Abstract
A blood crossmatch is essential to ensure RBC compatibility for previously transfused dogs. There is no gold standard crossmatch method for dogs, although the standards used most commonly by academic institutions and reference laboratories are the tube and gel-column crossmatches. Addition of anti-canine globulin (ACG) has been suggested to increase detection of RBC incompatibilities. Our objective was to determine if there is a correlation between results of a standard and an ACG-enhanced gel-column crossmatch in detecting post-transfusion RBC alloimmunization. Pre- and post-transfusion serum or plasma samples were obtained from 33 dogs for major crossmatches to 1–6 (median: 3) blood donors. Crossmatches were performed with (n = 202) and without (n = 202) ACG, with results scored by 4 observers, 3 of whom were anonymized. Ten of 33 (30%) dogs had major crossmatch incompatibilities post-transfusion. RBC incompatibilities (2–4+ agglutination) were detected only with ACG in 4 dogs, only without ACG in 3 dogs, and with both methods in 3 dogs. There was fair correlation between crossmatch methods for determination of compatibility (ρ = 0.34; p < 0.001) and incompatibility (ρ = 0.35; p < 0.001) scores. Among 4 observers, there was near-perfect agreement in determining compatibility (κ = 0.97; p < 0.001) and substantial agreement in overall scoring of incompatibility (κ = 0.77; p < 0.001). Our results suggest that detection of RBC incompatibilities in dogs can be maximized by performing a gel-column crossmatch both with and without ACG enhancement.
Keywords: alloimmunization, antiglobulin, blood transfusion, crossmatch, dogs
The primary goal of transfusion medicine is to provide safe and effective blood products to patients. In addition to careful screening of donors for blood-borne pathogens that could be transmitted via transfusion and proper blood collection and storage of blood components, pre-transfusion blood compatibility testing is essential. For humans, such testing includes ABO and Rh typing, an antibody screen (recipient’s serum incubated with selected cells expressing most RBC phenotypes), a standard (traditional) major crossmatch (recipient’s serum incubated with donor’s RBCs), and an anti-human globulin (AHG) crossmatch (traditional major crossmatch with addition of AHG reagent).8,16,27 The purpose of the AHG crossmatch is to detect antibodies directed against potential low-frequency antigens on the donor’s RBCs that are absent from RBCs used in the antibody screen. 16 When no clinically significant antibodies have been detected by an antibody screen, and review of the patient history and a validated computerized or electronic crossmatch to ensure ABO compatibility is available, packed RBC (pRBC) units can be released without the immediate spin (i.e., standard tube method) and AHG phases of the crossmatch. 8
For dogs, pre-transfusion compatibility testing, at a minimum, includes blood typing for dog erythrocyte antigen (DEA)1, clinically the most important blood group given its strong antigenicity and frequency of ~50% across many dog breeds. 13 Although there is controversy regarding the need for a blood crossmatch for transfusion-naïve dogs given their lack of clinically significant naturally occurring RBC alloantibodies, there is a consensus that a blood crossmatch is indicated for dogs that received RBCs ≥4 d earlier.6,7,11,13,18,20,26 The need for, or potential benefit of, an anti-canine globulin (ACG)-enhanced crossmatch has not been clearly established in veterinary medicine.
There is no gold standard blood crossmatch method for dogs, although the standards used most commonly by veterinary academic institutions and reference laboratories are the tube and gel-column crossmatches. The gel-column crossmatch is used commonly in human transfusion medicine 8 and has been documented to have near-perfect agreement among observers for determination of blood com-patibility in horses 4 and dogs. 29 Blood crossmatch kits containing gel tubes or immunochromatographic strips, both with and without ACG, have been developed for in-clinic use. Crossmatch methods to detect naturally occurring or post-transfusion RBC alloantibodies in dogs have been compared in several studies, with variable results.11,14,23,28,32 In a study comparing an ACG-enhanced immunochromatographic strip to an ACG-enhanced gel-column crossmatch for previously transfused dogs, the 2 methods gave entirely concordant results. 11 In contrast, in a study comparing 2 canine blood crossmatch kits, an ACG-enhanced immunochromatographic strip and a non–ACG-enhanced gel tube, to a laboratory-tube crossmatch (with and without ACG), there was no agreement between methods. 23 The gel-tube kit did not detect any of the 24 incompatibilities noted in the tube crossmatch, and the ACG-enhanced immunochromatographic strip detected only 3 of 26 inco-mpatibilities detected in the tube crossmatch. 23 The tube crossmatch included 4 phases of interpretation, with the phase in which incompatibility was first noted recorded: immediate (tube containing recipient serum and donor RBCs was centrifuged and evaluated for agglutination without an incubation period), incubation at room temperature for 30–60 min, incubation of a second tube at 37°C for 30–60 min, and after addition of ACG to tubes incubated at 37°C. 23 Of 145 incompatible tube crossmatches, 67 (46.2%) were identified only after addition of ACG. 23 Although these results suggest that addition of ACG to the tube crossmatch increases detection of RBC alloantibodies in dogs, the potential benefit of ACG in other crossmatch methods is unclear given that different crossmatch techniques (e.g., standard tube crossmatch and ACG-enhanced immunochromatographic strip) 32 or only ACG-enhanced crossmatch methods (e.g., ACG-enhanced immunochromatographic strip and ACG-enhanced gel column) 11 were used for comparison in other studies.
