Abstract
Background
Acute kidney injury (AKI) complicates 30% to 50% of cardiac surgeries in pediatric patients. Genetic variants that affect renal blood flow and inflammation have been associated with AKI after cardiac surgery in diverse populations of adults but have not been studied in children. The objective of this study is to test the hypothesis that common candidate genetic variants are associated with AKI following pediatric cardiac surgery.
Methods
This is a retrospective cohort study at a single tertiary referral children’s hospital of 2,062 individual patients undergoing surgery for congenital heart disease from September 2007 to July 2020. Pre-specified variants in candidate genes (AGTR1, APOE, IL6, NOS3, and TNF) were chosen. AKI was defined using Kidney Disease: Improving Global Outcomes serum creatinine criteria in the first week following surgery. Outcomes were analyzed by univariate and multivariable analysis of demographic, clinical, and genetic factors.
Results
The study population had median age of 6 (interquartile range [IQR], 1–53) months, 759 (37%) of whom met criteria for postoperative AKI. In unadjusted analyses of each genetic variant, only NOS3 (rs2070744) was associated with lower risk for AKI (OR 0.75, 95% CI 0.62–0.9, P = .002). In logistic regression analyses adjusting for body surface area, previously identified genetic syndrome, Society of Thoracic Surgeons-European Association for Cardio-Thoracic Surgery (STAT) score, cardiopulmonary bypass time, and nephrotoxic medication exposure, the NOS3 variant remained protective against AKI (OR 0.7, 95% CI 0.58–0.85, P < .001).
Conclusions
A common variant in NOS3 is associated with decreased incidence of AKI in children undergoing cardiac surgery. Further analysis of the genetic contributions to postoperative AKI may help identify individual risk in the pediatric population.
Acute kidney injury (AKI) is a common complication following pediatric cardiac surgery and is a significant cause of morbidity and resource utilization.1–6 There are a variety of estimates of AKI prevalence reported and is suggested to occur in the postsurgical course of 30% to 40% of pediatric patients.7 Clinical risk factors for AKI after cardiac surgery have been identified, including younger age, higher Society of Thoracic Surgeons-European Association for Cardio-Thoracic Surgery (STAT) score, and longer cardiopulmonary bypass (CPB) time.7 The mechanisms for development of AKI in children with congenital heart disease (CHD) are complex and likely include the effects of CPB, inflammatory changes, postoperative clinical changes, and neuroendocrine influences.
Several genetic variants have been associated with increased risk of AKI in adults,8–12 often through candidate gene studies based on physiologic factors known to play a role in the pathogenesis of renal injury. Renal blood flow is regulated by the renin-angiotensin-aldosterone system, known to be affected by variants in the angiotensin II receptor 1 (AGTR1) gene.8 Nitric oxide (NO) also plays an important role in the regulation of renal blood flow and is controlled by endothelial NO synthase (encoded by NOS3).9 Inflammation from surgery and CPB also is associated with increased renal and systemic cytokine production, influenced by variants in tumor necrosis factor alpha (TNF) and interleukin 6 (IL6).10–12 Apolipoprotein E (encoded by APOE) is a significant component of plasma lipoproteins and has been associated independently with the development and progression of several renal diseases.12, 13 Differences in the mechanisms of these pathways may influence risk for development of AKI across clinical scenarios.
The aim of this study is to test the hypothesis that common genetic variants associated with postoperative AKI in adults may predispose infants, children, and adolescents with CHD to develop AKI after surgery.
Methods
Patient population and electronic health record data extraction
This study was reviewed and approved by the institutional review board of Vanderbilt University Medical Center, Nashville, Tennessee. Patients were enrolled prospectively as participants in the observational study and written informed consent was obtained from parents or guardians, as well as written assent from study participants as appropriate by age. Inclusion criteria were patients undergoing surgery for CHD at the Monroe Carell Jr. Children’s Hospital at Vanderbilt and subsequently admitted to the pediatric cardiac intensive care unit from September 2007 to July 2020. Exclusion criteria were absence of either the baseline or postoperative serum creatinine measurements to determine AKI status, and use of extracorporeal membrane oxygenation (ECMO) during the postoperative course. If individuals had multiple cardiac surgical procedures, data from only the first surgery were included. Perioperative data were extracted from electronic health record (EHR) data sources and logged prospectively during the hospital admission. Data collection included patient demographics, prior medical history including known genetic syndrome(s) (DiGeorge, Down, Noonan, Turner, Holt-Oram, Williams, CHARGE, VACTERL syndromes, as well as any other identified genetic abnormalities), operative details including STAT score, aortic cross clamp and CPB times. Laboratory data and medications administered were extracted in bulk for the cohort retrospectively. Medications administered in the first week after surgery were categorized as high-risk nephrotoxins, moderate risk nephrotoxins, or non-nephrotoxins (Supplemental Table I). The nephrotoxin risk score was defined as the total number of unique high- or moderate-risk nephrotoxins administered to each patient in the first week after surgery.
