Abstract
Background
We assessed the impact of pre‐ and postprocedural plasma corin levels on the recurrence of atrial fibrillation (AF) after catheter ablation (CA).
Methods and Results
This prospective, single‐center, observational study included patients undergoing their first CA of AF. Corin was measured before and 1 day after CA. The primary end point was recurrent AF between 3 and 12 months after ablation. From April 2019 through May 2021, we analyzed 616 patients with AF (59.09% men) with a mean age of 62.86±9.42 years. Overall, 153 patients (24.84%) experienced recurrent AF. In the recurrence group, the pre‐ and postprocedure corin concentrations were 539.14 (329.24–702.08) and 607.37 (364.50–753.80) pg/mL, respectively, which were significantly higher than the nonrecurrence group's respective concentrations of 369.05 (186.36–489.28) and 489.12 (315.66–629.05) pg/mL (both P<0.0001). A multivariate Cox regression analysis with confounders found that elevated preablation corin levels were significantly associated with an increased risk of AF recurrence after CA. Receiver operating characteristic curve analysis identified that a preablation corin threshold of >494.85 pg/mL predicted AF recurrence at 1 year. An increase of 1 SD in corin concentrations before CA (264.94 pg/mL) increased the risk of recurrent AF by 54.3% after adjusting for confounding variables (hazard ratio, 1.465 [95% CI, 1.282–1.655]; P<0.0001).
Conclusions
Plasma corin levels at baseline is a valuable predictor of AF recurrence after CA, independent of established conventional risk factors. Risk stratification before ablation for AF may be useful in selecting treatment regimens for patients.
Keywords: atrial fibrillation, catheter ablation, corin, natriuretic peptides, recurrence
Subject Categories: Atrial Fibrillation, Diagnostic Testing, Biomarkers, Clinical Studies
Nonstandard Abbreviations and Acronyms
- AAD
antiarrhythmic drug
- FIRE AND ICE
Comparative Study of Two Ablation Procedures in Patients With Atrial Fibrillation
- PaAF
paroxysmal atrial fibrillation
- PeAF
persistent atrial fibrillation
- PVI
pulmonary vein isolation
- PVs
pulmonary veins
Clinical Perspective.
What Is New?
Elevated plasma corin levels at baseline are strongly associated with an increased incidence of atrial fibrillation (AF) recurrence after catheter ablation.
An increase of 1 SD in corin concentrations before ablation (264.94 pg/mL) increased the risk of recurrent AF by 54.3% after adjusting for confounding variables.
What Are the Clinical Implications?
A higher level of circulating corin at baseline could be a predictor of the risk of recurrent of AF after ablation.
Corin may play a considerable role in AF and may therefore serve as a candidate factor to predict recurrent AF after ablation.
Atrial fibrillation (AF) is a common cardiac arrhythmia that increases death and morbidity worldwide and has an estimated prevalence of 0.65% to 0.74% in the Chinese population. 1 , 2 Due to the complex molecular mechanisms of AF, the current therapeutic outcome of AF is not satisfactory.
Catheter ablation (CA) is a curative therapy for AF, but it has a substantial recurrence rate that varies between 50% and 75%, 3 , 4 and 24% to 39.4% in China. 5 , 6 Some patients may even require repeated CA or long‐term oral antiarrhythmic drugs (AADs). Therefore, the careful selection of patients is warranted to identify those likely to receive the greatest benefit. Several clinical factors have been identified as predictors of AF recurrence after CA, such as first‐pass isolation, 7 left atrial size and volume, 8 and biomarkers. The latter include myocardial injury markers, natriuretic peptides and inflammatory markers, oxidative stress biomarkers, and microRNA markers. 9
Corin is a type II transmembrane serine protease that is expressed primarily in cardiomyocytes. 10 It has been identified as the enzyme responsible for activating cardiac natriuretic peptides. 11 Corin is important in regulating the salt–water balance, blood pressure, and cardiac function. 12 , 13 Corin expressed at the cardiomyocyte surface could be shed through corin autocleavage and metalloproteinase‐mediated hydrolysis. 14 Therefore, corin molecules can enter the circulation, and soluble corin is detectable. 15 Soluble and membrane‐bound corin have similar physiological activities in processing proatrial natriuretic peptide (pro‐ANP). 16 Recent studies have demonstrated that circulating soluble corin has the potential to be a specific, sensitive biomarker for risk prediction and prognostic assessment in cardiovascular diseases (CVDs), such as hypertension, heart failure (HF), acute myocardial infarction (MI), preeclampsia, and AF. 17 , 18 , 19 , 20 Our previous studies found high plasma corin levels in patients with AF and a positive correlation between plasma corin levels and left atrial diameter or PR interval in patients with AF. 21 Variant rs3749585T in the 3′ untranslated region of CORIN has been significantly and negatively associated with AF, 22 but its utility in predicting AF recurrence after CA has not been investigated.
In this context, this prospective, single‐center, observational study assessed the impact of plasma corin levels (measured before and after CA for AF) on the rate of recurrence of AF at 1 year after the initial CA.
Methods
The data that support the findings of this study are available from the corresponding author upon reasonable request. We conducted a prospective, single‐center, observational study in the First Affiliated Hospital of Dalian Medical University from April 2019 through May 2021. The study protocol was approved by the hospital's ethics committee and adhered to the guidelines set forth by the Declaration of Helsinki. All the patients signed informed consent forms before enrollment.
Study Population
The inclusion criteria embraced patients aged >18 years with highly symptomatic paroxysmal AF (PaAF) or persistent AF (PeAF) who had been referred to the hospital for their first CA.
The exclusion criteria included patients with >1 CA or surgical ablation for AF; patients with significant valvular heart disease; those with a left ventricular ejection fraction of <40% or a recent (<1 month) decompensation of HF; those with severe coronary artery disease (CAD) or a recent (<1 month) MI; those with systemic inflammatory diseases, an impaired estimated glomerular filtration rate (eGFR) (<60 mL/min per 1.73 m2), hyperthyroidism, pulmonary hypertension with a pulmonary artery pressure of >45 mm Hg as determined by transthoracic echocardiography, or chronic respiratory disease; and patients aged <18 years.
Clinical Measurements and Definition of Explanatory Variables
The clinical data in the medical records included age, sex, body mass index (BMI), hypertension, CAD, HF, eGFR, diabetes, and AF type. The lipid factors included total cholesterol, triglyceride, low‐density lipoprotein cholesterol, and high‐density lipoprotein cholesterol (HDL‐C). Heart rate, PR interval, QRS, and corrected QT interval were recorded by standard 12‐lead ECG, while transthoracic echocardiography was employed to record left–right diameter of left atrium, superior–inferior diameter of left atrium, left–right diameter of right atrium, superior–inferior diameter of right atrium, interventricular septal thickness, right ventricular end‐diastolic diameter, left ventricular end‐diastolic diameter, and left ventricular ejection fraction. The data on pulmonary veins (PVs) were obtained by PV computed tomography angiography. Information on medications was also collected.
PaAF was defined as AF episodes lasting <7 days that terminated spontaneously. PeAF was defined as AF episodes lasting >7 days or requiring termination with pharmacologic or direct current cardioversion according to current guidelines. 23 Hypertension was defined as a systolic blood pressure ≥140 mm Hg or diastolic blood pressure ≥90 mm Hg or active use of antihypertensive drugs. Diabetes was diagnosed as either a fasting plasma glucose value of ≥7.0 mmol/L, a 2‐hour plasma glucose value of ≥11.1 mmol/L, or glycosylated hemoglobin of ≥6.5% according to the 2020 American Diabetes Association standards of medical care in diabetes. 24
Ablation Procedure
All the patients took effective oral anticoagulants for at least 1 month and underwent transesophageal echocardiography to verify the absence of thrombus before CA. Oral anticoagulants were taken continually in the periprocedural period, and the procedures were performed by qualified experienced operators. Only 1 type of procedure, radiofrequency CA, was performed throughout the whole study. The aim of the procedure was PV isolation (PVI). Additional lesions were made at the discretion of the operator. The details of the electrophysiological study and 3‐dimensional mapping have been described in previous studies. 25 After femoral venous access was obtained, 2 multipolar catheters were placed at the coronary sinus and right ventricle apex. A dual transseptal puncture was performed under fluoroscopic guidance for delivery of two 8.5‐F long sheaths (SL1, St. Jude Medical, St. Paul, MN) into the left atrium. An ablation catheter (ThermoCool SMARTTOUCH SF, Biosense Webster Inc., Irvine, CA) and a high‐density mapping catheter (PentaRay, Biosense Webster Inc.) were placed in the left atrium through the 2 sheaths. In patients with PaAF, circumferential PVI was achieved with the guidance of the CARTO system (Biosense Webster Inc.). In patients with PeAF, ablation was achieved by a stepwise approach. If AF continued after circumferential PVI, the following linear ablations were performed sequentially on the basis of the mapping results: roof line of left atrium, left atrial posterior wall line, mitral isthmus line, left atrial anterior wall line, inferior vena cava tricuspid annual isthmus line, and complex fractionated atrial electrogram ablation. If PeAF was not terminated after the procedure above, electric cardioversion was performed to restore sinus rhythm. The end point of ablation included AF termination; atrial arrhythmias could not be induced after cardioversion in sinus rhythm state; establishment of a bidirectional conduction block, and voltage reduction or disappearance of complex fractionated atrial electrogram disease. If the AF organized to atrial flutter, entrainment mapping was performed to target and ablate the critical isthmus. The radiofrequency energy was 35 to 45 W, with a temperature setting of no more than 43 to 48 °C. AADs could be continued at the operator's discretion to maintain sinus rhythm.
