Abstract
Background
Live attenuated vaccines alter immune functions and are associated with beneficial outcomes. We previously demonstrated that live attenuated yellow fever virus (YFV) vaccine (LA-YF-Vax) dampens T-cell receptor (TCR) signaling in vitro via an RNA-based mechanism. We examined study participants before and after LA-YF-Vax to assess TCR-mediated functions in vivo.
Methods
Serum samples and peripheral blood mononuclear cells (PBMCs) were obtained before and after LA-YF-Vax (with or without additional vaccines) or quadrivalent influenza vaccine. TCR-mediated activation was determined by interleukin 2 release or phosphorylation of the lymphocyte-specific Src kinase. TCR-regulating phosphatase (protein tyrosine phosphatase receptor type E [PTPRE]) expression was also measured.
Results
Compared with prevaccination findings, LA-YF-Vax recipient PBMCs demonstrated transient reduction in interleukin 2 release after TCR stimulation and PTPRE levels, unlike in control participants who received quadrivalent influenza vaccine. YFV was detected in 8 of 14 participants after LA-YF-Vax. After incubation of healthy donor PBMCs in serum-derived extracellular vesicles prepared from LA-YF-Vax recipients, TCR signaling and PTPRE levels were reduced after vaccination, even in participants without detectable YFV RNA.
Conclusions
LA-YF-Vax reduces TCR functions and PTPRE levels after vaccination. Extracellular vesicles from serum recapitulated this effect in healthy cells. This likely contributes to the reduced immunogenicity for heterologous vaccines after LA-YF-Vax administration. Identification of specific immune mechanisms related to vaccines should contribute to understanding of the “off-target,” beneficial effects of live vaccines.
Keywords: live attenuated yellow fever vaccine, T-cell receptor, T lymphocyte, vaccine, Off-target vaccine effects, yellow fever virus, PTPRE
Live attenuated vaccines induce beneficial clinical outcomes, thought to be due to immune modulation. After yellow fever virus vaccination, we observed a reduction in T-cell receptor signaling. This may be a potential “off target” effect that influences clinical outcomes.
Vaccines, in addition to reducing infections by the pathogen for which they are designed, induce alterations in immune responses resulting in “off-target” consequences [1–17]. While some of these off-target effects may be detrimental resulting in adverse events (eg, cardiomyopathy from severe acute respiratory syndrome vaccines) [18], several live attenuated vaccines (LAVs) appear to have beneficial, nonvaccine target outcomes. Specifically, LAVs are associated with a reduction in hospitalization rates, adverse cardiovascular events, and all-cause mortality in a variety of settings [2–8, 10, 11, 17]. A World Health Organization commission concluded in 2014 that BCG and measles vaccines are transiently associated with an almost 50% reduction in all-cause mortality rates and that these effects are unlikely explained to any large extent by the prevention of tuberculosis and measles [1, 2]. LAVs more closely model natural infection compared with subunit or killed vaccines, and interestingly, nonreplicating (subunit or killed) vaccines are not typically associated with beneficial outcomes [7, 12]. Although the mechanisms underlying these beneficial effects are poorly understood, they are generally thought to be related to uncharacterized immunological modulation induced by the replicating vaccine pathogen [19–21].
Although these “off-target” effects are often described as “nonspecific,” vaccine-induced immune modulation presumably uses specific but unrecognized mechanisms. Identification of how LAVs regulate immune function may provide insights into viral pathogenesis and identify novel approaches to ameliorate or treat inflammatory-based diseases. We previously found that several viruses within the Flaviviridae family, specifically human pegivirus (HPgV), hepatitis C virus (HCV), and yellow fever virus (YFV) infections, modulate immunity by inhibiting T-cell activation in vitro [22–26]. Three virus-specific mechanisms were identified that result in reduced signaling through the T-cell receptor (TCR) [24–26]. One mechanism was shared by HCV and YFV and involves a virus genome–derived, short RNA (vsRNA). Although the viral sequences are different, both HCV and YFV generate vsRNAs that target and reduce the expression of a regulatory phosphatase (protein tyrosine phosphatase receptor type E [PTPRE]), resulting in reduced TCR-induced activation [24–26]. This outcome likely contributes to the reduced immune responses to vaccines in individuals with HCV infection and after LAV administration [27–31]. To determine whether vaccination with live attenuated YFV vaccine (LA-YF-Vax) alters T-cell functions in vivo, we examined TCR signaling and PTPRE expression in individuals before and after immunization with LA-YF-Vax.
MATERIALS AND METHODS
Study Participants
Healthy study participants, including individuals attending the University of Iowa Travel Clinic who were traveling to countries requiring YFV vaccine or who had received seasonal inactivated quadrivalent influenza vaccine (QIV), were invited to participate. This study was approved by the University of Iowa Institutional Review Board, and all participants (healthy donors and those receiving vaccines) provided written informed consent. After written informed consent, blood samples were obtained for use in the studies outlined below. No participant had a history of human immunodeficiency virus (HIV) or hepatitis B or C virus infection or immune suppressive medication use. Laboratory personnel conducting the studies were not aware of the vaccines received or of the timing of sample acquisition relative to vaccination.
Cell Isolation and Stimulation
Serum, plasma, and peripheral blood mononuclear cells (PBMCs) were prepared from blood obtained from healthy donors within 4 hours of acquisition. as described elsewhere [32–34]. PBMCs were either used fresh or stored in liquid nitrogen, as described elsewhere [32]. For TCR stimulation studies, vaccine recipient PBMCs stored in liquid nitrogen (cell count, 1 × 106/mL) were incubated with plate-bound anti-CD3 (100 ng/mL, OKT3 clone, eBioscience), and interleukin 2 (IL-2) released into cell culture supernatants was quantified 16 hours after stimulation using a human IL-2 enzyme-linked immunosorbent assay kit (BD Biosciences), as described elsewhere, and according to the manufacturer's instructions [23, 24]. Each experiment was performed in triplicate.