A compatible blood crossmatch indicates that the recipient does not have detectable alloantibodies against the donor’s RBC antigens, decreasing the chances of an acute hemolytic transfusion reaction. However, a compatible crossmatch does not prevent the recipient from developing RBC alloantibodies post-transfusion. The reported incidence of post-transfusion RBC alloimmunization in dogs is 33–66%.11,14,18,20,23 Several factors could be involved in development of RBC alloimmunization, including the degree of antigenic difference between donor(s) and recipient, antigen immunogenicity, and environmental factors, including biologic changes in stored RBC units.9,17 In a murine transfusion model, RBCs became progressively more immunogenic as a function of storage time, with a 10- to 100-fold higher level of RBC alloantibodies in mice receiving RBCs stored for 14 d (considered “old” for mice) compared to those administered fresh RBCs. 17 To our knowledge, there is limited information regarding the effect of RBC storage duration on post-transfusion alloimmunization in dogs. 18
Our primary objective was to determine if there is a correlation between results of a standard gel-column crossmatch, currently used by our institution’s clinical laboratory, and an ACG-enhanced gel-column crossmatch. Our secondary objective was to evaluate the potential effect of duration of storage of canine RBCs on post-transfusion alloimmunization in a convenience sampling of a heterogeneous hospital population of dogs.
Materials and methods
Canine blood donors
Canine pRBCs were obtained exclusively from our institution’s blood bank, which has an established volunteer blood donor program. Canine donor requirements include: body weight ≥25 kg, 1–8 y-old, and good health, based on history, physical examination, and annual screening that includes a CBC, biochemistry profile, and testing for blood-borne pathogens, as outlined in the consensus statement of American College of Veterinary Internal Medicine. 31 In addition, blood donors must be transfusion-naïve and not receiving any medications, other than heartworm and flea–tick preventives. For our study, the term “original donor” refers to the blood donor(s) that provided pRBCs for transfusion to a particular recipient, whereas “random donor” refers to blood donors that were a source of RBCs for crossmatches only (i.e., a recipient was not transfused with pRBCs from these donors).
Blood transfusion recipients
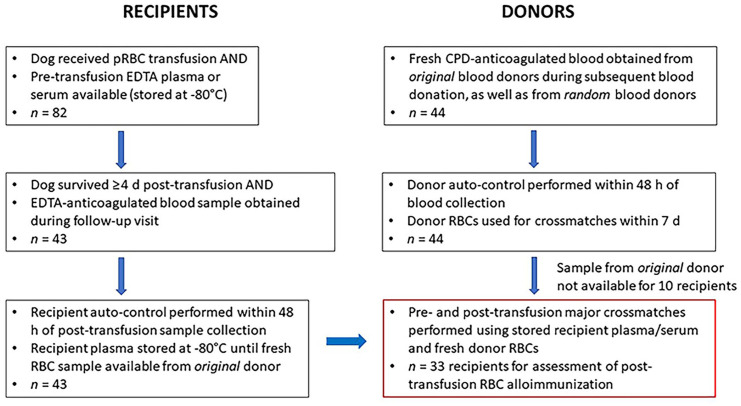
Dogs that received DEA1 type–compatible pRBCs were retrospectively enrolled in our observational study between July 2018 and February 2020 at the Matthew J. Ryan Veterinary Hospital of the University of Pennsylvania (MJR-VHUP). Pre-transfusion serum and/or EDTA-anticoagulated plasma from recipients were obtained from surplus samples in the clinical laboratory. Exclusion criteria included a previous blood transfusion, a diagnosis of immune-mediated hemolytic anemia (based on anemia and 1 or more of the following: persistent RBC agglutination, positive direct antiglobulin test, and moderate-to-marked spherocytosis), or insufficient pre-transfusion sample volume (<500 µL). Samples were stored at −80°C. When recipient dogs returned to the hospital for a follow-up visit, a 2-mL post-transfusion EDTA-anticoagulated blood sample was collected after obtaining owner consent. A recipient auto-control was performed within 48 h of collection of the post-transfusion sample, and the plasma was stored at −80°C until a fresh pRBC unit was available from the original blood donor(s) for a major crossmatch (Fig. 1). Data recorded included recipient’s signalment, DEA1 blood type, reason for transfusion, date of transfusion, donor identification number, date of collection of blood unit, number and volume of pRBC transfusions, concurrent blood products and immunosuppressive medications administered, and date on which post-transfusion plasma sample was collected. The study protocol was approved by the University of Pennsylvania Institutional Animal Care and Use Committee (protocol 806557) and the MJR-VHUP Privately Owned Animal Protocol Committee (protocol 538).
Figure 1.
Schematic demonstrating the sequence of collection and storage of blood samples from recipient and donor dogs for pre- and post-transfusion crossmatches and auto-controls. CPD = citrate–phosphate–dextrose; pRBC = packed RBC.
