Abstract
心肺疾病的发病率和病死率较高,已成为危害人们身心健康的主要疾病。经典名方当归补血汤组方精简,由黄芪和当归按5꞉1剂量组成,为气血双补之方剂,兼具通络之功效,其主要活性成分多糖类、皂苷类、黄酮类、挥发油等,具有良好的抗氧化、调节炎症、调节免疫和促进血液循环等作用。当归补血汤可改善心肌缺血再灌注损伤和心力衰竭,缓解糖尿病心肌病症状,抑制肺纤维化,保护肺损伤以及抗肺癌等多种心肺疾病。基础实验表明该方剂治疗心肺疾病的机制可能为通过多靶点通路发挥减轻炎症反应、抗氧化、调节自噬、增强免疫功能等作用。阐述当归补血汤的主要有效成分、药理作用及机制、临床应用,有助于为此方防治心肺疾病的临床应用提供更为全面的参考。
Keywords: 当归补血汤, 心肺疾病, 活性成分, 药理作用, 临床应用
Abstract
Cardiovascular and pulmonary diseases have become the major disease endangering people’s physical and mental health because of its high morbidity and mortality. Danggui Buxue Decoction, a noteworthy classical prescription, is composed of astragali radix and angelicae sinensis radix at a ratio of 5꞉1. It is a prescription for tonifying both vital energy and blood and has the effect of dredging collaterals. Its main active components, such as polysaccharides, saponins, flavonoids and volatile oils, have good effects on antioxidation, inflammation regulation, immune regulation and promotion of blood circulation. Danggui Buxue Decoction can improve myocardial ischemia-reperfusion injury and heart failure, relieve the symptoms of diabetic cardiomyopathy, inhibit pulmonary fibrosis, protect lung injury and fight against lung cancer and other cardiopulmonary diseases. Preclinical studies have showed that this prescription can inhibit inflammation and oxidative stress, regulate autophagy and enhance immune function through multi-target pathways. Reviewing the main effective components, pharmacological action and mechanism, and clinical application of Danggui Buxue Decoction are helpful to provide comprehensive information for the clinical application of Danggui Buxue Decoction in prevention and treatment of cardiovascular and pulmonary diseases.
Keywords: Danggui Buxue Decoction, cardiovascular and pulmonary diseases, active ingredients, pharmacological effect, clinical application
据世界卫生组织统计,在非传染性疾病中,以较高发病率、致残率、复发率和病死率为主要特征的心肺疾病严重危害人类身心健康[1]。心肺疾病具有生理结构和功能的潜在联系,且常存在共同的风险因素,如吸烟、喝酒、熬夜等[2]。随着工业化、城市化、人口老龄化进程的加快以及人们不良生活方式等危险因素的持续加剧,我国心肺疾病的患者人数呈上升趋势。目前,主要采用西药或手术予以治疗,但存在出血、肝肾功能损伤等不良反应,同时患者也承担着巨大经济压力[3]。中药以其多成分、多靶点、不良反应少、疗效好、医疗成本低等优势[4],逐渐得到患者的认可和青睐。中医理论将心肺疾病归纳于“胸痹、心痛、水肿”和“咳嗽、咯血、肺痈、肺痿”等辨治范畴。中药可以有效改善心肺功能,调节心肌能量代谢、微循环和血脂等,从而显著改善患者临床症状,提高生活质量。
当归补血汤(Danggui Buxue Decoction,DBD)是常用的气血双补方剂,最早记录在《内外伤辨惑论》一书中,为金元四大家之一李东垣创作。DBD由黄芪和当归两味中药按5꞉1的比例配伍组成,黄芪30 g、当归6 g,加8倍水,浸泡30 min,武火煮沸30 min,文火煎煮60 min后过滤,即得当归补血汤,常用于治疗血虚发热证。此方重用黄芪,补气固表,以急固浮阳而使热退,且补气又助生血,使阳生阴长,气旺血生,又配少量当归,养血和营,并得黄芪生血之助,使阳血渐充,则浮阳秘敛,虚热自退。研究[5]表明:DBD及其活性成分在抗炎、增强免疫、保护心血管、保护肺损伤、抗肿瘤等方面可发挥至关重要的作用。由于兼具药味简单、活性成分复杂、多种临床功效显著的特点,DBD已成为近年来国内外学者的研究热点[6]。本文旨在对DBD的活性成分、在心肺疾病中的作用与机制以及临床应用等进行总结,以期为DBD防治心肺疾病的临床应用提供更为全面的参考。
1. DBD的主要活性成分
目前对于DBD整体方剂化学成分研究较少,主要集中在对该方单味药的化学成分,而活性化合物是中药复方发挥药理作用的重要物质基础。笔者对DBD中黄芪和当归的主要化学成分进行整理(表1、图1)。
表1.
当归补血汤主要活性成分
Table 1 Active compounds of Danggui Buxue Decoction
| 化学名称 | 分子式 | CAS号 | 分子质量 | 来源 |
|---|---|---|---|---|
| 黄芪皂苷I(Astragaloside I) | C45H72O16 | 84680-75-1 | 840.45 | 黄芪 |
| 黄芪皂苷II(Astragaloside II) | C43H70O15 | 84676-89-1 | 826.47 | 黄芪 |
| 黄芪甲苷(Astragaloside IV) | C41H68O14 | 83207-58-3 | 784.46 | 黄芪 |
| 毛蕊异黄酮(Calycosin) | C16H12O5 | 20575-57-9 | 284.07 | 黄芪 |
| 毛蕊异黄酮苷(Calycosin 7-O-glucoside) | C22H22O10 | 20633-67-4 | 446.41 | 黄芪 |
| 芒柄花苷(Ononin) | C22H22O9 | 486-62-4 | 430.13 | 黄芪 |
| 芒柄花素(Formononetin) | C16H12O4 | 485-72-3 | 268.07 | 黄芪 |
| 藁本内酯(Ligustilide) | C12H14O2 | 4431-01-0 | 190.10 | 当归 |
| 丁烯基苯酞(N-butylidenephthalide) | C12H12O2 | 551-08-6 | 188.22 | 当归 |
| 新蛇床内酯(Neocnidilide) | C12H18O2 | 6415-59-4 | 194.13 | 当归 |
| 正丁基苯酞(3-N-butylphthalide) | C12H14O2 | 6066-49-5 | 190.10 | 当归 |
| 阿魏酸(Ferulic acid) | C10H10O4 | 1135-24-6 | 194.06 | 当归 |
| 原儿茶酸(Protocatechuic acid) | C7H6O4 | 99-50-3 | 154.03 | 当归 |
| 咖啡酸(Caffeic acid) | C9H8O4 | 331-39-5 | 180.04 | 当归 |
图1.
黄芪(A)和当归(B)的主要成分
Figure 1 Main components of astragalus (A) and angelica (B)
1.1. 黄芪
