Abstract
Context:
Patient misperceptions are a strong barrier to early palliative care discussions and referrals during advanced lung cancer treatment.
Objectives:
We developed and tested the acceptability of a web-based patient-facing palliative care education and screening tool intended for use in a planned multilevel intervention (i.e., patient, clinician, system-level targets).
Methods:
We elicited feedback from advanced lung cancer patients (n=6), oncology and palliative care clinicians (n=4), and a clinic administrator (n=1) on the perceived relevance of the intervention. We then tested the prototype of a patient-facing tool for patient acceptability and preliminary effects on patient palliative care knowledge and motivation.
Results:
Partners agreed that the intervention—clinician palliative care education and an electronic health record-integrated patient tool—is relevant and their feedback informed development of the patient prototype. Advanced stage lung cancer patients (n=20; age 60 ± 9.8; 40% male; 70% with a technical degree or less) reviewed and rated the prototype on a five-point scale for acceptability (4.48 ± 0.55), appropriateness (4.37 ± 0.62), and feasibility (4.43 ± 0.59). After using the prototype, 75% were interested in using palliative care and 80% were more motivated to talk to their oncologist about it. Of patients who had or were at risk of having misperceptions about palliative care (e.g., conflating it with hospice), 100% no longer held the misperceptions after using the prototype.
Conclusion:
The palliative care education and screening tool is acceptable to patients and may address misperceptions and motivate palliative care discussions during treatment.
Keywords: palliative care, improved referral process, patient-requested referral, advanced stage lung cancer, clinician education, palliative care misperceptions
INTRODUCTION
Despite guidelines and evidence supporting the benefits of early co-management between oncology and specialty palliative care (1), most patients with advanced lung cancer are not referred for ambulatory palliative care consultation during treatment—even in academic medical centers with well-established palliative care programs (2, 3). Oncology clinicians frequently cite patients’ misperception that palliative care is synonymous with hospice/end-of-life as a major barrier to early co-management (4–6). Due to this misperception, clinicians hesitate to initiate discussions about palliative care early in treatment (4, 7), creating a self-fulfilling prophecy wherein palliative care conversations are delayed until the transition to end-of-life.
Most interventions to improve outpatient palliative care utilization have focused on system- or clinician-level factors, including routine screening for palliative care needs (e.g., symptom concerns), referral triggers, and/or intensive clinician education, with varied results (4, 8–14). One trial conducted among gynecologic cancer patients tested the effects of patient education on palliative care referrals and yielded null results, possibly due to high baseline knowledge of palliative care among the sample and/or lack of simultaneous engagement with oncology clinicians (14). As patient–oncologist discussions about palliative care are critical to timely utilization (5, 7, 15, 16), there is a clear need for interventions that educate patients about palliative care during routine oncology visits to foster these discussions. Adding such a patient component to recommended system- and clinician-level interventions may help address well-documented multilevel barriers to palliative care use during advanced cancer treatment (15, 17).
The goals of this study were to refine the concept of a planned multilevel intervention to improve palliative care utilization and to develop and test the acceptability of the patient-facing intervention component, designed to educate patients about palliative care; assess relevant unmet needs; and facilitate discussion of the role of palliative care in advanced lung cancer treatment.
METHODS
Study Design
This was a two phase, formative intervention study. We began with a plan for a multilevel intervention (18), called “mPal,” that was informed by prior research (3), including our assessment of patient, clinician, and administrator perspectives on determinants of palliative care utilization during routine advanced lung cancer treatment (15), and a well-established behavior change framework previously used in describing palliative care determinants (19, 20). In this study, we collected feedback from patients, clinicians, and administrators on the relevance of the proposed intervention and feedback on the content of the intervention’s planned patient tool (phase I) (21). We then developed a full prototype of the patient tool and tested its acceptability with patients (phase II). The University of Kentucky Institutional Review Board (#63922) approved this study. Methods and results are reported by study phase for clarity.
Phase I Methods—Planned Multilevel Intervention (mPal)
Our plan for mPal was to target patient and clinician capability/knowledge, motivation, and opportunity to discuss and use palliative care during lung cancer treatment using core intervention functions of education and environmental restructuring (Figure 1) (19, 22). In our prior research (15), we identified an electronic health record (EHR)-integrated patient tool as a key potential intervention form to motivate patients and clinicians to discuss palliative care.
Figure 1. Theoretical model and planned multilevel intervention targets for mPal.
