Abstract
Background:
Celecoxib is a COX-2 nonsteroidal anti-inflammatory drug (NSAID). It is widely used for the treatment of osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis.
Objective:
This study aimed to explore the effect of long-term administration of celecoxib on kidney of male albino rats, and to study the potential effect of treatment discontinuation on such tissues. The study also examined the alleged ameliorative effect of royal jelly (RJ).
Methods:
Fifty, male albino rats were divided into 5 equal groups; 10 each. Group 1: rats received no drug (control group). Group 2: rats received celecoxib (50 mg/kg/day, orally for 30 successive days). Group 3: rats received celecoxib (50 mg/kg/day, orally) and royal jelly (300 mg/kg/day, orally) for 30 successive days. Group 4: rats received celecoxib for 30 successive days, then rats were left untreated for another 30 days. Group 5: rats received celecoxib and RJ for 30 successive days, then rats were left untreated for another 30 days.
Results:
Long-term celecoxib administration caused significant elevation in kidney function tests, with ameliorative effects of RJ against celecoxib-induced renal toxicity.
Conclusion:
Long-term celecoxib administration caused renal toxicity in male albino rats, with ameliorative effects of RJ.
Keywords: celecoxib, kidney, renal, royal jelly, toxicity
Résumé
Contexte:
Le célécoxib est un anti-inflammatoire non stéroïdien (AINS) inhibiteur de COX-2. Ce médicament est largement utilisé pour le traitement symptomatique de l’arthrose, de la polyarthrite rhumatoïde et de la spondylarthrite ankylosante.
Objectifs:
Cet essai visait à examiner l’effet d’une administration à long terme de célécoxib sur les reins de rats albinos mâles, à étudier les possibles effets de l’arrêt du traitement sur ces tissus et à vérifier l’effet d’amélioration allégué de la gelée royale.
Méthodologie:
Cinquante rats albinos mâles ont été répartis en cinq groupes égaux (10 rats par groupe). Groupe 1 (groupe témoin): rats n’ayant reçu aucun médicament. Groupe 2: rats ayant reçu du célécoxib (50 mg/kg/jour, par voie orale pendant 30 jours consécutifs). Groupe 3: rats ayant reçu du célécoxib (50 mg/kg/jour, par voie orale) et de la gelée royale (300 mg/kg/jour, par voie orale) pendant 30 jours consécutifs. Groupe 4: rats ayant reçu du célécoxib pendant 30 jours consécutifs, puis laissés sans traitement pendant 30 jours supplémentaires. Groupe 5: rats ayant reçu du célécoxib et de la gelée royale pendant 30 jours consécutifs, puis laissés sans traitement pendant 30 jours supplémentaires.
Résultats:
L’administration à long terme de célécoxib a entraîné une augmentation significative des tests de la fonction rénale; la gelée royale a montré des effets d’amélioration contre la toxicité rénale induite par le célécoxib.
Conclusion:
L’administration à long terme de célécoxib a provoqué une toxicité rénale chez les rats albinos mâles contre laquelle la gelée royale a montré des effets protecteurs.
Introduction
Celecoxib is a COX-2 nonsteroidal anti-inflammatory drug (NSAID). 1 It is widely used for the treatment of osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis.2,3 Celecoxib is a prospective alternative for traditional NSAIDs as it possesses anti-inflammatory, antipyretic, and analgesic properties with excellent gastrointestinal safety profile. 4 Celecoxib’s side effects still need to be further researched. Studies on celecoxib’s impact on the kidneys produced a wide range of findings. While some research verified that celecoxib is safe for the kidneys, 5 other studies even went as far as to claim that celecoxib has a protective impact on the kidneys. 6 On the contrary, some research suggests that celecoxib may be hazardous to the kidneys. 7
Royal jelly (RJ) is a nutrient-rich source of bioactive chemicals that are crucial to numerous biological activities, 8 including antibacterial, antioxidant, anti-inflammatory, anti-cancer, anti-hyperlipidemic, cardio-protective, and hepatorenal-protective health benefits. 9 The renal protection is assumed to be facilitated by free radical scavenging, antioxidant capacities, and anti-apoptotic activation of RJ.10,11
The stark discrepancy between the findings of several studies addressing celecoxib’s impact on the kidney served as a strong impetus for us to conduct this investigation in an effort to shed as much light as possible on the situation as feasible. This study was created to examine the effects of long-term celecoxib administration on renal tissue in male albino rats and to investigate the potential effects of treatment termination, with a focus on the potential effects of RJ administration.
Materials and Methods
Experimental Animals
Fifty, apparently healthy, male albino rats weighing 200 to 230 gm, randomly divided into 5 equal groups; each of 10 rats. They were kept in orderly cages with walls made of wire-bottomed galvanized metal. A healthy food and unrestricted access to clean drinking water were provided. They had a 7-day acclimatization period before starting the study.
Experimental Setting and Design
This study was conducted in the Lab animal unit, Pharmacology department, Faculty of Veterinary Medicine, Zagazig University, in the period from March 2023 to May 2023. All steps and procedures in the study were carried out following Zagazig University Institutional Animal Care and Use Committee regulations, with approval No. ZU-IACUC/2/F/522/2023.
Rats used in this study were allocated into the following groups:
Control group (Group 1, n = 10): Rats did not receive any medication. Rats were employed as the control group.
Celecoxib group (Group 2, n = 10): For 30 consecutive days, rats were given celecoxib (50 mg/kg/day, orally). 12
Celecoxib + RJ group (Group 3, n = 10): For 30 consecutive days, rats were given celecoxib plus RJ (300 mg/kg/day, orally). 9
Celecoxib recovery group (Group 4, n = 10): For 30 consecutive days, rats were given celecoxib, then rats were left untreated for another 30 days.
Celecoxib + RJ recovery group (Group 5, n = 10): For 30 consecutive days, rats were given celecoxib plus RJ, then rats were left untreated for another 30 days,
At the end, all group rats were euthanized and samples were taken.