黄芪为豆科植物蒙古黄芪或膜荚黄芪的干燥根,主产于山西、甘肃、内蒙古,甘、微温之品,归脾、肺经,具有补气升阳、固表、利水、消肿、养阴、行滞通痹的功效,为补脾益肺之要药[7]。研究[8]表明:黄芪蕴含三萜皂苷(黄芪皂苷I-VIII、异黄芪甲苷I和II、大豆皂苷I等)、黄酮类(芒柄花素、毛蕊异黄酮葡萄糖苷、毛蕊异黄酮等)、多糖、氨基酸和苯丙素等多种化学成分,可通过抑制炎症反应、细胞凋亡、心肌细胞和肺上皮细胞纤维化等多种途径改善心功能,防治肺损伤。
1.1.1. 黄芪多糖
黄芪多糖(astragalus polysaccharides,APS)是膜荚属植物中最重要的天然活性成分,主要由杂多糖、葡聚糖、酸性水溶性多糖等成分组成。其中葡聚糖分为水溶性和水不溶性,分别以α(1→4)(1→6)葡聚糖和α(1→4)葡聚糖命名,而杂多糖则多为水溶性酸性杂多糖,主要由L-鼠李糖、L-核糖、D-木糖、L-木糖、D-核糖、D-半乳糖和D-葡萄糖组成,其具有调节免疫、抗衰老、抗炎、抗氧化等多种生物学效应[9]。APS可调节Toll样受体4/核转录因子-κB p65(Toll-like receptor 4/nuclear factor-κB p65,TLR-4/NF-κB p65)信号通路,减轻炎症反应,从而改善病毒性心肌炎的症状[10],改善肺组织病理结构,减少中性粒细胞浸润,抑制肺部炎症反应[11]。
1.1.2. 黄芪皂苷
黄芪皂苷主要成分包括黄芪甲苷、异黄芪皂苷、乙酰基黄芪皂苷、大豆皂苷等,其中黄芪甲苷含量最丰富,具有抗炎、抗纤维化、抗氧化、抗肿瘤、调节免疫、调节代谢等作用[12]。黄芪甲苷能减轻心肌损伤、心肌细胞肥大,抑制心肌纤维化和炎症反应[13]。空气中可吸入细颗粒物PM2.5可促进炎症因子渗出,引起肺上皮细胞自噬和凋亡加剧,进而加重肺部损害,黄芪甲苷能够激活腺苷酸活化蛋白激酶/哺乳动物雷帕霉素靶蛋白(denosine monophosphate-activated protein kinase/mammalian target of rapamycin,AMPK/mTOR)信号通路预防PM2.5诱导的肺毒性[14]。
1.1.3. 黄酮类
黄芪所含的黄酮类化合物主要包括异黄酮类、异黄烷类、紫檀烷类等,主要有毛蕊异黄酮(calycoin,CA)、毛蕊异黄酮苷、毛蕊花素、芒柄花素、毛蕊花素-7-O-β-d-葡萄糖苷和芒柄花苷等化合物,能够透过血脑屏障,具有疏通微循环、扩张毛细血管、保护血管等多种生物学效应[15]。CA可通过抑制炎症和调控转化生长因子-β受体1(transforming growth factor-β receptor 1,TGFBRl)信号通路改善心功能,缓解心室重构,延缓心肌梗死(myocardial infarction,MI)的进展[16]。研究[17]表明CA可减轻肺组织炎症反应,改善肺功能,其机制与CA抑制脓毒症急性肺损伤大鼠肺组织中HMGB1/MyD88/NF-κB通路和NOD样受体热蛋白结构域相关蛋白-3(NOD-like receptor pyrin domain-related protein 3,NLRP3)炎症小体激活相关。
1.2. 当归
当归为伞形科植物当归Angelica sinensis (Oliv.) Diels的干燥根,主产于甘肃,甘、辛、温之品,归肝、心、脾三经,具有补血活血、调经止痛、润肠通便的功效,长于补血,为补血之圣药[7]。当归蕴含多种化学成分,如挥发性油、香豆素类、有机酸类、多糖和黄酮类等[18],对心肌缺血、急性肺损伤等具有良好的防治作用,机制可能与其抗氧化、抗炎、抗纤维化、调节免疫等有关。
1.2.1. 挥发油
当归属植物含有丰富的挥发油,主要包含醇、酮、内酯、苷等芳香化合物和脂肪烃及其衍生物等成分。而内酯类化合物,是当归挥发油的主要活性成分,主要由洋川芎内酯(A、D、H、I)、Z-藁本内酯、新蛇床内酯、Z-丁烯基苯酞、正丁基苯酞等活性成分组成,其中Z-藁本内酯的含量最高,具有抗炎、抗氧化、镇痛等作用[18]。藁本内酯能通过减轻H9C2细胞过度氧化损伤、炎症和纤维化改善心肌细胞功能障碍[19]。此外,藁本内酯还可以减少肺组织成纤维细胞数量,减轻氧化应激和细胞凋亡,维持Th1/Th2平衡,从而减轻肺组织纤维化[20]。
1.2.2. 有机酸类
当归中的有机酸类化合物主要由阿魏酸、绿原酸、邻苯二甲酸、咖啡酸、烟酸、香草酸、亚油酸等成分组成,其中阿魏酸的含量最高,具有抗氧化、抗炎、舒张血管、抗血栓等作用[21]。阿魏酸通过降低氧化应激和抑制心肌细胞凋亡,通过激活核因子E2相关因子2(nuclear factor E2-related factor 2,Nrf2)通路改善心力衰竭[22]。阿魏酸还可改善小鼠肺组织病理结构和补体沉积,抑制肺水肿,降低促炎细胞因子的产生,对急性肺损伤产生显著保护作用[23]。
1.2.3. 当归多糖
当归多糖(angelica sinensis polysaccharide,ASP)主要由葡萄糖、岩藻糖、木糖、鼠李糖、半乳糖、阿拉伯糖和半乳糖醛酸组成,具有造血、促进免疫、抗肿瘤、抗炎、抗氧化、抗衰老等作用[24]。ASP能通过减轻糖尿病心肌缺血再灌注大鼠的氧化应激以及炎症反应,上调血管内皮生长因子A(vascular endothelial growth factor A,VEGFA)表达和抑制caspase-3活化来抑制心肌细胞凋亡,达到保护心脏的作用[25];抑制大鼠肺纤维化和肺泡II型上皮细胞的纤维化,减轻急性肺损伤[26]。
综上,DBD中主要活性成分对心肺疾病具有重要的调节作用,活性成分的药理作用及其机制总结见表2 [13-14, 16-17, 19-20, 22-23, 25-42]。
表2.
活性成分用于心肺疾病的药理作用及机制
Table 2 Pharmacological effects and mechanisms of active ingredients for cardiovascular and pulmonary diseases
| 成分 | 实验动物/细胞 | 给药剂量及给药方式 | 生物活性及机制 |
|---|---|---|---|
| 黄芪皂苷I | 雄性SD大鼠 | 40、80、160 mg/kg,灌胃15 d | 降低炎症因子水平,抑制细胞凋亡,降低Bax/Bcl-2比值,抑制PI3K/AKT/mTOR通路减轻病毒性心肌炎心肌损害[27] |
| 黄芪甲苷 |
雄性C57BL/6 小鼠 |
40 mg/kg,灌胃4周 | 下调ROS水平、NLRP3/caspase-1/GSDMD蛋白表达,减轻心肌梗死引起的心肌纤维化和心脏重构[13] |
| 黄芪甲苷 | 雄性SD大鼠 |
50、100 mg/kg, 腹腔注射3 d |
下调GM-CSF、ICAM-1、IL-6等含量及Bax、caspase-3、PARP1和NF-κB蛋白表达,上调IL-2,IL-4和IL-10含量及Bcl-2蛋白,改善炎症,减轻自噬,改善PM2.5导致的急性肺毒性[14] |
| 毛蕊异黄酮 |
雄性C57/BL 小鼠 |
25、50 mg/kg, 灌胃4周 |
抑制TGFBR1、Smad2/3、I型胶原和III型胶原的表达及CFs的增殖和胶原沉积,减弱心肌梗死后的心肌纤维化和心室功能 障碍[16] |
| 毛蕊异黄酮 | 雄性SD大鼠 | 12.5、25、50 mg/kg,灌胃7 d | 抑制HMGB1/MyD88/NF-κB通路和NLRP3炎症小体,减轻 肺水肿[17] |
| 毛蕊异黄酮苷 |
雄性C57BL/6 小鼠 |
30 mg/kg, 尾静脉注射 |
上调IL-10、p-JAK2、p-STAT3,激活JAK2/STAT3信号通路,减轻心肌缺血/再灌注损伤[28] |
| 芒柄花素 | 雄性SD大鼠 |
10、30 mg/kg, 腹腔注射 |
下调TNF-α,IL-1β、IL-6,上调Bcl-2/Bax比值,抑制TXNIP/NLRP3炎症小体通路,减轻心肌缺血/再灌注损伤[29] |
| 芒柄花素 |
雌性BALB/c 小鼠 |
10、20、40 mg/kg, 灌胃 |
下调IL-4、IL-5、IL-13、IL-17A、IgE、CCL5、CCL11的表达,上调Nrf2/HO-1,减轻过敏性哮喘[30] |
| 藁本内酯 | H9C2细胞 | 2~100 μmol/L | 上调Bax和Bcl-2蛋白表达,提高SOD、GSH的水平,通过AMPK/GSK-3β/Nrf2途径调节细胞氧化损伤、炎症和纤维化[19] |
| 藁本内酯 | 雄性SD大鼠 | 20、40、80 mg/kg,灌胃4周 | 减少TGF-β、α-SMA的表达,减少纤维化;下调LDH、SOD、GSH、MDA,抑制细胞凋亡;下调Bax、Bcl-2、p65蛋白表达,减少凋亡[20] |
| α-蒎烯 | 雄性Wistar大鼠 | 25、50、100 mg/kg,灌胃7 d | 降低CK、CK-MB活性和TBARS,下调TNF-α、IL-6、NF-κB的表达,抑制氧化应激和改善炎症,延缓心肌梗死进程[31] |
| 阿魏酸 | 雄性SD大鼠 |
5、25、50 mg/kg, 灌胃4 d |
上调SOD和GSH-Px活性,下调MDA、LDH、caspase-3、caspase-9含量,激活Nrf2信号通路,抑制心肌细胞凋亡, 减轻心力衰竭[22] |
| 阿魏酸 |
雌性BALB/c 小鼠 |
25、50、100 mg/kg,灌胃3 d |
