et al., 1982):
In the above equation, FEfed is the total GE (kcal) fed to the roosters; EEfed and EEfasted are the total energy excreted (kcal) in fed and fasted states, respectively; Nfed and Nfasted are the amount of nitrogen (g) retained by the rooster in fed and fasted states, respectively; 8.22 (kcal) is the GE per gram of nitrogen in uric acid; FI is the amount of ingredient fed (g). The GE of rooster excreta was also determined by using bomb calorimetry (Model 6,200, Parr Instruments Co., Moline, IL, USA). Data calculated for TMEn were analyzed with MIXED model procedures of SAS version 9.4 (SAS Institute Inc., Cary, NC). Treatment was the fixed effect and rooster was the random effect. The alpha was set at 0.05 for significance.
Texture analyses.
Commercially canned foods and Loaf Test canned foods were opened right before testing and were analyzed in their original cans. The commercial canned foods used in the study were chosen for a similar container type and product type (loaf or pâté) to the test foods and are listed in the supplemental document. Canned foods were tested for texture profile indices with a modified back extrusion test (Hagen-Plantinga et al., 2017; Dainton et al., 2021). The analysis was performed with a texture analyzer (TA.HD Plus Connect, Stable Micro System Ltd., Godalming, UK) fitted with a 30 kg load cell. The conditions of the TA were as follows: pretest speed, 1.0 mm/s; test speed, 5.0 mm/s; post-test speed, 5.0 mm/s; strain, 50%; time, 5.0 s; trigger type, auto; and trigger force, 5 g. The probe used during these measurements was TA-3 (2.54 cm diameter acrylic cylinder, Stable Micro System Ltd., Godalming, UK). Values for hardness, adhesiveness, springiness, cohesiveness, gumminess, and resilience were determined according to the standard program in texture profile analysis. Hardness was the peak force that occurred during the first compression. Adhesiveness was the peak negative force when the probe was withdrawn, indicating the stickiness of the product. Springiness was the ability of the product to regain its former shape between the first and the second compression. Cohesiveness was the resistance to the second deformation of the product relative to the resistance to the first deformation. Resilience was the extent the product regained its original height. Gumminess was calculated by hardness × cohesiveness. With the same TA, the canned foods were also tested with a different probe TA-47 (pasta blade, Stable Micro System Ltd., Godalming, UK) for firmness/toughness, which was the peak force of the compression. The conditions were as follows: pretest speed, 1.0 mm/s; test speed, 2.0 mm/s; post-test speed, 5.0 mm/s; strain, 50%; time, 5.0 s; trigger type, auto; and trigger force, 5 g. Commercial foods were tested in triplicates while the replicates of loaf test foods ranged from one to three, based on availability.
Color analysis.
The color of commercially canned foods, water binding test canned foods, and gravy test canned foods were accessed by using a colorimeter (LabScan XE, Hunter Associates Laboratory Inc., Reston, VA, USA) in duplicates. The CIE L*a*b* was used for the color parameter. L* was calculated for brightness (0 = complete black to 100 = perfect white). a* was calculated for greenness (a* < 0) and redness (a* > 0). b* represented blueness (b* < 0) and yellowness (b* > 0). The color differences were also calculated as shown below, where dL* is the brightness difference, da* is the redness/greenness difference, db* is the difference in blueness/yellowness, and dE* is the total color difference. LSMP, aSMP, and bSMP are the measurements from the samples. LSTD, aSTD, and bSTD are the readings from the standards or controls. Commercially canned foods and water binding test canned foods were opened before the testing and were cut to fit the plate. gravy test canned foods that were chunks in gravy were added to fill the plate and 8 mL of gravy was added to the plate for those that were liquid gravy.
Study 2
In vitro experiment.
From study 1, GBF4 (Dried Banana, the Peterson Company, Kalamazoo, MI) was chosen for further analysis because of its commercial availability and its high RS content. Prior to the animal feeding trial, a third stage in vitro fiber fermentation experiment was conducted to test the fermentability of GBF along with the control fibers: cellulose (Millipore Sigma, St. Louis, MO) and beet pulp (Michigan Sugar Company, Bay City, MI). The control fibers were chosen because both cellulose and beet pulp are widely used in pet foods as fiber sources. In the current study, cellulose served as a control with mostly insoluble fiber of low fermentability and beet pulp served as a fiber with an ideal ratio of insoluble fiber and soluble fiber. Fecal samples were collected from three purpose-bred healthy dogs consuming a commercial extruded diet for adult dogs at maintenance and prepared as the inoculum (Bourquin et al., 1993). Substrates were added to the inoculum and fermented in triplicates. Tubes with no substrate were also included to allow for corrections. The tubes were incubated at 37 °C for 0, 2, 4, 8, or 12 h and submerged in an ice bath at the time points to stop fermentation. The samples were then analyzed for pH and short-chain fatty acid (SCFA) concentrations. Concentrations of SCFA were measured with gas chromatography using a modified method of Sunvold et al. (1995). Hewlett-Packard gas chromatograph (Model 5890A Hewlett Packard, Avondale, PA) equipped with a flame ionization detector on a column (1.8 m × 4 mm i.d.) packed with GP 10% SP-1,200/1% H3PO4 on Chromosorb W AW, 80 to 100 mesh (Supelco, Bellefonte, PA) was used. Nitrogen, the carrier gas, had a flow rate of 45 mL/min. Temperatures were set at 125 °C for the oven, 175 °C for the injection port, and 180 °C for the detector port.
Diets.
Five diets were formulated to meet or exceed the AAFCO (2020) nutrient profile for adult feline maintenance. The diets were formulated with similar ingredient compositions, except for the carbohydrate sources being tested, to achieve similar chemical compositions among all diets. The Peterson Dried Banana (GBF4) from study 1 was used to formulate the diets. The treatment diets were formulated with 4% rice flour (rice), 4% dehydrated potato flakes (potato), 1% GBF plus 3% rice flour (1% GBF), 2% GBF plus 2% rice flour (2% GBF), or 4% GBF (Table 4). All diets were made at the Food Science and Human Nutrition Pilot Processing Plant at the University of Illinois at Urbana-Champaign. Dry and wet ingredients were mixed and blended homogeneously together before being precooked in a jacketed steam kettle. The dough was heated to 50 °C for 10 min with the agitator speed set at 60 Hz before filling the standard number two cans. All diets were mixed and blended as a single batch by design to minimize nutritional differences among batches. Once the cans were filled (500 ± 5 g) and hermetically sealed, they were processed for approximately 1 h at 121.1 °C until achieving process lethality (F0) of eight in an Allpax Gentle Motion Retort (Allpax, Covington, LA). The diets were retorted in batches determined by the capacity of the machine, following the same processing conditions. The temperature during processing was monitored by wireless DataTrace temperature probes (Mesa Labs, Lakewood, CO). Three temperature probes were placed to measure the inner temperature of the canned foods in each batch to ensure consistent processing among cans and batches. Readings were continuous throughout the retort process.
Table 4.
Ingredient composition and chemical composition of treatment diets containing traditional starch sources and green banana flour for adult felines
| Treatment diet | |||||
|---|---|---|---|---|---|
| Green banana flour | |||||
| Rice | Potato | 1% | 2% | 4% | |
| Ingredient, % as-is | |||||
| Chicken mix1 | 39.30 | 39.30 | 39.30 | 39.30 | 39.30 |
| Water | 30.23 | 30.23 | 30.23 | 30.23 | 30.23 |
| Mechanical separated chicken | 20.00 | 20.00 | 20.00 | 20.00 | 20.00 |
| Chicken heart and liver | 5.00 | 5.00 | 5.00 | 5.00 | 5.00 |
| Dehydrated potato flakes | 0.00 | 4.00 | 0.00 | 0.00 | 0.00 |
| Rice flour | 4.00 | 0.00 | 3.00 | 2.00 | 0.00 |
| Green banana flour | 0.00 | 0.00 | 1.00 | 2.00 | 4.00 |
| Carrageenan | 0.35 | 0.35 | 0.35 | 0.35 | 0.35 |
| Cassia gum | 0.35 | 0.35 | 0.35 | 0.35 | 0.35 |
| Guar gum | 0.20 | 0.20 | 0.20 | 0.20 | 0.20 |
| Potassium chloride | 0.15 | 0.15 | 0.15 | 0.15 | 0.15 |
| Mineral premix2 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 |
| Salt | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 |
| Choline chloride 70% | 0.09 | 0.09 | 0.09 | 0.09 | 0.09 |
| Vitamin premix3 | 0.08 | 0.08 | 0.08 | 0.08 | 0.08 |
| Taurine | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 |
| Chemical composition | |||||
| Dry matter (DM), % | 23.0 | 23.1 | 22.8 | 23.1 | 22.7 |
| ----- % DM basis ----- | |||||
| Organic matter | 90.0 | 89.8 | 89.1 | 89.3 | 89.2 |
| Acid hydrolyzed fat | 37.0 | 37.3 | 37.6 | 37.8 | 37.2 |
| Crude protein | 36.6 | 37.0 | 36.7 | 36.3 | 36.8 |
| Total dietary fiber | 11.5 | 10.8 | 10.5 | 11.8 | 12.1 |
| Soluble dietary fiber | 5.7 | 5.0 | 5.1 | 5.6 | 4.5 |
| Insoluble dietary fiber | 5.8 | 5.9 | 5.4 | 6.2 | 7.6 |
| Gross energy, kcal/g | 6.2 | 6.2 | 6.4 | 6.3 | 6.3 |
1Chicken mix: chicken back and neck.
2Provided per kilogram diet: 30.0 mg iron (iron proteinate), 28.1 mg zinc (zinc proteinate), 3.2 mg copper (copper proteinate), 2.9 mg manganese (manganese proteinate), 0.7 mg potassium (potassium iodide), and 0.1 mg sodium (sodium selenite).
3Provided per kilogram diet: 30 IU vitamin E, 26.7 mg thiamin (thiamin mononitrate), 10.0 mg niacin, 2.7 mg pantothenic acid (d-calcium pantothenate), 1.5 mg pyridoxine (pyridoxine hydrochloride), 1.3 mg riboflavin, 1,560 IU vitamin A, 293.3 μg folic acid, 23.3 μg biotin, 555.5 IU vitamin D3 supplement, and 6.7 μg vitamin B12 supplement.
The bolded numbers are the main difference in the formulation which is the carbohydrate source.
Animals and experimental design.
Thirty-nine adult domesticated cats (15 females and 24 males, mean age 5.6 ± 2.11 yr, mean body weight 5.3 ± 1.24 kg, mean body condition score 6.6 ± 0.94) were included in the study and were randomized into one of the five treatment groups. At the beginning of the study, there was a 7-d diet adaptation period in which the rice diet was fed to all the cats. A fresh fecal collection was performed after the diet adaptation period. After the fresh fecal collection (day 0), all cats were fed their assigned diets for 21 d to maintain body weight. During the last 4 d of this treatment period, a total fecal collection was performed while a fresh fecal sample was also acquired.
Cats were group-housed except during feeding times. Feeding occurred twice a day from 0800 to 1000 hours and from 1500 to 1700 hours. During feeding times, cats were housed individually in stainless steel cages. Cats had free access to water at all times. Food was weighed right before feeding, and food refusals were weighed after each feeding time. Body weights and body condition scores were measured each week. Cats were housed at Kennelwood Inc. in temperature-controlled rooms with a 9 h light and 15 h dark cycle. Animals had access to enrichments in the room such as scratching posts and cat trees and daily social interactions with humans.
