Abstract
Background
Patients with coronavirus disease‐2019 (COVID‐19) are characterized by hyperinflammation. Calprotectin (S100A8/S100A9) is a calcium‐ and zinc‐binding protein mainly secreted by neutrophilic granulocytes or macrophages and has been suggested to be correlated with the severity and prognosis of COVID‐19.
Aim
To thoroughly evaluate the diagnostic and prognostic utility of calprotectin in patients with COVID‐19 by analyzing relevant studies.
Methods
PubMed, Web of Science, and Cochrane Library were comprehensively searched from inception to August 1, 2023 to retrieve studies about the application of calprotectin in COVID‐19. Useful data such as the level of calprotectin in different groups and the diagnostic efficacy of this biomarker for severe COVID‐19 were extracted and aggregated by using Stata 16.0 software.
Results
Fifteen studies were brought into this meta‐analysis. First, the pooled standardized mean differences (SMDs) were used to estimate the differences in the levels of circulating calprotectin between patients with severe and non‐severe COVID‐19. The results showed an overall estimate of 1.84 (95% confidence interval [CI]: 1.09–2.60). Diagnostic information was extracted from 11 studies, and the pooled sensitivity and specificity of calprotectin for diagnosing severe COVID‐19 were 0.75 (95% CI: 0.64–0.84) and 0.88 (95% CI: 0.79–0.94), respectively. The AUC was 0.89 and the pooled DOR was 18.44 (95% CI: 9.07–37.51). Furthermore, there was a strong correlation between elevated levels of circulating calprotectin and a higher risk of mortality outcomes in COVID‐19 patients (odds ratio: 8.60, 95% CI: 2.17–34.12; p < 0.1).
Conclusion
This meta‐analysis showed that calprotectin was elevated in patients with severe COVID‐19, and this atypical inflammatory cytokine might serve as a useful biomarker to distinguish the severity of COVID‐19 and predict the prognosis.
Keywords: biomarker, calprotectin, COVID‐19, meta‐analysis, systematic review
This article showed that calprotectin may be a useful biomarker to distinguish the severity and predict the prognosis of COVID‐19.

1. INTRODUCTION
In the last few years, more than 750 million cases of coronavirus disease‐2019 (COVID‐19) have been recorded worldwide and three billion people have been vaccinated. 1 , 2 Common symptoms at the onset of COVID‐19 include fever, cough without or without sputum, sore throat, nasal congestion, dizziness, shortness of breath, fatigue, myalgia, and headache. 3 In fact, the clinical course of this disease can range from asymptomatic infection to severe and even fatal illness. It is estimated that severe cases of COVID‐19 account for about 1/5 of all cases, and the fatality rate for patients over 85 years old is as high as 30%. 4 To date, seven million COVID‐19 deaths have been reported to WHO. 2 As the COVID‐19 pandemic continued, various SARS‐CoV‐2 variants emerged and rapidly spread. 5 A sequence abundance analysis revealed that the Omicron variant is the most widely distributed and transmissible strain, but the disease severity is less. 6 Hospitalization risk, ICU admission rate, mechanical ventilation use rate, and hospitalization period of Omicron variant infection were shorter. 7 COVID‐19 has been shown to be a highly inflammatory disease, and patients with severe symptoms are characterized by elevated serum levels of various cytokines. 8 , 9 Unfortunately, although many cytokines have been evaluated as markers for determining the severity or prognosis of COVID‐19, there is not a universal endorsement.
Calprotectin (S100A8/S100A9) is a calcium‐ and zinc‐bound heterodimeric molecule consisting of two heavy chains and one light chain noncovalently linked with a classical helix‐loop‐helix structural domain. 10 , 11 This atypical inflammatory cytokine is mainly produced by neutrophilic granulocytes or macrophages at sites of inflammation in a calcium‐dependent manner. 12 Calprotectin could modulate cyclooxygenase activity to regulate the expression of prostaglandin analogs, promoting the adhesion of phagocytes to the vascular endothelium and thrombi formation. 13 , 14 Moreover, it has potent antimicrobial effects against various bacterial and fungal pathogens. 15 As an acute phase reactant, the expression level of calprotectin is often increased following infection, trauma, and inflammatory diseases such as rheumatoid arthritis, ankylosing spondylitis, and inflammatory bowel disease, serving as a biomarker to help with the diagnosis. In particular, fecal calprotectin has become one of the most important indicators for monitoring inflammatory bowel disease. 16
For COVID‐19, which is highly transmissible, there is still an unmet need to determine its severity and predict mortality outcomes. Some biomarkers such as interleukin‐6 (IL‐6), C‐reactive protein (CRP), d‐dimer, ferritin, and serum amyloid A (SAA) have been recognized to assess the severity of COVID‐19. 17 , 18 Both circulating and fecal calprotectin were reported to be associated with COVID‐19, and this article focused on the clinical significance of serum and plasma calprotectin for COVID‐19. The aim of this study is to find a biomarker that can predict sudden and unexpected clinical deterioration and disease severity of COVID‐19. Not only for early screening and diagnosis but also to predict the risk of death. A pairwise meta‐analysis was used to reduce bias due to the insufficient number of studies and to improve the accuracy of the conclusion.
2. MATERIALS AND METHODS
The protocol for this review is registered on PROSPERO (International prospective register of systematic reviews) and the registration number is CRD42023452343. This study was reported in line with PRISMA (Preferred Reporting Items for Systematic Reviews and Meta‐Analyses) guidelines. 19 Electronic searches, reference lists screening, study selection, data extraction, methodological quality assessment and pooling of outcome estimates were performed by two authors (ZHR and ZQY) independently. For any ambiguities, a consensus was reached through active discussions or consulting with a third author (DJ). All required data were retrieved from published articles; thus, informed consent or ethical approval was not required.
2.1. Data source and search strategy
Three electronic databases including PubMed, EMBASE, and the Cochrane library (from inception to July 11, 2023) were searched by using a combination of keywords and mesh terms ([Calprotectin, S100A8/A9] AND [COVID‐19, SARS‐COV‐2, Novel coronavirus]) to obtain relevant studies. No restrictions on publication date, language, or the journal of publication were imposed. Titles and abstracts were independently screened by using the selection criteria to determine the eligible studies. Then, the full texts of the remaining records were carefully evaluated to include or exclude these studies accordingly. Negotiation and discussion were reached for any paper with controversial content to eliminate doubts. Lastly, a manual search was conducted in the reference list of related studies (especially reviews, meta‐analyses, and the included studies) for additional eligible literature.