下调MPO活性和MCP-1水平及TLR4、p-p65和p-IκBα的蛋白表达,降低TNF-α,IL-1β和IL-6的含量,抑制炎症反应, 减轻急性肺损伤[23] |
| 原儿茶酸 | 雄性Wistar大鼠 |
100、200 mg/kg, 灌胃14 d |
上调GSH、SOD和CAT,下调MDA、NO、TNF-α、IL-1β,Bax、caspase-3等,抑制炎症介质、凋亡和纤维化[32] |
| 原儿茶酸 | 雄性昆明小鼠 |
5、15、30 mg/kg, 腹腔注射 |
下调IL-1β、TNF-α、p38-MAPK蛋白水平,抑制HMGB1/MyD88/NF-κB途径和NLRP3炎症小体激活,改善炎症, 减轻急性肺损伤[33] |
| 绿原酸 | 雄性Wistar大鼠 | 40 mg/kg,灌胃19 d | 上调ICDH、SDH和MDH等的活性,下调ROS的表达,降低 氧化应激,改善心肌梗死症状[34] |
| 绿原酸 |
雄性BALB/c 小鼠 |
50、100、150 mg/kg,灌胃7 d | 抑制STAT3、AKT蛋白的活化及KAT2A的表达,从而抑制炎症反应[35] |
| 香草酸 | 雄性Wistar大鼠 | 5、10 mg/kg,灌胃10 d | 下调细胞凋亡上游信号Fas受体基因的表达,下调caspase-8、9和3以及caspase介导的凋亡通路表达,减少细胞凋亡[36] |
| 香草酸 | 雄性Swiss albino小鼠 | 200 mg/kg,灌胃给药16周,隔日1次 | 下调CEA和NSE的表达水平,上调SOD、CAT、GSH蛋白含量,抗氧化活性[37] |
| 成分 | 实验动物/细胞 | 给药剂量及给药方式 | 生物活性及机制 |
|---|---|---|---|
| 咖啡酸 | H9C2细胞 | 0.5、1.0、2.0、4.0、8.0 μmol/L | 上调Na+-K+-ATP酶、Ca2+-ATP酶的活性,下调ROS的表达,p65蛋白的表达量,LDH的含量,减轻氧化应激[38] |
| 咖啡酸 | 雄性昆明小鼠 | 15、30、60 mg/kg,腹腔注射 | 下调TNF-α、IL-6、IL-1β、Pro-/cleased-caspase1、Pro-/mature-IL-1β、Bax、caspase-3、caspase-9、p53,抑制NLRP3炎症小体活化,抑制NF-κB、MAPKs、NLRP3炎症小体通路的激活,减轻炎症反应[39] |
| 壬二酸 | H9C2细胞 | 10 μmol/L | 下调p62蛋白、BNIP3蛋白,上调LC3-II/LC3-I比值,抑制BNIP3介导的线粒体自噬和H9C2心肌细胞凋亡,增强细胞活力、减轻心肌纤维化[40] |
| 黄芪多糖 | 雄性C57BL/6小鼠 |
200 mg/kg,灌胃 给药2周 |
下调IL-1β、IL-6、TNF-α、INF-γ、MCP-1、TLR-4和磷酸化NF-κB p65的表达,抑制TLR-4/NF-κB通路的激活,减轻心肌炎症 损伤[10] |
| 黄芪多糖 | 雄性C57BL/6小鼠 | 200 mg/kg APS灌胃14 d | 下调IL-1β、IL-6、TNF-α、MPO、p65,抑制NF-κB信号通路和炎症因子渗出,减轻急性肺损伤[11] |
| 当归多糖 | 雄性SD大鼠 |
10 mg/kg,尾静脉 注射 |
下调TNF-α、IL-6、caspase-3,上调SOD、VEGFA,减轻氧化 应激、炎症反应及抗凋亡作用达到抗糖尿病心肌缺血再灌注 损伤[25] |
| 当归多糖 | 雄性SD大鼠 | 20 mg/kg,灌胃给药14 d | 下调DANCR、FOXO3,抑制大鼠肺纤维化和肺泡II型上皮细胞的纤维化[26] |
| 洋川芎内酯I | 雄性C57BL/6小鼠 | 36、200 mg/kg, 腹腔注射 |
降低TNF-α、IL-1β、IL-6、MDA以及MAPK和NF-κB激酶的 磷酸化水平,改善脓毒症急性肺损伤肺组织炎症和病理损害[41] |
| 正丁基苯酞 | 雄性C57BL/6小鼠 | 10、50 mg/kg, 尾静脉注射 |
通过激活SIRT1/Nrf2通路发挥抗氧化应激、抗炎作用,减轻 肺水肿[42] |
Bcl-2:B细胞淋巴瘤蛋白-2;Bax:Bcl-2相关X蛋白;PI3K:磷脂酰肌醇-3-激酶;AKT:蛋白激酶B;mTOR:哺乳动物雷帕霉素靶蛋白;ROS:活性氧;NLRP3:NOD样受体热蛋白结构域相关蛋白-3;caspase:天冬氨酸蛋白水解酶;GSDMD:Gasdermin D蛋白;GM-CSF:粒细胞-巨噬细胞集落刺激因子;ICAM-1:细胞间黏附分子-1;PARP1:多聚ADP核糖聚合酶1;NF-κB:核因子κB;TGFBR1:转化生长因子Β受体I;HMGB1:高迁移率族蛋白B1;MyD88:髓样分化因子;JAK2:两面神激酶;STAT3:重组人信号转导与转录激活因子3;TNF-α:肿瘤坏死因子α;TXNIP:硫氧还蛋白结合蛋白;IgE:免疫球蛋白E;CCL:趋化因子配体;Nrf2:核因子E2相关因子2;HO-1:血红素氧合酶1;SOD:超氧化物歧化酶;GSH:谷胱甘肽;AMPK:腺苷酸活化蛋白激酶;GSK-3β:糖原合成酶激酶3β;TGF-β:转化生长因子β; α-SMA:α-平滑肌肌动蛋白;LDH:乳酸脱氢酶;MDA:丙二醛;CK:肌酸激酶;CK-MB:肌酸激酶同工酶;TBARS:硫代巴比妥酸反应物质;MPO:髓过氧化物酶;MCP-1:单核细胞趋化蛋白-1;TLR-4:Toll样受体4;CAT:过氧化氢酶;NO:一氧化氮;ICDH:异柠檬酸脱氢酶;SDH:琥珀酸脱氢酶;MDH:小肠苹果酸脱氢酶;CEA:癌胚抗原;NSE:神经元特异性烯醇化酶;MAPK:丝裂原活化蛋白激酶;VEGFA:血管内皮生长因子A;BNIP3:BCL2相互作用蛋白3;LC3:微管相关蛋白1轻链3;DANCR:抗分化非编码RNA;FOXO3:叉头框蛋白O3;SIRT1:沉默信息调节因子1。
2. DBD药效物质基础和药代动力学
2.1. 药效物质基础
DBD化学成分较为复杂,且只有被人体吸收的成分才能起到治疗的作用,成为药效物质基础。Liu等[43]采用代谢组学和脂质组学的超高效液相色谱-四极杆-高效质谱的方法,分析得出DBD对嘧啶代谢和甘油磷脂代谢的调节主要与DBD的抗贫血作用有关。Li等[44]采用液相色谱-飞行时间质谱联用技术方法,研究DBD口服在大鼠胆汁和血浆中的代谢产物,发现大鼠胆汁中含有比尿液中更多的谷胱甘肽衍生的共轭物和皂苷化合物。研究[45]发现DBD及其敲除阿魏酸后都能刺激成骨细胞分化,而敲除CA后不能刺激MAPK/E成骨细胞分化,说明CA在DBD中起到协同多组分的作用。
2.2. 药代动力学
药代动力学(pharmacokinetics,PK)通过了解药物在机体内的浓度变化来分析影响药物吸收、分配、代谢和排泄过程的因素,是一种确定有效成分和发现质量控制标志的有效方法。研究[46]发现:与当归单煎液比较,给予兔DBD灌胃后,阿魏酸的达峰时间、曲线下面积、体内平均滞留时间均有变化,这表明DBD能使阿魏酸的达峰时间提前,生物利用度增加。Zhao等[47]研究了DBD中4种主要化学成分(阿魏酸、芒柄花素、芒柄花苷和毛蕊花素-7-O-葡萄糖苷)在血虚条件下,口服后不同时间点的血药浓度,结果表明阿魏酸的浓度在给药后15 min达到峰值,其最高浓度和曲线下面积最高,芒柄花素、芒柄花苷和毛蕊异黄酮-7-O-葡萄糖苷在心脏中含量最高,15 min达到峰值。Shi等[48]采用超高效液相色谱-三重四极杆质谱联用仪测定DBD中7种主要活性成分(阿魏酸、咖啡酸、丁苯酞、藁本内酯、毛蕊异黄酮-7-O-β-葡萄糖苷、毛蕊花甲苷和黄芪甲苷)的浓度,结果表明模型组的这7种活性成分浓度均小于或等于空白组,说明正常大鼠和血虚大鼠在某些PK特征上存在明显差异,表示血虚大鼠药物代谢的速度和程度都发生了改变。
3. DBD在心肺疾病中的保护作用
3.1. 对心血管系统的保护作用
3.1.1. 改善心肌缺血损伤
MI是因暂时或永久性冠状动脉闭塞,导致负性心肌重构、收缩功能障碍、心律失常,甚至心力衰竭的一种严重的心血管疾病。DBD可通过抑制心肌细胞凋亡、提高心肌细胞对氧化应激损伤的抵抗力、促进线粒体自噬改善线粒体功能,从而改善慢性间歇性低氧小鼠心功能。研究[49]通过左前降支冠状动脉结扎术结扎小鼠,构建小鼠MI模型,以灌胃的方式给予小鼠DBD,发现DBD通过上调沉默信息调节因子1(silent information regulator sirtuin 1,SIRT1)表达,发挥抗MI后心室重塑作用。还有研究[50]发现DBD能增强心肌抗凋亡蛋白B细胞淋巴瘤蛋白-2(B-cell lymphoma protein 2,Bcl-2)的表达,减少促凋亡蛋白Bcl-2相关X蛋白(Bcl-2 associated X protein,BAX)、caspase-3和caspase-9的表达,达到改善冠状动脉结扎诱导的大鼠缺血心肌细胞凋亡的作用。
3.1.2. 抗心肌缺血-再灌注损伤