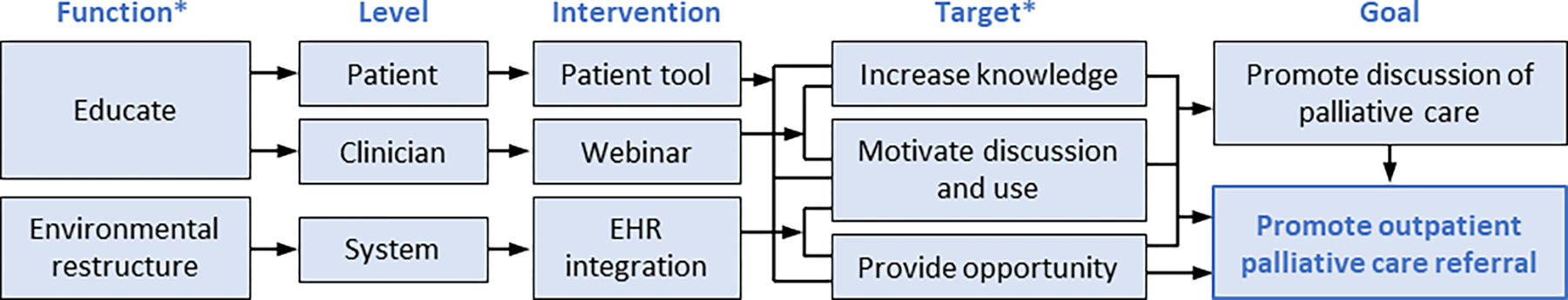
*COM-B (Capability/knowledge, Opportunity, Motivation–Behavior) Model and Behavior Change Wheel Constructs (19, 22); EHR: electronic health record.
Participants.
Phase I participants included patients, clinicians, and administrators. A planned sample size of 10 participants was based on the narrow scope of the research questions and homogeneous target population (23). Patients were eligible if they were English-speaking, had advanced stage lung cancer (extensive stage small cell or stage III or IV non-small cell), were age 18 or older, were at least 3 weeks into cancer treatment, and had an ECOG performance status of 0–3. Clinicians and administrators were eligible if they were an oncologist, advanced practice provider, nurse, pharmacist, or palliative care provider who treated advanced stage lung cancer patients, or an oncology practice manager. Participants were recruited between February and May 2021. Six of nine patients approached in clinic participated (67%). Five of ten clinicians and administrators who were approached (50%) participated. All participants received $50. Patients provided written informed consent; clinicians and administrators reviewed a study information sheet.
Surveys.
Patients completed a survey of self-report measures of sociodemographic factors (age, race, education, income, etc.), health literacy (BRIEF Health Literacy Screener (24); Cronbach’s α = .88), and palliative care knowledge and misperceptions from the Health Information National Trends Survey (25). Clinical information (e.g., lung cancer stage) was abstracted from the EHR. Clinicians and administrators self-reported demographic information, including years in practice, position, patient volume, etc.
Interviews.
A semi-structured interview guide queried proposed multilevel intervention operational flow, content, and delivery methods. The guide was developed based on our prior research on determinants of palliative care use among patients with advanced lung cancer (15). Patient interviews were conducted in-person by study staff during patients’ routine oncology appointments or infusions. Patient interviews queried feedback on potential palliative care education content, needs assessment domains (e.g., symptoms, psychosocial concerns), and delivery preferences. Clinician/administrator interviews were conducted via Zoom® and queried patient palliative care needs assessment domains, potential integration into the clinical workflow, and acceptability of potential clinician-facing education content. All interviews were recorded and transcribed verbatim. Patient interviews averaged 22 ± 8 minutes; clinician/administrator interviews averaged 33 ± 13 minutes.
Analysis.
Interview data analyses followed a codebook/template-guided approach (26, 27). First, three study team members (JLS, VB, KT) engaged in a data familiarization process, with each member reviewing transcripts and inserting memos into the data. The team members engaged in multiple discussions to compare memos and identify common codes to be used in labeling like responses within each broader interview question category (e.g., patients’ responses to the proposed palliative care needs assessment domains). VB and KT then applied these codes to the relevant text in each transcript and sorted text within these codes for review by the entire study team in guiding the intervention adaptation.
Phase II Methods—Patient Tool Prototype Review
Participants.
