Abstract
OBJECTIVES:
This systematic review and Bayesian network meta-analysis evaluated the efficacy and safety of hydrocortisone combined with fludrocortisone or hydrocortisone alone, compared with placebo in adult patients with septic shock.
DATA SOURCES:
By extending a prior Cochrane review, databases, including PubMed, Embase, the Cochrane Library, and ClinicalTrials.gov, along with other relevant websites, were searched until August 31, 2023.
STUDY SELECTION:
Randomized controlled trials (RCTs) and observational studies using target trial emulation were included.
DATA EXTRACTION:
The primary outcome was short-term mortality with an emphasis on 28- or 30-day mortality as the main measure and in-hospital or ICU mortality as the nearest surrogate of this measure. Three of the most common adverse events, namely, gastroduodenal bleeding, superinfection, and hyperglycemia, were also considered.
DATA SYNTHESIS:
A total of 19 studies involving 95,841 patients were included. Hydrocortisone plus fludrocortisone showed the lowest short-term mortality versus placebo (odds ratio [OR]: 0.79; 95% credible interval [CrI], 0.64–0.99; number needed to treat [NNT]: 21, range: 12–500; low certainty of evidence) in terms of informative priors. The surface under the cumulative ranking curve values for hydrocortisone plus fludrocortisone, hydrocortisone alone, and placebo were 0.9469, 0.4542, and 0.0989, respectively. Consistent results were observed in RCTs alone and those using a daily 200-mg dose of hydrocortisone. Although gastroduodenal bleeding or superinfection showed no clear increase, hyperglycemia risk increased. The ORs were 0.53 for placebo versus hydrocortisone plus fludrocortisone and 0.64 for placebo versus hydrocortisone alone, with very low certainty of evidence.
CONCLUSIONS:
In adults with septic shock, hydrocortisone plus fludrocortisone improved short-term survival with minimal adverse events compared with hydrocortisone alone or placebo. However, these findings are not definitive due to the limited certainty of evidence and wide NNT range. Additional large-scale, placebo-controlled RCTs are needed to provide conclusive evidence.
Keywords: corticosteroids, fludrocortisone, hydrocortisone, mineralocorticoids, network meta-analysis, septic shock
KEY POINTS.
Question: Is administering fludrocortisone in addition to hydrocortisone to adult patients with septic shock necessary?
Findings: We conducted an updated systematic review using Bayesian network meta-analysis to evaluate the efficacy and safety of hydrocortisone plus fludrocortisone, hydrocortisone alone, or placebo in adult patients with septic shock. Among the tested therapies, hydrocortisone plus fludrocortisone showed the highest likelihood of improving short-term survival.
Meaning: On the basis of the aforementioned evidence, the combined administration of hydrocortisone and fludrocortisone may be considered for adult patients with septic shock.
Despite the implementation of aggressive treatment approaches, septic shock continues to exhibit high mortality rates exceeding 30% in numerous studies (1). This is mainly result from activation of proinflammatory and procoagulant pathways (2). Corticosteroids have been considered as a potential strategy for modulating the dysregulated immune response associated with septic shock.
Growing concerns among intensivists are centered around critical-illness-related corticosteroid and mineralocorticoid deficiencies (3, 4). The International Guideline of Surviving Sepsis Campaign conditionally recommended administration of IV hydrocortisone at a typical dose of 200 mg/d to adult patients with septic shock necessitating vasopressor support (5). However, the 2020 Japanese Clinical Practice Guidelines for Sepsis and Septic Shock proposed the adjunctive use of fludrocortisone with hydrocortisone (6) based on a meta-analysis of two randomized controlled trials (RCTs) (7). The differences between these two recommendations warrant a thorough evaluation.
Hydrocortisone possesses glucocorticoid and mineralocorticoid properties. Fludrocortisone, a synthetic mineralocorticoid, is used to substitute for endogenous aldosterone under conditions of absent or insufficient endogenous synthesis. The comparative activity of hydrocortisone to that of fludrocortisone is approximately 1 to 125–150, a ratio that is primarily derived from the effects of hydrocortisone and fludrocortisone on sodium retention in the kidney (8, 9). Differences in the mineralocorticoid effects of hydrocortisone and fludrocortisone beyond this point have been reported in numerous investigations. In patients with sepsis, mineralocorticoid steroidogenesis is more impaired than glucocorticoid synthesis, and this deficiency is associated with sepsis nonsurvival (10). Therefore, the potential benefits of fludrocortisone in treating septic shock are beginning to be highly valued and are believed to stem from the restoration of homeostasis and augmentation of innate immunity (11). Thus, rigorously assessing the combined use of fludrocortisone and hydrocortisone in treating septic shock in adults is imperative.
MATERIALS AND METHODS
Literature Search Strategy
For assessing the efficacy and safety of hydrocortisone plus fludrocortisone, hydrocortisone alone, and placebo in adult patients with septic shock, we conducted a systematic review with network meta-analysis (NMA) by following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) 2020 guideline (12). Our research protocols were registered on INPLASY (registration number: INPLASY202360053, https://inplasy.com/inplasy-2023-6-0053/) and were updated on July 30, 2023, and August 31, 2023. By drawing from a Cochrane review (12), investigators P.C.L. and Y.T.H. searched databases, such as PubMed, Embase, Cochrane Library, and ClinicalTrials.gov, along with other relevant websites until August 31, 2023. We used free text and medical subject headings with keywords, such as “septic shock,” “hydrocortisone,” and “fludrocortisone,” without language restriction. Only parallel RCTs and observational studies with target trial emulation (TTE) were included. Discrepancies were addressed through group consensus.
