Visual Abstract
Keywords: pediatric kidney transplantation
Abstract
Background
Little is known about the time-varying determinants of kidney graft failure in children.
Methods
We performed a retrospective study of primary pediatric kidney transplant recipients (younger than 18 years) from the Eurotransplant registry (1990–2020). Piece-wise exponential additive mixed models were applied to analyze time-varying recipient, donor, and transplant risk factors. Primary outcome was death-censored graft failure.
Results
We report on 4528 kidney transplantations, of which 68% with deceased and 32% with living donor. One thousand six hundred and thirty-eight recipients experienced graft failure, and 168 died with a functioning graft. Between 2011 and 2020, the 5-year graft failure risk was 10% for deceased donor and 4% for living donor kidney transplant recipients. Risk of graft failure decreased five-fold from 1990 to 2020. The association between living donor transplantation and the lower risk of graft failure was strongest in the first month post-transplant (adjusted hazard ratio, 0.58; 95% confidence interval, 0.46 to 0.73) and remained statistically significant until 12 years post-transplant. Risk factors for graft failure in the first 2 years were deceased donor younger than 12 years or older than 46 years, potentially recurrent kidney disease, and panel-reactive antibody >0%. Other determinants of graft failure included dialysis before transplantation (until 5 years post-transplant), human leukocyte antigen mismatch 2–4 (0–15 years post-transplant), human leukocyte antigen mismatch 5–6 (2–12 years post-transplant), and hemodialysis (8–14 years post-transplant). Recipients older than 11 years at transplantation had a higher risk of graft failure 1–8 years post-transplant compared with other age groups, whereas young recipients had a lower risk throughout follow-up. Analysis of the combined effect of post-transplant time and recipient age showed a higher rate of graft failure during the first 5 years post-transplant in adolescents compared with young transplant recipients. In contrast to deceased donor younger than 12 years, deceased donor older than 46 years was consistently associated with a higher graft failure risk.
Conclusions
We report a long-term inverse association between living donor kidney transplantation and the risk of graft failure. The determinants of graft failure varied with time. There was a significant cumulative effect of adolescence and time post-transplant. The ideal donor age window was dependent on time post-transplant.
Introduction
Most pediatric kidney transplant recipients require multiple allografts throughout their lifetime. Retransplantation carries medical, psychological, and socioeconomic consequences, including a higher risk of graft failure than primary kidney transplantation.1 Graft longevity after primary transplantation is therefore of paramount importance. The reported 10-year graft survival rates in Europe, the United States, Australia, and New Zealand range from 61%–77% to 70%–75% in deceased and living donor transplantation, respectively.2 The donor, recipient, and transplant characteristics associated with a risk of graft failure after pediatric kidney transplantation in Europe include deceased donor, donor younger than 5 years especially in very young donors, poor human leukocyte antigen (HLA) matching, and long cold ischemia times.3,4
These characteristics are often nonproportional, meaning that the relative hazard is not constant over time. Data on the time-varying association, defined as a relationship where the relative risk can change over time from transplantation, between graft failure and its determinants in pediatric kidney transplantation are scarce. Larkins et al. reported a nonlinear relationship between age at transplantation and graft failure, dependent on time post-transplant in recipients younger than 5 years.5 These time-varying patterns of graft failure could be applied to fine-tune organ allocation policies and, in case of nonmodifiable factors such as recipient age, taken into account in clinical decision making. Still, most studies published to date have limited the follow-up to a certain period of time to address nonproportionality. With the use of Cox proportional hazard models, time-fixed associations between confounders and outcomes during a predefined period can be captured. Piece-wise exponential models enable the estimation of hazards with time-varying effects in time-to-event analyses.6
The objective of this study was to analyze the association between recipient, donor, and transplant characteristics and graft failure after primary kidney transplantation in pediatric patients across Eurotransplant countries.
Methods
Study Population and Data Collection
This retrospective, longitudinal study used anonymized data from the Eurotransplant Registry. All primary solitary kidney transplant recipients younger than 19 years at the time of transplantation between 1990 and 2020 were included. The data request was approved by the Scientific Committee of Eurotransplant. The national legislation of the member countries in regard to the ethical review and informed consent collection was followed (https://www.eurotransplant.org/gdpr/). Apart from the mandatory organ allocation-related data, Eurotransplant collects follow-up data on patient and transplantation outcomes. Transplant centers, except in Belgium and The Netherlands, are not obliged to provide this information. Data on the follow-up of German pediatric kidney transplant recipients are provided by the CERTAIN Registry (http://www.certain-registry.eu/).
Data on the following characteristics were collected: recipient sex, age, country, date of transplantation, dialysis vintage and modality before transplantation, panel-reactive antibody (PRA), primary kidney disease, date of graft failure, date of last follow-up or death if applicable, donor source (living versus deceased), age, sex, and number of HLA mismatches.
The study was conducted in accordance with the Declaration of Helsinki and the Declaration of Istanbul on Organ Trafficking and Transplant Tourism. The study was designed, analyzed, and reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (https://www.strobe-statement.org).
Outcomes and Definitions
Graft failure was defined as the requirement for dialysis or the date of preemptive retransplantation. Graft failure was censored for death with functioning graft at last follow-up. Death with functioning graft was assumed when the date of death matched the date of graft failure. Potentially recurrent kidney disease included focal segmental glomerulosclerosis, membranoproliferative glomerulonephritis, membranous nephropathy, rapidly progressive glomerulonephritis without systemic disease, and hemolytic uremic syndrome.7
Statistical Analysis
A piece-wise exponential additive mixed model was used to construct a model, where the hazard of graft failure was a piece-wise constant function depending on the confounders. The observation period was divided into a finite number of intervals, where the hazard rates were assumed to be piece-wise constant. A generalized additive model was then used to estimate the baseline hazard and covariate effects.8 We modeled the log hazard rate for patient i=1,…,n as
where represented the piece-wise constant baseline hazard for graft failure; the midpoint of the j-th interval; the potential smooth, time-varying effects of continuous covariates and time-varying effects of categorical covariates; and an independent, identically distributed, Gaussian random intercept term attributed to transplant country.