Sample collection.
For the duration of the 4 d total fecal collections, cats were housed individually. All feces were collected during the period and composited for each cat. Each fecal sample was weighed and evaluated for fecal score (1 = hard, dry pellets; 2 = hard formed, remains firm and soft; 3 = soft, formed and moist stool; 4 = soft, unformed stool; or 5 = watery, liquid that can be poured). Then, the samples were stored at −20 °C for later laboratory analysis.
A fresh fecal sample was collected from each cat after the diet adaptation period (day 0) and during the total fecal collection at the end of the experimental period. The fresh fecal samples were collected within 15 min of defecation. The samples were weighed and evaluated for pH and score. Then, they were aliquoted to determine DM, ammonia, SCFA, branched-chain fatty acid (BCFA), phenol, and indole concentrations. DM was determined by drying duplicates of approximately 2 g of feces in a forced-air oven at 105 °C. For ammonia, SCFA, and BCFA concentrations, 3 g of feces were mixed with 3 mL of 2 N hydrochloric acid in a Nalgene bottle and stored at −20 °C for later analysis. For phenol and indole concentrations, approximately 2 g of fecal samples were weighed into plastic test tubes in duplicates, covered with parafilm, and stored at −20 °C for later analysis. A subsample of each fresh fecal sample was also stored at −80 °C for microbiota analysis.
On days 0 and 21, a fasted blood sample was collected from each cat to evaluate their health status. Cats were sedated with 0.08 mL/kg BW of Dexdomitor (Orion Corporation, Espoo, Finland) and 5 mL of blood was collected via jugular vein puncture. The cats were reversed with 0.08 mL/kg BW of Antisedan (Orion Corporation, Espoo, Finland) after blood was collected. During collections, 1 mL of blood was placed in EDTA vacutainer tubes and 4 mL was placed in serum separator tubes (Becton, Dickinson and Company, Franklin Lakes, NJ). Blood samples were sent to the Clinical Pathology Laboratory at the College of Veterinary Medicine at the University of Illinois (Urbana, IL) for analysis.
Sample preparation.
Experimental diets were subsampled, freeze-dried in FreeZone Bulk Tray Dryer (Labconco Corporation, Kansas City, MO), and ground with a Wiley mini-mill (Thomas Scientific, Swedesboro, NJ) through a 2 -mm screen. Composited total fecal samples were partially dried at 57 °C in a forced-air oven, and also ground with a Wiley mini-mill with a 2 mm screen.
Chemical analysis.
Experimental diets and total fecal samples were determined for DM, ash, OM, AHF, CP, TDF, and GE. Diet samples were also analyzed for IDF and SDF. All analytic methods were the same as described in study 1.
Fecal SCFA and BCFA concentrations were measured with the same methods as described in the in vitro fermentation experiment. Measurements of fecal phenol and indole were adapted from Flickinger et al. (2003). Fecal ammonia concentration was measured according to Chaney and Marbach (1962).
Fecal microbiota analysis.
Total DNA was extracted from fresh fecal samples using a DNeasy PowerLyzer PowerSoil Kit (Qiagen, Germantown, MD). Invitrogen Qubit 4 Fluorometer (Thermo Fisher Scientific Inc., Waltham, MA) was used to measure DNA concentration. A Fluidigm Access Array (Fluidigm Corporation, South San Francisco, CA) combined with Roche High Fidelity Fast Start Kits (Roche, Indianapolis, IN) were used to amplify the 16S rRNA gene. The primers 515F (5ʹ-GTGYCAGC MGCCGCGGTAA-3ʹ) and 806R (5ʹ-GGACTACNVGG GTWTCTAAT-3ʹ), targeting a 292-bp fragment of V4 region, were used for amplification (primers synthesized by IDT Corp., Coralville, IA). The quality of the amplicons’ regions and sizes was confirmed by Fragment Analyzer (Advanced Analytics, Ames, IA). The pooled DNA samples were selected by size on a 1% agarose E-gel (Life Technologies, Grand Island, NY) and extracted using a gel extraction kit (QIAGEN, Germantown, MD). The pooled, size-selected, and cleaned products were then run on an Agilent Bioanalyzer to confirm the appropriate profile and mean size. The Roy J. Carver Biotechnology Center at the University of Illinois performed Illumina sequencing on a MiSeq using a V3 reagent (Illumina Inc., San Diego, CA). A FASTX-Toolkit removed the Fluidigm tags. Analysis of sequences was completed using QIIME 2.0. Alpha diversity was assessed by Pielou evenness, Faith’s phylogenetic diversity, and Shannon entropy. Bray-Curtis distance (Bray and Curtis, 1957) and UniFrac distance (Hamady et al., 2010), weighted and unweighted, were also calculated.
Texture analysis.
The consistency of the precooked batter from the jacketed steam kettle was measured with Bostwick Consistometer (CSC Scientific 24925000, Fairfax, VA) in triplicates. The batter was loaded in the cell, then the gate was opened, and a 30-s timer started. The distance the sample flowed during the 30-s interval was measured. Retorted experimental canned foods were opened right before the testing with a TA (TA.HD Plus Connect, Stable Micro System Ltd., Godalming, UK) and were evaluated in their original cans in triplicates. Canned foods were tested for texture profile indices with probes TA-3 (1-inch diameter acrylic cylinder, Stable Micro System Ltd., Godalming, UK) and TA-47 (pasta blade, Stable Micro System Ltd., Godalming, UK). The settings were the same as described in study 1.
Color analysis.
The color of each experimental diet before and after retort was accessed in triplicates by using a colorimeter (LabScan XE, Hunter Associates Laboratory Inc., Reston, VA, USA). Cans were opened right before measurements and subsamples of each canned food were placed on the measuring tray for analysis. The same parameters were measured as described in study 1.
Statistical analysis.
The MIXED model procedures of SAS version 9.4 (SAS Institute Inc., Cary, NC) were used to analyze all data. For the in vitro study, dietary fiber was included in the model as the fixed effect. For texture analysis and ATTD, the statistical model included dietary treatment as the fixed effect and animal as the random effect. For color analysis, fecal characteristics, and fecal metabolites, a two-factorial ANOVA was used with dietary treatment and time as the fixed effects and animal as the random effect. Data normality was checked with the UNIVARIATE procedure. Tukey adjustment was used to control experiment-wise error. Data with non-normal distribution were transformed to determine statistical significance. The alpha was set at 0.05 for significance. Pooled standard errors of the mean were calculated from the MIXED model procedures.
Results
Study 1
For the chemical composition (Table 5), overall, all 4 GBF were high in RS and also low in protein and fat. GBF 2 had a higher ash content (7.1%) compared with the others (3.3% to 4.6%), a higher AHF concentration (3.4%) while the others had a concentration close to 1%, and a higher TDF content of 28.9% as the others had 11% to 16%. The insoluble and soluble fiber content ratios were also variable. The ratio was 1.0 for GBF1 and GBF4, 2.0 for GBF2, and 0.8 for GBF3. The concentrations of RS were higher in GBF1 (38.3%) and GBF4 (40.9%) in contrast with GBF3 (33.5%) and GBF2 (28.4%). GE, 4.2 to 4.3 kcal/g, was similar among the GBF sources tested herein.
Table 5.
Chemical composition of green banana flour ingredients
| Ingredients1 | ||||
|---|---|---|---|---|
| Item | GBF1 | GBF2 | GBF3 | GBF4 |
| Dry matter (DM), % | 92.6 | 90.0 | 90.1 | 86.9 |
| % DM basis | ||||
| Organic matter | 96.7 | 92.9 | 95.5 | 96.0 |
| Ash | 3.3 | 7.1 | 4.6 | 4.0 |
| Acid hydrolyzed fat | 1.3 | 3.4 | 1.4 | 1.2 |
| Crude protein | 4.0 | 5.2 | 5.6 | 3.9 |
| Total dietary fiber | 12.9 | 28.9 | 15.5 | 11.5 |
| Soluble dietary fiber | 6.5 | 9.6 | 8.5 | 5.8 |
| Insoluble dietary fiber | 6.4 | 19.3 | 7.0 | 5.7 |
| Insoluble: soluble dietary fiber ratio | 1.0 | 2.0 | 0.8 | 1.0 |
| Starch | 40.2 | 27.0 | 40.7 | 31.0 |
| Resistant starch | 38.3 | 28.4 | 33.5 | 40.9 |
| Gross energy, kcal/g | 4.2 | 4.3 | 4.2 | 4.3 |
| Free sugar | ||||
| Fructose | 0.30 | 1.99 | 0.58 | 0.50 |
| Glucose | 0.60 | 2.04 | 1.01 | 0.78 |
| Sucrose | 1.25 | 3.43 | 1.73 | 0.50 |
| Lactose | 0.00 | 0.00 | 0.00 | 0.00 |
| Maltose | 0.00 | 0.00 | 0.00 | 0.00 |
| Hydrolyzed sugar | ||||
| Rhamnose | 0.00 | 0.00 | 0.00 | 0.00 |
| Ribose | 0.00 | 0.00 | 0.00 | 0.00 |
| Fucose | 0.00 | 0.00 | 0.00 | 0.00 |
| Arabinose | 0.16 | 0.67 | 0.42 | 0.28 |
| Xylose | 0.63 | 0.99 | 0.70 | 0.38 |
| Mannose | 2.68 | 3.81 | 2.62 | 2.90 |
| Glucose | 84.20 | 46.11 | 85.16 | 90.97 |
| Galactose | 3.12 | 4.46 | 3.72 | 4.47 |
1GBF1, Pereg Banana Multi-purpose Flour, Clifton, NJ; GBF2, Duraci Farinha de Banana Verde, Lençóis Paulista, Brazil; GBF3, Edward & Sons Let’s Do Organic Green Banana Flour, Carpinteria, CA; GBF4, Peterson Dried Banana, Kalamazoo, MI.
For free sugar concentrations (Table 5), GBF1, GBF3, and GBF4 had less than 1% fructose and 1% or less glucose. On the other hand, GBF2 had around 2% of both fructose and glucose. Sucrose concentrations varied among the GBF sources, from 3.43% for GBF2 to 0.50% for GBF4. None of the GBFs had free lactose or maltose. For hydrolyzed sugars, glucose concentrations were around 85% to 90% for GBF1, GBF3, and GBF4, but lower in GBF2 (46%). Galactose concentrations of all GBF were 3% to 5%. Mannose concentrations ranged from 2% to 4%. Arabinose and xylose concentrations were all less than 1%. Rhamnose, ribose, and fucose were not detected in these ingredient sources.
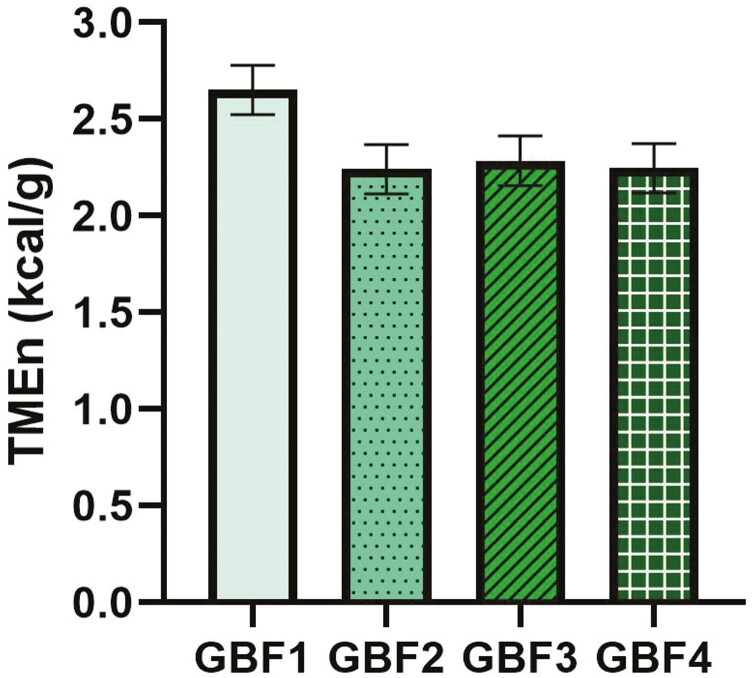
The TMEn values of the 4 GBF, ranging from 2.2 to 2.7 kcal/g, did not differ from each other (P > 0.05; Figure 1).