Study Eligibility and Exclusion Criteria
We included studies based on the following criteria: 1) population: critically ill adults with septic shock, 2) exposure: administration of hydrocortisone with or without fludrocortisone, or placebo, and 3) outcome: data on 28- or 30-day mortality, in-hospital and ICU mortality, or adverse events. We excluded: 1) reviews and case reports/series, 2) studies on sepsis without shock or vasopressor usage, 3) pediatric studies, 4) studies with data insufficient for meta-analysis, and 5) articles with unavailable full texts.
Risk-of-Bias Assessments
P.C.L. and Y.T.H. also assessed the quality of RCTs by using the revised Cochrane Risk of Bias (RoB 2.0) tool (13). Quality assessment was performed on non-RCT literature by applying the Risk of Bias in Nonrandomized Studies of Interventions (ROBINS-I) tool (14). Any discrepancies were resolved through group consensus. The results from RoB 2.0 and ROBINS-I were illustrated using the Risk-of-Bias Visualization tool (15).
Data Extraction and Outcome Measures
Two investigators (P.C.L. and Y.T.H.) independently extracted data, capturing study details and treatment strategies. Our primary focus is short-term mortality, specifically, 28- or 30-day mortality. In the absence of short-term mortality data, in-hospital or subsequent ICU mortality was used as the closest available surrogate for assessing patient outcomes. We also evaluated the three main adverse poststeroid administration events: gastroduodenal bleeding, superinfection, and hyperglycemia.
Statistical Analysis
We used NetMetaXL, V.1.6.1 (Canadian Agency for Drugs and Technologies in Health, Ottawa, Canada) alongside WinBUGS 1.4.3 (MRC Biostatistics Unit, Cambridge, and Imperial College, London, United Kingdom) adopting a Bayesian method with 10,000 simulations and a random-effects model with informative priors (16). Our sensitivity analysis used vague priors. Subgroup analysis focused on: 1) RCTs exclusively and 2) RCTs using 200 mg/d hydrocortisone as per the Surviving Sepsis Campaign guideline. Treatment effects were presented as odds ratios (OR) with 95% credible intervals (CrIs). Treatment rankings were obtained from surface under the cumulative ranking curve analysis (SUCRA), with high values denoting good outcomes.
Certainty of Evidence Assessment
We appraised evidence certainty for all outcomes by using the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) Working Group guideline for NMA (17) and the guideline for integrating RCTs and non-RCTs in evidence syntheses (18). We performed grading with GRADEpro software (gradepro.org). The number needed to treat (NNT) for short-term mortality was derived from the absolute risk reduction presented in the GRADE table.
RESULTS
Study Selection and Characteristics
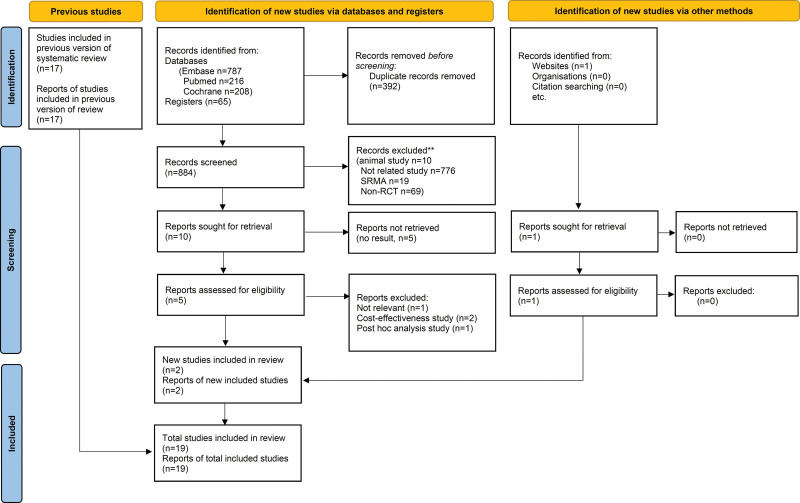
By following the PRISMA 2020 flow diagram and as detailed in Figure 1, we identified 17 RCTs through a Cochrane systematic review (12). We excluded two studies that were available only as abstracts (19, 20), one with a cross-over design (21), and one focusing on severe sepsis without septic shock (22). Our final analysis comprised 13 RCTs assessing low doses of hydrocortisone versus placebo (23–35) and three assessing hydrocortisone plus fludrocortisone versus placebo (36–38). Additionally, we included one RCT (39) and a TTE-based cohort study (40) to assess the efficacy of hydrocortisone plus fludrocortisone versus hydrocortisone alone. We also identified a three-armed RCT (41). The detailed summaries and maps of these studies are presented in Supplementary Tables 1 and 2 (http://links.lww.com/CCM/H472), respectively.
Figure 1.
Search in accordance with the flow diagram of Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020. RCT = randomized controlled trial, SRMA = systematic review and meta-analysis.
Risk-of-Bias Assessments
The overall risks of bias were rated as high, of some concern, and low in 4, 2, and 12 RCTs, respectively (Supplementary Fig. 1A, http://links.lww.com/CCM/H472). The risk of attrition bias was rated as high in one RCT (30). Three RCTs were assigned a high overall risk of bias due to risk of some concern in two domains (23, 28, 31). The cohort study using TTE was evaluated as having a serious risk of bias due to the moderate risk present in three domains (Supplementary Fig. 1B, http://links.lww.com/CCM/H472).