Diagnosis and classification of AKI
Diagnostic criteria for AKI were based on the 2012 Kidney Disease: Improving Global Outcomes (KDIGO) creatinine criteria guidelines. 14 KDIGO defines AKI as stage 1 (≥0.3 mg/dL or 1.5-fold increase), stage 2 (2-fold increase), and stage 3 (≥4 mg/dL or 3-fold increase). We added an additional requirement that peak creatinine be > 0.5 mg/dL to meet criteria for AKI to avoid classification of a patient with normal renal function as having AKI (e.g. increase from 0.2 to 0.4 mg/dL was not considered AKI). Baseline creatinine was defined as the lowest preoperative creatinine documented in the 6 months prior to surgery. Peak creatinine was defined as the highest postoperative creatinine value within 7 days after surgery. Only creatinine criteria were used in the definition for AKI since reliable urine output data are not available for all pediatric inpatients and there were not reliable retrospective height measurements available among pediatric inpatients to estimate glomerular filtration rate. Patients not meeting stage 1, 2, or 3 KDIGO criteria were classified as having no AKI.
Candidate variant selection and genotyping
Variants that are common in the general population (minor allele frequency, MAF, > 0.05) previously associated with postoperative AKI and associated with renal perfusion and inflammation in adults were identified via literature search using PubMed. These included variants in AGTR1 (rs5186, NC_000003.12:g.148742201A > C), APOE (rs429358, NC_000019.10:g.44908684T > C; and rs7412, NC_000019.10:g.44908822C > T), IL6 (rs1800796, NC_000007.14:g.22726627G > C), NOS3 (rs2070744, NC_000007.14:g.150992991C > T) and TNF (rs1800629, NC_000006.12:g.31575254G > A).8–13 For candidate genes, homozygous and heterozygous carriers of minor allele variants were grouped together and compared to patients without any variant alleles (ie, those homozygotes for major allele). DNA samples had been previously extracted by the Vanderbilt Center for Human Genetics Research DNA Resources Core using the Autopure instrument (Qiagen). Study participants had been previously genotyped using either the Affymetrix Axiom Precision Medicine Research Array or the Precision Medicine Diversity Array (Thermo Fisher Scientific, Waltham, MA). As part of genotype data quality control, variants were removed if genotype call rate was < 98%, if minor allele frequency was > 20% different from 1000 Genomes phase 3 European reference populations, or deviation from Hardy-Weinberg Equilibrium (P-value < 1 × 10−10). Data for individuals were removed if their genotype call rate was < 98%, the genetically estimated sex differed from parental-reported sex, or for relatedness (2nd degree or closer). Genotype data were imputed to the 1000 Genomes phase 3 reference panel. For this study, we extracted data for specific variants of interest from the study database.
Statistical analysis and covariates
Baseline descriptive statistics are represented by the medians (interquartile range [IQR]) for continuous variables or number (percentage) for categorical variables. Continuous variables were compared using the Kruskal-Wallis rank test across stages of AKI and categorical variables were compared using the Pearson χ2 test. Univariate and multivariable analyses were performed to test the association of covariates to the outcome of AKI as an ordinal outcome (by stage) and as a dichotomous outcome (no AKI vs. any AKI). These pre-specified covariates included age, sex, body surface area (BSA), STAT score,16 CPB time, aortic cross clamp time, concomitant nephrotoxic medications, and known genetic abnormality. Each of the candidate genetic variants was then tested individually for association with AKI. Logistic regression was used to determine the adjusted odds ratio of each clinical variable and genetic variant for AKI regardless of stage. Ordinal logistic regression was used to determine the adjusted odds ratio of each clinical variable and genetic variant by stage of AKI. For both the dichotomous and ordinal logistic regression analyses, STAT score was analyzed for this as an ordinal variable by score compared to STAT group 1 as the reference. The multivariable model included the subset of pre-selected covariates that were significant in univariate analysis. In the case of highly correlated pairs of pre-selected covariates (age and BSA, as well as CPB and cross-clamp time), a single variable was selected for each, based on the strength of each univariate association. The nephrotoxin risk score was included in the multivariable model as the number of unique nephrotoxins (Supplemental Table I). The STAT score was treated as a continuous variable in the multivariable analysis. A commercially available statistical soft-ware package was used for data analysis (STATA Version 17.0, College Station, Texas).
Results
During the study, a total of 2,062 individuals with baseline and peak serum creatinine measurements were included. Of those, 973 (47%) were female and 473 (23%) had a previously identified genetic anomaly. Seven-hundred and fifty-nine (37%) met criteria for postoperative AKI: 413 (54.4%) with stage 1; 239 (31.5%) with stage 2; and 107 (14.1%) with stage 3. Infants < 12 months of age (n = 1,228) had a 47% incidence of AKI while children > 12 months of age (n = 834) had 22% incidence of AKI. Other clinical characteristics of the cohort and those with AKI are listed in Table I. Primary surgical procedures are listed in Supplemental Table II. Genotype frequencies for the candidate variants are listed in Supplemental Table III.
Table I.
