Abstract
Lannea L. genus belongs to the Anacardiaceae botanical family and has long been used in traditional medicinal systems of many countries to manage several health conditions, but no studies have been conducted regarding its usefulness as a source of herbal medicine for human use. A literature review was conducted on scientific papers indexed on B-On, Pubmed, and Web of Science databases. Our results showed that medicinal plants from this botanical genus, mostly constituted by bark and leaf, are often used to approach a wide variety of disease symptoms, like fever, inflammatory states, pain, and gastrointestinal disorders. Phytochemical profiles of Lannea species revealed that phenolic acid derivatives including hydroquinones, phenolic acids, flavonoids, condensed tannins, and triterpenoids are the main classes of secondary metabolites present. Among the total of 165 identified compounds, 57 (34.5%) are flavonoids, mostly quercetin- and myricetin-derived flavonols and catechin and epicatechin flavan-3-ol derivatives also containing a galloyl group. In vitro and in vivo studies allowed the identification of 12 different biological activities, amongst which antimicrobial, antioxidant, anti-inflammatory, and cytotoxic activities were the most frequently cited and observed in in vitro essays. Our review contributes useful information for the scientifical validation of the use of Lannea species in traditional medicinal systems and shows that more research needs to be conducted to better understand the concrete utility of these as herbal medicines.
Keywords: Anacardiaceae, biological activity, ethnomedicine, Lannea, inflammation
1. Introduction
Lannea A. Rich. in Guill. is an important genus of flowering plants in Anacardiaceae, a botanical family comprising 81 genera and 800 species spread across tropical and subtropical regions with warm or temperate climates (tropical and South Africa, Saudi Arabian Peninsula, India, China, and Indochina) [1]. In addition to their importance in traditional medicinal systems, some species of Anacardiaceae have high economic value (e.g., Anacardium occidentale L., Mangifera indica L.) due to the use of their fruits and seeds in food and manufacture of beverages, being important to local communities and national economies as renewable forest resources and tradeable products [2,3]. The ancestral knowledge of Traditional Medicine Practitioners about the different medicinal proprieties of distinct species of this family has benefited many communities of tropical and sub-tropical countries where access to conventional primary health services is scarce and also in more developed countries as a complementary use to conventional medicine [4].
The Lannea genus was first described by Achille Richard and published in Florae Senegambiae Tentamen 153 in 1831 [5]. According to The World Flora Online [6], it includes a total of 36 accepted species (Table 1), of which 14.2% are classified as data deficient (DD), 30.9% as least concern (LC), 4.7% are classified as least concern (NT), and 7.1% as vulnerable (VU).
Table 1.
Lannea A. Rich. in Guill. accepted species.
| Lannea acida A.Rich. | Lannea humilis (Oliv.) Engl. |
| Lannea acuminata Engl. | Lannea katangensis Van der Veken |
| Lannea alata (Engl.) Engl. | Lannea ledermannii Engl. |
| Lannea ambacensis (Hiern) Engl. | Lannea malifolia (Chiov.) Sacleux |
| Lannea angolensis R. Fern. & Mendes | Lannea microcarpa Engl. & K.Krause |
| Lannea antiscorbutica (Hiern) Engl. | Lannea nigritana (Scott Elliot) Keay |
| Lannea asymmetrica R.E.Fr. | Lannea obovata (Hook.f. ex Oliv.) Engl. |
| Lannea barteri (Oliv.) Engl. | Lannea rivae (Chiov.) Sacleux |
| Lannea chevalieri Engl. | Lannea rubra (Hiern) Engl. |
| Lannea cinerascens Engl. | Lannea schimperi (Hochst. ex A.Rich.) Engl. |
| Lannea coromandelica (Houtt.) Merr. | Lannea schweinfurthii (Engl.) Engl. |
| Lannea cotoneaster (Chiov.) Sacleux | Lannea tibatensis Engl. |
| Lannea discolor (Sond.) Engl. | Lannea transulta (Balf.f.) Radcl. Sm. |
| Lannea edulis (Sond.) Engl. | Lannea triphylla (Hochst. ex A.Rich.) Engl. |
| Lannea fruticosa (Hochst. ex A.Rich.) Engl. | Lannea velutina A.Rich. |
| Lannea fulva (Engl.) Engl. | Lannea virgata R.Fern. & A.Fern. |
| Lannea glabrescens Engl. | Lannea welwitschii (Hiern) Engl. |
| Lannea gossweileri Exell & Mendonça | Lannea zastrowiana Engl. & Brehmer |
Adapted from: WFO (2023): Lannea A. Rich. in Guill. [7]
Lannea species are mainly trees, shrubs, or dioecious subshrubs up to 15 m high and are known for their great morphological diversity, distributed in the tropical and subtropical zones and native to tropical Africa and Asia. These species have characteristic imparipinnate leaves, opposite, entire leaflets, and a terminal panicle or raceme as the inflorescence. Most of them are deciduous and can be found in humid, arid, and dry environments but not in deserts or at altitudes over 3500 m [4,8].
To exemplify the botanical characteristics of Lannea species, we hereby provide a description of various species within the genus, including L. coromandelica, L. velutina, L. schimperii, L. acida, L. microcarpa, and L. welwitschii, focusing on their leaf morphology and anatomical characteristics. The leaves of Lannea species are compound and imparipinnate, consisting of petiolate leaflets that are oppositely arranged, forming a pseudo-verticillate pattern in the case of L. coromandelica. The leaves exhibit membranous texture, oval shape with asymmetric bases and pointed apices, and entire margins. Leaf size varies, and primary venation is pinnate. Secondary venation displays weak brochidodromous patterns with six basal veins, and intersecondary veins are faint. Tertiary venation demonstrates a mixed arrangement (opposite/alternate), while fourth-order venation is regularly polygonal reticulate. Fifth-order veins are dichotomous, and the highest order observed is the sixth. Marginal venation is free, forming incomplete arches. Stomata of the policytic-anomocytic type are located exclusively on the abaxial surface. Trichomes, which are moderate, multicellular, and stellate, are distributed throughout the leaf surface, and no prismatic crystals, druses, or resinous canals are observed. Regarding L. schimperii, L. acida, L. microcarpa, and L. welwitschii, these species exhibit several anatomical and morphological characteristics typical of the genus, despite the existing variability within and between some genera. Straight, curved, round, and wavy cell walls, as well as polygonal shapes, are observed on both leaf surfaces. Stomata are confined to the abaxial surface in all species, with only cyclocytic and anomocytic types identified. Trichomes are present in some Lannea species, with L. schimperii being the only one possessing trichomes on both adaxial and abaxial surfaces [9,10,11].
Modern medicine and scientific developments contribute to creating better health conditions in industrialized countries through constant breakthroughs in many areas. However, the global demographic distribution shows us that most of the world’s population lives in countries that do not have access to such healthcare. In these countries, people still rely almost exclusively on traditional medical systems, whose practices are based on the use of medicinal plants to treat illness or promote healthy conditions. Research shows that the ethnobotanical uses of Lannea species are well recognized in countries where they are native and includes their use as medicine, food, and ornamental and domestic lumber [12,13].
The use of Lannea species as medicinal plants in traditional medicinal systems is widely accepted, but there is a need for a critical assessment of their potential as a source of effective medicines based on quality, effectiveness, and safety data. A literature review of the available scientific information on Lannea species regarding their ethnomedical uses as well as their chemical, pharmacological, and toxicological data are hereby presented. This work is expected to provide a deep understanding of the potential of this botanical genus as a source of effective medicinal plants.
2. Results
2.1. Selection of Information
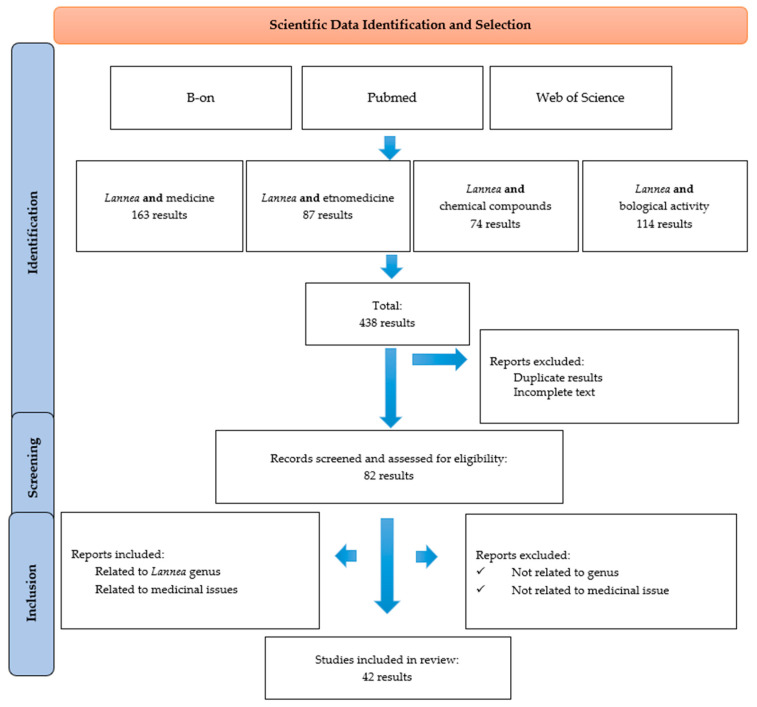
Data collection and selection were made according to the scheme presented in Figure 1. Initially, the database search of the scientific literature yielded 438 results. After excluding duplicate results, 82 scientific reports were assessed for relevance. Next, irrelevant reports were eliminated, and finally, 42 scientific publications were considered eligible for detailed analysis.
Figure 1.
Data screening based on the PRISMA methodology.
2.2. Ethnobotanical and Ethnomedical Data
2.2.1. Vernacular Names
The genus Lannea includes 16 species used in traditional medicinal systems (41.6% of the total number of accepted Lannea species) that are distributed in several countries, most of them in the African continent. Table 2 shows the vernacular names of the various species used in these traditional medicinal systems.
Table 2.
Lannea species used in traditional medicine and their vernacular names, countries, and ethnic groups.
| Species, (Synonyms) | Country | Vernacular Name (Ethnic Group) |
|---|---|---|
|
Lannea acida (Odina acida (A. Rich.) Oliv; Calesiam acidum (A.Rich.) Kuntze; Lannea glaucescens Engl.; Lannea lagdoensis (Engl. & K.Krause) Mildbr.; Sorindeia lagdoensis Engl. & K. Krause) [4,13,14] |
Benin | Tchemou (ta-kamba), yoronou (bariba), wansawatchemou (waama), zouzou (fon, goum) |
| Burkina Faso | Bembé (bambara), ébruhé, ébruké (attié), kondro (baoulé), sambagha, santuluga (mossi), siribu, sisubu (dagari), véké (senoufo) | |
| Ivory Coast | Béssomo (malinke), sinsàbgà (dagomba) | |
| Ghana | Gbentore (wale); manvora, vaaworo (lobi) | |
| Guinea-Bissau | Bembedja, bembem-hei, tchingole (fula); bémbô, irimusso (mandinga); betôlôdje (pepel); dôto (balanta); mantede (criolo); ututene (felupe do Senegal) | |
| Guinea-Conakry | Bembe nougou (malinké); tiouko, yiouko tioli, thionlli, touko (poular) | |
| Mali | Bembé (bambara); sìnsàbgà (dagomba); tinyoli (peuhl) | |
| Niger | Faru, tamarza (zarma) | |
| Nigeria | Faru (hausa) | |
| Senegal | Bembô (socé); bembéy (firdou, fouladou); tinoli (peul, tocolor); tuko (peul fouta–djalon) | |
| Togo | Eberg (gurmantché); gbednatun (moba); kisan, kizan (kabiyé); otchowé (akassélem) | |
|
Lannea alata (Calesiam alatum Engl.; Lannea minimifolia (Chiov.) Cufod.; Odina minimifolia Chiov.) [15] |
Kenya | Borana (wanreh); kumude (bejelo); samburu (mushiga); ngariso tharaka (mituungu) |
|
Lannea ambacensis [16] |
Angola | Mukumbu kakumbi, mucumbi, mukumbi, mungongolua, ngonjila, umbi |
|
Lannea angolensis [17] |
Angola | Bulukutu, omunthiwi (kikongo) |
|
Lannea barteri (Calesiam barteri Kuntze; Lannea kerstingii Engl. & K. Krause; Odina barteri Oliv; Lannea kertínger Engl. & K. Krause.) [18] |
Benin | Zuzugoto (fon) |
| Camaroon | Sorihi (fulfuldé) | |
| Guinea-Conakry | Tiuko (aub, fula-fulaar) | |
| Ivory Coast | Baule kondro, bembe, dinbé, peku (manding-maninka) | |
| Mali | Bembe, dagaari sisibigolo, sussuguté hausanamisinfara, moore sambituliga, sabagha (aub, begue) | |
| Nigeria | Báraá as mudas (bargery); tudi (hausa); faru (fulani, hausa) | |
| Sierra Leone | Dalalonke (susu) | |
| Togo | Benature, patandĕu, tingbatau (volkens); gurma (manga); met (tshaudjo); aku (yoruba-ife) | |
|
Lannea coromandelica (Calesiam grande (Dennst.) Kuntze, Dialium coromandelicum Houtt., Haberlia grandis Dennst., Odina gummifera Blume; Odina pinnata Rottler) [19,20] |
Bangladesh | Bhadi, bohar, ghadi, jail, jial bhandi, jiga, jigor, jiol, jir, jival, kasmala, lohar (-) |
