Abstract
Although the use of dermoscopy has markedly improved both the sensitivity and specificity for skin cancer detection, there is still opportunity for improvement. Ancillary techniques provide additional ways to assess a lesion with the aim of improving our diagnostic ability with little extra cost. Usage of these techniques can strengthen diagnosis and help reduce unnecessary biopsies of benign lesions.
Introduction
The use of dermoscopy has markedly improved both the sensitivity and specificity for skin cancer detection.1 However, some authors have posited that heightened sensitivity for melanoma may be contributing to an increase in the number of benign lesions biopsied.2 Therefore, some dermoscopy experts emphasize the importance of maintaining high diagnostic specificity, as implementation of dermoscopy can improve a clinician’s benign to malignant biopsy ratio.3,4
Although morphology provides the greatest insight into a lesion’s underlying diagnosis, some dermoscopy experts acknowledge that ancillary techniques may improve diagnostic accuracy. In this article, we review a selection of the ancillary techniques described in the literature to assist with diagnosis of both pigmented and nonpigmented lesions.
Pinch test for dermatofibroma
Dermatofibromas (DFs) are usually easily recognizable on clinical examination based on their morphology, location and consistency.5 On polarized dermoscopy, DFs often reveal peripheral network, vessels and shiny white lines,6,7 which can introduce diagnostic uncertainty as these findings are also associated with melanoma.8 Therefore, clinicians with a high suspicion for cutaneous malignancy may mistake these benign lesions for melanoma and biopsy them, even though a simple pinch test may provide the clinical insight to avoid a biopsy.
DFs are firm lesions that also characteristically dimple or pucker inward when squeezed. This is commonly referred to as the pinch test9 (Figure 1). Although commonly associated with DFs, other benign, neoplastic or inflammatory processes involving the upper dermis can also have a positive dimple sign such as pilomatrixoma.10 Therefore, results of the pinch test should be considered within the clinical and dermoscopic9,10 context of a lesion prior to biopsy.
Figure 1.
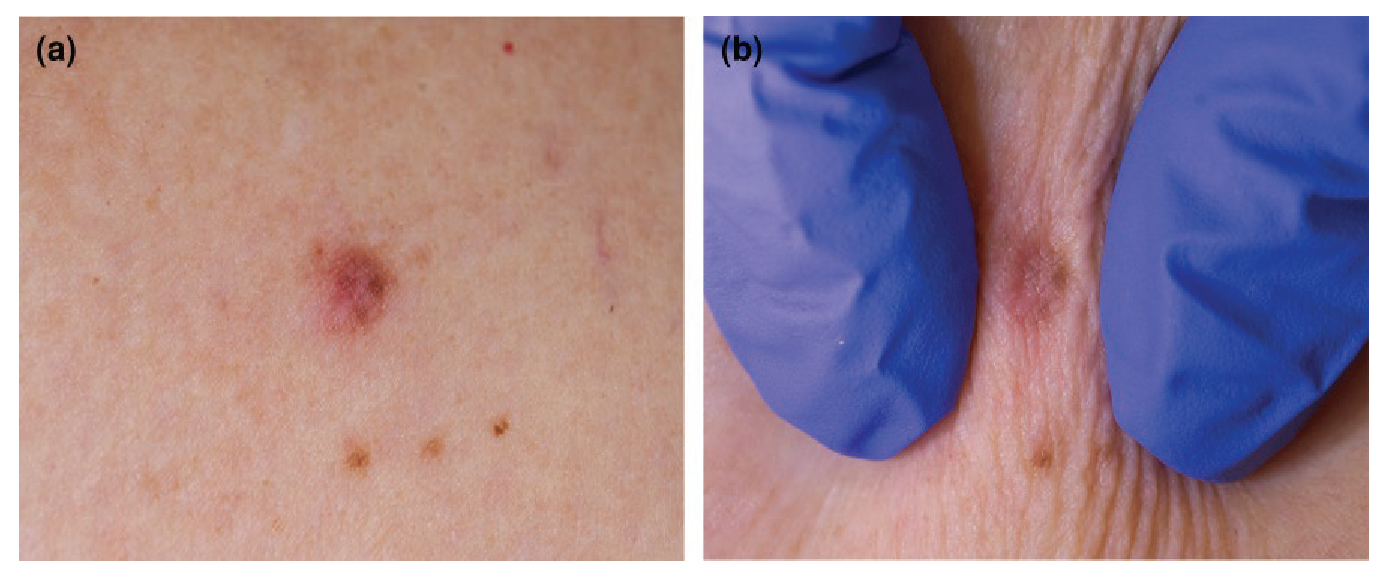
(a) Lesion suspected to be a dermatofibroma (DF); (b) the pinch test illustrates a positive dimple sign, supporting a diagnosis of DF.
Wobble sign for intradermal naevus or seborrhoeic keratosis
Intradermal naevus (IDN) is generally a clinical diagnosis, typically appearing as a dome-shaped papule or nodule on the face, often with hair and preserved skin markings.11 Morphologically, IDN can on occasion be difficult to differentiate from basal cell carcinoma11 (BCC), as similar vessels can be seen in both lesions.11,12
In this case, the wobble sign can aid in the diagnosis of IDN. With the dermatoscope in gentle contact with the lesion, horizontal pressure applied in a to-and-fro motion causes an IDN to wobble or roll (Figure 2; Supplementary Video S1). This motion is consistent with the dermal origin of IDN and is thought to be uncommon in cutaneous malignancies due to collagen deposition in the epidermis and dermis fixing the lesion in place. Although wobbling itself provides a valuable diagnostic clue, examination of the undersurface of pedunculated IDNs by wobbling the lesion allows the clinician to inspect the entire IDN surface, allowing for the identification of melanoma foci that can otherwise be easily missed.
Figure 2.
(a) Dermoscopic view of a lesion suspected to be an intradermal naevus (IDN); (b) application of horizontal pressure causes the lesion to wobble, supporting the diagnosis of IDN.