Samples Collection and Preservation
Blood samples were collected using a 3 mL syringe directly from the ventricular puncture of rats into centrifuge tubes and left to clot for 15 minutes at room temperature, then centrifuged at 3000 rpm for 10 minutes to allow serum separation, which was then aspirated into cryovials and stored at −20°C for serum biochemical assays.
The kidney tissues of each animal were dissected and collected immediately, transferred in liquid nitrogen, and kept at −80°C for total RNA extraction used for the determination of apoptotic and anti-apoptotic mRNA expression levels using reverse transcription polymerase chain reaction (RT-PCR). 13
Kidney Function Tests
Blood urea nitrogen (BUN) 14 and creatinine concentrations 15 were estimated via using commercially available Specific Pointe® Scientific Inc. colorimetric kits.
Oxidant/Antioxidant Status
Serum malondialdehyde (MDA) and superoxide dismutase (SOD) 16 were estimated via using commercially available kits supplied from Oxi Select™, USA.
Apoptotic and Anti-Apoptotic Genes Expression
Apoptotic genes (Bax) and anti-apoptotic gene (Bcl-2) 13 were estimated via using commercially available kits supplied from TOPreal™ qPCR 2X PreMIX (SYBR Green with low ROX) (Cat. # P725, Enzynomics, Korea).
The primer sequences 13 ;
Bax: (Size: 109 bp, Accession no. NM_017059.2)
5′-CGAATTGGCGATGAACTGGA-3′ (forward)
5′-CAAACATGTCAGCTGCCACAC-3′ (reverse);
Bcl-2: (Size: 135 bp, Accession no. NM_016993.1)
5′-GACTGAGTACCTGAACCGGCATC-3′ (forward)
5′-CTGAGCAGCGTCTTCAGAGACA-3′ (reverse);
Gene expressions were measured using the below formula and Ct (2–ΔΔCt) (fold change) method:
ΔΔCt = (Cttarget–Ctreference) test sample–(Cttarget–Ctreference) control sample
Finally, considering the primer efficiency value of ~2, the gene expression level was determined as 1–ΔΔCt.
Statistical Analysis
The Statistical Package for Social Sciences (SPSS) program (version 26.0; SPSS Inc., Illinois) for Microsoft Windows® was used to statistically analyze the collected data. The means of various groups were compared using one-way analysis of variance (ANOVA). The in-between group comparisons were performed using Tukey honestly significant difference (HSD) post hoc analysis. A P value of .05 or less is regarded as significant.
Results
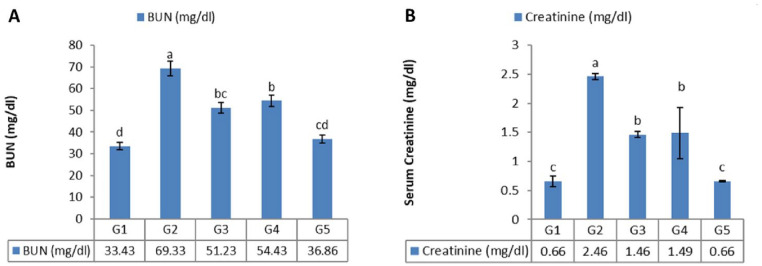
In the study rats, acute kidney injury was emerged by oral administration of celecoxib (50 mg/kg/day) for 30 consecutive days. This injury was allegedly characterized by a substantial rise in serum BUN and creatinine levels (P < .05), with RJ acting as an anti-nephrotoxic agent. When celecoxib and RJ were discontinued together, the effects of celecoxib’s nephrotoxicity were greatly reduced, and the BUN and creatinine levels returned to normal (Figure 1).
Figure 1.
Effects of celecoxib (50 mg/kg/day, orally), royal jelly (300 mg/kg/day, orally), and treatments discontinuation on kidney profile in male albino rats. A: BUN (mg/dL), B: creatinine (mg/dL). Data are expressed as mean ± SD, n = 10/group.
Group 1 received no drug (control). Group 2 received celecoxib (50 mg/kg/day, orally), for 30 successive days. Group 3 received celecoxib (50 mg/kg/day, orally) and royal jelly (300 mg/kg/day, orally) for 30 successive days. Group 4 received celecoxib (50 mg/kg/day, orally) for 30 successive days, then rats were left untreated for another 30 days. Group 5 received celecoxib (50 mg/kg/day, orally) and royal jelly (300 mg/kg/day, orally) for 30 successive days, then rats were left untreated for another 30 days.
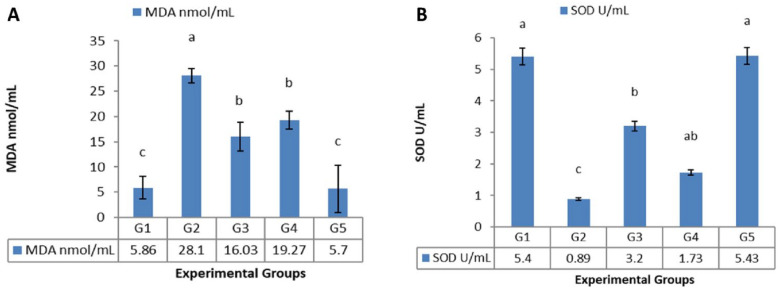
Celecoxib administration (50 mg/kg/day, orally), for 30 successive days was claimed to cause oxidative stress (OS) the study rats as manifested by significant elevation of serum MDA and significant decline in SOD levels, with ameliorative effects of RJ against celecoxib-induced OS. Celecoxib discontinuation significantly diminished the celecoxib-induced OS effects, and normal oxidative enzyme levels were regained in the case of dual medications (celecoxib + RJ) discontinuation (Figure 2).
Figure 2.
Effects of celecoxib (50 mg/kg/day, orally), royal jelly (300 mg/kg/day, orally), and treatments discontinuation on serum oxidative status profile in male albino rats, A: MDA (nmol/mL), B: SOD (U/mL). Data are expressed as mean ± SD, n = 10/group.