Patient characteristics.
| Variable | All (n = 2062) | No AKI (n = 1303) | Stage I AKI (n = 413) | Stage II AKI (n = 239) | Stage III AKI (n = 107) | P-value* |
|---|---|---|---|---|---|---|
|
| ||||||
| Age, Median (IQR), mo | 6.0 (1.0–52.5) | 12.7 (3.7–74.5) | 3.7 (0.2–18.5) | 2.5 (0.2–8.0) | 2.5 (0.2–6.5) | < .001 |
| BSA (m2 ) | 0.34 (0.24–0.67) | 0.41 (0.28–0.82) | 0.27 (0.22–0.45) | 0.26 (0.22–0.36) | 0.26 (0.22–0.36) | < .001 |
| Serum Creatinine, Median (IQR), mg/dL† | ||||||
| Preoperative Baseline | 0.42 (0.32–0.51) | 0.44 (0.35–0.54) | 0.41 (0.35–0.50) | 0.31 (0.25–0.40) | 0.24 (0.19–0.36) | < .001 |
| Postoperative Peak | 0.6 (0.50–0.74) | 0.54 (0.46–0.65) | 0.69 (0.59–0.83) | 0.73 (0.59–0.91) | 0.98 (0.69–1.36) | < .001 |
| Male Sex, No. (%) | 1089 (53%) | 664 (51%) | 237 (57%) | 133 (56%) | 55 (51%) | .12 |
| Genetic Syndrome, No. (%)‡ | 473 (23%) | 256 (20%) | 99 (24%) | 77 (32%) | 41 (38%) | < .001 |
| STAT Score, Median | 2 | 2 | 3 | 3 | 3 | < .001 |
| STAT 1 | 768 (37%) | 606 (47%) | 97 (23%) | 44 (18%) | 21 (20%) | |
| STAT 2 | 461 (22%) | 319 (24%) | 81 (20%) | 43 (18%) | 18 (17%) | |
| STAT 3 | 248 (12%) | 132 (10%) | 60 (15%) | 38 (16%) | 18 (17%) | |
| STAT 4 | 423 (21%) | 201 (15%) | 117 (28%) | 74 (31%) | 31 (29%) | |
| STAT 5 | 157 (8%) | 41 (3%) | 57 (13%) | 40 (17%) | 19 (18%) | |
| STAT Unknown | 4 (0.3%) | 1 (0.2%) | 0 (0%) | 0 (0%) | 0 (0%) | |
| CPB time, Median (IQR), min | 100 (62–150) | 87 (53–132) | 120 (76–170) | 132 (85–185) | 135 (103–189) | < .001 |
| Aortic Cross Clamp Time, Median (IQR), min | 42 (17–70) | 38 (15–65) | 49 (19–78) | 52 (26–78) | 51 (36–78) | < .001 |
| Nephrotoxin Risk Score, Median (IQR) | 3 (2–4) | 3 (2–4) | 4 (3–5) | 4 (3–5) | 4 (3–5) | < .001 |
P values are from the univariate Kruskal-Wallis rank test for continuous variables and from the univariate Pearson χ2 test for categorical variables by stage of AKI
SI conversion factor: To convert creatinine l evel to micromoles per liter, multiply by 88.4.
Genetic syndromes identified include DiGeorge, Down, Noonan, Turner, Holt-Oram, Williams, CHARGE, VACTERL, or other genetic abnormalityAbbreviations: BSA, body surface area; CPB, cardiopulmonary bypass; IQR, interquartile range; STAT, Society of Thoracic Surgeons-European Association for Cardio-Thoracic Surgery.
Table II shows univariate associations of each covariate with the development of postoperative AKI as an ordinal and as a dichotomous outcome. A significant association was present for AKI as a dichotomous outcome with years of age (OR = 0.92 [0.91–0.94], P < .001), BSA (OR = 0.36 [0.29–0.46], P < .001), male sex (OR = 1.2 [1.02–1.47], P = .027), genetic syndrome (OR = 1.64 [1.32–2.02], P < .001), STAT score (OR = 1.69 [1.58–1.82], P < .001), CPB time (OR 1.07 [1.05–1.08], P < .001) per 10 minutes, cross clamp time (OR = 1.07 [1.05–1.10], P < .001) per 10 minutes, and nephrotoxin risk score (OR = 1.21 [1.16–1.31], P < .001). Ordinal results for AKI outcomes by stage were similar to those of dichotomous outcomes (Table II). In univariate analysis, there was also a significant association of the candidate NOS3 variant and dichotomous AKI outcome, with the major allele conferring decreased risk (OR 0.75 [0.62–0.90], P = .002). The remaining candidate variants were not significantly associated with the outcome of AKI. For children > 12 months of age the NOS3 major allele had more protective effect for AKI (OR 0.74 [0.57–0.96], P = .021) vs. infants < 12 months of age (OR 0.84 [0.71–0.99], P = .021) when analyzing AKI as a dichotomous outcome.
Table II.