The wobble sign can also assist with differentiating verrucous melanoma from seborrhoeic keratoses (SK). Horizontal pressure applied in a to-and-fro manner causes SK to slide back and forth, whereas the dermal component of a melanoma may wobble.13
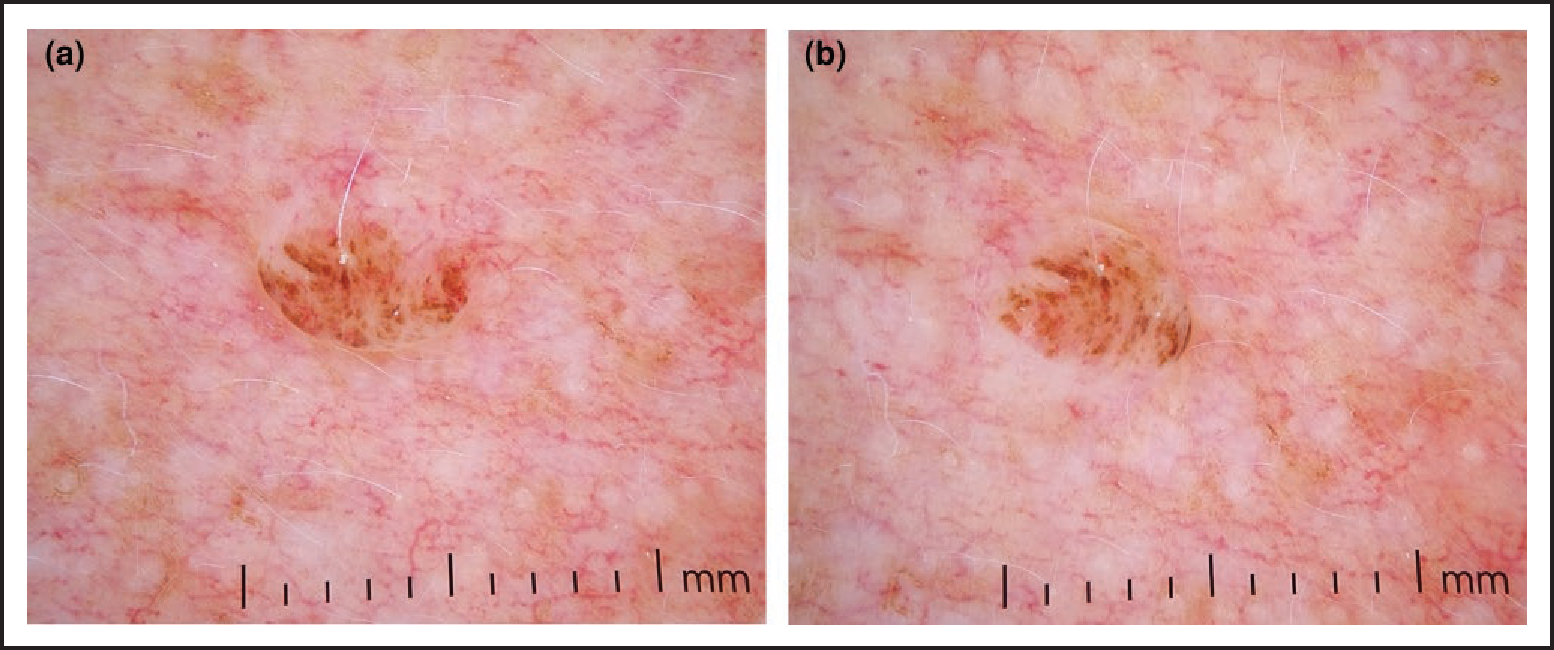
Scrape test for subcorneal haematoma
Subcorneal haematoma (SH) presents as a pigmented skin lesion occurring in response to trauma on the palms or soles. While SH commonly presents with homogeneous red–black pigmentation and satellite globules14 on dermoscopy, 40% of SHs have a parallel ridge pattern,14,15 a feature strongly associated with acral lentiginous melanoma.16 This may lead to a misdiagnosis of melanoma in the absence of a clear history of trauma to the area.
For these cases, the scrape test can be used, where the stratum corneum overlying an SH is pared down using a scalpel. Removal of the lesion in this manner demonstrates the pigment is confined to the corneal layer and confirms the diagnosis of SH14 (Figure 3). Exogenous pigment, such as that from dye, may also be removed with this method. By contrast, the lesion would not be scraped away if it were melanoma.
Figure 3.
(a) Dermoscopic image of a red/black lesion on the sole with a parallel ridge pattern; (b) scrape test performed using a blade to remove the stratum corneum from part of the lesion; (c) part of the lesion has been removed, confirming a diagnosis of subcorneal haematoma.
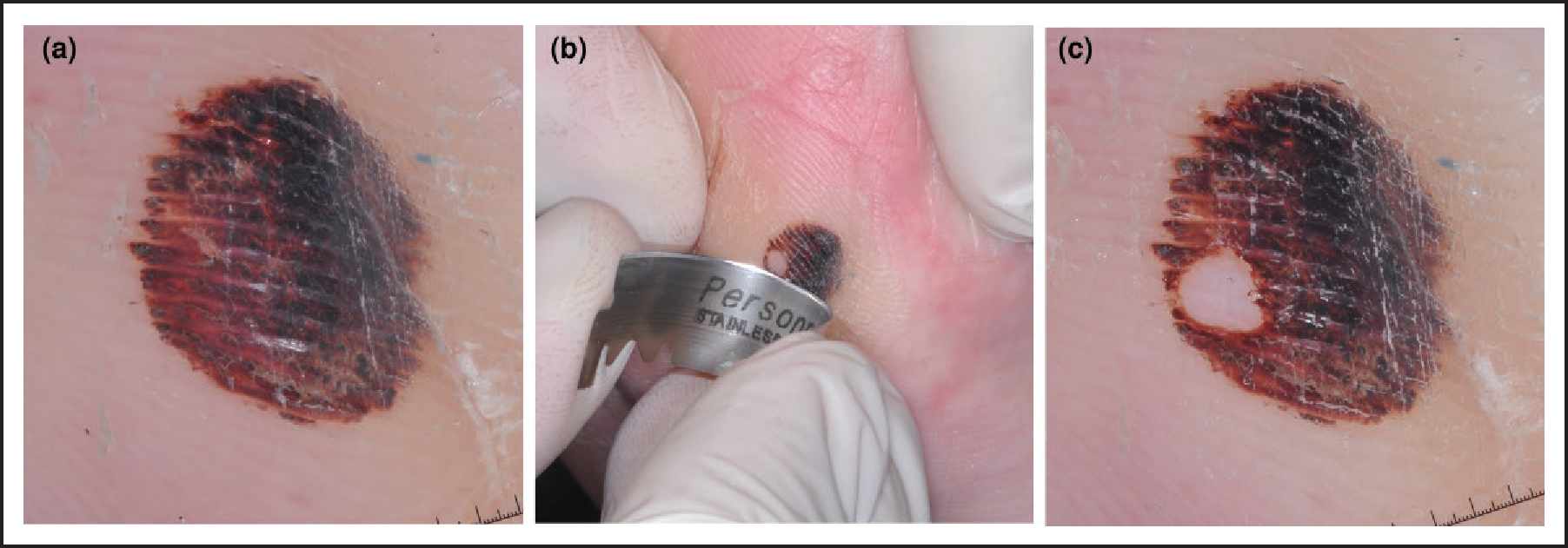
Adhesive tape test for hypermelanotic naevi
Hypermelanotic naevi are darkly pigmented lesions17 with a high concentration of melanin granules in the stratum corneum (black lamella), dermoscopically appreciable as a blotch. The blotch can render it challenging to view the deeper dermoscopic structures,18 commonly a reticular network, which would establish the diagnosis of hypermelanotic naevus over melanoma.17,19
The adhesive tape test (Figure 4) uses the repeated adhesion to and removal of tape from the lesion to remove the superficial layer of the stratum corneum. In hypermelanotic naevi, removal of the black lamella reveals the underlying structures. As the blotches in melanoma are due to heavy deposition of melanin throughout the dermis and epidermis, tape stripping will not reveal any underlying structures.18
Figure 4.
(a) Dermoscopic view of a darkly pigmented lesion with a central blotch; (b) appearance of the lesion following the adhesive tape test, revealing the reticular network of a benign naevus; (c) the pigmented stratum corneum has been removed and is visible on the tape.