Corin Measurements
The participants' peripheral venous blood samples were collected into sodium citrate coagulation test tubes before and 1 day after CA. Plasma was obtained by centrifugation at 845g for 10 minutes and then stored at −20 °C for later centralized analysis. Plasma corin concentrations were measured with human corin Quantikine ELISA kits (Catalog: DCRN00, R&D Systems, Minneapolis, MN). 21 , 26 , 27
Follow‐Up and Study End Points
All the patients were followed up at regular, predefined intervals (1, 3, 6, 9, and 12 months after the procedure) at our cardiology clinic or with their referring physician, with additional visits as required. Patients who did not attend the regular visits were contacted by telephone. The primary end point was recurrence of AF, defined as occurrence between 3 and 12 months after CA following a documented (by ECG, Holter monitoring) episode of AF, atrial tachycardia, or atrial flutter lasting >30 seconds, excluding the events during the blanking period of 3 months after ablation.
Statistical Analysis
Statistical analysis was performed with Statistical Package for Social Sciences, version 24.0 (IBM, Armonk, NY), R software version 4.3.1 (R Foundation for Statistical Computing, Vienna, Austria), and Prism version 9 (GraphPad Software, San Diego, CA). Categorical variables (sex, PaAF, hypertension, diabetes, CAD, HF, and medications) were expressed as percentages and were compared using Pearson's chi squared test. Continuous variables (age, BMI, eGFR, lipid factors, ECG parameters, transthoracic echocardiography parameters, and PV parameters) were expressed as means±SD and were compared between groups using Student's t test. Plasma corin levels were expressed as median (25th percentile [quartile 1] to 75th percentile [quartile 3]) and were compared using the Mann–Whitney test. Correlations between continuous variables were analyzed with Spearman's rank correlation coefficient. Receiver operating characteristic curves and Youden index were constructed to identify the threshold of corin that best predicted recurrence. Patients were categorized on the basis of their corin concentrations according to whether the concentration was above or below the threshold value. The threshold values differed according to sex and different AF types. Survival curves were generated using the Kaplan–Meier analysis, with a log‐rank test assessing the differences.
Cox regression analyses (adjusted for age, sex, BMI, eGFR, triglyceride, HDL‐C, AADs, β‐blockers, and corin levels before and after CA, which were statistically different at baseline between the groups) were performed for the primary end point, with corin as continuous and dichotomous variables (low versus high concentrations). The continuous variables including corin values were normalized by Z‐score normalization, and 1 SD was used to calculate the hazard ratio (HR). Binary logistic regression analysis (also adjusted for age, sex, BMI, eGFR, triglyceride, HDL‐C, AADs, β‐blockers, and corin levels before or after CA) was performed for the independent predictors of recurrent AF after CA. Statistical tests were 2‐sided, and P<0.05 was considered statistically significant.
Results
Baseline Characteristics
This study initially recruited 681 inpatients who intended to undergo a first CA for AF. Patients with valvular heart disease (n=6), hyperthyroidism (n=2), and severe kidney dysfunction (n=2) were excluded. Also excluded were 37 patients who refused CA, 6 patients with implanted pacemakers, 3 patients who had undergone concurrent pacemaker implantation and atrioventricular nodal ablation, 1 patient diagnosed with acute MI, 1 patient diagnosed with acute pulmonary thromboembolism, 6 patients who did not complete follow‐up, and 1 patient who died during follow‐up. Ultimately, a total of 616 patients were included in the study (Figure 1).
Figure 1. Flow diagram of inclusion and exclusion of study subjects.
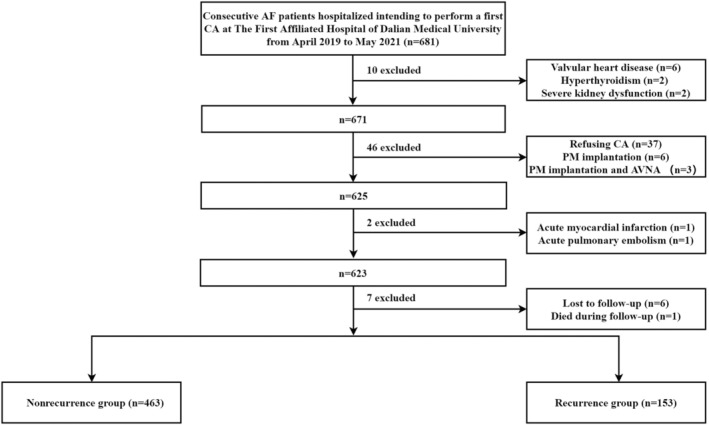
AF indicates atrial fibrillation; AVNA, atrioventricular nodal ablation; CA, catheter ablation; and PM, pacemaker.
The baseline characteristics of the study population are displayed in Table 1. The mean age was 62.86 years; 59.09% were men, and 61.36% had PaAF. During a follow‐up period of 1 year, 153 patients (24.84%) had recurrent AF (recurrence group). Overall, compared with the nonrecurrence group, patients in the recurrence group were younger, predominantly men, had higher BMI and triglyceride, and had lower HDL‐C. Patients in the recurrence group were more likely to take AADs (amiodarone, propafenone, and dronedarone) but less likely to take β‐blockers (metoprolol and bisoprolol).
Table 1.
Baseline and Corin Concentrations of the Total Cohort, Nonrecurrence, and Recurrence Group of Atrial Fibrillation After Catheter Ablation
| Variables | Total cohort (n=616) | Nonrecurrence group (n=463) | Recurrence group (n=153) | P value | |
|---|---|---|---|---|---|
| Age, y | 62.86±9.42 | 63.33±9.26 | 61.44±9.78 | 0.032 | |
| Male, n (%) | 364 (59.09) | 260 (56.16) | 104 (67.97) | 0.01 | |
| BMI, kg/m2 | 25.53±3.34 | 25.37±3.32 | 26.00±3.39 | 0.041 | |
| History | |||||
| PaAF, n (%) | 378 (61.36) | 292 (63.07) | 86 (56.21) | 0.151 | |
| Hypertension, n (%) | 320 (51.95) | 244 (52.7) | 76 (49.67) | 0.516 | |
| Diabetes, n (%) | 124 (20.13) | 97 (20.95) | 27 (17.65) | 0.417 | |
| Coronary artery disease, % | 79 (12.82) | 60 (12.96) | 19 (12.42) | 0.862 | |
| Heart failure, % | 98 (15.91) | 78 (16.85) | 20 (13.07) | 0.309 | |
| Laboratory values | |||||
| eGFR, mL/min per 1.73 m2 | 90.99±13.69 | 90.44±13.5 | 92.69±14.15 | 0.078 | |
| TC, mmol/L | 4.35±1.03 | 4.36±0.99 | 4.31±1.12 | 0.600 | |
| Triglyceride, mmol/L | 1.52±0.92 | 1.47±0.89 | 1.65±0.98 | 0.037 | |
| LDL‐C, mmol/L | 2.38±0.79 | 2.38±0.79 | 2.36±0.81 | 0.803 | |
| HDL‐C, mmol/L | 1.09±0.27 | 1.12±0.27 | 1.01±0.25 | <0.0001 | |
| ECG parameters | |||||
| Heart rate, bpm | 70.78±11.45 | 70.9±11.33 | 70.44±11.81 | 0.704 | |
| PR interval, ms | 168.45±25.67 | 167.71±24.57 | 170.76±28.83 | 0.258 | |
| QRS, ms | 89.85±15.21 | 89.22±14.45 | 91.83±17.27 | 0.103 | |
| QTc, ms | 446.86±31.38 | 447.16±31.56 | 445.95±30.94 | 0.716 | |
| TTE parameters | |||||
| LALRD, mm | 42.61±4.98 | 42.51±4.68 | 39.82±4.37 | 0.387 | |
| LASID, mm | 55.9±6.16 | 55.73±6.26 | 56.41±5.82 | 0.249 | |
| RALRD, mm | 38.48±4.92 | 38.42±5.01 | 38.68±4.63 | 0.583 | |
| RASID, mm | 50.36±5.78 | 50.2±5.90 | 50.84±5.38 | 0.251 | |
| IVST, mm | 10.41±1.51 | 10.44±1.60 | 10.29±1.22 | 0.302 | |
| RVEDD, mm | 17.55±1.76 | 17.47±1.78 | 17.79±1.70 | 0.054 | |
| LVEDD, mm | 47.54±4.48 | 47.37±4.55 | 48.06±4.20 | 0.106 | |
| LVEF, % | 56.25±5.21 | 56.23±5.16 | 56.32±5.38 | 0.847 | |
| PV parameters* | |||||
| LSPV | SID, mm | 17.83±3.73 | 17.84±3.78 | 17.82±3.61 | 0.958 |
| APD, mm | 15.96±4.14 | 16.03±4.21 | 15.73±3.91 | 0.461 | |
| LIPV | SID, mm | 15.93±2.81 | 15.9±2.83 | 15.99±2.78 | 0.753 |
| APD, mm | 12.84±3.57 | 12.8±3.66 | 12.96±3.29 | 0.661 | |
| RSPV | SID, mm | 18.11±3.71 | 18.21±3.78 | 17.82±3.48 | 0.288 |
| APD, mm | 16.58±3.51 | 16.66±3.52 | 16.33±3.47 | 0.348 | |
| RIPV | SID, mm | 16.97±3.38 | 16.96±3.37 | 16.98±3.42 | 0.952 |
| APD, mm | 15.21±3.51 | 15.21±3.47 | 15.21±3.64 | 0.990 | |
| Treatments | |||||
| AADs, n (%)† | 425 (68.99) | 303 (65.44) | 122 (79.74) | 0.001 | |
| Antiplates, n (%) | 17 (2.76) | 11 (2.38) | 6 (3.92) | 0.391 | |
| β‐blockers, n (%) | 152 (24.68) | 127 (27.43) | 25 (16.34) | 0.007 | |
| Digoxin, n (%) | 4 (0.65) | 4 (0.86) | 0 (0) | 0.577 | |
| Diltiazem, n (%) | 27 (4.38) | 19 (4.1) | 8 (5.23) | 0.648 | |
| Loop diuretics, n (%) | 61 (9.9) | 45 (9.72) | 16 (10.46) | 0.757 | |
| RAAS blockers, n (%)‡ | 208 (33.77) | 155 (33.48) | 53 (34.64) | 0.844 | |
| D‐CCB, n (%) | 137 (22.24) | 101 (21.81) | 36 (23.53) | 0.655 | |
| Statins, n (%) | 319 (51.79) | 238 (51.4) | 81 (52.94) | 0.780 | |
| Spironolactone, n (%) | 69 (11.2) | 50 (10.8) | 19 (12.42) | 0.558 | |
| Nitrates, n (%) | 26 (4.22) | 23 (4.97) | 3 (1.96) | 0.161 | |
| Corin concentrations | |||||
| Preablation, pg/mL | 411.30 (209.04, 552.32) | 369.05 (186.36, 489.28) | 539.14 (329.24, 702.08) | <0.0001 | |
| Postablation, pg/mL | 518.50 (324.50, 558.99) | 489.12 (315.66, 629.05) | 607.37 (364.50, 753.80) | <0.0001 | |
AADs indicates antiarrhythmic drugs; APD, anteroposterior diameter; BMI, body mass index; BNP, type B natriuretic peptide; D‐CCB, dihydropyridine calcium channel blocker; eGFR, estimated glomerular filtration rate; HDL‐C, high‐density lipoprotein cholesterol; IVST, interventricular septal thickness; LALRD, left–right diameter of left atrium; LASID, superior–inferior diameter of left atrium; LDL‐C, low‐density lipoprotein cholesterol; LIPV, left inferior pulmonary vein; LSPV, left superior pulmonary vein; LVEDD, left ventricular end‐diastolic diameter; LVEF, left ventricular ejection fraction; PaAF, paroxysmal atrial fibrillation; PVs, pulmonary veins; QTc, corrected QT interval; RAAS, renin–angiotensin–aldosterone system; RALRD, left–right diameter of right atrium; RASID, superior–inferior diameter of right atrium; RIPV, right inferior pulmonary vein; RSPV, right superior pulmonary vein; RVEDD, right ventricular end‐diastolic diameter; SID, superior–inferior diameter; TC, total cholesterol; TG, triglyceride; and TTE, transthoracic echocardiography.
