Abstract
目的
脓毒症是宿主对感染的反应失调引起的危及生命的器官功能障碍。由于缺乏有效的治疗手段,脓毒症一直是临床治疗的难点和挑战。研究表明脑源性神经营养因子前体(pro-brain-derived neurotrophic factor,proBDNF)可通过结合其高亲和力受体p75神经营养因子受体(p75 neurotrophin receptor,p75NTR)激活下游信号通路,扰乱免疫炎症微环境,在脓毒症病程的进展中发挥重要作用。本研究主要探讨淋巴细胞来源的proBDNF/p75NTR在脓毒症患者中的表达变化及其对淋巴细胞分化的影响。
方法
收集健康志愿者(对照组,n=40)和初次入院的脓毒症患者(脓毒症组,n=40)外周血样本,进行血常规临床指标检测,采用流式细胞术检测淋巴细胞亚群及其proBDNF/p75NTR的表达变化;体外分选对照组外周血淋巴细胞并采用脂多糖(lipopolysaccharide,LPS)刺激培养,流式细胞分析技术检测LPS刺激对淋巴细胞亚群proBDNF/p75NTR的表达影响;随后采用p75NTR的拮抗剂(p75ECD-Fc)抑制p75NTR观察对淋巴细胞分化的影响。
结果
与对照组相比,脓毒症组患者入院时白细胞计数、中性粒细胞计数和中性粒细胞百分比均显著升高,淋巴细胞计数与淋巴细胞百分比均显著降低(均P<0.001),中性粒细胞与淋巴细胞比值、单核细胞与淋巴细胞比值均显著升高(均P<0.05)。与对照组相比,proBDNF在脓毒症组外周血中CD19+ B细胞的表达上调(P<0.05),而p75NTR在CD19+ B细胞、CD4+ T细胞和CD8+ T细胞中的表达均上调(均P<0.05)。体外采用LPS刺激可诱导对照组外周血淋巴细胞的proBDNF/p75NTR表达上调(均P<0.05),趋势与在脓毒症组外周淋巴细胞的表达变化基本一致。抑制p75NTR可增加CD4+ T细胞和CD19+ B细胞的百分比,促进细胞因子IL-4和IL-10的表达,并降低IL-1β和IL-6的产生(均P<0.05)。
结论
脓毒症患者入院时即处于以淋巴细胞数量减少为特征的免疫抑制阶段,同时伴有中性粒细胞占比增加。ProBDNF/p75NTR在脓毒症患者外周血淋巴细胞表达增加,抑制p75NTR可能通过调节淋巴细胞的分化,参与脓毒症进展。
Keywords: 淋巴细胞, 脑源性神经营养因子前体, p75神经营养因子受体, 脓毒症
Abstract
Objective
Sepsis is a life-threatening organ dysfunction caused by the host’s imbalanced response to infection. Due to lack of effective treatments, it has always been the difficulty and focus of clinical treatment of sepsis. Studies have shown that pro-brain-derived neurotrophic factor (proBDNF) binds to the high-affinity total neurotrophic factor p75 neurotrophin receptor (p75NTR), which activates downstream signaling cascades and disrupts immunological inflammation and plays an important role in the progression of sepsis. This study aims to explore the expression changes of lymphocyte-derived proBDNF/p75NTR in patients with sepsis and its effect on lymphocyte differentiation.
Methods
From the healthy donors (control group, n=40) and sepsis patients (sepsis group, n=40) admitted to the hospital for the first time, peripheral blood samples and blood routine clinical detection indicators were obtained. By using flow cytometry, the proportion of lymphocyte subsets and their expression of proBDNF/p75NTR were examined. The peripheral blood lymphocytes were isolated from the control group and incubated with lipopolysaccharide (LPS). Flow cytometry analysis technology was used to detect the expression of proBDNF/p75NTR on LPS-treated lymphocyte subsets. On this basis, we investigated the effects on lymphocyte differentiation by inhibiting p75NTR.
Results
White blood cell count, neutrophil count, and neutrophil percentage of the patients in the sepsis group at admission were significantly higher than those in the control group; on the contrary, lymphocyte count and lymphocyte percentage in the sepsis group were lower than those in the control group (all P<0.001). The patients in the sepsis group had considerably greater neutrophil/lymphocyte and monocyte/lymphocyte ratios than those in the control group (both P<0.05). In the peripheral blood of sepsis patients, proBDNF expression was upregulated on CD19+ B cells, whereas p75NTR expression was elevated on B cells, CD4+ T cells, and CD8+ T cells (all P<0.05). ProBDNF/p75NTR expression was upregulated by LPS stimulation in vitro in peripheral blood cells of the control group (P<0.05), and this tendency was similar to the expression alterations in peripheral lymphocytes of the sepsis group. Inhibition of p75NTR increased CD4+ T cell and CD19+ B cell percentages, cytokine expression of IL-4 and IL-10, and reduced IL-1β and IL-6 production (all P<0.05).
Conclusion
The immunosuppressive state of sepsis patients is indicated by a reduction in lymphocyte count and an increase in the proportion of inactive neutrophils. ProBDNF/p75NTR expression is upregulated in the peripheral blood lymphocytes of sepsis patients, and p75NTR inhibition may control lymphocyte differentiation involved in sepsis progression.