Group 1 received no drug (control). Group 2 received celecoxib (50 mg/kg/day, orally), for 30 successive days. Group 3 received celecoxib (50 mg/kg/day, orally) and royal jelly (300 mg/kg/day, orally) for 30 successive days. Group 4 received celecoxib (50 mg/kg/day, orally) for 30 successive days, then rats were left untreated for another 30 days. Group 5 received celecoxib (50 mg/kg/day, orally) and royal jelly (300 mg/kg/day, orally) for 30 successive days, then rats were left untreated for another 30 days.
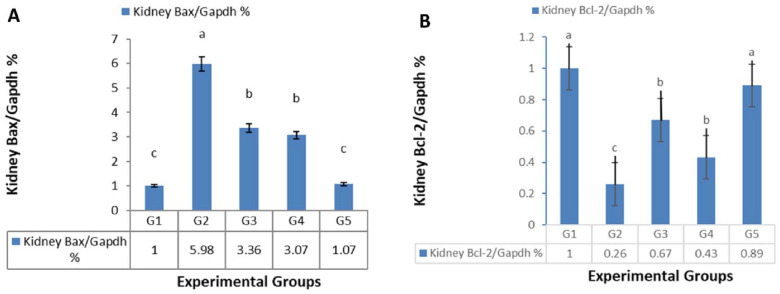
In the study rats, administration of celecoxib (50 mg/kg/day, orally) for 30 consecutive days was said to cause an apoptotic effect as evidenced by a significant increase in serum levels of the apoptotic gene Bax and a significant decrease in serum levels of the anti-apoptotic gene Bcl-2 in kidneys. Royal jelly was said to have protective effects against the celecoxib-induced apoptotic effect. Celecoxib withdrawal greatly reduced the celecoxib-induced apoptotic impact, and withdrawal of both drugs (celecoxib + RJ) restored normal expression of the apoptotic/anti-apoptotic genes (Figure 3).
Figure 3.
Effects of celecoxib (50 mg/kg/day, orally), royal jelly (300 mg/kg/day, orally), and treatments discontinuation on A: renal Bax (%), B: renal Bcl-2 (%). Data are expressed as mean ± SD, n = 10/group.
Group 1 received no drug (control). Group 2 received celecoxib (50 mg/kg/day, orally), for 30 successive days. Group 3 received celecoxib (50 mg/kg/day, orally) and royal jelly (300 mg/kg/day, orally) for 30 successive days. Group 4 received celecoxib (50 mg/kg/day, orally) for 30 successive days, then rats were left untreated for another 30 days. Group 5 received celecoxib (50 mg/kg/day, orally) and royal jelly (300 mg/kg/day, orally) for 30 successive days, then rats were left untreated for another 30 days.
Discussion
Celecoxib is a prospective alternative for the treatment of osteoarthritis and rheumatoid arthritis, 17 especially in individuals at high risk of gastrointestinal problems, due to its excellent gastrointestinal safety profile and long-term symptomatic improvement. 18
In our study, celecoxib administration (50 mg/kg/day, orally), for 30 successive days caused acute kidney injury in the study rats with ameliorative effects of RJ against celecoxib-induced nephrotoxicity.
In fact, both COX-1 and COX-2 are expressed in the kidney. 19 COX-1, which acts primarily in the regulation of renal hemodynamics and glomerular filtration rate (GFR), is expressed mainly as a constitutive isoform in normal conditions. 20 On the other hand, COX-2, which is responsible for salt and water excretion, is expressed not only initially as an inducible isoform in the presence of damaging stimuli, 21 but also constitutively for ensuring the tubuloglomerular feedback, contributing to the establish homeostasis. 22 Therefore, the impact of blocking one or both of these enzymes on renal functions is the consequence of inhibiting prostaglandin synthesis responsible for the maintenance of renal functions23,24 which can result in acute renal failure. Moreover, there is the possibility that long-term administration of any NSAIDs can cause chronic renal failure in some patients. 25
Selective COX-2 inhibitors, such as celecoxib, were developed, primarily, to produce the beneficial effects of NSAIDs, but spare the COX-1-mediated adverse events. 26 However, it has the same potential for adverse renal effects as traditional NSAIDs, 27 even in therapeutic doses, 28 especially in clinical situations associated with a renal impairment, such as sodium depletion, hypovolemia, cirrhosis, congestive heart failure, nephrotic syndrome, chronic kidney disease (CKD), 23 and old age. 29 Although there have been 2 cases of celecoxib-induced nephrotoxicity after 10 months of continuous usage, it has largely been also reported with short-term use, as fast as within 2 weeks. 30 Therefore, the same precautions in patients at risk for adverse renal effects probably apply to both the nonselective NSAIDs and COX-2 selective inhibitors. 19 The short duration of administration and close monitoring of renal functions, urine output, and fluid status are essential to avoid acute kidney injury because of celecoxib.31,32
The most frequent renal side effect associated with celecoxib administration is reversible renal insufficiency.33-36 In the early 1900s, BUN quantification replaced urea and served as a superior renal function biomarker. 37 Serum creatinine supplanted BUN in the mid-1900s and remains the “gold standard” laboratory test for the assessment of kidney function and GFR until now. 38 As celecoxib treatment continues, it may cause acute renal injury39,40 manifested by an abrupt reduction in kidney function: oliguria of 0.5 mL/kg/h for > 6 hours, increase in serum creatinine of either ≥ 0.3 mg/dL, or 1.5-fold from baseline. 41 Progressive functional or structural deterioration in renal tissue throughout a period of months or years is known as CKD42,43 manifested by a < 60 mL/min per 1.73 m2 reduction in glomerular filtration rate.44,45
The biochemical celecoxib-induced nephrotoxicity is attributed to OS where the balance between the generation of reactive oxygen species (ROS) and antioxidants defense is upset. 46 The OS pathophysiology is an increase in ROS levels with unbalanced extracellular antioxidant enzyme activity.40,47
In accordance with our findings, previous studies have demonstrated that the renal adverse effects of celecoxib are usually reversible. Most forms of acute renal failure from NSAID administration are short-term and reversible upon NSAID discontinuation. 48 Nonsteroidal anti-inflammatory drug administration for the short term for up to 6 weeks may preserve the chance for recovery; however, there has previously been no study to test the reversibility of renal adverse effects after long-term NSAID use. 49 Renal function regained following ceasing celecoxib medication after 2 weeks. 50
Royal jelly increased renal COX-2 protein expression and prostaglandin E2 (PGE-2) renal content, providing protection against celecoxib toxicity. It decreased iNOS protein expression, renal myeloperoxidase (MPO) levels, and increased ROS scavenging, inhibiting inflammatory and pro-inflammatory responses, reducing kidney damage and oxidative damage. 9 Prior studies described the protective effects of RJ against cisplatin-induced,51,52 doxorubicin- induced, 53 ethylene glycol-induced, 11 diclofenac-induced, 9 tyrosine kinase inhibitor-Induced, 54 fluoride-induced, 55 valproic acid-induced, 56 and CCl4-induced 57 renal toxicities in rats.
