Abstract
Cervical spinal cord injury impacts ventilatory and non‐ventilatory functions of the diaphragm muscle (DIAm) and contributes to clinical morbidity and mortality in the afflicted population. Periodically, integrated brainstem neural circuit activity drives the DIAm to generate a markedly augmented effort or sigh—which plays an important role in preventing atelectasis and thus maintaining lung function. Across species, the general pattern of DIAm efforts during a normal sigh is variable in amplitude and the extent of post‐sigh “apnea” (i.e., the post‐sigh inter‐breath interval). This post‐sigh inter‐breath interval acts as a respiratory reset, following the interruption of regular respiratory rhythm by sigh. We examined the impact of upper cervical (C2) spinal cord hemisection (C2SH) on the transdiaphragmatic pressure (P di) generated during sighs and the post‐sigh respiratory reset in rats. Sighs were identified in P di traces by their characteristic biphasic pattern. We found that C2SH results in a reduction of P di during both eupnea and sighs, and a decrease in the immediate post‐sigh breath interval. These results are consistent with partial removal of descending excitatory synaptic inputs to phrenic motor neurons that results from C2SH. Following cervical spinal cord injury, a reduction in the amplitude of P di during sighs may compromise the maintenance of normal lung function.
Keywords: diaphragm, respiration, spinal cord, transdiaphragmatic pressure, ventilation
1. INTRODUCTION
The diaphragm muscle (DIAm) is unique to mammals, and as a pump, it provides the major contribution to the generation of transdiaphragmatic pressure (P di) for ventilatory (breathing) and non‐ventilatory behaviors, including cough, sneeze, and straining maneuvers (Fogarty & Sieck, 2019; Sieck & Fournier, 1989). During ventilatory behaviors such as quiet breathing (eupnea), breaths in response to hypoxia and/or hypercapnia, or breathing efforts against an occluded airway, the P di generated is substantially less than the maximum P di generated by bilateral phrenic nerve stimulation (P dimax) (Fogarty et al., 2023; Khurram et al., 2018; Mantilla et al., 2010; Seven et al., 2018; Sieck, 1991). By contrast, the P di generated during non‐ventilatory airway clearance (e.g., cough and sneeze) or straining (e.g., defecation) behaviors are much higher, approaching P dimax (Fogarty et al., 2023; Khurram et al., 2018; Mantilla et al., 2010; Seven et al., 2018; Sieck, 1991). Often reported, but less empirically characterized in rats are sighs, also known as augmented breaths, which are biphasic with an increased flow/volume and an extended subsequent inter‐breath interval (Cherniack et al., 1981; Golder et al., 2005; van Lunteren et al., 1983). In cats, the post‐sigh breath is muted in duration and tidal volume (Cherniack et al., 1981; Reynolds Jr. & Hilgeson, 1965), whereas both the post‐sigh inter‐breath interval and tidal volume are increased in mice (Voituron et al., 2010) and unaltered in dogs (van Lunteren et al., 1983). This post‐sigh breath effectively serves as a respiratory reset, allowing for the resumption of normal eupnea post‐sigh (Ramirez, 2014; Ramirez et al., 2022).
It has been suggested that the main function of sighs is to prevent lung atelectasis (Housley et al., 1970; Katagiri et al., 1998; Lieske et al., 2000; Ramirez et al., 2022; Riede et al., 2020; Wuyts et al., 2011). Despite being sensitive to hypoxia, hypercapnia, and lung inflation, sighs still occur when carotid bodies are denervated, in vagotomized conditions and when lungs are transplanted (Severs et al., 2022). Sighs are involved in a variety of emotional states in experimental animals including rats and humans (Ajayi & Mills, 2017; Li et al., 2016; Ramirez, 2014; Ramirez et al., 2022; Riede et al., 2020; Severs et al., 2022; Soltysik & Jelen, 2005; Vlemincx et al., 2022). Sighs are often interchangeably used as a discrete behavior (Burns et al., 2019; Fuller et al., 2008; Seven et al., 2014), a basis for normalization for other breath data (Jensen et al., 2019; Mantilla et al., 2011; Seven et al., 2018; Wen & Lee, 2018), and a “filtered” behavior—normalized to greater than twice eupnea for DIAm EMG (Rana et al., 2017) or 2.5–3 times eupnea for plethysmography (Bell et al., 2009; Bell & Haouzi, 2010; Li et al., 2016; Sheikhbahaei et al., 2017). Many studies have used sighs to simultaneously serve as a combination of all these uses (Bell et al., 2009; Bell & Haouzi, 2010; Hernandez‐Torres et al., 2017; Jimenez‐Ruiz et al., 1985; Mantilla et al., 2011; Rana et al., 2017; Seven et al., 2018), severely curtailing any meaningful interpretation.
There is some contention as to whether the P di generated during sighs in rats has a “stereotypical” amplitude (e.g., >twice eupnea) or biphasic pattern (Khurram et al., 2017; Seven et al., 2018) similar to mice (Pareja‐Cajiao et al., 2021), or is variable (e.g., amplitudes <twice eupnea [Khurram et al., 2018]) such as in cats (Reynolds Jr. & Hilgeson, 1965). Additionally, little to no attention has been paid to the post‐sigh inter‐breath interval or respiratory reset in rats. The empirical characterization of sighs in rats would provide a basis for interpreting the neurophysiological underpinnings of DIAm motor unit recruitment, which currently stands as either lower than (Khurram et al., 2017, 2018) or greater than occlusion P di (Mantilla et al., 2010). The gamut of respiratory neuromotor P di generation in the rat ranges ~10‐fold from between eupnea (~10–15 cm H2O) to P dimax (~100 cm H2O). Where sighs fall within this range is currently unknown, as a winnowing of data by inclusion/exclusion criteria obscures interpretation.
Unilateral upper cervical spinal cord hemisection (C2SH) severs ipsilateral descending excitatory inputs to phrenic motor neurons (PhMNs) (Rana et al., 2020; Tai & Goshgarian, 1996) severely blunting PhMN and DIAm activation on the injured side (Miyata et al., 1995). As a result, P di and P dimax is reduced (Fogarty et al., 2023), with ventilation sustained primarily by compensatory activation of the contralateral DIAm (Fuller et al., 2006; Mantilla, Gransee, et al., 2013; Martinez‐Galvez et al., 2016). Although the effect of C2SH on P di generated during sighs is unknown, C2SH reduces DIAm EMG during sighs (Bezdudnaya et al., 2018), sigh tidal volume (Dougherty et al., 2012; Fuller et al., 2008; Golder et al., 2005), and post‐sigh inter‐breath interval (Golder et al., 2005).
The goal of the present study was to examine the impact of C2SH on the P di generated during sighs as well as inter‐breath intervals before and immediately after sighs. We also report the proportion of “stereotypical” sighs meeting common inclusion criteria (i.e., P di amplitudes and a post‐sigh inter‐breath interval of ≥twice eupnea) under normal pre‐injury, C2SH, and sham (laminectomy) conditions. We hypothesize that C2SH will reduce P di amplitude and the duration of the post‐sigh inter‐breath interval.
2. MATERIALS AND METHODS
2.1. Experimental animals
A total of 16 adult male Sprague‐Dawley (SD) rats (~325 g) obtained from Envigo (Indianapolis, IN) were used in the study. We have previously used male rats to examine the impact C2SH, with a highly repeatable pathological and physiological outcome (Fogarty et al., 2023; Mantilla, Gransee, et al., 2013; Sieck et al., 2021). Animals were acclimated for at least 7 days prior to any procedure and maintained on an alternating 12:12 h light–dark cycle with ad libitum access to fresh water and food. For all procedures, anesthesia was administered via intraperitoneal injections of xylazine (10 mg/kg) and ketamine (80 mg/kg).
2.2. Experimental design and timeline
Rats were placed into two experimental groups at random, sham laminectomy, or C2SH. These groups were further separated by timepoints into pre‐surgery (PRE‐SHAM and PRE‐C2SH) and post‐surgery (POST‐SHAM and POST‐C2SH). Anesthetized rats were placed in the prone position, where baseline P di was measures once depth of anesthesia was stable (absence of palpebral and deep pain reflexes). P di measures for both the sham laminectomy and C2SH took ~20 min, with the SHAM or C2SH surgeries taking ~20–30 min and the final P di recording sessions taking ~20 min. The sham group serves as a time control, as occasional redosing of xylazine (3 mg/kg) and ketamine (25 mg/kg) at ~one third of the initial dose was required (3 in 8 of the C2SH and 2 in 8 of the SHAM rats). Thus, all pre‐ and post‐surgery measurements were performed in one experimental session.