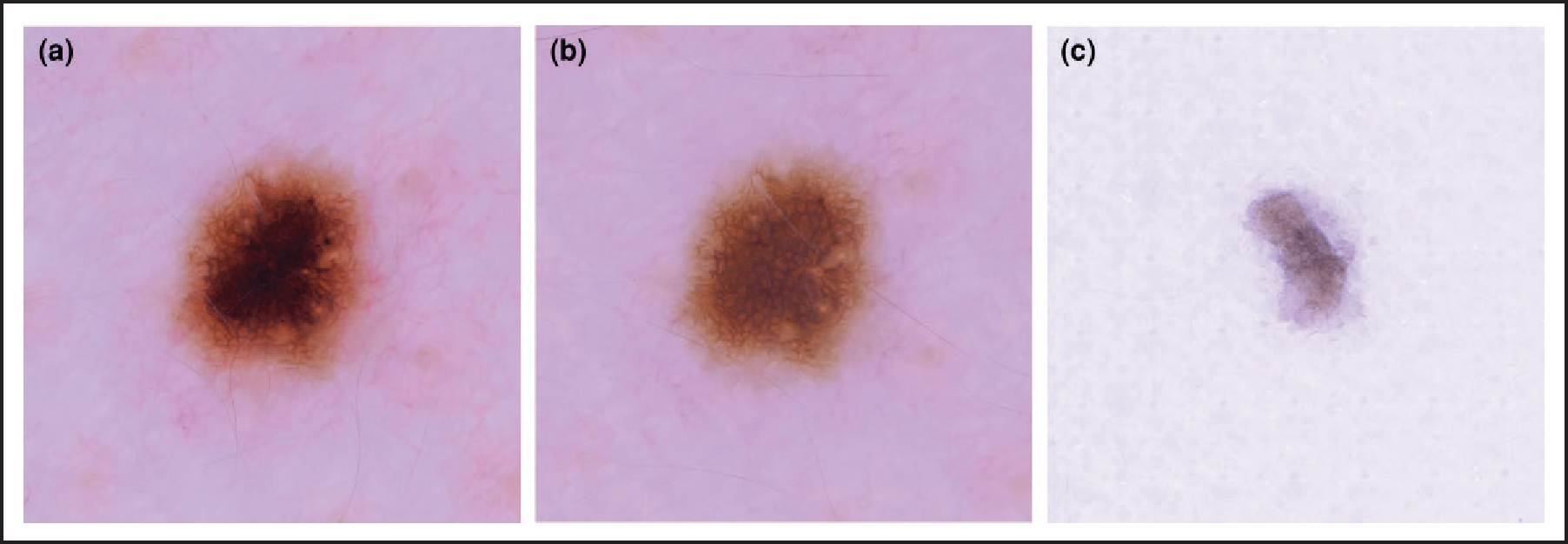
Ink test for seborrhoeic keratosis
On dermoscopy, SK may have milia-like cysts, comedo-like openings, gyri and sulci, sharp demarcation and moth-eaten borders and hairpin vessels.20,21 However, some SK can appear relatively featureless, thus raising concern for melanoma.
The ink test allows for improved visualization of structures highly specific for SK, i.e. gyri, sulci and comedo-like openings. The test involves marking the lesion with a surgical marker pen (gentian violet dye) followed by removal of the excess ink using an alcohol swab. On dermoscopic examination, the residual ink will adhere to the keratin-filled invaginations, thereby rendering the gyri, sulci and comedo-like openings more conspicuous22 (Figure 5). By contrast, melanomas do not retain the dye and remain featureless following the ink test.
Figure 5.
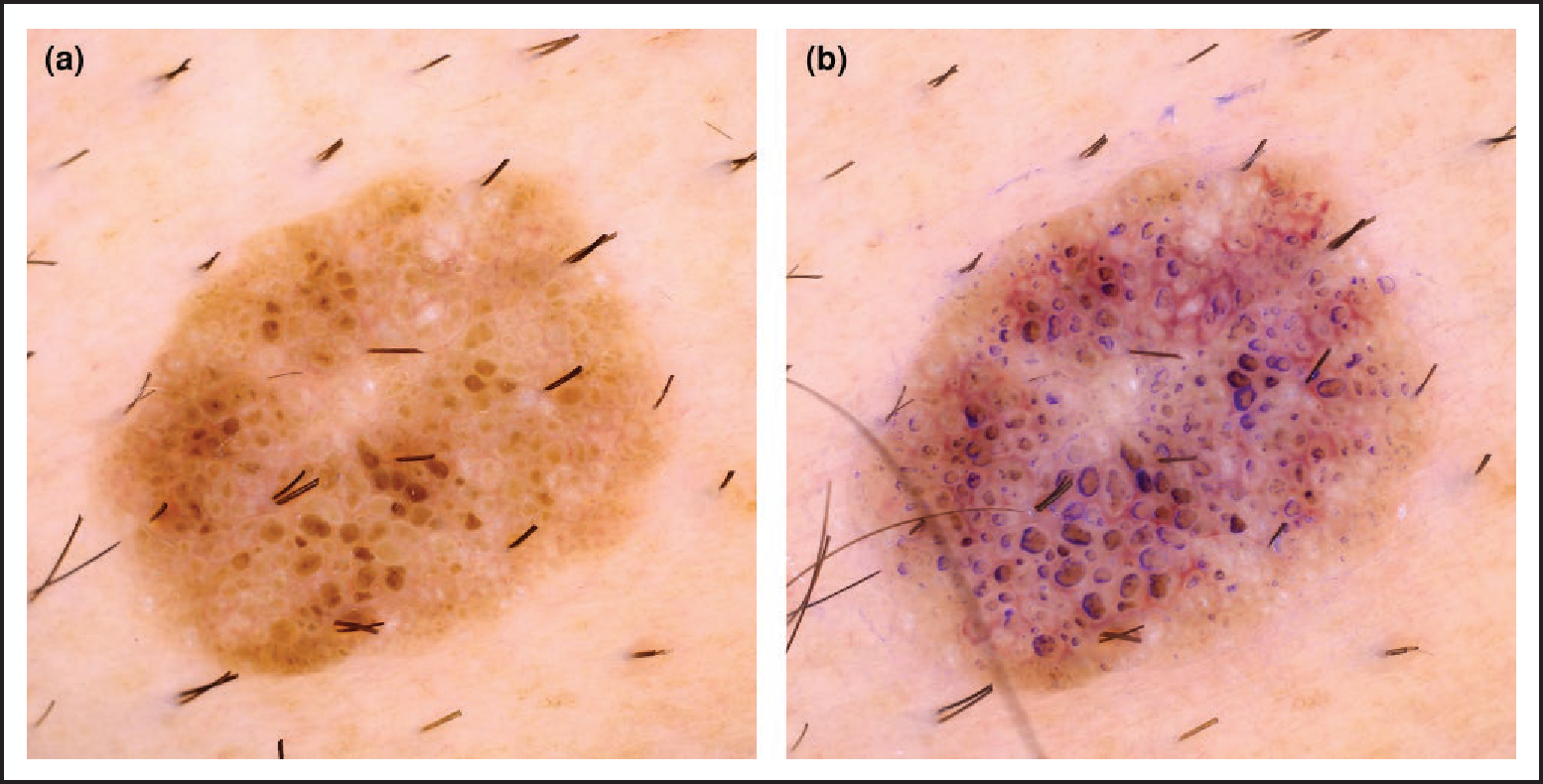
(a) Dermoscopic image of a lesion composed of nonspecific brown clods; (b) following the ink test, the comedo-like openings become easily apparent, aiding diagnosis of a seborrhoeic keratosis.
