Abstract
Objective
Although hysteroscopic adhesiolysis (HA) is the main treatment for intrauterine adhesion (IUA), postoperative management of IUA remains challenging because there is no consensus on how to mitigate the high rate of postoperative adhesions reformation. This study aims to compare the effectiveness and safety of 2 types of intrauterine stents with different thickness and hardness in treating moderate-to-severe IUA.
Methods
A retrospective clinical study was conducted in the Third Xiangya Hospital of Central South University from November 2020 to July 2021. A total of 191 patients with moderate-to-severe IUA who received surgical treatment and placed intrauterine stents after HA to prevent recurrence of postoperative adhesions were included. According to the hardness and thickness of the intrauterine stents, the participants were divided into a case group (placed the novel thin intrauterine stent, n=62) and a control group (placed the conventional stent, n=129). After 2-3 menstrual cycles, a second-look hysteroscopy was performed, and the intrauterine stents were removed. The postoperative efficacy [the reduction of American Fertility Society (AFS) scores, the adhesions reformation rate, the changes in menstrual pattern, and the pregnancy rate during the follow-up], safety (the adverse events), and applicability (the difficulty of stent removal) were compared between the 2 groups.
Results
No significant differences in preoperative clinical characteristics were observed between the 2 groups (all P>0.05). The menstrual volume of all patients was increased after the treatment. The reduction of AFS scores and the menstruation recovery rate were not significantly different between the 2 groups (P=0.519 and P=0.272, respectively). Notably, there was no case of displacement in the case group, while the displacement rate of the control group was 2.3% (P=0.552). Moreover, there was no significant difference in abdominal pain or postoperative abnormal vaginal bleeding between the 2 groups (P=0.823 and P=0.851, respectively). However, the difficulty rate of removing the thinner stents was significantly lower than that of removing the traditional stent (21.0% vs 38.8%, P=0.014). During the follow-up for half a year of the postoperative period, the pregnancy rate did not differ significantly in the case and control groups (45.0% vs 34.6%, P=0.173).
Conclusion
The novel intrauterine stent shows noninferior efficacy and had a good safety profile compared with conventional stents in treating moderate-to-severe IUA. Importantly, it was more convenient to be removed without increasing the rate of displacement and detachment. Therefore, it could reduce the amount of damage to the endometrium and has higher applicability than conventional stents.
Keywords: intrauterine stent, intrauterine adhesions, hysteroscopic adhesiolysis
Abstract
目的
宫腔镜下宫腔粘连分离术(hysteroscopic adhesiolysis,HA)是宫腔粘连(intrauterine adhesion,IUA)的主要治疗方法,但IUA术后管理仍然具有挑战性,主要在如何处理术后粘连高复发率的问题上缺乏共识。本研究旨在比较两种不同厚度和硬度的宫腔支架治疗中重度宫腔粘连的有效性和安全性。
方法
选择2020年11月至2021年7月在中南大学湘雅三医院接受手术治疗并于术后放置宫腔支架以防止术后粘连再发的中重度宫腔粘连患者191例进行回顾性研究。根据放置宫腔支架的厚度与硬度不同,分为研究组(放置新型宫腔支架,n=62)和对照组(放置传统宫腔支架组,n=129),2~3个月经周期后再次行宫腔镜探查并取出宫腔支架。比较两组患者的术后疗效[美国生育学会(American Fertility Society,AFS)评分下降程度、再粘连发生率、月经改善情况、随访期间的妊娠情况]、安全性(支架放置后的不良反应)和适用性(取出支架的难度)。
结果
2组间术前基线临床特征差异无统计学意义(均P>0.05)。所有患者治疗后月经量均增多。AFS分数的下降程度和月经恢复率2组间差异无统计学意义(分别P=0.519和 P=0.272)。研究组无支架移位发生,而对照组支架移位率为2.3%,但差异无统计学意义(P=0.552)。2组术后腹痛和异常阴道出血发生率差异无统计学意义(分别P=0.82和P=0.851)。新型支架较传统支架更容易取出(21.0% vs 38.8%,P=0.014)。支架取出后随访半年,2组妊娠率差异无统计学意义(45.0% vs 34.6%,P=0.173)。
结论
新款薄型支架用于治疗中重度宫腔粘连疗效不劣于传统宫腔支架,有较好的安全性并具有独特优势,即在不增加移位、脱落率的同时取出更加方便,因而能减少对子宫内膜的再次损伤,具有更好的临床适用性。
Keywords: 宫腔支架, 宫腔粘连, 宫腔镜下宫腔粘连分离术
Intrauterine adhesion (IUA) is predominately a consequence of endometrial injuries that cause abnormal fibrous adhesions. It remains a serious threat to women’s reproductive health worldwide. Incomplete statistics estimate that 90% of IUAs are associated with pregnancy[1]. The overall global incidence ranges from 1.73% to 40%[2]. Excessive and repeated uterine cavity operations lead to impaired endometrial function and regeneration, which destroys the normal anatomy of the uterus and reduces endometrial receptivity[3]. In addition to abnormal menstruation, IUA can also lead to infertility or missed abortion and may increase the risk of placenta accreta spectrum and products of pregnancy retention during subsequent pregnancies[4]. The standard treatment for IUA is transcervical resection of the adhesions by hysteroscopy to restore the normal anatomy of the uterus. Additionally, hormonal therapy and/or stem cell treatments can also be used to regenerate the endometrium[5]. The comprehensive treatment promotes endometrial receptivity and ultimately improves female fertility. However, the rate of IUA recurrence was reported to be as high as 66%[2, 6]. Moreover, the rate of postoperative successful pregnancy in severe cases only ranges from 22.5% to 33.3%[7-8]. Therefore, preventing the recurrence of IUA and improving fertility are great challenges that gynecological experts pay close attention to.