In our study, celecoxib administration (50 mg/kg/day, orally), for 30 successive days caused OS in the study rats as manifested by significant elevation of serum MDA and significant decline in SOD levels, with ameliorative effects of RJ against celecoxib-induced OS.
Oxidative stress is a balance shift between oxidant production and elimination through the antioxidant defense system. 58 Oxidative stress-mediated molecules are metabolites derived from ROS 59 and reactive nitrogen species (RNS). 60 Reactive oxygen species/reactive nitrogen species are produced from endogenous sources like immune system activation, inflammation, and mental stress61,62 while exogenous sources include air pollution, water pollution, alcohol, and ultraviolet radiation. 63 Antioxidants are the body’s primary defense against OS, inhibiting the oxidation reaction of molecules that produce free radicals. 64 They can be internally synthesized (endogenous) or externally supplied through foods like RJ, which acts as direct ROS scavengers and increase the antioxidant enzyme activities. 65 Endogenous antioxidant defenses involve a network of antioxidant enzymatic and non-enzymatic molecules. 66 Primary antioxidant enzymes involve SOD, catalase, and several peroxidases. Malondialdehyde is the end- product of polyunsaturated fatty acids oxidation in cellular membranes, thus, it acts as a dependable marker of OS. 67 and non-enzymatic molecules involve vitamins (A, C, E, and K), enzyme cofactors (Q10), and minerals (Zn, Mn, Cu, Se, etc). 68
Previous studies show that rats treated with celecoxib exhibit altered oxidant/antioxidant status, leading to excessive generation of free radicals, which cause disease development, peroxidation, and tissue destruction.69-72 In addition, previous studies have confirmed the beneficial and antioxidant effects of RJ,73-75 with in vitro administration inhibiting pro-inflammatory cytokines and suggesting anti-inflammatory and antioxidant properties. 76 These properties are attributed to free radical scavengers, inhibiting lipid peroxidation and cytochrome P450 expression, and reducing lipid peroxidation.77-79
In our study, celecoxib administration (50 mg/kg/day, orally), for 30 successive days induced an apoptotic effect in the study rats as manifested by a significant elevation of renal tissue apoptotic gene (Bax) and a significant decline in renal tissue anti-apoptotic gene (Bcl-2), with ameliorative effects of RJ against the celecoxib-induced apoptotic effect.
Apoptosis is a genetically determined process that involves the elimination of cells, regulated by 2 main proteins: caspases and the Bcl-2 family. 80 Caspases control cell degradation with minimal effect on surrounding tissues, 81 while Bcl-2 consists of anti-apoptotic and pro-apoptotic members. Anti-apoptotic members, such as Bcl-2 and Bcl-XL, sequester caspases or prevent the release of mitochondrial apoptogenic factors responsible for caspases activation, while pro-apoptotic members, such as Bax and Bak, trigger caspases by inducing the release of these factors. 82
Apoptosis is a physiological process that selectively eliminates individual cells without harming the entire organ. 83 It involves cell shrinkage, pyknosis, cytoplasmic and nuclear condensation, chromatin cleavage, apoptotic bodies formation, and phagocytosis. 84 Necrosis, on the other hand, is a passive, accidental cell death triggered by external factors or disease.85,86 The main morphological changes associated with necrosis include cell swelling, cytoplasmic vacuole formation, distended endoplasmic reticulum, cytoplasmic bleb formation, condensed, swollen, or ruptured mitochondria, disrupted organelle membranes, swollen and ruptured lysosomes, and ultimately disruption of the cell membrane. 87 Apoptosis is usually normal and beneficial, and can also occur as a protective process. 88
In accordance with our findings, previous studies have confirmed celecoxib-induced apoptosis. Celecoxib causes apoptosis by causing the loss of the mitochondrial transmembrane potential, the release of cytochrome c and apoptosis inducing factor (AIF), and the activation of caspase-9 and caspase-3. In addition, the anti-apoptotic protein Bcl-2 was reduced in abundance whereas the pro-apoptotic protein Bax was enhanced by celecoxib. The data showed that mitochondria-dependent signaling, not PPAR/NF-B signaling, was the mechanism through which celecoxib triggered apoptosis in mouse liver cancer cells. 89 In another study, celecoxib-induced apoptosis in 5-fluorouracil-resistant gastric cancer cells through protein kinase B (PKB) inhibition, 90 which is a key component of the phosphatidyl-inositol-3 kinase (PI3K) intracellular pathway that exerts a pivotal role in regulating cell proliferation, survival, and metabolism. 91 Celecoxib-induced apoptosis in glioblastoma tumor cells, the primary malignant tumor of the brain, via suppressing CIP2A/PP2A/Akt signaling axis. 92
Several previous studies confirmed the concept of the beneficial and anti-apoptotic effect of RJ against cisplatin-induced hepatorenal toxicity, 52 nicotine-induced testicular injury in mice, 93 doxorubicin-induced nephrotoxicity in male albino rats, 10 and hydroxyurea-induced hepatic injury in rats. 94 Moreover, RJ decreases the expression of the apoptotic gene (MMP-9) responsible for bladder cancer in humans. 95
The limitation of our study was the relatively small sample size, study performing on male rats only excluding the female rats, and focusing on a single dose of medication. These issues should be addressed in future studies.
