Abstract
Healthcare is presently experiencing a global workforce crisis, marked by the inability of hospitals to retain qualified healthcare workers. Indeed, poor working conditions and staff shortages have contributed to structural collapse and placed a heavy toll on healthcare workers’ (HCWs) well-being, with many suffering from stress, exhaustion, demoralization, and burnout. An additional factor driving qualified HCWs away is the repeated experience of moral distress, or the inability to act according to internally held moral values and perceived ethical obligations due to internal and external constraints. Despite general awareness of this crisis, we currently lack an organized understanding of how stress leads to poor health, wellbeing, and performance in healthcare workers. To address this critical issue, we first review the literature on moral distress, stress, and health in HCWs. Second, we summarize the biobehavioral pathways linking occupational and interpersonal stressors to health in this population, focusing on neuroendocrine, immune, genetic, and epigenetic processes. Third, we propose a novel Psychoneuroimmunological Model of Moral Distress and Health in HCWs based on this literature. Finally, we discuss evidence-based individual- and system-level interventions for preventing stress and promoting resilience at work. Throughout this review, we underscore that stress levels in HCWs are a major public health concern, and that a combination of system-level and individual-level interventions are necessary to address preventable health care harm and foster resilience in this population, including new health policies, mental health initiatives, and additional translational research.
Keywords: Moral distress, Occupational stress, Frontline healthcare workers, Mental and physical health, Psychoneuroimmunology, Genetics, Epigenetics, Resilience
Highlights
-
•
Moral distress is an occupational hazard in acute hospital care that can lead to long-lasting mental and physical health problems in healthcare workers (HCWs), workforce attrition, and an increase in patient safety incidents.
-
•
Despite the link between moral distress and impaired health, the immunological impact of moral distress remains poorly investigated.
-
•
This review, explores neuroendocrine, immune, genetic, and epigenetic mechanisms underlying occupational stressors in HCWs. Based on this literature, we propose a novel Psychoneuroimmunological Model of Moral Distress and Health in HCWs.
-
•
We suggest system-level and individual-level interventions to guide collective efforts in addressing preventable healthcare harm and fostering resilience in HCWs, including new health policies, mental health initiatives, and additional translational research.
1. Introduction
Hospitals across the world are currently facing a critical dearth of healthcare workers (HCWs) [1,2]. Such scarcity is especially severe among nurses [3]. The consequences, in turn, are devastating, as many hospitals have had to close units, cancel appointments, and divert patients due to inadequate numbers of professional medical personnel [4]. A unique and unprecedented event, the COVID-19 pandemic has additionally strained the already exhausted healthcare systems and placed a heavy toll on HCWs' well-being. Poor working conditions and staff shortages have contributed to structural collapse, with HCWs suffering from exhaustion, demoralization, burnout, and moral distress [[5], [6], [7], [8], [9]]. These experiences, and the consequent mental health problems, have reduced HCWs’ capacity to provide high-quality patient-centered care, resulting in higher rates of medical error and hospital-acquired infections, reduced patient satisfaction, increased job dissatisfaction, and greater attrition at work [[10], [11], [12], [13]]. Consequently, a vicious cycle has emerged in which stressful working environments, including understaffing, limited resources, risky working conditions, impaired ability to provide high quality patient care, and job dissatisfaction are both a cause and a consequence of poor mental and physical health in HCWs and employee attrition [14,15]. The healthcare crisis shows no signs of abating, with the World Health Organization (WHO) predicting a shortage of 15 million HCWs worldwide by 2030 [16]. Indeed, in October 2023, in the United States, a record-breaking strike took place at Kaiser Permanent in Modesto, California, involving 75,000 HCWs, marking the largest healthcare strike in U.S. history [17].
Working in the healthcare industry means working in high-stress, high-risk environments. Work demands and responsibilities in the healthcare sector are complex and have become increasingly challenging with the implementation of advanced medical technologies and rising prevalence of chronic diseases in the aging population [18]. Compared to other occupations, the medical professions include work stressors that are linked to sometimes grueling, often stressful environments that include repeated exposure to suffering, pain and death; significant responsibility for patient outcomes; unpredictable workflow, long and irregular working hours; exposure to personal health risks; a fast work pace; and the need for rapid adaptations to changing standards of care, and administrative and teaching duties [19]. Further complexities of healthcare work include navigating the permeable boundaries between work and non-work and family responsibilities, frequently resulting in work-life conflicts [20]. Finally, HCWs are vulnerable because they typically prioritize patients’ needs above their own [21,22].
Another significant factor driving qualified HCWs away from the bedside is the recurrent experience of moral distress, defined as an HCW's inability to act according to internally held moral values and perceived ethical obligations due to a combination of internal and external constraints [23]. Internal or personal constraints encompasses factors like self-doubt, fear of job loss, perceived lack of competence, the futility of past actions, or being socialized to follow orders. On the other hand, external constraints involve institutional policies and practices such as inadequate staffing ratios, overwhelming workloads, a poor working culture, political pressure, economization, or austerity measures [24,25]. Studies addressing moral distress in HCWs during the COVID-19 pandemic revealed that approximately 60 % – 80 % reported experiencing situations that caused moral distress [[26], [27], [28], [29]]. These experiences appear to be one of the most influential forces driving qualified HCWs away at present [1,9,30]. Ironically, this pattern of attrition also worsens resource scarcity, contributing to an increased likelihood of repeatedly morally distressing situations for those who remain at the bedside.
Despite substantial work examining the negative effects of stress on health, we currently lack an organized understanding of how stress leads to poor health and functioning in HCWs [31]. In this review, therefore, we apply the Job Demands-Resources (JD-R) health model [32], combining its core concept with a psychoneuroimmunological approach to elucidate neuroendocrine and neuroimmune mechanisms through which occupational stress may impact inflammation and immune dysregulation, resulting in long-lasting health consequences for HCWs. To accomplish this task, we first review the literature on moral distress, stress, and health in healthcare workers. Second, we summarize the main pathways linking occupational and interpersonal stressors to health in this population, focusing on neuroendocrine, immune, genetic, and epigenetic processes. Third, we propose a novel Psychoneuroimmunological Model of Moral Distress and Health in HCWs based on this literature. Finally, we discuss evidence-based individual- and system-level interventions for preventing stress at work and promoting resilience. In conducting this review, we argue that high stress levels in HCWs are a significant public health concern across the globe. Moreover, we suggest strategies to guide collective efforts for addressing preventable healthcare harm and fostering resilience in HCWs, including health policies, mental health initiatives, and fruitful avenues for additional research.
To accomplish this review, we conducted a comprehensive literature search on PubMed, Scopus, PsycInfo, and ClinicalTrials.gov to retrieve all relevant studies published through October 2023. This search strategy included a combination of the following keywords: occupational stress, moral distress, resilience, frontline HCWs, mental health, physical health, neuroendocrine, neuroimmune, inflammation, immunity, genetic, epigenetic, and evidenced-based interventions. Medical Subject Headings (MeSH) or equivalent and text word terms were used. To be considered for this review, articles had to be peer reviewed and written in English. This review was registered with the International Prospective Register of Systematic Reviews (PROSPERO) database (Registration No. CRD42023393564).
2. Moral distress in frontline healthcare workers
The experience of moral distress is a critical contributor to the current healthcare workforce shortage. As alluded to above, moral distress refers to a clinician's inability to take what they assess to be the ethically appropriate action due to either external (systemic, institutional, structural factors) or internal (intrapersonal, interpersonal factors) constraints (Table 1) [23]. Moral distress is distinct from other forms of psychological distress, as it refers to the violation of an individual's professional integrity and their inability to act according to their moral beliefs, core values, and professional obligations [23]. Two other defining features differentiate moral distress from other types of stress: first, it creates long-lasting, invisible moral wounds that contribute to mental suffering (i.e., moral residue) [33]; and second, it may build up over time when an individual is repeatedly exposed to morally-distressing situations (i.e., crescendo effect), resulting in moral injury [23].
Table 1.
Causes of Moral Distress: external and internal constraints.
| External constraints |
|
| Internal constraints |
|
Moral distress was initially reported in nurses [34], but can be experienced in HCWs across multiple different disciplines and professions [27,35,36]. It is particularly pronounced in clinicians who care for seriously and/or terminally ill patients [37]. Nurses are considered to have an increased risk of moral distress, particularly those working in high-stress environments, including intensive care units, neonatal intensive care units (NICU), emergency departments, and surgery, and oncology units [38]. The prevalence of moral distress in HCWs varies depending on the profession, clinical setting, and psychometric instrument used to screen for moral distress. In 2019, 20 % of HCWs reported considering leaving their current position due to moral distress and 33 % of new nurses left their position within two years due to moral distress [39]. Alarmingly, the ongoing experience of moral distress can cause severe mental health issues, including emotional exhaustion, burnout, depression, and an increased risk of suicide [[5], [6], [7],9,28]. These mental health problems reduce HCWs’ capacity to provide high-quality patient-centered care, resulting in higher rates of medical error and hospital-acquired infections, reduced patient satisfaction, and in turn, increased job dissatisfaction, and attrition at work [[10], [11], [12], [13]].
Major sources of moral distress in HCWs can be identified at a patient-level (e.g., potentially non-beneficial, life-prolonging interventions; conflicts over goals; proxy decision-making; withholding versus withdrawing treatment), provider-level (e.g., poor communication; lack of respect for a patient's dignity and/or autonomy; witnessing unethical behaviors by colleagues), and system-level (e.g., working under precarious conditions; inadequate resources; understaffing; austerity, rationing and economization; and value incongruence between the organization and HCWs) [[39], [40], [41]]. Healthcare system redesign and systemic and political austerity measures that include changes in primary care practices (e.g., insufficient staffing ratio, reduced resources, inadequate knowledge, reduced availability of certain treatments, lack of acute care beds, reduced time at the bedside) to attempt to improve the efficiency and effectiveness of patient care, have been shown to increase HCWs' moral distress and adversely impact patient outcomes [24]. For example, the risk of inpatient mortality increases by 7 % for every additional patient a nurse must care for [42]. Similarly, physicians with burnout have more than twice the odds of self-reporting medical error, even after adjusting for fatigue, specialty, work hours, and their work unit's safety rating [43]. These results are concerning, given that if HCWs are pushed past their physical and mental limits, they will not only make more mistakes but be at greater risk for developing serious mental health problems. In contrast, amongst nursing facilities, increasing the workforce by 20 min per day has been linked to 22 % fewer COVID infections and a 26 % reduction in COVID-19-associated mortality rate [44].
Taken together, the experience and consequences of moral distress in HCWs create an occupational hazard in healthcare that jeopardizes patient care, patient safety, HCWs' health, and functioning of the healthcare system itself. The high risk moral distress confers on comorbidity with psychiatric disorders is concerning, especially due to HCWs’ already-elevated risks of burnout, depression, and suicide.
Although moral distress is conceptualized differently than other forms of stress, the psychoneuroimmunological mechanisms underlying moral distress-related mental and physical health problems are likely similar. Studies on moral distress in HCWs started to increase in number since the early 2000s and peaked during the COVID-19 pandemic. Despite this surge in research, we were unable to identify any studies that investigated the relation between moral distress and changes in the neuroendocrine and neuroinflammatory markers in HCWs (Systematic Review registration PROSPERO Nr. CRD42023393564).
Occupational stress shares some similarities with moral distress in that both concepts explore job demands and its negative emotional impact within the work environment, which can result in chronic stress responses. Occupational stress is a broader concept related to work demands, whereas moral distress is specifically tied to moral and/or ethical conflicts in professional settings. Therefore, in the following section, we review the literature on the effect of occupational stressors on neuroendocrine, immune, genetic, and epigenetic processes, and overall health of HCWs. Subsequently, we use the results of this synthesis to develop a theoretical model of the psychoneuroimmunology of moral distress and health in HCWs, which can serve as a foundation to build a series of novel hypotheses.
3. Occupational stress pathways and its deleterious effects on health
Occupational stress can result from a misalignment between job demands, job resources, and an individual's capacity to cope with or meet these demands. Prolonged exposure to high levels of occupational stress can have negative effects on an employee's health, well-being, job performance, and overall quality of life [45]. The field of occupational health and safety is primarily concerned with understanding and addressing the impact of job characteristics and demands on the health and well-being of workers. Its overarching objective is to enhance workplace safety and promote the overall health of employees [46]. Throughout the 20th century and, particularly, in the aftermath of the COVID-19 pandemic, the introduction of new working methods and procedures has led to the establishment of various health and safety management standards and guidelines, and underscores the importance of management and leadership skills to protect HCWs [[47], [48], [49]]. Simultaneously, research has been conducted not only to identify occupational stressors but also to gain a deeper understanding of the underlying mechanisms explaining how job demands can lead to negative health outcomes [45,[50], [51], [52]].
The Job Demands-Resources (J-DR) health model [32] represents a further development of the Demand Control model (DCM) by Karasek [50] and classifies job characteristics into two general categories: job demands and job resources, encompassing both physical, mental, and social aspects. The J-DR health model posits that job resources interact with job demands and influence an employee's well-being. The model links job demands and job resources to positive and negative biopsychosocial health outcomes, as well as motivational processes, through both pathogenic and a salutogenic pathways.
It has been consistently demonstrated that, irrespective of the working context or specific task-related demands, job resources play a particularly beneficial role in maintaining work engagement under conditions of high job demands [53,54]. Consequently, employees exhibit more positive work attitudes (e.g., motivation, task enjoyment, commitment) when both job demands and job resources are high. In other words, under high job demands, job resources can enhance work engagement. Conversely, when job resources are scarce and job demands exceed a HCW's perceived capacity to cope, stress occurs, leading to negative health outcomes (e.g., insomnia, exhaustion, burnout, depression) and negative motivational processes (e.g., turnover intention) [55].
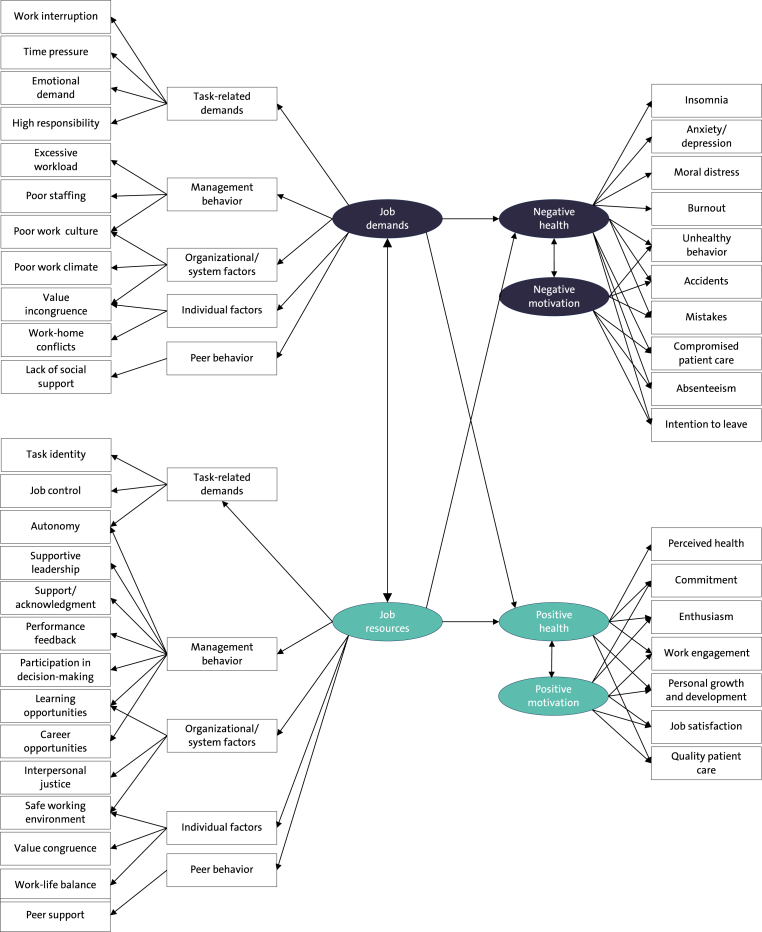
The J-DR health model is a useful instrument to analyze and identify factors contributing to health impairment and negative motivational processes at the workplace. It delineates key considerations for implementing public health interventions aimed at improving the work environment and safety of HCWs. Moreover, the model serves as a tool for studying the underlying biological mechanisms linking occupational stress to health [32]. Here, we present an adapted version of the J-DR health model that specifically addresses job demands and job resources of HCWs (Fig. 1).
Fig. 1.
Example of a Job Demands-Resources Health Model in Healthcare
The model links job demands and job resources through a pathogenic and a salutogenic path to both positive and negative health outcomes and motivational processes. Job demands and job resources interact and determine HCWs' well-being. The combination of high job demands and low job resources is a risk factor for exhaustion and burnout, whereas the combination of high job demands and high job resources promotes a heightened level of motivation, task enjoyment, and commitment.
Despite a large body of research examining the adverse health outcomes resulting from occupational stressors, understanding and mitigating stressor exposures and responses in HCWs in the current healthcare crisis continues to be overlooked. In the following section, therefore, we delve deeper into the psychoneuroimmunology of occupational stress, delineating the major stress pathways linking occupational and interpersonal stressors in HCWs with altered immune function and inflammation, which can in turn lead to serious mental and physical health problems. We then use this synthesis to model the negative effects of moral distress in HCWs.