| India | Annakara, dang paguel-kung, doka, doke, dumpidi, genjan, geru, ginyan, godda, gojal, gumpina, gumpini, jhingan, jingni, Jhingangummi, kalasan, kalayam, kamlai, kashmala, kekat, kiamil, ligna, magir, mohin, moi, mowen, moye, moyen, moyna, nanam, oddi, shimti, udi, uthi, vaddi, oti, ajasrngi (-) | |
| Myanmar | Maing (-) | |
| Nepal | Thulo dabdabe (halonre) | |
| Pakistan | Kembal (-) | |
|
Lannea edulis (Lannea nana Engl; Odina edulis Sond; Calesiam edule Kuntze.) [20,21] |
South Africa | Mutsambatsi (siswati); phepo (setswa-na); umtfokolovu, umgabunkhomo (isizulu); wildedruif (afrikaans) |
| Angola | Ngongolua, omungongolua (nyaneka); ngongwila, ungongwila (umubumbu) | |
| Burundi | Umutabataba (kirundi) | |
| Kenya | Masungubale (marachi) | |
| Rwanda | Imbatabata, umutabataba (kinyarwanda) | |
| Tanza-nia/Uganda | Lihambalimwe (kihehe); makavumba, navakumba (mbozi), mvumvu mkubwa (zaramo, tanzania), nekote (karamojong, ouganda), unahavumba (nyika) | |
| Zimbabwe | Mutsambatsi (shona) | |
|
Lannea gossweileri [22] |
Angola | African walnut, Gossweileri ash, Gossweileri false ash, Gossweileri Lannea |
|
Lannea humilis (Commiphora taborensis Engl.; Lannea bagir-mensis Engl.; Lannea tomentosa (Engl.) Engl.; Odina humilis Oliv.; Odina tomentosa Engl.; Tapirira humilis (Oliv.) Marchand.; Calesiam humile (Oliv.) Kuntze Calesiam tomentosum Engl.) [23] |
Nigeria | Kerwúlú, paàruú |
| Senegal | Ard a koy, habugan, béluki, ngonaro | |
| Uganda | Etopojo (ngakarimojong) | |
|
Lannea nigritana (Lannea afzelii Engl.; Lannea grossularia A. Chev.; Odina nigritana S. Elliot; Lannea glaberrima Engl. & K. Krause; Lannea nigritana var. nigritana Keay; Odina oghigee Hook.f.) [4,24] |
Guinea-Bissau | Bembedje, bembem-hei, tchingole (fula); bêmbô (mandinga); betôlôdje (pepel); mantede (criolo) |
| Guinea-Conakry | Bembé (malinké), lokouré (soussou) | |
|
Lannea rivcae (Commiphora tomentosa Engl; Lannea cufodontii Chiov; Lannea floccosa Sacleux; Odina rivae Chiov.) [20] |
Kenya | Kamba, kitharara, kithaala, kithaalua kya kiima, latat, lolowe, marakwet, muthaalwa |
|
L. schimperi (Lannea rufescens Engl.; Lannea ruspollii Engl.; Lannea schimperi var. glabrescens (Engl.) J.B. Gillett; Lannea stolzii Engl. & Brehmer; Odina schimperi Hochst. ex A. Rich.); Calesiam schimperi (Hochst. ex A.Rich.) Kuntze; Lannea schimperi var. peixe-boi; Lannea stolzii Engl. & Brehmer) [25] |
Burundi | Igifuto, umufute (kirundi) |
| Cameroon | Nkwelegito (babungo) | |
| Ethiopia | Enxxilif (afaan oromo) | |
| Mozambique | Munganikomo, xihumbunkany, xivombo nkanyi (changana) | |
| Namibia | Kangawa (lozi) | |
| Kenya | Kipng’etingwet, kumubumbu (nandi) | |
| Sudan | Tony (nuer) | |
| Tanzania | Mginkinywa (batemi); mugumbu (nyamwezi); navakumba (mbozi); ombumbo (haya) | |
|
Lannea schweinfurthii (Calesiam schweinfurthii (Engl.) Kuntze; Lannea schweinfurthii var. schweinfurthii; Odina schweinfurthii Engl.; Scassellatia heterophylla Chiov.) [26] |
South Africa | Mi-livhadza (luvenda); mulichadza (venda) |
| Mozambique | M’sutototo (chindau) | |
| Namibia | Rungomba (lozi) | |
| Kenya | Kuogo (luo); mnyumbu (kilifi); omusalu (suba); mumongoo (pokomo) | |
| Somalia | Arusha (eravande); gogo (muwumbu); lugu (muhingilo); mate (ndelamwana); mnyamendi, mribwampara, muhondobogo (zinza); msayu, nsayu (suku); mumendo, omosaruwa (kuria); mwera (mpupi); nyam (mnyumbu); pare (msighe); rangi (msakawa); swah (mtundu); tambaragi, thigii (iraqw); zara (mpiwipwi); zigua (mumbu) | |
| Tanzania | Mbu, mfupapo, mmongo, muumbu, nago (swahili); orpadwa (masaï) | |
| Uganda | Musinga bakali (bulamogi) | |
| Zambia | Musamba (silozi) | |
|
Lannea velutina (Calesiam velutinum (A. Rich.) Kuntze; Odina velutina Engl. ex Walp.; Tapirira velutina Marchand) [4,27] |
Benin | (-) |
| Burkina Faso | kruntoni (sanan), tougô-dâ, zinzam-tougô (bis-sa), wâamsâbga (mossi) | |
| Ghana | (-) | |
| Guinea-Bissau | Aionque (bijagós); ambi-lire (tanda); balêbári (the fruit); bembei, dembei, mantede (criolo); bem-bedje, bembei, bembem- hei, tchucó, tchingole (fula); bémbô (mandinga); be-tôlôdje (pel); coxolourô, cupote-cuxolourô (felupe do senegal); dôtô (balanta); lagari (manjaco); m’riuol (balanta); n’taluass, n’tchalúas, untchalbinass (nalu); n’tata, untata (pepel); sandje-bombo, sand-ji-bombro(fula); undêbári (cobiana) | |
| Guinea-Conakry | Bembé (malinké), tiouko, tiouko niadouko, tiouko niabé (poular) | |
| Mali | Bakororonpeku, fégou-ganiè, surukunnpeku (malinke); nteku-bangènyè, bakoro npeku (bambara); satungo npege, saanci jonon (minyanka); satungo vègè (senoufo); sa’ui-nyinu (bwa) | |
| Niger | (-) | |
| Senegal | Bemmbeyi (peul), bubu-ka (diola), ndabarndoki (serer), ndogot (wolof), tinolipoley (peul) | |
| Togo | (-) | |
|
Lannea welwitschii (Calesiam welwitschii Hiern; Lannea acidíssima A. Chev.; Lannea longifoliolata Engl. & K. Krause; Lannea zenkeri Engl. & K. Krause; Odina welwitschii K. Schum.; Ricinodendron staudtii Pax) [28] |
Angola | Nkumbi (kikongo) |
| Democratic Republic of the Congo | Kumbi (kikongo) | |
| Ivory Coast | Loloti, ngdongoloti (abe); kakoro (akanfante); n-nu, nu, tchico, tchiwo (akye); baiséguma, baopiré, bore pore (anyi); trongba (baule); tobero (gagu); tétégné (kru-guere); duko, durgo, duruku (kulango); adubruhia, atukruhia, dugbruhia (kyama); kakoro (nzema) | |
| Gabon | Okum-nini, kumenini, kum-anini (enti) | |
| Ghana | Kum-anini, kumenini, kum-onini, kuntunkuni (akan-asante); kakoro (fante); aberewa nyansiŋ, kum-anini, okum-nini (twi); kumenini (wasa); bopire (anyi-sehwi); abalapuli (nzema) | |
| Nigeria | Abe (loloti); anyisehwi (bopire); anyi (bai-séguma); asante (kuntunkuri); baule (trongba); ekika, ekika-ajá (yoruba); fante (kakoro); gagu (tobero); kulango (duko); kru-guere (tétégné); kyama (adubruhia) |
(-)—vernacular name or ethnic group not found.
2.2.2. Traditional Uses
Table 3 summarises the obtained data on the traditional medicinal uses of the 14 Lannea species. Results showed that for most of them there is little information about the exact methodology and duration of treatment. Bark (29%) and leaf (17%) were the most used plant parts, and the most reported symptoms and illnesses were related to infection symptoms (31%), gastrointestinal discomfort (14%), pain (12%), diarrhoea (9%), and inflammation (7%). Lannea coromandelica and Lannea edulis are the most reported species and are used in traditional medicine systems of 15 and 14 countries, respectively.
Other Lannea species, like L. acida, are employed in tropical Africa to treat and manage bacterial, fungal, and viral infections, fever, and mental and gastrointestinal disorders. For example, L. acida is used to treat dysentery, stomach pain, and other gastrointestinal pathologies [29]; L. microcarpa is used for the treatment of mouth blisters, rheumatism, dysentery, diarrhoea, gastroenteritis, malaria, and bacterial infections [30]; L. schweinfurthii is used for the treatment of diseases related to the reproductive system, circulatory system, and gastrointestinal diseases, headaches, and against opportunistic diseases related to HIV, such as malaria, diarrhoea, tuberculosis, and skin infections [31].
Lannea ambacensis is known to be used in traditional Angolan medicine, particularly in the treatment of diabetes, rheumatism, and symptoms of respiratory, gastrointestinal, and urogenital diseases [32].
Table 3.
Traditional uses of Lannea species by geographical region.
| Species | Distribution | Medicinal Uses | Plant Part |
|---|---|---|---|
|
Lannea acida [33] |
Benin, Burkina Faso, Ghana, Guinea-Bissau, Ivory Coast, Niger, Nigeria, Senegal, Togo | Antipyretic, gastrointestinal tract disorder, malaria, pain, skin disease, and sexually transmitted disease (gonorrhoea, syphilis) | Branch, root, stem, stem bark |
|
Lannea alata [15] |
Kenya, Somalia, South Africa, Tanzania | Fever, fractures, malaria | Stem |
|
Lannea ambacensis [16] |
Angola | Asthma, colitis, cough, eye diseases, ulcer | Root |
|
Lannea angolensis [17] |
Angola | Bronchitis, pleuropneumonia, pneumonia, rhinitis, tuberculosis | Bark |
|
Lannea barteri [34] |
Benin, Burkina Faso, Burundi, Democratic Republic of the Congo, Ethiopia, Ghana, Guinea-Conakry, Ivory Coast, Mali, Nigeria, Uganda, Zaire |
Anaemia, convulsions, diabetes, oedema, epilepsy, leprosy, madness, paralysis, salmonellosis, spasms, vermifuge | Bark, leaf Stem bark |
|
Lannea coromandelica [19,20] |
Andaman, Assa, Bangladesh, Cambodia, Guangdong, Guangxi Hainan, India, Laos, Myanmar, Nepal, Sri Lanka, Thailand, Vietnam, Yunnan | Heart disease, inflammations, leprous ulcers, mouth sores, pain, rashes, sprains, toothache | Bark, leaf |
|
Lannea edulis [35] |
Angola, Botswana, Burundi, Democratic Republic of the Congo, Ethiopia, Kenya, Malawi, Mozambique, Rwanda, South Africa, Tanzania, Uganda, Zambia, Zimbabwe | Bilhárzia and other parasitoses, cholera, contusion, diarrhoea, fever, food, haematoma, malaria, sexually transmitted disease (gonorrhoea, syphilis), swelling, tuberculosis, wound | Fruit, leaf, root, root bark, stem |
|
Lannea humilis [23] |
Ethiopia, Senegal, Zambia, Zimbabwe | Body aches, cholera, cough, diarrhoea, dysentery, nausea, weakness | Bark |
|
Lannea nigritana [24] |
Benin, Cameroon, Central African Republic, Congo (Brazzaville), Equatorial Guinea, Gambia, Ivory Coast, Liberia, Mali, Nigeria, Senegal, Sierra Leone, Togo | Anaemia, bad odour, cachexia, chest stiffness, drepanocytosis, dysentery, impotence, intestinal pain, purgative, rickets, tiredness | Bark |
|
Lannea rivae [36] |
Ethiopia, Kenya, Tanzania, Uganda | Cold, cough, stomach-ache | Bark |
|
Lannea schimperi [37] |
Burundi, Cameroon, Congo, Ethiopia, Kenya, Malawi, Mozambique, Nigeria, Rwanda, Tanzania, Togo, Uganda, Zambia | Back pain and general weakness, diarrhoea, dysentery, infections, stomach pain, tuberculosis | Bark, branch, leaf, stem, trunk |
|
Lannea schweinfurthii [38] |
Botswana, Ethiopia, Kenya, Malawi, Mozambique, Rwanda, Somalia, Sudan, Tanzania, Uganda, Zambia, Zimbabwe | Abdominal pain, anaemia, diarrhoea, food, gastric ulcer, headaches, sexually transmitted diseases (chlamydia, gonorrhoea, syphilis), stomach problems, tonic | Bark, leaf, stem bark |
|
Lannea velutina [27] |
Burkina Faso, Central African Republic, Chad, Ghana, Guinea-Bissau, Senegal | Anaemia, asthenia, cachexia, cholera, conjunctivitis, cuts, diarrhoea, dysentery, ectoparasites (flea, leech, lice, mite, tick), fever, impotence, inflammation, pain, rash, renal colic, skin growths, tuberculosis, wound | Bark, fruit, leaf, root |
|
Lannea welwitschii [39] |
Angola, Cameroon, Central African Republic, Congo, Ethiopia, Gabon, Ghana, Ivory Coast, Liberia, Nigeria, Uganda, Zaïre | Diarrhoea, dysentery, oedema, epilepsy, food, gout, haemorrhoids, hypertension, laxative, nasopharyngeal disorders, pulmonary diseases, purgative, venereal diseases | Bark, root |
2.3. Phytochemical Studies
The results of the chemical studies conducted on Lannea species are summarised in Table 4. Most studies focused on leaf and bark plant parts. Polyphenolic compounds, including hydroquinones, phenolic acids, flavonoids, and terpenoids, namely triterpenoids, are the major classes of compounds identified in this botanical genus. Other terpenoid derivatives and fatty acids were also commonly identified.
Among the total 160 compounds identified in Lannea species, 57 (34.5%) are flavonoids (quercetin and myricetin flavonols) and condensed tannins like catechin and epicatechin, also containing a galloyl group. As in other Anacardiaceae species, proanthocyanidins are representative secondary metabolites found in all parts of the plant, mainly in the bark.
Table 4.
