Abstract
Background:
The treatment of end-stage chronic renal failure involves substantial costs for health care, which could be higher considering hemodialysis access complications management costs. Complications could be addressed by cannulation technique, but also by the needle. In particular, the use of a metal needle for cannulation is responsible for several complications that compromise dialysis delivery and require interventions. This analysis aims to evaluate, from a hospital perspective, the direct costs related to complications that may occur in hemodialysis patients in Italy.
Methods:
To identify the main complications to consider in our analysis and related patient pathway for their management, we conducted an international literature search on PubMed and validated the data for Italy with an Italian Key Opinion Leader (KOL). A micro-costing analysis was developed to precisely assess the economic costs of healthcare interventions to manage complications due to vascular access cannulation.
Results:
The major complications identified, and the average cost/per episode for their management are the following ones: Local infection € 1455 (min € 745 and max € 2160); Sepsis, that requires hospitalization in ward € 4401 (min € 3693 and max € 4623); Sepsis (ICU) € 17,190; Hematoma € 304 (min € 252 and max € 728); Aneurysm € 3632 (min € 3139 and max € 4014); Pseudoaneurysm € 3695 (min € 3615 and max € 4014); Stenosis € 2229 (min € 1874 and max € 4857); Thrombosis € 2151 (min € 1941 and max € 3395).
Conclusions:
In making decisions, hospital administration, and payer should take into consideration, not just the price of devices, but the entire patient’s pathway. The use of plastic cannulae in hemodialysis patients shows improved outcomes compared to traditional metal needles. Furthermore, combining it with accurate and efficient cannulation techniques reduces complication rates, improves patients’ quality of life, and reduces healthcare resource consumption.
Keywords: Dialysis access, dialysis, AV fistula, economic and health services, vascular access, economic analysis, complications costs, plastic needle, cannulation, micro-costing
Introduction
Hemodialysis (HD) is a complex attritional disease state that afflicts almost 40,000 people with end-stage renal disease (ESRD) 1 in Italy, and leads to a 45% annual increase in excess mortality. 2 The treatment of end-stage chronic renal failure involves substantial costs for health care. If we consider an average cost for each session of € 281 for three weekly sessions, the annual healthcare cost of an HD patient in Italy is € 43,800, without considering complications that may occur. 3
Perhaps uniquely, the outcome and morbidity of HD are inherently related and may be altered through the method of providing HD, whether it is through a central venous catheter (CVC), an arteriovenous fistula (AVF), or an arteriovenous graft (AVG). 2 It is widely accepted that the optimal method of providing vascular access is through a native AVF due to the best long-term patency, low complication and reintervention rates, and better long-term patient survival than alternative methods such as a CVC. 4 It has been reported by Vachharajani et al. 5 that in 2017, globally, 80% of patients were using a CVC at HD initiation, whereas the use of AVF at HD initiation rose from 12% to 17% from 2005 to 2017. In Italy the prevalence of patients using different vascular access has been reported by Pisoni et al. 6 in particular, AVF was 68%, AVG was 8%, and CVC was 25%.
