Abstract
Background
Pediatric brain tumor patients are at risk of developing neurocognitive impairments and associated white matter alterations. In other populations, post-traumatic stress symptoms (PTSS) impact cognition and white matter. This study aims to investigate the effect of PTSS on neurocognitive functioning and limbic white matter in pediatric brain tumor patients.
Methods
Sixty-six patients (6–16 years) completed neuropsychological assessment and brain MRI (1-year post-diagnosis) and parents completed PTSS proxy questionnaires (CRIES-13; 1–3 months and 1-year post-diagnosis). Mean Z-scores and percentage impaired (>1SD) for attention, processing speed, executive functioning, and memory were compared to normscores (t-tests, chi-square tests). Multi-shell diffusion MRI data were analyzed for white matter tractography (fractional anisotropy/axial diffusivity). Effects of PTSS on neurocognition and white matter were explored with linear regression models (FDR correction for multiple testing), including age at diagnosis, treatment intensity, and tumor location as covariates. Neurocognition and limbic white matter associations were explored with correlations.
Results
Attention (M = −0.49, 33% impaired; P < .05) and processing speed (M = −0.57, 34% impaired; P < .05) were significantly lower than healthy peers. PTSS was associated with poorer processing speed (β = −0.64, P < .01). Treatment intensity, age at diagnosis, and tumor location, but not PTSS, were associated with limbic white matter metrics. Neurocognition and white matter metrics were not associated.
Conclusions
Higher PTSS was associated with poorer processing speed, highlighting the need for monitoring, and timely referrals to optimize psychological well-being and neurocognitive functioning. Future research should focus on longitudinal follow-up and explore the impact of PTSS interventions on neurocognitive performance.
Keywords: limbic system, neurocognition, pediatric brain tumor, PTSS, white matter
Key Points.
Higher post-traumatic stress symptoms (PTSS) in pediatric brain tumor patients are associated with poorer processing speed 1 year after diagnosis.
Limbic white matter integrity was related to treatment intensity, age at diagnosis, and tumor location (not PTSS).
Importance of the Study.
Of all pediatric cancers, children with a brain tumor are at highest risk for developing neurocognitive impairments, which may worsen over time and impact educational attainment and employment rates. These cognitive impairments have been associated with changes in age-expected white matter growth, but interventions to prevent or reverse cognitive declines are limited. In other populations, post-traumatic stress symptoms (PTSS) are linked to both neurocognition and white matter, and has not been researched in pediatric brain tumor patients. This study provides insights into these relationships and demonstrates that higher PTSS negatively impacts processing speed, independent of treatment intensity, age at diagnosis, and tumor location. This suggests that interventions targeting PTSS may be able to enhance cognitive functioning and health-related quality of life, which is a suggested target for future research. Future longitudinal studies to examine relationships with brain metrics in the long term are suggested.
Survivors of pediatric brain tumors report the poorest health-related quality of life among childhood cancer survivors,1,2 which are partly driven by neurocognitive problems. Furthermore, these problems may continue to worsen over time after cancer treatment.3,4 Earlier research has shown that declines are mostly seen in the domains of attention, executive functioning, processing speed, and memory.5–8 Also, these impairments have been associated with altered brain structure, such as abnormalities in white matter, including a loss of white matter volume and a deficit in age-expected white matter growth.9–11 Neurocognition and white matter integrity impairments can be caused by factors such as the tumor itself, neurosurgery, and radiotherapy, and greater risk is associated with higher-intensity therapies.8,12–16 To date, interventions to prevent or reverse neurocognitive impairments are limited.7
Besides medical factors, there is increasing evidence that psychological factors can also influence both neurocognition and the structure of the brain. In typically developing children, it was seen that post-traumatic stress symptoms (PTSS) negatively impacted neurocognitive functioning on overall IQ and several domains such as executive functioning.17 The diagnosis of a life-threatening disease, and undergoing intensive medical procedures inevitably cause stress responses in children with a brain tumor. Although stress is a normal response and many children and families are resilient, research has shown that PTSS is common and can even become chronic in some children.18–21 Kosir et al. showed that a significant number of survivors are at risk of developing symptoms of post-traumatic stress.19 The traumatic stress model of Price et al.22 proposes that, on average, stress levels peak at the beginning after a medical diagnosis and decrease over time. However, some people (30%) have higher than average rates of stress levels close after a medical diagnosis, and this group may experience persistent stress for a longer period. After some time, in this patient group, stress levels can either increase further, be persistent (chronic) or decrease.18 It has been suggested that early life experiences, particularly persistent stress, can increase the risk of cognitive dysfunction.23 However, the early relationship between PTSS and neurocognitive functioning in children with a brain tumor remains unclear.
Chronic PTSS has also been linked to abnormalities in the structure and function of the brain, such as changes in white matter in the limbic regions, which are crucial for emotional regulation and cognitive processing.17,18,24–26 In typically developing children, PTSS has been associated with altered white matter fractional anisotropy (FA) and axial diffusivity (AD) metrics, suggesting potential disruptions in connectivity and structural integrity in these areas after exposure to stressful events.17 Relationships between PTSS and limbic white matter integrity have not been previously investigated in pediatric brain tumor patients.
Considering the challenges of physical and psychological burden faced by children with brain tumors, it is important to gain insight into the impact of PTSS on neurocognitive functioning and limbic white matter integrity at an early stage after diagnosis. Understanding the relationships could be a starting point in developing timely, much-needed tools and interventions for healthcare practitioners that target PTSS, to improve long-term neurocognitive functioning, and health-related quality of life. The overall aim of this research is to examine relationships between PTSS, neurocognitive functioning, and limbic white matter in pediatric brain tumor patients within the first year after diagnosis. First, we investigate the occurrence of PTSS in pediatric brain tumor patients. Second, we investigate the neurocognitive performance of our sample and how it relates to a normative age-matched healthy population. This is followed by an investigation into the effect of PTSS on neurocognition and PTSS on limbic white matter. Finally, we evaluate the relationship between PTSS and neurocognition. Based on the literature, we hypothesize that pediatric brain tumor patients will have higher rates of PTSS and have impaired neurocognitive performance 1 year after diagnosis. Additionally, we expect an effect of PTSS on neurocognitive performance and on limbic white matter.