Keywords: lymphocytes, pro-brain-derived neurotrophic factor, p75 neurotrophin receptor, sepsis
脓毒症是由宿主对感染的反应失调引起的危及生命的器官功能障碍,是创伤、烧伤和感染等临床急危重患者的严重并发症之一[1]。淋巴细胞介导的适应性免疫反应在脓毒症进展中发挥了重要作用。T淋巴细胞数目和功能异常与脓毒症患者的病情严重程度明显相关[2]。T细胞数量减少及免疫功能持续低下是预测脓毒症患者结局不良的2个重要指标[3]。此外,脓毒症患者外周B淋巴细胞的增殖抑制表现为进行性减少[4-5],其中感染性休克死亡的脓毒症患者外周CD23+ B细胞百分比显著低于幸存患者。探究脓毒症各淋巴细胞表型/功能的改变并寻找脓毒症免疫调控因素,对于改善脓毒症患者预后具有重要意义。
脑源性神经营养因子前体蛋白(pro-brain-derived neurotrophic factor,proBDNF)是成熟BDNF合成过程中的中间体,广泛分布于中枢神经系统的神经营养因子。与成熟BDNF的神经保护作用相反,proBDNF与其高亲和力受体p75神经营养因子受体(p75 neurotrophin receptor,p75NTR)结合,介导促进神经元凋亡和轴突修剪,以及负向调节学习和记忆[6]。本研究团队的前期研究表明:在系统性红斑狼疮[7]、多发性硬化[8]等自身免疫性疾病中,proBDNF/p75NTR信号在活化的淋巴细胞表达上调并通过自分泌或者旁分泌的形式扰乱免疫炎症微环境,加重器官损伤。此外,脂多糖(lipopolysaccharide,LPS)腹腔注射诱导的脓毒症模型小鼠外周血中CD4+ T细胞和CD19+ B细胞比例降低。外源性重组proBDNF蛋白处理剂量依赖性地降低LPS诱导的脓毒症模型小鼠脾CD4+ T细胞的百分比[9]。以上研究提示proBDNF可能通过作用p75NTR调控淋巴细胞的功能表型,参与脓毒症的病程进展。
本研究拟通过比较分析脓毒症患者与健康志愿者的外周血中淋巴细胞亚型变化和对比分析proBDNF及其受体p75NTR在脓毒症患者外周血中淋巴细胞上的表达差异。体外采用LPS刺激外周血淋巴细胞并检测proBDNF/p75NTR信号的表达变化,在此基础上加入p75NTR的拮抗剂(p75ECD-Fc)联合干预2 d[7],进一步观察拮抗p75NTR对淋巴细胞分化的影响。
1. 对象与方法
1.1. 对象
本研究于2023年1月1日至2023年4月1日纳入在中南大学湘雅二医院(以下简称“我院”)重症监护室的确诊脓毒症患者(脓毒症组,n=40)和我院体检中心的健康志愿者(对照组,n=40)。脓毒症患者的纳入标准:年龄>18岁;符合《中国严重脓毒症/脓毒性休克治疗指南(2014)》[10]中的相关诊断。排除标准:慢性肝病(Child-Pugh评分>12)患者;有活动性恶性肿瘤病史的患者;感染免疫缺陷病毒的患者;4周内进行过化学治疗(以下简称“化疗”)或激素治疗的患者;怀孕和哺乳患者;确诊后非脓毒症致死的患者。所有患者家属均签署知情同意书。对照组纳入标准:纳入与脓毒症组的年龄和性别匹配的健康志愿者,体检报告各项临床指标均在正常范围内。所有参与者均签署捐赠血液用于实验研究的知情同意书。本研究已通过我院医学伦理委员会批准,审批号为(2021)伦审[临研]第(078)号,并在中国临床试验注册中心注册(注册号:ChiCTR2000033592)。
1.2. 主要试剂
红细胞裂解液RBC Lysis Buffer、流式染色封闭试剂Fc受体封闭试剂、APC-Cy7标记的抗人CD45抗体(APC-Cy7-CD45)、PE-Cy7标记的抗CD3抗体(PE-Cy7-CD3)、PerCP/Cy5.5标记的抗人CD4抗体(PerCP-Cy5.5-CD4)、APC标记的抗人CD19抗体(APC-CD19)、固定液Fixation buffer均购自美国Biolegend 公司。BV510标记的抗CD8抗体购自美国BD公司。Biotin anti-proBDNF抗体、p75ECD-Fc和Control-Fc均由上海易乐生物技术有限公司赠予。FITC标记的荧光二抗购自美国Abcam公司。PE标记的抗人和鼠CD271(p75NTR)抗体购自美国Miltenyi Biotec公司。缓冲液PBS购自美国Boston BioProducts公司。淋巴细胞分离液购自美国Stemcell公司。
1.3. 方法
1.3.1. 收集血常规分析检测指标
收集对照组和脓毒症组患者初次入院的外周血细胞计数检测(即血常规分析)结果,检测指标包括白细胞、中性粒细胞、淋巴细胞、单核细胞的百分比及绝对计数值。
1.3.2. 流式细胞术检测淋巴细胞亚群百分比及其proBDNF/p75NTR的表达变化