Figure 1.
True metabolizable energy corrected to zero nitrogen retention of different sources of green banana flour (GBF). GBF1, Pereg Banana Multi-purpose Flour; GBF2, Duraci Farinha de Banana Verde; GBF3, Let’s Do Organic GBF; GBF4, Peterson Dried Banana. No treatment effect was observed (P > 0.05).
Hardness, adhesiveness, springiness, resilience, and firmness/toughness of Loaf Test canned foods were within the values determined from commercial diets (Table 6–7). Cohesiveness was generally lower in the loaf test diets than in the commercial foods. Comparing loaf tests with the same source of GBF, higher inclusion concentrations tended to result in higher springiness.
Table 6.
Texture profile analysis of commercial diets and loaf test treatments containing green banana flour
| Treatments1 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Texture parameters2 | CD1 | CD2 | CD3 | CD4 | LCO | LT1 | LT2 | LT3 | LT4 | LT5 | LT6 | LT7 | LT8 | LT9 | LT10 |
| Hardness, kg | 0.46 | 0.80 | 0.87 | 1.25 | 1.33 | 1.22 | 1.21 | 1.24 | 1.17 | 1.32 | 0.94 | 0.66 | 0.68 | 1.40 | 0.72 |
| Adhesiveness, kg × s | −0.87 | −0.59 | −0.03 | −0.04 | −0.04 | −0.06 | −0.06 | −0.02 | −0.05 | −0.05 | −0.31 | −0.03 | −0.02 | −0.01 | −0.02 |
| Springiness, % | 0.75 | 0.67 | 0.80 | 0.91 | 0.68 | 0.64 | 0.80 | 0.70 | 0.82 | 0.66 | 0.74 | 0.74 | 0.76 | 1.15 | 2.85 |
| Cohesiveness, % | 0.45 | 0.44 | 0.40 | 0.36 | 0.21 | 0.19 | 0.21 | 0.22 | 0.23 | 0.20 | 0.25 | 0.19 | 0.20 | 0.21 | 0.19 |
| Resilience, % | 0.05 | 0.10 | 0.14 | 0.11 | 0.06 | 0.06 | 0.06 | 0.08 | 0.08 | 0.06 | 0.06 | 0.04 | 0.05 | 0.05 | 0.04 |
| Gumminess | 0.21 | 0.35 | 0.34 | 0.45 | 0.28 | 0.23 | 0.26 | 0.27 | 0.27 | 0.27 | 0.24 | 0.12 | 0.13 | 0.29 | 0.14 |
1CD1, blue buffalo divine delights porterhouse flavor; CD2, purina friskies indoor pate chicken dinner; CD3, nutro steamed chicken and turkey recipe; CD4, cesar oven roasted chicken flavor; LCO, loaf test control; LT, loaf test.
2Hardness: the peak force that occurs during the first compression; adhesiveness: the stickiness of the product; springiness: the ability of the product to regain its former shape during the wait time between the first deformation and the second compression; cohesiveness: the resistance to the second deformation of the product relative to the resistance to the first deformation; resilience: the extent the product regain its original height; gumminess: applies to semi-solids, hardness * cohesiveness.
Table 7.
Firmness/toughness from knife cutting of commercial diets and loaf tests containing green banana flour
| Treatments | Firmness/toughness, kg |
|---|---|
| Cesar filet mignon flavor | 1.07 |
| Cesar beef recipe | 1.34 |
| Cesar porterhouse steak flavor | 1.01 |
| Cesar grilled chicken flavor | 1.01 |
| Cesar oven-roasted chicken flavor | 0.92 |
| Blue buffalo divine delights porterhouse flavor | 0.17 |
| Purina friskies indoor pate chicken dinner | 0.50 |
| Nutro steamed chicken and Turkey recipe | 0.61 |
| Loaf test control (LCO) | 1.01 |
| Loaf test 1 (LT1) | 0.94 |
| Loaf test 2 (LT2) | 0.80 |
| Loaf test 3 (LT3) | 0.99 |
| Loaf test 4 (LT4) | 1.02 |
| Loaf test 5 (LT5) | 0.86 |
| Loaf test 6 (LT6) | 0.82 |
| Loaf test 7 (LT7) | 0.98 |
| Loaf test 8 (LT8) | 1.03 |
| Loaf test 9 (LT9) | 0.62 |
| Loaf test 10 (LT10) | 0.56 |
For water binding test canned diets, hardness, adhesiveness, and firmness/toughness were higher with higher GBF4 concentrations (Table 8). Compared to water binding test positive control (+WCO), all the other water binding test diets and the commercial diets were darker on the top surface (dL* = −11.88 to −38.86) and in the middle (dL* = −7.23 to −34.73; Table 9). The water binding tests were lighter than the commercial diets both on the top surface and in the middle. On the top surface, water binding tests and commercial diets had more redness than + WCO. The redness was similar between water binding tests and commercial diets. For yellowness, there was a high variance among all the diets. The water binding test canned diets containing GBF3 had the most similar extent of yellowness compared with the commercial diets.
Table 8.
Texture profile analysis and firmness/toughness from knife cutting for water binding test treatments containing green banana flour
| Treatments1 | |||||
|---|---|---|---|---|---|
| Texture parameters | WT4 | WT5 | WT6 | WT7 | WT8 |
| Hardness, kg | 3.54 | 1.59 | 2.63 | 2.00 | 0.90 |
| Adhesiveness, kg × s | 0.00 | −0.05 | −0.06 | −0.09 | −0.10 |
| Springiness, % | 0.79 | 0.83 | 0.98 | 0.98 | 0.88 |
| Cohesiveness, % | 0.15 | 0.18 | 0.39 | 0.31 | 0.32 |
| Resilience, % | 0.10 | 0.07 | 0.13 | 0.14 | 0.11 |
| Gumminess | 0.52 | 2.92 | 1.02 | 0.62 | 0.29 |
| Firmness/toughness, kg | 5.18 | 1.66 | 4.57 | 0.35 | 0.49 |
1WT, water binding test.
2Hardness: the peak force that occurs during the first compression; adhesiveness: the stickiness of the product; springiness: the ability of the product to regain its former shape during the wait time between the first deformation and the second compression; cohesiveness: the resistance to the second deformation of the product relative to the resistance to the first deformation; resilience: the extent the product regain its original height; gumminess: applies to semi-solids, hardness * cohesiveness.
Table 9.
Color analysis of the top surface and middle of commercial diets and water binding tests containing green banana flour
| Color parameter | |||||||
|---|---|---|---|---|---|---|---|
| Treatments1 | L* | a* | b* | dL* | da* | db* | dE* |
| Top surface | |||||||
| +WCO top (standard) | 63.41 | 4.67 | 17.16 | NA | NA | NA | NA |
| −WCO top | 51.53 | 11.13 | 20.17 | −11.88 | 6.47 | 3.01 | 13.86 |
| WT1 top | 44.70 | 6.03 | 16.68 | −18.72 | 1.37 | −0.48 | 18.77 |
| WT2 top | 35.60 | 4.81 | 13.58 | −27.82 | 0.15 | −3.58 | 28.04 |
| WT3 top | 40.38 | 8.71 | 20.15 | −23.03 | 4.04 | 3.00 | 23.57 |
| CD1 top | 31.04 | 7.99 | 21.29 | −32.38 | 3.32 | 4.13 | 32.81 |
| CD5 top | 31.55 | 7.98 | 17.64 | −31.86 | 3.31 | 0.48 | 32.04 |
| CD6 top | 34.24 | 8.42 | 18.25 | −29.18 | 3.76 | 1.09 | 29.44 |
| CD7 top | 24.55 | 11.29 | 24.26 | −38.86 | 6.63 | 7.10 | 40.05 |
| Middle | |||||||
| +WCO mid (standard) | 61.85 | 7.89 | 15.90 | NA | NA | NA | NA |
| −WCO mid | 54.62 | 9.92 | 17.19 | −7.23 | 2.03 | 1.30 | 7.62 |
| WT1 mid | 48.81 | 8.76 | 15.28 | −13.04 | 0.88 | −0.62 | 13.08 |
| WT2 mid | 41.02 | 7.58 | 14.06 | −20.83 | −0.32 | −1.84 | 20.91 |
| WT3 mid | 45.70 | 10.00 | 17.54 | −16.15 | 2.11 | 1.65 | 16.36 |
| CD1 mid | 34.20 | 7.61 | 17.58 | −27.65 | −0.29 | 1.69 | 27.70 |
| CD5 mid | 30.17 | 8.65 | 18.14 | −31.68 | 0.76 | 2.25 | 31.77 |
| CD6 mid | 36.75 | 7.47 | 16.20 | −25.10 | −0.43 | 0.31 | 25.10 |
| CD7 mid | 27.12 | 10.04 | 19.59 | −34.73 | 2.15 | 3.70 | 34.99 |
1+WCO, water binding test positive control; −WCO, water binding test negative control; WT, water binding test; CD1, blue buffalo divine delights porterhouse flavor; CD5, purina friskies indoor pate poultry platter; CD6, cesar Turkey recipe; CD7, cesar filet mignon flavor.
For the liquid gravy, gravy test canned food containing GBF1, GBF2, and GBF3 had more redness and yellowness than the control which had no GBF. Gravy test canned food containing GBF1 and GBF3 were lighter than the control whereas the gravy with GBF2 was darker (Table 10). For chunks in gravy, the commercial diets were darker, redder, and more yellow than the gravy test diets.
Table 10.
Color analysis of commercial diets and gravy tests containing green banana flour
| Color parameter | |||||||
|---|---|---|---|---|---|---|---|
| Treatments1 | L* | a* | b* | dL* | da* | db* | dE* |
| Liquid gravy | |||||||
| GCO (standard) | 22.87 | −0.52 | −0.06 | NA | NA | NA | NA |
| GT1 | 25.49 | 1.60 | 4.10 | 2.61 | 2.13 | 4.16 | 5.35 |
| GT2 | 20.85 | 3.85 | 8.72 | −2.03 | 4.37 | 8.78 | 10.01 |
| GT3 | 25.24 | 1.64 | 6.13 | 2.36 | 2.16 | 6.19 | 6.97 |
| Chunks in gravy | |||||||
| GCO (standard) | 41.80 | 4.03 | 15.00 | NA | NA | NA | NA |
| GT4 | 41.87 | 4.93 | 14.50 | 0.07 | 0.90 | −0.51 | 1.03 |
| GT5 | 41.46 | 4.52 | 14.13 | −0.35 | 0.49 | −0.88 | 1.06 |
| GT6 | 42.48 | 4.72 | 15.22 | 0.68 | 0.69 | 0.22 | 0.99 |
| GT7 | 41.27 | 4.47 | 14.73 | −0.54 | 0.44 | −0.28 | 0.74 |
| CD8 | 31.03 | 9.32 | 18.95 | −10.78 | 5.29 | 3.95 | 12.63 |
| CD9 | 34.11 | 10.34 | 19.86 | −7.69 | 6.31 | 4.86 | 11.07 |
| CD10 | 33.53 | 10.23 | 20.33 | −8.27 | 5.33 | 5.33 | 11.63 |
1GCO, gravy test control; GT1, gravy test 1; GT2, gravy test 2; GT3, gravy test 3; GT4, gravy test 4; GT5, gravy test 5; GT6, gravy test 6; GT7, gravy test 7; CD8, Cesar Filets in Sauce Prime Rib Flavor; CD9, Cesar Filets in Sauce Filet Mignon Flavor; CD10, Cesar Filets in Sauce New York Strip Flavor.