Materials and Methods
Participants
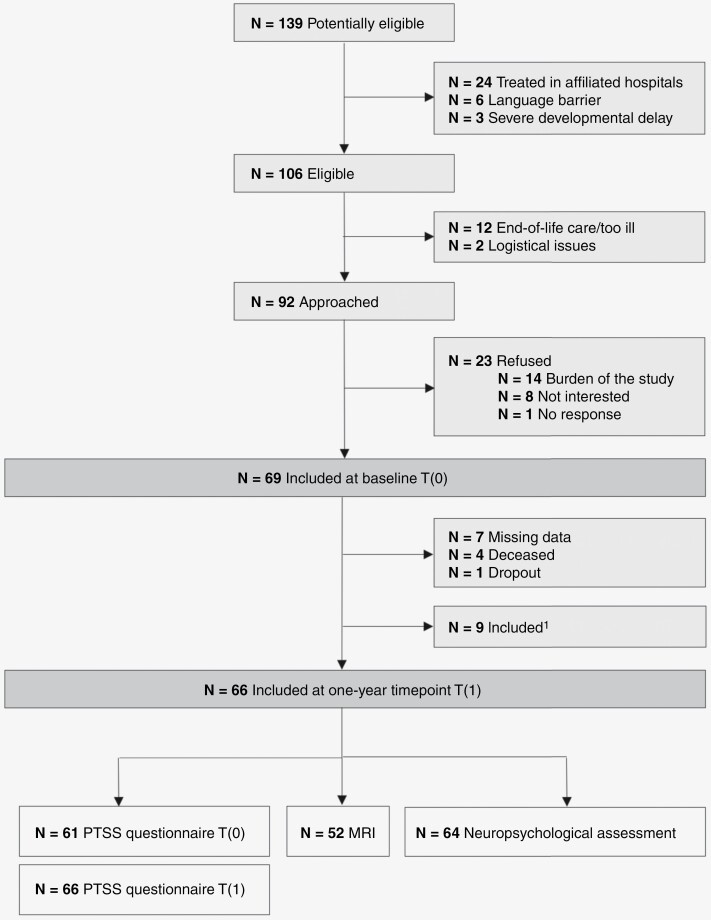
The SuSPeCT-study is a longitudinal, prospective, and observational study at the Princess Máxima Center for Pediatric Oncology in the Netherlands. Between January 2020 and December 2021, children with a newly diagnosed primary brain tumor were invited for study participation. All children and parents provided written informed consent. This study was approved by the Clinical Research Committee of the Princess Maxima Center (PMC CRC 2019-065) and confirmed subsequently by the Medical Research Ethics Committee of the University Medical Center Utrecht (19/728). The Institutional Review Board of the University Medical Center Utrecht classified this study as exempt from the Medical Research Involving Human Subjects Act. Eligible patients were between 6 and 16 years old at the time of diagnosis, spoke Dutch sufficiently, did not have a developmental delay, and were not receiving end-of-life care. See Figure 1 for flowchart of patient enrollment. All patients were followed for 2 years and completed 3 testing moments, consisting of neuropsychological assessments, actigraphic sleep measures, questionnaires, and a brain MRI.
Figure 1.
Flowchart of participant enrollment and available data. 1Patients who were initially treated at shared care centers, and continued treatment at the Princess Máxima Center, were invited for participation for the 1-year timepoint.
This was an explorative study and therefore we did not conduct an a priori power calculation. Rather, we based the calculation on the expected number of patients eligible for the study. The expected number of eligible patients was 60 per year. With an inclusion period of one and half years and a participation rate of about 70–75%, based on previous experiences with these types of studies, we expected that a total of approximately 65 participants would participate.
Procedure
Measures of interest for this study were at the time points shortly after diagnosis (1–3 months; T0) and 1 year later (9–15 months; T1). For T0, proxy report PTSS questionnaires were administered. For T1, the same proxy report PTSS questionnaires were administered, plus a multi-shell diffusion MRI scan (standard care), and neuropsychological assessment (standard care for a part of the participants) were completed. Actigraphic sleep measures were reported in a separate paper.27
Proxy-Reported PTSS
PTSS was assessed using the Children’s Revised Impact of Event Scale (CRIES-13) proxy report.28 The CRIES-13 is a brief measure designed to screen children at risk for post-traumatic stress disorder (PTSD). It has good face and construct validity, a stable factor structure, correlates well with other indices of distress, and has been used to screen very large samples of at-risk children following a wide range of traumatic events. The internal consistency and test–retest reliability of the CRIES are high: ɑ = 0.89 and a test–retest reliability of.85.29 The questionnaire is completed by parents, using the online “KLIK PROM” portal (www.hetklikt.nu). It contains 13 questions on 3 subscales; re-experience, avoidance, and increased irritability; these can be captured in a total score range of 0–65. The clinical threshold for children at risk for PTSD is established at 31, however, to examine the effect of proxy-reported PTSS, rather than a PTSD diagnosis, we used a subclinical threshold of 25. Patients were split into 2 groups based on their scores: the “no PTSS” group obtained scores ≤ 25 on both time points, and patients who obtained a score > 25 on at least 1 of the 2 time points were allocated to the “PTSS” group.
Neuropsychological Assessment
Neuropsychological assessments were administered by a trained neuropsychologist around 1 year after diagnosis, N = 24 (36%) were completed within standard care (called Brain CARE program). We tested the domains of attention, executive functioning, processing speed, and memory, using norm-referenced Dutch tests (see below). All scores were converted to Z-scores, a measure of how much an individual deviates from the normative sample with M = 0, SD = 1 (higher scores indicate better performance). For each neurocognitive domain, composite scores were calculated by averaging 2 sub-scores. When only 1 of the 2 domain scores was available for calculating domain scores, only 1 score was used. For this analysis, we used the same composite scores as our other papers of the SuSPeCT-study (unpublished manuscript, Kremer—Hooft van Huijsduijnen, E.1).
Attention was measured with the sustained attention dots (SAD) task of The Amsterdam Neuropsychological Tasks (ANT), a computerized test battery.30 The neurocognitive composite attention score was computed by calculating the mean of the average completion time (ANT SAD) and the SD of the completion time (ANT SAD). Executive functioning was measured by the number of misses of the ANT SAD, as an indicator of inhibition, and the Digit Span task (WISC-V-NL/WAIS-IV-NL), as an indicator for working memory.31,32 The processing speed composite score consisted of the mean reaction speed (ANT Baseline Speed) and the Processing Speed Index (WISC-V-NL/WAIS-IV-NL, subtests “Symbol Search” and “Coding”).31,32 Memory was measured by averaging the immediate recall (short-term memory) and delayed recall (long-term memory) scores of the 15-word task (15WT). For children up to 12 years old, the child version was administered, and for adolescents, there was the 15WT adult version.33
MRI Image Acquisition and Processing
MRI data was acquired 1 year (±3 months) after diagnosis using a standard clinical MRI-protocol. Data acquisition included an anatomical 3D T1 scan and multi-shell diffusion MRI from a 3.0T Philips Ingenia Elition X MR scanner (Philips Healthcare Medical Systems, Best, The Netherlands).
Whole-brain multi-shell diffusion MRI was collected with a single-shot echo-planar imaging sequence. 56 directions; 4x b = 0, 52x b = 1000 s/mm2, and 56x b = 2000 s/mm2; TE = 8.9 ms, TR = 323.97 ms; 66 slices; slice acquisition matrix = 112 × 112 with FOV = 224 × 224 mm2; voxel-size = 2 × 2 × 2 mm3. Slices were acquired on the axial plane, with a total scan duration of 5 min and 57 s.