CVCs remain important in access provision, and after the failure of vascular access, they remain the first choice. 7 The problem of prevalent patients on a CVC remains complex due to hostile anatomy, central vein stenosis, a reluctance to change established access modality, and lower autologous access success rates in patients already dialyzing through a CVC. AVGs provide an alternative to AVF or CVC, with long-term survival outcomes that approach those of an AVF, along with better primary patency. 2 AVG is the second viable option in the construction of vascular access but has a higher rate of complications caused by intimal hyperplasia.8,9
The most common method of cannulation among maintenance HD patients is the rope ladder technique, but it is actively discouraged due to progressive weakness in the vessel wall and subsequent aneurysm formation. Buttonhole cannulation is more often used in home HD, even if it involves a constant site of needling that, over time, leads to an epithelized track into which a blunt needle is inserted. 10
Unfortunately, cannulation is a problem-ridden procedure for multiple reasons including nonstandard geometries of AVFs, lack of training opportunities for patient care technicians (PCTs), and a high turnover rate among PCTs in dialysis clinics. Lack of cannulation skill results in poor clinical outcomes due to infiltration and other cannulation-related trauma that could potentially lead to unusable vascular access. 11
Hemodialysis access complications are common. The cannulation technique influences AVF and AVG survival. 12 Needle-related cannulation injuries and complications arise from various causes and can manifest in a variety of outcomes. 13 Needle injury (mechanical or hemodynamic) may cause complications that compromise dialysis delivery, requiring interventions, radiological or surgical procedures, to maintain or restore patency. 14 The trauma associated with HD needle insertions is complex. The mechanical trauma related directly to the needle placement includes the biological injury to the wall of the vessel. In addition, blood extravasation due to accidental piercing through the vessel wall or trauma at the time of needle removal may generate hematomas that distort the geometry of the access which, when severe, may cause flow obstruction with increased risk of thrombosis and access loss. 14 Routine needle access to AVGs and AVFs for purposes of hemodialysis can result in material or tissue traumatic degradation over time even when proper cannulation technique and guidelines are practiced. Most commonly, pseudoaneurysm or aneurysmal development is the resulting outcome even with routine cannulation due to disruption of hemodialysis access (HAD) conduit integrity, resulting in extravascular blood flow, or a chronic outward remodeling process occurs. This process is accelerated and can lead to more complex or dangerous complications when area cannulation is confined to focal regions of the access. In contrast to degenerative complications and outward remodeling, needle-related injury (NRI) can also result in focal intraconduit stenosis, and these irregular areas of stenosis can lead to an increased potential for thrombosis. Furthermore, repetitive localized puncture, pseudoaneurysm formation, localized puncture site infection from skin contamination, or infection of a peri-access hematoma (from acute NRI, or improper use of an early access graft) can all be traced back to needle cannulation. 13
Accordingly, it was demonstrated that potential needle-related complications accounted for 6% of all complication cases. In particular, the use of a metal needle is responsible for several complications, high resource consumption, and a reduction in patients’ quality of life. 13
At the moment, in Italy, there is no economic data available on needle-related complication management costs.
Aim
This analysis aims to evaluate, from a hospital perspective, the unitary direct costs related to complications that may occur in hemodialysis patients in Italy. Additional economic analysis is also developed, reporting the potential cost savings in moving from sharp metal to plastic cannulae as a strategy for reducing access-related complications.
Methods
Perspective
In accordance with our major goal, which was to identify the complication management costs in hemodialysis patients, the hospital’s perspective was adopted. Unitary direct costs per adverse event (AE) were computed, considering the cost per unit of the items included in the complication management.
Clinical input
To identify the main complications to consider in the analysis and corresponding patient’s pathway for their management, an international literature search on PubMed was conducted, and the data were validated at the National level with an Italian KOL, Professor Tozzi, based on his clinical experience developed at ASST-Sette Laghi Varese.
The rate of complications that occur in HD patients was assessed initially from the literature, and then adapted and estimated reflecting Professor Tozzi’s experience. In fact, it can be considered an underestimation due to the KOL’s extensive clinical practice, it counts about 600 procedures/year (surgical and endovascular procedures) related to vascular access for HD patients. Those percentages are presented as the incident of the AEs applied to the total HD patients.
The data validation and adaptation were performed by interviews with the KOL, to calibrate and assess the incidence of the annual complications considered, to define the patient pathway to treat every AE, and consequently the in-hospital resources consumption (pre-post and intraoperative resources). Without stratifying patients per specific characteristics or per risk factors, the patient pathway was described as an estimation of the usual management applied by the KOL for those AEs, including the following items: setting of care (hospitalization in ICU, Ward, or outpatient setting, and Theater room), length of stay, diagnostic analysis, procedural materials, and staff (physicians, anesthesiologists, surgeons, nurses) for surgical procedures, and drugs (intra-procedural and post-procedural).