Ink test for porokeratosis
Porokeratosis is characterized by the presence of a ridged, hyperkeratotic border associated with a longitudinal furrow, which corresponds histologically to the cornoid lamella. Dermoscopically, the cornoid lamella is seen as a peripheral white rim.23,24 However, porokeratosis can also appear as pink lesions with dotted to glomerular vessels, and with a cornoid lamella that is not readily obvious, leading to the incorrect clinical diagnosis of a squamous cell carcinoma (SCC).
The ink test, performed in the same manner as for SK, deposits residual ink within the furrow of the cornoid lamella, producing a visible ring around the lesion25,26 (Figure 6). Identification of the cornoid lamella in this way helps confirm the diagnosis of porokeratosis, thereby avoiding the need for biopsy.
Figure 6.
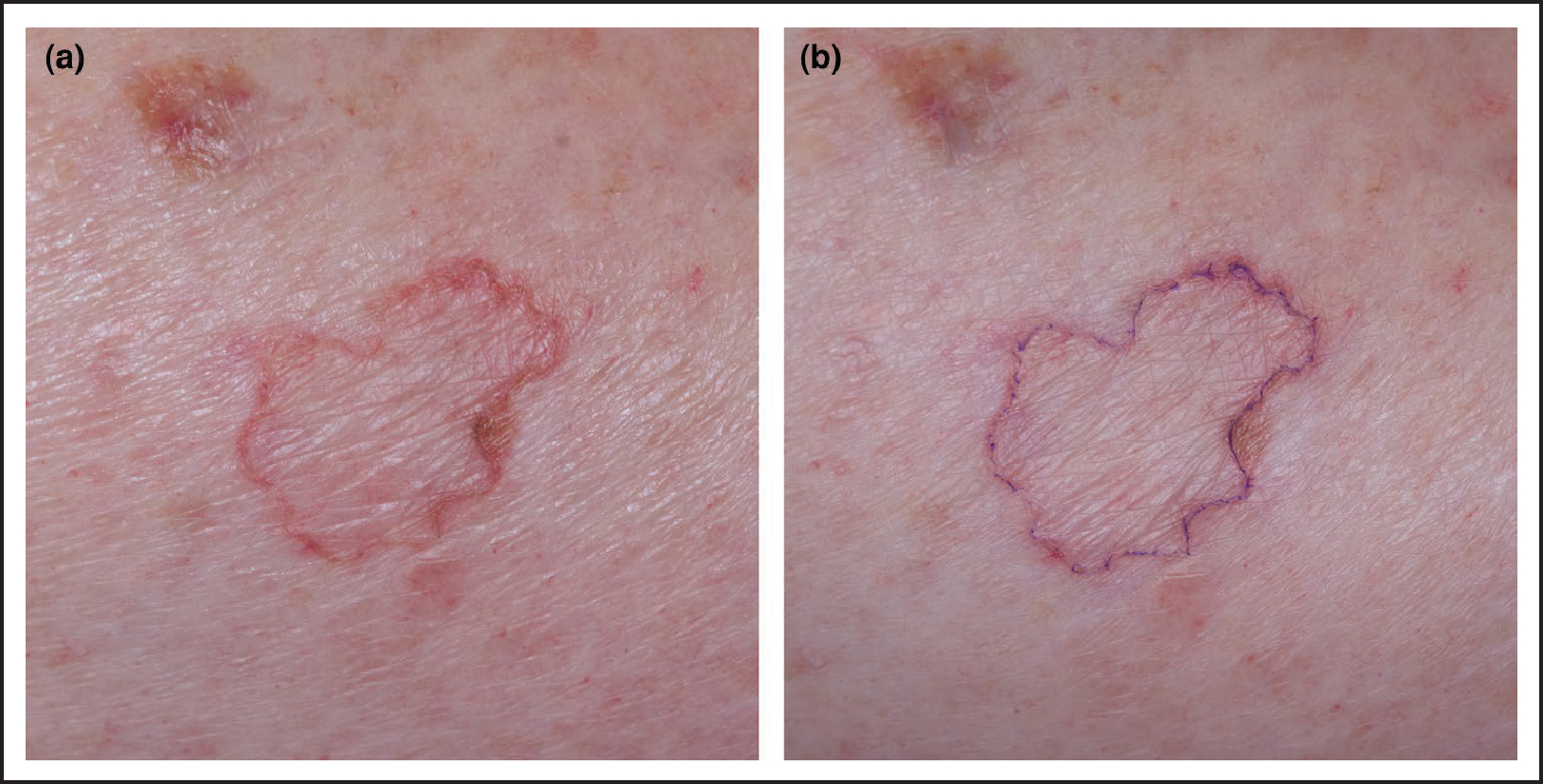
(a) An annular skin lesion with unclear diagnosis; (b) following the ink test, deposition of ink within the furrow of the cornoid lamella confirms the diagnosis of porokeratosis.
Furrow ink test for palmoplantar pigmented lesions
The appearance of palmoplantar skin is unique due to the dermatoglyphics creating alternating (wider) ridges and (narrower) furrows, with multiple eccrine openings found linearly along the ridges.16,27 Histological analysis has demonstrated that melanocytic naevus cells cluster near the dermoepidermal junction of the crista limitans and deposit melanin in the keratinocytes directly above. This corresponds to the parallel furrow appearance of acral melanocytic naevus. In contrast, melanocytes in early acral melanoma accumulate near the crista intermedia and deposit melanin in keratinocytes that correspond to the ridges.28
It is imperative to determine whether on dermoscopy the pigment predominates along the furrows (parallel furrow pattern or lattice-like pattern), which is highly specific for a benign melanocytic naevus (specificity 93·2%), or whether the pigment predominates along the ridges (parallel ridge pattern), highly specific for melanoma in non-black patients (specificity 99·0%).16 However, distinguishing between the two can be challenging in some cases, such as with the double-line furrow pattern.
The furrow ink test, performed in the same manner as the ink tests described above, leads to preferential staining of the furrows, facilitating their identification.27 The ink also accentuates the eccrine openings, thus helping to emphasize the ridges27 and perhaps even preventing an unnecessary biopsy (Figure 7).
Figure 7.
