Abstract
The diagnosis of primary and secondary CNS neoplasms of dogs and cats relies on histologic examination of autopsy or biopsy samples. In addition, many neoplasms must be further characterized by immunohistochemistry (IHC) for a more refined diagnosis in specific cases. Given the many investigations assessing the diagnostic and prognostic IHC profile of CNS neoplasms in the veterinary literature, it may be difficult for the diagnostic pathologist or pathology trainee to narrow the list of reliable diagnostic IHCs when facing a challenging case. Here we compile a comprehensive list of the most diagnostically relevant immunomarkers that should be utilized for the diagnostic support or confirmation of the most common primary and secondary CNS neoplasms of dogs and cats.
Keywords: cats; CNS tumors; dogs; glioma; immunohistochemistry, meningioma; neuropathology
Immunohistochemistry (IHC) is a powerful tool to identify specific cell populations in the CNS, including stem cells (which can differentiate into neurons, astrocytes, and oligodendrocytes) and adult cells, such as neurons, astrocytes, oligodendrocytes, microglia, ependymal and choroid plexus epithelium, meningothelial cells, and endothelial cells, among others.20,81 Understanding the basic immunolabeling pattern in different cell populations in the fetal and adult CNS can be helpful when interpreting IHC features of primary and secondary CNS neoplasms.80,81
A few investigations have assessed the role of IHC in identifying stem cells and neuroglial precursor cells as well as mature neuroglial cells and other non-CNS cell populations in the brain of dogs and cats. In the fetal canine brain, neuronal nuclear protein (NeuN) and β-3 tubulin nuclear immunolabeling can be used to highlight neuron intermediate progenitor cells in the ventricular and subventricular zone, as well as immature neurons in the cortical plate. 81 Cytoplasmic immunolabeling for nestin and glial fibrillary acidic protein (GFAP) highlights radial glia in the ventricular zone and their cytoplasmic processes extending into the subventricular zone. 81 Cytoplasmic and/or membranous platelet-derived growth factor receptor α (PDGFR-α) and nuclear oligodendrocyte transcription factor 2 (OLIG2) immunolabeling is reported in oligodendrocyte precursor cells in the subventricular and intermediate zone. 81 Immature astrocytes in the subventricular and intermediate zone have cytoplasmic GFAP and nuclear OLIG2 immunolabeling. 81 In the fetal feline brain, NeuN and β-3 tubulin can be used to identify neuron intermediate progenitor cells in the subventricular zone and immature neurons in the cortical plate. 80 Nuclear SRY-box transcription factor 2 (SOX2) immunolabeling is reported in radial glial cells in the ventricular and subventricular zone (which also have GFAP and nestin immunolabeling) and cortical plate neurons. 80 Subventricular and intermediate zone cells (which were not specified by the authors) are immunopositive for OLIG2 and PDGFR-α. 80 Membranous Na+/H+ exchanger regulatory factor 1 (NHERF-1) immunolabeling is reported in the apical surface of ventricular zone cells and choroid plexus epithelium. 80
In the adult canine brain, cytoplasmic immunolabeling for β-3 tubulin and doublecortin (DCX), as well as nuclear immunolabeling for NeuN, highlights neuron intermediate precursors in the ventricular and subventricular zone, immature neurons in the cortical plate, and mature neurons throughout.15,81 PDGFR-α immunolabeling is reported in oligodendrocyte precursor cells in the subventricular and intermediate zone, as well as in vascular walls. 81 Nuclear OLIG2 immunolabeling highlights immature astrocytes in the subventricular and intermediate zone, as well as oligodendrocyte precursor cells throughout the CNS. 81 Cytoplasmic nestin immunolabeling highlights neural stem cells in the subventricular zone, as well as vascular walls.15,81 DCX, NeuN, nestin, and β-3 tubulin immunolabeling is observed in neuron intermediate progenitor cells in the subventricular zone of adult cats. 80
Numerous neuroglial immunomarkers are available to identify adult cell populations in the CNS. Mature neurons have cytoplasmic immunolabeling for neurofilament (NF), neuron-specific enolase (NSE), S100 protein, synaptophysin (SYN), and DCX; these cells also have nuclear immunolabeling for NeuN.15,50,81 Immunolabeling for vimentin and GFAP is often utilized to highlight astrocytes throughout the gray and white matter. Oligodendrocytes can be identified by their cytoplasmic immunolabeling for vimentin and 2′,3′-cyclic-nucleotide 3'-phosphodiesterase (CNPase), and robust nuclear immunolabeling for OLIG2.15,81 Although GFAP is traditionally used to label astrocytes, cytoplasmic immunolabeling is also detected in normal ependymal and radial glial cells.26,41 In cats, astrocytic immunolabeling with cytokeratin AE1/ AE3 (CKAE1/3) has been reported. 80 Membranous NHERF-1 immunolabeling is observed in the apical surface of the ependyma, choroid plexus epithelium, and the glia limitans of cats. 80 Nuclear SOX2 immunolabeling is reported in the feline ependyma and oligodendrocytes. 80 Membranous and cytoplasmic inward rectifier potassium channel (Kir7.1) is a highly specific immunomarker for the normal canine choroid plexus epithelium but has not been tested in cats. 8 Less specific choroid plexus epithelial immunomarkers include different types of cytokeratins, E-cadherin, N-cadherin, and β-catenin. 21 Ionized calcium-binding adaptor molecule 1 (Iba1) is a reliable antibody for microglia and intratumoral macrophages. 60 However, other immunomarkers, including CD18, CD11d, CD204, CD68, CD163, TLR4, TLR9, HLA-DR, and MAC387 can be used to stratify infiltrating macrophages within the CNS and CNS tumors.4,40,60,67
Most classification and grading systems of CNS neoplasms of dogs and cats are based on the World Health Organization classification guidelines for human CNS tumors, which utilizes routine histology and IHC but relies heavily on molecular tools to provide a more objective tumor diagnosis and prognosis. 78 In contrast, the diagnosis of canine and feline CNS neoplasms relies heavily on histologic examination of autopsy or biopsy tumor samples. Given that molecular diagnostic techniques are not routinely available in veterinary medicine, IHC is the gold standard test for characterization of neoplastic cells when morphology is not sufficient for a diagnosis.26,69,70 For these reasons, many publications have assessed the diagnostic and prognostic IHC profile of CNS neoplasms in the veterinary literature. However, it can be difficult for the diagnostic pathologist or pathology trainee to sift through all of the information available and narrow the list of potentially useful and reliable diagnostic IHCs when facing a challenging case.
Here we compile a list of immunomarkers that can be utilized for diagnostic support or confirmation of the most common primary and secondary CNS neoplasms of dogs and cats. The immunolabeling profile of a CNS neoplasm (and any other neoplasms) should always be interpreted in light of tumor morphology and not as a standalone diagnostic feature. Given that differences in the IHC protocols among publications likely exist and may have implications for the immunolabeling profile of individual neoplasms, readers are encouraged to search for the details of particular IHC methods in the original investigations, if necessary.
Primary CNS neoplasms
Meningioma
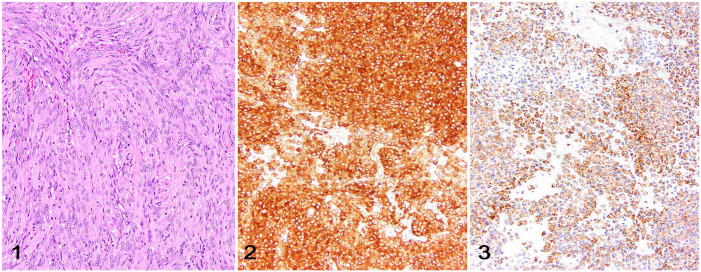
Meningioma is the most commonly diagnosed primary CNS neoplasm of companion animals, accounting for nearly 50% and 60% of all intracranial neoplasms of dogs and cats, respectively (Fig. 1).70,73 Spinal cord meningiomas are reported less often.37,48 Meningiomas arise from the meningothelial cells of the arachnoid layer, which have cellular constituents of both mesenchymal and neural crest origin, 20 resulting in neoplasms with a wide variety of morphologic patterns and IHC profiles. 44 Granular cell tumors that arise in the meninges likely represent a subtype of meningioma, which is supported by their similar IHC profile. 64 The neuroanatomic localization and histologic features of canine and feline meningioma typically suffice for a routine diagnosis in most cases, but IHC may be needed for diagnostic differentiation of meningiomas and neoplasms that may share similar histologic features.2,26,73
Figures 1–3.
Meningioma in the brain of a cat. Figure 1. Meningothelial meningioma characterized by sheets and lobules of neoplastic meningothelial cells with abundant eosinophilic cytoplasm and round-to-oval nuclei. H&E. Figure 2. Neoplastic meningothelial cells with widespread membranous immunolabeling for vimentin. Figure 3. Neoplastic meningothelial cells with widespread membranous immunolabeling for E-cadherin.