The annual complications rate was then used to assess a scenario analysis to evaluate the economic burden for National Healthcare System (NHS) in the short term.
Economic input
A micro-costing analysis was performed in Microsoft Excel and was developed with the support of professional health economists to precisely assess the economic costs of healthcare interventions to manage every complication. The process has been developed in three stages: the identification of all resources involved in the provision of care (human-resources/theater-time/consumables); accurate measurement of each resource (using time-and-motion studies), and valuation of the resources used.
Cost data were collected based on patient pathway identified by ASST-Sette Laghi clinical practice for every complication considered. We adopted a micro-costing procedure to identify cost-generating components and to attribute appropriate unit costs for the direct healthcare resources.
In line with the hospital perspective of the analysis, the direct costs for the single AE were estimated. Direct healthcare costs included hospitalization, clinical consultations, diagnostic tests and procedures, medical therapies, and surgical procedures (theater room costs, materials, physicians, and nurses).
The unit costs data, when available, came from the ASST-Sette Laghi administration internal database. Personnel costs for surgeons, anesthetists, and nurses were calculated as costs per minute. For estimating hospital personnel and overhead costs, an internal Medtronic database was used.
A weighted average of complication management costs was applied to define each complication’s total cost.
An additional scenario analysis was performed. Based on the results presented by Gage and Reichert, 13 the average of 6% in overall complication rates needle-related was assumed to be reduced by shifting 100 and 1000 HD patients from sharp metal to plastic cannulae.
The clinical reduction in complication rates reflects later the reduction in the resources consumption, with the intent to simulate a potential strategy to reduce health care costs, improving patient’s quality of life.
Results
In accordance with literature research,12–16 and as confirmed by Italian clinical practice and experience, the major complications identified, that occur in HD patients, can be associated to metal needles and include: infections, that can be local, sepsis that requires hospitalization in Ward, or severe sepsis that require hospitalization in ICU, hematoma, aneurysm and pseudoaneurysm, stenosis and thrombosis.
Complications relative risks and treatments are the following:
Infections
Local infections are related in 5% to arteriovenous grafts/year (AVGy) and in 1.5% to native access: they are treated in an outpatient setting (dialysis room). 60% of them required a CVC implant and 30% of them required AVG/AVF surgical revision.
Sepsis occurs in 2.5% of AVGy: required hospitalization with a length of stay of 5 days; in 100% of cases CVC implant and 50% of cases surgical revision are needed.
Severe sepsis occurs at 0.5% of AVGy: required hospitalization in ICU for 15 days; in 100% of cases, CVC implant and surgical revision are needed.
Hematoma
Hematoma occurs in 2% of AVF and 7% of AVG cases, and it is treated in an outpatient setting (dialysis room) and 11% of cases require a CVC implant.
Aneurysm/pseudoaneurysm
Aneurysm occurs in 5% of AVF: it requires 5 days of hospitalization. In 30% of cases CVC is needed, and in 40% of cases (symptomatic or >3.5 cm) surgical treatment is performed.
Pseudoaneurysm occurs in 1.2% of native and prosthetic fistula: it requires 5 days of hospitalization. In 100% of cases surgical treatment is performed, of that 80% are CVC implants.
Stenosis
Stenosis occurs in 20% of the cases and requires a surgical procedure: 80% PTA endovascular intervention; 7% stent graft endovascular intervention; 13% AVF surgical revision.
Thrombosis
Thrombosis occurs in 15% of AVG and AVF cases, and requires 2 days of hospitalization.
Endovascular intervention is needed in 10% of the cases. Other surgical procedures are performed in 90% of the cases: 11% temporary CVC is implanted (90% of the time with locoregional anesthesia), 2%–5% AVF surgical revision.