2.3. P di measurements
P di was measured as the difference between esophageal and gastric pressures, with two 3.5 French solid‐state pressure catheters (SPR‐524; Millar Instruments, Houston, TX) inserted through the mouth and into the esophagus or stomach (Fogarty et al., 2023; Khurram et al., 2018; Sieck & Fournier, 1989), both before (uninjured) and after C2SH or sham laminectomy. The abdomen was bound to approximate isometric conditions. Esophageal and abdominal pressure signals were recorded and digitized (400 Hz) with PowerLab 4/35, with P di calculated and visualized in real‐time with LabChart 9 (ADInstruments, Colorado Springs, CO). From the P di tracings, P di amplitude, respiratory rate (and inter‐breath intervals), and inspiratory duration were determined in a manner identical to previous reports (Fogarty et al., 2022, 2023). All behaviors were recorded with initial O2 saturation of >90%, in accordance with past studies (Khurram et al., 2019).
2.4. C2SH surgery
The surgical methods for C2SH have been previously described in detail (Beth Zimmer et al., 2015; Fuller et al., 2006, 2008; Miyata et al., 1995; Streeter et al., 2020), with the lesion functionally validated using DIAm EMG (with electrodes implanted 3 days prior to C2SH) during the time of surgery and 3 days post‐surgery, in accordance with previous studies (Fogarty et al., 2023; Mantilla, Gransee, et al., 2013; Sieck et al., 2021). Briefly, following a dorsal laminectomy, the C2 spinal cord was cut using a surgical microknife, beginning anterior to the dorsal root entry zone fissure and proceeding ventrally taking care to preserve the dorsal funiculus on the right side, ~7 mm from the midline at the level of the dorsal subdural. The sham group experienced a dorsal laminectomy alone, serving as a time control.
2.5. Statistical analyses
The data sets comprise eupnea (with ~130 individual breaths assessed per rat per surgery status [range: 102–154]), sigh (identified manually as a biphasic breath, with a superimposed additional effort causing a point of inflection at the plateauing inspiratory phase [Cherniack et al., 1981; Reynolds Jr. & Hilgeson, 1965]), and post‐sigh eupnea (the breath immediately following a sigh). For sighs and post‐sigh breaths, ~8 breaths were analyzed per rat and only during eupneic breathing (range: 5–12), as sigh incidence and amplitudes may be affected by baseline respiratory drive (Jimenez‐Ruiz et al., 1985; Wen & Lee, 2018). With eight rats, we were powered (α = 0.05, β = 0.80) to detect a significant difference of >20% in sigh P di amplitude and the eupneic inter‐breath intervals following C2SH, with mean and variability based on our past study of P di in SD rats (Khurram et al., 2017). Prism 9 was used for all statistical analyses (Graphpad, Carlsbad, CA). We performed paired t‐tests, two‐ or three‐way ANOVAs with a Geiser‐Greenhouse correction (n.b., we did not assume equal variability between data sets) on all outcome measures. All paired data were done between individual rats pre‐surgery (PRE‐C2SH) and post‐C2SH (POST‐C2SH). Similarly, data were paired within behaviors across sham rats pre‐surgery (PRE‐SHAM) and post laminectomy (POST‐SHAM). When appropriate within group differences were evaluated using Bonferroni post hoc tests. When comparing our empirical data generated within this study to the “stereotypical sigh,” we are referring to twice eupneic amplitudes and twice eupneic post‐sigh inter‐breath intervals (Bell et al., 2009; Bell & Haouzi, 2010; Mantilla et al., 2011; Seven et al., 2018). Statistical significance was set at p < 0.05, with p values reported to three significant figures in the results. In the interest or reliability, rigor and robustness of our work, the coefficients of variation of each data set, the relative changes reported (when statistically warranted) and the effect size of each PRE‐C2SH and POST‐C2SH comparison are reported in tabular form (Table 1). All data are reported as the mean ± 95% confidence intervals (CI), unless indicated.
TABLE 1.
Variance and effect sizes between the PRE‐C2SH and POST‐C2SH states across rat means.
| Parameter | Variance (CV%) | Group differences (%; p) | Cohen's d (effect size) |
|---|---|---|---|
| Breaths per minute | PRE: 14.7; POST: 10.7 | NA; p = 0.16 | NA |
| Duty cycle (%) | PRE: 11.8; POST: 13.1 | NA; p = 0.18 | NA |
| P di (cm H2O) of rat means within behavior | Eupnea PRE: 23.6; POST: 30.4 | −42.4%; p = 0.0121 | 2.04 (large) |
| Sigh PRE: 14.1; POST: 33.0 | −51.9%; p = 0.0001 | 3.47 (large) | |
| Post‐sigh PRE: 29.8; POST: 31.9 | −40.9%; p = 0.0291 | 1.63 (large) | |
| P di (% Eupnea) of rat means within behavior | Sigh PRE: 14.5; POST: 14.2 | NA | NA |
| Post‐sigh PRE: 16.2; POST: 6.4 | NA | NA | |
| Breath duration (s), rat means within behavior | Eupnea PRE: 15.5; POST: 12.9 | NA | NA |
| Sigh PRE: 22.6; POST: 16.0 | NA | NA | |
| Post‐sigh PRE: 16.2; POST: 19.2 | NA | NA | |
| Breath duration (% Eupnea), rat means within behavior | Sigh PRE: 25.3; POST: 13.1 | NA | NA |
| Post‐sigh PRE: 11.3; POST: 9.7 | NA | NA | |
| Inter‐breath interval (s), rat means within behavior | Eupnea PRE: 10.7; POST: 14.0 | NA | NA |
| Sigh PRE: 27.6; POST: 30.7 | NA | NA | |
| Post‐sigh PRE: 28.5; POST: 22.9 | NA | NA | |
| Inter‐breath interval (% Eupnea), rat means within behavior | Sigh PRE: 22.0; POST: 21.9 | NA | NA |
| Post‐sigh PRE: 25.7; POST: 19.2 | NA | NA |
3. RESULTS
3.1. Ventilatory parameters following sham or C2SH surgery
The C2SH lesion used in the present study is highly consistent both structurally and functionally (Fogarty et al., 2023; Fuller et al., 2006, 2008; Mantilla, Gransee, et al., 2013; Miyata et al., 1995; Rana et al., 2020; Sieck et al., 2021; Warren et al., 2018). C2SH severed the right lateral and ventral funiculi resulting in the silencing of eupneic DIAm EMG activity on the right ipsilateral side in anesthetized rats. During eupnea, there was no effect of laminectomy or C2SH (F = 2.2; p = 0.16, two‐way ANOVA) on respiratory rate (PRE‐SHAM: 65.8 ± 14.2 breaths per min; POST‐SHAM: 62.5 ± 8.9 breaths per min; PRE‐C2SH: 61.3 ± 7.9 breaths per min; POST‐C2SH: 57.6 ± 5.2 breaths per min; n = 8 per group). There was also no effect of laminectomy or C2SH (F = 2.0; p = 0.18, two‐way ANOVA) on the DIAm duty cycle during eupnea (PRE‐SHAM: 45.8 ± 5.0%; POST‐SHAM: 47.0 ± 10.7%; PRE‐C2SH: 44.5 ± 4.4%; POST‐C2SH: 49.8 ± 5.4%; n = 8 per group).
3.2. P di amplitude decreases following C2SH during eupnea, sigh, and the immediate post‐sigh breath
Amplitudes of P di during eupnea, sigh, and the post‐sigh breath (i.e., DIAm motor behaviors) were assessed at PRE and POST timepoints in SHAM and C2SH rats (Figure 1a). When the mean of each behavior was calculated for each rat, P di amplitude was affected by behavior (F = 353.5; p < 0.0001), type of surgery (SHAM vs. C2SH; F = 6.3; p = 0.0228), and time (PRE‐ vs. POST; F = 24.0; p = 0.0002; n = 8 per group, three‐way ANOVA; Figure 1b). P di amplitude during eupnea was reduced by ~42% in POST‐C2SH (8.3 ± 2.1 cm H2O) compared to PRE‐C2SH (14.4 ± 2.9 cm H2O; p = 0.0121), with no difference observed between PRE‐SHAM (13.5 ± 1.4 cm H2O) and POST‐SHAM (13.0 ± 0.8 cm H2O; p > 0.99; Bonferroni post‐tests; Figure 1b). P di amplitude during sigh was reduced by ~52% in POST (12.9 ± 3.6 cm H2O) compared to PRE (26.8 ± 3.2 cm H2O; p < 0.0001), with no difference observed between PRE‐SHAM (24.1 ± 2.8 cm H2O) and POST‐SHAM (23.3 ± 1.5 cm H2O; p > 0.99; Bonferroni post‐tests; Figure 1b) timepoints. P di amplitude during the post‐sigh breath was reduced by ~41% in POST (8.1 ± 2.2 cm H2O) compared to PRE (13.7 ± 3.4 cm H2O; p = 0.0291), with no difference observed between PRE‐SHAM (12.2 ± 1.0 cm H2O) and POST‐SHAM (11.7 ± 1.6 cm H2O; p > 0.99; Bonferroni post‐tests; Figure 1b) timepoints.