Conclusion
Long-term celecoxib administration caused renal toxicity in male albino rats, with ameliorative effects of RJ against celecoxib-induced oxidative and apoptotic stress.
Acknowledgments
None.
Footnotes
Ethics Approval and Consent to Participate: Animals were not exposed to unnecessary pain or stress and animal manipulation was performed with maximal care and hygiene. Zagazig University Scientific Research and Publications Ethics Committee approved the study. All steps and procedures in the study were carried out following Zagazig University Institutional Animal Care and Use Committee regulations, with approval no. ZU-IACUC/2/F/ 522 /2023. All methods were carried out in accordance with relevant guidelines and regulations. It was performed according to the recommendations of Good Clinical Practice and the Declaration of Helsinki (2013).
Consent for Publication: Not applicable.
Availability of Data and Materials: All relevant data are included in this published article.
Author Contributions: All authors are responsible for the concept and design of the study; N.Z., and H.K. contributed to data acquisition; H.N statistical analysis; N.Z., H.K., and H.N. interpreted the results; H.N analyzed the data and drafted the manuscript. All authors critically revised the manuscript, approved the final version to be published, and agreed to be accountable for all aspects of the work. All authors contributed to the creation of the manuscript.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD: Heba A. Nazim  https://orcid.org/0009-0009-2005-5088
https://orcid.org/0009-0009-2005-5088
References
- 1. Park SB, Jee BC, Kim SH, Cho YJ, Han M. Cyclooxygenase-2 inhibitor, celecoxib, inhibits leiomyoma cell proliferation through the nuclear factor κB pathway. Reprod Sci. 2014;21(9):1187-1195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Clemett D, Goa KL. Celecoxib: a review of its use in osteoarthritis, rheumatoid arthritis and acute pain. Drugs. 2000;59(4):957-980. [DOI] [PubMed] [Google Scholar]
- 3. McCormack PL. Celecoxib: a review of its use for symptomatic relief in the treatment of osteoarthritis, rheumatoid arthritis and ankylosing spondylitis. Drugs. 2011;71:2457-2489. [DOI] [PubMed] [Google Scholar]
- 4. Zarghi A, Arfaei S. Selective COX-2 inhibitors: a review of their structure-activity relationships. Iran J Pharm Res. 2011;10(4):655-683. [PMC free article] [PubMed] [Google Scholar]
- 5. Whelton A, Maurath CJ, Verburg KM, Geis GS. Renal safety and tolerability of celecoxib, a novel cyclooxygenase-2 inhibitor. Am J Ther. 2000;7(3):159-175. [DOI] [PubMed] [Google Scholar]
- 6. Suddek GM, El-Kenawi AE, Abdel-Aziz A, El-Kashef HA. Celecoxib, a selective cyclooxygenase-2 inhibitor, attenuates renal injury in a rat model of Cisplatin-induced nephrotoxicity. Chemotherapy. 2011;57(4):321-326. [DOI] [PubMed] [Google Scholar]
- 7. Shin S. Safety of celecoxib versus traditional nonsteroidal anti-inflammatory drugs in older patients with arthritis. J Pain Res. 2018;11:3211-3219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Ahmad S, Campos MG, Fratini F, Altaye SZ, Li J. New insights into the biological and pharmaceutical properties of royal jelly. Int J Mol Sci. 2020;21(2):382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Mostafa RE, El-Marasy SA, Abdel Jaleel GA, Bakeer RM. Protective effect of royal jelly against diclofenac-induced hepato-renal damage and gastrointestinal ulcerations in rats. Heliyon. 2020;6(2):e03330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Mohamed HK, Mobasher MA, Ebiya RA, et al. Anti-inflammatory, anti-apoptotic, and antioxidant roles of honey, royal jelly, and propolis in suppressing nephrotoxicity induced by doxorubicin in male albino rats. Antioxidants. 2022;11(5):1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Aslan Z, Aksoy L. Anti-inflammatory effects of royal jelly on ethylene glycol induced renal inflammation in rats. Int Braz J Urol. 2015;41(5):1008-1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Koçkaya EA, Selmanoğlu G, Kismet K, Akay MT. Pathological and biochemical effects of therapeutic and supratherapeutic doses of celecoxib in Wistar albino male rats. Drug Chem Toxicol. 2010;33(4):410-414. [DOI] [PubMed] [Google Scholar]
- 13. Eleawa SM, Alkhateeb MA, Alhashem FH, et al. Resveratrol reverses cadmium chloride-induced testicular damage and subfertility by downregulating p53 and Bax and upregulating gonadotropins and Bcl-2 gene expression. J Reprod Dev. 2014;60(2):115-127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Fawcett JK, Scott JE. A rapid and precise method for the determination of urea. J Clin Pathol. 1960;13(2):156-159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Young D, Postaner L, Giberrman V. Colorimetric determination of serum creatinine. J Clin Chem. 1975;21112. [Google Scholar]
- 16. Wang M, Utell MJ, Schneider A, et al. Does total antioxidant capacity modify adverse cardiac responses associated with ambient ultrafine, accumulation mode, and fine particles in patients undergoing cardiac rehabilitation? Environ Res. 2016;149:15-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Schönthal AH, Chen TC, Hofman FM, Louie SG, Petasis NA. Celecoxib analogs that lack COX-2 inhibitory function: preclinical development of novel anticancer drugs. Expert Opin Investig Drugs. 2008;17(2):197-208. [DOI] [PubMed] [Google Scholar]
