Abstract
Background:
Oral cancer remains one of the most dreadful diseases in developing nations. Currently, there has been a rise in the prevalence of tongue squamous cell carcinoma (SCC), with a poor prognosis. The use of standard treatment approaches against oral cancer patients brings about several side effects. In recent years, nanomedicine has provided a versatile platform for developing new targeted therapeutic modalities. However, safety remains a concern in the synthesis of nanoparticles (NPs). Therefore, the present study aims to synthesize safer phytoconstituent-mediated gold NPs (AuNPs) utilizing leaf extracts of Annona muricata, where the biochemical components of the plant leaf act as the reducing and capping agents in the synthesis of NPs, and to evaluate its anti-cancer activity against SCC.
Materials and Methods:
In this in vitro experimental study, AuNPs were synthesized through an effective, simple, and ecologically sound green synthesis method. After characterization of these synthesized AuNPs, in vitro assays such as 3-(4, 5-dimethylthiazole2-yl)-2, 5-biphenyl tetrazolium bromide, wound healing, and clonogenic assays were carried out to investigate the anti-cancer potential of green synthesized AuNPs in the human tongue SCC cell line (SCC-15), and the possible mechanism of action was evaluated through gene and protein expression analysis of Bax, Bcl-2, and p53 genes. The results were expressed as mean ± standard deviation using Statistical Package for Social Sciences (SPSS) 20.0 software and Student's t-test was performed for experimental data. P ≤0.05 were considered statistically significant.
Results:
The in vitro assays demonstrated that the synthesized AuNPs are exhibiting anti-cancer activity by apoptosis of SCC-15 cells in a dose-dependent manner. Further, it also revealed a highly significant decrease in anti-apoptotic Bcl-2 gene expression, whereas pro-apoptotic genes p53 and Bax revealed a highly significant increase, which is statistically significant compared to the control (P < 0.05).
Conclusion:
Our findings demonstrated that the AuNPs synthesized from A. muricata leaf extract could act as a novel anticancer agent, particularly against SCC, after further scrutiny.
Keywords: Anticancer agent, apoptosis, nanoparticles, oral tongue squamous cell carcinoma
INTRODUCTION
According to GLOBOCAN 2020 estimates, there were 19.3 million new cases of cancer and about 10 million deaths due to cancer. It is also estimated that the total number of cancer cases worldwide is predicted to reach 28.4 million in 2040, an increase of 47% from 2020. This spike will be significantly greater in developing (64%–95%) than in developed (32%–56%) countries because of changing demographics, an expanding economy, and new risk concerns brought forth by globalization.[1]
Among various cancers, recent reports indicate that tongue squamous cell carcinoma (SCC) in young people is on the rise, especially in females. Despite the various treatment modalities available, the survival rate of patients has remained unchanged over the past 30 years.[2,3] Hence, there is a need for a novel, safe, and potent targeted anticancer therapy against oral SCC.
Nanotechnology is concerned with the synthesis and creation of materials at the nanoscale that display novel behaviors that are different from those of molecules or bulk materials. Researchers from every branch of science have paid intense attention to this peculiar feature.[4]
Among the inorganic nanoparticles (NPs), gold NPs (AuNPs) are the most widely used due to their versatile, inert, biocompatible, and stable properties. Due to their well-defined surface chemistry, AuNPs can be easily conjugated with different molecules and are not hazardous to humans in low doses. These attributes make them a great option for a variety of medicinal applications, particularly cancer treatment.[5]
The synthesis of NPs can be done through different physical and chemical approaches. However, these methods are expensive and unsafe for human health due to the use of toxic components and the generation of byproducts, either during the synthesis or during the capping/stabilization process of NPs.[6]
Recently, AuNPs synthesized utilizing plant extract have been found to be more effective and beneficial. In addition, it produces a significant amount of NPs and lessens the environmental contamination. They may serve as stabilizing and reducing agents in the creation of NPs. Hence, the advantages of plant extract-mediated AuNPs production over alternative approaches are its ease of use, safety, and compatibility with the environment.[7]
Annona muricata L., otherwise commonly called soursop, is one of the 2300 species in the Annonaceae family, which has roughly 130 genera. Originally, it belonged to the hottest tropical regions of South and North America and is now found around the world.[8] Previous studies demonstrated significant cytotoxicity for A. muricata leaves through induction of apoptosis against various cancer cell lines including MCF7 breast cancer cells, A549 lung cancer cells, HL-60 leukemia cells, and HepG2 liver cancer cells.[9,10,11]
Apoptosis is the process of programmed cell death. The resistance to cell death constitutes one of the important hallmarks of cancer.[12] By altering the expression of anti- or pro-apoptotic genes, cancer cells may be able to evade apoptosis. Apoptosis is controlled by proteins from the Bcl-2 family. Cell death inducers and inhibitors are members of this family of interfering partners. Together, they govern and mediate the intrinsic apoptosis pathway, which is the mechanism through which mitochondria cause cell death. The normal development of the embryo as well as the prevention of cancer mainly depends on this pathway. The Bcl-2 gene is one of the most notable anti-apoptotic proteins expressed in oral SCC that promotes cell survival, while the Bax gene acts as a pro-apoptotic gene. Mutant p53-gene cells have a greater possibility of escaping apoptosis.[13]
According to the results of some research, AuNPs were synthesized from A. muricata leaf extract and tested for both antibacterial and anticancer properties.[14,15] However, the anti-cancer activities of A. muricata leaf extract mediated (ALEM) AuNP on oral cancer have not been evaluated so far.
Hence, the present study aims to investigate the anti-cancer potential of the ALEM-AuNP in oral cancer cells, particularly SCC-15, and to elucidate its potential pro-apoptotic properties that may have contributed to this effect through gene and protein expression analysis of Bax, Bcl-2, and p53 genes through in vitro assays.
MATERIALS AND METHODS
The study was approved by the Institutional Ethical Committee of Sree Mookambika Institute of Medical Sciences, Kulasekharam, Tamil Nadu, dated March 25, 2021.
Study design
This was in vitro experimental study.