PTPRE, Lymphocyte-specific protein tyrosine kines (Lck), and phosphorylated active site (Y394) Lck were measured either in vaccine recipient PBMCs or in healthy donor PBMCs before and 10 minutes after TCR stimulation with anti-CD3, as described elsewhere [23, 24, 26]. Proteins were separated by polyacrylamide gel electrophoresis and transferred to nitrocellulose membranes (Bio-Rad). Membranes were incubated in protein-free blocking buffer (Thermo Scientific) for 1 hour at room temperature, followed by incubation with primary antibodies. Activated (phosphorylated) and total Lck and PTPRE were detected using immune blot, as described elsewhere [25, 26]. Primary antibodies (Origene) included antibodies binding phosphorylated Lck [(Lck-p;] Y394/pSrcY416), total Lck, PTPRE (clone 4B2), and β-actin (clone 8H10). Proteins were detected with Amersham enhanced electrochemiluminescence (GE Healthcare) using a Kodak imager. Immunoblots were quantified using ImageJ software IJ 1.46r [24].
Cells, Virus, and Viral Quantification
YFV (vaccine strain 17D; Sanofi) was purchased and stock virus preparations were grown in Vero cells (American Type Culture Collection), maintained as described elsewhere [26]. YFV RNA was detected and quantified with real-time RT-PCR, and YFV infectivity was measured using the median tissue culture infective dose, as described elsewhere [26]. Serum YFV neutralization was determined using a YFV microneutralization method [33, 35]. Briefly, heat-inactivated serum samples from study participants were diluted 1:100 in medium and then mixed with YFV (1 × 106 infectious doses) for 1 hour at 37°C before quantification by median tissue culture infective dose, as described elsewhere [33]. Positive results were defined as a reduction in YFV titer by >90% at the 1:100 dilution. Genomic YFV RNA was detected with real-time RT-PCR, as described elsewhere [33, 35].
Extracellular Vesicle Isolation
Extracellular vesicles (EVs) were purified from human serum using the ExoQuick reagent (Systems Biosciences), according to the manufacturer's instructions and as described elsewhere [24, 36, 37]. The serum EV pellets were resuspended in Roswell Park Memorial Institute 1640 medium and stored at −20°C until use. ExoQuick is a commercial reagent that has been previously reported to yield EVs from cell culture supernatant and human serum, and results have been validated using alternative methods of vesicle purification (eg, gradient centrifugation) [38–40].
Statistical Analysis
All data represent the average of 3 technical replicates unless otherwise stated, and error bars are provided to show the standard deviation. Experiments using health blood donors were independently performed with ≥3 donors. Postvaccination results were normalized using the prevaccination value as 100% for individual participants, and the percentage change on subsequent days for each individual experiment was calculated for each data point. Protein expression (activated Lck and PTPRE) were normalized to actin for each sample, and within participant’s protein expression levels after immunization were normalized to the prevaccine value, which was assigned as 100% expression. All experiments were conducted in triplicate. Postvaccination values were compared statistically using t tests for each time point (GraphPad software, version 9.4.1; GraphPad Prism Software). Differences were considered statistically significant at P < .05. All experiments that were statistically significant by parametric approaches were also significant by nonparametric statistical analyses.
RESULTS
Study Participants
Those recruited for the study included 14 individuals receiving vaccines to prevent disease during travel were recruited from the University of Iowa Travel Clinic, 5 who had received seasonal influenza vaccine, and nonvaccinated healthy blood donors. Demographic and vaccine information are summarized in Table 1. Participants who received LA-YF-Vax (Sanofi) were required to receive this vaccine based on their travel plans. Participants ranged in age from 24 to 71 years, and half of those receiving LA-YF-Vax were women. Three participants had received LA-YF-Vax previously, >10 years before the current immunization. Six received only LA-YF-Vax, while 8 received ≥1 additional vaccine (Table 1). Initially, 5 participants were enrolled in a pilot study to determine whether there was an effect on T-cell function and to examine the kinetics of this response; they had blood samples obtained before vaccination and 4, 7, and 10 days later. Based on the results of this pilot study, the study was expanded and the timing of blood sampling was changed to before and 7, 14, 21, and 28 days after vaccination.
Table 1.