Study 2
For the in vitro experiment, there was a significant interaction of treatment fiber and time (P < 0.05) for pH change and SCFA production. There was no difference in SCFA production or pH among groups at 2 h (P > 0.05); however, differences were seen at 4, 8, and 12 h (P < 0.05). Over the 12 h fermentation period, there was a numerical decrease in pH for beet pulp and GBF (Figure 2). At the same time points of 4, 8, and 12 h, the pH decrease in GBF samples was less than in beet pulp (P < 0.05). There were increased total SCFA concentrations, including acetate, propionate, and butyrate, over the 12 h seen in beet pulp and GBF samples. The production in all SCFAs was greater for beet pulp than GBF at the same time points 8 and 12 h (P < 0.05), which corresponded to the pH change.
Figure 2.
Short-chain fatty acid production and pH change during the in vitro fermentation experiment for green banana flour. * Mean values are significantly different among treatments (P < 0.05).
All treatment diets were formulated to have similar ingredient compositions, except for the test carbohydrate sources. The test carbohydrate sources (rice flour, dehydrated potato, and GBF) were analyzed for their macronutrient contents (Table 11). All three carbohydrate sources had lower than 2% of AHF concentration. Rice flour and dehydrated potato had higher CP contents (8.6% and 9.4%, respectively) than GBF (3.9%) while dehydrated potato and GBF had higher TDF contents (12.8% and 11.5%, respectively) than rice flour (4.9%). The chemical compositions of the treatment diets were confirmed by proximate analysis and the results showed that the nutrient compositions were similar among treatments (Table 4). DM contents ranged from 22.7% to 23.1% and OM contents ranged from 89.1% to 90.0%. AHF contents were between 37.2% and 37.8%. CP content ranged from 36.3% to 37.0%. TDF ranged from 10.5% to 11.8%. The rice, potato, 1% GBF, and 2% GBF diets had similar soluble and IDF ratios, whereas the 4% GBF diet had a slightly higher insoluble fiber content.
Table 11.
Chemical composition of traditional carbohydrate sources and green banana flour ingredient
| Item | Rice flour1 | Dehydrated potato flakes2 | Green banana flour (GBF4)3 |
|---|---|---|---|
| Dry matter (DM), % | 89.5 | 90.9 | 86.9 |
| % DM basis | |||
| Organic matter | 99.2 | 96.9 | 96.0 |
| Acid hydrolyzed fat | 1.6 | 1.0 | 1.2 |
| Crude protein | 8.6 | 9.4 | 3.9 |
| Total dietary fiber | 4.9 | 12.8 | 11.5 |
| Soluble dietary fiber | 1.8 | 2.8 | 5.8 |
| Insoluble dietary fiber | 3.1 | 10.0 | 5.7 |
| Insoluble: soluble dietary fiber ratio | 1.7 | 3.6 | 1.0 |
| Gross energy, kcal/g | 4.3 | 4.2 | 4.3 |
1Carbohydrate content of rice flour according to USDA: 91.0% DM basis.
2Carbohydrate content of dehydrated potato flakes according to USDA: 86.9% DM basis.
3GBF4, Peterson Dried Banana, Kalamazoo, MI. Total starch content of green banana flour: 71.9% DM basis.
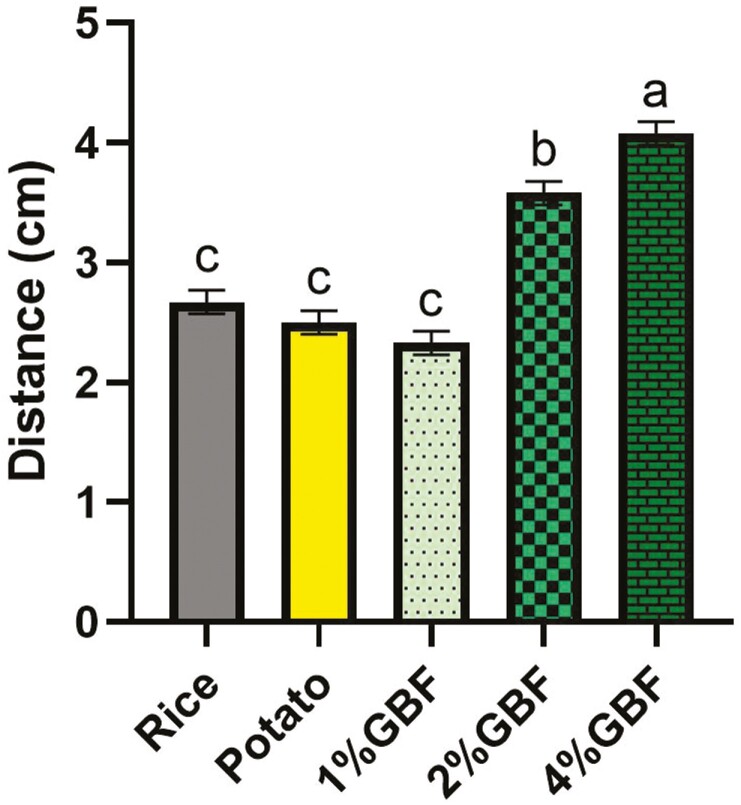
The consistency of the batter was the thinnest in the 4% GBF diet with a longer distance measured (P < 0.05), followed by the 2% GBF diet, while the rice, potato, and 1% GBF diets did not differ (P > 0.05) from each other (Figure 3). The texture profile analysis showed there were differences (P < 0.05) in hardness, adhesiveness, cohesiveness, resilience, and gumminess among diets (Table 12). The rice and potato diets had higher hardness (P < 0.05) of 1.64 and 1.58 kg, respectively, whereas the hardness of the three GBF diets ranged from 1.15 to 1.28 kg. The adhesiveness of the three GBF diets (−1.35 to −1.57 kg × s) was between the rice (−1.92 kg × s) and potato (−1.21 kg × s) diets. A treatment effect was noted for cohesiveness with 4% GBF (0.57) having greater cohesiveness in comparison with the potato diet (0.50; P < 0.05). In contrast, the potato diet showed a higher (P < 0.05) resilience (0.06) than the 2% GBF and 4% GBF diets (0.04). The rice diet had a higher gumminess of 0.87 while the potato and GBF diets were comparable, ranging from 0.65 to 0.79 (P < 0.05). Firmness/toughness analyzed with the knife probe showed that the rice diet was the highest in this parameter (0.87 kg) compared with the other diets (0.68 to 0.76 kg; P < 0.05).
Figure 3.
Consistency of the treatment diet batter containing traditional starch source and green banana flour measured by distance traveled in Bostwick Consistometer. a-c Means with different superscript letters are different (P < 0.05).
Table 12.
Texture and color analyses of treatment diets containing traditional starch source and green banana flour for adult felines
| Treatment diet | SEM1 | |||||
|---|---|---|---|---|---|---|
| Green banana flour | ||||||
| Item2 | Rice | Potato | 1% | 2% | 4% | |
| Texture analysis | ||||||
| Hardness, kg | 1.64a | 1.58a | 1.28b | 1.27b | 1.15b | 0.058 |
| Adhesiveness, kg × s | −1.92a | −1.21b | −1.57ab | −1.46a,b | −1.35a,b | 0.127 |
| Springiness, % | 0.99 | 0.94 | 1.00 | 0.92 | 1.00 | 0.035 |
| Cohesiveness, % | 0.53a,b | 0.50b | 0.56a,b | 0.56a | 0.57a | 0.011 |
| Resilience, % | 0.04a,b | 0.06a | 0.04a,b | 0.04b | 0.04b | 0.003 |
| Gumminess | 0.87a | 0.79a,b | 0.71b | 0.71b | 0.65b | 0.033 |
| Firmness/toughness, kg | 0.87a | 0.68c | 0.76b | 0.73b,c | 0.70b,c | 0.016 |
| Color analysis | ||||||
| Raw canned food | ||||||
| L* | 57.98a | 53.09c | 53.59b | 51.40d | 45.67e | 0.056 |
| a* | 4.47c | 4.73a | 4.77a | 4.61b | 3.73d | 0.013 |
| b* | 18.53c | 21.39a | 21.21a | 20.32b | 18.40c | 0.038 |
| Retorted canned food | ||||||
| L* | 52.34a | 47.40b,c | 48.40b | 46.07c | 46.36c | 0.320 |
| a* | 5.96b | 7.91a | 6.71b | 6.05b | 4.35c | 0.222 |
| b* | 19.50b | 25.80a | 22.55b | 22.13b | 16.32c | 0.422 |
1Standard error of the mean.
2Hardness: the peak force that occurs during the first compression; adhesiveness: the stickiness of the product; springiness: the ability of the product to regain its former shape during the wait time between the first deformation and the second compression; cohesiveness: the resistance to the second deformation of the product relative to the resistance to the first deformation; resilience: the extent the product regain its original height; gumminess: applies to semi-solids, hardness * cohesiveness.
a, b,c,d,eMeans in the same row with different superscript letters are different (P < 0.05).
The results of the color analysis were separated into two parts, before and after retort, as shown in Table 12. Before retort, the rice diet had the highest brightness of 57.98 and the 4% GBF diet had the lowest brightness of 45.67 (P < 0.05). For a* (redness), the 4% GBF diet was the lowest being 3.73, whereas the other diets ranged from 4.47 to 4.77 (P < 0.05). The b* (yellowness) was highest in the potato and 1% GBF diets (21.39 and 21.21, respectively) with the rice and 4% GBF diets being the lowest (18.53 and 18.40, respectively; P < 0.05). After retort, the rice diet remained to have the highest brightness of 52.3 among all diets (P < 0.05). The potato diet had the highest redness and yellowness (7.91 and 25.80, respectively) and the 4% GBF diet had the lowest redness and yellowness (4.35 and 16.32, respectively; P < 0.05).
Treatment diets did not have a significant effect on food intake (as-is and DM basis), fecal output (as-is and DM basis), or macronutrient ATTD (Table 13; P > 0.05). DM digestibility ranged from 76.2% to 82.3%. Digestibility of OM and CP were above 80% for all treatment groups. AHF digestibility ranged from 82.4% to 91.8%. TDF digestibility ranged from 49.0% to 62.3%. The digestible energy (DE) for the treatment diets was between 5.1 and 5.5 kcal/g.
Table 13.
