Abstract
Objectives
Inhaled corticosteroids (ICS) are widely used in patients with chronic obstructive pulmonary disease (COPD). However, ICS are associated with an increased risk of adverse effects.
We aimed to determine whether an association between a lower respiratory tract culture with Stenotrophomonas maltophilia and increasing ICS dosing in patients with COPD exists.
Design
An observational cohort study of outpatients with COPD in Denmark between 2010 and 2018.
ICS exposure was categorised into four groups based on average daily consumption 1 year prior to inclusion: no use, low ICS dose (≤400 µg), moderate ICS dose (400–800 µg) and high ICS dose (>800 µg). Dose–response relationship was investigated by a multivariable Cox proportional hazards regression.
Results
Of the total 22 689 patients, 459 had lower respiratory tract cultures positive for S. maltophilia. The HR of S. maltophilia increased with increasing daily ICS dose: low ICS dose HR 2.6 (95% CI 1.6 to 4.0), moderate ICS dose HR 3.0 (95% CI 1.9 to 4.6) and high ICS dose HR 5.7 (95% CI 3.8 to 8.5).
Conclusions
We found that ICS was associated with a high, dose-dependent increased hazard of S. maltophilia in outpatients with COPD. High dose users had a nearly six times increased hazard compared with non-users of ICS. When appropriate, attempts at de-escalating ICS treatment should be made.
Keywords: COPD epidemiology; COPD Pathology; Pulmonary Disease, Chronic Obstructive
WHAT IS ALREADY KNOWN ON THIS TOPIC
Treatment with inhaled corticosteroids has been linked to a higher prevalence of pneumonia in patients with chronic obstructive pulmonary disease (COPD) in both clinical trials and cohort studies but has never been investigated in relation to colonisation of the lower respiratory tract by Stenotrophomonas maltophilia.
WHAT THIS STUDY ADDS
This study finds a dose–response relationship between increasing doses of inhaled corticosteroid and hazard for S. maltophilia in the lower respiratory tract. The clear dose–response relationship supports a causal relation.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
This study underlines the importance of continuous monitoring treatment and treatment response in patients with COPD, and when possible, step down or stop inhaled corticosteroid treatment.
Introduction
Chronic obstructive pulmonary disease (COPD) affects approximately 400 million people worldwide and is a leading cause of death.1 COPD is associated with airway inflammation,2 and oral and inhaled corticosteroids (ICS) are often used in stable state disease as well as during acute exacerbation as a mean to reduce mucosal swelling and inflammation.3
ICS are associated with an increased risk of adverse effects such as pneumonia,4 5 osteoporosis and diabetes.6 7 Additionally, infections with other Gram-negative opportunistic bacteria, such as Pseudomonas aeruginosa and mycobacterial infections, have been associated with ICS use in patients with COPD, both associated with worsened outcome.8–14 However, no knowledge on ICS in relation to Stenotrophomonas maltophilia exists.
S. maltophilia, an ubiquitous, aerobic, non-fermentative, Gram-negative bacillus is a global opportunistic pathogen.15 Further, S. maltophilia is an important nosocomial pathogen associated with a crude mortality in hospitalised ranging between 14% and 69% with an attributable mortality rate of up to 37.5%.16 17
The role of S. maltophilia in patients with COPD is poorly understood, and it is debated whether it should be considered a marker of disease progression or regarded as a pathogen in chronic lung diseases such as cystic fibrosis.18 In a Canadian population of outpatients with COPD, S. maltophilia was detected in 10% of patients and associated with a twofold increase in mortality.19 In a recent study from our group, S. maltophilia in the lower respiratory tract was associated with a threefold increased risk of both hospitalisation-requiring exacerbation of COPD and death.20
The aim of this study was to determine, among all outpatients with COPD from the Eastern part of Denmark, and with 100% follow-up on microbiological sampling and vital status, whether the risk of acquiring a lower respiratory tract culture with S. maltophilia was associated with increasing daily ICS doses. Specifically, we aimed to investigate if there was a dose–response relationship between ICS dose and S. maltophilia in the lower respiratory tract, as our hypothesis was that ICS use was associated with a dose-dependent risk of isolation of S. maltophilia in the lower respiratory tract.