2.2. Inclusion and exclusion criteria
Articles in which adults had COVID‐19 were enrolled and tested for serum or plasma calprotectin were included. These studies should delineate mild and severe, or can indirectly differentiate mild and severe based on intensive care unit (ICU) care, acute respiratory distress syndrome (ARDS), mechanical ventilation, or death. Our exclusion criteria were articles with incomplete information, non‐English language articles, and articles with the control group involved other inflammatory diseases.
2.3. Data extraction
Following data were extracted from qualified research: name of first author, study design, region, inclusion interval of patients, number of patients, comorbidities, outcome of interest, gender, age, death, diagnostic information of calprotectin (true positive/TP, false positive/FP, false negative/FN, and true negative/TN), mean and associated standard deviation (SD) of calprotectin, and odds ratio of mortality.
2.4. Quality assessment
The QUADAS‐2 tool was used to assess the quality of individual studies and the Review Manager software (version 5.3) was used to draw the graph of the quality. The QUADAS‐2 tool consisted of the following four parts: Patients Selection, Index Test, Reference Standard, and Flow and Timing. All parts were evaluated in terms of risk of bias and the former three parts were evaluated in terms of applicability concerns.
2.5. Approach to evidence synthesis and analysis
STATA 16.0 software was used to merge the effect sizes of the diagnostic test, calculating the combined sensitivity, specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR), diagnostic odds ratio (DOR). The range of DOR was 0 to infinity, and the larger the value, the better the diagnostic efficiency. 20 The summary receiver operating characteristic curve (SROC) was constructed to obtain the area under the curve (AUC). The AUC of SROC ranged from 0 to 1: AUC of <0.5 indicates no diagnostic significance; AUC of 0.5–0.7, low diagnostic accuracy; AUC of 0.7–0.9, moderate diagnostic performance; and AUC of >0.9, high diagnostic accuracy. 21 The SROC curve was observed to assess whether there is apparent shoulder‐arm effect. The Spearman correlation coefficient is calculated, and if the p > .05, the heterogeneity caused by the threshold effect is considered to be absent. Then we performed the Cochran Q test and I² test to estimate the existence and severity of heterogeneity. Significant heterogeneity is considered when p < .1 and I² > 50%. Deek's funnel plot was used to assess whether there was publication bias. Symmetry of the funnel plot suggested no publication bias, asymmetry of the funnel plot, and p < .05 of linear regression analysis suggested the existence of publication bias.
If continuous variables appear as mean and standard deviation, these data were extracted directly. If they appear as median and interquartile range, they need to be converted to mean and standard deviation before extraction according to Luo's and Wan's methods. 22 , 23 Due to the different methods and units for assessing the calprotectin level, the standardized mean difference was adopted as the effect size.
3. RESULTS
3.1. Search results
The flow chart of the literature screening process is shown in Figure 1. A total of 647 records were identified from electronic database and 134 studies were left after eliminating duplicates. Through assessing titles/abstracts according predefined criteria, 109 records were excluded. Because of the absence of information about COVID‐19, mechanical ventilation, mortality, ICU, or ARDS, which were outcomes of our interest, four studies were further discarded. Since there was no sufficient data of serum or plasma calprotectin, six others studies were also considered ineligible for this meta‐analysis. Finally, 15 studies 24 , 25 , 26 , 27 , 28 , 29 , 30 , 31 , 32 , 33 , 34 , 35 , 36 , 37 , 38 involving 2753 patients were included in the quantitative analysis. Among the 15 studies, 11 24 , 26 , 27 , 28 , 29 , 31 , 32 , 33 , 34 , 35 , 38 assessed the diagnostic value of calprotectin, five 25 , 31 , 34 , 35 , 36 reported the mortality and eight 24 , 27 , 29 , 30 , 32 , 35 , 36 , 37 presented the level of calprotectin. The methods used to detect calprotectin in all 15 studies were enzyme‐linked immunosorbent assay (ELISA), chemiluminescent immunoassay, turbidimetric assay, and quantitative reverse‐transcription polymerase chain reaction (RT‐qPCR). ELISA was used in eight papers, chemiluminescent immunoassay was used in two papers, turbidimetric assay was used in five papers and RT‐qPCR was used in one paper. And one article used both turbidimetric assays and chemiluminescent immunoassays. The baseline characteristics of enrolled patients are shown in Table 1.
Figure 1.
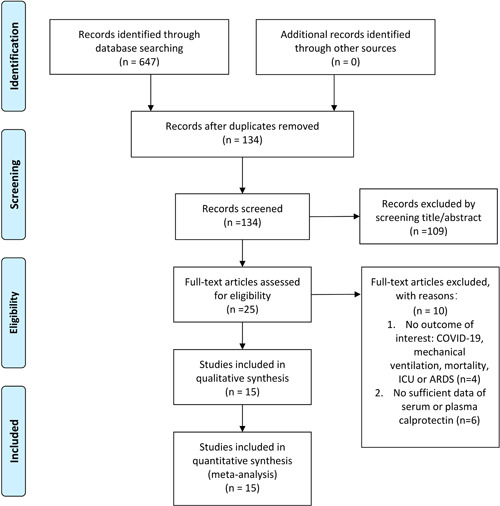
PRISMA 2009 flow diagram. A total of 647 records were retrieved, after inclusion and exclusion criteria, the final 15 studies were included.
Table 1.