Food intake, fecal output, and total tract apparent macronutrient digestibility of adult felines fed dietary treatments containing traditional starch source and green banana flour
| Treatment | SEM | |||||
|---|---|---|---|---|---|---|
| Green banana flour | ||||||
| Item | Rice | Potato | 1% | 2% | 4% | |
| Food intake, g/d as-is | 155.1 | 160.9 | 160.3 | 175.2 | 159.6 | 27.22 |
| Food intake, g/d DM basis | 37.2 | 38.9 | 38.4 | 42.3 | 38.0 | 6.55 |
| Fecal output, g/d as-is | 24.6 | 18.6 | 34.2 | 29.4 | 24.6 | 7.93 |
| Fecal output, g/d DM basis | 8.8 | 7.1 | 10.3 | 8.1 | 7.2 | 1.74 |
| Digestibility, % | ||||||
| Dry matter (DM) | 76.2 | 82.3 | 75.2 | 79.7 | 80.2 | 1.93 |
| % DM basis | ||||||
| Organic matter | 82.8 | 88.3 | 81.7 | 85.3 | 86.3 | 1.77 |
| Acid hydrolyzed fat | 84.0 | 91.8 | 82.4 | 90.0 | 91.2 | 3.17 |
| Crude protein | 81.4 | 87.5 | 81.5 | 84.8 | 84.8 | 1.88 |
| Total dietary fiber | 52.3 | 60.4 | 49.0 | 62.5 | 57.6 | 3.83 |
| Digestible energy, kcal/g (DM basis) | 5.1 | 5.5 | 5.2 | 5.5 | 5.5 | 1.20 |
No treatment effect was observed (P > 0.05).
There was no interaction between treatment and time for fecal score, pH, or metabolites (Table 14; P > 0.05). No main effects of treatment or time were present either (P > 0.05). The average fecal score of each group ranged from 1.9 to 3.2 on day 0 and 1.9 to 2.5 on day 21. The average fecal pH ranged from 6.4 to 6.9 on days 0 and 21. The concentration of total fecal SCFA ranged from 184.1 to 270.0 μmol/g on a DM basis with acetate being the predominant SCFA, followed by propionate, and then butyrate. The concentrations of BCFA ranged from 27.7 to 44.4 μmol/g on a DM basis. Total phenol and indole concentrations ranged from 1.7 to 3.8 μmol/g on a DM basis with indole being the predominant metabolite. Fecal ammonia concentration ranged from 149.7 to 225.4 μmol/g on a DM basis.
Table 14.
Fecal characteristics of adult felines fed dietary treatments containing traditional starch source and green banana flour
| Treatment diet | SEM | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Green banana flour | |||||||||||
| Rice | Potato | 1% | 2% | 4% | |||||||
| Item | D0 | D21 | D0 | D21 | D0 | D21 | D0 | D21 | D0 | D21 | |
| Fecal score | 2.1 | 2.4 | 1.9 | 1.9 | 2.6 | 2.4 | 3.2 | 2.5 | 2.4 | 2.4 | 0.36 |
| Fecal pH | 6.5 | 6.5 | 6.9 | 6.7 | 6.5 | 6.5 | 6.6 | 6.5 | 6.6 | 6.4 | 0.13 |
| Short-chain fatty acid, μmol/g DMB 1 | |||||||||||
| Acetate | 103.1 | 112.8 | 103.4 | 117.4 | 130.3 | 116.5 | 152.0 | 145.4 | 163.3 | 140.9 | 32.36 |
| Propionate | 43.8 | 43.8 | 44.2 | 53.5 | 55.4 | 51.7 | 63.0 | 55.0 | 67.9 | 56.4 | 10.92 |
| Butyrate | 37.2 | 30.2 | 30.4 | 27.7 | 34.9 | 27.2 | 38.4 | 32.8 | 38.8 | 31.8 | 4.45 |
| Total SCFA | 184.1 | 186.8 | 178.0 | 198.6 | 220.5 | 195.5 | 253.4 | 233.2 | 270.0 | 229.1 | 46.03 |
| Branched-chain fatty acid, μmol/g DMB 1 | |||||||||||
| Isobutyrate | 10.0 | 6.8 | 6.7 | 6.3 | 8.1 | 5.9 | 9.1 | 6.5 | 11.7 | 6.5 | 1.53 |
| Isovalerate | 13.9 | 12.0 | 11.5 | 11.0 | 12.0 | 10.0 | 12.9 | 10.9 | 14.9 | 11.5 | 1.57 |
| Valerate | 18.3 | 13.9 | 16.3 | 13.3 | 15.0 | 11.8 | 16.3 | 14.1 | 17.9 | 12.4 | 1.90 |
| Total BCFA | 42.2 | 32.7 | 34.5 | 30.6 | 35.1 | 27.7 | 38.3 | 31.5 | 44.4 | 30.4 | 4.68 |
| Phenol/indole, μmol/g DMB 1 | |||||||||||
| Phenol | 1.3 | 0.0 | 0.6 | 0.4 | 0.2 | 0.0 | 0.0 | 0.0 | 0.1 | 0.1 | 0.34 |
| Indole | 2.5 | 2.7 | 2.2 | 1.7 | 1.5 | 1.9 | 3.1 | 2.7 | 2.0 | 2.7 | 0.55 |
| Total phenol and indole | 3.8 | 2.7 | 2.8 | 2.1 | 1.7 | 1.9 | 3.1 | 2.7 | 2.2 | 2.8 | 0.78 |
| Ammonia, μmol/g DMB2 | 212.1 | 157.4 | 164.6 | 139.5 | 186.1 | 154.7 | 225.4 | 200.0 | 199.2 | 149.7 | 32.55 |
1DMB, dry matter basis.
No treatment effect was observed at D0 or D21 (P > 0.05).
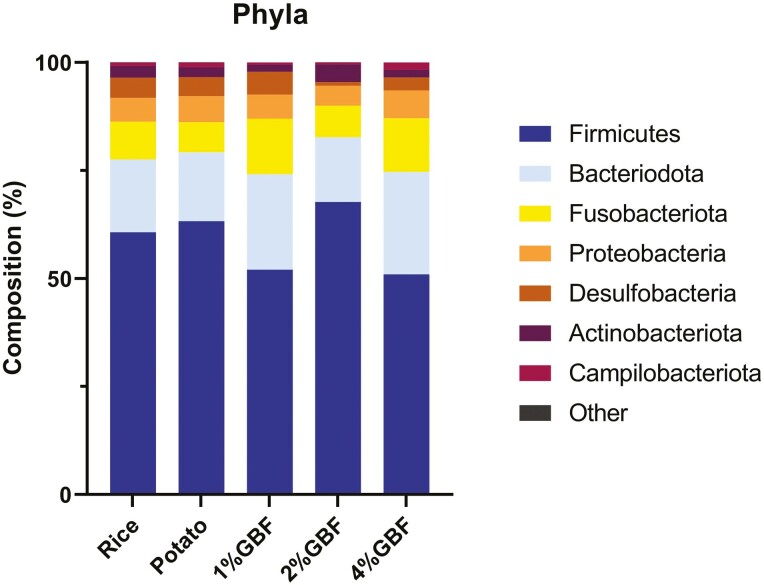
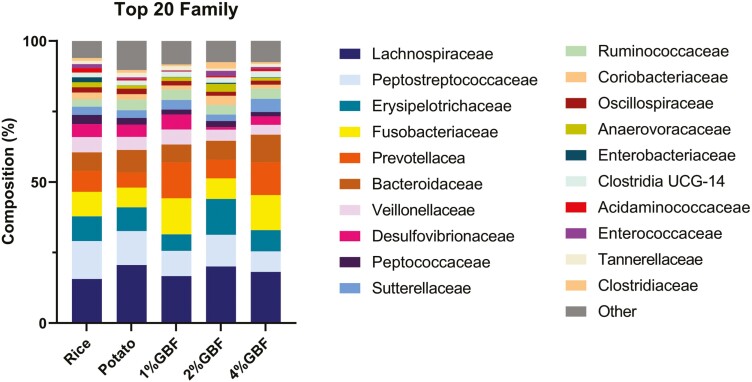
No microbiota parameters measured on day 0 were different among treatment groups (P > 0.05); therefore, only data from day 21 are shown. The fecal microbiota composition plots of cats consuming treatment diets on day 21 are shown in Figure 4–6, including the phyla, top 20 families, and top 20 genera. Some taxa showed differences in relative abundance among treatments (P < 0.05; Table 15). On the phylum level, cats consuming 2% GBF had higher abundances of Firmicutes and Actinobacteria than the 1% and 4% GBF diets as well as lower Bacteriodota abundance than 4% GBF (P < 0.05). However, cats fed the three GBF-containing diets were comparable to the rice and potato controls in microbiota phyla composition (P > 0.05). On the family level, the 2% GBF group had a higher (P < 0.05) relative abundance of Coriobacteriaceae than the 1% and 4% GBF groups while all three GBF-containing diets were comparable to the controls (P > 0.05). The Tannerellaceae family was less abundant in the 2% GBF group than potato group (P < 0.05); however, the 2% GBF group was comparable to the rice control, 1% GBF, and 4% GBF (P > 0.05). On the genus level, the 2% GBF group had a higher abundance of Collinsella than 1% and 4% GBF groups, a higher abundance of Eubacterium coprostanoligenes group than potato control, and a lower abundance of Parabacteroides than potato control (P < 0.05). The relative abundance of Oscillospiraceae UCG-005 was higher (P < 0.05) in the rice control than in the potato control while the three GBF groups were comparable to both of the controls (P > 0.05). Even though there were a few differences in taxa relative abundance, no difference was observed with any alpha or beta diversity parameters among the groups at days 0 or 21 (Supplementary Figure S1; P > 0.05).
Figure 4.
Phylum composition of fecal microbiota from adult felines fed dietary treatments containing traditional starch source and green banana flour.
Figure 5.
Family composition of fecal microbiota from adult felines fed dietary treatments containing traditional starch source and green banana flour.
Figure 6.
Genus composition of fecal microbiota from adult felines fed dietary treatments containing traditional starch source and green banana flour.
Table 15.
Fecal microbiota relative abundance in cats consuming treatment diets containing traditional starch source and green banana flour
| Treatment | SEM | ||||||
|---|---|---|---|---|---|---|---|
| Green banana flour | |||||||
| Taxa, % | Rice | Potato | 1% | 2% | 4% | ||
| Phylum | |||||||
| Firmicutes | 60.7a,b | 63.2a,b | 52.0b | 67.7a | 50.9b | 3.30 | |
| Bacteriodota | 16.8a,b | 16.0a,b | 22.1a,b | 14.9b | 23.7a | 1.97 | |
| Actinobacteriota | 2.7a,b | 2.3a,b | 1.7b | 4.1a | 1.8b | 0.47 | |
| Phylum | Family | ||||||
| Actinobacteriota | Coriobacteriaceae | 2.4a,b | 1.9a,b | 1.4b | 3.3a | 1.4b | 0.37 |
| Bacteroidota | Tannerellaceae | 1.2a,b | 1.6a | 1.5a,b | 0.8b | 1.1a,b | 0.19 |
| Phylum | Genus | ||||||
| Actinobacteriota | Collinsella | 2.4a,b | 1.9a,b | 1.4b | 3.3a | 1.4b | 0.37 |
| Bacteroidota | Parabacteroides | 1.2a,b | 1.6a | 1.5a,b | 0.8b | 1.1a,b | 0.19 |
| Firmictues | Eubacterium coprostanoligenes group | 0.7a,b | 0.3b | 0.5a,b | 1.0a | 0.5a,b | 0.15 |
| Firmictues | Oscillospiraceae UCG-005 | 0.7a | 0.3b | 0.4a,b | 0.4a,b | 0.3a,b | 0.11 |
a,bMeans in the same row with different superscript letters are different (P < 0.05). Only the taxa with significant differences among groups are listed.