缺血是由组织或器官局部血流减少引起,由于负责心肌灌注的冠状动脉闭塞,心肌通常会出现缺血。心肌缺血-再灌注损伤(myocardial ischemia reperfusion injury,MIRI)是指在急性冠状动脉闭塞后,再灌注所导致的心肌损伤较缺血本身更为严重,包括缺血诱导和再灌注2种损伤。吴伟等[51]通过结扎大鼠左降前支致缺血30 min,然后再灌注120 min构建MIRI模型,以10 mL/kg的DBD灌胃给药,发现DBD可通过抑制炎症因子的释放并改善心肌缺血等作用,达到对MIRI心肌的保护。王时光等[52]通过构建MIRI模型,发现DBD通过降低血清中丙二醛(malondialdehyde,MDA)的含量,恢复过氧化氢酶和谷胱甘肽过氧化物酶(glutathione peroxidase,GPx)的活力,使得心肌病理学变化减轻,炎症细胞浸润减少,调节氧化应激相关细胞因子含量及蛋白质表达水平,对大鼠心肌缺血有保护作用。
3.1.3. 改善糖尿病心肌病
糖尿病心肌病(diabetic cardiomyopathy,DCM)是糖尿病引起的可导致心力衰竭的病理生理状态,主要与胰岛素抵抗、高胰岛素血症和高血糖症密切相关,而与冠心病和高血压等其他心脏危险因素无关。DCM的致病因素有氧化应激、线粒体功能障碍、晚期糖基化终末产物的产生、炎症、钙稳态损害等。DBD可通过促进线粒体自噬,抑制细胞凋亡,抑制大鼠内质网应激反应,平衡心肌细胞内钙离子稳态环境,从而显著缓解DCM大鼠心室重构,减轻心肌损伤,改善心功能[53-54]。
3.1.4. 抗心力衰竭
心脏无法为外周组织提供所需的血液和氧气以满足其代谢需求,这种情况在临床上常常表现为呼吸困难或疲劳,以及颈静脉压升高、心动过速或周围水肿等,通常是由于潜在的心肌疾病引起的心力衰竭。DBD是中医临床治疗慢性心力衰竭的常用方剂,其能有效地提高体内促红细胞生成素的水平,改善造血干细胞功能和心肌功能。蒲秀瑛等[55]采用不同浓度的阿霉素(随时间依次递增)腹腔注射,构建大鼠心力衰竭模型,发现DBD通过降低慢性心力衰竭大鼠心肌组织中内皮素和提高降钙素基因相关肽,改善慢性心力衰竭大鼠机体免疫功能,达到防治慢性心力衰竭的目的。
3.2. 对肺疾病的保护作用
3.2.1. 抑制肺纤维化
肺纤维化(pulmonary fibrosis,PF)是一种慢性进行性肺疾病,其病因是纤维化导致肺泡壁增厚,使气体交换减弱,通气受限,从而使呼吸衰竭。PF的病理生理过程包括炎症反应、效应细胞激活和迁移以及细胞外基质过度沉积,从而导致肺功能受损[56]。Zhao等[57]发现DBD总苷(total glucosides of DBD,TGD)可能通过抑制NADPH氧化酶4(NADPH oxidase 4,NOX4)来控制氧化应激水平,从而减轻博莱霉素所致大鼠PF程度。研究[58]发现DBD可通过调节PKD1/NF-κB/MnSOD线粒体核抗氧化通路,提高机体抗氧化能力,从而减轻纤维化程度。
3.2.2. 对放射性肺损伤的保护作用
癌症患者在接受胸部放射治疗(以下简称“放疗”)时,可能会产生放射性肺损伤(radiation-induced lung injury,RILI),可分为早期放射性肺炎和晚期放射性肺纤维化。早期RILI通常为短期,其病理特征为肺泡液渗出,肺泡壁充血水肿,肺泡膜损伤等,表现为磨玻璃样混浊,而晚期RILI发生于放射后6~24个月,其病理特征为肺泡间隔增厚,肺泡壁或间质纤维化[59]。研究[60]采用辐射大鼠,构建急性RILI模型,以低、中高剂量DBD灌胃,发现DBD能抑制大鼠肺NF-κB p65和内皮缩血管肽(endothelin,ET-1)的表达而防护RILI。
3.2.3. 抗肺癌活性
肺癌严重威胁人类的健康,主要包括小细胞肺癌(small-cell lung cancer,SCLC)和非小细胞肺癌(non-small-cell lung cancer,NSCLC),而NSCLC是最常见的肺癌类型,其症状有咳嗽、咯血、胸痛和呼吸困难等[61]。目前,治疗肺癌主要采用放疗的手段,但在杀死癌细胞的同时也对人体正常细胞造成了伤害,引起骨髓抑制,使机体免疫功能降低,而中医药治疗放疗引起的骨髓抑制具有较好的效果。研究[62]采用皮下接种A549细胞,以建立裸鼠异种移植模型,以3.6 g/kg的DBD联合吉西他滨灌胃,发现DBD可通过提高人类抗原R(human antigen R,HuR)、脱氧胞苷激酶(deoxycytidine kinase,dCK)和Nrf2的表达,使其对吉西他滨的敏感性增加,促进细胞周期转化和调节周期相关蛋白,缓解吉西他滨诱导的骨髓抑制。
4. DBD治疗心肺疾病的作用机制
4.1. DBD保护心血管机制
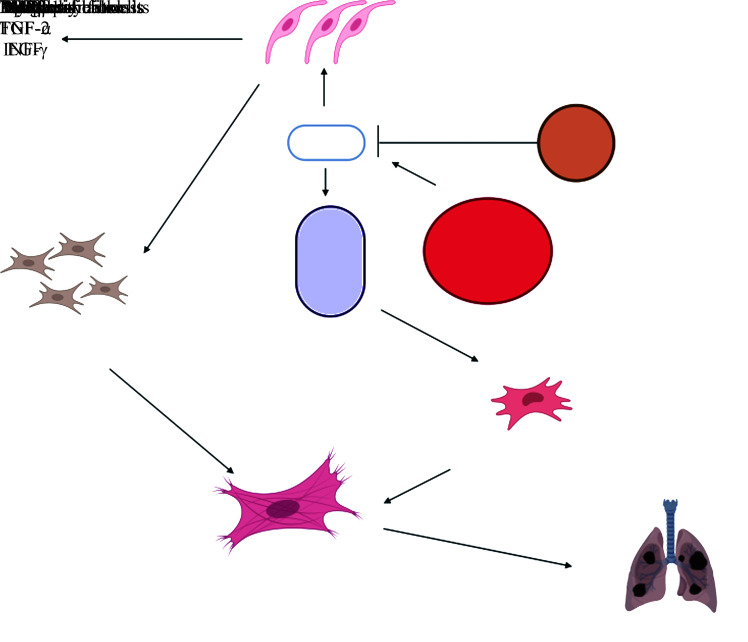
心血管疾病的发病机制较为复杂,其中包括氧化应激、钙超载、线粒体紊乱、细胞自噬、炎症等一系列反应[63]。对心血管疾病保护作用的分子机制包括抑制心肌细胞自噬、促进血管生成、抑制氧化应激和炎症反应及抗细胞凋亡等(图2)。
图2.
当归补血汤治疗心肌疾病的作用机制
Figure 2 Activity mechanism of Danggui Buexue Decoction (DBD) in the treatment of myocardial diseases
This figure was created with BioRender.com. LC3: Microtubule-associated protein l light chain 3; SOD: Super oxide dismutase; MDA: Malondialdehyde; Bax: Bcl-2-associated X protein; Caspase-3: Cysteinyl aspartate specific proteinase 3; Bcl-2: B-cell lymphoma protein-2.
4.1.1. 调控心肌细胞自噬
细胞自噬是自我降解的过程,其作用有清除错误折叠或聚集的蛋白质、细胞内病原体和受损的细胞器,此外还可促进细胞衰老和细胞表面抗原呈递,防止基因组不稳定和坏死,通常自噬被认为是一种生存机制[64]。研究[65]表明DBD可逆转MI诱导的自噬相关蛋白质表达异常,从而上调自噬水平、改善心肌损伤。有研究[66]发现DBD通过激活磷脂酰肌醇-3-激酶(phosphatidylinositol 3-kinase,PI3K)介导Akt/mTOR信号通路,调控心肌细胞自噬、减轻心肌细胞损伤,对阿霉素诱导的小鼠心肌损伤具有保护作用。
4.1.2. 促进血管生成
研究[67]发现DBD能提高血清中一氧化氮和VEGF的含量,从而促进衰老MI大鼠冠状动脉侧枝血管生成,改善大鼠的心功能。
4.1.3. 抑制氧化应激和炎症反应
氧化应激是指机体在面对各种风险因子的刺激时,其正常代谢产生的活性氧(reactive oxygen species,ROS)的生成速率大于清除速率,氧化产物MDA等增多,超氧化物歧化酶(superoxide dismutase,SOD)的含量减少,细胞内氧化物的产生与抗氧化作用不平衡,导致ROS在机体蓄积,最终损伤机体。研究[68]发现DBD能激活PI3K/Akt通路并抑制氧化应激反应及炎症反应,达到减轻老年MI大鼠的心肌损害,保护心脏的作用。
4.1.4. 抑制细胞凋亡
研究[69]表明DBD含药血清能通过促进间歇性低氧H9C2细胞的线粒体自噬,抑制细胞凋亡,改善心肌细胞损伤。还有研究[70]发现DBD通过抑制凋亡相关因子配体(factor associated suicide ligand,FasL)/肿瘤坏死因子-α(tumor necrosis factor-α,TNF-α)信号通路来抑制心肌细胞凋亡,从而改善放射性心肌损伤。
4.2. DBD对肺疾病的作用机制
4.2.1. 免疫调节作用
Sun等[71]构建NSCLC模型,以1.8 g/kg剂量的DBD灌胃12 d,发现DBD能上调dCK和下调P-gp蛋白的表达,且能提高小鼠血浆中IL-12和GM-CSF的表达,说明DBD对荷瘤小鼠具有潜在的免疫调节作用,可以用于治疗NSCLC。
4.2.2. 抑制氧化应激和炎症反应
研究[57]发现TGD可通过抑制NOX4减轻氧化应激损伤,延缓PF进展。此外,DBD能通过减少炎性细胞因子的分泌和胶原沉积,抑制TLR4/NLRP3信号通路,从而抑制PF[72]。DBD治疗PF的作用机制见图3。
图3.
当归补血汤治疗肺纤维化作用机制
Figure 3 Activity mechanism of Danggui Buexue Decoction (DBD) in the treatment of pulmonary fibrosis
This figure was created with BioRender.com. ROS: Robot operating system; TGF-β1: Transforming growth factor-β1; ER stress: Endoplasmic reticulum stress; FGF-2: Fibroblast growth factor 2; EGF: Epidermal growth factor; IL-1β: Interleukin-1β; TNF-α: Tumor necrosis factor-α; INF-γ: Interferon-γ; EMT: Epithelial-mesenchymal transition.