Excluding 18 patients with 5 pulmonary veins and 19 patients with 3 pulmonary veins.
Including amiodarone, propafenone, and dronedarone.
Including angiotensin‐converting enzyme inhibitor, angiotensin receptor blocker, and angiotensin receptor/neprilysin inhibitor.
CA Procedures
The PVs were successfully isolated in all 616 patients (100%). The ablation frequencies of the left atrial anterior wall, mitral isthmus, and coronary sinus ablation in the patients in the recurrence group were greater than those of patients in the nonrecurrence group. A total of 58 patients (9.42%) had left atrial appendage occlusion, including 46 patients (9.94%) in the nonrecurrence group and 11 (7.19%) in the recurrence group (Table 2). Regarding complications, 1 patient had cardiac perforation, 1 had a groin site complication, and 1 had hemorrhagic stroke.
Table 2.
Procedural Data in Patients With and Without Recurrence of Atrial Fibrillation After Catheter Ablation
| Catheter ablation techniques | Total cohort (n=616) | Nonrecurrence group (n=463) | Recurrence group (n=153) | P value |
|---|---|---|---|---|
| Pulmonary vein isolation (%) | 616 (100) | 463 (100) | 153 (100) | 1 |
| LA roof (%) | 43 (7.14) | 28 (6.25) | 15 (9.8) | 0.114 |
| LA posterior wall (%) | 96 (15.58) | 65 (14.04) | 31 (20.26) | 0.066 |
| LA anterior wall (%) | 39 (6.33) | 21 (4.54) | 18 (11.76) | 0.001 |
| IVC–tricuspid annulus isthmus (%) | 88 (14.29) | 67 (14.47) | 21 (13.73) | 0.819 |
| SVC (%) | 19 (3.08) | 15 (3.24) | 4 (2.61) | 0.698 |
| Mitral isthmus (%) | 37 (6.01) | 19 (4.1) | 18 (11.76) | 0.001 |
| Coronary sinus (%) | 23 (3.73) | 13 (2.81) | 10 (6.54) | 0.035 |
| CFAEs (%) | 2 (0.32) | 1 (0.22) | 1 (0.65) | 0.409 |
| LAAO (%) | 58 (9.42) | 46 (9.94) | 11 (7.19) | 0.31 |
CFAEs indicates complex fractionated atrial electrograms; IVC, inferior vena cava; LA, left atrial; LAAO, left atrial appendage occlusion and SVC, superior vena cava.
Pre‐/Postprocedural Plasma Corin Levels in Patients Treated With CA for AF
In the total cohort, the plasma corin concentrations before the procedure were significantly lower than those after the procedure (411.30 [209.04–552.32] versus 518.50 [324.50–558.99] pg/mL, respectively; P<0.0001). In the recurrence group, the corin concentrations drawn before and after the procedure were 539.14 (329.24–702.08) and 607.37 (364.50–753.80) pg/mL, respectively, and those were significantly higher than those in the nonrecurrence group (369.05 [186.36–489.28] and 489.12 [315.66–629.05], respectively; both P<0.0001; Table 3, Figure 2A).
Table 3.
Pre‐/Postprocedural Plasma Corin Levels in Patients Treated With Catheter Ablation for AF
| Total group | ||||
|---|---|---|---|---|
| Total cohort | Nonrecurrence group | Recurrence group | P value* | |
| Total | n=616 | n=463 | n=153 | |
| Before ablation, pg/mL | 411.30 (209.04–552.32) | 369.05 (186.36–489.28) | 539.14 (329.24–702.08) | <0.0001 |
| After ablation, pg/mL | 518.50 (324.50–558.99) | 489.12 (315.66–629.05) | 607.37 (364.50–753.80) | <0.0001 |
| P value† | <0.0001 | <0.0001 | <0.0001 | … |
| AF type subgroup | ||||
| PaAF | n=378 | n=292 | n=86 | |
| Before ablation, pg/mL | 344.72 (191.74–489.66) | 304.06 (165.40–455.94) | 468.05 (302.80–647.67) | <0.0001 |
| After ablation, pg/mL | 442.57 (302.51–599.71) | 423.33 (288.29–572.70) | 527.61 (315.67–698.21) | 0.003 |
| PeAF | n=238 | n=171 | n=67 | |
| Before ablation, pg/mL | 435.91 (261.18–637.29) | 404.68 (235.93–572.66) | 565.07 (347.99–786.97) | <0.0001 |
| After ablation, pg/mL | 527.60 (367.78–728.85) | 508.00 (352.10–690.43) | 565.37 (440.83–855.09) | 0.018 |
| P value‡ (before ablation) | <0.0001 | <0.0001 | 0.024 | … |
| P value§ (after ablation) | <0.0001 | <0.0001 | 0.032 | … |
| Sex subgroup | ||||
| Male | n=364 | n=260 | n=104 | |
| Before ablation, pg/mL | 445.7 (260.2–642.0) | 405.2 (241.0–591.4) | 568.5 (397.9–793.2) | <0.0001 |
| After ablation, pg/mL | 556.3 (409.7–741.6) | 523.3 (372.5–717.8) | 630.9 (485.1–849.8) | 0.001 |
| Female | n=252 | n=203 | n=49 | |
| Before ablation, pg/mL | 302.2 (164.7–436.0) | 281.1 (153.3–409.4) | 353.5 (253.4–521.1) | 0.002 |
| After ablation, pg/mL | 372.3 (266.6–505.5) | 372.6 (261.8–499.5) | 360.7 (294.1–537.8) | 0.354 |
| P value|| (before ablation) | <0.0001 | <0.0001 | <0.0001 | … |
| P value# (after ablation) | <0.0001 | <0.0001 | <0.0001 | … |
Values are provided as median (quartile 1–quartile 3). AF indicates atrial fibrillation; PaAF, paroxysmal atrial fibrillation; and PeAF, persistent atrial fibrillation.
Nonrecurrence group vs recurrence group.
Preablation vs postablation.
Paroxysmal atrial fibrillation vs persistent atrial fibrillation of corin levels before ablation.
Paroxysmal atrial fibrillation vs persistent atrial fibrillation of corin levelsafter ablation.
Male vs female corin levels before ablation.
Male vs female corin levels postablation.
Figure 2. Pre‐ and postprocedural plasma corin levels in patients treated with catheter ablation for atrial fibrillation.