The baseline hazard represented how the relative risk of graft failure changed over time compared with the reference (in our analysis, 1 year post-transplant) when the values of all predictor variables were set to their reference levels. The hazard was set to be piece-wise constant monthly up until 1 year post-transplant and annually thereafter. Follow-up was censored at 15 years post-transplant. All variables were fitted as potentially nonlinear and time-varying, except for transplant year. Nonlinear associations were estimated using penalized splines, and the smoothing penalty was selected using fast restricted maximum likelihood.9 We applied a Gaussian random frailty effect for transplant countries to account for potential heterogeneity. To detect and remove irrelevant time-varying associations and maintain a balance between goodness of fit and parsimony, parameter selection was performed using the double penalty approach.10 A complete case analysis was conducted.
All models were adjusted for potential time-varying effects of recipient and donor sex, donor source, PRA, primary kidney disease, donor and recipient age, and time-fixed effects of transplant year and country. The analysis of donor age was performed separately for living and deceased donor kidney transplantation. Given the large proportion of missing values on HLA mismatch in living donor kidney transplantation (52%), this was only analyzed in deceased donor transplant recipients (n=3079). In 93% of cases, data on dialysis vintage before 2001 was missing. Therefore, it was only analyzed in deceased donor kidney transplant recipients between 2001 and 2020 (n=1849) and adjusted for HLA mismatch. Dialysis modality was analyzed in deceased donor transplant recipients between 2001 and 2020 (n=1614) and adjusted for HLA mismatch and dialysis vintage. Kaplan–Meier analysis and Cox proportional hazards regression were respectively used to explore differences in graft failure and estimate unadjusted hazard ratios (HR) between subgroups. The models were estimated using the R packages pammtools8,11 and mgcv.9 R was used for visualization and data analysis.12
Results
Patient and Donor Characteristics
In the Eurotransplant area, 4528 primary pediatric kidney transplantations were performed between 1990 and 2020; of these, 3079 with deceased donor and 1449 with living donor. Median follow-up was 85.5 months (interquartile range [IQR], 37–150). One thousand six hundred and thirty-eight recipients experienced graft failure, and 168 died with a functioning graft after median time of 31 months (IQR, 3.75–117.5). The median time to graft failure was 91 months (IQR, 29–145). A detailed overview of patient, donor, and transplant characteristics stratified by donor source is shown in Table 1 and Supplemental Figure 1.
Table 1.
Demographics of primary pediatric kidney transplant recipients, stratified by donor type
| Donor Source | Deceased Donor (3079) | Living Donor (1449) |
|---|---|---|
| Female recipient | 1283 (42) | 577 (40) |
| Recipient age at transplantation | ||
| Younger than 5 yr | 626 (20) | 268 (18) |
| 5–11 yr | 1098 (36) | 462 (32) |
| Older than 11 yr | 1355 (44) | 719 (50) |
| Kidney disease | ||
| CAKUT | 911 (30) | 457 (32) |
| Cystic | 344 (11) | 156 (11) |
| Glomerular | 384 (12) | 156 (11) |
| Potentially recurrent | 434 (14) | 149 (10) |
| Other/unknown | 1006 (33) | 531 (37) |
| PRA a | ||
| 0% | 2688 (90) | 1168 (89) |
| 1%–25% | 236 (8) | 125 (9) |
| 26%–100% | 67 (2) | 18 (1) |
| Missingb | 68 (3) | 138 (10) |
| Preemptivec | 235 (12) | 564 (52) |
| Non-preemptivec | 1653 (88) | 519 (46) |
| Missing | 0 (0) | 25 (2) |
| Months dialysisc,d | 21 (12–32) | 10 (5–17) |
| Missingb | 0 (0) | 28 (3) |
| Dialysis modality a , c | ||
| Hemodialysis | 713 (43) | 286 (55) |
| Peritoneal dialysis | 928 (56) | 230 (44) |
| Dialysis, type unknown | 12 (1) | 3 (1) |
| Median donor age (IQR) | 41 (36–46) | 26 (12–43) |
| Donor age | ||
| Younger than 5 yr | 327 (11) | 0 (0) |
| 5–10 yr | 364 (12) | 0 (0) |
| 11–20 yr | 601 (20) | 4 (<1) |
| 21–30 yr | 403 (13) | 100 (7) |
| 31–40 yr | 472 (15) | 570 (39) |
| 41–50 yr | 630 (20) | 617 (43) |
| 51–60 yr | 248 (8) | 131 (9) |
| Older than 60 yr | 34 (1) | 27 (2) |
| Female donor | 1349 (44) | 786 (54) |
| Transplant year | 2004 (1997–2012) | 2007 (2001–2014) |
| Transplant country | ||
| Austria | 202 (7) | 127 (9) |
| Belgium | 350 (11) | 119 (8) |
| Croatia | 41 (1) | 14 (1) |
| Germany | 2016 (65) | 830 (57) |
| Hungary | 61 (2) | 27 (2) |
| Luxembourg | 3 (<1) | 0 (0) |
| The Netherlands | 396 (13) | 332 (23) |
| Slovenia | 10 (<1) | 0 (0) |
| HLA mismatch a | ||
| 0–1 | 447 (15) | 119 (17) |
| 2–4 | 2476 (80) | 542 (78) |
| 5–6 | 148 (5) | 37 (5) |
| Missingb | 8 (0) | 751 (52) |
| Living donor type | NA | |
| Nonrelated | 24 (2) | |
| Related | 1425 (98) |
Categorical data are presented as number (% of cases). CAKUT, congenital anomalies of the kidney and urinary tract; HLA, human leukocyte antigen; IQR, interquartile range, presented as Q1–Q3; NA, not available; PRA, panel-reactive antibody.