Collection and conservation of plant material
The mature leaves of A. muricata L. were collected (8°21’32.7”N and 77°15’56.7”E) during the flowering stage. The plant has been authenticated by Scientist-E and Head of Office, Botanical Survey of India, as A. muricata Linn. (Family: Annonaceae). The voucher specimen is maintained in our institution for further reference (BSI/SRC/5/23/2021/Tech/301).
Preparation of aqueous leaf extract of Annona muricata L.
The leaves of A. muricata were carefully washed with water and allowed to dry in the shade. The withered leaves were pulverized in a blender to form a granular powder. The 10 g of powdered plant material sample was extracted with 170 mL of distilled water at 80°C–100°C, depending on the evaporation point of the solvent by Soxhlet extraction. The process of extraction continues for 24 h or until the solvent in the syphon tube of an extractor becomes colorless. After that, the extract was taken in a beaker and kept in a water bath at 30°C –40°C till all the solvent evaporated, finally we got the extract in a semisolid state. The extract was stored in the refrigerator at 4°C for further use.
Green synthesis method for gold nanoparticles
The AuNP were synthesized using the technique described by Folorunso et al.[15] The green synthesis of AuNP was achieved by adding 20 mL of a 1 mM solution of gold chloride to 100 mL of freshly prepared aqueous A. muricata leaf extract and well agitated at room temperature till the color changed to deep purple, confirming the synthesis of AuNPs. The combination was incubated for about 24 h, to achieve complete bioreduction. Finally, the green synthesized ALEM-AuNPs were subjected to characterization.
Characterization of the synthesized gold nanoparticles
Here, field emission scanning electron microscopy (SEM) (JSM-7100F, field emission scanning electron microscope [FESEM] JEOL, USA) was used to evaluate the morphology and energy-dispersive X-ray (EDX) analysis was used for measuring the elemental composition.
Cell line and cell culture
The human tongue SCC cell line, SCC-15 was obtained from the American type culture collection (CRL-1623). Cells were cultured in Dulbecco's modified eagles medium and supplemented with 10% fetal bovine serum, 1% L-Glutamine, 0.1 mM nonessential amino acid, 100 U/mL of penicillin, and 100 μg/mL of streptomycin. Cultures were incubated in a humidified incubator at 37°C with 5% CO2.
Cytotoxicity assay
The 3-(4, 5-dimethylthiazole2-yl)-2, 5-biphenyl tetrazolium bromide (MTT) assay was used to determine the percentage of cell inhibition. Cells at 70%–80% confluence were trypsinized and plated into a 96-well plate. After overnight incubation, cells were treated with various concentrations of AuNPs at concentrations 10, 20, 100, 200, 500, and 1000 µg/mL for 24 h. After that, the medium was aspirated and the cells were washed with phosphate-buffered saline (PBS). Each well received 0.5 mg/mL of MTT solution. After 4 h of dark incubation at 37°C, 200 µL of dimethyl sulfoxide was replaced in the place of MTT solution. An enzyme-linked immunosorbent assay reader measured the absorbance at 570 nm after agitation at 150 rpm for 5 min.
Colony formation/clonogenic assay
The effect of ALEM-AuNPson colony formation was determined in parallel with the control. SCC-15 cells (300 cells/well) were seeded in 12-well plates and treated with ALEM-AuNPs. The cells were maintained in a humidified incubator at 37°C and 5% CO2. After 14 days of culture, the medium was discarded, and cells were washed with PBS. The cells were fixed with 4% paraformaldehyde at room temperature for 30 min and stained with 0.1% crystal violet for 30 min. Then, the crystal violet was removed and, the plates were air-dried. The plates were placed under a microscope to count clones with more than 50 cells. The experiment was repeated three times.
Wound healing/cell migration assay
Confluent monolayers of cells were grown in 6-well plates at 1–100 cells per well, and after overnight starvation in serum-free media, the cells were harvested. Two hundred microliter pipette tips were used to create straight wounds. Then, control and ALEM-AuNPs-treated SCC-15 cells were maintained in a humidified incubator at 37°C and 5% CO2 for 24 h. The wound gaps were imaged at both 0 and 24 h. Using image analysis software, we compared the wound closure rates between the control and treated groups and calculated the percentage of wound closure relative to the initial wound size. Results were obtained after three separate experiments.
Apoptosis gene expression profiling using a polymerase chain reaction array
Real-time polymerase chain reaction (RT-PCR) was used to assess the levels of Bcl-2, Bax, and p53 mRNA expression. Total RNA from treated and untreated cells was extracted using the Trizol reagent (Invitrogen, Waltham, MA, USA). With the help of the Prime Script 1st Strand cDNA synthesis kit from Takara (Japan), the total extracted RNA (1 g) was reverse transcribed. Using the IQ SYBR Green PCR master mix kit, real-time PCR was performed in the Bio-Rad CFX96 Real-Time PCR Machine. RT-PCR was carried out as follows: 40 cycles of 94°C for 30 s and 55°C for 30 s were performed following 95°C for 5 min. The 2∆∆Ct technique was used to calculate the fold changes in gene expression between the control and treatment samples. GAPDH was used as an internal control, and each reaction was carried out in triplicate.
Western blot analysis
After treatment of SCC-15 cells with ALEM-AuNPs, the cells were collected and lysed with radioimmunoprecipitation assay buffer. Protein concentration was measured using a bicinchoninic acid protein assay kit (Thermo Fisher Scientific, USA) with standard protein concentrations of bovine serum albumin (BSA). A 10% sodium dodecyl sulfate polyacrylamide gel was prepared and loaded with an equal amount of protein samples along with a protein molecular weight marker. Electrophoresis was performed at a constant voltage until the dye front reached the bottom of the gel. The proteins from the gel were carefully transferred to a polyvinylidene fluoride membrane using a semi-dry system (Bio-Rad). The membrane was blocked with a blocking buffer (5% BSA) for 1 h at room temperature. In addition, the membrane was treated with appropriate primary monoclonal antibodies specific to target proteins (P53, Bcl-2, Bax, and GAPDH; Santa Cruz, USA) overnight at 4°C. Then, the membrane was gently washed with the appropriate washing buffer (Tris-buffered saline with Tween-20) to remove unbound antibodies. The membrane was incubated with a suitable secondary antibody conjugated with a horseradish peroxidase enzyme for 2 h at room temperature. Finally, the membrane was washed properly with tris-buffered saline with tween buffer to remove unbound secondary antibodies. A chemiluminescent substrate was used (ECL, Bio-Rad), and the protein bands were visualized using a ChemiDoc imaging system (Bio-Rad). Finally, the protein bands were analyzed and the protein expression levels were quantified using image lab software (Bio-Rad).
