Abstract
Background
In France, the potential benefit of new treatments is initially evaluated by the Haute Autorité de Santé to determine reimbursement and pricing, but rarely afterwards. Although immunotherapies (ITs) have considerably improved the survival of patients, few data are available on their long-term benefit at a population-treated level. The present retrospective study aimed to assess the clinical benefit of ITs compared to the previous standards of care (SoCs) in France from 2014 to 2021.
Materials and methods
To do this, we analyzed all ITs from the anti-programmed cell death protein 1/programmed death-ligand 1 [anti-PD-(L)1] class used in monotherapy or in association with another treatment available in early access or reimbursed in France between 2014 and 2021, regardless of indication. The number of patients initiating an IT was retrieved by year, drug and indication. Using extrapolated Kaplan–Meier curves, utility scores and the population treated, the clinical benefit was expressed as the number of deaths prevented (DP), life-years (LYs) and quality-adjusted life years (QALYs) gained compared to previous SoC.
Results
Across the period, five ITs were marketed in 21 indications related to eight primary tumor sites. Between 2014 and 2021, 132 924 patients initiated an IT. By December 2021, 16 173 (13 804-17 141) deaths were delayed compared to previous SoC, mainly in lung cancer. Compared to their SoC, ITs provided a gain of 37 316 (33 581-41 048) additional LYs and 27 709 (23 784-30 450) additional QALYs. Lung cancer was the driver indication with 70.6% of LYs and 68.4% of QALYs gained followed by melanoma with 18.7% and 20.4% of the gain, respectively.
Conclusions
Significant gains in DP, LYs and QALYs have been observed in France following the introduction of ITs.
Key words: clinical benefit, life-years, immunotherapies, oncology
Highlights
-
•
From 2014 to 2021, five ITs corresponding to 21 indications were available in France.
-
•
Between 2014 and 2021, 132 924 patients initiated an IT.
-
•
By the end of 2021, ITs were estimated to prevent or delay 16 173 deaths compared to previous SoC.
-
•
Over the follow-up, IT provided a gain of 37 316 LYs and 27 709 QALYs'.
Introduction
In 2020, the worldwide incidence of cancer was estimated at 19.3 million cases and almost 10.0 million cancer deaths occurred globally.1 In France, 157 000 deaths were related to cancer in 2018,2 making it the leading cause of deaths.3 Between 1990 and 2018, cancer mortality rates have decreased yearly by 1.8% for men and 0.8% for women. Multiple factors have participated in cancer mortality reduction, including prevention and control of risk factors, early screening and therapeutic advances.2,4, 5, 6 Prevention and control of risk factors like the elimination of tobacco may avoid 20% of cancer mortality by 2050.7 In parallel, cancer treatment innovations, such as targeted therapies or immunotherapies (ITs) have changed the landscape of cancer treatment.8,9 Anti-programmed cell death protein 1 (PD-1)/programmed death-ligand 1 (PD-L1) have demonstrated their efficacy in extending survival and improving quality of life10 in many advanced cancers, such as melanoma, lung cancer, renal cell carcinoma or head and neck cancer.11, 12, 13, 14 By the end of 2021, five ITs of the anti-PD-1/PD-L1 class used in monotherapy or in combination (atezolizumab, avelumab, durvalumab, nivolumab and pembrolizumab) have been approved in France in a growing number of indications, with an even increasing number of patients treated over time.
In France, the benefit of new treatments is evaluated by the Haute Autorité de Santé (HAS), the French national Health Technology Agency (HTA), before they are marketed to determine their price and reimbursement status.15 The assessment is composed of an evaluation of the actual benefit and the clinical added value of the drug in each indication compared to actual standards of care assessed by the Transparency Committee (TC). A cost-effectiveness (CE) and a budget-impact analysis may be required under certain conditions for an assessment by the Economic and Public Health Evaluation Committee (CEESP).16 As progression-free survival (PFS) and overall survival (OS) data from clinical trials are usually immature (i.e. some patients are censored), different extrapolations of the original PFS and OS Kaplan–Meier curves are proposed in the CE model, using different parametric distributions (such as exponential, log-normal or Weibull) to estimate the lifetime benefit of the drug. The best extrapolation is selected based on statistical parameters, visual inspection and external validation. All relevant comparators are included in the CE analysis. The CEESP assesses the methodology and the uncertainties associated with modelling aspects. TC and CEESP reports are published on the HAS website. However, drugs are rarely re-assessed after this initial evaluation, to ensure that the ‘observed’ benefits are consistent with the initial expectations. Moreover, if CE analyses provided information regarding the expected efficiency of the new drug at an individual patient level [expressed as the incremental cost per quality-adjusted life years (QALYs) gained and per patient], little is known on the overall benefit of the drug at the population level. Some studies demonstrated that innovative cancer drug approvals were associated with a reduction from 10% to 30% in cancer mortality between 1990 and 2011.17, 18, 19 These studies are few, were interested in different countries with heterogeneous HTA processes and are not specific to a given class of drugs. Thus, while ITs are now largely available in France and in >20 cancer indications, little is known about their benefit at a population level, in reducing mortality and increasing life-years (LYs) gained.