Characteristics of Vaccine Recipients
| Participant | Sex | Race | Age, y | Prior YFV | Vaccines | RNA Titer/TCID50a |
|---|---|---|---|---|---|---|
| 1 | F | W | 60 | No | LA-YF-Vax | 3.72/72 |
| 2 | M | AA | 36 | Yes | LA-YF-Vax | ND/ND |
| 3 | F | W | 29 | No | LA-YF-Vax, Ty21a | 4.41/118 |
| 4 | M | AA | 32 | No | LA-YF-Vax, HAV, Ty21a | 4.04/88 |
| 5 | M | A/PI | 71 | Yes | LA-YF-Vax, ViCPS | ND/ND |
| 6 | F | W | 70 | No | LA-YF-Vax, HAV, ViCPS | 3.61/39 |
| 7 | F | A/PI | 30 | No | LA-YF-Vax, HAV, ViCPS | ND/ND |
| 8 | M | W | 20 | No | LA-YF-Vax, ViCPS, Mening, Rabies | 2.91/41 |
| 9 | M | AA | 24 | Yes | LA-YF-Vax, QIV | 1.88/ND |
| 10 | F | W | 52 | No | LA-YF-Vax, HAV-HBV, Mening, MMR, QIV | ND/ND |
| 11 | M | A/PI | 44 | No | LA-YF-Vax | 3.24/ND |
| 12 | F | W | 36 | No | LA-YF-Vax | ND/ND |
| 13 | F | W | 28 | No | LA-YF-Vax | ND/ND |
| 14 | M | W | 33 | No | LA-YF-Vax | 4.67/94 |
| 15 | F | W | 42 | No | QIV | ND/ND |
| 16 | F | AA | 53 | No | QIV | ND/ND |
| 17 | M | W | 29 | No | QIV | ND/ND |
| 18 | F | W | 61 | No | QIV | ND/ND |
| 19 | M | AA | 38 | No | QIV | ND/ND |
Abbreviations: AA, African American; A/PI, Asian-Pacific Islander; F, female; HAV, hepatitis A vaccine; HBV, hepatitis B vaccine; LA-YF-Vax, live attenuated yellow fever virus vaccine; M, male; Mening, meningococcal vaccine; MMR, measles, mumps, rubella vaccine; ND, not detected; QIV, quadrivalent influenza vaccine; TCID50, median tissue culture infective dose; Ty21a, oral typhoid vaccine; ViCPS, Vi capsular polysaccharide intramuscular typhoid vaccine; W, white; YFV, yellow fever vaccine.
aSerum RNA titer (log10 × genome equivalents/mL) and TCID50 (infectious titer).
YFV Viremia and Neutralizing Antibody Responses in Vaccine Recipients
It was previously shown that low-level YFV viremia is detected in some, but not all, LA-YF-Vax recipients during the first 7 days after vaccination [41]. We detected YFV genomic RNA in the day 4 and/or day 7 postvaccination serum samples obtained from 8 of the 14 vaccine recipients (range, 76–46 774 genome equivalents/mL). Five of these 8 participants had infectious virus detected, although the serum titers were very low (range, 39–118 infectious units/mL) (Table 1). Of note, 6 participants did not have detectable viremia by either real-time RT-PCR or infectivity assays. Two of these them had received LA-YF-Vax previously. Viral RNA but not infectious virus was detected in one participant who had been previously immunized. All vaccine recipient serum samples neutralized YFV by >90% by day 21 at a 1:100 dilution. The 2 individuals receiving their second LA-YF-Vax who did not have viremia detected (participants 2 and 5) had neutralizing antibodies present on day 7 after vaccination, while only 2 of the remaining 12 vaccine recipients had neutralizing antibodies detected on day 7. Viral RNA and viremia findings after LA-YF-Vax are summarized in Table 1.
T-Cell Function and PTPRE Regulation After LA-YF-Vax Vaccination
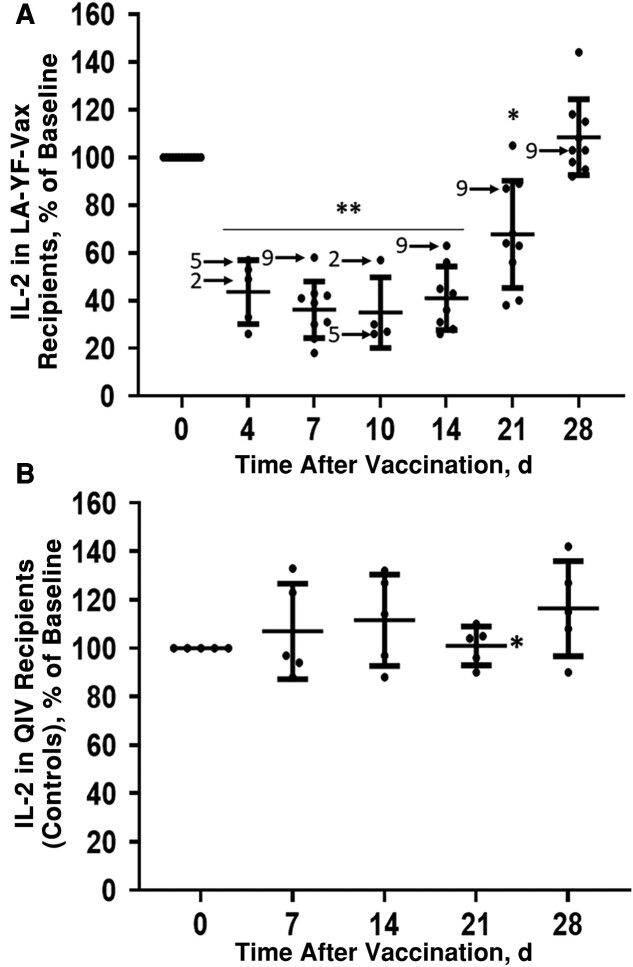
The effect of LA-YF-Vax on TCR signaling was assessed by measuring IL-2 release 16 hours after stimulation of PBMCs with anti-CD3. IL-2 release was normalized to the prevaccination level for each participant to allow comparison. Figure 1A demonstrates that IL-2 production was significantly lower than baseline on days 4–21 after vaccination but returned to baseline by day 28 after vaccination. IL-2 nadir levels occurred on days 7 and 10 after vaccination. In 2 of the 3 previously vaccinated individuals, the IL-2 levels returned to normal by day 21. Individuals who received QIV did not have a reduction in TCR-mediated IL-2 release during the first 4 weeks after vaccination (Figure 1B). These data are summarized in Table 1 and indicate that T-cell function stimulated through the TCR was transiently diminished by LA-YF-Vax.
Figure 1.
