Abstract
Introduction:
Uncomplicated Staphylococcus aureus bacteremia remains a leading cause of morbidity and mortality in hospitalized patients. Current guidelines recommend a minimum of 14 days of treatment.
Objective:
To evaluate the efficacy and safety of short versus usual antibiotic therapy in adults with uncomplicated S. aureus bacteremia (SAB).
Methods:
We developed a search strategy to identify systematic review and meta-analysis of non-randomized studies (NRS), comparing short versus usual or long antibiotic regimens for uncomplicated SAB in MEDLINE, Embase, and the Cochrane Register up to June 2023. The risk of bias was assessed using the ROBINS I tool. The meta-analysis was performed using Review Manager software with a random effect model.
Results:
Six NRS with a total of 1700 patients were included. No significant differences were found when comparing short versus prolonged antibiotic therapy as defined by the authors for 90-day mortality [odds ratio (OR): 1.09; 95% confidence interval (CI): 0.82–1.46, p: 0.55; I2 = 0%] or 90-day recurrence or relapse of bacteremia [OR: 0.72; 95% CI: 0.31–1.68, p: 0.45; I2 = 26%]. Sensitivity analysis showed similar results when comparing a predefined duration of <14 days versus ⩾14 days and when excluding the only study with a high risk of bias.
Conclusion:
Shorter-duration regimens could be considered as an alternative option for uncomplicated SAB in low-risk cases. However, based on a small number of studies with significant methodological limitations and risk of bias, the benefits and harms of shorter regimens should be analyzed with caution. Randomized clinical trials are needed to determine the best approach regarding the optimal duration of therapy.
Keywords: antibiotic duration, bacteremia, meta-analysis, Staphylococcus aureus, systematic review
Plain language summary
Comparing short and regular antibiotic treatment duration, for a type of blood infection caused by S. aureus
We investigated the optimal duration of antibiotic treatment for adults with a specific type of blood infection (uncomplicated Staphylococcus aureus), a condition with a significant global impact on mortality and costs. After a thorough search, only six trials involving 1700 patients were identified. We therefore decided to perform a meta-analysis (a type of statistical analysis). The results showed that the duration of antibiotics, whether short or long (less or more than 14 days), did not significantly affect mortality or recurrence of infection within 90 days. Consequently, we suggested that shorter courses of antibiotics might be appropriate for less severe cases. However, we emphasized caution because of the limitations of the studies. We recommended further research with improved methods to determine the optimal approach to treating this type of infection.
Introduction
Bacteremia due to Staphylococcus aureus (S. aureus) is one of the three leading causes of healthcare-associated bacteremia worldwide.1,2 Its presence is associated with mortality of up to 10.4% at 7 days and 27% at 30 days. 3 It is also estimated that each episode of bacteremia due to methicillin-sensitive S. aureus (MSSA) can cost approximately 10,000 euros, and up to twice that for methicillin-resistant S. aureus (MRSA). 4
Reducing the duration of antibiotic treatment for bacterial infections is being evaluated in various clinical settings, due to its potential to decrease adverse events, costs, and hospitalization duration while also maintaining comparable effectiveness regarding mortality, relapse, and infection complications.5–8 For example, in community-acquired pneumonia with clinical stability, one study demonstrated non-inferiority of a 3-day versus 8-day regimen. 9 Similar findings were reported in afebrile males with urinary tract infections, comparing 7 versus 14 days. 10 Recently published clinical trials have evaluated the efficacy of shortened antibiotic regimens for uncomplicated Gram-negative bacteremia, showing non-inferiority in efficacy-related outcomes and a lower incidence of adverse effects compared to longer antibiotic regimens.5,7,11–13
Current guidelines recommend a minimum of 14 days of antibiotic treatment for uncomplicated S. aureus bacteremia (SAB) 14 ; however, evidence supporting this recommendation is scarce and mainly based on small observational studies15,16 with several methodological limitations. New evidence on relevant clinical and microbiological outcomes of shorter antibiotic regimens is emerging and may change this recommendation.
This systematic review evaluates whether shortened antibiotic treatment in adult patients with uncomplicated SAB differs from standard therapy (14 days or longer) in terms of efficacy and safety-related outcomes.
Methods
Protocol
The protocol was registered in the International Prospective Registry of Systematic Reviews: CRD42023440714.
Search strategy
We developed a search strategy to identify randomized clinical trials (RCTs) and non-randomized studies (NRS), comparing short versus usual or long antibiotic regimens for S. aureus uncomplicated bacteremia in MEDLINE, Embase, and the Cochrane Central Register of Controlled Trials (CENTRAL), from inception to June 2023. It included controlled vocabulary and free-text terms using field labels, Boolean, and proximity operators adapted for each search engine, without language restrictions. Search strategies are available in Supplemental Table 1. In addition, we searched for clinical trial registries in the International Clinical Trials Registry Platform of the World Health Organization; and we hand-searched a reference list of the selected studies.