U nivariate analyses of factors associated with AKI after surgery for congenital heart disease.
| Variable | Ordinal AKI outcome |
Dichotomous AKI outcome |
|||||
|---|---|---|---|---|---|---|---|
| OR | 95% CI | P-value | OR | 95% CI | P-value | ||
|
| |||||||
| Age, years | 0.92 | [0.90–0.94] | <.001 | 0.92 | [0.91 –0.94] | < .001 | |
| BSA (m2) | 0.35 | [0.28–0.44] | <.001 | 0.36 | [0.29–0.46] | < .001 | |
| Male Sex | 1.19 | [1.00–1.41] | .056 | 1.22 | [1.02–1.47] | .027 | |
| Genetic Syndrome STAT Score | 1.74 | [1.42–2.12] | <.001 | 1.64 | [1.32–2.02] | < .001 | |
| STAT Score 1 | 1.00 | Reference | - | 1.00 | Reference | - | |
| STAT Score 2 | 1.67 | [1.29–2.17] | <.001 | 1.67 | [1.28–2.17] | < .001 | |
| STAT Score 3 | 3.32 | [2.47–4.46] | <.001 | 3.29 | [2.43–4.45] | < .001 | |
| STAT Score 4 | 4.01 | [3.13–5.14] | <.001 | 4.13 | [3.19–5.35] | < .001 | |
| STAT Score 5 | 8.19 | [5.91–11.36] | <.001 | 10.58 | [7.12–15.73] | < .001 | |
| CPB time (per ten min) | 1.06 | [1.05–1.07] | <.001 | 1.07 | [1.05–1.08] | < .001 | |
| Aortic Cross Clamp time (per ten min) | 1.07 | [1.05–1.10] | <.001 | 1.07 | [1.05–1.10] | < .001 | |
| Nephrotoxin Risk score | 1.23 | [1.16–1.30] | <.001 | 1.23 | [1.16–1.31] | < .001 | |
| rs429358 | APOE | 1.06 | [0.83–1.35] | .635 | 1.06 | [0.82–1.37] | .655 |
| rs7412 | APOE | 1.05 | [0.87–1.26] | .637 | 1.08 | [0.89–1.31] | .455 |
| rs5186 | AGTR1 | 0.99 | [0.83–1.18] | .953 | 1.00 | [0.84–1.20] | .973 |
| rs2070744 | NOS3 | 0.80 | [0.67–0.95] | .012 | 0.75 | [0.62–0.90] | .002 |
| rs1800796 | IL6 | 1.00 | [0.79–1.27] | .986 | 0.99 | [0.77–1.26] | .927 |
| rs1800629 | TNF | 0.92 | [0.75–1.12] | .392 | 0.89 | [0.73–1.09] | .278 |
Abbreviations: AKI, acute kidney injury; BSA, body surface area; CPB, cardiopulmonary bypass; OR, Odds Ratio; STAT, Society of Thoracic Surgeons-European Association for Cardio-Thoracic Surgery.
In order to test for gene-dose effect, we performed a post hoc subanalysis using those homozygous for the NOS3 major allele as a reference group. We calculated ORs for homozygotes for the minor NOS3 alleles and for NOS3 heterozygotes (Supplemental Table IV). The decreased risk of AKI was similar in the homozygous and heterozygous minor allele carriers.
Given the significant association with genetic syndromes, a post hoc subanalysis of recognized syndromes or associations was performed to test the association of DiGeorge, Down, Noonan, Turner, Holt-Oram, Williams, CHARGE, VACTERL, or other genetic abnormality syndromes with AKI (Supplemental Table V). Of the identified syndromes only VACTERL (n = 19) had a significant association with AKI (OR = 2.59 [1.04–6.46], P = .04). Other genetic abnormalities identified on chromosomal or genetic testing were also significantly associated with AKI (OR = 1.76 [1.25–2.48], P < .01).
Results of the multivariable analysis using BSA, genetic syndrome, STAT category, bypass time (per ten minutes), nephrotoxin risk score and the NOS3 variant are shown in Figure 1. In this multivariable analysis, all covariates remained significant with NOS3 OR = 0.70, [0.58–0.85, P < .0001].
Figure 1.