We used FreeSurfer 7.3.2 to define cortical and subcortical regions in the T1-weighted images of each individual.34 Briefly, this processing included motion correction and averaging,35 removal of non-brain tissue using a hybrid watershed/surface deformation procedure,36 automated Talairach transformation, and segmentation of the subcortical white matter and deep gray matter volumetric structures.34,37 Freesurfer morphometric procedures have been shown to have good test–retest reliability across scanner manufacturers and field strengths.38,39
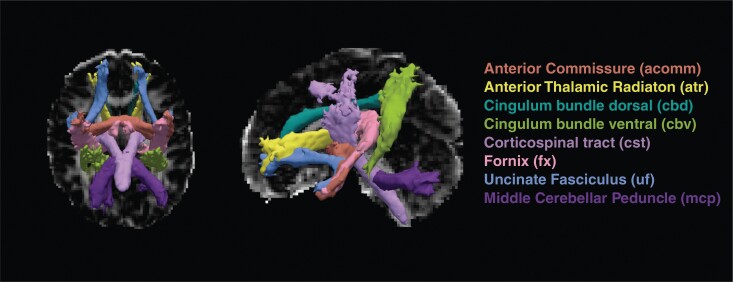
We then performed global probabilistic tractography with anatomical priors using TRACULA (Tracts Constrained by Underlying Anatomy). This tool reconstructs major white matter pathways by incorporating prior information on the structural segmentation labels that each pathway goes through or next to as a function of position along the length of the pathway. We used the ball and stick single-shell model in the bedpostx step.40 TRACULA generated averaged fractional anisotropy (FA) and axial diffusivity (AD) of all 7 tracts around the limbic system and the middle cerebellar peduncle as control. We chose to only examine FA and AD, since we wanted to reduce the number of comparisons, and radial diffusivity (RD) and mean diffusivity (MD) were both highly negatively correlated with FA. Additionally, we generated head motion data, of which we calculated the total motion index (TMI) for quality control. One participant had > 3SD deviance for the TMI and therefore was excluded from the analysis, resulting in a sample size of N = 52. FA and AD scores were averaged across left and right. AD values were multiplied by 1000 (to help with interpretation). The 8 tracts were Anterior Commissure, Anterior Thalamic Radiation, Cingulum Bundle Dorsal, Cingulum Bundle Ventral, Corticospinal tract, Fornix, Uncinate Fasciculus, and Middle Cerebellar Peduncle (Figure 2).
Figure 2.
White matter tracts that were constructed using TRACULA. Seven tracts are in/around the limbic system and the MCP is added as control tract. FA and AD scores were averaged across left and right.
Medical Variables
The Neurological Predictor Scale (NPS) indicates treatment intensity by combining cumulative neurological risk factors and tumor treatments into a single total score.41,42 The NPS considers information on radiotherapy, chemotherapy, neurosurgery, and the presence of hydrocephalus, endocrine dysfunction, and seizure medications. The score ranges from 0 (lowest level of risk) to 11 (highest level of risk). Studies have established the reliability and validity of this measure in childhood cancer survivors. For example, the total NPS score is significantly associated with intelligence, processing speed, working memory, attention, and adaptive functioning, which was independent of individual risk factors.41
Statistical Analysis
Participant characteristics were descriptively reported. Demographics were compared between participants and non-participants, and the PTSS group and no PTSS group (t-tests for sex, age at assessment, days since diagnosis, NPS score, and TMI) to assess potential demographic differences between the groups of interest. For each neurocognitive domain, the percentage impaired (participants scoring > 1SD lower than norm scores) was compared to 16% (as expected in the general population) by using non-parametric chi-square tests. Average Z-scores were compared to a norm score of zero with one sample t-tests. These tests were performed for the complete sample and subsequently for the separate PTSS groups. We built 4 linear regression models to test the effect of PTSS (dichotomous) on the 4 neurocognitive domains (attention, executive functioning, processing speed, and memory; continuous) and corrected for multiple testing using false discovery rate (FDR).43 For the limbic white matter outcomes, 16 linear regression models were built and corrected for multiple testing using FDR. We tested the effect of PTSS (dichotomous) on FA and AD (continuous) in the 7 different white matter tracts around the limbic system and the MCP. For all linear regression models, covariates were age at diagnosis (continuous), NPS score (continuous), and whether the tumor was in the limbic system area or not (supratentorial medial structures vs. other locations; dichotomous) and corrected for multiple testing using FDR. We reported on significant effects of covariates in the linear models on both neurocognition and the limbic white matter. To explore the relation between limbic white matter and neurocognitive functioning, Pearson correlation tests were performed and corrected for multiple testing using FDR.
Results
Sample Characteristics
Ninety-two patients and parents were approached for the study; 69 (75%) participated at T0 and 66 (72%) participated at T1 (Figure 1). Demographics and medical characteristics are shown in Table 1. Participants (35% female, mean age 12.53 (SD = 2.81) years, 33% supratentorial medial structure tumors) did not differ from non-participants (N = 43; 40% female, mean age 10.61 (SD = 3.41) years, 33% supratentorial medial structure tumors, all P > 0.10). Tumors in the supratentorial medial structures included low-grade glioma (n = 3), germ cell tumor (n = 9), and craniopharyngioma (n = 10). Posterior fossa tumors included low-grade glioma (n = 20), medulloblastoma (n = 7), ependymoma (n = 2), germ cell tumor (n = 1), and other (n = 2). Tumors located in the cerebral lobes were low-grade glioma (n = 8), high-grade glioma (n = 2), and other (n = 2). Furthermore, there were no differences in demographics and medical variables between the PTSS and no PTSS groups (Table 1). Of the participants, 64 (91%) patients had data on PTSS and neuropsychological functioning, and 52 (74%) patients had proxy data on PTSS and limbic white matter structure.
Table 1.
Demographic and medical variables
| N (%) or mean (SD) | Overall (N = 66) | No PTSS (N = 43) | PTSS (N = 23) | P-value1 |
|---|---|---|---|---|
| Female sex | 23 (35) | 15 (35) | 8 (35) | >0.99 |
| Age at assessment (years) | 12.53 (2.81) | 12.04 (2.83) | 13.40 (2.62) | 0.06 |
| Time since diagnosis (days) | 393 (76) | 390 (86) | 400 (52) | 0.57 |
| NPS score | 4.11 (2.11) | 4.00 (2.06) | 4.30 (2.22) | 0.59 |
| Tumor location | ||||
| Cerebral lobes | 12 (18) | 10 (23) | 2 (9) | |
| Supratentorial medial structures | 22 (33) | 15 (35) | 7 (30) | |
| Posterior fossa | 32 (48) | 18 (42) | 14 (61) | |
| Tumor type | ||||
| Low-grade glioma | 31 (47) | 21 (49) | 10 (43) | |
| High-grade glioma | 2 (3) | 1 (2) | 1 (4) | |
| Medulloblastoma | 7 (11) | 4 (9) | 3 (13) | |
| Ependymoma | 2 (3) | 1 (2) | 1 (4) | |
| Germ cell tumor | 10 (15) | 7 (16) | 3 (13) | |
| Craniopharyngioma | 10 (15) | 7 (16) | 3 (13) | |
| Other2 | 4 (6) | 2 (5) | 2 (9) | |
| Neurosurgery | ||||
| No | 8 (12) | 6 (14) | 2 (9) | |
| Biopsy only | 5 (8) | 4 (9) | 1 (4) | |
| One resection | 48 (73) | 31 (72) | 17 (74) | |
| Several resections | 5 (8) | 2 (5) | 3 (13) | |
| Radiotherapy | ||||
| No radiotherapy | 43 (65) | 31 (72) | 12 (52) | |
| Focal radiotherapy | 11 (17) | 5 (12) | 6 (26) | |
| Whole brain or craniospinal | 4 (6) | 2 (5) | 2 (9) | |
| Whole brain + boost | 8 (12) | 5 (12) | 3 (13) | |
| Radiotherapy type | ||||
| Photon | 7 (11) | 2 (5) | 5 (22) | |
| Proton | 16 (24) | 10 (23) | 6 (26) | |
| Chemotherapy | 18 (27) | 11 (26) | 7 (30) | |
| Cerebellar mutism syndrome | 5 (8) | 2 (5) | 3 (13) | |
| Metastases at diagnosis | 9 (14) | 7 (16) | 2 (9) | |
| Hormone deficiency | 20 (31) | 15 (35) | 5 (23) | |
| Epilepsy with seizure medication | 7 (11) | 6 (14) | 1 (4) | |
| Obstructive hydrocephalus | 30 (45) | 19 (44) | 11 (48) | |
| Total Motion Index (MRI) | 1.10 (2.06) | 1.28 (2.16) | 0.75 (1.85) | 0.35 |
1Independent t-tests between no PTSS and PTSS group. t-Tests were only performed on variables that influenced the sample selection.