Secondary metabolites of Lannea species.
| Species, Ref. | Plant Part | Chemical Class | Compound |
|---|---|---|---|
| L. acida [40,41,42,43,44,45] | Whole plant | Flavonol | Quercetin |
| Leaf | Flavanone | 6,7-(2″,2″ -dimethyl chromene)-8-γ, γ-dimethyl allyl flavanone | |
| Flavonol | 3′,4′dihydroxy-7,8(2″,2″-dimethyl chromene)-6-γ, γ dimethyl allyl flavonol | ||
| Isoflavone | 7-methyltectorigenin | ||
| Isoflavone | Irisolidone | ||
| Flavonol glycoside | Myricetin-3-O-α-L-rhamnopyranoside | ||
| Flavonol glycoside | Myricetin-3-O-β-D-glucopyranoside | ||
| Flavonol glycoside | Myricetin-3-(6″-galloylgalactoside) | ||
| Gallic acid derivative | 3,4,5-trigalloylquinic acid | ||
| Flavan-3-ol | (-)-Epicatechin-3-gallate | ||
| Flavan-3-ol | (-)-Epigallocatechin-3-gallate | ||
| Flavan-3-ol | (-)-Epigallocatechin | ||
| Flavan-3-ol | (-)-Epicatechin | ||
| Flavone | Lanceolatin B | ||
| Flavanone | 7,2′-dimethoxy-4′,5′methylenedioxyflavanone | ||
| Eugenol derivative | Eugenyl-O-β-D-(6′-sulphonylglucoside) | ||
| Flavonol glycoside | Quercetin-3-O-β-D-glucuronic acid | ||
| Flavonol glycoside | Quercetin-3-O-β-D-glucopyranoside | ||
| Flavonol glycoside | Quercetin-3-(6″-galloylglucopyranoside) | ||
| Stem bark | Flavone | Luteolin | |
| Flavonol | Kaempferol | ||
| Fatty acid | Hexadecanoic acid (20.59%) | ||
| Fatty acid | Trans-13-octadecenoic acid decanoic acid (2.16%). | ||
| Fatty acid | 7,10-octadecanoyl acid | ||
| Fatty acid | Hexadecanoic acid | ||
| Fatty acid | Ecadienoic acid | ||
| Fatty acid | Eicosanoic acid (7.62%) | ||
| Fatty acid | Dodecanoic acid (8.51%) | ||
| Fatty acid | Octadecanoic acid (13.77%) | ||
| Fatty acid | Tetradecanoic acid (18.18%) | ||
| Methyl ester | Methyl ester (4.86%) | ||
| Methyl ester | Methyl ester (7.70%) | ||
| Ester | Methoxy acetic acid, 2-tetradecyl ester | ||
| Phthalate ester | Dibutyl phthalate (4.12%) | ||
| Root bark | Phenol derivative | (E)-3-(hepatic-14-enyl)phenol | |
| Phenol derivative | (E)-3-(nonadec-16-enyl)phenol | ||
| Benzene derivative | (E)-2-(heptadec-14-enyl)benzene-1,4-diol | ||
| Cyclohexenone | (5R,14E)-5-(heptadec-14-enyl)-5-hydroxycyclohex-2-en-1-one | ||
| Cyclohexenone | (5R,16E)-5-(nonadec-16-enyl)-5-hydroxycyclohex-2-en-1-one | ||
| Cyclohexene diol | (1S,3S)-1-((E)-heptadec-14-enyl)cyclohex-4-ene-1,3-diol | ||
| Cyclohexene diol | (1S,3S)-1-((E)-nonadec-16-enyl)cyclohex-4-ene-1,3-diol | ||
| Cyclohexene diol | (1S,3S)-1-((E)-heneicos-18-enyl)cyclohex-4-ene-1,3-diol | ||
| Bicyclic alcohol | (1S,3S,6R)-1-((E)-heptadec-14-enyl)-7-oxabicyclo [4.1.0]hept-4-en-3-ol | ||
| Bicyclic alcohol | (1R,3R,6S)-1-((E)-nonadec-16-enyl)-7-oxabicyclo[4.1.0]hept-4-en-3-ol | ||
| Cyclohexenone | (4R,5S)-5-((E)-heptadec-14-en-1-yl)-4,5-dihydroxy-cyclohex-2-en-1-one | ||
| L. alata [46,47] | Whole plant | Flavonol | Lanneaflavonol |
| Flavonol | Dihydrolanneaflavonol | ||
| Flavonol glycoside | Myricetin-3-O-α ramnopyranoside | ||
| Flavonol glycoside | Myricetin-3-O-α-arabinofuranoside (betmidin) | ||
| Triterpene | Lupeol | ||
| Phytosterol | ß-sitosterol | ||
| L. barteri [48] | Leaf | Flavonol glycoside | Kaempferol-3-O-rhamnoside |
| Flavonol glycoside | Myricetin-3-O-rhamnoside | ||
| Flavonol | Quercetin-3,7,3′,4′-tetramethyl | ||
| Flavonol glycoside | Quercetin-3- O-arabinofuranoside | ||
| Flavonol glycoside | Quercetin-3-O-galactoside (hysperoside) | ||
| Flavonol glycoside | Quercetin-3-O-rhamnoside (quercetrin) | ||
| L. coromandelica [49,50,51] | Bark | Lipid derivative | (2S,3S,4R,10E)-2-[(2R)-2-hydroxytetracosanoyl amino]-10-octadecene-1,3,4-triol |
| Phenolic aldehyde | Isovanillin | ||
| Glycosphingolipid | Aralia cerebroside | ||
| Saturated fatty acid | Palmitic acid | ||
| Saturated fatty acid | Stearic acid | ||
| Phenolic acid | Protocatechuic acid | ||
| Oestrogenic compound | P-hydroxybenzoic acid ethyl ester | ||
| Organic compound | 5,5-dibuthoxy-2,2-bifuran | ||
| Phytosterol ester | Phytosterol-β-sitosterol palmitate | ||
| Sterol glycoside | Β-sitosteryl-3β-glucopyranoside-6-O-palmitate | ||
| Triterpene | Myricadiol | ||
| Leaf | Flavonol | Quercetin | |
| Flavonol glycoside | Quercetin-3-arabinoside | ||
| Flavan-3-ol | Leucocyanidin | ||
| Flavan-3-ol | Leucodelphinidin | ||
| Flower, stem bark | Phytosterol | ß-Sitosterol | |
| Flavonol glycoside | Isoquercetin | ||
| Flavonol | (2R, 3S)-(+)-4,7-di-O-methylhydroquercetin | ||
| Flavonol | (2R, 3S)-(+))-4-O-methyldihydroquercetin | ||
| Flavonol | (2R, 3S)-(+) 3,5-dihydroxy-4,7dimethoxydihydroflavonol | ||
| Flavonol | (2R, 3S)-(+)-4,5,7-trimethoxydihydroflavonol | ||
| Flavonol | (2R, 3S)-(+)-4,7-di-O-methyldihydrokaemferol | ||
| Flavonol | Morin | ||
| Oligosaccharide | 4-O-(α-D-galactopyranosyluronic acid)-D-galactose | ||
| Oligosaccharide | 6-O-(ß-D-glucopyranosyluronic acid)-D-galactose | ||
| Oligosaccharide | 6-O-(4-O-methyl-D-glucopyranosyluronic acid)-D-galactose. | ||
| L. edulis [52] | Root bark | Phenolic lipid | Cardonol 7 |
| Phenolic lipid | Cardonol 13 | ||
| Cyclohexenone | 5-[14-heptadecenyl]-4,5-dihydroxy-2-cyclohexenone | ||
| Cyclohexenone | 5-[16-nonadecenyl]-4S,5S-dihydroxy-2-cyclohexenone | ||
| Cyclohexenone | 5-[16-Nonadecenyl]-4,5-dihydroxy-2-cyclohexenone. | ||
| L. humilis [53] | Bark | Dicarboxylic acid | Malic acid |
| Hydroxycinnamic acid | Quinic acid | ||
| Gallotannin | Gallic acid glucoside | ||
| Flavan-3-ol | (Epi)gallocatechin | ||
| Flavan-3-ol sulfate ester | (epi)gallocatechin 5-O-methyl 7-O-sulphate | ||
| Flavan-3-ol | (Epi)catechin | ||
| Flavan-3-ol gallate | (Epi)-gallocatechin gallate | ||
| Flavan-3-ol sulfate ester | 3-flavan 3-,4-,5- trihydroxy5-O-methyl 7-O-sulphate | ||
| Sulfated phenolic acid | Syringic acid sulphate | ||
| Flavan-3-ol sulfate ester | (epi)catechin 5-O-ethyl 7-O-sulphate-3-O-hexoside | ||
| Flavan-3-ol sulfate ester | (epi)catechin 5-O-ethyl 7-O-sulphate | ||
| Flavan-3-ol gallate | Procyanidin dimer mono gallate | ||
| Flavan-3-ol gallate sulfate ester | (epi)gallocatechin gallate 5-O-ethyl 7-O- sulphate. | ||
| L. rivae [46,54] | Root | Carotenoid | E-lutein |
| Flavan-3-ol gallate | (-)-epicatechin-3-O-gallate | ||
| Flavonol | Myricetin | ||
| Phenol derivative | 3-nonadec-14′-Z-enyl phenol | ||
| Phenol derivative | 3-heptadec-12′-Z-enyl phenol | ||
| Phenol derivative | 3-pentadec-10′-Z-enyl phenol | ||
| Phenol derivative | 3-pentadecyl phenol | ||
| Furanone | 4,5-dihydroxy-4,5-furan-2′-[16′-(Z)-18′-(E)-heneicosenyldiene] cyclohex-2-enone | ||
| Cyclohexanone | 2,4,5-trihydroxy-2-[16′-(Z)-heneicosenyl] cyclohexanone | ||
| Cyclohexenone | 4S,6R-dihydroxy-6-(12′(Z)-heptadecenyl) 2-cyclohexenone | ||
| Cyclohexenone | 4S,6R-dihydroxy-6-(14′(Z)-nonadecenyl) 2-cyclohexenone | ||
| Cyclohexane | 1,2,4-trihydroxy-4-[16′(Z)-heneicosenyl] cyclohexane. | ||
| Sterol glycoside | Sitosterol glucoside | ||
| Triterpenoid | Β-sitosterol | ||
| Triterpenoid | Taraxerol | ||
| Triterpenoid | Taraxerone | ||
| L. schimperi [54,55,56] | Whole plant | Phenol derivative | 3-[12′(E)-pentadecenyl] fenol |
| Phenol derivative | 3-[14′(E)-heptadecenyl] fenol | ||
| Phenol derivative | 3-[16′(E)-nonadecenyl] fenol | ||
| Phenol derivative | 3-[18′(E)-heneicosenyl] fenol | ||
| Cyclohexenone | 5-[12′(E)-pentadecenyl] 4,5-dihydroxyciclohex-2-enone | ||
| Cyclohexenone | 5-[14′(E)-heptadecenyl] 4,5-dihydroxyciclohex-2-enone | ||
| Cyclohexenone | 5-[16′(E)-nonadecenil] 4,5-dihydroxyciclohex-2-enone | ||
| Cyclohexenone | 5-[18′(E)-heneicosenyl] 4,5-dihydroxycyclohex-2-enone | ||
| Cyclohexenol | 1-[12′(E)-pentadecenyl] cyclohex-3-en-1,2,5-triol | ||
| Cyclohexenol | 1-[14′(E)-heptadecenyl] cyclohex-3-en-1,2,5-triol | ||
| Cyclohexenol | 1-[16′(E)-nonadecenyl] cyclohex-3-en-1,2,5-triol | ||
| Cyclohexenol | 1-[14′(E)-heptadecenyl] 4-cyclohex-4-en-1,3-diol | ||
| Cyclohexenol | 1-[16′(E)-nonadecenyl] 4-cyclohex-4-en-1,3-diol | ||
| Cyclohexenol | 1-[18′(E)-heneicosenyl] 4-cyclohex-4-en-1,3-diol. | ||
| Leaf | Lipid | Ceramide | |
| Alkaloid | Forsskamide | ||
| Isoprenoid | A-tocopherol | ||
| Triterpenoid | Betulinic acid | ||
| Triterpenoid | Lupeol | ||
| Triterpenoid | Oleanolic acid | ||
| Triterpenoid | 23-hydroxyoleanolic acid. | ||
| L. schweinfurthii [46] | Root | Phenol derivative | 3-[tridecyl] phenol |
| Phenol derivative | 3-[heptadecyl] phenol | ||
| Phenol derivative | 3-[heptadec-12′(Z),14′(E)-dienyl] phenol | ||
| Phenol derivative | 3-[nonadec-14′(Z),16′(E)-dienyl] phenol | ||
| Phenol derivative | 3-[heneicos-16′(Z),18′(E)-dienyl] phenol | ||
| Flavan-3-ol | Catechin | ||
| Flavan-3-ol | Epicatechin | ||
| Favonol rutinoside | Rutin | ||
| Triterpenoid | Lupenone | ||
| Cyclohexenol | 1-[tridecyl] cyclohex-3-en-1,2,5-triol | ||
| Cyclohexenol | 1-[heptadecyl] cyclohex-3-en-1,2,5-triol | ||
| Cyclohexenol | 1-[tridecyl] cyclohex-4-en-1,3-diol | ||
| Cyclohexenol | 1-[nonadecyl] cyclohex-4-en-1,3-diol | ||
| Cyclohexenol | 1-[heneicosyl] cyclohex-4-en-1,3-diol | ||
| Cyclohexenol | 1-[tricosyl] cyclohex-4-en-1,3-diol | ||
| Cyclohexenol | 1-[pentadec-12′(E)-enyl] cyclohex-4-en-1,3-diol | ||
| Cyclohexenol | 1-[nonadec-14′(Z),16′(E)-dienyl] cyclohex-4-en-1,3-diol | ||
| Cyclohexenol | 1-[heneicosen-16′(Z),18′(E)-dienyl] cyclohex-4-en-1,3 diol | ||
| Cyclohexenone | 5-hydroxy-5-[tridecyl] cyclohex-2-enone | ||
| Cyclohexenone | 5-hydroxy-5-[pentadecyl] cyclohex-2-enone | ||
| Cyclohexenone | 5-hydroxy-5-[heptadecyl] cyclohex-2-enone | ||
| Cyclohexenone | 5-hydroxy-5-[pentadec-12′(E)-enyl] cyclohex-2-enone | ||
| L. velutina [57,58,59] | Root bark | Flavan-3-ol | Catechin (as starting unit) |
| Flavan-3-ol | Epicatechin (as an extender unit). | ||
| Leaf | Phenolic lipid | Anacardic acid | |
| Phenolic acid | Gallic acid | ||
| Flower | Sesquiterpenoid | Beta-caryophyllene 22 to 36% | |
| Alkane | Heneicosane 4 to 10%. | ||
| L. welwitschii [42,60] | Whole plant | Phenolic compound | Lanneaquinol |
| Phenolic compound | 2′(R)-hydroxylanneaquinol. | ||
| Leaf | Flavonol | Mearnsetin | |
| Flavonol glycoside | Myricetin 3-O-β-D-arabinofuranoside | ||
| Flavonol glycoside | Myricetin-3-O-β-D-glucuronic acid | ||
| Flavonol glycoside | Myricetin-3-O-β-D-xylofuranoside | ||
| Flavonol glycoside | Myricetin-3-O-β-D-galactopyranoside |
2.4. Biological Studies
Biological studies were conducted in vitro and in vivo using extracts prepared with different plant parts of Lannea species using, namely, the aerial part, bark, leaf, stem, root, stem and root bark, and the whole plant (Table 5). Most plant extracts were prepared with methanol or ethanol as solvents, and the bark and leaf of Lannea species were the most frequently used plant parts.