The objective of the present study was to assess the clinical benefit conferred by anti-PD-(L)1 ITs in monotherapy or in association with another treatment in advanced cancers at a population level in France, from their introduction in 2014 until the end of 2021 in terms of LYs gained, QALYs and the number of deaths avoided, compared to previous standards of care.
Materials and methods
We developed a dynamic open-cohort model, suitable for estimating the clinical benefit of ITs for treated populations in France between 2014 and 2021. Outcomes were expressed in LYs and QALYs gained, and the number of deaths avoided (or delayed) with IT, compared to the previous standard of care in each indication. To do that, the following steps were taken: firstly, we identified the different indications of IT approved during the period with their respective authorization dates. Secondly, we searched for associated CE reports to extract PFS, OS and utility values. We also searched the number of patients treated per year. Finally, we built a dynamic model to estimate the outcomes per indication or treatment line.
Identification of immunotherapies and their associated indications
The first step consisted of identifying all ITs and associated indications available either in the early-access process or reimbursed for the treatment of advanced or metastatic cancer in France before the end of 2021 (Figure 1, step 1). The HAS website was searched for all TC assessments and CE assessments of ITs during this period. To be included in the analysis, each immunotherapy indication had to fulfil the following criteria: (i) IT eligible for reimbursement or for an early-access program, (ii) extrapolated PFS and OS curves based on pivotal trials and no major methodological reservations on the modelling approach in the CE reports.
Figure 1.
Study methods. CEESP, Economic and Public Health Evaluation Committee; OS, overall survival; PFS, progression-free survival.
Reports relating to adjuvant and neo-adjuvant indications were excluded, as well as indications refused for reimbursement.
Data extraction
In the CE assessment reports publicly available on the HAS website, we first identified the comparator(s) of the IT drug in the given indication, by retaining only the comparator(s) considered as cost-effective for the HAS, defined as those on the CE frontier. In case of multiple comparators, the most cost-effective one was selected, which may not always be the standard of care. The following information was extracted both for the IT and its comparator in the CE assessment reports: indication, original PFS and OS Kaplan–Meier curves, extrapolated PFS and OS curves and utility values per health state. In the present study, we retained the extrapolated curves for PFS and OS that were validated by the HAS in the CE dossier and digitized them. Extrapolated OS curves were used to estimate LY. Extrapolated PFS and OS curves as well as utility values were used for the estimation of QALYs. Utility values are numerical values applied to health state based on the preference for this specific state relative to perfect health. QALYs are calculated by multiplying the time spent in PFS and post-progression survival by the utility score of the corresponding health state. One QALY corresponds to one LY in perfect health.
The published extrapolated PFS and OS curves of the IT and its comparator were digitized (Engauge Digitizer®, version 3.0, http://markummitchell.github.io/engauge-digitizer) and restructured (R® version 4.0.0, R Foundation, Indianapolis, IN) (Figure 1, steps 2 and 3).
Regarding the number of patients treated by indication, we searched for the number of patients initiating an IT (incident population) in French real-world cohorts from publications, studies or reports based on the French Hospital Medical Information database [Programme de Médicalisation des Systèmes d’Information (PMSI)] or official documents. When only the number of treated patients (prevalent population) was available, we applied the share of incident patients of the prevalent population of the closest year with the information available. Population estimates are described in Supplementary Table S1, available at https://doi.org/10.1016/j.esmoop.2024.102240.
The date of reimbursement or date of early-access authorization (if applicable) were identified and the length of time the treatment has been available in France was calculated.
Data analysis
For each indication, we created monthly cohorts of patients who initiated an IT during a given month. For each cohort, the number of patients concerned was calculated by dividing the annual number of incident patients by the number of months when the molecule was available in the indication considered. Thus, a multiple cohort model was used to represent the whole population who initiated IT treatment per month, and to follow their outcomes across the evaluation period. For each cohort, the evaluation period was defined as the time between the date of initiation of IT (index date) and 31 December 2021.
To calculate the number of deaths avoided, we used the survival rate from extrapolated curves for OS restructured in R software for the IT evaluated and its comparator. For each cohort, we retained the difference in survival probability between the two treatments as of 31 December 31 2021. This difference was multiplied by the number of incident patients from each cohort.
For the estimation of LYs gained, we calculated for each cohort and treatment the restricted mean survival time (RMST) between the index date and 31 December 31 2021. The RMST is a measure of average survival from index date to a specified time point, and may be estimated as the area under the survival curve up to that point. LYs gained were then obtained by multiplying the number of incident patients from each cohort by the difference in RMST between the IT and its comparator (Figure 1, step 4).
For the estimation of QALYs, we also estimated RMST based on the PFS curve between the index date and 31 December 31 2021, per cohort and per treatment. For each cohort, the time spent in the progression-free state was obtained as the difference in RMST based on PFS curve between the IT and its comparator. Similarly, the time spent in progressive disease state was calculated as the difference in RMST between PFS and OS. These differences were multiplied by the specific utility scores for the health state and the treatment considered, to obtain the mean QALY gained by the patient. This was then multiplied by the number of incident patients of the cohort.
The total number of avoided deaths, LYs and QALYs gained over the period is the sum of the estimations for all IT indication cohorts.