4.2.3. 抑制血管新生
研究[73]发现DBD可通过下调缺氧诱导因子,上调内皮抑素的表达,增加肺部供氧,抑制血管新生,改善肺小动脉血管状态。
5. DBD在心肺疾病中的临床应用
5.1. 在心血管疾病中的应用
研究[74]将120例冠心病患者随机分成对照组和观察组,对照组给予卡维地洛,观察组给予DBD联合卡维地洛,发现2组患者在治疗后均有好转,但观察组患者在心电图ST段压低次数、持续时间及心肌缺血总负荷下降比对照组明显;在左室舒张功能上,对照组治疗前后变化不明显,观察组下降明显,结果表明DBD联合卡维地洛能有效治疗冠心病患者,使患者早日恢复健康。栾海燕等[75]将80例急性MI患者随机平均分成对照组和观察组,对照组给予急性MI常规治疗,而观察组在对照组基础上联合使用DBD治疗,发现治疗后观察组降低左室后壁厚度和舒张末期室间隔厚度优于对照组,且观察组心血管不良事件的总发生率显著低于对照组,结果表明DBD能有效帮助患者心室重塑、改善心功能和减少心血管不良事件的发生。此外,DBD能明显改善DCM患者心功能,改善患者透析期的低血压,减轻心肌损害[54]。
5.2. 在肺疾病中的应用
研究[76]将进行放疗后的肺癌患者随机分为观察组和对照组,对照组给予常规治疗,观察组给予加味DBD(黄芪、鸡血藤、当归、薏苡仁、女贞子、枸杞子、太子参、山药、白术)治疗,结果表明采用加味DBD进行辅助治疗,能有效改善肺癌放疗后白细胞减少,提升治疗总有效率,疗效比常规治疗更显著。研究[77]将140例放、化疗后的肺癌患者随机分为对照组与实验组,对照组给予常规治疗,实验组在对照组治疗的基础上给予DBD辅助治疗,结果表明DBD能有效缓解患者骨髓抑制,改善其临床症状,提高患者的生存质量。
6. DBD的指纹图谱研究
目前DBD已被开发成各种剂型的中成药以满足不同的临床需要,例如当归补血口服液、当归补血浓缩丸、当归补血胶囊、当归补血颗粒等。DBD及其衍生物质量控制的优劣是临床疗效的基础。以合适的方法和质量控制标志物建立质量标准是质量控制的关键。DBD中仅由黄芪和当归两味中药配伍而成,组方相对简单,其内含有多种有效成分。DBD不同制剂主要采用薄层色谱法(thin layer chromatography,TLC)和高效液相色谱法(high performance liquid chromatography,HPLC)进行质量检测。当归补血丸采用HPLC在323 nm波长处建立了一种基于指纹的质量控制方法,并同时采用HPLC法测定其中阿魏酸、毛蕊异黄酮葡萄糖苷和黄芪甲苷的含量[78];采用TLC对当归补血颗粒组方药物当归有效成分藁本内酯进行鉴别,采用HPLC分别对组方药当归中的阿魏酸和黄芪中的有效成分黄芪甲苷进行含量测定[79]。采用反相HPLC法测定当归补血胶囊中阿魏酸的含量,采用薄层色谱鉴别法分别检测当归补血口服液组方药物当归和黄芪的有效成分蒿本内酯和黄芪苷含量。以上所确定的TLC鉴别和HPLC含量测定方法可对DBD及其衍生物的质量进行有效控制,为进一步制订质量标准奠定基础。
7. 结 语
近年来,随着人口老龄化的逐步加深,心肺疾病在中国的患病率逐年上升,其发病机制非常复杂,如果不能及时有效的治疗,可能会对患者造成相应的后遗症甚至死亡。中医药作为中国的传统医学,具有独特的理论和长期的临床实践,越来越多的人选择中医药治疗心肺疾病,且具有较好的疗效。DBD作为经典的中药配方,治疗血虚风燥已有一千多年的历史,能改善心肺疾病。但是,在DBD作为心肺疾病治疗药物的开发过程中还面临一些挑战,如DBD中有效成分研究不足,导致部分化合物的作用可能被忽略;DBD治疗心肺疾病的药效学和药理机制研究不足。随着现代科学技术的高速发展,出现了许多有助于DBD治疗心肺疾病机制研究的软件与系统,如基因组学、转录组学、网络药理学等,这些将会推进DBD的深入研究,在临床上发挥更安全和有效的作用。
基金资助
甘肃省科技计划项目(23ZDFA013-5);第九四〇医院部队面上项目(2021yxky082)。
This work was supported by the Science and Technology Program Project of Gansu Province (23ZDFA013-5) and the 940th Hospital Troop Project (2021yxky082), China.
利益冲突声明
作者声称无任何利益冲突。
作者贡献
姜琳 文献查阅和整理,论文撰写和修改;陈丽萍 论文写作指导及修改;李文斌、王荣 论文审阅与修订。所有作者阅读并同意最终的文本。
原文网址
http://xbyxb.csu.edu.cn/xbwk/fileup/PDF/2023101479.pdf
参考文献
- 1. Joseph P, Leong D, McKee M, et al. Reducing the global burden of cardiovascular disease, part 1: The epidemiology and risk factors[J]. Circ Res, 2017, 121(6): 677-694. 10.1161/CIRCRESAHA.117.308903. [DOI] [PubMed] [Google Scholar]
- 2. Ramalho SHR, Shah AM. Lung function and cardiovascular disease: a link[J]. Trends Cardiovasc Med, 2021, 31(2): 93-98. 10.1016/j.tcm.2019.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Brieler J, Breeden MA, Tucker J. Cardiomyopathy: an overview[J]. Am Fam Physician, 2017, 96(10): 640-646. [PubMed] [Google Scholar]
- 4. 王栋, 于雪, 黄凯, 等. 清热解毒中药调节炎症反应对心血管疾病异病同治的意义[J]. 中国中药杂志, 2022, 47(20): 5418-5423. 10.19540/j.cnki.cjcmm.20220601.701. [DOI] [PubMed] [Google Scholar]; WANG Dong, YU Xue, HUANG Kai, et al. Common anti-inflammatory effects of heat-clearing and toxin-removing Chinese medicines on diverse cardiovascular diseases[J]. China Journal of Chinese Materia Medica, 2022, 47(20): 5418-5423. 10.19540/j.cnki.cjcmm.20220601.701. [DOI] [PubMed] [Google Scholar]
- 5. 杨飞霞, 王玉, 夏鹏飞, 等. 当归补血汤化学成分、药理作用、临床应用的研究进展及质量标志物的预测分析[J]. 中国中药杂志, 2021, 46(11): 2677-2685. 10.19540/j.cnki.cjcmm.20200828.201. [DOI] [PubMed] [Google Scholar]; YANG Feixia, WANG Yu, XIA Pengfei, et al. Review of chemical constituents, pharmacological effects and clinical applications of Danggui Buxue Decoction and prediction and analysis of its Q-markers[J]. China Journal of Chinese Materia Medica, 2021, 46(11): 2677-2685. 10.19540/j.cnki.cjcmm.20200828.201. [DOI] [PubMed] [Google Scholar]
- 6. 曾宇, 张三印, 胡冠英. 当归补血汤的研究进展[J]. 时珍国医国药, 2016, 27(2): 422-424. [Google Scholar]; ZENG Yu, ZHANG Sanyin, HU Guanying. Research progress of Danggui Buxue Decoction[J]. Lishizhen Medicine and Materia Medica Research, 2016, 27(2): 422-424. [Google Scholar]
- 7. 国家药典委员会 . 中华人民共和国药典一部: 2020年版[M]. 北京: 中国医药科技出版社, 2020. [Google Scholar]; National Pharmacopoeia Commission . People’s republic of China (PRC) pharmacopoeia-part I: 2020 edition[M]. Beijing: China Medical Science and Technology Press, 2020. [Google Scholar]
- 8. Su HF, Shaker S, Kuang Y, et al. Phytochemistry and cardiovascular protective effects of Huang-Qi (Astragali Radix)[J]. Med Res Rev, 2021, 41(4): 1999-2038. 10.1002/med.21785. [DOI] [PubMed] [Google Scholar]
- 9. 左晓利, 毕凌云, 曹宏敏. 黄芪多糖调控微RNA-16/NF-κB轴对阿霉素肾病大鼠多药耐药基因1和P-糖蛋白170的影响[J]. 中南大学学报(医学版), 2022, 47(1): 26-34. 10.11817/j.issn.1672-7347.2022.201001. [DOI] [PMC free article] [PubMed] [Google Scholar]; ZUO Xiaoli, BI Lingyun, CAO Hongmin. Astragalus polysaccharide affects multidrug resistance gene 1 and P-glycoprotein 170 in adriamycin nephropathy rats via regulating microRNA-16/NF-κB axis[J]. Journal of Central South University. Medical Science, 2022, 47(1): 26-34. 10.11817/j.issn.1672-7347.2022.201001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Liu TL, Zhang MJ, Niu HY, et al. Astragalus polysaccharide from Astragalus Melittin ameliorates inflammation via suppressing the activation of TLR-4/NF-κB p65 signal pathway and protects mice from CVB3-induced virus myocarditis[J]. Int J Biol Macromol, 2019, 126: 179-186. 10.1016/j.ijbiomac.2018.12.207. [DOI] [PubMed] [Google Scholar]
- 11. Ming K, Zhuang S, Ma N, et al. Astragalus polysaccharides alleviates lipopolysaccharides-induced inflammatory lung injury by altering intestinal microbiota in mice[J]. Front Microbiol, 2022, 13: 1033875. 10.3389/fmicb.2022.1033875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Xiao LM, Cao PH, Luo ZH, et al. Cycloartane-type triterpenoids from the root of Astragalus membranaceus var. mongholicus[J]. J Asian Nat Prod Res, 2020, 22(10): 905-913. 10.1080/10286020.2020.1787994. [DOI] [PubMed] [Google Scholar]
- 13. Zhang XQ, Qu HY, Yang T, et al. Astragaloside IV attenuate MI-induced myocardial fibrosis and cardiac remodeling by inhibiting ROS/caspase-1/GSDMD signaling pathway[J]. Cell Cycle, 2022, 21(21): 2309-2322. 10.1080/15384101.2022.2093598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Wang ZX, Wu YC, Pei CX, et al. Astragaloside IV pre-treatment attenuates PM2.5-induced lung injury in rats: impact on autophagy, apoptosis and inflammation[J]. Phytomedicine, 2022, 96: 153912. 10.1016/j.phymed.2021.153912. [DOI] [PubMed] [Google Scholar]
- 15. Li XL, Zhao TT, Gu JL, et al. Intake of flavonoids from Astragalus membranaceus ameliorated brain impairment in diabetic mice via modulating brain-gut axis[J]. Chin Med, 2022, 17(1): 22. 10.1186/s13020-022-00578-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Chen GH, Xu HL, Xu T, et al. Calycosin reduces myocardial fibrosis and improves cardiac function in post-myocardial infarction mice by suppressing TGFBR1 signaling pathways[J]. Phytomedicine, 2022, 104: 154277. 10.1016/j.phymed.2022.154277. [DOI] [PubMed] [Google Scholar]
- 17. Chen GY, Hou Y, Li XG, et al. Sepsis-induced acute lung injury in young rats is relieved by calycosin through inactivating the HMGB1/MyD88/NF-κB pathway and NLRP3 inflammasome[J]. Int Immunopharmacol, 2021, 96: 107623. 10.1016/j.intimp.2021.107623. [DOI] [PubMed] [Google Scholar]