Frequency on the basis of total amount of nonmissing cases.
Frequency on the basis of total amount of cases (3079 for deceased donor transplants, 1449 for live donor transplants).
Numbers reported for transplants performed between 2001 and 2020 (1888 deceased donor transplants, 1108 live donor transplants).
Dialysis vintage reported for non-preemptive transplants only.
Probability of Graft Failure by Donor Source
As shown in Figure 1 and Supplemental Table 1, the probability of graft failure improved per decade and was consistently lower in living than in deceased donor transplantation. The 5-year probability of graft failure decreased from 25% in 1990–2000 to 10% in 2011–2020 (unadjusted HR, 0.36; 95% confidence intervals [95% CI], 0.28 to 0.47) and from 14% in 1990–2000 to 4% in 2011–2020 (unadjusted HR, 0.26; 95% CI, 0.15 to 0.46) in deceased and living donor transplant recipients, respectively. The improvement in unadjusted 5-year probability of graft failure per decade was more pronounced in living than in deceased donor transplant recipients (unadjusted HR, 0.54; 95% CI, 0.40 to 0.74 in 1990–2000 versus unadjusted HR, 0.38; 95% CI, 0.23 to 0.64 in 2011–2020).
Figure 1.
Death-censored graft survival after primary kidney transplantation with 95% confidence intervals (CI), stratified by transplant period and donor type. Shades represent 95% CI. DD, deceased donor; LD, living donor.
Transplant Characteristics and Graft Failure
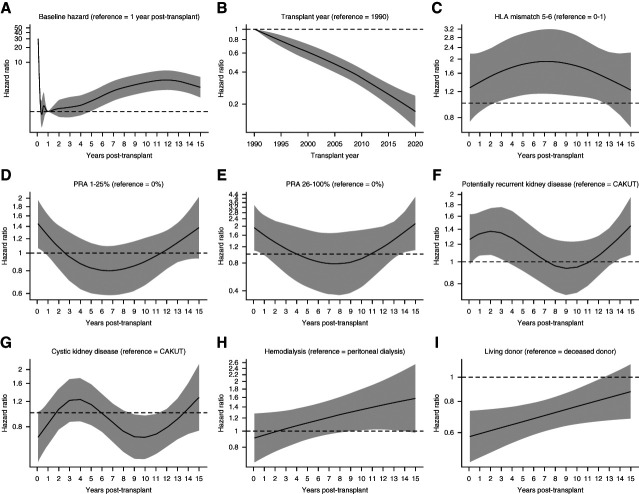
As shown in Figure 2A, the risk of graft failure was highest in the first months post-transplant and decreased rapidly with time up until 1 year post-transplant. From 1 year post-transplant, it steadily increased with time. The HR of graft failure was 30.01 (95% CI, 19.23 to 46.83) in the first month, 2.1 (95% CI, 1.33 to 3.31) at 5 years, and 4.24 (95% CI, 2.7 to 6.64) at 10 years post-transplant as compared with 1 year post-transplant (Supplemental Table 2).
Figure 2.
Baseline hazard, time-fixed effect of transplant year, and hazard ratios for graft failure by time post-transplant for HLA mismatch 5–6, PRA, potentially recurrent kidney disease, cystic kidney disease, hemodialysis, and living donor source. Baseline hazard (A), time-fixed effect of transplant year (B), and hazard ratios for graft failure by time post-transplant for HLA mismatch 5–6 (C), PRA (D and E), potentially recurrent kidney disease (F), cystic kidney disease (G), hemodialysis (H), and living donor source (I). Solid lines represent HR; shades represent 95% CI. All models were adjusted for potential time-varying effects of recipient and donor sex, donor source, PRA, kidney disease, donor/recipient age, and time-fixed effects of transplant year and transplant country and included all primary kidney transplants unless otherwise noted. (C) Analysis restricted to all deceased donor transplants and additionally adjusted for potential time-varying effect of HLA mismatch. (H) Analysis restricted to all non-preemptive deceased donor transplants between 2001 and 2020 and additionally adjusted for potential time-varying effect of HLA mismatch, dialysis vintage, and modality. CAKUT, congenital anomalies of the kidney and urinary tract; HLA, human leukocyte antigen; HR, hazard ratio; PRA, panel-reactive antibody.
The rate of graft failure improved with calendar year of transplantation (Figure 2B, Supplemental Table 3). The risk of graft failure halved from 1990 to 2005 (adjusted hazard ratio [aHR], 0.48; 95% CI, 0.4 to 0.57), and recipients who underwent transplantation in 2018 had 80% lower risk of graft failure (95% CI, 0.15 to 0.26).
Compared with HLA mismatch 0–1, there was a time-constant association between HLA mismatch 2–4 and graft failure in deceased donor transplantation (aHR, 1.29; 95% CI, 1.08 to 1.54) (Supplemental Figure 2C). HLA mismatch 5–6 was associated with a higher risk of graft failure between 2 and 12 years post-transplant (Figure 2C, Supplemental Table 4).
Recipient Characteristics and Graft Failure
As shown in Figure 3A and Supplemental Table 5, ascending recipient age was associated with a higher risk of graft failure between 1 and 9 years after transplantation. For example, at 5 years post-transplant, a 6-year-old recipient had a 38% lower risk (aHR, 0.62; 95% CI, 0.52 to 0.76), and a 15-year-old recipient had a 55% higher risk (aHR, 1.55; 95% CI, 1.34 to 1.81) of graft failure compared with an 11-year-old recipient.
Figure 3.
