Abstract
Introduction:
People living with HIV are considered at higher risk of developing severe forms of tuberculosis (TB) disease. Providing HIV testing to TB-exposed people is therefore critical. We present the results of integrating HIV testing into a community-based intervention for household TB contact management in Cameroon and Uganda.
Methods:
Trained community health workers visited the households of index patients with TB identified in 3 urban/semiurban and 6 rural districts or subdistricts as part of a cluster-randomized trial and provided TB screening to all household contacts. Voluntary HIV counseling and testing were offered to contacts aged 5 years or older with unknown HIV status. We describe the cascade of care for HIV testing and the factors associated with the acceptance of HIV testing.
Results:
Overall, 1983 household contacts aged 5 years or older were screened for TB. Of these contacts, 1652 (83.3%) did not know their HIV status, 1457 (88.2%) accepted HIV testing, and 1439 (98.8%) received testing. HIV testing acceptance was lower among adults than children [adjusted odds ratio (aOR) = 0.35, 95% confidence interval (CI): 0.22 to 0.55], those living in household of an HIV-positive vs HIV-negative index case (aOR = 0.56, 95% CI: 0.38 to 0.83), and contacts requiring a reassessment visit after the initial TB screening visit vs asymptomatic contacts (aOR = 0.20, 95% CI: 0.06 to 0.67) and was higher if living in Uganda vs Cameroon (aOR = 4.54, 95% CI: 1.17 to 17.62) or if another contact of the same index case was tested for HIV (aOR = 9.22, 95% CI: 5.25 to 16.18).
Conclusion:
HIV testing can be integrated into community-based household TB contact screening and is well-accepted.
Key Words: HIV, tuberculosis, integration, community, contact tracing, community health workers
INTRODUCTION
Despite major progress in the access to antiretroviral therapy (ART), a considerable number of people living with HIV (PLHIV) are diagnosed with tuberculosis (TB), and TB remains the most frequent cause of death worldwide among PLHIV.1 According to the 2022 United Nations Programme on HIV/AIDS Global AIDS update, about 38.4 million people were living with HIV in 2021 worldwide.1 The 2022 World Health Organization (WHO) Global TB report reported an estimated 10.6 million persons who developed TB the same year, of whom 6.7% were living with HIV, the majority in sub-Saharan Africa.2 In addition, an estimated 214,000 TB-related deaths occurred among PLHIV in 2020.1
PLHIV have an increased risk of developing TB disease after exposure, especially severe forms of TB.3–5 WHO recommends systematic TB symptom screening for PLHIV and provision of TB preventive treatment (TPT) in TB-endemic areas.6,7 TPT substantially reduces the risk of TB infection progressing to TB disease, especially for those with recent TB exposure.8,9 Hence, providing HIV testing to TB-exposed people is therefore an essential component of the TB contact investigation cascade, especially in high HIV burden settings because it might help identify people unaware of their HIV infection among those exposed to TB.
WHO also recommends using differentiated HIV testing service (HTS) delivery approaches to improve the identification of hard-to-reach PLHIV who do not know their status.10,11 In high HIV burden settings, community-based HIV testing is recommended as a complement to facility-based services, with a focus on efficiently and effectively reaching those in need of HTSs.10 Furthermore, WHO recommends implementing adapted programming practices and strategies to expand HTS thereby and make it more accessible, efficient, and cost-effective. This includes the integration of HTS with other testing and service decentralization.10 Over the recent years, adoption and implementation of strategies integrating routine TB screening and TPT delivery into differentiated ART delivery models in HIV national programs helped improve TPT coverage among PLHIV in high TB burden countries.12,13 So far, national TB programs have focused their TPT strategies primarily on household contacts younger than 5 years, by asking TB index cases to bring their young child contacts to facility for TB screening and TPT eligibility assessment.14 However, with this strategy, older children and adult contacts potentially infected with TB or HIV may not be reached. Community-based approaches for the identification and management of household TB contacts provide an ideal opportunity to identify TB-exposed HIV-infected people who otherwise would not have received TPT, making earlier linkage to care possible.15–17 There is little evidence on the effectiveness and feasibility of such an integrated approach. The aim of this study was to assess the integration of voluntary HIV counseling and testing (VCT) in a community-based intervention for household TB contact management and examined factors associated with the acceptance of home-based HIV VCT among household TB contacts.
METHODS
Study Design, Setting, and Population
We performed a cross-sectional analysis using data from the Community Intervention for Tuberculosis Active Contact Tracing and Preventive Therapy (CONTACT) study, a cluster randomized controlled trial performed in 2 high TB incidence, resource-limited African countries (Cameroon and Uganda) between October 2019 and January 2022. The CONTACT study evaluated the impact of a community-based intervention, as compared with the standard of care, for the contact investigations and management of TPT for household contacts of patients with bacteriologically confirmed pulmonary TB older than 15 years (index patients).18 The community intervention of the CONTACT study was implemented in 9 clusters defined by health facilities with TB diagnostic and treatment capacity and their catchment areas: 4 clusters in the Center and Littoral regions in Cameroon (3 urban/semiurban and 1 rural) and 5 clusters in the South–West region in Uganda (all rural). Facilities selected were district hospitals in Cameroon and primary health centers or district hospitals in Uganda, all supported by the CaP-TB UNITAID funded project.19 Details of the CONTACT study settings, population, and methodology have previously been published.18
We included in our analysis all contacts aged 5 years or older enrolled in the intervention arm of the CONTACT study.