取外周血100 μL,加入红细胞裂解液1 mL,混匀后于室温放置10 min。加入4 mL PBS缓冲液终止反应,350 g离心5 min后弃上清。加入0.5 μL的Fc受体封闭试剂,于室温封闭15 min。再依次加入抗APC-Cy7-CD45、PE-Cy7-CD3、PerCP-Cy5.5-CD4、BV510-CD8、APC-CD19、PE-CD271(p75NTR)各0.5 μL旋涡混匀,于4 ℃避光反应30 min。随后每管加入200 μL固定液,旋涡混匀,4 ℃反应20 min。固定破膜后,每管加入1 μL Biotin anti-proBDNF单克隆抗体,混匀,于4 ℃避光反应30 min。同型对照管则加入Biotin anti-IgG。随后每管加入1 mL PBS缓冲液清洗,混匀,以350 g离心5 min后弃上清液。加入1 μL FITC标记的荧光二抗,混匀,于室温避光反应30 min。PBS缓冲液清洗,清洗完毕后,每管加入300 μL PBS定容。采用流式细胞分析仪器进行检测,数据采用Flow jo 7.6软件进行分析。
1.3.3. 外周血淋巴细胞分离、体外刺激培养检测proBDNF/p75NTR表达水平
收集对照组外周血2 mL,并获取外周血淋巴细胞。体外加入LPS(1 μg/mL)[8]刺激。培养2 d后,收集各组细胞用于检测proBDNF/p75NTR表达变化。具体染色与检测步骤同1.3.2。
1.3.4. 干预p75NTR对外周血淋巴细胞分化的检测
参考课题组前期研究[7],加入p75NTR胞外段融合蛋白(extracellular domain of p75NTR,p75ECD-Fc)阻断内源性p75NTR激活,用于观察拮抗p75NTR对淋巴细胞分化的影响。在1.3.3的基础上联合加入p75ECD-Fc (500 ng/mL)或对照Fc干预处理。2 d后,收集各组细胞用于下列2组实验,或对照Fc干预处理。
1)流式细胞分析检测淋巴细胞亚群分化百分比:实验操作流程依次为各管加入100 μL PBS缓冲液,加入0.5 μL流式染色封闭试剂,于室温封闭15 min。然后各管加入抗PE-Cy7-CD3,PerCP-Cy5.5-CD4,BV510-CD8和APC-CD19各0.5 μL旋涡混匀,于4 ℃避光反应30 min。每管加入1 mL PBS缓冲液清洗,混匀,以350 g离心5 min后弃上清液。重复此操作2次。清洗、检测与数据分析同1.3.2。
2)实时反转录聚合酶链反应(real-time reverse transcription PCR,real-time RT-PCR)检测T细胞亚群相关细胞因子的转录水平差异:采用商业化试剂盒提取RNA并反转录为cDNA,通过使用定量检测试剂盒进行RT-qPCR。使用β-actin校正目的基因,通过2-ΔΔct计算目的基因的mRNA相对表达水平。PCR引物序列见表1。
表1.
实时反转录聚合酶链反应引物序列
Table 1 Sequences of primers for real-time reverse transcription PCR
| 基因名称 | 引物序列(5'→3') |
|---|---|
| IFNg |
正向:ACTAGGCAGCCAACCTAAGCAAGA 反向:CATCAGGGTCACCTGACACATTCA |
| IL-4 |
正向:CCAACTGCTTCCCCCTCTG 反向:TCTGTTACGGTCAACTCGGTG |
| IL-17 |
正向:CAGGATGCCCAAATTCTGAGG 反向:CAAGGTGAGGTGGATCGGTT |
| IL-10 |
正向:GACTTTAAGGGTTACCTGGGTTG 反向:TCACATGCGCCTTGATGTCTG |
| TGF-β |
正向:GGCCAGATCCTGTCCAAGC 反向:GTGGGTTTCCACCATTAGCAC |
| IL-1β |
正向:ATCCAGCTACGAATCTCCGA 反向:CCACTTGTTGCTCCATATCC |
| IL-6 |
正向:CTTCGGTCCAGTTGCCTTCT 反向:GTGCCTCTTTGCTGCTTTCA |
| β-actin |
正向:GATCATTGCTCCTCCTGAGC 反向:CGTCATACTCCTGCTTGCTG |
1.4. 统计学处理
采用Graphpad Prism 8.0 进行数据处理。计量资料用均数±标准误(mean±SEM)表示;非正态分布的计量资料用中位数(第1四分位数,第3四分位数) [M(P 25, P 75)]表示。服从正态分布且方差齐的数据的组间比较采用t检验,若不服从正态分布或方差不齐则采用非参数秩和检验。P<0.05为差异具有统计学意义。
2. 结 果
2.1. 脓毒症组与对照组免疫细胞数目的比较
40例脓毒症患者中男性20例,女性20例,年龄为(67.85±13.63)岁。脓毒症患者入院时白细胞计数(11.97×109/L)、中性粒细胞计数(10.92×109/L)和中性粒细胞百分比(89.76%)均明显高于对照组(均P<0.001,表2)。相反,脓毒症患者淋巴细胞计数(0.92×109/L)、淋巴细胞百分比(6.74%)和单核细胞百分比(3.63%)均低于对照组(均P<0.001),但单核细胞计数的差异无统计学意义(P>0.05, 表2)。表明脓毒症患者入院时可能已进入以淋巴细胞大量凋亡为特征的免疫抑制阶段。
表2.