Studies selection
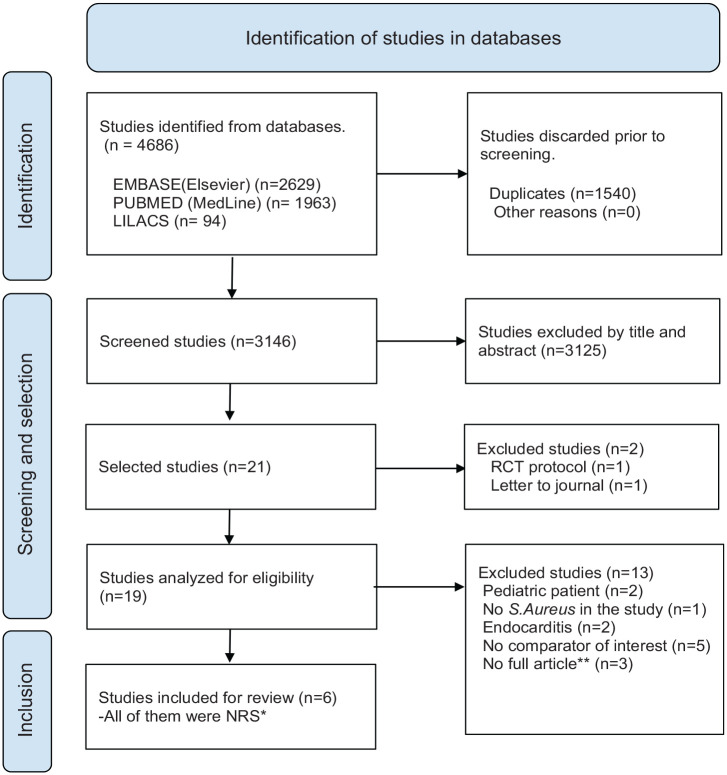
Two authors (CD-B and SGP) independently reviewed the identified studies, initially by title and abstract and later by full text. Disagreements in the selection process were resolved by consensus or by involving a third reviewer (OMMV). The selection process is presented in the PRISMA (Preferred reporting items for systematic review and meta-analysis) flowchart (Figure 1).
Figure 1.
PRISMA flowchart.
*NRS, non-randomized studies.
**Only the abstract was found.
Studies were included if they considered hospitalized patients older than 18 years with uncomplicated SAB confirmed by at least one positive blood culture. Studies were excluded if they reported on polymicrobial bacteremia, sources of bacteremia requiring prolonged treatment, evidence of metastatic sites of infection, and prolonged bacteriemia.
Studies had to compare a short course with a longer or usual duration of antibiotic treatment, as defined by the author, versus the standard of care for the treatment of uncomplicated SAB at the time of the study.
In addition, studies had to report at least one of the following outcomes: in-hospital mortality from any cause, outpatient mortality from any cause at 30/90 days, recurrence of bacteremia, distant relapse (defined as evidence of metastatic sites of infection after completion of antibiotic treatment before 90 days), or adverse events related to antibiotic therapy. Bacteremia recurrence was determined by the presence of a new bloodstream infection of S. aureus after the initial 90-day proposed antibiotic regimen completion. If genetic testing was available, it had to confirm that the microorganism was identical to the initial one. Otherwise, it was classified as a reinfection after the 90-day threshold. Adverse events associated with antibiotics for MRSA and MSSA included acute kidney injury (as defined by KDIGO criteria), hypersensitivity reactions, infusion-related reactions, gastrointestinal symptoms, rash, phlebitis, chills, and less frequently neutropenia (<500/mm3).
Risk of bias assessment
We planned to assess the risk of bias for RCT using the ROB 2 tool 17 and for NRS using the ROBINS I tool 18 proposed by the Cochrane Collaboration. This assessment was performed by two investigators (CD-B and SGP) in parallel and independently, with discrepancies resolved by a third investigator (OMMV).
The NRS were categorized as having low, moderate, high, or critical risk of bias according to seven domains: bias due to confounding, bias in the selection of participants for the study, bias in the classification of interventions, bias due to deviations from the intended interventions, bias due to missing data, and bias due to the measurement of outcomes. 17
Data extraction
The following information was extracted for each study: author, country in which the study was conducted, study date, baseline characteristics of the population (including sex, age, prevalence of diabetes, intravenous drug use, immunosuppression, the presence of a catheter, hemodialysis, and patients with recent surgery), intervention and comparators, percentage of MRSA, most commonly used antibiotic, and the frequency of outcomes. If necessary, the corresponding authors were contacted to obtain missing data; if not available, the study was discarded.
Analysis
Meta-analysis was carried out using Review Manager software 5.4 version. 19 A random effects model was used, as proposed by Valentine for meta-analyses including NRS because it takes into account the unexplained heterogeneity of results associated with different sources of bias between studies. 20 For dichotomous outcomes, results are presented as odds ratio (OR) with their 95% confidence intervals (CIs), displayed in forest plot figures.
However, we recognize that in these circumstances, the CI around a summary meta-analytic estimate represents only the identifiable statistical variation and does not fully reflect the uncertainty due to the unknown direction and magnitude of bias in each study. Therefore, we choose not only to perform the meta-analysis but also to include a description of possible confounding to allow the reader to make their assessment of heterogeneity. 20
Clinical heterogeneity was assessed by analyzing the variability between studies due to differences in the characteristics of the participants, the interventions, the comparators, and the way the outcomes were measured. Statistical heterogeneity was assessed by visual inspection of the forest plot and the χ2, I2, and TAU 2 statistical tests. A publication bias analysis was planned in case there were at least 10 studies evaluating a certain outcome, but it was not executed as this criterion was not fulfilled. 21 We planned a subgroup analysis differentiating short versus long according to the definition of the studies, and according to our definition of < or ⩾14 days. Likewise, a sensitivity analysis was performed excluding those with a high risk of bias.