4. Effects of occupational stress on mental and physical health in healthcare workers
During clinical practice, HCWs frequently experience both acute and chronic stress and are at particularly high risk of developing stress-related health problems. Research that has been conducted over the past decade on chronic occupational stress exposure in HCWs has revealed very high risk of burnout, depression, and suicide relative to the general population. Prevalence rates of burnout in HCWs was as high as 67 % during the pre-COVID period [56,57], whereas the overall prevalence of depression in nurses and physicians range between 25 % and 43 % [19,58], versus just 4 % in the general population [59]. Occupational burnout in physician is, in turn, associated with poor mental health, including exhaustion, reduced productivity, detachment from one's work, depression, suicidal ideation, early job retirement, and lower patient satisfaction [60]. In addition, accumulative evidence links burnout in physicians with an increased risk of somatic diseases, particularly cardiovascular diseases, chronic lung diseases, and skin diseases [61,62].
Both burnout and depression are substantial risk factors for suicide in HCWs [63]. Alarmingly, both nurses and physicians have a two-fold increased risk of suicide relative to the general population [64]. Young nurses and physicians are especially vulnerable to developing mental health issues because they often are required to manage excessive workloads, which can increase their stress levels; moreover, elevated levels of stress may compromise patient care, which may further exacerbate HCWs’ stress experience and impair their mental health [65,66]. Depression occurs in roughly 30 % of residents and young nurses [[67], [68], [69]]. Moreover, mental health issues in HCWs are a serious patient-safety issue, contributing to medical errors, patient dissatisfaction, and high healthcare worker turnover rates, and in turn, causing financial difficulties for hospitals [19,70].
In addition, the COVID-19 pandemic – as such a highly stressful event – has taken a serious toll on HCWs’ mental health. Results from a recent meta-analysis of eight studies revealed that HCWs have been disproportionally affected by mental health problems, including distress, anxiety, insomnia, depression and traumatization during the COVID-19 pandemic, relative to other occupational groups [71]. According to recent estimates, during the COVID-19 pandemic, 39 % of HCWs had insomnia, 23% had anxiety, and 23 % had depressive disorders [72].
5. Pathways linking moral distress, stress, and health in healthcare workers
5.1. HPA axis
Under basal conditions, the hypothalamic-pituitary-adrenal (HPA) axis continuously regulates the release of glucocorticoids (GCs) from the adrenal glands into the blood circulation, depending on circadian variations [73]. Glucocorticoids are potent anti-inflammatory and immunosuppressive agents and, with their ubiquitous expression of GC receptors throughout the body, regulate a wide variety of cellular processes, including immunologic, metabolic, reproductive, cardiovascular, and cognitive functions, mainly through glucocorticoid-receptor-mediated activation [74]. Their effect is largely mediated by protein-protein interactions that involve the transcription factor NF-κB [75].
However, excessive or insufficient release of GCs caused, for example, by chronic stress, exerts negative feedback on secretion-driving mechanisms via the activation of glucocorticoid receptors. The prolonged stress response disrupts the glucocorticoid signaling pathway and induces abnormalities in glucocorticoid receptor sensitivity, contributing to an increased inflammatory response, which is generally immunosuppressive [75]. Increased levels of glucocorticoids in the blood and glucocorticoid resistance are strongly linked to suppressed immune function, indicated by suppressed natural killer (NK) cell activity, antigen presentation, and T-cell proliferation, and a reduced number of circulating lymphocytes (B cells and T cells). Importantly, chronic, prolonged exposure to high circulating levels of GCs can result in various adverse health effects, including osteoporosis, hypertension, diabetes, dyslipidemia, and even neurodegeneration, and depression [[76], [77], [78], [79]].
HCWs are often engaged in shift work. Shift work, particularly night shifts, interferes with the physiological circadian rhythm and has the unwanted potential to alter autonomic nervous system, neuroendocrine, and immune processes that, in turn, have been linked to the development of many chronic health conditions like cardiovascular disorders [80]. Salivary cortisol is commonly used as a biomarker of psychological stress. Cortisol release follows a diurnal circadian rhythm, which is typically elevated after waking, then drops rapidly within subsequent hours and ultimately reaches a nadir in the evening [81]. Working night shifts and sleep deprivation have been associated with HPA-axis dysregulation, overall elevated levels of daily cortisol [82,83], suppressed gonadal steroids [84], and reduced endothelial function [85] in HCWs across different disciplines and occupational groups (nurses, physicians, residents), reflecting elevated physiological stress. Concerningly, night shifts and sleep deprivation not only affect mood, but also concentration and vigilance, which results in more mistakes during shifts [84].
Hair cortisol is another useful marker for the retrospective assessment of stress [86]. Consistent with the findings summarized above, excessive workloads have been associated with higher hair cortisol concentrations in HCWs, and HCWs who reported early life stress or stress later in life, as well as depressive symptoms, have been shown to have higher hair cortisol concentrations levels than those not reporting adverse life events [87]. Similar findings have been reported for palliative care clinicians suffering from burnout: they have an augmented cortisol awakening response and higher levels of diurnal cortisol secretion [88]. Analogously, medical residents have been shown to have an increased stress response when they are on call in an emergency department, including increased heart-rate, blood-pressure, elevated salivary alpha-amylase, and increases in their serum level of the key inflammatory cytokine interleukin (IL)-1β. This association is independent of gender [89]. Of note, elevated diurnal cortisol concentration has been shown to correlate with an increased risk of occupational injury in HCWs [90].
Triage refers to the categorization of patients with respect to their disease severity and prognosis for the purpose of determining priority for care, as was the case during both the first and second waves of the COVID-19 pandemic. Typically, triage during mass casualty incidents is linked with higher stress levels among HCWs [91]. Similar results have been observed in HCWs during the COVID-19 pandemic. Findings from a recent study found increased hair cortisol concentrations levels in HCWs who, during the current pandemic, experienced high distress due to an increased risk of exposure to the virus, fear of risk to their own and their family's health, greater workloads, and ethical and moral dilemmas during triage and patient care [92]. Additionally, changes in hair cortisol concentrations levels among HCWs three months after the outbreak of the COVID-19 pandemic predicted future burnout among HCWs [93]. Similar findings were reported for female nurses during the first wave of the COVID-19 pandemic: they had higher hair cortisol concentrations levels in the hair during the first wave relative to prior to the pandemic. In addition, nurses who worked in high-risk environments had higher hair cortisol concentrations levels than nurses who worked in low-risk settings [94].
Gender plays a crucial role in the inflammatory response to stress and cytokine expression [95]. However, few studies have evaluated the effects of stress and changes in biomarkers isolated in female HCWs, even though women are at the frontline of many healthcare professions, including nurses and physicians, and other healthcare professionals in social sector and administrative jobs. The U.S. Bureau of Labor Statistics estimated that, in 2021, approximately 78 % of HCWs were women [96]. Moreover, although both men and women can be affected by mental health problems, women are nearly twice as likely as men to develop depression [97]. Some studies in female HCWs have revealed higher levels of perceived distress, elevated hair cortisol concentrations, and impaired diurnal cortisol patterns [94,98].
Another stressor frequently reported among HCWs is the challenge to balance work, non-work, and family responsibilities. At this time, female physicians with children continue to be disproportionally represented in lower-status and lower-paid medical specialties, like family medicine, pediatrics, and psychiatry, whereas male physicians who are also fathers are more likely to work full-time and benefit from their spouses working part-time [99]. This said, more and more men working in healthcare continue to engage in multiple family roles (father, informal caregiver, or both) alongside pursuing an academic career; yet, little is known about the health effects of various family responsibilities for men relative to women [100]. One Canadian study collected salivary cortisol levels in physicians to assess the interactive effects of job strain and family responsibilities on physical functioning in 18 female and 23 male physicians. Interestingly, although both female and male physicians showed changes in their levels of the stress-sensitive hormone cortisol, no significant gender differences in diurnal mean salivary cortisol levels were observed, indicating that both female and male physicians react to the increasing responsibilities stemming from managing the work-family balance; and that this might be reflected by disruptions in HPA functioning (lower early morning cortisol levels; higher evening levels, a flattened stress response) [101]. Contrary to this finding, other studies have found that having a family can help to create meaning and enhance satisfaction in life, and contribute to a positive life orientation, and this may have a protective effect against cumulative work stress [102].
Moreover, violence against HCWs, especially against nurses – including verbal threats, physical and sexual abuse, and racial and ethnic harassment – has become an occupational hazard in acute patient care that can increase HCWs’ distress and feelings of fear or anger, contributing to increased stress levels, emotional exhaustion, and burnout [103]. Consistent with this finding, HCWs who have experienced verbal and physical aggression have been found to have higher hair cortisol concentrations levels, and to report increased burdens and more burnout symptoms [104].
5.2. Sympathetic nervous system
The catecholamines epinephrine and norepinephrine are the major mediators of the acute stress response released by the sympathetic nervous system (SNS), causing increases in blood pressure and heart rate, and the release of glucose from energy stores [77]. Catecholamines bind to G-coupled protein receptors, which are divided into α-adrenergic receptors and β-adrenergic receptors. Adrenergic receptors are expressed on many tissues throughout the body and are responsible for transmitting information from the extracellular environment to the interior of cells. Through these pathways, epinephrine and norepinephrine regulate immune and inflammatory function by secreting inflammatory factors [105]. During chronic stress, epinephrine and norepinephrine levels are elevated, which can foster a pro-inflammatory microenvironment in both the periphery and brain via the release of pro-inflammatory cytokines, including IL-1β, interleukin-6 (IL-6), and tumor necrosis factor alpha (TNF-α), as well as the inflammatory biomarker, C-reactive protein (CRP) [106,107], all of which exert immunosuppressive effects. In addition, the constant release of cytokines into the circulation may trigger an over-regulated glucocorticoid response that induces glucocorticoid resistance, contributing to cytokine over-secretion and increased inflammation [106]. Increased and sustained levels of catecholamines have been linked to T-lymphocyte apoptosis, altered distribution of NK cells and granulocytes, suppressed NK cell activity [108,109], and DNA damage [110], all of which are important defense mechanisms against different diseases. These effects may have long-lasting consequences, including DNA mutations, and thereby increase the potential for abnormal cellular transformation [[111], [112], [113]].
A substantial body of research highlights plausible mechanisms through which chronic stress and low-grade inflammation may contribute to adverse health consequences in HCWs. For instance, partial and total sleep deprivation due to night shifts and 24-h shift-work impair immune function, resulting in the elevated excretion of epinephrine [114] and several pro-inflammatory cytokines, namely IL-1β, IL-6, and TNF-α [[115], [116], [117]]. Low-grade systemic inflammation in turn, as reflected by increased CRP and elevated concentrations of cytokines (e.g., IL-1β, IL-6; TNF-α) is a robust risk factor for the onset of many diseases, including major depression, cardiovascular disease, and stroke [[118], [119], [120]].
Neuroendocrine changes also are observed in female HCWs working under high job strain, which is associated with increased peripheral levels of neuroendocrine stress hormone (e.g., epinephrine, norepinephrine, cortisol) that persists over time relative to HCWs with low job strain [121]. Similar results have been reported for surgical nurses who met the clinical criteria for PTSD and depression, who also demonstrated higher inflammatory cytokine levels; moreover, this association is thought to be mediated through immune dysregulation [122]. Sustained low-grade inflammation also has been observed in emergency physicians (age range 32–46 years) at least three days following a 24-h shift, while IL-8 secretion was found to be considerably higher with advancing age [123]. Lastly, a similar association has been identified between stress exposure and the sympathetic regulation of immune responses. Salivary alpha-amylase activity (sAA) is a surrogate marker of sympathetic nervous system activity in response to stress. Nurses with self-reported stress have been found to have more pronounced changes in sAA, and smoking and medication use strengthened this link [124]. Interestingly, job resources – defined as social support through the supervisor and job autonomy – seem to buffer the negative impact of job demand (i.e., emotional demands, interpersonal conflicts) on health, as manifested by a decreased sAA awakening response and lower serum IL-6 levels [125,126].
Lastly, the negative effect of stress on health and work performance can be mediated by other life stressors, including personal, social, and environmental factors experienced earlier in life [120,127]. A recent study investigating stress and health in nursing students found a close relationship between nurses’ lifetime stress exposure with several biomarkers, including IL-6, IL-8, and TNF-a, which in turn increased the risk for various health conditions, including hypertension, diabetes, and depression [128].
5.3. Vagus nerve
The vagus nerve is a major pathway within the parasympathetic nervous system (PNS) which, in conjunction with the SNS, forms the autonomic nervous system (ANS). The vagus nerve plays a critical role in regulating metabolic homeostasis by controlling digestion, heart and respiratory rates, and gastrointestinal motility, and secretion. Its main function is to down-regulate stress responses and restore homeostasis [129]. The vagus nerve also has important immune modulating functions [130]. Any disruption of this finely-tuned brain-gut-immune axis can result into various disorders, including inflammatory, cardiovascular, metabolic, and neuropsychiatric disorders [131,132].
Chronic stress and the sustained release of glucocorticoids and catecholamines inhibit the vagus nerve and, thus, impede its anti-inflammatory effects, leading to increased concentrations of cytokines (e.g., IL-6, TNF-α) in the gut. Furthermore, stress-induced release of glucocorticoids and catecholamines affects the gut by binding to glucocorticoid and adrenergic receptors in the enteric nervous system, inducing intestinal inflammation, thereby modulating gut microbiota, intestinal motility and permeability that, all together, compromise intestinal barrier integrity [[133], [134], [135]].
Given its complexity, vagal function is difficult to measure directly. However, because some vagus nerves innervate the heart, vagus nerve activation can be determined by measuring heart rate and heart-rate variability (HRV), or variations in the time interval between consecutive heartbeats. During conditions of chronic stress, heart rate increases and HRV decreases, leading to dysregulation of the HPA axis and SNS [136,137]. For example, in highly-stressful situations, such as working in an NICU, cortisol values correlate positively with HR and inversely with HRV, suggesting that the HPA axis and the autonomic system work in synergy under conditions of high stress [138]. Furthermore, reduced HRV and endothelial dysfunction have been reported in HCWs as long-term effects of their job strain and shift work, underlying an important pathway linking chronic stress with cardiovascular disease [85]. These results are consistent with robust evidence linking prothrombotic changes – including elevated heart-rate, lower heart rate variability, and higher systolic blood pressure – with an increased risk of cardiovascular disease and mortality [139].
5.4. Immunity
Immunity refers to the natural and acquired resistance of an organism to bacterial and viral pathogens, infections or disease, while at the same time being adequately tolerant to avoid allergic reactions and autoimmune disease [140]. Chronic and elevated concentrations of stress hormones (e.g., catecholamines; corticosteroids) can dysregulate or suppress cellular and humoral immune responses by altering the type 1/type 2 cytokine balance and, by doing so, induce low-grade inflammation, which as a consequence suppresses the immune-protective function of the cells [141]. Moreover, T and B lymphocytes, macrophages, neutrophiles, and natural killer cells have β-adrenergic receptors. Through these β-adrenergic pathways, chronic stress can modulate and suppress different aspects of immune function, including lymphocyte migration and proliferation, antibody secretion, cytotoxic activity, and macrophage activation, principally through the stress-induced release of catecholamines and glucocorticoids [105,142].
Natural killer cells have a critical role in the defense against viral infections and their malfunction can have severe health consequences; for instance, through impaired protection against neoplastic growths [77,140]. Stress-induced immune dysfunction can also be observed in otherwise healthy nurses, among whom job stress and workload have been linked to increased concentrations of catecholamines and decreased NK cell activity [143], thereby negatively impacting immune function.
5.5. Genetic and epigenetic changes
Individuals vary substantially in their response to adversity, with a considerable proportion manifesting resilience [144]. Epigenetic mechanisms, especially alterations in DNA methylation and abnormal microRNAs (miRNA) expression, may be an important molecular pathway explaining individual differences in stress responses and through which psychopathology and human diseases occurs [[145], [146], [147], [148], [149], [150], [151]].
Epigenetic modifications refer to heritable phenotype changes that do not involve changes in the underlying DNA sequence but rather impact gene activity and expression. These epigenetic modifications result from external or environmental influences, such as exposure to temperature changes, chemical compounds, diet, or other forms of stress [152]. Stress-induced epigenetic modifications in a genome (i.e., changes in gene expression) are associated with altered DNA methylation patterns, altered histone modifications, and several non-coding RNA-mediated processes [153]. Specifically, miRNAs play an important role in the regulation of gene expression at the posttranscriptional level [154]. miRNAs are expressed in the central nervous system and in all body fluids (serum, plasma, saliva), crucial for the development of neural structures, and have been implicated in inflammatory and autoimmune processes [151].
Chronic stress, via activation of the SNS and HPA axis and release of catecholamines and glucocorticoids, can affect gene expression and cellular and molecular functions in part through epigenetic mechanisms, thereby dysregulating immunity [155,156]. For instance, elevated glucocorticoid levels have been linked to the deacetylation of histones and reduced expression of immune response genes [156,157]. Moreover, stress hormones can mediate immunosuppression by altering the expression of the transcription factors AP-1 and NFκB, both of which have critical roles regulating and activating the immune system, including lymphocyte function [158].
Multiple factors including genetic, epigenetic, and environmental influences and their interactions synergistically contribute to the manifestation of vulnerability and resilience following early-life adversity [159]. The three-hit concept of vulnerability and resilience suggests that interactions between someone's genetic predisposition (hit 1), early life environment (hit 2), and later-life environment (hit 3) determines vulnerability or resilience in different scenarios. Accordingly, depending upon a person's genetics and early-life environment(s), later-life environments may compromise mental functioning and vulnerability may arise, though the same individual might be resilient to stress when it occurs within supportive environmental conditions [160]. This is especially crucial when we consider how to establish work environments for HCWs that allow them to bring out their best selves, enhance their commitment, work engagement, and job satisfaction, and promote human flourishing.