FIGURE 1.
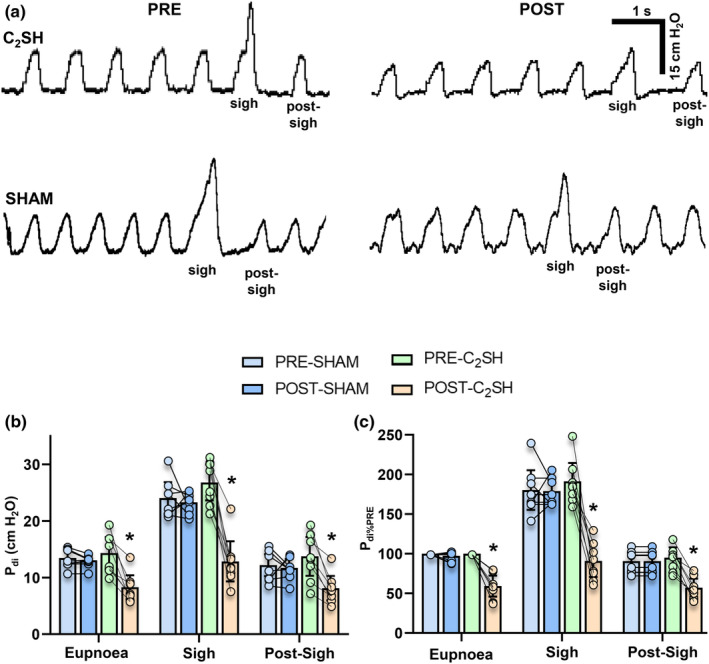
(a) Representative tracing of P di during eupnea, sigh, and the immediate post‐sigh breath in PRE‐SHAM, POST‐SHAM, PRE‐C2SH, and POST‐C2SH conditions. (b) Scatterplot of P di (cm H2O) showing reduction in the POST‐C2SH (orange) compared to the PRE‐C2SH (green) condition during eupnea, sigh, and the post‐sigh breath, with no changes between PRE‐SHAM and POST‐SHAM with sigh (except during POST‐C2SH) having greater P di than all other groups (behavior: p < 0.0001; type of surgery: p = 0.0002; and time: p < 0.0001). (c) Scatterplot of normalized P di (P di%PRE) shows reductions in the POST‐C2SH compared to PRE‐C2SH condition during eupnea, sigh, and the post‐sigh breath, with no changes between PRE‐SHAM and POST‐SHAM (behavior: p < 0.0001; type of surgery: p = 0.0002; and time: p < 0.0001). All tests are paired three‐way ANOVAs with Bonferroni post‐tests, n = 8 for all groups. Each scatter point represents the mean value of each rat, * denotes significant differences within behavior between PRE‐C2SH and POST‐C2SH groups for P di during eupnea (p = 0.012), sigh (p < 0.0001) and the post‐sigh breath (p = 0.029) and for normalized P di%PRE during eupnea (p = 0.011), sigh (p = 0.0001), and the post‐sigh breath (p = 0.010).
Within each rat, when P di amplitude during each motor behavior was expressed as a % of PRE eupnea (P di%PRE), P di%PRE was dependent on behavior (F = 230.5; p < 0.0001), type of surgery (SHAM vs. C2SH; F = 34.7; p = 0.0002), and time (PRE‐ vs. POST; F = 55.9; p < 0.0001; n = 8 per group, three‐way ANOVA; Figure 1c), with P di%PRE reduced POST‐C2SH compare to PRE‐C2SH during eupnea (PRE‐C2SH: 100 ± 0%; POST‐C2SH: 59 ± 13%; p = 0.0108), sigh (PRE‐C2SH: 191 ± 24%; POST‐C2SH: 91 ± 20%; p = 0.0001), and the post‐sigh breath (PRE‐C2SH: 95 ± 13%; POST‐C2SH: 57 ± 12%; p = 0.0096; Bonferroni post‐tests; Figure 1c). There were no differences between PRE‐SHAM and POST‐SHAM in P di%PRE during eupnea (p > 0.99), sigh (p > 0.99), and post‐sigh (p > 0.99; Bonferroni post‐tests; Figure 1c).
Note that the amplitude of P di for PRE‐C2SH sighs was ~double the amplitude of P di for PRE‐C2SH eupnea, whereas POST‐C2SH sigh P di amplitude was no different compared to PRE‐C2SH eupnea P di amplitude (p > 0.99; Figure 1c). Overall, these results show the P di amplitudes of eupnea, sigh, and the immediate post‐sigh breath were reduced in POST‐C2SH rats compared to the PRE‐C2SH condition, with no differences in SHAM time controls.
3.3. Breath durations are unaltered following C2SH during eupnea, sigh, and the immediate post‐sigh breath
When the mean of each behavior was calculated for each rat, breath duration was dependent on behavior (F = 145.1; p < 0.0001), but not type of surgery (SHAM vs. C2SH; F = 0.02; p = 0.787) or time (PRE‐ vs. POST; F = 1.5; p = 0.227; n = 8 per group, three‐way ANOVA; Figure 2a). In all rats, sigh duration was longer compared to eupnea and the post‐sigh breath, regardless of pre‐ or post‐injury timepoint (p < 0.05; Bonferroni post‐tests). However, there was no difference between C2SH and SHAM rats (Figure 2a).
FIGURE 2.
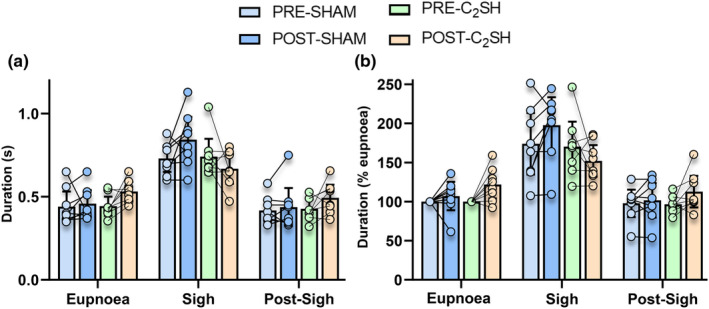
(a) Scatterplot of breath duration (s) showing increased breath duration of sigh compared to eupnea and the immediate post‐sigh breath, regardless of injury (behavior: p < 0.0001; type of surgery: p = 0.787; and time: p = 0.227). (b) Scatterplot of breath durations normalized within rats to PRE‐injury eupnea values shows extended breath durations during sigh compared to eupnea and the post‐sigh breath, regardless of injury (behavior: p < 0.0001; type of surgery: p = 0.469; and time: p = 0.270). All tests are paired three‐way ANOVAs with Bonferroni post‐tests, n = 8 for all groups. Each scatter point represents the mean value of each rat.
When breath durations were expressed as a % of PRE eupnea, breath duration was dependent on behavior (F = 104.4; p < 0.0001) but not type of surgery (SHAM vs. C2SH; F = 0.3; p = 0.469) or time (PRE‐ vs. POST; F = 1.3; p = 0.270; n = 8 per group, three‐way ANOVA; Figure 2b), with sigh having increased durations compared to eupnea and the post‐sigh breath in all rats (p < 0.05, Bonferroni post‐tests; Figure 2b). Overall, these results show the breath duration of eupnea, sigh, and the immediate post‐sigh breath was unchanged with C2SH or sham.
3.4. Inter‐breath intervals are unaltered following C2SH during eupnea, sigh, and the immediate post‐sigh breath
When the mean of each behavior was calculated for each rat, the inter‐breath interval was dependent on behavior (F = 88.9; p < 0.0001), but not type of surgery (SHAM vs. C2SH; F = 0.2; p = 0.659) or time (PRE‐ vs. POST; F = 0.3; p = 0.610; n = 8 per group, three‐way ANOVA; Figure 3a). Inter‐breath intervals of the post‐sigh breath were greater than compared to sigh across all groups (Figure 3a).