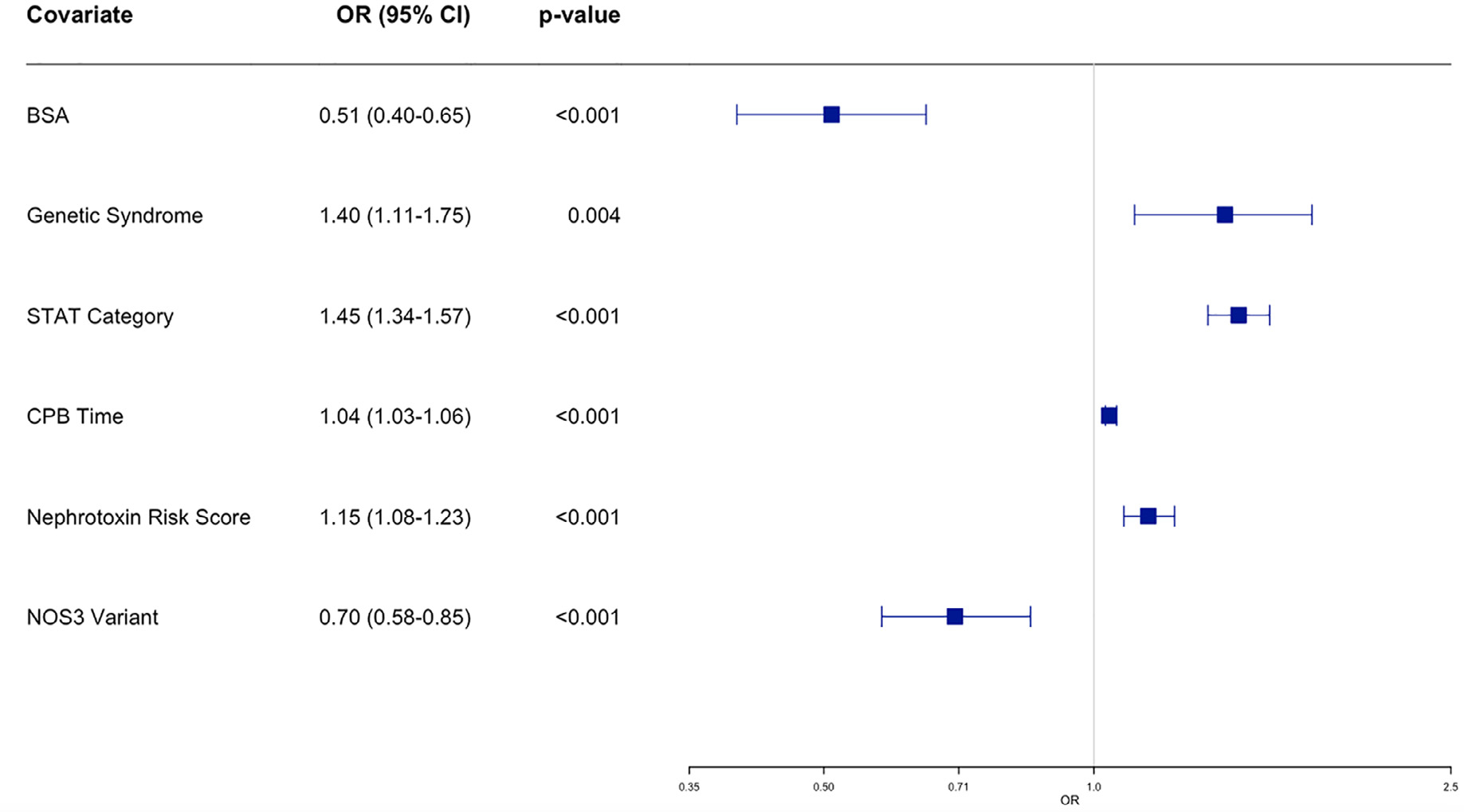
Multivariable Analysis for AKI as Dichotomous Predictor. Odds ratios (ORs) and 95% CIs for each of the clinical variables in the dichotomous multivariable logistic regression analysis for association with acute kidney injury. Odds ratios are for the following: Body surface area (BSA), identified preexisting genetic syndrome including (DiGeorge, Down, Noonan, Turner, Holt-Oram, Williams, CHARGE, VACTERL syndrome, or other identified genetic abnormality), Society of Thoracic Surgeons-European Association for Cardio-Thoracic Surgery (STAT) score, Cardiopulmonary bypass (CPB) time (per 10 minutes), Nephrotoxin Risk score which was equal to total number of unique exposures to high and moderate risk nephrotoxic medications, and presence of NOS3 rs2070744 allele variant. Point estimates and 95% CIs are shown to the left of each plot.
Discussion
AKI following pediatric cardiac surgery remains a significant component of postoperative morbidity. In our cohort, 37% of patients developed postoperative AKI, with most (54%) being categorized as stage 1 AKI. Age, BSA, sex, genetic syndromes, nephrotoxin exposure, and surgical complexity (as represented by STAT score, by-pass time, and cross clamp time) represent important risk factors for AKI in this study. A common variant in NOS3 was found to have a protective effect against the development of AKI in children with OR of 0.75 [0.62–0.90] in those with the presence of at least 1 minor allele.
Renal blood flow, cytokine production, and plasma lipoproteins are hypothesized to influence AKI risk.10,11 Previous studies in adults have demonstrated that common variants in genes that regulate renal blood flow and inflammation are associated with increased risk of postoperative AKI.8–13 To our knowledge, these associations for postoperative AKI have not been assessed in children, and the differences in individual risk remain challenging to predict. The candidate gene analysis performed did not demonstrate a significant association with common variants of APOE, AGTR, IL6, or TNF, but there was a protective effect of NOS3.