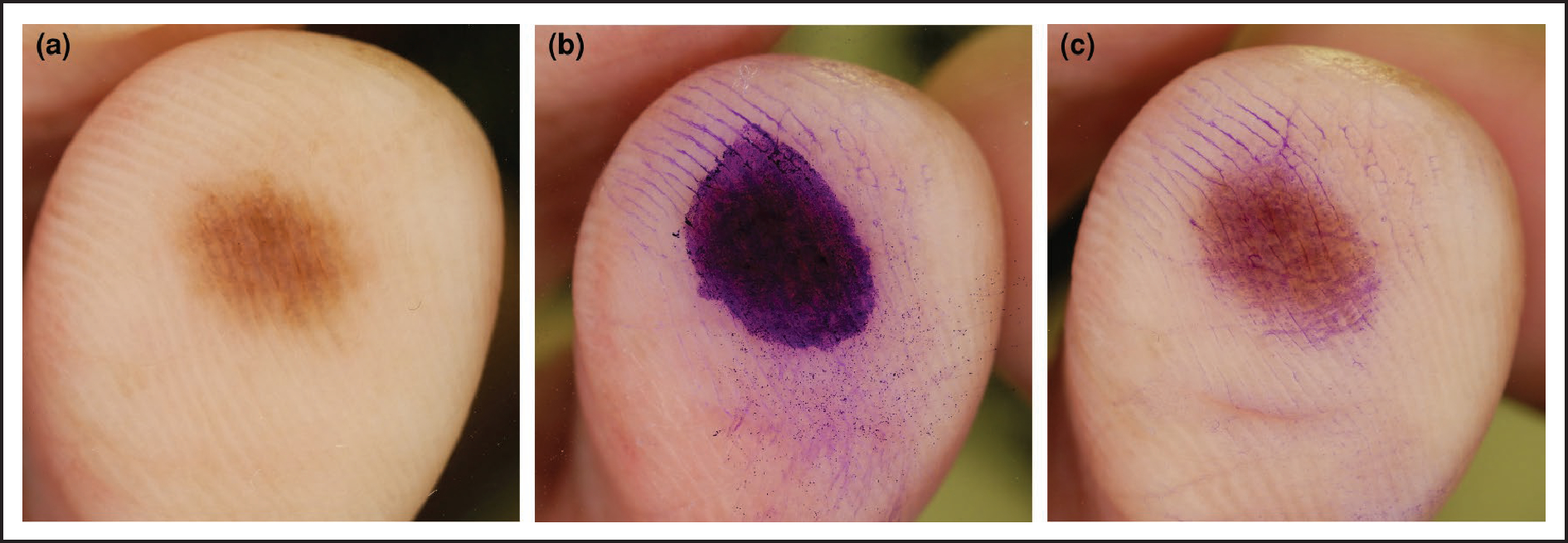
(a) Dermoscopic view of a pigmented acral lesion; it is unclear whether the pigment predominates in the furrows or the ridges. (b) Ink is applied to the lesion as part of the furrow ink test; (c) when the ink is wiped away a parallel furrow pattern becomes evident, indicating a benign naevus.
Lateral pressure technique for fibrillar-patterned pigmented lesions on the sole
The fibrillar pattern is regarded as an artefact of the parallel furrow pattern, as chronic unidirectional frictional forces on the skin, such as that on the weight-bearing area of the sole,29–32 result in uniformly distributed, obliquely arranged columns of melanin in a slanting stratum corneum.29,30,33 However, the fibrillar pattern can also be seen in acral lentiginous melanoma with either focal or heterogeneous distribution, explaining why a large proportion of lesions with this morphology are biopsied.34
Applying lateral pressure with the dermatoscope in the direction opposite to the frictional forces allows alignment of pigmentation back to a vertical plane. Visualization of the parallel furrow pattern can reassure a clinician that the lesion is benign31,32 (Figure 8). Alternatively, the lesion can be viewed through the dermatoscope without contact via an oblique angle to reveal any underlying parallel furrow pattern.35
Figure 8.
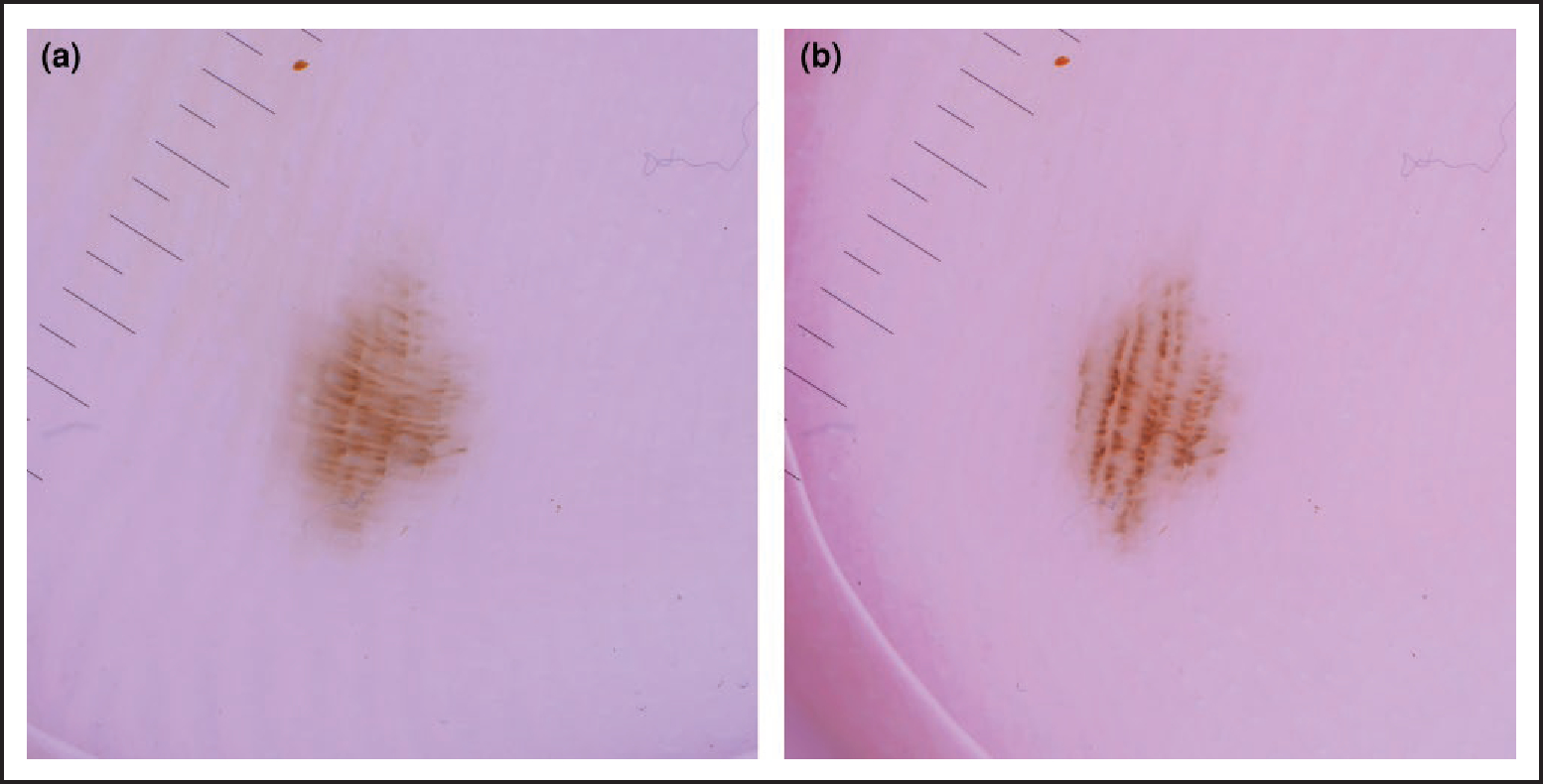
(a) Dermoscopic image of a fibrillar-patterned pigmented lesion on the sole; (b) lateral pressure with the dermatoscope aligns the pigment back to a parallel furrow pattern, indicating a benign naevus.