Results
A total of 3146 individual studies were identified, of which 6 non-RCTs22–27 with a total of 1700 patients were finally included. No RCTs met the inclusion criteria. Of the selected studies, four were retrospective cohorts22,25–27 conducted in Switzerland, Denmark, the Netherlands, and the United States. Another study was a prospective cohort 23 from South Korea, and finally one was a post hoc analysis 24 from the United Kingdom. Zeylemaker et al. 25 included three different cohorts with different moments of data collection, assessment of comorbidities, and sample size. A table with the inclusion and exclusion criteria for the individual studies has been addressed in Supplemental Table 2. The selection process is shown in the PRISMA diagram 28 (Figure 1).
The characteristics of the studies and the patients included in each study are shown in Table 1. The median age was between 50 and 70 years, and the proportion of male patients was more than 60%. The proportion of patients with diabetes ranged from 12% to 30%, and the proportion of patients with immunosuppression was variable, ranging from 5% to 40% related to different definitions in each study. Hemodialysis and surgery were present in <10% of patients in most studies.
Table 1.
Included studies and patients’ characteristics.
| Author, year, country | Type of study | Uncomplicated SAB definition | Short group a | Usual group a | Assessed outcomes | AB Most Used Name, % | N | Age, years, median IQR | Male% | MRSA% | DM% | IDU% | IS% | HD% | CX % | CVC % |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Abbas, 2019, Switzerland |
RC | Absence of: EC, IP. Fever <3 days ,SAB duration < 2 days. |
⩽14 | >14 | Mortality 90 days, Relapse 90 days. |
MRSA Vanc, 48.6 MSSA Flux 24.3 |
185 | ST 74.3 (51.2–83.4) UT 70.4 (53.5–81.2) |
ST 66.7 UT 66.6 |
ST 32 UT 15 |
ST 13.3 UT 13.9 |
ST 2.7 UT 5.4 |
ST 44.0
b
UT 42.4 b |
ST 9.3 UT 7.3 |
NR | ST 20 UT 12 |
| Thorlacius-Ussing, 2020, Denmark d |
RC | Absence of: EC, IP, OM, MG, AR, SP, PNEU. Positive BC <48 h AT. |
CH1 8 (7–10) CH2 9 (8–10) CH3 8 (7–10) |
CH1 14 (13–15) CH2 14 (13–15) CH3 13 (12–15) |
Mortality 30 days , Mortality 90 days , Relapse 90 days. |
NR | CH1 645 CH2 219 CH3 141 |
CH1 ST 71(58–81) UT 71(59–83) CH2 ST 53(49–77) UT 63(52–73) CH3 ST 62 (52–75) UT 70 (55–80) |
CH1 ST 63 UT 64 CH2 ST 68 UT 66 CH3 ST 63 UT 57 |
CH1 ST 63 UT 64 CH2 ST 68 UT 66 CH3 ST 63 UT 57 |
CH1 ST 25 UT 24 CH2 ST 14 UT 20 CH3 NR |
CH1 ST 4 UT 3 CH2 ST 5 UT 3 CH3 ST 2 UT 3 |
CH1
c
ST 5 UT 6 CH2 c ST 19 UT 17 CH3 NR |
CH1 ST 2 UT 3 CH2 NR CH3 ST 12 UT 12 |
CH1 ST 2 UT 2 CH2 ST 3 UT 4 CH3 ST 0 UT 1 |
CH1 ST 30 UT 21 CH2 ST 32 UT 26 CH3 ST 22 UT 14 |
| Zeylemaker, 2001, Netherlands |
RC | Absence of EC, OM, MA, SS. | ⩽14 | >14 | Favorable outcome
e
Mortality due to complications ,Mortality due to underlying disease. |
NR | 44 | 57 (20–92) | 47 | NR | 12.2 | NR | 30.6 f | NR | NR | 100 |
| Evans, 2022, United Kingdom |
Post hoc | Absence of: EC, IP. | <10 | 10–18 | Mortality 28 days | MRSA Vanc 77.2 MSSA Flux 78.0 |
291 | ST 67(45–77) UT 66(51–76) |
ST 60.9 UT 68.3 |
ST 16 UT 12.2 |
ST 25.4 UT 21.7 |
NR | NR | ST 9.6 UT 11.7 |
ST 8.6 UT 12.2 |
ST 12.2 UT 13.9 |
| Chong, 2013, South Korea |
PC | Absence of EC, MA ,Fever <3 days ,SAB duration <2–4 days. |
<14 | ⩾14 | Relapse 90 days ,Mortality 90 days |
MRSA Vanc 85 MSSA Cfz 48.3 |
111 | ST 63(51–73) UT 59(49–67) |
ST 57.9 UT 61.6 |
ST 47.4 UT 47.9 |
ST 23.7 UT 24.7 |
NR | ST 18.4
f
UT 16.4 f |
ST 5.3 UT 13.7 |
NR | ST 34.2 UT 53.4 |
| Taupin, 2020, United States |
RC | Absence of EC, AR, OM, MA, PNEU, IP, Fever <3 days ,SAB duration <3 days. |
<14 | ⩾14 | Complicated bacteremia , Relapse and 90-day mortality. |
NR | 64 | ST 60(51–70) GU 64(50–74) |
ST 48 UT 53 |
ST 19 UT 37 |
ST 29 UT 40 |
ST 0 UT 2 |
ST 14
g
UT 44 g |
ST 0 UT 12 |
NR | ST 10 UT 47 |
The results are displayed as days.