Sensitivity analyses
Sensitivity analyses were carried out by varying the RMST or survival rate, the utility values (Supplementary Table S2, available at https://doi.org/10.1016/j.esmoop.2024.102240), the size of the population initiating an IT and the period of availability.
Results
Identification of indication
Of the 188 CE assessment reports available on the HAS website up to 2021, 34 concerned ITs in advanced or metastatic cancer (Supplementary Figure S1, available at https://doi.org/10.1016/j.esmoop.2024.102240). Fifteen were excluded from the analysis due to the absence of reimbursement (or early access) between 2014 and 2021 (n = 12), due to the modeling not being based on clinical trials data (n = 1) or because the extrapolated curves were not accepted by the HAS (n = 2). The characteristics of the remaining 19 assessments relating to 21 treatment indications are presented in Table 1 and Supplementary Table S3, available at https://doi.org/10.1016/j.esmoop.2024.102240.
Table 1.
Description of each case study
| Cancer location and stage | Treatment line | Immunotherapy | Most cost-effective comparator | Early access date | Official reimbursement date of the immunotherapy | Utility values used in the cost-effectiveness assessment report |
ESMO-MCBS score | |
|---|---|---|---|---|---|---|---|---|
| Progression-free survival | Post-progression | |||||||
| Metastatic hepatocellular cancer | First line | Atezolizumab (+bevacizumab) | Sorafenib | Mid-July 2020 | October 2021 | 0.8933 | 0.8634 | 5 |
| Metastatic melanoma | First line | Nivolumab | Fotemustine | January 2015 | January 2017 | 0.823 | 0.729 | A/4 |
| First line | Nivolumab (+ ipilimumab) | Fotemustine | NA | May 2019 | 0.823 | 0.729 | A/4 | |
| First line | Pembrolizumab | Fotemustine | Mid-September 2014 | January 2017 | 0.810 | 0.680 | A/4 | |
| Mesothelioma | First line | Nivolumab (+ ipilimumab) | Chemotherapy | April 2021 | February 2022 | 0.716 | 0.580 | 3 |
| Advanced or metastatic non-small-cell lung cancer | Second-line squamous | Nivolumab | Docetaxel | Mid-May 2015 | January 2017 | 0.723 | 0.530 | 5 |
| Second-line non-squamous | Nivolumab | Docetaxel | June 2015 | March 2017 | 0.743 | 0.659 | 5 | |
| Second line | Pembrolizumab | Docetaxel | NA | Mid-May 2017 | 0.737 | 0.628 | 5 | |
| Second line | Atezolizumab | Docetaxel | NA | Mid-February 2019 | 0.7043 | 0.550 | 5 | |
| First-line squamous (PD-L1 ≥50%) | Pembrolizumab | Platinum-based chemotherapy | NA | December 2017 | 0.760 | 0.641 | 5 | |
| First-line non-squamous (PD-L1 ≥50%) | Pembrolizumab | Bevacizumab + paclitaxel | NA | December 2017 | 0.760 | 0.641 | 5 | |
| First-line non-squamous | Pembrolizumab (+ chemotherapy) | Platinum + gemcitabine/vinorelbine | NA | Mid-November 2019 | 0.726 | 0.644 | 4 | |
| First-line squamous | Pembrolizumab (+ chemotherapy) | Platinum + paclitaxel | NA | June 2020 | 0.741 | 0.618 | 4 | |
| First-line maintenance locally | Durvalumab | Placebo | April 2018 | May 2020 | 0.795 | 0.751 | 4 | |
| Renal cell carcinoma metastatic | Second line | Nivolumab | Sorafenib | NA | January 2017 | 0.824 | 0.744 | 5 |
| First line | Nivolumab (+ ipilimumab) | Pazopanib | NA | March 2020 | 0.749 | 0.687 | 4 | |
| First line | Pembrolizumab (+ axitinib) | Pazopanib | NA | June 2020 | 0.7846 | 0.7529 | 4 | |
| Advanced Squamous cell cancer of head and neck | Second line | Nivolumab | Standard treatments | NA | June 2018 | 0.743 | 0.628 | 5 |
| First line | Pembrolizumab | Platinum + 5-FU + cetuximab | NA | November 2020 | 0.764 | 0.676 | 4 | |
| Metastatic small-cell lung cancer | First line | Atezolizumab (+ chemotherapy) | Platinum + etoposide | May 2019 | Mid-June 2022 | 0.7291 | 0.7118 | 3 |
| Urothelial cancer | First-line maintenance | Avelumab | Placebo | July 2020 | Mid-September 2022 | 0.894 | 0.840 | 4 |
5-FU, 5-fluorouracil; MCBS, Magnitude of Clinical Benefit Scale; NA, not applicable; PD-L1, programmed death-ligand 1.