Statistical analysis
The results were expressed as mean ± standard deviation (SD) using IBM SPSS (Statistical Package for Social Sciences), Version 20.0. Armonk, NY: IBM Corp.
All graphs present the mean and standard variations of at least three independent experiments and a Student's t-test has been performed for experimental data. P ≤0.05 were considered statistically significant.
RESULTS
Macroscopic analysis
The synthesis of metal NPs is indicated by the color change of the solution. As depicted in Figure 1, the ALEM, after the addition of gold chloride, gradually changes its color from brownish yellow to deep purple after 24 h. The color change is due to the excitation of free electrons in the NPs. After 24 h, no further color change was observed due to the stabilization of synthesized NPs.
Figure 1.

Photograph showing color change in the Annona muricata leaf extract mediatedgold nanoparticles. (a) Gold chloride (b) Gold chloride added with A.muricata leaf extract showing brownish yellow color (c) Gold chloride added with A.muricata leaf extract showing deep purple color after 24 h.
Energy dispersive X-ray spectrum analysis
The elemental spectrum technique of EDX analysis is used to confirm the presence of gold in a zero-valent form. The EDX spectrum revealed that Au possesses a high peak confirming the synthesized AuNPs. The weight percent composition of Au was 22.9%, respectively.
In this work, the EDX analysis indicated the phytoreduction of gold chloroaurate into AuNPs through strong signals of elemental gold atoms that were detected at several energy levels mainly at 1.5 KeV and also at 2.2, 8.5, 9.8, 11.5, and 13.5 KeV. Moreover, signals for other elements such as carbon and oxygen were observed which are suggested to be of plant origin. Furthermore, signals for copper were detected that could originate from the carbon-coated copper grid used for this analysis [Figure 2].
Figure 2.

Graph showing the presence of Au in the elemental spectrum technique of Energy Dispersive X-ray analysis.
Morphological characteristics of Annona muricata leaf extract mediated-gold nanoparticles using scanning electron microscopy analysis
Figure 3 illustrates the analysis of the surface morphology of ALEM-AuNPs. Figure 3a-d, which is FESEM images, demonstrated well-defined nano-cubes of varying sizes. Overall, the ALEM-AuNPs synthesized using the green synthesis method had a particle size ranging from 100 to 500 nm.
Figure 3.

(a-d) 3D-Photomicrographs showing the surface morphology of Annona muricata leaf extract mediated-AuNPs in Scanning electron microscopy analysis.
In vitro cytotoxic effects of Annona muricata leaf extract mediated-gold nanoparticles on squamous cell carcinoma-15 cells
In the current research, the cytotoxic effect of ALEM-AuNPs was evaluated preliminarily by MTT assay. The percentage of inhibition was calculated using the following formula.[16]
Percentage of inhibition = (Control OD−Sample OD) ×100/Control OD
A linear regression curve is used for the determination of the half-maximal inhibitory concentration value, which was found to be 416.23 µg/mL. It was observed that ALEM-AuNPs showed significant cytotoxic effects (P ≤ 0.05) on SCC-15 cell lines that were represented as mean ± SD in triplicate [Figure 4 and Table 1]. The results also showed a significant cytotoxic effect when the concentration was increased. After 24 h, at a concentration of 10 µg/mL the treated cells showed 10.9% inhibition whereas at 1000 µg/mL, 86.95% inhibition of the treated SCC-15 cell line was observed [Figure 5].
Figure 4.
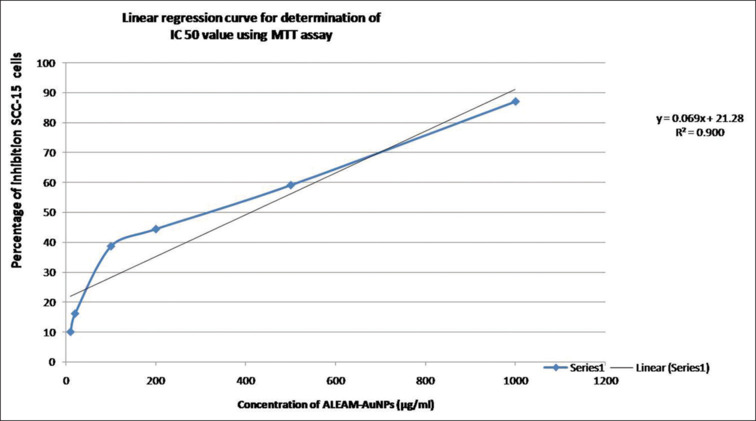
Graph showing the linear regression curve at 50% inhibition of squamous cell carcinoma-15 cells in 3-(4, 5-dimethylthiazole2-yl)-2, 5-biphenyl tetrazolium bromide assay using the straight-line equation. SCC: Squamous cell carcinoma, MTT: 3-(4, 5-dimethylthiazole2-yl)-2, 5-biphenyl tetrazolium bromide, IC50: Half-maximal inhibitory concentration, ALEM: Aqueous leaf extract of Annona muricata mediated, AuNPs: Gold nanoparticles.
Table 1.
Evaluation of cytotoxic effect of aqueous leaf extract of Annona muricata mediated gold nanoparticles by 3-(4, 5-dimethylthiazole2-yl)-2, 5-biphenyl tetrazolium bromide assay
| Extract used | ALEM concentrations (µg/mL) | OD at 540 nm | Percentage of inhibition (SCC - 15 cells) |
|---|---|---|---|
| ALEM-AuNPs | Control | 0.525 | 0.00 |
| 10 | 0.472 | 10.09 | |
| 20 | 0.440 | 16.19 | |
| 100 | 0.322 | 38.66 | |
| 200 | 0.292 | 44.38 | |
| 500 | 0.215 | 59.04 | |
| 1000 | 0.069 | 86.95 |
ALEM: Aqueous leaf extract of Annona muricata mediated; ALEM-AuNPs: ALEM gold nanoparticles; OD: Optical density; SCC: Squamous cell carcinoma
Figure 5.