Immunosuppression includes any of the following: HIV (CD4 200/mm3), an end-stage renal disease requiring dialysis, insulin-dependent DM, active malignancy, cytotoxic chemotherapy 6 months prior, prednisone therapy 10 mg/day, child C cirrhosis, neutropenia 500/mm3, hematopoietic stem cell transplantation, solid organ transplantation.
Immunosuppression was defined as the use of immunosuppressive medication.
This study has three cohorts which were named CH1, CH2, and CH3, respectively.
Defined as no complications and no death during the 1-year follow-up period.
Immunosuppression had no specific definition.
Defined as transplant recipients, active cancer, corticosteroid use, or use of other immunomodulatory agents.
AB, antibiotic; AR, arthritis; AT, after treatment initiation; BC, blood culture; Cfz, Cefazolin; CVC, central venous catheter; CX, surgery; DM, diabetes mellitus; EC, endocarditis; Flux, flucloxacillin; HD, hemodialysis; IDU, intravenous drug user; IP, implanted prosthetics; IQR, interquartile range; IS, immunosuppression; MA, metastatic abscesses; MG, meningitis; N, sample size; NR, unreported; OM, osteomyelitis; PC, prospective cohort; PNEU, pneumonia; RC, retrospective cohort; MRSA, methicillin-resistant Staphylococcus aureus; MSSA, methicillin-sensitive S. aureus; SAB, S. aureus bacteremia; SP, spondylodiscitis; SS, severe sepsis; ST, shortened treatment group; UT, treatment group as usual; Vanc, vancomycin.
The duration of the shortened treatment group was <14 days in most studies, but two counted as 14 days in this group25,27 and one counted as short <10 days. 24 The same study classified medium duration as 10–18 days and long duration as >18 days. The most commonly used antibiotic in both groups was vancomycin for MRSA and Flux for MSSA, except in one study 23 where cefazolin was the most commonly used treatment.
Risk of bias
Some studies22–25 did not evaluate confounding factors like patients’ clinical status or comorbidities. In addition, there were clinical and statistically significant differences in basal characteristics between groups, which could have resulted in selection bias. In the study by Taupin et al., 22 there were no differences in variables such as age, sex, sensitivity to oxacillin, and community origin, but there were differences in terms of immunosuppression and the presence of valvular heart disease, which were greater in the long-course group. In the study by Chong et al., 23 no differences were found in both comparisons regarding age, sex, community origin, antibiotic treatment, MRSA isolation, and comorbidities such as diabetes mellitus (DM), immunosuppression, hemodialysis dependence, liver cirrhosis, prosthetic device, among others. The only difference was found in peripheral catheter-related infection, which was greater in the short-course arm. In the study by Evans et al., 24 baseline characteristics were broadly similar between groups using standardized mean differences (SMD) to quantify disparities. The only variables for which the SMD was greater than 10% were estimated glomerular filtration rate (better renal function in patients in the short-course group) and source of infection (more often skin and soft tissue or uncertain in the short-course group). In the study by Zeylemaker et al., 25 comorbidities such as DM, renal failure, and neoplasm were greater in the long-course group.
Some cases of complicated SAB could potentially have been misclassified as low-risk SAB in one study. 26 Two studies22,25 had a risk of bias due to missing data, where 7.8% and 12.3% of the participants, respectively, were excluded because of lack of available data. Finally, the risk of bias associated with the selection of the reported result was observed in one study, where 11 patients were excluded from analysis due to mortality during antibiotic therapy, with some having serious comorbidities. 23
One study was assessed as low risk, 27 four studies as moderate risk,22–24,26 and one as serious risk of bias. 25 The results of the risk of bias assessment are presented in Figure 2.
Figure 2.
Risk of bias assessment.