Primary Outcome: Short-Term Mortality
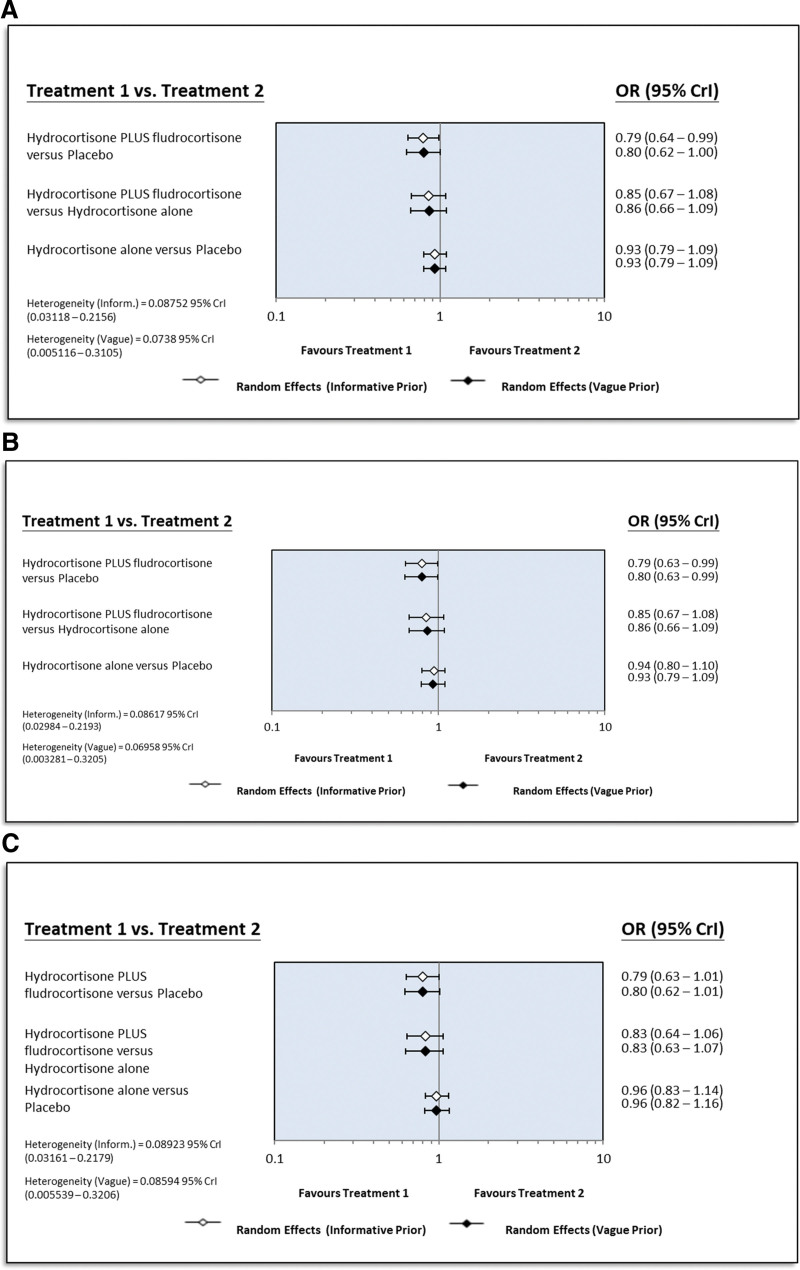
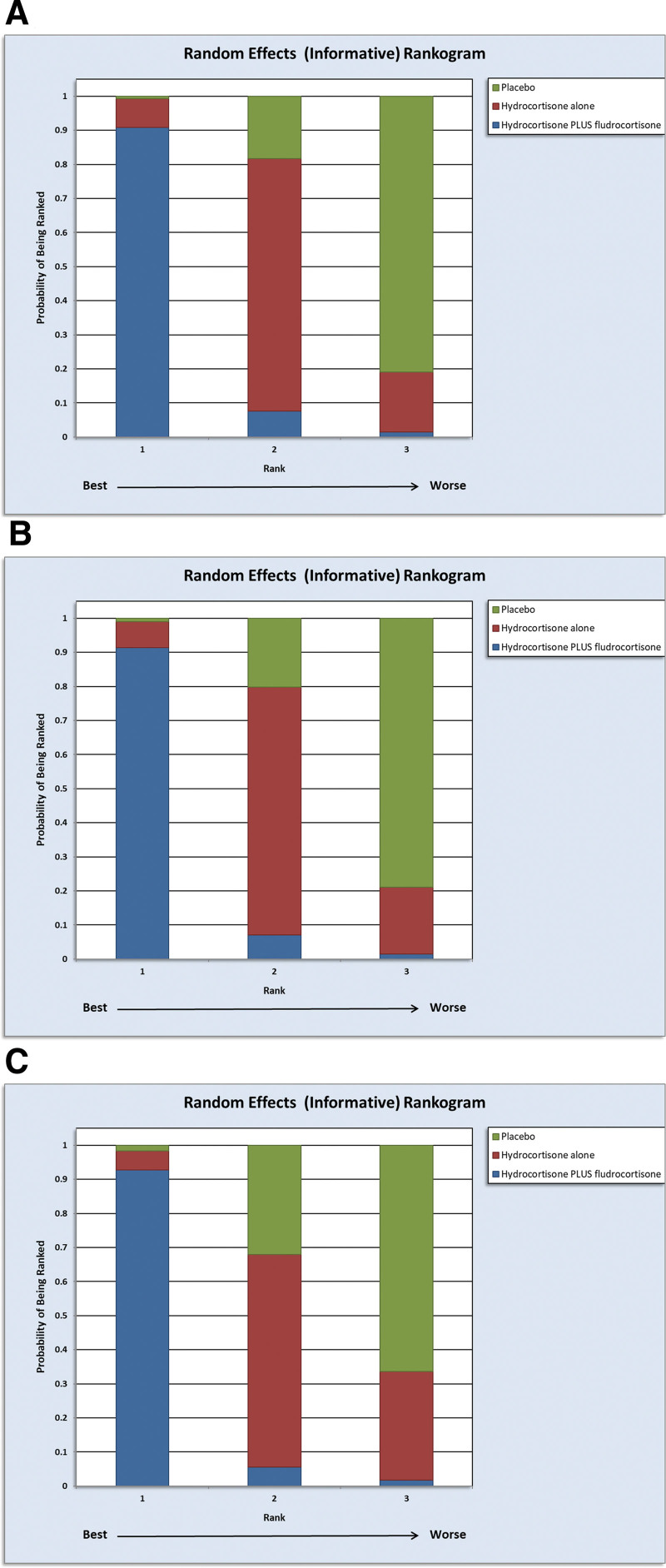
Data from 19 studies and 95,841 patients (shown in Fig. 2A) revealed that hydrocortisone plus fludrocortisone reduced the risk of short-term mortality. In informative and vague priors, the reduction had an OR of 0.79 (95% CrI, 0.64–0.99) and 0.80 (95% CrI, 0.62–1.00) relative to the placebo. In the comparison of hydrocortisone plus fludrocortisone with hydrocortisone alone, the ORs were 0.85 (95% CrI, 0.67–1.08) using informative priors and 0.86 (95% CrI, 0.66–1.09) using vague priors. Hydrocortisone plus fludrocortisone appeared to be more effective than hydrocortisone alone. The probability that substantive benefit is absent is small. Hydrocortisone monotherapy exhibited marginal benefits over placebo (OR: 0.93; 95% CrI, 0.79–1.09 using informative and vague priors). The SUCRA evaluations distinctly favor hydrocortisone plus fludrocortisone (score: 0.9469) over hydrocortisone alone (score: 0.4542) and placebo (score: 0.0989). The rankogram in Figure 3A further emphasizes the therapeutic superiority of hydrocortisone plus fludrocortisone.
Figure 2.
Forest plots presenting the odds ratios for short-term mortality among treatment groups receiving either combined hydrocortisone and fludrocortisone, hydrocortisone alone, or placebo: includes all studies (A); has been narrowed down to randomized controlled trials (B); and specifically examines randomized controlled trials administering an initial daily hydrocortisone dosage of 200 mg (C). CrI = credible interval, OR = odds ratio.
Figure 3.
Rankogram illustrating the probability rankings for improving short-term mortality across treatments: combined hydrocortisone and fludrocortisone, hydrocortisone alone, or placebo: encompasses all studies (A); is limited to randomized controlled trials (B); and highlights randomized controlled trials starting with a daily hydrocortisone dose of 200 mg (C).