Blood crossmatch procedure
To detect RBC alloimmunization, major crossmatches were performed using the recipient’s pre- and post-transfusion serum or plasma samples and fresh RBCs (citrate–phosphate–dextrose [CPD]-anticoagulated whole blood tube segments stored <1 wk) from the original blood donor(s) obtained during a subsequent blood donation, noting any new RBC incompatibilities and the degree of incompatibility. In addition, donor and recipient auto-controls were performed. All major crossmatches and auto-controls were performed using a standard gel column (ID-micro typing system gel test cards; Ortho-Clinical Diagnostics), both with and without a polyspecific ACG reagent containing anti-IgG, anti-IgM, and anti-C3 (ImmunO; MP Biomedicals), and graded as described previously. 11 Briefly, RBCs were washed 3 times in buffered blood bank 0.85% isotonic saline solution, and a 0.8% RBC suspension was prepared by adding 10 µL of washed pRBCs to 1 mL of low ionic strength saline solution (ID-micro typing system MTS diluent 2; Ortho-Clinical). For the standard (non–ACG-enhanced) crossmatch, 25 µL of plasma or serum and then 50 µL of RBC suspension were pipetted onto the top of gel columns. Gel column cards were incubated (ID-incubator 37 SI, ID micro typing system; DiaMed) at 37°C for 15 min and then centrifuged (ID-centrifuge 12 S II, ID micro typing system; DiaMed) for 10 min. For the ACG-enhanced crossmatch, 50 µL of RBC suspension and 50 µL of plasma or serum were added to a 3-mL tube, mixed, and incubated at 37°C for 15 min. After incubation, the RBC suspension was washed twice in buffered saline to remove free antibodies. After the second wash, the supernatant was removed, and 15 µL of buffered saline was added to resuspend the RBC pellet, which was aspirated and added to the top of a gel column. ACG diluted 1:2 in 10 µL of buffered saline was then added to the gel column and centrifuged as above. The agglutination was graded as follows: 0 (negative, or compatible) = all RBCs were at the bottom of the gel column; 1± a few RBC agglutinates were dispersed in the gel, but most of the RBCs were at the bottom of the gel column; 2± all RBC agglutinates were dispersed in the gel; 3± some RBC agglutinates were dispersed in the upper part of the gel, most of the RBCs remained on the top of the gel column; and 4± all RBCs were agglutinated and remained on the top of the gel column. Any RBC agglutination noted in the gel column was recorded, including the degree of incompatibility. If there was a sufficient volume of pre- and post-transfusion serum or plasma after performing a major crossmatch to original blood donor(s), additional crossmatches were performed using random donors.
Interobserver variation
Results of all auto-controls and crossmatches performed were recorded by photographing gel columns immediately after centrifugation. Crossmatch results were independently scored by A. Thomas-Hollands (a medicine resident with no prior crossmatch experience), 2 clinical laboratory technicians (S. Fromm, N. Chappini) with 10–14 y of experience performing and interpreting crossmatches, and 1 board-certified clinical pathologist (N. Weinstein) with 20 y of experience in blood compatibility testing. All observers, except A. Thomas-Hollands, were anonymized to the identification of samples. All crossmatch results are reported as the median score of the 4 observers.
Statistical analysis
Most continuous variables were not normally distributed, as determined visually and by skewness and kurtosis tests for normality. Therefore, results are reported as median (range) for continuous variables, and as counts and percentages for categorical variables. The Spearman correlation test was used to evaluate the degree of correlation between variables that were not normally distributed. Logistic regression was utilized to investigate the correlation between binary outcomes and continuous predictor variables. The Fisher exact test was employed to examine the relationship between 2 categorical variables because 1 or more cells had a frequency of ≤5. The kappa-statistic was used to determine interobserver variation, with kappa (κ) values of 0 and 1 indicating random and perfect agreement, respectively. For intermediate values, the following interpretations were used: <0 = poor agreement; 0–0.2 = slight agreement; 0.21–0.4 = fair agreement; 0.41–0.60 = moderate agreement; 0.61–0.80 = substantial agreement; and 0.81–1.0 = almost-perfect agreement. A p ≤ 0.05 was considered significant for all tests. All statistical evaluations were performed using a statistical software package (v.14.0 for Mac; Stata).
Results
Transfusion recipient characteristics
With exclusion criteria applied, pre-transfusion plasma or serum samples were available for 82 dogs. Thirteen dogs died or were euthanized <4 d post-transfusion, and 26 dogs did not return for a follow-up visit. Of the remaining 43 dogs, fresh blood samples from donors were not available for post-transfusion crossmatches for 10 recipients. Therefore, 33 recipient dogs for which both pre- and post-transfusion plasma or serum samples were available were included in our study (Fig. 1). The median age was 8 y (range: 9 wk to 16 y), and 17 breeds were represented, with mixed-breed dogs (n = 8) and Golden Retrievers (n = 6) being most common (Suppl. Table 1). There were 15 castrated and 3 intact males, and 13 spayed and 2 intact females. The median body weight was 26 kg (range: 4.6–47.6 kg). Nineteen dogs were blood-typed as DEA1+ and 14 dogs as DEA1–.
RBC transfusions and concurrent treatments
Causes of anemia resulting in the administration of pRBC transfusions included blood loss (n = 21), ineffective erythropoiesis (n = 11), and unknown (n = 1; Suppl. Table 1). A total of 51 pRBC units were administered to 33 dogs, with a median of 1 (range: 1–4) pRBC transfusion per patient. During our study (19 mo), blood donations were obtained from some donor dogs more than once. In total, RBCs from 44 blood donors were used in post-transfusion crossmatches. The median storage duration of pRBC units was 21 d (range: 1–33). Four dogs received concurrent blood products (fresh frozen plasma, n = 2; platelet-rich plasma, n = 1; canine albumin, n = 1). Ten dogs received prednisone for suspected immune-mediated disease (e.g., immune thrombocytopenia, precursor immune-mediated anemia) or neoplasia, and 7 of the 10 dogs also received a second immunosuppressive medication (cyclosporine, n = 3; mycophenolate, n = 3; and cyclophosphamide, doxorubicin, vincristine, and prednisone, n = 1; Suppl. Table 2).