Hazard ratios for graft failure by time post-transplant for recipient age at transplantation, dialysis vintage, deceased donor age, and living donor age. Hazard ratios for graft failure by time post-transplant for recipient age at transplantation (A), dialysis vintage (B), deceased donor age (C), and living donor age (D). All models were adjusted for potential time-varying effects of recipient and donor sex, donor source, PRA, kidney disease, donor/recipient age, and time-fixed effects of transplant year and transplant country and included all primary kidney transplants unless otherwise noted. (B) Analysis restricted to all deceased donor transplants between 2001 and 2020 and additionally adjusted for potential time-varying effect of HLA mismatch and dialysis vintage. (C) Analysis restricted to all deceased donor transplants and additionally adjusted for potential time-varying effect of HLA mismatch. (D) Analysis restricted to all living donor transplants.
Figure 4 (Supplemental Table 6) illustrates the combined effect of post-transplant time and recipient age on graft failure and indicates that the risk of graft failure in recipients who underwent transplantation during adolescence increased up until 7 years post-transplant, after which it reached a plateau. The risk of graft failure in recipients who underwent transplantation during childhood increased at a slower rate up until 10 years post-transplant, and afterward, it was comparable with those who underwent transplantation in adolescence (Figure 4D). Figure 3A shows that the risk of graft failure at the age of 18 years was higher for those who underwent transplantation at the age of 5 years (aHR, 3.56; 95% CI, 2.26 to 5.6) as compared with the age at transplant of 15 years (aHR, 2.3; 95% CI, 1.46 to 3.64).
Figure 4.
Baseline hazard ratio for graft failure for a 5-, 10-, and 15-year-old recipient at transplantation between 1 and 15 years post-transplant. Baseline hazard ratio for graft failure for a 5- (A and D), 10- (B and D), and 15-year-old (C and D) recipient at transplantation between 1 and 15 years post-transplant. Facets illustrate the combined effect of time post-transplant (baseline hazard) and the time-varying effect of recipient age. Solid lines represent HR; shades represent 95% CI. The model was adjusted for potential time-varying effects of recipient and donor sex, donor source, PRA, kidney disease, donor/recipient age, and time-fixed effects of transplant year and transplant country and included all primary kidney transplants.
At 1 month, recipients with PRA between 1% and 25% (aHR, 1.44; 95% CI, 1.07 to 1.96) and 26–100% (aHR, 1.92; 95% CI, 1.09 to 3.38) had a higher risk of graft failure, and this was statistically significant in the first year post-transplant (Figure 2, D and E, Supplemental Table 7). Graft failure was not associated with male recipient sex (aHR, 0.97; 95% CI, 0.86 to 1.08) (Supplemental Figure 2A). Potentially recurrent kidney disease was associated with a higher risk of graft failure up to 4.5 years post-transplant as compared with congenital anomalies of the kidney and urinary tract (CAKUT) (Figure 2F, Supplemental Table 8). Cystic kidney disease was associated with a lower risk of graft failure between 8 and 11 years post-transplant as compared with CAKUT (Figure 2F, Supplemental Table 8).
In the first years after deceased donor transplantation, increasing time on dialysis was associated with a higher rate of graft failure (Figure 3B, Supplemental Table 9). At 1-year post-transplant, the respective aHR of graft failure in deceased donor transplant recipients with 1, 2, and 3 years dialysis vintage was 1.22 (95% CI, 1.08 to 1.39), 1.46 (95% CI, 1.17 to 1.82), and 1.66 (95% CI, 1.25 to 2.22) compared with preemptive transplantation with deceased donors. This association had dissipated by 3–5 years post-transplant. Compared with peritoneal dialysis, hemodialysis (Figure 2H, Supplemental Table 10) was associated with a higher risk of graft failure between 8 and 14 years post-transplant (aHR, 1.40; 95% CI, 1.02 to 1.9 at 10 years).
Donor Characteristics and Graft Failure
As shown in Figure 2I and Supplemental Table 11, the association between living donor transplantation and a lower risk of graft failure was strongest in the first month post-transplant (aHR, 0.58; 95% CI, 0.46 to 0.73). Over time, this relationship became weaker, and it was no longer statistically significant at 12 years (aHR, 0.83; 95% CI, 0.67 to 1.02).
Deceased donor age showed a U-shaped relationship with graft failure in the first 3 years post-transplant, with the lowest rate of graft failure at 1 month in recipients of organs from donors aged 12 (aHR, 1.28; 95% CI, 1.02 to 1.6) to 47 years (aHR, 1.36; 95% CI, 1.01 to 1.84), where donor age of 25 years was the reference (Figure 3, C and D and Supplemental Tables 12 and 13). The risk of graft failure in recipients from donors younger than 12 years became lower over time, whereas the higher risk in recipients from donors older than 46 years persisted (Figure 3C). Three years post-transplant, recipients with a 6- and 50-year-old deceased donor experienced a 12% (aHR, 1.28; 95% CI, 0.87 to 1.45) and 49% (aHR, 1.49; 95% CI, 1.13 to 1.97) greater risk of graft failure than the reference. Beyond 5 years, the lowest risk of graft failure was for recipients of organs from deceased donors aged 2–36 years. There was no association between living donor age and graft failure (Figure 3D). Male donor sex (aHR, 1.04; 95% CI, 0.93 to 1.16) was not time-varying, nor was it associated with graft failure.
Clinical Application of the Model
Kidney transplantation from deceased donors aged 12–45 years was associated with a lower rate of graft failure compared with younger and older donors. This could offset the graft failure risk in recipients with multiple risk factors. If 100 identical transplant candidates (an 11-year-old male recipient with CAKUT, PRA 0%, transplanted in Germany in 2005 with HLA mismatch 2–4) received a graft from a 6-year-old male deceased donor, then on the basis of our model, nine recipients would experience graft failure within 2 years (predicted graft survival, 0.91; 95% CI, 0.87 to 0.93). Considering multiple risk factors for lower short-term graft survival (PRA 1%–25% and potentially recurrent primary kidney disease), 17 recipients would experience graft failure (predicted graft survival, 0.83; 95% CI, 0.74 to 0.89). If these recipients were transplanted with a 20-year-old deceased donor, 11 recipients would experience graft failure within 2 years (predicted graft survival, 0.89; 95% CI, 0.83 to 0.93).