Methods
Data sources
Data were collected from the following databases, which were accessible through the Danish Health Data Authority:
The Danish Register of COPD (DrCOPD) was used to identify patients with specialist-verified COPD. DrCOPD is a nationwide register that comprises individual patient data, for example, severity of airflow obstruction, body mass index (BMI), Medical Research Council (MRC) Dyspnoea Scale and smoking status, at all respiratory outpatient clinic visits since 2010.21
The Danish National Patient Registry contains data on all hospital contacts since 1995, including diagnoses and length of contact.22
The Danish National Database of Reimbursed Prescriptions is nationwide and includes data on all redeemed prescriptions. The register includes information on the strength, dose, product name and Anatomical Therapeutic Chemical classification of each prescription.23
Microbiological data from the Clinical Microbiology Departments in Eastern Denmark (Region Zealand and Capital Region) consist of approximately 2.6 million inhabitants. The register contains information on all microbiological samples including lower respiratory tract cultures, and by that used to identify patients with S. maltophilia. Samples containing sputum, tracheal secretion and bronchoalveolar wash were considered representative for the lower respiratory tract.
Study design
This was a retrospective, registry-based cohort study of Danish outpatients with COPD.
The study cohort comprised all patients registered with an outpatient clinic visit from 1 January 2010 to 31 October 2017 in DrCOPD. The majority of patients with COPD in Denmark attend their general practitioner for management of COPD, but those with most severe disease are referred to hospital-based outpatient clinics and included in DrCOPD. We had only access to microbiological data on patients residing in Eastern Denmark; hence, patients residing in Western Denmark were excluded as we had no access to microbiological data on these patients. However, we judged that the power of the study was sufficient despite only using the Eastern Danish data, due to the large number of cases in the cohort.
We excluded patients with a history of immunodeficiency and cancer, except for non-melanoma malignancies of the skin, within the last 5 years prior to cohort entry. Also, patients with a lower respiratory tract sample positive for S. maltophilia within 365 days of cohort entry were excluded.
Cohort entry was defined as the date for the patient’s first outpatient clinic visit in DrCOPD. We only included patients with outpatient registrations, as in-hospital registrations do not contain information on essential patient characteristics: severity of airflow obstruction (forced expired volume in the first second (FEV1)), degree of dyspnoea (MRC Dyspnoea Scale), BMI and smoking status.
Patients were followed for 5 years or until the first of either (1) end of microbiological sample follow-up on 1 July 2018, (2) event of S. maltophilia or (3) death.
Exposure to ICS
All prescriptions for ICS, alone or in fixed combination with long-acting bronchodilators, redeemed 365 days prior to cohort entry and during the study period were identified.
ICS was handled as a time-dependant variable; cumulated dose was based on prescriptions redeemed within 365 days prior to cohort entry to assess an average daily dose and updated for every 365 days of follow-up.
All doses of ICS were converted to budesonide-equivalent doses, as shown in table 1, as the potency varies as much as a factor 10 regarding local treatment effect.24 Patients with no ICS use during the study period were used as the reference category, while ICS users were separated into three subgroups: low ICS dose (≤400 µg), moderate ICS dose (400–800 µg) and high ICS dose (>800 µg) daily dose according to international guidelines.25
Table 1.
Equipotent doses of the different ICS drugs analysed24
| Drug | Budesonide | Momethasone | Beclomethasone | Beclomethasone HFA | Fluticasone propionate | Fluticasone furoate | Ciclesonide |
| Dose (µg) | 100 | 100 | 100 | 50 | 50 | 10 | 40 |
ICS, inhaled corticosteroids.
Statistical analyses
The risk of acquiring a lower respiratory tract culture positive for S. maltophilia associated with use of ICS was estimated using a Cox proportional hazards regression, censoring for death. Both unadjusted and adjusted results are presented. The multivariable model was adjusted for known and suspected confounders as well as markers of disease: age (continuous (year)), sex (male vs female), FEV1 (continuous (%)), BMI (continuous (kg/m2)), smoking status (active vs not active) and cumulated dose of oral corticosteroids (none, low: ≤250 mg prednisolone, high: >250 mg prednisolone) and calendar year for entry in DrCOPD. Cumulated oral corticosteroids were calculated by redeemed prescriptions 365 days prior to cohort entry, with ≤250 mg prednisolone corresponding to treatment for one exacerbation of COPD and >250 mg prednisolone corresponding to treatment for two or more exacerbation episodes. Prior exacerbation, FEV1, BMI and smoking status have earlier been showed to be strong predictors of exacerbation of COPD.26 Results are shown as HRs with 95% CIs.