Study Procedures
As part of the community intervention of the CONTACT study, community TB and HIV services were delivered to household TB contacts at home by trained community health workers (CHWs) and community nurses (CNs), all supervised by the cluster facility TB focal person. Home visits were conducted by a team comprising the CHW and a research assistant in the household(s) of consenting index patients identified by the TB focal person within 2 weeks after enrollment. During the screening visit, all household members (adults and children) were screened for TB by the CHW using a symptom-based screening questionnaire.18 Contacts with TB suggestive symptoms were referred to the facility for further investigations. Children with symptoms that did not meet the TB suggestive symptoms list were reassessed within 2 weeks in the household by the CHW. Asymptomatic children who were eligible for TPT (aged younger than 5 years and 5–14 years if HIV-positive) received a second household visit within 1 week for TPT initiation by the CN and CHW.
To identify older children (5–14 years) who could be eligible for TPT in the context of the CONTACT study, HIV VCT was proposed by the CHWs during the first TB screening visit to all child contacts aged 5 years or older with an unknown HIV status (not known to be HIV positive or without a documented HIV-negative test in the previous 3 months). HIV VCT was also proposed to all contacts aged 15 years or older with unknown HIV status who could be initiated on TPT at the HIV clinic under routine of cares. CHWs were in charge of performing the HIV VCT at the first household screening visit in Uganda. In Cameroon, HIV VCT was performed by the CN during a second household visit to comply with the national guidelines for HIV VCT. In both countries, the CHWs and CNs were trained before the intervention to perform HIV counseling, testing, and reporting results in household in compliance with national guidelines. Emphasis was placed on confidentiality and informed consent. Before the test, the CHWs and CNs had to identify a confidential location where they were to speak individually to each contact to ensure privacy and confidentiality of the testing procedure. HIV testing was performed only in persons who accepted to be tested using rapid HIV test kits Alere Determine VIH-1/2 (Abbott Diagnostics Scarborough, Inc. Scarborough, Maine, United States) recommended by the national HIV program in both countries and was followed by post-test counseling. Contacts with a positive test were referred to the cluster facility for confirmatory test using OraQuick VIH-1/2 (OraSure Technologies, Inc.) in Cameroon and HIV 1/2 STAT-PAKTM Assay (Chembio Diagnostic Systems) in Uganda. Participants confirmed with HIV were linked to the HIV clinic and initiated on ART and HIV care as per national guidelines.
Data were collected by the TB focal persons at facility level and by CHWs and CNs at household level. Data were entered by research assistants into an electronic Case Report Form on REDCap (version 8.10) using tablets.
Statistical Analysis
General characteristics of household contacts were analyzed overall and by 3 age groups: children (5–9 years), adolescents (10–19 years), and adults (20 years or older). Categorical variables were expressed as number (n) and percentages (%), and we calculated median with interquartile range (IQR) for continuous variable. Comparison between the different groups was performed using the Pearson χ2 test.
To assess the uptake of HIV VCT among household TB contacts, we described the following steps of the cascade of care for HIV testing globally, by age groups and by country: (1) enrolled contacts with unknown HIV status and who were offered VCT, (2) contacts who accepted VCT among those with unknown HIV status, (3) contacts tested for HIV among those who accepted VCT, (4) contacts with an HIV-positive test in the household among those tested, (5) contacts with a positive HIV test confirmed at the HIV clinic among those with positive results on first test, and (6) contacts with a positive HIV test initiated on ART among those confirmed with HIV infection. For stratified comparison of endpoints in cascade of care (by age or by country), generalized linear mixed models with a binomial distribution were used including country as fixed effects and clusters as random effect.
We assessed the association between index patients' characteristics (HIV status, household size, and other contact of the same index patient tested for HIV) and contact patients' characteristics (sex, age, country, rural versus urban or semiurban residence area, relation to the index patient, and TB screening result) and the acceptance of HIV testing by household contacts aged 5 years or older with unknown HIV status using a generalized linear mixed model with country as fixed effects and clusters as random effect. For the acceptance in children, we considered parental acceptance for children aged 5–7 years and for children older than 7 years, acceptance corresponded to the acceptance of the child (assent) and the parent. Variables with a P-value <0.20 in univariate models were included in the multivariate model; a manual backward stepwise approach was used to obtain the final multivariate model. Odds ratio and adjusted odds ratio were reported with their 95% confidence interval. P-value <0.05 were considered significant.
All statistical analyses were performed using R version 4.3.0 statistical software (R Development Core Team, Vienna, Austria).
Ethical Consideration
The study protocol was approved by the Advarra Institutional Review Board in the United States, the WHO Ethics Research Committee, the Cameroon National Ethics Committee for Human Health Research, the Research Ethics Committee of the Mbarara University of Science and Technology, and the Uganda National Council for Science and Technology in Uganda. Written informed consent was obtained from index patients and contacts. In addition, children older than 7 years in Cameroon and 8 years in Uganda provided written informed assent.
RESULTS
Between October 2019 and January 2022, 2206 eligible household contacts aged 5 years or older were identified (from 496 index patients), and 1983 (89.9%) of them were enrolled in the study and screened for TB. The median age of not-enrolled contacts was 34 years (IQR 17–50), and 71% aged 20 years or older. The median age of enrolled contacts was 19 years (IQR 10–33) with 522 (26.3%) 5–9 years, 503 (25.4%) 10–19 years, and 958 (48.3%) 20 years or older. Girls and women represented 60.9% of contacts; 588 contacts (29.7%) were immediate family members (parents, spouse, sibling, son, or daughter) of the index patients. The index patient was HIV-positive for 21.0% of contacts, and 331 contacts (16.7%) had a known HIV status (known HIV positive or documented HIV-negative results of less than 3 months) at enrollment and among them, 32 (1.6%) were HIV-positive. Among the 1983 contacts, 117 (5.9%) had TB suggestive symptoms at the first TB screening visit, 13 (0.7%) required a second TB screening visit, 1849 (93.2%) were asymptomatic at the first screening visit, and the screening result was not available in 4 children (0.2%). (Table 1).