Mortality at 90 days
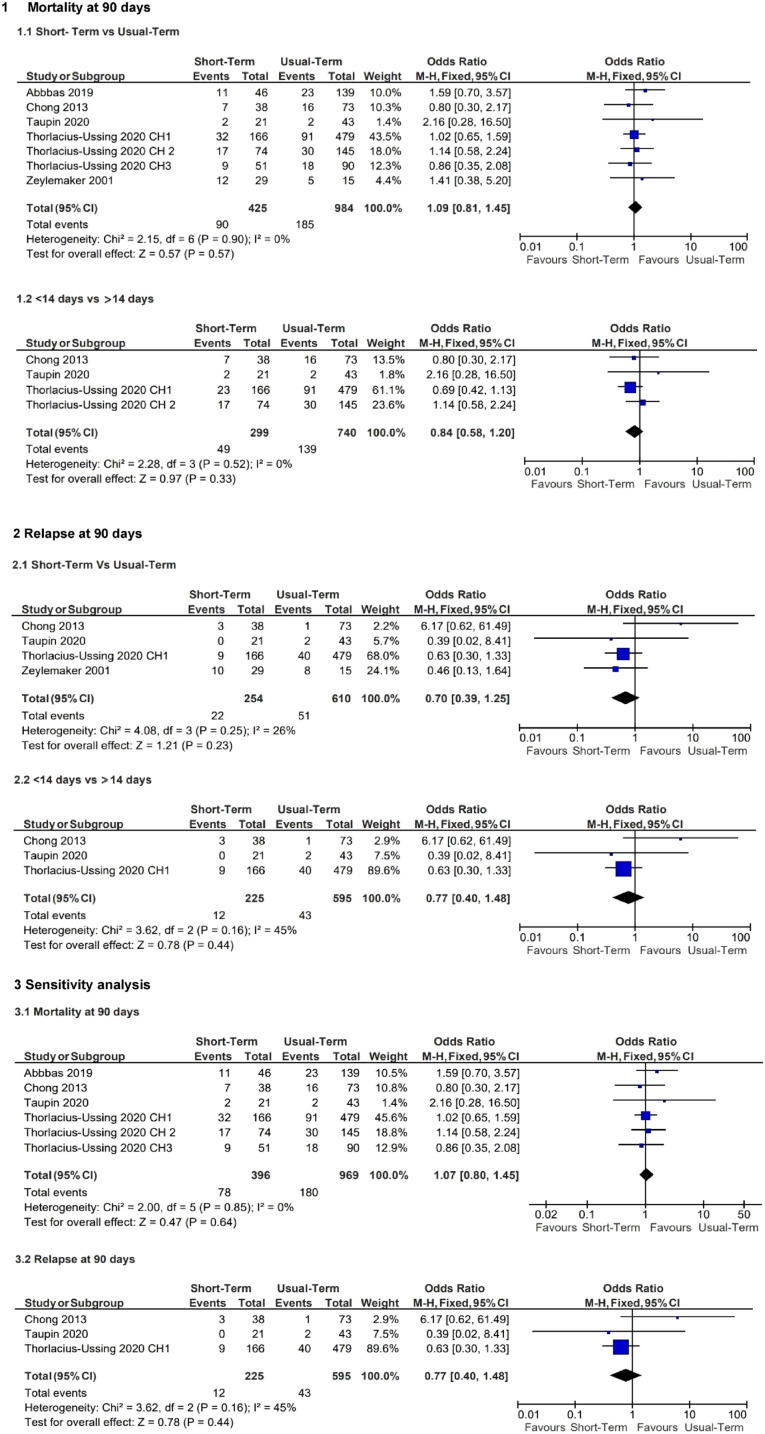
Five trials evaluated this outcome in a total of 1409 patients. An initial meta-analysis was performed using the authors’ definition of abbreviated versus standard therapy (Figure 3) and showed no statistical difference (OR: 1.09, 95% CI, 0.82–1.46, p: 0.55; I2 = 0%). Subsequently, a subgroup analysis was performed defining shortened therapy as <14 days versus ⩾ 14 days, for a total of 3 studies with 1039 patients, also showing no statistical difference (OR: 0.84, 95% CI: 0.59–1.22, p = 0.36; I2 = 0%). Finally, a sensitivity analysis excluding those with a high risk of bias was consistent with the results above (OR: 1.08, 95% CI: 0.80–1.45, p: 0.62; I2 = 0%).
Figure 3.
Forest plot.
30-Day mortality
Only one study 23 evaluated 30-day mortality in a total of 291 patients. Compared with the extended regimens, the shortened course antibiotic had higher mortality (OR: 7.82, 95% CI: 2.10–29.17) but the intermediate course (OR: 1.37, 95% CI: 0.38–4.97) did not reach statistical significance.
Relapse 90 days
This outcome was reported in five of the selected studies, with a total of 864 patients. The initial meta-analysis using the authors’ definition of shortened versus standard therapy (Table 1), yielded an OR of 0.72 (95% CI: 0.31–1.68, p: 0.45; I2 = 26%). Subgroup analysis with a predefined length of therapy <14 days versus ⩾14 days, with three studies and a total of 820 patients found OR 1.03 (95% CI: 0.24–4.41, p: 0.97; I2 = 45%). The sensitivity analysis without the high-risk study showed a similar result (OR: 1.03, 95% CI: 0.24–4.41, p: 0.97; I2 = 45%).
Adverse events
Only one study reported associated adverse events. 22 In the shortened treatment group (<14 days), 5 adverse events occurred in 21 patients, 4 of which were due to Clostridium difficile infection, and 1 was not specified, while in the usual or long therapy group (⩾14 days), 7 events were documented in 43 patients, 1 Clostridium difficile infection and 6 not specified, with no statistically significant differences.
Discussion
The objective of this systematic review was to assess whether shortened versus standard antibiotic treatment differs in terms of mortality, recurrence of bacteremia, or occurrence of adverse events in adults with uncomplicated S. aureus bacteremia. In the six NRS included, we did not find significant differences in the odds of 90-day mortality or 90-day relapse or recurrence of bacteremia with the administration of shorter versus longer regimens. Only one study 22 reported adverse events at follow-up, with no significant differences between the two comparisons. No serious adverse events were reported in the included study.
To our knowledge, this is the first systematic review assessing the safety and efficacy of shortened versus extended antibiotic therapy in adults with uncomplicated S. aureus bacteremia. Regarding 30- and 90-day mortality, our results support the findings of a retrospective cohort study 29 in which there was no difference in survival rate between patients with S. aureus bacteremia treated for more than 14 days compared to those receiving shorter antibiotic courses: That study was not included in our review because it did not differentiate between complicated and not complicated bacteremia but the results were similar. Similarly, in terms of relapse of bacteremia, our findings are in line with evidence from a previous study 30 in which treatment for less than 14 days among patients with a short duration of bacteremia (less than 3 days) did not show an increase in relapse rate of bacteremia where also uncomplicated bacteremia was not well differentiated. It is worth noting that Abbas et al.’s 27 study also evaluated mortality rates by comparing short versus usual duration antibiotic treatments for complicated S. aureus bacteremia. A mortality reduction was observed in the antibiotic treatment regimen lasting >14 days (hazard ratio: 0.32, 95% CI: 0.16–0.64). This finding implies that shorter therapy time may only be suitable for uncomplicated bacteremia.