In accordance with the GRADE Working Group’s guideline on integrating evidence from RCTs and non-RCTs, the initial evaluation of evidence derived from RCTs is recommended (18). The forest plots in Figure 2B and the rankogram in Figure 3B reveal that the NMA limited to RCTs closely aligns with the results obtained by combining RCTs and cohort studies using TTE. Hydrocortisone plus fludrocortisone is the top treatment choice, with a SUCRA score of 0.9501, surpassing hydrocortisone alone (0.4397) and placebo (0.1102).
The GRADE Working Group advises prioritizing RCTs due to consistent results when determining evidence certainty (18). In contrast to placebo, hydrocortisone plus fludrocortisone and hydrocortisone alone received a low certainty rating (Supplementary Table 2, http://links.lww.com/CCM/H472). The main downgrade reason was imprecision (18), with 95% CrIs approaching the minimally important difference threshold (defined as an OR of 0.9) (Supplementary Table 3, http://links.lww.com/CCM/H472). The NNT of hydrocortisone plus fludrocortisone compared with placebo was 21, with a range of 12–500.
Figures 2C and 3C underscore the superior efficacy of hydrocortisone plus fludrocortisone (SUCRA score: 0.9546) over that of hydrocortisone (0.3693) or placebo (0.1761) in the RCT analysis involving 7432 patients from 15 RCTs using 200 mg/d hydrocortisone.
Adverse Events
Gastroduodenal Bleeding
Eight RCTs involving 2023 participants examined gastroduodenal bleeding. The wide 95% CrIs obtained when comparing hydrocortisone plus fludrocortisone with placebo, hydrocortisone plus fludrocortisone with hydrocortisone alone, and hydrocortisone alone with placebo failed to highlight any obvious difference in gastroduodenal bleeding risk among these treatments (Supplementary Fig. 2A, http://links.lww.com/CCM/H472). The certainty of evidence was assessed as very low (Supplementary Table 4, http://links.lww.com/CCM/H472). In the rankogram in Supplementary Figure 2B (http://links.lww.com/CCM/H472), hydrocortisone plus fludrocortisone ranked the lowest among all treatments for the occurrence of gastroduodenal bleeding. In terms of SUCRA scores, hydrocortisone plus fludrocortisone (0.7052) had a similar rank as placebo (0.6758), but was notably ranked higher than hydrocortisone alone (0.119). This result suggests that hydrocortisone plus fludrocortisone could be the least favorable option when considering the risk of gastroduodenal bleeding.