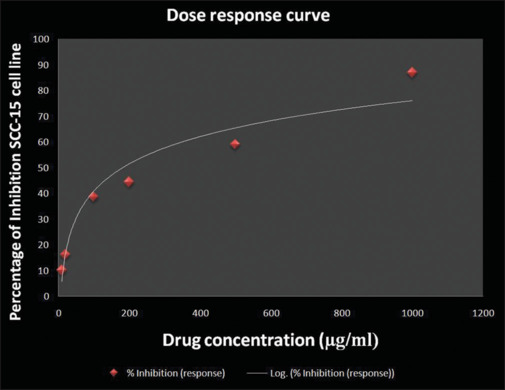
Graph showing the dose response curve for 3-(4, 5-dimethylthiazole2-yl)-2, 5-biphenyl tetrazolium bromide assay. SCC: Squamous cell carcinoma.
Annona muricata leaf extract mediated-gold nanoparticles inhibit the clonogenic potential of squamous cell carcinoma-15 cells
The ability of a single cell to multiply into a colony is the foundation of the clonogenic assay, also known as the colony formation assay. At least 50 cells must be present for the colony to be considered. Every cell in the population is essentially examined as part of the experiment to determine whether it has the capacity for unlimited division.[17] Here, a colony formation assay was performed to evaluate the effect of green synthesized AuNPs on the ability of cancer cells to form colonies. The findings showed that ALEM-AuNPs treatment significantly reduced the number of colonies formed in SCC-15 cell lines [Figure 6] compared to their control (P < 0.05). These findings confirmed that treatment with green synthesized AuNPs inhibits the clonogenic potential of oral cancer cells.
Figure 6.
(a) Photograph showing reduced number of colonies formed in Annona muricata- gold nanoparticles (AuNPs) treated squamous cell carcinoma-15 cell lines. (b) Bar graph of squamous cell carcinoma-15 cell colonies at 14 days after treated with control and Annona muricata-AuNPs. AuNPs: Gold nanoparticles.
Annona muricata leaf extract mediated-gold nanoparticles inhibit the migration of squamous cell carcinoma-15 cells
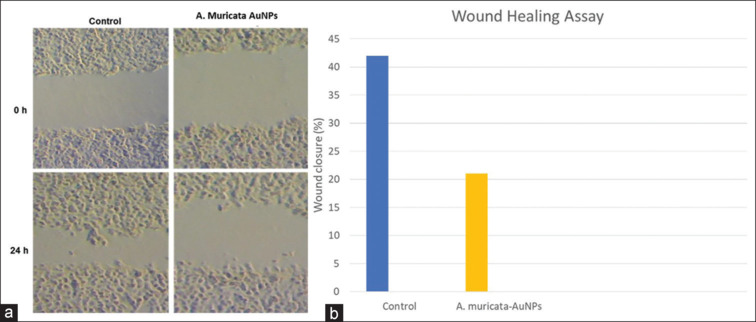
To determine the functional impact of green synthesized AuNP treatment on cancer cell migration, a wound healing assay was performed. Our results showed that ALEM-AuNPs treatment effectively inhibited the migration of SCC-15 cells when compared with their respective control groups after 24 h [Figure 7].
Figure 7.
(a and b) Wound healing assay in squamous cell carcinoma-15 cells. Photomicrograph and bar chart show that treatment with Annona muricata-Gold nanoparticles dramatically reduced the migration of squamous cell carcinoma-15 cells at 0 and 24 h compared to their control cells. AuNPs: Gold nanoparticles.
Effect of Annona muricata leaf extract mediated-gold nanoparticles on the expression of apoptosis-related genes and proteins
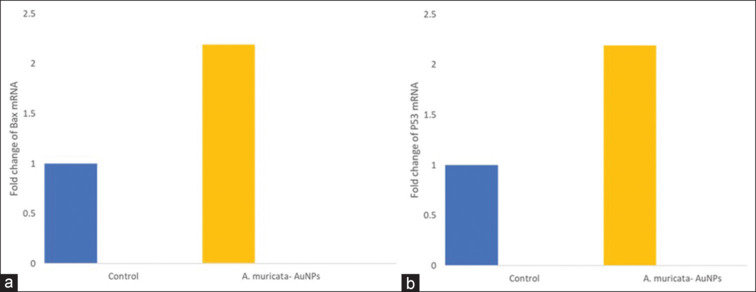
After preliminary in vitro assays, gene and protein expression analysis was performed. Compared with the negative control, ALEM-AuNPs increased the mRNA expression of the pro-apoptotic gene Bax, and p53 and decreased the expression of anti-apoptotic gene Bcl-2 in SCC-15 cells particularly at the treatment dose [Figures 8 and 9]. The results were statistically significant with a P < 0.05 as compared with the control. All these findings suggest that the anti-cancer activity executed by ALEM-AuNPs in oral cancer cells, particularly SCC-15 is mediated through apoptotic pathways.
Figure 8.