Meningiomas typically have robust and uniform immunolabeling for vimentin (Fig. 2),1,26,43,51 but because vimentin immunolabeling occurs in a wide variety of mesenchymal and occasionally epithelial neoplasms, it adds little support to the diagnosis. 20 In addition, an investigation using a larger number of samples reported vimentin immunolabeling in less than half of feline meningiomas, 63 suggesting that this IHC may be more useful for diagnostic support of canine meningiomas than feline meningiomas. 51 For these reasons, vimentin immunolabeling should be interpreted in the context of tumor morphology and in conjunction with other immunomarkers.
Co-immunolabeling for vimentin, CD34, and E-cadherin is proposed to support a diagnosis of canine and feline meningioma; nearly 95% of canine and feline meningiomas had cytoplasmic immunolabeling for CD34 and ~80% had membranous and/or cytoplasmic E-cadherin immunolabeling. 51 However, the immunolabeling profile varied among cases and between canine and feline tumor samples. 51 A subsequent study revealed that over 90% of feline meningiomas had immunolabeling for E-cadherin, confirming this cellular adhesion molecule as a useful diagnostic immunomarker of meningioma in cats, regardless of tumor subtype and grade (Fig. 3). 63 E-cadherin immunolabeling is also reported commonly in meningeal granular cell tumors. 64
Variable or inconsistent meningioma immunolabeling intensity and distribution are reported with CKAE1/3, laminin, claudin-1, glucose transporter 1 (GLUT-1), progesterone receptor (PR), protein gene product 9.5 (PGP9.5), NSE, S100, and GFAP in dogs and cats.51,63 Less than 50% of canine meningiomas reportedly have cytoplasmic immunolabeling for CKAE1/3 or other cytokeratins (such as 8 and 18), but the number of immunolabeled neoplastic cells is often small, indicating that, despite the ubiquitous anecdotal categorization of meningiomas as tumors that have cytokeratin and vimentin immunolabeling, cytokeratin IHC is not reliable for the diagnosis, particularly during examination of small surgical biopsies.1,43,51 In cats, CKAE1/3 immunolabeling is either negative or rarely detected.51,63
Most canine and feline meningiomas have cytoplasmic and membranous immunolabeling for laminin, a basement membrane and extracellular matrix protein that is also expressed by human meningiomas and schwannomas. 51 The diagnostic utility of laminin IHC or its potential specificity to canine and feline meningiomas has not been explored widely, but it remains an excellent diagnostic choice for nerve sheath neoplasms. 31
Nearly 50% of canine meningiomas and 75% of feline meningiomas have cytoplasmic and/or membranous immunolabeling for claudin-1 and cytoplasmic immunolabeling for GLUT-1. 51 No claudin-1 immunolabeling was reported when additional feline meningiomas were assessed, which was attributed to the use of 2 different claudin-1 antibodies. 63 Claudin-1 IHC is utilized to distinguish human meningiomas (immunopositive) from schwannomas (immunonegative). 51 In humans, GLUT-1 is expressed by meningiomas and perineuriomas. The utility of claudin-1 and GLUT-1 IHC for the diagnosis of meningioma in veterinary medicine remains undetermined. 51
Less than half of canine and feline meningiomas have nuclear immunolabeling for PR and PGP9.5. 51 Although lack of PR immunolabeling has been associated with aggressive behavior in human meningiomas, the significance of these findings for tumor behavior in canine and feline meningiomas remains unknown.35,71 PR expression is reported to be inversely correlated with immunolabeling for Ki67; however, interpretation of Ki67 immunolabeling in meningioma samples can be challenging because the antibody will widely label infiltrating inflammatory cells and proliferating endothelial cells, making the proliferative index seem higher than it really is. 35 PGP9.5 immunolabeling is reported in canine nerve sheath tumors and theoretically could be useful in their differentiation from meningiomas. However, in a small study of 28 canine and 8 feline meningiomas, approximately half had immunolabeling for PGP9.5. 51 The intensity and distribution of NSE and S100 immunolabeling is highly variable across canine and feline meningioma cases.1,51 GFAP immunolabeling is rarely reported and has little or no use in making the diagnosis.1,43,51
Cytoplasmic N-cadherin and DCX immunolabeling, along with nuclear immunolabeling for β-catenin, is reported in anaplastic canine meningiomas, suggesting that these IHCs could be useful in the identification of more invasive tumors in canine tumor biopsies. 24 Similarly, cytoplasmic osteopontin (OPN) immunolabeling has been reported in canine meningioma independent of type and grade. 25 OPN immunolabeling was also correlated negatively with tumor infiltration, suggesting that it could be used as a predictor of tumor recurrence. 25 However, these findings need to be validated and no clear associations between canine and feline meningioma type or grade and different IHC profiles have been found to date. 1
Additional immunohistochemical studies of canine meningioma have assessed the immunolabeling profile of cyclooxygenase-2 (COX-2). COX-2 immunolabeling is expressed in the majority of canine meningiomas; however, there is no association between tumor type and COX-2 expression. 62 Similarly, feline meningiomas commonly have COX-2 immunolabeling, which is more prominent in areas of tumor necrosis. 65 Matrix metalloproteinase-2 and -9 have been studied in canine and feline meningiomas in which sporadic cytoplasmic labeling can be seen unrelated to the proliferation index (Ki67) and PR expression. 34 Vascular endothelial-derived growth factor (VEGF) is expressed in most canine meningiomas; however, it is unassociated with Ki67 immunolabeling, and its relevance to patient outcome is unknown.39,49 In human neuropathology, epithelial membrane antigen (EMA) is an important immunomarker of meningioma, but it has not been studied widely in dogs outside of a single investigation reporting EMA immunolabeling in a number of different canine meningioma types. 33 However, we have had no success replicating these published results (J.W. Koehler, A.D. Miller, pers. comm., 2023 Aug 1). Somatostatin receptor 2 (SSTR2) is expressed in canine meningiomas and potentially has lower expression in higher-grade meningiomas. 17 This is a potential targetable receptor and may yield further therapeutic strategies. The cytoplasmic granules in canine meningeal granular cell tumors are typically labeled for lysosomal immunomarkers, such as ubiquitin, p62, and LC3. 64
Numerous immunomarkers have been applied to canine and feline meningiomas. However, no single IHC can prove the diagnosis of meningioma, and diagnosticians should continue to rely on the clinicopathologic and histologic features rather than over- or misinterpreting IHC results in a specific case.
Glioma
Glioma is the second most commonly diagnosed primary CNS neoplasm of dogs and the third most commonly diagnosed in cats.28,61,69,70 Glioma accounts for nearly 35% and 8% of all canine and feline intracranial neoplasms, respectively.37,70,73 Spinal cord gliomas are described uncommonly.37,57 Oligodendroglioma is the most commonly reported glioma of dogs (~70% of all canine gliomas), whereas astrocytoma is less common.28,69,70 Ependymomas are more commonly encountered in cats.61,77 Canine ependymoma is exceedingly rare and shares key morphologic features with many other neoplasms, such as oligodendrogliomas, choroid plexus tumors, pituitary corticotroph adenomas, papillary meningiomas, and suprasellar germ cell tumors. 41 For these reasons, the diagnosis of canine ependymoma is particularly dependent on IHC support. 41 The cell of origin of oligodendroglioma and astrocytoma remains undetermined, but neoplasms likely arise from neural stem cells or oligodendrocyte precursor cells that have the potential to differentiate into adult cells with glial or neuronal phenotype. 23 This often is supported by the intraventricular and/or periventricular growth pattern of canine glioma. Ependymoma is thought to arise from subventricular radial glial cells.41,70,77
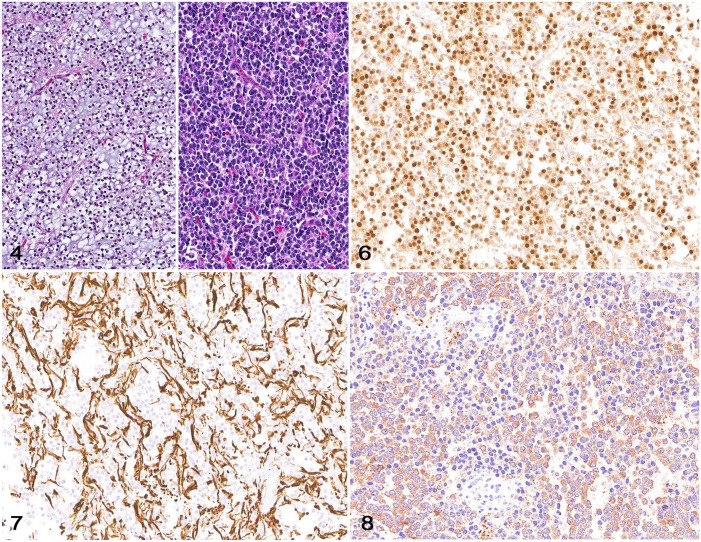
OLIG2 and GFAP are the 2 IHCs that are used most widely to confirm the diagnosis of oligodendroglioma and astrocytoma in dogs and cats.23,26,28,61 Although OLIG2 IHC is not specific for oligodendroglial or astrocytic neoplasms, nuclear OLIG2 immunolabeling is a highly reliable initial step to confirm the glial origin of a CNS neoplasm.11,27,28,61 A glioma with oligodendroglial morphology typically has widespread, robust, nuclear OLIG2 immunolabeling, supportive of an oligodendroglioma (Figs. 4–6).11,27,28 Cytoplasmic GFAP immunolabeling can be observed occasionally in neoplastic cells but is more evident in entrapped astrocytes within the neoplasm (Fig. 7). A suspect oligodendroglioma with no OLIG2 immunolabeling should be immunostained with round-cell immunomarkers to rule out the possibility of lymphoma or, less likely, other round-cell neoplasms. 61 Further support for an oligodendroglioma diagnosis in dogs can be achieved using CNPase IHC (Fig. 8). 28 CNPase cytoplasmic immunolabeling has been reported as particularly sensitive to formalin fixation delay, uneven formalin fixation, or prolonged formalin fixation, which could decrease its diagnostic utility on autopsy samples. 28 However, this assertion could not be confirmed in a study investigating the effects of experimentally induced postmortem autolysis in the normal canine brain by controlled delayed tissue fixation and prolonged formalin fixation times on a panel of immunomarkers, including CNPase. 29 The reason for these discrepancies remains unknown, but it is possible that CNPase IHC immunolabeling may have different labeling affinity for normal brain tissue when compared to neoplastic cells.