Although stress-induced epigenetic changes can occur during all life phases [[161], [162], [163], [164]], the epigenome is particularly sensitive to stress exposure during the early phase of conception, and during the prenatal and postnatal periods. This is important as the healthcare workforce is predominantly female [96] and the quota of female doctors has increased in all OECD countries over the past two decades while female doctors are on average younger than male doctors. Most likely, a big proportion of the female workforce in healthcare are mothers [165]. Animal models indicate that stress exposure during these sensitive periods may lead to stress-induced changes in DNA methylation in the hippocampus and prefrontal cortex in the offspring that, in turn, may influence several downstream neuroendocrine, neuroimmune, and neurocognitive pathways, contributing to variances in individual stress vulnerability and resilience [[166], [167], [168]]. For instance, prenatal maternal depression and postnatal exposure to adversity are both linked to alterations in the offspring's DNA methylation of several genes, including the glucocorticoid receptor gene NR3C1, serotonin-transporter gene SLC6A4, and oxytocin receptor OXTR [[169], [170], [171]]. It is hypothesized that these variations in methylation patterns may confer disease susceptibility or resilience for psychopathology in the face of adversity during later life [172].
Epigenetic modifications in response to stress exposure have been observed in HCWs, particularly when working night shifts that entail significant disruptions in the chronotype (sleeping patterns), which may lead to alterations in HCWs biological aging. Findings indicate an association between the duration of night shifts and reduced telomere length [173,174], alterations in DNA methylation, and accelerated aging in HCWs [175]. These epigenetic modifications have been found to be especially prevalent in HCWs who are stressed, obese, or both. A more recent study compared the onset of depression and PTSD with methylation levels among frontline HCWs working in COVID-19 wards versus those working in non-COVID-19 wards [176]. In this study, frontline HCWs more often met the criteria for clinically-relevant depression and PTSD and also exhibited upregulated methylation of corticotropin-releasing hormone receptor 1 (CRHR1), which is involved in regulating the HPA axis and the dopamine receptor D2 (DRD2), which may account for the development of behavioral and substance disorders like anhedonia, depressed mood, and psychotic symptoms. DNA methylation in CRHR1 and DRD2 also was associated with increased cortisol levels [177]. These results highlight the neuroplasticity of gene expression involved in the stress response and suggest that CRHR1 and DRD2 methylation can be modulated through the experience of chronic stress and that epigenetic modification in these genes might be implicated in the development of depression and PTSD [176,178].
As a corollary to this, it might be possible to reverse stress-induced methylation modifications and thereby improve mood and symptom burden in HCWs. To give an example, in animal models, prolonged periods of social isolation have been found to cause neuroinflammation and altered immune functioning, including elevated levels of serum glucocorticoids and cytokines [179], as well as alterations in the DNA methylation of various genes in the hippocampus and prefrontal cortex (e.g., the CRF-promoter and serotonin transporter or the brain-derived neurotrophic factor (BDNF) genes) [178,180]. These stress effects are partially mediated through NF-κB, a key pro-brain-derived neurotrophic transcription factor [181]. It is plausible to assume that social distancing measures during the early COVID-19 likely exacerbated these neuropsychobiological effects in HCWs’ stress. Intriguingly, these stress-induced epigenetic alterations and immune system deregulation could be reverted, at least in part, with environmental changes [144], the provision of care and social support [182,183], and antidepressant treatment with imipramine or escitalopram [180,184,185]. These results highlight the enormous potential that the brain has, which enables it to continuously form new neural connections (i.e., neuroplasticity) following adversity and the important role that social and environmental influences have on neuroplasticity and resilience.
6. Psychoneuroimmunological Model of moral distress and health
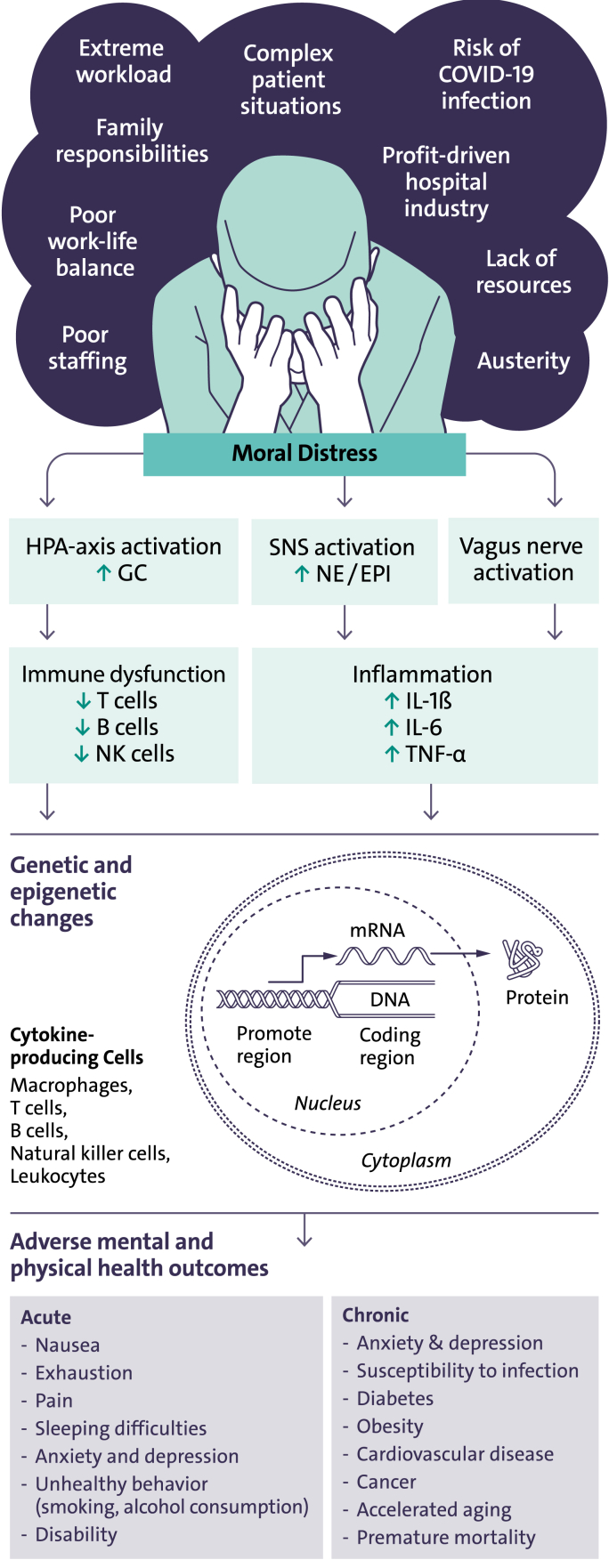
Our literature review indicates that HCWs have an increased risk for low-grade chronic inflammation and dysregulated immune function secondary to occupational stress. Low-grade systemic inflammation and immune dysregulation have been consistently associated with an increased risk of adverse health outcomes, including burnout, depression, cardiovascular disease, stroke, and early mortality [120,186]. In accordance with the literature on occupational stress, we argue that moral distress in HCWs is a significant source of stress that has the potential to dysregulate the autonomic, neuroendocrine, and immune systems, leading to long-lasting negative health outcomes. Building upon this premise, we propose a Psychoneuroimmunological Model of Moral Distress and Health elucidating the impact of moral distress on neuroendocrine, immune, genetic, and epigenetic processes, providing a hypothetical explanation for the increased risk of morbidity and mortality among HCWs (Fig. 2). Alongside the J-DR health model in HCWs (Fig. 1), this theoretical model can serve as a foundation for providing a deeper understanding of the clinical significance of moral distress in HCWs and its implication for the immune system and health. The testable model, which links moral distress in HCWs to immune dysregulation, generates a multitude of hypotheses, paving the way for future research avenues linking moral distress with psychoneuroimmunological outcomes. The results of this work may, in turn, serve as a catalyst for ethics quality improvement projects, thereby aiding in the transformation and improvement of the healthcare environment through evidence-based practice.
Fig. 2.
Psychoneuroimmunological Model of Moral Distress and Health
The stress response is mediated, in part, by three bidirectional pathways that enable the brain to communicate with the peripheral immune system and vice versa. These signaling pathways include the (a) hypothalamic-pituitary-adrenal (HPA) axis, (b) sympathetic nervous system (SNS), and (c) vagus nerve. Exposure to a stressor activates the HPA axis and the SNS, which in turn triggers the release of hormones that modulate immune functions, including catecholamines (epinephrine and norepinephrine), glucocorticoids, and pro-inflammatory cytokines (interleukin-1β, IL-1β; interleukin-6, IL-6; tumor necrosis factor-α, TNF-α). During acute stress, the release of these neuroendocrine modulators enhances immune function, and thus is an adaptive psychophysiological mechanism that confers immune protection in response to an infection or an injury and aims to restore homeostasis. Conversely, exposure to chronic or prolonged stress results in upregulated autonomic, neuroendocrine, and inflammatory pathways and, by doing so, increases inflammation and induces immune dysregulation that may alter immune defense mechanisms and elevate someone's susceptibility to certain diseases and mental illnesses.
7. Call to action: Responding to stress in healthcare environments and clinical practice
The ramification of stress in HCWs is a public health concern. Stress-related disorders in HCWs, including burnout, depression, and increased risk of suicide, are major contributors to disease burden, disability, and suffering, with significant personal, professional, and financial consequences [70]. The impact of stress on the mental and physical health of HCWs are tragic outcomes, particularly given their onset is largely avoidable.
Stress is deeply entrenched in healthcare environments and clinical practice. In particular, situations in which HCWs are unable to take what they assess to be the ethically appropriate action due to lack of resources or other system-level factors, trigger moral stress that compromise their well-being. The current healthcare crisis highlights the urgent need for a paradigm shift from the long-held standard of crisis management to a focus on prevention and health promotion for HCWs. Efforts to improve HCWs' working conditions with evidence-based interventions to preserve their health and thereby ensure high-quality patient care and patients’ safety are paramount to prevent healthcare harm. A multidimensional approach is necessary to address the complex underlying systemic and social factors that give rise to moral stress. Meaningful changes to improve healthcare environments require a shift from interventions occurring at an individual level to a system-wide effort. Furthermore, the implementation of a public health response necessitates the coordination and partnerships among different stakeholders, including policy agencies, healthcare organizations, insurers, healthcare leaders, HCWs, and other members of the healthcare workforce.
In the following sections, we will summarize system-level and evidence-based individual-level interventions that could guide collective efforts to improve work environments, mitigate deleterious effects of stress, and promote resilience in HCWs.
8. System-level interventions to improve work environments, reduce stress, and promote resilience
8.1. Health policy responses
Attracting and retaining the nursing workforce will require a more nuanced understanding of sources of stress and what nurses are looking for in their profession and employer. Stressful work conflicts emerged in part because HCWs' family and work responsibilities collide: the increased work exposure to communicable diseases often put at risk their families’ health, and their increased workload conflicts with their family responsibilities, while at the same time, HCWs feel torn between their duty at work and responsibilities toward their patients. According to a nursing survey over the past two years, flexibility, meaning, and balance emerged as the most important factors to stay in direct patient care [187]. Following these gleaned insights, system-level interventions and structural solutions that help ensure a manageable workload and reduce the risk of moral distress and burnout in HCWs include, among other things, sufficient staffing, limited duration of shifts at the frontline, a safe environment, protected biobreaks during shifts for sitting down, eating and drinking, predictability of schedule, ability to take paid time off, and compensation with practical support, such as paid childcare services, elderly or animal care, as well as leadership training. These system-level interventions continue to be critical in retraining the healthcare workforce [[188], [189], [190]].
8.2. Employee participatory programs
Given the increased risk of mental health issues in HCWs and the negative impact of occupational stress on immune function and disease susceptibility, a holistic, system-based approach is needed to address and mitigate moral distress and burnout in HCWs. We, therefore, advocate for a system-based approach to creating working environments that aim to reduce stress in HCWs. Consistent with this goal, research has demonstrated that targeting organizations rather than individuals is more effective at reducing moral distress resulting from austerity, because such interventions are tailored toward improving the workplace climate, health behaviors, and absenteeism [191]. Employee participatory programs are one example of an effective organizational intervention. Such interventions prompt employees to actively engage in identifying workplace problems and feasible solutions/actions to improve their working environment [192]. One recently published study investigated the effects of participatory workplace improvement programs on stress biomarkers and found a significant decrease in different inflammatory markers (i.e., IL-6, IL-12) both one week and three months following completion of the program [192], underscoring the potential and effectiveness of such programs at improving mental and physical health among HCWs. In support of these results, a national study among U.S. internal medicine physicians found that perceived organizational support and the feeling of being valued by their healthcare institution could significantly lower moral distress [28].
8.3. Ethics consultations
Ethics consultations may be another potential bridge that can be used to mitigate the negative effects of stress in HCWs. Evidence indicates that involving clinical ethicists in ethical deliberations helps to diminish moral distress in HCWs [193]. In particular, the creation of “safe spaces” is an opportunity to facilitate conversations about ethical difficulties and distressing events where diverse moral insights, voices, and perspectives can be heard, considered, and discussed, without ulterior motives, as a means to finding a path forward to navigate these difficult ethical issues [194]. By viewing situations through an ethical lens, clinical ethicists may also help others to recognize mechanisms or parts of a system that are not working well and identify solutions to either fix or overcome them.
8.4. Narrative medicine
Finally, narrative medicine could make a powerful contribution to improving healthcare practices, and reducing stress and promoting resilience in HCWs in many ways [195,196]. Narrative training prompts health care providers to write about a clinical encounter that had struck them at the core and elicited a strong feeling in them, be it sadness, frustration, anger, or joy. Health care workers are instructed to focus on the inside (their emotions) rather than on the outside, and thereby reflect on patient encounters and clinical situations and why, in part, they reacted the way they did. Upon sharing the text with their peers, the group analyzes the story's structure, and thereby adopting multiple perspectives, recognizing metaphors and allusions, gaining new insights, and reflecting on their own feelings. Telling stories provides meaning, context, and improves the understanding of what matters, while prompting providers to reflect on their own feelings, actions, and modes of expression [197].
The mechanisms through which narrative training benefits HCWs’ health may be multifold: narrative medicine allows for self-reflection in a safe, collective space. It encourages a shift into a state of vulnerability, in which an individual may find themselves appealed by sharing their strongly felt experiences with peers. This, in turn, may encourage colleagues to express their own experiences, in which insecurity, unsettlement, frustration, or any other type of uneasy reaction were experienced in the clinical environment. We believe that it is the human connection that occurs while sharing stories and the confrontation with our own vulnerability and the vulnerability of others that reduces stress, promote meaning-making and resilience, and provides hope. As a result of narrative competence, individuals may experience validation and increased awareness of their own subjective emotions, thoughts, and experience. Much like their patients, articulating the challenges they encounter in their medical practice may yield a healing effect, restoring their compromised professional and personal integrity, and fostering moral repair [198]. This process can assist HCWs in maintaining a sense of moral agency and control [199].
9. Individual-level interventions to reduce stress and promote resilience
A number of psychosocial interventions and lifestyle modifications including dietary changes, environmental improvements, psychological support, mind-body interventions, and even pharmacological treatments have documented potential therapeutic benefits for mitigating the negative effects of stress and reducing inflammation, thereby improving immune function and promoting wellbeing. In particular, interventions that activate vagal function (and thus increase/normalize heart rate variability and improve HPA axis function) such as relaxation techniques and mindfulness-based stress reduction, but also exercise, a balanced diet, and psychosocial support have shown promise for restoring balance between SNS and PNS arousal [128,[200], [201], [202], [203]]. Therefore, such interventions may have the potential to mitigate stress, including moral distress, in HCWs and may be of particular relevance to those experiencing high job strain.
9.1. Mind-body interventions
The vagus nerve can be influenced through slow breathing and mind-body interventions like meditation and yoga, and mindfulness-based therapy can increase vagal tone and has been shown to improve mood, reduce anxiety, and restore resilience in HCWs [204]. Furthermore, its stimulation has been documented to attenuate glucocorticoid and cytokine production [130,205], enhance telomerase activity [203], and improve neuroplasticity and functional connectivity within the hippocampus and prefrontal cortex, both of which are key sites of pathology in major depressive disorder [206]. However, such positive health effects only are sustained if stress-management techniques are continued [205], and finding time for such exercise is challenging in the fast-paced healthcare environment. In addition, vagus nerve stimulation has demonstrated promising results in treating treatment-refractory depression, PTSD, and inflammatory bowel disease, its therapeutic effect argued to be mediated through its relaxing and anti-inflammatory properties [207].
9.2. Exercise
Regular physical exercise of moderate intensity influences neurobiological mechanisms and promotes resilience against stress through multiple mechanisms. First, physical exercise activates vagal tone, whereas prolonged periods of sedentary time reduce heart rate variability and vagal nerve function [208]. Second, physical activity has a positive influence on endocrine and immune function. Physical exercise can activate the HPA axis and trigger cortisol secretion, which has a beneficial anti-inflammatory effect. Likewise, physical activity reduces levels of pro-inflammatory cytokines and CRP [208,209]. Interestingly, individuals who train on a regular basis have better protection against stress-induced increases in cortisol [210]. If implemented correctly, exercise may have several health benefits relevant for HCWs, as exercise may have the potential to prevent infections and strengthen the immune system [211]. The implementation of workplace wellness programs, aiming to strengthen the immune system, overall health, and increase job satisfaction despite numerous work constraints, could be a valuable resource [212]. However, without protected biobreaks for HCWs to sit, eat, and drink, any additional activities added to their already limited personal time do not yield discernible health benefits. Therefore, these interventions require careful consideration.