Superinfection
The same eight RCTs involving 2023 participants were included in the NMA for the risk of superinfection. Once again, the expansive 95% CrIs in forest plots illustrating the comparison of hydrocortisone plus fludrocortisone with placebo, hydrocortisone plus fludrocortisone with hydrocortisone alone, and hydrocortisone alone with placebo, suggest the absence of a clear distinction in superinfection rates (Supplementary Fig. 3A, http://links.lww.com/CCM/H472) with a very low certainty of evidence (Supplementary Table 5, http://links.lww.com/CCM/H472). In the rankogram in Supplementary Figure 3B (http://links.lww.com/CCM/H472), placebo, followed by hydrocortisone plus fludrocortisone, ranked the lowest for the occurrence of superinfection. In accordance with SUCRA scores, placebo (0.7522) showed a slightly higher ranking than hydrocortisone plus fludrocortisone (0.5964) and apparently ranked higher than hydrocortisone alone (0.1514). This result hints that hydrocortisone plus fludrocortisone might not elevate superinfection risk.
Hyperglycemia
In seven RCTs involving 5365 patients assessing hyperglycemia risk, the placebo group had a lower likelihood for hyperglycemia than the hydrocortisone plus fludrocortisone group (OR: 0.53 [95% CrI, 0.37–0.73] using informative priors, Supplementary Fig. 4A, http://links.lww.com/CCM/H472) with moderate evidence certainty (Supplementary Table 6, http://links.lww.com/CCM/H472). The comparison between hydrocortisone alone and hydrocortisone plus fludrocortisone revealed broad CrIs (OR: 0.82 [95% CrI, 0.48–1.37] using informative priors; additional details in Supplementary Fig. 4A, http://links.lww.com/CCM/H472). The comparison of placebo with hydrocortisone alone showed that the former had decreased odds for hyperglycemia (OR: 0.64 [95% Crl, 0.42–0.96]) albeit with very low evidence certainty (Supplementary Table 6, http://links.lww.com/CCM/H472). SUCRA scores and rankograms consistently showed that among the investigated therapies, hydrocortisone plus fludrocortisone, which had the lowest score of 0.1073, might be the most likely to cause hyperglycemia. Among the investigated therapies, hydrocortisone alone was the next most likely to cause hyperglycemia with a score of 0.4003, and placebo had the least likelihood to cause hyperglycemia with a score of 0.9924 (Supplementary Fig. 4B, http://links.lww.com/CCM/H472).
DISCUSSION
Our NMAs show that hydrocortisone plus fludrocortisone improves short-term mortality outcomes for septic shock in adults, whereas hydrocortisone alone has limited effects. Mineralocorticoids have roles beyond sodium retention. Heming et al (42) have highlighted how these substances affect immune responses, which we discuss further below. Different mineralocorticoid receptor (MR) isoforms exist. Some bind to glucocorticoids and mineralocorticoids, whereas others bind only to mineralocorticoids. The deactivation of glucocorticoids by 11β-hydroxysteroid dehydrogenase ensures that mineralocorticoids have strong MR interactions. Additionally, MRs are present in immune cells, and their activation enhances leukocyte adhesion, aiding in bacterial infection eradication. Experimental sepsis studies have also demonstrated that mineralocorticoids reduce the plasma levels of histamine, serotonin, and bradykinin, thereby accelerating the reversal of shock. These findings support the conclusion that hydrocortisone plus fludrocortisone is the most effective treatment for improving short-term mortality, as evidenced by our NMA results.
A recent study performed a fixed-effects NMA using a frequentist approach to compare hydrocortisone administered as an IV bolus or via continuous infusion with hydrocortisone plus fludrocortisone (43). Consistent with our data, the combination reduced 90-day all-cause mortality compared with the control, yielding a risk ratio (RR) of 0.88 (95% CI, 0.81–0.97). We did not assess long-term mortality due to the myriad factors influencing septic shock in adults. The potential of hydrocortisone plus fludrocortisone to improve 90-day mortality further strengthens the case for its use in adult patients with septic shock. We used a Bayesian approach in our statistical analysis because of its strong alignment with clinical reasoning. Bayesian analysis has been recognized as a practical methodology for interpreting clinical trials and formulating clinical practice guidelines (44). Direct comparisons between hydrocortisone plus fludrocortisone and hydrocortisone alone are limited. In our NMA, we identified only two RCTs and one TTE-style study. By using indirect data from the network of studies that compared hydrocortisone plus fludrocortisone and hydrocortisone alone with placebo, we strengthened the evidence base for comparing the two treatment options directly. Our NMA results demonstrate that hydrocortisone plus fludrocortisone is more likely to reduce short-term mortality than hydrocortisone alone as indicated by the 95% CrIs and SUCRA values. The aforementioned frequentist NMA (43) included only a single RCT that compared the combination regimen with hydrocortisone alone. Generally, large sample sizes enhance study power by providing clear effect size estimates and minimizing chance results, a principle that is equally applicable to (network) meta-analysis (45). A notable feature of our NMA is the inclusion of a three-armed RCT (41) and a recent cohort study (88,275 patients) with TTE (40). The use of TTE in observational studies stimulates researchers to formulate research questions for hypothetical RCTs that compare drug effectiveness (46). Therefore, we believe that enrolling an increased number of cases in this topic will lead to pooled estimates with increased precision.