- 18. Long Y, Li D, Yu S, et al. Medicine-food herb: Angelica sinensis, a potential therapeutic hope for Alzheimer’s disease and related complications[J]. Food Funct, 2022, 13(17): 8783-8803. 10.1039/d2fo01287a. [DOI] [PubMed] [Google Scholar]
- 19. Cao YQ, Dong Z, Yang DP, et al. Alleviation of glucolipotoxicity-incurred cardiomyocyte dysfunction by Z-ligustilide involves in the suppression of oxidative insult, inflammation and fibrosis[J]. Chem Phys Lipids, 2021, 241: 105138. 10.1016/j.chemphyslip.2021.105138. [DOI] [PubMed] [Google Scholar]
- 20. Luo S, Gong JZ, Cao XP, et al. Ligustilide modulates oxidative stress, apoptosis, and immunity to avoid pathological damages in bleomycin induced pulmonary fibrosis rats via inactivating TLR4/MyD88/NF-κB P65[J]. Ann Transl Med, 2020, 8(15): 931. 10.21037/atm-20-4233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Li D, Rui YX, Guo SD, et al. Ferulic acid: a review of its pharmacology, pharmacokinetics and derivatives[J]. Life Sci, 2021, 284: 119921. 10.1016/j.lfs.2021.119921. [DOI] [PubMed] [Google Scholar]
- 22. Zhang XJ, Cui ZH, Zhao YX, et al. Ferulic acid ameliorates isoproterenol-induced heart failure by decreasing oxidative stress and inhibiting cardiocyte apoptosis via activating Nrf2 signaling pathway in rats[J]. Biol Pharm Bull, 2021, 44(3): 396-403. 10.1248/bpb.b20-00783. [DOI] [PubMed] [Google Scholar]
- 23. Wu XL, Lin LY, Wu HB. Ferulic acid alleviates lipopolysaccharide-induced acute lung injury through inhibiting TLR4/NF-κB signaling pathway[J/OL]. J Biochem Mol Toxicol, 2021, 35(3): e22664[2023-05-02]. 10.1002/jbt.22664. [DOI] [PubMed] [Google Scholar]
- 24. Nai JJ, Zhang C, Shao HL, et al. Extraction, structure, pharmacological activities and drug carrier applications of Angelica sinensis polysaccharide[J]. Int J Biol Macromol, 2021, 183: 2337-2353. 10.1016/j.ijbiomac.2021.05.213. [DOI] [PubMed] [Google Scholar]
- 25. 刘永升, 魏梅, 宋春梅, 等. 当归多糖通过上调VEGF抑制细胞凋亡[J]. 基因组学与应用生物学, 2020, 39(3): 1307-1313. 10.13417/j.gab.039.001307. [DOI] [Google Scholar]; LIU Yongsheng, WEI Mei, SONG Chunmei, et al. Angelica polysaccharides inhibit apoptosis by up-regulating VEGF[J]. Genomics and Applied Biology, 2020, 39(3): 1307-1313. 10.13417/j.gab.039.001307. [DOI] [Google Scholar]
- 26. Qian WB, Cai XR, Qian QH, et al. Angelica sinensis polysaccharide suppresses epithelial-mesenchymal transition and pulmonary fibrosis via a DANCR/AUF-1/FOXO3 regulatory axis[J]. Aging Dis, 2020, 11(1): 17-30. 10.14336/AD.2019.0512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. 郑恒, 张聪子, 徐金军, 等. 黄芪皂苷对病毒性心肌炎大鼠PI3K/AKT/mTOR信号通路和心肌细胞凋亡的影响[J]. 中华医院感染学杂志, 2022, 32(23): 3521-3526. 10.11816/cn.ni.2022-212318. [DOI] [Google Scholar]; ZHENG Heng, ZHANG Congzi, XU Jinjun, et al. Effects of saponins of astragalus on PI3K/AKT/mTOR signaling pathway and cardiomyocytes apoptosis in rats with viral myocarditis[J]. Chinese Journal of Nosocomiology, 2022, 32(23): 3521-3526. 10.11816/cn.ni.2022-212318. [DOI] [Google Scholar]
- 28. Liu YJ, Che GY, Di ZX, et al. Calycosin-7-O-β-D-glucoside attenuates myocardial ischemia-reperfusion injury by activating JAK2/STAT3 signaling pathway via the regulation of IL-10 secretion in mice[J]. Mol Cell Biochem, 2020, 463(1/2): 175-187. 10.1007/s11010-019-03639-z. [DOI] [PubMed] [Google Scholar]
- 29. Wang DS, Yan LY, Yang DZ, et al. Formononetin ameliorates myocardial ischemia/reperfusion injury in rats by suppressing the ROS-TXNIP-NLRP3 pathway[J]. Biochem Biophys Res Commun, 2020, 525(3): 759-766. 10.1016/j.bbrc.2020.02.147. [DOI] [PubMed] [Google Scholar]
- 30. Yi L, Cui J, Wang WQ, et al. Formononetin attenuates airway Inflammation and oxidative stress in murine allergic asthma[J]. Front Pharmacol, 2020, 11: 533841. 10.3389/fphar.2020.533841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Zhang B, Wang H, Yang Z, et al. Protective effect of alpha-pinene against isoproterenol-induced myocardial infarction through NF-κB signaling pathway[J]. Hum Exp Toxicol, 2020, 39(12): 1596-1606. 10.1177/0960327120934537. [DOI] [PubMed] [Google Scholar]
- 32. Li L, Ma H, Zhang YC, et al. Protocatechuic acid reverses myocardial infarction mediated by β-adrenergic agonist via regulation of Nrf2/HO-1 pathway, inflammatory, apoptotic, and fibrotic events[J/OL]. J Biochem Mol Toxicol, 2023, 37(3): e23270[2023-05-02]. 10.1002/jbt.23270. [DOI] [PubMed] [Google Scholar]
- 33. Zhang XL, Li CL, Li J, et al. Protective effects of protocatechuic acid on acute lung injury induced by lipopolysaccharide in mice via p38MAPK and NF-κB signal pathways[J]. Int Immunopharmacol, 2015, 26(1): 229-236. 10.1016/j.intimp.2015.03.031. [DOI] [PubMed] [Google Scholar]
- 34. Akila P, Asaikumar L, Vennila L. Chlorogenic acid ameliorates isoproterenol-induced myocardial injury in rats by stabilizing mitochondrial and lysosomal enzymes[J]. Biomedecine Pharmacother, 2017, 85: 582-591. 10.1016/j.biopha.2016.11.067. [DOI] [PubMed] [Google Scholar]
- 35. Lv B, Guo JH, Du YD, et al. Chlorogenic acid reduces inflammation by inhibiting the elevated expression of KAT2A to ameliorate lipopolysaccharide-induced acute lung injury[J]. Br J Pharmacol, 2023, 180(16): 2156-2171. 10.1111/bph.16069. [DOI] [PubMed] [Google Scholar]
- 36. Stanely Mainzen Prince P, Dhanasekar K, Rajakumar S. Vanillic acid prevents altered ion pumps, ions, inhibits Fas-receptor and caspase mediated apoptosis-signaling pathway and cardiomyocyte death in myocardial infarcted rats[J]. Chem Biol Interact, 2015, 232: 68-76. 10.1016/j.cbi.2015.03.009. [DOI] [PubMed] [Google Scholar]
- 37. Velli SK, Sundaram J, Murugan M, et al. Protective effect of vanillic acid against benzo(a)pyrene induced lung cancer in Swiss albino mice[J/OL]. J Biochem Mol Toxicol, 2019, 33(10): e22382[2023-05-02]. 10.1002/jbt.22382. [DOI] [PubMed] [Google Scholar]
- 38. 唐炜, 叶鹏林, 刘坤, 等. 咖啡酸对阿霉素心肌细胞损伤的保护作用及机制研究[J]. 安徽医科大学学报, 2022, 57(3): 402-407. 10.19405/j.cnki.issn1000-1492.2022.03.012. [DOI] [Google Scholar]; TANG Wei, YE Penglin, LIU Kun, et al. Protective effects of caffeic acid on adriamycin-induced myocardial injury and potential mechanisms[J]. Journal of Anhui University of Chinese Medicine, 2022, 57(3): 402-407. 10.19405/j.cnki.issn1000-1492.2022.03.012. [DOI] [Google Scholar]
- 39. 孙雅丽. 咖啡酸对脂多糖诱导急性肺损伤的保护作用及机制研究[D]. 哈尔滨: 东北林业大学, 2022. 10.27009/d.cnki.gdblu.2022.001063. [DOI] [Google Scholar]; SUN Yali. Protective effect and mechanism of caffeic acid on lipopolysaccharide-induced acute lung injury[D]. Harbin: Northeast Forestry University, 2022. [Google Scholar]
- 40. 高远真, 原阳, 叶婷, 等. 壬二酸对阿霉素诱导的心肌细胞损伤的保护作用及其机制[J]. 精准医学杂志, 2022, 37(4): 288-293. 10.13362/j.jpmed.202204002. [DOI] [Google Scholar]; GAO Yuanzhen, YUAN Yang, YE Ting, et al. Protective effect of azelaic acid against doxorubicin-induced cardiomyocyte injury and its mechanism[J]. Journal of Precision Medicine, 2022, 37(4): 288-293. 10.13362/j.jpmed.202204002. [DOI] [Google Scholar]