Discussion
In this study comprising one of the largest European cohorts of pediatric kidney transplant recipients with long-term follow-up, we report a substantial and ongoing improvement in graft survival over time from 1990 to 2020. The unique feature of this report is the application of statistical methods, allowing the identification of time-varying determinants of graft failure. Living donor transplantation showed a sustained benefit over deceased donor transplantation, which remained statistically significant until 12 years post-transplant. Risk factors for graft failure in the first 2 years post-transplant were deceased donor younger than 12 years or older than 46 years, potentially recurrent primary disease, and PRA >0%. Covariates associated with a higher risk of graft failure included HLA mismatch 2–4 (0–15 years post-transplant), HLA mismatch 5–6 (2–12 years post-transplant), dialysis before transplant (until 5 years post-transplant), and hemodialysis as opposed to peritoneal dialysis (8–12 years post-transplant). In contrast to deceased donor younger than 12 years, deceased donor older than 46 years was persistently associated with a higher risk of graft failure. Between 1 and 9 years post-transplant, recipients older than 11 years had a higher risk of graft failure than younger recipients. At 10 years from transplantation, the risk of graft failure was comparable for those who underwent transplantation at a young age and in adolescence.
The risk of graft failure was high in the first month post-transplant, most likely because of surgical complications. Therefore, surgically challenging transplantation, such as in very small recipients, should ideally be undertaken in large-volume centers. While this policy may theoretically work in large European countries, it is not feasible in smaller countries with a limited number of pediatric kidney transplantations.
The association between living donor transplantation and a lower risk of graft failure has been well-documented in the literature.4,13–15 We showed that this association was most pronounced in the first month post-transplant, became weaker over time, and was not significant at 12 years. The initial lower risk of graft failure can be explained by better patient and organ preparation and lower exposure to ischemia reperfusion injury.16,17 In addition, it has been reported that children growing up in families with a higher socioeconomic status are more likely to receive an organ from a living donor.18,19 These patients are more likely to have access to health care and medication, higher education level, and better therapy adherence.20 This can explain the superior long-term outcome of living donor transplantation.21 The lack of statistical significance beyond 12 years post-transplant likely denotes a lower number of patients still in follow-up, rather than a lack of clinical significance. In addition, we showed that the difference in outcome between living and deceased donor transplantation persisted, despite the overall improvement in transplant outcome.
Adolescence has been recognized as a determinant of graft failure.3,22,23 In our study, increasing age at transplantation was associated with a higher risk of graft failure between 1 and 9 years post-transplant. From then on, young recipient age was no longer associated with lower rates of graft failure as compared with adolescents. Potential explanations include lower adherence to immunosuppression, transition to adult care, and a maturation of the immune system.3,13,22,24 Therefore, clinicians should stress the importance of immunosuppression and explain the risks of nonadherence to adolescents and their families.
The previously reported high-risk window age for graft failure between 17 and 24 years25 was confirmed in our analysis when combining the effects of recipient age and post-transplant time. In this analysis, the risk of graft failure was highest at 12 years post-transplant and comparable in all recipient age groups. Furthermore, a 5-year-old recipient experienced a higher risk of graft failure at the age of 18 as compared with a 15-year-old recipient. This shows a significant cumulative effect of adolescence and time post-transplant.
In line with previous reports, deceased donor transplantation with donors younger than 12 years was associated with higher risk of graft failure in the first 4 years post-transplant.3,26 This association can be explained by surgical complications, acute tubular necrosis, hyperfiltration injury, and early rejection.27,28 Beyond the critical period of 4 years, the long-term outcome was favorable. In contrast to young deceased donors, the association between deceased donor older than 46 years and higher rates of graft failure persisted. Elaborating on the work of Chesnaye et al.,3 we report that the ideal donor age window was dependent on time post-transplant. In the first 3 years, deceased donor transplantation with donors aged 12–46 years was associated with the lowest risk of graft failure as compared with a 25-year-old deceased donor. Beyond 5 years, the ideal donor age window was 2–36 years.
PRA >0% and potentially recurrent primary kidney disease were associated with graft failure in the first 2 years post-transplant. High pretransplant PRA levels not only increase the risk of acute allograft rejection but also reduce the pool of suitable donors, which may hamper access to transplantation and lead to the acceptance of inferior-quality organs.29 Therefore, close monitoring of transplant recipients with detectable PRA to facilitate early diagnosis of antibody-mediated allograft rejection should particularly be undertaken in the first year post-transplant. In accordance with previous studies,30–33 we reported that HLA mismatch 2–4 and 5–6 was associated with a higher risk of graft failure over the complete follow-up period and between 2 and 12 years after transplantation, respectively. Notably, the estimated adjusted HR of graft failure before 2 years and after 12 years for HLA mismatch 5–6 was higher as compared with HLA mismatch 2–4. We believe the nonsignificance was due to the relative low number of patients with HLA mismatch 5–6 (n=148).