To date, several methods of repairing endometrial injury and preventing the recurrence of IUA are widely available for use in clinical practice. Currently, comprehensive therapies are adopted to prevent the recurrence of postoperative adhesions. These therapies include the intrauterine device (IUD), Foley balloon, hormonal therapy, sodium hyaluronate gel, and amniotic membrane or stem cell transplantation in addition to hysteroscopic adhesiolysis (HA)[9-11]. Although these therapies can alleviate the recurrence of IUA to a certain degree, no specific and effective strategy exists to significantly improve the postoperative pregnancy rate in patients with moderate to severe IUA. Our previous studies[12-13] have reported a patented intrauterine stent that can effectively maintain the normal anatomical shape of the uterine cavity and enable sufficient drainage to prevent the reformation of postoperative IUA. In the preliminary study, a hysteroscopy with an outer sheath of 5.4 mm in diameter was adopted to diagnose and treat IUA, without cervical dilation. It is relatively easy to insert the intrauterine stent into the uterine cavity after HA, but we have observed that the stent can be difficult to remove because it curls if the cervix is not sufficiently dilated. Generally, hysteroscopic micro-forceps have been used to facilitate stent removal, but several failed attempts may cause secondary endometrial injury to the lateral wall of the uterine cavity by repeated friction. To resolve the problems associated with stent removal, we have continuously modified and improved various stents of varying degrees of thickness and hardness without affecting the therapeutic efficacy. Therefore, we conducted a retrospective analysis among moderate to severe IUA patients treated with 2 types of stents that differed in thickness and hardness and compared the efficacy, removal difficulty, and side effects. The purpose of this study was to select an intrauterine stent with better efficacy that created less secondary endometrial damage and was more applicable for clinical use. Moreover, we also compared the efficacy and pregnancy rate of 2 types of stents in patients with cervical canal adhesions and cornual adhesions given the special anatomy and poor prognosis.
1. Subjects and methods
1.1. Participants
A total of 433 patients had undergone HA at the Third Xiangya Hospital of Central South University from November 2020 to July 2021. IUA is diagnosed and scored by the same experienced surgeon using hysteroscopy. According to the American Fertility Society (AFS) classification system, IUA is scored as follows: 1-4 (mild), 5-8 (moderate), and 9-12 (severe)[14].
The inclusion criteria were: (I) IUA confirmed by hysteroscopy and underwent HA; (II) age between 18-45 years; (III) the size of the intrauterine stent matching the uterine cavity after HA; (IV) complete data and information of hospitalization, particularly clear records of surgery; (V) all hysteroscopies were performed by experienced experts; (VI) moderate to severe (AFS score≥5) IUA. Meanwhile, the exclusion criteria were: (I) abnormal endocrine function; (II) tubercular IUA; (III) grossly abnormal partner semen; (IV) severe systemic disease or contraindications to surgery; (V) congenital reproductive tract abnormalities; (VI) cervical insufficiency.
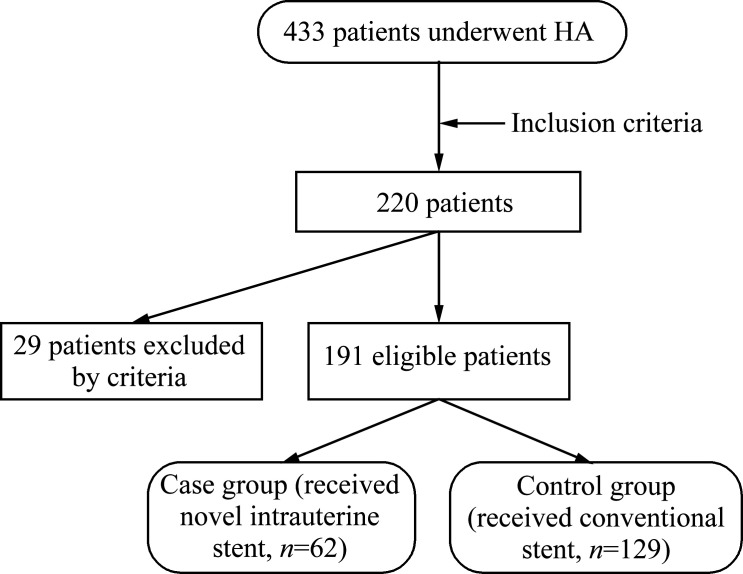
Finally, 191 eligible patients with IUA were selected retrospectively according to the inclusion criteria and exclusion criteria in our study (Figure1). This study was approved by the Ethics Committee of the Third Xiangya Hospital of Central South University (No. 22074). Written informed consent was obtained voluntarily from all participants.
Figure 1. Flowchart of the study population HA: Hysteroscopic adhesiolysis.