The NOS3 variant is in the promotor region of the gene which likely impacts gene expression and causes variations in the amount of NO produced and affects renal medullary blood flow.15,17 This risk allele has been studied by candidate gene approach in a wide variety of diseases including coronary artery disease,18,19 diabetes,20 preeclampsia,21 multiple sclerosis22 as well as others. While presence of the minor NOS3 allele variant has been associated with reduced transcription and NO production, Data et al.23 and Augeri et al 24 demonstrated that the presence of this allele was associated with a more favorable blood pressure response to exercise in adults. The functional characteristics of this variant have been suggested as a more dynamic contributor to hyper-emic effects after shear stress,23 particularly in otherwise healthy young adults. We hypothesize that the carriers of the NOS allele variant may have more dynamic effects in renal medullary blood flow creating a protective response to CPB and stress after surgery. The differences in pediatric physiology may also demonstrate the variations in this risk allele and its protective effect. In young infants, the kidneys receive as low as 2.5% to 4% of systemic blood flow as compared to 20% to 25% in adults. While systemic vascular resistance decreases and allows increased renal blood flow, there is subsequent stress on renal function in children.25, 26 In children > 12 months of age, the NOS3 variant demonstrated increased protective effects compared to infants < 12 months of age (OR 0.74 v. OR 0.84). The NOS3 variant protective effect shown in this study may be related to protective effects on regulating the dynamic changes in renal blood flow that cause increased stress and may lead to AKI. As physiologic renal blood flow increases, we hypothesize that individuals with this variant may have improved regulation of dynamic effects with decreased risk of AKI.
Our analysis also demonstrated an association of increased risk of AKI with children who have an already identified genetic syndrome. A recent meta-analysis by Van den Eynde et al.7 which included 11 studies with genetic syndrome as a baseline variable did not demonstrate a significant association with postoperative AKI. A post hoc analysis of our study population with sub-groups including patients with DiGeorge, Downs, Noonan, Turner, Holt-Oram, Williams, CHARGE, VACTERL syndrome, as well as other genetic abnormalities. Of the identified syndromes, VACTERL was significantly associated with postoperative AKI which we hypothesize is related to its associated renal anomalies, although the sample size for this subgroup was small (n = 19).
Identifying individual risk factors for pediatric patients who are at higher risk for postoperative AKI pose the possibility for alternative treatments. Patients who are homozygous for the NOS3 minor allele may be more predisposed to AKI due to increased shear stress and may benefit from interventions such as close blood pressure monitoring and control, careful fluid balance, minimization of nephrotoxic exposures, and high dose acetaminophen.27 Modern biomarkers including urinary neutrophil gelatinase-associated lipocalin (uNGAL) to creatinine ratio as well as others have been shows to be early predictors of cardiac-AKI in children.28,29 The use of risk stratification using genetic variants and these biomarkers as the relationship between these predictors may provide benefit to patient management. Understanding genetic differences that may affect risk could also help drive the discovery and use of other biomarkers, such as those related to renal blood flow and stress. This data may also be useful in postoperative and medical interventions to help decrease renal damage. There have been several studies analyzing potential therapeutic benefits to altering NOS3 to improve cardiovascular and renal outcomes in adults.30,31 Pharmacologic agents and nutritional applications to alter NO bioactivity and reduce oxidative stress have the potential to alter disease development and progression but are not well understood in children.32 Future directions may include development of treatment strategies based on this novel genetic association in this pediatric population.
Although there was a large study group analyzed, further prospective studies are required to further analyze the observed results. Other genetic contributions that were not tested in this study may help to explain the differences in individual risk. Understanding how genetic associations contribute to individual risks remain an important factor in helping reduce the morbidity and mortality incurred by renal injury in the postoperative period.
Limitations
This retrospective observational study does not inform causality of AKI. Only a limited number of variants were analyzed and it does not preclude the likelihood that there are other genetic associations with postoperative AKI. Additionally, this study was performed at a single tertiary care center which may represent potential selection bias. Our definition of AKI was based on KDIGO creatinine criteria and not urine output due to difficulty in measurement in children and inaccuracy of retrospective data. The NOS3 variant may be in linkage disequilibrium with another variant that contributes directly to AKI risk. Multivariable analysis was performed to adjust for potential confounders including size, nephrotoxin exposure, STAT score, CPB time, however there may be additional unmeasured confounders.
Conclusion
In this study, younger age, smaller BSA, male sex, known genetic syndrome, STAT score, CBP time, aortic cross clamp time, nephrotoxin risk score were all associated with outcome of AKI in children undergoing cardiac surgery. On genetic analysis of 6 common variants that affect renal blood flow and inflammation, a variant in NOS3 was found to be protective of AKI. These findings emphasize the importance of genetic risk prediction developing prediction models for pediatric postoperative AKI.
Supplementary Material
Acknowledgments
We thank study nurses Kim Crum, RN, BS, Carla Hissam, RN, CPN, and Darlene Fountain, RN, BSN (Vanderbilt University Medical Center Department) as well as all the study participants and families.