The main recurrent surgical procedures are: CVC implant, AVF surgical revision, and surgical treatment. They were considered in addition to the length of stay, laboratory exams, and the pharmaceutical therapy treatment (like antibiotic therapy).
A CVC implant requires 30 min of intervention in the angiography room, where a nephrologist and a nurse would perform the implant of a CVC, for a total cost of € 476. Based on the type of CVC implanted (permanent or temporary), there could be a consistent variation in presented costs.
The cost for AVF surgical revision per se is € 1414. This operation requires 60 min in the Theater room, where a surgeon and a nurse, supported by an anesthesiologist, would implant the prosthesis using biological material. The prosthesis and the biological material are the main items responsible for the high resource consumption. Note that anesthesiology drugs are not included due to a lack of information.
Surgical treatment requires the same amount of time, staff, and materials as AVF surgical revision (without including the prosthesis), with an impact on total costs of € 875.
According to the results of the analysis, the average cost/per episode is the following (Figure 1):
Figure 1.
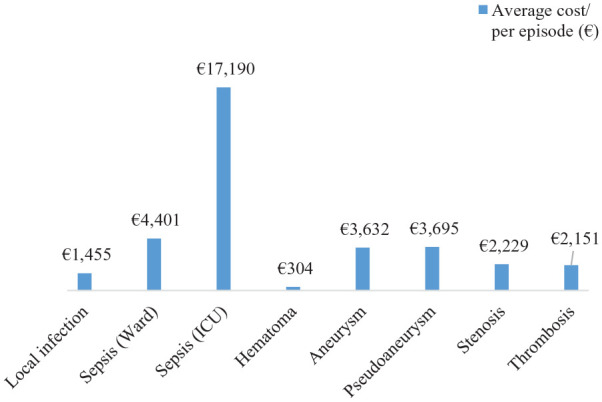
Average cost/per episode.
Local infection € 1455 (min € 745 and max € 2160); sepsis, that requires hospitalization in Ward € 4401 (min € 3693 and max € 4623); sepsis (ICU) € 17,190; Hematoma € 304 (min € 252 and max € 728); Aneurysm € 3632 (min € 3139 and max € 4014); Pseudoaneurysm € 3695 (min € 3615 and max € 4014); Stenosis € 2229 (min € 1874 and max € 4857); Thrombosis € 2151 (min € 1941 and max € 3395).
In Table 1 percentage of every item (setting, staff, materials, analysis, therapy) that occur at every average complication cost per episode was collected.
Table 1.
Items impact costs distribution.
| Average cost/per episode (€) | Setting (%) | Staff (%) | Materials (%) | Analysis (%) | Therapy (%) | |
|---|---|---|---|---|---|---|
| Local infection | 1455 | 10 | 20 | 34 | 8 | 28 |
| Sepsis (Ward) | 4401 | 51 | 7 | 19 | 5 | 19 |
| Sepsis (ICU) | 17,190 | 82 | 1 | 8 | 4 | 5 |
| Hematoma | 304 | 4 | 31 | 11 | 30 | 24 |
| Aneurysm | 3632 | 72 | 14 | 7 | 6 | 1 |
| Pseudoaneurysm | 3695 | 72 | 13 | 9 | 6 | 1 |
| Stenosis | 2229 | 16 | 9 | 61 | 13 | 1 |
| Thrombosis | 2151 | 58 | 10 | 25 | 6 | 2 |
Setting: setting of care (operating room, Ward, ICU); Staff: physician, nurse, anesthesiologist; Materials: devices, procedural kit; Analysis: pre-operational test; Therapy: pharmacological therapy and drugs administered during and/or post-procedures.
For local infection materials used for AVF surgical revision play the main role in resource consumption (38% of total cost), followed by antibiotics therapy.
For sepsis in Ward, the setting is the main player in resource consumption (51% of total cost).