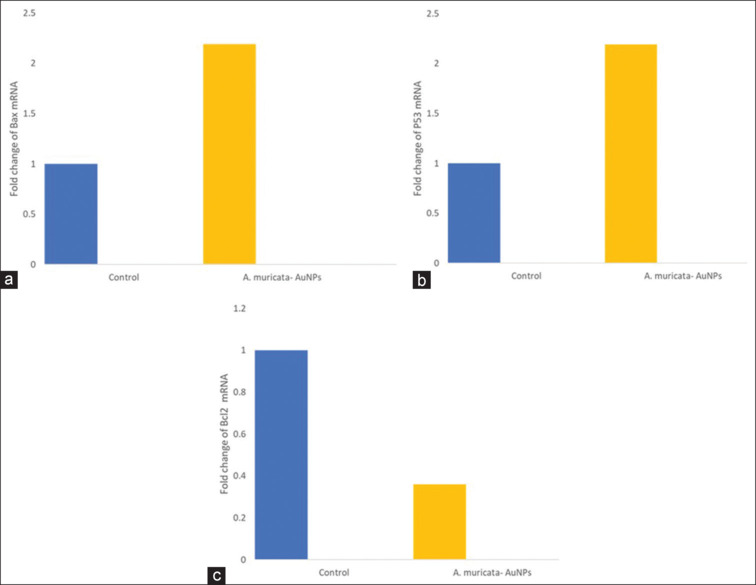
(a-c) Representative graphs comparing the fold changes in the mRNA expression of Bax, P53, and Bcl-2 in squamous cell carcinoma-15 cells treated with Annona muricata- Gold nanoparticles to the control. AuNPs: Gold nanoparticles.
Figure 9.
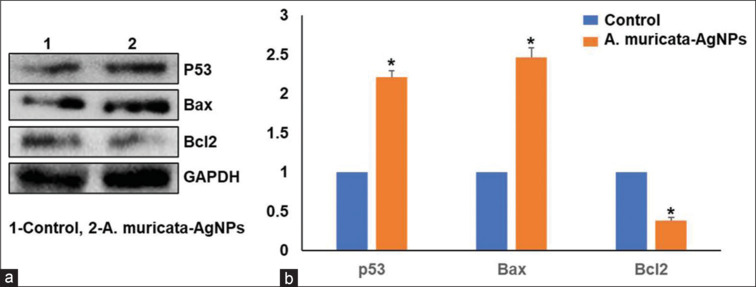
(a) Protein expression of p53, Bax, and Bcl2 in squamous cell carcinoma (SCC)-15 cells treated with control and Annona muricata-gold nanoparticles (AuNPs) in the Western blots analysis. (b) Bar graph of p53, Bax, and Bcl2 expression in SCC-15 cells treated with control and Annona muricata-AuNPs. AuNPs: Gold nanoparticles.
DISCUSSION
The current conventional treatment options for early-stage oral SCC include surgery, radiotherapy, or combination therapy with chemotherapy in advanced cases.[18] Apart from these treatment modalities, it was found that the overall median 5-year survival rate is 54.1%. Moreover, <50.0% in lesions involving the base of the tongue and oropharynx.[19] In addition, they produce certain adverse reactions and affect healthy cells.[20] Despite improving the patient's quality of life, they result in morbidity and mortality of the patient.[21]
This led to the notion of chemopreventive agents, particularly targeting cancer cells; one among them is herbal nanomedicines. They are safer, have higher bioavailability, and have enhanced therapeutic value than conventional herbal and synthetic agents.[22] Hence, in the present study, we employed the green synthesis approach of nanoparticle synthesis and this could be a safer method of choice due to the nontoxic nature of the reactions used in this technique.[23]
In the current research work, AuNPs were synthesized using an aqueous leaf extract of A. muricata that has shown promising results as an anti-cancer agent.[24] This was the first study to evaluate the anticancer effects of ALEM-AuNPs on oral cancer cells. In this study, the reduction of gold chloride to AuNPs is initially confirmed by the visual observation method indicated by color change.
The term “nanotechnology” refers to the designing and use of elements that are produced at the nanoscale of 1 nm or up to 1000 nm in size. In biomedical applications, the characteristics and performance of AuNPs are heavily influenced by the size of the nanoparticles. Particles <2 nm are highly toxic and at sizes more than 5 nm, they are considered to be chemically inert.[25]
Previous studies on other plant extracts mostly showed spherical-shaped nanoparticles with the least size of 2–1000 nm.[26,27] In a study conducted by Wang et al., images from the TEM study revealed that the green synthesized AuNPs from an aqueous extract of Scutellaria barbata had a spherical form, and their usual sizes ranged from 400 nm to 1000 nm.[27]
In the present study, to determine the morphology and size of the green synthesized AuNPs, they are characterized using SEM and ERD analysis. According to our study's FESEM images, the ALEM-AuNPs showed anisotropic morphologies with notable square-shaped nanocubes that ranged in size from 100 to 500 nm. The size range of the nanoparticles in our investigation was in accordance with that of the study by Wang et al.[27] In addition, triangular and tetragonal NPs were also observed. They were distributed uniformly and well dispersed without aggregation. This indicated that the phytochemicals present in the leaf extract possess a good capping effect and prevent clumping of the particles.
In the present study, the elemental composition was determined through EDX analysis, a typical method used to determine the total quantity of the synthesized AuNPs using biological components as a source, especially plants.[28] The results of the present study showed that Au possesses a high peak with a weight percentage of 22.9% [Figure 2].
In the current study, we preliminarily evaluated the cytotoxic effects of ALEM-AuNPs on SCC-15 cell lines through MTT assay, and the results showed promising cytotoxic effects. Then, we further investigated the potential long-term and functional impact of ALEM-AuNPs on SCC-15 cells using clonogenic and wound healing in vitro assays. The results were confounding and positive, showing that ALEM-AuNPs exhibit potent anti-proliferative and anti-metastatic activity. No previous studies were reported using ALEM-AuNPs on SCC-15 cells. However, studies on other plant extract-mediated AuNPs have shown potential cytotoxic effects on various cancer cells including head and neck cancer.[6,29,30,31]
The findings of the clonogenic assay in our study were in accordance with previous studies. In a study conducted by Chamcheu et al. on the Human NonMelanoma Skin Cancer cell line, the Graviola leaf and stem extract (GLSE) showed dose-dependent inhibition of colony formation at concentrations of GLSE (0-160 µg/mL) on UW-BCC1 and A431 cells after an incubation period of 14–16 days. They also observed a significant reduction in the number of colonies formed relative to untreated controls.[32]
Another research work by Kim et al. found that the triple-negative breast cancer (TNBC) cell line, MDA-MB-231 showed a good reduction in clonogenicity when compared to the non-TNBC cell line MCF-7 cell line. They also stated that the total progression of the clonogenicity was observed in 24 h to the 100 µg/mL concentrations.[33]
Similar findings were observed when the MCF-7 breast cancer cell line, and the BPH-1 prostate cancer cell line, were treated with an aqueous leaf extract of A. muricata in the studies conducted by Hadisaputri et al.[9] and Asare et al.[34]
In another in vivo study conducted by Syed Najmuddin et al., A. muricata leaves crude aqueous extract (AMCE) showed a decrease in the number of colonies formed in the lung of 6–8-week-old female BALB/c mice in the AMCE treatment group (15 ± 0.82) compared to the control group (67 ± 2.05).[35]
The invasion and migration of tumor cells have long been recognized as serious obstacles to effective treatment. To determine the functional impact of ALEM on the metastatic potential of the SCC-15 cell line, a wound healing assay was performed in the present research work as the progression of a tumor is not only reliant on the pace at which it proliferates. The research findings demonstrated that ALEM and ALEM-AuNPs demonstrated their potential to stop the migration of cancer cells in the wound healing experiment by delaying the growth of treated SCC-15 cells towards the center of the wound area when compared to the control.