For sensitivity analysis, we performed a multinomial propensity score (PS)-weighted Cox proportional hazards regression, as we had four levels of exposure.27
Model validation
Proportional hazards assumption was tested as an interaction with time, in addition to testing for linearity of the continuous covariates. Continuous covariates failing test for linearity were handled using penalised splines, which did not alter the results.
Missing data were handled by the substantive model for multiple imputation, with 100 imputations with each 20 iterations.28
Data management and statistical analyses were performed using Statistical Analysis Software V.9.4 (SAS Institute). Multiple imputation, Cox proportional hazards regression and inverse probability of treatment weighting were completed in R Studio V.4.1.3 (R Foundation for Statistical Computing, Vienna, Austria) with the SMCFCS V.1.6.1, MITOOLS V.2.4, SURVIVAL V.3.3.1 and TWANG V.2.5 packages respectively.
Patient and public involvement
In the Steering Committee of our organisation (COP:TRIN), we have patient representation to ensure that patients’ opinions and interpretations are taken seriously. This allows them to influence the design and conduct of trials and observational studies. For example, studies like this one.
Further, we have recently, as chairs and initiators, conducted a global Delphi survey on outcomes in COPD trials, in which patients, relatives and patient organisations were invited and participated.
We seek to inform patients, whenever we conduct nationwide observational studies, by making press releases and by discussing results with the Danish lung patient organisation.
Results
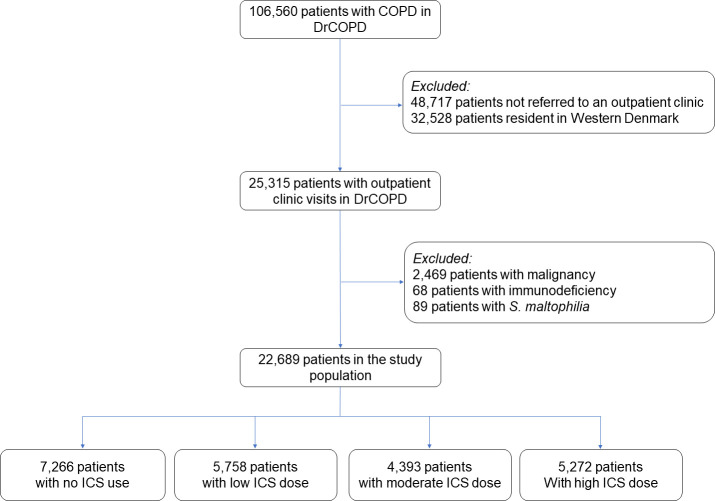
A total of 106 560 patients with COPD were identified in DrCOPD. Of these, 57 843 had at least one outpatient visit during the study period. Patients from Western Denmark (n=32 528) were excluded due to no accessible microbiological data. Additionally, patients with a diagnosis of malignant disease (n=2469) or immunodeficiency (n=68) within 5 years before study entry were excluded, as well as those with a lower respiratory tract sample positive for S. maltophilia (n=89) within 365 days prior to potential cohort entry.
In total, 22 689 patients were included and contributed 70 896 person-years of risk-time for the analysis (figure 1). A lower respiratory tract sample positive for S. maltophilia was found in 459 (2.0%) patients (no ICS use: 35, low ICS dose: 67, moderate ICS dose: 114 and high ICS dose: 243). Baseline characteristics are listed in table 2, and definitions of comorbidities are in online supplemental appendix 1. In general, with increasing daily ICS dose, a decrease in FEV1 and increase in high-dose oral corticosteroids as well as an increase in prevalence of asthma were observed.
Figure 1.
Study flowchart. DrCOPD: Danish Register of COPD, ICS: Inhaled corticosteroid.
Table 2.