Figures 4–8.
Oligodendroglioma in the brain of a dog. Figure 4. Low-grade oligodendroglioma characterized by sheets of neoplastic cells and a mucinous and fine capillary stroma. H&E. Figure 5. High-grade oligodendroglioma characterized by dense sheets of neoplastic cells and a fine capillary stroma. H&E. Figure 6. Neoplastic cells with widespread nuclear immunolabeling for OLIG2. Figure 7. Reactive astrocytes among neoplastic cells have cytoplasmic immunolabeling for GFAP. Figure 8. Neoplastic cells with widespread cytoplasmic immunolabeling for CNPase.
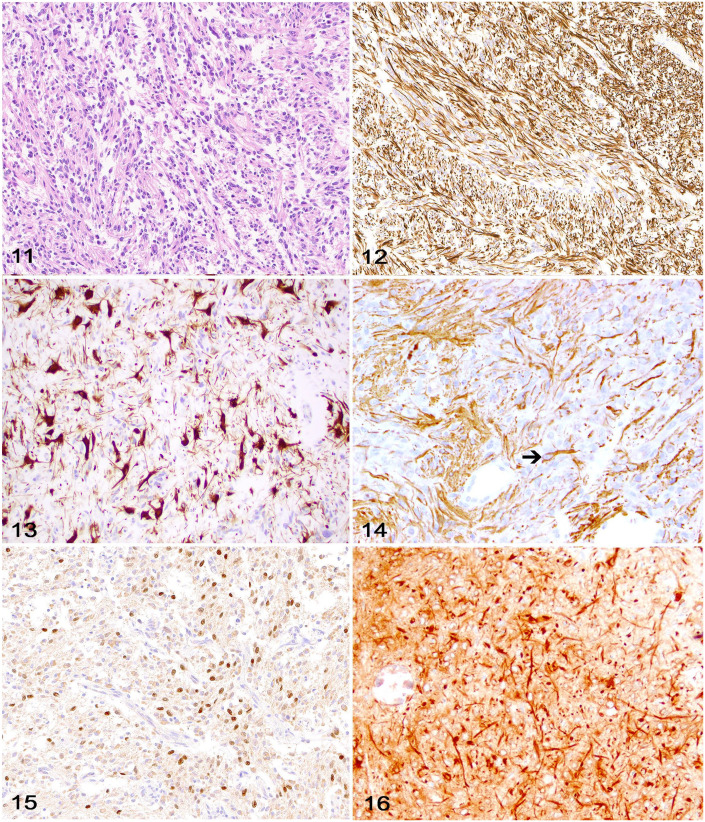
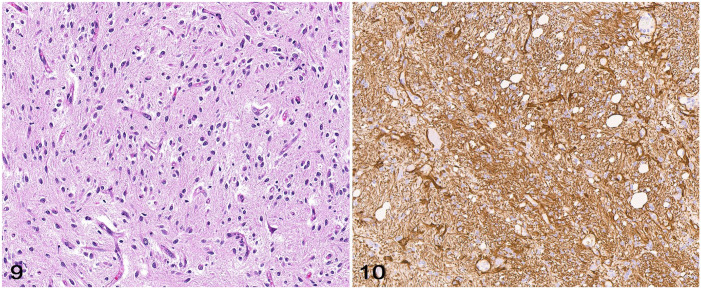
GFAP and nestin are intermediate filament proteins expressed by mature and immature astrocytes, respectively. 23 Although diagnostically useful, cytoplasmic GFAP and nestin immunolabeling has been reported in other neuroepithelial neoplasms, including oligodendrogliomas, and should not be relied upon for diagnostic confirmation of an astrocytoma (or any glioma subtype). 23 GFAP immunolabeling is more common in canine and feline gliomas with astrocytic or undefined morphology (gliomas with nearly equal proportions of neoplastic cells with astrocytic and oligodendroglial morphology).28,61 Although GFAP does not confirm a diagnosis of astrocytoma, cytoplasmic immunolabeling offers diagnostic support when in combination with astrocytic tumor morphology and variable or negative OLIG2 immunolabeling (Figs. 9–15).11,28,61 When interpreting GFAP immunolabeling, it is important for the diagnostic pathologist to be aware of several facts: 1) there may be more consistent immunolabeling of cell processes as opposed to the cell body; 2) immunolabeling can be heterogeneous within the neoplasm, and 3) immunolabeling does not have to be present in 100% of the cells to consider a tumor GFAP-positive. When attempting to distinguish entrapped reactive astrocytes from neoplastic cells (a common dilemma with diffusely infiltrative tumors), a close examination of the morphology of the immunolabeled processes can be helpful because entrapped astrocytes are more likely to have long, slender processes whereas neoplastic astrocytes, if not possessing a pilocytic morphology, are more likely to have irregular, short, stocky processes. As with CNPase, altered GFAP immunolabeling typically occurs as a result of improper formalin fixation, although in comparative studies it was noted that canine tumors with astrocytic morphology seem to lose GFAP immunolabeling more commonly than do human astrocytomas. 28 This appears especially true in high-grade astrocytomas. Microtubule-associated protein 2 (MAP2) immunolabeling is consistently detected in canine glioma (Fig. 16) and can be useful to distinguish astrocytoma (immunolabeling of ramified cytoplasmic processes) from oligodendroglioma (perinuclear and cytoplasmic immunolabeling). 11 Caution should be exercised when interpreting immunolabeling in tumors with highly ambiguous morphology or potential metastases from extracranial sites, given that MAP2 is reported in human thyroid carcinoma, prostate carcinoma, neuroendocrine neoplasms, small-cell lung carcinoma, and melanoma, as well as neuroblastomas, 72 pineal tumors, and glioneuronal tumors. 3
Figures 11–16.
Astrocytoma in the brain of a dog. Figure 11. High-grade astrocytoma characterized by bundles of neoplastic cells and a neuroparenchyma-like stroma. H&E. Figure 12. Robust cytoplasmic immunolabeling for GFAP throughout the astrocytic cell bodies and processes. Figure 13. Robust cytoplasmic immunolabeling for GFAP is more prominent in the astrocytic cell bodies. Figure 14. Robust cytoplasmic immunolabeling for GFAP is more prominent in the astrocytic cell processes, with only rare cell bodies labeled (arrow). Figure 15. Neoplastic cells with patchy nuclear immunolabeling for OLIG2. Figure 16. Neoplastic cells (particularly cell processes) with widespread cytoplasmic immunolabeling for MAP2.
Figures 9–10.
Astrocytoma in the brain of a dog. Figure 9. Low-grade astrocytoma characterized by scattered elongate neoplastic cells and a fibrillary, neuroparenchyma-like stroma. H&E. Figure 10. Neoplastic cells with widespread cytoplasmic immunolabeling for GFAP.