2Choroid plexus tumor, atypical teratoid rhabdoid tumor, mesenchyoma, and meningioma.
Proxy-Reported PTSS
Twenty-three (34%) patients had PTSS above the threshold of 25 at either T0 and/or T1, reported by their parents. This included 15 (22%) patients at T0, 12 (18%) patients at T1, and 4 (6%) patients at both T0 and T1. Of note, 54 (82%) participants had PTSS data on both time points. Of the 12 participants that had data at either one of both time points, 3 scored above the subclinical threshold. There were 9 patients who were not included at T0, and this was mostly because of burden at that time. At T1, PTSS scores for these patients suggest that stress may have also played a role (3 out of 9 later included patients have > 25 score at T1). This is a limitation to our study, however, we chose not to exclude these patients from analyses because they are an especially vulnerable group.
Neurocognition
Results on neuropsychological performance are presented in Table 2. Mean Z-scores included attention (M = −0.49, SD = 1.12), executive functioning (M = −0.21, SD = 1.02), processing speed (M = −0.57, SD = 0.98), and memory (M = −0.23, SD = 1.42). Attention (t(57) = −3.29, P < .01) and processing speed (t(60) = −4.55, P < .001) deviated significantly from 0, indicative of lower performance compared to healthy peers. Additionally, attention (33% impaired; χ2 = 5.98, P = .01) and processing speed (34% impaired; χ2 = 7.27, P < .01) had a significantly higher proportion of impaired patients (>1SD) compared to what would be expected in the healthy, age-matched population (16%). In the overall group, 59% of patients were impaired on at least one domain.
Table 2.
Neurocognitive performance
| Overall group (N = 66) | No PTSS group (N = 43) | PTSS group (N = 23) | ||||
|---|---|---|---|---|---|---|
| Z-score, M (SD)1 | Impaired, N (%)2 | Z-score, M (SD)1 | Impaired, N (%)2 | Z-score, M (SD)1 | Impaired, N (%)2 | |
| Attention (N = 58) | −0.49 (1.12) | 19 (33) | −0.38 (1.11) | 11 (30) | −0.67 (1.14) | 8 (38) |
| Executive functioning (N = 63) | −0.21(1.02) | 11 (17) | −0.06 (1.01) | 5 (12) | −0.47 (0.99) | 6 (27) |
| Processing speed (N = 61) | −0.57 (0.98) | 21 (34) | −0.29 (0.92) | 10 (26) | −1.04 (0.92) | 11 (48) |
| Memory (N = 59) | −0.23 (1.42) | 16 (27) | −0.17 (1.58) | 9 (24) | −0.34 (1.07) | 7 (32) |
Significant numbers (P < 0.05) are in bold.
1One-sample t-test for testing mean Z-scores against 0.
2Chi square tests to compare impaired patient proportions with those expected in a healthy, age-matched population.
In the PTSS group specifically, attention (38% impaired; χ2 = 5.33, P = .02) and processing speed (48% impaired; χ2 = 11.06, P < .001) had an even higher proportion of impaired children. Average scores of attention (M = −0.67, SD = 1.14, t(20) = −2.68, P = .01), executive functioning (M = −0.47, SD = 0.99, t(21) = −2.22, P = .04), and processing speed (M = −1.04, SD = 0.92, t(22) = −5.43, P < .001) were lower than expected compared to what would be expected in the healthy, age-matched population (16%). In the no PTSS group, only attention (M = −0.38, SD = 1.11, t(36) = −2.09, P = .04) was significantly lower than expected, but the percentages impaired were not different from the general population.
PTSS and Neurocognition
Results on the effect of proxy-reported PTSS on neurocognitive functioning are presented in Table 3. The overall model for processing speed was statistically significant (R2 = 0.24, F(4,56) = 4.30, PFDRcorrected = 0.017). It was found that the PTSS group (β = −0.64, P = .01) and higher NPS score (β = −0.12, P = .03) were significantly related to lower processing speed. Models for the other 3 neurocognitive domains demonstrated no significant effects of PTSS, and overall models were not statistically significant.
Table 3.
Multivariable regression models of PTSS and neurocognitive functioning
| B (95% CI) | Attention (N = 58) | Executive functioning (N = 63) | Processing speed (N = 61)* | Memory (N = 59) |
|---|---|---|---|---|
| (Intercept) | 0.54 (−0.85 to 1.94) | 0.86 (−0.30 to 2.02) | 0.41 (−0.66 to 1.48) | −0.36 (−1.97 to 1.25) |
| PTSS group Yes vs. no |
−0.13 (−0.78 to 0.52) | −0.22 (−0.77 to 0.32) | −0.64 (−1.12 to −0.15)** | −0.13 (−0.89 to 0.62) |
| Age at diagnosis Continuous |
−0.05 (−0.16 to 0.06) | −0.08 (−0.17 to 0.01) | −0.03 (−0.11 to 0.05) | 0.07 (−0.06 to 0.19) |
| NPS score Continuous |
−0.11 (−0.25 to 0.04) | −0.04 (−0.17 to 0.08) | −0.12 (−0.24 to −0.01)* | −0.16 (−0.34 to 0.01) |
| Tumor location Suprat. vs. rest |
0.14 (−0.50 to 0.77) | 0.41 (−0.13 to 0.95) | 0.44 (−0.06 to 0.93) | 0.16 (−0.60 to 0.93) |
Abbreviations. Suprat. = supratentorial medial structures.
* Significant at the P < 0.05 level.
** Significant at the P < 0.01 level.
PTSS and Limbic White Matter
Results on the effect of PTSS on FA and AD in 8 white matter tracts are presented in Table 4. After FDR correction for multiple testing, models were not statistically significant. There was no effect of PTSS group in any of the white matter regression models.
Table 4.