RBC alloimmunization
Major crossmatches using pre-transfusion plasma or serum
Recipient auto-controls were not performed for pre-transfusion crossmatches because of the retrospective identification and collection of pre-transfusion plasma and serum samples from the clinical laboratory. In major crossmatches performed using recipients’ pre-transfusion plasma or serum samples and RBCs from original and random blood donors, RBC agglutination was observed for 7 of 33 dogs (21%; Suppl Table 3). The median number of blood donors to which these 7 recipient dogs were crossmatched was 3 (range: 2–4). Of these 7 dogs, 6 (dogs 1, 2, 4–7) had RBC agglutination only with the addition of ACG (agglutination scores of 1.5–4+) and 1 (dog 3) only without ACG (agglutination score 4+) in crossmatches with 1 or more donors. Despite RBC agglutination noted on major crossmatches using pre-transfusion plasma or serum, 6 of the 7 recipient dogs did not have evidence of RBC agglutination in major crossmatches using post-transfusion plasma. The 7th recipient dog had RBC agglutination (2+ and 3+, with and without ACG enhancement, respectively) on the major crossmatch with only 1 random donor using post-transfusion plasma. This is the same recipient dog (dog 1) that had 4+ RBC agglutination with 3 donors (with ACG enhancement only) using pre-transfusion plasma or serum. In light of the RBC agglutination noted using pre-transfusion plasma or serum, the agglutination noted in this post-transfusion crossmatch was not considered to be evidence of development of RBC alloimmunization.
Major crossmatches using post-transfusion plasma
EDTA-anticoagulated blood samples were collected from the 33 recipient dogs at the following times after the first pRBC transfusion: <2 wk (n = 5), 2–8 wk (n = 22), and >8 wk (n = 6). The median time to sample collection was 24 d (range: 6–388 d) after the first pRBC transfusion. Recipient auto-controls performed on all post-transfusion samples, as well as all donor auto-controls, were negative (i.e., they did not show evidence of RBC agglutination in the gel columns with or without addition of ACG). The median number of blood donors evaluated per recipient in major crossmatches was 3 (range: 1–6).
Ten of 33 dogs (30%) developed major crossmatch incompatibilities post-transfusion (Suppl. Table 4). None of these 10 dogs had evidence of RBC agglutination on major crossmatches performed using pre-transfusion plasma or serum. Among these 10 recipient dogs, the total number of crossmatches performed using RBCs from original donors was 13, with 8 incompatibilities observed; using RBCs from random donors, 18 crossmatches were performed, with 8 incompatibilities observed (Suppl. Table 4). Seven of the 10 recipient dogs (dogs 8–11, 13, 14, 16) were crossmatch-incompatible with their original donor(s), and 2 of these 7 dogs (dogs 8, 14) also were incompatible with random donors. Three of the 10 recipient dogs (dogs 12, 15, 17) developed major crossmatch incompatibilities with random donors but not their original donor(s). RBC agglutination (2–3.25+) was detected only with addition of ACG in 4 of the 10 dogs (dogs 8, 9, 14, 17), in a total of 9 major crossmatches (Fig. 2). Three of the 10 recipient dogs (dogs 12, 13, 16) had RBC agglutination (2.25–3+) detected only without addition of ACG in a single major crossmatch each. Two of the 10 recipient dogs (dogs 10, 11) had RBC incompatibilities detected both with (2.75, 4+) and without (both 2+) addition of ACG in a single major crossmatch each. Finally, 1 of the 10 recipient dogs (dog 15) had detectable RBC agglutination in a major crossmatch with 1 donor only with addition of ACG (3+) and both with and without ACG enhancement (4 and 3+, respectively) with a second donor.
Figure 2.

Gel column major crossmatches using pre- and post-transfusion plasma samples (53A and 53B [dog 22], respectively) with RBCs from donor 5243 without (columns 1, 2) and with (columns 3, 4) anti-canine globulin (ACG), demonstrating detection of RBC alloantibodies post-transfusion (3.5+ incompatibility) only with the addition of ACG.
Agreement between standard gel-column crossmatches with and without ACG enhancement
In light of the RBC agglutination noted in pre-transfusion major crossmatches of 7 dogs and the absence of agglutination in the corresponding post-transfusion crossmatches for 6 of the 7 dogs, the observed RBC agglutination was not interpreted as evidence of naturally occurring RBC alloantibodies (at least in 6 dogs) but rather nonspecific agglutination associated with the recipients’ underlying conditions. Therefore, pre-transfusion crossmatches were not included in the assessment of correlation between crossmatch methods. With 33 recipients each crossmatched to 1–6 donors (median: 3), there were 101 post-transfusion crossmatch pairs performed with and without ACG, for a total of 202 crossmatches. For determination of compatibility versus incompatibility, there was fair correlation (ρ = 0.34; p < 0.001) between gel-column crossmatches performed with and without ACG enhancement. Comparing incompatibility scores, there was also fair correlation (ρ = 0.35; p < 0.001) between crossmatch methods.
Interobserver variation
A total of 562 gel columns were available for review, including pre- and post-transfusion major crossmatches, recipient and donor auto-controls with and without ACG, and 4 incompatible pre-transfusion crossmatches run in duplicate to confirm results. Results of all gel columns were interpreted by 4 individuals. There was near-perfect agreement among the 4 observers in determining compatibility (κ = 0.97) and substantial agreement in overall scoring of incompatibility (κ = 0.77). There was moderate agreement for crossmatches with an incompatibility score of 1+ (κ = 0.41), 2+ (κ = 0.51), or 3+ (κ = 0.57); the agreement was substantial for crossmatches with an incompatibility score of 4+ (κ = 0.73). The measure of agreement was significantly different than random agreement for all agglutination scores (p < 0.001).