Neural stem cells (which give rise to neuron precursor cells, oligodendrocyte precursor cells, and astrocyte precursor cells) are likely involved in the oncogenesis of glioma. 27 Cytoplasmic immunolabeling for nestin and CD133 and nuclear and cytoplasmic immunolabeling for activating transcription factor 5 (ATF5) supports the presence of neural stem cells in canine glioma, with no associations between immunolabeling and tumor type and grade.15,79
DCX, a microtubule-associated protein that regulates neuroblast migration during embryonal life, can be utilized to detect neuronal progenitor cells (neuronal differentiation) in canine and feline gliomas.15,23,55 In addition, strong cytoplasmic DCX immunolabeling at the tumor margins is associated with tumor infiltration in human glioma, but no evidence has yet supported that association in canine and feline gliomas.14,55
Nuclear immunolabeling for NeuN and cytoplasmic immunolabeling for β-3 tubulin (both immunomarkers for mature neurons) occurs most often in high-grade canine oligodendrogliomas compared to low-grade oligodendrogliomas,27,81 but the clinical significance of these findings remains elusive. Feline gliomas lack NeuN immunolabeling. 14
Nestin, CD133, ATF5, DCX, NeuN, and β-3 tubulin, together with NSE and SYN IHC, label neural stem cells and support neuronal differentiation within gliomas.15,23,27 However, the diagnostic and prognostic implications of neuronal differentiation within gliomas (if any) remain undetermined.23,58
Nuclear immunolabeling for stem cell transcription factors SOX2 and SOX10 is consistently reported in canine oligodendroglioma and may be useful in the identification of intratumoral oligodendrocyte precursor cells and in support of its oligodendroglial origin.27,42 Additional support for the presence of intratumoral oligodendrocyte precursor cells in canine glioma includes membranous and cytoplasmic immunolabeling for PDGF-α and neural/glial antigen 2 (NG2).27,81
Specific cytokeratin antibodies (AE3, AE5, and KS-1A3) have been demonstrated to stain normal human brain tissue and a variable proportion of astrocytomas. 30 Further, the staining pattern of these cytokeratins was identical to GFAP staining in both normal brain and astrocytomas. 30 A similar pancytokeratin and GFAP immunolabeling pattern has been sporadically observed in canine and feline brain tissue (particularly glia limitans) and astrocytomas (J.W. Koehler, A.D. Miller, pers. comm., 2023 Aug 1), which could complicate interpretation of pancytokeratin and GFAP IHC immunolabeling of normal brain and, potentially, of tumor samples in companion animals.
Diagnostic confirmation of oligodendrogliomas and astrocytomas in dogs and cats should rely primarily on tumor morphology associated with OLIG2 immunolabeling (robust nuclear immunolabeling in oligodendrogliomas and variable nuclear immunolabeling in astrocytomas) and GFAP immunolabeling (robust cytoplasmic immunolabeling in astrocytomas and rare or no cytoplasmic immunolabeling in oligodendrogliomas).28,61 CNPase could lend further support to a diagnosis of oligodendroglioma. 28
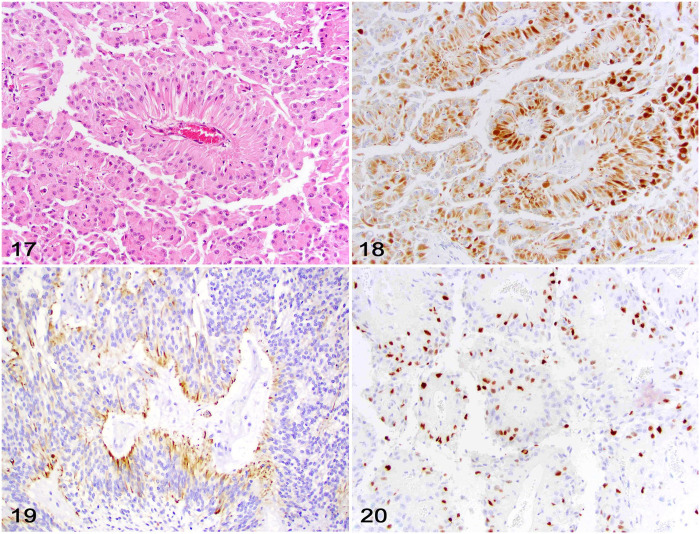
The routine diagnosis of ependymoma has historically relied on the presence of ependymal rosettes and pseudorosettes, as well as ependymal canals, characteristic of many ependymal neoplasms (Fig. 17).41,77 However, these morphologic features are not always present in all tumors and are not unique to ependymomas (rosettes and pseudorosettes can be observed in many other CNS neoplasms), and diagnostic confirmation may be challenging without the aid of IHC. 41 Diagnostic IHC features of ependymoma include robust cytoplasmic immunolabeling for GFAP (Fig. 18) and CKAE1/3 (Fig. 19) that is predominantly concentrated in neoplastic cells forming rosettes and pseudorosettes, with scattered immunolabeling in other portions of the neoplasm. Variable cytoplasmic S100 immunolabeling can be also detected. In cats, cytoplasmic GFAP immunolabeling is also variable, and CKAE1/3 immunolabeling is only occasionally detected. 77 Importantly, whereas intranuclear OLIG2 immunolabeling is rarely observed in canine ependymoma, it is widespread in most feline ependymomas (Fig. 20).12,41
Figures 17–20.
Ependymoma in the brain of a cat. Figure 17. Classic ependymoma characterized by neoplastic cells surrounding a blood vessel (pseudorosette) supported by a fine fibrovascular stroma. H&E. Figure 18. Patchy cytoplasmic immunolabeling for GFAP predominantly within neoplastic cells forming pseudorosettes. Figure 19. Neoplastic cells with patchy cytoplasmic immunolabeling for CKAE1/3, predominantly in cells forming pseudorosettes. Figure 20. Neoplastic cells with widespread nuclear immunolabeling for OLIG2.
Diagnostic support for canine ependymoma should rely on tumor location and morphology associated with GFAP and CKAE1/3 immunolabeling, and lack of OLIG2 immunolabeling. 41 Feline ependymoma should be suspected based on tumor location and morphology associated with variable GFAP and CKAE1/3 immunolabeling and widespread OLIG2 immunolabeling. 12
Choroid plexus neoplasms
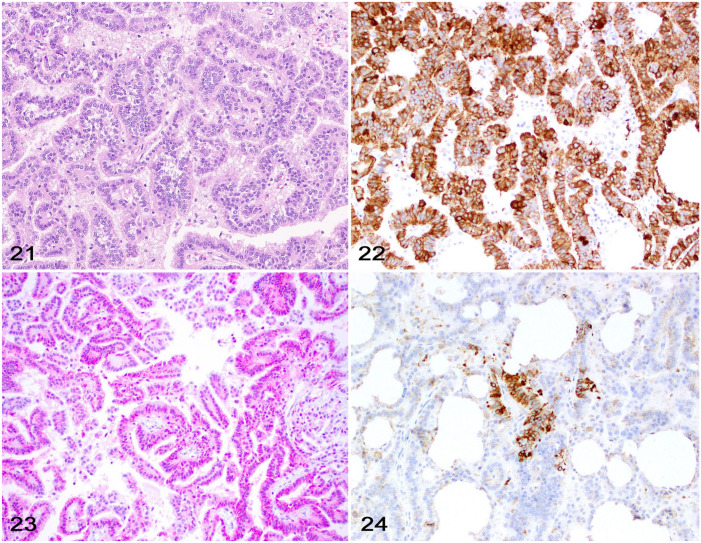
Choroid plexus tumors (CPTs; Fig. 21) are uncommon in dogs (~6% of all intracranial neoplasms) and not convincingly reported in cats. 70 Although a routine diagnosis may be straightforward in most cases, choroid plexus carcinomas can disseminate through the ventricular system to other CNS locations and may need to be differentiated from a metastatic carcinoma, especially when biopsy samples are evaluated. 8 Further, choroid plexus carcinomas may need to be differentiated from other neoplasms such as papillary ependymoma. 41
Figures 21–24.
Choroid plexus tumor in the brain of a dog. Figure 21. Choroid plexus papilloma characterized by neoplastic cells forming papillary projections supported by a stalk of fibrovascular stroma. H&E. Figure 22. Neoplastic cells with widespread cytoplasmic immunolabeling for CKAE1/3. Figure 23. Neoplastic cells with widespread cytoplasmic immunolabeling for E-cadherin. Figure 24. A cluster of neoplastic cells with cytoplasmic immunolabeling for GFAP.
CPTs have been immunolabeled with CKAE1/3, CAM5.2, or Lu-5, E-cadherin, N-cadherin, β-catenin, and GFAP (Figs. 22, 23).7,21,26,45,54 However, these antibodies are not specific for the choroid plexus epithelium and have unreliable results with highly variable immunolabeling among tumors.