9.3. Diet
A healthy, balanced diet has the potential to positively impact the gut microbiota and, thereby, reduce gut inflammation while enhancing body metabolism, and physical and emotional functioning [213]. Robust evidence links specific nutrients [214] and probiotics [215] with an improved vagal messenger pathway that strengthens the communication between the gut and brain, specifically brain regions that include the prefrontal cortex, hippocampus and amygdala, thereby promoting resilience in the face of adversity [216]. As a corollary to this point, high-carbohydrate and high-fat diets have been related to inhibited vagus activity [217]. In fact, busy HCWs are particularly bad at eating well, especially when working clinically [218]. Particular attention has focused on omega-3-polyunsatured fatty acids (O3PUFAs), which can be found in fish and other seafood, in some plant oils, and in nuts, because of several health benefits—namely, they regulate blood pressure, inflammatory processes, and several cellular functions, including cell-signaling, cell membrane fluidity, and structural maintenance. In addition, O3PUFAs are known to have cardioprotective effects [219] and are effective at reducing depressive symptoms [220]. Daily intake of O3PUFAs also appears to have positive health effects in both female and male nurses: after O3PUFAs intake for eight consecutive weeks, both genders had lower diurnal saliva cortisol concentrations and reported diminished symptoms of occupational burnout, including reduced emotional exhaustion and increased feelings of personal accomplishment [221]. Taken together, these results suggest beneficial effects of O3PUFAs on HPA function, immune function, and regional brain activity [222].
9.4. Psychosocial support
Results from several other studies suggest promising avenues to counterbalance the negative effects of stress while building resilience through behavioral, psychosocial, pharmacological, or phytochemical modulation of several factors implicated in HPA-axis function, the immune system, DNA methylation, and neuronal plasticity [[223], [224], [225], [226]]. Social support is an important protective factor when one is confronted with stress and adversity and it exhibits potentially modifying effects on stress-induced DNA methylation [182,183]. Consistent with these results, during the COVID-19 pandemic, perceived social support significantly reduced psychological distress in HCWs [227,228], underscoring the importance of facilitating opportunities to create social ties with colleagues during stressful situations and times. Emerging evidence also supports the therapeutic potential of psychological interventions to positively influence epigenetic processes in HCWs [226,229]. Furthermore, some experimental drug trials have revealed the ability for medication to partially reverse stress-induced HPA-axis dysregulation and DNA methylation, suggesting the potential benefit of the prophylactic or early use of antidepressants as protection against depressive symptoms in high-risk or frontline HCWs [180,184,185,230].
10. Discussion
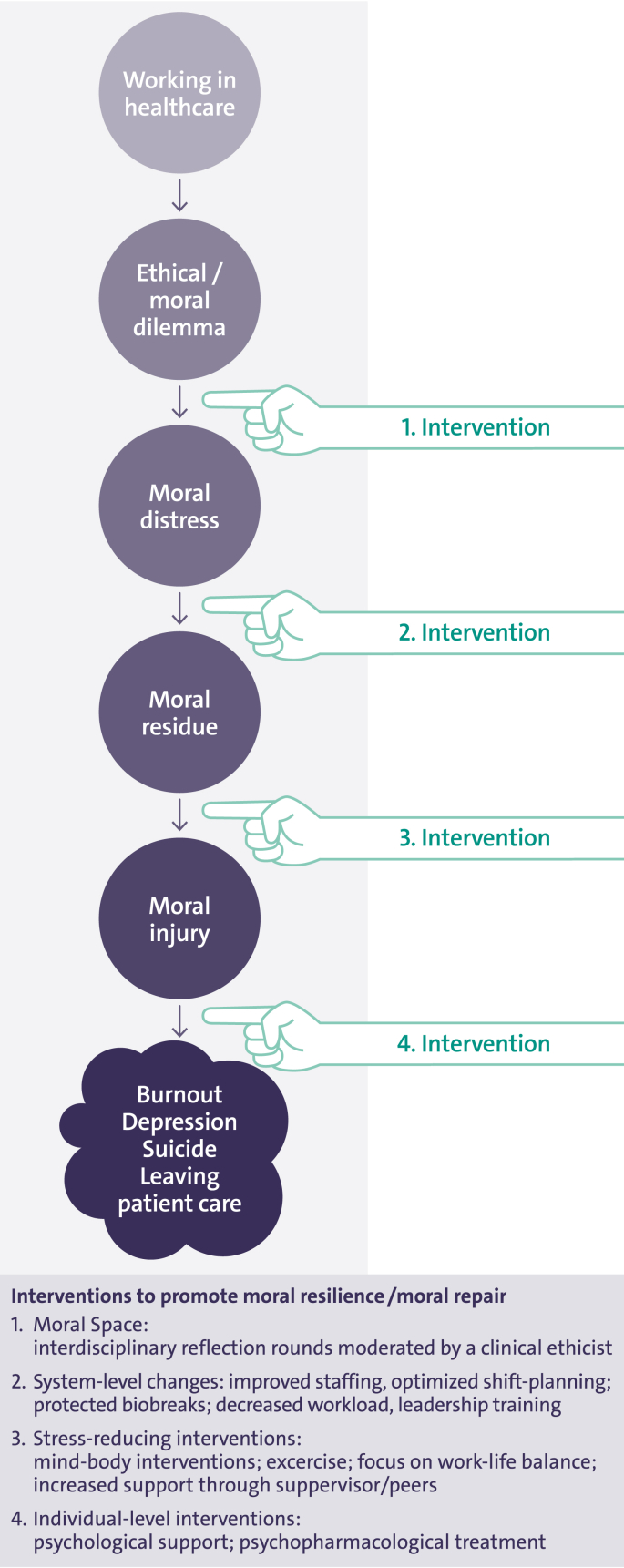
Moral distress is an occupational hazard that can translate into severe mental health problems in HCWs, jeopardizing the quality of patient care and likely being responsible in part for the current healthcare turnover crisis. Despite the robust link between moral distress, mental health issues, and HCWs’ intent to leave patient care, the immunological impact of moral distress in HCWs remain poorly investigated. In this review, we thoroughly examined both psychological and biological factors contributing to health impairment in HCWs. We have proposed an adapted version of the J-DR health model that specifically reflects the distinct job demands and job resources of HCWs, drawing insights form the occupational health psychology literature. Expanding on this model, which links occupational stress to health, we present a testable Psychoneuroimmunological Model of Moral Distress and Health in HCWs (Fig. 2), illustrating the potential neuroendocrine and neuroimmune mechanisms underlying moral distress in HCWs that may be used as a framework for future research to better understand how moral distress can affect psychobiological processes that impact health. Built upon our premise that moral distress in HCWs is a significant source of stress with the potential to impact the immune system and health, we have designed a conceptional framework featuring targeted health interventions aimed at mitigating the adverse effects of moral distress and promoting resilience in HCWs (Fig. 3).
Fig. 3.
Interventions for promoting resilience and moral repair.
Recent insights from studies on the neurobiology of stress and its impact on vulnerability and resilience indicate that stress-induced alterations in HPA function, neuroendocrine and immune markers, epigenetics regulation, and brain activity may be partially reversed through lifestyle modification, psychosocial support and even psychopharmacological interventions. This suggests several potential targets for interventions designed to promote resilience and mitigate the effects of moral distress in HCWs. Therefore, interventions that aim to improve heart rate variability and, thereby, improve HPA axis function and SNS activity represent promising avenues for building resilience and counterbalancing the adverse effects of moral distress among HCWs.
The creation of “safe spaces” seems to be one of the most effective solutions to prevent and mitigate stress in HCWs. Balint and support groups as part of the institutional infrastructure demonstrate an institutional, not individual response, to stress. In support of this notion, Balint-like groups as a low-cost model have shown effectiveness in attenuating stress through mechanisms like building connections, validating HCWs’ experiences, and gaining an accurate understanding how colleagues navigate these challenges [231]. Although many HCWs may think they lack the time to engage in supervision, Balint and support groups, seeking help and having conversations with others may help them to achieve different perspectives on morally-distressing situations, which in turn might reduce their overall stress levels.
In addition, narrative medicine practices may lessen feelings of distress and prevent moral injury. Through the training of narrative competence, including reflective writing, close reading, telling their stories, and listening to the narratives of others, HCWs can strengthen the commitment to their own health and health behaviors, improve empathy and care towards themselves and their colleagues, and experience improved self-awareness and increased work satisfaction [232]. By bridging emotions, vulnerability, and human connection with medical practice, narrative approaches can help HCWs offer accurate, engaged, authentic, compassionate, and effective patient care [197], while at the same time reducing emotional exhaustion and burnout [195,196]. We believe that reintegrating the perspectives of humanities and narrative medicine into clinical practice, could greatly improve the healthcare environment, which currently is driven by economization and rationalization as its central values, often at the expense of patient care, quality of care, and patient safety.
One limitation we encountered while writing this review was the inherent ambiguity in the terminology associated with occupational stress and moral distress, making it challenging to differentiate between these concepts. Moral distress could be viewed as a subcomponent within the broader realm of occupational stress, highlighting the need for further clarification. Indeed, a shift in “framing” moral distress away from an individual problem and towards framing as an occupational hazard may result in novel ways of thinking about its reduction and mitigation.
From our perspective, the pandemic has given rise to a unique phenomenon where, despite HCWs knowing what to do in the moment, and even being able to perform what they perceive as the right action, they may still experience stress due to the context surrounding their decisions. We believe that a reconceptualization of moral distress in HCWs is essential; one that can provide a framework for addressing the complex social and structural factors contributing to the current healthcare crisis. The concept of entrapment stressors [233,234], which refers to working situations where individuals feel unable to escape from distressing circumstances, often characterized by a perceived lack of control and a feeling of hopelessness, may offer an alternative perspective on moral distress in HCWs in the post-pandemic transformation. This conceptualization could open up new avenues to explore public health responses to improve the health and well-being of HCWs and address preventable healthcare harm.
Today, the main triggers for occupational stress in HCWs originate from organizational and system-level issues (e.g., excessive work load demands; poor staffing; poor workplace environment, misalignment between individual, team and organizational goals etc.) [235,236]. To achieve systemic changes within healthcare, it will be important that leaders are aware of the impact of occupational stressors on the immune system and health. Systemic changes can only be implemented if leaders hold themselves accountable to ensure justice in our institutions and advocate for policies and resources that support a healthy workforce. Lastly, only when HCWs are valued and acknowledged by their leaders and the healthcare system, they can provide high-quality patient care and stay mentally and physically healthy [237]. Given the considerable contribution of HCWs to our healthcare system and patient-care, healthcare systems have an ethical obligation to take care of their employees’ health and create healthy, safe, and sustainable working environments for them.
11. Clinical implications and avenues for future research
The adaptation of the J-DR health model for HCWs is a valuable tool for analyzing and identifying existing job demands and job resources. This facilitates the development and implementation of public health policies and targeted mental health interventions to improve the work environment, ensure the safety, health, and well-being of HCWs and better protect them from the negative effects of stress. Furthermore, the J-DR health model can serve as a valuable framework for initiating an ethics quality improvement project withing a hospital system. By employing this model, healthcare institutions can strategically address occupational stressors, fostering a work environment that prioritizes both the well-being of HCWs and the quality of patient care.
Furthermore, we suggest a testable model linking moral distress in HCWs to immune dysregulation (Fig. 2). In fact, the impact of moral distress on immunity and health in HCWs and its underlying biological mechanisms remain poorly investigated. Longitudinal, large-scale studies are warranted that include validated psychometric instruments to screen for moral distress, anxiety, depression, and burnout in combination with biological stress markers (e.g., CRP, serum level of IL-6; TNF-α) to better illustrate the prevalence and severity of moral distress in HCWs and its implications over time for psychoneuroendocrine and psychoneuroimmune outcomes.
Immune and, more specifically, inflammatory markers of stress may be important for identifying HCWs at higher risk for adverse health outcomes in response to stress, thereby providing an opportunity for early intervention. Because gender plays a critical role in cytokine expression, future studies on moral distress and biological stress markers should also account for gender differences. Furthermore, using validated and reliable instruments to assess moral distress in research is critical. Recently, Epstein and colleagues [39] revised the Moral Distress Scale (MDS-R) and now recommend using the Measure of Moral Distress for Health Care Professionals (MMD-HP) to assess the impact of moral distress on healthcare professionals. Lastly, from a public health ethics perspective, it will be equally important to collect data regarding HCW disability, job turnover, and healthcare costs, as well as to track the career trajectories of those HCWs who leave their healthcare positions due to moral distress.
12. Conclusion
In conclusion, occupational stress is prevalent in HCWs, leading to severe mental and physical health issues, compromised patient care, and high attrition rates. Our review has explored neuroendocrine, immune, genetic, and epigenetic mechanisms of occupational stressors in HCWs, offering insights into how moral distress can affect HCWs' immune system and health. Investigating the immunological impact of moral distress in HCWs is a promising area of study, given the public health implications of stress in this population. This review underscores the imperative for healthcare systems to prioritize HCWs’ mental health and invest in burnout prevention. We advocate for a comprehensive approach to prevent and mitigate stress and its adverse health effects on HCWs. Such interventions should encompass both organizational and structural strategies, such as the creation of reflective spaces, along with individual-level interventions like counseling programs or stress-reduction initiatives. Target high-risk groups, including trainees, early career professionals, women, frontline HCWs (especially nurses), and those in high-stress areas like emergency departments, intensive care units, and palliative care, is crucial. Lastly, we must collectively take responsibility for addressing the root causes of occupational stressors and burnout in our institutions. Upholding moral standards and fostering supportive environment where HCWs feel valued and acknowledged are essential for enabling them to carry out the much-needed work for our patients.
Funding
G.M.S. was supported by grant #OPR21101 from the California Governor's Office of Planning and Research/California Initiative to Advance Precision Medicine. These organizations had no role in planning, writing, editing, or reviewing this article, or in deciding to submit this article for publication.
CRediT authorship contribution statement
Annina Seiler: Conceptualization, Writing – original draft. Aimee Milliken: Conceptualization, Supervision, Writing – review & editing. Richard E. Leiter: Conceptualization, Supervision, Writing – review & editing. David Blum: Writing – review & editing. George M. Slavich: Conceptualization, Supervision, Writing – review & editing.
Declaration of competing interest
The authors declare no conflicts of interest with respect to this work.
Acknowledgments
We thank Scientific Visualization and Visual Communication at the University of Zurich, Switzerland (SIVIC) for helping create the graphics contained in this article.