TABLE 1.
Characteristics of Household Contacts by Age Group and Country
| Characteristic | Overall N = 1983 n (%) |
Age Group | P | Country | P | |||
| 5–9 yrs N = 522 n (%) |
10–19 yrs N = 503 n (%) |
20 yrs or older N = 958 n (%) |
Cameroon N = 1326 n (%) |
Uganda N = 657 n (%) |
||||
| Sex (N = 1982) | ||||||||
| Male | 775 (39.1) | 264 (50.6) | 238 (47.3) | 273 (28.6) | <0.001 | 563 (42.5) | 212 (32.3) | <0.001 |
| Female | 1207 (60.9) | 258 (49.4) | 265 (52.7) | 684 (71.4) | 763 (57.5) | 444 (67.7) | ||
| Age | ||||||||
| 5–9 | 522 (26.3) | — | — | — | — | 349 (26.3) | 173 (26.3) | |
| 10–19 | 503 (25.4) | 379 (28.6) | 124 (18.9) | <0.001 | ||||
| 20 yrs or older | 958 (48.3) | 598 (45.1) | 360 (54.8) | |||||
| Country | ||||||||
| Cameroon | 1326 (67.9) | 349 (66.9) | 379 (75.3) | 598 (62.4) | <0.001 | — | — | — |
| Uganda | 657 (33.1) | 173 (33.1) | 124 (24.7) | 360 (37.6) | ||||
| Residence area | ||||||||
| Rural | 816 (41.1) | 219 (42.0) | 172 (34.2) | 425 (44.4) | 0.001 | 159 (12.0) | 657 (100) | <0.001 |
| Urban/semiurban | 1167 (58.9) | 303 (58.0) | 331 (65.8) | 533 (55.6) | 1167 (88.0) | 0 (0.0) | ||
| Household size | ||||||||
| 1–5 | 865 (43.6) | 209 (40.0) | 201 (40.0) | 455 (47.5) | 0.003 | 544 (41) | 321 (48.9) | 0.001 |
| ≥6 | 1118 (56.4) | 313 (60.0) | 302 (60.0) | 503 (52.5) | 782 (59) | 336 (51.1) | ||
| Relation to the index | ||||||||
| Immediate family member | 588 (29.7) | 205 (39.3) | 184 (36.6) | 199 (20.8) | <0.001 | 481 (36.3) | 107 (16.3) | <0.001 |
| Other family member | 869 (43.8) | 300 (57.5) | 248 (49.3) | 321 (33.5) | 717 (54.1) | 152 (23.1) | ||
| Not family member | 64 (3.2) | 17 (3.3) | 22 (4.4) | 25 (2.6) | 60 (4.5) | 4 (0.6) | ||
| Unknown | 462 (23.3) | 0 (0.0) | 49 (9.7) | 413 (43.1) | 68 (5.1) | 394 (60.0) | ||
| Contact HIV status (initial) | ||||||||
| Negative | 299 (15.1) | 68 (13.0) | 46 (9.1) | 185 (19.3) | <0.001 | 105 (7.9) | 194 (29.5) | <0.001 |
| Positive | 32 (1.6) | 2 (0.4) | 1 (0.2) | 29 (3.0) | 11 (0.8) | 21 (3.2) | ||
| Unknown | 1652 (83.3) | 452 (86.6) | 456 (90.7) | 744 (77.7) | 1210 (91.3) | 442 (67.3) | ||
| Index patient HIV status | ||||||||
| Negative | 1559 (78.6) | 409 (78.4) | 403 (80.1) | 747 (78.0) | 0.684 | 1065 (80.3) | 494 (75.2) | <0.001 |
| Positive | 417 (21.0) | 112 (21.5) | 99 (19.7) | 206 (21.5) | 261 (19.7) | 156 (23.7) | ||
| Unknown | 7 (0.4) | 1 (0.2) | 1 (0.2) | 5 (0.5) | 0 (0.0) | 7 (1.1) | ||
| Other contact of the same index patient tested for HIV | ||||||||
| No | 341 (17.2) | 79 (15.1) | 58 (11.5) | 204 (21.3) | <0.001 | 79 (6.0) | 262 (39.9) | <0.001 |
| Yes | 1642 (82.8) | 443 (84.9) | 445 (88.5) | 754 (78.7) | 1247 (94.0) | 395 (60.1) | ||
| TB screening result* | ||||||||
| Asymptomatic | 1849 (93.2) | 478 (91.6) | 472 (93.8) | 899 (93.8) | 0.207 | 1236 (93.2) | 613 (93.3) | 0.99 |
| Needs reassessment | 13 (0.7) | 7 (1.3) | 6 (1.2) | 0 (0.0) | 13 (1.0) | 0 (0.0) | ||
| Suggestive of TB | 117 (5.9) | 37 (7.1) | 25 (5.0) | 55 (5.7) | 76 (5.7) | 41 (6.2) | ||
| Not done | 4 (0.2) | 0 (0.0) | 0 (0.0) | 4 (0.4) | 1 (0.1) | 3 (0.5) | ||
Owing to small numbers per categories, the “TB screening result” variable has been grouped into 2 categories (asymptomatic and others) for statistical comparison by age group
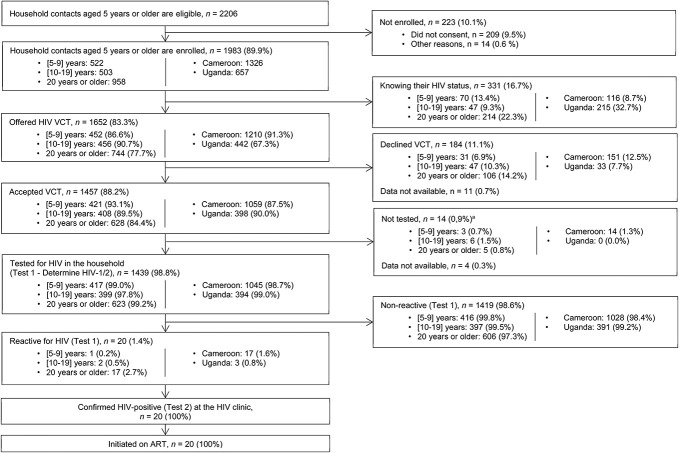
Of 1983 household contacts, 1652 (83.3%) were unaware of their HIV status, with a higher proportion among children (86.6%) and adolescents (90.7%) as compared with adults (77.7%), P < 0.001. The proportion was also higher in Cameroon (91.3%) than in Uganda (67.3%), P < 0.001. Of the 1652 contacts unaware of their HIV status, 1457 (88.2%) accepted HIV VCT. The acceptance of HIV VCT was lower among adults (84.4%) as compared with children (93.1%), P < 0.001; there was no significant difference among adolescents (89.5%) as compared with children, P = 0.100. HIV VCT acceptance was also lower in Cameroon (87.5%) compared with Uganda (90.0%), but this difference was not statistically significant in bivariate analyses (P = 0.285). Of 1439 contacts tested, 20 (1.4%) were HIV-positive, and of them, 17 (85.5%) were adults. They were all confirmed with the second HIV test at a facility, linked to HIV care, and initiated on ART (Fig. 1 and Table 2).