Potential factors influencing development or detection of RBC alloimmunization
We considered various factors that could influence development or detection of RBC alloantibodies by a recipient post-transfusion. There was no association identified between major crossmatch incompatibilities and the number of pRBC transfusions, the duration of pRBC storage (whether comparing first pRBC unit or oldest pRBC unit administered), or the time of collection of plasma samples after the first transfusion (Suppl. Table 5). However, there was a significant association between time of collection of post-transfusion plasma samples for all pRBC units administered and absence of crossmatch incompatibilities when sample collection was <2 wk post-transfusion (p = 0.02). Concurrent administration of other blood products or immunosuppressive medications was not associated with development of RBC incompatibilities, although the numbers were too small for meaningful statistical analysis. One of 4 dogs that received concurrent blood products and 2 of 10 dogs that received immunosuppressants developed RBC incompatibilities (Suppl. Table 2). In addition, in contrast to a study that identified a DEA1+ blood type as a risk factor for incompatible tube crossmatches, 23 there was no association between recipient blood type (DEA1+ vs. DEA1–) and development of post-transfusion crossmatch incompatibilities; 6 of 10 dogs with new RBC incompatibilities were DEA1+.
Discussion
Our results indicate that post-transfusion RBC alloimmunization is common in dogs, with major crossmatch incompatibilities detected in 30% of our sample population. Comparing results of post-transfusion major crossmatches performed using a standard and ACG-enhanced gel-column crossmatch, there was fair but significant correlation between methods. Notably, of 10 dogs with post-transfusion crossmatch incompatibilities noted in either crossmatch method, 4 dogs had RBC agglutination noted only in the ACG-enhanced crossmatch, and 3 dogs had RBC agglutination noted only in the standard (non–ACG-enhanced) crossmatch. There was substantial agreement among 4 observers in determining compatibility and scoring incompatibility with the gel-column crossmatch method. Finally, in this small heterogeneous population of dogs receiving RBC transfusions, duration of pRBC storage was not associated with development of RBC alloimmunization.
The reported prevalence of post-transfusion RBC alloimmunization in dogs is 33–66%, with differences in crossmatch technique possibly contributing to the variability.11,14,20,23 Standard (non–ACG-enhanced) tube and gel-column crossmatches produce comparable results for compatibility testing for dogs. 1 In 2 studies using ACG-enhanced crossmatches (ACG-enhanced gel column ± immunochromatographic strip kit), post-transfusion RBC incompatibilities were detected in 11 of 25 (44%) 11 and 6 of 9 (66%) dogs. 20 In a study in which a tube crossmatch was performed sequentially without (standard) and then with addition of ACG, major crossmatch incompatibilities were noted in 57 of 164 previously transfused dogs (34.7%). 23 Interestingly, among 145 incompatible donor-recipient pairings for which there was a notation about the phase of crossmatch during which incompatibility was first detected, 67 (46.2%) were identified only after addition of ACG to the tube crossmatch. 23 Our findings of a post-transfusion alloimmunization rate of 30% (10 of 33 dogs) and detection of RBC agglutination in 4 of the 10 dogs only with addition of ACG to the gel-column crossmatch are similar to the tube crossmatch study. 23 However, 3 of 10 dogs in our study had major crossmatch incompatibilities (>2+) noted only in standard (i.e., non–ACG-enhanced) gel columns. A potential explanation for the detection of RBC agglutination only in the absence of ACG is that the recipient developed an IgM antibody that can agglutinate donor RBCs without the need for a secondary antibody. Other potential explanations include dilution or removal of weakly bound RBC antibodies in the post-incubation washing step, neutralization of the ACG reagent by residual unbound antibodies as a result of inadequate washing, a prozone effect caused by excessive ACG interfering with RBC agglutination, and insufficient time for ACG to bind to RBC-bound IgG before centrifugation of gel columns. Interestingly, false-negative compatible AHG crossmatches have been documented in samples from humans with well-characterized alloantibodies intentionally crossmatched against donor RBCs expressing the cognate antigen, a finding attributed to the antibody titer (i.e., low titer is more likely to yield a false-negative result) and donor cell zygosity (i.e., RBCs heterozygous rather than homozygous for antigen are more likely to yield a false-negative result). 25
The optimal time to detect post-transfusion RBC alloimmunization in dogs is uncertain and likely variable for a given dog and RBC antigen, although the general recommendation is to perform a blood crossmatch for dogs transfused ≥4 d earlier.11,20 In a study in which 2 Dal-negative research Beagles were sensitized to the Dal antigen, antibodies appeared on days 4 and 21 in the respective dogs, lending support to the recommendation of performing crossmatches beginning on day 4 post-transfusion. 10 In clinical case reports, the specific day on which alloantibodies first could be detected is typically unknown, but RBC incompatibilities have been noted on crossmatches performed 8–37 d post-transfusion.2,6,13,24 In the 10 dogs of our study that developed major crossmatch incompatibilities, plasma samples were collected 14–388 d post-transfusion. In dogs with and without RBC agglutination noted on crossmatches, there was no difference in the median number of days from the first RBC transfusion to the collection of plasma samples. It is possible, however, that for recipient dogs for which plasma samples were collected <2–3 wk post-transfusion, alloantibodies were not yet present in sufficient quantity to be detected. If plasma samples were obtained at a later time, evidence of RBC alloimmunization may have been detected, as demonstrated by the research dog with anti-Dal antibodies not detected until day 21 post-transfusion. 10 Furthermore, given that maximum titers of anti-Dal antibodies were documented at 4 and 8 wk post-sensitization in 2 research dogs, 10 it is possible that RBC alloantibodies decreased below the level of detection in the dogs of our study for which plasma samples were collected >8 wk post-transfusion. Therefore, the prevalence of RBC alloimmunization in our study could have been underestimated, depending on the time of collection of the recipient’s plasma relative to the pRBC transfusion.