脓毒症组与对照组免疫细胞数目的比较
Table 2 Comparison of peripheral blood immune cells in sepsis patients and control donors
| 组别 | n |
白细胞计数/ (×109·L-1) |
中性粒细胞计数/ (×109·L-1) |
中性粒细胞 百分比/% |
淋巴细胞计数/ (×109·L-1) |
淋巴细胞 百分比/% |
|---|---|---|---|---|---|---|
| 对照组 | 40 | 6.02±1.25 | 3.62±1.21 | 59.03±11.11 | 1.75±0.59 | 30.15±10.69 |
| 脓毒症组 | 40 | 11.97(7.25, 17.75) | 10.92(5.70, 16.18) | 89.76(83.06, 92.18) | 0.92(0.54, 1.11) | 6.74(4.32, 11.63) |
| Z | -4.691 | -4.697 | -4.973 | -4.698 | -5.350 | |
| P | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| 组别 |
单核细胞计数/ (×109·L-1) |
单核细胞百分比/% |
中性粒细胞与 淋巴细胞比值 |
单核细胞与 淋巴细胞比值 |
血小板与淋巴细胞比值 |
|---|---|---|---|---|---|
| 对照组 | 0.40(0.32, 0.53) | 6.44(5.33, 9.21) | 1.80(1.56, 2.78) | 0.26(0.16, 0.28) | 106.85(92.71, 163.61) |
| 脓毒症组 | 0.37(0.25, 0.65) | 3.63(1.84, 4.92) | 13.47(7.12, 21.74) | 0.45(0.30, 0.68) | 146.40(69.43, 240.10) |
| Z | -0.342 | -4.476 | -5.094 | -3.186 | -1.505 |
| P | 0.732 | <0.001 | <0.001 | 0.001 | 0.132 |
正态分布的计量资料用均数±标准误表示;非正态分布的计量资料用中位数(第1四分位数,第3四分位数)表示。
进一步对比分析脓毒症组与对照组的免疫细胞与淋巴细胞比值,结果显示:脓毒症患者的中性粒细胞与淋巴细胞比值为13.47(7.12, 21.74),单核细胞与淋巴细胞比值为0.45(0.30, 0.68),均高于对照组 (均P<0.001,表2)。表明脓毒症患者入院时存在高水平炎症反应和免疫功能紊乱。
2.2. 脓毒症组与对照组淋巴细胞亚群百分比的比较
采用流式细胞术,比较脓毒症患者和对照组外周血淋巴细胞亚群的差异。淋巴细胞亚群的门控策略如下(图1A):从CD45+白细胞分析CD3+ CD19- T淋巴细胞和CD3- CD19+ B淋巴细胞进行门控,再从CD3+ CD19- T淋巴细胞继续观察CD8+ T淋巴细胞(Q1)和CD4+ T淋巴细胞亚群(Q3)。与对照组相比,脓毒症组患者外周血中CD3+ T淋巴细胞(P<0.05,图1B)和CD19+ B淋巴细胞(P<0.01,图1E)在CD45+白细胞中的百分比呈现一致性的减少。在CD3+ T淋巴细胞中,进一步将T淋巴细胞分为CD4- CD8+细胞(CD8+ T细胞,Q1)、CD4+ CD8+细胞(Q2)、CD4+ CD8-细胞(CD4+ T细胞,Q3)和CD4- CD8-细胞(Q4)4个亚群。其中,与对照组相比,脓毒症组患者外周血CD4+ T淋巴细胞百分比显著减少(P<0.05,图1C),而脓毒症组患者外周血CD8+ T淋巴细胞百分比(图1D)有下降趋势,但差异无统计学意义(P>0.05)。
图1.
脓毒症组和对照组的淋巴细胞亚群百分比
Figure 1 Percentage of lymphocytes subsets of peripheral blood in the sepsis group and the control group A: Gateing strategy; B: Percentage of CD3+ T cells; C: Percentage of CD4+ T cells; D: Percentage of CD8+ T cells; E: Percentage of CD19+ B cells. *P<0.05, **P<0.01. ns: No differences; Con: Control; SSC: Side scatter; APC-Cy7-CD45: APC/Cyanine 7 anti-human CD45 antibody; PE-Cy7-CD3: PE/Cyanine 7 anti-human CD3 antibody; BV510-CD8: BV510 anti-human CD8 antibody.