The blink sign
Some dermoscopic structures may be more conspicuous under nonpolarized dermoscopy (NPD) while others may be more visible under polarized dermoscopy (PD), reflecting the importance of both modes.36 NPD is superior for identifying superficial structures including a blue–white veil due to orthokeratosis, milia-like cysts and comedo-like openings, and granularity/peppering on thin skin, whereas PD is better able to identify deeper structures including blood vessels and shiny white structures (SWS).36
Toggling between the nonpolarized and polarized light modes on a hybrid dermatoscope causes structures more visible under either NPD or PD to rapidly come into view and then disappear, giving the impression of a ‘blink’. The optical contrast provided by the ‘blink’ allows for the improved detection of structures such as shiny white lines for melanoma and milia-like cysts for SK37 (Figure 9; Supplementary Video S2).
Figure 9.
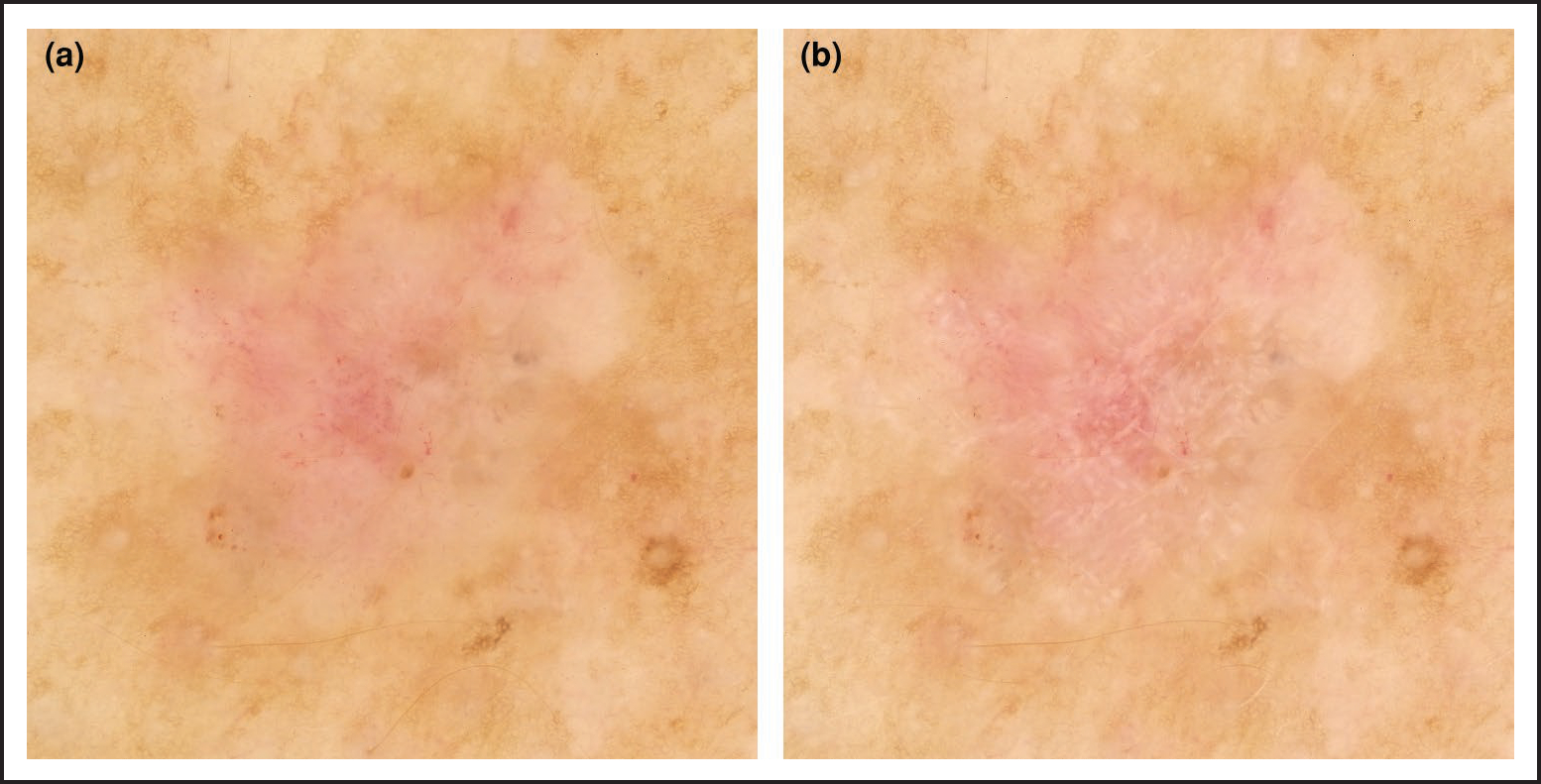
(a) Nonpolarized dermoscopy revealed a largely featureless skin lesion; (b) toggling to polarized dermoscopy allowed identification of shiny white structures, aiding with the diagnosis of skin cancer, in this case a basal cell carcinoma.
Angular dependence of polarized light
SWS correspond to altered collagen within skin lesions and are accentuated by PD.36,38 They are seen in DF, BCC, Spitz naevi and melanoma.39–41 As collagen is birefringent and rapidly randomizes polarized light, the visibility of SWS is dependent on the viewing angle.38 Therefore, it is important to be mindful that using the dermatoscope in just one orientation may lead to failure to identify SWS. Slowly rotating the dermatoscope by 180 degrees while using polarized light will ensure that SWS are not missed during examination as they can become more or less prominent depending on the angle of polarization, thus demonstrating the angular dependence of polarized light38 (Figure 10).
Figure 10.