L. acida stem bark aqueous extract showed anti-diarrhoeal and anti-inflammatory activity-inhibition of prostaglandin E2 in the paw oedema method [61]; hydroalcoholic extract of the bark and the whole plant showed in vitro antioxidant activity and cytotoxic and anti-Mycobacterium tuberculosis H37Rv activities [62,63]; ethanolic extract of L. acida bark revealed in vitro antibacterial properties against Gram-negative (Escherichia coli and Pseudomonas aeruginosa) and Gram-positive (Staphylococcus aureus, Enterococcus faecalis, Streptococcus pyogenes, and Bacillus subtilis), including against resistant antibiotic strains and also oestrogenic activity and anti-osteoporotic potential in the ovariectomized Wistar rat model [64].
The in vitro antibacterial activity against S. aureus and antioxidant activity exhibited by L. alata were attributed to the presence of prenylated flavonoids, epicatechin gallate, betamidine, and myricetin [47].
Quantitative evaluation of the inhibitory (MIC) and bactericidal (MBC) concentrations of methanolic extracts of the bark, stem, and root of L. barteri, against S. aureus, Staphylococcus epidermidis, Proteus mirabilis, E. faecalis, E. coli, and P. aeruginosa, confirmed that this medicinal plant has significant antibacterial and antifungal activities [65,66,67].
The biological properties of L. coromandelica are numerous (Table 5). A stem bark extract has shown in vitro antimicrobial, hypotensive, and sporicidal activities [68]; studies on the bark revealed in vivo anti-diarrhoeal activity and in vitro antimicrobial activities [69,70,71]; the presence in stem bark of dihydroflavonols and terpenoids, polyphenols, flavonoids, kaempferol, and quercetin provided in vivo hepatoprotective and antioxidant activities to this medicinal plant [72].
According to Sohni et al., 1995, L. edulis whole plant water extract showed low in vitro mutagenic activity against Salmonella typhimurium and antioxidant activity [73].
Ethanolic and methanolic extracts of different parts of L. velutina showed selective in vitro antimicrobial activity against Cladosporium cucumerinum and Candida albicans; larvicidal against Aedes aegypti, Anophelis gambiae, and Culex quinquefasciatus; molluscidal against Biomphalaria glabrata, Biomphalaria pfeifferi, and Bulinus truncatus; and antioxidant activity; lipophilic root bark and hydroalcoholic stem extracts showed in vivo antioxidant and 15-lipoxygenase inhibitory activities [57,58,74].
In vitro decoction of L. nigritana leaf showed selective antimicrobial activity against seven reference strains and clinical isolates of M. ulcerans [63].
Root and stem extracts of L. rivae containing 2,4,5-trihydroxy-2-[16′-(Z)-heneicosenyl] cyclohexanone and 4,5-dihydroxy-4,5-furan-2’-[16′-(Z)-18-(E)-heneicosenyldiene] cyclohex-2-enone as marker compounds showed significant in vitro cytotoxicity in human tumour cell lines; root and stem hexane and dichloromethane extracts showed antibacterial activity against E. faecalis and S. aureus; dichloromethane/methanol (1:1) root extracts and the isolated compounds epicatechin gallate and (4R, 6S)-4,6-dihydroxy-6-((Z)-nonadec-14′-en-1-yl)cyclohex-2-en-1-one reduced carrageenan-induced oedema [36,54,73,75].
According to Mikail H. et al., 2016, L. schimperi methanolic leaf extracts demonstrated in vitro and in vitro anticoccidial activities [37,76].
Methanol, hexane, and ethyl acetate stem bark extracts of L. schweinfurthii showed significant in vitro antimicrobial activity against C. albicans, B. subtilis, E. coli, P. aeruginosa, and S. aureus [77]. In vitro acetylcholinesterase inhibitory activity (ACHE) was exhibited by a root ethyl acetate extract (IC50 = 0.3 ± 0.0 µg/mL), as being higher than that of galantamine control (0.53 µg/mL) [78].
L. velutina bark and leaf ethanolic extracts showed antioxidant and antimicrobial in vitro activities. Anacardic acid has previously been identified as one of the major compounds present in this medicinal plant [57,58,62,74,79,80].
L. welwitschii was also the object of different biological activity studies, like analgesic, in which the total analgesic effect of the hydroethanolic stem bark extract significantly increased in a dose-dependent manner; antibacterial activity was observed for the methanolic leaf extract against Enterococcus faecalis, Klebsiella pneumoniae, Proteus mirabilis, P. aeruginosa, Staphylococcus aureus, and Escherichia coli strains resistant to pefloxacin, with MIC values of 5, 10, 5, 2.5, and 2.5 mg mL−1, respectively against E. coli, P. aeruginosa, S. aureus, and B. subtilis compared to ciprofloxacin (0.025, 0.055, 0.025, and 0.02 mg/mL); antioxidant activity was exhibited by the free radical scavenging method (2,2-diphenyl-1-picrylhydrazyl (DPPH)), with an IC50 value of 81.8 µg/mL compared to that of α-tocopherol (1.5 µg/mL); antidiarrhoeal activity was observed for the bark aqueous extract (50–400 mg/kg), with a significant (p < 0.05) delay in the onset of profuse diarrhoea and reduction in intestinal fluid volume; anti-inflammatory activity at 200 mg/kg dose had an inhibition of 14.49 ± 2.43% compared to the control in the paw oedema method, while the total oedema induced over the 6 h was 37.19 ± 4.38%. The maximum inhibitory effects were verified with a dose of 400 mg/kg. Myricetin, a common phenolic compound present in several plants, has previously been identified in L. welwitschii [60,81,82].
L. acida was the most studied Lannea species, followed by L. coromandelica and L. velutina. Different biological activities were observed, but the predominant ones were by far antimicrobial, antioxidant, and anti-inflammatory activities.
Table 5.
In vitro and in vivo biological studies on Lannea species.
| Species | Plant Part | Extract | Test | Results | Refs |
|---|---|---|---|---|---|
| Lannea acida | Wp | EtOH | In vitro: antibacterial activity | Potential source of new antibacterial agents against Gram-negative (Escherichia coli and Pseudomonas aeruginosas) and Gram-positive (Staphylococcus aureus, Enterococcus faecalis, Streptococcus pyogenes and Bacillus subtilis); crude extract showed bactericidal and bacteriostatic activity (IC50 values between 12 and 94 µg/mL). | [64] |
| Wp | H2O, MeOH | In vivo: reproductive toxicity of colibri in adult male rats | Treatment with L. acida extracts was significant (p ≤ 0.05–0.001) because it reversed the reproductive system-induced damage, especially after 28 days of treatment with aqueous solution (340 mg/kg) and methanol extracts (170 mg/kg). | [83] | |
| Wp | EtOH | In vivo: antibacterial activity by microdilution in broths of bacterial strains | Selective antibacterial activity against Gram-negative (E. coli and P. aeruginosa) and Gram-positive (S. aureus, E. faecalis, S. pyogenes, and B. subtilis), including against resistant strains, with MICs/MBCs ranging from 7.80 to 125 µg/mL. The highest sensitivity was seen against Bacillus subtilis and Pseudomonas aeruginosa. | [62] | |
| B | EtOH | In vitro: Folin Method–Ciocalteu (antioxidant activity) | Determination of total phenolic compounds and flavonoids by the Folin Ciocalteu method, expressed in mg of gallic acid equivalents and quercetin equivalents, respectively (total phenols vary between 34.4 to 40.55; total flavonoids vary between 6.4 and 11.02). | [40] | |
| B | EtOH | In vitro and in vivo: evaluation of oestrogenic activity and anti-osteoporotic potential in ovariectomized Wistar rats | L. acida bark extract induced proliferation of MCF-7 cells. At 200 mg/kg, prolonged treatment with the extract prevented ovariectomy-induced body weight gain and loss of bone mass and/or density. The ethanol extract induced a significant increase in MCF-7 cell production at concentrations of 10 (p < 0.05), 100 (p < 0.05), and 200 (p < 0.01)/g/mL compared to control DMSO. | [84] | |
| StB | Hx, Chl, Ace | In vitro: antimicrobial activity | The antimicrobial test result showed that stem bark extracts exhibited antimicrobial activity against several microorganisms (Bacillus cereus, Escherichia coli, Klebsiella pneumonia, Pseudomonas aeruginosa, Staphylococcus aureus, and Streptococcus pyogenes), with clear zones of inhibition ranging from 6 mm to 21 mm. | [85] | |
| StB | H2O | In vivo: anti-inflammatory activities by method PGE E-2-induced paw oedema | The extract inhibited paw oedema significantly (F(3,96) = 25.02; p < 0.05) and (F(5,96) = 16.46; p < 0.01) at doses of 100 mg/kg and 300 mg/kg, respectively. However, the extract did not show significant inhibition at 30 mg/kg (F(15,96) = 1.12; p = 0.3505). Aqueous extract inhibited prostaglandin E2-anti-inflammatory activity. | [61] | |
| B | EtOH | In vitro: antioxidant activity by DPPH | Antioxidant activity through DPPH method using quercetin and gallic acid as positive controls. The IC50 value of each extract was determined and all tests were performed in triplicate. The bark extract of Lannea acida showed IC50 = 345.72 ± 7.76 μg mL−1 while that of Lannea velutina IC50 = 478. 68 ± 8.55. | [40] | |
| R B | DCM | In vitro: antiproliferative activity | The XTT assay was used to evaluate the antiproliferative activity of the extract, fractions, and compounds on three multiple myeloma cell lines: RPMI 8226, MM.1S, and MM.1R. Fractions were considered active when they inhibited at least 50% of cell growth at 20 μg/mL; two compounds showed activity on all cell lines with IC50 values < 5 µM. Bortezomib was used as a positive control. | [44] | |
| Wp | EtOH | In vitro: cytotoxic and anti-Mycobacterium tuberculosis H37Rv activities | The rate of monocytes at different stages of mitosis was corrected in the absence and presence of the extract as follows: G0/G1 58.83–59.83%; synthesis 21.95–18.64%; mitosis 16.67–15.97%; necrosis 2.65–5.64%. The percentage of inhibition of Mycobacterium tuberculosis proliferation was 77.6 and 36.8%, respectively, for 1.2 and 0.6 mg mL−1 of extract. | [62] | |
| Lannea barteri | L and St | MeOH | In vitro: antibacterial activity using the agar well diffusion method | MBC determination showed that the MBC ranges for methanolic and ethanolic extracts of L. barteri leaves were 6.25 to 50 mg/mL and 6.25 to 12.5 mg/mL, respectively. The rapid death of S. aureus was verified in the range of 1.45 × 106 CFU of minimum bactericidal concentration (MBC) of methanolic leaf extract of L. barteri. | [66] |
| L, StB | DCM, MeOH, H2O | In vitro: anticancer activity | The extracts and fractions were tested for anticancer activity by using the crystal violet cell proliferation on four adherent human carcinoma cell lines. The inhibitory concentration (IC50) of fractions IH, 1I, 2E, and 2F were: 3.75 ± 1.33, 3.88 ± 2.15, 0.53 ± 0.41, and 0.42 ± 0.45 µg/mL against KYSE 70 and 1.04 ± 0.94, 2.69 ± 1.17, 2.38 ± 3.64, and 2.17 ± 1.92 µg/mL against SiSo cell lines, respectively. Fraction 2E showed weak apoptotic activity at double the IC50 and some sign of cell cycle arrest in the G2/M phase | [86] | |
| Lannea coromandelica | L | EtOAc, MeOH, H2O | In vitro: antioxidant activity by DPPH method | The ethyl acetate fraction had stronger DPPH scavenging activity than the methanolic extract and aqueous extract fractions. The DPPH clearing effect of both standards and plant extracts occurred in the order of BHT > EAF > CME > AqF and was 91.9%, 71.4%, 56.2%, and 42.2% at a concentration of 100 µg/mL, respectively. | [87] |
| Wp | EtOH | In vivo: hypotensive activity | The ethanolic extract of L. coromandelica was administered to dogs and rats at doses 5–100 mg/kg and 1–25 mg/kg, respectively, and a reduction in blood pressure was observed. | [88] | |
| L | EtOH:H2O | In vivo: anti-ulcer activity model | L. coromandelica anti-ulcer activity was evaluated in two different in vivo models of induced gastric ulcer. Leaf hydroethanolic extract showed significant levels of ulcer inhibition and gastric protection. | [89] | |
| L | MeOH | In vitro: neuropharmacological and antidiabetic activity | Rats received doses of 100, 150, and 200 mg/kg of body weight in an elevated plus maze and motor coordination; 100 and 200 mg/kg of body weight in sleep time, hole crossing, hole plate, and open field testing; and 200 and 400 mg/kg body weight in the antidiabetic activity test. The results obtained were all significant and dose dependent. L. coromandelica extracts possess significant neuromodulatory properties, had no significant effect on normal blood sugar levels, but corrected alloxan-induced changes in blood sugar and pancreas. | [90] | |
| B | MeOH | In vitro: antioxidant activity by DPPH method | The percentage of free radical scavenging by the DPPH, with IC50 12.12 ± 0.48 µg/mL compared to the ascorbic acid standard 8.66 ± 0.11 µg. | [84] | |
| L | EtOH | In vitro: antidiabetic activity in rats | Blood glucose levels in normal rats reached high levels 60 min after oral glucose administration (3 g/kg) and gradually decreased to 125 mg/dL in 2 h. Groups pretreated with ethanolic extract of L. coromandelica (100 and 200 mg/kg) and metformin (250 mg/kg) had induced decreased blood glucose levels significantly (p < 0.05) compared with that of the control group. | [56] | |
| B | MeOH | In vivo: castor oil-induced antidiarrhoeal activity | The extract considerably reduced the number of diarrhoeal episodes compared to control animals. The bark extract of L. coromandelica at a dose of 200 mg/kg showed a significant reduction (p < 0.05) of 68.86% in the number of faecal episodes, compared to the antidiarrheal drug, loperamide which has 89, 14% protection. | [69] | |
| L | MeOH | In vivo: aspirin-induced antiulcer activity | The test was performed on albino rats weighing between 150 and 200 g, using an aqueous suspension of aspirin at a dose of 200 mg/kg orally for 8 days. The result was a significant decrease in the ulcer index, with the percentage of gastric protection of 17.3% (standard), 78.29% (positive control), 30.57% (low dose), and 62.76% (high dose), and a significant reduction in the volume of gastric juice and acidity and increase in pH. | [91] | |
| B | MeOH | In vitro: antibacterial activity | Methanolic extract of L. coromandelica revealed a significant moderate antibacterial activity against Staphylococcus aureus, Salmonella typhi, Shigella dysenteriae, Pseudomonas aeruginosa, and Escherichia coli; there was no activity against Shigella boydii, however, there was a greater zone of inhibition against Escherichia coli (inhibition zone of 15.59 ± 0.22 mm), followed by Staphylococcus aureus and Shigella dysenteriae. | [69] | |