Patients who completed phase I were not eligible for phase II; all other eligibility criteria were the same. Patients were recruited between June and August 2021. We approached 24 patients in clinic and 20 consented (83%). Patients provided written informed consent and received $50 for participating. Patients were asked to use the web-based mPal patient tool on a Wi-Fi-connected iPad and complete pre- and post-surveys in clinic.
Surveys.
Patients completed the same survey measures as in phase I. For analysis, palliative care knowledge items were recoded and examined individually (e.g., “The goal of palliative care is to manage pain…”; “strongly disagree” or “somewhat disagree” = incorrect; “strongly agree” or “somewhat agree” = correct; “don’t know” = “don’t know”) and misperception items were recoded and examined individually (e.g., “If you accept palliative care, you must stop other treatments”; “agree” or “strongly agree” = misperception endorsed; “strongly disagree” or “disagree” = misperception not endorsed; “don’t know” = “don’t know”). Next, patients interacted with the patient tool prototype. Finally, patients were asked to retake the measure of palliative care knowledge and rate the intervention’s acceptability and potential effectiveness. Acceptability was assessed with items from Weiner et al.’s Feasibility of Intervention (2 items), Intervention Appropriateness Measure (3 items), and Acceptability of Intervention (3 items) measures (28). Items were responded to on a scale of 1 (completely disagree) to 5 (completely agree). Scores were averaged by subscale, with higher scores indicating greater feasibility, appropriateness, and acceptability (Cronbach’s α = .96, .96, and .83, respectively). These items were supplemented with additional investigator-generated items (e.g., “Using the mPal app in clinic would be convenient”), which were responded to on the same Likert-type scale and examined individually. Investigator-generated items assessed the extent to which the intervention content could affect palliative care interest (“After using the mPal app, I would be interested in using palliative care”), knowledge (“After using the mPal app, I learned something about what palliative care was”), attitudes (“After using the mPal app, palliative care seems less scary”), and motivation (“After using the mPal app, I feel more motivated to talk to my cancer doctor about symptoms or concerns I have”). Items were responded to on a scale of 1 (completely disagree) to 5 (completely agree) and examined individually.
Analysis.
We computed means, medians, standard deviations, and percentages for survey data. Binary variable correlations were calculated using phi coefficient. Pre–post changes in palliative care knowledge and misperceptions in phase II were assessed using exact McNemar’s test with a two-tailed alpha of 0.05. “Don’t know” responses were counted as “incorrect” or “endorsing a misperception” when dichotomizing responses. Survey data were analyzed using SPSS v.29.
RESULTS
Phase I Results—Refining the Multilevel Intervention
Patient Sample Characteristics.
Phase I participants (n = 6) were on average 60.83 years of age (SD 3.06; range 57–65 years). Most (67%) were male; 100% were non-Hispanic White; 50% had a technical/vocational degree or less education. Three were retired, one was on disability, and two were working full-time. Half had a household income of $25,000 or less. Two had limited health literacy, one marginal, and three adequate health literacy. Three had stage IV non-small cell lung cancer and three had extensive stage small cell lung cancer. Half had never heard of palliative care; half thought they knew a little bit about it.
Clinician and Administrator Sample Characteristics.
Participants (n = 5) included two medical oncologists, a palliative care physician, a registered oncology nurse, and an oncology clinic manager. Participants were on average 52.2 (SD 7.46) years of age; 60% were female; 40% had been in practice 11–20 years; 60% had been in practice 20+ years; 60% saw 11–30 patients with advanced lung cancer in a typical week; 40% saw 10 or fewer per week.
Interview Results.
Supplemental Table 1 summarizes the primary participant recommendations for mPal intervention concept content and delivery, with illustrative quotes that were influential in our intervention refinement. Most patients lacked pre-existing knowledge of palliative care, supporting the need for an educational component; clinicians were agreeable to a pre-recorded webinar to remind them of palliative care guidelines and evidence. Key recommendations that informed the intervention refinement and patient prototype development included: providing video and text educational content for patients, utilizing clinical flowsheets in the EHR to integrate screening tool results, and not automatically referring patients to palliative care.
Multilevel Intervention Concept Refinement and Patient Tool Prototype Development.