Baseline characteristics at study entry of outpatients with COPD according to use of inhaled corticosteroid
| Inhaled corticosteroid group | ||||
| None (n=7266) | Low (n=5758) | Moderate (n=4393) | High (n=5272) | |
| Daily budesonide equivalents (µg), median (IQR) | 0 | 184 (110–316) | 579 (473–658) | 1315 (986–1808) |
| Sex female, n (%) | 3543 (48.8) | 3049 (53.0) | 2511 (57.2) | 3276 (62.1) |
| Age (years), median (IQR) | 68.5 (60.0–76.3) | 69.3 (61.1–76.9) | 70.5 (63.5–77.6) | 71.0 (63.9–78.0) |
| FEV1 (%), median (IQR), missing (%) | 57.0 (44.0–70.0) (16.4) | 52.0 (40.0–65.0) (10.7) | 45.0 (33.0–58.0) (8.5) | 40.0 (29.0–53.0) (8.0) |
| BMI (kg/m2), median (IQR), missing (%) | 25.0 (22.0–29.0) (17.7) | 25.0 (22.0–29.0) (11.5) | 25.0 (21.0–29.0) (8.8) | 24.0 (21.0–28.0) (7.8) |
| MRC Dyspnoea Scale, median (IQR) | 2 (2–3) | 3 (2–3) | 3 (2–4) | 3 (3–4) |
| Smoking status | ||||
| Not active smoker, n (%) | 3292 (45.3) | 3167 (55.0) | 2764 (62.9) | 3309 (62.8) |
| Active smoker, n (%) | 2773 (38.2) | 1981 (34.4) | 1281 (29.3) | 1568 (29.7) |
| Smoking status missing, n (%) | 1201 (16.5) | 610 (10.6) | 342 (7.8) | 395 (7.5) |
| Oral corticosteroid* | ||||
| None, n (%) | 5992 (82.5) | 3685 (64.0) | 2409 (54.8) | 2327 (44.1) |
| Low dose, n (%) | 444 (6.1) | 631 (11.0) | 489 (11.1) | 657 (12.5) |
| High dose, n (%) | 830 (11.4) | 1442 (25.0) | 1495 (34.0) | 2288 (43.4) |
| Comorbidities† | ||||
| Cerebrovascular disease, n (%) | 695 (9.6) | 529 (9.2) | 353 (8.0) | 419 (7.9) |
| Asthma, n (%) | 479 (6.6) | 894 (15.5) | 834 (19.0) | 1124 (21.3) |
| Atrial fibrillation, n (%) | 1086 (14.9) | 836 (14.5) | 648 (14.8) | 691 (13.1) |
| Depression, n (%) | 327 (4.5) | 270 (4.7) | 211 (4.8) | 272 (5.2) |
| Diabetes mellitus, n (%) | 945 (13.0) | 685 (11.9) | 446 (10.2) | 599 (11.4) |
| Congestive heart failure, n (%) | 1265 (17.4) | 947 (16.4) | 712 (16.2) | 847 (16.1) |
| Ischaemic heart disease, n (%) | 534 (7.3) | 463 (8.1) | 285 (6.5) | 374 (7.1) |
| Renal disease, n (%) | 398 (5.5) | 275 (4.8) | 161 (3.7) | 203 (3.9) |
| Peripheral vascular disease, n (%) | 828 (11.4) | 595 (10.3) | 406 (9.2) | 438 (8.3) |
*Oral corticosteroids accumulated dose 365 days prior to study entry: none: no use, low dose: ≤250 mg prednisolone, high dose: >250 mg prednisolone.
†Comorbidities registered in the Danish National Patient Registry prior to study entry (online supplemental appendix 1).
BMI, body mass index; COPD, chronic obstructive pulmonary disease; FEV1, forced expired volume in the first second; MRC, Medical Research Council; n, number.
bmjresp-2023-001929supp001.pdf (33.3KB, pdf)
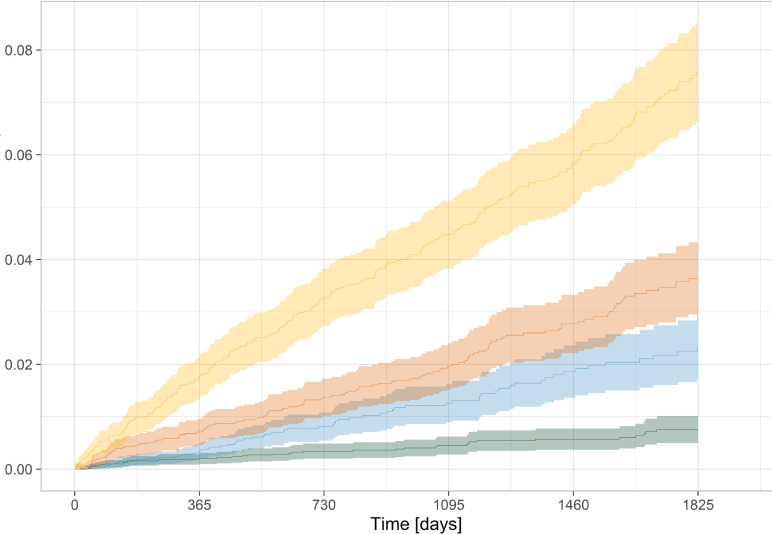
The adjusted Cox proportional hazards regression showed an increased HR of acquiring a lower respiratory tract sample positive for S. maltophilia with a dose–response relationship in the low, moderate and high ICS dose group compared with the no use of ICS group: low dose: HR 2.6 (CI 1.6 to 4.0), moderate dose: HR 3.0 (CI 1.9 to 4.6) and high dose: HR 5.7 (CI 3.8 to 8.5), which was an attenuation of the unadjusted results (table 3 and figure 2; for full adjusted analysis, see online supplemental appendix 2).