2.3. 脓毒症组和对照组淋巴细胞亚群上proBDNF/p75NTR 的表达变化
进一步分析外周血中CD3+ T细胞、CD4+ T细胞、CD8+ T细胞和CD19+ B细胞上proBDNF/p75NTR在脓毒症组和对照组的表达水平差异(图2)。与对照组相比,脓毒症组患者外周血中T细胞上proBDNF的平均荧光强度(mean fluorescence intensity,MFI)差异无统计学意义(均P>0.05,图2A~2C),而脓毒症组CD19+ B细胞上proBDNF的MFI明显增加(P<0.05,图2D)。与proBDNF表达变化不同的是,受体p75NTR的MFI在脓毒症组患者外周血中CD4+ T细胞(P<0.01,图2F)、CD8+ T细胞(P<0.05,图2G)和CD19+ B细胞(P<0.01,图2H)均显著增加,而CD3+ T细胞来源的p75NTR MFI差异无统计学意义(P>0.05,图2E)。以上结果提示,脓毒症组患者外周血中CD19+ B细胞来源的proBDNF/p75NTR表达增加,CD4+ T细胞和CD8+ T细胞来源的受体p75NTR表达上调。
图2.
脓毒症组和对照组淋巴细胞亚群上proBDNF/p75NTR 的表达
Figure 2 Expression of proBDNF/p75NTR on lymphocytes subpopulations of the sepsis group and the control group A-D: ProBDNF MFI on CD3+ T cells (A), CD4+ T cells (B), CD8+ T cells (C), and CD19+ B cells (D). E-H: P75NTR MFI on CD3+ T cells (E), CD4+ T cells (F), CD8+ T cells (G), and CD19+ B cells (H). *P<0.05, **P<0.01. ns: No differences; Con: Control; MFI: Mean fluorescence intensity; proBDNF: Pro-brain-derived neurotrophic factor; p75NTR: P75 neurotrophin receptor.
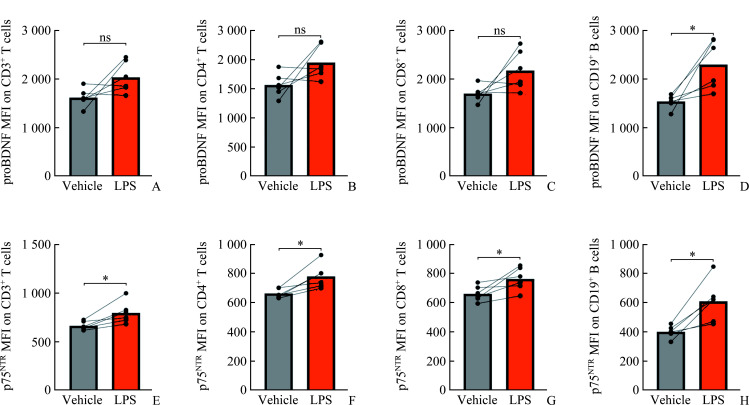
2.4. LPS激活外周血淋巴细胞上proBDNF/p75NTR 的表达
本研究继续采用LPS体外刺激培养健康对照组外周血淋巴细胞2 d,流式细胞术检测淋巴细胞各亚群的proBDNF/p75NTR的表达情况(图3)。与对照组(Vehicle)相比,LPS刺激促进B淋巴细胞上proBDNF MFI表达增加(P<0.05,图3D),而淋巴细胞来源的p75NTR的表达均上调(P<0.05,图3E~3H),这提示LPS刺激可诱导对照组外周血淋巴细胞来源的proBDNF/p75NTR表达上调。
图3.
LPS刺激上调健康对照组外周血淋巴细胞proBDNF/p75NTR 的表达
Figure 3 Expression of proBDNF/p75NTR of lymphocytes subpopulations in healthy control after LPS stimulation
A-D: ProBDNF MFI on CD3+ T cells (A), CD4+ T cells (B), CD8+ T cells (C), and CD19+ B cells (D). E-H: P75NTR MFI on CD3+ T cells (E), CD4+ T cells (F), CD8+ T cells (G), and CD19+ B cells (H). *P<0.05. ns: No differences; MFI: Median fluorescence intensity; proBDNF: Pro-brain-derived neurotrophic factor; p75NTR: P75 neurotrophin receptor; LPS: Lipopolysaccharide.
2.5. 拮抗p75NTR 对淋巴细胞分化的影响
最后采用p75ECD-Fc和对照Fc(Control-Fc)体外干预LPS刺激培养的健康对照组的外周血淋巴细胞,观察抑制p75NTR信号对淋巴细胞分化的影响。与Control-Fc组相比,p75ECD-Fc处理导致CD4+ T细胞百分比(P<0.05,图4A和4C)在CD3+ T淋巴细胞中的占比增加和CD19+ B细胞的百分比(P<0.05,图4A和4E)在CD45+白细胞中的占比增加。对CD3+ T细胞百分比在CD45+白细胞占比和CD8+ T细胞百分比在CD3+ T淋巴细胞中的占比无明显作用(图4A,4B,4D)。
图4.
P75ECD-Fc干预对淋巴细胞分化的影响
Figure 4 Effects of p75ECD-Fc therapy on lymphocyte differentiation
A: Method of lymphocyte subsets’ gating; B-E: Proportion of CD3+ (B), CD4+ (C), CD8+ (D) T cells, and CD19+ B cells (E) in the control-Fc group and the p75ECD-Fc group, respectively; F: Relative mRNA levels of lymphocyte-related cytokines. *P<0.05, **P<0.01, ***P<0.001. IFN-γ: Interferon-γ; p75ECD-Fc: Extracellular domain of p75NTR; TGF-β: Transforming growth factor-β; ns: No differences.