For sepsis in ICU, 82% of the resource is absorbed by the setting.
For hematoma, nephrologist time and labor analysis are the main items to use resources, respectively 31% and 30% of total costs.
For aneurysms and pseudoaneurysms, the main costs are for the setting, due to hospitalizations for at least 5 days/episode (72% of total costs).
For stenosis, the main costs are for the materials used in the operating room setting (61% of the total costs).
For thrombosis, the main costs are for the setting, due to hospitalizations for at least 2 days/episode (58% of total costs).
Considering the Italian HD patients population, the incidence of the complications (without distinguishing between the type of HAD), and the costs identified in this analysis, the total amount spent every year by the hospitals would be approximately € 51 million (mln) (Table 2).
Table 2.
Total complications episodes and total impact costs/year.
| Average cost/per episode (€) | Incidence (%) | n° Episodes | Total costs/total episodes (€) | |
|---|---|---|---|---|
| Local infection | 1455 | 5 | 2000 | 2,910,042 |
| Sepsis (Ward) | 4401 | 3 | 1000 | 4,400,935 |
| Sepsis (ICU) | 17,190 | 1 | 200 | 3,438,056 |
| Hematoma | 304 | 2 | 800 | 243,251 |
| Aneurysm | 3632 | 5 | 2000 | 7,263,206 |
| Pseudoaneurysm | 3695 | 1 | 480 | 1,773,401 |
| Stenosis | 2229 | 20 | 8000 | 17,834,789 |
| Thrombosis | 2151 | 15 | 6000 | 12,905,590 |
| Total | 20,480 | 50,769,270 |
It could be underestimated considering that some patients could experience more than one complication during the duration of their hemodialysis care.
The same consideration has been performed for a hospital population of 100 and 1000 HD patients. The total estimated complication costs are respectively around € 127,000 and € 1270 mln (Tables 3 and 4).
Table 3.
Total complications episodes and total impact costs (100 patients).
| Average cost/per episode (€) | Incidence (%) | n° Episodes | Total costs/total episodes (€) | |
|---|---|---|---|---|
| Local infection | 1455 | 5 | 5 | 7275 |
| Sepsis (Ward) | 4401 | 3 | 3 | 11,002 |
| Sepsis (ICU) | 17,190 | 1 | 1 | 8595 |
| Hematoma | 304 | 2 | 2 | 608 |
| Aneurysm | 3632 | 5 | 5 | 18,158 |
| Pseudoaneurysm | 3695 | 1 | 1 | 4434 |
| Stenosis | 2229 | 20 | 20 | 44,587 |
| Thrombosis | 2151 | 15 | 15 | 32,264 |
| Total | 51 | 126,923 |
Table 4.
Total complications episodes and total impact costs (1000 patients).
| Average cost/per episode (€) | Incidence (%) | n° Episodes | Total costs/total episodes (€) | |
|---|---|---|---|---|
| Local infection | 1455 | 5 | 50 | 72,751 |
| Sepsis (Ward) | 4401 | 3 | 25 | 110,023 |
| Sepsis (ICU) | 17,190 | 1 | 5 | 85,951 |
| Hematoma | 304 | 2 | 20 | 6081 |
| Aneurysm | 3632 | 5 | 50 | 181,580 |
| Pseudoaneurysm | 3695 | 1 | 12 | 44,335 |
| Stenosis | 2229 | 20 | 200 | 445,870 |
| Thrombosis | 2151 | 15 | 150 | 322,640 |
| Total | 512 | 1,269,232 |
A clinical study conducted by Marticorena et al. 14 demonstrated that procedures to treat complications along cannulation segments increased from 0.41 to 1.29 per patient when using metal needles and decreased from 1.25 to 0.69 per patient when using plastic needles. The relative risks of having an intervention and having an infiltration during HD were higher for metal needles.