Our findings indicate that hydrocortisone alone demonstrated marginal benefits over placebo. In a systematic review conducted in 2019, Siddiqui et al (47) concluded that the administration of hydrocortisone significantly reduced the 28-day mortality rate among patients with refractory septic shock. However, their results were based on a meta-analysis of only five RCTs. Systematic reviews with meta-analyses published after 2018 have shown inconsistent results regarding short-term mortality linked to hydrocortisone use in patients with sepsis (12, 47–50). These disparities may arise from factors, such as the use of different steroids, application of varied statistical methods, and inclusion of participants beyond just adults with septic shock. Of course, the effect of hydrocortisone alone may be minimal, which might cause fluctuations in the results under the influence of the aforementioned covariates. Pirracchio et al (43) reported a patient-level meta-analysis of low-dose hydrocortisone therapy in patients with septic shock. The comparison of hydrocortisone with placebo by using individual patient data (IPD) yielded marginal results with an RR of 0.93 and 95% CIs of 0.82–1.04 for 90-day mortality. IPD meta-analysis typically yields considerable enhancements in the quantity and quality of data through increased statistical power, the ability to adjust for confounding factors, broadened analytical possibilities, and the generation of results with increased comprehensiveness and robustness (51). The results mentioned above are consistent with the findings of our Bayesian NMA. This consistency prompts us to reconsider whether the guidelines for the treatment of septic shock in adults should be revised to recommend the use of hydrocortisone plus fludrocortisone instead of hydrocortisone alone.
Previous Bayesian NMAs have suggested that a daily dosage of 200–400 mg of hydrocortisone or equivalent steroids is likely to provide the greatest benefit to patients with sepsis (52). We also attempted to analyze the effect of various corticosteroids on short-term mortality by using NMA. We identified only two additional three-armed RCTs comparing dexamethasone, methylprednisolone, and placebo that were published in 1976 (53) and 1984 (54). These studies are limited by their methodologies, which are outdated relative to current critical care practices. Thus, caution is advised when interpreting the NMA results indicating potential short-term mortality benefits from dexamethasone and methylprednisolone treatments due to intransitivity concerns (Supplementary Fig. 5, http://links.lww.com/CCM/H472).
Our study has limitations. First, we only considered adults. Whether different corticosteroids have varying effects on pediatric patients with septic shock cannot be determined from our study. If RCTs examining fludrocortisone alone exist, then conducting a component NMA could provide an in-depth understanding of the roles of fludrocortisone in septic shock management. It is worth noting that most of the RCTs investigating hydrocortisone plus fludrocortisone originated from the same group and geographic region. Expanding research to include studies from diverse regions and research teams is crucial for a more comprehensive understanding of the efficacy and generalizability of this treatment strategy. Furthermore, we excluded certain outcomes, such as time to wean off vasopressors, ICU stay length, ventilator-free days, and kidney replacement needs, given their variability based on institutional practices. Another constraint was our focus on just three adverse events of high prevalence because events with low incidences can reduce the robustness of NMA results. Notably, we found elevated risks of hyperglycemia under treatments with hydrocortisone plus fludrocortisone and hydrocortisone alone. Assessing the effect of steroid-induced hyperglycemia through NMA remains challenging.
CONCLUSIONS
In adults with septic shock, hydrocortisone plus fludrocortisone improves short-term survival with an increased risk of hyperglycemia. Given the affordability and accessibility of fludrocortisone, its coadministration with IV hydrocortisone is an appropriate option for patients receiving enteral nutrition. Nevertheless, the uncertainty and wide NNT range for short-term mortality underscore the urgent need for well-conducted, triple-blinded RCTs to provide clear guidance.
ACKNOWLEDGMENTS
We express our gratitude to the English editors at KG Support for their valuable assistance in language editing.
Supplementary Material
Footnotes
*See also p. 678.
Dr. Lai contributed to database search, data extraction, and drafting of the article. Dr. Lai contributed to the design and process of this research and editing of the article. Dr. Lai contributed to consultation in concepts, methodologies, and interpretation of the data. Dr. Huang contributed to database search, data extraction, statistical analyses, and drafting of the article. All authors approved the final revision of the article.
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal’s website (http://journals.lww.com/ccmjournal).
Grant-in-aid from the National Cheng Kung University Hospital, Tainan, Taiwan (NCKUH-11209002 and NCKUH-11210042).
Dr. Huang received support for article research from the National Cheng Kung University Hospital, Tainan, Taiwan (NCKUH-11209002). The remaining authors have disclosed that they do not have any potential conflicts of interest.
This article is original, has not been previously published in any journal, and is not currently under consideration by any other journal.
All authors of the article have read and agreed to its content, and they are responsible for all aspects of the article’s accuracy and integrity in accordance with International Committee of Medical Journal Editors criteria.
The datasets supporting the conclusions of this article are included within the article itself and its additional files.