| B | EtOH | In vivo: thioacetamide-induced hepatoprotective and antioxidant activity in rats | Hepatotoxicity was induced by thiocetamide 100 mg/kg subcutaneously in male Wistar rats, causing marked changes in serum AST, ALT, ALP, and serum bilirubin and reduced serum concentration of total proteins, albumin, sodium, and potassium compared to those in the control (p < 0.05). The results showed that the hydroalcoholic extracts of the bark of L. coromandelica used at a higher dose (400 mg/kg) reduced AST ((138 ± 5.1) IU/L) to the maximum ((71 ± 5.1) IU/L), ALT ((71 ± 2.7) IU/L), ALP ((140 ± 1.9) IU/L), and serum levels of bilirubin, cholesterol, sugar, and LDH. | [72] | |
| L | EtOH | In vivo: antidiabetic activity in rats induced by alloxan | The ethanolic extract of L. coromandelica (100 to 200 mg/kg) reduced the glucose level (123 ± 2.2 and 115 ± 2.6, respectively) both in diabetic animals and in those induced with alloxan when compared to normal animals (74 ± 1.7 and 70 ± 1.4). | [92] | |
| R | EtOH | In vitro: antioxidant activity | The crude extract of ethyl acetate at concentrations 200; 100; 50; 25; 12.5; and 6.25 µg/mL, in 3 mL of methanolic DPPH solution. Ascorbic acid was used as a positive control. The compound isolated from the extract (citrinin) showed moderate antioxidant activity (AAI 0.671 and IC50 145.9 ppm). | [93] | |
| Wp | EtOAc | Antimicrobial activity agar diffusion method | The antimicrobial activity demonstrated that the isolated compound was not active against Escherichia coli ATCC25922, Salmonella typhi ATCC 14028, Staphylococcus aureus ATCC25923, and Pseudomonas aeruginosa ATCC 27853 (MIC: 1000 μg/mL). | [93] | |
| Lannea edulis | Wp | H2O | In vitro: mutagenicity test | The mutagenicity test was performed using Salmonella typhimurium strains TA97a, TA98, and TA100, and marginal-type displacement mutations (marginal mutagenicity) were observed in the TA97a strain. | [73] |
| L | H2O | Antidiabetic activity by alloxan induction method | Daily dosing of L. edulis resulted in significant reductions in blood glucose levels compared to those in the diabetic control from day 3; only the 300 mg/kg and 500 mg/kg L. edulis diabetic positive control groups had significant differences (p < 0.05) in mean blood glucose levels. The 100 mg/kg diabetic positive control group kg of L. edulis showed significant difference (p < 0.05) compared to diabetic control group from day 5. | [75] | |
| H2O | In vitro: cytotoxic activity | The cytotoxic effect of aqueous extracts was evaluated on U937, MeWo, and Vero cell lines tested. L. edulis at the highest tested concentration was seen to be significantly toxic (p = 0.007). L. edulis (p < 0.007) showed a similar toxic effect in the MeWo and Vero cell lines. | [94] | ||
| Wp | H2O | In vitro: anti-inflammatory activity | The anti-inflammatory potential of the extract was evaluated on RAW 264.7 cells, and there was no anti-inflammatory activity observed for the plants tested. However, in the absence of LPS stimulation, there was an increase of NO production, indicating that the extracts might have pro-inflammatory properties. | [94] | |
| Lannea humilis | B | MeOH | In vitro: antioxidant activity by DPPH and FRAP methods | DPPH = 9.3 (EC50 µg/mL); FRAP = 19.77 (mM FeSO4 equivalent/mg sample). | [53] |
| Stem bark | MeOH | In vitro: antioxidant activity by DPPH method | The antioxidant activity of plant extracts demonstrated dose-dependent behaviour. The ethyl acetate extract displayed the most noteworthy antioxidant activity of 98% at 240 µg/mL, followed by the hexane extract with antioxidant activity of 92% at 240 µg/mL. Methanol extract showed antioxidant activity of 71% at 240 µg/mL. | [95] | |
| Lannea nigritana | R | H2O | In vitro: proportional method for MIC determination | Leaf decoction showed activity on 7 M. ulcerans strains and isolates with mean MIC values of 40 μg/mL. | [63] |
| StB | EtOH | In vitro: cytotoxic activity of the ethanolic extract by the HeLa method | Extracts can be classified as being of low cytotoxicity, showing less than 40% activity at 500 µg/mL. | [96] | |
| Lannea rivae | B | DCM/MeOH | In vivo: anti-inflammatory activity by method paw oedema in Wistar rats | Extract of L. rivae roots and epicatechin gallate and (4R, 6S)-4,6-dihydroxy-6-((Z)-nonadec14’-en-1-yl)cyclohex-2-en-1 -one at 200 mg/kg using Indomethacin as the standard showed anti-inflammatory activity; both the extract and the 2 compounds moderately inhibited the oedema induced by carrageenan, however, none of them reached the level of inhibition of the Indomethacin standard. | [36] |
| R | DCM/MeOH | In vitro: antibacterial activity | The new compounds isolated (4R,6S)-4,6-dihydroxy-6-((Z)-nonadec-14′-en-1-yl)cyclohex-2-en-1-one and (2S*,4R*,5S*)-2,4,5-trihydroxy-2-((Z)-nonadec-14′-en-1-yl)cyclohexanone were tested against Staphylococcus aureus and Escherichia coli. Compound 1, taraxerol, β-sitosterol, taraxerone, and lupeol showed moderate activity against E. coli (56.64% inhibition), while only compound 2 and β-sitosterol showed activity against S. aureus (43.56%). | [36] | |
| R, St | Hx, DCM, EtOAc, MeOH | In vitro: antibacterial activity of selected compounds | The hexane extracts of L. rivae exhibited intermediate antibacterial activity against E. faecalis, while the DCM extracts showed intermediate activity against both Gram-positive bacteria E. faecalis and S. aureus, but no activity against Gram-negative bacteria. The EtOAc and MeOH extracts demonstrated a broader spectrum of activity, with better activity being observed with the Gram-positive bacteria. | [46] | |
| Lannea schimperi | Ap | EtOH | In vivo: effect of ethanolic extract on ethanol/HCl-induced gastric ulcers in rats | Doses of ethanolic extract of 100, 200, 400, and 800 mg/kg were tested in rats against gastric ulcer induced by ethanol-HCl and the effects were compared to those of pantoprazole 40 mg; after removal and analysis of the stomach, it was found that the ethanolic extract of L. schimperi showed an average protection of 81.7% compared to 87.5% for the drug pantoprazole. | [55] |
| L | MeOH | In vitro: anticoccidial activity in Eimeria tenella oocysts | This activity was carried out using oocysts isolated from infected chicks, and three doses of methanolic extract of L. schimperi leaves were used, 25 mg/mL, 50 mg/mL, and 100 mg/mL. Anticoccidial activity was determined by counting lysed and non-sporulated oocysts and sporulated oocysts. The extract dose at 100 mg/mL exhibited 98% higher anticoccidial activity and an inhibition of 97.92%. Doses 25 and 50 mg/mL of extract showed activities and inhibitions against non-sporulated oocysts of E. tenella of 68% and 89% and 66.65 and 88.5, respectively. | [37] | |
| R, St | MeOH, H2O | In vitro: cytotoxic activity colorimetric test | MTT was used to measure all growth and cellular chemosensitivity. The samples were prepared for a stock solution of 20 mg/mL in 100% DMSO, and emetine was used as a positive control. The 5-[alkenyl]-4,5-dihydroxycyclohex-2-enone mixture (1a-d) exhibited good in vitro cytotoxicity against the Chinese Hamster Ovarian mammalian cell line. | [97] | |
| MeOH | MeOH | In vivo: anti-inflammatory activity | The test was carried out using the egg albumin induction method in rats. Tested doses were 12 and 24 mg/kg, and acetylsalicylic acid 80 mg was used as standard. The anti-inflammatory response was significant (p< 0.05); however, there was no significant difference (p > 0.05) between the extract-treated groups and the standard drug-treated group (positive control). | [98] | |
| Lannea schweinfurthii | Wp | Hx, MeOH, EtOAc | In vitro: antibacterial and antifungal activity | The extracts were tested against S. aureus, Bacillus subtilis, P. aeruginosa, Escherichia coli, and Candida albicans. Measured inhibition zone showed significant differences: 7 mm hexane extract (α = 0.05); methanolic and ethyl acetate showed high activity (13 mm inhibition and above). Both extracts showed moderate activity, with inhibition between 7 and 14 mm against bacteria and fungi. | [77] |
| R | EtOAc | In vitro: ACHE inhibitory activity | The ethyl acetate extract of L. schweinfurthii showed an IC50 value higher than that of galanthamine (standard) 0.00053 mg/mL. The extract has ACHE inhibitory activity with an IC50 of 0.0030 ± 0.000 mg/mL. | [78] | |
| R | Hx | In vitro: antibacterial activity | The extract was active against Enterococcus faecalis and Enterococcus faecium with10 mm zone of inhibition. | [31] | |
| R, St | MeOH | In vitro: antibacterial activity | Active against Salmonella typhimurium, Enterococcus faecalis, Enterococcus faecium, Pseudomonas aeruginosa, and Staphylococcus aureus with zone of inhibition ranging from 8 mm to 15 mm. | [31] | |
| B | MeOH | In vitro: anti-HIV-2 activity | The methanolic extract of the stem bark of L. Schweinfurthii was active against HIV type 2, with IC50 values < 10 µg/mL and 9.9 µg/mL against HIV-1, respectively. | [99] | |
| Lannea velutina | R B | MeOH, EtOH | In vitro: DPPH radical scavenging activities and 15-LOX inhibition | The concentrations of extracts and fractions that provide 50% radical scavenging are (12 ± 2 and 17 ± 2) and 50% enzyme inhibition (14 ± 1 and 18 ± 2), respectively; scavenging activity and inhibitory effect were statistically very significant; p < 0.001. | [74] |
| R B | EtOH:H2O | In vitro: antioxidant activity DPPH method | 50% radical scavenging, at concentrations of 5–7 micrograms/mL, and 15-lipoxygenase inhibitors (50% inhibition at 10–18 micrograms/mL). L. velutina extract possessed a weak DPPH radical scavenging action. | [40] | |
| Wp | EtOH, DCM, MeOH, H2O | In vitro. Antimicrobial activity tested on mosquito larvae; molluscicidal activity with molluscs | Positive results were obtained for antioxidant activity (methanolic extracts of bark and roots), antifungal activity (dichloromethane extract active against Candida albicans and Cladosporium cucumerinum); larvicidal activity against the malarial mosquito Anopheles gambiae (dichloromethane extract of bark and methanolic extract of leaves); and molluscicidal activity directed at the snail Biomphalaria pfeifferi, transmitter of schistosiasis. The ethanol extract of the bark showed greater antibacterial activity against Bacillus subtilis, Staphylococcus aureus (Gram-positive), Pseudomonas aeruginosa, and Salmonella typhimurium (Gram-negative). | [57,100] | |
| R B, StB | EtOH, MeOH, H2O | In vitro: antioxidant activity by DPPH method | Petroleum ether, chloroform, and dichloromethane extracts are inactive as DPPH radical scavengers; the aqueous extract had moderate activity while the methanolic and hydroalcoholic extracts of root bark and stem bark were very active. | [57] | |
| B | EtOH | In vitro: antioxidant activity by DPPH method | For the test on the free radical potential on the radical DPPH, o L. velutina, which showed a percentage inhibition of 52.8125 ± 2.16% lower than that of the gallic acid, was used as reference substance. | [79] | |
| B | EtOH | In vitro: antimicrobial activity by inhibition method | Shigella dysenteria, S. aureus were sensitive to Lannea velutina extracts with inhibition diameters of 10 mm; Bacillus cereus and Escherichia coli were also sensitive to the extract with 8 mm and Salmonella thyphi with 7 millimetres. | [79] | |
| L | Hx, EtOAc, DCM, MeOH, H2O | In vitro: antioxidant activity by DPPH method | The L. velutina leaf methanol extract showed IC50 15.42 g/mL. | [80] | |
| L | Hx, EtOAc, DCM, MeOH, H2O | In vivo: acute toxicity | The acute oral toxicity test of ethyl acetate, methanol, and aqueous extracts on mice exhibit a lethal dose (LD50) estimated to be higher than 2000 mg/kg body weight. | [80] | |
| Lannea welwitschii | B | H2O | In vivo: anti-diarrhoeal activity in mice | Bark aqueous extract (50–400 mg/kg) caused a significant delay (p < 0.05) in the onset of profuse diarrhoea, decreased purging frequency, wet stool weight, and diarrhoea severity. Oral administration of castor oil produced an intestinal fluid volume of 2.33 ± 0.17 mL; Lw bark aqueous extract at 400 mg/kg significantly (p < 0.05) reduced intestinal fluid volume to 1.40 ± 0.25. | [60] |
| B | H2O | In vivo: anti-diarrhoeal activity in mice | The acute toxicity tests carried out showed a well-tolerated effect of the drug via oral route, a dose of 20 g/kg produced no death in the animals. LD50 was estimated to be 631 mg/kg. | [82] | |
| L | MeOH | In vivo: analgesic activity | In doses of 50, 200, and 400 mg/kg, L. welwitschii extract caused a significant increase (p < 0.0001) in the mean reaction time of treated mice (49.67 ± 2.18%, 63.20 ± 2.54%, and 59.42 ± 0.84%) respectively compared to the control group, while the total analgesic effect (AUC) was significant (p < 0.0001) and the dose-dependent increase was to 159.20 ± 19.65, 202.30 ± 12.44 and 228.8 ± 11.29, respectively. There was no statistical difference in the analgesia produced with 100 mg/kg aspirin. | [60] | |
| L | MeOH | In vitro: antioxidant activity by DPPH method | MeOH extract showed antioxidant activity with IC50 81.8 µg mL−1 compared to α-tocopherol 1.5 µg/mL. | [81] | |
| L | MeOH | In vitro: antimicrobial activity by agar diffusion and microdilution methods | The extract showed activity against Enterococcus faecalis, Klebsiella pneumoniae, Proteus mirabilis, Pseudomonas aeruginosa, Staphylococcus aureus, and some strains of Escherichia coli resistant to pefloxacin. The methanolic extract of L. welwitschii showed MICs of 5, 10, 5, 2.5, and 2.5 mg/mL, respectively, against E. coli, P. aeruginosa, S. aureus, and B. subtilis compared to Ciprofloxacin which was 0.025; 0.055; 0.025; 0.02 mg/mL while the MICs of methanolic leaf extract and clotrimazole against C. albicans were 2.5 and 0.025 mg/mL, respectively. | [60] | |
| StB | EtOH:H2O | In vivo: anti-inflammatory activity by method carrageenan-induced paw oedema | The L. welwitschii extract was administered at doses of 50, 200, and 400 mg/kg. The 200 mg/kg dose had an inhibition of 14.49 ± 2.43% compared to the control, while the total oedema induced over 6 h was 37.19 ± 4.38% The maximum inhibitory effects were seen with 400 mg/kg dose. | [60] | |
| Wp | DCM, MeOH | In vitro: antioxidant activity by spectrophotometric methodology | The antioxidant activity of identified Compound 4 (IC50 18.6 ± 4.5 µg/mL) and 2 (IC50 20.0 ± 0.1 µg/mL) showed better activity than the controls, ascorbic acid (IC50 23.17 ± 2.02), and quercetin (IC50 31.67 ± 2.88 µg/mL) | [42] |
Aerial part—Ap; Ace—acetone; AgNps—green silver nanoparticles; AP—aerial part; Ba—bark; Be—berries; ButOH—butanol; C6H14—petroleum ether; CFU—Per milliliter colony forming unit; Chl—chloroform; DCM—dichloromethane; DMSO—dimethyl sulfoxide; Et2O—diethyl ether; EtOAc—ethyl acetate; EtOH—ethanol; Fl—flower; Fr—fruit; H2O—water; Hx—hexane; IC50—median inhibition concentration; Iz—inhibition zone; L—leaf; MBC—minimum bactericide concentration; MeOH—methanol; MIC—minimum inhibitory concentration; NA; Na2SO4—sodium sulfate; N-Hx—N-hexane; P—pulp; R—root; Se—seed; Sf—supercritical fluid; St—stem; StB—stem bark; StO—steam distilled oil; whole plant—Wp.