Multivariable regression models of FA and AD of the 8 white matter tracts
| B (95%CI) | Fractional anisotropy (N = 52) | |||
|---|---|---|---|---|
| Anterior commissure | Anterior thalamic radiation | Cingulum bundle dorsal | Cingulum bundle ventral | |
| (Intercept) | 0.28 (0.23 to 0.34)*** | 0.30 (0.23 to 0.36)*** | 0.35 (0.27 to 0.43)*** | 0.34 (0.23 to 0.45)*** |
| PTSS group Yes vs. no |
0.01 (−0.02 to 0.04) | −0.00 (−0.04 to 0.03) | 0.03 (−0.00 to 0.07) | 0.00 (−0.05 to 0.06) |
| Age at diagnosis Continuous |
0.00 (−0.00 to 0.00) | 0.01 (0.00 to 0.01)* | −0.01 (−0.01 to 0.00) | −0.00 (−0.01 to 0.01) |
| NPS score Continuous |
0.00 (−0.00 to 0.01) | −0.01 (−0.01 to −0.00)* | 0.01 (0.00 to 0.02)** | 0.01 (−0.00 to 0.02) |
| Tumor location Suprat. vs. rest |
−0.03 (−0.05 to 0.00) | −0.02 (−0.05 to 0.01) | −0.01 (−0.05 to 0.02) | −0.01 (−0.06 to 0.04) |
| Corticospinal tract | Fornix | Uncinate fasciculus | Middle cerebellar peduncle | |
|---|---|---|---|---|
| (Intercept) | 0.49 (0.43 to 0.55)*** | 0.37 (0.31 to 0.42)*** | 0.31 (0.26 to 0.36)*** | 0.42 (0.36 to 0.48)*** |
| PTSS group Yes vs. no |
0.00 (−0.02 to 0.03) | −0.01 (−0.03 to 0.02) | 0.01 (−0.02 to 0.03) | 0.02 (−0.01 to 0.04) |
| Age at diagnosis Continuous |
0.01 (0.00 to 0.01)* | 0.00 (−0.00 to 0.01) | 0.00 (−0.00 to 0.01) | 0.00 (−0.00 to 0.01) |
| NPS score Continuous |
0.00 (−0.00 to 0.01) | −0.00 (−0.01 to 0.00) | 0.00 (−0.01 to 0.01) | 0.00 (−0.00 to 0.01) |
| Tumor location Suprat. vs. rest |
−0.01 (−0.04 to 0.02) | −0.04 (−0.06 to −0.01)** | −0.03 (−0.05 to −0.00)* | 0.01 (−0.02 to 0.04) |
| Axial diffusivity (N = 52) | ||||
|---|---|---|---|---|
| Anterior commissure | Anterior thalamic radiation | Cingulum bundle dorsal | Cingulum bundle ventral | |
| (Intercept) | 0.88 (0.82 to 0.94)*** | 0.88 (0.84 to 0.93)*** | 0.87 (0.75 to 0.99)*** | 0.92 (0.84 to 1.00)*** |
| PTSS group Yes vs. no |
0.00 (−0.02 to 0.03) | 0.00 (−0.02 to 0.03) | 0.02 (−0.04 to 0.07) | 0.02 (−0.02 to 0.06) |
| Age at diagnosis Continuous |
−0.01 (−0.01 to −0.00)** | −0.00 (−0.01 to −0.00)* | −0.01 (−0.02 to 0.00) | −0.01 (−0.01 to −0.00)* |
| NPS score Continuous |
0.00 (−0.00 to 0.01) | 0.00 (−0.00 to 0.01) | 0.01 (−0.00 to 0.03) | 0.00 (−0.01 to 0.01) |
| Tumor location Suprat. vs. rest |
−0.01 (−0.04 to 0.02) | −0.01 (−0.03 to 0.01) | 0.05 (−0.00 to 0.11) | −0.01 (−0.04 to 0.03) |
| Corticospinal tract | Fornix | Uncinate Fasciculus | Middle Cerebellar Peduncle | |
|---|---|---|---|---|
| (Intercept) | 0.87 (0.79 to 0.94)*** | 0.99 (0.94 to 1.05)*** | 0.90(0.84 to 0.97)*** | 0.80 (0.74 to 0.86)*** |
| PTSS group Yes vs. no |
0.01 (−0.03 to 0.04) | 0.00 (−0.02 to 0.03) | 0.02 (−0.01 to 0.05) | 0.00 (−0.02 to 0.03) |
| Age at diagnosis Continuous |
−0.00 (−0.01 to 0.00) | −0.00 (−0.01 to −0.00)* | −0.00 (−0.01 to 0.00) | −0.00 (−0.01 to 0.00) |
| NPS score Continuous |
0.00 (−0.01 to 0.01) | 0.00 (−0.01 to 0.01) | 0.00 (−0.01 to 0.01) | 0.00 (−0.00 to 0.01) |
| Tumor location Suprat. vs. rest |
−0.01 (−0.05 to 0.02) | −0.01 (−0.04 to 0.01) | −0.03 (−0.06 to −0.00)* | −0.04 (−0.07 to −0.01)** |
Abbreviations. Suprat. = supratentorial medial structures.
* Significant at the P < .05 level.
** Significant at the P < .01 level.
*** Significant at the P < .001 level.
For exploratory reasons, we report the effects of the individual predictors. Older age at diagnosis was associated with higher FA of the anterior thalamic radiation (β = 0.01, P = .03) and corticospinal tract (β = 0.01, P = .03), and lower AD of the anterior commissure (β = −0.01, P < .01), anterior thalamic radiation (β = −0.00, P = .02), ventral part of the cingulum bundle (β = −0.01, P = .02), and fornix (β = −0.01, P = .03). Higher NPS (treatment intensity) score was associated to lower FA of the anterior thalamic radiation (β = −0.01, P = .05) and higher FA of the dorsal part of the cingulum bundle (β = 0.01, P < .01). Tumors in the supratentorial medial structures were related to lower FA in the fornix (β = −0.04, P < 0.01) and uncinate fasciculus (β = −0.03, P = .04), and lower AD of the uncinate fasciculus (β = −0.03, P = .03) and middle cerebellar peduncle (β = −0.04, P < .01).
Neurocognition and Limbic White Matter
To explore the relation between neurocognitive functioning and limbic white matter integrity, we calculated Pearson correlations. After correction for multiple testing, no significant correlations were found.
Discussion
This study aimed to investigate the effect of PTSS on neurocognitive functioning and limbic white matter integrity during the first year after pediatric brain tumor diagnosis. Higher PTSS was associated with poorer processing speed, and no effect of PTSS on limbic white matter integrity was found. White matter integrity was, however, associated with treatment intensity, age at diagnosis, and tumor location.
In general, the overall group had poorer attention and processing speed performance compared to healthy peers, and in the PTSS group, executive functioning was also lower than peers. In our sample, per domain, between 17% and 34% experienced these problems, and 59% of the patients were impaired in at least one neurocognitive domain. These neurocognitive impairments amongst pediatric brain tumor patients are common and frequently described, highlighting the clinical need to develop targeted interventions and prevent or reverse deterioration in a timely manner.16
Relationship Between PTSS and Neurocognition
The finding that PTSS was associated with poorer processing speed suggests that children with PTSS are at risk for declines in intellectual and psychosocial functioning.3 In our clinic, we have observed that slow movements and reaction speeds are often seen in pediatric brain tumor patients, and potentially there are interventions that could reduce the processing speed problems. For example, processing speed has been associated with PTSS in the literature in different populations.44,45 Earlier research45 in trauma survivors showed significant, small- to medium-sized improvements in several cognitive domains, including processing speed, after trauma-focused psychotherapy such as Eye Movement Desensitization and Reprocessing (EMDR)46. This earlier research has not been done in a childhood population. Still, EMDR is known to be helpful as a trauma intervention in children,47 and therefore, trauma therapy such as EMDR could potentially help cognitive problems, including in children with brain tumors.