Baseline characteristics of included studies.
| Autor | Study design | Region | Inclusion interval of patients | Inclusion number of patients | Comorbidities | Outcome of interest | Male, n (%) | Age, years | % Deaths | Gold standard | Mild COVID‐19 calprotectin range | Severe |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Shokri‑Afra et al. 28 | Prospective study | Iran | 2020.09–2020.11 | COVID‐19 n = 76, non‐COVID n = 24 | Diabetes, hypertension, cardiovascular disease, chronic kidney disease, chronic liver disease | Serum calprotectin | COVID‐19 n = 39(51.3%), non‐COVID‐19 n = 16 (66.7%) | COVID‐19 52.1 ± 17.2, non‐COVID‐19 52.83 ± 22.61a | 5.30% | ELISA | NR | NR |
| Cardiero et al. 25 | Retrospective study | Italy | 2020.03.10–2020.04.30; 2020.12.08–2021.04.30 | COVID‐19 n = 195 | Decreased lung function, diabetes, heart disease, hypertension | Circulating calprotectin | 94 (48.2%) | 55 (34–67)b | 5.13% | Particle‐enhanced turbidimetric immunoassay and chemiluminescent immunoassay | NR | NR |
| Norman et al. 26 | NR | USA | 2020 spring | COVID n = 157 | Diabetes, heart disease, renal disease, lung disease, autoimmune, cancer, history of stroke, obesity, hypertension, history of smoking | Circulating calprotectin | 92 (59%) | 58 ± 17a | 22% | Chemiluminescent immunoassay | NR | NR |
| Kaya et al. 27 | Retrospective cross‐sectional cohort study | Turkey | 2020.11.01–2020.12.10 | COVID‐19 n = 80 | Diabetes mellitus, hypertension, coronary artery disease, renal failure, chronic respiratory disease | Circulating calprotectin | ICU n = 24 (63.2%); non‐ICU n = 19(45.2%) | ICU 70.8 ± 12.8, non‐ICU 62.5 ± 17.2a | ICU 57.1% | Immunoturbidimetric assay | 37.9 (30.3–46.2)b (ng/mL) | 44.8 (34.5–61.5)b |
| Shokri‑Afra et al. 24 | Prospective study | Iran | 2020.09–2020.11 | COVID‐19 n = 70, non‐COVID 19 n = 19 | Hypertension, Diabetes mellitus, cardiovascular disease, dyslipidemia chronic kidney disease, Chronic liver disease, | Serum calprotectin | COVID‐19 n = 36 (51.4); non‐COVID 19 n = 12 (63.2) | COVID‐19 51.47 ± 17.28, non‐COVID 19 47.74 ± 21.27a | COVID‐19 5.71% | ELISA | 2013 ± 1074a (ng/mL) | 5436 ± 940a (ng/mL) |
| Lee et al. 29 | Retrospective study | Korea | 2020.07.04–2020.11.15 | COVID‐19 n = 105 | Diabetes, hypertension, cardiovascular diseases, renal disease, cancer, autoimmune disease, dementia/mental retardation, pregnancy, hyperlipidemia, hepatitis B virus infection, obesity | Serum calprotectin | 45 (42.9%) | 68.0 ± 18.8a | 8.60% | ELISA | 2.60 (1.40–5.28)b (μg/mL) | 12.60 (8.10–18.50)b |
| Shrivastava et al. 30 | Retrospective study | India | 2020.05–2020.09 | Healthy n = 19, mild n = 32, severe n = 31 | NR | Circulating calprotectin | Healthy n = 10 (53%), mild n = 19 (59%); severe n = 21 (68%) | Healthy 27 (18–33), mild 40 (15‐67), severe 59 (30–85)b | NR | ELISA | 16,589 ± 2651a (ng/mL) | 18,183 ± 2512a |
| Morgane Ducastle et al. 31 | Retrospective monocentric study | France | 2020.04‐2020.05 | Mild n = 31, moderate n = 36, severe n = 36, critical n = 57 | Overweight, hypertension, diabetes, chronic kidney disease, cardiovascular disease, chronic respiratory failure, systemic autoimmune Disease | Plasma calprotectin | Mild n = 11 (35.5%); moderate n = 20 (55.6%); severe n = 18 (50%); critical n = 43 (75.4%) | Mild 47.4 (32.4–57.1), moderate 63.9 (49.1–75.8), severe 64.7 (58.5–74.9), critical 61.9 (51.2–73.9)b | Critical 29.8% | Turbidimetric assay | NR | NR |
| Shi et al. 32 | Prospective study | USA | NR | Room air n = 41, noninvasive supplemental oxygen n = 71, mechanical ventilation n = 60 | Diabetes, ischemic heart disease, renal disease, lung disease, autoimmune, cancer, history of stroke, obesity, hypertension, immune deficiency, history of smoking | Circulating calprotectin | COVID‐19 n = 97 (56%) | 61.48 ± 17.7a | NR | ELISA | 3365 ± 3146a (ng/mL) | 8039 ± 7031a |
| Silvin et al. 33 | Cohort | France | NR | Negative control n = 10, mild n = 3, moderate n = 4, severe n = 7 | Overweight, cardiovascular, diabetes, respiratory, cancer | Plasma calprotectin | All n = 12 (50%); negative control n = 6 (60%), mild n = 1 (33%), moderate n = 1 (25%), severe n = 4 (57%) | 60 (54–68)b | All n = 1 (4%), severe n = 1(14%) | RT‐qPCR | NR | NR |
| Chen et al. 34 | Retrospective study | China | NR | ICU n = 40; non‐ICU n = 81 | Hypertension, diabetes, coronary artery heart disease, cancer, COPD, immunodeficiency | Serum calprotectin | All n = 77 (63.6%); non‐ICU n = 49 (60.5%), ICU n = 28 (70.0%) | 63 (53–70)b | All n = 36 (29.8%); non‐ICU n = 3 (3.7%), ICU n = 33 (82.5%) | ELISA | NR | NR |
| Bauer et al. 35 | Prospective study | Germany | NR | ICU n = 8, MOF within 72 h n = 4, total MOF n = 6, 90‐day mortality n = 2 | NR | Serum calprotectin | COVID‐19 n = 8 (42%) | 67.6 (53.9–72.0)b | 10.50% | Turbidimetric assay | 2.08 (1.36–2.59)b (mg/L) | 3.77 (1.90–5.16)b |
| Luis et al. 36 | Observational study | Spain | 2020.03‐2020.04 | Survivors n = 58; nonsurvivors n = 8 | Hypertension, diabetes mellitus, non‐asthma respiratory disease, cardiovascular disease, chronic kidney disease, immunosuppression | Circulating calprotectin | 43 (65.2%) | 61 ± 16a | 12.1% (8/66) | Particle‐enhanced turbidimetric immunoassay | 6.0 (2.8–10.8) b (mg/dL) | 19.3 (10.4–30.5)b |
| Gupta et al. 37 | NR | Dubai | 2020.05‐ 2020.06 | Moderate n = 15, severe n = 15, control n = 10 | NR | Plasma calprotectin | NR | Moderate 48 ± 8.1, severe 55 ± 8.3a | 33.3% (5/15) | ELISA | 0.8972 ± 0.1868a (ng/mL) | 3.626 ± 0.748a (ng/mL) |
| Kassianidis et al. 38 | NR | Greece | 2020.04‐2020.11 | Control n = 40, asymptomatic n = 19; moderate n = 42, severe n = 78, ARDS and were on MV n = 42 | Type 2 diabetes mellitus, chronic heart failure, Chronic renal disease, coronary heart disease, dyslipidemia, hypothyroidism, hypertension, stroke, atrial fibrillation, COPD | Serum calprotectin | Control n = 28(70%), asymptomatic n = 12(63.2%), moderate n = 25(59.5%), severe n = 56(71.8%), ARDS and were on MV n = 35(83.3%) | Control 58.3 ± 15.8, asymptomatic 59.5 ± 10.8, moderate 55.3 ± 14.8, severe 61.4 ± 13.9, ARDS and were on MV 64.9 ± 12.8a | NR | ELISA | NR | NR |
Abbreviations: ARDS, acute respiratory distress syndrome; COPD, chronic obstructive pulmonary disease; COVID‐19, coronavirus disease‐2019; ELISA, enzyme‐linked immunosorbent assay; ICU, intensive care unit; MOF, multiorgan failure; MV, mechanical ventilation; NR, not reported.