1.2. Surgical procedure
All participants had undergone preoperative 3-dimensional transvaginal ultrasound (3D-TVUS) examination, which evaluated the shape of the uterus, intracorneal distance, endometrial thickness, number of visible fallopian tubal ostia, and endometrial blood flow grade. The HA was conducted within 3–7 days after the menstruation, except in those patients identified with uterine amenorrhea, who underwent HA at any time. All patients fasted for 8 hours before surgery and underwent preoperative vaginal preparation and conventional sterilization. Sterile saline solution was used as the distention medium. Distension pressure was set at 110-120 mmHg (1 mmHg=0.133 kPa) with a flow rate of 300-350 mL/min. HA was performed under intravenous anesthesia. Hysteroscopies were performed by experienced experts and monitored under transabdominal sonographic guidance. The phases and steps of the procedure were detailed below.
(I) HA: A diagnostic hysteroscopy with a 5Fr working channel and an outer sheath of 5.4 mm in diameter was used to pass through the cervical canal and evaluate the AFS scores of adhesions before performing HA. The adhesions and scars in the uterine cavity were separated by using 5Fr scissors, combined with double-action forceps that were used for the “blunt spreading dissection technique”, resulting in the restoration of the correct anatomical uterine cavity[15-16].
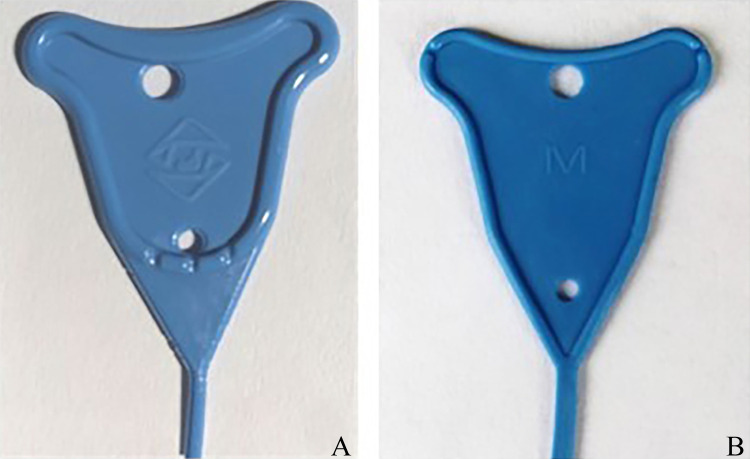
(II) Insertion of intrauterine stent: Intrauterine stents were used as barriers to prevent IUA recurrence after HA. The size of the stent was determined by an experienced expert according to the preoperative 3D-TVUS. Among them, the thin stent is the novel stent group (case group), and the thick stent is the conventional stent group (control group) (Table 1, Figure 2). Each group had various sizes of intrauterine stents from XXXS to XXXL. The stent was inserted into the uterine cavity with a pushrod and adjusted with micro-forceps under hysteroscopy to confirm whether the position and size of the stent matched the uterine cavity. Next, 3 mL of sodium hyaluronate gel was injected into the uterine cavity through the catheter.
Table 1.
Comparison of the thickness and hardness of the 2 types of intrauterine stents
| Types | Central thin film thickness/mm | Periphery stiffener thickness/mm | Hardness/Shore A |
|---|---|---|---|
| Novel stent | 0.25 | 0.75 | 70 |
| Conventional stent | 0.50 | 2.00 | 60 |
Figure 2. Structure of the 2 types of intrauterine stents.
A: Conventional stent; B: Novel stent.
(III) Postoperative management: If the patient had hormonal contraindications, postoperative treatment with estrogen therapy was not used. Otherwise, the application of estrogen-progestin sequential therapy or combined with traditional Chinese medicine after HA was considered to promote endometrial growth. During the stent placement, the menstrual pattern changes, abdominal pain, and abnormal uterine bleeding were recorded, and 3D-TVUS was performed again to evaluate the position of the intrauterine stent.
(IV) The second-look hysteroscopy and the stent removal: Postoperative assessment of the uterine cavity was conducted with hysteroscopy after 2-3 menstrual cycles after HA to evaluate the efficacy of treatment (e.g., postoperative AFS scores, the reduction of AFS scores, the position of the intrauterine stent, the adhesions reformation and the compression of the endometrium).
(V) Postoperative follow-up: All patients were advised to conceive as soon as possible after the removal of the stent. Patients were followed up to record the pregnancy rate within 6 months after the stent removal.
1.3. Statistical analysis
All data analyses were performed using SPSS 26.0 software (IBM Corp, Armonk, NY, USA). Data distribution was determined by the one-sample Kolmogorov-Smirnov test, combined with the histogram, P-P diagram and Q-Q diagram. When the P value was greater than 0.05, data were considered to be normally distributed. Continuous variables were presented as mean±standard deviation and analyzed using Student t-test. Categorical variables were described as percentages and were evaluated using the Chi-squared test or Fisher exact test. Additionally, the Mann-Whitney U test was used to compare the differences in nonparametric data between the groups. A P value less than 0.05 (2-sided) was considered statistically significant.
2. Results
2.1. Baseline characteristics of the participants
A total of 191 patients with IUA were recruited in this study, including 62 patients in the case group and 129 patients in the control group. A history of recurrence of IUA after HA was observed in 87 (45.5%) patients, of whom 23 (26.4%) had previously undergone HA over 3 times, with 1 patient having undergone HA 11 times. In the case group, there were 34 (54.8%) cases of moderate IUA and 28 (45.2%) cases of severe IUA. In the control group, there were 75 (58.1%) cases of moderate IUA and 54 (41.9%) cases of severe IUA patients. The ages of women were (33.65±5.20) and (33.09±4.67) years old for the case group and the control group, respectively (P=0.456). Preoperative AFS scores were (8.82±1.21) and (8.67±1.37) for the case group and the control group, respectively (P=0.446). Additionally, no significant differences were observed in terms of BMI, pregnancy history, history of uterine cavity operation, preoperative HA times, and previous HA times (all P>0.05), which means the 2 groups were comparable. The demographic and clinical characteristics of the participants are shown in Table 2.