Based on participant feedback, we developed the proposed multilevel intervention workflow and patient tool prototype. The planned multilevel intervention will include a webinar to educate oncology clinicians about the evidence for palliative care in advanced lung cancer treatment, clinical guidelines pertaining to palliative care, and an orientation to the patient EHR-integrated tool (Figure 2). Clinics can then implement the patient tool, which includes: (a) education about palliative care (text and ~1.5-minute video describing what palliative care is, how it differs from hospice, and benefits of using it during advanced lung cancer treatment); (b) a 16-item assessment of palliative care needs (e.g., physical, mental health symptom, prognostic, and family/caregiver psychosocial concerns; informed by interviews as well as our previous research on unmet care needs (29) and concerns patients indicated they would want to track with their oncology team (unpublished data)); and (c) an assessment of interest in discussing palliative care needs and its role in treatment, receiving more information about palliative care, and interest in a referral (see Supplemental Figure 1 for screenshots). Based on concerns raised by oncology clinicians and palliative care physicians regarding the potential for miscommunication between clinicians about co-management and their prior experiences with automatic referrals in the inpatient setting, we decided not to automatically trigger a palliative care referral based on patient needs or patient interest. The patient tool is intended to be completed at or prior to a routine oncology visit through the patient EHR portal or on a tablet in clinic. Results will be entered into the EHR via a clinical flowsheet where they can be viewed by the oncology team with other flowsheet data, such as lab values. The prototype of the patient tool described above was developed as a web app for testing in phase II.
Figure 2. Multilevel intervention workflow after Phase I.
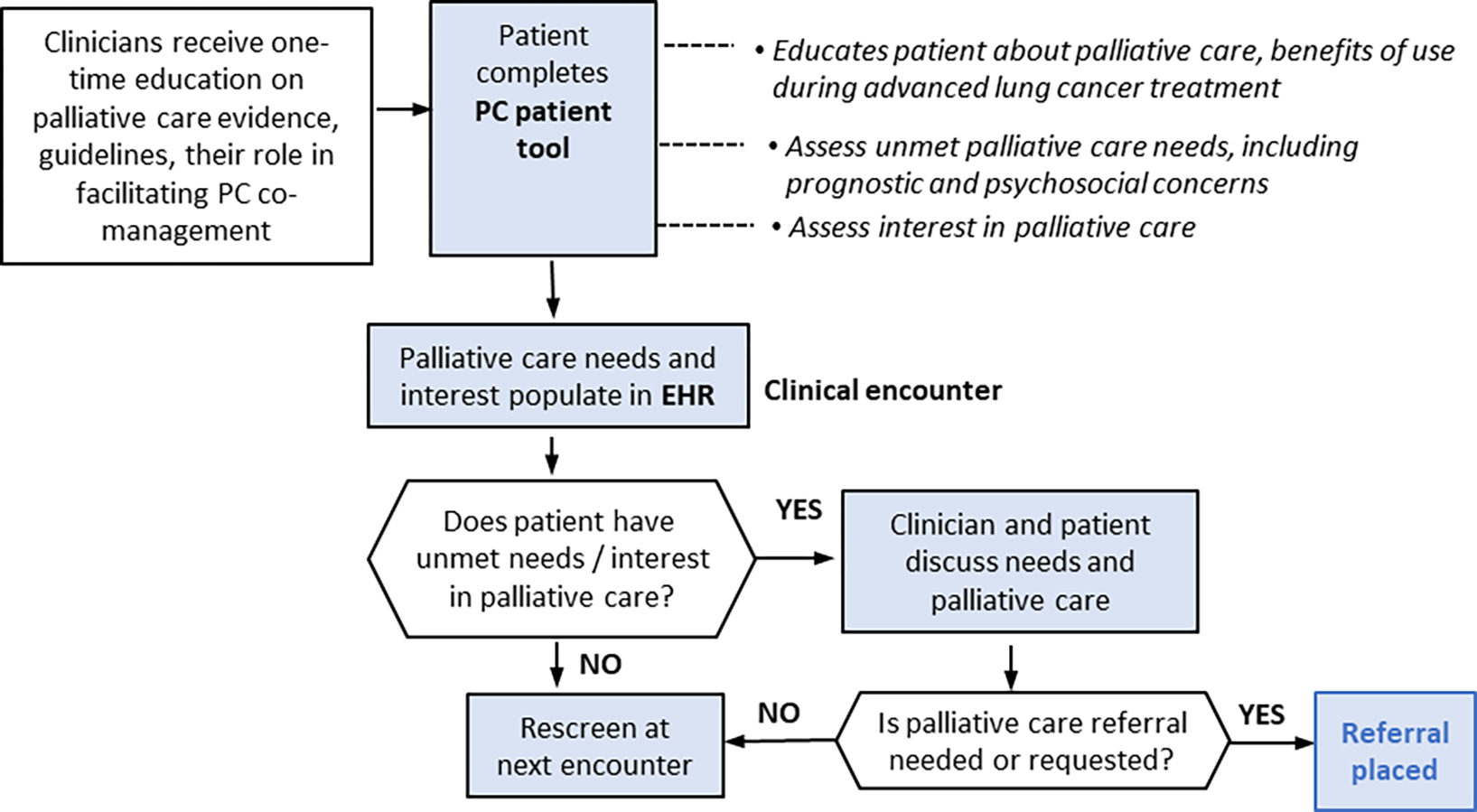
Phase II—Patient Tool Prototype Testing
Sample Characteristics.