FIGURE 3.
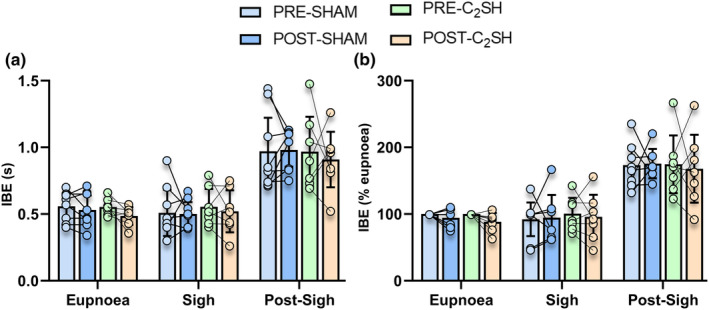
(a) Scatterplot of inter‐breath intervals (s) showing increased inter‐breath intervals (IBE) of the post‐sigh breath compared to all other behaviors, regardless of injury (behavior: p < 0.0001; type of surgery: p = 0.659; and time: p = 0.610). (b) Scatterplot of IBE normalized within rats to PRE eupnea values shows longer inter‐breath intervals in the post‐sigh breath regardless of injury (behavior: p < 0.0001; type of surgery: p = 0.924; and time: p = 0.661). All tests are paired three‐way ANOVAs, n = 8 for all groups. Each scatter point represents the mean value of each rat.
When the inter‐breath interval was expressed as a % of PRE eupnea, inter‐breath interval was dependent on behavior (F = 89.2; p < 0.0001) but not type of surgery (SHAM vs. C2SH; F = 0.002; p = 0.924) or time (PRE‐ vs. POST; F = 0.2; p = 0.661; n = 8 per group, three‐way ANOVA; Figure 3b), with greater post‐sigh inter‐breath interval compared to sigh in all groups (p < 0.05, Bonferroni post‐tests; Figure 3b). Overall, these results show the inter‐breath intervals of eupnea, sigh, and the immediate post‐sigh breath were unchanged with C2SH or sham.
3.5. Sigh variability and comparisons to stereotypical sigh in C2SH rats
As we did not observe a significant effect of time (PRE vs. POST) alone on P di from SHAM rats, we ignored these for the paired analyses of PRE‐ and POST‐C2SH rats. Variability across rats was readily apparent in both the PRE‐C2SH and POST‐C2SH injury timepoints (Table 1). Within rats, we characterized the % of all sighs within a rat that had P di magnitudes of ≥twice eupnea (i.e., the “stereotypical” sigh amplitude) at PRE‐ and POST‐C2SH timepoints (Figure 4a). At the PRE‐C2SH timepoint, the mean % of sighs within a rat that exhibited stereotypical P di amplitudes was ~48%, ~10 times the % of stereotypical amplitudes observed in the POST‐C2SH timepoint (~4%; p = 0.0078, Wilcoxon matched‐pairs signed‐rank test; Figure 4a).
FIGURE 4.
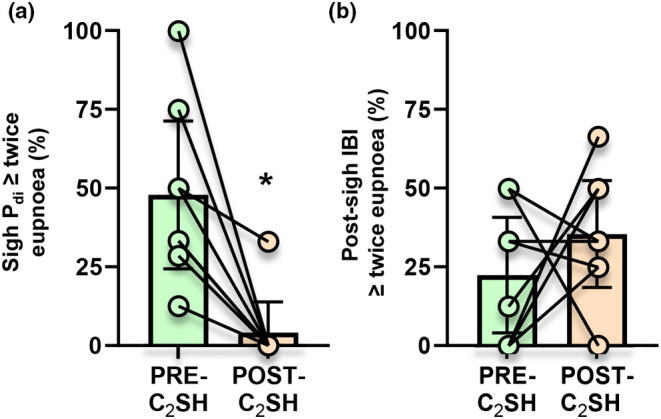
(a) Scatterplot showing the % of all sighs exhibiting “stereotypical” twice eupnea P di shows reduction in the % of sighs with canonical P di amplitudes in POST‐C2SH compared to PRE‐C2SH rats (p = 0.0078, Wilcoxon matched‐pairs signed‐rank test). (b) Scatterplot showing the % of all sighs exhibiting “stereotypical” twice eupnea inter‐breath intervals (i.e., post‐sigh respiratory reset) of the post‐sigh breath shows no change with injury in the % of sighs with canonical post‐sigh respiratory reset in PRE‐ and POST‐C2SH groups (p = 0.373, paired t‐test). Each scatter point represents the mean value of each rat, n = 8 for all groups, * denotes significant differences (p = 0.0078) within behavior between PRE‐ and POST‐C2SH groups.
Within rats, we characterized the % of all sighs within a rat that had post‐sigh inter‐breath intervals of ≥twice eupnea (i.e., the “stereotypical” apneic period) at PRE‐ and POST‐C2SH timepoints (Figure 4b). At the PRE‐C2SH timepoint, the mean % of sighs within a rat that exhibited stereotypical post‐sigh inter‐breath intervals was ~22%, with no significant difference observed in the POST‐C2SH timepoint (~35%; p = 0.373, paired t‐test; Figure 4b).
When all sighs were pooled and considered independent events, the 51% reduction in P di amplitude in the POST‐C2SH (13.4 ± 1.8 cm H2O, n = 28) timepoint was apparent compared to PRE‐C2SH amplitudes (27.6 ± 2.0 cm H2O, n = 36; p < 0.0001, unpaired t‐test; Figure 5a). When all sighs were considered independent events, there was no difference in the post‐sigh inter‐breath interval between PRE‐C2SH (0.891 ± 0.110 s, n = 36) and POST‐C2SH (0.893 ± 0.152 s, n = 28) timepoints (p = 0.979, unpaired t‐test; Figure 5b).
FIGURE 5.

(a) Scatterplot of all individual sigh P di (cm H2O), showing reduced amplitudes in the POST‐C2SH compared to PRE‐C2SH timepoints (p < 0.0001). (b) Scatterplot of all individual inter‐breath intervals (s) between sigh and the post‐sigh breath, showing no difference between PRE‐ and POST‐C2SH timepoints (p = 0.979). In all plots, green symbols represent “stereotypical” sighs of >twice eupneic P di and post‐sigh respiratory reset, purple symbols represent sighs satisfying “stereotypical” P di criteria, yellow symbols represent sighs satisfying inter‐breath interval criteria and red symbols represent sighs where neither criterion was satisfied. All tests are unpaired t‐tests, * denotes p < 0.0001, n = 36 (PRE‐C2SH) and n = 24 (POST‐C2SH).
The mean duration, inter‐breath interval, and amplitudes of the sigh and post‐sigh breath were compared to the “stereotypical” sigh at PRE‐ and POST‐C2SH timepoints (Figure 5; Table 2), with the reduction in POST‐C2SH sigh P di amplitude readily observed. Even in the PRE‐C2SH condition, there was a marked departure of the sigh and post‐sigh breath from the “stereotypical” sigh. For sigh amplitudes, only 53% (19/36) from the PRE‐C2SH and 4% (1/28) from the POST‐C2SH timepoints satisfied “stereotypical” criteria, with significant differences between PRE‐ and POST‐C2SH timepoints (p < 0.0001, Fischer's exact test; Figure 5; Table 2). Likewise, for post‐sigh inter‐breath intervals, only 22% (8/36) from the PRE‐C2SH and 32% (9/28) from the POST‐C2SH timepoints satisfied “stereotypical” criteria, with no differences between timepoints (p = 0.406, Fischer's exact test; Figure 5; Table 2). Incredibly, only 17% of sighs from the PRE‐C2SH timepoint and not a single sigh from the POST‐C2SH timepoint satisfied both criteria, with significant differences between PRE‐ and POST‐C2SH timepoints (p = 0.031, Fischer's exact test; Figure 5; Table 2). A rather large portion of sighs satisfied neither criterion 25% at PRE‐C2SH (9/36) and 36% at POST‐C2SH (10/28) timepoints, with a significant increase between PRE‐ and POST‐C2SH timepoints (p = 0.001, Fischer's exact test; Figure 5; Table 2).
TABLE 2.