Table 2.
Baseline characteristics of the participants
| Groups | n | Age/years | BMI/(kg·m-2) | Gravidity | Parity | Preoperative AFS score |
|---|---|---|---|---|---|---|
| P | 0.456 | 0.899 | 0.494 | 0.545 | 0.446 | |
| Case group | 62 | 33.65±5.20 | 21.79±2.74 | 2.89±1.80 | 0.52±0.56 | 8.82±1.21 |
| Control group | 129 | 33.09±4.67 | 21.73±2.69 | 3.08±1.80 | 0.57±0.64 | 8.67±1.37 |
| Groups | History of uterine cavity operations | Preoperative HA times | Recurrent IUA/[No.(%)] | ||
|---|---|---|---|---|---|
| D&C for retained products of conception/[No.(%)] | Non-pregnancy-related surgeries*/[No.(%)] | D&C for abortion and induced labor/[No.(%)] | |||
| Case group | 5(8.1) | 14(22.6) | 43(69.3) | 0.82±1.03 | 30(48.4) |
| Control group | 11(8.5) | 33(25.6) | 85(65.9) | 0.98±1.60 | 57(44.2) |
| P | 0.887 | 0.489 | 0.585 | ||
D&C: Dilation and curettage; HA: Hysteroscopic adhesiolysis; AFS: American Fertility Society; IUA: Intrauterine adhesion. *Non-pregnancy-related surgeries include hysteroscopic endometrial polypectomy, hysteroscopic myomectomy for submucosal fibroids, and hysteroscopic resection of the uterine septum, and so on.
2.2. Efficacy of treatment
Postoperative AFS scores were 2.85±1.04 and 2.88±1.33 for the case group and the control group, respectively (P=0.917). A significant reduction was observed between the preoperative and second-look AFS scores in both case and control groups. There were no significant differences in postoperative AFS score and reduction of AFS score between the case group and the control group (both P>0.05). One participant in the control group had a displacement of the conventional stent into the cervical canal, and this stent was removed in the outpatient clinic without a second-look hysteroscopy to assess IUA. Adhesions reformation at second-look hysteroscopy in the case group was not significantly different from that in the control group (P=0.091, Table 3).
Table 3.
Comparison of postoperative AFS scores, adhesions reformation, and menstrual pattern improvement between the 2 groups
| Groups | n | Postoperative AFS score | Reduction of AFS score | Adhesions reformation/[No.(%)] |
|---|---|---|---|---|
| P | 0.917 | 0.519 | 0.091 | |
| Case group | 62 | 2.85±1.04 | 5.97±1.48 | 13(21.0) |
| Control group | 128* | 2.88±1.33 | 5.80±1.70 | 42(32.8) |
| Groups | Menstrual improvement | |
|---|---|---|
| Improved not up to normal/[No.(%)] | Restore of normal menstruation/[No.(%)] | |
| Case group | 21(33.9) | 41(66.1) |
| Control group | 54(42.2) | 74(57.8) |
| P | 0.272 | |
AFS: American Fertility Society. *One participant in the control group was not performed a second look hysteroscopy because the stent was removed in the clinic outpatient after displacement into the cervical canal.
The menstrual volume of all patients increased after the treatment. A total of 66.1% (41/62) of individuals in the case group restored normal menstruation, compared with 57.8% (74/128) in the control group. The change in the menstrual pattern after surgery in the case group was not significantly different from that of the control group (P=0.272, Table 3).
2.3. Safety comparison of intrauterine stents
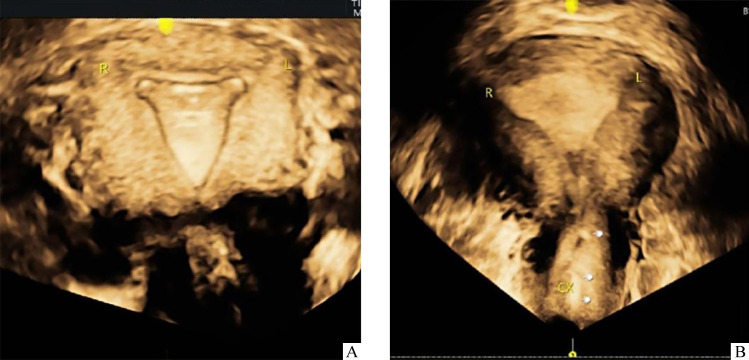
All participants underwent the procedure without any surgical complications (e.g., fever, allergy, uterine perforation, heavy uterine bleeding, or hyperhydration syndrome). After insertion of the intrauterine stent, no instances of spontaneous expulsion of the stent occurred in the 2 groups. A total of 7 patients experienced abdominal pain, including 2 (3.2%) in the case group and 5 (3.9%) in the control group. There were 3 (4.8%) cases of abnormal uterine bleeding, and 1 (1.6%) case of embedment into the myometrium but no cases of displacement in the case group, while in the control group, there were 4 (3.1%) cases of abnormal uterine bleeding and 2 (1.6%) cases of embedment. The postoperative 3D-TVUS showed that the stents were in a normal position except for 3 (2.3%) cases of downward displacement in the control group (Figure3). There were no significant differences in safety between the 2 groups (P>0.05, Table4).