FIGURE 1.
Testing cascade per age group and per country. The proportions are calculated using the total number (n) from previous steps as the denominator. VCT, voluntary HIV counseling and testing. aReasons not documented.
TABLE 2.
Factors Associated With Acceptance of HIV Voluntary Counseling and Testing Among Household Contacts With Unknown HIV Status
| Overall (N = 1641)* | Accepted VCT n (%) |
OR (CI 95%) | P | aOR (CI 95%) | P | |
| Sex | ||||||
| Female | 974 | 861 (88.4) | 1 | |||
| Male | 667 | 596 (89.4) | 1.17 (0.85 to 1.63) | 0.336 | ||
| Age (yrs) | ||||||
| 5–9 | 452 | 421 (93.1) | 1 | 1 | ||
| 10–19 | 455 | 408 (89.7) | 0.67 (0.41 to 1.08) | 0.100 | 0.71 (0.43 to 1.18) | 0.183 |
| 20 yrs or older | 734 | 628 (85.6) | 0.35 (0.23 to 0.54) | <0.001 | 0.35 (0.22 to 0.55) | <0.001 |
| Country | ||||||
| Cameroon | 1210 | 1059 (87.5) | 1 | 1 | ||
| Uganda | 431 | 398 (92.3) | 2.16 (0.53 to 8.87) | 0.285 | 4.54 (1.17 to 17.62) | 0.029 |
| Index patient HIV status | ||||||
| Negative | 1301 | 1174 (90.2) | 1 | 1 | ||
| Positive | 336 | 280 (83.3) | 0.54 (0.37 to 0.77) | 0.001 | 0.56 (0.38 to 0.83) | 0.003 |
| Unknown | 4 | 3 (75.0) | 0.47 (0.05 to 4.84) | 0.525 | 0.37 (0.03 to 4.11) | 0.417 |
| Contact TB screening result (N = 1639) | ||||||
| Asymptomatic | 1533 | 1362 (88.8) | 1 | 1 | ||
| Need reassessment | 12 | 6 (50.0) | 0.31 (0.10 to 0.98) | 0.045 | 0.20 (0.06 to 0.67) | 0.009 |
| Suggestive of TB | 94 | 89 (94.7) | 2.22 (0.87 to 5.67) | 0.094 | 2.55 (0.95 to 6.83) | 0.063 |
| Household size | ||||||
| 1–5 | 709 | 633 (89.3) | 1 | |||
| ≥6 | 932 | 824 (88.4) | 0.91 (0.65 to 1.27) | 0.578 | ||
| Relation with the index patient | ||||||
| Direct family member | 512 | 469 (91.6) | 1 | |||
| Other family member | 764 | 668 (87.4) | 0.73 (0.49 to 1.09) | 0.126 | ||
| Not family member | 50 | 41 (82.0) | 0.66 (0.30 to 1.49) | 0.318 | ||
| Unknown | 315 | 279 (88.6) | 0.45 (0.24 to 0.81) | 0.008 | ||
| Other contact of the same index patient tested for HIV | ||||||
| No | 123 | 84 (68.3) | 1 | 1 | ||
| Yes | 1518 | 1373 (90.4) | 8.28 (4.86 to 14.11) | <0.001 | 9.22 (5.25 to 16.18) | <0.001 |
| District location | ||||||
| Rural | 586 | 533 (91.0) | 1 | |||
| Urban/semiurban | 1055 | 924 (87.6) | 0.64 (0.14 to 2.90) | 0.560 | ||
All models used for analysis included clusters as random effect.
Information about VCT acceptance was missing in 11 (0.7%) of the 1652 participants who were offered VCT.
After multivariate analysis, factors independently associated with the acceptance of HIV VCT residing in Uganda (aOR = 4.54, 95% CI: 1.17 to 17.62) and having another contact of the same index patient tested for HIV (aOR = 9.22, 95% CI: 5.25 to 16.18). Adults as compared with children (aOR = 0.35, 95% CI: 0.22 to 0.55, P < 0.001), being a contact of a HIV-positive index patient (aOR = 0.56, 95% CI: 0.38 to 0.83) and contacts needing a reassessment TB screening visit (aOR = 0.20, 95% CI: 0.06 to 0.67) were less likely to accept HIV VCT.