Of the 21 IT indications retained, three concerned metastatic melanoma, nine non-small-cell lung cancer (NSCLC), three renal cell carcinoma (RCC) and two squamous cell cancer of head and neck (SCCHN). The remaining indications concerned one approval each in small-cell lung cancer (SCLC), urothelial carcinoma (UC), hepatocellular carcinoma (HCC) and mesothelioma (n = 1). The treatment was indicated as first-line treatment in 13 cases, as second-line in 6 cases and as first-line maintenance in 2 cases. Most indications involved pembrolizumab in monotherapy (n = 5) or in combination (n = 3), and nivolumab in monotherapy (n = 5) or in combination (n = 3). The remaining treatments were atezolizumab (n = 3), durvalumab (n = 1) and avelumab (n = 1). An early access was implemented for nine (43%) indications.
Number of patients initiating an anti-PD(L)1 immunotherapy
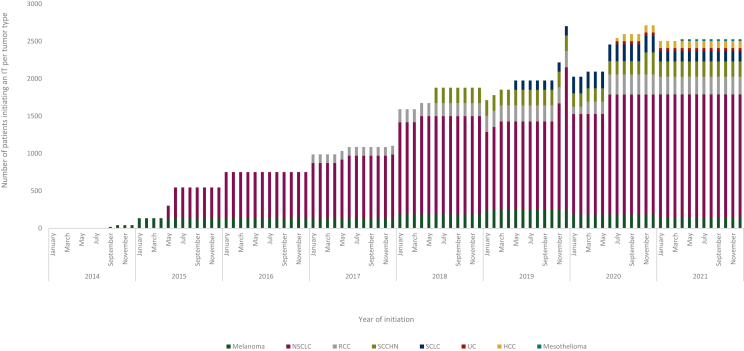
The number of patients initiating an IT increased over time, from 18 patients in September 2014 to 2600 patients in December 2021 (Figure 2). Overall, 132 924 patients initiated an IT (Table 2). The earliest indication in which patients initiated an IT was metastatic melanoma (88 months of use up to 31 December 2021), followed by NSCLC (79.5 months of use). For all indications, the number of patients initiating an IT tended to increase over time, except in melanoma where a decrease was observed in early 2020 (Supplementary Figure S2, available at https://doi.org/10.1016/j.esmoop.2024.102240). From its launch, until the end of 2021, NSCLC was the tumor with most patients initiating an IT, representing 66.5% of all patients. All other tumors accounted for <10% of patients, except for melanoma.
Figure 2.
Patients initiating per month per tumor type over the follow-up. HCC, hepatocellular carcinoma; ICI, immune checkpoint inhibitor; NSCLC, non-small-cell lung cancer; RCC, renal cell carcinoma; SCCHN, squamous cell cancer of head and neck; SCLC, small-cell lung cancer; UC, urothelial carcinoma.
Table 2.
Number of avoided deaths, life years gained and QALYs gained by immunotherapy compared to the most cost-effective comparator up to 31 December 2021
| Duration between December 2021 and first availability (months) | Number of patients initiating an immunotherapy n (%) | Deaths avoided n (%) | Percentage of avoided deaths in patients initiating an immunotherapy (%) | Life years gained n (%) | QALYs gained n (%) | ||
|---|---|---|---|---|---|---|---|
| Total | 88 | 132 924 | 16 173 | 12.2 | 37 316 | 27 709 | |
| Tumor type | Melanoma | 88 | 16 297 (12.3) | 2555 (15.8) | 15.7 | 6990 (18.7) | 5645 (20.4) |
| Non-small-cell lung | 79.5 | 88 443 (66.5) | 11 368 (70.3) | 12.9 | 26 334 (70.6) | 18 952 (68.4) | |
| Renal cell carcinoma | 60 | 11 506 (8.8) | 853 (5.3) | 7.4 | 1902 (5.1) | 1636 (5.9) | |
| Squamous cell cancer of head and neck | 43 | 8757 (6.7) | 708 (4.4) | 8.1 | 1457 (3.9) | 995 (3.6) | |
| Small-cell lung | 32 | 5268 (4.0) | 465 (2.9) | 8.8 | 520 (1.4) | 375 (1.4) | |
| Urothelial | 18 | 788 (0.6) | 76 (0.5) | 9.6 | 38 (0.1) | 37 (0.1) | |
| Hepatocellular | 18 | 1651 (1.3) | 146 (0.9) | 8.9 | 75 (0.2) | 70 (0.3) | |
| Mesothelioma | 9 | 215 (0.2) | 4 (<0.01) | 1.9 | 0.5 (<0.1) | −0.7 (<0.1) | |
| Treatment lines | Second line | 79.5 | 67 562 (50.8) | 6911 (42.7) | 10.2 | 22 384 (60.0) | 15 689 (56.6) |
| First line | 88 | 59 597 (44.8) | 8577 (53.0) | 14.4 | 14 170 (38.0) | 11 353 (41.0) | |
| Other treatment lines | 45 | 5764 (4.3) | 670 (4.2) | 11.9 | 761 (2.0) | 667 (2.4) | |
| Treatment | Atezolizumab | 35 | 11 145 (8.4) | 1063 (6.6) | 9.5 | 1136 (3.0) | 757 (2.7) |
| Avelumab | 18 | 788 (0.6) | 76 (0.5) | 9.6 | 38 (0.1) | 37 (0.1) | |
| Durvalumab | 45 | 4977 (3.7) | 610 (3.8) | 12.3 | 723 (1.9) | 631 (2.3) | |
| Nivolumab | 84 | 68 405 (51.5) | 7357 (45.5) | 10.8 | 24 115 (64.6) | 17 484 (63.1) | |
| Pembrolizumab | 88 | 47 609 (35.8) | 7068 (43.7) | 14.8 | 11 303 (30.3) | 8800 (31.8) | |
| Early access | NA | 24 594 (18.5) | 2369 (14.6) | 9.6 | 2254 (6.0) | 1639 (5.9) | |
NA, not applicable; QALYs, quality-adjusted life years.