2.4. Differences in the difficulty of stent removal
Except for a conventional stent with downward displacement into the cervix which was removed in the outpatient clinic, others were removed under the second-look hysteroscopy. If the surgeon failed to remove the stent using hysteroscopic micro-forceps but needed to use a vascular clamp or fully dilate the cervix, this was considered difficult to remove the stent. A total of 13 (21.0%) cases had difficulty in removing the novel stent. Among the 128 participants of the control group, 49 (38.3%) cases had stents that were difficult to remove. There was a statistically significant difference between the 2 groups (P=0.014).
Figure 3. Three-dimensional transvaginal ultrasound (3D-TVUS) showing the position of the intrauterine stent in uterine cavity after surgery.
A: Normal position of the intrauterine stent; B: Displacement of one conventional stent into the cervix.
Table 4.
Results of adverse events
| Groups | n | Abdominal pain/[No.(%)] | Abnormal uterine bleeding/[No.(%)] | Displacement/[No.(%)] | Embedment/[No.(%)] |
|---|---|---|---|---|---|
| P | 0.823 | 0.851 | 0.552 | 0.694 | |
| Case group | 62 | 2(3.2) | 3(4.8) | 0(0.0) | 1(1.6) |
| Control group | 129 | 5(3.9) | 4(3.1) | 3(2.3) | 2(1.6) |
2.5. Fertility result
A total of 187 participants were followed up for half a year, and 4 participants (2 in each group) were lost to follow-up. Given that the postoperative follow-up time was short, we could only follow up on the pregnancy rate within half a year, but the pregnancy outcomes could not be followed up. Among the 60 patients of the case group, 27 (45.0%) patients succeeded in pregnancy, and the pregnancy rate was higher than that of the control group (44/127, 34.6%). No statistical difference was observed in the pregnancy rate between the 2 groups (P=0.173).
2.6. Analysis of individuals with cervical canal adhesions and uterine horn adhesions
2.6.1. Effectiveness and pregnancy rate in patients with cervical canal adhesions
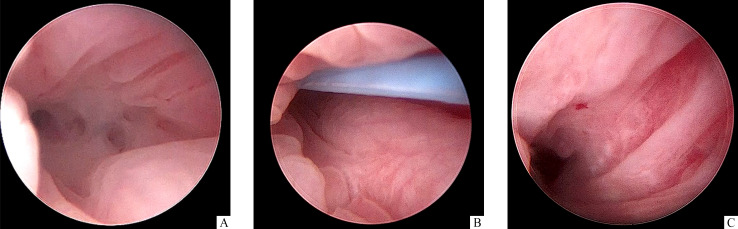
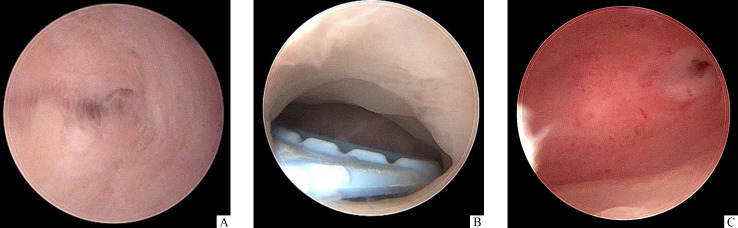
Among all participants, cervical canal adhesions were observed in 74 individuals under hysteroscopy. The postoperative AFS score, its reduction, and menstrual improvement showed no statistical differences between the case group and the control group (all P>0.05). Adhesions reformation was observed in only 4 (16.0%) patients in the case group, while there were 16 (32.7%) cases in the control group (P=0.127). The overall pregnancy rate did not differ between the 2 groups (P=0.454, Table 5). The comparison of cervical canal images between diagnostic hysteroscopy and second-look hysteroscopy is shown in Figure 4.
Table 5.
Comparison of therapeutic efficacy and pregnancy rate between the 2 groups in patients with preoperative cervical canal adhesions
| Groups | n | Preoperative AFS scores | Postoperative AFS scores | Reduction of AFS scores |
|---|---|---|---|---|
| P | 0.072 | 0.729 | 0.170 | |
| Case group | 25 | 8.76±1.30 | 2.68±0.85 | 6.08±1.53 |
| Control group | 49 | 9.32±1.14 | 2.78±1.23 | 6.55±1.31 |
| Groups |
Adhesions reformation/ [No.(%)] |
Menstrual improvement/[No.(%)] |
Pregnancy rate/ [No.(%)] |
|
|---|---|---|---|---|
| Improved not up to normal | Restore of normal menstruation | |||
| P | 0.127 | 0.903 | 0.454 | |
| Case group | 4(16.0) | 8(32.0) | 17(68.0) | 7(30.4) |
| Control group | 16(32.7) | 15(30.6) | 34(69.4) | 19(39.6) |
Figure 4. Hysteroscopy images of cervical canal.
A: Cervical canal adhesions before hysteroscopic adhesiolysis is shown. B: Stent tail is shown to prevent cervical canal to reform adhesions under second-look hysteroscopy. C: No adhesions is observed in cervical canal after the stent removal.