进一步通过RT-qPCR检测淋巴细胞亚群相关的细胞因子的变化,结果显示:与Control-Fc组相比,p75ECD-Fc处理增加IL-4(P<0.001)和IL-10的转录水平(P<0.05),降低IL-1β和IL-6的表达(均P<0.05,图4F)。对其他细胞因子无明显影响。这一结果提示抑制p75NTR可能调控外周淋巴细胞CD4+ T细胞和CD19+ B细胞的分化。
3. 讨 论
脓毒症的病理生理过程为坏死组织和病原体释放的损伤相关分子模式和病原体相关分子模式,与模式识别受体结合并激活先天性免疫系统,先天性免疫细胞开始高度增殖以清除机体内病原体,同时分泌细胞因子包括IL-1、IL-6、IL-10、肿瘤坏死因子-α(tumor necrosis factor-α,TNF-α)、血管活性肽、补体和活性氧等共同引发全身性炎症[11]。接着辅助CD4+ T细胞和细胞毒性CD8+ T细胞通过T细胞受体激活、分化和增殖作出高度特异性的适应性免疫应答[12]。
本研究发现脓毒症患者外周血中性粒细胞的数目和百分比显著增加,其原因可能与脓毒症期间炎症环境促进中性粒细胞的生成和释放[13]、中性粒细胞迁移障碍导致其滞留在循环系统[14]和凋亡抑制导致寿命延长[15]有关。同时,脓毒症患者淋巴细胞计数和百分比、CD3+ T细胞、CD4+ T细胞和CD19+ B细胞百分比呈现一致性的减少。其原因可能为脓毒症期间持续的抗原刺激和高水平细胞因子通过上调T淋巴细胞内半胱天冬酶8(caspase-8)和半胱天冬酶9(caspase-9)的表达[16],从而促进T淋巴细胞程序性死亡[17],同时抑制T淋巴细胞增殖活化,二者共同导致T细胞耗竭[18-19],表现为脓毒症晚期外周循环中T淋巴细胞的长期减少和功能障碍。脓毒症免疫抑制阶段B淋巴细胞发生进行性消耗[20]。此外,本研究还发现中性粒细胞与淋巴细胞比值及单核细胞与淋巴细胞比值在脓毒症患者中显著增加。研究[21]表明中性粒细胞与淋巴细胞比值、单核细胞与淋巴细胞比值以及血小板与淋巴细胞比值可作为评价危急重症患者的感染、高炎症水平以及预后的可靠标志物。这些结果均表明,高炎症水平和长期免疫抑制是脓毒症的重要病理特征,并伴随淋巴细胞免疫功能紊乱。
ProBDNF是BDNF基因的前体产物,该前体在高尔基体内装配后可被细胞内外的弗林蛋白酶或蛋白酶原转化酶裂解为成熟BDNF[22]。近年研究[23]发现proBDNF不仅是成熟BDNF的前体物质,同时也可通过与其高亲和力受体p75NTR结合发挥生物学效应。本课题组围绕proBDNF信号在LPS模拟的脓毒症动物模型中的作用研究[24]发现:proBDNF/p75NTR信号在LPS诱导的脓毒症模型中肠系膜淋巴结表达明显上调,其肠系膜皮质区域CD3+ T细胞和CD4+ T细胞来源的proBDNF上调尤为显著。此外,腹腔注射LPS促进脓毒症模型小鼠脑膜和外周血上调免疫细胞proBDNF,后者下调循环CD4+ T细胞比例,减少CD4+ T细胞向脑膜浸润,加重脑膜免疫微环境紊乱,从而促进脓毒症脑病病程[9]。抗proBDNF单克隆抗体干预可以改善脓毒症脑病模型小鼠的学习记忆障碍[9, 25]。本研究通过收集临床样本,观察proBDNF/p75NTR在脓毒症患者外周血淋巴细胞上的表达,初步证明proBDNF/p75NTR在脓毒症患者中的可能作用。
P75NTR在抗原呈递细胞非依赖性途径中参与T细胞受体介导的T细胞活化共刺激,增加T细胞增殖以及干扰素γ(interferon γ,IFN-γ)和TNF-α等细胞因子的分泌[26-27]。多发性硬化患者外周血中B淋巴细胞上proBDNF/p75NTR的表达上调,给予自主研发的抗proBDNF单克隆抗体干预可抑制B细胞增殖,其机制与负性调节p75NTR下游的核因子κB(nuclear factor-κB,NF-κB)信号激活有关[8]。中和proBDNF或条件性敲除B细胞上的p75NTR可抑制系统性红斑狼疮患者抗体分泌细胞的分化和抗体生成[7, 28]。本研究发现脓毒症患者外周血中CD4+ T细胞和CD8+ T细胞上p75NTR的表达上调,而B细胞上proBDNF/p75NTR表达上调。LPS体外刺激培养上调健康对照组外周血淋巴细胞proBDNF/p75NTR的表达,这一结果与脓毒症患者淋巴细胞上的改变基本一致。在此基础上,阻断p75NTR信号可增加CD4+ T细胞和CD19+ B细胞的百分比、促进细胞因子IL-4和IL-10的表达,并减少IL-1β和IL-6的产生。p75NTR主要激活下游3个信号转导通路(NF-κB、JNK、RhoA),参与调节免疫细胞的凋亡[29]、细胞因子的产生[30]和调控淋巴细胞功能[8]等生物学效应。采用高通量测序、RNA免疫共沉淀等技术可确定淋巴细胞在激活分化的条件下proBDNF/p75NTR信号与靶基因有关的调控机制。
综上所述,脓毒症患者入院时即处于以淋巴细胞数量减少为特征的免疫抑制阶段,同时伴有中性粒细胞占比增加。ProBDNF/p75NTR在脓毒症患者外周血淋巴细胞表达增加,抑制p75NTR可能通过调节淋巴细胞的分化,参与脓毒症进展。
基金资助
国家自然科学基金(82271379);湖南省杰出青年科学基金(2023JJ10088)。
This work was supported by the National Natural Science Foundation (82271379) and the Science Foundation of Hunan Province for Distinguished Young Scholars (2023JJ10088), China.