3. Discussion
Our analysis found that 14 Lannea species are reportedly used in traditional medicinal systems of over 35 countries to treat a variety of disease signals and symptoms. Among these, fever, inflammation, diabetes-related symptoms, gastrointestinal disorders, and sexually transmitted diseases are the most common diseases treated with various extracts of Lannea species. Although not all Lannea species have been studied for their biological activity, those that have been showed antimicrobial, antioxidant, and anti-inflammatory properties, mainly observed in vitro. These results support the use of Lannea medicinal plants in traditional medicinal systems, as most of their applications are in the treatment of disease symptoms related to the biological activities observed in vitro.
In the genus Lannea, some characteristic Anacardiaceae compounds such as anacardic acid, as well as common natural products such as gallic acid and derivatives, flavonol derivatives such as quercetin and rutin, kaempferol, myricetin, and flavones like luteolin, have been identified [49,58,97,101].
Twelve different biological activities have been reported in vitro and/or in vivo for Lannea species, with antimicrobial, antioxidant, anti-inflammatory, and cytotoxic activities being the most common. In many cases, the observed activity was considered significant when compared to the positive controls used in the studies. Most extracts were prepared with methanol, ethanol, and water, suggesting that most extracted compounds have a relatively high polarity.
Previous research on anacardic acid showed that this natural compound can exhibit a wide variety of other biological activities. For instance, antibacterial activity was observed against bacteria species like Bacilus subtilis, Helycobacter pylori, Propionibacterium acnes, and Staphylococcus aureus. Antimicrobial activity exhibited by L. velutina ethanolic leaf extracts, in which this compound has previously been identified, thus may be related to anacardic acid [100,101].
In an in vivo mouse model of inflammation induced by carrageenan, prostaglandin E2, dextran, and histamine, the effects of pretreatment with anacardic acid (administered at doses of 10, 25, and 50 mg/kg intraperitoneally) were investigated. The study revealed that anacardic acid exhibited inhibitory effects on carrageenan-induced oedema, with a significant efficacy observed at a dose of 25 mg/kg, surpassing that of the positive control, indomethacin. Histological examination of tissue specimens from the anacardic acid-treated group indicated reduced neutrophil infiltration compared to the carrageenan-treated group. Furthermore, anacardic acid demonstrated inhibitory properties against carrageenan-induced depletion of glutathione and reduced levels of malondialdehyde, a pivotal marker of oxidative stress. Taken together, these results suggest that the anti-inflammatory effect of anacardic acid is due to its ability to inhibit inflammatory mediators, mitigate chemotaxis, and alleviate oxidative stress. In addition, the assessment of antinociceptive activity showed a reduction in pain symptoms in the anacardic acid-treated group. Mechanistic insights into this activity revealed a link to opioid receptors, as demonstrated using the nonselective opioid receptor antagonist naloxone as a control [102].
Anacardic acid also exhibited modulatory activity in gene expression, cell death, and cell proliferation; selective cytotoxicity against human cancer cell lines was also observed, indicating that this compound may be a useful focus of study for the development of new therapeutic anticancer agents [101].
Quercetin, a common flavonol abundantly present in numerous plant species, has a significant antioxidant activity and has been described to prevent diseases like osteoporosis, cancer, tumours, and lung and cardiovascular diseases. In vivo studies have shown that this antioxidant activity is mainly exerted through the effect on gluthathione reactive oxygen species, enzymatic activity (namely acetylcholinesterase), and signal transduction pathways. Quercetin has also shown to be able to prevent lipopolysaccharide (LPS)-induced heart damage by clearing oxygen-free radicals and consequently preventing myocardium damage. Its activity is also exerted in several steps of signal transduction pathways, decreasing the impact of oxidative stress. In a LPS-induced acute liver injury in vivo mouse model, quercetin inhibited NF-κB and MAPK signalling pathways and inhibited the expression of apoptosis-related proteins, which led to decreased oxidative stress and tissue damage. Antioxidant and anti-inflammatory properties have been demonstrated for L. acida and L. coromandelica, from which quercetin has previously been identified [103]. This natural product demonstrated selective in vitro antibacterial efficacy against various infectious strains of both Gram-positive and Gram-negative bacteria. Notably, Staphylococcus aureus, Staphylococcus epidermidis, and clinical strains of methicillin-resistant Staphylococcus aureus (MRSA) exhibited significant susceptibility to quercetin. Furthermore, when administered concomitantly with antibiotics such as ampicillin, erythromycin, gentamycin, oxacillin, and vancomycin, quercetin significantly potentiated the antibacterial activity of these drugs against clinical MRSA strains, implying a synergistic interaction between quercetin and antibiotics. This observed phenomenon underscores the potential of quercetin as a promising therapeutic agent for the treatment of infectious diseases [104].
In other antibacterial studies, quercetin has showed inhibitory activity on pathogenic bacteria growth, namely E. coli, P. mirabilis, Aspergillus flavus, P. aeruginosa, Salmonella enteritidis, and S. aureus. Synthetic derivatives of this compound also showed growth inhibitory activity against E. coli, S. aureus, and P. aeruginosa. The current research proposes that the antibacterial mechanism is related to cell wall destruction and cell permeability deregulation, compromising metabolic pathways crucial for bacterial survival, like protein synthesis and expression, enzyme activity, and nucleic acid synthesis. This mechanism may justify the synergistic effect observed when quercetin was administered in combination with antibiotics [105].
Myricetin is a flavonol with a wide distribution in many plants and is highly recognised for its nutritional value. Previously conducted studies on this compound showed that it can display different biological activities, such as antioxidant activity, being able to reduce oxidative stress through mechanisms like radical scavenging, decreasing production of pro-inflammatory agents, and disrupting inflammatory pathways. Similar activities have also been observed for L. welwitschii and L. rivae, where this compound was previously identified. Anticancer activity has also been reported, with myricetin exhibiting selective cytotoxic activity against human hepatic, pancreatic, skin, colon, and leukaemia cancer cell lines with clinical relevance. Research showed that myricetin can also interfere with different mechanisms related to tumour proliferation, namely modulating gene expression and inhibiting enzymes and other agents that directly promote cell division. Other studies showed that myricetin can act as an anti-platelet aggregation agent, supressing thromboxane formation and inhibiting specific receptor binding of platelet activating factor, and as an antihypertensive agent, reducing systolic blood pressure and vascular reactivity; immunomodulatory activity has been described in vivo and in vitro, with myricetin acting on stimulating antibody formation and regulating TNF-α, IL-2, IL-6, and IL-12 expression and lymphocyte proliferation [106].
Flavonoid compounds like catechins and its derivatives, found in L. alata, and terpenoid compounds like b-sitosterol, found in L. coromoandelica, have previously been studied for their biological activities. While catechins have shown antioxidant activity in in vitro essays, b-sitosterol has exhibited several in vitro biological activities like antimicrobial, anti-inflammatory, antioxidant, and antidiabetic activities [96,107].
Understanding the biological activities of plant extracts represents a significant challenge due to their complex composition, which includes a variety of natural products derived from the secondary metabolism of plants. It is often believed that the observed activities of plant extracts are associated with the presence of the most common occurring compounds or classes of compounds; however, this association often occurs based on an equilibrium between concentrations of compounds belonging to different classes. In particular, synergistic and other complex interactions may play a role, and numerous reports documented in the literature indicate that the biological activities of isolated major compounds can be inferior to those of all extracts.
Our research showed that different plant parts of Lannea species are used as medicinal plants for the preparation of traditional herbal preparations through decoction and maceration. Phytochemical studies on this genus have shown that phenolic compounds are the chemical class with higher representativity, and that Lannea species have in vitro/in vivo biological activities (antibacterial, antidiabetic, antifungal, antimicrobial, anti-inflammatory, antioxidant, antipyretic). Since these activities reported in the literature are aligned with their use in traditional medicine, we can thus consider that this use is totally or partially scientifically valid.
Given that a significant proportion of the identified secondary metabolites in Lannea species belong to the chemical class of polyphenols, it is plausible to correlate the observed biological activities with phenolic compounds in general. Nevertheless, this hypothesis requires empirical validation through specific studies aimed at a comprehensive characterization of these activities.
4. Materials and Methods
This review was performed following the criteria described in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement 2020 (https://prisma-statement.org/prismastatement/flowdiagram.aspx; accessed on 1 February 2023).
A literature search covering articles published between January 1995 and June 2023 was conducted using databases from, B-on, Google Schoolar, Prelude Medicinal Plants database, Pubmed, Web of science, and primary bibliographic sources. These bibliographic sources were searched using different key words: “Lannea”; “Ethnomedicinal”; “Chemical”; “Biological activity”, and the Boolean connectors AND/OR.
The studies that were related to plants belonging to the Lannea genus and were concerned with their medicinal importance were selected and included in this review.
5. Conclusions
Lannea species may represent an important source of natural products with relevant biological activities that can contribute to the development of new drugs. This study of this genus highlights its importance for traditional medicine in developing countries where access to primary health care is still poor. Despite this wide utilization, more multidisciplinary (taxonomic, conservational, ethnopharmacological) studies are needed to validate their concrete use as herbal medicines for the specific treatment of pathologies to which they are traditionally indicated.
Author Contributions
Q.M.: methodology, validation, investigation, writing—review and editing. G.I.C.: methodology, investigation, writing—review and editing. L.C.: methodology, validation. B.I.: methodology, validation. I.M.d.S.: investigation, writing—review. B.L.: writing—review and editing, project administration, funding acquisition. O.S.: conceptualization, methodology, validation, formal analysis, investigation, resources, writing—original draft, writing—review and editing, visualization, supervision, project administration, funding acquisition. All authors have read and agreed to the published version of the manuscript.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding Statement
This work was supported by the Foundation for Science and Technology (FCT, Portugal) through national funds FCT/MCTES to iMed.ULisboa (UIDP/04138/2020; https://doi.org/10.54499/UIDP/04138/2020 accessed on 25 December 2023) and cE3c (UIDB/00329/2020; https://doi.org/10.54499/UIDB/00329/2020 accessed on 25 December 2023).
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.WHO. 2023. [(accessed on 27 February 2023)]. Available online: http://www.worldfloraonline.org/taxon/wfo-4000020561.
- 2.Santos C.C., Borba E.L., Queiroz L.P. A família Anacardiaceae no semi-árido do Estado da Bahia, Brasil. Sitientibus Série Ciências Biológicas. 2008;8:189–219. doi: 10.13102/scb8091. [DOI] [Google Scholar]
- 3.Jahurul M.H., Zaidul I.S., Ghafoor K., Al-Juhaimi F.Y., Nyam K.L., Norulaini N.A., Sahena F., Omar A.M. Mango (Mangifera indica L.) by-products and their valuable components: A review. Food Chem. 2015;183:173–180. doi: 10.1016/j.foodchem.2015.03.046. [DOI] [PubMed] [Google Scholar]
- 4.Catarino L., Indjai B. Rvores Florestais da Guiné-Bissau. IBAP; Bissau, Guinea-Bissau: 2019. [Google Scholar]
- 5.Efloras Lannea in Flore d’Afrique Centrale. [(accessed on 27 February 2023)]. Available online: https://www.floredafriquecentrale.be.
- 6.Efloras Lannea in Flora of China. [(accessed on 27 February 2023)]. Available online: http://www.efloras.org/flora_page.aspx?flora_id=2.
- 7.Kew R.B.G. Plants of the World Online. [(accessed on 27 February 2023)]. Available online: https://powo.science.kew.org/taxon/urn:isid:ipni.org:names:331697-2.