If we assume an HD hospital population of 100 and 1000 patients and apply a total complications reduction of 6% on the annual complication incidence, as Gage and Reichert 13 analysis reported to be needle-related, the total savings for the hospital would be respectively around € 7600 and € 76,000.
Thus, taken all together, the use of plastic needles for cannula fistula would not only be safer, but more cost-effective.
The average cost for a plastic needle for a cannulae fistula is approximately € 2.5, multiplying it by the number of weekly sessions (#3), per number of cannulae fistula for a session (#2), for months/year, the annual cost per patient/year would be € 288. It is clear that defining a target population that could benefit the most from plastic needles for cannulae fistula, due to the higher risk of complications, would be the best option to match clinical benefits and economic sustainability. Based on clinical practice, patients who have just started hemodialysis, patients with vascular access prosthesis, and patients with complex vascular access (too deep or too short) are the best candidates to receive selective treatment. Assuming they represent from 15% to 30% of the total HD patients, a reduction of at least 6% of complication rate, 13 in the same populations, could make a consistent difference in patients’ quality of life and related management costs.
Discussion
The results confirmed that management of adverse events in HD patients in Italy has a considerable impact on resource consumption.
Clinical evidence gathered over the last two decades has shifted the dogma from “fistula first” to the “right access” and has given equal importance to alternative types of access. 5 In particular, the National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative (KDOQI) 2019 vascular access guideline suggested a patient-centered approach to access selection. 4 The emphasis is on implementing a patient-centered approach that offers improved patient satisfaction and is aligned with individual goals of care. 5
In line with this innovative approach, more relevance and importance should be attributed to patient-reported outcomes measure (PROMs) including this parameter in every clinical study to have a full picture of all the parameters that contribute to improve the quality of care for patients’ perspective.
Furthermore, after the publication and validation of a new measure for vascular access-specific quality of life (VASQoL), which demonstrated to have good internal consistency, test-retest reliability, convergent validity, and responsiveness to change for clinically relevant vascular access outcomes. 17 Those characteristics made the VASQoL the perfect fit for future trials of vascular access, evaluation of new technologies, and routine use as a PROM. 17
As a matter of fact, the vascular outcomes of plastic cannulae were demonstrated by Choi et al. 16 to be more favorable compared to metal needles in incident HD patients, with a lower overall incidence of vessel injury and lower time for hemostasis. However, the patients’ pain scores and nursing staff’s cannulation difficulty scores were higher in the plastic group, emphasizing the great importance of practice in using plastic cannulae. In conclusion, plastic cannulae were identified as a new and innovative way to improve the quality of dialysis.
Before choosing the intervention and the technology to use, healthcare decision-makers should consider the entire patient pathway including their life plan, 4 to make an accurate assessment. 2 The complications that occur because of cannulation not only cause significant discomfort and distress 15 to patients but in some cases, may negatively impact their ability to have functioning dialysis access in the future. 4 History has shown that the use of metal needles on cannulation has contributed to extravasation of the vessel, thrombus, aneurysm, pseudoaneurysm, and stenosis formation, in addition to patient anxiety when starting HD, particularly related to the pain of needle insertion. 18 Several new grafts with more refined properties, such as permitting immediate cannulation, heparin bonding, tapered designs, spiral flow, etc. have been brought to the market. 2 The use of plastic cannulae showed improved outcomes with no aborted dialysis sessions and no mid-dialysis extravasations compared to the traditional metal needles, 19 which combined with accurate and efficient cannulation techniques,4,11,12 reduces complication rate, improves patients’ quality of life, and reduces healthcare costs consumption. 20
A limitation of this analysis is that to the data analysis method did not allow for the evaluation of long-term risks associated with vascular access complications and longevity; the costs considered were per single episode, and it was not able to consider the possibility of developing more than one adverse event simultaneously.
Despite this limitation, the manuscript represents original research developed to cover the gap of knowledge, at both the Italian level as well as at the international level, regarding complications management costs in HD patients, and could offer the basis for future considerations including an extensive time perspective.