Table 3.
Hazard for acquisition of Stenotrophomonas maltophilia with use of inhaled corticosteroid
| Unadjusted | Adjusted | |||
| HR | CI | HR | CI | |
| Inhaled corticosteroid group* | ||||
| None | Ref | – | Ref | – |
| Low dose | 3.0 | 1.9 to 4.7 | 2.6 | 1.6 to 4.0 |
| Moderate dose | 4.4 | 2.9 to 6.6 | 3.0 | 1.9 to 4.6 |
| High dose | 9.9 | 6.8 to 14.5 | 5.7 | 3.8 to 8.5 |
The analysis was adjusted for: age, sex, forced expired volume in the first second, body mass index, smoking status and oral corticosteroid use.
*Inhaled corticosteroid cumulated daily dose in budesonide equivalents: none: no use, low dose: ≤400 µg, moderate dose: 400–800 µg, high dose: >800 µg.
Figure 2.
Cumulative incidence of positive lower respiratory tract sample for S. maltophilia within 5 years of first outpatient clinic visit according to exposure to inhaled corticosteroid use (green: no use, blue: low dose, red: moderate dose, and yellow: high dose. Mean in solid, 95% confidence interval in transparent).
bmjresp-2023-001929supp002.pdf (61.8KB, pdf)
The PS sensitivity analysis confirmed the main finding: low dose: HR 2.4 (CI 1.5 to 3.8), moderate dose: HR 2.6 (CI 1.6 to 4.1) and high dose: HR 5.2 (CI 3.4 to 8.0), respectively.
Of the included patients, 2932 (13%) had one or more clinical values missing from the outpatient clinic visit. In most cases, when missing data were seen, FEV1, BMI, MRC Dyspnoea Scale and smoking status were missing together, telling us that somehow the values were not correctly reported from the outpatient clinic. This indicates that data were missing at random, and thus allowed for handling by multiple imputation.
Discussion
In this regional registry-based cohort of outpatients with COPD, we found that ICS treatment was associated with a marked, dose–response-related, increased hazard of a lower respiratory tract sample positive for S. maltophilia. The result was robust for adjustment and by an inverse probability of treatment-weighted sensitivity analysis.
The evidence on COPD and the role of S. maltophilia is sparse, and even slimmer when considering ICS use in patients with COPD. A single study has found an association between ICS and infection with S. maltophilia, but not differentiated by ICS doses.19 A meta-analysis covering eight studies on S. maltophilia in intensive care units shows an association between COPD and S. maltophilia pneumonia.29
Other larger population-based studies have demonstrated increased risk of pneumonia, infection with P. aeruginosa and mycobacterial infections with ICS use in patients with COPD.8–12 Results from these studies cannot be directly compared, but several similarities are apparent. First, the high proportion of ICS users in our cohort is comparable with the previous studies, and like ours, these studies also report a robust dose-related risk. Several COPD trials have also reported increased risk of pneumonia with ICS use30 31 and the risk of pneumonia was also seen when lower daily doses of ICS are used.32 33
It is debated whether S. maltophilia in the lower respiratory tract represents a pathogen-causing infection or is merely a benign colonising bacterium found in those with the most advanced degree of COPD. A recent study from our group has demonstrated that S. maltophilia certainly is associated with a steep increase in disease burden following infection.