Age, mean ± SD.
Age, median (first quantile‐third quantile).
3.2. Quality assessment
The studies we included were all of high quality (Figure 2). The risk assessment of patient selection was not satisfactory, four studies did not avoid case‐control studies, and six studies did not include consecutive or random sample of patients. This is the most important cause of patient selection bias.
Figure 2.

Review authors' judgements about each risk of bias item for included studies. (A) Risk of bias summary; (B) risk of bias graph presented as percentages.
3.3. Severity of COVID‐19 and calprotectin levels
On account of the I 2 = 91.2%, a random effect model was used and the pooled SMD of calprotectin between patients with severe COVID‐19 and those with nonsevere COVID‐19 was 1.84 (95% confidence interval [CI]: 1.09–2.60) (Figure 3). The symmetric distribution of Deek's funnel plot and the result of Egger's test result (p = .124) both indicated low publication bias (Figure SS1). Next, a sensitivity analysis of the pooled results of changes in calprotectin levels was performed by excluding the eight included studies individually (Figure SS2). The results showed no significant change in the final effect size, which demonstrated that the conclusion was robust and reliable.
Figure 3.
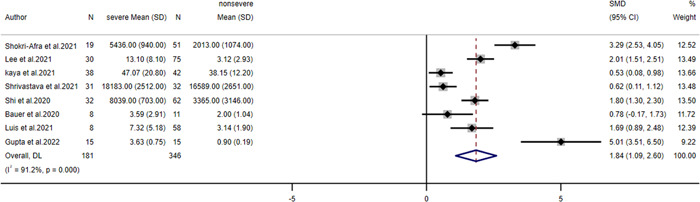
Forest plot of calprotectin levels.
3.4. Diagnostic performance of calprotectin in predicting COVID‐19 severity
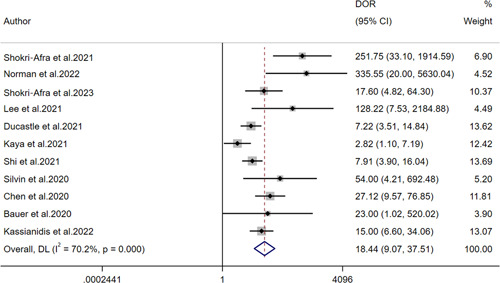
The pooled sensitivity, specificity, PLR, NLR, and DOR as well as the associated 95% CI were 0.75 (95% CI: 0.64–0.84), 0.88 (95% CI: 0.79–0.94), 6.4 (95% CI: 3.5–11.6), 0.28 (95% CI: 0.19–0.42), 18.44 (95% CI: 9.07–37.51), respectively (Figures 4 and 5). All the above indicators suggested that calprotectin had a satisfactory efficiency for determining the severity of COVID‐19. Furthermore, the SROC curve gave an AUC of 0.89 (95% CI: 0.86–0.91) (Figure 6). The publication bias was assessed by plotting Deek's funnel plot (Figure SS3). It is obvious that the 11 included studies were evenly distributed on both sides of the regression line. The difference was not statistically significant (p = .83), suggesting that the included studies had a low risk of publication bias.
Figure 4.
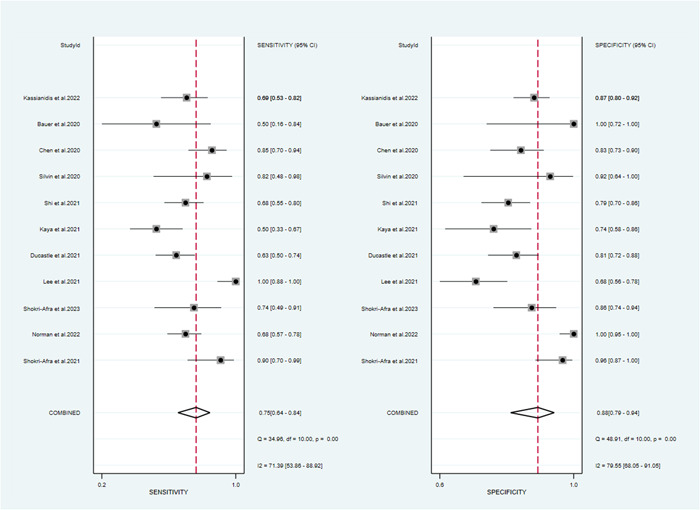
Forest plots of sensitivity and specificity of calprotectin diagnosis value for COVID‐19 severity.
Figure 5.
Forest plot for the combined diagnostic odds ratio of calprotectin.
Figure 6.
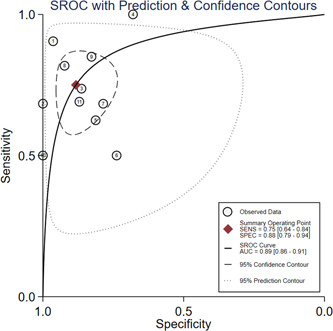
Summary receiver operating characteristic curve of COVID‐19 severity detected by calprotectin.