Decreased membranous immunolabeling or relocation of E-cadherin to the cytoplasm or the nucleus of neoplastic cells is associated with higher invasiveness and metastatic potential in canine mast cell tumors, canine melanoma, and feline mammary carcinomas. 50 Most CPTs are reported to have variable membranous and aberrant cytoplasmic and nuclear E-cadherin and β-catenin immunolabeling, regardless of tumor grade.45,54 Other studies have reported cytoplasmic N-cadherin immunolabeling that was more frequent in low-grade CPTs. 54 Cytoplasmic CAM5.2 and GFAP immunolabeling is variable and reported in nearly 50% of CPTs (Fig. 24). 45 A panel of antibodies including E-cadherin and GFAP might be useful to support the diagnosis of CPT and differentiate it from other tumors, such as ependymomas and metastatic epithelial tumors.
Kir7.1 is a specific and reliable antibody that can be used for both diagnostic confirmation and differentiation of CPTs (positive immunolabeling) and other carcinomas (negative immunolabeling) in dogs.8,10 Robust Kir7.1 immunolabeling occurs at the apical cell membrane in benign CPTs and the apical cell membrane and cytoplasm in choroid plexus carcinomas.8,10 Although this IHC marker is specific to the choroid plexus epithelium, the antibody originally described to work in the dog is no longer available (A.D. Miller, pers. comm., 2023 Aug 1), and further research needs to be done to identify a new Kir7.1 IHC that works in our domesticated species.
The diagnosis of canine and feline CPTs should rely on tumor location and histology in most cases. When necessary, diagnostic support could be achieved based on CK, E-cadherin, and GFAP immunolabeling. Diagnostic confirmation could be achieved with Kir7.1 or another specific antibody against the choroid plexus epithelium.
Neuronal and neuronal-glial neoplasms
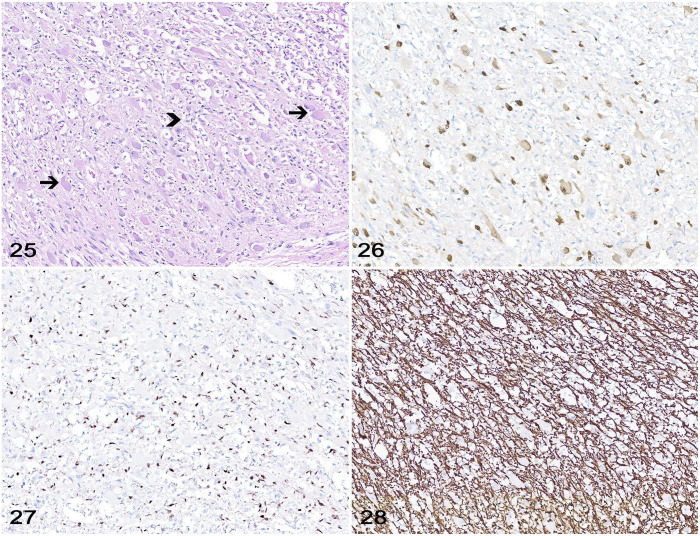
Neuronal or mixed neuronal-glial neoplasms are rarely described in veterinary medicine and consist mainly of gangliocytoma and ganglioglioma (Fig. 25).20,46,74 Not enough cases have been reported to account for a detailed IHC assessment of each tumor category, but it is widely accepted that diagnostic confirmation can be achieved based on tumor morphology associated with IHC for neuronal immunomarkers such as NF, NeuN, and SYN, and/or glial immunomarkers such as OLIG2 and GFAP (Figs. 26–28).20,76
Figures 25–28.
Ganglioglioma in the spinal cord of a dog. Figure 25. Neoplastic neurons (arrows) are interspersed with neoplastic glial cells (arrowhead) throughout. Figure 26. Neoplastic neurons with moderate cytoplasmic immunolabeling for NeuN. Figure 27. Neoplastic glial cells with robust nuclear immunolabeling for OLIG2. Figure 28. Neoplastic glial cells with robust cytoplasmic immunolabeling for GFAP.
Embryonal and germ-cell neoplasms
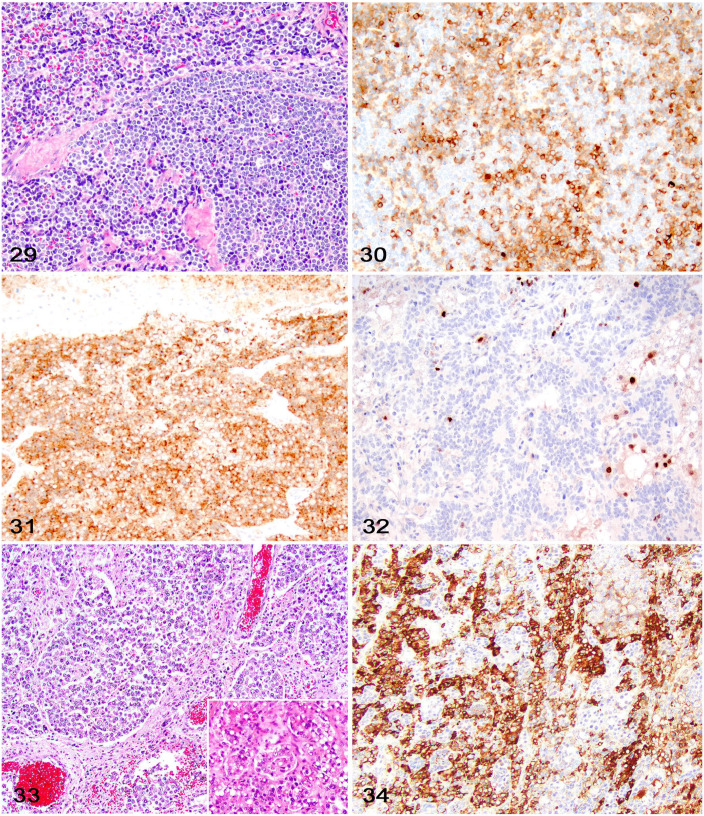
Embryonal CNS neoplasms (including olfactory neuroblastoma, neuroblastoma, medulloblastoma, and embryonal neoplasms, not otherwise specified [NOS]) likely arise from germinal neuroepithelial cells (Fig. 29). 20 Primitive neuroectodermal tumors (PNETs), a controversial term that is no longer commonly used in human neuropathology, is still often used in veterinary medicine and falls under this category. Because these neoplasms are rare in veterinary medicine, their morphologic and IHC profile has not been well characterized.9,13,20,23
Figures 29–34.
Embryonal and germ cell neoplasms in the brains of dogs. Figure 29. Embryonal neoplasm characterized by sheets of neoplastic round cells. Figure 30. Neoplastic embryonal cells with cytoplasmic immunolabeling for MAP2. Figure 31. Neoplastic embryonal cells with cytoplasmic immunolabeling for SYN. Figure 32. Scattered neoplastic embryonal cells with nuclear immunolabeling for OLIG2. Figure 33. Suprasellar germ cell tumor consisting of sheets of round germ cells (left) and hepatocyte-like cells with cytoplasmic lipid vacuoles and occasional acinar structures (inset). Figure 34. Neoplastic cells in a suprasellar germ cell tumor have robust cytoplasmic immunolabeling for α-fetoprotein.
Olfactory neuroblastomas arise from the olfactory neuroepithelium and typically occur in the nasal cavity and paranasal sinuses.6,32 In a study examining olfactory neuroblastomas in 13 dogs and 9 cats, cytoplasmic immunolabeling for MAP2 and NSE, as well as nuclear immunolabeling for NeuN was reported in nearly all cases. 6 Canine and feline olfactory neuroblastomas also have strong immunolabeling for other markers of neuronal differentiation, including cytoplasmic immunolabeling for SYN, Tuj-1 (a specific antibody clone against neuron-specific class β-3 tubulin), PGP9.5, and NF. 9 Variable immunolabeling can be found for chromogranin A, GFAP, and CKAE1/3.6,9,47
Cytoplasmic immunolabeling for β-3 tubulin and DCX is reported in a small set of canine neuroblastomas. 23 Strong cytoplasmic immunolabeling for MAP2 and SYN within neuronal-like neoplastic cells has been reported in embryonal CNS neoplasms of dogs regardless of the diagnosis, suggesting that these immunomarkers may be useful in the diagnostic support of such tumors. Neoplasms had rare immunolabeling for NSE, NeuN, and OLIG2, and no immunolabeling for NF.