- 18. Zweers MC, de Boer TN, van Roon J, Bijlsma JW, Lafeber FP, Mastbergen SC. Celecoxib: considerations regarding its potential disease-modifying properties in osteoarthritis. Arthritis Res Ther. 2011;13(5):239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Harris RC. COX-2 and the kidney. J Cardiovasc Pharmacol. 2006;47(suppl 1):S37-S42. [DOI] [PubMed] [Google Scholar]
- 20. Weir MR. Renal effects of nonselective NSAIDs and coxibs. Cleve Clin J Med. 2002;69(suppl 1):SI53-S158. [DOI] [PubMed] [Google Scholar]
- 21. Akyazi I, Eraslan E, Gülçubuk A, et al. Long-term aspirin pretreatment in the prevention of cerulein-induced acute pancreatitis in rats. World J Gastroenterol. 2013;19(19):2894-2903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Lomas AL, Grauer GF. The renal effects of NSAIDs in dogs. J Am Anim Hosp Assoc. 2015;51(3):197-203. [DOI] [PubMed] [Google Scholar]
- 23. Hörl WH. Nonsteroidal anti-inflammatory drugs and the kidney. Pharmaceuticals. 2010;3(7):2291-2321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Khan S, Andrews KL, Chin-Dusting JPF. Cyclo-oxygenase (COX) inhibitors and cardiovascular risk: are non-steroidal anti-inflammatory drugs really anti-inflammatory? Int J Mol Sci. 2019;20(17):4262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Nderitu P, Doos L, Jones PW, Davies SJ, Kadam UT. Non-steroidal anti-inflammatory drugs and chronic kidney disease progression: a systematic review. Fam Pract. 2013;30(3):247-255. [DOI] [PubMed] [Google Scholar]
- 26. LeLorier J, Bombardier C, Burgess E, et al. Practical considerations for the use of nonsteroidal anti-inflammatory drugs and cyclo-oxygenase-2 inhibitors in hypertension and kidney disease. Can J Cardiol. 2002;18(12):1301-1308. [PubMed] [Google Scholar]
- 27. Aziz ND, Ouda MH, Ubaid MM. Comparing the toxic effects of nonsteroidal anti-inflammatory drugs (celecoxib and ibuprofen) on heart, liver, and kidney in rats. Asian J Pharm Clin Res. 2018;11(6):482-485. [Google Scholar]
- 28. Zhang X, Donnan PT, Bell S, Guthrie B. Non-steroidal anti-inflammatory drug induced acute kidney injury in the community dwelling general population and people with chronic kidney disease: systematic review and meta-analysis. BMC Nephrol. 2017;18(1):256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Maruyama K, Tanaka S, Hiramoto K, Ooi K. Effect of celecoxib administration on the skin of 40-week-old mice. Biol Pharm Bull. 2022;45(12):1857-1861. [DOI] [PubMed] [Google Scholar]
- 30. Tabibian JH, Tabibian N, Kaufman DM. Late-onset celecoxib-induced combined hepato-nephrotoxicity. Br J Clin Pharmacol. 2008;66(1):150-151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Drożdżal S, Lechowicz K, Szostak B, et al. Kidney damage from nonsteroidal anti-inflammatory drugs-Myth or truth? review of selected literature. Pharmacol Res Perspect. 2021;9(4):e00817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Chan MTH, Chan TCW, Mak HCY, et al. Incidence, severity and reversibility of acute kidney injury after elective hip and knee arthroplasty in patients receiving celecoxib perioperatively as one of the standard multimodal analgesic protocols. J Orthop Sports Med. 2023;5:199-206. [Google Scholar]
- 33. Ahmad SR, Kortepeter C, Brinker A, Chen M, Beitz J. Renal failure associated with the use of celecoxib and rofecoxib. Drug Saf. 2002;25(7):537-544. [DOI] [PubMed] [Google Scholar]
- 34. Alper AB, Jr, Meleg-Smith S, Krane NK. Nephrotic syndrome and interstitial nephritis associated with celecoxib. Am J Kidney Dis. 2002;40(5):1086-1090. [DOI] [PubMed] [Google Scholar]
- 35. Markowitz GS, Falkowitz DC, Isom R, et al. Membranous glomerulopathy and acute interstitial nephritis following treatment with celecoxib. Clin Nephrol. 2003;59(2):137-142. [DOI] [PubMed] [Google Scholar]
- 36. Akhund L, Quinet RJ, Ishaq S. Celecoxib-related renal papillary necrosis. Arch Intern Med. 2003;163(1):114-115. [DOI] [PubMed] [Google Scholar]
- 37. Bonventre JV, Vaidya VS, Schmouder R, Feig P, Dieterle F. Next-generation biomarkers for detecting kidney toxicity. Nat Biotechnol. 2010;28(5):436-440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Malyszko J, Lukaszyk E, Glowinska I, Durlik M. Biomarkers of delayed graft function as a form of acute kidney injury in kidney transplantation. Sci Rep. 2015;5:11684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Ejaz P, Bhojani K, Joshi VR. NSAIDs and kidney. J Assoc Physicians India. 2004;52:632-640. [PubMed] [Google Scholar]
- 40. Gupta S, Sarotra P, Aggarwal R, Dutta N, Agnihotri N. Role of oxidative stress in celecoxib-induced renal damage in wistar rats. Dig Dis Sci. 2007;52(11):3092-3098. [DOI] [PubMed] [Google Scholar]
- 41. Kellum JA, Lameire N; KDIGO AKI Guideline Work Group. Diagnosis, evaluation, and management of acute kidney injury: a KDIGO summary (part 1). Crit Care. 2013;17(1):204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Melgaco SS, Saraiva MI, Lima TT, Silva Junior GB, Daher EF. Nonsteroidal antiinflammatory drugs nephrotoxicity. Med. 2010;43(4):382-390. [Google Scholar]
- 43. Chiu HY, Huang HL, Li CH, et al. Increased risk of chronic kidney disease in rheumatoid arthritis associated with cardiovascular complications: a national population-based cohort study. PLoS ONE. 2015;10(9):e0136508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Maulood IM, Mahmud AM. Effects of COX-1 and COX-2 inhibitors in L-nitro-l-arginine methyl ester induced hypertensive rats. Jordan J Biol Sci. 2013;147(898):1-6. [Google Scholar]