We reported that the association between increasing dialysis vintage and higher risk of graft failure was most pronounced in the first 5 years post-transplant. This is in line with the work of Amaral et al., who showed that any dialysis exposure was associated with a 32% higher rate of graft failure in comparison with preemptive transplantation.15 During long-term dialysis, disturbed mineral metabolism and bone disease, inflammation, oxidative stress, and the accumulation of uremic toxins contribute to irreversible changes such as reduced heart function, vascular stiffness, and calcification.34 Moreover, we found an association between lower rates of graft failure and pretransplant peritoneal dialysis as opposed to hemodialysis. Further studies are needed to clarify whether this association is causal or reflects confounding by disease severity. Peritoneal dialysis is associated with better preservation of venous access and residual kidney function. A higher mortality risk has also been reported in pretransplant children on hemodialysis compared with peritoneal dialysis.35–37
The strengths of our study included a large international cohort of pediatric transplant recipients with long-term follow-up. Our study also has several limitations. We cannot exclude residual confounding because of incomplete or unreported data. More recently transplanted patients were less represented in the cohort. The analysis was not adjusted for other relevant predictors of graft failure, including cold and warm ischemia time, socioeconomic status, and allograft rejection. It is possible that socioeconomic status has confounded our results regarding dialysis vintage. Furthermore, we did not have details on the etiologies of certain primary kidney diseases, such as rapidly progressive glomerulonephritis. There is evidence that outcomes after pediatric kidney transplantation have improved over time for specific recipient subgroups, especially for highly sensitized recipients.38 Our findings should be interpreted with caution outside the Eurotransplant area because of differences in ethnicity, access to health care, and therapeutic approach.
In conclusion, we reported a long-term inverse association between living donor kidney transplantation and the risk of graft failure. Patients with PRA >0% and HLA mismatch 2–6 had higher risk of graft failure. There was a significant cumulative effect of adolescence and time post-transplant. The ideal donor age window was dependent on time post-transplant.
Supplementary Material
Acknowledgments
The authors gratefully acknowledge the funding of the CERTAIN Registry by a grant from the Dietmar Hopp Stiftung, the European Society for Pediatric Nephrology (ESPN), the German Society for Pediatric Nephrology (GPN), and by grants from the pharmaceutical companies Astellas and Novartis. The authors would like to thank all the contributors to the CERTAIN Registry for providing patient data: M. Bald (Stuttgart), H. Fehrenbach (Memmingen), D. Haffner (Hannover), M. Hansen (Frankfurt), C. Hempel (Leipzig), U. John (Jena), G. Klaus (Marburg), J. König (Münster), B. Lange-Sperandio (Munich), D. Müller (Berlin), J. Oh (Hamburg), L. Pape (Essen), M. Pohl (Freiburg), K. Sauerstein (Erlangen), G. Schalk (Bonn), H. Staude (Rostock), P. Strotmann (Munich TUM), LT Weber (Cologne), M. Weitz (Tübingen), L. Berta (Budapest, Hungary), and K. Heindl-Rusai (Vienna, Austria).
Footnotes
M. Bald, H. Fehrenbach, D. Haffner, M. Hansen, C. Hempel, U. John, G. Klaus, J. König, B. Lange-Sperandio, D. Müller, J. Oh, L. Pape, M. Pohl, K. Sauerstein, G. Schalk, H. Staude, P. Strotmann, LT Weber, M. Weitz, L. Berta, and K. Heindl-Rusai.
See related editorial, “Embracing Complexity to Better Serve Pediatric Kidney Transplant Recipients,” on pages 286–288.
Contributor Information
Collaborators: M. Bald, H. Fehrenbach, D. Haffner, M. Hansen, C. Hempel, U. John, G. Klaus, J. König, B. Lange-Sperandio, D. Müller, J. Oh, L. Pape, M. Pohl, K. Sauerstein, G. Schalk, H. Staude, P. Strotmann, L.T. Weber, M. Weitz, L. Berta, and K. Heindl-Rusai
Disclosures
J.J. Kim acknowledges funding from a MRC NIHR fellowship (MR/V037900/1). N. Knops reports research funding from Sandoz. L. Pape reports consultancy for Alexion, Alnylam, Chiesi, Neovii, and Novartis; research funding from Chiesi, Germany; and honoraria from Alexion and Chiesi. A. Prytula reports research funding from Astellas and Novo Nordisk. I. Tieken reports other interests or relationships with Eurotransplant. B. Toenshoff reports consulting fees from Bristol-Myers Squibb, Chiesi, CSL Behring Biotherapies for Life, and Vifor; research grants from Astellas, Chiesi, and Novartis; and participation in advisory boards of Bristol-Myers Squibb, Chiesi, and Vifor. S. Vogelaar reports employment with Eurotransplant International. Cooperative European Paediatric Renal TransplAnt INitiative (CERTAIN) registry acknowledges financial contribution from the Dietmar Hopp Stiftung, the European Society for Pediatric Nephrology (ESPN), the German Society for Pediatric Nephrology (GPN), and by grants from the pharmaceutical companies Astellas and Novartis. All remaining authors have nothing to disclose.
Funding
None.
Author Contributions
Conceptualization: Ferran Coens, Agnieszka Prytula
Data curation: Ineke Tieken, Serge Vogelaar.
Formal analysis: Andreas Bender, Ferran Coens.
Methodology: Andreas Bender, Ferran Coens, Agnieszka Prytula.
Writing – original draft: Ferran Coens, Agnieszka Prytula.
Writing – review & editing: Andreas Bender, Jon Jin Kim, Noël Knops, Kai Krupka, Lars Pape, Ann Raes, Ineke Tieken, Burkhard Tönshoff, Serge Vogelaar.
Data Sharing Statement
Data cannot be shared. Eurotransplant policy restricts it.
Supplemental Material
This article contains the following supplemental material online at http://links.lww.com/CJN/B838.
Supplemental Table 1. Death-censored graft survival after first kidney transplantation with 95% CI, stratified by transplant period and donor source.
Supplemental Table 2. Hazard ratios with 95% CI by post-transplant time interval (months).
Supplemental Table 3. Time-fixed hazard ratio with 95% CI for transplant year.
Supplemental Table 4. Hazard ratio of graft failure with 95% CI for HLA mismatch level, by post-transplant time interval (months).
Supplemental Table 5. Hazard ratio of graft failure with 95% CI per year of recipient age at transplant, by post-transplant time interval (months).
Supplemental Table 6. Baseline hazard ratio for graft failure for a 5-, 10-, and 15-year-old recipient at kidney transplantation between 1 and 15 years post-transplant.