Owing to the various stages of differentiation in these neoplasms, a panel that includes SYN, NeuN, MAP2, PGP9.5, and NF can greatly aid in defining embryonal neoplasms (Figs. 30, 31). The sparse immunolabeling for OLIG2 in a limited number of cases suggests that this IHC can be useful for the differentiation of embryonal CNS neoplasms (other than medulloblastomas) and gliomas in dogs (Fig. 32). 13
Germ cell tumors (germinomas) of the CNS are uncommon in dogs, with suprasellar germ cell tumors encountered most frequently. These tumors arise in the skull base and acquire a diverse histologic pattern including epithelial, germ cell, and hepatocyte-like differentiation, with the latter often having intracytoplasmic lipid vacuoles (Fig. 33). 75 Although the IHC features of these tumors are not published widely, neoplastic cells consistently have cytoplasmic immunolabeling for α-fetoprotein (Fig. 34). 75
Ectopic nephroblastoma of the thoracolumbar spinal cord
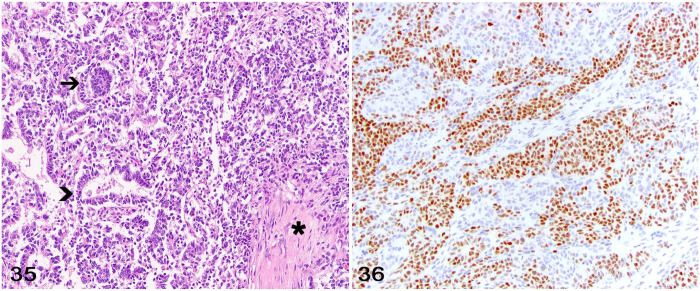
These uncommon neoplasms occur in the thoracolumbar spinal cord segments of young dogs. 5 Tumors arise from remnants of ectopic renal metanephric blastema trapped between the dura mater and the spinal cord during embryonic development. 5 Neoplasms are typically intradural and extraparenchymal, and most commonly affect the T9–L3 spinal segments. Multiple spinal cord nephroblastomas or metastatic tumors occur rarely.5,19 The location of the neoplasm and the presence of triphasic histologic elements consisting of epithelial (tubules and primitive glomeruli), blastemal, and mesenchymal components is highly supportive of the diagnosis (Fig. 35). However, IHC may be needed. Cytoplasmic immunolabeling for CK and vimentin is often reported within the epithelial and mesenchymal neoplastic cell components, respectively. 20 Nuclear immunolabeling for Wilms tumor protein 1 (WT1) is often regarded as confirmatory for the diagnosis (Fig. 36). In a study with 11 dogs, WT1 was reported in neoplastic epithelial cells forming primitive glomeruli and tubules, as well as neoplastic blastemal cells in 9 cases. 5 However, a more in-depth IHC assessment of a larger number of cases would likely be useful to determine the overall frequency of WT1 immunolabeling among tumors.
Figures 35, 36.
Ectopic nephroblastoma in the spinal cord of a dog. Figure 35. The neoplasm consists of an epithelial component forming tubules (arrowhead) and primitive glomeruli (arrow), and a mesenchymal component (asterisk). Figure 36. Neoplastic cells with nuclear immunolabeling for WT1.
Round-cell neoplasms
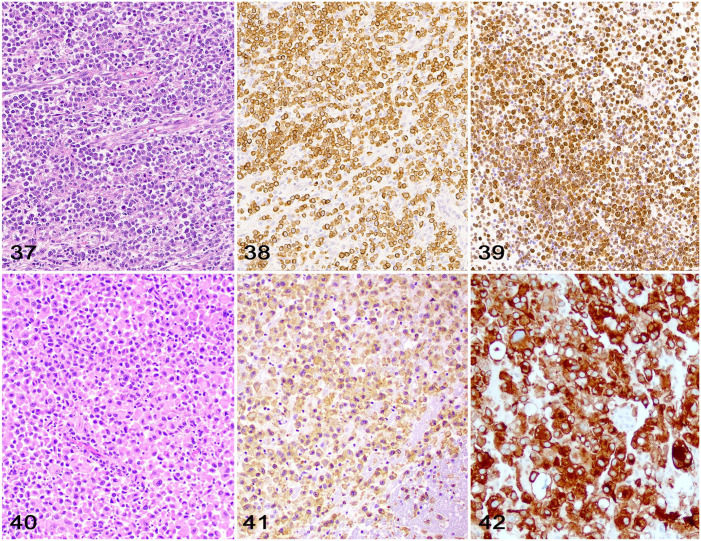
Round-cell neoplasms are rare in the CNS of dogs and cats, and can be either primary to the CNS or part of multicentric disease. 20 The most commonly reported primary round-cell neoplasms in the brain and spinal cord include lymphoma (Figs. 37–39) and histiocytic sarcoma (Figs. 40–42).56,59 Regardless of the tumor type, diagnostic support for these neoplasms depends on the use of a panel of round-cell immunomarkers, including CD18 (leukocyte immunomarker), CD3 (T-lymphocyte immunomarker), CD79, CD20, CD21, and Pax5 (B-lymphocyte immunomarkers), 59 Iba1 and CD204 (histiocytic cell immunomarkers),22,36,56 and MUM1 (plasma cell immunomarker),56,59 among others.
Figures 37–42.
Round cell neoplasms in the brains of cats. Figure 37. Encephalic lymphoma characterized by sheets of neoplastic lymphocytes supported by a fine fibrovascular stroma. H&E. Figure 38. Encephalic T-cell lymphoma with widespread membranous immunolabeling for CD3. Figure 39. Encephalic B-cell lymphoma with widespread membranous immunolabeling for CD79. Figure 40. Encephalic histiocytic sarcoma characterized by sheets of neoplastic histiocytes supported by a fine fibrovascular stroma. H&E. Figure 41. Encephalic histiocytic sarcoma with widespread cytoplasmic immunolabeling for Iba1. Figure 42. Encephalic histiocytic sarcoma with widespread cytoplasmic immunolabeling for CD204.
Given the wide range of possibilities, diagnostic confirmation of round-cell neoplasms should be based on neoplastic cell morphology and specific IHCs to confirm a particular suspicion (e.g., CD3 and CD79 for lymphoma) or a wide IHC panel targeting all possibilities.
Secondary CNS neoplasms
Secondary neoplasms encompass metastatic tumors that spread hematogenously to the CNS, tumors that are part of disseminated neoplasia, and tumors that compress and/or infiltrate the CNS by direct extension from adjacent tissues.18,20 Although a few studies suggest that secondary CNS neoplasms may be as frequent as primary CNS neoplasms,69,70 the true incidence of secondary CNS tumors is not well enumerated in the literature.
Hemangiosarcoma is the most common secondary CNS neoplasm of dogs and can be identified by characteristic histologic features of abortive vascular structures that are often accompanied or predominated by hemorrhage. Although IHC is often not necessary for hemangiosarcoma, diagnosis of poorly differentiated or unusual morphologic phenotypes can be aided by IHC for CD31 or factor 8–related antigen. 38 In one study, 38% of dogs with melanoma had CNS metastases and, because of the diverse histomorphology associated with canine melanoma, IHC can be critical in diagnosing these tumors. 53 A number of melanocytic IHCs have utility in the dog, including melan-A, PNL2, and TRP1/2. 68 Diagnostically, these IHC markers do not work as well in the cat and should not be relied upon for confirming the diagnosis.
Epithelial neoplasms (such as sinonasal carcinomas) can infiltrate the CNS, whereas others (such as pulmonary and mammary carcinomas) can metastasize to the CNS. No specific IHCs to identify nasal-origin carcinomas from other carcinomas have been published, and diagnosis should rely on clinical findings and imaging. Pulmonary carcinomas can be identified by finding cilia along the apical surface of the neoplastic cells; however, this is a challenging feature to recognize and may not be present. Pulmonary carcinomas can be identified via IHC for thyroid transcription factor 1 (TTF-1), which reveals strong intranuclear immunolabeling in canine and feline pulmonary carcinoma.16,52 A number of different immunohistochemical studies have been employed in feline and canine mammary carcinoma and most are not specific to carcinomas of this location. Although a comprehensive review of mammary carcinoma IHC is not the purpose of our review, IHCs such as PR and estrogen receptor could be useful in a diagnostic setting to establish the origin of the primary tumor if the morphology supports the diagnosis of carcinoma. 66
Secondary CNS round-cell neoplasms typically occur as part of widespread neoplasia and include mainly lymphoma and histiocytic sarcoma.18,20,36,70,73 Diagnostic confirmation for these neoplasms is similar to that described above for primary CNS round-cell neoplasms.