DISCUSSION
In this study, we describe a model of service delivery integrating voluntary HIV testing and counseling into a community-based intervention for household TB contact management in Cameroon and Uganda. Integrating VCT with household TB contact screening enabled 88.2% of household contacts aged 5 years or older to learn their HIV status and helped to identify 20 TB-exposed PLHIV who did not know their HIV status, linking them successfully to treatment services. This intervention increased access to HIV cares and TB prevention services among TB contacts. This is particularly important for these persons given that TB-exposed PLHIV have an increased risk of developing TB disease.3–5 Despite the scaling up and strong adoption of integrated delivery models of TB and HIV care in resource-limited settings over the recent past years, most of these models are facility-based, requiring exposed or at-risk people to come to the health facility for HIV and/or TB testing and treatment.20 Our results reinforce the WHO recommendation to integrate TB/HIV activities in community.21 This has the advantage of offering the opportunity to screen the household members and identify all individuals at increased risk of developing TB after exposure, including PLHIV.
Our results show that most of the TB-exposed individuals in households were not aware of their HIV status and that integrating VCT with TB screening was highly accepted by household contacts. The results were consistent across different age groups and in the 2 countries. The HIV testing acceptance rate (88.2%) in our study was consistent with general population-based studies performed in Uganda and Kenya, which reported high acceptance rates for home-based HIV counseling and testing.22,23 Our findings were also consistent with the result of a similar study performed in Uganda and assessing the integration of home HIV counseling and testing into household TB contact investigation, which observed an acceptance rate of VCT of 80.3% among household contacts with unknown HIV status.24 The high level of acceptance among household contacts may be explained by the counseling performed in this study that included information on the relation between HIV infection and TB exposure and the study context that included the assessment of a parent and child/adolescent for participation in the CONTACT trial. Our results show that several factors that could have been barriers to acceptance of VCT did not influence test acceptance. The association between the household size and the acceptance of the HIV test was not statistically significant. This result could be explained by the fact that the CN and CHW had the possibility to schedule another visit to complete the contact screening in case of high workload because of the size of the household. This is reassuring for future implementation but may need to be verified under routine conditions. The geographic location of the household was not associated with the acceptance, suggesting that this integrated approach could work well in a rural or urban area. However, this intervention was implemented exclusively in rural area in Uganda and more in urban or semiurban area in Cameroon. The effect of the geographic location on the acceptance of VCT might be different when considering each country. The type of familiar relationship between the contact and the index patient was not associated with VCT acceptance. We identified some factors that were significantly associated with acceptance of VCT. Our results show that household contacts in Cameroon were less likely to accept the VCT than in Uganda. These findings align with the current situation of community HIV testing in the 2 countries. Community-based service delivery is among key strategies of Uganda's national AIDS policy, and home-based HIV counseling and testing is widely implemented in Uganda compared with Cameroon.25–28 This suggests that acceptance of integration is increased in settings where community-based interventions are widespread. In this study, child (aged 5–9 years) household contacts were more likely to accept VCT than adults, which may reflect the willingness of parents to test their younger children in home. Adolescents (90.7%) had also high level of VCT acceptance in our study. Similar findings have been reported in other home-based HIV testing programs and studies in sub-Saharan Africa.29–31 Several studies reported lower HIV VCT acceptance rates among adolescents when testing strategies are centered on care at the level of health facilities.32,33 The effectiveness of community-based approaches for improving HIV test uptake among adolescents has been proven by several previous studies and is recommended by WHO as important strategy for increasing VCT coverage among adolescents.34 The results from our study shows that community-based interventions for VCT integrated into community TB service delivery remains effective in adolescents. Our findings suggest that household contacts from HIV-positive index patients were less likely to accept testing during home visits than household contacts from HIV-negative index cases. Our hypothesis is that some contacts living in the same household than a HIV-positive index patient may not have disclosed their HIV status and may accept to be enrolled in the study to benefit from the TB contact screening but refuse to be tested for HIV. TB contacts needing a second visit for TB screening were less likely to accept VCT compared with contacts with only 1 screening visit. This association is difficult to explain and should be taken with caution knowing the very low number of contacts that needed to be reassessed (n = 13, 0.7%). Having another contact of the same index patient tested for HIV was positively associated with the acceptance of HIV testing. This finding suggests that other attitudes of others in the household may affect testing acceptance, referring to the concept of collective acceptance.35
Several key implementation aspects were crucial for the success of this integrated approach and must be take into consideration for routine implementation. This includes the selection of CHWs. CHWs must be experienced in community-based activities on health issues with good reading and writing skills and ideally members of the same community.36 Another aspect is the training of CHWs and CNs to conduct VCT according to national algorithms and to the respect of ethics and confidentiality that are critical to reduce the stigma. Furthermore, it is crucial to implement an efficient referral system and communication with the health facilities for contacts with TB suggestive symptoms and to ensure linkage to care. In this study, the existing referral system was strengthened and did not include transport reimbursement. It is also important to anticipate resource implications of household visits to ensure logistical aspects (supply of HIV testing kits), cost of transporting CHWs for household visits, and eventually financial incentives.
A few limitations should be noted. Because in the study, VCT was proposed to contacts aged 5 years or older and that the facility-based standard of care (control) was focusing on young children (younger than 5 years), we were not able to compare acceptance rate between both approaches. This would have been helpful to inform on the effectiveness of this intervention. Second, in this study, the VCT was not proposed by the CHWs to children older than 5 years because this activity was performed as part of a community intervention targeting priority populations for TPT, which included young children younger than 5 years, regardless of their HIV status. Third, the evaluation of the integration of the HIV testing with household contact TB screening was not part of the main outcomes of the CONTACT study explaining the limited number of variables to explore factors associated with HIV acceptance and the absence of information about the barriers and facilitators of the integration of the HIV testing with household TB contact screening. Fourth, we cannot exclude the possibility that some contacts refused to participate in the study because of the HIV testing, which could result in a potential overestimation of the VCT acceptance. Finally, although the CONTACT trial was a pragmatic study, it does not reflect the on-the-ground routine practices.