2.6.2. Therapeutic efficacy and fertility results in patients with uterine horn adhesions
Preoperative uterine horn adhesions were reported in 65 participants, including 19 in the case group and 46 in the control group. Similarly, no significant differences were observed in postoperative AFS score, its reduction, and pregnancy rate (all P>0.05). The proportion of patients with normal menstruation in the case group was about 1.5 times that of the control group after treatment (73.7% vs 50.0%, P=0.079; Table 6). The comparison of uterine horn images between diagnostic hysteroscopy and second-look hysteroscopy is shown in Figure 5.
Table 6.
Comparison of therapeutic efficacy and pregnancy rate between the 2 groups in patients with preoperative cornual adhesions
| Groups | n | Preoperative AFS scores | Postoperative AFS scores | Reduction of AFS scores |
|---|---|---|---|---|
| P | 0.254 | 0.689 | 0.213 | |
| Case group | 19 | 9.32±1.25 | 3.11±0.81 | 6.21±1.23 |
| Control group | 46 | 8.93±1.20 | 3.22±1.41 | 5.72±1.83 |
| Groups | Adhesions reformation/[No.(%)] | Menstrual improvement/[No.(%)] | Pregnancy rate/[No.(%)] | |
|---|---|---|---|---|
| Improved not up to normal | Restore of normal menstruation | |||
| P | 0.064 | 0.079 | 0.499 | |
| Case group | 4(21.1) | 5(26.3) | 14(73.7) | 5(31.3) |
| Control group | 21(45.7) | 23(50.0) | 23(50.0) | 12(27.3) |
Figure 5. Hysteroscopic images of unilateral uterine horn.
A: Right uterine horn adhesions before hysteroscopic adhesiolysis; B: Two edges of the stent approximately reached the uterine horns to prevent adhesions reformation; C: A right uterine horn without adhesions after the stent removal.
3. Discussion
Although HA is an effective and standard method in the treatment of IUA, postoperative management of IUA remains controversial, mainly because there is no consensus on how to manage the high rate of postoperative adhesions recurrence. The high adhesions reformation rate is mainly alleviated by using a postoperative insertion of intrauterine mechanical barriers, which separates the anterior and posterior uterine walls and maintains the normal postoperative uterine shape, which is combined with the application of medications to promote endometrial growth[17]. Given the limitation of some barriers, the therapeutic efficacy of the combination of 2 or more devices is significantly better than that of the treatment alone, such as the combination of an IUD, Foley balloon, and hyaluronic acid gel[18-20]. According to our previous studies[12-13], the conventional patented intrauterine stent had obvious advantages in separating uterine cavity walls and maintaining the normal shape of the uterine cavity in the treatment of patients with recurrent and refractory IUA. In our still-to-be-published multicenter, randomized, prospective clinical study on the conventional intrauterine stent, the efficacy of the stent alone in the treatment of IUA was not inferior to that of the combination of an IUD, Foley balloon, or hyaluronic acid gel. However, it is not clear whether the thickness and hardness of intrauterine stents affect the treatment and prognosis of IUA.
All participants in this study had the hysteroscopic diagnosis of moderate-to-severe IUA with an average preoperative AFS greater than 8. A history of recurrence of IUA after HA was observed in 87 (45.5%) patients, of whom 23 (26.4%) had previously undergone HA over 3 times, with 1 patient having undergone HA 11 times. Therefore, we attempted to use our patented intrauterine stents for patients with initial or recurrent IUA. It is an obvious advantage that all patients could remove the novel or conventional stent after only 2-3 menstrual cycles without repeated HA procedures. In our study, the mean postoperative AFS score was 2.8, with a significant reduction of AFS score (>5) in all cases, and the efficacy of the treatment was 100%. Zhang, et al[21] used a Foley balloon combined with an IUD to prevent the postoperative recurrence of IUA. The results showed that the combination of a Foley intrauterine balloon and an IUD was more effective than IUD alone. Additionally, the extension of placement time could prevent the recurrence of IUA in patients with moderate IUA. Cai, et al[22] found that no adhesions reformation was observed in only 17.1% of patients with moderate-to-severe IUA treated with a physical barrier. However, 95.1% of patients needed to undergo 4 HAs, and they used Interceed (an oxidized, regenerative fibrin-bonded barrier) combined with an IUD to prevent adhesions recurrence. Therefore, we believe that the use of intrauterine stents to prevent the recurrence of IUA can achieve the effect of the combined treatment without repeated operations.