References
- 1.King Lucy, Kessel Jonah M. New York Times. 2022. We know the real cause of the crisis in our hospitals. It's greed. January 19. [Google Scholar]
- 2.Berlin G., Lapointe M., Murphy M. McKinsey & Company; 2023. Nursing in 2023: How Hospitals Are Confronting Shortages. [Google Scholar]
- 3.McKinsey, Company . 2022. Surveyed Nurses Consider Leaving Direct Patient Care at Elevated Rates.https://www.mckinsey.com/industries/healthcare-systems-and-services/our-insights/surveyed-nurses-consider-leaving-direct-patient-care-at-elevated-rates [Google Scholar]
- 4.Sachin Ravikumar. 2022. Britain's Health Service Facing Worst Staffing Crisis, Say Lawmakers.https://www.reuters.com/world/uk/britains-health-service-facing-worst-staffing-crisis-say-lawmakers-2022-07-25/ Retrieved 08/16/2022, from REUTERS. [Google Scholar]
- 5.Antonsdottir I., Rushton C.H., Nelson K.E., Heinze K.E., Swoboda S.M., Hanson G.C. Burnout and moral resilience in interdisciplinary healthcare professionals. J. Clin. Nurs. 2022;31(1–2):196–208. doi: 10.1111/jocn.15896. [DOI] [PubMed] [Google Scholar]
- 6.Johnson-Coyle L., Opgenorth D., Bellows M., Dhaliwal J., Richardson-Carr S., Bagshaw S.M. Moral distress and burnout among cardiovascular surgery intensive care unit healthcare professionals: a prospective cross-sectional survey. Can J Crit Care Nurs. 2016;27(4):27–36. [PubMed] [Google Scholar]
- 7.Kok Niek, Van Gurp Jelle, van der Hoeven Johannes G., Fuchs Malaika, Cornelia Hoedemaekers, Marieke Zegers. Complex interplay between moral distress and other risk factors of burnout in ICU professionals: findings from a cross-sectional survey study. BMJ Qual. Saf. 2021 doi: 10.1136/bmjqs-2020-012239. bmjqs-2020-012239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Moss M., Good V.S., Gozal D., Kleinpell R., Sessler C.N. An official critical care societies collaborative statement-burnout syndrome in critical care health-care professionals: a call for action. Chest. 2016;150(1):17–26. doi: 10.1016/j.chest.2016.02.649. [DOI] [PubMed] [Google Scholar]
- 9.Petrișor C., Breazu C., Doroftei M., Mărieș I., Popescu C. Association of moral distress with anxiety, depression, and an intention to leave among nurses working in intensive care units during the COVID-19 pandemic. Healthcare. 2021;9(10) doi: 10.3390/healthcare9101377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Moss M., Good V.S., Gozal D., Kleinpell R., Sessler C.N. An official critical care societies collaborative statement: burnout syndrome in critical care healthcare professionals: a call for action. Crit. Care Med. 2016;44(7):1414–1421. doi: 10.1097/ccm.0000000000001885. [DOI] [PubMed] [Google Scholar]
- 11.Hamric A.B., Blackhall L.J. Nurse-physician perspectives on the care of dying patients in intensive care units: collaboration, moral distress, and ethical climate. Crit. Care Med. 2007;35(2):422–429. doi: 10.1097/01.Ccm.0000254722.50608.2d. [DOI] [PubMed] [Google Scholar]
- 12.Austin C.L., Saylor R., Finley P.J. Moral distress in physicians and nurses: impact on professional quality of life and turnover. Psychol Trauma. 2017;9(4):399–406. doi: 10.1037/tra0000201. [DOI] [PubMed] [Google Scholar]
- 13.Vahey D.C., Aiken L.H., Sloane D.M., Clarke S.P., Vargas D. Nurse burnout and patient satisfaction. Med. Care. 2004;42(2 Suppl):Ii57–66. doi: 10.1097/01.mlr.0000109126.50398.5a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Beck Emily, Jo Daniels. Intolerance of uncertainty, fear of contamination and perceived social support as predictors of psychological distress in NHS healthcare workers during the COVID-19 pandemic. Psychol. Health Med. 2022:1–13. doi: 10.1080/13548506.2022.2092762. [DOI] [PubMed] [Google Scholar]
- 15.Newman K.L., Jeve Y., Majumder P. Experiences and emotional strain of NHS frontline workers during the peak of the COVID-19 pandemic. Int. J. Soc. Psychiatr. 2022;68(4):783–790. doi: 10.1177/00207640211006153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.(WHO) World Health Organization . World Health Organization (WHO); 2022. Global Strategy on Human Resources for Health: Workforce 2030: Reporting at Seventy-Fifth World Health Assembly. (WHA 75.17)https://www.who.int/news/item/02-06-2022-global-strategy-on-human-resources-for-health--workforce-2030 Retrieved 08/15/2022. [Google Scholar]
- 17.Michael Sainato. The Guardian. 2023. Over 75,000 workers poised for largest healthcare strike in US history. Kaiser Permanente workers push for improvements to staffing levels and wage increases after union contract expires. [Google Scholar]
- 18.El Haj Alicia J. The grand challenges of medical technology. [Field grand challenge] Frontiers in Medical Technology. 2020;2 doi: 10.3389/fmedt.2020.00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bernadette Melnyk. Burnout, depression and suicide in nurses/clinicians and learners: an urgent call for action to enhance professional well-being and healthcare safety. Worldviews Evidence-Based Nurs. 2020;17:2–5. doi: 10.1111/wvn.12416. [DOI] [PubMed] [Google Scholar]
- 20.Kislov R., Harvey G., Jones L. Boundary organising in healthcare: theoretical perspectives, empirical insights and future prospects. J. Health Organisat. Manag. 2021;35(2):133–140. doi: 10.1108/JHOM-04-2021-475. [DOI] [Google Scholar]
- 21.Cutler David M. Challenges for the beleaguered health care workforce during COVID-19. JAMA Health Forum. 2022;3(1) doi: 10.1001/jamahealthforum.2022.0143. [DOI] [PubMed] [Google Scholar]
- 22.Linton M., Koonmen J. Self-care as an ethical obligation for nurses. Nurs. Ethics. 2020;969733020940371 doi: 10.1177/0969733020940371. [DOI] [PubMed] [Google Scholar]
- 23.Epstein E.G., Hamric A.B. Moral distress, moral residue, and the crescendo effect. J. Clin. Ethics. 2009;20(4):330–342. [PubMed] [Google Scholar]
- 24.Georgina Morley, Jonathan Ives, Caroline Bradbury-Jones. Moral distress and austerity: an avoidable ethical challenge in healthcare. Health Care Anal. 2019;27(3):185–201. doi: 10.1007/s10728-019-00376-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ruta Karaliuniene, Sachin Nagendrappa, Chonnakarn Jatchavala, Isioma Ojeahere Margaret, Irfan Ullah, Gashi Bytyçi Drita, Peter Ogunnubi Oluseun, Michele Cherro, Mohammadreza Shalbafan, Camille Noël, Gonzalez-Diaz Jairo M., Ramya Vadivel, Pereira-Sanchez Victor, Mohammad Abu Slaih, Sheikh Shoib, de Filippis Renato Support the frontliners – good initiatives during the COVID-19 pandemic for healthcare workers across the world: is this what we really need? BJPsych Int. 2022:1–4. doi: 10.1192/bji.2022.6. [DOI] [Google Scholar]
- 26.Lake Eileen T., Narva Aliza M., Holland Sara, Smith Jessica G., Cramer Emily, Fitzpatrick Rosenbaum Kathleen E., French Rachel, Clark Rebecca R.S., Rogowski Jeannette A. Hospital nurses' moral distress and mental health during COVID-19. J. Adv. Nurs. 2022;78(3):799–809. doi: 10.1111/jan.15013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Norman S.B., Feingold J.H., Kaye-Kauderer H., Kaplan C.A., Hurtado A., Kachadourian L., Feder A., Murrough J.W., Charney D., Southwick S.M., Ripp J., Peccoralo L., Pietrzak R.H. Moral distress in frontline healthcare workers in the initial epicenter of the COVID-19 pandemic in the United States: relationship to PTSD symptoms, burnout, and psychosocial functioning. Depress. Anxiety. 2021;38(10):1007–1017. doi: 10.1002/da.23205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sonis J., Pathman D.E., Read S., Gaynes B.N. A national study of moral distress among U.S. internal medicine physicians during the COVID-19 pandemic. PLoS One. 2022;17(5) doi: 10.1371/journal.pone.0268375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Whitehead P.B., Herbertson R.K., Hamric A.B., Epstein E.G., Fisher J.M. Moral distress among healthcare professionals: report of an institution-wide survey. J. Nurs. Scholarsh. 2015;47(2):117–125. doi: 10.1111/jnu.12115. [DOI] [PubMed] [Google Scholar]
- 30.Alexandra Hootnick. August 12, 2021. Nurses Deserve Better. So Do Their Patients. (New York Times) [Google Scholar]
- 31.Xu Minzhi, Yin Xiaoxv, Gong Yanhong. Lifestyle factors in the association of shift work and depression and anxiety. JAMA Netw. Open. 2023;6(8) doi: 10.1001/jamanetworkopen.2023.28798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Brauchli R., Jenny G.J., Füllemann D., Bauer G.F. Towards a job demands-resources health model: empirical testing with generalizable indicators of job demands, job resources, and comprehensive health outcomes. BioMed Res. Int. 2015;2015 doi: 10.1155/2015/959621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Epstein Elizabeth G., Delgado Sarah A. Understanding and addressing moral distress. Online J. Issues Nurs. 2010;15(3) doi: 10.3912/OJIN.Vol15No03Man01. [DOI] [Google Scholar]
- 34.Jameton A. Dilemmas of moral distress: moral responsibility and nursing practice. AWHONNS Clin. Issues Perinat. Womens Health Nurs. 1993;4(4):542–551. [PubMed] [Google Scholar]
- 35.St Ledger U., Reid J., Begley A., Dodek P., McAuley D.F., Prior L., Blackwood B. Moral distress in end-of-life decisions: a qualitative study of intensive care physicians. J. Crit. Care. 2021;62:185–189. doi: 10.1016/j.jcrc.2020.12.019. [DOI] [PubMed] [Google Scholar]
- 36.Giulia Lamiani, Lidia Borghi, Piergiorgio Argentero. When healthcare professionals cannot do the right thing: a systematic review of moral distress and its correlates. J. Health Psychol. 2015;22(1):51–67. doi: 10.1177/1359105315595120. [DOI] [PubMed] [Google Scholar]
- 37.Carnevale F.A. Moral distress in the ICU: it's time to do something about it. Minerva Anestesiol. 2020;86(4):455–460. doi: 10.23736/s0375-9393.19.14021-7. [DOI] [PubMed] [Google Scholar]
- 38.Mealer M., Jones J. Posttraumatic stress disorder in the nursing population: a concept analysis. Nurs. Forum. 2013;48(4):279–288. doi: 10.1111/nuf.12045. [DOI] [PubMed] [Google Scholar]
- 39.Epstein E.G., Whitehead P.B., Prompahakul C., Thacker L.R., Hamric A.B. Enhancing understanding of moral distress: the measure of moral distress for health care professionals. AJOB Empir Bioeth. 2019;10(2):113–124. doi: 10.1080/23294515.2019.1586008. [DOI] [PubMed] [Google Scholar]
- 40.Milliken A., Courtwright A., Grace P., Eagan-Bengston E., Visser M., Jurchak M. Ethics consultations at a major academic medical center: a retrospective, longitudinal analysis. AJOB Empir Bioeth. 2020;11(4):275–286. doi: 10.1080/23294515.2020.1818879. [DOI] [PubMed] [Google Scholar]
- 41.Liu X., Xu Y., Chen Y., Chen C., Wu Q., Xu H., Zhu P., Waidley E. Ethical dilemmas faced by frontline support nurses fighting COVID-19. Nurs. Ethics. 2022;29(1):7–18. doi: 10.1177/09697330211015284. [DOI] [PubMed] [Google Scholar]
- 42.Aiken L.H., Sloane D.M., Bruyneel L., Van den Heede K., Griffiths P., Busse R., Diomidous M., Kinnunen J., Kózka M., Lesaffre E., McHugh M.D., Moreno-Casbas M.T., Rafferty A.M., Schwendimann R., Scott P.A., Tishelman C., van Achterberg T., Sermeus W. Nurse staffing and education and hospital mortality in nine European countries: a retrospective observational study. Lancet. 2014;383(9931):1824–1830. doi: 10.1016/s0140-6736(13)62631-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tawfik Daniel S., Profit Jochen, Morgenthaler Timothy I., Satele Daniel V., Sinsky Christine A., Dyrbye Liselotte N., Tutty Michael A., West Colin P., Shanafelt Tait D. Physician burnout, well-being, and work unit safety grades in relationship to reported medical errors. Mayo Clin. Proc. 2018;93(11):1571–1580. doi: 10.1016/j.mayocp.2018.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Yue Li, Temkin-Greener Helena, Gao Shan, Cai Xueya. COVID-19 infections and deaths among Connecticut nursing home residents: facility correlates. J. Am. Geriatr. Soc. 2020;68(9):1899–1906. doi: 10.1111/jgs.16689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Schaufeli Wilmar B., Bakker Arnold B. Job demands, job resources, and their relationship with burnout and engagement: a multi-sample study. J. Organ. Behav. 2004;25(3):293–315. doi: 10.1002/job.248. [DOI] [Google Scholar]
- 46.Hofmann D.A., Burke M.J., Zohar D. 100 years of occupational safety research: from basic protections and work analysis to a multilevel view of workplace safety and risk. J. Appl. Psychol. 2017;102(3):375–388. doi: 10.1037/apl0000114. [DOI] [PubMed] [Google Scholar]
- 47.Michaels D., Wagner G.R., Ryan L. Lessons from COVID-19 for protecting workers in the next pandemic. JAMA. 2023;330(1):23–24. doi: 10.1001/jama.2023.8229. [DOI] [PubMed] [Google Scholar]
- 48.Peters Susan E., Dennerlein Jack T., Wagner Gregory R., Sorensen Glorian. Work and worker health in the post-pandemic world: a public health perspective. Lancet Public Health. 2022;7(2):e188–e194. doi: 10.1016/S2468-2667(21)00259-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Williams Jessica A. R., Collins Jamie E., Gandhi Ashvin, Yu Huizi, Boden Leslie I., Katz Jeffrey N., Wagner Gregory R., Sorensen Glorian Can better leadership reduce nursing home staff turnover? J. Am. Med. Dir. Assoc.. doi: 10.1016/j.jamda.2023.05.021. [DOI] [PMC free article] [PubMed]
- 50.Karasek Robert A. Job demands, job decision latitude, and mental strain: implications for job redesign. Adm. Sci. Q. 1979;24(2):285–308. doi: 10.2307/2392498. [DOI] [Google Scholar]
- 51.Bakker A.B., Demerouti E. The job demands-resources model: state of the art. J. Manag. Psychol. 2007;22:309–328. doi: 10.1108/02683940710733115. [DOI] [Google Scholar]
- 52.1981). Health, stress and coping. By A. Antonovsky. Jossey-Bass: London. 1979. Psychological Medicine, vol. 11(1), 206-206. doi: 10.1017/S003329170005371X.
- 53.Jari Hakanen, Evangelia Demerouti, Despoina Xanthopoulou. Job resources boost work engagement, particularly when job demands are high. J. Educ. Psychol. 2007;99 doi: 10.1037/0022-0663.99.2.274. [DOI] [Google Scholar]
- 54.Bakker Arnold B., van Veldhoven Marc, Despoina Xanthopoulou. Beyond the demand-control model. J. Person. Psychol. 2010;9(1):3–16. doi: 10.1027/1866-5888/a000006. [DOI] [Google Scholar]
- 55.Lazarus R.S., Folkman S. Springer; New York, NY: 1984. Stress, Appraisal, and Coping. [Google Scholar]
- 56.Rotenstein Lisa S., Matthew Torre, Ramos Marco A., Rosales Rachael C., Guille Constance, Sen Srijan, Mata Douglas A. Prevalence of burnout among physicians: a systematic review. JAMA. 2018;320(11):1131–1150. doi: 10.1001/jama.2018.12777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Holly Wei, Julia Aucoin, Kuntapay Gabrielle R., Amber Justice, Jones Abigail, Zhang Chongben, Santos Hudson P., Jr., Hall Lynne A. The prevalence of nurse burnout and its association with telomere length pre and during the COVID-19 pandemic. PLoS One. 2022;17(3) doi: 10.1371/journal.pone.0263603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Maharaj S., Lees T., Lal S. Prevalence and risk factors of depression, anxiety, and stress in a cohort of Australian nurses. Int. J. Environ. Res. Publ. Health. 2018;16(1) doi: 10.3390/ijerph16010061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.World Health Organization (WHO) World Health Organization (WHO); 2021. Depression.https://www.who.int/news-room/fact-sheets/detail/depression Retrieved 09/17/2022. [Google Scholar]
- 60.West C.P., Dyrbye L.N., Shanafelt T.D. Physician burnout: contributors, consequences and solutions. J. Intern. Med. 2018;283(6):516–529. doi: 10.1111/joim.12752. [DOI] [PubMed] [Google Scholar]
- 61.von Känel R., Princip M., Holzgang S.A., Fuchs W.J., van Nuffel M., Pazhenkottil A.P., Spiller T.R. Relationship between job burnout and somatic diseases: a network analysis. Sci. Rep. 2020;10(1) doi: 10.1038/s41598-020-75611-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.von Känel R., Princip M., Holzgang S.A., Garefa C., Rossi A., Benz D.C., Giannopoulos A.A., Kaufmann P.A., Buechel R.R., Zuccarella-Hackl C., Pazhenkottil A.P. Coronary microvascular function in male physicians with burnout and job stress: an observational study. BMC Med. 2023;21(1):477. doi: 10.1186/s12916-023-03192-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Menon Nikitha K., Shanafelt Tait D., Sinsky Christine A., Mark Linzer, Lindsey Carlasare, Brady Keri J.S., Stillman Martin J., Trockel Mickey T. Association of physician burnout with suicidal ideation and medical errors. JAMA Netw. Open. 2020;3(12) doi: 10.1001/jamanetworkopen.2020.28780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Davis Matthew A., Cher Benjamin A.Y., Friese Christopher R., Bynum Julie P.W. Association of US nurse and physician occupation with risk of suicide. JAMA Psychiatr. 2021;78(6):651–658. doi: 10.1001/jamapsychiatry.2021.0154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Pereira-Lima K., Loureiro S.R. Burnout, anxiety, depression, and social skills in medical residents. Psychol. Health Med. 2015;20(3):353–362. doi: 10.1080/13548506.2014.936889. [DOI] [PubMed] [Google Scholar]