Community-based approaches have shown their effectiveness in contributing to broader health service delivery in context of limited access to care.15,37 WHO recognizes that these approaches represent opportunities to adapt or develop new ways to integrate HIV and TB care.15 Among them, integrating HIV testing into household TB contact screening visits offers great potential to increase access to both HIV and TB care for PLHIV who do not know their HIV status. This study brings some evidence on the effectiveness and feasibility of this integrated community-based approach using CHWs in 2 high TB and HIV burden countries with limited resources.
CONCLUSIONS
HIV testing can be integrated into community-based household TB contact screening and is well-accepted. This is a key approach to identifying PLHIV at high risk of developing TB that would otherwise be missed. Consideration may be given to integrating HIV and TB activities with other community-directed interventions.
ACKNOWLEDGMENTS
The authors gratefully acknowledge the contributions of the study participants and dedication of the staff at the CONTACT study sites. The authors also thank the Ministry of Health and National Tuberculosis Program of Cameroon and Uganda who supported the implementation of the CaP-TB project and the CONTACT study.
APPENDIX. CONTACT Study Group
Savine Chauvet, Elisabete de Carvalho, Sayouba Ouedraogo, Gwenvael Leguicher (IRD, France), Appolinaire Tiam, Maria Oziemkowska, Elizabeth Atieno Ayuo (Elizabeth Glaser Pediatrics AIDS Foundation, USA [Sponsor]), Nyashadzaishe Mafirakureva (Sheffield University, UK), Mikhael de Souza, Maude Berset, Jean-Francois Lemaire (Elizabeth Glaser Pediatrics AIDS Foundation,Switzerland), Collette Sih, Rogacien Kana, Eric Youm, Jinette Lorraine Guedem Nekame, Paul Williams Manguele, Paul Bindzi, Marie Louise Aimée Ndongo, Doline Ndjang Kombou, Philippe Narcisse Tsigaing, Muhamed Mbunka Awolu, Leticia Grace Seuleu Ndjamakou, Narcisse Sitamze Kaptue (Elizabeth Glaser Pediatrics AIDS Foundation, Cameroon), Dany Flora Ngounou Moyo, Raimatou Patouokoumche Ngouh, Joseph Stéphane Kouotou Mouliom, Henriette Alida Abogo Abatsong, Rose Cynthia Essebe Ngangue, Rodrigue Djeumene, Laure Tatiane Maguia Tatiane Kouam, Lydie Flore Djilo Nono, Marie-Josée Bakmano Raïssa, Kong Derick Njikeh (Ministry of Health facilities, district hospitals Cameroon), Anne-Cécile Bissek (Division of operational research on health, Cameroon), Rinah Arinaitwe, David Otai, Hamidah Kamanzi, Agnes Natukunda, Eva Natukunda, Rose Kyarimpa, Doreen Kyomuhendo, Scovia Sanyu, John Ssemanya, Jane Nabbuto, Ssuan Lugoose, Kiconco Rachael, Jonanita Tebylwa Berytah, Frank Kitakule, Syson Atuhaire, Moreen Kembabazi, Florence Abok (Epicentre, Uganda), Michael Kakinda, Dickens Odongo, Henri Ijjo (Elizabeth Glaser Pediatrics AIDS Foundation, Uganda), Clemencia Kyomugisha, Johnan Aryatuhwera, Beth Ashaba, Patrick Nuwamanya, Merdard Arinaitwe, Prisca Natukunda, Collins Muhangi, Deogratious Muhumuza, Gordon Ndyeimuka, Johnson Bagabe, Justus Tiboruhanga, Francis Tibaijuka, Marion Nahabwe (Ministry of Health facilities, health centres Uganda).
Footnotes
This work was funded by Unitaid.
The authors have no funding or conflicts of interest to disclose.
The data that support the findings of this study are available on request from the corresponding author. Data will be shared after approval by a committee of relevant persons involved in the study and after examining the relevance of the request and considering ethical and legal implications.
B.T.Y. wrote the original draft of the manuscript and interpreted results. M.B. and A.V. conceived the study and designed the study protocol. M.C., B.K.T., D.A., and J.C. contributed to the study design. M.B., A.V., D.A., B.K.T., B.T.Y., B.S., D.O., and C.S. supervised the study implementation. R.K., R.A., L.S., R.O., P.T., A.K.K., and S.T. provided support to the study implementation. B.T.Y. did the statistical analysis with the support of B.C. B.K.T. and M.B. provided substantial input in the manuscript. B.C., C.S., A.V., R.K., M.C., and D.A. critically revised the manuscript. All authors have read and approved the final manuscript.
Contributor Information
Daniel Atwine, Email: daniel.atwine@must.ac.ug.
David Otai, Email: David.OTAI@epicentre.msf.org.
Anca Vasiliu, Email: Anca.Vasiliu@bcm.edu.
Bob Ssekyanzi, Email: sekyanzi@gmail.com.
Colette Sih, Email: sihcolette91@gmail.com.
Rogacien Kana, Email: rkana@pedaids.org.
Rinah Arinaitwe, Email: Rinah.ARINAITWE@epicentre.msf.org.
Benjamin Cuer, Email: benjamin.cuer@ird.fr.
Leonie Simo, Email: lsimo@pedaids.org.
Richard Okello, Email: rokello@pedaids.org.
Patrice Tchendjou, Email: ptchendjou@pedaids.org.
Martina Casenghi, Email: mcasenghi@pedaids.org.