Discussion
Characteristics of GBF and experimental diets
There were differences in chemical compositions seen among the sources of GBF which were predicted since many factors would affect the nutrient profile, including manufacturing methods, ripeness of the banana, and type of banana (Falade and Oyeyinka, 2014; Khoozani et al., 2019; Menezes et al., 2010; Moongngarm et al., 2014; Rodríguez-Damián et al., 2013). However, even with the variability in chemical composition, the tested GBF corresponded to reported values in the literature and were all high in RS (30% to 50%) while low in free sugar (<2%), protein (3% to 4%), and fat (<1%; Menezes et al., 2010; Moongngarm et al., 2014; Khoozani et al., 2019). The RS content also corresponded with the TMEn results from the precision-fed rooster assay. With more than 30% of RS in GBF, the TMEn was 35% to 49% lower than the GE. This indicated high proportions of non-digestible fiber in the GBF ingredients.
The range of texture in the tested commercial pet canned foods varied widely for all parameters. Therefore, the determination of a specific texture profile might be based on the manufacturer’s strategy and decision since the wide variety of textures is accepted by pet animals and is available in the market. Comparing the texture among the treatment diets with traditional carbohydrate sources and GBF, the hardness was lower in GBF-containing diets. As for the tested batter consistency measured by Bostwick Consistometer, the results were comparable to a previous study of feline canned diets (Dainton et al., 2021). The final product texture corresponded to the batter consistency measured as the batter with GBF was thinner. This could be due to the lack of gelling function from the RS content (Apostolidis et al., 2021) and possibly low pectin content since the GBF was included in a small amount. Since the water binding capacity contributes to the texture (Voller-Reasonover et al., 1997), it was not surprising to see the difference in texture. There have been studies showing the correlation between texture and palatability in cats and piglets (Sola-Oriol et al., 2009; Hagen-Plantinga et al., 2017). Cats preferred canned foods that were less firm for the first week of being exposed to different diets but there was no difference in preference after a longer exposure, whereas adhesiveness and chewing work did not have a significant effect on preference (Hagen-Plantinga et al., 2017). In piglets, they showed a similar preference for firmness as cats but they also preferred foods with less chewing work (Sola-Oriol et al., 2009). The discrepancy between species could result from less chewing required when cats are ingesting wet food (Watanabe et al., 2005). Although a palatability test was not performed in the current study, the food intake did not differ among treatment groups. Since the treatment period was 21 d and the numerical difference of the hardness may not be great enough for the cats to differentiate, the cats might not have preferred one diet over another in the current study.
The color of commercial wet pet foods also had a wide range. It can be concluded that most canned foods have different shades of brown but the brightness, redness, and yellowness may differ. It is not a new finding that ingredients could change the color of canned food. Even a 2.5% binder ingredient change could lead to significant differences in color (Polo et al., 2009). Another study also showed color differences with the inclusion of different fiber sources (barley, wheat, and oat fiber) in canned foods (Słowiński et al., 2021). Since the GBF ingredient had a darker color than rice flour, it was expected that the final diet with GBF would have a darker color. As the range of color could vary greatly based on the formulation, it is difficult to directly compare the current treatment diets with data from other studies or commercial diets; however, the results of the color analysis were comparable with the chicken-based canned foods from a recent study by Dainton et al. (2021). According to Hobbs and Shanoyan (2018), color was not a main determinant for customers’ choice of pet food. Nonetheless, “ingredient”, “health”, and “natural” were important for the customers. In commercial wet foods, color additives are commonly used to enhance the appearance. With the trend of “health” and “natural”, adding GBF to wet foods could be beneficial since it could serve as both a carbohydrate source and a color additive to create a simpler label for better customer appeal.
In vitro study
Regarding the pH change and SCFA production of the in vitro fermentation experiment, GBF was less fermentable than beet pulp but more fermentable than cellulose. Since cellulose is more than 90% composed of insoluble fiber (de Godoy et al., 2013), it was expected that GBF would be more fermentable. However, the difference between GBF and beet pulp was not fully expected since the GBF source tested had a 40.9% RS content which should be fermentable (Nugent, 2005). This could potentially be due to a numerically higher SCFA production at baseline (0 h) for the GBF samples. There is a possibility that the ice bath at baseline did not stop fermentation abruptly enough and some easily accessible energy sources were fermented in a short period of time. In addition, an in vitro study on retrograded maize starch using human feces showed most acetate production occurred between 12 and 18 h (Zhu and Zhao, 2013). This finding corresponds to the current study as the SCFA production did not reach a plateau at the end of the fermentation study at 12 h. A longer fermentation period could be beneficial to gain a more extensive view of the fermentability of GBF. A previous 12 h in vitro study by Panasevich et al. (2015) on soluble corn fiber, containing 78.3% SDF and 7.8% RS, showed that the soluble corn fiber was more fermentable than cellulose but less than pectin; however, the study did not include beet pulp for comparison. There have been several other in vitro studies on SDF using cat or dog fecal inoculum (Sunvold et al., 1995; de Godoy et al., 2015; Bosch et al., 2008; Traughber et al., 2020) showing high fermentability of different substrates. However, it is difficult to directly compare the results with the GBF in the current study; nonetheless, these studies all detected acetate production as the most abundant SCFA which was consistent with the present result. The current study used canine feces since they are more feasible to collect and composite to a pooled sample than feline feces. In addition, the main goal of the in vitro study was to examine the potential of GBF as a fiber source in pet foods, which was accomplished using the current method. However, future in vitro studies of GBF using feline fecal inoculum would be interesting for possible differences between species.
Macronutrient digestibility
There has been continuous conflict about how dietary fiber would affect macronutrient digestibility in companion animal research. In the present study, the digestibility of DM, OM, AHF, CP, TDF, and DE was not significantly different. When discussing the effect of fiber on digestibility, the inclusion rate and fiber source should be put into consideration. Many sources of dietary fiber are used by the pet food industry and even more novel fibers are being tested in research (de Godoy et al., 2013). These fibers have different compositions and physicochemical properties which could lead to different results in digestibility. Murray et al. (1999) concluded that an inclusion rate of 50% potato flour, a common grain-free starch source with high RS, as the main starch source in canine kibbles could lead to lower CP digestibility when compared with diets with 44% rice flour inclusion. Beloshapka et al. (2021) found a linear decrease in digestibility of DM, OM, CP, AHF, and DE with increasing RS content, from 6.25% to 10.25% DM basis, in the canine diets. On the other hand, results from Panasevich et al. (2013) showed no difference in CP digestibility up to a 6% as-is inclusion rate of potato fiber as a fiber source in dry extruded dog food. The current research included 4% of different carbohydrate sources (as-is basis) in the wet food formulation. With a low inclusion rate, it was understandable that no difference in ATTD was observed. However, the diet type could also be another factor affecting digestibility, since the formulation and processing conditions are vastly different between extruded diets and canned diets. To our knowledge, there was no published literature on the effects of starch sources in canned foods for companion animals. Therefore, a direct comparison of the results from the present study to previous data on dry foods would not be fully translatable. The results of the current research could provide more insights into how carbohydrates could affect digestibility in wet diets.
Fecal metabolites and microbiota
RS type 2 from potatoes has been reported to increase the concentration of butyrate in humans and rats (Cummings et al., 1996; Ferguson et al., 2000; Martin et al., 2000; Henningsson et al., 2003). Some studies in companion animals showed the effect of RS on increasing fecal total SCFA concentration and butyrate concentration as well (Peixoto et al., 2018; Ribeiro et al., 2019; Jackson et al., 2020). Peixoto et al. (2017) and Ribeiro et al. (2019) reported an increase in total fecal SCFA concentration with a diet containing 1.5% (DM basis) RS from extrusion processing compared with the 0.2% (DM basis) RS diet in dogs. Panasevich et al. (2013) found linear increases in all individual and total fecal SCFA with diets of increasing potato RS content in dogs. Jackson et al. (2020) found an increase in fecal butyrate concentration and a decrease in fecal ammonia concentration in cats fed a diet with 7.9% (DM basis) RS from extrusion processing. However, Beloshapka et al. (2021) and Panasevich et al. (2015) found null or few differences in fermentative end-products with the inclusions of 1% to 4% RS from corn and 1.5% to 6% potato fiber, respectively. The current study also showed no difference in fecal metabolites. The discrepancy could be due to the source and/or amount of RS, processing methods of diets, and individual variations of animal response to treatment (Nugent, 2005). The low inclusion rate of carbohydrate sources and diet type in the current study could be a factor for causing no difference in fecal metabolites among groups. As previously mentioned, the effect of retorts on canned foods should also be taken into consideration; thus, a direct comparison may not be fair. In addition, fecal metabolites are not a direct indication of the colon environment since there is a high absorption rate of SCFA in the large intestine (von Englehardt et al., 1989). Thus, if a more proximal fermentation of RS occurs, it might be difficult to detect differences in fecal SCFA concentrations.
The lower inclusion rate of GBF could have also led to the finding of similar fecal microbiota from the cats in the current study. Only 4 out of the 132 identified genera were different among groups. Despite the statistical differences in the relative abundance of a few taxa, the small difference may not bear physiological significance, which would correspond to the comparable fecal metabolites among treatment groups. Previously, Jackson et al. (2020) observed a higher alpha diversity in cats fed a high RS diet (7.9% RS on a DM basis) when compared with a low RS diet (0.5% RS on a DM basis) after 3 wk. This was attributed to the higher fermentable fiber content from the high RS diet that could provide more substrate for saccharolytic bacteria in the gut. Therefore, a higher inclusion level of GBF has the potential of increasing saccharolytic microbes in cats. Nonetheless, overall, the microbiota composition of the present study corresponded with previous research with the most abundant phyla being Firmicutes, Bacteriodota, Fusobacteriota, Proteobacteria, Desulfobacteria, Actinobacteriota, and Campilobacteriota. (Jackson et al., 2020; Li et al., 2023).
One of the main limitation of the current research was the low inclusion rate of GBF since carbohydrates could only account for a lower proportion of canned diets to be manufacturable and achieve the physical characteristics of wet foods. Therefore, a higher inclusion rate in other forms of diets could potentially lead to more significant physiological responses in cats. Another limitation was that the RS content was not tested in the final canned products. Future research on the effect of manufacturing on the RS in GBF could be beneficial to understand more about this novel ingredient.
Conclusions
The current study confirmed that GBF is a fiber source with high RS. Since the addition of GBF caused no negative effect on ATTD in cats, GBF could be used as a comparable carbohydrate source with rice flour and potato in feline wet food formulation. Even though there were no significant differences seen in the fecal metabolites and microbiota with GBF-incorporated diets, the in vitro assay showed an intermediate degree of fermentability of GBF in contrast with beet pulp and cellulose. Therefore, at a higher dietary concentration, GBF could potentially demonstrate modulatory effects on hindgut fermentation in companion animals. From a manufacturing standpoint, it can be concluded that GBF could serve as a natural coloring agent to darken the retorted pet foods, but not as an optimal thickening agent. The addition of GBF to canned foods would result in a lower hardness which could be utilized to produce the desired texture. In conclusion, the addition of GBF could provoke interest in customers as a novel ingredient and contribute to the colorization of canned foods while serving as a comparable carbohydrate source and a label-friendly ingredient in feline diets.
Supplementary Material
Acknowledgments
We thank Simmons Pet Food, Inc. for the financial support of the study.