- 66.González-Cabrera J.M., Fernández-Prada M., Iribar C., Molina-Ruano R., Salinero-Bachiller M., Peinado J.M. Acute stress and anxiety in medical residents on the emergency department duty. Int. J. Environ. Res. Publ. Health. 2018;15(3) doi: 10.3390/ijerph15030506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Mata D.A., Ramos M.A., Bansal N., Khan R., Guille C., Di Angelantonio E., Sen S. Prevalence of depression and depressive symptoms among resident physicians: a systematic review and meta-analysis. JAMA. 2015;314(22):2373–2383. doi: 10.1001/jama.2015.15845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Arica Brandford, Reed Deborah. Depression in registered nurses: a state of the science. Workplace Health & Saf. 2016;64:488–511. doi: 10.1177/2165079916653415. [DOI] [PubMed] [Google Scholar]
- 69.Rotenstein Lisa S., Ramos Marco A., Matthew Torre, Bradley Segal J., Peluso Michael J., Guille Constance, Sen Srijan, Mata Douglas A. Prevalence of depression, depressive symptoms, and suicidal ideation among medical students: a systematic review and meta-analysis. JAMA. 2016;316(21):2214–2236. doi: 10.1001/jama.2016.17324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tait Shanafelt, Goh Joel, Christine Sinsky. The business case for investing in physician well-being. JAMA Intern. Med. 2017;177(12):1826–1832. doi: 10.1001/jamainternmed.2017.4340. [DOI] [PubMed] [Google Scholar]
- 71.da Silva F.C.T., Neto M.L.R. Psychiatric symptomatology associated with depression, anxiety, distress, and insomnia in health professionals working in patients affected by COVID-19: a systematic review with meta-analysis. Prog. Neuro-Psychopharmacol. Biol. Psychiatry. 2021;104 doi: 10.1016/j.pnpbp.2020.110057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Sofia Pappa, Vasiliki Ntella, Timoleon Giannakas, Giannakoulis Vassilis G., Eleni Papoutsi, Paraskevi Katsaounou. Prevalence of depression, anxiety, and insomnia among healthcare workers during the COVID-19 pandemic: a systematic review and meta-analysis. Brain Behav. Immun. 2020;88:901–907. doi: 10.1016/j.bbi.2020.05.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lightman S.L., Conway-Campbell B.L. The crucial role of pulsatile activity of the HPA axis for continuous dynamic equilibration. Nat. Rev. Neurosci. 2010;11(10):710–718. doi: 10.1038/nrn2914. [DOI] [PubMed] [Google Scholar]
- 74.Bellavance M.A., Rivest S. The HPA - immune Axis and the immunomodulatory actions of glucocorticoids in the brain. Front. Immunol. 2014;5:136. doi: 10.3389/fimmu.2014.00136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Smoak K.A., Cidlowski J.A. Mechanisms of glucocorticoid receptor signaling during inflammation. Mech. Ageing Dev. 2004;125(10–11):697–706. doi: 10.1016/j.mad.2004.06.010. [DOI] [PubMed] [Google Scholar]
- 76.Amaya J.M., Suidgeest E., Sahut-Barnola I., Dumontet T., Montanier N., Pagès G., Keller C., van der Weerd L., Pereira A.M., Martinez A., Meijer O.C. Effects of long-term endogenous corticosteroid exposure on brain volume and glial cells in the AdKO mouse. Front. Neurosci. 2021;15 doi: 10.3389/fnins.2021.604103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Glaser R., Kiecolt-Glaser J.K. Stress-induced immune dysfunction: implications for health. Nat. Rev. Immunol. 2005;5(3):243–251. doi: 10.1038/nri1571. [DOI] [PubMed] [Google Scholar]
- 78.O'Donovan A., Hughes B.M., Slavich G.M., Lynch L., Cronin M.T., O'Farrelly C., Malone K.M. Clinical anxiety, cortisol and interleukin-6: evidence for specificity in emotion-biology relationships. Brain Behav. Immun. 2010;24(7):1074–1077. doi: 10.1016/j.bbi.2010.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.O'Donovan A., Slavich G.M., Epel E.S., Neylan T.C. Exaggerated neurobiological sensitivity to threat as a mechanism linking anxiety with increased risk for diseases of aging. Neurosci. Biobehav. Rev. 2013;37(1):96–108. doi: 10.1016/j.neubiorev.2012.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Ferri P., Guadi M., Marcheselli L., Balduzzi S., Magnani D., Di Lorenzo R. The impact of shift work on the psychological and physical health of nurses in a general hospital: a comparison between rotating night shifts and day shifts. Risk Manag. Healthc. Pol. 2016;9:203–211. doi: 10.2147/rmhp.S115326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hellhammer D.H., Wüst S., Kudielka B.M. Salivary cortisol as a biomarker in stress research. Psychoneuroendocrinology. 2009;34(2):163–171. doi: 10.1016/j.psyneuen.2008.10.026. [DOI] [PubMed] [Google Scholar]
- 82.Brand M.C., Shippey H., Hagan J., Hanneman S.K., Levy B., Range S., Wongsuwan N., Zodin A., Walden M. Comparison of psychological and physiological stress in NICU nurses: effects of unit design and shift. Adv. Neonatal Care. 2021;21(4):E93–e100. doi: 10.1097/anc.0000000000000837. [DOI] [PubMed] [Google Scholar]
- 83.Minelli A., Di Palma M., Rocchi M.B.L., Ponzio E., Barbadoro P., Bracci M., Pelusi G., Prospero E. Cortisol, chronotype, and coping styles as determinants of tolerance of nursing staff to rotating shift work. Chronobiol. Int. 2021;38(5):666–680. doi: 10.1080/07420528.2021.1887883. [DOI] [PubMed] [Google Scholar]
- 84.Osterode W., Schranz S., Jordakieva G. Effects of night shift on the cognitive load of physicians and urinary steroid hormone profiles - a randomized crossover trial. Chronobiol. Int. 2018;35(7):946–958. doi: 10.1080/07420528.2018.1443942. [DOI] [PubMed] [Google Scholar]
- 85.Wong I.S., Ostry A.S., Demers P.A., Davies H.W. Job strain and shift work influences on biomarkers and subclinical heart disease indicators: a pilot study. J. Occup. Environ. Hyg. 2012;9(8):467–477. doi: 10.1080/15459624.2012.693831. [DOI] [PubMed] [Google Scholar]
- 86.Russell Evan, Gideon Koren, Rieder Michael, Van Uum Stan Hair cortisol as a biological marker of chronic stress: current status, future directions and unanswered questions. Psychoneuroendocrinology. 2012;37(5):589–601. doi: 10.1016/j.psyneuen.2011.09.009. [DOI] [PubMed] [Google Scholar]
- 87.Behnke A., Karabatsiakis A., Krumbholz A., Karrasch S., Schelling G., Kolassa I.T., Rojas R. Associating Emergency Medical Services personnel's workload, trauma exposure, and health with the cortisol, endocannabinoid, and N-acylethanolamine concentrations in their hair. Sci. Rep. 2020;10(1) doi: 10.1038/s41598-020-79859-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Fernández-Sánchez J.C., Pérez-Mármol J.M., Blásquez A., Santos-Ruiz A.M., Peralta-Ramírez M.I. Association between burnout and cortisol secretion, perceived stress, and psychopathology in palliative care unit health professionals. Palliat. Support Care. 2018;16(3):286–297. doi: 10.1017/s1478951517000244. [DOI] [PubMed] [Google Scholar]
- 89.Daglius Dias R., Scalabrini Neto A. Stress levels during emergency care: a comparison between reality and simulated scenarios. J. Crit. Care. 2016;33:8–13. doi: 10.1016/j.jcrc.2016.02.010. [DOI] [PubMed] [Google Scholar]
- 90.Valent F., Mariuz M., Liva G., Verri S., Arlandini S., Vivoli R. Salivary melatonin and cortisol and occupational injuries among Italian hospital workers. Neurol. Sci. 2016;37(10):1613–1620. doi: 10.1007/s10072-016-2630-x. [DOI] [PubMed] [Google Scholar]
- 91.Nieto Fernández-Pacheco A., Castro Delgado R., Arcos González P., Navarro Fernández J.L., Cerón Madrigal J.J., Juguera Rodriguez L., Perez Alonso N., Armero-Barranco D., Lidon López Iborra M., Damian E.T., Pardo Rios M. Analysis of performance and stress caused by a simulation of a mass casualty incident. Nurse Educ. Today. 2018;62:52–57. doi: 10.1016/j.nedt.2017.12.016. [DOI] [PubMed] [Google Scholar]
- 92.Ibar C., Fortuna F., Gonzalez D., Jamardo J., Jacobsen D., Pugliese L., Giraudo L., Ceres V., Mendoza C., Repetto E.M., Reboredo G., Iglesias S., Azzara S., Berg G., Zopatti D., Fabre B. Evaluation of stress, burnout and hair cortisol levels in health workers at a University Hospital during COVID-19 pandemic. Psychoneuroendocrinology. 2021;128 doi: 10.1016/j.psyneuen.2021.105213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Marcil M.J., Cyr S., Marin M.F., Rosa C., Tardif J.C., Guay S., Guertin M.C., Genest C., Forest J., Lavoie P., Labrosse M., Vadeboncoeur A., Selcer S., Ducharme S., Brouillette J. Hair cortisol change at COVID-19 pandemic onset predicts burnout among health personnel. Psychoneuroendocrinology. 2022;138 doi: 10.1016/j.psyneuen.2021.105645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Rajcani J., Vytykacova S., Solarikova P., Brezina I. Stress and hair cortisol concentrations in nurses during the first wave of the COVID-19 pandemic. Psychoneuroendocrinology. 2021;129 doi: 10.1016/j.psyneuen.2021.105245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Prather A.A., Carroll J.E., Fury J.M., McDade K.K., Ross D., Marsland A.L. Gender differences in stimulated cytokine production following acute psychological stress. Brain Behav. Immun. 2009;23(5):622–628. doi: 10.1016/j.bbi.2008.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.The U.S. Bureau of Labor Statistics (BLS) The U.S. Bureau of Labor Statistics (BLS); 2022. Over 16 Million Women Worked in Health Care and Social Assistance in 2021.https://www.bls.gov/opub/ted/2022/over-16-million-women-worked-in-health-care-and-social-assistance-in-2021.htm Retrieved 09/18/2022. [Google Scholar]
- 97.Kendler K.S., Gardner C.O., Prescott C.A. Toward a comprehensive developmental model for major depression in women. Am. J. Psychiatr. 2002;159(7):1133–1145. doi: 10.1176/appi.ajp.159.7.1133. [DOI] [PubMed] [Google Scholar]
- 98.Bani-Issa W., Radwan H., Al Marzooq F., Al Awar S., Al-Shujairi A.M., Samsudin A.R., Khasawneh W., Albluwi N. Salivary cortisol, subjective stress and quality of sleep among female healthcare professionals. J. Multidiscip. Healthc. 2020;13:125–140. doi: 10.2147/jmdh.S229396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Shelly Lachish, Elena Svirko, Goldacre Michael, Lambert Trevor. Factors associated with less-than-full-time working in medical practice: results of surveys of five cohorts of UK doctors, 10 years after graduation. Hum. Resour. Health. 2016;14 doi: 10.1186/s12960-016-0162-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Nicole DePasquale, Polenick Courtney A., Jesse Hinde, Bray Jeremy W., Zarit Steven H., Phyllis Moen, Hammer Leslie B., Almeida David M. Health behavior among men with multiple family roles: the moderating effects of perceived partner relationship quality. Am. J. Men's Health. 2016;12(6):2006–2017. doi: 10.1177/1557988316660088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Bergman B., Ahmad F., Stewart D.E. Work family balance, stress, and salivary cortisol in men and women academic physicians. Int. J. Behav. Med. 2008;15(1):54–61. doi: 10.1007/bf03003074. [DOI] [PubMed] [Google Scholar]
- 102.Dich N., Lange T., Head J., Rod N.H. Work stress, caregiving, and allostatic load: prospective results from the Whitehall II cohort study. Psychosom. Med. 2015;77(5):539–547. doi: 10.1097/psy.0000000000000191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Honarvar B., Ghazanfari N., Raeisi Shahraki H., Rostami S., Lankarani K.B. Violence against nurses: a neglected and health-threatening epidemic in the university affiliated public hospitals in Shiraz, Iran. Int. J. Occup. Environ. Med. 2019;10(3):111–123. doi: 10.15171/ijoem.2019.1556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Kind N., Eckert A., Steinlin C., Fegert J.M., Schmid M. Verbal and physical client aggression - a longitudinal analysis of professional caregivers' psychophysiological stress response and burnout. Psychoneuroendocrinology. 2018;94:11–16. doi: 10.1016/j.psyneuen.2018.05.001. [DOI] [PubMed] [Google Scholar]
- 105.Sanders V.M. The beta2-adrenergic receptor on T and B lymphocytes: do we understand it yet? Brain Behav. Immun. 2012;26(2):195–200. doi: 10.1016/j.bbi.2011.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Irwin M.R., Cole S.W. Reciprocal regulation of the neural and innate immune systems. Nat. Rev. Immunol. 2011;11(9):625–632. doi: 10.1038/nri3042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Miller A.H., Maletic V., Raison C.L. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol. Psychiatr. 2009;65(9):732–741. doi: 10.1016/j.biopsych.2008.11.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Sun Z., Hou D., Liu S., Fu W., Wang J., Liang Z. Norepinephrine inhibits the cytotoxicity of NK92-MI cells via the β2-adrenoceptor/cAMP/PKA/p-CREB signaling pathway. Mol. Med. Rep. 2018;17(6):8530–8535. doi: 10.3892/mmr.2018.8872. [DOI] [PubMed] [Google Scholar]
- 109.Witek-Janusek L., Gabram S., Mathews H.L. Psychologic stress, reduced NK cell activity, and cytokine dysregulation in women experiencing diagnostic breast biopsy. Psychoneuroendocrinology. 2007;32(1):22–35. doi: 10.1016/j.psyneuen.2006.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Hara M.R., Kovacs J.J., Whalen E.J., Rajagopal S., Strachan R.T., Grant W., Towers A.J., Williams B., Lam C.M., Xiao K., Shenoy S.K., Gregory S.G., Ahn S., Duckett D.R., Lefkowitz R.J. A stress response pathway regulates DNA damage through β2-adrenoreceptors and β-arrestin-1. Nature. 2011;477(7364):349–353. doi: 10.1038/nature10368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Flint M.S., Baum A., Episcopo B., Knickelbein K.Z., Liegey Dougall A.J., Chambers W.H., Jenkins F.J. Chronic exposure to stress hormones promotes transformation and tumorigenicity of 3T3 mouse fibroblasts. Stress. 2013;16(1):114–121. doi: 10.3109/10253890.2012.686075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Sloan E.K., Priceman S.J., Cox B.F., Yu S., Pimentel M.A., Tangkanangnukul V., Arevalo J.M., Morizono K., Karanikolas B.D., Wu L., Sood A.K., Cole S.W. The sympathetic nervous system induces a metastatic switch in primary breast cancer. Cancer Res. 2010;70(18):7042–7052. doi: 10.1158/0008-5472.Can-10-0522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Thaker P.H., Han L.Y., Kamat A.A., Arevalo J.M., Takahashi R., Lu C., Jennings N.B., Armaiz-Pena G., Bankson J.A., Ravoori M., Merritt W.M., Lin Y.G., Mangala L.S., Kim T.J., Coleman R.L., Landen C.N., Li Y., Felix E., Sanguino A.M., Newman R.A., Lloyd M., Gershenson D.M., Kundra V., Lopez-Berestein G., Lutgendorf S.K., Cole S.W., Sood A.K. Chronic stress promotes tumor growth and angiogenesis in a mouse model of ovarian carcinoma. Nat. Med. 2006;12(8):939–944. doi: 10.1038/nm1447. [DOI] [PubMed] [Google Scholar]
- 114.Boettcher C., Sommer G., Peitzsch M., Zimmer K.P., Eisenhofer G., Wudy S.A. Differential responses of urinary epinephrine and norepinephrine to 24-h shift-work stressor in physicians. Front. Endocrinol. 2020;11 doi: 10.3389/fendo.2020.572461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Copertaro A., Bracci M., Gesuita R., Carle F., Amati M., Baldassari M., Mocchegiani E., Santarelli L. Influence of shift-work on selected immune variables in nurses. Ind. Health. 2011;49(5):597–604. doi: 10.2486/indhealth.ms1210. [DOI] [PubMed] [Google Scholar]
- 116.Eldin A.S., Sabry D., Abdelgwad M., Ramadan M.A. Some health effects of work-related stress among nurses working in critical care units. Toxicol. Ind. Health. 2021;37(3):142–151. doi: 10.1177/0748233720977413. [DOI] [PubMed] [Google Scholar]
- 117.Bjorvatn B., Axelsson J., Pallesen S., Waage S., Vedaa Ø., Blytt K.M., Buchvold H.V., Moen B.E., Thun E. The association between shift work and immunological biomarkers in nurses. Front. Public Health. 2020;8:415. doi: 10.3389/fpubh.2020.00415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Osimo E.F., Cardinal R.N., Jones P.B., Khandaker G.M. Prevalence and correlates of low-grade systemic inflammation in adult psychiatric inpatients: an electronic health record-based study. Psychoneuroendocrinology. 2018;91:226–234. doi: 10.1016/j.psyneuen.2018.02.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Luca Liberale, Stefano Ministrini, Federico Carbone, Camici Giovanni G., Fabrizio Montecucco. Cytokines as therapeutic targets for cardio- and cerebrovascular diseases. Basic Res. Cardiol. 2021;116(1):23. doi: 10.1007/s00395-021-00863-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Slavich G.M., Irwin M.R. From stress to inflammation and major depressive disorder: a social signal transduction theory of depression. Psychol. Bull. 2014;140(3):774–815. doi: 10.1037/a0035302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Fujiwara K., Tsukishima E., Kasai S., Masuchi A., Tsutsumi A., Kawakami N., Miyake H., Kishi R. Urinary catecholamines and salivary cortisol on workdays and days off in relation to job strain among female health care providers. Scand. J. Work. Environ. Health. 2004;30(2):129–138. doi: 10.5271/sjweh.770. [DOI] [PubMed] [Google Scholar]