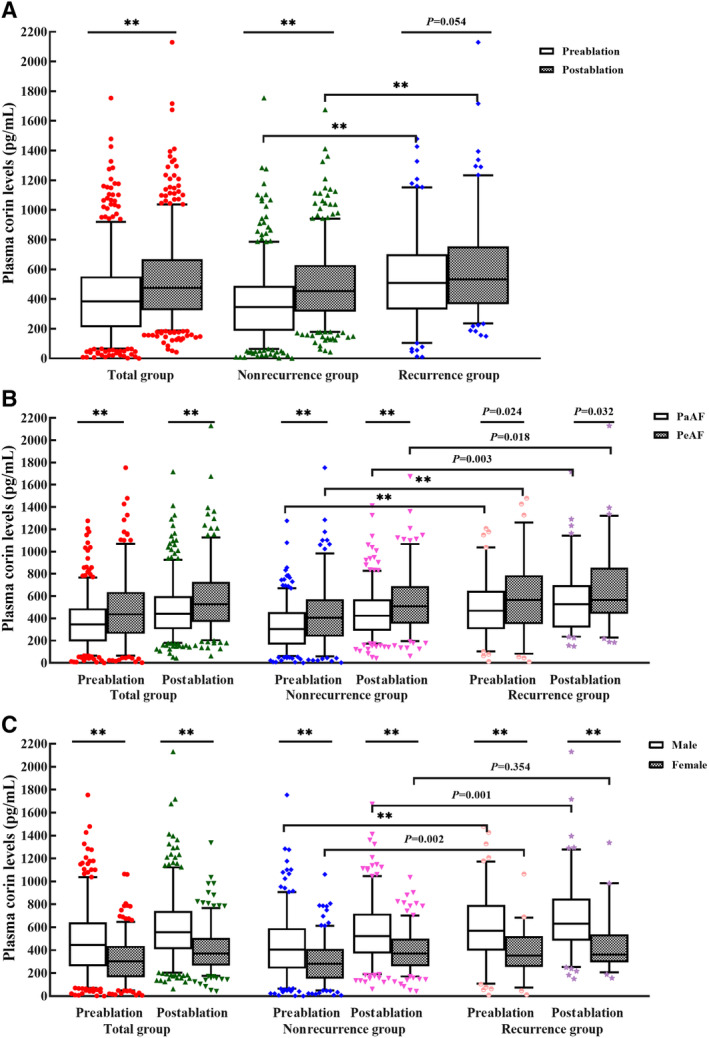
A, Total group; (B) subgroup analysis of atrial fibrillation type; (C) subgroup analysis of sex. The box diagram represents the 5% to 95% range of values. **P<0.0001.
The plasma corin concentrations were higher in patients with PeAF than in patients with PaAF 21 and higher in men than in women. 21 , 28 In a subgroup analysis of AF types, we found the trend of plasma corin levels was similar to that of the general population. Plasma corin concentrations were significantly higher in patients with PeAF than in patients with PaAF, whether measured in peripheral venous blood drawn before or after ablation (before ablation, 435.91 [261.18–637.29] versus 344.72 [191.74–489.66] pg/mL, respectively; P<0.0001; after ablation, 527.60 [367.78–728.85] versus 442.57 [302.51–599.71] pg/mL, respectively; P<0.0001). In the PaAF subgroup, corin concentrations were significantly higher in patients with recurrence than in those without recurrence (before ablation, 468.05 [302.80–647.67] versus 304.06 [165.40–455.94] pg/mL, respectively; P<0.0001; after ablation, 527.61 [315.67–698.21] versus 423.33 [288.29–572.70] pg/mL, respectively; P=0.003). In the PeAF subgroup, corin concentrations were also significantly higher in patients with recurrence than in those without recurrence (before ablation, 565.07 [347.99–786.97] versus 404.68 [235.93–572.66] pg/mL, respectively; P<0.0001; after ablation, 565.37 [440.83–855.09] versus 508.00 [352.10–690.43] pg/mL, respectively; P=0.018; Table 3, Figure 2B).
In a subgroup analysis by sex, plasma corin concentrations were significantly higher in men than in women, whether measured in peripheral venous blood drawn before or after ablation (before ablation, 445.7 [260.2–642.0] versus 302.2 [164.7–436.0] pg/mL, respectively; P<0.0001; after ablation 556.3 [409.7–741.6] versus 372.3 [266.6–505.5] pg/mL, respectively; P<0.0001). In the male subgroup, corin concentrations were significantly higher in patients with recurrence than in those without recurrence (before ablation, 568.5 [397.9–793.2] versus 405.2 [241.0–591.4] pg/mL, respectively; P<0.0001; after ablation, 630.9 [485.1–849.8] versus 523.3 [372.5–717.8] pg/mL, respectively; P=0.001). In the female subgroup, corin concentrations drawn before ablation were also higher in patients with recurrence than in those without recurrence (353.5 [253.4–521.1] versus 281.1 [153.3–409.4] pg/mL, respectively; P=0.002), but this was not the case after ablation (360.7 [294.1–537.8] versus 372.6 [261.8–499.5] pg/mL, respectively; P=0.354; Table 3, Figure 2C).
A significant positive association was found between corin levels before and after ablation, including in the total group (R=0.604, P<0.0001) (Figure 3A), the nonrecurrence group (R=0.568, P<0.0001) (Figure 3B), and the recurrence group (R=0.688, P<0.0001; Figure 3C).
Figure 3. The association between corin levels before and after ablation.
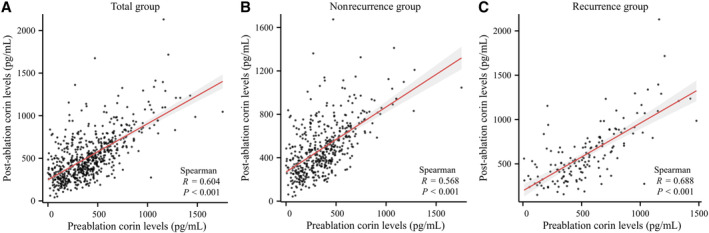
A, Total group; (B) nonrecurrence group; (C) recurrence group. The shaded region indicates the 95% CIs of Spearman r value.
Plasma Corin Threshold of Best Predictive Value for Recurrent AF at 1 Year After CA
The receiver operating characteristic curve analysis of plasma corin drawn before and after ablation identified threshold values of >494.85 pg/mL and >526.57 pg/mL, respectively, as having the best predictive value for recurrent AF at 1 year after CA, with a sensitivity of 52.9%, a specificity of 76.5%, and an area under the curve (AUC) of 0.676 before ablation and a sensitivity of 53.6%, a specificity of 63.9%, and an AUC of 0.610 after ablation (both P<0.0001; Table 4, Figure 4A).
Table 4.
Plasma Corin Threshold of Best Predictive Value for Recurrent AF at 1 Year After Catheter Ablation
| Total | Male | Female | PaAF | PeAF | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Before ablation | After ablation | Before ablation | After ablation | Before ablation | After ablation | Before ablation | After ablation | Before ablation | After ablation | |
| AUC (95% CI) | 0.676 (0.626–0.727) | 0.610 (0.558–0.662) | 0.681 (0.619–0.743) | 0.612 (0.548–0.675) | 0.644 (0.556–0.731) | 0.543 (0.455–0.631) | 0.683 (0.617–0.749) | 0.606 (0.537–0.675) | 0.659 (0.580–0.738) | 0.603 (0.523–0.683) |
| Threshold, pg/mL | 494.85 | 526.57 | 642.56 | 503.43 | 291.62 | 272.80 | 490.375 | 526.573 | 648.655 | 792.172 |
| Sensitivity (%) | 52.9 | 53.6 | 46.2 | 70.2 | 73.5 | 83.7 | 47.7 | 51.2 | 44.8 | 34.3 |
| Specificity (%) | 76.5 | 63.9 | 83.8 | 48.5 | 53.2 | 28.6 | 82.2 | 70.5 | 84.2 | 84.2 |
| P value | <0.0001 | <0.0001 | <0.0001 | 0.0009 | 0.0018 | 0.3539 | <0.0001 | 0.0029 | 0.0001 | 0.0132 |
AF indicates atrial fibrillation; AUC, area under the receiver operating characteristic curve; PaAF, paroxysmal atrial fibrillation; and PeAF, persistent atrial fibrillation.
Figure 4. Plasma corin threshold of best predictive value for recurrent atrial fibrillation at 1 year after catheter ablation.
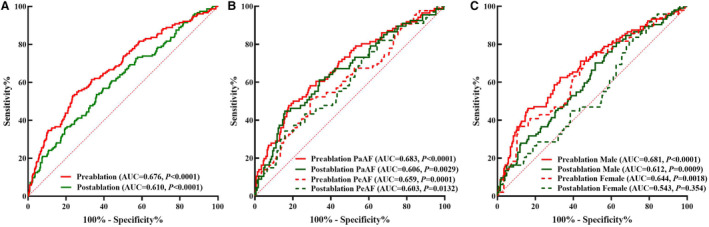
A, Total group; (B) subgroup analysis of atrial fibrillation type; (C) subgroup analysis of sex. AUC indicates area under the curve; PaAF, paroxysmal atrial fibrillation; and PeAF, persistent atrial fibrillation.
We also analyzed plasma corin's best predictive threshold for recurrent AF at 1 year after CA according to AF type and sex. In patients with PaAF, the pre‐ and postablation thresholds were >490.375 pg/mL and >526.573 pg/mL, respectively. The sensitivity was 47.7% and the specificity 82.2% before ablation (P<0.0001), whereas the sensitivity was 51.2% and the specificity 70.5% after ablation (P=0.0029). The AUC was 0.682 before ablation and 0.606 after ablation. In patients with PeAF, the pre‐ and postablation thresholds were >648.655 pg/mL and >792.172 pg/mL, respectively. The sensitivity was 44.8% and the specificity 84.2% before ablation (P=0.0001), whereas the sensitivity was 34.3% and the specificity 84.2% after ablation (P=0.0132). The AUC was 0.659 before ablation and 0.603 after ablation (Table 4, Figure 4B).
In male patients, the pre‐ and postablation thresholds were >642.56 pg/mL and >503.43 pg/mL, respectively. The sensitivity was 46.2% and the specificity 83.8% before ablation (P<0.0001), while the sensitivity was 70.2% and the specificity 48.5% after ablation (P=0.0009). The AUC was 0.681 before ablation and 0.612 after ablation. In female patients, the preablation plasma corin level predicted the recurrence of AF after CA at a threshold of >291.62 pg/mL (P=0.0018). However, the predictive value of corin levels in the recurrence of AF after ablation was not found (P=0.354; Table 4, Figure 4C).
Relationship Between Plasma Corin Levels and AF Recurrence
We classified low and high corin levels according to the thresholds in different groups. The Kaplan–Meier survival curve shows that high corin concentrations measured before the procedure indicated a significantly higher risk of AF recurrence than low corin concentrations, whether in the total group, different types of AF, or different sex (Figure 5A through 5E). The same results were found in the blood after the procedure (Figure 5F through 5I) except in the female group (HR, 1.872 [95% CI, 0.995–3.524]; P=0.0987; Figure 5J).