Patients were on average 59.80 years old (SD 9.82; range 32–72). All were non-Hispanic White; 40% were male; 70% had a technical degree or less; 70% were receiving disability or retired; 63.2% had an annual household income of $25,000 or less. Most (80%) had non-small cell lung cancer, 70% of which were stage IV. Patient health literacy was low: 30% had limited health literacy, 40% marginal, and 30% adequate.
Patient Tool Responses.
Responses to the participant assessment tool are shown in Table 1. The most common concerns were fatigue, wondering about how long they would live, worry about loved ones; the most common concerns patients wanted help with were financial problems, fatigue, worry about the future, and appetite problems. Over half wanted more information about palliative care. Several of the palliative care interest items were correlated (Table 1); desire for more information about palliative care and desire to talk with a doctor about palliative care trended toward being correlated (ϕ = .452, p = .091).
Table 1.
Participant Responses to Prototype Patient Tool Assessment (N = 20 advanced lung cancer patients)
| Potential Concern | n (%)“Yes” (experienced in the past 30 days) | n who wanted help with the concern/ of those who experienced the concern (%) | |||
|---|---|---|---|---|---|
|
| |||||
| Feeling tired | 18 (90%) | 9/18 (50%) | |||
| Wondering about how long I might live | 17 (85%) | 5/17 (29%) | |||
| Worry about loved ones | 16 (80%) | 6/16 (38%) | |||
| Nausea | 15 (75%) | 4/15 (27%) | |||
| Difficulty breathing | 14 (70%) | 4/14 (29%) | |||
| Appetite problems | 14 (70%) | 7/14 (50%) | |||
| Feeling down | 14 (70%) | 3/14 (21%) | |||
| Depression or anxiety | 13 (65%) | 5/13 (38%) | |||
| Worry about the future | 13 (65%) | 7/13 (54%) | |||
| Pain | 11 (55%) | 4/11 (36%) | |||
| Constipation | 10 (50%) | 2/10 (20%) | |||
| Sleep problems | 10 (50%) | 4/10 (40%) | |||
| Unsure about treatment options | 10 (50%) | 2/10 (20%) | |||
| Diarrhea | 6 (30%) | 1/6 (17%) | |||
| Financial problems | 6 (30%) | 4/6 (67%) | |||
| Spiritual/religious concerns | 2 (10%) | 1/2 (50%) | |||
|
| |||||
| Palliative Care Referral/Interest Questi ms | n (%) “Yes” | Phi Coefficient (p-value) | |||
|
| |||||
| 2 | 3 | 4 | |||
|
| |||||
| 1. Would you like to schedule a visit with a palliative care provider as part of your cancer care? | 8 (40%) | .583 (.009) | .492 (.028) | .452 (.092) | |
| 2. Would you like to talk to your doctor about palliative care? | 8 (40%) | .698 (.002) | .452 (.091) | ||
| 3. Would you like to talk with your doctor about the concerns you noted? | 9 (45%) | .101 (.707) | |||
| 4. Would you like more information about palliative care? | 11 (55%) | ||||
Effects of mPal Patient Tool on Palliative Care Knowledge and Misperceptions.
After interacting with the mPal patient tool, nine (81.8%) of the eleven patients who had never heard of palliative care at baseline reported they knew a little bit about it; two (18.2%) reported they knew what palliative care was and could explain it to someone else. Of the eight who said they knew a little bit about palliative care at baseline, seven (87.5%) reported they knew a little bit about it after using the patient tool; one (12.5%) reported they knew what palliative care was and could explain it. Only six to seven patients correctly identified functions of palliative care at baseline. Of the 13–14 who did not, nearly all correctly identified them as palliative care functions after viewing education in the patient tool (Table 2). One patient who “did not know” and one patient who correctly identified that a goal of palliative care was to help friends and family cope with a patient’s illness at baseline incorrectly omitted it as a goal of palliative care after viewing the tool’s educational content.