During the application, we found that conventional stents needed to be improved in certain aspects. Given that the insertion hole of the stents’ pushrod deviated from the uterine midline, the stents could easily pass through the uterine cavity, which is characterized by a wide uterine fundus and narrow cervix. On the contrary, when the conventional stent is removed by hysteroscopic microforceps, it is difficult for the periphery stiffener to curl, resulting in the wide upper part of the stent having difficulty passing through the narrow cervical canal. It is possible that the repeated friction due to the difficult removal of the conventional stent caused endometrial damage to the lateral wall of the uterus. Compared with the conventional stent (a 0.5 mm central thin film and 2 mm periphery stiffener), the modified novel stent has a 0.25 mm thin film in thickness in the center and a periphery stiffener of about 0.75 mm. Theoretically, both types of intrauterine stents with different thicknesses can completely separate the anterior and posterior walls of the uterine cavity and support the lateral walls to prevent the recurrence of central and peripheral IUA. Furthermore, the novel thinner stent does not affect the efficacy of the treatment. In this retrospective study, 49 (38.3%) cases in the conventional stent group had difficulty in removing the stent. Therefore, the novel thinner stent was modified from the conventional stent by reducing the thickness of the central thin film and periphery stiffener to allow the stent to maintain proper curvature and enable easier insertion and removal. Our results showed that the novel stents were significantly easier to remove than the conventional stents. Given the possibility of curling the novel thinner intrauterine stent in the uterine cavity, the improvement was made by increasing the hardness (70 Shore A hardness) to reduce stent deformation and displacement. In our study, downward displacement and expulsion were not observed in the new stent group, while 3 cases in the conventional stent group reported downward displacement. We speculated that the lower displacement rate of novel stents might be related to the appropriate increase of stent hardness or the higher size/shape matching with the uterus, which needs to be verified by further studies. Although the thickness and hardness of the 2 kinds of stents differed, there was no significant difference in the recurrence of IUA or the reduction of the AFS score between the 2 groups. Additionally, only a few patients showed adverse events, such as abdominal pain, vaginal bleeding, displacement, and embedment, which did not increase the discomfort of the patients, and there was no significant difference in safety between the 2 groups. Therefore, the novel thin intrauterine stents were not inferior to the conventional stents in efficacy or safety of treatment for moderate-to-severe IUA.
The ultimate goal of the treatment of IUA is to meet fertility needs, and pregnancy outcomes after treatment are the focus of obstetricians and gynecologists. A recent Meta-analysis showed a 62.3% pregnancy rate after HA in patients with mild IUA[23]. Chen, et al[24] reported an overall pregnancy rate of 48.2% after HA, which included 53.4% in patients with moderate IUA but only 25% in patients with severe IUA. In our study, due to the short follow-up period, the pregnancy rate was only recorded within 6 months after stent removal. Unfortunately, the final pregnancy outcome was not followed. The pregnancy rates in the case and control groups were 45.0% and 34.6%, respectively, which were close to the results of a study conducted by Chen, et al[24]. We estimated that the final pregnancy rate might be higher within the proper and longer follow-up period. The pregnancy rate of novel stents was slightly higher than that of conventional stents, which might be related to the decreased compression of the endometrium to the lateral walls of the uterus, benefiting endometrial tolerance and endometrial blood flow. However, certain limitations of the present study should also be considered. We used a retrospective design with limited sample size and insufficient follow-up duration, from which we could not observe the final pregnancy outcomes. Future studies will focus on fertility, including increasing pregnancy rates and improving pregnancy outcomes based on existing studies.
Funding Statement
This work was supported by the Natural Science Foundation of Hunan Province (2021JJ40953) and the Science and Technology Project of Hunan Province (2020SK53605), China.
Conflict of Interest
The authors declare that they have no conflicts of interest to disclose.
AUTHORS’CONTRIBUTIONS
HUANG Huan and MAO Xuetao Data collection, statistical analysis, thesis writing and revision; YU Yang, XIAO Bingxin, and ZHAO Xingping Image analysis and interpretation of data; ZHANG Aiqian Research design, critical review, and thesis revision; XU Dabao Research design and thesis writing. All authors have read and agreed to the final text.
Note
http://xbyxb.csu.edu.cn/xbwk/fileup/PDF/2022111575.pdf
References
- 1. Schenker JG, Margalioth EJ. Intrauterine adhesions: an updated appraisal[J]. Fertil Steril, 1982, 37(5): 593-610. 10.1016/s0015-0282(16)46268-0. [DOI] [PubMed] [Google Scholar]
- 2. Yu D, Wong YM, Cheong Y, et al. Asherman syndrome—one century later[J]. Fertil Steril, 2008, 89(4): 759-779. 10.1016/j.fertnstert.2008.02.096. [DOI] [PubMed] [Google Scholar]