The first two ITs available in France were indicated for first-line treatment of metastatic melanoma. After that, all indications were for second-line treatment until December 2017. Second-line treatments represented 69.0% of the patients initiating an IT until the end of 2019. Thereafter, the patients mostly initiate an IT in the first line (Supplementary Figure S2, available at https://doi.org/10.1016/j.esmoop.2024.102240). Over the period, 50.8% of patients initiated an IT in the second line, 44.8% in first line and <5% in first-line maintenance (Table 2). Nivolumab was the treatment initiated by more than half of the patients (51.5%), followed by pembrolizumab (35.8%). However, treatment patterns evolved over time: nivolumab was the largest contributor until the end of 2019, whereafter pembrolizumab was the most widely used (Supplementary Figure S3, available at https://doi.org/10.1016/j.esmoop.2024.102240). Other treatments were used by <10% of initiating patients.
Avoided deaths, life-years and QALYs gained
In the end of 2021, IT contributed to 16 173 avoided (or delayed) deaths, as compared to the standard care (Table 2). The largest contributors to these avoided deaths were NSCLC (70.3%) and melanoma (15.8%). RCC and SCCHN represented 5.3% and 4.4% of avoided deaths, respectively, since ITs have been available more recently in these indications. UC, HCC and mesothelioma contributed to <1% of avoided deaths. Most avoided deaths concerned first-line (53.0%) indications. Treatments contributing the most to avoided deaths were nivolumab (45.5%) and pembrolizumab (43.7%). Of the 88 443 patients initiating an IT for NSCLC, 11 368 (12.9%) were still alive at the end of 2021.
By the end of 2021, ITs contributed to 37 316 LYs gained in France, as compared to the comparator. NSCLC and melanoma were the indications with the largest gains with 70.6% and 18.7%, respectively. RCC accounted for 5.1% and SCCHN for 3.9 % of the LYs gained, whereas all indications available after 2019 (SCLC, UC, HCC and mesothelioma) accounted for <5% of LYs gained. Second-line indications accounted for most LYs gained (60.0%). Nivolumab accounted for more than half the LYs gained (64.6%) and pembrolizumab for a third of the gain (30.3%). Treatments launched later (avelumab, atezolizumab and durvalumab) accounted for <5% of the LYs.
By the end of 2021, 27 709 QALYs had been gained using IT, as compared to their comparator. NSCLC and melanoma were the indications with the largest gains (68.4% and 20.4% of QALYs, respectively). As for LYs, second-line indications accounted for most QALYs gained (56.6%). Nivolumab contributed to more than half (63.1%) of the QALYs gained and pembrolizumab around a third of the gain (31.8%). Treatments launched later accounted for <5% of the QALYs.
Early-access contribution
Nine indications benefited from an early-access program over the follow-up (Table 1). These programs allowed the initiation of an IT for 24 594 patients (18.5%) over the period (Table 2). Patients participating in these programs contributed in 14.6% of deaths avoided, 6.0% of LYs gained and 5.9% of QALYs gained.
Sensitivity analyses
The deterministic sensitivity analyses demonstrated little impact on avoided deaths, LYs and QALYs gained (Supplementary Figure S4, available at https://doi.org/10.1016/j.esmoop.2024.102240). With respect to deaths (Supplementary Figure S4A, available at https://doi.org/10.1016/j.esmoop.2024.102240), the hypothesis that had the largest impact was a decrease of 14.6% of deaths avoided when the benefits associated with the early-access program were not considered. The other parameters tested had an impact of <7%. Based on these analyses, the avoided deaths ranged from 13 804 to 17 141.
With respect to LYs gained (Supplementary Figure S4B, available at https://doi.org/10.1016/j.esmoop.2024.102240), the largest impact is associated with the variation of ±10% of the RMST for OS, which have a linear impact on the LYs. The other parameters had an impact of ≤5%. Based on these estimations, the LYs gained vary from 33 581 to 41 048.
With respect to QALYs gained (Supplementary Figure S4C, available at https://doi.org/10.1016/j.esmoop.2024.102240), three analyses had an impact of >5%: no difference in PFS (−14%), variation by 10% of the RMST of PFS and post-progression (±10%), deleting the early-access gains (−6%). Based on these analyses, the QALYs gained vary from 23 784 to 30 450.
Discussion
Based on official and institutional reports, we estimated the clinical benefits since the introduction of a new class of drugs, compared to the previous standard of care, at a population level. We captured avoided deaths and LYs and QALYs gained since the availability of IT (2014) until December 2021. This study does not address comparisons of the difference of efficacy between ITs, since they are not always reimbursed in the same indications and treatment line but also because we estimated benefit at the population-treated level, so results depend on population size. We estimated that 132 924 patients initiated one of the ITs available in France in one of the 21 indications between 2014 and 2021.