Funding
The project described was supported by CTSA award UL1 TR002243 from the National Institutes of Health (NIH) National Center for Advancing Translational Sciences (NCATS) and R01 HD084461 from the NIH Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD). SK is supported by T32HL105334. SLV and PJK are supported by P50 HD106446 from NICHD. Genotyping for some individuals was funded by R01 GM132204 from NIH National Institute of General Medical Sciences (NIGMS). This project utilized REDCap, funded by UL1 TR000445 from NIH NCATS. The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication. The authors are solely responsible for the design and conduct of this study, all study analyses, the drafting and editing of the paper and its final contents.
Footnotes
Conflict of interest
None reported.
Reprint requests: Sandra Kikano, MD, 2220 Children’s Way, Suite 5230, Nashville, TN 37232-9119.
Supplementary materials
Supplementary material associated with this article can be found, in the online version, at doi: 10.1016/j.ahj.2022.08.003.
References
- 1.Van den Eynde J, Rotbi H, Gewillig M, et al. In-hospital outcomes of acute kidney injury after pediatric cardiac surgery: a meta-analysis. Front Pediatr 2021;9. doi: 10.3389/fped.2021.733744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tóth R, Breuer T, Cserép Z, et al. Acute kidney injury is associated with higher morbidity and resource utilization in pediatric patients undergoing heart surgery. Ann Thorac Surg 2012;93:1984–90. doi: 10.1016/j.athoracsur.2011.10.046. [DOI] [PubMed] [Google Scholar]
- 3.Bastin AJ, Ostermann M, Slack AJ, et al. Acute kidney injury after cardiac surgery according to risk/injury/failure/loss/end-stage, acute kidney injury network, and kidney disease: improving global outcomes classifications. J Crit Care 2013;28:389–96. doi: 10.1016/j.jcrc.2012.12.008. [DOI] [PubMed] [Google Scholar]
- 4.Lex DJ, Tóth R, Cserép Z, et al. A comparison of the systems for the identification of postoperative acute kidney injury in pediatric cardiac patients. Ann Thorac Surg 2014;97:202–10. doi: 10.1016/j.athoracsur.2013.09.014. [DOI] [PubMed] [Google Scholar]
- 5.Sethi SK, Sharma R, Gupta A, et al. Long-term renal outcomes in children with acute kidney injury post cardiac surgery. Kidney Int Rep 2021;6:1850–7. doi: 10.1016/j.ekir.2021.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Blinder JJ, Goldstein SL, Lee VV, et al. Congenital heart surgery in infants: effects of acute kidney injury on outcomes. J Thorac Cardiovasc Surg 2012;143:368–74. doi: 10.1016/j.jtcvs.2011.06.021. [DOI] [PubMed] [Google Scholar]
- 7.Van den Eynde J, Delpire B, Jacquemyn X, et al. Risk factors for acute kidney injury after pediatric cardiac surgery: a meta-analysis. Pediatr Nephrol 2022;37:509–19. doi: 10.1007/s00467-021-05297-0. [DOI] [PubMed] [Google Scholar]
- 8.Isbir SC, Tekeli A, Ergen A, et al. Genetic polymorphisms contribute to acute kidney injury after coronary artery bypass grafting. Heart Surg Forum 2007;10:E439–44. doi: 10.1532/HSF98.20071117. [DOI] [PubMed] [Google Scholar]
- 9.Vilander LM, Kaunisto MA, Pettilä V. Genetic predisposition to acute kidney injury–a systematic review. BMC Nephrol 2015;16:197. doi: 10.1186/s12882-015-0190-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gaudino M, Di Castelnuovo A, Zamparelli R, et al. Genetic control of postoperative systemic inflammatory reaction and pulmonary and renal complications after coronary artery surgery. J Thorac Cardiovasc Surg 2003;126:1107–12. doi: 10.1016/s0022-5223(03)00396–9. [DOI] [PubMed] [Google Scholar]
- 11.Boehm J, Eichhorn S, Kornek M, et al. Apolipoprotein E genotype, TNF-α 308G/A and risk for cardiac surgery associated-acute kidney injury in Caucasians. Ren Fail 2014;36:237–43. doi: 10.3109/0886022X.2013.835267. [DOI] [PubMed] [Google Scholar]
- 12.Hsu CC, Kao WHL, Coresh J, et al. Apolipoprotein E and progression of chronic kidney disease. JAMA 2005;293:2892–9. doi: 10.1001/jama.293.23.2892. [DOI] [PubMed] [Google Scholar]
- 13.Stafford-Smith M, Podgoreanu M, Swaminathan M, et al. Association of genetic polymorphisms with risk of renal injury after coronary bypass graft surgery. Am J Kidney Dis 2005;45:519–30. doi: 10.1053/j.ajkd.2004.11.021. [DOI] [PubMed] [Google Scholar]
- 14.The kidney diseases improving global outcomes (KDIGO) working group. definition and classification of acute kidney injury. Kidney Int 2012(suppl 2):19–36.22437409 [Google Scholar]