Figure 5. Kaplan–Meier curves showing recurrent atrial fibrillation at 1 year after catheter ablation for different corin levels.
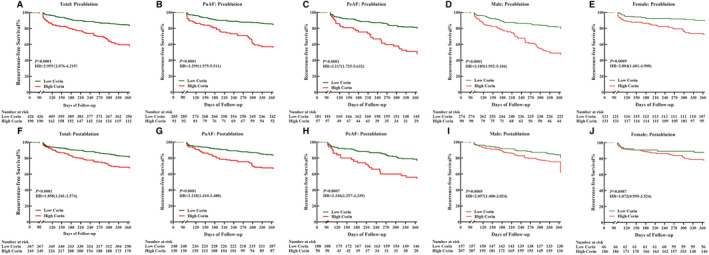
A through E, Corin levels before ablation in total, PaAF, PeAF, male and female groups; F through J, corin levels after ablation in total, PaAF, PeAF, male and female. HR indicates hazard ratio; PaAF, paroxysmal atrial fibrillation; and PeAF, persistent atrial fibrillation.
The Cox regression and forest plot found that high corin levels, whether before or after the procedure, PaAF or PeAF, man or woman, brought high risks of recurrent AF after CA without adjusting for confounding factors (Table 5, Figure 6). In the general population, 1 SD increase in corin concentration before CA increased the risk of recurrent AF by 54.3% after adjusting for age, sex, BMI, triglyceride, HDL‐C, AADs, β‐blockers, and corin levels after CA (HR, 1.465 [95% CI: 1.282–1.655]; P<0.0001; Table 5, Figure 6). The results remained significant in the PaAF (HR, 1.603 [95% CI, 1.314–1.954]; P<0.0001) and PeAF group (HR, 1.521 [95% CI, 1.43–1.86]; P<0.0001), and in both male (HR, 1.497 [95% CI, 1.272–1.762]; P<0.0001) and female patients (HR, 1.428 [95% CI, 1.121–1.82]; P<0.0001) after adjusting for confounding factors. However, corin concentration after CA was not associated with the risk of recurrent AF after adjusting for confounding factors, whether in the total group, in the PaAF or PeAF group, or in the male or female group (Table 5, Figure 6).
Table 5.
Cox Regression Analysis for Corin Concentrations Between Recurrent AF and Nonrecurrent AF After Catheter Ablation
| Corin concentrations (Per SD, pg/mL) | Unadjusted | Adjusted* | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | P value | HR | 95% CI | P value | |
| Total group | ||||||
| Before ablation (264.94) | 1.559 | 1.378–1.763 | <0.0001 | 1.457 | 1.282–1.655 | <0.0001 |
| After ablation (266.39) | 1.382 | 1.215–1.572 | <0.0001 | 1.031 | 0.831–1.28 | 0.78 |
| Male group | ||||||
| Before ablation (289.61) | 1.563 | 1.334–1.83 | <0.0001 | 1.497 | 1.272–1.762 | <0.0001 |
| After ablation (283.52) | 1.359 | 1.156–1.597 | <0.0001 | 1.022 | 0.803–1.300 | 0.861 |
| Female group | ||||||
| Before ablation (187.33) | 1.501 | 1.176–1.915 | 0.001 | 1.428 | 1.121–1.82 | 0.004 |
| After ablation (184.54) | 1.289 | 0.99–1.679 | 0.06 | 0.918 | 0.611–1.379 | 0.658 |
| PaAF group | ||||||
| Before ablation (226.47) | 1.803 | 1.478–2.199 | <0.0001 | 1.603 | 1.314–1.954 | <0.0001 |
| After ablation (239.41) | 1.44 | 1.186–1.748 | <0.0001 | 0.92 | 0.671–1.26 | 0.603 |
| PeAF group | ||||||
| Before ablation (303.66) | 1.493 | 1.222–1.823 | <0.0001 | 1.521 | 1.243–1.86 | <0.0001 |
| After ablation (293.45) | 1.353 | 1.105–1.655 | 0.003 | 1.11 | 0.805–1.531 | 0.525 |
AF indicates atrial fibrillation; HR, hazard ratio; PaAF, paroxysmal atrial fibrillation; and PeAF, persistent atrial fibrillation.
Adjusted for age, sex, body mass index, estimated glomerular filtration rate, triglyceride, high‐density lipoprotein cholesterol, antiarrhythmic drugs, β‐blockers, and corin levels before or after catheter ablation.
Figure 6. Hazard ratios with 95% CIs for 1 SD increase in corin concentration of recurrent atrial fibrillation at 1 year after catheter ablation.
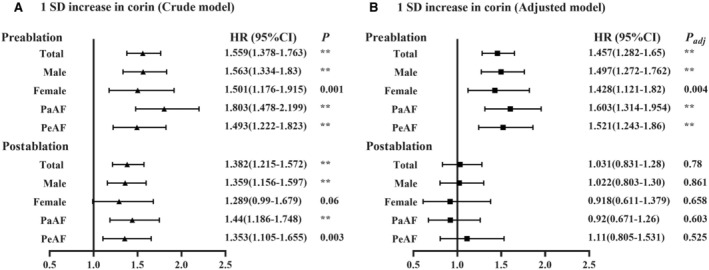
A, Crude model; (B) adjusted model (adjusted for age, sex, body mass index, triglyceride, high‐density lipoprotein cholesterol, antiarrhythmic drugs, β‐blockers and corin levels before or after catheter ablation). HR indicates hazard ratio; PaAF, paroxysmal atrial fibrillation; and PeAF, persistent atrial fibrillation. **P<0.0001.
Independent Predictors of Recurrent AF After CA
In the adjusted models, higher preablation corin levels (odds ratio [OR], 1.796 [95% CI, 1.368–2.358]; P<0.0001) and lower preablation HDL‐C levels (OR, 0.691 [95% CI, 0.544–0.878]; P=0.003) were associated with recurrent AF after ablation in the total group (Figure 7). The same results were found in the male group for higher preablation corin levels (OR, 1.183 [95% CI, 1.307–2.514]; P<0.0001) and lower preablation HDL‐C levels (OR, 0.689 [95% CI, 0.517–0.918; P=0.011). In the female and PeAF groups, only higher preablation corin levels were associated with recurrent AF after ablation (female group: OR, 1.742 [95% CI, 1.105–2.745]; P=0.017; PeAF group: OR, 1.614 [95% CI, 1.073–2.427]; P=0.022). In the PaAF group, in addition to higher preablation corin levels (OR, 2.208 [95% CI, 1.454–3.352]; P<0.0001) and lower preablation HDL‐C levels (OR, 0.65 [95% CI, 0.47–0.898]; P=0.009), oral AADs were associated with a high risk of recurrent AF after ablation (OR, 1.936 [95% CI, 1.024–3.66]; P=0.042). Table S1 shows the independent predictors of recurrent AF after CA in the crude models.
Figure 7. Predictors of recurrent atrial fibrillation at 1 year after catheter ablation in adjusted models.

Adjusted for age, sex, body mass index, triglyceride, high‐density lipoprotein cholesterol, antiarrhythmic drugs, β‐blockers, and corin levels before or after catheter ablation. The continuous variables were log‐transformed and 1 SD was used for odds ratio calculation. AADs indicates antiarrhythmic drugs; AF, atrial fibrillation; BMI, body mass index; HDL‐C, high‐density lipoprotein cholesterol; OR, odds ratio; PaAF, paroxysmal atrial fibrillation; PeAF, persistent atrial fibrillation; and TG, triglyceride. **P<0.0001.
Discussion
To the best of our knowledge, this is the first prospective, single‐center study to investigate the impact of plasma corin levels before and after CA for AF on the risk of recurrence of AF at 1 year after the procedure. Preablation levels of corin were higher in patients with recurrent AF at 1 year, whether in the general population, male or female group, or PaAF or PeAF group. A multivariate Cox regression showed that a 1 SD increase (264.94 pg/mL) in corin concentrations before CA increased the risk of recurrent AF by 54.3%. A higher level of circulating corin at baseline could be a predictor of the risk of recurrent of AF after CA. These findings suggest that corin may play a considerable role in AF and may therefore serve as a candidate factor to predict recurrent AF after CA. It may also be a potential therapeutic target for AF.
Soluble corin represents a potential biomarker for risk prediction and prognostic assessment of CVDs. Currently, some studies have demonstrated an association between soluble corin levels and CVDs. A higher serum corin at baseline predicted a higher risk of CVD events and stroke, but not CAD. 29 A prospective study of a cohort of patients with chronic HF found that those with low soluble corin tended to have a better outcome. 18 The results were consistent with the present study, in which high corin concentrations predicted adverse clinical outcomes. However, a report of an HF population and a report of an acute MI population, both from China, found that those with high corin concentrations had a lower incidence of major adverse cardiac events and all‐cause death. 19 , 30 This discrepancy may be related to the acute and chronic state of HF or CAD, the use of renin–angiotensin–aldosterone system inhibitors, and different ethnic groups. Regarding AF, our previous and present studies found that plasma corin concentrations in patients with PeAF were higher than in patients with PaAF. 21 An increase in the expression of A disintegrin and metalloprotease 10 (ADAM10) was reported in human atria during AF, 31 and ADAM10 was primarily responsible for shedding corin in its juxtamembrane region. 14 This suggests that more corin shedding, particularly ADAM10‐mediated shedding, occurs in the atrial tissue with increased fibrosis and remodeling. In the cultured medium from transfected human embryonic kidney 293 cells and mouse atrial HL‐1 cardiomyocytes, 3 distinct corin fragments of ≈100, ≈160, and ≈180 kDa were detected. 14 The majority of the observed activity in processing natriuretic peptides can be attributed to the ≈180‐kDa soluble corin fragments, whereas the other 2 fragments have little biological activity. 14 We did not test plasma corin activity or the kind of fragments in the present study.