We could identify a moderate statistical heterogeneity among the studies for the outcome of 90-day relapse of bacteremia (I2 = 45%), with non-significant results (p: 0.97). We believe that these findings may be explained in part by the differences in the proportion of included patients with MRSA, DM status, and hemodialysis requirement in shortened compared to prolonged antibiotic therapy groups among the summarized studies.
An important finding of our review was the differences in the definitions used to classify shortened or prolonged duration of antibiotic therapy between studies. While the definition of standard treatment was more homogeneous among the included studies (14 days or longer) in all included studies except the study by Evans et al. 24 (defined as 10–18 days), in the case of a shortened regimen, it was as short as 7–14 days of antibiotic treatment, with one study 26 being 8–9 days. The fact that there is no uniform and universally accepted definition of what is considered a short or prolonged regimen underscores the need to standardize these definitions in further clinical trials to reduce the heterogeneity of the interventions and comparators evaluated and to draw more precise conclusions on this topic. There is a novel proposal for the classification and management of patients with S. aureus bacteremia to systematically identify those patients at high risk of developing infectious complications and treat them accordingly rather than classifying patients as having complicated or uncomplicated bacteremia. 31 It consists of four steps: an initial risk stratification for the presence of metastatic infection, a diagnostic work-up in search of these infections based on the initial risk assessment, a final clinical diagnosis, and finally a general direction for treatment that can be individualized according to favorable clinical features.
The results of this systematic literature review provide interesting data on the prescription of shorter antibiotic regimens compared with standard therapy in adults with uncomplicated S. aureus bacteremia. However, there are some considerations regarding our findings: First, only one study was assessed as overall low risk of bias. To mitigate this concern, a sensitivity analysis was performed including only low and moderate risk of bias studies, with similar findings. Second, the number of total events was limited, presented in 77 out of 864 patients for relapse of bacteremia at 90 days and in 266 out of 1409 patients for 90-day mortality. This could explain a potential insufficient precision of the analyzed outcomes, which implies the need to generate studies with a greater sample size to corroborate our results. Third, among the included studies, we could identify little representation of patients with chronic and debilitating diseases such as DM, chronic renal failure, and immunosuppressive states, which may contribute to the prognosis of bacteremia, It is important to mention that in some studies,22–25 there were differences in basal clinical characteristics (DM, immunosuppression, renal failure) that could have contributed to selection bias. The difficulties in the lack of appropriate description of these variables could be explained by the retrospective nature of the included studies.
A limitation was that relatively few studies were available to be included in this meta-analysis, and some outcomes could only include a subset of these, which may limit our conclusions. Future studies are needed to overcome this limitation. Currently, there is an ongoing randomized, non-blinded interventional study (NCT03514446) that aims to evaluate if 7 days of antibiotic treatment in patients with uncomplicated S. aureus bacteremia is non-inferior to 14 days of treatment, which results may contribute to determining the best time duration of antibiotic therapy in these population. Nowadays, there is an urgent need for well-designed, controlled clinical trials with a robust sample size, evaluating clinical and microbiologic outcomes associated with S. aureus uncomplicated bacteremia to elucidate the best course of action regarding the optimal duration of therapy.
Conclusion
In summary, considering that no differences were found in the outcomes of 90-day mortality, 90-day relapse or recurrence of bacteremia, or the frequency of adverse events according to the length of therapy comparing shorter regimens with prolonged antibiotic therapy, shorter duration regimens could be considered as an alternative option in individual low-risk cases. Some of the included studies had important limitations regarding bias due to confounding factors and showed an imbalance in basal clinical characteristics such as DM, immunosuppression, and renal failure. This population remains underrepresented in the studies, which limits the generalizability of the results in these conditions. Because of the risk of bias, imprecision for several of the outcomes analyzed, and the few studies taken into account in this study, the benefits and harms of using shorter treatment regimens for uncomplicated S. aureus bacteremia should be analyzed with caution and on an individual basis.
Supplemental Material
Supplemental material, sj-docx-1-tai-10.1177_20499361241237615 for Short-term versus usual-term antibiotic treatment for uncomplicated Staphylococcus aureus bacteremia: a systematic review and meta-analysis by Santiago Grillo Perez, Candida Diaz-Brochero, Javier Ricardo Garzon Herazo and Oscar Mauricio Muñoz Velandia in Therapeutic Advances in Infectious Disease
Supplemental material, sj-docx-2-tai-10.1177_20499361241237615 for Short-term versus usual-term antibiotic treatment for uncomplicated Staphylococcus aureus bacteremia: a systematic review and meta-analysis by Santiago Grillo Perez, Candida Diaz-Brochero, Javier Ricardo Garzon Herazo and Oscar Mauricio Muñoz Velandia in Therapeutic Advances in Infectious Disease
Supplemental material, sj-docx-3-tai-10.1177_20499361241237615 for Short-term versus usual-term antibiotic treatment for uncomplicated Staphylococcus aureus bacteremia: a systematic review and meta-analysis by Santiago Grillo Perez, Candida Diaz-Brochero, Javier Ricardo Garzon Herazo and Oscar Mauricio Muñoz Velandia in Therapeutic Advances in Infectious Disease
Acknowledgments
None.
Footnotes
ORCID iDs: Santiago Grillo Perez  https://orcid.org/0000-0002-3457-565X
https://orcid.org/0000-0002-3457-565X
Candida Diaz-Brochero  https://orcid.org/0000-0002-2176-7388
https://orcid.org/0000-0002-2176-7388
Supplemental material: Supplemental material for this article is available online.