- 45. Wheeler DC, Winkelmayer WC; Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int Suppl (2011). 2017;7(3):1-59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Natarajan SK, Basivireddy J, Ramachandran A, et al. Renal damage in experimentally-induced cirrhosis in rats: role of oxygen free radicals. Hepatology. 2006;43(6):1248-1256. [DOI] [PubMed] [Google Scholar]
- 47. Sozer S, Diniz G, Lermioglu F. Effects of celecoxib in young rats: histopathological changes in tissues and alterations of oxidative stress/antioxidant defense system. Arch Pharm Res. 2011;34(2):253-259. [DOI] [PubMed] [Google Scholar]
- 48. Harirforoosh S, Asghar W, Jamali F. Adverse effects of nonsteroidal antiinflammatory drugs: an update of gastrointestinal, cardiovascular and renal complications. J Pharm Pharm Sci. 2013;16(5):821-847. [DOI] [PubMed] [Google Scholar]
- 49. Shukla A, Rai MK, Prasad N, Agarwal V. Short-term non-steroid anti-inflammatory drug use in spondyloarthritis patients induces subclinical acute kidney injury: biomarkers study. Nephron. 2017;135(4):277-286. [DOI] [PubMed] [Google Scholar]
- 50. Henao J, Hisamuddin I, Nzerue CM, Vasandani G, Hewan-Lowe K. Celecoxib-induced acute interstitial nephritis. Am J Kidney Dis. 2002;39(6):1313-1317. [DOI] [PubMed] [Google Scholar]
- 51. Silici S, Ekmekcioglu O, Kanbur M, Deniz K. The protective effect of royal jelly against cisplatin-induced renal oxidative stress in rats. World J Urol. 2011;29(1):127-132. [DOI] [PubMed] [Google Scholar]
- 52. Karadeniz A, Simsek N, Karakus E, et al. Royal jelly modulates oxidative stress and apoptosis in liver and kidneys of rats treated with cisplatin. Oxid Med Cell Longev. 2011;2011:981793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. El-Kott AF, Omar NA, El-Hawary HK, Mansy SE. The protective effect of royal jelly against Doxorubicin-induced renal oxidative stress, histopathological and immunohistochemiacal alterations in Ehrlich Ascites tumor bearing mice. Egypt Acad J Biolog Sci. 2014;5(1):9-21. [Google Scholar]
- 54. Araki K, Miyata Y, Ohba K, et al. Oral intake of royal jelly has protective effects against tyrosine kinase inhibitor-induced toxicity in patients with renal cell carcinoma: a randomized, double-blinded, placebo-controlled trial. Medicines. 2018;6(1):2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Aslan A, Beyaz S, Gok O, et al. Protective effect of royal jelly on fluoride-induced nephrotoxicity in rats via the some protein biomarkers signalling pathways: a new approach for kidney damage. Biomarkers. 2022;27(7):637-647. [DOI] [PubMed] [Google Scholar]
- 56. El-Sayed AAA. Anti-inflammatory and protective effects of royal jelly against hepatic and renal damage induced by valproic acid in rats. Genet Mol Res. 2013;22(1):GMR19063. [Google Scholar]
- 57. Focak M, Suljevic D. Ameliorative effects of propolis and royal jelly against ccl4-induced hepatotoxicity and nephrotoxicity in Wistar rats. Chem Biodiv. 2023;20(1):e202200948. [DOI] [PubMed] [Google Scholar]
- 58. Trevisan M, Browne R, Ram M, et al. Correlates of markers of oxidative status in the general population. Am J Epidemiol. 2001;154(4):348-356. [DOI] [PubMed] [Google Scholar]
- 59. Bernabucci U, Ronchi B, Lacetera N, Nardone A. Markers of oxidative status in plasma and erythrocytes of transition dairy cows during hot season. J Dairy Sci. 2002;85(9):2173-2179. [DOI] [PubMed] [Google Scholar]
- 60. Rao PS, Kalva S, Yerramilli A, Mamidi S. Free radicals and tissue damage: role of antioxidants. Free Radic Antioxid. 2011;1(4):2-7. [Google Scholar]
- 61. Dupré-Crochet S, Erard M, Nüβe O. ROS production in phagocytes: why, when, and where? J Leukoc Biol. 2013;94(4):657-670. [DOI] [PubMed] [Google Scholar]
- 62. Valko M, Leibfritz D, Moncol J, Cronin MT, Mazur M, Telser J. Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol. 2007;39(1):44-84. [DOI] [PubMed] [Google Scholar]
- 63. Pham-Huy LA, He H, Pham-Huy C. Free radicals, antioxidants in disease and health. Int J Biomed Sci. 2008;4(2):89-96. [PMC free article] [PubMed] [Google Scholar]
- 64. Anand David AV, Arulmoli R, Parasuraman S. Overviews of biological importance of quercetin: a bioactive flavonoid. Pharmacogn Rev. 2016;10(20):84-89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Ďuračková Z. Some current insights into oxidative stress. Physiol Res. 2010;59(4):459-469. [DOI] [PubMed] [Google Scholar]
- 66. Reiter RJ, Paredes SD, Manchester LC, Tan DX. Reducing oxidative/nitrosative stress: a newly-discovered genre for melatonin. Crit Rev Biochem Mol Biol. 2009;44(4):175-200. [DOI] [PubMed] [Google Scholar]
- 67. Ziech D, Franco R, Georgakilas AG, et al. The role of reactive oxygen species and oxidative stress in environmental carcinogenesis and biomarker development. Chem Biol Interact. 2010;188(2):334-339. [DOI] [PubMed] [Google Scholar]
- 68. Mirończuk-Chodakowska I, Witkowska AM, Zujko ME. Endogenous non-enzymatic antioxidants in the human body. Adv Med Sci. 2018;63(1):68-78. [DOI] [PubMed] [Google Scholar]
- 69. Burak Cimen MY, Cimen OB, Eskandari G, Sahin G, Erdoğan C, Atik U. In vivo effects of meloxicam, celecoxib, and ibuprofen on free radical metabolism in human erythrocytes. Drug Chem Toxicol. 2003;26(3):169-176. [DOI] [PubMed] [Google Scholar]
- 70. Ozgocmen S, Ardicoglu O, Erdogan H, Fadillioglu E, Gudul H. In vivo effect of celecoxib and tenoxicam on oxidant/ anti-oxidant status of patients with knee osteoarthritis. Ann Clin Lab Sci. 2005;35(2):137-143. [PubMed] [Google Scholar]