Albert Kuate Kuate, Email: akuate2001@yahoo.com.
Stavia Turyahabwe, Email: turyahabwestavia@gmail.com.
Jennifer Cohn, Email: jecohn6@gmail.com.
Maryline Bonnet, Email: maryline.bonnet@ird.fr.
Boris Kevin Tchounga, Email: btchounga@pedaids.org.
Collaborators: Savine Chauvet, Elisabete de Carvalho, Sayouba OuAppolinaire Tiam, Maria Oziemkowska, Elizabeth Atieno Ayuoedraogo, Gwenvael Leguicher, Nyashadzaishe Mafirakureva, Mikhael de Souza, Maude Berset, Jean-Francois Lemaire, Collette Sih, Rogacien Kana, Eric Youm, Jinette Lorraine Guedem Nekame, Paul Williams Manguele, Paul Bindzi, Marie Louise Aimée Ndongo, Doline Ndjang Kombou, Philippe Narcisse Tsigaing, Muhamed Mbunka Awolu, Leticia Grace Seuleu Ndjamakou, Narcisse Sitamze Kaptue, Dany Flora Ngounou Moyo, Raimatou Patouokoumche Ngouh, Joseph Stéphane Kouotou Mouliom, Henriette Alida Abogo Abatsong, Rose Cynthia Essebe Ngangue, Rodrigue Djeumene, Laure Tatiane Maguia Tatiane Kouam, Lydie Flore Djilo Nono, Marie-Josée Bakmano Raïssa, Kong Derick Njikeh, Anne-Cécile Bissek, Rinah Arinaitwe, David Otai, Hamidah Kamanzi, Agnes Natukunda, Eva Natukunda, Rose Kyarimpa, Doreen Kyomuhendo, Scovia Sanyu, John Ssemanya, Jane Nabbuto, Ssuan Lugoose, Kiconco Rachael, Jonanita Tebylwa Berytah, Frank Kitakule, Syson Atuhaire, Moreen Kembabazi, Florence Abok, Michael Kakinda, Dickens Odongo, Henri Ijjo, Clemencia Kyomugisha, Johnan Aryatuhwera, Beth Ashaba, Patrick Nuwamanya, Merdard Arinaitwe, Prisca Natukunda, Collins Muhangi, Deogratious Muhumuza, Gordon Ndyeimuka, Johnson Bagabe, Justus Tiboruhanga, Francis Tibaijuka, and Marion Nahabwe
REFERENCES
- 1.Publications UN. In Danger: UNAIDS Global AIDS Update 2022. UN; 2022:376. [Google Scholar]
- 2.World Health Organization. Global Tuberculosis Report 2022 [Internet]. Geneva: World Health Organization; 2022:68. Available at: https://www.who.int/publications/i/item/9789240061729. 3/15/2023 [Google Scholar]
- 3.Getahun H, Gunneberg C, Granich R, et al. HIV infection-associated tuberculosis: the epidemiology and the response. Clin Infect Dis. 2010;50(suppl 3):S201–S207. [DOI] [PubMed] [Google Scholar]
- 4.Pawlowski A, Jansson M, Sköld M, et al. Tuberculosis and HIV co-infection. PLoS Pathog. 2012;8:e1002464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Letang E, Ellis J, Naidoo K, et al. Tuberculosis-HIV co-infection: progress and challenges after two decades of global antiretroviral treatment roll-out. Arch Bronconeumol. 2020;56:446–454. [DOI] [PubMed] [Google Scholar]
- 6.WHO policy on collaborative TB/HIV activities : guidelines for national programmes and other stakeholders [Internet]. 2022. World Health Organization; https://www.who.int/publications/i/item/9789241503006. 03/01/24 [PubMed] [Google Scholar]
- 7.WHO Consolidated Guidelines on Tuberculosis: Tuberculosis Preventive Treatment: Module 1: Prevention [Internet]. Geneva: World Health Organization; 2020 WHO Guidelines Approved by the Guidelines Review Committee. Available at: http://www.ncbi.nlm.nih.gov/books/NBK554956/ (2020, 08/20/2022) [Google Scholar]
- 8.Getahun H, Matteelli A, Abubakar I, et al. Management of latent Mycobacterium tuberculosis infection: WHO guidelines for low tuberculosis burden countries. Eur Respir J. 2015;46:1563–1576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zenner D, Beer N, Harris RJ, et al. Treatment of latent tuberculosis infection: an updated network meta-analysis. Ann Intern Med. 2017;167:248–255. [DOI] [PubMed] [Google Scholar]
- 10.World Health Organization. Consolidated guidelines on HIV testing services [Internet]; 2019:286. Available at: https://www.who.int/publications-detail-redirect/978-92-4-155058-1 (2019, 05/26/2023)
- 11.World Health Organization. Service Delivery Approaches to HIV Testing and Conselling (HTC): A Strategic HTC Programme Framework. World Health Organization. 2012
- 12.González Fernández L, Casas EC, Singh S, et al. New opportunities in tuberculosis prevention: implications for people living with HIV. J Int AIDS Soc. 2020;23:e25438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Boyd AT, Moore B, Shah M, et al. Implementing TB preventive treatment within differentiated HIV service delivery models in global programs. Public Health Action. 2020;10:104–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vasiliu A, Salazar-Austin N, Trajman A, et al. Child contact case management—a major policy-practice gap in high-burden countries. Pathogens. 2021;11:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.World Health Organization. WHO Operational Handbook on Tuberculosis: Module 1: Prevention: Tuberculosis Preventive Treatment [Internet]. World Health Organization; 2020:ix, 121. Available at: https://apps.who.int/iris/handle/10665/331525 (2020, 10/11/2020) [Google Scholar]
- 16.Shapiro AE, Variava E, Rakgokong MH, et al. Community-based targeted case finding for tuberculosis and HIV in household contacts of patients with tuberculosis in South Africa. Am J Respir Crit Care Med. 2012;185:1110–1116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lala SG, Little KM, Tshabangu N, et al. Integrated source case investigation for tuberculosis (TB) and HIV in the caregivers and household contacts of hospitalised young children diagnosed with TB in South Africa: an observational study. PLoS One. 2015;10:e0137518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vasiliu A, Eymard-Duvernay S, Tchounga B, et al. Community intervention for child tuberculosis active contact investigation and management: study protocol for a parallel cluster randomized controlled trial. Trials. 2021;22:180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Unitaid. Catalyzing paediatric tuberculosis innovations [Internet]. Available at: https://unitaid.org/project/catalyzing-paediatric-tuberculosis-services-cap-tb/