This analysis could objectively inform decision-makers and support evidence-based medicine and NHS resource optimization, matching clinical aspects to economic considerations.
However, further studies, ideally multi-country, large, randomized comparisons of different forms of needles for vascular access, including cannulation techniques, and PROMs, are required to statistically demonstrate the reduction in complication rate associated with cannulae needles and better inform shared decision-making.
Other national and international studies were conducted to assess NHS costs related to the different types of dialysis,21,22 but none of them included the relative risk of developing AEs related to them. This analysis offers an additional point of view to the NHS’s often underestimated, hospital point of view. Every complication and hospitalization that could be avoided somehow would serve to increase hospitals’ efforts, and reduce their capacity constraints, which are nowadays stressed even more due to COVID-19.
Many factors need to be taken into consideration: patient’s clinical characteristics, dialysis treatment, cannulation techniques, and vascular accesses, related relative complications risk. Taken all together, research and development (R&D) in new technology, associated with evidence-based medicine, is the key to progress in improving patients’ quality of care and life.
Conclusions
Based on the analysis conducted, it is clear that the use of plastic cannulae for at least selected HD patients should be mandatory, replacing metal needles. Plastic cannulae are considered safe and reduce complications management costs in the short term. The limit is that it represents only a small part of the entire patient pathway; however, it is one of the strategies that can contribute to optimize HD patient management and to improve patients’ quality of life. It is urgent the need for more comprehensive and accurate data from longer-term studies to be able to show the potential costs and benefits for future investments.
Footnotes
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: the manuscript was developed by the Health Economics department of Medtronic Italy, S.p.A., with the collaboration of Professor Tozzi, Director of vascular surgery of ASST Sette Laghi, and Associate Professor at Insubria University of Varese, who declared there is no conflict of interest for this specific work, and that he has been involved by Medtronic in other circumstances for lectures, presentations, speakers’ bureaus, manuscript writing, or educational events after honoraria.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval/Informed consent: The economic analysis developed does not require an informed consent statement, since the percentage of adverse events considered came from literature, and it was validated and adjusted according to the experience of Professor Tozzi. A specific patients population analysis of ASST Sette Laghi Varese hospital was not performed, but only items related to costs come from the state hospital.
ORCID iD: Federica Tito  https://orcid.org/0000-0002-7984-0349
https://orcid.org/0000-0002-7984-0349
References
- 1. Cusinato S. 62° SIN National Congress, Italian Nephrology Society (SIN), https://congresso2021.eventisin.it/ (2021).
- 2. Kingsmore DB, Stevenson KS, Jackson A, et al. Arteriovenous access graft infection: standards of reporting and implications for comparative data analysis. Ann Vasc Surg 2020; 63: 391–398. [DOI] [PubMed] [Google Scholar]
- 3. Ministero della salute. Documento di indirizzo per la malattia renale cronica, https://www.salute.gov.it/imgs/C_17_pubblicazioni_2244_allegato.pdf (2020, accessed 28 April 2022).