Also, in patients with cystic fibrosis, S. maltophilia is a well-established contributor to irreversible decline in lung function and increase in exacerbation, and the pathogenesis is closely linked to well-studied and complex virulence mechanisms, cross-infection and multiresistance.18
Strengths of the current study include observations based on a large and well-characterised population of patients with a respiratory specialist-verified and spirometry-confirmed diagnosis of COPD. The patients have one or more outpatient clinic visits but are in all other aspects unselected. Also, we have data giving us information on actual ICS doses redeemed at the pharmacy and not only prescriptions. Last, there is a 100% follow-up for lower respiratory tract sample positive for S. maltophilia, as all patients, we argue, with S. maltophilia would have relevant symptoms and, thus have a lower respiratory tract sample analysed.
Despite the noted strengths, our study has some important limitations. First, we cannot report actual intake of the ICS, since the data did not contain information on adherence. Some patients may fail to administer the inhalation correctly. However, repetitive collection of the prescribed medicine suggests some degree of adherence and we have no reason to suspect different degrees of adherence in the three strata of ICS users. Additionally, the proportion of patients with COPD on ICS correlates well with previous population studies.9 10 Second, ICS seems to correlate with more severe COPD and confounding by indication may be a concern; however, our registries contain many important confounders such as smoking status, oral corticosteroid use, lung function by FEV1 and nutrition status, allowing us to account for these. We cannot rule out residual confounding, but the paramount effect of ICS after adjusting indicates that confounding by no mean can be the only explanation of the found association between ICS and S. maltophilia. Third, we do not have lower respiratory tract sample from all patients, but we argue that patients followed in an outpatient clinic with relevant symptoms (phlegm, coughing, elevated markers of infection, fever) would have a sample taken.
To conclude, use of ICS in patients with COPD who were followed in outpatient clinics was associated with a substantial dose–response-related hazard of getting a lower respiratory tract culture positive for S. maltophilia. The large and seemingly strong dose–response relationship of this hazard suggests a causal relationship. Hence, we argue that ICS should only be administered specifically to patients with COPD who have a documented need as per guideline recommendations, and caution should be taken when prescribing higher doses of ICS, especially since it is unclear whether high doses are more effective in patients with COPD than low and moderate doses.5 34
Acknowledgments
We thank the COP:TRIN (coptrin.dk) Steering Committee and the CURE group for providing input to this study during meetings.
Footnotes
Contributors: Conceptualisation—CR, PS, JE, CSU and J-USJ. Methodology—all authors. Software—CR and PK. Formal analysis—CR and PK. Resources—J-USJ. Data curation—CR and PK. Writing (original draft preparation)—CR, PK, CKH, PS, JE, CSU and J-USJ. Writing (review and editing)—all authors. Visualisation—CR. Supervision—PS, CSU and J-USJ. Project administration—CR, PS, JE, CSU and J-USJ. Funding acquisition—J-USJ. Guarantor—J-USJ.
Funding: The study was financed by grants from the Novo Nordisk Foundation (no. NNF20OC0060657).
Disclaimer: The funding source had no influence on study design, data collection, analysis or reporting of data.
Competing interests: Outside the submitted work, RBD has been on an advisory board for Pfizer. CSU has received grants from Sanofi, Boehringer Ingelheim, AstraZeneca and Novartis; consulting fees from Chiesi, Orion Pharma, AstraZeneca, GSK and TEVA; speaker fees from Orion Pharma, AstraZeneca and TEVA; and been on advisory boards for Novartis, Sanofi, Glaxo-Smith Kline, Chiesi, AstraZeneca and Boehringer Ingelheim.
Patient and public involvement: Patients and/or the public were involved in the design, or conduct, or reporting, or dissemination plans of this research. Refer to the Methods section for further details.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data may be obtained from a third party and are not publicly available. The data supporting the findings of this study are available from the Danish Health Data Authority following application and approval from the Danish Health Authority. Restrictions apply to the availability of these data, which were used under the licence for this study.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
In Denmark, retrospective use of registry data does not require patient consent nor approval from the Danish National Committee on Health Research Ethics.