3.5. The mortality rates
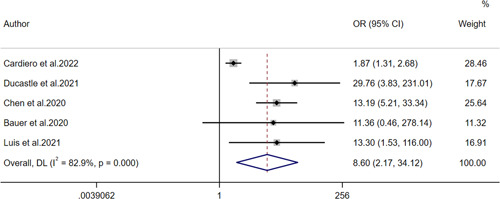
Five studies provided data related to mortality outcomes, and OR with 95%CI was used to evaluate mortality outcomes (Figure 7). High heterogeneity can be observed in the forest plots (I 2 = 82.9%, p < .001). Therefore, the random effects model was selected. The pooled OR and 95% CI were 8.60 (95% CI: 2.17–34.12), unveiling that COVID‐19 mortality is positively correlated with the calprotectin level.
Figure 7.
Forest plot for the odds ratio of mortality.
4. DISCUSSION
After infection, neutrophil counts increased and natural killer cells decreased, proinflammatory cytokines such as tumor necrosis factor‐α (TNF‐α), IL‐1, IL‐2, IL‐6, and IFN‐γ‐inducible protein 10 (IP‐10) was also abnormally elevated. 39 , 40 Due to the intrinsic cytotoxic and proinflammatory properties of calprotectin, the level of this atypical cytokine is demonstrated to be associated with the development of inflammatory/autoimmune diseases (e.g., rheumatoid arthritis, systemic lupus erythematosus, and connective tissue diseases). 41 , 42 , 43 , 44 COVID‐19 is actually characterized by a cytokine storm, in which the immune system plays a regulatory role in summoning cytokines to attack pathogens. 45 This meta‐analysis was performed to assess the diagnostic value of circulating calprotectin in the severity of COVID‐19. Eventually, we found that levels of circulating calprotectin were positively associated with the severity and mortality outcome of COVID‐19 patients. Circulating calprotectin has clinical significance and practical value in diagnosing the severity and predicting the prognosis of patients with COVID‐19.
Calprotectin is widely distributed in peripheral blood and fecal. At present, fecal calprotectin has been studied more, which is recognized to be related to intestinal injury. 46 The cause of gastrointestinal reaction caused by SARS‐CoV‐2 virus may be related to angiotensin‐converting enzyme 2 (ACE2) receptor binding. ACE2 receptors are highly expressed in intestinal cells (e.g., small intestine epithelial cells and large intestine epithelial cells) and are the main route by which viruses enter the intestine. 47 In addition, severe COVID‐19 patients often have gastrointestinal complications such as ileus, gastrointestinal bleeding, intestinal ischemia, and even intestinal perforation. 48 Fecal calprotectin is often considered to be an indicator associated with intestinal inflammation, and its most important use is in the differential diagnosis of inflammatory bowel diseases (IBD) and irritable bowel syndrome. 49 When inflammation occurs in the intestinal tract, fecal calprotectin can be detected in the feces due to the increased permeability of the intestinal mucosa, which allows neutrophils to infiltrate and release calprotectin. 50 Intestinal injury responds to both acute and chronic inflammatory conditions. Many studies have shown that fecal calprotectin levels are highly expressed in patients with COVID‐19. 51 , 52 , 53 Therefore, SARS‐CoV‐2 virus attacks the ACE2 receptor of gastrointestinal cells after infection, causing gastrointestinal inflammation and complications, which may be the main reason for the increase of calprotectin. But the study had also shown high levels of fecal calprotectin in patients with COVID‐19, regardless of gastrointestinal symptoms. 54 However, the correlation between the specific mechanism and infection diagnosis is still unclear, which is an important line for future research. Circulating calprotectin has been recognized by some studies as one of the most relevant predictors of COVID‐19 disease severity, and transcriptome analysis has shown that S100A8 and S100A9 are the few genes that are significantly upregulated after infection of SARS‐CoV‐2. 55 , 56 From the studies searched, calprotectin is expected to be an accurate and effective biomarker for COVID‐19.
In a meta‐analysis conducted by Udeh et al., 57 eight quantitative literatures were included, and the pooled SMD of calprotectin between the cases with severe COVID‐19 and other cases was 1.34 (95% CI: 0.77–1.91). Mao et al. 58 also included eight quantitative literatures for meta‐analysis, and the pooled SMD was 1.18 (95% CI: 0.74–1.62). The current study included eight articles for meta‐analysis with an effect size of SMD. Compared with the above two studies, the results obtained were more timely, more comprehensive and more persuasive. In the included paper, four methods were applied to detect the scope of calprotectin. Because the applied methods and units were not uniform, we applied SMD to combine them. It can be observed that calprotectin of severe patients is significantly higher than that of mild patients. The normal values of calprotectin measured by different methods can be seen in Table 1. This meta‐analysis showed that the pooled SMD was 1.84 (95% CI: 1.09–2.60). These results suggest that the higher the levels of circulating calprotectin, the more likely patients were severe COVID‐19. In a meta‐analysis of adverse outcomes, OR and its 95% CI were used to assess the significance of circulating calprotectin in predicting mortality in COVID‐19 patients. The results of the meta‐analysis showed that OR and 95% CI were 8.60 (2.17–34.12), which suggested that high expression of circulating calprotectin was a poor prognostic marker for COVID‐19.
In recent years, studies on calprotectin and COVID‐19 have been widely carried out worldwide and were increasing year by year. However, our study was the first meta‐analysis to assess the diagnostic value of calprotectin in the severity of COVID‐19. In this study, the effect results of 11 diagnostic tests in 11 articles were combined, and the pooled sensitivity and specificity of calprotectin in diagnosing the severity of COVID‐19 were 0.75 (95% CI: 0.64–0.84), 0.88 (95% CI: 0.79–0.94), respectively. The PLR for circulating calprotectin on COVID‐19 severity was 6.4 (95% CI: 3.5–11.6). Patients with severe COVID‐19 were 6.4 times more likely to detect positive for circulating calprotectin than patients with mild COVID‐19. The NLR for circulating calprotectin on COVID‐19 severity was 0.28 (95% CI: 0.19–0.42). This indicated that the probability of circulating calprotectin being diagnosed as mild when the gold standard diagnosis was severe was 0.28. The pooled DOR for circulating calprotectin on COVID‐19 severity was 18.44 (95% CI: 9.07–37.51). It suggested that circulating calprotectin had a high diagnostic value for COVID‐19 severity. In this meta‐analysis, circulating calprotectin was moderately effective in the diagnosis of COVID‐19 severity and had outstanding diagnostic value for COVID‐19.