Live attenuated yellow fever virus vaccine (LA-YF-Vax) specifically reduces T-cell receptor (TCR)–mediated interleukin 2 (IL-2) release after vaccination. A, All 14 study participants receiving LA-YF-Vax had significantly less IL-2 released from their peripheral blood mononuclear cells 4–21 days after vaccination, compared with their prevaccination values. IL-2 was measured 16 hours after TCR stimulation. B, In contrast, none of the controls who received quadrivalent influenza vaccine (QIV) had significant changes in TCR-mediated IL-2 release. Arrows identify participants who received LA-YF-Vax ≥10 years before this immunization (participants 2, 5, and 9). *P < .05; **P < .01.
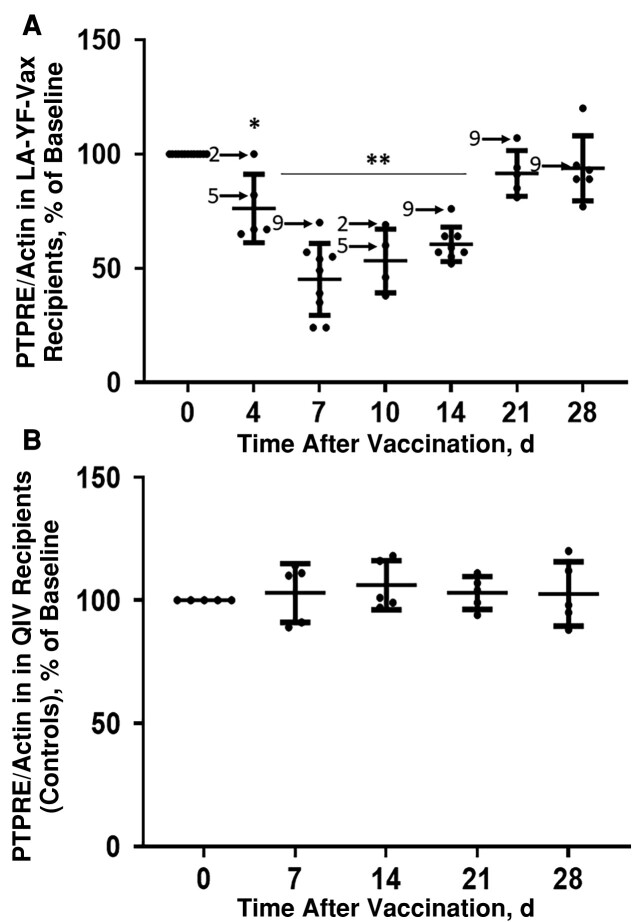
To determine whether the reduction in TCR-induced IL-2 release was associated with down-regulation of the Src kinase–activating phosphatase (PTPRE) that was previously shown to be regulated by YFV vsRNA during in vitro infection and in a murine animal model infection [26], PTPRE and actin were quantified in fresh PBMC lysates obtained before and after vaccination. Figure 2A demonstrates that PTPRE levels relative to PBMC actin levels were significantly reduced between days 4 and 14 compared with prevaccination levels for those who received LA-YF-Vax. This was specific, as none of the 5 QIV recipients had significant reductions in PTPRE after vaccination (Figure 2B).
Figure 2.
Live attenuated yellow fever virus vaccine (LA-YF-Vax) specifically reduces the Src kinase–activating protein tyrosine phosphatase receptor type E (PTPRE). A, PTPRE levels relative to actin within samples in circulating peripheral blood mononuclear cells were reduced in study participants receiving LA-YF-Vax 4–14 days after vaccination, compared with their prevaccination values. B, In contrast, none of the controls who received quadrivalent influenza vaccine (QIV) had significant changes in PTPRE levels relative to actin within samples. Arrows identify participants who received LA-YF-Vax ≥10 years before this immunization (participants 2, 5, and 9). *P < .05; **P < .01.
Of note, 8 of the 14 participants receiving LA-YF-Vax received additional vaccines, while 6 received only LA-YF-Vax (Table 1). All 14 participants demonstrated reduced TCR signaling and PTPRE expression Figures 1 and 2), including 2 who received QIV (participants 9 and 10). None of the 5 participants who received only quadrivalent, inactivated flu vaccine demonstrated reduced TCR signaling by IL-2 release or had reduced PTPRE expression, illustrating the specificity of this reaction to LA-YF-Vax.
Serum and Serum Particles Reduction of T-Cell Function and PTPRE Levels After LA-YF-Vax
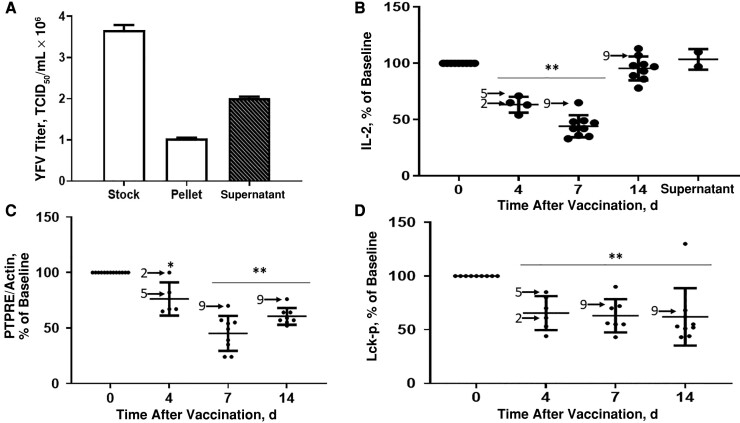
Since PTPRE protein levels were reduced in all 14 participants after LA-YF-Vax administration and IL-2 release after TCR stimulation was reduced between days 4 and 14 in all participants including those with little or no YFV viremia, we purified particles from participant’s serum samples using a commercial exosome purification method (ExoQuick) to determine whether serum particles could recapitulate the T-cell signaling effects. First, to determine whether ExoQuick precipitates infectious viral particles, infectious YFV in media was precipitated and residual infectivity of the precipitate and supernatant was measured. Figure 3A demonstrates that >28% of infectious YFV was precipitated by the ExoQuick reagent; thus, ExoQuick presumably precipitates infectious and defective, noninfectious YFV particles in addition to EVs present in serum.