- 15.James MT, Hobson CE, Darmon M, et al. Applications for detection of acute kidney injury using electronic medical records and clinical information systems: workgroup statements from the 15(th) ADQI consensus conference. Can J Kidney Health Dis 2016;3:9. doi: 10.1186/s40697-016-0100-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.O’Brien SM, Clarke DR, Jacobs JP, et al. An empirically based tool for analyzing mortality associated with congenital heart surgery. J Thorac Cardiovasc Surg 2009;138:1139–53. doi: 10.1016/j.jtcvs.2009.03.071. [DOI] [PubMed] [Google Scholar]
- 17.Jenkins KJ, Gauvreau K, Newburger JW, et al. Consensus-based method for risk adjustment for surgery for congenital heart disease. J Thorac Cardiovasc Surg 2002;123:110–18. doi: 10.1067/mtc.2002.119064. [DOI] [PubMed] [Google Scholar]
- 18.Nakayama M, Yasue H, Yoshimura M, et al. T-786–>C mutation in the 5’-flanking region of the endothelial nitric oxide synthase gene is associated with coronary spasm. Circulation 1999;99:2864–70. doi: 10.1161/01.cir.99.22.2864. [DOI] [PubMed] [Google Scholar]
- 19.Cowley AW, Mori T, Mattson D, Zou AP. Role of renal NO production in the regulation of medullary blood flow. Am J Physiol Regul Integr Comp Physiol 2003;284:R1355–69. doi: 10.1152/ajpregu.00701.2002. [DOI] [PubMed] [Google Scholar]
- 20.Dong J, Ping Y, Wang Y, Zhang Y. The roles of endothelial nitric oxide synthase gene polymorphisms in diabetes mellitus and its associated vascular complications: a systematic review and meta-analysis. Endocrine 2018;62:412–22. doi: 10.1007/s12020-018-1683-4. [DOI] [PubMed] [Google Scholar]
- 21.Zhou L, Hui X, Yuan H, et al. Combination of genetic markers and age effectively facilitates the identification of people with high risk of preeclampsia in the han chinese population. Biomed Res Int 2018;2018. doi: 10.1155/2018/4808046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Heidari MM, Khatami M, Tahamtan Y. Molecular analysis of rs2070744 and rs1799983 polymorphisms of NOS3 gene in iranian patients with multiple sclerosis. Basic Clin Neurosci 2017;8:279–84. doi: 10.18869/nirp.bcn.8.4.279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Data SA, Roltsch MH, Hand B, et al. eNOS T-786C genotype, physical activity, and peak forearm blood flow in females. Med Sci Sports Exerc 2003;35:1991–7. doi: 10.1249/01.MSS.0000099105.99682.8B. [DOI] [PubMed] [Google Scholar]
- 24.Augeri AL, Tsongalis GJ, Van Heest JL, et al. The endothelial nitric oxide synthase −786 T>C polymorphism and the exercise-induced blood pressure and nitric oxide responses among men with elevated blood pressure. Atherosclerosis 2009;204:e28–34. doi: 10.1016/j.atherosclerosis.2008.12.015. [DOI] [PubMed] [Google Scholar]
- 25.Selewski DT, Charlton JR, Jetton JG, et al. Neonatal acute kidney injury. Pediatrics 2015;136:e463–73. doi: 10.1542/peds.2014-3819. [DOI] [PubMed] [Google Scholar]
- 26.Yao LP, Jose PA. Developmental renal hemodynamics. Pediatr Nephrol 1995;9:632–7. doi: 10.1007/BF00860962. [DOI] [PubMed] [Google Scholar]
- 27.Van Driest SL, Jooste EH, Shi Y, et al. Association between early postoperative acetaminophen exposure and acute kidney injury in pediatric patients undergoing cardiac surgery. JAMA Pediatr 2018;172:655–63. doi: 10.1001/jamapediatrics.2018.0614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Van den Eynde J, Schuermans A, Verbakel JY, et al. Biomarkers of acute kidney injury after pediatric cardiac surgery: a meta-analysis of diagnostic test accuracy. Eur J Pediatr 2022;181:1909–21. doi: 10.1007/s00431-022-04380-4. [DOI] [PubMed] [Google Scholar]
- 29.Hazle MA, Gajarski RJ, Aiyagari R, et al. Urinary biomarkers and renal near-infrared spectroscopy predict intensive care unit outcomes after cardiac surgery in infants younger than 6 months of age. J Thorac Cardiovasc Surg 2013;146:861–7. doi: 10.1016/j.jtcvs.2012.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Carlström M Nitric oxide signalling in kidney regulation and cardiometabolic health. Nat Rev Nephrol 2021;17:575–90. doi: 10.1038/s41581-021-00429-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rochette L, Lorin J, Zeller M, et al. Nitric oxide synthase inhibition and oxidative stress in cardiovascular diseases: possible therapeutic targets? Pharmacol Ther 2013;140:239–57. doi: 10.1016/j.pharmthera.2013.07.004. [DOI] [PubMed] [Google Scholar]
- 32.Hsu CN, Tain YL. Asymmetric dimethylarginine (ADMA) in pediatric renal diseases: from pathophysiological phenomenon to clinical biomarker and beyond. Children (Basel) 2021;8:837. doi: 10.3390/children8100837. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