REFERENCES
- 1.Bauer M, Gerlach H, Vogelmann T, et al. : Mortality in sepsis and septic shock in Europe, North America and Australia between 2009 and 2019—results from a systematic review and meta-analysis. Crit Care 2020; 24:239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gotts JE, Matthay MA: Sepsis: Pathophysiology and clinical management. BMJ 2016; 353:i1585. [DOI] [PubMed] [Google Scholar]
- 3.Fowler C, Raoof N, Pastores SM: Sepsis and adrenal insufficiency. J Intensive Care Med 2023; 38:987–996 [DOI] [PubMed] [Google Scholar]
- 4.Nethathe GD, Lipman J, Anderson R, et al. : CIRMI—a new term for a concept worthy of further exploration: A narrative review. Ann Transl Med 2022; 10:646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Evans L, Rhodes A, Alhazzani W, et al. : Surviving sepsis campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Crit Care Med 2021; 49:e1063–e1143 [DOI] [PubMed] [Google Scholar]
- 6.Egi M, Ogura H, Yatabe T, et al. : The Japanese clinical practice guidelines for management of sepsis and septic shock 2020 (J-SSCG 2020). J Intensive Care 2021; 9:53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yamamoto R, Nahara I, Toyosaki M, et al. : Hydrocortisone with fludrocortisone for septic shock: A systematic review and meta-analysis. Acute Med Surg 2020; 7:e563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liu D, Ahmet A, Ward L, et al. : A practical guide to the monitoring and management of the complications of systemic corticosteroid therapy. Allergy Asthma Clin Immunol 2013; 9:30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schimmer BP, Funder JW: Adrenocorticotropic hormone, adrenal steroids, and the adrenal cortex. In: Goodman & Gilman’s: The Pharmacological Basis of Therapeutics. Thirteen Edition. Brunton LL, Hilal-Dandan R, Knollmann BC. (Eds). New York, McGraw-Hill, 2017. Available at: https://accessmedicine.mhmedical.com/content.aspx?bookid=2189§ionid=172482605. Accessed October 28, 2023 [Google Scholar]
- 10.Briegel J, Mohnle P, Keh D, et al. ; SepNet Critical Care Trials Group: Corticotropin-stimulated steroid profiles to predict shock development and mortality in sepsis: From the HYPRESS study. Crit Care 2022; 26:343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Matthay MA, Dahabreh IJ, Thompson BT: Should we add fludrocortisone to hydrocortisone for treatment of septic shock? JAMA Intern Med 2023; 183:460–461 [DOI] [PubMed] [Google Scholar]
- 12.Annane D, Bellissant E, Bollaert PE, et al. : Corticosteroids for treating sepsis in children and adults. Cochrane Database Syst Rev 2019; 12:CD002243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sterne JAC, Savovic J, Page MJ, et al. : RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019; 366:l4898. [DOI] [PubMed] [Google Scholar]
- 14.Sterne JA, Hernan MA, Reeves BC, et al. : ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016; 355:i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.McGuinness LA, Higgins JPT: Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res Synth Methods 2021; 12:55–61 [DOI] [PubMed] [Google Scholar]
- 16.Brown S, Hutton B, Clifford T, et al. : A microsoft-excel-based tool for running and critically appraising network meta-analyses—an overview and application of NetMetaXL. Syst Rev 2014; 3:110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Brignardello-Petersen R, Bonner A, Alexander PE, et al. ; GRADE Working Group: Advances in the GRADE approach to rate the certainty in estimates from a network meta-analysis. J Clin Epidemiol 2018; 93:36–44 [DOI] [PubMed] [Google Scholar]
- 18.Cuello-Garcia CA, Santesso N, Morgan RL, et al. : GRADE guidance 24 optimizing the integration of randomized and non-randomized studies of interventions in evidence syntheses and health guidelines. J Clin Epidemiol 2022; 142:200–208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mirea L, Ungureanu R, Pavelescu D, et al. : Continuous administration of corticosteroids in septic shock can reduce risk of hypernatremia. Crit Care 2014; 18:P239 [Google Scholar]
- 20.Chawla K, Kupfer Y, Goldman I, et al. : Hydrocortisone reverses refractory septic shock. Crit Care Med 1999; 27 (Suppl):33A [Google Scholar]
- 21.Keh D, Boehnke T, Weber-Cartens S, et al. : Immunologic and hemodynamic effects of “low-dose” hydrocortisone in septic shock: A double-blind, randomized, placebo-controlled, crossover study. Am J Respir Crit Care Med 2003; 167:512–520 [DOI] [PubMed] [Google Scholar]
- 22.Keh D, Trips E, Marx G, et al. ; SepNet–Critical Care Trials Group: Effect of hydrocortisone on development of shock among patients with severe sepsis: The HYPRESS randomized clinical trial. JAMA 2016; 316:1775–1785 [DOI] [PubMed] [Google Scholar]
- 23.Arabi YM, Aljumah A, Dabbagh O, et al. : Low-dose hydrocortisone in patients with cirrhosis and septic shock: A randomized controlled trial. CMAJ 2010; 182:1971–1977 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bollaert PE, Charpentier C, Levy B, et al. : Reversal of late septic shock with supraphysiologic doses of hydrocortisone. Crit Care Med 1998; 26:645–650 [DOI] [PubMed] [Google Scholar]
- 25.Briegel J, Forst H, Haller M, et al. : Stress doses of hydrocortisone reverse hyperdynamic septic shock: A prospective, randomized, double-blind, single-center study. Crit Care Med 1999; 27:723–732 [DOI] [PubMed] [Google Scholar]
- 26.Gordon AC, Mason AJ, Perkins GD, et al. : The interaction of vasopressin and corticosteroids in septic shock: A pilot randomized controlled trial. Crit Care Med 2014; 42:1325–1333 [DOI] [PubMed] [Google Scholar]
- 27.Gordon AC, Mason AJ, Thirunavukkarasu N, et al. ; VANISH Investigators: Effect of early vasopressin vs norepinephrine on kidney failure in patients with septic shock: The VANISH randomized clinical trial. JAMA 2016; 316:509–518 [DOI] [PubMed] [Google Scholar]