- 4. Lok C, Huber TS, Lee T, et al. KDOQI clinical practice guideline for vascular access: 2019 update. Am J Kidney Dis 2020; 75(4 Suppl 2): S1–S164. [DOI] [PubMed] [Google Scholar]
- 5. Vachharajani TJ, Taliercio JJ, Anvari E. New devices and technologies for hemodialysis vascular access: a review. Am J Kidney Dis 2021; 78(1): 116–124. [DOI] [PubMed] [Google Scholar]
- 6. Pisoni RL, Zepel L, Port FK, et al. Trends in US vascular access use, patient preferences, and related practices: an update from the US DOPPS practice monitor with international comparisons. Am J Kidney Dis 2015; 65(6): 905–915. [DOI] [PubMed] [Google Scholar]
- 7. Pisoni RL, Arrington CJ, Albert JM, et al. Facility hemodialysis vascular access use and mortality in countries participating in DOPPS: an instrumental variable analysis. Am J Kidney Dis 2009; 53(3): 475–491. [DOI] [PubMed] [Google Scholar]
- 8. Roy-Chaudhury P, Sukhatme VP, Cheung AK. Hemodialysis vascular access dysfunction: a cellular and molecular viewpoint. J Am Soc Nephrol 2006; 17(4): 1112–1127. [DOI] [PubMed] [Google Scholar]
- 9. Swedberg SH, Brown BG, Sigley R, et al. Intimal fibromuscular hyperplasia at the venous anastomosis of PTFE grafts in hemodialysis patients. Clinical, immunocytochemical, light and electron microscopic assessment. Circulation 1989; 80(6): 1726–1736. [DOI] [PubMed] [Google Scholar]
- 10. MacRae JM. Should buttonhole cannulation of arteriovenous fistulas be used? CON. Kidney360 2020; 1(5): 322–325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Liu Z, Bible J, Petersen L, et al. Measuring cannulation skills for hemodialysis: objective versus subjective assessment. Front Med (Lausanne) 2021; 8: 777186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Parisotto MT, Schoder VU, Miriunis C, et al. Cannulation technique influences arteriovenous fistula and graft survival. Kidney Int 2014; 86(4): 790–797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Gage SM, Reichert H. Determining the incidence of needle-related complications in hemodialysis access: we need a better system. J Vasc Access 2021; 22(4): 521–532. [DOI] [PubMed] [Google Scholar]
- 14. Marticorena RM, Dacouris N, Donnelly SM. Randomized pilot study to compare metal needles versus plastic cannulae in the development of complications in hemodialysis access. J Vasc Access 2018; 19(3): 272–282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Staaf K, Fernström A, Uhlin F. Cannulation technique and complications in arteriovenous fistulas: a Swedish Renal Registry-based cohort study. BMC Nephrol 2021; 22(1): 256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Choi YS, Lee HS, Joo N, et al. Efficacy and safety of plastic cannulae compared with metal needles in the initial use of an arteriovenous fistulae in incident hemodialysis patients: a randomized controlled study. Am J Nephrol 2021; 52(6): 479–486. [DOI] [PubMed] [Google Scholar]
- 17. Richarz S, Greenwood S, Kingsmore DB, et al. Validation of a vascular access specific quality of life measure (VASQoL). J Vasc Access. Epub ahead of print 5 October 2021. DOI: 10.1177/11297298211046746. [DOI] [PubMed] [Google Scholar]
- 18. Kim MK, Kim HS. Clinical effects of buttonhole cannulation method on hemodialysis patients. Hemodial Int 2013; 17(2): 294–299. [DOI] [PubMed] [Google Scholar]
- 19. Smith V, Schoch M. Do plastic cannulae have better outcomes than metal needles in haemodialysis? A retrospective review. Ren Soc Australas J 2017; 13(3): 78–82. [Google Scholar]
- 20. De Barbieri I, Sisti D, Strini V, et al. Plastic cannulae versus metal needle cannulation in haemodialysis: results of an international survey from the nurse perspective. J Vasc Access. Epub ahead of print 30 December 2021. DOI: 10.1177/11297298211066763. [DOI] [PubMed] [Google Scholar]
- 21. Roberts G, Holmes J, Williams G, et al. Current costs of dialysis modalities: a comprehensive analysis within the United Kingdom. Perit Dial Int. Epub ahead of print 24 January 2022. DOI: 10.1177/08968608211061126. [DOI] [PubMed] [Google Scholar]
- 22. Vaccaro CM, Sopranzi F. A comparison between the costs of dialysis treatments in Marche region, Italy: Macerata and Tolentino hospitals. Ann Ist Super Sanita 2017; 53(4): 344–349. [DOI] [PubMed] [Google Scholar]