The relationship between corin activity in the circulatory system and AF recurrence after CA is unknown. Corin plays a central role in the processing of pro‐ANP. ANP released from cardiomyocytes inhibits collagen synthesis as a paracrine factor. 32 ANP/cGMP/protein kinase G signaling disrupts the transforming growth factor‐β1–induced nuclear translocation of phosphorylated Smad3 and downstream events, including myofibroblast transformation, proliferation, and expression of extracellular matrix molecules in cardiac fibroblasts. 33 ANP also plays a key role in cardiac electrophysiology, modulating the autonomic nervous system and regulating the function of cardiac ion channels. 34 The ultimate electrophysiological effect of ANP is to shorten the action potential duration and reduce calcium influx into the cell through L‐type calcium current. Furthermore, cGMP facilitates the sarco/endoplasmic reticulum Ca2+ ATPase uptake of calcium into the sarcoplasmic reticulum. 35 Therefore, we speculate that corin may also suppress atrial fibrosis and has a similar electrophysiological effect to that of ANP in light of discoveries connecting corin and vulnerability to AF. A statistically significant association has been found between baseline ANP level and postablation AF recurrence. 36 In the future, preablation detection of corin and ANP may help physicians optimize patient selection, improve treatment strategies, and provide novel targets for pharmacological intervention against AF recurrence. Validation in AF cohorts undergoing ablation is warranted. The association of 1‐day postablation level of corin with AF recurrence after CA was not detected in the present study. This may be explained by ablation's interventional effect on corin shedding from atrial myocytes. Thermal procedures may lead to ischemia, coagulation necrosis, edema, and local inflammation of atrial tissue, 37 which may result in the increased shedding of corin from atrial myocytes. More studies are warranted. The dynamic change of corin level after ablation, for example, at the first, third, or sixth month, may be related to AF recurrence. Therefore, dynamically monitoring corin in the circulation is necessary to better understand the role of corin in AF.
Most studies have demonstrated a sex difference in circulating corin similar to that in our study, with a level higher in men than in women whether in a state of health or disease. 21 , 26 , 28 , 38 In our study, men's plasma corin levels were higher than women's, and the association between plasma corin and recurrent AF after CA was stronger in men than in women. Similar associations have also been found in metabolic syndrome 38 and stroke, 39 but the mechanisms underlying the sex‐specific contribution of corin to AF recurrence and other CVDs remain unclear. Some studies indicate that sex seems to be involved in a different response to the AF ablation procedure, including a higher risk of AF recurrence rate and increased periprocedural complications and hospitalization in women than in men. 40 , 41 , 42 , 43 There are several possible explanations. First, female patients have a greater symptom burden. 44 , 45 , 46 Second, population‐based studies have demonstrated that, on average, new‐onset AF starts 5 years later in women than in men, 47 so female patients are older than male patients. 48 , 49 Third, female patients show more advanced atrial remodeling on high‐density electroanatomic mapping than male patients. 41 The rates of AF recurrence after CA were higher in men than in women in our study (28.57% versus 19.44%, respectively; P=0.01), but female sex was not an independent risk factor for AF recurrence after adjusting for confounders. Corin levels may be an important mechanism with regard to the sex difference in AF recurrence after ablation. The sex‐specific difference in plasma corin levels may be 1 mechanism underlying the different rates of AF recurrence between men and women. Before the integration of corin assessment in daily clinical practice, it should be noted that the method of soluble corin assessment needs to be improved and standardized according to sex. 17 Some large and prospective studies are warranted to measure the levels of soluble corin in the healthy or morbid population. Most importantly, other characteristics need to be well matched in men and women.
The AF recurrence rate after CA in our study was 24.84%, which was lower than that reported in most studies. 3 , 50 , 51 This may be related to several factors. First, the methods of assessing AF recurrence may have led to an underestimation of actual recurrence rates. The patients in the study did not receive an implantable loop recorder. The assessments of recurrence relied only on symptoms and intermittent ECGs, which may be the main reason for a potential underestimation of the recurrence rate. However, 1 randomized controlled trial found a 1‐year recurrence of symptomatic atrial tachyarrhythmia (defined by continuous rhythm monitoring) of 20.9% in PaAF after radiofrequency CA, 50 which was even lower than in the present study. This indicates that the recurrence rate in the present study reflects reality, which may be explained by the fact that all the patients were symptomatic, making the detection of recurrence easier. Second, some of the patients in our study were not refractory to AADs yet had a strong desire for CA. Therefore, they may have responded well to CA and had a lower recurrence rate. Third, the proportion of postoperative oral AADs in this study was 68.99%, which was higher than the 59.8% in the FIRE AND ICE (Comparative Study of Two Ablation Procedures in Patients With Atrial Fibrillation) trial. 3 Additionally, the mean BMI in this study was significantly lower than in other studies (25.53 kg/m2 versus 27.8, 31.5, and 30 kg/m2). 3 , 51 , 52 The conclusion has been validated that a high BMI is associated with a higher risk of AF recurrence in patients undergoing CA. In one meta‐analysis, the highest BMI group had a higher AF recurrence (OR, 1.37 [95% CI, 1.18–1.58]; P<0.001). The dose–response relationship for BMI and AF recurrence was nonlinear, and the curve became steeper at 30 to 35 kg/m2. 53 Fourth, our center is experienced in the ablation of AF, and, in addition to routine PVI, we mapped and ablated as far as possible all the ectopic sites of AF, including the left atrium, inferior vena cava, and so on. Fifth, the follow‐up time in the study was 1 year, which was shorter than in other studies. 3 , 50
Our study has some limitations. First, it was a single‐center, observational study and was subject to the inherent limitations associated with residual confoundings. However, our prospective design confirmed the chronological order in which elevated corin preceded the recurrence of AF after ablation, thereby increasing the likelihood of causality. Second, the actual AF recurrence rates may be underestimated in the present study as described in the discussion section. In addition, if underestimation occurred, it likely affected both the recurrence and nonrecurrence groups equivalently. We therefore believe that the monitoring modalities likely had little effect on the results. Third, some clinical studies have found that elevated ANP, or midregional N‐terminal pro‐ANP, type B natriuretic peptide, and N‐terminal pro‐BNP levels were significantly associated with the risk factor of AF recurrence after ablation. 54 , 55 , 56 , 57 , 58 We did not discuss the association between those factors and recurrent AF after ablation. Fourth, plasma corin levels are correlated with immediate heart rhythm, as confirmed by higher concentrations in AF than in sinus rhythm. 21 We found that the corin levels 1 day after ablation were higher than at baseline. However, we did not dynamically monitor the changes in corin level after ablation, for example, at the first, third, sixth, ninth, or 12th month after ablation. It is unclear when corin levels decrease and to what extent the decrease in corin levels after ablation is associated with AF recurrence. This needs to be confirmed in future trials. Finally, we acknowledge that this study is the first to demonstrate an independent association of corin levels with outcome and should be considered as a hypothesis‐generating study. Validation in other AF cohorts undergoing ablation is warranted.
Conclusions
Our study demonstrates that baseline plasma corin level is a prognostic biomarker of AF recurrence after ablation, independent of established conventional risk factors. Although more studies are warranted, high corin levels may have important implications for the interpretation and design of studies in search of optimal methods to enhance response well to CA for AF. Risk stratification before ablation for AF may be useful in selecting treatment regimens for patients.
Sources of Funding
This work was supported by the National Natural Science Foundation of China (grant number 81700301), Science and Technology Project of Education Department of Liaoning Province (grant number LZ2020058) and Scientific Research Project of Dalian Medical Key Specialty “Climbing Peak Plan” (grant number 2022DF016).
Disclosures
None.
Supporting information
Table S1.
Acknowledgments
The authors gratefully acknowledge the statistical support by Dr Yongkui Ren, also from The First Affiliated Hospital of Dalian Medical University.
Y. Zhao, X. Yuan and Y. Xie contributed equally and are co‐first authors.
Supplemental Material is available at https://www.ahajournals.org/doi/suppl/10.1161/JAHA.123.031928
For Sources of Funding and Disclosures, see page 14.
Contributor Information
Jinqiu Liu, Email: 18098875755@163.com.
Feifei Chen, Email: sdaqchenfei@126.com.