It is possible that attention, executive functioning, and memory domains may also be impacted by PTSS, if PTSS levels are present for a longer period and with severe symptoms.44 In the first 3 months after diagnosis, we found no relationship between PTSS and neurocognitive performance, including processing speed, suggesting that time is an important factor (unpublished manuscript, Kremer—Hooft van Huijsduijnen, E.2). Also, cognitive domains seem to deteriorate years after cancer treatment, and PTSS may play a role in this, underlining the importance for future research to look at these associations over time.16 These findings suggest a critical time-window for clinical interventions between 3 months and 1 year after diagnosis to prevent long-term deterioration of neurocognitive functioning.
We hypothesized PTSS to induce both neurobiological changes in limbic white matter as well as neurocognitive problems, in which it is assumed that neurocognitive problems could be (partly) caused by those white matter alterations. Our findings, however, suggest that PTSS is related to neurocognition and not white matter, at least in the short term on processing speed. This phenomenon is in line with the limited capacity model of working memory,48 where cognitive representations of stressful life events compete with task demands for attentional resources. In other words, these unwanted thoughts from stressful life events compete for limited activation resources, hindering normal cognitive functioning.48 When working memory is compromised, it can potentially affect other aspects of neurocognitive performance as well. The lower processing speed scores in the PTSS group, compared to the no PTSS group that we found in our sample, could point to this mechanism. Overall, our results suggest that targeting PTSS may be useful for improving cognitive performance in these pediatric brain tumor groups.
Relationship Between PTSS and White Matter
In this study, there was no effect of PTSS on either FA or AD of the limbic white matter tracts. Earlier research with 17 maltreated children with PTSD showed reduced FA in the medial and posterior corpus callosum compared to controls, and in pediatric acute lymphoblastic leukemia survivors, higher re-experiencing PTSS was linked to increased functional connectivity of the amygdala.49,50 Possibly, effects in our sample can occur in the long-term, and/or different brain regions are impacted by PTSS. Limbic areas play a key role in the fear neural circuitry. However, other regions, such as the ventral anterior cingulate cortex and ventromedial prefrontal cortex, can extinguish fear responses of the amygdala by inhibitory control.51 Processing speed, for example, is known to be related to prefrontal processes in the brain.44 An alternative explanation is that children with lower processing speed may be more irritable, which resulted in higher PTSS scores. In this case, the relationship between processing speed and PTSS may be due to irritability, and therefore, a relationship between irritability and limbic white matter may not be found (due to different underlying mechanisms).
In addition, it could be that the changes in white matter integrity are not reflected in changes in FA or AD, but could be a change in connectivity and reflect in for example shorter tracts. Finally, earlier neuroimaging studies propose exposure to childhood trauma, even in the absence of PTSS, alters neurodevelopment fundamentally, and changes in the fear neural circuitry induce susceptibility to the development of PTSS/PTSD up to decades later. This further emphasizes the importance of unraveling the pathophysiology of PTSS in this vulnerable patient group18 using longer-term follow-up.
Strengths and Limitations
Strengths of this study are a relatively large sample size within a specific patient group, namely children with the highest risk of developing neurocognitive problems from all cancer diagnoses. It provides insight into different neurocognitive domains, where earlier research often focuses on global intellectual functioning and considers many different white matter tracts.16 In earlier research, neuropsychological deficits, medical factors, and white matter alterations resulting from cancer treatment are well-described as opposed to psychological factors. Psychological factors such as PTSS may be more targetable with interventions compared to white matter alterations and neuropsychological deficits. This study contributes to our understanding of the effect of PTSS on the white matter alterations and neuropsychological deficits. Additionally, the study highlights the importance of early testing on cognitive performance and PTSS. This was also the first study using the NPS score in brain tumor patients at an early phase, as previous studies focused on survivors.41,52 The NPS score is used as an indicator for treatment intensity and has been validated for survivors. Our analysis shows that the NPS is useful at an earlier stage in treatment and survivorship, particularly within the first year after diagnosis. However, future research is needed to validate the validity and reliability of using the NPS at different time points.
Limitations may be the estimates of PTSS, which were completed by proxy (parent) reported questionnaires. We know from our unpublished manuscript that proxy-reports and self-reports for child PTSS are correlated, but that proxy-reports yield lower PTSS scores than self-reports and this may be underestimating PTSS (unpublished manuscript, Kremer—Hooft van Huijsduijnen, E2). This could potentially induce a group bias where the ‘no PTSS’ group may then in fact contain participants with considerable PTSS symptoms. That could in turn explain why no relation was found between PTSS scores and limbic white matter integrity. In this analysis, we chose to focus on proxy-reported scores because of sample size and to limit data imputation. Additionally, patients who were included at T1 (and not T0) may be because they were experiencing stress; the fact that 3 out of 9 later included patients had subclinical scores (>25) at T1 endorse this statement. Some participants only completed the PTSS questionnaire at one time point, and therefore, changes over time are unknown. Additionally, no data on psychosocial care was collected. Consequently, we do not know to what extent the patients had anxiety problems or received psychotherapy or other psychological interventions regarding their medical traumatic stress.
One limitation of the relatively large sample size is that the sample is heterogeneous regarding the tumor type. All brain tumor patients from 6 years and older were eligible for the study, which resulted in 18 different diagnoses. Therefore, including tumor type as a covariate was not statistically feasible. In future studies, it is suggested to add tumor type (e.g., histological diagnosis) as potential covariates to further explore these relationships.
The MRI TRACULA pipeline may have been negatively impacted by the tumor, tumor cavities, and potentially disrupted white matter pathways due to neurosurgery. Studies utilizing TRACULA with pediatric brain tumor patients have not been conducted before, making it difficult to determine how this has impacted the outcomes. We do, however, replicate findings from the literature on the covariates (age, tumor, treatment), indicating reliable processing of the MRI analysis pipeline.
Lastly, as mentioned above, we were interested in early brain changes caused by heightened PTSS, although 1 year after diagnosis may be too early to detect subtle changes, and thus, we recommend longer follow-up designs for future research. Furthermore, we recommend PTSS questionnaires to be administered more frequently, including self-reports and/or objective stress measures such as hair cortisol. MRI analyses can include gray matter volumetric analyses of limbic areas such as the hippocampus and amygdala. Finally, it will be important to assess the effect of trauma interventions on neurocognitive functioning.18,53
Conclusions
In conclusion, PTSS during the first year after pediatric brain tumor diagnosis was associated with lower processing speed performance, but not with limbic white matter integrity. Ongoing efforts to unravel the relationship of psychological well-being with neurocognitive and neurobiological outcomes are important. This is of clinical importance as it may guide the development of targeted interventions to prevent or reverse functional decline and optimize health-related quality of life. PTSS and neurocognitive impairments were frequently observed in this sample and therefore regular screening and timely referrals for psychological and educational support are important throughout brain tumor treatment and survivorship.
Acknowledgments
We thank the patients and their families who contributed to the SuSPeCT study.