We did not evaluate the RBC antigens to which dogs developed antibodies post-transfusion in our study. In a study in which extended blood typing of both donors and recipients for DEA1, 3, 4, 7, and Dal was performed, an association could not be made between any of these RBC antigens and post-transfusion crossmatch incompatibilities noted among 10 donor-recipient pairings, 20 suggesting that additional RBC antigens of potential clinical significance are yet to be defined. In our study, of the 10 dogs that had RBC agglutination in major crossmatches post-transfusion, 7 dogs were deemed incompatible with their original blood donors, with 2 of the 7 dogs also deemed incompatible with random blood donors. An unexpected finding was detectable RBC agglutination noted in major crossmatches only with random donors (i.e., dogs appeared compatible with their original blood donors) for 3 recipient dogs. The reason for this finding is uncertain, but a potential explanation could be greater antigen expression on the surface of the RBCs of the random donors compared to the original donors, resulting in obvious RBC agglutination only with the random donors. Although anti-DEA1 antibodies are known not to be the cause of any RBC incompatibilities in our study given the administration of only DEA1 type–compatible pRBCs, it has been documented that transfusion of blood from a weakly DEA1+ donor to a DEA1– recipient induces strong alloimmunization. 13 Whether other weakly positive canine RBC antigens elicit formation of alloantibodies in an antigen-negative recipient is unknown. The clinical significance of the crossmatch incompatibilities noted with or without ACG in our study is uncertain given that the dogs were not in need of a RBC transfusion at the time of the crossmatch. Furthermore, it would have been unethical to transfuse incompatible pRBC units to determine if crossmatch incompatibilities were associated with decreased in vivo RBC survival or acute hemolytic transfusion reactions.
Although there is uniform agreement that a blood crossmatch should be performed for all previously transfused (≥4 d) dogs in need of another transfusion because of the risk of RBC alloimmunization, there is controversy over the need for a crossmatch before a dog’s first RBC transfusion given the lack of documentation of clinically significant naturally occurring RBC alloantibodies in dogs.11,23,26 All 33 recipients in our study were transfusion naïve. Seven of the dogs, using pre-transfusion serum or plasma samples, had major crossmatch incompatibilities of 1–4+, with RBC agglutination only evident in 6 of the dogs after addition of ACG. However, these major crossmatch incompatibilities did not persist with the post-transfusion plasma samples from 6 dogs, suggesting that the RBC agglutination observed with the pre-transfusion samples could have been associated with the dog’s underlying condition rather than naturally occurring RBC alloantibodies. Given that auto-controls were not performed for pre-transfusion crossmatches, we cannot rule out RBC autoagglutination. However, dogs with a diagnosis of immune-mediated hemolytic anemia were excluded from our study, and RBC agglutination was not observed in pre-transfusion crossmatches with all donors tested for 5 of the 7 dogs.
The ACG that we used was a polyspecific reagent containing anti-IgG, anti-IgM, and anti-C3 antibodies. The use of polyspecific AHG reagents for pre-transfusion compatibility testing has been shown to produce clinically insignificant positive crossmatch reactions (i.e., false agglutination) in samples from humans with certain diseases associated with high titers of cold agglutinins (e.g., mycoplasma pneumonia, lymphoma), an effect attributed to the anti-complement component. 30 The 6 dogs with incompatibilities noted only in pre-transfusion crossmatches had diagnoses of hemoabdomen secondary to neoplasia (n = 2), vehicular trauma, surgical blood loss, acute kidney injury, and blood-loss anemia secondary to primary immune thrombocytopenia. It is uncertain if use of a polyspecific ACG reagent is the cause of the transient RBC agglutination observed for these dogs.
The fair correlation between crossmatch methods in our study is in contrast to the strong correlation noted between gel-column crossmatch techniques with and without ACG for detection of naturally occurring alloantibodies in cats. 12 Notable differences between studies include species (feline vs. canine), use of a gel column containing anti-feline globulin within the gel (rather than addition of ACG to the top of saline gel column at the final step), and direct placement of the recipient plasma and donor RBC mixture on the gel column without a post-incubation washing step to remove free antibodies. 12 In addition, most incompatible crossmatches in the feline study were a result of pairing plasma from type B cats containing strong anti-A antibodies with RBCs from type A cats, yielding RBC incompatibility scores of 4+, 12 compared to our range of median post-transfusion agglutination scores of 2.25–4+. A similarity between studies is the detection of some RBC incompatibilities only in the ACG-enhanced gel-column crossmatch, with 14 of 15 such incompatibilities in the feline study noted outside the expected A-B mismatches. 12
Interpretation of crossmatch results is somewhat subjective with respect to grading the degree of incompatibility. In our study, which included a total of 562 gel columns, there was near-perfect agreement (97%) among 4 observers in determining compatibility, similar to results of our previous canine study 29 and an equine study 4 using the gel-column crossmatch method. For the individual scores (1–4+), there was substantial agreement (73%) only for an incompatibility score of 4+, defined as all RBCs being agglutinated and remaining on the top of the gel column. There was moderate agreement for 1–3+ incompatibilities, highlighting the subjectivity of the grading system even among experienced observers. However, variability in the interpretation of degree of incompatibility is less important than agreement in determining compatibility in a clinical situation given that the goal is always to administer compatible RBCs, if available.