- 28.Hu B, Li JG, Liang H, et al. : The effect of low-dose hydrocortisone on requirement of norepinephrine and lactate clearance in patients with refractory septic shock. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue 2009; 21:529–531 [PubMed] [Google Scholar]
- 29.Lv QQ, Gu XH, Chen QH, et al. : Early initiation of low-dose hydrocortisone treatment for septic shock in adults: A randomized clinical trial. Am J Emerg Med 2017; 35:1810–1814 [DOI] [PubMed] [Google Scholar]
- 30.Oppert M, Schindler R, Husung C, et al. : Low-dose hydrocortisone improves shock reversal and reduces cytokine levels in early hyperdynamic septic shock. Crit Care Med 2005; 33:2457–2464 [DOI] [PubMed] [Google Scholar]
- 31.Rinaldi S, Adembri C, Grechi S, et al. : Low-dose hydrocortisone during severe sepsis: Effects on microalbuminuria. Crit Care Med 2006; 34:2334–2339 [DOI] [PubMed] [Google Scholar]
- 32.Sprung CL, Annane D, Keh D, et al. ; CORTICUS Study Group: Hydrocortisone therapy for patients with septic shock. N Engl J Med 2008; 358:111–124 [DOI] [PubMed] [Google Scholar]
- 33.Tongyoo S, Permpikul C, Mongkolpun W, et al. : Hydrocortisone treatment in early sepsis-associated acute respiratory distress syndrome: Results of a randomized controlled trial. Crit Care 2016; 20:329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Venkatesh B, Finfer S, Cohen J, et al. ; ADRENAL Trial Investigators and the Australian — New Zealand Intensive Care Society Clinical Trials Group: Adjunctive glucocorticoid therapy in patients with septic shock. N Engl J Med 2018; 378:797–808 [DOI] [PubMed] [Google Scholar]
- 35.Talebi Doluee M, Salehi M, Mahmoudi Gharaee A, et al. : The effect of physiologic dose of intravenous hydrocortisone in patients with refractory septic shock: A randomized control trial. J Emerg Pract Trauma 2018; 4:29–33 [Google Scholar]
- 36.Aboab J, Polito A, Orlikowski D, et al. : Hydrocortisone effects on cardiovascular variability in septic shock: A spectral analysis approach. Crit Care Med 2008; 36:1481–1486 [DOI] [PubMed] [Google Scholar]
- 37.Annane D, Sebille V, Charpentier C, et al. : Effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. JAMA 2002; 288:862–871 [DOI] [PubMed] [Google Scholar]
- 38.Annane D, Renault A, Brun-Buisson C, et al. ; CRICS-TRIGGERSEP Network: Hydrocortisone plus fludrocortisone for adults with septic shock. N Engl J Med 2018; 378:809–818 [DOI] [PubMed] [Google Scholar]
- 39.Investigators CS, Annane D, Cariou A, et al. : Corticosteroid treatment and intensive insulin therapy for septic shock in adults: A randomized controlled trial. JAMA 2010; 303:341–348 [DOI] [PubMed] [Google Scholar]
- 40.Bosch NA, Teja B, Law AC, et al. : Comparative effectiveness of fludrocortisone and hydrocortisone vs hydrocortisone alone among patients with septic shock. JAMA Intern Med 2023; 183:451–459 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Labib HA, Hassan AI, Kamaly AM, et al. : Evaluation of the role of hydrocortisone either alone or combined with fludrocortisone in the outcome of septic shock in adults. Ain-Shams J Anesthesiol 2022; 14:60 [Google Scholar]
- 42.Heming N, Sivanandamoorthy S, Meng P, et al. : Immune effects of corticosteroids in sepsis. Front Immunol 2018; 9:1736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pirracchio R, Annane D, Waschka AK, et al. : Patient-level meta-analysis of low-dose hydrocortisone in adults with septic shock. NEJM Evid 2023; 2:EVIDoa2300034. [DOI] [PubMed] [Google Scholar]
- 44.Bittl JA, He Y: Bayesian analysis: A practical approach to interpret clinical trials and create clinical practice guidelines. Circ Cardiovasc Qual Outcomes 2017; 10:e003563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Turner RM, Bird SM, Higgins JP: The impact of study size on meta-analyses: Examination of underpowered studies in Cochrane reviews. PLoS One 2013; 8:e59202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hernan MA, Wang W, Leaf DE: Target trial emulation: A framework for causal inference from observational data. JAMA 2022; 328:2446–2447 [DOI] [PubMed] [Google Scholar]
- 47.Siddiqui WJ, Iyer P, Aftab G, et al. : Hydrocortisone reduces 28-day mortality in septic patients: A systemic review and meta-analysis. Cureus 2019; 11:e4914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Fang F, Zhang Y, Tang J, et al. : Association of corticosteroid treatment with outcomes in adult patients with sepsis: A systematic review and meta-analysis. JAMA Intern Med 2019; 179:213–223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lyu QQ, Chen QH, Zheng RQ, et al. : Effect of low-dose hydrocortisone therapy in adult patients with septic shock: A meta-analysis with trial sequential analysis of randomized controlled trials. J Intensive Care Med 2020; 35:971–983 [DOI] [PubMed] [Google Scholar]
- 50.Aletreby WT, Alharthy AM, Madi AF, et al. : Impact on efficacy and safety of hydrocortisone in sepsis and septic shock—a systematic literature review and meta-analysis. Arch Iran Med 2019; 22:394–402 [PubMed] [Google Scholar]
- 51.Riley RD, Lambert PC, Abo-Zaid G: Meta-analysis of individual participant data: Rationale, conduct, and reporting. BMJ 2010; 340:c221. [DOI] [PubMed] [Google Scholar]
- 52.Zhang S, Chang W, Xie J, et al. : The efficacy, safety, and optimal regimen of corticosteroids in sepsis: A Bayesian network meta-analysis. Crit Care Explor 2020; 2:e0094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Schumer W: Steroids in the treatment of clinical septic shock. Ann Surg 1976; 184:333–341 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sprung CL, Caralis PV, Marcial EH, et al. : The effects of high-dose corticosteroids in patients with septic shock: A prospective, controlled study. N Engl J Med 1984; 311:1137–1143 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.