- 41. Zha YF, Xie J, Ding P, et al. Senkyunolide I protect against lung injury via inhibiting formation of neutrophil extracellular trap in a murine model of cecal ligation and puncture[J]. Int Immunopharmacol, 2021, 99: 107922. 10.1016/j.intimp.2021.107922. [DOI] [PubMed] [Google Scholar]
- 42. Gong QM, Xue Y, Li XM, et al. DL-3-n-butylphthalide attenuates lipopolysaccharide-induced acute lung injury via SIRT1-dependent and-independent regulation of Nrf2[J]. Int Immunopharmacol, 2019, 74: 105658. 10.1016/j.intimp.2019.05.043. [DOI] [PubMed] [Google Scholar]
- 43. Liu YT, Li XQ, Li AP, et al. UHPLC Q-Exactive MS-based spleen metabolomics and lipidomics to explore the effect mechanisms of Danggui Buxue Decoction in anemia mice[J]. J Pharm Biomed Anal, 2020, 185: 113234. 10.1016/j.jpba.2020.113234. [DOI] [PubMed] [Google Scholar]
- 44. Li CY, Qi LW, Li P. Correlative analysis of metabolite profiling of Danggui Buxue Tang in rat biological fluids by rapid resolution LC-TOF/MS[J]. J Pharm Biomed Anal, 2011, 55(1): 146-160. 10.1016/j.jpba.2010.12.034. [DOI] [PubMed] [Google Scholar]
- 45. Gong AGW, Duan R, Wang HY, et al. Calycosin orchestrates osteogenesis of Danggui Buxue Tang in cultured osteoblasts: evaluating the mechanism of action by omics and chemical knock-out methodologies[J]. Front Pharmacol, 2018, 9: 36. 10.3389/fphar.2018.00036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. 王华伟, 王文萍. 给予当归补血汤或当归后兔血浆中阿魏酸的药动学研究[J]. 辽宁中医杂志, 2022, 49(8): 166-169. 10.13192/j.issn.1000-1719.2022.08.046. [DOI] [Google Scholar]; WANG Huawei, WANG Wenping. Determination of ferulic acid in rabbits plasma administrated with Danggui Buxue Decoction or Danggui (angelicae sinensis radix)[J]. Liaoning Journal of Traditional Chinese Medicine, 2022, 49(8): 166-169. 10.13192/j.issn.1000-1719.2022.08.046. [DOI] [Google Scholar]
- 47. Zhao HD, Qi M, Gong Y, et al. Danggui Buxue Decoction: comparative pharmacokinetic research on six bio-active components in different states by ultra-performance liquid chromatography-tandem mass spectrometry after oral administration[J/OL]. J Sep Sci, 2023, 46(6): e2200794[2023-05-02]. 10.1002/jssc.202200794. [DOI] [PubMed] [Google Scholar]
- 48. Shi XQ, Tang YP, Zhu HX, et al. Pharmacokinetic comparison of seven major bio-active components in normal and blood deficiency rats after oral administration of Danggui Buxue Decoction by UPLC-TQ/MS[J]. J Ethnopharmacol, 2014, 153(1): 169-177. 10.1016/j.jep.2014.02.004. [DOI] [PubMed] [Google Scholar]
- 49. 范智文, 赵一俏, 凌龙, 等. miR-34a在缺氧复氧介导心肌细胞凋亡中的作用以及当归补血汤的干预效果[J]. 广东医学, 2017, 38(22): 3401-3404. 10.13820/j.cnki.gdyx.20171117.004. [DOI] [Google Scholar]; FAN Zhiwen, ZHAO Yiqiao, LING Long, et al. Study on the role of miR-34a in cardiomyocyte apoptosis induced by reoxygenation after hypoxia and the intervention of Danggui Buxue Decoction[J]. Guangdong Medical Journal, 2017, 38(22): 3401-3404. 10.13820/j.cnki.gdyx.20171117.004. [DOI] [Google Scholar]
- 50. 王时光, 梁国庆, 朱静, 等. 当归补血汤对冠脉结扎致心肌梗死模型大鼠心肌细胞凋亡的保护作用[J]. 中成药, 2016, 38(11): 2458-2461. 10.3969/j.issn.1001-1528.2016.11.030. [DOI] [Google Scholar]; WANG Shiguang, LIANG Guoqing, ZHU Jing, et al. Protective effect of Danggui Buxue Decoction on myocardial cell apoptosis in myocardial infarction model rats caused by coronary artery ligation[J]. Chinese Traditional Patent Medicine, 2016, 38(11): 2458-2461. 10.3969/j.issn.1001-1528.2016.11.030. [DOI] [Google Scholar]
- 51. 吴伟, 刘新灿. 当归补血汤对大鼠心肌缺血再灌注损伤的保护作用[J]. 中国中医药现代远程教育, 2018, 16(1): 99-100. 10.3969/j.issn.1672-2779.2018.01.043. [DOI] [Google Scholar]; WU Wei, LIU Xincan. The protective effect of Danggui Buxue Decoction on myocardial ischemia reperfusion injury in rats[J]. Chinese Medicine Modern Distance Education of China, 2018, 16(1): 99-100. 10.3969/j.issn.1672-2779.2018.01.043. [DOI] [Google Scholar]
- 52. 王时光, 杨帆, 梁国庆, 等. 当归补血汤对心肌缺血模型大鼠心肌组织氧化应激的影响[J]. 时珍国医国药, 2016, 27(8): 1850-1852. 10.3969/j.issn.1008-0805.2016.08.020. [DOI] [Google Scholar]; WANG Shiguang, YANG Fan, LIANG Guoqing, et al. Effect of antioxidant activity of Dangguibuxuetang on coronary artery ligation-induced myocardial ischemia rat[J]. Lishizhen Medicine and Materia Medica Research, 2016, 27(8): 1850-1852. 10.3969/j.issn.1008-0805.2016.08.020. [DOI] [Google Scholar]
- 53. Zaveri MP, Perry JC, Schuetz TM, et al. Diabetic cardiomyopathy as a clinical entity: is it a myth?[J/OL]. Cureus, 2020, 12(10): e11100[2023-05-02]. 10.7759/cureus.11100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. 王琳娜, 郭存霞, 陈小永. 当归补血汤联合可调钠曲线血液透析对糖尿病性心肌病患者心肌跨壁复极离散度的影响[J]. 中国现代医学杂志, 2015, 25(23): 52-56. 10.3969/j.issn.1005-8982.2015.23.011. [DOI] [Google Scholar]; WANG Linna, GUO Cunxia, CHEN Xiaoyong. Effect of Danggui Buxue Decoction combined with adjustable sodium curve hemodialysis on transmural dispersion of repolarization in patients with diabetic cardiomyopathy[J]. China Journal of Modern Medicine, 2015, 25(23): 52-56. 10.3969/j.issn.1005-8982.2015.23.011. [DOI] [Google Scholar]
- 55. 蒲秀瑛, 王恒瑞, 许珊丽. 当归补血汤对心力衰竭大鼠心肌组织的影响[J]. 中医药学报, 2013, 41(3): 102-105. 10.19664/j.cnki.1002-2392.2013.03.035. [DOI] [Google Scholar]; PU Xiuying, WANG Hengrui, XU Shanli. Influence of Danggui Buxue Decoction on cardiac tissue of rats with heart failure[J]. Acta Chinese Medicine and Pharmacology, 2013, 41(3): 102-105. 10.19664/j.cnki.1002-2392.2013.03.035. [DOI] [Google Scholar]
- 56. Savin IA, Zenkova MA, Sen’kova AV. Pulmonary fibrosis as a result of acute lung inflammation: molecular mechanisms, relevant in vivo models, prognostic and therapeutic approaches[J]. Int J Mol Sci, 2022, 23(23): 14959. 10.3390/ijms232314959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Zhao P, Zhou WC, Li DL, et al. Total glucosides of Danggui Buxue Tang attenuate BLM-induced pulmonary fibrosis via regulating oxidative stress by inhibiting NOX4[J]. Oxid Med Cell Longev, 2015, 2015: 645814. 10.1155/2015/645814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. 刘娜, 王杰鹏, 鲁辰希, 等. 当归补血汤对博莱霉素致肺纤维化大鼠PKD1/NF-κB/MnSOD信号通路的影响[J]. 中国实验方剂学杂志, 2020, 26(13): 66-72. 10.13422/j.cnki.syfjx.20201303. [DOI] [Google Scholar]; LIU Na, WANG Jiepeng, LU Chenxi, et al. Effect of Danggui buxuetang on PKD1/NF-κB/MnSOD signal pathway in bleomycin-induced pulmonary fibrosis in rats[J]. Chinese Journal of Experimental Traditional Medical Formulae, 2020, 26(13): 66-72. 10.13422/j.cnki.syfjx.20201303. [DOI] [Google Scholar]
- 59. Arroyo-Hernández M, Maldonado F, Lozano-Ruiz F, et al. Radiation-induced lung injury: current evidence[J]. BMC Pulm Med, 2021, 21(1): 9. 10.1186/s12890-020-01376-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. 李重, 吴洪斌, 雷章, 等. 当归补血汤对急性放射性肺损伤大鼠NF-κB和ET-1的影响[J]. 辽宁中医杂志, 2019, 46(5): 1059-1062, 1119. 10.13192/j.issn.1000-1719.2019.05.053. 31092714 [DOI] [Google Scholar]; LI Zhong, WU Hongbin, LEI Zhang, et al. Effect of Danggui Buxue Decoction on NF-κB and ET-1 in rats with acute radiation-induced lung injury[J]. Liaoning Journal of Traditional Chinese Medicine, 2019, 46(5): 1059-1062, 1119. 10.13192/j.issn.1000-1719.2019.05.053. [DOI] [Google Scholar]