Number and percentages of sighs exhibiting stereotypical criteria in PRE‐C2SH and POST‐C2SH conditions.
| Group | P di ≥ twice eupnea | Post‐sigh inter‐breath interval ≥ twice eupnea | Both criteria satisfied | Neither criteria satisfied | Total sighs |
|---|---|---|---|---|---|
| PRE | 19 (53%) | 8 (22%) | 6 (17%) | 9 (25%) | 36 |
| POST | 1 (4%) | 9 (32%) | 0 (0%) | 10 (36%) | 28 |
Note: Italicized POST values are significantly different from PRE, Fischer's exact test, p < 0.05.
4. DISCUSSION
This study was the first to characterize the P di generated during sighs and the respiratory reset PRE‐ and POST‐C2SH. We present four major findings: (i) P di amplitudes, but not breath durations or inter‐breath intervals were altered POST‐C2SH during eupnea, sigh, and the immediate post‐sigh breath; (ii) the amplitudes of P di during sighs were highly variable, with only ~53% of sigh P di amplitudes two times greater than P di during eupnea PRE‐C2SH, while only ~4% of POST‐C2SH sighs exhibited P di amplitudes that were > twice eupnea; (iii) while post‐sigh inter‐breath intervals were longer compared to eupnea, only ~22% of sighs PRE‐C2SH and ~32% of sighs POST‐C2SH displayed inter‐breath intervals that were twice eupnea; and (iv) overall, PRE‐C2SH only 17% of all sighs satisfied both “stereotypical” criteria (i.e., amplitudes and inter‐breath intervals greater than twice eupnea), while POST‐C2SH “stereotypical” sighs were absent. These findings have important consequences for the study of respiratory function more generally, and for the effects of C2SH and DIAm impairments more specifically.
Sighs have been reported in a myriad of reduced and intact experimental preparations and in a variety of species such as humans (Vlemincx et al., 2010; Wuyts et al., 2011), cats (Cherniack et al., 1981; Orem & Trotter, 1993; van Lunteren et al., 1988), dogs (Katagiri et al., 1998; van Lunteren et al., 1983), rats (Ajayi & Mills, 2017; Bell et al., 2009, 2011; Bell & Haouzi, 2010; Fogarty et al., 2023; Fuller et al., 2006, 2008; Golder et al., 2005; Janssen et al., 2000; Jimenez‐Ruiz et al., 1985; Khurram et al., 2017, 2018; Seven et al., 2018), and mice (Li et al., 2016; Pareja‐Cajiao et al., 2021; Voituron et al., 2010). To date, the focus has been primarily on the pulmonary and EMG characteristics of sigh and the post‐sigh breath, with information on rat P di during sigh influenced by the “filtering” effect of inclusion criteria. There is also a total absence of quantitative P di information on the post‐sigh breath and whether P di characteristics of sighs are in accordance with plethysmographic results, where duration and tidal volumes are reduced compared to eupnea in cats (Cherniack et al., 1981; Reynolds Jr. & Hilgeson, 1965), but not dogs (van Lunteren et al., 1983). Previously, sigh P di in anesthetized rats was reported to be either less than that generated during airway occlusion in rats (Gill et al., 2015; Khurram et al., 2017, 2018; Seven et al., 2018) or greater than occlusion P di (Mantilla et al., 2010). Sigh P di in anesthetized cats was also reported to be less than occlusion P di (Reynolds Jr. & Hilgeson, 1965). In the present study we evaluated augmented breaths with characteristic waveforms without iexclusion criteria of amplitudes or the duration of the post‐sigh respiratory reset. We found the mean sigh P di to be ~27 cm H2O compared to 14.4 cm H2O during eupnea. The mean sigh P di amplitude observed in the present study was moderately higher than that previously reported in Sprague‐Dawley rats (Gill et al., 2015; Khurram et al., 2017). The P di generated during airway occlusion in anesthetized Sprague‐Dawley rats of the same sex/age and weight range is ~30 cm H2O across studies (Fogarty et al., 2023; Gill et al., 2015; Khurram et al., 2017; Seven et al., 2018). On balance, it appears that P di amplitude sighs are generally equivalent to those generated during airway occlusion.
The post‐sigh inter‐breath intervals were longer compared to inter‐breath intervals for eupnea both PRE‐ and POST‐C2SH. In rats, it appears that that the post‐sigh respiratory reset is almost immediate, compared to cats, where 5–6 post‐sigh breaths remain depressed compared to normal eupnea (Cherniack et al., 1981). An important limitation of the present study is that P di measurements were performed while animals were anesthetized, which largely abolishes the post‐inspiratory phase of breathing. It remains to be seen in rats whether the duration of the post‐sigh respiratory reset is influenced by post‐inspiration DIAm activity.
In the present study, the amplitude of P di during sighs was reduced POST‐C2SH. Previously in rats, we reported that C2SH reduced the P di generated during eupnea, hypoxia/hypercapnia and occlusion (Fogarty et al., 2023). This remarkable effect of C2SH on ventilatory P di is consistent with past reports of reduced tidal volume during eupnea and augmented breaths in unanesthetized rats (Fuller et al., 2006, 2008). Importantly, with C2SH, reduced P di during ventilatory behaviors may not be entirely dependent on impaired DIAm function. Reduced stiffening of the thoracic cage by the chest wall musculature (intercostals) during inspiration (Beth Zimmer et al., 2015; Denton & McKinlay, 2009) may impair P di due to increased thoracic compliance. In other studies evaluating the impact of C2SH on sighs, the amplitude of DIAm EMG (Bezdudnaya et al., 2018) and tidal volume was reduced (Dougherty et al., 2012; Fuller et al., 2008; Golder et al., 2005). It is important to note that different behaviors of the DIAm are affected by C2SH differently depending on the method of assessment. For example, the amplitude of DIAm EMG during airway occlusion has been reported to be unchanged (Martinez‐Galvez et al., 2016) or reduced (Fogarty et al., 2023; Warren et al., 2018) following C2SH, depending on the normalization procedure. In the present study, we have focused on the immediate effects of C2SH; however, there is a large body of literature chronicling spontaneous recovery of the injured side in the weeks following the lesion (Brown et al., 2022; Fogarty et al., 2023; Fuller et al., 2006, 2008; Goshgarian et al., 1991; Gransee et al., 2015, 2017; Hernandez‐Torres et al., 2017; Mantilla, Gransee, et al., 2013; Mantilla, Greising, et al., 2013; Martinez‐Galvez et al., 2016; Miyata et al., 1995; Moreno et al., 1992; Sieck et al., 2021; Singh et al., 2012). In rats, we have consistently observed ~30% spontaneous recovery in DIAm EMG during eupnea between 7‐ and 14‐days POST‐C2SH (Brown et al., 2022; Fogarty et al., 2023; Gransee et al., 2015; Hernandez‐Torres et al., 2017; Mantilla, Gransee, et al., 2013; Martinez‐Galvez et al., 2016; Sieck et al., 2021). Notably, the C2SH lesion in rat spares respiratory premotor inputs from the contralateral side and local interneurons (Fogarty & Sieck, 2020), which may be provide the substrate for de novo synaptogenesis and recovery (Gransee et al., 2017; Hadley et al., 1999; Mantilla et al., 2012; Porter, 1895; Streeter et al., 2017, 2020; Sunshine et al., 2020; Zhou & Goshgarian, 1999). Enhancement of this phenomena has been shown to occur by promoting neurotrophic, neuromodulatory, and synaptic formations, which can increase the level of eupneic recovery to >80% (Fogarty et al., 2023; Fuller et al., 2003; Gransee et al., 2013, 2015; Hernandez‐Torres et al., 2017; Mantilla, Gransee, et al., 2013; Martinez‐Galvez et al., 2016; Sieck et al., 2021; Wollman et al., 2020; Zhou & Goshgarian, 1999). However, few of these (Fogarty et al., 2023) have evaluated recovery across the full gamut of ventilatory and non‐ventilatory behaviors.