Figure 3.
Serum extracellular vesicles (EVs) from live attenuated yellow fever virus (YFV) vaccine (LA-YF-Vax) recipients dampen T-cell receptor–mediated T-cell function and reduce protein tyrosine phosphatase receptor type E (PTPRE) expression. A, YFV titer in stock virus, the pellet, and supernatant from a commercial exosome purification method (ExoQuick). B–D, Incubation of healthy donor peripheral blood mononuclear cells with EVs in serum before and after vaccination led to reduction in interleukin 2 (IL-2) release (B), PTPRE expression relative to actin (C), and Lck activation (Lck-p) (D) after vaccination compared with prevaccination values. Arrows identify study participants who received LA-YF-Vax ≥10 years before this immunization (participants 2, 5, and 9). *P < .05; **P < .01. Abbreviation: TCID50, median tissue culture infective dose.
Because 6 participants did not have detectable viral RNA and 8 did not have infectious YFV identified in serum, we sought to examine whether infectious or defective viral particles or EVs elicited by LA-YF-Vax were responsible for PTPRE and TCR reduction. EVs were purified from 0.5 mL of serum from participants on days 4, 7, and 14 after vaccination and resuspended to 0.5 mL of phosphate-buffered saline; thus, the concentration of EVs pulled down by ExoQuick was equal to or less than that present in serum. Limited samples precluded testing of all samples and time points. These serum-derived EV preparations were incubated with PBMCs obtained from healthy, nonvaccinated blood donors and stimulated with anti-CD3, as described above. EVs obtained 4 or 7 days after vaccination reduced TCR-stimulated IL-2 release when added to healthy donor PBMCs for 24 hours before stimulation, compared with prevaccination values (Figure 3B), and levels returned to baseline by 14 days after vaccination. These serum EVs from vaccine recipients also reduced PBMC PTPRE levels normalized to cellular actin concentrations (Figure 3C) on days 4, 7, and 14 after vaccination compared with prevaccination levels, including the 6 participants who had neither YFV RNA nor infectious virus detected in blood samples with our real-time RT-PCR method. Unfortunately, sufficient serum did not remain from days 21 and 28 after vaccination to assess these later time points.
To examine early TCR signaling event, serum EVs were incubated with healthy donor PBMCs, and phosphorylation of Y394 of Lck was measured in PBMCs after 10 minutes of anti-CD3 treatment. Lck is an Src kinase that is phosphorylated at Y505 in the nonactive state. After TCR activation, Y505 is dephosphorylated, resulting in a conformational change of the kinase leading to autophosphorylation in trans by Lck at Y394 [23]. Serum EVs obtained 4, 7, and 14 days after LA-YF-Vax receipt inhibited the activation of Lck compared with serum obtained before vaccination (Figure 3D). Supplementary Figure 1 presents a representative immune blot showing that EVs prepared from serum after vaccination mediated a reduction in PTPRE and Lck activation compared with prevaccine levels. The effects of EVs on TCR-mediated IL-2 release, Lck activation, and PTPRE levels was specific, as EVs prepared from individuals who received only QIV did not change after immunization (Supplementary Figure 2). Thus, serum particles, including those from individuals with no detectable viral RNA or infectious virus detected, specifically reduced TCR signaling and cellular PTPRE levels in vitro.
DISCUSSION
Extensive evidence demonstrates that LAVs have important effects that are associated with beneficial clinical outcomes beyond prevention of the pathogen for which the vaccines were administered (reviewed in [1, 2, 42]). While typically described as “nonspecific” or “off-target,” presumably these beneficial effects are mediated by specific mechanisms induced by the LAVs that alter immune functions leading to improved disease severity [5, 6, 8] and/or survival [3, 4, 7, 9, 11]. Despite these benefits potentially saving an “extra million lives a year” [42], few studies have identified specific mechanisms by which LAVs modulate host immunity [19, 20]. It is important to point out that some natural viral infections leave hosts susceptible to serious secondary infections (ie, bacterial pneumonia after influenza infection), thus viral-induced immune modulation may lead to serious and even fatal health outcomes after natural infection. The extent and mechanisms of natural infection and attenuated immunization may differ. Characterizing these potential differences may offer insights into the beneficial or detrimental effect of virus-mediated immune modulation.
We and others identified mechanisms by which many DNA and RNA viruses modulate T-cell functions mediated by the TCR (reviewed in [43–45]). One mechanism shared by HCV and YFV involves a genome-derived short, noncoding RNA that targets an activating phosphatase involved in Src kinase signaling [24–26]. In the current study, we demonstrate that infection with LA-YF-Vax reproduces the in vitro findings. We observed a transient reduction in signaling after stimulation of the TCR, with reduced cytokine (IL-2) release comparing results before and after vaccination (Figure 1). Furthermore, the activating phosphatase PTPRE was reduced after LA-YF-Vax receipt (Figure 2). Although multiple vaccines were administered to some, all LA-YF-Vax recipients demonstrated a reduction in TCR function, including 6 who received only LA-YF-Vax, and none of the 5 who received only QIV demonstrated TCR signaling reduction.
As expected, neutralizing antibodies developed more quickly for 2 of the 3 participants who had previously received LA-YF-Vax and neither of them had detectable YFV RNA in serum after immunization (Table 1). There was a trend for reduced PTPRE levels and TCR inhibition in these participants (Figures 1 and 2); however, there are too few participants to allow conclusions to be drawn. The reduced levels of PTPREs and reduced inhibition of TCR in these participants may be due to preexisting immunity. We speculate that the memory including YFV-specific TCRs may respond more efficiently to YFV antigens, at least partially overcoming the inhibitory effects compared with YFV-naive T cells.