Glossary
Abbreviations
- AHF
acid hydrolyzed fat
- AAFCO
Association of American Feed Control Officials
- ATTD
apparent total tract digestibility
- BCFA
branched-chain fatty acid
- CP
crude protein
- DE
digestible energy
- DM
dry matter
- GE
gross energy
- IDF
insoluble dietary fiber
- OM
organic matter
- RS
resistant starch
- SCFA
short-chain fatty acid
- SDF
soluble dietary fiber
- TDF
total dietary fiber
- TMEn
true metabolizable energy corrected to zero nitrogen retention
Contributor Information
Clare Hsu, Department of Animal Sciences, University of Illinois at Urbana-Champaign, Urbana, IL 61801, USA.
Brittany White, Simmons Pet Food, Inc., Siloam Springs, AR 72761, USA.
Leah Lambrakis, Simmons Pet Food, Inc., Siloam Springs, AR 72761, USA.
Patricia M Oba, Department of Animal Sciences, University of Illinois at Urbana-Champaign, Urbana, IL 61801, USA.
Fei He, Department of Animal Sciences, University of Illinois at Urbana-Champaign, Urbana, IL 61801, USA.
Pamela Utterback, Department of Animal Sciences, University of Illinois at Urbana-Champaign, Urbana, IL 61801, USA.
Carl M Parsons, Department of Animal Sciences, University of Illinois at Urbana-Champaign, Urbana, IL 61801, USA.
Maria R C de Godoy, Department of Animal Sciences, University of Illinois at Urbana-Champaign, Urbana, IL 61801, USA; Division of Nutritional Sciences, University of Illinois at Urbana-Champaign, Urbana, IL 61801, USA.
Conflict of Interest Statement
C. H., P. M. O., F. H., P. U., C. M. P., and M. R. C. G. have no conflict of interest to declare. B. W. and L. L. are employed by Simmons Pet Food, Inc.
Literature Cited
- American Association of Cereal Chemists (AACC). 1983. Approved methods. 8th ed. St. Paul (MN): AACC. [Google Scholar]
- American Association of Cereal Chemists (AACC). 2010. Approved methods. 11th ed. St. Paul (MN): AACC. [Google Scholar]
- Apostolidis, E., Kioupis D., Kakali G., Stoforos N. G., and Mandala I... 2021. Effect of starch concentration and resistant starch filler addition on the physical properties of starch hydrogels. J. Food Sci. 86:5340–5352. doi: 10.1111/1750-3841.15954 [DOI] [PubMed] [Google Scholar]
- Association of American Feed Control Officials (AAFCO). 2020. Official Publication. Champaign, IL: Association of American Feed Control Officials. [Google Scholar]
- Association of Analytical Chemists (AOAC). 2006. Official methods of analysis. 17th ed. Arlington (VA): AOAC. [Google Scholar]
- Beloshapka, A. N., Cross T. L., and Swanson K. S... 2021. Graded dietary resistant starch concentrations on apparent total tract macronutrient digestibility and fecal fermentative end products and microbial populations of healthy adult dogs. J. Anim. Sci. 99:skaa409. doi: 10.1093/jas/skaa409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ble-Castillo, J. L., Aparicio-Trápala M. A., Francisco-Luria M. U., Córdova-Uscanga R., Rodríguez-Hernández A., Méndez J. D., and Díaz-Zagoya J. C... 2010. Effects of native banana starch supplementation on body weight and insulin sensitivity in obese type 2 diabetics. Int. J. Environ. Res. Public Health 7:1953–1962. doi: 10.3390/ijerph7051953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosch, G., Pellikaan W. F., Rutten P. G., van der Poel A. F., Verstegen M. W., and Hendriks W. H... 2008. Comparative in vitro fermentation activity in the canine distal gastrointestinal tract and fermentation kinetics of fiber sources. J. Anim. Sci. 86:2979–2989. doi: 10.2527/jas.2007-0819 [DOI] [PubMed] [Google Scholar]
- Bourquin, L. D., Titgemeyer E. C., and Fahey G. C.. 1993. Vegetable fiber fermentation by human fecal bacteria: Cell wall polysaccharide disappearance and short-chain fatty acid production during in vitro fermentation and water-holding capacity of unfermented residues. J. Nutr. 123:860–869. doi: 10.1093/jn/123.5.860 [DOI] [PubMed] [Google Scholar]
- Bray, J. R., and Curtis J. T.. 1957. An ordination of the upland forest communities of Southern Wisconsin. Ecol. Monogr. 27:325–349. doi: 10.2307/1942268 [DOI] [Google Scholar]
- Brown, I. L., McNaught K. J., and Moloney E... 1995. Hi-maize: new directions in starch technology and nutrition. Food Aust. 47:272–275. [Google Scholar]
- Budde, E. F. 1952. The determination of fat in baked biscuit type of dog foods. J. Assoc. Off. Agric. Chem. 35:799–805. doi: 10.1093/jaoac/35.3.799 [DOI] [Google Scholar]
- Chaney, A. L., and Marbach E. P... 1962. Modified reagents for determination of urea and ammonia. Clin. Chem. 8:130–132. doi: 10.1093/clinchem/8.2.130 [DOI] [PubMed] [Google Scholar]
- Chiang, B. H., Chu W. C., and Chu C. L... 1987. A pilot scale study for banana starch production. Starch. 39:5–8. doi: 10.1002/star.19870390103 [DOI] [Google Scholar]
- Churms, S.C. 1982. “Carbohydrates” In: by Zweig Gunter, and Sherma Joseph, Eds Handbook of Chromatography. Boca Raton, FL: CRC Press. [Google Scholar]
- Cummings, J. H., Beatty E. R., Kingman S. M., Bingham S. A., and Englyst H. N... 1996. Digestion and physiological properties of resistant starch in the human large bowel. Br. J. Nutr. 75:733–747. doi: 10.1079/bjn19960177 [DOI] [PubMed] [Google Scholar]
- Dai, F. J., and Chau C. F... 2016. Classification and regulatory perspectives of dietary fiber. J. Food Drug Anal. 25:37–42. doi: 10.1016/j.jfda.2016.09.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dainton, A. N., Dogan H., and Aldrich C. G... 2021. The effects of select hydrocolloids on the processing of pâté-style canned pet food. Foods. 10:2506. doi: 10.3390/foods10102506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dan, M. C., Cardenette G. H., Sardá F. A., Giuntini E. B., Bello-Pérez L. A., Carpinelli R., Lajolo F. M., and Menezes E. W... 2015. Colonic fermentation of unavailable carbohydrates from unripe banana and its influence over glycemic control. Plant Foods Hum. Nutr. 70:297–303. doi: 10.1007/s11130-015-0493-6 [DOI] [PubMed] [Google Scholar]
- da Silva, A. R., Cerdeira C. D., Brito A. R., Salles B. C., Ravazi G. F., Moraes G. O., Rufino L. R., Oliveira R. B., and Santos G. B... 2016. Green banana pasta diet prevents oxidative damage in liver and kidney and improves biochemical parameters in type 1 diabetic rats. Arch. Endocrinol. Metab. 60:355–366. doi: 10.1590/2359-3997000000152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Godoy, M. R., Kerr K. R., and G. C.Fahey, Jr. 2013. Alternative dietary fiber sources in companion animal nutrition. Nutrients. 5:3099–3117. doi: 10.3390/nu5083099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Godoy, M. R., Mitsuhashi Y., Bauer L. L., G. C.Fahey, Jr, Buff P. R., and Swanson K. S... 2015. In vitro fermentation characteristics of novel fibers, coconut endosperm fiber and chicory pulp, using canine fecal inoculum. J. Anim. Sci. 93:370–376. doi: 10.2527/jas.2014-7962 [DOI] [PubMed] [Google Scholar]
- Dunjic, B. S., Svensson I., Axelson J., Adlercreutz P., Ar’rajab A., Larsson K., and Bengmark S... 1993. Green banana protection of gastric mucosa against experimentally induced injuries in rats. Scand. J. Gastroenterol. 28:894–898. doi: 10.3109/00365529309103131 [DOI] [PubMed] [Google Scholar]
- Eleazu, C. O., and Okafor P... 2015. Use of unripe plantain (Musa paradisiaca) in the management of diabetes and hepatic dysfunction in streptozotocin induced diabetes in rats. Interv. Med. Appl. Sci. 7:9–16. doi: 10.1556/IMAS.7.2015.1.2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Englyst, H. N., Kingman S. M., and Cummings J. H... 1992. Classification and measurement of nutritionally important starch fractions. Eur. J. Clin. Nutr. 46:S33–S50. [PubMed] [Google Scholar]
- Falade, K. O., and Oyeyinka S. A... 2014. Color, Chemical and functional properties of plantain cultivars and cooking banana flour as affected by drying method and maturity. J. Food Process. Preserv. 39:816–828. doi: 10.1111/jfpp.12292 [DOI] [Google Scholar]
- Falcomer, A. L., Riquette R. F. R., de Lima B. R., Ginani V. C., and Zandonadi R. P... 2019. Health benefits of green banana consumption: a systematic review. Nutrients. 11:1222. doi: 10.3390/nu11061222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferguson, L. R., Tasman-Jones C., Englyst H., and Harris P. J... 2000. Comparative effects of three resistant starch preparations on transit time and short-chain fatty acid production in rats. Nutr. Cancer 36:230–237. doi: 10.1207/S15327914NC3602_13 [DOI] [PubMed] [Google Scholar]
- Flickinger, E. A., Schreijen E. M., Patil A. R., Hussein H. S., Grieshop C. M., Merchen N. R., and G. C.Fahey, Jr. 2003. Nutrient digestibilities, microbial populations, and protein catabolites as affected by fructan supplementation of dog diets. J. Anim. Sci. 81:2008–2018. doi: 10.2527/2003.8182008x [DOI] [PubMed] [Google Scholar]
- Food and Agriculture Organization of the United Nations. 2018. Banana market review 2018.– accessed [15 January 15, 2021]. https://www.fao.org/markets-and-trade/publications/detail/en/c/1438765/.