In contrast to the results of a murine transfusion model study in which RBCs became progressively more immunogenic as a function of storage time, 17 we found no apparent association between duration of pRBC storage and development of RBC alloantibodies in the dogs of our study. In the murine study, 27 and 25 mice were infused with fresh (i.e., same day as collected) or old (14-d stored) leukoreduced RBCs, respectively, with all of the mice receiving old RBCs having a 10- to 100-fold higher level of RBC alloantibodies compared to those administered fresh RBCs. 17 There is no clear definition of storage duration for “fresh” or “old” human or canine pRBCs, although several studies have used <14 or ≥14 d, respectively, as cutoffs.3,15,19,21,22 The median duration of pRBC storage in our study was 21 d (range: 1–33 d). Of 33 dogs, only 7 dogs received pRBCs stored <14 d given the nature of blood banking, in which the oldest pRBC units are dispensed first to reduce wastage. Whether analyzed based on the number of days of storage of first or oldest pRBC unit or a comparison of first pRBC units stored for <14 or ≥14 d, there was no association between storage duration and development of RBC alloantibodies. In another study of post-transfusion alloimmunization in dogs, there was also no association between blood storage duration and RBC incompatibilities in a standard tube crossmatch, although the median RBC storage duration was 7 d (range: 0–28 d). 18 It is possible that a larger homogeneous study population of dogs administered pRBC units stored for periods of time that are more clearly divided as fresh (e.g., <7 d) or old (e.g., >28 d) could yield different results.
Limitations of our study include a small sample size for evaluation of post-transfusion RBC alloimmunization as a result of discontinuation of patient and donor sample collection at the start of the COVID-19 pandemic. We evaluated only the gel-column crossmatch, hence results comparing standard to ACG-enhanced crossmatches cannot be extrapolated to other crossmatch methods. Also, although post-transfusion crossmatches were performed consistently with EDTA plasma, pre-transfusion crossmatches were performed using either EDTA plasma or serum, whichever was available as a surplus sample in the clinical laboratory. However, a study comparing serum versus EDTA plasma in canine major crossmatch reactions concluded that either was acceptable, although EDTA plasma might be more sensitive in detecting weak agglutination. 5 In addition, recipient auto-controls were performed for all post-transfusion but not pre-transfusion crossmatches given the nature of sample collection (i.e., recipient needed to survive and return to hospital for follow-up evaluation and blood collection to be included in our study). We could not determine the clinical relevance of RBC agglutination noted in the gel-column crossmatches, with or without ACG. Finally, although an association between duration of pRBC storage and RBC alloimmunization was not detected in our convenience sampling of a heterogeneous hospital population of dogs, a disproportionate number of dogs receiving old rather than fresh pRBCs and concurrent administration of immunosuppressive medications to 10 of 33 dogs are potential confounding factors that could have affected results. Another study limitation is that a sample size calculation was not performed because data related to the main study objective, which was to determine if there is a correlation between results of a standard and ACG-enhanced gel-column crossmatch to detect post-transfusion RBC alloimmunization in dogs, were not available at the time our study was designed. To our knowledge, such data are still not published, but our study may help facilitate future sample size calculations for other studies.
Although the aim of our study was not to determine the gold standard method for canine crossmatches, we demonstrated that some RBC incompatibilities are evident in the gel-column crossmatch only with the addition of ACG or only in the absence of ACG with the methodologies used. As with any crossmatch technique, the clinical relevance of any observed RBC agglutination is unclear. However, to maximize detection of potential RBC incompatibilities that could be of clinical significance, it is reasonable to perform both the standard and ACG-enhanced gel-column crossmatches to identify compatible RBC units for previously transfused dogs.
Supplemental Material
Supplemental material, sj-pdf-1-vdi-10.1177_10406387231222895 for Evaluation of post-transfusion RBC alloimmunization in dogs using a gel-column crossmatch with and without anti-canine globulin enhancement by Alison Thomas-Hollands, Rebecka S. Hess, Nicole M. Weinstein, Samantha Fromm, Nicole A. Chappini, Kimberly Marryott and Mary Beth Callan in Journal of Veterinary Diagnostic Investigation
Acknowledgments
We gratefully acknowledge the many clinicians who facilitated collection of blood samples from patients, as well as the clients who consented to have their dogs participate in this study.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: This study was supported by a generous donation from Jack and Theresa Price, grateful PennVet clients, to support resident research.
ORCID iD: Mary Beth Callan  https://orcid.org/0000-0002-4508-9980
https://orcid.org/0000-0002-4508-9980
Supplemental material: Supplemental material for this article is available online.