How does an attenuated vaccine with minimal systemic replication lead to reduced global TCR signaling and PTPRE expression? To address this question, we incubated healthy donor T cells with serum-derived particles (EVs) obtained before and after vaccination. Particles obtained at early time points after LA-YF-Vax administration were sufficient to inhibit TCR-mediated IL-2 release and Lck activation (Figure 3B and 3D) compared with prevaccination levels, even though several of the serum samples had no detectable YFV RNA or infectious virus. We found that the EV purification method precipitates infectious viral particles (Figure 3A), and presumably defective YFV particles are also precipitated. Because EVs are released from virus-infected cells and can carry viral RNAs, it is tempting to speculate that a combination of defective YFV particles and EVs containing viral proteins and/or genome-derived YFV vsRNAs are present at levels sufficient to cause the reductions in TCR signaling and PTPRE expression (Figure 3C). EVs from participants with detectable YFV RNA in serum (Table 1) had detectable YFV RNA using the real-time RT-PCR methods; however, those from participants without detectable YFV RNA were negative as expected (data not shown). Studies are underway to attempt to identify viral RNA or proteins within EVs after vaccination to better characterize the mechanisms of TCR inhibition.
Prior studies found that HIV-infected participants who are also infected with dengue virus have reduced HIV viral loads [46], and EVs are known regulators of HIV replication [40]. While dengue virus serotype 2 envelope protein does not inhibit TCR signaling [26], dengue virus NS5 protein does interfere with it [47]. Thus, different mechanisms used by viruses to interfere with TCR signaling may influence these clinical outcomes [46]. In the current study, our finding that EVs generated by LA-YF-Vax administration reduce TCR-mediated immune functions may contribute to the dengue and YFV NS5A protein-mediated reduction in T-cell CD4 expression and HIV inhibition observed in vitro [33, 47] and in vivo [46]. Furthermore, the mechanisms that have evolved in different viruses may well contribute to other beneficial outcomes for both the host and the virus, including facilitation of the establishment of infection and replication benefits that are yet to be ascertained.
Human T cells incubated with several viruses (YFV, HCV, or HPgV) though not all (not Zika, mumps, or dengue virus 2) have significantly reduced IL-2 release after TCR stimulation. However, IL-2 release is not completely inhibited after TCR stimulus, and thus TCR-mediated cell activation is not abolished [22–26]. Similarly, BCG vaccination leads to reduced interferon γ responsiveness to heterologous stimulation, and BCG-induced changes in cytokine responses to heterologous stimulation vary by pathogen [16]. The reduction in response as opposed to abrogation of the response may explain why most infections with these viral pathogens do not result in severe immune suppression. Nevertheless, immune activation is an important mediator of viral pathogenesis, and the dampening of acute inflammatory responses is likely beneficial. Infection with HPgV, for example, reduces mortality rates in HIV- and Ebola-infected people [48–50]. We posit that the immune modulation identified for LA-YF-Vax may contribute to the beneficial outcomes in studies of LA-YF-Vax recipients [7] and other LAV recipients [1, 2, 42]. These data support the need to identify additional novel mechanisms of immune modulation by LAVs and viral pathogens, which will provide new insights into beneficial outcomes of immune modulation.
Supplementary Data
Supplementary materials are available at The Journal of Infectious Diseases online. Consisting of data provided by the authors to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the authors, so questions or comments should be addressed to the corresponding author.
Supplementary Material
Contributor Information
J Xiang, Iowa City Department of Veterans Affairs Healthcare System, University of Iowa, Iowa City, Iowa, USA.
Q Chang, Iowa City Department of Veterans Affairs Healthcare System, University of Iowa, Iowa City, Iowa, USA.
J H McLinden, Iowa City Department of Veterans Affairs Healthcare System, University of Iowa, Iowa City, Iowa, USA.
N Bhattarai, Division of Cellular and Gene Therapies, Office of Tissues and Advanced Therapies, Center for Biologics Evaluation and Research, Food and Drug Administration, Silver Spring, Maryland, USA.
J L Welch, Iowa City Department of Veterans Affairs Healthcare System, University of Iowa, Iowa City, Iowa, USA.
T M Kaufman, Iowa City Department of Veterans Affairs Healthcare System, University of Iowa, Iowa City, Iowa, USA.
Jack T Stapleton, Iowa City Department of Veterans Affairs Healthcare System, University of Iowa, Iowa City, Iowa, USA.
Notes
Acknowledgments. We thank Professor Judy Streit and the University of Iowa Travel Clinic for facilitating recruitment of study participants receiving yellow fever virus vaccine, Professor Jon Houtman for helpful discussions related to T-cell receptor signaling, and Professor Patricia Winokur for providing research space in the University of Iowa Vaccine Treatment and Evaluation Unit for visits. In addition, we thank A. J. Carr for assistance with phlebotomy.
Financial support. This work was supported by the U.S. Department of Veterans Affairs (Merit Review grant BX000207 to J. T. S.), the University of Iowa Carver College of Medicine (pilot grant to J. T. S. and Professor Jon Houtman), and the National Institute of Allergy and Infectious Diseases, National Institutes of Health (training grant 5T32AI343 to J. T. S.; also supporting J. L. W.).