References
- 1. Zhou Z, Hu D. An epidemiological study on the prevalence of atrial fibrillation in the Chinese population of mainland China. J Epidemiol. 2008;18:209–216. doi: 10.2188/jea.JE2008021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Zhang S. Atrial fibrillation in mainland China: epidemiology and current management. Heart. 2009;95:1052–1055. doi: 10.1136/hrt.2008.146589 [DOI] [PubMed] [Google Scholar]
- 3. Kuck KH, Brugada J, Fürnkranz A, Metzner A, Ouyang F, Chun KRJ, Elvan A, Arentz T, Bestehorn K, Pocock SJ, et al. Cryoballoon or radiofrequency ablation for paroxysmal atrial fibrillation. N Engl J Med. 2016;374:2235–2245. doi: 10.1056/NEJMoa1602014 [DOI] [PubMed] [Google Scholar]
- 4. Mont L, Bisbal F, Hernández‐Madrid A, Pérez‐Castellano N, Viñolas X, Arenal A, Arribas F, Fernández‐Lozano I, Bodegas A, Cobos A, et al. Catheter ablation vs. antiarrhythmic drug treatment of persistent atrial fibrillation: a multicentre, randomized, controlled trial (SARA study). Eur Heart J. 2014;35:501–507. doi: 10.1093/eurheartj/eht457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Chen H, Li C, Han B, Xiao F, Yi F, Wei Y, Jiang C, Zou C, Shi L, Ma W, et al. Circumferential pulmonary vein isolation with vs without additional low‐voltage‐area ablation in older patients with paroxysmal atrial fibrillation: a randomized clinical trial. JAMA Cardiol. 2023;8:765–772. doi: 10.1001/jamacardio.2023.1749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Wu G, Huang H, Cai L, Yang Y, Liu X, Yu B, Tang Y, Jiang H, Huang C; CAPA Study Investigators . Long‐term observation of catheter ablation vs. pharmacotherapy in the management of persistent and long‐standing persistent atrial fibrillation (CAPA study). Europace. 2021;23:731–739. doi: 10.1093/europace/euaa356 [DOI] [PubMed] [Google Scholar]
- 7. Osorio J, Hunter TD, Rajendra A, Zei P, Silverstein J, Morales G. Predictors of clinical success after paroxysmal atrial fibrillation catheter ablation. J Cardiovasc Electrophysiol. 2021;32:1814–1821. doi: 10.1111/jce.15028 [DOI] [PubMed] [Google Scholar]
- 8. Liżewska‐Springer A, Dąbrowska‐Kugacka A, Lewicka E, Drelich Ł, Królak T, Raczak G. Echocardiographic predictors of atrial fibrillation recurrence after catheter ablation: a literature review. Cardiol J. 2020;27:848–856. doi: 10.5603/CJ.a2018.0067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Tsiachris D, Giannopoulos G, Deftereos S, Kossyvakis C, Tsioufis C, Siasos G, Oikonomou E, Gatzoulis K, Tousoulis D, Stefanadis C. Biomarkers determining prognosis of atrial fibrillation ablation. Curr Med Chem. 2019;26:925–937. doi: 10.2174/0929867325666180320122930 [DOI] [PubMed] [Google Scholar]
- 10. Ichiki T, Huntley BK, Heublein DM, Sandberg SM, McKie PM, Martin FL, Jougasaki M, Burnett JC Jr. Corin is present in the normal human heart, kidney, and blood, with pro‐B‐type natriuretic peptide processing in the circulation. Clin Chem. 2011;57:40–47. doi: 10.1373/clinchem.2010.153908 [DOI] [PubMed] [Google Scholar]
- 11. Yan W, Sheng N, Seto M, Morser J, Wu Q. Corin, a mosaic transmembrane serine protease encoded by a novel cDNA from human heart. J Biol Chem. 1999;274:14926–14935. doi: 10.1074/jbc.274.21.14926 [DOI] [PubMed] [Google Scholar]
- 12. Potter LR, Abbey HS, Dickey DM. Natriuretic peptides, their receptors, and cyclic guanosine monophosphate dependent signaling functions. Endocr Rev. 2006;27:47–72. doi: 10.1210/er.2005-0014 [DOI] [PubMed] [Google Scholar]
- 13. Yan W, Wu F, Morser J, Wu Q. Corin, a transmembrane cardiac serine protease, acts as a pro‐atrial natriuretic peptide‐converting enzyme. Proc Natl Acad Sci USA. 2000;97:8525–8529. doi: 10.1073/pnas.150149097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Jiang J, Wu S, Wang W, Chen S, Peng J, Zhang X, Wu Q. Ectodomain shedding and autocleavage of the cardiac membrane protease corin. J Biol Chem. 2011;286:10066–10072. doi: 10.1074/jbc.M110.185082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Dong N, Chen S, Wang W, Zhou Y, Wu Q. Corin in clinical laboratory diagnostics. Clin Chim Acta. 2012;413:378–383. doi: 10.1016/j.cca.2011.10.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Knappe S, Wu F, Masikat MR, Morser J, Wu Q. Functional analysis of the transmembrane domain and activation cleavage of human corin: design and characterization of a soluble corin. J Biol Chem. 2003;278:52363–52370. doi: 10.1074/jbc.M309991200 [DOI] [PubMed] [Google Scholar]
- 17. Rong Y, Han X, Zhang X, Wang Y, Wang T. Circulating soluble corin as a potential biomarker for cardiovascular diseases: a translational review. Clin Chim Acta. 2018;485:106–112. doi: 10.1016/j.cca.2018.06.036 [DOI] [PubMed] [Google Scholar]
- 18. Gommans DHF, Revuelta‐Lopez E, Lupon J, Cserkóová A, Domingo M, Vart P, van Royen N, Bayés‐Genis A, van Kimmenade RRJ. Soluble neprilysin and corin concentrations in relation to clinical outcome in chronic heart failure. JACC Heart Fail. 2021;9:85–95. doi: 10.1016/j.jchf.2020.08.015 [DOI] [PubMed] [Google Scholar]
- 19. Zhou X, Chen JC, Liu Y, Yang H, Du K, Kong Y, Xu XH. Plasma corin as a predictor of cardiovascular events in patients with chronic heart failure. JACC Heart Fail. 2016;4:664–669. doi: 10.1016/j.jchf.2016.03.006 [DOI] [PubMed] [Google Scholar]
- 20. Chen L, Zhang Q, Zhang M, Yu J, Ren L, Li J, Ma S, He Y, Hu W, Peng H. Soluble corin predicts the risk of cardiovascular disease: a 10‐year follow‐up study. JACC Asia. 2022;2:490–501. doi: 10.1016/j.jacasi.2022.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Chen F, Xia Y, Liu Y, Zhang Y, Song W, Zhong Y, Gao L, Jin Y, Li S, Jiang Y, et al. Increased plasma corin levels in patients with atrial fibrillation. Clin Chim Acta. 2015;447:79–85. doi: 10.1016/j.cca.2015.05.017 [DOI] [PubMed] [Google Scholar]
- 22. Zhao Y, Yuan X, Zhong Y, Zhang Y, Zhang S, Li S, Zhao Y, Zheng W, Liu J, Xia Y, et al. Single‐nucleotide polymorphisms in the 3′ untranslated region of CORIN associated with cardiovascular diseases in a Chinese Han population: a case‐control study. Front Cardiovasc Med. 2021;8:625072–625085. doi: 10.3389/fcvm.2021.625072 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomström‐Lundqvist C, Boriani G, Castella M, Dan GA, Dilaveris PE, et al. 2020 ESC guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio‐Thoracic Surgery (EACTS): the task force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J. 2021;42:373–498. doi: 10.1093/eurheartj/ehaa612 [DOI] [PubMed] [Google Scholar]
- 24. American Diabetes Association . Classification and diagnosis of diabetes: standards of medical care in diabetes‐2020. Diabetes Care. 2020;43:S14–S31. doi: 10.2337/dc20-S002 [DOI] [PubMed] [Google Scholar]
- 25. Ouyang F, Bänsch D, Ernst S, Schaumann A, Hachiya H, Chen M, Chun J, Falk P, Khanedani A, Antz M, et al. Complete isolation of left atrium surrounding the pulmonary veins: new insights from the double‐lasso technique in paroxysmal atrial fibrillation. Circulation. 2004;110:2090–2096. doi: 10.1161/01.CIR.0000144459.37455.EE [DOI] [PubMed] [Google Scholar]
- 26. Peng H, Zhang Q, Cai XQ, Liu Y, Ding JS, Tian HG, Chao XQ, Shen HS, Jiang L, Jin JH, et al. Association between high serum soluble corin and hypertension: a cross‐sectional study in a general population of China. Am J Hypertens. 2015;28:1141–1149. doi: 10.1093/ajh/hpv002 [DOI] [PubMed] [Google Scholar]
- 27. Zhang H, Mo XB, Zhou ZY, Zhu ZB, HuangFu XF, Xu T, Wang AL, Guo ZR, Zhang YH. Associations among NPPA gene polymorphisms, serum ANP levels, and hypertension in the Chinese Han population. J Hum Hypertens. 2019;3:641–647. doi: 10.1038/s41371-019-0219-6 [DOI] [PubMed] [Google Scholar]
- 28. Dong N, Dong J, Liu P, Xu L, Shi S, Wu Q. Effects of anticoagulants on human plasma soluble corin levels measured by ELISA. Clin Chim Acta. 2010;411:1998–2003. doi: 10.1016/j.cca.2010.08.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Chen L, Zhang Q, Zhang M, Yu J, Ren L, Li J, Ma S, He Y, Hu W, Peng H. Soluble corin predicts the risk of cardiovascular disease: a 10‐year follow‐up study. JACC Asia. 2022;2:490–501. doi: 10.1016/j.jacasi.2022.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Zhou X, Chen J, Zhang Q, Shao J, Du K, Xu X, Kong Y. Prognostic value of plasma soluble corin in patients with acute myocardial infarction. J Am Coll Cardiol. 2016;67:2008–2014. doi: 10.1016/j.jacc.2016.02.035 [DOI] [PubMed] [Google Scholar]
- 31. Arndt M, Lendeckel U, Röcken C, Nepple K, Wolke C, Spiess A, Huth C, Ansorge S, Klein HU, Goette A. Altered expression of ADAMs (a disintegrin and metalloproteinase) in fibrillating human atria. Circulation. 2002;105:720–725. doi: 10.1161/hc0602.103639 [DOI] [PubMed] [Google Scholar]