References
- 1. Adeloye D, Chua S, Lee C, et al. Global and regional estimates of COPD prevalence: systematic review and meta-analysis. J Glob Health 2015;5:020415. 10.7189/jogh.05.020415 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Stănescu D, Sanna A, Veriter C, et al. Airways obstruction, chronic expectoration, and rapid decline of fev1 in smokers are associated with increased levels of sputum neutrophils. Thorax 1996;51:267–71. 10.1136/thx.51.3.267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Falk JA, Minai OA, Mosenifar Z. Inhaled and systemic corticosteroids in chronic obstructive pulmonary disease. Proc Am Thorac Soc 2008;5:506–12. 10.1513/pats.200707-096ET [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Crim C, Dransfield MT, Bourbeau J, et al. Pneumonia risk with inhaled fluticasone furoate and vilanterol compared with vilanterol alone in patients with COPD. Ann Am Thorac Soc 2015;12:27–34. 10.1513/AnnalsATS.201409-413OC [DOI] [PubMed] [Google Scholar]
- 5. Agusti A, Fabbri LM, Singh D, et al. Inhaled corticosteroids in COPD: friend or foe. Eur Respir J 2018;52:1801219. 10.1183/13993003.01219-2018 [DOI] [PubMed] [Google Scholar]
- 6. Price DB, Voorham J, Brusselle G, et al. Inhaled corticosteroids in COPD and onset of type 2 diabetes and osteoporosis: matched cohort study. NPJ Prim Care Respir Med 2019;29:38. 10.1038/s41533-019-0150-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Suissa S, Kezouh A, Ernst P. Inhaled corticosteroids and the risks of diabetes onset and progression. Am J Med 2010;123:1001–6. 10.1016/j.amjmed.2010.06.019 [DOI] [PubMed] [Google Scholar]
- 8. Brassard P, Suissa S, Kezouh A, et al. Inhaled corticosteroids and risk of tuberculosis in patients with respiratory diseases. Am J Respir Crit Care Med 2011;183:675–8. 10.1164/rccm.201007-1099OC [DOI] [PubMed] [Google Scholar]
- 9. Suissa S, Patenaude V, Lapi F, et al. Inhaled corticosteroids in COPD and the risk of serious pneumonia. Thorax 2013;68:1029–36. 10.1136/thoraxjnl-2012-202872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Eklöf J, Ingebrigtsen TS, Sørensen R, et al. Use of inhaled corticosteroids and risk of acquiring pseudomonas aeruginosa in patients with chronic obstructive pulmonary disease. Thorax 2022;77:573–80. 10.1136/thoraxjnl-2021-217160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Andréjak C, Nielsen R, Thomsen VØ, et al. Chronic respiratory disease, inhaled corticosteroids and risk of non-tuberculous mycobacteriosis. Thorax 2013;68:256–62. 10.1136/thoraxjnl-2012-201772 [DOI] [PubMed] [Google Scholar]
- 12. Ernst P, Gonzalez AV, Brassard P, et al. Inhaled corticosteroid use in chronic obstructive pulmonary disease and the risk of hospitalization for pneumonia. Am J Respir Crit Care Med 2007;176:162–6. 10.1164/rccm.200611-1630OC [DOI] [PubMed] [Google Scholar]
- 13. Hassan MM, Tahir MH, Ameeq M, et al. Risk factors identification of COVID-19 patients with chronic obstructive pulmonary disease: a retrospective study in Punjab-Pakistan. Immun Inflamm Dis 2023;11:e981. 10.1002/iid3.981 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Eklöf J, Sørensen R, Ingebrigtsen TS, et al. Pseudomonas aeruginosa and risk of death and exacerbations in patients with chronic obstructive pulmonary disease: an observational cohort study of 22 053 patients. Clin Microbiol Infect 2020;26:227–34. 10.1016/j.cmi.2019.06.011 [DOI] [PubMed] [Google Scholar]
- 15. Denton M, Kerr KG. Microbiological and clinical aspects of infection associated with stenotrophomonas maltophilia. Clin Microbiol Rev 1998;11:57–80. 10.1128/CMR.11.1.57 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Brooke JS. Stenotrophomonas maltophilia: an emerging global opportunistic pathogen. Clin Microbiol Rev 2012;25:2–41. 10.1128/CMR.00019-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Falagas ME, Kastoris AC, Vouloumanou EK, et al. Attributable mortality of stenotrophomonas maltophilia infections: a systematic review of the literature. Future Microbiol 2009;4:1103–9. 10.2217/fmb.09.84 [DOI] [PubMed] [Google Scholar]