Evidently, the anti-metastatic potential of ALEM is in accord with the previous findings from other studies by Syed Najmuddin et al.,[35] Kim et al.[33] however on different cancer cell lines. Another study conducted by Torres et al. on pancreatic cancer (PC) cells found that A. muricata inhibited the migratory capacity of metastatic PC cell lines FG/COLO357 and CD18/HPAF both in vitro and in vivo thereby substantiating its anti-metastatic potential.[36]
Apoptosis is a complex process that involves two main pathways (extrinsic and intrinsic), each of which is controlled at various levels. The extrinsic pathway depends on the cell surface receptors, its inhibitory competitors, and associated cytoplasmic proteins whereas the intrinsic pathway focuses on the mitochondria and its prime apoptogenic elements.[37] The key regulators of the intrinsic pathway are the pro- and anti-apoptotic members of the Bcl-2 family.[38,39] The key regulators in both the extrinsic and intrinsic pathways can be modulated by the apoptotic process regulator p53.[40]
Previous studies have shown that AuNPs synthesized from various plant extracts cause apoptosis by inhibiting the expression of apoptotic proteins in multiple cell lines.[7,27,41] In this study, the pro-apoptotic properties of ALEM-AuNPs were confirmed by the gene and protein expression analysis of the pro-apoptotic and anti-apoptotic proteins, Bax, Bcl2, and p53. The results revealed that ALEM-AuNPs treatment on SCC-15 cell lines exhibited upregulation of Bax, P53, and downregulation of Bcl-2 at the gene and protein expression levels. These findings imply that ALEM-AuNPs effectively induce apoptosis by regulating the intrinsic apoptotic pathway [Figure 10].
Figure 10.
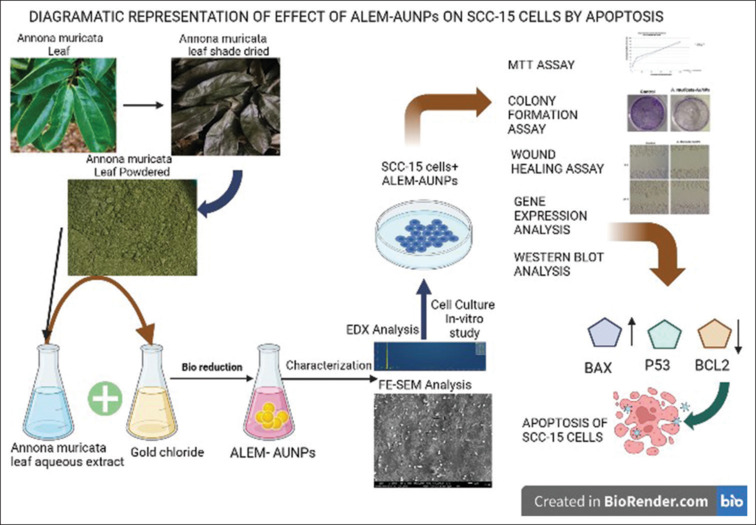
Schematic representation of the effect of Annona muricata leaf extract mediated-AuNPs on squamous cell carcinoma-15 cells by apoptosis. SCC: Squamous cell carcinoma, ALEM: Aqueous leaf extract of Annona muricata mediated, AuNPs: Gold nanoparticles, EDX: Energy-dispersive X-ray, FESEM: Field emission scanning electron microscope.
Limitations and future perspectives
This study was carried out on crude leaf extract and further investigation is needed, nevertheless, to ascertain which component of A. muricata leaves is actually active and thereby acts as a pro-apoptotic agent. Moreover, the majority of the long-promised benefits have been supported by in vitro assays; however, they still need to be confirmed by preclinical in vivo research, as well as human clinical trials. Furthermore, the effects on normal oral epithelial cells, possible drug interactions, mechanism of action, and appropriate dosage are yet to be evaluated to employ the green synthesized leaf extract of A. muricata-mediated AuNPs as a viable therapeutic agent to treat oral cancer. Analysing AuNPs’ safety and biocompatibility is essential before using them in long-term applications. Further, for greater reliability, the toxicological profile must be recorded. To establish and validate the model, in vitro data generation is usually combined with in silico methods.[42]
CONCLUSION
The study of the anticancer effect of ALEM-AuNPs is very limited so far and to the best of our knowledge from the literature search, this was the first study to evaluate the anticancer effect of ALEM-AuNPs on oral cancer cell lines, particularly oral SCC. Interestingly, our study has proven that ALEM-AuNPs show potent anti-cancer efficacy, which is exhibited through their pro-apoptotic properties. Further, the green synthesis approach of AuNPs is found to be safe, cost-effective, eco-friendly, and less time-consuming. Hence, our study demonstrates that ALEM-AuNPs could be developed as a novel anti-cancer drug for the treatment of oral SCC after further clinical trials.
Financial support and sponsorship
Nil.
Conflicts of interest
The authors of this manuscript declare that they have no conflicts of interest, real or perceived, financial or non-financial in this article.