As a potential COVID‐19 biomarker, calprotectin has multiple advantages. First, calprotectin could be used both as a diagnostic tool and as a severity stratification tool for COVID‐19 patients. The levels of circulating calprotectin could discriminate patients who develop a severe form of COVID‐19. Meanwhile, it had an excellent predictive value for adverse outcomes in patients with COVID‐19. Second, the levels of circulating calprotectin correlated better with the severity of COVID‐19 compared with a host of other traditional biomarkers including IL‐6, CRP, ESR, and D‐dimer. 32 , 34 , 36 , 59 Third, circulating calprotectin offers a clear kinetic advantage as the first indication of acute inflammation since it does not require de novo synthesis. 60 Moreover, compared with other more common biomarkers, calprotectin had the advantage that marker levels did not increase significantly during ICU due to potential bacterial infection and other influences. 61 Last but not least, given the encouraging results in treating inflammatory disorders, 62 S100A8/S100A9 inhibitors (e.g., paquinimod) may be a promising treatment option for severe or critically ill COVID‐19 individuals. 63
Although there were multiple original studies evaluating the value of calprotectin in COVID‐19 from the aspects of expression level, diagnostic, and prognostic value, their results were controversial. This was the first time to comprehensively assess this issue by using a method of pair‐wise meta‐analysis. By utilizing rigorous statistical methods and multiple outcome estimates, the pooled results were robust and convincing. However, there were limitations existing in the current meta‐analysis. First, all included studies were observational ones and did not specify whether blinding was used or not. Second, after pooling effect sizes, high heterogeneity was found across studies. We guessed that it was caused by different races and various parts and ways of detecting calprotectin. Third, the small number of included patients reduced the strength of the current meta‐analysis. Currently, separate analysis assessing the prognostic value of serum calprotectin regarding intubation is unavailable. Fourth, the mechanism of calprotectin is still in the initial stage. The specific reasons for the elevated expression of calprotectin are unclear as to how it works in viral infections.
5. CONCLUSION
In a word, calprotectin was significantly elevated in patients with severe COVID‐19. This atypical cytokine may serve as a novel biomarker for predicting the severity of COVID‐19 with satisfactory diagnostic efficiency. Meanwhile, mortality in patients with COVID‐19 tends to be positively correlated with calprotectin levels. More high‐quality studies are still needed to validate the value of calprotectin as a diagnostic and prognostic marker for COVID‐19, and clarify the function of this heterodimer in disease progression.
AUTHOR CONTRIBUTIONS
Haoran Zhang: Conceptualization; data curation; formal analysis; investigation; methodology; project administration; software; writing—original draft. Qingyu Zhang: Conceptualization; data curation; formal analysis; funding acquisition; investigation; methodology; project administration; software; writing—original draft. Kun Liu: Formal analysis; software; writing—original draft. Zenong Yuan: Writing—review and editing. Xiqiang Xu: Conceptualization; investigation; methodology; software; supervision; writing—review and editing. Jun Dong: Conceptualization; funding acquisition; project administration; software; supervision; validation; writing—review and editing.
CONFLICT OF INTEREST STATEMENT
The authors declare no conflict of interest.
Supporting information
Supplementary Figure 1 Deeks’ funnel plot of calprotectin levels
Supplementary Figure 2 Sensitivity analysis was used to assess whether there was a unstable experiment
Supplementary Figure 3 Deeks’ funnel plot of calprotectin diagnosis value for COVID‐19 severity
ACKNOWLEDGMENTS
This work was partially supported by the Young Taishan Scholars Program of Shandong Province (Grant number tsqn201909183), the Academic Promotion Program of Shandong First Medical University (Grant number 2020RC008), the Natural Science Foundation of Shandong Province (Grant numbers ZR2020QH072 and ZR2020MH098), the National Natural Science Foundation of China (Grant numbers 82302682), and the Jinan Clinical Medical Science and Technology Innovation Program (grant number 202328067).
Zhang H, Zhang Q, Liu K, Yuan Z, Xu X, Dong J. Elevated level of circulating calprotectin correlates with severity and high mortality in patients with COVID‐19. Immun Inflamm Dis. 2024;12:e1212. 10.1002/iid3.1212
Haoran Zhang and Qingyu Zhang contributed equally to this study.
Contributor Information
Xiqiang Xu, Email: xxqsdjn@126.com.
Jun Dong, Email: slyydj2019@163.com.
DATA AVAILABILITY STATEMENT
All data generated or analyzed in this study are available in the article.