Table 2.
Effects of mPal patient tool on patient palliative care knowledge (N = 20 advanced lung cancer patients)
| Pre-mPa* | Post-mPal* | ||||
|---|---|---|---|---|---|
| Goals of Palliative Care | Correctly identified | “Don’t know” | Incorrectly omitted | Changed from “don’t know” or incorrectly omitted to correctly identified |
Changed from correctly identified to incorrectly omitted |
| Offer social and emotional support | 7 | 13 | 0 | 13 | 0 |
| Manage pain and other symptoms a | 6 | 13 | 0 | 13 | 0 |
| Help friends and family to cope with a patient’s illness | 7 | 13 | 0 | 12 | 1 |
| Misperception | Did not endorse | “Don’t know” | Endorsed | Chinged from “don’t know” or endorsed to not endorsed | Changed from not endorsed to endorsed or “don’t know” |
| If you accept palliative care, must stop other treatments | 7 | 13 | 12 | 0 | |
| Accepting palliative care means giving up | 6 | 14 | 0 | 14 | 0 |
| Same as hospice | 4 | 14 | 2 | 16 | 0 |
Pre-post changes were assessed with exact McNemar tests in which “Don’t know” responses were counted as either “incorrectly omitting for knowledge items or “endorsed misperception” for misperception items. Exact McNemar tests showed the pre/post proportions were different for each knowledge and misperception item, p <.005 (two-sided alpha).
Response was missing for one person.
Only three patients did not endorse misperceptions about palliative care at baseline. Of those who either “did not know” or endorsed a misperception, all but one did not endorse the misperception after viewing the tool’s content (see Table 2); one patient maintained a “do not know” response for the misperception that accepting palliative care would mean stopping other treatments. All who accurately did not endorse a misperception prior to completing the tool maintained accurate responses post.
Acceptability of mPal Patient Tool and Effects on Palliative Care Interest and Motivation.
Patients rated the mPal patient tool highly regarding acceptability, appropriateness, and feasibility (Table 3). Most (75–100%) agreed or strongly agreed that the tool improved their knowledge of palliative care, increased their interest in using palliative care, increased their motivation to discuss it with their oncology team, and made palliative care seem less scary.
Table 3.
Acceptability of mPal patient tool and effects on palliative care interest and motivation (N = 20 advanced lung cancer patients)
| Acceptability | Mean ± SD; Observed range; Median (IQR) or n (%) Agree or Strongly Agree |
|---|---|
| Acceptabilitya of mPal | 4.48 ± 0.55; 3.33–5.00; 4.50 (1.00) |
| Appropriateness a of mPal | 4.37 ± 0.62; 3.33–5.00; 4.00 (1.00) |
| Feasibility a of mPal | 4.43 ± 0.59; 3.00–5.00; 4.25 (1.00) |
| Using the mPal app in clinic would be doable | 20 (100%) |
| Using the mPal app in clinic would be convenient | 18 (90%) |
| Potential Effectiveness of mPal Patient Tool | |
| After using mPal, I would be interested in using palliative care during my cancer treatment | 15 (75%) |
| After using mPal, I learned something about palliative care | 20 (100%) |
| After using mPal, palliative care seems less scary | 16 (80%) |
| After using mPal, I feel more motivated to talk to my docto, about a symptom or concern I have | 16 (80%) |
Note. From Weiner et al.’s measures (28); possible range = 1 – 5, with higher scores indicating greater acceptability, appropriateness, and feasibility.
DISCUSSION
To help oncology clinicians refer patients with advanced lung cancer to palliative care during cancer treatment, patient misperceptions must be addressed and their palliative care needs must be identified (17). The goal of this study was to develop patient-facing components of a multilevel intervention (18) to address patient misperceptions and facilitate discussions of patients’ palliative care needs and its role in advanced lung cancer treatment during routine oncology visits. Consistent with best practices for intervention development to ensure relevance and patient-centered care (21), key partners provided feedback on the intervention concept and content, which we then tested for patient usability, feasibility, acceptability, and appropriateness. Overall, results suggest the patient tool holds promise as an intervention component to facilitate discussions of patients’ needs and the role of palliative care during routine oncology visits.