Acknowledgments
We thank Dr. Marc Kent, Department of Small Animal Medicine & Surgery, College of Veterinary Medicine, University of Georgia (Athens, GA, USA) for providing us with a feline ependymoma (Fig. 17). We also thank Dr. Emily K. Swan (Captain, U.S. Army Veterinary Corps), Veterinary Pathology Services, Joint Pathology Center (Silver Spring, MD, USA) for providing us with Figs. 25–28.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The authors declare that they received no financial support for their research and/or authorship of this article.
ORCID iDs: Daniel R. Rissi  https://orcid.org/0000-0003-4574-2836
https://orcid.org/0000-0003-4574-2836
Andrew D. Miller  https://orcid.org/0000-0001-6350-5581
https://orcid.org/0000-0001-6350-5581
Jennifer W. Koehler  https://orcid.org/0000-0002-2287-2702
https://orcid.org/0000-0002-2287-2702
Contributor Information
Daniel R. Rissi, Athens Veterinary Diagnostic Laboratory, Department of Pathology, College of Veterinary Medicine, University of Georgia, Athens, GA, USA.
Andrew D. Miller, Department of Population Medicine and Diagnostic Sciences, Section of Anatomic Pathology, College of Veterinary Medicine, Cornell University, Ithaca, NY, USA.
Elena A. Demeter, Department of Population Medicine and Diagnostic Sciences, Section of Anatomic Pathology, College of Veterinary Medicine, Cornell University, Ithaca, NY, USA
Molly E. Church, Department of Pathobiology, School of Veterinary Medicine, University of Pennsylvania, Philadelphia, PA, USA
Jennifer W. Koehler, Department of Pathobiology, College of Veterinary Medicine, Auburn University, Auburn, AL, USA
References
- 1. Barnhart KF, et al. Immunohistochemical staining patterns of canine meningiomas and correlation with published immunophenotypes. Vet Pathol 2002;39:311–321. [DOI] [PubMed] [Google Scholar]
- 2. Belluco S, et al. Standardisation of canine meningioma grading: inter-observer agreement and recommendations for reproducible histopathologic criteria. Vet Comp Oncol 2022;20:509–520. [DOI] [PubMed] [Google Scholar]
- 3. Blümcke I, et al. Microtubule-associated protein-2 immunoreactivity: a useful tool in the differential diagnosis of low-grade neuroepithelial tumors. Acta Neuropathol 2004;108:89–96. [DOI] [PubMed] [Google Scholar]
- 4. Boozer LB, et al. Characterization of immune cell infiltration into canine intracranial meningiomas. Vet Pathol 2012;49:784–795. [DOI] [PubMed] [Google Scholar]
- 5. Brewer DM, et al. Spinal cord nephroblastoma in dogs: 11 cases (1985–2007). J Am Vet Med Assoc 2011;238:618–624. [DOI] [PubMed] [Google Scholar]
- 6. Brosinski K, et al. Olfactory neuroblastoma in dogs and cats—a histological and immunohistochemical analysis. J Comp Pathol 2012;146:152–159. [DOI] [PubMed] [Google Scholar]
- 7. Cantile C, et al. Pathological and immunohistochemical studies of choroid plexus carcinoma of the dog. J Comp Pathol 2002;126:183–193. [DOI] [PubMed] [Google Scholar]
- 8. Choi EJ, et al. Kir7.1 immunoreactivity in canine choroid plexus tumors. J Vet Diagn Invest 2016;28:464–468. [DOI] [PubMed] [Google Scholar]
- 9. Church ME, et al. Clinical outcomes, ultrastructure and immunohistochemical features of canine high-grade olfactory neuroblastoma. Vet Comp Oncol 2019;17:578–584. [DOI] [PubMed] [Google Scholar]
- 10. Dalton MF, et al. Clinicopathologic features, diagnosis, and characterization of the immune cell population in canine choroid plexus tumors. Front Vet Sci 2019;6:224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Demeter EA, et al. Microtubule-associated protein 2 expression in canine glioma. Front Vet Sci 2019;6:395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Demeter EA, et al. OLIG2 immunolabeling in feline ependymoma. J Vet Diagn Invest 2022;34:898–901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Demeter EA, et al. Immunohistochemical features of canine intracranial embryonal neoplasia: 10 cases. Am Coll Vet Path Ann Meet; Washington, DC; 2018. https://cdn.ymaws.com/www.acvp.org/resource/resmgr/meetings_&_events/2018/acvp_2018-10-25_abstracts.pdf
- 14. Elbert JA, Rissi DR. Doublecortin immunolabeling and lack of neuronal nuclear protein immunolabeling in feline gliomas. J Vet Diagn Invest 2022;34:757–760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Fernández F, et al. Presence of neural progenitors in spontaneous canine gliomas: a histopathological and immunohistochemical study of 20 cases. Vet J 2016;209:125–132. [DOI] [PubMed] [Google Scholar]
- 16. Finotello R, et al. Role of thyroid transcription factor-1 in the diagnosis of feline lung-digit syndrome. J Feline Med Surg 2017;19:477–483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Foiani G, et al. Somatostatin receptor 2 expression in canine meningioma. J Comp Pathol 2019;166:59–68. [DOI] [PubMed] [Google Scholar]
- 18. Heck LC, et al. Pathological features of 78 metastatic or multicentric neoplasms involving the central nervous system in dogs. Pesq Vet Bras 2018;38:1989–1998. [Google Scholar]
- 19. Henker LC, et al. Multifocal spinal cord nephroblastoma in a dog. J Comp Pathol 2018;158:12–16. [DOI] [PubMed] [Google Scholar]
- 20. Higgins RJ, et al. Tumors of the nervous system. In: Meuten DJ, ed. Tumors in Domestic Animals. 5th ed. Wiley, 2017:834–891. [Google Scholar]
- 21. Hirose N, et al. Expression of cell adhesion molecules in canine choroid plexus tumors. J Vet Med Sci 2015;77:255–259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Ide T, et al. Pathological and immunohistochemical features of subdural histiocytic sarcomas in 15 dogs. J Vet Diagn Invest 2011;23:127–132. [DOI] [PubMed] [Google Scholar]
- 23. Ide T, et al. Immunohistochemical characterization of canine neuroepithelial tumors. Vet Pathol 2010;47:741–750. [DOI] [PubMed] [Google Scholar]
- 24. Ide T, et al. Expression of cell adhesion molecules and doublecortin in canine anaplastic meningiomas. Vet Pathol 2011;48:292–301. [DOI] [PubMed] [Google Scholar]
- 25. Janssen J, et al. Osteopontin and Ki-67 expression in World Health Organization graded canine meningioma. J Comp Pathol 2023;201:41–48. [DOI] [PubMed] [Google Scholar]
- 26. Johnson GC, et al. Diagnostic immunohistochemistry of canine and feline intracalvarial tumors in the age of brain biopsies. Vet Pathol 2014;51:146–160. [DOI] [PubMed] [Google Scholar]
- 27. Kishimoto TE, et al. Expression of oligodendrocyte precursor cell markers in canine oligodendrogliomas. Vet Pathol 2018;55:634–644. [DOI] [PubMed] [Google Scholar]
- 28. Koehler JW, et al. A revised diagnostic classification of canine glioma: towards validation of the canine glioma patient as a naturally occurring preclinical model for human glioma. J Neuropathol Exp Neurol 2018;77:1039–1054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Koehler JW, et al. Effects of autolysis and prolonged formalin fixation on histomorphology and immunohistochemistry of normal canine brain tissue: an experimental study. J Vet Diagn Invest 2024;36(2). In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Kriho VK, et al. Keratin expression in astrocytomas: an immunofluorescent and biochemical reassessment. Virchows Arch 1997;431:139–147. [DOI] [PubMed] [Google Scholar]
- 31. Lanigan LG, et al. Comparative pathology of the peripheral nervous system. Vet Pathol 2021;58:10–33. [DOI] [PubMed] [Google Scholar]
- 32. Lubojemska A, et al. Of mice and men: olfactory neuroblastoma among animals and humans. Vet Comp Oncol 2016;14:e70–e82. [DOI] [PubMed] [Google Scholar]
- 33. Mandara MT, et al. Immunoexpression of epithelial membrane antigen in canine meningioma: novel results for perspective considerations. Vet Comp Oncol 2021;19:115–122. [DOI] [PubMed] [Google Scholar]
- 34. Mandara MT, et al. Matrix metalloproteinase-2 and matrix metalloproteinase-9 expression in canine and feline meningioma. Vet Pathol 2009;46:836–845. [DOI] [PubMed] [Google Scholar]
- 35. Mandara MT, et al. Immunohistochemical identification and image analysis quantification of oestrogen and progesterone receptors in canine and feline meningioma. J Comp Pathol 2002;127:214–218. [DOI] [PubMed] [Google Scholar]