- 122.Ke Y., Zhu H., Huang Y., Cheng L., Wu X., Chen N. Correlation between monoamine neurotransmitter and cytokine levels and the occurrence of post-traumatic stress disorder among operating room nurses. Ann. Palliat. Med. 2020;9(6):3947–3956. doi: 10.21037/apm-20-1829. [DOI] [PubMed] [Google Scholar]
- 123.Dutheil F., Trousselard M., Perrier C., Lac G., Chamoux A., Duclos M., Naughton G., Mnatzaganian G., Schmidt J. Urinary interleukin-8 is a biomarker of stress in emergency physicians, especially with advancing age--the JOBSTRESS* randomized trial. PLoS One. 2013;8(8) doi: 10.1371/journal.pone.0071658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Santos Svmd, Silva L.A.D., Terra F.S., Souza A.V., Espindola F.S., Marziale M.H.P., Teixeira R.R., Mldcc Robazzi. Association of salivary alpha-amylase with anxiety and stress in nursing professionals. Rev Lat Am Enfermagem. 2021;29:e3468. doi: 10.1590/1518-8345.4859.3468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Heckenberg R.A., Hale M.W., Kent S., Wright B.J. Empathy and job resources buffer the effect of higher job demands on increased salivary alpha amylase awakening responses in direct-care workers. Behav. Brain Res. 2020;394 doi: 10.1016/j.bbr.2020.112826. [DOI] [PubMed] [Google Scholar]
- 126.Falco A., Dal Corso L., Girardi D., De Carlo A., Comar M. The moderating role of job resources in the relationship between job demands and interleukin-6 in an Italian healthcare organization. Res. Nurs. Health. 2018;41(1):39–48. doi: 10.1002/nur.21844. [DOI] [PubMed] [Google Scholar]
- 127.Slavich G.M. Life stress and health: a review of conceptual issues and recent findings. Teach. Psychol. 2016;43(4):346–355. doi: 10.1177/0098628316662768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Olvera Alvarez H.A., Provencio-Vasquez E., Slavich G.M., Laurent J.G.C., Browning M., McKee-Lopez G., Robbins L., Spengler J.D. Stress and health in nursing students: the nurse engagement and wellness study. Nurs. Res. 2019;68(6):453–463. doi: 10.1097/nnr.0000000000000383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Carabotti M., Scirocco A., Maselli M.A., Severi C. The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems. Ann. Gastroenterol. 2015;28(2):203–209. [PMC free article] [PubMed] [Google Scholar]
- 130.Johnston G.R., Webster N.R. Cytokines and the immunomodulatory function of the vagus nerve. Br. J. Anaesth. 2009;102(4):453–462. doi: 10.1093/bja/aep037. [DOI] [PubMed] [Google Scholar]
- 131.Ondicova K., Mravec B. Multilevel interactions between the sympathetic and parasympathetic nervous systems: a minireview. Endocr. Regul. 2010;44(2):69–75. doi: 10.4149/endo_2010_02_69. [DOI] [PubMed] [Google Scholar]
- 132.Herselman M.F., Bailey S., Bobrovskaya L. The effects of stress and diet on the "Brain-Gut" and "Gut-Brain" pathways in animal models of stress and depression. Int. J. Mol. Sci. 2022;23(4) doi: 10.3390/ijms23042013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Vodička M., Ergang P., Hrnčíř T., Mikulecká A., Kvapilová P., Vagnerová K., Šestáková B., Fajstová A., Hermanová P., Hudcovic T., Kozáková H., Pácha J. Microbiota affects the expression of genes involved in HPA axis regulation and local metabolism of glucocorticoids in chronic psychosocial stress. Brain Behav. Immun. 2018;73:615–624. doi: 10.1016/j.bbi.2018.07.007. [DOI] [PubMed] [Google Scholar]
- 134.Lyte M., Vulchanova L., Brown D.R. Stress at the intestinal surface: catecholamines and mucosa-bacteria interactions. Cell Tissue Res. 2011;343(1):23–32. doi: 10.1007/s00441-010-1050-0. [DOI] [PubMed] [Google Scholar]
- 135.Hart A., Kamm M.A. Review article: mechanisms of initiation and perpetuation of gut inflammation by stress. Aliment. Pharmacol. Ther. 2002;16(12):2017–2028. doi: 10.1046/j.1365-2036.2002.01359.x. [DOI] [PubMed] [Google Scholar]
- 136.Schnall P.L., Schwartz J.E., Landsbergis P.A., Warren K., Pickering T.G. A longitudinal study of job strain and ambulatory blood pressure: results from a three-year follow-up. Psychosom. Med. 1998;60(6):697–706. doi: 10.1097/00006842-199811000-00007. [DOI] [PubMed] [Google Scholar]
- 137.Horsten M., Ericson M., Perski A., Wamala S.P., Schenck-Gustafsson K., Orth-Gomér K. Psychosocial factors and heart rate variability in healthy women. Psychosom. Med. 1999;61(1):49–57. doi: 10.1097/00006842-199901000-00009. [DOI] [PubMed] [Google Scholar]
- 138.Looser R.R., Metzenthin P., Helfricht S., Kudielka B.M., Loerbroks A., Thayer J.F., Fischer J.E. Cortisol is significantly correlated with cardiovascular responses during high levels of stress in critical care personnel. Psychosom. Med. 2010;72(3):281–289. doi: 10.1097/PSY.0b013e3181d35065. [DOI] [PubMed] [Google Scholar]
- 139.Su-Chen Fang, Wu Yu-Lin, Pei-Shan Tsai. Heart rate variability and risk of all-cause death and cardiovascular events in patients with cardiovascular disease: a meta-analysis of cohort studies. Biol. Res. Nurs. 2019;22(1):45–56. doi: 10.1177/1099800419877442. [DOI] [PubMed] [Google Scholar]
- 140.Segerstrom S.C., Miller G.E. Psychological stress and the human immune system: a meta-analytic study of 30 years of inquiry. Psychol. Bull. 2004;130(4):601–630. doi: 10.1037/0033-2909.130.4.601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Glaser R., MacCallum R.C., Laskowski B.F., Malarkey W.B., Sheridan J.F., Kiecolt-Glaser J.K. Evidence for a shift in the Th-1 to Th-2 cytokine response associated with chronic stress and aging. J Gerontol A Biol Sci Med Sci. 2001;56(8):M477–M482. doi: 10.1093/gerona/56.8.m477. [DOI] [PubMed] [Google Scholar]
- 142.Miller A.H., Ancoli-Israel S., Bower J.E., Capuron L., Irwin M.R. Neuroendocrine-immune mechanisms of behavioral comorbidities in patients with cancer. J. Clin. Oncol. 2008;26(6):971–982. doi: 10.1200/jco.2007.10.7805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Morikawa Y., Kitaoka-Higashiguchi K., Tanimoto C., Hayashi M., Oketani R., Miura K., Nishijo M., Nakagawa H. A cross-sectional study on the relationship of job stress with natural killer cell activity and natural killer cell subsets among healthy nurses. J. Occup. Health. 2005;47(5):378–383. doi: 10.1539/joh.47.378. [DOI] [PubMed] [Google Scholar]
- 144.Smeeth D., Beck S., Karam E.G., Pluess M. The role of epigenetics in psychological resilience. Lancet Psychiatr. 2021;8(7):620–629. doi: 10.1016/s2215-0366(20)30515-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Barker E.D. Epigenetics, early adversity and child and adolescent mental health. Psychopathology. 2018;51(2):71–75. doi: 10.1159/000486683. [DOI] [PubMed] [Google Scholar]
- 146.McEwen B.S. Allostasis and the epigenetics of brain and body health over the life course: the brain on stress. JAMA Psychiatr. 2017;74(6):551–552. doi: 10.1001/jamapsychiatry.2017.0270. [DOI] [PubMed] [Google Scholar]
- 147.Slavich G.M., Cole S.W. The emerging field of human social genomics. Clin. Psychol. Sci. 2013;1(3):331–348. doi: 10.1177/2167702613478594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Slavich G.M., Mengelkoch S., Cole S.W. Human social genomics: concepts, mechanisms, and implications for health. Lifestyle Medicine. 2024 doi: 10.1002/lim2.75. (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Wang F., Pan F., Tang Y., Huang J.H. Editorial: early life stress-induced epigenetic changes involved in mental disorders. Front. Genet. 2021;12 doi: 10.3389/fgene.2021.684844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Starnawska A., Demontis D. Role of DNA methylation in mediating genetic risk of psychiatric disorders. Front. Psychiatr. 2021;12 doi: 10.3389/fpsyt.2021.596821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Dai Rujuan, Ansar Ahmed S. MicroRNA, a new paradigm for understanding immunoregulation, inflammation, and autoimmune diseases. Transl. Res. 2011;157(4):163–179. doi: 10.1016/j.trsl.2011.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Robert Feil, Fraga Mario F. Epigenetics and the environment: emerging patterns and implications. Nat. Rev. Genet. 2012;13(2):97–109. doi: 10.1038/nrg3142. [DOI] [PubMed] [Google Scholar]
- 153.Rudolf Jaenisch, Adrian Bird. Epigenetic regulation of gene expression: how the genome integrates intrinsic and environmental signals. Nat. Genet. 2003;33(3):245–254. doi: 10.1038/ng1089. [DOI] [PubMed] [Google Scholar]
- 154.Bartel D.P. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136(2):215–233. doi: 10.1016/j.cell.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Eddy Justin L., Karen Krukowski, Linda Janusek, Mathews Herbert L. Glucocorticoids regulate natural killer cell function epigenetically. Cell. Immunol. 2014;290(1):120–130. doi: 10.1016/j.cellimm.2014.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Krukowski K., Eddy J., Kosik K.L., Konley T., Janusek L.W., Mathews H.L. Glucocorticoid dysregulation of natural killer cell function through epigenetic modification. Brain Behav. Immun. 2011;25(2):239–249. doi: 10.1016/j.bbi.2010.07.244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Marco Cippitelli, Antonio Sica, Vincenzo Viggiano, Ye Jianping, Ghosh Paritosh, Birrer Michael J., Young Howard A. Negative transcriptional regulation of the interferon-γ promoter by glucocorticoids and dominant negative mutants of c-jun. J. Biol. Chem. 1995;270(21):12548–12556. doi: 10.1074/jbc.270.21.12548. [DOI] [PubMed] [Google Scholar]
- 158.Moolky Nagabhushan, Mathews Herbert L., Linda Witek-Janusek. Aberrant nuclear expression of AP-1 and NFkB in lymphocytes of women stressed by the experience of breast biopsy. Brain Behav. Immun. 2001;15(1):78–84. doi: 10.1006/brbi.2000.0589. [DOI] [PubMed] [Google Scholar]
- 159.Jiang S., Postovit L., Cattaneo A., Binder E.B., Aitchison K.J. Epigenetic modifications in stress response genes associated with childhood trauma. Front. Psychiatr. 2019;10:808. doi: 10.3389/fpsyt.2019.00808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Daskalakis N.P., Bagot R.C., Parker K.J., Vinkers C.H., de Kloet E.R. The three-hit concept of vulnerability and resilience: toward understanding adaptation to early-life adversity outcome. Psychoneuroendocrinology. 2013;38(9):1858–1873. doi: 10.1016/j.psyneuen.2013.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Zhao Mingyue, Wang Wei, Jiang Zhijun, Zhu Zemeng, Liu Dexiang, Pan Fang. Long-term effect of post-traumatic stress in adolescence on dendrite development and H3K9me2/BDNF expression in male rat Hippocampus and prefrontal cortex. [Original research] Front. Cell Dev. Biol. 2020;8 doi: 10.3389/fcell.2020.00682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Benoit Jamie D., Pasko Rakic, Frick Karyn M. Prenatal stress induces spatial memory deficits and epigenetic changes in the hippocampus indicative of heterochromatin formation and reduced gene expression. Behav. Brain Res. 2015;281:1–8. doi: 10.1016/j.bbr.2014.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Rowson Sydney A., Mandakh Bekhbat, Kelly Sean D., Binder Elisabeth B., Hyer Molly M., Shaw Gladys, Bent Maria Alexis, Georgia Hodes, Gregory Tharp, David Weinshenker, Qin Zhouhui, Neigh Gretchen N. Chronic adolescent stress sex-specifically alters the hippocampal transcriptome in adulthood. Neuropsychopharmacology. 2019;44(7):1207–1215. doi: 10.1038/s41386-019-0321-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Borges Juliano Viana, Betânia Souza de Freitas, Vinicius Antoniazzi, de Souza dos Santos Cristophod, Kelem Vedovelli, Pires Vivian Naziaseno, Leticia Paludo, Maria Noêmia Martins de Lima, Elke Bromberg. Social isolation and social support at adulthood affect epigenetic mechanisms, brain-derived neurotrophic factor levels and behavior of chronically stressed rats. Behav. Brain Res. 2019;366:36–44. doi: 10.1016/j.bbr.2019.03.025. [DOI] [PubMed] [Google Scholar]
- 165.Organisation for Economic Co-operation and Development (OECD) 2022. Gender Equality.https://www.oecd.org/gender/data/the-proportion-of-female-doctors-has-increased-in-all-oecd-countries-over-the-past-two-decades.htm# Retrieved 11/20/2022. [Google Scholar]
- 166.Ismael Palacios-García, Lara-Vásquez Ariel, Montiel Juan F., Díaz-Véliz Gabriela F., Hugo Sepúlveda, Elías Utreras, Martín Montecino, González-Billault Christian, Francisco Aboitiz. Prenatal stress down-regulates reelin expression by methylation of its promoter and induces adult behavioral impairments in rats. PLoS One. 2015;10(2) doi: 10.1371/journal.pone.0117680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Dunn E.C., Soare T.W., Zhu Y., Simpkin A.J., Suderman M.J., Klengel T., Smith Adac, Ressler K.J., Relton C.L. Sensitive periods for the effect of childhood adversity on DNA methylation: results from a prospective, longitudinal study. Biol. Psychiatr. 2019;85(10):838–849. doi: 10.1016/j.biopsych.2018.12.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Markham J.A., Taylor A.R., Taylor S.B., Bell D.B., Koenig J.I. Characterization of the cognitive impairments induced by prenatal exposure to stress in the rat. Front. Behav. Neurosci. 2010;4:173. doi: 10.3389/fnbeh.2010.00173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Nemoda Z., Massart R., Suderman M., Hallett M., Li T., Coote M., Cody N., Sun Z.S., Soares C.N., Turecki G., Steiner M., Szyf M. Maternal depression is associated with DNA methylation changes in cord blood T lymphocytes and adult hippocampi. Transl. Psychiatry. 2015;5(4) doi: 10.1038/tp.2015.32. e545-e545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Murgatroyd C., Quinn J.P., Sharp H.M., Pickles A., Hill J. Effects of prenatal and postnatal depression, and maternal stroking, at the glucocorticoid receptor gene. Transl. Psychiatry. 2015;5(5) doi: 10.1038/tp.2014.140. e560-e560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Choi Damee, Tsuchiya Kenji J., Takei Nori. Interaction effect of oxytocin receptor (OXTR) rs53576 genotype and maternal postpartum depression on child behavioural problems. Sci. Rep. 2019;9(1):7685. doi: 10.1038/s41598-019-44175-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Kraaijenvanger Eline J., He Yujie, Spencer Hannah, Smith Alicia K., Bos Peter A., Boks Marco P.M. Epigenetic variability in the human oxytocin receptor (OXTR) gene: a possible pathway from early life experiences to psychopathologies. Neurosci. Biobehav. Rev. 2019;96:127–142. doi: 10.1016/j.neubiorev.2018.11.016. [DOI] [PubMed] [Google Scholar]
- 173.Carugno M., Maggioni C., Crespi E., Bonzini M., Cuocina S., Dioni L., Tarantini L., Consonni D., Ferrari L., Pesatori A.C. Night shift work, DNA methylation and telomere length: an investigation on hospital female nurses. Int. J. Environ. Res. Publ. Health. 2019;16(13) doi: 10.3390/ijerph16132292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Samulin Erdem J., Notø H.Ø., Skare Ø., Lie J.S., Petersen-Øverleir M., Reszka E., Pepłońska B., Zienolddiny S. Mechanisms of breast cancer risk in shift workers: association of telomere shortening with the duration and intensity of night work. Cancer Med. 2017;6(8):1988–1997. doi: 10.1002/cam4.1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Carugno M., Maggioni C., Ruggiero V., Crespi E., Monti P., Ferrari L., Pesatori A.C. Can night shift work affect biological age? Hints from a cross-sectional study on hospital female nurses. Int. J. Environ. Res. Publ. Health. 2021;18(20) doi: 10.3390/ijerph182010639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Tabano S., Tassi L., Cannone M.G., Brescia G., Gaudioso G., Ferrara M., Colapietro P., Fontana L., Miozzo M.R., Croci G.A., Seia M., Piuma C., Solbiati M., Tobaldini E., Ferrero S., Montano N., Costantino G., Buoli M. Mental health and the effects on methylation of stress-related genes in front-line versus other health care professionals during the second wave of COVID-19 pandemic: an Italian pilot study. Eur. Arch. Psychiatr. Clin. Neurosci. 2022:1–10. doi: 10.1007/s00406-022-01472-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Chajka A.V., Ablyakimova V.L., Kormochi K.A., Khusainov D.R., Shadrina M.I., Filatova E.V. The role of DRD2-dependent genes in the formation of depressive behavior. Mol. Genet. Microbiol. Virol. 2021;36(4):181–188. doi: 10.3103/S0891416821040054. [DOI] [Google Scholar]
- 178.Bakusic J., Schaufeli W., Claes S., Godderis L. Stress, burnout and depression: a systematic review on DNA methylation mechanisms. J. Psychosom. Res. 2017;92:34–44. doi: 10.1016/j.jpsychores.2016.11.005. [DOI] [PubMed] [Google Scholar]
- 179.Al Omran Alzahra J., Shao Amy S., Watanabe Saki, Zhang Zeyu, Zhang Jifeng, Chen Xue, Watanabe Junji, Davies Daryl L., Shao Xuesi M., Liang Jing. Social isolation induces neuroinflammation and microglia overactivation, while dihydromyricetin prevents and improves them. J. Neuroinflammation. 2022;19(1):2. doi: 10.1186/s12974-021-02368-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Elliott Evan, Ezra-Nevo Gili, Limor Regev, Neufeld-Cohen Adi, Chen Alon. Resilience to social stress coincides with functional DNA methylation of the Crf gene in adult mice. Nat. Neurosci. 2010;13(11):1351–1353. doi: 10.1038/nn.2642. [DOI] [PubMed] [Google Scholar]