Acknowledgments
We would like to acknowledge Molecular Biology Lab, Saveetha Institute of Medical and Technical Sciences for conducting SEM analysis. We also want to convey our gratitude to Dr. Isaac Jospeh, Dr. Girish, and Dr. Angelin for their guidance and support. Special thanks to Dr. Beyanso CP Daniel for his immense help in the completion of this study.
REFERENCES
- 1.Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Feller L, Lemmer J. Oral squamous CELL carcinoma: Epidemiology, clinical presentation and treatment. J Cancer Ther. 2012;4:263–8. [Google Scholar]
- 3.Paderno A, Morello R, Piazza C. Tongue carcinoma in young adults: A review of the literature. Acta Otorhinolaryngol Ital. 2018;38:175–80. doi: 10.14639/0392-100X-1932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kedi PB, Meva FE, Kotsedi L, Nguemfo EL, Zangueu CB, Ntoumba AA, et al. Eco-friendly synthesis, characterization, in vitro and in vivo anti-inflammatory activity of silver nanoparticle-mediated Selaginella myosurus aqueous extract. Int J Nanomedicine. 2018;13:8537–48. doi: 10.2147/IJN.S174530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hu X, Zhang Y, Ding T, Liu J, Zhao H. Multifunctional gold nanoparticles: A novel nanomaterial for various medical applications and biological activities. Front Bioeng Biotechnol. 2020;8:990.. doi: 10.3389/fbioe.2020.00990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Santhosh PB, Genova J, Chamati H. Green synthesis of gold nanoparticles: An eco-friendly approach. Chemistry. 2022;4:345–69. [Google Scholar]
- 7.Sun B, Hu N, Han L, Pi Y, Gao Y, Chen K. Anticancer activity of green synthesised gold nanoparticles from Marsdenia tenacissima inhibits A549 cell proliferation through the apoptotic pathway. Artif Cells Nanomed Biotechnol. 2019;47:4012–9. doi: 10.1080/21691401.2019.1575844. [DOI] [PubMed] [Google Scholar]
- 8.Ilango S, Sahoo DK, Paital B, Kathirvel K, Gabriel JI, Subramaniam K, et al. A review on Annona muricata and its anticancer activity. Cancers (Basel) 2022;14:4539.. doi: 10.3390/cancers14184539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hadisaputri YE, Habibah U, Abdullah FF, Halimah E, Mutakin M, Megantara S, et al. Antiproliferation activity and apoptotic mechanism of soursop (Annona muricata L.) leaves extract and fractions on MCF7 breast cancer cells. Breast Cancer (Dove Med Press) 2021;13:447–57. doi: 10.2147/BCTT.S317682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pieme CA, Kumar SG, Dongmo MS, Moukette BM, Boyoum FF, Ngogang JY, et al. Antiproliferative activity and induction of apoptosis by Annona muricata (Annonaceae) extract on human cancer cells. BMC Complement Altern Med. 2014;14:516.. doi: 10.1186/1472-6882-14-516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Moghadamtousi SZ, Kadir HA, Paydar M, Rouhollahi E, Karimian H. Annona muricata leaves induced apoptosis in A549 cells through mitochondrial-mediated pathway and involvement of NF-κB. BMC Complement Altern Med. 2014;14:299.. doi: 10.1186/1472-6882-14-299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hanahan D, Weinberg RA. Hallmarks of cancer: The next generation. Cell. 2011;144:646–74. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 13.Dwivedi R, Pandey R, Chandra S, Mehrotra D. Apoptosis and genes involved in oral cancer – A comprehensive review. Oncol Rev. 2020;14:472.. doi: 10.4081/oncol.2020.472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Heyam Saad Ali, Imran Muhammad Husseini, Ghaleb Awad, Nahid S. Paul Vinod El-Haj, Babiker M, et al. An effective anticancer nano-approach for melanoma and breast cancers using Annona muricate gold nanoparticles. Acta Pharm Sci. 2021;5:46–54. [Google Scholar]
- 15.Aderonke F, Sunday A, Kolawole AB, Samuel A, Babawale A, Ibrahim A, et al. Biosynthesis, characterization and antimicrobial activity of gold nanoparticles from leaf extracts of Annona muricata. J Nanostructure Chem. 2019;9:111–7. [Google Scholar]
- 16.Nadri MH, Salim Y, Basar N, Yahya A, Zulkifli RM. Antioxidant activities and tyrosinase inhibition effects of Phaleria macrocarpa extracts. Afr J Tradit Complement Altern Med. 2014;11:107–11. doi: 10.4314/ajtcam.v11i3.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Franken NA, Rodermond HM, Stap J, Haveman J, van Bree C. Clonogenic assay of cells in vitro. Nat Protoc. 2006;1:2315–9. doi: 10.1038/nprot.2006.339. [DOI] [PubMed] [Google Scholar]
- 18.Huang SH, O’Sullivan B. Oral cancer: Current role of radiotherapy and chemotherapy. Med Oral Patol Oral Cir Bucal. 2013;18:e233–40.. doi: 10.4317/medoral.18772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jehn P, Dittmann J, Zimmerer R, Stier R, Jehn M, Gellrich NC, et al. Survival rates according to tumour location in patients with surgically treated oral and oropharyngeal squamous cell carcinoma. Anticancer Res. 2019;39:2527–33. doi: 10.21873/anticanres.13374. [DOI] [PubMed] [Google Scholar]
- 20.Tolentino Ede S, Centurion BS, Ferreira LH, Souza AP, Damante JH, Rubira-Bullen IR. Oral adverse effects of head and neck radiotherapy: Literature review and suggestion of a clinical oral care guideline for irradiated patients. J Appl Oral Sci. 2011;19:448–54. doi: 10.1590/S1678-77572011000500003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Naidu MU, Ramana GV, Rani PU, Mohan IK, Suman A, Roy P. Chemotherapy-induced and/or radiation therapy-induced oral mucositis – Complicating the treatment of cancer. Neoplasia. 2004;6:423–31. doi: 10.1593/neo.04169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Teja PK, Mithiya J, Kate AS, Bairwa K, Chauthe SK. Herbal nanomedicines: Recent advancements, challenges, opportunities and regulatory overview. Phytomedicine. 2022;96:153890.. doi: 10.1016/j.phymed.2021.153890. [DOI] [PubMed] [Google Scholar]