REFERENCES
- 1. Harky A, Ala'Aldeen A, Butt S, Duric B, Roy S, Zeinah M. COVID‐19 and multiorgan response: the long‐term impact. Curr Probl Cardiol. 2023;48(9):101756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. WHO . Coronavirus Disease (COVID‐19) Dashboard. 2020. Accessed November 26, 2023. https://data.who.int/dashboards/covid19/deaths?n=c
- 3. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497‐506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Tian S, Hu N, Lou J, et al. Characteristics of COVID‐19 infection in Beijing. J Infect. 2020;80(4):401‐406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Zhang Y, Zhang H, Zhang W. SARS‐CoV‐2 variants, immune escape, and countermeasures. Front Med. 2022;16(2):196‐207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Chatterjee S, Bhattacharya M, Nag S, Dhama K, Chakraborty C. A detailed overview of SARS‐CoV‐2 omicron: its sub‐variants, mutations and pathophysiology, clinical characteristics, immunological landscape, immune escape, and therapies. Viruses. 2023;15(1):167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Sun C, Xie C, Bu G‐L, Zhong L‐Y, Zeng M‐S. Molecular characteristics, immune evasion, and impact of SARS‐CoV‐2 variants. Signal Transduct Target Ther. 2022;7(1):202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Maity S, Santra A, Hebbani AV, et al. Targeting cytokine storm as the potential anti‐viral therapy: implications in regulating SARS‐CoV‐2 pathogenicity. Gene. 2023;881:147612. [DOI] [PubMed] [Google Scholar]
- 9. Zheng M, Gao Y, Wang G, et al. Functional exhaustion of antiviral lymphocytes in COVID‐19 patients. Cell Mol Immunol. 2020;17(5):533‐535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Fagerhol MK, Dale I, Anderson T. Release and quantitation of a leucocyte derived protein (L1). Scand J Haematol. 1980;24(5):393‐398. [Google Scholar]
- 11. Heizmann W, Heizmann CW. S100 proteins: structure, functions and pathology. Front Biosci. 2002;7:d1356‐d1368. [DOI] [PubMed] [Google Scholar]
- 12. Vogl T, Pröpper C, Hartmann M, et al. S100A12 is expressed exclusively by granulocytes and acts independently from MRP8 and MRP14. J Biol Chem. 1999;274(36):25291‐25296. [DOI] [PubMed] [Google Scholar]
- 13. Ehrchen JM, Sunderkötter C, Foell D, Vogl T, Roth J. The endogenous Toll‐like receptor 4 agonist S100A8/S100A9 (calprotectin) as innate amplifier of infection, autoimmunity, and cancer. J Leukoc Biol. 2009;86(3):557‐566. [DOI] [PubMed] [Google Scholar]
- 14. Inciarte‐Mundo J, Frade‐Sosa B, Sanmartí R. From bench to bedside: calprotectin (S100A8/S100A9) as a biomarker in rheumatoid arthritis. Front Immunol. 2022;13:1001025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ostermann L, Seeliger B, David S, et al. S100A9 is indispensable for survival of pneumococcal pneumonia in mice. PLoS Pathog. 2023;19(7):e1011493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Ashton JJ, Beattie RM. Inflammatory bowel disease: recent developments. Arch Dis Child. 2023. Epub ahead of print. 10.1136/archdischild-2023-325668 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Huang I, Pranata R, Lim MA, Oehadian A, Alisjahbana B. C‐reactive protein, procalcitonin, D‐dimer, and ferritin in severe coronavirus disease‐2019: a meta‐analysis. Therap Adv Resp Dis. 2020;14:175346662093717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Qin R, He L, Yang Z, et al. Identification of parameters representative of immune dysfunction in patients with severe and fatal COVID‐19 infection: a systematic review and meta‐analysis. Clin Rev Allergy Immunol. 2023;64(1):33‐65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Moher D. Preferred reporting items for systematic reviews and meta‐analyses: the PRISMA statement. Ann Intern Med. 2009;151(4):264‐269. [DOI] [PubMed] [Google Scholar]
- 20. Glas AS, Lijmer JG, Prins MH, Bonsel GJ, Bossuyt PMM. The diagnostic odds ratio: a single indicator of test performance. J Clin Epidemiol. 2003;56(11):1129‐1135. [DOI] [PubMed] [Google Scholar]
- 21. Jones CM, Athanasiou T. Summary receiver operating characteristic curve analysis techniques in the evaluation of diagnostic tests. Ann Thorac Surg. 2005;79(1):16‐20. [DOI] [PubMed] [Google Scholar]
- 22. Luo D, Wan X, Liu J, Tong T. Optimally estimating the sample mean from the sample size, median, mid‐range, and/or mid‐quartile range. Stat Methods Med Res. 2018;27(6):1785‐1805. [DOI] [PubMed] [Google Scholar]
- 23. Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014;14:135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Shokri‐Afra H, Alikhani A, Moradipoodeh B, Noorbakhsh F, Fakheri H, Moradi‐Sardareh H. Elevated fecal and serum calprotectin in COVID‐19 are not consistent with gastrointestinal symptoms. Sci Rep. 2021;11(1):22001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Cardiero G, Palma D, Vano M, et al. Calprotectin levels and neutrophil count are prognostic markers of mortality in COVID‐19 patients. Diagnostics. 2022;12(10):2554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Norman GL, Navaz SA, Kanthi Y, et al. Circulating calprotectin as a predictive and severity biomarker in patients with COVID‐19. Diagnostics. 2022;12(6):1324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Kaya T, Yaylacı S, Nalbant A, et al. Serum calprotectin as a novel biomarker for severity of COVID‐19 disease. Irish J Med Sci. 2022;191(1):59‐64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Shokri‐Afra H, Moradi M, Musavi H, et al. Serum calprotectin can indicate current and future severity of COVID‐19. J Clin Lab Anal. 2023;37(1):e24809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Lee A, Nahm CH, Lee JS, Lee MK, Lee KR. Assessment of antiphospholipid antibodies and calprotectin as biomarkers for discriminating mild from severe COVID‐19. J Clin Lab Anal. 2021;35(11):e24004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Shrivastava S, Chelluboina S, Jedge P, et al. Elevated levels of neutrophil‐activated proteins, alpha‐defensins (DEFA1), calprotectin (S100A8/A9) and myeloperoxidase (MPO) are associated with disease severity in COVID‐19 patients. Front Cell Infect Microbiol. 2021;11:751232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Ducastel M, Chenevier‐Gobeaux C, Ballaa Y, et al. Oxidative stress and inflammatory biomarkers for the prediction of severity and ICU admission in unselected patients hospitalized with COVID‐19. Int J Mol Sci. 2021;22(14):7462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Shi H, Zuo Y, Yalavarthi S, et al. Neutrophil calprotectin identifies severe pulmonary disease in COVID‐19. J Leukoc Biol. 2021;109(1):67‐72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Silvin A, Chapuis N, Dunsmore G, et al. Elevated calprotectin and abnormal myeloid cell subsets discriminate severe from mild COVID‐19. Cell. 2020;182(6):1401‐1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Chen L, Long X, Xu Q, et al. Elevated serum levels of S100A8/A9 and HMGB1 at hospital admission are correlated with inferior clinical outcomes in COVID‐19 patients. Cell Mol Immunol. 