Supplemental Table 7. Hazard ratio of graft failure with 95% CI for PRA level, by post-transplant time interval (months).
Supplemental Table 8. Hazard ratio of graft failure with 95% CI for primary kidney disease level, by post-transplant time interval (months).
Supplemental Table 9. Hazard ratio of graft failure with 95% CI per month of dialysis vintage, by post-transplant time interval (months).
Supplemental Table 10. Hazard ratio of graft failure with 95% CI for dialysis modality, by post-transplant time interval (months).
Supplemental Table 11. Hazard ratio of graft failure with 95% CI for donor type, by post-transplant time interval (months).
Supplemental Table 12. Hazard ratio of graft failure with 95% CI per year of deceased donor age, by post-transplant time interval (months).
Supplemental Table 13. Hazard ratio of graft failure with 95% CI per year of living donor age, by post-transplant time interval (months).
Supplemental Figure 1. Distribution of transplant year, donor age, recipient age, HLA mismatch, and dialysis vintage stratified by donor source.
Supplemental Figure 2. Hazard ratios for graft failure by time post-transplant for HLA mismatch 2–4 (A), recipient sex (B), glomerular kidney disease (C), other/unknown kidney disease (D), and donor sex (E).
References
- 1.Van Arendonk KJ Garonzik Wang JM Deshpande NA, et al. Practice patterns and outcomes in retransplantation among pediatric kidney transplant recipients. Transplantation. 2013;95(11):1360–1368. doi: 10.1097/TP.0b013e31828c6d64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fernandez HE, Foster BJ. Long-term care of the pediatric kidney transplant recipient. Clin J Am Soc Nephrol. 2022;17(2):296–304. doi: 10.2215/CJN.16891020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chesnaye NC van Stralen KJ Bonthuis M, et al.. The association of donor and recipient age with graft survival in paediatric renal transplant recipients in a European Society for Paediatric Nephrology/European Renal Association-European Dialysis and Transplantation Association Registry study. Nephrol Dial Transplant. 2017;32(11):1949–1956. doi: 10.1093/ndt/gfx261 [DOI] [PubMed] [Google Scholar]
- 4.Mumford L, Maxwell H, Ahmad N, Marks SD, Tizard J. The impact of changing practice on improved outcomes of paediatric renal transplantation in the United Kingdom: a 25 years review. Transpl Int. 2019;32(7):751–761. doi: 10.1111/tri.13418 [DOI] [PubMed] [Google Scholar]
- 5.Larkins NG Wong G Alexander SI, et al. Survival and transplant outcomes among young children requiring kidney replacement therapy. Pediatr Nephrol. 2021;36(8):2443–2452. doi: 10.1007/s00467-021-04945-9 [DOI] [PubMed] [Google Scholar]
- 6.Wey A, Salkowski N, Kremers W, Ahn YS, Snyder J. Piecewise exponential models with time-varying effects: estimating mortality after listing for solid organ transplant. Stat. 2020;9(1):e264. doi: 10.1002/sta4.264 [DOI] [Google Scholar]
- 7.Cochat P Fargue S Mestrallet G, et al. Disease recurrence in paediatric renal transplantation. Pediatr Nephrol. 2009;24(11):2097–2108. doi: 10.1007/s00467-009-1137-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bender A, Groll A, Scheipl F. A generalized additive model approach to time-to-event analysis. Stat Model. 2018;18(3–4):299–321. doi: 10.1177/1471082x17748083 [DOI] [Google Scholar]
- 9.Wood S. mgcv: Mixed GAM Computation Vehicle with GCV/AIC/REML Smoothness Estimation; 2012. [Google Scholar]
- 10.Marra G, Wood SN. Practical variable selection for generalized additive models. Comput Stat Data Anal. 2011;55(7):2372–2387. doi: 10.1016/j.csda.2011.02.004 [DOI] [Google Scholar]
- 11.Bender A, Scheipl F. Pammtools: Piece-wise Exponential Additive Mixed Modeling Tools. arXiv; 2018. [Google Scholar]
- 12.R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; 2022. https://www.R-project.org/ [Google Scholar]
- 13.Dharnidharka VR, Fiorina P, Harmon WE. Kidney transplantation in children. N Engl J Med. 2014;371(6):549–558. doi: 10.1056/NEJMra1314376 [DOI] [PubMed] [Google Scholar]
- 14.Smith JM, Martz K, Blydt-Hansen TD. Pediatric kidney transplant practice patterns and outcome benchmarks, 1987-2010: a report of the North American Pediatric Renal Trials and Collaborative Studies. Pediatr Transplant. 2013;17(2):149–157. doi: 10.1111/petr.12034 [DOI] [PubMed] [Google Scholar]
- 15.Amaral S, Sayed BA, Kutner N, Patzer RE. Preemptive kidney transplantation is associated with survival benefits among pediatric patients with end-stage renal disease. Kidney Int. 2016;90(5):1100–1108. doi: 10.1016/j.kint.2016.07.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chandar J, Chen L, Defreitas M, Ciancio G, Burke G, III. Donor considerations in pediatric kidney transplantation. Pediatr Nephrol. 2021;36(2):245–257. doi: 10.1007/s00467-019-04362-z [DOI] [PubMed] [Google Scholar]
- 17.Mehrabi A Golriz M Khajeh E, et al. Surgical outcomes after pediatric kidney transplantation at the University of Heidelberg. J Pediatr Urol. 2019;15(3):221.e1–221.e8. doi: 10.1016/j.jpurol.2019.01.007 [DOI] [PubMed] [Google Scholar]
- 18.Sigurjonsdottir VK, Grimm PC. Living or deceased donor kidney transplantation in children. Curr Opin Pediatr. 2019;31(2):232–236. doi: 10.1097/mop.0000000000000740 [DOI] [PubMed] [Google Scholar]