Kremer-Hooft van Huijsduijnen et. al, “Posttraumatic stress, sleep, and neurocognitive problems in children newly diagnosed with a pediatric brain tumor.” In preparation. 2Kremer-Hooft van Huijsduijnen et. al, “Posttraumatic stress symptoms in children and parents shortly after pediatric brain tumor: prevalence and risk factors." In preparation.
Contributor Information
Anne E M Leenders, Princess Máxima Center for Pediatric Oncology, Utrecht, The Netherlands.
Eva Kremer-Hooft van Huijsduijnen, Princess Máxima Center for Pediatric Oncology, Utrecht, The Netherlands.
Bruno Robalo, Princess Máxima Center for Pediatric Oncology, Utrecht, The Netherlands.
Rosa van Male, Princess Máxima Center for Pediatric Oncology, Utrecht, The Netherlands.
Alberto De Luca, University Medical Center Utrecht, Utrecht, The Netherlands.
Rachèl Kemps, Princess Máxima Center for Pediatric Oncology, Utrecht, The Netherlands.
Eelco Hoving, Princess Máxima Center for Pediatric Oncology, Utrecht, The Netherlands.
Maarten H Lequin, Princess Máxima Center for Pediatric Oncology, Utrecht, The Netherlands; University Medical Center Utrecht, Utrecht, The Netherlands.
Martha A Grootenhuis, Princess Máxima Center for Pediatric Oncology, Utrecht, The Netherlands.
Marita Partanen, Princess Máxima Center for Pediatric Oncology, Utrecht, The Netherlands.
Funding
We gratefully acknowledge funding of the Dutch Children Cancer-free Foundation (KIKA) and the Princess Maxima Center for Pediatric Oncology.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Authorship statement
Study design: E.K.H.v.H., A.D.L., R.K., E.H., M.L., M.G., M.P. Data collection: A.L., E.K.H.v.H., R.v.M, M.L. Data analysis: A.L., E.K.H.v.H., B.R., R.v.M. Technical support: B.R., A.D.L. Draft of the manuscript: A.L., E.K.H.v.H. Critically review manuscript: A.L., E.K.H.v.H., B.R., R.v.M., A.D.L., R.K., E.H., M.L., M.G., M.P. All authors read and approved the final manuscript.
Data availability
This includes data from an ongoing study, and therefore data is under embargo (as of February 2024).
References
- 1. Schreiber JE, Gurney JG, Palmer SL, et al. Examination of risk factors for intellectual and academic outcomes following treatment for pediatric medulloblastoma. Neuro-oncology. 2014;16(8):1129–1136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Zeltzer LK, Recklitis C, Buchbinder D, et al. Psychological status in childhood cancer survivors: a report from the childhood cancer survivor study. J Clin Oncol. 2009;27(14):2396–2404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Ruiter MA de, Meeteren AYNS van, Vuurden DG van, et al. Psychosocial profile of pediatric brain tumor survivors with neurocognitive complaints. Qual Life Res. 2016;25(2):435–446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Stavinoha PL, Askins MA, Powell SK, Smiley NP, Robert RS.. Neurocognitive and psychosocial outcomes in pediatric brain tumor survivors. Bioeng. 2018;5(3):73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Brinkman TM, Reddick WE, Luxton J, et al. Cerebral white matter integrity and executive function in adult survivors of childhood medulloblastoma. Neuro-oncology. 2012;14(suppl_4):iviv25–iviv36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Brinkman TM, Krasin MJ, Liu W, et al. Long-term neurocognitive functioning and social attainment in adult survivors of pediatric CNS tumors: results from the St Jude lifetime cohort study. J Clin Oncol. 2016;34(12):1358–1367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Krull KR, Hardy KK, Kahalley LS, Schuitema I, Kesler SR.. Neurocognitive outcomes and interventions in long-term survivors of childhood cancer. J Clin Oncol. 2018;36(21):2181–2189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Ruiter MAD, Mourik RV, Meeteren AYNSV, Grootenhuis MA, Oosterlaan J.. Neurocognitive consequences of a paediatric brain tumour and its treatment: a meta-analysis. Dev Medicine Child Neurology. 2013;55(5):408–417. [DOI] [PubMed] [Google Scholar]
- 9. Kesler SR, Sleurs C, McDonald BC, et al. Brain imaging in pediatric cancer survivors: correlates of cognitive impairment. J Clin Oncol. 2021:39(16):1775–1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Ailion AS, Hortman K, King TZ.. Childhood brain tumors: a systematic review of the structural neuroimaging literature. Neuropsychol Rev. 2017;27(3):220–244. [DOI] [PubMed] [Google Scholar]
- 11. Nelson MCB, O’Neil SH, Tanedo J, et al. Brain biomarkers and neuropsychological outcomes of pediatric posterior fossa brain tumor survivors treated with surgical resection with or without adjuvant chemotherapy. Pediatr Blood Cancer. 2020;68(2):e28817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Aarsen FK, Dongen HRV, Paquier PF, Mourik MV, Catsman-Berrevoets CE.. Long-term sequelae in children after cerebellar astrocytoma surgery. Neurology. 2004;62(8):1311–1316. [DOI] [PubMed] [Google Scholar]
- 13. Hanzlik E, Woodrome SE, Abdel-Baki M, Geller TJ, Elbabaa SK.. A systematic review of neuropsychological outcomes following posterior fossa tumor surgery in children. Child’s Nerv Syst. 2015;31(10):1869–1875. [DOI] [PubMed] [Google Scholar]
- 14. Hardy KK, Bonner MJ, Willard VW, Watral MA, Gururangan S.. Hydrocephalus as a possible additional contributor to cognitive outcome in survivors of pediatric medulloblastoma. Psycho Oncol. 2008;17(11):1157–1161. [DOI] [PubMed] [Google Scholar]
- 15. Mulhern RK, Palmer SL, Reddick WE, et al. Risks of young age for selected neurocognitive deficits in medulloblastoma are associated with white matter loss. J Clin Oncol. 2001;19(2):472–479. [DOI] [PubMed] [Google Scholar]
- 16. Oyefiade A, Paltin I, Luca CRD, et al. Cognitive risk in survivors of pediatric brain tumors. J Clin Oncol. 2021:39(16):1718–1726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bellis MDD, Zisk A.. The biological effects of childhood trauma. Child Adol Psych Cl. 2014;23(2):185–222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Marusak HA, Harper FW, Taub JW, Rabinak CA.. Pediatric cancer, posttraumatic stress and fear-related neural circuitry. Int J Hematologic Oncol. 2019;8(0):IJH17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Kosir U, Wiedemann M, Wild J, Bowes L.. Psychiatric disorders in adolescent cancer survivors: A systematic review of prevalence and predictors. Cancer Reports. 2019;2(e1168):1–15. [Google Scholar]
- 20. Maas A, Maurice-Stam H, Kremer LCM, et al. Psychosocial outcomes in long-term Dutch adult survivors of childhood cancer: The DCCSS-LATER 2 psycho-oncology study. Cancer. 2023;129(16):2553–2567. [DOI] [PubMed] [Google Scholar]
- 21. Bruce M. A systematic and conceptual review of posttraumatic stress in childhood cancer survivors and their parents. Clin Psychol Rev. 2006;26(3):233–256. [DOI] [PubMed] [Google Scholar]