References
- 1. Jensen KJ, Benn CS, van Crevel R. Unravelling the nature of non-specific effects of vaccines-a challenge for innate immunologists. Semin Immunol 2016; 28:377–83. [DOI] [PubMed] [Google Scholar]
- 2. Benn CS, Netea MG, Selin LK, Aaby P. A small jab—a big effect: nonspecific immunomodulation by vaccines. Trends Immunol 2013; 34:431–9. [DOI] [PubMed] [Google Scholar]
- 3. Biering-Sørensen S, Aaby P, Napirna BM, et al. Small randomized trial among low-birth-weight children receiving bacillus Calmette–Guerin vaccination at first health center contact. Pediatr Infect Dis J 2012; 31:306–8. [DOI] [PubMed] [Google Scholar]
- 4. Benn CS, Jacobsen LH, Fisker AB, et al. Campaigns with oral polio vaccine may lower mortality and create unexpected results. Vaccine 2017; 35:1113–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Sorup S, Benn CS, Poulsen A, Krause TG, Aaby P, Ravn H. Simultaneous vaccination with MMR and DtaP-IPV-Hib and rate of hospital admissions with any infections: a nationwide register based cohort study. Vaccine 2016; 34:6172–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Sorup S, Benn CS, Poulsen A, Krause TG, Aaby P, Ravn H. Live vaccine against measles, mumps, and rubella and the risk of hospital admissions for nontargeted infections. JAMA 2014; 311:826–35. [DOI] [PubMed] [Google Scholar]
- 7. Fisker AB, Ravn H, Rodrigues A, et al. Co-administration of live measles and yellow fever vaccines and inactivated pentavalent vaccines is associated with increased mortality compared with measles and yellow fever vaccines only: an observational study from Guinea-Bissau. Vaccine 2014; 32:598–605. [DOI] [PubMed] [Google Scholar]
- 8. Sorup S, Villumsen M, Ravn H, et al. Smallpox vaccination and all-cause infectious disease hospitalization: a Danish register-based cohort study. Int J Epidemiol 2011; 40:955–63. [DOI] [PubMed] [Google Scholar]
- 9. Aaby P, Martins CL, Garly ML, et al. Non-specific effects of standard measles vaccine at 4.5 and 9 months of age on childhood mortality: randomised controlled trial. BMJ 2010; 341:6495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Jensen ML, Dave S, Schim van der Loeff M, et al. Vaccinia scars associated with improved survival among adults in rural Guinea-Bissau. PloS One 2006; 1:e101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Aaby P, Samb B, Simondon F, Seck AM, Knudsen K, Whittle H. Non-specific beneficial effect of measles immunisation: analysis of mortality studies from developing countries. BMJ 1995; 311:481–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Vaugelade J, Pinchinat S, Guiella G, Elguero E, Simondon F. Non-specific effects of vaccination on child survival: prospective cohort study in Burkina Faso. BMJ 2004; 329:1309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Aaby P, Ravn H, Benn CS, et al. Randomized trials comparing inactivated vaccine after medium- or high-titer measles vaccine with standard titer measles vaccine after inactivated vaccine: a meta-analysis. Pediatr Infect Dis J 2016; 35:1232–41. [DOI] [PubMed] [Google Scholar]
- 14. Aaby P, Martins CL, Garly ML, Rodrigues A, Benn CS, Whittle H. The optimal age of measles mmunization in low-income countries: a secondary analysis of the assumptions underlying the current policy. BMJ Open 2012; 2:e000761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Prentice S, Dockrell HM. BCG specific and nonspecific effects: different questions, similar challenges. J Infect Dis 2021; 224:1105–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Messina NL, Pittet LF, Gardiner K, et al. Neonatal bacille Calmette-Guerin vaccination and infections in the first year of life: the MIS BAIR randomized controlled trial. J Infect Dis 2021; 224:1115–27. [DOI] [PubMed] [Google Scholar]
- 17. Aaby P, Roth A, Ravn H, et al. Randomized trial of BCG vaccination at birth to low-birth-weight children: beneficial nonspecific effects in the neonatal period? J Infect Dis 2011; 204:245–52. [DOI] [PubMed] [Google Scholar]
- 18. Husby A, Hansen JV, Fosbol E, et al. SARS-CoV-2 vaccination and myocarditis or myopericarditis: population based cohort study. BMJ 2021; 375:e068665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Kleinnijenhuis J, Quintin J, Preijers F, et al. Bacille Calmette–Guerin induces NOD2-dependent nonspecific protection from reinfection via epigenetic reprogramming of monocytes. Proc Natl Acad Sci USA 2012; 109:17537–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Welsh RM, Selin LK. No one is I: the significance of heterologous T-cell immunity. Nat Rev Immunol 2002; 2:417–26. [DOI] [PubMed] [Google Scholar]
- 21. Netea MG, Quintin J, van der Meer JW. Trained immunity: a memory for innate host defense. Cell Host Microbe 2011; 9:355–61. [DOI] [PubMed] [Google Scholar]
- 22. Bhattarai N, McLinden JH, Xiang J, Kaufman TM, Stapleton JT. GB virus C envelope protein E2 inhibits TCR-induced IL-2 production and alters IL-2-signaling pathways. J Immunol 2012; 189:2211–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Bhattarai N, McLinden JH, Xiang J, Landay A, Chivero ET, Stapleton JT. GB virus C particles inhibit T cell activation via envelope E2 protein-mediated inhibition of T cell receptor signaling. J Immunol 2013; 190:6351–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Bhattarai N, McLinden JH, Xiang J, Kaufman TM, Stapleton JT. Conserved motifs within hepatitis C virus envelope (E2) RNA and protein independently inhibit T cell activation. PloS Pathog 2015; 11:e1005183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Bhattarai N, McLinden JH, Xiang J, et al. Hepatitis C virus infection inhibits a Src-kinase regulatory phosphatase and reduces T cell activation in vivo. PloS Pathog 2017; 13:e1006232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. McLinden JH, Bhattarai N, Stapleton JT, et al. Yellow fever virus, but not Zika virus or dengue virus, inhibits T-cell receptor-mediated T-cell function by an RNA-based mechanism. J Infect Dis 2017; 216:1164–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Wiedmann M, Liebert UG, Oesen U, et al. Decreased immunogenicity of recombinant hepatitis B vaccine in chronic hepatitis C. Hepatology 2000; 31:230–4. [DOI] [PubMed] [Google Scholar]