The National Institute of Cancer has identified an annual decrease in cancer mortality between −0.7% and −2% in France attributable to earlier diagnosis and significant therapeutic advances.19 At 31 December 2021 landmark, ITs enabled 16 173 (range: 13 804-17 141) deaths to be avoided, corresponding to 12.2% of additional patients surviving compared to the previous standard of care at this date. The number of initiating patients in our study corresponded to 4.2% of incident cancer patients in France, but deaths avoided represents 10% of annual deaths meaning ITs have had a large positive impact on cancer mortality.2 In comparison, before the introduction of ITs, an Australian study showed 16 833 avoided deaths between the period 2005 and 2014 in 13 tumor types, mainly by early diagnosis and screening.20 Their results considered a longer follow-up period and more tumor but similar outcomes in deaths avoided, confirming the benefits of immune checkpoint inhibitors (ICIs). In our study, most of the deaths avoided (70.3%) concerned NSCLC, as it is the location with higher number of indications and with the largest number of patients (66.5%). We estimated that death was avoided for 12.9% of patients initiating an IT for NSCLC. Previous studies exploring deaths avoided in Australia and the United Kingdom,21,22 have described lung cancer (NSCLC and SCLC) as one of the cancers with the lowest number of avoidable deaths (<6% reduction in deaths between 2005 and 2014 compared to those between 1985 and 1994).22 IT have had a major impact on avoided deaths in NSCLC since their launch. First-line treatments have the largest impact on deaths avoided, which is consistent with previous results explaining the importance of early cancer care.
As the number of avoided deaths are calculated at a specific landmark (31 December 31 2021) and did not represent cured patients but difference in deaths between the IT and previous standard of care, we also expressed the benefit in the LYs and QALYs gained. Overall, 37 316 (range: 33 581-41 048) LYs and 27 709 (range: 23 784-30 450) QALYs were gained in France since the availability of ITs. As for avoided deaths, NSCLC was the largest contributor with 70.6% of LYs and 68.4% of QALYs gained. The share of LYs and QALYs gained in melanoma, 18.7% and 20.4%, respectively, are superior to the share of avoided death (15.8%). This is partly correlated with the longer availability of ITs in this indication, since estimation of LYs and QALYs gained is cumulative and considers the period for which treatment was available. Different patterns of LYs and QALYs gained are observed in NSCLC, RCC and melanoma. The larger shares of QALYs for melanoma and RCC can be explained by the higher unit utility values in these indications. Second-line treatments accounted for most LYs and QALYs gained but only 42.7% of avoided deaths. This is caused by the longer time of availability considered favorably for LY and QALY analyses.
In mesothelioma, ITs seem to reduce the number of QALYs gained. However, IT was only available for 9 months of follow-up in our analysis; in the clinical trials, their relative benefits only appear after around 10 months in this indication.
An early-access program was available in France for nine treatment indications and allowed 24 594 patients to start an IT earlier, representing almost 20% of all initiating patients. This program accounted for 14.6% of avoided deaths, 6.0% LYs and 5.9% QALYs gained. The larger share in avoided deaths compared to LYs and QALYs is explained by the recent introduction of five of them (since 2018). In these cases, less hindsight is available reducing the number of LYs gained compared to previous indications. These results confirm the benefit of the recent extension of early-access programs in France.23,24
The principal limitation of this study is related to the data sources. The number of incident patients is derived from heterogeneous sources: the PMSI, official published documents and ad hoc hypotheses in some cases, which leads to assumptions. In some cases, we only had the number of prevalent patients per year and applied a percentage to generate an estimate of the number of incident patients. However, these hypotheses have been tested in the sensitivity analyses. Moreover, we considered an equal distribution of patients over the availability period per year. The second limitation is for the long-term survival estimation. Our study was based on extrapolated PFS and OS curves from HAS economic evaluations which are projections made at the time of the claim of reimbursement. These may incorrectly estimate clinical benefit over time since, in clinical trials, the study population is selected through more or less stringent inclusion and exclusion criteria, compared with everyday practice, where all eligible patients may be treated. To limit the risk of misestimation, we used only the economic evaluations validated by the HAS leading to the exclusion of some IT indications and tested the results in sensitivity analyses. Moreover, a previous study indicated that extrapolation tends to underestimate long-term OS in patients treated with ICIs25 suggesting that we present here a minimal estimation of the benefit. In addition, extrapolations were carried out using data from results of phase III studies (as these data have the highest internal validity), whereas a large difference may exist between the trial population and treated population with reimbursement, especially regarding performance status and comorbidity indexes. We carried out a comparative review of patient characteristics and outcomes between clinical trials and French real-life studies which showed minimal difference in outcomes even with more comorbidities and lower functional status in real-life practice (Supplementary Table S4, available at https://doi.org/10.1016/j.esmoop.2024.102240). The third limitation is that in anti-PD-(L)1 association with other treatments, we were not able to isolate the specific benefit of the anti-PD-(L)1 in the overall benefit of the regimen. In these cases, the result obtained corresponds to the benefit of the treatment association. The fourth limitation concerns the selected comparator. Indeed, since CE analysis includes all relevant comparators, the most cost-effective is not always the standard of care in real-world practice. The last limitation concerns toxicities and their impact on utility values. We did not integrate disutility values associated with short- or long-term toxicities since some CE reports only integrate short-term disutility values and these data were not estimated with the same methods. However, the utility values were tested in sensitivity analyses and had a limited impact on the results. Consequently, QALY analyses only reflect to clinical outcomes and not adverse events.