Contributor Information
Santiago Grillo Perez, Department of Internal Medicine, Hospital Universitario San Ignacio, Carrera 7 No 40-62, 7th Floor, Bogotá 110231, Colombia; School of medicine, Pontifical Xavierian University, Carrera 7 No 40-62, 8th Floor, Bogotá 110231, Colombia.
Candida Diaz-Brochero, Pontifical Xavierian University, Bogotá, Colombia; Department of Internal Medicine, Hospital Universitario San Ignacio, Bogotá, Colombia; Infectious Diseases Unit, Hospital Universitario San Ignacio, Bogotá, Colombia.
Javier Ricardo Garzon Herazo, Pontifical Xavierian University, Bogotá, Colombia; Department of Internal Medicine, Hospital Universitario San Ignacio, Bogotá, Colombia; Infectious Diseases Unit, Hospital Universitario San Ignacio, Bogotá, Colombia.
Oscar Mauricio Muñoz Velandia, Pontifical Xavierian University, Bogotá, Colombia; Department of Internal Medicine, Hospital Universitario San Ignacio, Bogotá, Colombia.
Declarations
Ethics approval and consent to participate: The present study is subject to the ethical norms of Helsinki and the Scientific, Technical, and Administrative Norms for Health Research Resolution No. 008430 of 1993. According to the same resolution, it is considered research without risk. The confidentiality and reserve of the data obtained were maintained through secure databases.
Consent for publication: Not applicable.
Author contributions: Santiago Grillo Perez: Conceptualization; Data curation; Formal analysis; Investigation; Writing – original draft; Writing – review & editing.
Candida Diaz-Brochero: Conceptualization; Data curation; Formal analysis; Investigation; Writing – original draft; Writing – review & editing.
Javier Ricardo Garzon Herazo: Supervision; Writing – review & editing.
Oscar Mauricio Muñoz Velandia: Data curation; Formal analysis; Methodology; Supervision; Writing – original draft; Writing – review & editing.
Funding: The authors received no financial support for the research, authorship, and/or publication of this article.
The authors declare that there is no conflict of interest.
Availability of data and materials: All the material is owned by the authors.
References
- 1. Mun SJ, Kim SH, Kim HT, et al. The epidemiology of bloodstream infection contributing to mortality: the difference between community-acquired, healthcare-associated, and hospital-acquired infections. BMC Infect Dis 2022; 22: 336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Cisterna R, Ezpeleta C, Sota M. Epidemiology of bacteremia and of nosocomial infections. Clinical Microbiology and Infection. 1997; 3: 209–213. [Google Scholar]
- 3. Inagaki K, Lucar J, Blackshear C, et al. Methicillin-susceptible and methicillin-resistant Staphylococcus aureus bacteremia: nationwide estimates of 30-day readmission, in-hospital mortality, length of stay, and cost in the United States. Clin Infect Dis 2019; 69: 2112–2118. [DOI] [PubMed] [Google Scholar]
- 4. Thampi N, Showler A, Burry L, et al. Multicenter study of health care cost of patients admitted to hospital with Staphylococcus aureus bacteremia: impact of length of stay and intensity of care. Am J Infect Control 2015; 43: 739–744. [DOI] [PubMed] [Google Scholar]
- 5. Tamma PD, Avdic E, Li DX, et al. Association of adverse events with antibiotic use in hospitalized patients. JAMA Intern Med 2017; 177: 1308–1315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Mo Y, Oonsivilai M, Lim C, et al. Implications of reducing antibiotic treatment duration for antimicrobial resistance in hospital settings: a modelling study and meta-analysis. PLoS Med 2023; 20: e1004013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Turjeman A, von Dach E, Molina J, et al. Duration of antibiotic treatment for Gram-negative bacteremia – systematic review and individual participant data (IPD) meta-analysis. EClinicalMedicine 2023; 55: 101750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Opmeer BC, El Moussaoui R, Bossuyt PMM, et al. Costs associated with shorter duration of antibiotic therapy in hospitalized patients with mild-to-moderate severe community-acquired pneumonia. J Antimicrob Chemother 2007; 60: 1131–1136. [DOI] [PubMed] [Google Scholar]
- 9. Dinh A, Ropers J, Duran C, et al. Discontinuing β-lactam treatment after 3 days for patients with community-acquired pneumonia in non-critical care wards (PTC): a double-blind, randomised, placebo-controlled, non-inferiority trial. Lancet 2021; 397: 1195–1203. [DOI] [PubMed] [Google Scholar]
- 10. Drekonja DM, Trautner B, Amundson C, et al. Effect of 7 vs 14 days of antibiotic therapy on resolution of symptoms among afebrile men with urinary tract infection: a randomized clinical trial. JAMA 2021; 326: 324–331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Barrasa-Villar JI, Aibar-Remón C, Prieto-Andrés P, et al. Impact on morbidity, mortality, and length of stay of hospital-acquired infections by resistant microorganisms. Clin Infect Dis 2017; 65: 644–652. [DOI] [PubMed] [Google Scholar]
- 12. Bae M, Jeong Y, Bae S, et al. Short versus prolonged courses of antimicrobial therapy for patients with uncomplicated Pseudomonas aeruginosa bloodstream infection: a retrospective study. J Antimicrob Chemother 2021; 77: 223–228. [DOI] [PubMed] [Google Scholar]