Given the prevalence and salience of patient misperceptions (3, 15, 30, 31), a primary consideration in designing the patient tool was to provide education that could improve attitudes toward palliative care. Based on our prior research with patients with advanced lung cancer and our phase I results (3, 15), we described palliative care in terms of what it is not (i.e., not hospice), what it is, and how it can be helpful. The pre–post changes observed in patients’ palliative care knowledge and misperceptions during phase II suggest that this approach effectively addressed misperceptions. After interacting with the mPal patient tool, patients correctly identified the functions of palliative care and did not endorse misperceptions. Additionally, patients reported increased motivation to use palliative care and discuss their it with their oncology team. These positive outcomes were particularly significant considering the sample’s limited educational attainment and health literacy, which puts them at higher risk for misperceptions (31). However, if future trials show 75% of patients are interested in palliative care after interacting with the tool (as observed in this pilot), most clinics will clearly need to further develop procedures for implementing referrals (e.g., specifying thresholds for referral, identifying which concerns will be managed primarily through oncology and/or other supportive oncology services), as specialty palliative care is a limited resource.
The palliative care needs screener component of the patient tool that was developed and piloted in this study was mostly viewed as comprehensive and appropriate by patients, clinicians, and administration. Usability testing suggested the tool is feasible, appropriate, and acceptable to patients. However, the screener is not psychometrically validated, and some items, namely symptom items, overlap with validated tools that are recommended to screen for palliative care needs (e.g., Edmonton Symptom Assessment Scale; ESAS (32)) (17). Thus, in future studies we plan to incorporate a validated screening tool such as the ESAS and supplement it with the additional unique items developed and piloted in this study (e.g., “wondering how long I might live”).
We plan to deliver the patient tool via the patient EHR portal (MyChart® in EPIC®) and/or via tablet in clinic. Based on clinician feedback, we also plan to integrate patients’ palliative care needs screening and interest responses in the EHR by leveraging clinical flowsheets and tools to embed results in outpatient oncology notes (e.g., a SmartPhrase in EPIC®). Considerations for future studies include determining whether to display all vs. “key” results (e.g., only the concerns patients want help with or clinical thresholds for symptom scales), especially given the possibility of alert fatigue (33).
A unique feature of the mPal patient tool is its assessment of patients’ desire to discuss their palliative care needs, the role of palliative care, and their interest in a referral by their oncology team. We originally intended for the response to interest in a palliative care referral to trigger an automatic palliative care consultation. However, oncology clinicians and palliative care physicians expressed concern due to clinician variability in comfort with automatic referrals. This finding is consistent with a recent study that indicated oncology clinicians should remain gatekeepers to outpatient palliative care (34). We decided to keep the assessment of a patient’s interest in a palliative care referral in the mPal patient tool to document to the oncology team whether a patient is already interested in a referral, hopefully enabling the oncology team to discuss palliative care and place the referral with a request tied to a patient palliative care need identified from the assessment. Whether this has its intended effect remains to be tested.
This study is limited by its conduct at a single academic medical center. However, the mPal patient tool concept was informed by our prior research with advanced lung cancer patients, clinicians, and administrators representing multiple community oncology practices and our academic medical center (15). Thus, we expect the tool to be widely generalizable. Other limitations include a patient sample that, though consistent with our catchment area (35) and representative of the broader lung cancer patient population in terms of many sociodemographic factors (36), was non-Hispanic White. Additional tailoring may be needed to reach racially and ethnically minoritized patient populations (37). Finally, we did not test the integration of the tool in the EHR or clinician workflow. We are currently doing so as part of a feasibility trial (NCT05270395) of the entire multilevel intervention, which also includes one-time oncology clinician education to synergistically address barriers to palliative care utilization during advanced lung cancer treatment. Whether one-time oncology clinician education is sufficient to address provider misconceptions about palliative care will be an important outcome in that pilot trial. We selected one-time education for the trial to minimize provider burden, but phase I interviews also raise the possibility that one-time education may address misconceptions, but be insufficient for building necessary trust between Oncology and Palliative Care to effectively support integration. Thus, an important secondary outcome of the feasibility trial of the multilevel intervention will be to begin to identify potential contextual factors that may influence successful implementation and ultimately the effectiveness of the intervention (e.g., how long specialty palliative care has been available in a clinic as a contextual factor for trust among providers, patient health literacy as a factor that may influence ability to complete the mPal patient tool independently vs. requiring additional aid).