- 3. Lee WL, Liu CH, Cheng M, et al. Focus on the primary prevention of intrauterine adhesions: Current concept and vision[J]. Int J Mol Sci, 2021, 22(10): 5175. 10.3390/ijms22105175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Vomstein K, Voss P, Molnar K, et al. Two of a kind? Immunological and clinical risk factors differ between recurrent implantation failure and recurrent miscarriage[J]. J Reprod Immunol, 2020, 141: 103166. 10.1016/j.jri.2020.103166. [DOI] [PubMed] [Google Scholar]
- 5. Huang J, Zhang W, Yu J, et al. Human amniotic mesenchymal stem cells combined with PPCNg facilitate injured endometrial regeneration[J]. Stem Cell Res Ther, 2022, 13(1): 17. 10.1186/s13287-021-02682-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Salma U, Xue M, Md Sayed AS, et al. Efficacy of intrauterine device in the treatment of intrauterine adhesions[J]. Biomed Res Int, 2014, 2014: 589296. 10.1155/2014/589296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Yu D, Li TC, Xia E, et al. Factors affecting reproductive outcome of hysteroscopic adhesiolysis for Asherman’s syndrome[J]. Fertil Steril, 2008, 89(3): 715-722. 10.1016/j.fertnstert.2007.03.070. [DOI] [PubMed] [Google Scholar]
- 8. Roy KK, Baruah J, Sharma JB, et al. Reproductive outcome following hysteroscopic adhesiolysis in patients with infertility due to Asherman’s syndrome[J]. Arch Gynecol Obstet, 2010, 281(2): 355-361. 10.1007/s00404-009-1117-x. [DOI] [PubMed] [Google Scholar]
- 9. AAGL Elevating Gynecologic Surgery. AAGL Practice Report: Practice Guidelines on Intrauterine Adhesions Developed in Collaboration With the European Society of Gynaecological Endoscopy (ESGE)[J]. J Minim Invasive Gynecol, 2017, 24(5): 695-705. 10.1016/j.jmig.2016.11.008. [DOI] [PubMed] [Google Scholar]
- 10. Dou Y, Yu T, Li Z, et al. Short- and long-term outcomes of postoperative intrauterine application of hyaluronic acid gel: A Meta-analysis of randomized controlled trials[J]. J Minim Invasive Gynecol, 2022, 29(8): 934-942. 10.1016/j.jmig.2022.05.006 [DOI] [PubMed] [Google Scholar]
- 11. Li X, Li P, Wang C, et al. A thermo-sensitive and injectable hydrogel derived from a decellularized amniotic membrane to prevent intrauterine adhesion by accelerating endometrium regeneration[J]. Biomater Sci, 2022, 10(9): 2275-2286. 10.1039/d1bm01791h. [DOI] [PubMed] [Google Scholar]
- 12. Huang H, Xu B, Cheng C, et al. A novel intrauterine stent for prevention of intrauterine adhesions[J]. Ann Transl Med, 2020, 8(4): 61. 10.21037/atm.2019.12.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Huang H, Zou L, Zhang A, et al. A preliminary study on a patented intrauterine stent in the treatment of recurrent intrauterine adhesions with poor prognosis[J]. Ann Transl Med, 2020, 8(4): 57. 10.21037/atm.2020.01.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Valle RF, Sciarra JJ. Intrauterine adhesions: hysteroscopic diagnosis, classification, treatment, and reproductive outcome[J]. Am J Obstet Gynecol, 1988, 158(6Pt1): 1459-1470. 10.1016/0002-9378(88)90382-1. [DOI] [PubMed] [Google Scholar]
- 15. Huang H, Cheng C, Johnson G, et al. Hysteroscopic intrauterine adhesiolysis using a blunt spreading dissection technique with double-action forceps[J]. J Minim Invasive Gynecol, 2018, 25(4): 583-584. 10.1016/j.jmig.2017.10.011. [DOI] [PubMed] [Google Scholar]
- 16. Zhu K, Jamail G, Xu D, et al. Hysteroscopic intrauterine adhesiolysis using “ploughing” technique with cold scissors[J/OL]. J Minim Invasive Gynecol, 2015, 22(6S): S122[2022-04-08]. 10.1016/j.jmig.2015.08.357. [DOI] [PubMed] [Google Scholar]
- 17. Azumaguchi A, Henmi H, Saito T. Efficacy of silicone sheet as a personalized barrier for preventing adhesion reformation after hysteroscopic adhesiolysis of intrauterine adhesions[J]. Reprod Med Biol, 2019, 18(4): 378-383. 10.1002/rmb2.12294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Myers EM, Hurst BS. Comprehensive management of severe Asherman syndrome and amenorrhea[J]. Fertil Steril, 2012, 97(1): 160-164. 10.1016/j.fertnstert.2011.10.036. [DOI] [PubMed] [Google Scholar]
- 19. Huang XW, Lin MM, Zhao HQ, et al. A prospective randomized controlled trial comparing two different treatments of intrauterine adhesions[J]. Reprod Biomed Online, 2020, 40(6): 835-841. 10.1016/j.rbmo.2020.02.013. [DOI] [PubMed] [Google Scholar]
- 20. Kou L, Jiang X, Xiao S, et al. Therapeutic options and drug delivery strategies for the prevention of intrauterine adhesions[J]. J Control Release, 2020, 318: 25-37. 10.1016/j.jconrel.2019.12.007. [DOI] [PubMed] [Google Scholar]
- 21. Zhang X, Liu W, Zhou Y, et al. Comparison of therapeutic efficacy of three methods to prevent re-adhesion after hysteroscopic intrauterine adhesion separation: a parallel, randomized and single-center trial[J]. Ann Palliat Med, 2021, 10(6): 6804-6823. 10.21037/apm-21-1296. [DOI] [PubMed] [Google Scholar]
- 22. Cai H, Qiao L, Song K, et al. Oxidized, regenerated cellulose adhesion barrier plus intrauterine device prevents recurrence after adhesiolysis for moderate to severe intrauterine adhesions[J]. J Minim Invasive Gynecol, 2017, 24(1): 80-88. 10.1016/j.jmig.2016.09.021. [DOI] [PubMed] [Google Scholar]
- 23. Hooker AB, Mansvelder FJ, Elbers RG, et al. Reproductive outcomes in women with mild intrauterine adhesions; a systematic review and meta-analysis[J]. J Matern Fetal Neonatal Med, 2022, 35(25): 6933-6941. 10.1080/14767058.2021.1931103. [DOI] [PubMed] [Google Scholar]
- 24. Chen L, Zhang H, Wang Q, et al. Reproductive outcomes in patients with intrauterine adhesions following hysteroscopic adhesiolysis: Experience from the largest women’s hospital in China[J]. J Minim Invasive Gynecol, 2017, 24(2): 299-304. 10.1016/j.jmig.2016.10.018. [DOI] [PubMed] [Google Scholar]