- 181.Miller G.E., Chen E., Sze J., Marin T., Arevalo J.M., Doll R., Ma R., Cole S.W. A functional genomic fingerprint of chronic stress in humans: blunted glucocorticoid and increased NF-kappaB signaling. Biol. Psychiatr. 2008;64(4):266–272. doi: 10.1016/j.biopsych.2008.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Shields Alexandra E., Wise Lauren A., Ruiz-Narvaez Edward A., Bobak Seddighzadeh, Hyang-Min Byun, Cozier Yvette C., Rosenberg Lynn, Palmer Julie R., Baccarelli Andrea A. Childhood abuse, promoter methylation of leukocyte NR3C1 and the potential modifying effect of emotional support. Epigenomics. 2016;8(11):1507–1517. doi: 10.2217/epi-2016-0074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Provenzi L., Brambilla M., Scotto di Minico G., Montirosso R., Borgatti R. Maternal caregiving and DNA methylation in human infants and children: systematic review. Gene Brain Behav. 2020;19(3) doi: 10.1111/gbb.12616. [DOI] [PubMed] [Google Scholar]
- 184.Xu Hang, Wang Jiesi, Zhang Ke, Zhao Mei, Bart Ellenbroek, Shao Feng, Wang Weiwen. Effects of adolescent social stress and antidepressant treatment on cognitive inflexibility and Bdnf epigenetic modifications in the mPFC of adult mice. Psychoneuroendocrinology. 2018;88:92–101. doi: 10.1016/j.psyneuen.2017.11.013. [DOI] [PubMed] [Google Scholar]
- 185.Wang Peipei, Zhang Cuizhen, Lv Qinyu, Bao Chenxi, Sun Hong, Guo Ma, Fang Yiru, Yi Zhenghui, Cai Weimin. Association of DNA methylation in BDNF with escitalopram treatment response in depressed Chinese Han patients. Eur. J. Clin. Pharmacol. 2018;74(8):1011–1020. doi: 10.1007/s00228-018-2463-z. [DOI] [PubMed] [Google Scholar]
- 186.Furman D., Campisi J., Verdin E., Carrera-Bastos P., Targ S., Franceschi C., Ferrucci L., Gilroy D.W., Fasano A., Miller G.W., Miller A.H., Mantovani A., Weyand C.M., Barzilai N., Goronzy J.J., Rando T.A., Effros R.B., Lucia A., Kleinstreuer N., Slavich G.M. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019;25(12):1822–1832. doi: 10.1038/s41591-019-0675-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Berlin G., Burns F., Essick C., Lapointe M., Murphy M. 2023. Nursing in 2023: How Hospitals Are Confronting Shortages.https://www.mckinsey.com/industries/healthcare/our-insights/nursing-in-2023 Retrieved 12/23/2023. [Google Scholar]
- 188.Miner S.A., Berkman B.E., Altiery de Jesus V., Jamal L., Grady C. Navigating pandemic moral distress at home and at work: frontline workers' experiences. AJOB Empir Bioeth. 2022:1–11. doi: 10.1080/23294515.2022.2064000. [DOI] [PubMed] [Google Scholar]
- 189.Leo C.G., Sabina S., Tumolo M.R., Bodini A., Ponzini G., Sabato E., Mincarone P. Burnout among healthcare workers in the COVID 19 era: a review of the existing literature. Front. Public Health. 2021;9 doi: 10.3389/fpubh.2021.750529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Rupal Malani. 2023. 2024 Health Systems Outlook: A Host of Challenges Ahead.https://www.mckinsey.com/industries/healthcare/our-insights/2024-health-systems-outlook-a-host-of-challenges-ahead Retrieved 12/23/2023. [Google Scholar]
- 191.Lamontagne A.D., Keegel T., Louie A.M., Ostry A., Landsbergis P.A. A systematic review of the job-stress intervention evaluation literature, 1990-2005. Int. J. Occup. Environ. Health. 2007;13(3):268–280. doi: 10.1179/oeh.2007.13.3.268. [DOI] [PubMed] [Google Scholar]
- 192.Tsutsumi A., Nagami M., Yoshikawa T., Kogi K., Kawakami N. Participatory intervention for workplace improvements on mental health and job performance among blue-collar workers: a cluster randomized controlled trial. J. Occup. Environ. Med. 2009;51(5):554–563. doi: 10.1097/JOM.0b013e3181a24d28. [DOI] [PubMed] [Google Scholar]
- 193.Georgina Morley, Sankary Lauren R., Cole Horsburgh Cristie. Mitigating moral distress through ethics consultation. Am. J. Bioeth. 2022;22(4):61–63. doi: 10.1080/15265161.2022.2044555. [DOI] [PubMed] [Google Scholar]
- 194.Hamric A.B., Wocial L.D. Institutional ethics resources: creating moral spaces. Hastings Cent. Rep. 2016;46(Suppl 1):S22–S27. doi: 10.1002/hast.627. [DOI] [PubMed] [Google Scholar]
- 195.Sands S.A., Stanley P., Charon R. Pediatric narrative oncology: interprofessional training to promote empathy, build teams, and prevent burnout. J. Support Oncol. 2008;6(7):307–312. [PubMed] [Google Scholar]
- 196.Winkel A.F., Feldman N., Moss H., Jakalow H., Simon J., Blank S. Narrative medicine workshops for obstetrics and gynecology residents and association with burnout measures. Obstet. Gynecol. 2016;128(Suppl 1):27s–33s. doi: 10.1097/aog.0000000000001619. [DOI] [PubMed] [Google Scholar]
- 197.Rita Charon. Narrative MedicineA model for empathy, reflection, profession, and trust. JAMA. 2001;286(15):1897–1902. doi: 10.1001/jama.286.15.1897. [DOI] [PubMed] [Google Scholar]
- 198.Charon R. Narrative and medicine. N. Engl. J. Med. 2004;350(9):862–864. doi: 10.1056/NEJMp038249. [DOI] [PubMed] [Google Scholar]
- 199.Naehrig D., Schokman A., Hughes J.K., Epstein R., Hickie I.B., Glozier N. Effect of interventions for the well-being, satisfaction and flourishing of general practitioners-a systematic review. BMJ Open. 2021;11(8) doi: 10.1136/bmjopen-2020-046599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Dedoncker J., Vanderhasselt M.A., Ottaviani C., Slavich G.M. Mental health during the COVID-19 pandemic and beyond: the importance of the vagus nerve for biopsychosocial resilience. Neurosci. Biobehav. Rev. 2021;125:1–10. doi: 10.1016/j.neubiorev.2021.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Shields G.S., Spahr C.M., Slavich G.M. Psychosocial interventions and immune system function: a systematic review and meta-analysis of randomized clinical trials. JAMA Psychiatr. 2020;77(10):1031–1043. doi: 10.1001/jamapsychiatry.2020.0431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Slavich G.M. Social Safety Theory: understanding social stress, disease risk, resilience, and behavior during the COVID-19 pandemic and beyond. Curr Opin Psychol. 2022;45 doi: 10.1016/j.copsyc.2022.101299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Black D.S., Slavich G.M. Mindfulness meditation and the immune system: a systematic review of randomized controlled trials. Ann. N. Y. Acad. Sci. 2016;1373(1):13–24. doi: 10.1111/nyas.12998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Gerritsen Roderik J.S., Band Guido P.H. Breath of life: the respiratory vagal stimulation model of contemplative activity. [Hypothesis and theory] Front. Hum. Neurosci. 2018;12 doi: 10.3389/fnhum.2018.00397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Errazuriz A., Schmidt K., Undurraga E.A., Medeiros S., Baudrand R., Cussen D., Henriquez M., Celhay P., Figueroa R.A. Effects of mindfulness-based stress reduction on psychological distress in health workers: a three-arm parallel randomized controlled trial. J. Psychiatr. Res. 2022;145:284–293. doi: 10.1016/j.jpsychires.2020.11.011. [DOI] [PubMed] [Google Scholar]
- 206.Bär K.J., Herbsleb M., Schumann A., de la Cruz F., Gabriel H.W., Wagner G. Hippocampal-brainstem connectivity associated with vagal modulation after an intense exercise intervention in healthy men. Front. Neurosci. 2016;10:145. doi: 10.3389/fnins.2016.00145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Breit S., Kupferberg A., Rogler G., Hasler G. Vagus nerve as modulator of the brain-gut Axis in psychiatric and inflammatory disorders. Front. Psychiatr. 2018;9:44. doi: 10.3389/fpsyt.2018.00044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Lujan H.L., DiCarlo S.E. Physical activity, by enhancing parasympathetic tone and activating the cholinergic anti-inflammatory pathway, is a therapeutic strategy to restrain chronic inflammation and prevent many chronic diseases. Med. Hypotheses. 2013;80(5):548–552. doi: 10.1016/j.mehy.2013.01.014. [DOI] [PubMed] [Google Scholar]
- 209.Metsios G.S., Moe R.H., Kitas G.D. Exercise and inflammation. Best Pract. Res. Clin. Rheumatol. 2020;34(2) doi: 10.1016/j.berh.2020.101504. [DOI] [PubMed] [Google Scholar]
- 210.Duclos M., Tabarin A. Exercise and the hypothalamo-pituitary-adrenal Axis. Front. Horm. Res. 2016;47:12–26. doi: 10.1159/000445149. [DOI] [PubMed] [Google Scholar]
- 211.Wang Meizi, Baker Julien S., Quan Wenjing, Shen Siqin, Gusztáv Fekete, Gu Yaodong. A preventive role of exercise across the coronavirus 2 (SARS-CoV-2) pandemic. Front. Physiol. 2020;11 doi: 10.3389/fphys.2020.572718. Review. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Reif J., Chan D., Jones D., Payne L., Molitor D. Effects of a workplace wellness program on employee health, health beliefs, and medical use: a randomized clinical trial. JAMA Intern. Med. 2020;180(7):952–960. doi: 10.1001/jamainternmed.2020.1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Taylor A.M., Thompson S.V., Edwards C.G., Musaad S.M.A., Khan N.A., Holscher H.D. Associations among diet, the gastrointestinal microbiota, and negative emotional states in adults. Nutr. Neurosci. 2020;23(12):983–992. doi: 10.1080/1028415x.2019.1582578. [DOI] [PubMed] [Google Scholar]
- 214.Toyoda A. Nutritional interventions for promoting stress resilience: recent progress using psychosocial stress models of rodents. Anim. Sci. J. 2020;91(1) doi: 10.1111/asj.13478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Westfall S., Caracci F., Zhao D., Wu Q.L., Frolinger T., Simon J., Pasinetti G.M. Microbiota metabolites modulate the T helper 17 to regulatory T cell (Th17/Treg) imbalance promoting resilience to stress-induced anxiety- and depressive-like behaviors. Brain Behav. Immun. 2021;91:350–368. doi: 10.1016/j.bbi.2020.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Holzer P., Reichmann F., Farzi A. Neuropeptide Y, peptide YY and pancreatic polypeptide in the gut-brain axis. Neuropeptides. 2012;46(6):261–274. doi: 10.1016/j.npep.2012.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Hailley Loper, Monique Leinen, Logan Bassoff, Jack Sample, Romero-Ortega Mario, Gustafson Kenneth J., Taylor Dawn M., Schiefer Matthew A. Both high fat and high carbohydrate diets impair vagus nerve signaling of satiety. Sci. Rep. 2021;11(1) doi: 10.1038/s41598-021-89465-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Jennifer Utter, Sally McCray, Simon Denny. Work site food purchases among healthcare staff: relationship with healthy eating and opportunities for intervention. Nutr. Diet. 2022;79(2):265–271. doi: 10.1111/1747-0080.12724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Jin Endo, Arita Makoto. Cardioprotective mechanism of omega-3 polyunsaturated fatty acids. J. Cardiol. 2016;67(1):22–27. doi: 10.1016/j.jjcc.2015.08.002. [DOI] [PubMed] [Google Scholar]
- 220.Liao Yuhua, Xie Bo, Zhang Huimin, He Qian, Guo Lan, Mehala Subramanieapillai, Fan Beifang, Lu Ciyong, McIntyre Roger S. Efficacy of omega-3 PUFAs in depression: a meta-analysis. Transl. Psychiatry. 2019;9(1):190. doi: 10.1038/s41398-019-0515-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Jahangard L., Hedayati M., Abbasalipourkabir R., Haghighi M., Ahmadpanah M., Faryadras M., Mikoteit T., Sadeghi Bahmani D., Brand S. Omega-3-polyunsatured fatty acids (O3PUFAs), compared to placebo, reduced symptoms of occupational burnout and lowered morning cortisol secretion. Psychoneuroendocrinology. 2019;109 doi: 10.1016/j.psyneuen.2019.104384. [DOI] [PubMed] [Google Scholar]
- 222.Murrough J.W., Abdallah C.G., Anticevic A., Collins K.A., Geha P., Averill L.A., Schwartz J., DeWilde K.E., Averill C., Jia-Wei Yang G., Wong E., Tang C.Y., Krystal J.H., Iosifescu D.V., Charney D.S. Reduced global functional connectivity of the medial prefrontal cortex in major depressive disorder. Hum. Brain Mapp. 2016;37(9):3214–3223. doi: 10.1002/hbm.23235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Wang J., Hodes G.E., Zhang H., Zhang S., Zhao W., Golden S.A., Bi W., Menard C., Kana V., Leboeuf M., Xie M., Bregman D., Pfau M.L., Flanigan M.E., Esteban-Fernández A., Yemul S., Sharma A., Ho L., Dixon R., Merad M., Han M.H., Russo S.J., Pasinetti G.M. Epigenetic modulation of inflammation and synaptic plasticity promotes resilience against stress in mice. Nat. Commun. 2018;9(1):477. doi: 10.1038/s41467-017-02794-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Shields A.E., Wise L.A., Ruiz-Narvaez E.A., Seddighzadeh B., Byun H.M., Cozier Y.C., Rosenberg L., Palmer J.R., Baccarelli A.A. Childhood abuse, promoter methylation of leukocyte NR3C1 and the potential modifying effect of emotional support. Epigenomics. 2016;8(11):1507–1517. doi: 10.2217/epi-2016-0074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Pape Julius C., Tania Carrillo-Roa, Rothbaum Barbara O., Nemeroff Charles B., Darina Czamara, Zannas Anthony S., Dan Iosifescu, Mathew Sanjay J., Neylan Thomas C., Mayberg Helen S., Dunlop Boadie W., Binder Elisabeth B. DNA methylation levels are associated with CRF1 receptor antagonist treatment outcome in women with post-traumatic stress disorder. Clin. Epigenet. 2018;10(1):136. doi: 10.1186/s13148-018-0569-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Benjamin Rosen, Mary Preisman, Hunter Jonathan, Robert Maunder. Applying psychotherapeutic principles to bolster resilience among health care workers during the COVID-19 pandemic. Am. J. Psychother. 2020;73(4):144–148. doi: 10.1176/appi.psychotherapy.20200020. [DOI] [PubMed] [Google Scholar]
- 227.Azizeh Alizadeh, Reza Khankeh Hamid, Mohammad Barati, Yazdan Ahmadi, Arash Hadian, Maryam Azizi. Psychological distress among Iranian health-care providers exposed to coronavirus disease 2019 (COVID-19): a qualitative study. BMC Psychiatr. 2020;20(1):494. doi: 10.1186/s12888-020-02889-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Grzegorz Józef Nowicki, Barbara Ślusarska, Kinga Tucholska, Katarzyna Naylor, Chrzan-Rodak Agnieszka, Barbara Niedorys. The severity of traumatic stress associated with COVID-19 pandemic, perception of support, sense of security, and sense of meaning in life among nurses: research protocol and preliminary results from Poland. Int. J. Environ. Res. Publ. Health. 2020;17(18):6491. doi: 10.3390/ijerph17186491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Duan L., Zhu G. Psychological interventions for people affected by the COVID-19 epidemic. Lancet Psychiatr. 2020;7(4):300–302. doi: 10.1016/s2215-0366(20)30073-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Brachman Rebecca A., McGowan Josephine C., Perusini Jennifer N., Lim Sean C., Pham Thu Ha, Charlene Faye, Gardier Alain M., Mendez-David Indira, David Denis J., René Hen, Denny Christine A. Ketamine as a prophylactic against stress-induced depressive-like behavior. Biol. Psychiatr. 2016;79(9):776–786. doi: 10.1016/j.biopsych.2015.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Schwartz R., Shanafelt T.D., Gimmler C., Osterberg L. Developing institutional infrastructure for physician wellness: qualitative Insights from VA physicians. BMC Health Serv. Res. 2020;20(1):7. doi: 10.1186/s12913-019-4783-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Milota M.M., van Thiel G.J.M.W., van Delden J.J.M. Narrative medicine as a medical education tool: a systematic review. Med. Teach. 2019;41(7):802–810. doi: 10.1080/0142159X.2019.1584274. [DOI] [PubMed] [Google Scholar]
- 233.Brown G.W., Harris T.O., Hepworth C. Loss, humiliation and entrapment among women developing depression: a patient and non-patient comparison. Psychol. Med. 1995;25(1):7–21. doi: 10.1017/s003329170002804x. [DOI] [PubMed] [Google Scholar]
- 234.Lee Hyun-Jung, Park Bom-Mi. Feelings of entrapment during the COVID-19 pandemic based on ace star model: a concept analysis. Healthcare. 2021;9:1305. doi: 10.3390/healthcare9101305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.National Academies of Sciences Engineering, Medicine, National Academy of Medicine, Committee on Systems Approaches to Improve Patient Care by Supporting Clinician Well-Being . National Academies Press (US) Copyright; Washington (DC): 2019. Taking Action against Clinician Burnout: A Systems Approach to Professional Well-Being. 2019 by the National Academy of Sciences. All rights reserved. [PubMed] [Google Scholar]
- 236.Shanafelt T.D., Gorringe G., Menaker R., Storz K.A., Reeves D., Buskirk S.J., Sloan J.A., Swensen S.J. Impact of organizational leadership on physician burnout and satisfaction. Mayo Clin. Proc. 2015;90(4):432–440. doi: 10.1016/j.mayocp.2015.01.012. [DOI] [PubMed] [Google Scholar]
- 237.Epstein Elizabeth G., Haizlip Julie, Liaschenko Joan, Zhao David, Bennett Rachel, Marshall Mary Faith. Moral distress, mattering, and secondary traumatic stress in provider burnout: a call for moral community. AACN Adv. Crit. Care. 2020;31(2):146–157. doi: 10.4037/aacnacc2020285. [DOI] [PubMed] [Google Scholar]