This is the first study to estimate the clinical benefits to the overall population treated brought by anti-PD-(L)1 ITs in monotherapy or in association. This is also the first study to consider deaths avoided, as well as LYs and QALYs gained which are complementary measures of health benefit. The number of deaths avoided can provide a tangible estimate of improvements in cancer survival. The number of LYs gained can provide information on the duration of the treatment effect and the number of QALYs gained can provide information on quality of life during the survival period. This analysis is likely to underestimate the full potential benefits of ICIs, since it did not include indications in adjuvant settings (melanoma), stopped at the end of 2021 and did not take into account gains accrued or anticipated thereafter nor new indications that have or will become available after this date; indeed, more than 50 phase III trials are ongoing (see http://clinicaltrials.gov).
In conclusion, this study enabled us to estimate increases in cancer survival attributable to IT independently from other factors. The introduction of ITs in advanced cancer has had an important public health impact at the population level in France and confirmed the importance of early access to these treatments. This is a new approach to estimate overall clinical benefits of a therapeutic class at the population level. Further analyses including survival results in the early-stage setting would be interesting to justify the importance of implementing ITs in these settings.
Acknowledgements
We acknowledge Adam Doble from SARL Foxymed for his editorial assistance.
Funding
This work was supported by Bristol-Myers Squibb (no grant number).
Disclosure
VG has a contract at Bristol Myers Squibb for a PhD. FEC and AFG are employed by Bristol Myers Squibb. EGL received personal fees (advisory boards) from Bristol Myers Squibb. CL reports personal fees and nonfinancial support from Amgen, Bristol Myers Squibb, Incyte, Merck Sharp & Dohme, Novartis, Pfizer, Pierre Fabre, Roche and Sanofi, outside the submitted work. IB has declared no conflicts of interest.
Data sharing
Number of patients treated was identified in public reports or publication. However, in some cases, the population were identified in the National Health Data System (SNDS, Système national des Données de Santé) and are available from the HDH (Health Data Hub https://www.health-data-hub.fr/). Restrictions apply to the availability of these data containing potentially identifying and sensitive patient information.
Since this was a retrospective study of an anonymized database and had no influence on patient care, ethics committee approval was not required. The study was carried out according to the MR-006 guideline of the French data protection agency (CNIL, Commission Nationale de l’Informatique et des Libertés). This framework allows pharmaceutical industries to access PMSI data under strict conditions. The study must be of public interest. The data controllers—Bristol Myers Squibb—must declare the study process in an activity registry supervised by the SNDS (declaration no. F20210623164242/no. I57141211192019). Patient anonymity and data confidentiality must be guaranteed; patient data linkage with other datasets is impossible. Access to the data is only possible through certified third-party research consultancy/laboratories—HEVA. The third party must comply with a reference system set by the CNIL (Decree of 17 July 2017), is independently audited every third year and complies with the ban on prohibited purposes.
This study is a retrospective study based on claims data. Patient consent was not necessary (article R.1461-9 of the French Public Health Code).
All data relevant to the study are included in the article or uploaded as Supplementary Material, available at https://doi.org/10.1016/j.esmoop.2024.102240.
Supplementary data
References
- 1.Sung H., Ferlay H., Siegel C., et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Defossez G., Le Guyader-Peyrou S., Uhry Z., et al. Santé Publique France; Saint-Maurice: 2019. Estimations nationales de l’incidence et de la mortalité par cancer en France métropolitaine entre 1990 et 2018. Étude à partir des registres des cancers du réseau Francim. Volume 1 - Tumeurs solides. Compléments par localisation tumorale et sous-sites ou sous-types histologiques. [Google Scholar]
- 3.Direction de la recherche, des études de l’évaluation et des statistiques. L’état de santé de la population en France. Rapport 2017. Principales causes de décès et de morbités; 2017. [Google Scholar]
- 4.Cardoso R., Guo F., Heisser T. Colorectal cancer incidence, mortality, and stage distribution in European countries in the colorectal cancer screening era: an international population-based study. Lancet Oncol. 2021;22(7):1002–1013. doi: 10.1016/S1470-2045(21)00199-6. [DOI] [PubMed] [Google Scholar]