- 22. Price J, Kassam-Adams N, Alderfer MA, Christofferson J, Kazak AE.. Systematic Review: A reevaluation and update of the integrative (Trajectory) model of pediatric medical traumatic stress. J Pediatr Psychol. 2016;41(1):86–97. [DOI] [PubMed] [Google Scholar]
- 23. Kaplan Z, Iancu I, Bodner E.. A Review of psychological debriefing after extreme stress. Psychiatr Serv. 2001;52(6):824–827. [DOI] [PubMed] [Google Scholar]
- 24. Siehl S, King JA, Burgess N, Flor H, Nees F.. Structural white matter changes in adults and children with posttraumatic stress disorder: a systematic review and meta-analysis. Neuroimage Clin. 2018;19:581–598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Ewing-Cobbs L, DeMaster D, Watson CG, et al. Post-traumatic stress symptoms after pediatric injury: relation to pre-frontal limbic circuitry. J Neurotrauma. 2019;36(11):1738–1751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Kribakaran S, Danese A, Bromis K, Kempton MJ, Gee DG.. Meta-analysis of structural magnetic resonance imaging studies in pediatric posttraumatic stress disorder and comparison with related conditions. Biol Psychiat Cognit Neurosci Neuroimag. 2020;5(1):23–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Hooft van Huijsduijnen E, Partanen M, Grootenhuis M, et al. Sleep problems and impact of obstructive hydrocephalus in newly diagnosed pediatric brain tumor patients. EJC Paediatr Oncol. 2023;2:100124. [Google Scholar]
- 28. Perrin S, Meiser-Stedman R, Smith P.. The Children’s Revised Impact of Event Scale (CRIES): validity as a screening instrument for PTSD. Behavioural and Cognitive Psychotherapy. 2005;33(33):487–498. [Google Scholar]
- 29. Verlinden E, Meijel EPM van, Opmeer BC, et al. Characteristics of the children’s revised impact of event scale in a clinically referred Dutch sample. J Trauma Stress. 2014;27(3):338–344. [DOI] [PubMed] [Google Scholar]
- 30. Sonneville L de. Handboek Amsterdamse Neuropsychologische Taken. Boom; 2014. [Google Scholar]
- 31. Wechsler D. Wechsler Adult Intellgience Scale - Fourth Edition (WAIS-IV-NL). Pearson: Amsterdam. Published online 2012. [Google Scholar]
- 32. Wechsler D. Wechsler Intelligence Scale for Children - Fifth Edition (WISC-V-NL). Pearson: Amsterdam. Published online 2018. [Google Scholar]
- 33. Magalhães S de S, Malloy-Diniz LF, Hamdan AC.. Validity convergent and reliability test-retest of the Rey Auditory Verbal Learning Test. Clinical Neuropsychiatry. 2012;9(3):129–137. [Google Scholar]
- 34. Fischl B, Salat DH, Busa E, et al. Whole brain segmentation automated labeling of neuroanatomical structures in the human brain. Neuron. 2002;33(3):341–355. [DOI] [PubMed] [Google Scholar]
- 35. Reuter M, Rosas HD, Fischl B.. Highly accurate inverse consistent registration: a robust approach. Neuroimage. 2010;53(4):1181–1196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Segonne F, Dale AM, Busa E, et al. A hybrid approach to the skull stripping problem in MRI. Neuroimage. 2004;22(3):1060–1075. [DOI] [PubMed] [Google Scholar]
- 37. Fischl B, Salat DH, Kouwe AJW van der, et al. Sequence-independent segmentation of magnetic resonance images. Neuroimage. 2004;23(Supplement 1):S69–S84. [DOI] [PubMed] [Google Scholar]
- 38. Han X, Jovicich J, Salat D, et al. Reliability of MRI-derived measurements of human cerebral cortical thickness: the effects of field strength, scanner upgrade and manufacturer. Neuroimage. 2006;32(1):180–194. [DOI] [PubMed] [Google Scholar]
- 39. Reuter M, Schmansky NJ, Rosas HD, Fischl B.. Within-subject template estimation for unbiased longitudinal image analysis. Neuroimage. 2012;61(4):1402–1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Yendiki A, Panneck P, Srinivasan P, et al. Automated probabilistic reconstruction of white-matter pathways in health and disease using an atlas of the underlying anatomy. Front Neuroinform. 2011;5(23):1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Taiwo Z, Na S, King TZ.. The neurological predictor scale: a predictive tool for long-term core cognitive outcomes in survivors of childhood brain tumors. Pediatr Blood Cancer. 2017;64(1):172–179. [DOI] [PubMed] [Google Scholar]
- 42. Micklewright JL, King TZ, Morris RD, Krawiecki N.. Quantifying pediatric neuro-oncology risk factors: development of the neurological predictor scale. J Child Neurol. 2008;23(4):455–458. [DOI] [PubMed] [Google Scholar]
- 43. Benjamini Y, Hochberg Y.. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Statist Soc Ser B (Methodological). 1995;57(1):289–300. [Google Scholar]
- 44. Scott JC, Matt GE, Wrocklage KM, et al. A quantitative meta-analysis of neurocognitive functioning in posttraumatic stress disorder. Psychol Bull. 2015;141(1):105–140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Nijdam MJ, Martens IJM, Reitsma JB, Gersons BPR, Olff M.. Neurocognitive functioning over the course of trauma-focused psychotherapy for PTSD: Changes in verbal memory and executive functioning. Br J Clin Psychol. 2018;57(4):436–452. [DOI] [PubMed] [Google Scholar]
- 46. Shapiro F. Eye movement desensitization: a new treatment for post-traumatic stress disorder. J Behav Ther Exp Psychiatry. 1989;20(3):211–217. [DOI] [PubMed] [Google Scholar]
- 47. Rodenburg R, Benjamin A, Roos C de, Meijer AM, Stams GJ.. Efficacy of EMDR in children: a meta-analysis. Clin Psychol Rev. 2009;29(7):599–606. [DOI] [PubMed] [Google Scholar]
- 48. Klein K, Boals A.. The relationship of life event stress and working memory capacity. Appl Cognit Psychol. 2001;15(5):565–579. [Google Scholar]
- 49. Marusak HA, Iadipaolo AS, Paulisin S, et al. Emotion-related brain organization and behavioral responses to socioemotional stimuli in pediatric cancer survivors with posttraumatic stress symptoms. Pediatr Blood Cancer. 2019;66(1):e27470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Jackowski AP, Douglas-Palumberi H, Jackowski M, et al. Corpus callosum in maltreated children with posttraumatic stress disorder: a diffusion tensor imaging study. Psychiatry Res. 2008;162(3):256–261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Keding TJ, Herringa RJ.. Abnormal structure of fear circuitry in pediatric post-traumatic stress disorder. Neuropsychopharmacology. 2015;40(3):537–545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Srsich AR, McCury MD, Fantozzi PM, Hocking MC.. Predicting neuropsychological late effects in pediatric brain tumor survivors using the neurological predictor scale and the pediatric neuro-oncology rating of treatment intensity. J Int Neuropsychol Soc. 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Heuvel LL van den, Stalder T, Plessis S, et al. Hair cortisol levels in posttraumatic stress disorder and metabolic syndrome. Stress. 2020;23(5):577–589. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
This includes data from an ongoing study, and therefore data is under embargo (as of February 2024).