- 8.Cevallos-Ferriz S. Leaf architecture of Anacardiaceae. Rev. Mex. Biodivers. 2005;76:137–190. [Google Scholar]
- 9.Martinez-Millán M., Cevallos-Ferriz S. Arquitectura foliar de Anacardiaceae. Rev. Mex. Biodiv. 2005;76:137–190. [Google Scholar]
- 10.AbdulRahaman A.A., Kolawole O.S., Oladele F.A. Leaf epidermal features as taxonomic characters in some Lannea spieces (Anacardiaceae) from Nigeria. Phytol. Balc. 2014;20:227–231. [Google Scholar]
- 11.Diniz M.A., Martins E.S., Gomes E., Silva O. Contribuição para o conhecimento de plantas medicinais da Guiné-Bissau. Port. Acta Biol. 2000;19:417–427. [Google Scholar]
- 12.Etuk E.U., Ugwah M.O., Ajagbonna O.P., Onyeyili P.A. Ethnobotanical survey and preliminary evaluation of medicinal plants with antidiarrhoea properties in sokoto state, nigeria. J. Med. Plant. Res. 2009;3:763–766. [Google Scholar]
- 13.Zerbo P., Millogo-Rasolodimby J., Nacoulma-Ouedraogo O.G., Van Damme P. Plantes médicinales et pratiques médicales au Burkina Faso: Cas des Sanan. Bois Forets Trop. 2011;307:41–53. doi: 10.19182/bft2011.307.a20481. [DOI] [Google Scholar]
- 14.Online W.F. World Flora Online. [(accessed on 27 February 2023)]. Available online: https://wfoplantlist.org/taxon/wfo-4000020561-2022-12?page=1.
- 15.Figueiredo E., Smith G. Common Names of Angolan Plants. Protea Book House; Pretoria, South Africa: 2017. p. 399. [Google Scholar]
- 16.Urso V., Signorini M.A., Tonini M., Bruschi P. Wild medicinal and food plants used by communities living in MOPANE woodlands of southern angola: Results of an ethnobotanical field investigation. J. Ethnopharmacol. 2016;177:126–139. doi: 10.1016/j.jep.2015.11.041. [DOI] [PubMed] [Google Scholar]
- 17.Salihu Shinkafi T., Bello L., Wara Hassan S., Ali S. An ethnobotanical survey of antidiabetic plants used by Hausa-Fulani tribes in Sokoto, Northwest Nigeria. J. Ethnopharmacol. 2015;172:91–99. doi: 10.1016/j.jep.2015.06.014. [DOI] [PubMed] [Google Scholar]
- 18.Rahman L., Hossain M.K. Distribution pattern of medicinal tree species in Chunati wildlife sanctuary of Chittagong. J. Trop. Med. Plants. 2002;3:65–72. [Google Scholar]
- 19.Fern K. Tropical Plants Database. [(accessed on 27 February 2023)]. Available online: https://tropical.theferns.info/
- 20.Durand R.P.J. d’Astrida G.S., editor. Les Plantes Bienfaisantes du Ruanda et de l’Urundi. 1960. [(accessed on 27 February 2023)]. Available online: http://www.ethnopharmacologia.org/recherche-dans-prelude/?plant_id=551.
- 21.Wamucii S. Lannea Gossweileri–Uses, Benefits & Care. [(accessed on 27 February 2023)]. Available online: https://www.selinawamucii.com/plants/anacardiaceae/lannea-gossweileri/#common-names.
- 22.Grade J.T., Tabuti J.R., Van Damme P. Ethnoveterinary knowledge in pastoral Karamoja, Uganda. J. Ethnopharmacol. 2009;122:273–293. doi: 10.1016/j.jep.2009.01.005. [DOI] [PubMed] [Google Scholar]
- 23.Carrière M. Plantes de Guinée à L’usage des Éleveurs et des Vétérinaires. CIRAD-EMVT; Montpellier, France: 2000. [Google Scholar]
- 24.Ribeiro A., Romeiras M.M., Tavares J., Faria M.T. Ethnobotanical survey in Canhane village, district of Massingir, Mozambique: Medicinal plants and traditional knowledge. J. Ethnobiol. Ethnomed. 2010;6:33. doi: 10.1186/1746-4269-6-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bruschi P., Morganti M., Mancini M., Signorini M.A. Traditional healers and laypeople: A qualitative and quantitative approach to local knowledge on medicinal plants in Muda (Mozambique) J. Ethnopharmacol. 2011;138:543–563. doi: 10.1016/j.jep.2011.09.055. [DOI] [PubMed] [Google Scholar]
- 26.Kerharo J., Adam J.G. Plantes médicinales et toxiques des peul et des toucouleur du sénégal. J. Agric. Trop. Bot. Appl. 1964;11:384–444. [Google Scholar]
- 27.Lautenschlager T., Monizi M., Pedro M., Mandombe J.L., Branquima M.F., Heinze C., Neinhuis C. First large-scale ethnobotanical survey in the province of Uige, northern Angola. J. Ethnobiol. Ethnomed. 2018;14:51. doi: 10.1186/s13002-018-0238-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sulaiman A.N., Arzai A.H., Taura D.W. Ethnobotanical survey: A comprehensive review of medicinal plants used in treatment of gastro intestinal diseases in Kano state, Nigeria. Phytomed. Plus. 2022;2:100180. doi: 10.1016/j.phyplu.2021.100180. [DOI] [Google Scholar]
- 29.Mangla B., Kohli K., Rabiu S. Review of medicinal uses, phytochemistry, pharmacological properties, extraction methods and toxicology of Lannea microcarpa (African grapes) Curr. Tradit. Med. 2021;7:125–137. doi: 10.2174/2215083805666190626095609. [DOI] [Google Scholar]
- 30.Maroyi A. Review of Ethnomedicinal, Phytochemical and Pharmacological Properties of Lannea schweinfurthii (Engl.) Engl. Molecules. 2019;24:732. doi: 10.3390/molecules24040732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bossard E. La Medecine Traditionnelle au Centre et a l’ouest de l’Angola. Instituto de Investigação Científica Tropical; Lisboa, Portugal: 1996. [Google Scholar]
- 32.Adam J.G., Echard N., Lescot M. Plantes médicinales Hausa de l’Ader (République du Niger) J. Agric. Trop. Bot. Appl. 1972;19:259–399. doi: 10.3406/jatba.1972.3119. [DOI] [Google Scholar]
- 33.Adomou A.C., Yedomonhan H., Djossa B., Legba S.I., Oumorou M., Akoegninou A. Etude Ethnobotanique des plantes médicinales vendues dans le marché d’Abomey-Calavi au Bénin. Int. J. Biol. Chem. Sci. 2012;6:745–772. doi: 10.4314/ijbcs.v6i2.18. [DOI] [Google Scholar]
- 34.Bossard E. Quelques Notes sur l’ Alimentation et les Apports Nutritionnels Occultes en Angola Garcia de Orta. Sér. Bot. 1996;13:7–41. [Google Scholar]
- 35.Yaouba S., Koch A., Guantai E.M., Derese S., Irungu B., Heydenreich M., Yenesew A. Alkenyl cyclohexanone derivatives from Lannea rivae and Lannea schweinfurthii. Phytochem. Lett. 2018;23:141–148. doi: 10.1016/j.phytol.2017.12.001. [DOI] [Google Scholar]
- 36.Mikail H., Yusuf M., Hussain G. In vitro anticoccidial activity of methanolic leaves extract of Lannea schimperi against oocysts of Eimeria tenella. J. Pharm. Biol. Sci. 2016;11:35–38. [Google Scholar]
- 37.Gathirwa J.W., Rukunga G.M., Mwitari P.G., Mwikwabe N.M., Kimani C.W., Muthaura C.N., Kiboi D.M., Nyangacha R.M., Omar S.A. Traditional herbal antimalarial therapy in kilifi district, kenya. J. Ethnopharmacol. 2011;134:434–442. doi: 10.1016/j.jep.2010.12.043. [DOI] [PubMed] [Google Scholar]
- 38.Ngbolua K., Bongo G., Ashande M. Ethno-botanical survey and ecological study of plants resources used in Folk medicine to treat symptoms of Tuberculosis in Kinshasa City, Democratic Republic of the Congo. J. Modern Drug Discov. Drug Deliv. Res. 2014;1:2348–3776. [Google Scholar]
- 39.Ouattara L., Koudou J., Zongo C., Barro N., Savadogo A., Bassole I.H.N., Ouattara A.S., Traore A.S. Antioxidant and antibacterial activities of three species of Lannea from Burkina Faso. J. Appl. Sci. 2011;11:157–162. doi: 10.3923/jas.2011.157.162. [DOI] [Google Scholar]
- 40.Muhaisen H.M.H. Chemical constituents from the bark of Lannea acida (Anacardiaceae) Pharma Chem. 2013;5:88–96. [Google Scholar]
- 41.Kouamé J.M., Yao-Kouassi A.P., Alabdul M.A., Voutquenne-Nazabadioko L. A new sulfonyl glucoside eugenol and antioxidant phenolic compounds from Lannea acida and Lannea welwitschii. J. Pharm. Biol. Sci. 2022;9:31–36. [Google Scholar]
- 42.Olusegun O.A., Isaiah O.O., Samson A.O., Olufunmi O.A. Characterization and evaluations for antioxidant properties of flavonoids from Lannea acida extract. Medrech. 2020;7:287–296. [Google Scholar]
- 43.Saraux N., Bruna L., Ebrahimi S.N., Karimou S., Christen P., Cuendet M. Antiproliferative activity of compounds isolated from the root bark of Lannea acida in multiple myeloma cell lines. Phytochemistry. 2023;209:113641. doi: 10.1016/j.phytochem.2023.113641. [DOI] [PubMed] [Google Scholar]
- 44.Kashyap R., Pachwarya R.B., Hidaya E.N., Meena P.K., Sharma R. Constitution and synthetic study of a flavanone Lannea acida pigment A. Heterocycl. Lett. 2022;12:321–328. [Google Scholar]
- 45.Okoth A.D. Doctoral Dissertation. University of KwaZulu-Natal; Durban, South Africa: 2014. Phytochemistry and Bioactive Natural Products from Lannea alata, Lannea rivae, Lannea schimperi and Lannea schweinfurthii (Anacardiaceae) [Google Scholar]
- 46.Okoth D.A., Chenia H.Y., Koorbanally N.A. Antibacterial and antioxidant activities of flavonoids from Lannea alata (Engl.) Engl. (Anacardiaceae) Phytochem. Lett. 2013;6:476–481. doi: 10.1016/j.phytol.2013.06.003. [DOI] [Google Scholar]
- 47.Mbaoji F.N., Nweze J.A. Antioxidant and hepatoprotective potentials of active fractions of Lannea barteri Oliv. (Anarcadiaceae) in rats. Heliyon. 2020;6:e04099. doi: 10.1016/j.heliyon.2020.e04099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yun X.-J., Shu H.-M., Chen G.-Y., Ji M.-H., Ding J.-Y. Chemical constituents from barks of Lannea coromandelica. Chin. Herb. Med. 2014;6:65–69. doi: 10.1016/S1674-6384(14)60009-5. [DOI] [Google Scholar]
- 49.Islam T., Tahara S. Dihydrofavonols from Lannea coromandelica. Phytochemistry. 2000;54:901–907. doi: 10.1016/S0031-9422(00)00048-0. [DOI] [PubMed] [Google Scholar]
- 50.Anderson D.M.W., Hendrie A. The structure of Lannea coromandelica gum. Carbohydr. Res. 1973;26:105–115. doi: 10.1016/S0008-6215(00)85027-2. [DOI] [Google Scholar]
- 51.Queiroz E.F., Kuhl C., Terreaux C., Mavi S., Hostettmann K. New dihydroalkylhexenones from Lannea edulis. J. Nat. Prod. 2003;66:578–580. doi: 10.1021/np0202428. [DOI] [PubMed] [Google Scholar]
- 52.Sobeh M., Mahmoud M.F., Hasan R.A., Abdelfattah M.A.O., Sabry O.M., Ghareeb M.A., El-Shazly A.M., Wink M. Tannin-rich extracts from Lannea stuhlmannii and Lannea humilis (anacardiaceae) exhibit hepatoprotective activities in vivo via enhancement of the anti-apoptotic protein bcl-2. Sci. Rep. 2018;8:9343. doi: 10.1038/s41598-018-27452-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Okoth D.A., Akala H.M., Johnson J.D., Koorbanally N.A. Alkyl phenols, alkenyl cyclohexenones and other phytochemical constituents from Lannea rivae (chiov) Sacleux (Anacardiaceae) and their bioactivity. Med. Chem. Res. 2016;25:690–703. doi: 10.1007/s00044-016-1521-2. [DOI] [Google Scholar]
- 54.Haule E.E., Moshi M.J., Nondo R.S., Mwangomo D.T., Mahunnah L. A study of antimicrobial activity, acute toxicity and cytoprotective effect of a polyherbal extract in a rat ethanol-HCl gastric ulcer model. BMC Res. Notes. 2012;5:546. doi: 10.1186/1756-0500-5-546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Nguemo R.T., Mbouangouere R., Bitchagno G.T.M., Tchuenguem R., Temgoua E.V.N., Ndontsa B.L., Mpetga J.S., Opatz T., Ngouela A.S., Tane P. A new ceramide from the leaves of Lannea schimperi (hochst. Ex a.Rich.) engl. Nat. Prod. Res. 2022;36:515–522. doi: 10.1080/14786419.2020.1789632. [DOI] [PubMed] [Google Scholar]
- 56.Malterud K.E. Ethnopharmacology, chemistry and biological properties of four malian medicinal plants. Plants. 2017;6:11. doi: 10.3390/plants6010011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Malu Q., Lima K., Malmir M., Pinto R., da Silva I.M., Catarino L., Duarte M.P., Serrano R., Rocha J., Lima B.S., et al. Contribution to the preclinical safety assessment of Lannea velutina and sorindeia juglandifolia leaves. Plants. 2022;12:130. doi: 10.3390/plants12010130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Bouare S., Traore N., Sidibe L., Fofana B., Chalard P., Figueredo G., Chalchat J.C. Composition chimique de l’huile essentielle des fleurs de Lannea velutina (Anacardiaceae) du Mali. Int. J. Biol. Chem. Sci. 2013;6:2274–2279. [Google Scholar]
- 59.Osafo N., Boakye Y.D. A review: Ethnomedicinal, phytochemical and pharmacological investigations of Lannea welwitschii (Hiern) Engl. J. Adv. Med. Pharm. Sci. 2017;11:1–10. doi: 10.9734/JAMPS/2016/30431. [DOI] [Google Scholar]
- 60.Owusu G., Ofori-Amoah J. Anti-inflammatory and analgesic effects of an aqueous extract of Lannea acida Stem Bark. Br. J. Pharm. Res. 2017;16:1–8. doi: 10.9734/BJPR/2017/33266. [DOI] [Google Scholar]
- 61.Ouattara L., Koudou J., Karou D.S., Giacò L., Capelli G., Simpore J., Fraziano M., Colizzi V., Traore A.S. In vitro Anti Mycobacterium tuberculosis H37Rv activity of Lannea acida A. Rich. Freom burkina faso. Pak. J. Biol. Sci. 2011;14:47–52. doi: 10.3923/pjbs.2011.47.52. [DOI] [PubMed] [Google Scholar]