It is likely that spontaneous sighs utilize or interact with the canonical brainstem respiratory centers (Housley et al., 1970; Ramirez, 2014; Ramirez et al., 2022; Soltysik & Jelen, 2005). The results of the present study indicate that the generation of P di during sighs is sensitive to C2SH removal of ipsilateral descending excitatory input to PhMNs. The P di generated POST‐C2SH are in stark contrast to the P di generated following unilateral phrenicotomy, where P di generated during eupnea and hypoxia/hypercapnia are preserved with an increase in contralateral DIAm EMG, while the P di generated during sigh and airway occlusion are reduced, with no compensatory increase in DIAm EMG on the uninjured side (Gill et al., 2015; Khurram et al., 2017). Following C4 unilateral contusion, the P di generated during airway occlusion is reduced (Khurram et al., 2019), with no effects on DIAm EMG (Rana et al., 2017). Differences in the effects of C2SH and unilateral C4 contusion are likely related to the differential disruption of descending excitatory inputs to PhMN and the extent of contusion induced PhMN death, for example, with C2SH the extent of sparing of ipsilateral versus contralateral inputs to PhMNs is exceedingly important (Fogarty & Sieck, 2020; Rana et al., 2020; Tai & Goshgarian, 1996). With C4 contusion, frank PhMN death and DIAm denervation occurs on the injured side (Alvarez‐Argote et al., 2016; Rana et al., 2017) together with disruption of synaptic inputs to some PhMNs below the lesion.
Overall, this study shows that, in the absence of winnowing out non‐“stereotypical” sighs, the sigh is quite variable within and across rats and is highly sensitive to C2SH. Likewise the P di of the post‐sigh breath is also affected by C2SH. In the spinal cord injury population, maintenance of cardiorespiratory health is essential (Fogarty & Sieck, 2020; Raab et al., 2021; Randelman et al., 2021), with sighs highly important in maintaining lung ventilatory function (Housley et al., 1970; Katagiri et al., 1998; Lieske et al., 2000; Ramirez et al., 2022; Riede et al., 2020; Wuyts et al., 2011). Our results of reduced sigh P di amplitude likely impair the effectiveness of this behavior to stave of lung atelectasis. The positive effects of breathing exercises (e.g., breath‐stacking) post cervical spinal cord injury may be one way to accomplish what spontaneous sigh cannot in this population (Jeong & Yoo, 2015), although this probably is insufficient to mitigate any risks of pneumonias and airway infections due to impaired coughing (Berlowitz et al., 2016).
Our study also has important consequences for respiratory neurobiology in general, particularly with using sighs as an exemplar for the function of the entire respiratory neuromotor system. We caution against experimental approaches where atypical sighs are arbitrarily excluded and then subsequently used as a normalization, as this may artificially reduce variability, essentially doing nothing other than normalizing to twice eupnea. This is particularly important for conditions where sighs and the post‐sigh breaths are particularly vulnerable (e.g., Rett syndrome) (Fogarty, 2023; Huang et al., 2016; Robinson et al., 2012; Voituron et al., 2010). Here, we show that using an inclusive shape‐based, not amplitude‐based sigh sampling, we are able to capture the true variability within the behavior while also being sensitive enough to see an effect of injury. In the future as evaluations of sigh become less phenomenological, with more attention paid to their genesis within neural circuits, it will be important to pay attention to both the sigh itself and the immediate post‐sigh breath, as various species exhibit different P di and temporal relationships to eupnea and maximal DIAm behaviors.
FUNDING INFORMATION
This work was supported by National Institutes of Health grants HL146114 (GCS) and HL166204 (MJF).
CONFLICT OF INTEREST STATEMENT
None of the authors has any conflicts of interest, real nor perceived, to disclose.
ETHICS STATEMENT
All procedures were performed in accordance with the American Physiological Society's Animal Care Guidelines and the National Institutes of Health (NIH) guide for use and care of laboratory animals. These procedures were approved by the Institutional Animal Care and Use Committee (IACUC) at Mayo Clinic (A00003105‐17‐R20).
ACKNOWLEDGMENTS
We would like to thank Rebecca Macken for her assistance in this project.
Fogarty, M. J. , Zhan, W.‐Z. , Mantilla, C. B. , & Sieck, G. C. (2024). Cervical spinal cord hemisection impacts sigh and the respiratory reset in male rats. Physiological Reports, 12, e15973. 10.14814/phy2.15973
Contributor Information
Matthew J. Fogarty, Email: fogarty.matthew@mayo.edu.
Gary C. Sieck, Email: sieck.gary@mayo.edu.
REFERENCES
- Ajayi, I. E. , & Mills, P. C. (2017). Effects of the hippocampus on the motor expression of augmented breaths. PLoS One, 12, e0183619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alvarez‐Argote, S. , Gransee, H. M. , Mora, J. C. , Stowe, J. M. , Jorgenson, A. J. , Sieck, G. C. , & Mantilla, C. B. (2016). The impact of midcervical contusion injury on diaphragm muscle function. Journal of Neurotrauma, 33, 500–509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bell, H. J. , Azubike, E. , & Haouzi, P. (2011). The “other” respiratory effect of opioids: Suppression of spontaneous augmented (“sigh”) breaths. Journal of Applied Physiology, 111, 1296–1303. [DOI] [PubMed] [Google Scholar]
- Bell, H. J. , Ferguson, C. , Kehoe, V. , & Haouzi, P. (2009). Hypocapnia increases the prevalence of hypoxia‐induced augmented breaths. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology, 296, R334–R344. [DOI] [PubMed] [Google Scholar]
- Bell, H. J. , & Haouzi, P. (2010). The hypoxia‐induced facilitation of augmented breaths is suppressed by the common effect of carbonic anhydrase inhibition. Respiratory Physiology & Neurobiology, 171, 201–211. [DOI] [PubMed] [Google Scholar]
- Berlowitz, D. J. , Wadsworth, B. , & Ross, J. (2016). Respiratory problems and management in people with spinal cord injury. Breathe (Sheffield, England), 12, 328–340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beth Zimmer, M. , Grant, J. S. , Ayar, A. E. , & Goshgarian, H. G. (2015). Ipsilateral inspiratory intercostal muscle activity after C2 spinal cord hemisection in rats. The Journal of Spinal Cord Medicine, 38, 224–230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bezdudnaya, T. , Hormigo, K. M. , Marchenko, V. , & Lane, M. A. (2018). Spontaneous respiratory plasticity following unilateral high cervical spinal cord injury in behaving rats. Experimental Neurology, 305, 56–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown, A. D. , Fogarty, M. J. , Davis, L. A. , Dasgupta, D. , Mantilla, C. B. , & Sieck, G. C. (2022). Mitochondrial adaptations to inactivity in diaphragm muscle fibers. Journal of Applied Physiology, 133, 191–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burns, D. P. , Murphy, K. H. , Lucking, E. F. , & O'Halloran, K. D. (2019). Inspiratory pressure‐generating capacity is preserved during ventilatory and non‐ventilatory behaviours in young dystrophic mdx mice despite profound diaphragm muscle weakness. The Journal of Physiology, 597, 831–848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cherniack, N. S. , von Euler, C. , Glogowska, M. , & Homma, I. (1981). Characteristics and rate of occurrence of spontaneous and provoked augmented breaths. Acta Physiologica Scandinavica, 111, 349–360. [DOI] [PubMed] [Google Scholar]
- Denton, M. , & McKinlay, J. (2009). Cervical cord injury and critical care. Continuing Education in Anaesthesia Critical Care & Pain, 9, 82–86. [Google Scholar]