- 61. Schabath MB, Cote ML. Cancer progress and priorities: lung cancer[J]. Cancer Epidemiol Biomarkers Prev, 2019, 28(10): 1563-1579. 10.1158/1055-9965.EPI-19-0221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. 刘玉才, 张焕珍, 于凤玲, 等. 加味当归补血汤治疗肺癌放疗后白细胞减少的临床疗效[J]. 山东医学高等专科学校学报, 2016, 38(2): 112-114. 10.3969/j.issn.1674-0947.2016.02.013. [DOI] [Google Scholar]; LIU Yucai, ZHANG Huanzhen, YU Fengling, et al. Clinical effect of modified Danggui blood-enriching decoction on leukopenia induced by radiotherapy in patients with lung cancer[J]. Journal of Shandong Medical College, 2016, 38(2): 112-114. 10.3969/j.issn.1674-0947.2016.02.013. [DOI] [Google Scholar]
- 63. Yang YK, Li J, Rao TC, et al. The role and mechanism of hyperoside against myocardial infarction in mice by regulating autophagy via NLRP1 inflammation pathway[J]. J Ethnopharmacol, 2021, 276: 114187. 10.1016/j.jep.2021.114187. [DOI] [PubMed] [Google Scholar]
- 64. Sies H. Oxidative stress: a concept in redox biology and medicine[J]. Redox Biol, 2015, 4: 180-183. 10.1016/j.redox.2015.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. 陈前, 申楚翘, 程凯, 等. 当归补血汤调节自噬干预心肌梗死模型大鼠心肌损伤的作用机制[J]. 安徽中医药大学学报, 2023, 42(1): 92-97. 10.3969/j.issn.2095-7246.2023.01.021. [DOI] [Google Scholar]; CHEN Qian, SHEN Chuqiao, CHENG Kai, et al. Mechanism of action of Danggui Buxue Decoction in intervention against myo-cardial injury by regulating autophagy in a rat model of myocardial infarction[J]. Journal of Anhui University of Chinese Medicine, 2023, 42(1): 92-97. 10.3969/j.issn.2095-7246.2023.01.021. [DOI] [Google Scholar]
- 66. 毛启远, 刘兰椿, 王瑾琨, 等. 当归补血汤对阿霉素心肌损伤小鼠的保护作用及对心肌细胞自噬的影响[J]. 中华中医药杂志, 2021, 36(5): 2515-2520. [Google Scholar]; MAO Qiyuan, LIU Lanchun, WANG Jinkun, et al. Protective effect of Danggui Buxue Decoction on myocardial injury induced by adriamycin in mice and its effect on autophagy of cardiomyocytes[J]. China Journal of Traditional Chinese Medicine and Pharmacy, 2021, 36(5): 2515-2520. [Google Scholar]
- 67. 王嫔, 李丹, 田昕. 当归补血汤对衰老心肌梗死大鼠冠状动脉侧枝血管生成的影响及其机制[J]. 中药材, 2016, 39(7): 1651-1653. 10.13863/j.issn1001-4454.2016.07.048. 27307068 [DOI] [Google Scholar]; WANG Pin, LI Dan, TIAN Xin. Effect of Danggui Buxue Decoction on coronary artery collateral angiogenesis in aged rats with myocardial infarction and its mechanism[J]. Journal of Chinese Medicinal Materials, 2016, 39(7): 1651-1653. 10.13863/j.issn1001-4454.2016.07.048. [DOI] [Google Scholar]
- 68. 武春莲, 邓翠东, 吴俊才. 当归补血汤通过PI3K/Akt通路减轻老年心肌梗死大鼠心肌损害的实验研究[J]. 中国中医急症, 2023, 32(5): 781-785, 805. 10.3969/j.issn.1004-745X.2023.05.006. [DOI] [Google Scholar]; WU Chunlian, DENG Cuidong, WU Juncai. Danggui Buxue Decoction alleviates myocardial damage in aged rats with myocardial infarction through PI3K/Akt pathway[J]. Journal of Emergency in Traditional Chinese Medicine, 2023, 32(5): 781-785, 805. 10.3969/j.issn.1004-745X.2023.05.006. [DOI] [Google Scholar]
- 69. 李亭亭, 陈捷, 吉恩生, 等. 当归补血汤含药血清对间歇性低氧暴露H9c2心肌细胞损伤的作用及机制[J]. 中国中药杂志, 2023, 48(21): 5881-5887. 10.19540/j.cnki.cjcmm.20230614.703. [DOI] [PubMed] [Google Scholar]; LI Tingting, CHEN Jie, JI Ensheng, et al. Effect and mechanism of Danggui Buxue Decoction-containing serum in mitigating H9c2 cell injury caused by exposure to intermittent low oxygen[J]. China Journal of Chinese Materia Medica, 2023, 48(21): 5881-5887. 10.19540/j.cnki.cjcmm.20230614.703. [DOI] [PubMed] [Google Scholar]
- 70. 赵信科, 汪鸣, 邱勇玉, 等. 基于FasL/TNF-α信号通路的当归补血汤对放射性心肌损伤凋亡反应的影响探讨[J]. 时珍国医国药, 2020, 31(6): 1312-1314. [Google Scholar]; ZHAO Xinke, WANG Ming, QIU Yongyu, et al. Effect of Danggui Buxue decoction on apoptosis of radiation-induced myocardial injury based on FasL/TNF-α signal pathway[J]. Lishizhen Medicine and Materia Medica Research, 2020, 31(6): 1312-1314. [Google Scholar]
- 71. Sun XY, Xu X, Chen YF, et al. Danggui Buxue Decoction sensitizes the response of non-small-cell lung cancer to gemcitabine via regulating deoxycytidine kinase and P-glycoprotein[J]. Molecules, 2019, 24(10): 2011. 10.3390/molecules24102011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Wang JP, Wang H, Fang F, et al. Danggui Buxue Tang ameliorates bleomycin-induced pulmonary fibrosis by suppressing the TLR4/NLRP3 signaling pathway in rats[J]. Evid Based Complement Alternat Med, 2021, 2021: 8030143. 10.1155/2021/8030143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. 鲁辰希, 王杰鹏, 刘娜, 等. 当归补血汤对特发性肺纤维化大鼠血管新生因子HIF-1α和endostatin的影响[J]. 中华中医药杂志, 2021, 36(3): 1683-1687. [Google Scholar]; LU Chenxi, WANG Jiepeng, LIU Na, et al. Effects of Danggui Buxue Decoction on HIF-1α and endostatin of angiogenesis factors in experimental idiopathic pulmonary fibrosis rats[J]. China Journal of Traditional Chinese Medicine and Pharmacy, 2021, 36(3): 1683-1687. [Google Scholar]
- 74. 崔爽. 当归补血汤联合卡维地洛治疗心肌缺血疗效观察[J]. 中药药理与临床, 2017, 33(2): 197-199. 10.13412/j.cnki.zyyl.2017.02.056. [DOI] [Google Scholar]; CUI Shuang. Efficacy of dangguibuxue decoction combined with carvedilol on myocardial ischemia[J]. Pharmacology and Clinics of Chinese Materia Medica, 2017, 33(2): 197-199. 10.13412/j.cnki.zyyl.2017.02.056. [DOI] [Google Scholar]
- 75. 栾海燕, 李铮, 杜毅. 当归补血汤对急性心肌梗死病人心室重塑及心功能的影响[J]. 中西医结合心脑血管病杂志, 2019, 17(16): 2466-2468. 10.12102/j.issn.1672-1349.2019.16.018. [DOI] [Google Scholar]; LUAN Haiyan, LI Zheng, DU Yi. Effect of Danggui Buxue Decoction on ventricular remodeling and cardiac function in patients with acute myocardial infarction[J]. Chinese Journal of Integrative Medicine on Cardio/Cerebrovascular Disease, 2019, 17(16): 2466-2468. 10.12102/j.issn.1672-1349.2019.16.018. [DOI] [Google Scholar]
- 76. 郭桂明, 王小利, 王卫华. 加味当归补血汤治疗肺癌放疗后白细胞减少的临床疗效观察[J]. 实用中医内科杂志, 2022, 36(6): 122-124. 10.13729/j.issn.1671-7813.Z20211393. [DOI] [Google Scholar]; GUO Guiming, WANG Xiaoli, WANG Weihua. Clinical observation of modified Danggui Buxue Decoction in the treatment of leukopenia after radiotherapy for lung cancer[J]. Journal of Practical Traditional Chinese Internal Medicine, 2022, 36(6): 122-124. 10.13729/j.issn.1671-7813.Z20211393. [DOI] [Google Scholar]
- 77. 张喜峰. 当归补血汤对肺癌放化疗后骨髓抑制的影响[J]. 北方药学, 2017, 14(9): 39. [Google Scholar]; ZHANG Xifeng. Effect of Danggui Buxue decoction on bone marrow suppression after radiotherapy and chemotherapy for lung cancer[J]. Journal of North Pharmacy, 2017, 14(9): 39. [Google Scholar]
- 78. 范佳儿, 刘银榕, 晁志, 等. 当归补血丸指纹图谱及3种指标成分含量测定方法的建立[J]. 中国药房, 2022, 33(11): 1343-1347, 1354. 10.6039/j.issn.1001-0408.2022.11.11. [DOI] [Google Scholar]; FAN Jia’er, LIU Yinrong, CHAO Zhi, et al. Establishment of the fingerprints for Danggui Buxue pills and the method for the content determination of three indicative constituents[J]. China Pharmacy, 2022, 33(11): 1343-1347, 1354. 10.6039/j.issn.1001-0408.2022.11.11. [DOI] [Google Scholar]
- 79. 李思聪, 李旭廷, 李金良, 等. 当归补血颗粒的质量标准研究[J]. 动物医学进展, 2018, 39(4): 42-48. 10.16437/j.cnki.1007-5038.2018.04.008. [DOI] [Google Scholar]; LI Sicong, LI Xuting, LI Jinliang, et al. Study on quality standard of Danggui Buxue Keli[J]. Progress in Veterinary Medicine, 2018, 39(4): 42-48. 10.16437/j.cnki.1007-5038.2018.04.008. [DOI] [Google Scholar]