- 20.Legido-Quigley H, Montgomery CM, Khan P, et al. Integrating tuberculosis and HIV services in low- and middle-income countries: a systematic review. Trop Med Int Health. 2013;18:199–211. [DOI] [PubMed] [Google Scholar]
- 21.World Health Organization. WHO Consolidated Guidelines on Tuberculosis: Module 5: Management of Tuberculosis in Children and Adolescents [Internet]. World Health Organization; 2022. Available at: https://www.ncbi.nlm.nih.gov/books/NBK579380/ (2022, 10/18/2022) [PubMed] [Google Scholar]
- 22.Sekandi JN, Sempeera H, List J, et al. High acceptance of home-based HIV counseling and testing in an urban community setting in Uganda. BMC Public Health. 2011;11:730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dalal W, Feikin DR, Amolloh M, et al. Home-based HIV testing and counseling in rural and urban Kenyan communities. J Acquir Immune Defic Syndr. 2013;62:e47-e54. [DOI] [PubMed] [Google Scholar]
- 24.Ochom E, Meyer AJ, Armstrong-Hough M, et al. Integrating home HIV counselling and testing into household TB contact investigation: a mixed-methods study. Public Health Action. 2018;8:72–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Asiimwe S, Ross JM, Arinaitwe A, et al. Expanding HIV testing and linkage to care in southwestern Uganda with community health extension workers. J Int AIDS Soc. 2017;20(suppl 4):21633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Uganda AIDS Commission. National HIV and AIDS Priority Action Plan 2018; Kampala, Uganda: Ministry of Health; 2019. [Google Scholar]
- 27.Division of Health Operations Research of the Cameroon Ministry of Health; The National AIDS Control Committee of the Cameroon Ministry of Health; National Institute of Statistics of Cameroon; U.S. President's Emergency Plan for AIDS Relief; Centers for Disease Control and Prevention (U.S.); Westat, inc.; ICAP at Columbia University. Cameroon Population-Based HIV Impact Assessment CAMPHIA 2017-2018; Cameroon: Ministry of Health; 2020. [Google Scholar]
- 28.Ware NC, Wyatt MA, Asiimwe S, et al. How home HIV testing and counselling with follow-up support achieves high testing coverage and linkage to treatment and prevention: a qualitative analysis from Uganda. J Int AIDS Soc. 2016;19:20929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wachira J, Ndege S, Koech J, et al. HIV testing uptake and prevalence among adolescents and adults in a large home-based HIV testing program in Western Kenya. J Acquir Immune Defic Syndr. 2014;65:e58–e66. [DOI] [PubMed] [Google Scholar]
- 30.Shanaube K, Schaap A, Chaila MJ, et al. Community intervention improves knowledge of HIV status of adolescents in Zambia: findings from HPTN 071-PopART for youth study. AIDS. 2017;31(suppl 3):S221–S232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Govindasamy D, Ferrand RA, Wilmore SM, et al. Uptake and yield of HIV testing and counselling among children and adolescents in sub-Saharan Africa: a systematic review. J Int AIDS Soc. 2015;18:20182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lugada E, Levin J, Abang B, et al. Comparison of home and clinic-based HIV testing among household members of persons taking antiretroviral therapy in Uganda: results from a randomized trial. J Acquir Immune Defic Syndr. 2010;55:245–252. [DOI] [PubMed] [Google Scholar]
- 33.Sweat M, Morin S, Celentano D, et al. Community-based intervention to increase HIV testing and case detection in people aged 16-32 years in Tanzania, Zimbabwe, and Thailand (NIMH Project Accept, HPTN 043): a randomised study. Lancet Infect Dis. 2011;11:525–532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.World Health Organization. HIV and Adolescents: Guidance for HIV Testing and Counselling and Care for Adolescents Living with HIV: Recommendations for a Public Health Approach and Considerations for Policy-Makers and Managers [Internet]. World Health Organization; 2013;xi, 81. Available at: https://apps.who.int/iris/handle/10665/94334 (2013, 05/26/2023). [PubMed] [Google Scholar]
- 35.Tuomela R. Collective acceptance and the formation of group attitudes. In: Tuomela R, ed. Social Ontology: Collective Intentionality and Group Agents [Internet]. Oxford University Press; 2013. Available at: 10.1093/acprof:oso/9780199978267.003.0005 (2013, 03/16/2023). [DOI] [Google Scholar]
- 36.Vasiliu A, Tiendrebeogo G, Awolu MM, et al. Feasibility of a randomized clinical trial evaluating a community intervention for household tuberculosis child contact management in Cameroon and Uganda. Pilot Feasibility Stud. 2022;8:39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Moh DR, Bangali M, Coffie P, et al. Community health workers. Reinforcement of an outreach strategy in rural areas aimed at improving the integration of HIV, tuberculosis and malaria prevention, screening and care into the health systems. “Proxy-Santé” study. Front Public Health. 2022;10:801762. [DOI] [PMC free article] [PubMed] [Google Scholar]