- 32. Maki T, Horio T, Yoshihara F, Suga S, Takeo S, Matsuo H, Kangawa K. Effect of neutral endopeptidase inhibitor on endogenous atrial natriuretic peptide as a paracrine factor in cultured cardiac fibroblasts. Br J Pharmacol. 2000;131:1204–1210. doi: 10.1038/sj.bjp.0703679 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Li P, Wang D, Lucas J, Oparil S, Xing D, Cao X, Novak L, Renfrow MB, Chen YF. Atrial natriuretic peptide inhibits transforming growth factor beta‐induced Smad signaling and myofibroblast transformation in mouse cardiac fibroblasts. Circ Res. 2008;102:185–192. doi: 10.1161/CIRCRESAHA.107.157677 [DOI] [PubMed] [Google Scholar]
- 34. Perrin MJ, Gollob MH. The role of atrial natriuretic peptide in modulating cardiac electrophysiology. Heart Rhythm. 2012;9:610–615. doi: 10.1016/j.hrthm.2011.11.019 [DOI] [PubMed] [Google Scholar]
- 35. Zhang Q, Yan L, Weiss HR, Scholz PM. Cyclic GMP‐induced reduction in cardiac myocyte function is partially mediated by activation of the sarcoplasmic reticulum Ca(2+)‐ATPase. Pharmacology. 2002;64:106–112. doi: 10.1159/000056158 [DOI] [PubMed] [Google Scholar]
- 36. Jiang H, Wang W, Wang C, Xie X, Hou Y. Association of pre‐ablation level of potential blood markers with atrial fibrillation recurrence after catheter ablation: a meta‐analysis. Europace. 2017;19:392–400. doi: 10.1093/europace/euw088 [DOI] [PubMed] [Google Scholar]
- 37. Gottlieb LA, Dekker LRC, Coronel R. The blinding period following ablation therapy for atrial fibrillation. JACC Clin Electrophysiol. 2021;7:416–430. doi: 10.1016/j.jacep.2021.01.011 [DOI] [PubMed] [Google Scholar]
- 38. Li H, Zhang Q, He Y, Shi J, Hu W, Peng H. Sex‐specific association between soluble corin and metabolic syndrome in Chinese adults. Hypertens Res. 2019;42:1029–1035. doi: 10.1038/s41440-019-0228-4 [DOI] [PubMed] [Google Scholar]
- 39. Peng H, Zhu F, Shi J, Han X, Zhou D, Liu Y, Zhi ZW, Zhang FD, Shen Y, Ma JJ, et al. Serum soluble corin is decreased in stroke. Stroke. 2015;46:1758–1763. doi: 10.1161/STROKEAHA.114.008368 [DOI] [PubMed] [Google Scholar]
- 40. Hermida A, Burtin J, Kubala M, Fay F, Lallemand PM, Buiciuc O, Lieu A, Zaitouni M, Beyls C, Hermida JS. Sex differences in the outcomes of cryoablation for atrial fibrillation. Front Cardiovasc Med. 2022;9:893553–893564. doi: 10.3389/fcvm.2022.893553 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Wong GR, Nalliah CJ, Lee G, Voskoboinik A, Chieng D, Prabhu S, Parameswaran R, Sugumar H, Al‐Kaisey A, McLellan A, et al. Sex‐related differences in atrial remodeling in patients with atrial fibrillation: relationship to ablation outcomes. Circ Arrhythm Electrophysiol. 2022;15:e9925–e9937. doi: 10.1161/CIRCEP.121.009925 [DOI] [PubMed] [Google Scholar]
- 42. Li H, Wang Z, Cheng Z, Zhu Y, Yuan Z, Gao J, Zhang X, Wu Y. Sex differences involved in persistent atrial fibrillation recurrence after radiofrequency ablation. BMC Cardiovasc Disord. 2022;22:549–558. doi: 10.1186/s12872-022-03002-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Sugumar H, Nanayakkara S, Chieng D, Wong GR, Parameswaran R, Anderson RD, Al‐Kaisey A, Nalliah CJ, Azzopardi S, Prabhu S, et al. Arrhythmia recurrence is more common in females undergoing multiple catheter ablation procedures for persistent atrial fibrillation: time to close the gender gap. Heart Rhythm. 2020;17:692–698. doi: 10.1016/j.hrthm.2019.12.013 [DOI] [PubMed] [Google Scholar]
- 44. Grecu M, Blomström‐Lundqvist C, Kautzner J, Laroche C, Van Gelder IC, Jordaens L, Tavazzi L, Cihak R, Campal JM, Kalarus Z, et al. In‐hospital and 12‐month follow‐up outcome from the ESCEORP EHRA atrial fibrillation ablation long‐term registry: sex differences. EP Europace. 2020;22:66–73. doi: 10.1093/europace/euz225 [DOI] [PubMed] [Google Scholar]
- 45. Blum S, Muff C, Aeschbacher S, Ammann P, Erne P, Moschovitis G, Di Valentino M, Shah D, Schläpfer J, Fischer A, et al. Prospective assessment of sex‐related differences in symptom status and health perception among patients with atrial fibrillation. J Am Heart Assoc. 2017;6:e005401–e005415. doi: 10.1161/JAHA.116.005401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Kloosterman M, Crijns HJGM, Mulder BA, Groenveld HF, Van Veldhuisen DJ, Rienstra M, Van Gelder IC. Sex‐related differences in risk factors, outcome, and quality of life in patients with permanent atrial fibrillation: results from the RACE II study. Europace. 2019;12:1619–1627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Benjamin EJ, Levy D, Vaziri SM, D'Agostino RB, Belanger AJ, Wolf PA. Independent risk factors for atrial fibrillation in a populationbased cohort: the Framingham heart study. JAMA. 1994;271:840–844. doi: 10.1001/jama.1994.03510350050036 [DOI] [PubMed] [Google Scholar]
- 48. Russo AM, Zeitler EP, Giczewska A, Silverstein AP, Al‐Khalidi HR, Cha YM, Monahan KH, Bahnson TD, Mark DB, Packer DL, et al. Association between sex and treatment outcomes of atrial fibrillation ablation vs drug therapy. Circulation. 2021;143:661–672. doi: 10.1161/CIRCULATIONAHA.120.051558 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Singh SM, D'Avila A, Aryana A, Kim YH, Mangrum JM, Michaud GF, Dukkipati SR, Heist EK, Barrett CD, Thorpe KE, et al. Persistent atrial fibrillation ablation in females: insight from the MAGIC‐AF trial: gender and persistent AF. J Cardiovasc Electrophysiol. 2016;27:1259–1263. doi: 10.1111/jce.13051 [DOI] [PubMed] [Google Scholar]
- 50. Andrade JG, Champagne J, Dubuc M, Deyell MW, Verma A, Macle L, Leong‐Sit P, Novak P, Badra‐Verdu M, Sapp J, et al. Cryoballoon or radiofrequency ablation for atrial fibrillation assessed by continuous monitoring: a randomized clinical trial. Circulation. 2019;140:1779–1788. doi: 10.1161/CIRCULATIONAHA.119.042622 [DOI] [PubMed] [Google Scholar]
- 51. Packer DL, Mark DB, Robb RA, Monahan KH, Bahnson TD, Poole JE, Noseworthy PA, Rosenberg YD, Jeffries N, Mitchell LB, et al. Effect of catheter ablation vs antiarrhythmic drug therapy on mortality, stroke, bleeding, and cardiac arrest among patients with atrial fibrillation: the CABANA randomized clinical trial. JAMA. 2019;321:1261–1274. doi: 10.1001/jama.2019.0693 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Liang JJ, Elafros MA, Chik WW, Santangeli P, Zado ES, Frankel DS, Supple GE, Schaller RD, Lin D, Hutchinson MD, et al. Early recurrence of atrial arrhythmias following pulmonary vein antral isolation: timing and frequency of early recurrences predicts long‐term ablation success. Heart Rhythm. 2015;12:2461–2468. doi: 10.1016/j.hrthm.2015.07.015 [DOI] [PubMed] [Google Scholar]
- 53. Pranata R, Henrina J, Yonas E, Putra ICS, Cahyadi I, Lim MA, Munawar DA, Munawar M. BMI and atrial fibrillation recurrence post catheter ablation: a dose‐response meta‐analysis. Eur J Clin Investig. 2021;51:e13499–e13506. doi: 10.1111/eci.13499 [DOI] [PubMed] [Google Scholar]
- 54. Yamada T, Murakami Y, Okada T, Okamoto M, Shimizu T, Toyama J, Yoshida Y, Tsuboi N, Ito T, Muto M, et al. Plasma atrial natriuretic peptide and brain natriuretic peptide levels after radiofrequency catheter ablation of atrial fibrillation. Am J Cardiol. 2006;97:1741–1744. doi: 10.1016/j.amjcard.2005.12.071 [DOI] [PubMed] [Google Scholar]
- 55. Kurosaki K, Tada H, Hashimoto T, Ito S, Miyaji K, Naito S, Oshima S, Taniguchi K. Plasma natriuretic peptide concentrations as a predictor for successful catheter ablation in patients with drug‐refractory atrial fibrillation. Circ J. 2007;71:313–320. doi: 10.1253/circj.71.313 [DOI] [PubMed] [Google Scholar]
- 56. Baba M, Yoshida K, Naruse Y, Hattori A, Yui Y, Kimata A, Ito Y, Tsumagari Y, Tsuneoka H, Shinoda Y, et al. Predictors of recurrence after catheter ablation of paroxysmal atrial fibrillation in different follow‐up periods. Medicina (Kaunas). 2020;56:465–472. doi: 10.3390/medicina56090465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Fan JQ, Cao H, Su L, Ling ZY, Liu ZZ, Lan XB, Xu YP, Chen WJ, Yin YH. NT‐proBNP, but not ANP and C‐reactive protein, is predictive of paroxysmal atrial fibrillation in patients undergoing pulmonary vein isolation. J Interv Card Electrophysiol. 2012;33:93–100. doi: 10.1007/s10840-011-9606-9 [DOI] [PubMed] [Google Scholar]
- 58. Badoz M, Serzian G, Favoulet B, Sellal JM, De Chillou C, Hammache N, Laurent G, Mebazaa A, Ecarnot F, Bardonnet K, et al. Impact of midregional N‐terminal pro‐atrial natriuretic peptide and soluble suppression of tumorigenicity 2 levels on heart rhythm in patients treated with catheter ablation for atrial fibrillation: the Biorhythm study. J Am Heart Assoc. 2021;10:e020917–e020923. doi: 10.1161/JAHA.121.020917 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1.