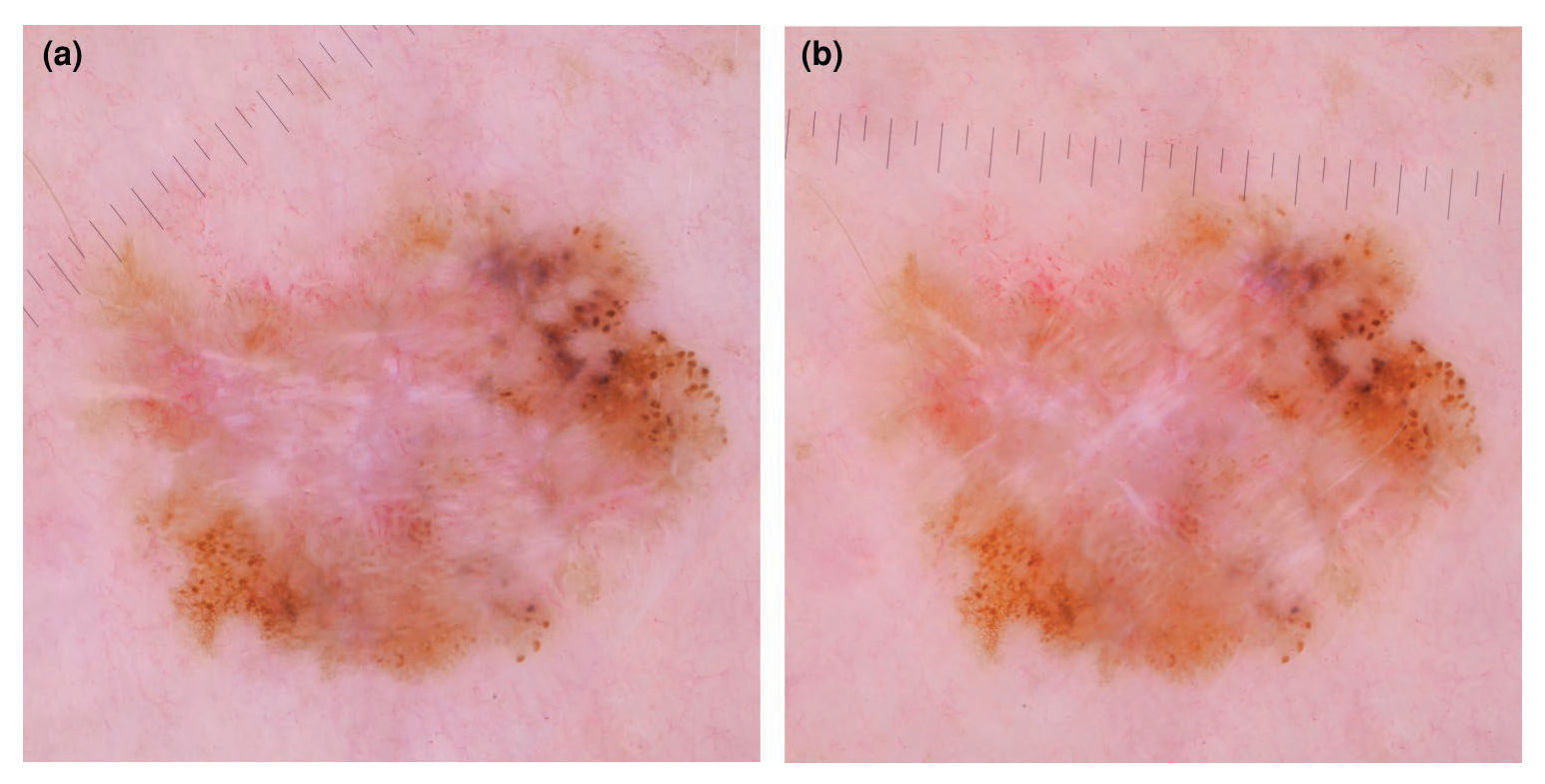
(a) Shiny white structures (SWS) are visible with polarized dermoscopy (PD) but less prominent due to the orientation of the dermatoscope; (b) slow rotation of the dermatoscope up to 180 degrees while using PD can enhance identification of the SWS, in this case supporting a diagnosis of melanoma. This phenomenon is due to the angular dependence of polarized light.
Learning points
Ancillary techniques aim to improve the diagnostic accuracy of dermoscopic assessment of skin lesions.
The pinch test for DF and the wobble sign for IDN take advantage of the dermal component of each lesion for improved identification of each.
The scrape test for SH and the tape test for hypermelanotic naevi allow for improved diagnosis by virtue of involvement of the stratum corneum in both lesions.
Ink tests, such as those for SK, porokeratosis and palmoplantar pigmented lesions use differences in the structural nature of each lesion to allow for improved detection.
The lateral pressure technique attempts to correct for the underlying pathophysiological change responsible for fibrillar pattern, thus improving the differentiation of benign from malignant lesions with fibrillar pattern on clinical examination.
The blink sign and the angular dependence of polarized light demonstrate the differences in light absorption and reflection in the presumed collagen bundles within lesions such as melanoma, SK and DF.
Supplementary Material
Funding
The study was supported in part by a support grant (P30 CA008748) from Memorial Sloan Kettering Cancer Center’s NIH/National Cancer Institute Cancer Center.
Footnotes
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics statement
Ethics approval: not applicable. The patients provided informed consent for publication of their case details and images.
Supporting Information
Additional Supporting Information can be found in the online version of this article at the publisher’s website.
Data availability
Data are available on request from the corresponding author.
References
- 1.Vestergaard ME, Macaskill P, Holt PE, Menzies SW. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol 2008; 159:669–76. [DOI] [PubMed] [Google Scholar]
- 2.Welch HG, Mazer BL, Adamson AS. The rapid rise in cutaneous melanoma diagnoses. N Engl J Med 2021; 384:72–9. [DOI] [PubMed] [Google Scholar]
- 3.Argenziano G, Cerroni L, Zalaudek I et al. Accuracy in melanoma detection: a 10-year multicenter survey. J Am Acad Dermatol 2012; 67:54–9. [DOI] [PubMed] [Google Scholar]
- 4.Carli P, De Giorgi V, Crocetti E et al. Improvement of malignant/benign ratio in excised melanocytic lesions in the ‘dermoscopy era’: a retrospective study 1997–2001. Br J Dermatol 2004; 150:687–92. [DOI] [PubMed] [Google Scholar]
- 5.Han TY, Chang HS, Lee JHK et al. A clinical and histopathological study of 122 cases of dermatofibroma (benign fibrous histiocytoma). Ann Dermatol 2011; 23:185–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Agero AL, Taliercio S, Dusza SW et al. Conventional and polarized dermoscopy features of dermatofibroma. Arch Dermatol 2006; 142:1431–7. [DOI] [PubMed] [Google Scholar]
- 7.Zaballos P, Puig S, Llambrich A, Malvehy J. Dermoscopy of dermatofibromas: a prospective morphological study of 412 cases. Arch Dermatol 2008; 144:75–83. [DOI] [PubMed] [Google Scholar]
- 8.Jaimes N, Marghoob AA. Triage amalgamated dermoscopic algorithm. J Am Acad Dermatol 2020; 82:1551–2. [DOI] [PubMed] [Google Scholar]
- 9.Fitzpatrick TB, Gilchrest BA. Dimple sign to differentiate benign from malignant pigmented cutaneous lesions. N Engl J Med 1977; 296:1518. [DOI] [PubMed] [Google Scholar]
- 10.Meffert JJ, Peake MF, Wilde JL. ‘Dimpling’ is not unique to dermatofibromas. Dermatology 1997; 195:384–6. [DOI] [PubMed] [Google Scholar]
- 11.Greco V, Cappello M, Megna M et al. Dermoscopic patterns of intradermal naevi. Australas J Dermatol 2020; 61:337–41. [DOI] [PubMed] [Google Scholar]
- 12.Caccavale S, Lupoli A, Alfano R et al. Vascular structures in dermal nevi: a reappraisal. Int J Dermatol 2017; 56:e68–70. [DOI] [PubMed] [Google Scholar]
- 13.Braun RP, Krischer J, Saurat JH. The ‘wobble sign’ in epiluminescence microscopy as a novel clue to the differential diagnosis of pigmented skin lesions. Arch Dermatol 2000; 136:940–2. [DOI] [PubMed] [Google Scholar]
- 14.Zalaudek I, Argenziano G, Soyer HP et al. Dermoscopy of subcorneal hematoma. Dermatologic Surg 2004; 30:1229–32. [DOI] [PubMed] [Google Scholar]
- 15.Elmas ÖF. Subcorneal hematoma as an imitator of acral melanoma: dermoscopic diagnosis. North Clin Istanbul 2019; 7:56–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Saida T, Miyazaki A, Oguchi S et al. Significance of dermoscopic patterns in detecting malignant melanoma on acral volar skin: results of a multicenter study in Japan. Arch Dermatol 2004; 140:1233–8. [DOI] [PubMed] [Google Scholar]
- 17.Cohen LM, Bennion SD, Johnson TW, Golitz LE. Hypermelanotic nevus: clinical, histopathologic, and ultrastructural features in 316 cases. Am J Dermatopathol 1997; 19:23–30. [DOI] [PubMed] [Google Scholar]