- 18. Hansen CR. Stenotrophomonas maltophilia: to be or not to be a cystic fibrosis pathogen. Curr Opin Pulm Med 2012;18:628–31. 10.1097/MCP.0b013e328358d4f8 [DOI] [PubMed] [Google Scholar]
- 19. Soumagne T, Levesque F, Milot J, et al. Significance of stenotrophomonas maltophilia when detected in sputum of ambulatory patients with COPD. Int J Chron Obstruct Pulmon Dis 2021;16:2895–900. 10.2147/COPD.S325419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Rønn C, Kamstrup P, Eklöf J, et al. Mortality and exacerbations associated with stenotrophomonas maltophilia in chronic obstructive pulmonary disease. A regional cohort study of 22,689 outpatients. Respir Res 2023;24:232. 10.1186/s12931-023-02544-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Lange P, Tøttenborg SS, Sorknæs AD, et al. Danish register of chronic obstructive pulmonary disease. Clin Epidemiol 2016;8:673–8. 10.2147/CLEP.S99489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Lynge E, Sandegaard JL, Rebolj M. The Danish national patient register. Scand J Public Health 2011;39(7 Suppl):30–3. 10.1177/1403494811401482 [DOI] [PubMed] [Google Scholar]
- 23. Johannesdottir SA, Horváth-Puhó E, Ehrenstein V, et al. Existing data sources for clinical epidemiology: the Danish national database of reimbursed prescriptions. Clin Epidemiol 2012;4:303–13. 10.2147/CLEP.S37587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. (CHMP) CfMPfHU . Type II variation assessment report: European medicines agency; 2015.
- 25. Reddel HK, Bacharier LB, Bateman ED, et al. Global initiative for asthma strategy 2021: executive summary and rationale for key changes. Eur Respir J 2022;59:2102730. 10.1183/13993003.02730-2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Hurst JR, Vestbo J, Anzueto A, et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N Engl J Med 2010;363:1128–38. 10.1056/NEJMoa0909883 [DOI] [PubMed] [Google Scholar]
- 27. Rosenbaum PR. Model-based direct adjustment. J Am Stat Assoc 1987;82:387–94. 10.1080/01621459.1987.10478441 [DOI] [Google Scholar]
- 28. Bartlett JW, Seaman SR, White IR, et al. Multiple imputation of covariates by fully conditional specification: accommodating the substantive model. Stat Methods Med Res 2015;24:462–87. 10.1177/0962280214521348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wang N, Tang C, Wang L. n.d. Risk factors for acquired stenotrophomonas maltophilia pneumonia in intensive care unit: a systematic review and meta-analysis. Front Med;8. 10.3389/fmed.2021.808391 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Crim C, Calverley PMA, Anderson JA, et al. Pneumonia risk in COPD patients receiving inhaled corticosteroids alone or in combination: TORCH study results. Eur Respir J 2009;34:641–7. 10.1183/09031936.00193908 [DOI] [PubMed] [Google Scholar]
- 31. Calverley PMA, Stockley RA, Seemungal TAR, et al. Reported pneumonia in patients with COPD: findings from the INSPIRE study. Chest 2011;139:505–12. 10.1378/chest.09-2992 [DOI] [PubMed] [Google Scholar]
- 32. Ferguson GT, Anzueto A, Fei R, et al. Effect of fluticasone propionate/salmeterol (250/50 microg) or salmeterol (50 microg) on COPD exacerbations. Respir Med 2008;102:1099–108. 10.1016/j.rmed.2008.04.019 [DOI] [PubMed] [Google Scholar]
- 33. Anzueto A, Ferguson GT, Feldman G, et al. Effect of fluticasone propionate/Salmeterol (250/50) on COPD exacerbations and impact on patient outcomes. COPD 2009;6:320–9. 10.1080/15412550903140881 [DOI] [PubMed] [Google Scholar]
- 34. Archontakis Barakakis P, Tran T, You JY, et al. High versus medium dose of inhaled corticosteroid in chronic obstructive lung disease: a systematic review and meta-analysis. Int J Chron Obstruct Pulmon Dis 2023;18:469–82. 10.2147/COPD.S401736 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjresp-2023-001929supp001.pdf (33.3KB, pdf)
bmjresp-2023-001929supp002.pdf (61.8KB, pdf)
Data Availability Statement
Data may be obtained from a third party and are not publicly available. The data supporting the findings of this study are available from the Danish Health Data Authority following application and approval from the Danish Health Authority. Restrictions apply to the availability of these data, which were used under the licence for this study.