利益冲突声明
作者声称无任何利益冲突。
作者贡献
王双、曾秋明 实验操作,论文撰写;高海亮、高山 数据采集及统计分析;戴茹萍 论文指导;胡招兰 实验设计及指导,论文撰写及修改。所有作者阅读并同意最终的文本。
Footnotes
http://dx.chinadoi.cn/
原文网址
http://xbyxb.csu.edu.cn/xbwk/fileup/PDF/2023111629.pdf
参考文献
- 1. Rubio I, Osuchowski MF, Shankar-Hari M, et al. Current gaps in sepsis immunology: new opportunities for translational research[J/OL]. Lancet Infect Dis, 2019, 19(12): e422-e436[2023-05-02]. 10.1016/S1473-3099(19)30567-5. [DOI] [PubMed] [Google Scholar]
- 2. Martin MD, Badovinac VP, Griffith TS. CD4 T cell responses and the sepsis-induced immunoparalysis state[J]. Front Immunol, 2020, 11: 1364. 10.3389/fimmu.2020.01364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Inoue S, Suzuki-Utsunomiya K, Okada Y, et al. Reduction of immunocompetent T cells followed by prolonged lymphopenia in severe sepsis in the elderly[J]. Crit Care Med, 2013, 41(3): 810-819. 10.1097/CCM.0b013e318274645f. [DOI] [PubMed] [Google Scholar]
- 4. Hotchkiss RS, Tinsley KW, Swanson PE, et al. Sepsis-induced apoptosis causes progressive profound depletion of B and CD4+ T lymphocytes in humans[J]. J Immunol, 2001, 166(11): 6952-6963. 10.4049/jimmunol.166.11.6952. [DOI] [PubMed] [Google Scholar]
- 5. Monserrat J, de Pablo R, Diaz-Martín D, et al. Early alterations of B cells in patients with septic shock[J]. Crit Care, 2013, 17(3): R105. 10.1186/cc12750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Eggert S, Kins S, Endres K, et al. Brothers in arms: proBDNF/BDNF and sAPPα/Aβ-signaling and their common interplay with ADAM10, TrkB, p75NTR, sortilin, and sorLA in the progression of Alzheimer’s disease[J]. Biol Chem, 2021, 403(1): 43-71. 10.1515/hsz-2021-0330. [DOI] [PubMed] [Google Scholar]
- 7. Shen WY, Luo C, Hurtado PR, et al. Up-regulation of proBDNF/p75NTR signaling in antibody-secreting cells drives systemic lupus erythematosus[J]. Sci Adv, 2022, 8(3): eabj2797. 10.1126/sciadv.abj2797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Hu ZL, Luo C, Hurtado PR, et al. Brain-derived neurotrophic factor precursor in the immune system is a novel target for treating multiple sclerosis[J]. Theranostics, 2021, 11(2): 715-730. 10.7150/thno.51390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Luo RY, Luo C, Zhong F, et al. ProBDNF promotes sepsis-associated encephalopathy in mice by dampening the immune activity of meningeal CD4+ T cells[J]. J Neuroinflammation, 2020, 17(1): 169. 10.1186/s12974-020-01850-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. 中华医学会重症医学分会 . 中国严重脓毒症/脓毒性休克治疗指南(2014)[J]. 中华内科杂志, 2015, 54(6): 557-581. 10.3760/cma.j.issn.0578-1426.2015.06.021. [DOI] [Google Scholar]; Chinese Society of Critical Care Medicine . Chinese Guidelines for the Treatment of Severe Sepsis/Septic Shock (2014)[J]. Chinese Journal of Internal Medicine, 2015, 54(6): 557-581. 10.3760/cma.j.issn.0578-1426.2015.06.021. [DOI] [Google Scholar]
- 11. Fitzgerald KA, Kagan JC. Toll-like receptors and the control of immunity[J]. Cell, 2020, 180(6): 1044-1066. 10.1016/j.cell.2020.02.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Rodrigues PRS, Picco N, Morgan BP, et al. Sepsis target validation for repurposing and combining complement and immune checkpoint inhibition therapeutics[J]. Expert Opin Drug Discov, 2021, 16(5): 537-551. 10.1080/17460441.2021.1851186. [DOI] [PubMed] [Google Scholar]
- 13. Eash KJ, Greenbaum AM, Gopalan PK, et al. CXCR2 and CXCR4 antagonistically regulate neutrophil trafficking from murine bone marrow[J]. J Clin Invest, 2010, 120(7): 2423-2431. 10.1172/JCI41649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. de Kleijn S, Kox M, Sama IE, et al. Transcriptome kinetics of circulating neutrophils during human experimental endotoxemia[J/OL]. PLoS One, 2012, 7(6): e38255[2023-05-01]. 10.1371/journal.pone.0038255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Luan YY, Yao YM, Xiao XZ, et al. Insights into the apoptotic death of immune cells in sepsis[J]. J Interferon Cytokine Res, 2015, 35(1): 17-22. 10.1089/jir.2014.0069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hotchkiss RS, Swanson PE, Knudson CM, et al. Overexpression of Bcl-2 in transgenic mice decreases apoptosis and improves survival in sepsis[J]. J Immunol, 1999, 162(7): 4148-4156. [PubMed] [Google Scholar]