- 28. Leroy V, Bourliere M, Durand M, et al. The antibody response to hepatitis B virus vaccination is negatively influenced by the hepatitis C virus viral load in patients with chronic hepatitis C: a case-control study. Eur J Gastroenterol Hepatol 2002; 14:485–9. [DOI] [PubMed] [Google Scholar]
- 29. Silva JRN, Camacho LA, Siqueira MM, et al. Mutual interference on the immune response to yellow fever vaccine and a combined vaccine against measles, mumps and rubella. Vaccine 2011; 29:6327–34. [DOI] [PubMed] [Google Scholar]
- 30. Nasveld PE, Marjason J, Bennett S, et al. Concomitant or sequential administration of live attenuated Japanese encephalitis chimeric virus vaccine and yellow fever 17D vaccine: randomized double-blind phase II evaluation of safety and immunogenicity. Hum Vaccin 2010; 6:906–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Moorman JP, Zhang CL, Ni L, et al. Impaired hepatitis B vaccine responses during chronic hepatitis C infection: involvement of the PD-1 pathway in regulating CD4+ T cell responses. Vaccine 2011; 29:3169–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Stapleton JT, Chaloner K, Martenson JA, et al. GB virus C infection is associated with altered lymphocyte subset distribution and reduced T cell activation and proliferation in HIV-infected individuals. PloS One 2012; 7:e50563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Xiang J, McLinden JH, Rydze RA, et al. Viruses within the Flaviviridae decrease CD4 expression and inhibit HIV replication in human CD4+ cells. J Immunol 2009; 183:7860–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Chivero ET, Bhattarai N, Rydze RT, Winters MA, Holodniy M, Stapleton JT. Human pegivirus RNA is found in multiple blood mononuclear cells in vivo and serum-derived viral RNA-containing particles are infectious in vitro. J Gen Virol 2014; 95:1307–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Taketa-Graham M, Powell Pereira JL, Baylis E, et al. High throughput quantitative colorimetric microneutralization assay for the confirmation and differentiation of west Nile virus and St. Louis encephalitis virus. Am J Trop Med Hyg 2010; 82:501–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Welch JL, Kaddour H, Winchester L, Fletcher CV, Stapleton JT, Okeoma CM. Semen extracellular vesicles from HIV-1-infected individuals inhibit HIV-1 replication in vitro, and extracellular vesicles carry antiretroviral drugs in vivo. J Acquir Immune Defic Syndr 2020; 83:90–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Chivero ET, Bhattarai N, McLinden JH, Xiang J, Stapleton JT. Human pegivirus (HPgV; formerly known as GBV-C) inhibits IL-12 dependent natural killer cell function. Virology 2015; 485:116–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Fabbri M, Paone A, Calore F, et al. MicroRNAs bind to Toll-like receptors to induce prometastatic inflammatory response. Proc Natl Acad Sci U S A 2012; 109:E2110-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Zhuang G, Wu X, Jiang Z, et al. Tumour-secreted miR-9 promotes endothelial cell migration and angiogenesis by activating the JAK-STAT pathway. EMBO J 2012; 31:3513–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Welch JL, Stapleton JT, Okeoma CM. Vehicles of intercellular communication: exosomes and HIV-1. J Gen Virol 2019; 100:350–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Wheelock EF, Sibley WA. Circulating virus, interferon and antibody after vaccination with the 17-d strain of yellow-fever virus. N Engl J Med 1965; 273:194–8. [DOI] [PubMed] [Google Scholar]
- 42. Shann F. A live-vaccine-last schedule: saving an extra million lives a year? Clin Infect Dis 2021; 72:1437–9. [DOI] [PubMed] [Google Scholar]
- 43. Jerome KR. Viral modulation of T-cell receptor signaling. J Virol 2008; 82:4194–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Bhattarai N, Stapleton JT. GB virus C: the good boy virus? Trends Microbiol 2012; 20:124–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Stapleton JT. Human pegivirus type 1: a common human virus that is beneficial in immune-mediated disease? Front Immunol 2022; 13:887760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Watt G, Kantipong P, Jongsakul K. Decrease in human immunodeficiency virus type 1 load during acute dengue fever. Clin Infect Dis 2003; 36:1067–9. [DOI] [PubMed] [Google Scholar]
- 47. McLinden JH, Stapleton JT, Chang Q, Xiang J. Expression of the dengue virus type 2 NS5 protein in a CD4+ T cell line inhibits HIV replication. J Infect Dis 2008; 198:860–3. [DOI] [PubMed] [Google Scholar]
- 48. Xiang J, Wunschmann S, Diekema DJ, et al. Effect of coinfection with GB virus C (hepatitis G virus) on survival among patients with HIV infection. N Engl J Med 2001; 345:707–14. [DOI] [PubMed] [Google Scholar]
- 49. Williams CF, Klinzman D, Yamashita TE, et al. Persistent GB virus C infection and survival in HIV-infected men. N Engl J Med 2004; 350:981–90. [DOI] [PubMed] [Google Scholar]
- 50. Lauck M, Bailey AL, Andersen KG, Goldberg TL, Sabeti PC, O’Connor DH. GB virus C coinfections in West African Ebola patients. J Virol 2015; 89:2425–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.