- 19.Van Cauwenberghe K Raes A Pauwels L, et al. The choice between deceased- vs living-donor renal transplantation in children: analysis of data from a Belgian tertiary center. Pediatr Transplant. 2018;22(2). doi: 10.1111/petr.13140 [DOI] [PubMed] [Google Scholar]
- 20.Axelrod DA Dzebisashvili N Schnitzler MA, et al. The interplay of socioeconomic status, distance to center, and interdonor service area travel on kidney transplant access and outcomes. Clin J Am Soc Nephrol. 2010;5(12):2276–2288. doi: 10.2215/CJN.04940610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Anand A Malik TH Dunson J, et al. Factors associated with long-term graft survival in pediatric kidney transplant recipients. Pediatr Transplant. 2021;25(4):e13999. doi: 10.1111/petr.13999 [DOI] [PubMed] [Google Scholar]
- 22.Foster BJ, Dahhou M, Zhang X, Platt RW, Samuel SM, Hanley JA. Association between age and graft failure rates in young kidney transplant recipients. Transplantation. 2011;92(11):1237–1243. doi: 10.1097/TP.0b013e31823411d7 [DOI] [PubMed] [Google Scholar]
- 23.Ritchie AG, Clayton PA, McDonald SP, Kennedy SE. Age-specific risk of renal graft loss from late acute rejection or non-compliance in the adolescent and young adult period. Nephrology (Carlton). 2018;23(6):585–591. doi: 10.1111/nep.13067 [DOI] [PubMed] [Google Scholar]
- 24.Matsuda-Abedini M, Marks SD, Foster BJ. Transition of young adult kidney transplant recipients. Pediatr Nephrol. 2023;38(2):383–390. doi: 10.1007/s00467-022-05582-6 [DOI] [PubMed] [Google Scholar]
- 25.Van Arendonk KJ James NT Boyarsky BJ, et al. Age at graft loss after pediatric kidney transplantation: exploring the high-risk age window. Clin J Am Soc Nephrol. 2013;8(6):1019–1026. doi: 10.2215/CJN.10311012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Opelz G, Döhler B. Pediatric kidney transplantation: analysis of donor age, HLA match, and posttransplant non-Hodgkin lymphoma: a collaborative transplant study report. Transplantation. 2010;90(3):292–297. doi: 10.1097/TP.0b013e3181e46a22 [DOI] [PubMed] [Google Scholar]
- 27.Beetz O Weigle CA Nogly R, et al. Surgical complications in pediatric kidney transplantation-incidence, risk factors, and effects on graft survival: a retrospective single-center study. Pediatr Transplant. 2021;25(2):e13871. doi: 10.1111/petr.13871 [DOI] [PubMed] [Google Scholar]
- 28.Irtan S Maisin A Baudouin V, et al. Renal transplantation in children: critical analysis of age related surgical complications. Pediatr Transplant. 2010;14(4):512–519. doi: 10.1111/j.1399-3046.2009.01260.x [DOI] [PubMed] [Google Scholar]
- 29.Sypek MP Kausman JY Watson N, et al. The introduction of cPRA and its impact on access to deceased donor kidney transplantation for highly sensitized patients in Australia. Transplantation. 2021;105(6):1317–1325. doi: 10.1097/TP.0000000000003410 [DOI] [PubMed] [Google Scholar]
- 30.Opelz G, Döhler B, Middleton D, Süsal C, A Collaborative Transplant Study Report. HLA matching in pediatric kidney transplantation: HLA poorly matched living donor transplants versus HLA well-matched deceased donor transplants. Transplantation. 2017;101(11):2789–2792. doi: 10.1097/TP.0000000000001811 [DOI] [PubMed] [Google Scholar]
- 31.Trnka P, McTaggart SJ, Francis A. The impact of donor/recipient age difference and HLA mismatch on graft outcome in pediatric kidney transplantation. Pediatr Transplant. 2018;22(7):e13265. doi: 10.1111/petr.13265 [DOI] [PubMed] [Google Scholar]
- 32.Shi X Liu R Xie X, et al.. Effect of human leukocyte antigen mismatching on the outcomes of pediatric kidney transplantation: a systematic review and meta-analysis. Nephrol Dial Transplant. 2017;32(11):1939–1948. doi: 10.1093/ndt/gfx259 [DOI] [PubMed] [Google Scholar]
- 33.Kim JJ Curtis RMK Reynolds B, et al. The UK kidney donor risk index poorly predicts long-term transplant survival in paediatric kidney transplant recipients. Front Immunol. 2023;14:1207145. doi: 10.3389/fimmu.2023.1207145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vila Cuenca M, Hordijk PL, Vervloet MG. Most exposed: the endothelium in chronic kidney disease. Nephrol Dial Transplant. 2020;35(9):1478–1487. doi: 10.1093/ndt/gfz055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chesnaye NC Schaefer F Groothoff JW, et al. Mortality risk in European children with end-stage renal disease on dialysis. Kidney Int. 2016;89(6):1355–1362. doi: 10.1016/j.kint.2016.02.016 [DOI] [PubMed] [Google Scholar]
- 36.Kong J, Davies M, Mount P. The importance of residual kidney function in haemodialysis patients. Nephrology (Carlton). 2018;23(12):1073–1080. doi: 10.1111/nep.13427 [DOI] [PubMed] [Google Scholar]
- 37.Rees L, Schaefer F, Schmitt CP, Shroff R, Warady BA. Chronic dialysis in children and adolescents: challenges and outcomes. Lancet Child Adolescent Health. 2017;1(1):68–77. doi: 10.1016/S2352-4642(17)30018-4 [DOI] [PubMed] [Google Scholar]
- 38.Van Arendonk KJ Boyarsky BJ Orandi BJ, et al. National trends over 25 years in pediatric kidney transplant outcomes. Pediatrics. 2014;133(4):594–601. doi: 10.1542/peds.2013-2775 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data cannot be shared. Eurotransplant policy restricts it.