- 36. Mariani CL, et al. Histiocytic sarcoma with central nervous system involvement in dogs: 19 cases (2006–2012). J Vet Intern Med 2015;29:607–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Marioni-Henry K, et al. Tumors affecting the spinal cord of cats: 85 cases (1980–2005). J Am Vet Med Assoc 2008;232:237–243. [DOI] [PubMed] [Google Scholar]
- 38. Marr J, et al. A review of proliferative vascular disorders of the central nervous system of animals. Vet Pathol 2021;58:864–880. [DOI] [PubMed] [Google Scholar]
- 39. Matiasek LA, et al. Ki-67 and vascular endothelial growth factor expression in intracranial meningiomas in dogs. J Vet Intern Med 2009;23:146–151. [DOI] [PubMed] [Google Scholar]
- 40. McBride R, et al. Immune cell infiltration in feline meningioma. J Comp Pathol 2017;156:162–168. [DOI] [PubMed] [Google Scholar]
- 41. Miller AD, et al. Canine ependymoma: diagnostic criteria and common pitfalls. Vet Pathol 2019;56:860–867. [DOI] [PubMed] [Google Scholar]
- 42. Miranda IC, Miller AD. SOX2 expression in canine neoplasia. Vet Pathol 2021;58:964–970. [DOI] [PubMed] [Google Scholar]
- 43. Montoliu P, et al. Histological and immunohistochemical study of 30 cases of canine meningioma. J Comp Pathol 2006;135:200–207. [DOI] [PubMed] [Google Scholar]
- 44. Motta L, et al. Canine and feline intracranial meningiomas: an updated review. Vet J 2012;192:153–165. [DOI] [PubMed] [Google Scholar]
- 45. Nentwig A, et al. Aberrant E-cadherin, β-catenin, and glial fibrillary acidic protein (GFAP) expression in canine choroid plexus tumors. J Vet Diagn Invest 2012;24:14–22. [DOI] [PubMed] [Google Scholar]
- 46. Nyska A, et al. Intracranial gangliocytoma in a dog. Vet Pathol 1995;32:190–192. [DOI] [PubMed] [Google Scholar]
- 47. Parker VJ, et al. Olfactory neuroblastoma in a cat. J Feline Med Surg 2010;12:867–871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Petersen SA, et al. Canine intraspinal meningiomas: imaging features, histopathologic classification, and long-term outcome in 34 dogs. J Vet Intern Med 2008;22:946–953. [DOI] [PubMed] [Google Scholar]
- 49. Platt SR, et al. Vascular endothelial growth factor expression in canine intracranial meningiomas and association with patient survival. J Vet Intern Med 2006;20:663–668. [DOI] [PubMed] [Google Scholar]
- 50. Ramos-Vara JA, Borst LB. Immunohistochemistry: fundamentals and applications in oncology. In: Meuten DJ, ed. Tumors in Domestic Animals. 5th ed. Wiley, 2017:44–87. [Google Scholar]
- 51. Ramos-Vara JA, et al. Immunohistochemical detection of CD34, E-cadherin, claudin-1, glucose transporter 1, laminin, and protein gene product 9.5 in 28 canine and 8 feline meningiomas. Vet Pathol 2010;47:725–737. [DOI] [PubMed] [Google Scholar]
- 52. Ramos-Vara JA, et al. Usefulness of thyroid transcription factor-1 immunohistochemical staining in the differential diagnosis of primary pulmonary tumors of dogs. Vet Pathol 2005;42:315–320. [DOI] [PubMed] [Google Scholar]
- 53. Razmara AM, et al. Prevalence and clinicopathologic features of canine metastatic melanoma involving the central nervous system: a retrospective analysis and comparative review. Front Oncol 2022;12:868004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Reginato A, et al. E-cadherin, N-cadherin expression and histologic characterization of canine choroid plexus tumors. Vet Pathol 2016;53:788–791. [DOI] [PubMed] [Google Scholar]
- 55. Reyes VAA, et al. Doublecortin immunolabeling in canine gliomas with distinct degrees of tumor infiltration. J Vet Diagn Invest 2023;35:187–192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Riker J, et al. Histiocytic sarcoma with central nervous system involvement in 6 cats. J Vet Diagn Invest 2023;35:87–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Rissi DR, et al. Canine spinal cord glioma. J Vet Diagn Invest 2017;29:126–132. [DOI] [PubMed] [Google Scholar]
- 58. Rissi DR, et al. Cerebral oligodendroglioma mimicking intraventricular neoplasia in three dogs. J Vet Diagn Invest 2015;27:396–400. [DOI] [PubMed] [Google Scholar]
- 59. Rissi DR, et al. Primary nervous system lymphoma in cats. J Vet Diagn Invest 2022;34:712–717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Rissi DR, et al. Immunohistochemical characterization of immune cell infiltration in feline glioma. J Comp Pathol 2018;160:15–22. [DOI] [PubMed] [Google Scholar]
- 61. Rissi DR, Miller AD. Feline glioma: a retrospective study and review of the literature. J Feline Med Surg 2017;19:1307–1314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Rossmeisl JH, Jr, et al. Cyclooxygenase-2 (COX-2) expression in canine intracranial meningiomas. Vet Comp Oncol 2009;7:173–180. [DOI] [PubMed] [Google Scholar]
- 63. Saito R, et al. Pathological and immunohistochemical features of 45 cases of feline meningioma. J Vet Med Sci 2021;83:1219–1224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Saito R, et al. Immunohistochemical study of autophagy associated molecules and cell adhesion molecules in canine intracranial granular cell tumors. J Vet Med Sci 2022;84:1474–1479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Samarani F, et al. Immunohistochemical expression of cyclooxygenase-2 (COX-2) is not associated with tumor grade in feline meningiomas. Vet J 2018;241:20–23. [DOI] [PubMed] [Google Scholar]
- 66. Sammarco A, et al. Wnt/β-catenin and hippo pathway deregulation in mammary tumors of humans, dogs, and cats. Vet Pathol 2020;57:774–790. [DOI] [PubMed] [Google Scholar]
- 67. Sloma EA, et al. Characterization of inflammatory changes associated with canine oligodendroglioma. J Comp Pathol 2015;153:92–100. [DOI] [PubMed] [Google Scholar]
- 68. Smedley RC, et al. Immunohistochemical diagnosis of canine oral amelanotic melanocytic neoplasms. Vet Pathol 2011;48:32–40. [DOI] [PubMed] [Google Scholar]
- 69. Snyder JM, et al. Canine intracranial primary neoplasia: 173 cases (1986–2003). J Vet Intern Med 2006;20:669–675. [DOI] [PubMed] [Google Scholar]
- 70. Song RB, et al. Postmortem evaluation of 435 cases of intracranial neoplasia in dogs and relationship of neoplasm with breed, age, and body weight. J Vet Intern Med 2013;27:1143–1152. [DOI] [PubMed] [Google Scholar]
- 71. Speciale J, et al. Identification of gonadal steroid receptors in meningiomas from dogs and cats. Am J Vet Res 1990;51:833–835. [PubMed] [Google Scholar]
- 72. The Human Protein Atlas, 2023. Oct 3. https://www.proteinatlas.org
- 73. Troxel MT, et al. Feline intracranial neoplasia: retrospective review of 160 cases (1985–2001). J Vet Intern Med 2003;17:850–859. [DOI] [PubMed] [Google Scholar]
- 74. Uchida K, et al. Ganglioglioma in the thalamus of a puppy. J Vet Med Sci 2003;65:113–115. [DOI] [PubMed] [Google Scholar]
- 75. Valentine BA, et al. Suprasellar germ cell tumors in the dog: a report of five cases and review of the literature. Acta Neuropathol 1988;76:94–100. [DOI] [PubMed] [Google Scholar]
- 76. Vandevelde M, et al. Veterinary Neuropathology: Essentials of Theory and Practice. Wiley, 2012. [Google Scholar]
- 77. Woolford L, et al. Ventricular and extraventricular ependymal tumors in 18 cats. Vet Pathol 2013;50:243–251. [DOI] [PubMed] [Google Scholar]
- 78. World Health Organization (WHO). Central Nervous System Tumours. WHO Classification of Tumours. Vol. 6. 5th ed. International Agency for Research on Cancer, 2022. [Google Scholar]
- 79. York D, et al. Expression and targeting of transcription factor ATF5 in dog gliomas. Vet Comp Oncol 2018;16:102–107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Yoshida K, et al. Immunohistochemical analyses of neural stem cell lineage markers in normal feline brains and glial tumors. Vet Pathol 2024;61:46–57. [DOI] [PubMed] [Google Scholar]
- 81. Yoshida K, et al. Immunohistochemical study of neural stem cell lineage markers in canine brains, gliomas, and a glioma cell line. Vet Pathol 2023;60:35–46. [DOI] [PubMed] [Google Scholar]