- Dougherty, B. J. , Lee, K. Z. , Lane, M. A. , Reier, P. J. , & Fuller, D. D. (2012). Contribution of the spontaneous crossed‐phrenic phenomenon to inspiratory tidal volume in spontaneously breathing rats. Journal of Applied Physiology, 112, 96–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fogarty, M. J. (2023). Inhibitory synaptic influences on developmental motor disorders. International Journal of Molecular Sciences, 24, 6962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fogarty, M. J. , Brandenburg, J. E. , Zhan, W. Z. , & Sieck, G. C. (2022). Diaphragm muscle function in a mouse model of early onset spasticity. Journal of Applied Physiology, 133, 60–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fogarty, M. J. , & Sieck, G. C. (2019). Evolution and functional differentiation of the diaphragm muscle of mammals. Comprehensive Physiology, 9, 715–766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fogarty, M. J. , & Sieck, G. C. (2020). Spinal cord injury and diaphragm neuromotor control. Expert Review of Respiratory Medicine, 14, 453–464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fogarty, M. J. , Zhan, W.‐Z. , Simmon, V. F. , Vanderklish, P. W. , Sarraf, S. T. , & Sieck, G. C. (2023). Novel regenerative drug, SPG302 promotes functional recovery of diaphragm muscle activity after cervical spinal cord injury. The Journal of Physiology, 601, 2513–2532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuller, D. D. , Doperalski, N. J. , Dougherty, B. J. , Sandhu, M. S. , Bolser, D. C. , & Reier, P. J. (2008). Modest spontaneous recovery of ventilation following chronic high cervical hemisection in rats. Experimental Neurology, 211, 97–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuller, D. D. , Golder, F. J. , Olson, E. B., Jr. , & Mitchell, G. S. (2006). Recovery of phrenic activity and ventilation after cervical spinal hemisection in rats. Journal of Applied Physiology, 100, 800–806. [DOI] [PubMed] [Google Scholar]
- Fuller, D. D. , Johnson, S. M. , Olson, E. B., Jr. , & Mitchell, G. S. (2003). Synaptic pathways to phrenic motoneurons are enhanced by chronic intermittent hypoxia after cervical spinal cord injury. The Journal of Neuroscience, 23, 2993–3000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gill, L. C. , Mantilla, C. B. , & Sieck, G. C. (2015). Impact of unilateral denervation on transdiaphragmatic pressure. Respiratory Physiology & Neurobiology, 210, 14–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Golder, F. J. , Davenport, P. W. , Johnson, R. D. , Reier, P. J. , & Bolser, D. C. (2005). Augmented breath phase volume and timing relationships in the anesthetized rat. Neuroscience Letters, 373, 89–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goshgarian, H. G. , Ellenberger, H. H. , & Feldman, J. L. (1991). Decussation of bulbospinal respiratory axons at the level of the phrenic nuclei: A possible substrate for the crossed‐phrenic phenomenon. Experimental Neurology, 111, 135–139. [DOI] [PubMed] [Google Scholar]
- Gransee, H. M. , Gonzalez Porras, M. A. , Zhan, W. Z. , Sieck, G. C. , & Mantilla, C. B. (2017). Motoneuron glutamatergic receptor expression following recovery from cervical spinal hemisection. The Journal of Comparative Neurology, 525, 1192–1205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gransee, H. M. , Zhan, W. Z. , Sieck, G. C. , & Mantilla, C. B. (2013). Targeted delivery of TrkB receptor to phrenic motoneurons enhances functional recovery of rhythmic phrenic activity after cervical spinal hemisection. PLoS One, 8, e64755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gransee, H. M. , Zhan, W. Z. , Sieck, G. C. , & Mantilla, C. B. (2015). Localized delivery of brain‐derived neurotrophic factor‐expressing mesenchymal stem cells enhances functional recovery following cervical spinal cord injury. Journal of Neurotrauma, 32, 185–193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadley, S. D. , Walker, P. D. , & Goshgarian, H. G. (1999). Effects of serotonin inhibition on neuronal and astrocyte plasticity in the phrenic nucleus 4 h following C2 spinal cord hemisection. Experimental Neurology, 160, 433–445. [DOI] [PubMed] [Google Scholar]
- Hernandez‐Torres, V. , Gransee, H. M. , Mantilla, C. B. , Wang, Y. , Zhan, W. Z. , & Sieck, G. C. (2017). BDNF effects on functional recovery across motor behaviors after cervical spinal cord injury. Journal of Neurophysiology, 117, 537–544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Housley, E. , Louzada, N. , & Becklake, M. R. (1970). To sigh or not to sigh. The American Review of Respiratory Disease, 101, 611–614. [DOI] [PubMed] [Google Scholar]
- Huang, T. W. , Kochukov, M. Y. , Ward, C. S. , Merritt, J. , Thomas, K. , Nguyen, T. , Arenkiel, B. R. , & Neul, J. L. (2016). Progressive changes in a distributed neural circuit underlie breathing abnormalities in mice lacking MeCP2. The Journal of Neuroscience, 36, 5572–5586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janssen, P. L. , Williams, J. S. , & Fregosi, R. F. (2000). Consequences of periodic augmented breaths on tongue muscle activities in hypoxic rats. Journal of Applied Physiology, 88, 1915–1923. [DOI] [PubMed] [Google Scholar]
- Jensen, V. N. , Seedle, K. , Turner, S. M. , Lorenz, J. N. , & Crone, S. A. (2019). V2a neurons constrain extradiaphragmatic respiratory muscle activity at rest. eNeuro, 6, ENURO.0492‐18.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeong, J. H. , & Yoo, W. G. (2015). Effects of air stacking on pulmonary function and peak cough flow in patients with cervical spinal cord injury. Journal of Physical Therapy Science, 27, 1951–1952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jimenez‐Ruiz, F. , Khurram, O. U. , Zhan, W. Z. , Gransee, H. M. , Sieck, G. C. , & Mantilla, C. B. (1985). Diaphragm muscle activity across respiratory motor behaviors in awake and lightly anesthetized rats. Journal of Applied Physiology, 2018, 915–922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katagiri, H. , Katagiri, M. , Kieser, T. M. , & Easton, P. A. (1998). Diaphragm function during sighs in awake dogs after laparotomy. American Journal of Respiratory and Critical Care Medicine, 157, 1085–1092. [DOI] [PubMed] [Google Scholar]
- Khurram, O. U. , Fogarty, M. J. , Rana, S. , Vang, P. , Sieck, G. C. , & Mantilla, C. B. (2019). Diaphragm muscle function following midcervical contusion injury in rats. Journal of Applied Physiology, 126, 221–230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khurram, O. U. , Fogarty, M. J. , Sarrafian, T. L. , Bhatt, A. , Mantilla, C. B. , & Sieck, G. C. (2018). Impact of aging on diaphragm muscle function in male and female Fischer 344 rats. Physiological Reports, 6, e13786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khurram, O. U. , Sieck, G. C. , & Mantilla, C. B. (2017). Compensatory effects following unilateral diaphragm paralysis. Respiratory Physiology & Neurobiology, 246, 39–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, P. , Janczewski, W. A. , Yackle, K. , Kam, K. , Pagliardini, S. , Krasnow, M. A. , & Feldman, J. L. (2016). The peptidergic control circuit for sighing. Nature, 530, 293–297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lieske, S. P. , Thoby‐Brisson, M. , Telgkamp, P. , & Ramirez, J. M. (2000). Reconfiguration of the neural network controlling multiple breathing patterns: Eupnea, sighs and gasps. Nature Neuroscience, 3, 600–607. [DOI] [PubMed] [Google Scholar]
- Mantilla, C. B. , Bailey, J. P. , Zhan, W. Z. , & Sieck, G. C. (2012). Phrenic motoneuron expression of serotonergic and glutamatergic receptors following upper cervical spinal cord injury. Experimental Neurology, 234, 191–199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mantilla, C. B. , Gransee, H. M. , Zhan, W. Z. , & Sieck, G. C. (2013). Motoneuron BDNF/TrkB signaling enhances functional recovery after cervical spinal cord injury. Experimental Neurology, 247C, 101–109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mantilla, C. B. , Greising, S. M. , Zhan, W. Z. , Seven, Y. B. , & Sieck, G. C. (2013). Prolonged C2 spinal hemisection‐induced inactivity reduces diaphragm muscle specific force with modest, selective atrophy of type IIx and/or IIb fibers. Journal of Applied Physiology, 114, 380–386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mantilla, C. B. , Seven, Y. B. , Hurtado‐Palomino, J. N. , Zhan, W. Z. , & Sieck, G. C. (2011). Chronic assessment of diaphragm muscle EMG activity across motor behaviors. Respiratory Physiology & Neurobiology, 177, 176–182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mantilla, C. B. , Seven, Y. B. , Zhan, W. Z. , & Sieck, G. C. (2010). Diaphragm motor unit recruitment in rats. Respiratory Physiology & Neurobiology, 173, 101–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez‐Galvez, G. , Zambrano, J. M. , Diaz Soto, J. C. , Zhan, W. Z. , Gransee, H. M. , Sieck, G. C. , & Mantilla, C. B. (2016). TrkB gene therapy by adeno‐associated virus enhances recovery after cervical spinal cord injury. Experimental Neurology, 276, 31–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyata, H. , Zhan, W. Z. , Prakash, Y. S. , & Sieck, G. C. (1995). Myoneural interactions affect diaphragm muscle adaptations to inactivity. Journal of Applied Physiology, 79, 1640–1649. [DOI] [PubMed] [Google Scholar]