- 71. Melekh B, Ilkiv I, Lozynskyi A, Sklyarov A. Antioxidant enzyme activity and lipid peroxidation in rat liver exposed to celecoxib and lansoprazole under epinephrine-induced stress. J Appl Pharm Sci. 2017;7(10):094-099. [Google Scholar]
- 72. Hamza RZ, Al-Eisa RA, El-Shenawy NS. Possible ameliorative effects of the royal jelly on hepatotoxicity and oxidative stress induced by molybdenum nanoparticles and/or cadmium chloride in male rats. Biology. 2022;11(3):450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Cotter TG, Rinella M. Nonalcoholic fatty liver disease 2020: the state of the disease. Gastroenterology. 2020;158(7):1851-1864. [DOI] [PubMed] [Google Scholar]
- 74. You MM, Liu YC, Chen YF, et al. Royal jelly attenuates nonalcoholic fatty liver disease by inhibiting oxidative stress and regulating the expression of circadian genes in ovariectomized rats. J Food Biochem. 2020;44(3):e13138. [DOI] [PubMed] [Google Scholar]
- 75. Karimi E, Khorvash F, Arab A, Sepidarkish M, Saadatnia M, Amani R. The effects of royal jelly supplementation on oxidative stress, inflammatory mediators, mental health, cognitive function, quality of life, and clinical outcomes of patients with ischemic stroke: study protocol for a randomized controlled trial. BMC Nutr. 2023;9(1):32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Kohno K, Okamoto I, Sano O, et al. Royal jelly inhibits the production of proinflammatory cytokines by activated macrophages. Biosci Biotechnol Biochem. 2004;68(1):138-145. [DOI] [PubMed] [Google Scholar]
- 77. Hadi A, Najafgholizadeh A, Aydenlu ES, et al. Royal jelly is an effective and relatively safe alternative approach to blood lipid modulation: a meta-analysis. J Funct Foods. 2018;41:202-209. [Google Scholar]
- 78. Maleki V, Jafari-Vayghan H, Saleh-Ghadimi S, Adibian M, Kheirouri S, Alizadeh M. Effects of Royal jelly on metabolic variables in diabetes mellitus: a systematic review. Complement Ther Med. 2019;43:20-27. [DOI] [PubMed] [Google Scholar]
- 79. Kanbur M, Eraslan G, Silici S, Karabacak M. Effects of sodium fluoride exposure on some biochemical parameters in mice: evaluation of the ameliorative effect of royal jelly applications on these parameters. Food Chem Toxicol. 2009;47(6):1184-1189. [DOI] [PubMed] [Google Scholar]
- 80. Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol. 2007;35(4):495-516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Rathore S, Datta G, Kaur I, Malhotra P, Mohmmed A. Disruption of cellular homeostasis induces organelle stress and triggers apoptosis like cell-death pathways in malaria parasite. Cell Death Dis. 2015;6(7):e1803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Hussar P. Apoptosis regulators bcl-2 and caspase-3. Encyclopedia. 2022;2(4):1624-1636. [Google Scholar]
- 83. Lockshin RA, Zakeri Z. Programmed cell death and apoptosis: origins of the theory. Nat Rev Mol Cell Biol. 2001;2(7):545-550. [DOI] [PubMed] [Google Scholar]
- 84. Blagosklonny MV. Cell death beyond apoptosis. Leukemia. 2000;14(8):1502-1508. [DOI] [PubMed] [Google Scholar]
- 85. Fink SL, Cookson BT. Apoptosis, pyroptosis, and necrosis: mechanistic description of dead and dying eukaryotic cells. Infect Immun. 2005;73(4):1907-1916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Xu X, Lai Y, Hua ZC. Apoptosis and apoptotic body: disease message and therapeutic target potentials. Biosci Rep. 2019;39(1):BSR20180992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. D’Arcy MS. Cell death: a review of the major forms of apoptosis, necrosis and autophagy. Cell Biol Int. 2019;43(6):582-592. [DOI] [PubMed] [Google Scholar]
- 88. Chi H, Chang HY, Sang TK. Neuronal cell death mechanisms in major neurodegenerative diseases. Int J Mol Sci. 2018;19(10):3082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Shao D, Kan M, Qiao P, et al. Celecoxib induces apoptosis via a mitochondriadependent pathway in the H22 mouse hepatoma cell line. Mol Med Rep. 2014;10(4):2093-2098. [DOI] [PubMed] [Google Scholar]
- 90. Choi SM, Cho YS, Park G, Lee SK, Chun KS. Celecoxib induces apoptosis through Akt inhibition in 5-fluorouracil-resistant gastric cancer cells. Toxicol Res. 2020;37(1):25-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Manning BD, Toker A. AKT/PKB signaling: navigating the network. Cell. 2017;169(3):381-405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Gao D, Nyalali AMK, Hou Y, et al. 2,5-Dimethyl celecoxib inhibits proliferation and cell cycle and induces apoptosis in glioblastoma by suppressing CIP2A/PP2A/Akt signaling axis. J Mol Neurosci. 2021;71(8):1703-1713. [DOI] [PubMed] [Google Scholar]
- 93. Azad F, Nejati V, Shalizar-Jalali A, Najafi G, Rahmani F. Antioxidant and anti-apoptotic effects of royal jelly against nicotine-induced testicular injury in mice. Environ Toxicol. 2019;34(6):708-718. [DOI] [PubMed] [Google Scholar]
- 94. Tohamy HG, El-Neweshy MS, Soliman MM, et al. Protective potential of royal jelly against hydroxyurea -induced hepatic injury in rats via antioxidant, anti-inflammatory, and anti-apoptosis properties. PLoS ONE. 2022;17(3):e0265261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Fazili N, Soheili ZS, Malekzadeh-Shafaroudi S, et al. Royal jelly decreases MMP-9 expression and induces apoptosis in human bladder cancer 5637 cells. J Cell Mol Res. 2021;13(1):36-43. [Google Scholar]