- 23.Roy A, Datta S, Luthra R, Khan MA, Gacem A, Hasan MA, et al. Green synthesis of metalloid nanoparticles and its biological applications: A review. Front Chem. 2022;10:994724.. doi: 10.3389/fchem.2022.994724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yajid AI, Ab Rahman HS, Wong MP, Wan Zain WZ. Potential benefits of Annona muricata in combating cancer: A review. Malays J Med Sci. 2018;25:5–15. doi: 10.21315/mjms2018.25.1.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Akkam N, Aljabali AA, Akkam Y, Abo Alrob O, Al-Trad B, Alzoubi H, et al. Investigating the fate and toxicity of green synthesized gold nanoparticles in albino mice. Drug Dev Ind Pharm. 2023;49:508–20. doi: 10.1080/03639045.2023.2243334. [DOI] [PubMed] [Google Scholar]
- 26.Amina SJ, Guo B. A review on the synthesis and functionalization of gold nanoparticles as a drug delivery vehicle. Int J Nanomedicine. 2020;15:9823–57. doi: 10.2147/IJN.S279094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang L, Xu J, Yan Y, Liu H, Karunakaran T, Li F. Green synthesis of gold nanoparticles from Scutellaria barbata and its anticancer activity in pancreatic cancer cell (PANC-1) Artif Cells Nanomed Biotechnol. 2019;47:1617–27. doi: 10.1080/21691401.2019.1594862. [DOI] [PubMed] [Google Scholar]
- 28.Scimeca M, Bischetti S, Lamsira HK, Bonfiglio R, Bonanno E. Energy Dispersive X-ray (EDX) microanalysis: A powerful tool in biomedical research and diagnosis. Eur J Histochem. 2018;62:2841.. doi: 10.4081/ejh.2018.2841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kumar SA, Peter YA, Nadeau JL. Facile biosynthesis, separation and conjugation of gold nanoparticles to doxorubicin. Nanotechnology. 2008;19:495101.. doi: 10.1088/0957-4484/19/49/495101. [DOI] [PubMed] [Google Scholar]
- 30.Geetha R, Ashokkumar T, Tamilselvan S, Govindaraju K, Sadiq M, Singaravelu G. Green synthesis of gold nanoparticles and their anticancer activity. Cancer Nanotechnol. 2013;4:91–8. doi: 10.1007/s12645-013-0040-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mi XJ, Park HR, Dhandapani S, Lee S, Kim YJ. Biologically synthesis of gold nanoparticles using Cirsium japonicum var. maackii extract and the study of anti-cancer properties on AGS gastric cancer cells. Int J Biol Sci. 2022;18:5809–26. doi: 10.7150/ijbs.77734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chamcheu JC, Rady I, Chamcheu RN, Siddique AB, Bloch MB, Banang Mbeumi S, et al. Graviola (Annona muricata) exerts anti-proliferative, anti-clonogenic and pro-apoptotic effects in human non-melanoma skin cancer UW-BCC1 and A431 cells in vitro: Involvement of hedgehog signaling. Int J Mol Sci. 2018;19:1791.. doi: 10.3390/ijms19061791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kim JY, Dao TT, Song K, Park SB, Jang H, Park MK, et al. Annona muricata leaf extract triggered intrinsic apoptotic pathway to attenuate cancerous features of triple negative breast cancer MDA-MB-231 Cells. Evid Based Complement Alternat Med. 2018;2018:7972916.. doi: 10.1155/2018/7972916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Asare GA, Afriyie D, Ngala RA, Abutiate H, Doku D, Mahmood SA, et al. Antiproliferative activity of aqueous leaf extract of Annona muricata L. On the prostate, BPH–1 cells, and some target genes. Integr Cancer Ther. 2015;14:65–74. doi: 10.1177/1534735414550198. [DOI] [PubMed] [Google Scholar]
- 35.Syed Najmuddin SU, Romli MF, Hamid M, Alitheen NB, Nik Abd Rahman NM. Anti-cancer effect of Annona Muricata Linn Leaves Crude Extract (AMCE) on breast cancer cell line. BMC Complement Altern Med. 2016;16:311.. doi: 10.1186/s12906-016-1290-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Torres MP, Rachagani S, Purohit V, Pandey P, Joshi S, Moore ED, et al. Graviola: A novel promising natural-derived drug that inhibits tumorigenicity and metastasis of pancreatic cancer cells in vitro and in vivo through altering cell metabolism. Cancer Lett. 2012;323:29–40. doi: 10.1016/j.canlet.2012.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Elmore S. Apoptosis: A review of programmed cell death. Toxicol Pathol. 2007;35:495–516. doi: 10.1080/01926230701320337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jan R, Chaudhry GE. Understanding apoptosis and apoptotic pathways targeted cancer therapeutics. Adv Pharm Bull. 2019;9:205–18. doi: 10.15171/apb.2019.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wong RS. Apoptosis in cancer: From pathogenesis to treatment. J Exp Clin Cancer Res. 2011;30:87.. doi: 10.1186/1756-9966-30-87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Shen Y, White E. p53-dependent apoptosis pathways. Adv Cancer Res. 2001;82:55–84. doi: 10.1016/s0065-230x(01)82002-9. [DOI] [PubMed] [Google Scholar]
- 41.Baharara J, Ramezani T, Divsalar A, Mousavi M, Seyedarabi A. Induction of apoptosis by green synthesized gold nanoparticles through activation of caspase-3 and 9 in human cervical cancer cells. Avicenna J Med Biotechnol. 2016;8:75–83. [PMC free article] [PubMed] [Google Scholar]
- 42.Ekins S, Mestres J, Testa B. In silico pharmacology for drug discovery: Methods for virtual ligand screening and profiling. Br J Pharmacol. 2007;152:9–20. doi: 10.1038/sj.bjp.0707305. [DOI] [PMC free article] [PubMed] [Google Scholar]