- 5.Levin T., Corley D., Jensen C., et al. Effects of organized colorectal cancer screening on cancer incidence and mortality in a large community-based population. Gastroenterology. 2018;155(5):1383–1391.e5. doi: 10.1053/j.gastro.2018.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Arbryn M., Raifu A., Weiderpass E., et al. Trends of cervical cancer mortality in the member states of the European Union. Eur J Cancer. 2009;45(15):2640–2648. doi: 10.1016/j.ejca.2009.07.018. [DOI] [PubMed] [Google Scholar]
- 7.La Vecchia C., Negri E., Carioli G., et al. Progress in cancer epidemiology: avoided deaths in Europe over the last three decades. Eur J Cancer Prev. 2022;31(4):388–392. doi: 10.1097/CEJ.0000000000000714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Martin-Liberal J., Ochoa de Olza M., Hierro C., et al. The expanding role of immunotherapy. Cancer Treat Rev. 2017;54:74–86. doi: 10.1016/j.ctrv.2017.01.008. [DOI] [PubMed] [Google Scholar]
- 9.Mittra A., Takebe N., Florou V., Chen A.P., Naqash A.R. The emerging landscape of immune checkpoint inhibitor based clinical trials in adults with advanced rare tumors. Hum Vaccin Immunother. 2021;17:1935–1939. doi: 10.1080/21645515.2020.1854604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Boutros A., Bruzzone M., Tanda T.E., et al. Health-related quality of life in cancer patients treated with immune checkpoint inhibitors in randomised controlled trials: a systematic review and meta-analysis. Eur J Cancer. 2021;159:154–166. doi: 10.1016/j.ejca.2021.10.005. [DOI] [PubMed] [Google Scholar]
- 11.Topalian S.L., Hodi F.S., Brahmer J.R., et al. Five-year survival and correlates among patients with advanced melanoma, renal cell carcinoma, or non-small cell lung cancer treated with nivolumab. JAMA Oncol. 2019;5:1411–1420. doi: 10.1001/jamaoncol.2019.2187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Debieuvre D., Juergens R.A., Asselain B., et al. Two-year survival with nivolumab in previously treated advanced non-small-cell lung cancer: a real-world pooled analysis of patients from France, Germany, and Canada. Lung Cancer. 2021;157:40–47. doi: 10.1016/j.lungcan.2021.04.022. [DOI] [PubMed] [Google Scholar]
- 13.Polkowska M., Ekk-Cierniakowski P., Czepielewska E., et al. Survival of melanoma patients treated with novel drugs: retrospective analysis of real-world data. J Cancer Res Clin Oncol. 2017;143:2087–2094. doi: 10.1007/s00432-017-2453-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Assie J.B., Corre R., Levra M.G., et al. Nivolumab treatment in advanced non-small cell lung cancer: real-world long-term outcomes within overall and special populations (the UNIVOC study) Ther Adv Med Oncol. 2020;12 doi: 10.1177/1758835920967237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Haute Autorité de santé description. https://www.has-sante.fr/jcms/c_415958/en/about Available at.
- 16.Journal Officiel:Décret n° 2012-1116 du 2 octobre 2012 relatif aux missions de la Haute Autorité de santé médico-économiques. https://www.legifrance.gouv.fr/jorf/id/JORFTEXT000026453514 Available at.
- 17.Dubois P., Kyle M. Toulouse School of Economics (TSE); 2016. The Effects of Pharmaceutical Innovation on Cancer Mortality Rates. [Google Scholar]
- 18.Lichtenberg F. National Bureau of Economic Research; Cambridge (MA): 2010. Has Medical Innovation Reduced Cancer Mortality? [Google Scholar]
- 19.MacEwan J., Dennen S., Kee R., et al. Changes in mortality associated with cancer drug approvals in the United States from 2000 to 2016. J Med Econ. 2020;23(12):1558–1569. doi: 10.1080/13696998.2020.1834403. [DOI] [PubMed] [Google Scholar]
- 20.Institut National du Cancer Panorama des cancers en France. 2022. https://www.e-cancer.fr/pdf_inca/preview/303372/4327939/file/Panorama%20des%20cancers%20en%20France_2021.pdf Available at.
- 21.Dasgupta P., Cramb S.M., Kou K., et al. Quantifying the number of cancer deaths avoided due to improvements in cancer survival since the 1980s in the Australian Population, 1985-2014. Cancer Epidemiol Biomarkers Prev. 2020;29(9):1825–1831. doi: 10.1158/1055-9965.EPI-20-0299. [DOI] [PubMed] [Google Scholar]
- 22.Richards M.A., Stockton D., Babb P., et al. How many deaths have been avoided through improvements in cancer survival? BMJ. 2000;320(7239):895–898. doi: 10.1136/bmj.320.7239.895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jacquet E., Kerouani-Lafaye G., Grude F., et al. Comparative study on anticancer drug access times between FDA, EMA and the French temporary authorisation for use program over 13 years. Eur J Cancer. 2021;149:82–90. doi: 10.1016/j.ejca.2021.03.008. [DOI] [PubMed] [Google Scholar]
- 24.Albin N., Chassagnol F., Bergmann J.F., et al. Early access to health products in France: major advances of the French "Conseil stratégique des industries de santé" (CSIS) to be implemented (modalities, regulations, funding) Therapie. 2019;74(1):103–117. doi: 10.1016/j.therap.2018.12.002. [DOI] [PubMed] [Google Scholar]
- 25.Grumberg V., Roze S., Chevalier J., et al. A review of overall survival extrapolations of immune-checkpoint inhibitors used in health technology assessments by the French Health Authorities. Int J Technol Assess Health Care. 2022;38(1):e28. doi: 10.1017/S0266462322000125. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.