- 17. Auffray C, Fogg D, Garfa M, et al. Monitoring of blood vessels and tissues by a population of monocytes with patrolling behavior[J]. Science, 2007, 317(5838): 666-670. 10.1126/science.1142883. [DOI] [PubMed] [Google Scholar]
- 18. Sjaastad FV, Condotta SA, Kotov JA, et al. Polymicrobial sepsis chronic immunoparalysis is defined by diminished Ag-specific T cell-dependent B cell responses[J]. Front Immunol, 2018, 9: 2532. 10.3389/fimmu.2018.02532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Boomer JS, To K, Chang KC, et al. Immunosuppression in patients who die of sepsis and multiple organ failure[J]. JAMA, 2011, 306(23): 2594-2605. 10.1001/jama.2011.1829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Gustave CA, Gossez M, Demaret J, et al. Septic shock shapes B cell response toward an exhausted-like/immunoregulatory profile in patients[J]. J Immunol, 2018, 200(7): 2418-2425. 10.4049/jimmunol.1700929. [DOI] [PubMed] [Google Scholar]
- 21. Ng WW, Lam SM, Yan WW, et al. NLR, MLR, PLR and RDW to predict outcome and differentiate between viral and bacterial pneumonia in the intensive care unit[J]. Sci Rep, 2022, 12(1): 15974. 10.1038/s41598-022-20385-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Foltran RB, Diaz SL. BDNF isoforms: a round trip ticket between neurogenesis and serotonin?[J]. J Neurochem, 2016, 138(2): 204-221. 10.1111/jnc.13658. [DOI] [PubMed] [Google Scholar]
- 23. Gibon J, Barker PA. Neurotrophins and proneurotrophins: focus on synaptic activity and plasticity in the brain[J]. Neuroscientist, 2017, 23(6): 587-604. 10.1177/1073858417697037. [DOI] [PubMed] [Google Scholar]
- 24. Wang Z, Wu JL, Zhong F, et al. Upregulation of proBDNF in the mesenteric lymph nodes in septic mice[J]. Neurotox Res, 2019, 36(3): 540-550. 10.1007/s12640-019-00081-3. [DOI] [PubMed] [Google Scholar]
- 25. Cui YH, Zhou SF, Liu Y, et al. Injection of anti-proBDNF attenuates hippocampal-dependent learning and memory dysfunction in mice with sepsis-associated encephalopathy[J]. Front Neurosci, 2021, 15: 665757. 10.3389/fnins.2021.665757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Kim EY, Priatel JJ, Teh SJ, et al. TNF receptor type 2 (p75) functions as a costimulator for antigen-driven T cell responses in vivo[J]. J Immunol, 2006, 176(2): 1026-1035. 10.4049/jimmunol.176.2.1026. [DOI] [PubMed] [Google Scholar]
- 27. Aspalter RM, Eibl MM, Wolf HM. Regulation of TCR-mediated T cell activation by TNF-RII[J]. J Leukoc Biol, 2003, 74(4): 572-582. 10.1189/jlb.0303112. [DOI] [PubMed] [Google Scholar]
- 28. Zha AH, Luo C, Shen WY, et al. Systemic blockade of proBDNF inhibited the expansion and altered the transcriptomic expression in CD3+B220+ cells in MRL/lpr lupus mice[J/OL]. Lupus Sci Med, 2022, 9(1): e000836[2023-05-02]. 10.1136/lupus-2022-000836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Fauchais AL, Lalloué F, Lise MC, et al. Role of endogenous brain-derived neurotrophic factor and sortilin in B cell survival[J]. J Immunol, 2008, 181(5): 3027-3038. 10.4049/jimmunol.181.5.3027. [DOI] [PubMed] [Google Scholar]
- 30. Düsedau HP, Kleveman J, Figueiredo CA, et al. p75NTR regulates brain mononuclear cell function and neuronal structure in Toxoplasma infection-induced neuroinflammation[J]. Glia, 2019, 67(1): 193-211. 10.1002/glia.23553. [DOI] [PMC free article] [PubMed] [Google Scholar]