2020;17(9):992‐994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Bauer W, Diehl‐Wiesenecker E, Ulke J, et al. Outcome prediction by serum calprotectin in patients with COVID‐19 in the emergency department. J Infect. 2021;82(4):84‐123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Luis García de Guadiana Romualdo R, Mulero MDR, Olivo MH, et al. Circulating levels of GDF‐15 and calprotectin for prediction of in‐hospital mortality in COVID‐19 patients: a case series. J Infect. 2021;82(2):e40‐e42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Gupta A, Al‐Tamimi AO, Halwani R, Alsaidi H, Kannan M, Ahmad F. Lipocalin‐2, S100A8/A9, and cystatin C: potential predictive biomarkers of cardiovascular complications in COVID‐19. Exp Biol Med. 2022;247(14):1205‐1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Kassianidis G, Siampanos A, Poulakou G, et al. Calprotectin and imbalances between acute‐phase mediators are associated with critical illness in COVID‐19. Int J Mol Sci. 2022;23(9):4894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Del Valle DM, Kim‐Schulze S, Huang HH, et al. An inflammatory cytokine signature predicts COVID‐19 severity and survival. Nat Med. 2020;26(10):1636‐1643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Mastboim NS, Angel A, Shaham O, et al. An immune‐protein score combining TRAIL, IP‐10 and CRP for predicting severe COVID‐19 disease. Cytokine. 2023;169:156246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Gernert M, Schmalzing M, Tony HP, Strunz PP, Schwaneck EC, Fröhlich M. Calprotectin (S100A8/S100A9) detects inflammatory activity in rheumatoid arthritis patients receiving tocilizumab therapy. Arthritis Res Ther. 2022;24(1):200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Davies JC, Midgley A, Carlsson E, et al. Urine and serum S100A8/A9 and S100A12 associate with active lupus nephritis and may predict response to rituximab treatment. RMD Open. 2020;6(2):e001257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Šumová B, Cerezo LA, Szczuková L, et al. Circulating S100 proteins effectively discriminate SLE patients from healthy controls: a cross‐sectional study. Rheumatol Int. 2019;39(3):469‐478. [DOI] [PubMed] [Google Scholar]
- 44. Manfredi M, Van Hoovels L, Benucci M, et al. Circulating calprotectin (cCLP) in autoimmune diseases. Autoimmun Rev. 2023;22(5):103295. [DOI] [PubMed] [Google Scholar]
- 45. Tyagi K, Rai P, Gautam A, et al. Neurological manifestations of SARS‐CoV‐2: complexity, mechanism and associated disorders. Eur J Med Res. 2023;28(1):307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Adriana DN, Sugihartono T, Nusi IA, et al. Role of fecal calprotectin as a hypoxic intestinal damage biomarker in COVID‐19 patients. Gut Pathog. 2022;14(1):34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Saviano A, Brigida M, Petruzziello C, et al. Intestinal damage, inflammation and microbiota alteration during COVID‐19 infection. Biomedicines. 2023;11(4):1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Kaafarani HMA, El Moheb M, Hwabejire JO, et al. Gastrointestinal complications in critically ill patients with COVID‐19. Ann Surg. 2020;272(2):e61‐e62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Chang MH, Chou JW, Chen SM, et al. Faecal calprotectin as a novel biomarker for differentiating between inflammatory bowel disease and irritable bowel syndrome. Mol Med Rep. 2014;10(1):522‐526. [DOI] [PubMed] [Google Scholar]
- 50. Seethaler B, Basrai M, Neyrinck AM, et al. Biomarkers for assessment of intestinal permeability in clinical practice. Am J Physiol Gastrointest Liver Physiol. 2021;321(1):G11‐G17. [DOI] [PubMed] [Google Scholar]
- 51. Toma L, Dodot M, Zgura A, et al. Calprotectin in viral systemic infections—COVID‐19 versus hepatitis C virus. Clin Exp Med. 2021;22(2):311‐317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Giuffrè M, Di Bella S, Sambataro G, et al. COVID‐19‐induced thrombosis in patients without gastrointestinal symptoms and elevated fecal calprotectin: hypothesis regarding mechanism of intestinal damage associated with COVID‐19. Trop Med Infect Dis. 2020;5(3):147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Effenberger M, Grabherr F, Mayr L, et al. Faecal calprotectin indicates intestinal inflammation in COVID‐19. Gut. 2020;69(8):1543‐1544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Zerbato V, Di Bella S, Giuffrè M, et al. High fecal calprotectin levels are associated with SARS‐CoV‐2 intestinal shedding in COVID‐19 patients: a proof‐of‐concept study. World J Gastroenterol. 2021;27(22):3130‐3137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Mahler M, Meroni PL, Infantino M, Buhler KA, Fritzler MJ. Circulating calprotectin as a biomarker of COVID‐19 severity. Expert Rev Clin Immunol. 2021;17(5):431‐443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Chandrashekar DS, Athar M, Manne U, Varambally S. Comparative transcriptome analyses reveal genes associated with SARS‐CoV‐2 infection of human lung epithelial cells. Sci Rep. 2021;11(1):16212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Udeh R, Advani S, de Guadiana Romualdo LG, Dolja‐Gore X. Calprotectin, an emerging biomarker of interest in COVID‐19: a systematic review and meta‐analysis. J Clin Med. 2021;10(4):775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Mao Q, Wang C, Wen W, et al. A meta‐analysis of the association between calprotectin and the severity of COVID‐19. J Infect. 2022;84(3):e31‐e33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Mago S, Vaziri H, Tadros M. The usefulness of fecal calprotectin in the era of the COVID‐19 pandemic. Gastroenterology. 2021;160(7):2623‐2625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Giuffrè M, Vetrugno L, Di Bella S, Moretti R, Berretti D, Crocè LS. Calprotectin and SARS‐CoV‐2: a brief‐report of the current literature. Healthcare. 2021;9(8):956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Nevejan L, Strypens T, Van Nieuwenhove M, et al. Serial measurement of circulating calprotectin as a prognostic biomarker in COVID‐19 patients in intensive care setting. Clin Chem Lab Med (CCLM). 2023;61(3):494‐502. [DOI] [PubMed] [Google Scholar]
- 62. Wu M, Chen Y, Xia H, et al. Transcriptional and proteomic insights into the host response in fatal COVID‐19 cases. Proc Natl Acad Sci USA. 2020;117(45):28336‐28343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Bengtsson AA, Sturfelt G, Lood C, et al. Pharmacokinetics, tolerability, and preliminary efficacy of paquinimod (ABR‐215757), a new quinoline‐3‐carboxamide derivative: studies in lupus‐prone mice and a multicenter, randomized, double‐blind, placebo‐controlled, repeat‐dose, dose‐ranging study in patients with systemic lupus erythematosus. Arthritis Rheum. 2012;64(5):1579‐1588. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Figure 1 Deeks’ funnel plot of calprotectin levels
Supplementary Figure 2 Sensitivity analysis was used to assess whether there was a unstable experiment
Supplementary Figure 3 Deeks’ funnel plot of calprotectin diagnosis value for COVID‐19 severity
Data Availability Statement
All data generated or analyzed in this study are available in the article.