References
- 1. Blais M-C, et al. Canine Dal blood type: a red cell antigen lacking in some Dalmatians. J Vet Intern Med 2007;21:281–286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Callan MB, et al. Hemolytic transfusion reactions in a dog with an alloantibody to a common antigen. J Vet Intern Med 1995;9:277–279. [DOI] [PubMed] [Google Scholar]
- 3. Callan MB, et al. Transfusion of 28-day-old leucoreduced or non-leucoreduced stored red blood cells induces an inflammatory response in healthy dogs. Vox Sang 2013;105:319–327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Casenave P, et al. Modified stall-side crossmatch for transfusions in horses. J Vet Intern Med 2019;33:1775–1783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Caudill MN, et al. Comparison of serum versus EDTA plasma in canine major crossmatch reactions. Vet Clin Pathol 2021;50:319–326. [DOI] [PubMed] [Google Scholar]
- 6. Conti-Patara A, et al. Dal-induced red blood cell incompatibilities in a Doberman Pinscher with von Willebrand factor deficiency and ehrlichiosis. J Vet Emerg Crit Care (San Antonio) 2021;31:274–278. [DOI] [PubMed] [Google Scholar]
- 7. Davidow EB, et al. Association of Veterinary Hematology and Transfusion Medicine (AVHTM) Transfusion Reaction Small Animal Consensus Statement (TRACS) part 2: prevention and monitoring. J Vet Emerg Crit Care (San Antonio) 2021;31:167–188. [DOI] [PubMed] [Google Scholar]
- 8. Downes KA, Shulman IA. Pretransfusion testing. In: Fung MK, et al., eds. AABB Technical Manual. 18th ed. AABB, 2014:367–390. [Google Scholar]
- 9. Evers D, et al. Red-blood-cell alloimmunisation in relation to antigens’ exposure and their immunogenicity: a cohort study. Lancet Haematol 2016;3:e284–e292. [DOI] [PubMed] [Google Scholar]
- 10. Goulet S, Blais MC. Characterization of anti-Dal alloantibodies following sensitization of two Dal-negative dogs. Vet Pathol 2018;55:108–115. [DOI] [PubMed] [Google Scholar]
- 11. Goy-Thollot I, et al. Pre- and post-transfusion alloimmunization in dogs characterized by 2 antiglobulin-enhanced cross-match tests. J Vet Intern Med 2017;31:1420–1429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Goy-Thollot I, et al. Detection of naturally occurring alloantibody by an in-clinic antiglobulin-enhanced and standard crossmatch gel column test in non-transfused domestic shorthair cats. J Vet Intern Med 2019;33:588–595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Guidetti M, et al. Alloimmunization of a dog erythrocyte antigen 1- dog transfused with weakly dog erythrocyte antigen 1+ blood. J Vet Intern Med 2019;33:2037–2045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Guzman LR, et al. Comparison of a commercial blood cross-matching kit to the standard laboratory method for establishing blood transfusion compatibility in dogs. J Vet Emerg Crit Care (San Antonio) 2016;26:262–268. [DOI] [PubMed] [Google Scholar]
- 15. Hann L, et al. Effect of duration of packed red blood cell storage on morbidity and mortality in dogs after transfusion: 3,095 cases (2001–2010). J Vet Intern Med 2014;28:1830–1837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Heddle NM, et al. A prospective study to determine the safety of omitting the antiglobulin crossmatch from pretransfusion testing. Br J Haematol 1992;81:579–584. [DOI] [PubMed] [Google Scholar]
- 17. Hendrickson JE, et al. Storage of murine red blood cells enhances alloantibody responses to an erythroid-specific model antigen. Transfusion 2010;50:642–648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Herter L, et al. Alloimmunization in dogs after transfusion: a serial cross-match study. J Vet Intern Med 2022;36:1660–1668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Karam O, et al. Association between length of storage of red blood cell units and outcome of critically ill children: a prospective observational study. Crit Care 2010;14:R57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Kessler RJ, et al. Dog erythrocyte antigens 1.1, 1.2, 3, 4, 7, and Dal blood typing and cross-matching by gel column technique. Vet Clin Pathol 2010;39:306–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Koch CG, et al. Duration of red-cell storage and complications after cardiac surgery. N Engl J Med 2008;358:1229–1239. [DOI] [PubMed] [Google Scholar]
- 22. Koch CG, et al. Effect of red blood cell storage duration on major postoperative complications in cardiac surgery: a randomized trial. J Thorac Cardiovasc Surg 2020;160:1505–1514.e3. [DOI] [PubMed] [Google Scholar]
- 23. Marshall H, et al. Accuracy of point-of-care crossmatching methods and crossmatch incompatibility in critically ill dogs. J Vet Intern Med 2021;35:245–251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Melzer KJ, et al. A hemolytic transfusion reaction due to DEA 4 alloantibodies in a dog. J Vet Intern Med 2003;17:931–933. [PubMed] [Google Scholar]
- 25. Nixon CP, et al. False-negative compatible antiglobulin crossmatches in samples with alloantibodies to cognate red blood cell antigens. Transfusion 2018;58:2022–2026. [DOI] [PubMed] [Google Scholar]
- 26. Odunayo A, et al. Incidence of incompatible crossmatch results in dogs admitted to a veterinary teaching hospital with no history of prior red blood cell transfusion. J Am Vet Med Assoc 2017;250:303–308. [DOI] [PubMed] [Google Scholar]
- 27. Pokhrel A, et al. Outcome of type and screen versus crossmatch in cardiovascular surgery patients: a comparative study. Asian J Transfus Sci 2022;16:1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Spada E, et al. Comparison of cross-matching method for detection of DEA 7 blood incompatibility. J Vet Diagn Invest 2018;30:911–916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Thomas-Hollands A, et al. Effect of donor blood storage on gel column crossmatch in dogs. Vet Clin Pathol 2023;52:30–37. [DOI] [PubMed] [Google Scholar]
- 30. Wang S-S, et al. A renewed understanding of anti-human globulin reagents: interference constraints using an optimization method in pretransfusion compatibility tests. J Clin Lab Anal 2021;35:e23695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Wardrop KJ, et al. Update on canine and feline blood donor screening for blood-borne pathogens. J Vet Intern Med 2016;30:15–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Zaremba RM, et al. Comparison of a commercial immunochromatographic strip crossmatch kit and standard laboratory crossmatch methods for blood transfusion compatibility in dogs. J Vet Emerg Crit Care (San Antonio) 2022;32:582–591. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-pdf-1-vdi-10.1177_10406387231222895 for Evaluation of post-transfusion RBC alloimmunization in dogs using a gel-column crossmatch with and without anti-canine globulin enhancement by Alison Thomas-Hollands, Rebecka S. Hess, Nicole M. Weinstein, Samantha Fromm, Nicole A. Chappini, Kimberly Marryott and Mary Beth Callan in Journal of Veterinary Diagnostic Investigation