- 13. Giannella M, Pascale R, Toschi A, et al. Treatment duration for Escherichia coli bloodstream infection and outcomes: retrospective single-centre study. Clin Microbiol Infect 2018; 24: 1077–1083. [DOI] [PubMed] [Google Scholar]
- 14. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis 2011; 52: e18–e55. [DOI] [PubMed] [Google Scholar]
- 15. Jensen AG, Wachmann CH, Espersen F, et al. Treatment and outcome of Staphylococcus aureus bacteremia: a prospective study of 278 cases. Arch Intern Med 2002; 162: 25–32. [DOI] [PubMed] [Google Scholar]
- 16. Eichenberger EM, Fowler VG, Holland TL. Duration of antibiotic therapy for Staphylococcus aureus bacteremia: the long and the short of it. Clin Microbiol Infect 2020; 26: 536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 2019; 366: 14898. [DOI] [PubMed] [Google Scholar]
- 18. Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016; 355: i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Review Manager 5 (RevMan 5) [Computer program]. Version 5.4. Copenhagen: The Cochrane Collaboration, 2020. https://training.cochrane.org/system/files/uploads/protected_file/RevMan5.4_user_guide.pdf [Google Scholar]
- 20. Valentine JC, Thompson SG. Issues relating to confounding and meta-analysis when including non-randomized studies in systematic reviews on the effects of interventions. Res Synth Methods 2013; 4: 26–35. [DOI] [PubMed] [Google Scholar]
- 21. Deeks JJ, Higgins JPT, Altman DG. (eds). Chapter 10: Analysing data and undertaking meta-analyses. In: Higgins JPT, Thomas J, Chandler J, et al. (eds) Cochrane handbook for systematic reviews of interventions version 6.4 (updated August 2023). Cochrane, 2023. www.training.cochrane.org/handbook [Google Scholar]
- 22. Taupin D, Karchmer AW, Davis RB, et al. Uncomplicated Staphylococcus aureus bacteremia treatment duration and outcomes at an academic medical center. Open Forum Infect Dis 2020; 7: 1–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Chong YP, Moon SM, Bang KM, et al. Treatment duration for uncomplicated Staphylococcus aureus bacteremia to prevent relapse: analysis of a prospective observational cohort study. Antimicrob Agents Chemother 2013; 57: 1150–1156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Evans RN, Harris J, Rogers CA, et al. The effect of duration of therapy for treatment of Staphylococcus aureus blood stream infection: an application of cloning to deal with immortal-time bias in an analysis of data from a cohort study (BSI-FOO). J Antimicrob Chemother 2023; 78: 196–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Zeylemaker MMP, Jaspers CAJJ, Van Kraaij MGJ, et al. Long-term infectious complications and their relation to treatment duration in catheter-related Staphylococcus aureus bacteremia. Eur J Clin Microbiol Infect Dis 2001; 20: 380–384. [DOI] [PubMed] [Google Scholar]
- 26. Thorlacius-Ussing L, Sandholdt H, Nissen J, et al. Comparable outcomes of short-course and prolonged-course therapy in selected cases of methicillin-susceptible Staphylococcus aureus bacteremia: a pooled cohort study. Clin Infect Dis 2021; 73: 866–872. [DOI] [PubMed] [Google Scholar]
- 27. Abbas M, Rossel A, de Kraker MEA, et al. Association between treatment duration and mortality or relapse in adult patients with Staphylococcus aureus bacteraemia: a retrospective cohort study. Clin Microbiol Infect 2020; 26: 626–631. [DOI] [PubMed] [Google Scholar]
- 28. Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ; 339: 332–336. [PMC free article] [PubMed] [Google Scholar]
- 29. Fätkenheuer G, Preuss M, Salzberger B, et al. Long-term outcome and quality of care of patients with Staphylococcus aureus bacteremia. Eur J Clin Microbiol Infect Dis 2004; 23: 157–162. [DOI] [PubMed] [Google Scholar]
- 30. Johnson LB, Almoujahed MO, Ilg K, et al. Staphylococcus aureus bacteremia: compliance with standard treatment, long-term outcome and predictors of relapse. Scand J Infect Dis 2003; 35: 782–789. [DOI] [PubMed] [Google Scholar]
- 31. Kouijzer IJE, Fowler VG, Ten Oever J. Review redefining Staphylococcus aureus bacteremia: a structured approach guiding diagnostic and therapeutic management. J Infect 2023; 86: 9–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-docx-1-tai-10.1177_20499361241237615 for Short-term versus usual-term antibiotic treatment for uncomplicated Staphylococcus aureus bacteremia: a systematic review and meta-analysis by Santiago Grillo Perez, Candida Diaz-Brochero, Javier Ricardo Garzon Herazo and Oscar Mauricio Muñoz Velandia in Therapeutic Advances in Infectious Disease
Supplemental material, sj-docx-2-tai-10.1177_20499361241237615 for Short-term versus usual-term antibiotic treatment for uncomplicated Staphylococcus aureus bacteremia: a systematic review and meta-analysis by Santiago Grillo Perez, Candida Diaz-Brochero, Javier Ricardo Garzon Herazo and Oscar Mauricio Muñoz Velandia in Therapeutic Advances in Infectious Disease
Supplemental material, sj-docx-3-tai-10.1177_20499361241237615 for Short-term versus usual-term antibiotic treatment for uncomplicated Staphylococcus aureus bacteremia: a systematic review and meta-analysis by Santiago Grillo Perez, Candida Diaz-Brochero, Javier Ricardo Garzon Herazo and Oscar Mauricio Muñoz Velandia in Therapeutic Advances in Infectious Disease