- Moreno, D. E. , Yu, X. J. , & Goshgarian, H. G. (1992). Identification of the axon pathways which mediate functional recovery of a paralyzed hemidiaphragm following spinal cord hemisection in the adult rat. Experimental Neurology, 116, 219–228. [DOI] [PubMed] [Google Scholar]
- Orem, J. , & Trotter, R. H. (1993). Medullary respiratory neuronal activity during augmented breaths in intact unanesthetized cats. Journal of Applied Physiology, 74, 761–769. [DOI] [PubMed] [Google Scholar]
- Pareja‐Cajiao, M. , Gransee, H. M. , Sieck, G. C. , & Mantilla, C. B. (2021). TrkB signaling contributes to transdiaphragmatic pressure generation in aged mice. Journal of Neurophysiology, 125, 1157–1163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porter, J. (1895). The path of the respiratory impulse from the bulb to the phrenic nuclei. The Journal of Physiology (London), 17, 455–485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raab, A. M. , Mueller, G. , Elsig, S. , Gandevia, S. C. , Zwahlen, M. , Hopman, M. T. E. , & Hilfiker, R. (2021). Systematic review of incidence studies of pneumonia in persons with spinal cord injury. Journal of Clinical Medicine, 11, 211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramirez, J. M. (2014). The integrative role of the sigh in psychology, physiology, pathology, and neurobiology. Progress in Brain Research, 209, 91–129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramirez, J. M. , Vlemincx, E. , Baertsch, N. A. , & Severs, L. J. (2022). The sigh and related behaviors. Handbook of Clinical Neurology, 188, 357–372. [DOI] [PubMed] [Google Scholar]
- Rana, S. , Sieck, G. C. , & Mantilla, C. B. (2017). Diaphragm electromyographic activity following unilateral midcervical contusion injury in rats. Journal of Neurophysiology, 117, 545–555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rana, S. , Zhan, W. Z. , Mantilla, C. B. , & Sieck, G. C. (2020). Disproportionate loss of excitatory inputs to smaller phrenic motor neurons following cervical spinal hemisection. The Journal of Physiology, 598, 4693–4711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Randelman, M. , Zholudeva, L. V. , Vinit, S. , & Lane, M. A. (2021). Respiratory training and plasticity after cervical spinal cord injury. Frontiers in Cellular Neuroscience, 15, 700821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reynolds, L. B., Jr. , & Hilgeson, M. D. (1965). Increase in breathing frequency following the reflex deep breath in anesthetized cats. Journal of Applied Physiology, 20, 491–495. [DOI] [PubMed] [Google Scholar]
- Riede, T. , Schaefer, C. , & Stein, A. (2020). Role of deep breaths in ultrasonic vocal production of Sprague‐Dawley rats. Journal of Neurophysiology, 123, 966–979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson, L. , Guy, J. , McKay, L. , Brockett, E. , Spike, R. C. , Selfridge, J. , De Sousa, D. , Merusi, C. , Riedel, G. , Bird, A. , & Cobb, S. R. (2012). Morphological and functional reversal of phenotypes in a mouse model of Rett syndrome. Brain, 135, 2699–2710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seven, Y. B. , Mantilla, C. B. , & Sieck, G. C. (2014). Recruitment of rat diaphragm motor units across motor behaviors with different levels of diaphragm activation. Journal of Applied Physiology, 117, 1308–1316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seven, Y. B. , Nichols, N. L. , Kelly, M. N. , Hobson, O. R. , Satriotomo, I. , & Mitchell, G. S. (2018). Compensatory plasticity in diaphragm and intercostal muscle utilization in a rat model of ALS. Experimental Neurology, 299, 148–156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Severs, L. J. , Vlemincx, E. , & Ramirez, J. M. (2022). The psychophysiology of the sigh: I: The sigh from the physiological perspective. Biological Psychology, 170, 108313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheikhbahaei, S. , Gourine, A. V. , & Smith, J. C. (2017). Respiratory rhythm irregularity after carotid body denervation in rats. Respiratory Physiology & Neurobiology, 246, 92–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sieck, G. C. (1991). Neural control of the inspiratory pump. NIPS, 6, 260–264. [Google Scholar]
- Sieck, G. C. , & Fournier, M. (1989). Diaphragm motor unit recruitment during ventilatory and nonventilatory behaviors. Journal of Applied Physiology, 66, 2539–2545. [DOI] [PubMed] [Google Scholar]
- Sieck, G. C. , Gransee, H. M. , Zhan, W. Z. , & Mantilla, C. B. (2021). Acute intrathecal BDNF enhances functional recovery after cervical spinal cord injury in rats. Journal of Neurophysiology, 125, 2158–2165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh, L. P. , Devi, T. S. , & Nantwi, K. D. (2012). Theophylline regulates inflammatory and neurotrophic factor signals in functional recovery after C2‐hemisection in adult rats. Experimental Neurology, 238, 79–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soltysik, S. , & Jelen, P. (2005). In rats, sighs correlate with relief. Physiology and Behavior, 85, 598–602. [DOI] [PubMed] [Google Scholar]
- Streeter, K. A. , Sunshine, M. D. , Patel, S. , Gonzalez‐Rothi, E. J. , Reier, P. J. , Baekey, D. M. , & Fuller, D. D. (2017). Intermittent hypoxia enhances functional connectivity of midcervical spinal interneurons. The Journal of Neuroscience, 37, 8349–8362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Streeter, K. A. , Sunshine, M. D. , Patel, S. R. , Gonzalez‐Rothi, E. J. , Reier, P. J. , Baekey, D. M. , & Fuller, D. D. (2020). Mid‐cervical interneuron networks following high cervical spinal cord injury. Respiratory Physiology & Neurobiology, 271, 103305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sunshine, M. D. , Sutor, T. W. , Fox, E. J. , & Fuller, D. D. (2020). Targeted activation of spinal respiratory neural circuits. Experimental Neurology, 328, 113256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tai, Q. , & Goshgarian, H. G. (1996). Ultrastructural quantitative analysis of glutamatergic and GABAergic synaptic terminals in the phrenic nucleus after spinal cord injury. The Journal of Comparative Neurology, 372, 343–355. [DOI] [PubMed] [Google Scholar]
- van Lunteren, E. , Prabhakar, N. R. , Cherniack, N. S. , Haxhiu, M. A. , & Dick, T. E. (1988). Inhibition of expiratory muscle EMG and motor unit activity during augmented breaths in cats. Respiratory Physiology & Neurobiology, 72, 303–314. [DOI] [PubMed] [Google Scholar]
- van Lunteren, E. , van de Graaff, W. B. , Parker, D. M. , Strohl, K. P. , Mitra, J. , Salamone, J. , & Cherniack, N. S. (1983). Activity of upper airway muscles during augmented breaths. Respiratory Physiology & Neurobiology, 53, 87–98. [DOI] [PubMed] [Google Scholar]
- Vlemincx, E. , Severs, L. , & Ramirez, J. M. (2022). The psychophysiology of the sigh: II: The sigh from the psychological perspective. Biological Psychology, 173, 108386. [DOI] [PubMed] [Google Scholar]
- Vlemincx, E. , van Diest, I. , Lehrer, P. M. , Aubert, A. E. , & van den Bergh, O. (2010). Respiratory variability preceding and following sighs: a resetter hypothesis. Biological Psychology, 84, 82–87. [DOI] [PubMed] [Google Scholar]
- Voituron, N. , Zanella, S. , Menuet, C. , Lajard, A. M. , Dutschmann, M. , & Hilaire, G. (2010). Early abnormalities of post‐sigh breathing in a mouse model of Rett syndrome. Respiratory Physiology & Neurobiology, 170, 173–182. [DOI] [PubMed] [Google Scholar]
- Warren, P. M. , Steiger, S. C. , Dick, T. E. , MacFarlane, P. M. , Alilain, W. J. , & Silver, J. (2018). Rapid and robust restoration of breathing long after spinal cord injury. Nature Communications, 9, 4843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen, M. H. , & Lee, K. Z. (2018). Diaphragm and intercostal muscle activity after mid‐cervical spinal cord contusion in the rat. Journal of Neurotrauma, 35, 533–547. [DOI] [PubMed] [Google Scholar]
- Wollman, L. B. , Streeter, K. A. , Fusco, A. F. , Gonzalez‐Rothi, E. J. , Sandhu, M. S. , Greer, J. J. , & Fuller, D. D. (2020). Ampakines stimulate phrenic motor output after cervical spinal cord injury. Experimental Neurology, 334, 113465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wuyts, R. , Vlemincx, E. , Bogaerts, K. , van Diest, I. , & van den Bergh, O. (2011). Sigh rate and respiratory variability during normal breathing and the role of negative affectivity. International Journal of Psychophysiology, 82, 175–179. [DOI] [PubMed] [Google Scholar]
- Zhou, S. Y. , & Goshgarian, H. G. (1999). Effects of serotonin on crossed phrenic nerve activity in cervical spinal cord hemisected rats. Experimental Neurology, 160, 446–453. [DOI] [PubMed] [Google Scholar]