- 18.Babino G, Specchio F, Lallas A et al. Tape stripping: a very short-term follow-up procedure for suspicious black lesions. J Am Acad Dermatol 2015; 72:e151–2. [DOI] [PubMed] [Google Scholar]
- 19.Yadav S, Vossaert KA, Kopf AW et al. Histopathologic correlates of structures seen on dermoscopy (epiluminescence microscopy). Am J Dermatopathol 1993; 15:297–305. [DOI] [PubMed] [Google Scholar]
- 20.Braun RP, Rabinovitz HS, Krischer J et al. Dermoscopy of pigmented seborrheic keratosis: a morphological study. Arch Dermatol 2002; 138:1556–60. [DOI] [PubMed] [Google Scholar]
- 21.Minagawa A Dermoscopy–pathology relationship in seborrheic keratosis. J Dermatol 2017; 44:518–24. [DOI] [PubMed] [Google Scholar]
- 22.Yagerman S, Marghoob AA. The ink test: identifying 3-dimensional features of seborrheic keratoses under dermoscopy. JAMA Dermatol 2013; 149:497–8. [DOI] [PubMed] [Google Scholar]
- 23.Zaballos P, Puig S, Malvehy J. Dermoscopy of disseminated superficial actinic porokeratosis. Arch Dermatol 2004; 140:1410. [DOI] [PubMed] [Google Scholar]
- 24.Pizzichetta MA, Canzonieri V, Massone C, Soyer HP. Clinical and dermoscopic features of porokeratosis of Mibelli. Arch Dermatol 2009; 145:91–2. [DOI] [PubMed] [Google Scholar]
- 25.Navarrete-Dechent C, Uribe P, Marghoob A. Ink-enhanced dermoscopy is a useful tool to differentiate acquired solitary plaque porokeratosis from other scaly lesions. J Am Acad Dermatol 2019; 80:e137–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Thomas CJ, Elston DM. Medical pearl: gentian violet to highlight the cornoid lamella in disseminated superficial actinic porokeratosis. J Am Acad Dermatol 2005; 52:513–14. [DOI] [PubMed] [Google Scholar]
- 27.Braun RP, Thomas L, Kolm I et al. The furrow ink test: a clue for the dermoscopic diagnosis of acral melanoma vs nevus. Arch Dermatol 2008; 144:1618–20. [DOI] [PubMed] [Google Scholar]
- 28.Ishihara Y, Saida T, Miyazaki A et al. Early acral melanoma in situ: correlation between the parallel ridge pattern on dermoscopy and microscopic features. Am J Dermatopathol 2006; 28:21–7. [DOI] [PubMed] [Google Scholar]
- 29.Palleschi GM, Cipollini EM, Torchia DA et al. Fibrillar pattern of a plantar acquired melanocytic naevus: correspondence between epiluminescence microscopy and transverse section histology. Clin Exp Dermatol 2006; 31:449–51. [DOI] [PubMed] [Google Scholar]
- 30.Miyazaki A, Saida T, Koga H et al. Anatomical and histopathological correlates of the dermoscopic patterns seen in melanocytic nevi on the sole: a retrospective study. J Am Acad Dermatol 2005; 53:230–6. [DOI] [PubMed] [Google Scholar]
- 31.Braun RP, Kopf AW, Marghoob AA. Comment on dermoscopy patterns of melanocytic nevi on the sole. J Am Acad Dermatol 2006; 55:162–3. [DOI] [PubMed] [Google Scholar]
- 32.Bowling J Fibrillar pattern of an acquired plantar acral melanocytic naevus: correspondence between epiluminescence light microscopy and transverse section histology. Clin Exp Dermatol 2007; 32:103. [DOI] [PubMed] [Google Scholar]
- 33.Kimoto M, Sakamoto M, Iyatomi H, Tanaka M. Three-dimensional melanin distribution of acral melanocytic nevi is reflected in dermoscopy features: analysis of the parallel pattern. Dermatology 2008; 216:205–12. [DOI] [PubMed] [Google Scholar]
- 34.Costello CM, Ghanavatian S, Temkit M et al. Educational and practice gaps in the management of volar melanocytic lesions. J Eur Acad Dermatol Venereol 2018; 32:1450–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Maumi Y, Kimoto M, Kobayashi K et al. Oblique view dermoscopy changes regular fibrillar pattern into parallel furrow pattern. Dermatology 2009; 218:385–6. [DOI] [PubMed] [Google Scholar]
- 36.Benvenuto-Andrade C, Dusza SW, Agero AL et al. Differences between polarized light dermoscopy and immersion contact dermoscopy for the evaluation of skin lesions. Arch Dermatol 2007; 143:329–38. [DOI] [PubMed] [Google Scholar]
- 37.Braun RP, Scope A, Marghoob AA. The ‘blink sign’ in dermoscopy. Arch Dermatol 2011; 147:520. [DOI] [PubMed] [Google Scholar]
- 38.Marghoob AA, Cowell L, Kopf AW, Scope A. Observation of chrysalis structures with polarized dermoscopy. Arch Dermatol 2009; 145:618. [DOI] [PubMed] [Google Scholar]
- 39.Di Stefani A, Campbell TM, Malvehy J et al. Shiny white streaks: an additional dermoscopic finding in melanomas viewed using contact polarised dermoscopy. Australas J Dermatol 2010; 51:295–8. [DOI] [PubMed] [Google Scholar]
- 40.Balagula Y, Braun RP, Rabinovitz HS et al. The significance of crystalline/chrysalis structures in the diagnosis of melanocytic and nonmelanocytic lesions. J Am Acad Dermatol 2012; 67:194.e1–8. [DOI] [PubMed] [Google Scholar]
- 41.Liebman TN, Rabinovitz HS, Dusza SW, Marghoob AA. White shiny structures: dermoscopic features revealed under polarized light. J Eur Acad Dermatology Venereol 2012; 26:1493–7. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data are available on request from the corresponding author.