- 62.Tsouh Fokou P.V., Nyarko A.K., Appiah-Opong R., Tchokouaha Yamthe L.R., Ofosuhene M., Boyom F.F. Update on medicinal plants with potency on Mycobacterium ulcerans. BioMed Res. Int. 2015;2015:917086. doi: 10.1155/2015/917086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kone W.M., Atindehou K.K., Terreaux C., Hostettmann K., Traore D., Dosso M. Traditional medicine in north Cote-d’Ivoire: Screening of 50 medicinal plants for antibacterial activity. J. Ethnopharmacol. 2004;93:43–49. doi: 10.1016/j.jep.2004.03.006. [DOI] [PubMed] [Google Scholar]
- 64.Koné M.W., Dramane S., Dro B., Yao K. Chemical composition, antioxidant, antimicrobial and acetylcholinesterase inhibitory properties of Lannea barteri (Anacardiaceae) Aust. J. Basic Appl. Sci. 2011;5:1516–1523. [Google Scholar]
- 65.Adegoke S.A., Agada F.D., Ogundipe L.O. Antibacterial activity of methanol and ethanol leaf extracts of Antidesma venosum and Lannea barteri. Afr. J. Microbiol. Res. 2013;7:3442–3447. [Google Scholar]
- 66.Njinga N.S., Sule M.I., Pateh U.U., Hassan H.S., Ahmad M.M., Abdullahi S.T., Danja B.A., Bawa B. Phytochemical and antimicrobial activity of the leaves of Lannea kerstingii engl & krause (anacardiaceae) Nitte Univ. J. Health Sci. 2014;4:4–9. [Google Scholar]
- 67.Reddy A.K., Joy J.M., Kumara C.K.A. Lannea coromandelica: The researcher’s tree. J. Pharm. Res. 2011;4:577–579. [Google Scholar]
- 68.Majumder R., Alam B. Antidiarrheal activity of Lannea coromandelica Linn. Bark Extract. Am. Eurasian J. Sci. Res. 2013;8:128–134. [Google Scholar]
- 69.Kaur R., Jaiswal M.L., Jain V. Protective effect of Lannea coromandelica Houtt. Merrill. against three common pathogens. J. Ayurveda. Integr. Med. 2013;4:224–228. doi: 10.4103/0975-9476.123706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Syamsurya S., Ahmad A., Firdau F. Potential of methanol extracts the stem bark Lannea coromandelica (Houtt.) merr. against Staphylococcus aureus and analysis of the main secondary metabolites. Indo. J. Chem. Res. 2016;4:362–366. [Google Scholar]
- 71.Rao V.S., Einstein J.W., Das K. Hepatoprotective and antioxidant activity of Lannea coromandelica Linn. On thioacetamide induced hepatotoxicity in rats. Int. Lett. Nat. Sci. 2014;3:30–43. doi: 10.56431/p-ha9scc. [DOI] [Google Scholar]
- 72.Sohni Y.R., Davis C.L., Deschamps A.B., Kale P.G. Frameshift mutations in salmonella induced by the extracts of medicinal herbs Lannea edulis (Sond.) Engl. And monotes glaber sprague. Environ. Mol. Mutagen. 1995;25:77–82. doi: 10.1002/em.2850250111. [DOI] [PubMed] [Google Scholar]
- 73.Maiga A., Malterud K.E., Diallo D., Paulsen B.S. Antioxidant and 15-lipoxygenase inhibitory activities of the Malian medicinal plants Diospyros abyssinica (Hiern) F. White (Ebenaceae), Lannea velutina A. Rich (anacardiaceae) and Crossopteryx febrifuga (afzel) Benth. (Rubiaceae) J. Ethnopharmacol. 2006;104:132–137. doi: 10.1016/j.jep.2005.08.063. [DOI] [PubMed] [Google Scholar]
- 74.Banda M., Nyirenda J., Muzandu K., Sijumbila G., Mudenda S. Antihyperglycemic and Antihyperlipidemic effects of aqueous extracts of Lannea edulis in Alloxan-induced diabetic rats. Front. Pharmacol. 2018;9:1099. doi: 10.3389/fphar.2018.01099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Mohammed B.R., Mikail H. Effects of methanolic leaf extract of Lannea schimperi on some organs histopathology in experimentally induced coccidiosis in broiler chickens. Int. J. Vet. Sci. Anim. Husb. 2019;6:602. [Google Scholar]
- 76.Wamuyu K.R., Machocho A.K., Wafula A.W. Antimicrobial and phytochemical screening of Lannea schweinfurthii (Engl.) Engl. Asian J. Trop. Biotechnol. 2020;17:1–13. [Google Scholar]
- 77.Adewusi E.A., Steenkamp V. In vitro screening for acetylcholinesterase inhibition and antioxidant activity of medicinal plants from southern Africa. Asian Pac. J. Trop. Med. 2011;4:829–835. doi: 10.1016/S1995-7645(11)60203-4. [DOI] [PubMed] [Google Scholar]
- 78.Hilou A., Nikiema M., Guenne S., N’do J.Y.-p., Pare D. Phytochemical study and biological activities of two medicinal plants used in burkina faso: Lannea velutina a. Rich (Anacardiaceae) and Ximenia americana L. (Olacaceae) Asian J. Chem. Sci. 2019;6:1–9. [Google Scholar]
- 79.Kabore B., Koala M., Belemnaba L., Nitiema M. High-performance thin-layer chromatography phytochemical profiling, antioxidant activities, and acute toxicity of leaves extracts of Lannea velutina a. Rich. J. Med. Chem. Sci. 2023;6:410–423. [Google Scholar]
- 80.Agyare C., Bempah S.B., Boakye Y.D., Ayande P.G., Adarkwa-Yiadom M., Mensah K.B. Evaluation of antimicrobial and wound healing potential of Justicia flava and Lannea welwitschii. J. Evid. Based Complement. Altern. Med. 2013;2013:632927. doi: 10.1155/2013/632927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Olatokunboh A.O., Mofomosara S.H., Ekene O.A. Evaluation of the antidiarrhoeal effect of Lannea welwitschii Hiern (Anacardiaceae) bark extract. Afr. J. Pharm. Pharmacol. 2010;4:165–169. [Google Scholar]
- 82.Tetsatsi A.C.M., Nkeng-Effouet P.A., Alumeti D.M., Bonsou G.R.F., Kamanyi A., Watcho P. Colibri(r) insecticide induces male reproductive toxicity: Alleviating effects of Lannea acida (Anacardiaceae) in rats. Basic Clin. Androl. 2019;29:16. doi: 10.1186/s12610-019-0096-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Oumarou M.R., Zingue S., Bakam B.Y., Ateba S.B., Foyet S.H., Mbakop F.T.T., Njamen D. Lannea acida a. Rich. (Anacardiaceae) ethanol extract exhibits estrogenic effects and prevents bone loss in an ovariectomized rat model of osteoporosis. J. Evid. Based Complement. Altern. Med. 2017;2017:7829059. doi: 10.1155/2017/7829059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Oladipo A.D., Edet S.E., Akala A.O., Bolaji O.W., Ihediuche C.I., Olatunji O.A. Phytochemical screening and antimicrobial activities of Lannea acida (A. Rich) stem bark extract. J. Adv. Biol. Biotechnol. 2020;23:21–26. [Google Scholar]
- 85.Mbaoji F.N., Behnisch-Cornwell S., Ezike A.C., Nworu C.S., Bednarski P.J. Pharmacological evaluation of the anticancer activity of extracts and fractions of Lannea barteri Oliv. (Anacardiaceae) on adherent human cancer cell lines. Molecules. 2020;25:849. doi: 10.3390/molecules25040849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kumar T., Jain V. Appraisal of total phenol, flavonoid contents, and antioxidant potential of folkloric Lannea coromandelica using in vitro and in vivo assays. Scientifica. 2015;2015:203679. doi: 10.1155/2015/203679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Singh S., Singh G.B. Hypotensive activity of Lannea coromandelica bark extract. Phytother. Res. 1996;10:429–430. doi: 10.1002/(SICI)1099-1573(199608)10:5<429::AID-PTR862>3.0.CO;2-2. [DOI] [Google Scholar]
- 88.Baisya O. Preclinical evaluation of hydro-alcoholic extract of Lannea coromandelica leaves for anti-ulcer activity on albino wistar rat. Int. J. Biol. Pharm. Allied Sci. 2022;11:5759–5768. [Google Scholar]
- 89.Islam F., Mitra S., Nafady M.H., Rahman M.T., Tirth V., Akter A., Emran T.B., Mohamed A.A., Algahtani A., El-Kholy S.S. Neuropharmacological and antidiabetic potential of Lannea coromandelica (Houtt.) merr. Leaves extract: An experimental analysis. J. Evid. Based Complement. Altern. Med. 2022;2022:6144733. doi: 10.1155/2022/6144733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Priya N.S., Aruna M.S., Tony D.E., NAdendla R.R. Pharmacological evaluation of extract of Lannea coromandelica (Linn) for its antiulcer activity in rodents. Sch. Acad. J. Pharm. 2015;4:217–221. [Google Scholar]
- 91.Allenki V., Vasantha G. Antidiabetic activity of Lannea coromandelica Houtt. leaves in alloxan induced diabetic rats. Int. J. Pharm. Biol. Sci. 2014;4:108–114. [Google Scholar]
- 92.Amelia P., Ivada P., Fitriana N., Komala I., Bahri S., Hanafi M. Antioxidant and antimicrobial activity of secondary metabolites produced by endophytic fungi isolated from Lannea coromandelica (Houtt.) merr. Int. J. Pharm. Sci. Res. 2021;12:1588–1592. [Google Scholar]
- 93.Sigidi M.T., Anokwuru C.P., Zininga T., Tshisikhawe, Shonai A., Ramaite I.D.I., Traoré A.N., Potgieter N. Comparative in vitro cytotoxic, anti-inflammatory and anti-microbiological activities of two indigenous venda medicinal plants. Transl. Med. Commun. 2016;1:9. doi: 10.1186/s41231-016-0009-x. [DOI] [Google Scholar]
- 94.Achika J. Chemical investigation and antioxidant activity of fractions of Lannea humilis (Oliv.) Engl. J. Turk. Chem. Soc. 2017;4:563–572. doi: 10.18596/jotcsa.288249. [DOI] [Google Scholar]
- 95.Sowemimo A., Van de Venter M., Baatjies L., Koekemoer T. Cytotoxic activity of selected Nigerian plants. Afr. J. Tradit. Complement. Altern. Med. 2009;6:526–528. doi: 10.4314/ajtcam.v6i4.57186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Okoth D.A., Koorbanally N.A. Cardanols, long chain cyclohexenones and cyclohexenols from Lannea schimperi (Anacardiaceae) Nat. Prod. Commun. 2014;10:103–106. doi: 10.1177/1934578X1501000126. [DOI] [PubMed] [Google Scholar]
- 97.Egbe E.O., Akumka D.D., Adamu M., Mikail H. Phytochemistry, antinociceptive and anti-inflammatory actvities of methanolic leaves extract of Lannea schimperi (hoschst. Ex rich) eng. Recent Pat. Biotechnol. 2016;9:145–152. doi: 10.2174/187220830902160308195034. [DOI] [PubMed] [Google Scholar]
- 98.Maregesi S., Van Miert S., Pannecouque C., Feiz Haddad M.H., Hermans N., Wright C.W., Vlietinck A.J., Apers S., Pieters L. Screening of tanzanian medicinal plants against plasmodium falciparum and human immunodeficiency virus. Planta Med. 2010;76:195–201. doi: 10.1055/s-0029-1186024. [DOI] [PubMed] [Google Scholar]
- 99.Diallo D., Marston A., Terreaux C., Touré Y., Paulsen B.S., Hostettmann K. Screening of Malian medicinal plants for antifungal, larvicidal, molluscicidal, antioxidant and radical scavenging activities. Phytother. Res. 2001;15:401–406. doi: 10.1002/ptr.738. [DOI] [PubMed] [Google Scholar]
- 100.Hemshekhar M., Sebastin Santhosh M., Kemparaju K., Girish K.S. Emerging roles of anacardic acid and its derivatives: A pharmacological overview. Basic Clin. Pharmacol. Toxicol. 2012;110:122–132. doi: 10.1111/j.1742-7843.2011.00833.x. [DOI] [PubMed] [Google Scholar]
- 101.Gomes Junior A.L., Islam M.T., Nicolau L.A.D., de Souza L.K.M., Araujo T.S.L., Lopes de Oliveira G.A., de Melo Nogueira K., da Silva Lopes L., Medeiros J.R., Mubarak M.S., et al. Anti-inflammatory, antinociceptive, and antioxidant properties of anacardic acid in experimental models. ACS Omega. 2020;5:19506–19515. doi: 10.1021/acsomega.0c01775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Xu D., Hu M.J., Wang Y.Q., Cui Y.L. Antioxidant activities of quercetin and its complexes for medicinal application. Molecules. 2019;24:1123. doi: 10.3390/molecules24061123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Hirai I., Okuno M., Katsuma R., Arita N., Tachibana M., Yamamoto Y. Characterisation of anti-staphylococcus aureus activity of quercetin. Int. J. Food Sci. Technol. 2010;45:1250–1254. doi: 10.1111/j.1365-2621.2010.02267.x. [DOI] [Google Scholar]
- 104.Yang D., Wang T., Long M., Li P. Quercetin: Its main pharmacological activity and potential application in clinical medicine. Oxid. Med. Cell. Longev. 2020;2020:8825387. doi: 10.1155/2020/8825387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Semwal D.K., Semwal R.B., Combrinck S., Viljoen A. Myricetin: A dietary molecule with diverse biological activities. Nutrients. 2016;8:90. doi: 10.3390/nu8020090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Khan Z., Nath N., Rauf A., Emran T.B., Mitra S., Islam F., Chandran D., Barua J., Khandaker M.U., Idris A.M., et al. Multifunctional roles and pharmacological potential of beta-sitosterol: Emerging evidence toward clinical applications. Chem. Biol. Interact. 2022;365:110117. doi: 10.1016/j.cbi.2022.110117. [DOI] [PubMed] [Google Scholar]
- 107.Gallo M., Sarachine M. Biological activities of Lupeol. Int. J. Biomed. Pharm. 2009;3:46–66. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.