- Food and Drug Administration, HHS. 2016. Food labeling: revision of the nutrition and supplement facts labels. Fed. Regist. 81:33741–33999. https://www.federalregister.gov/documents/2016/05/27/2016-11867/food-labeling-revision-of-the-nutrition-and-supplement-facts-labels [PubMed] [Google Scholar]
- Fox, A., Morgan, S.L. and Gilbert, J.. 1989. “Preparation of alditol acetates and their analysis by gas chromatography (GC) and mass spectrometry (MS)”, Chapter 5, In: by GLC and MS, Biermann, CJ and McGinnis, eds. Analysis of Carbohydrates. Boca Raton, FL: CRC Press. 87–117. [Google Scholar]
- Fuentes-Zaragoza, E., Riquelme-Navarrete M. J., Sánchez-Zapata E., and Pérez-Álvarez J. A... 2010. Resistant starch as functional ingredient: a review. Food Res. Int. 43:931–942. doi: 10.1016/j.foodres.2010.02.004 [DOI] [Google Scholar]
- Hagen-Plantinga, E. A., Orlanes D. F., Bosch G., Hendriks W. H., and van der Poel A. F. B... 2017. Retorting conditions affect palatability and physical characteristics of canned cat food. J. Nutr. Sci. 6:1–5. doi: 10.1017/jns.2017.17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamady, M., Lozupone C., and Knight R.. 2010. Fast UniFrac: facilitating high-throughput phylogenetic analyses of microbial communities including analysis of pyrosequencing and PhyloChip data. ISME J. 4:17–27. doi: 10.1038/ismej.2009.97 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henningsson, A. M., Margareta E., Nyman G. L., and Björck I. M. E... 2003. Influences of dietary adaptation and source of resistant starch in the hindgut of rats. Br. J. Nutr. 89:319–327. doi: 10.1079/bjn2003782 [DOI] [PubMed] [Google Scholar]
- Hobbs, L., and Shanoyan A... 2018. Analysis of consumer perception of product attributes in pet food: implications for marketing and brand strategy. Agric. Appl. Econ. Assoc. Annual Meeting. doi: 10.22004/ag.econ.274070 [DOI] [Google Scholar]
- Hudson, J. R., Morgan S. L., and Fox A... 1982. High resolution glass capillary columns for the gas chromatographic analysis of alditol acetates of neutral and amino sugars. J. High Res. Chromatog. 5:285. doi: 10.1002/jhrc.1240050602 [DOI] [PubMed] [Google Scholar]
- Jackson, M. I., Waldy C., and Jewell D. E... 2020. Dietary resistant starch preserved through mild extrusion of grain alters fecal microbiome metabolism of dietary macronutrients while increasing immunoglobulin A in the cat. PLoS One 15:e0241037. doi: 10.1371/journal.pone.0241037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kakehi, K. and Honda S... 1989. “Silyl ethers of carbohydrates”, Chapter 4. In: by GLC and MS, Biermann, CJ and McGinnis, Eds., Analysis of Carbohydrates. Boca Raton, FL: CRC Press. 43–85. [Google Scholar]
- Khoozani, A. A., Bekhit A. E. A., and Birch J... 2019. Effects of different drying conditions on the starch content, thermal properties and some of the physicochemical parameters of whole green banana flour. Int. J. Biol. Macromol. 130:938–946. doi: 10.1016/j.ijbiomac.2019.03.010 [DOI] [PubMed] [Google Scholar]
- Kiho, T., Ukai S., and Hara C... 1986. Simultaneous determination of the alditol acetate derivatives of amino and neutral sugars by gas-liquid chromatography. J. Chromatogr. A 369:415. doi: 10.1016/S0021-9673(00)90149-3 [DOI] [Google Scholar]
- Langkilde, A. M., Champ M., and Andersson H... 2002. Effects of high-resistant-starch banana flour (RS2) on in vitro fermentation and the small-bowel excretion of energy, nutrients, and sterols: an ileostomy study. Am. J. Clin. Nutr. 75:104–111. doi: 10.1093/ajcn/75.1.104 [DOI] [PubMed] [Google Scholar]
- Li, Y., Ali I., Lei Z., Li Y., Yang M., Yang C., and Li L... 2023. Effect of a multistrain probiotic on feline gut health through the fecal microbiota and its metabolite SCFAs. Metabolites. 13:228. doi: 10.3390/metabo13020228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin, L. J., Dumon H. J., Lecannu G., and Champ M. M... 2000. Potato and high-amylose maize starches are not equivalent producers of butyrate for the colonic mucosa. Br. J. Nutr. 84:689–696. doi: 10.1017/S0007114500002038 [DOI] [PubMed] [Google Scholar]
- McCleary, B. V., and Monaghan D. A... 2002. Measurement of resistant starch. J. AOAC Int. 85:665–675. doi: 10.1093/jaoac/85.3.665 [DOI] [PubMed] [Google Scholar]
- Menezes, E. W., Dan M. C. T., Cardenette G. H. L., Goñi I., Bello-Pérez L. A., and Lajolo F. M... 2010. In vitro colonic fermentation and glycemic response of different kinds of unripe banana flour. Plant Foods Hum. Nutr. 65:379–385. doi: 10.1007/s11130-010-0190-4 [DOI] [PubMed] [Google Scholar]
- Moongngarm, A., Tiboonbun W., Sanpong M., Sriwong P., Phiewtong L., Prakitrum R., and Huychan N... 2014. Resistant starch and bioactive contents of unripe banana flour as influenced by harvesting periods and its application. Am. J. Agric. Biol. Sci. 9:457–465. doi: 10.3844/ajabssp.2014.457.465 [DOI] [Google Scholar]
- Murray, S. M., G. C.Fahey, Jr., Merchen N. R., Sunvold G. D., and Reinhart G. A... 1999. Evaluation of selected high-starch flours as ingredients in canine diets. J. Anim. Sci. 77:2180–2186. doi: 10.2527/1999.7782180x [DOI] [PubMed] [Google Scholar]
- Nugent, A. P. 2005. Health properties of resistant starch. Nutr. Bull. 30:27–54. doi: 10.1111/j.1467-3010.2005.00481.x [DOI] [Google Scholar]
- Oxley, D., Currie, G., and Bacic, A.. 2004. Monosaccharide composition analysis: alditol acetates. chapter 25, In: Simpson ed. purifying proteins for proteomics. Cold Spring Harbor (NY): Cold Spring Harbor Laboratory Press. [Google Scholar]
- Panasevich, M. R., Rossoni Serao M. C., de Godoy M. R., Swanson K. S., Guérin-Deremaux L., Lynch G. L., Wils D., G. C.Fahey, Jr., and Dilger R. N... 2013. Potato fiber as a dietary fiber source in dog foods. J. Anim. Sci. 91:5344–5352. doi: 10.2527/jas.2013-6842 [DOI] [PubMed] [Google Scholar]
- Panasevich, M. R., Kerr K. R., Dilger R. N., G. C.Fahey, Jr, Guérin-Deremaux L., Lynch G. L., Wils D., Suchodolski J. S., Steer J. M., Dowd S. E.,. et al. 2015. Modulation of the faecal microbiome of healthy adult dogs by inclusion of potato fibre in the diet. Br. J. Nutr. 113:125–133. doi: 10.1017/S0007114514003274 [DOI] [PubMed] [Google Scholar]
- Parsons, C. M. 1985. Influence of caecectomy on digestibility of amino acids by roosters fed distillers’ dried grains with solubles. J. Agric. Sci. 104:469–472. doi: 10.1017/s0021859600044178 [DOI] [Google Scholar]
- Parsons, C. M., Potter L. M., and Bliss B. A... 1982. True metabolizable energy corrected to nitrogen equilibrium. Poult. Sci. 61:2241–2246. doi: 10.3382/ps.0612241 [DOI] [Google Scholar]
- Peixoto, M. C., Ribeiro M., Maria A. P. J., Loureiro B. A., di Santo L. G., Putarov T. C., Yoshitoshi F. N., Pereira G. T., Sá L. R. M., and Carciofi A. C... 2018. Effect of resistant starch on the intestinal health of old dogs: fermentation products and histological features of the intestinal mucosa. J. Anim. Physiol. Anim. Nutr. (Berl) 102:e111–e121. doi: 10.1111/jpn.12711 [DOI] [PubMed] [Google Scholar]
- Polo, J., Rodríguez C., Ródenas J., Morera S., and Saborido N... 2009. The use of spray-dried animal plasma in comparison with other binders in canned pet food recipes. Anim. Feed Sci. Technol. 154:241–247. doi: 10.1016/j.anifeedsci.2009.08.009 [DOI] [Google Scholar]
- Prosky, A., Asp N. G., Schweizer T. F., Devries J. W., and Furda I... 1992. Determination of insoluble and soluble dietary fiber in foods and food products: collaborative study. J. AOAC. 75:360–367. doi: 10.1093/jaoac/75.2.360 [DOI] [PubMed] [Google Scholar]
- Ribeiro, M., Peixoto M. C., Putarov T. C., Monti M., Pacheco P., Loureiro B. A., Pereira G. T., and Carciofi A. C... 2019. The effects of age and dietary resistant starch on digestibility, fermentation end products in faeces and postprandial glucose and insulin responses of dogs. Arch. Anim. Nutr. 73:485–504. doi: 10.1080/1745039X.2019.1652516 [DOI] [PubMed] [Google Scholar]
- Rodríguez-Damián, A. R., Millán J. R., Acevedo E. A., Díaz P. O., and Pérez L. A. B... 2013. Effect of different thermal treatments and storage on starch digestibility and physicochemical characteristics of unripe banana flour. J. Food Process. Preserv. 37:987–998. doi: 10.1111/j.1745-4549.2012.00737.x [DOI] [Google Scholar]
- Sardá, F. A. H., Giuntini E. B., Gomez M. L. P. A., Lui M. C. Y., Negrini J. A. E., Tadini C. C., Lajolo F. M., and Menezes E. W... 2016. Impact of resistant starch from unripe banana flour on hunger, satiety, and glucose homeostasis in healthy volunteers. J. Funct. Foods 24:63–74. doi: 10.1016/j.jff.2016.04.001 [DOI] [Google Scholar]
- Słowiński, M., Miazek J., Dasiewicz K., and Chmiel M... 2021. The effect of the addition of fiber preparations on the color of medium-grounded pasteurized and sterilized model canned meat products. Molecules 26:2247. doi: 10.3390/molecules26082247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sola-Oriol, D., Roura E., and Torrallardona D... 2009. Feed preference in pigs: relationship with feed particles size and texture. J. Anim. Sci. 87:571–582. doi: 10.2527/jas.2008-0951 [DOI] [PubMed] [Google Scholar]
- Sunvold, G. D., G. C.Fahey, Jr, Merchen N. R., Bourquin L. D., Titgemeyer E. C., Bauer L. L., and Reinhart G. A... 1995. Dietary fiber for cats: in vitro fermentation of selected fiber sources by cat fecal inoculum and in vivo utilization of diets containing selected fiber sources and their blends. J. Anim. Sci. 73:2329–2339. doi: 10.2527/1995.7382329x [DOI] [PubMed] [Google Scholar]
- Traughber, Z. T., He F., Hoke J. M., Davenport G. M., and de Godoy M. R... 2020. Chemical composition and in vitro fermentation characteristics of legumes using canine fecal inoculum. Transl. Anim. Sci. 4:txaa200. doi: 10.1093/tas/txaa200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voller-Reasonover, L., Han I. Y., Acton J. C., Titus T. C., Bridges W. C., and Dawson P. L... 1997. High temperature processing effects on the properties of fowl meat gels. Poult. Sci. 76:774–779. doi: 10.1093/ps/76.5.774 [DOI] [PubMed] [Google Scholar]
- von Englehardt, W., Rönnau K., Rechkemmer G., and Sakata T... 1989. Absorption of short-chain fatty acids and their role in the hindgut of monogastric animals. Anim. Feed Sci. Technol. 23:43–53. doi: 10.1016/0377-8401(89)90088-6 [DOI] [Google Scholar]
- Watanabe, S., Izawa M., Kato A., Ropert-Coudert Y., and Naito Y... 2005. A new technique for monitoring the detailed behaviour of terrestrial animals: a case study with the domestic cat. Appl. Anim. Behav. Sci. 94:117–131. doi: 10.1016/j.applanim.2005.01.010 [DOI] [Google Scholar]
- Zhang, P., Whistler R., Bemiller J., and Hamaker B... 2005. Banana starch: production, physicochemical properties and digestibility-a review. Carbohydr. Polym. 59:443–458. doi: 10.1016/j.carbpol.2004.10.014 [DOI] [Google Scholar]
- Zhu, C. L., and Zhao X. H... 2013. In vitro fermentation of a retrograded maize starch by healthy adult fecal extract and impacts of exogenous microorganisms on three acids production. Starch. 65:330–337. doi: 10.1002/star.201200100 [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.