In conclusion, the mPal patient tool is acceptable, feasible, and appropriate to patients and may be promising approach to help educate patients about palliative care, assess their palliative care needs, and prime patients and oncologists to discuss the role of palliative care as part of routine oncology visits.
Supplementary Material
Key Message:
A patient palliative care education and assessment tool demonstrated acceptability and has strong potential to help address palliative care misperceptions and support referrals if integrated into multilevel interventions to improve primary and specialty palliative care receipt.
ACKNOWLEDGEMENTS
The Markey Cancer Center’s Research Communications office assisted with manuscript preparation. We would like to thank Kimberly Haney, MLS, for her assistance with qualitative data coding.
Funding:
This work was supported by the University of Kentucky Markey Cancer Center (P30 CA177558); National Center for Advancing Translational Sciences, National Institutes of Health KL2TR001996; and the University of Kentucky Igniting Research Collaborations Grant.
Footnotes
Conflicts of Interest: None to declare.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Data Statement:
Survey data will be made available upon reasonable request to the corresponding author.
REFERENCES
- 1.Ferrell BR, Temel JS, Temin S, et al. Integration of Palliative Care Into Standard Oncology Care: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol 2017;35:96–112. [DOI] [PubMed] [Google Scholar]
- 2.Huo J, Hong Y-R, Turner K, et al. Utilization pattern and service settings of palliative care for patients with metastatic non–small cell lung cancer. Cancer 2019;125:4481–4489. [DOI] [PubMed] [Google Scholar]
- 3.McLouth LE, Gabbard J, Levine BJ, et al. Prognostic Awareness, Palliative Care Use, and Barriers to Palliative Care in Patients Undergoing Immunotherapy or Chemo-Immunotherapy for Metastatic Lung Cancer. Journal of Palliative Medicine 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sedhom R, Gupta A, Shah M, et al. Oncology Fellow–Led Quality Improvement Project to Improve Rates of Palliative Care Utilization in Patients With Advanced Cancer. JCO Oncology Practice 2020;16:e814–e822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Feld E, Singhi EK, Phillips S, et al. Palliative care referrals for advanced non–small-cell lung cancer (NSCLC): patient and provider attitudes and practices. Clinical Lung Cancer 2019;20:e291–e298. [DOI] [PubMed] [Google Scholar]
- 6.Dunn S, Earp MA, Biondo P, et al. Oncology Clinicians’ Challenges to Providing Palliative Cancer Care—A Theoretical Domains Framework, Pan-Cancer System Survey. Current Oncology 2021;28:1483–1494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Meritens ABd, Margolis B, Blinderman C, et al. Practice Patterns, Attitudes, and Barriers to Palliative Care Consultation by Gynecologic Oncologists. Journal of Oncology Practice 2017;13:e703–e711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Conduit C, Thompson M, Thomas R, Nott L, Wuttke M. Implementing “Goals of Care” discussion and palliative care referral for patients with advanced lung cancer – an outpatient-based pilot project. Internal Medicine Journal;n/a. [DOI] [PubMed] [Google Scholar]
- 9.Hong NJL, Liu N, Wright FC, et al. Assessing the Impact of Early Identification of Patients Appropriate for Palliative Care on Resource Use and Costs in the Final Month of Life. JCO Oncology Practice 2020;16:e688–e702. [DOI] [PubMed] [Google Scholar]
- 10.Kim JY, Peters KB, Herndon JE 2nd, Affronti ML. Utilizing a Palliative Care Screening Tool in Patients With Glioblastoma. J Adv Pract Oncol 2020;11:684–692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Reville B, Reifsnyder J, McGuire DB, Kaiser K, Santana AJ. Education and referral criteria: impact on oncology referrals to palliative care. J Palliat Med 2013;16:786–9. [DOI] [PubMed] [Google Scholar]
- 12.Hui D, Titus A, Curtis T, et al. Implementation of the Edmonton Symptom Assessment System for Symptom Distress Screening at a Community Cancer Center: A Pilot Program. Oncologist 2017;22:995–1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sridharan K, Paul E, Stirling RG, Li C. Impacts of multidisciplinary meeting case discussion on palliative care referral and end-of-life care in lung cancer: a retrospective observational study. Intern Med J 2021;51:1450–1456. [DOI] [PubMed] [Google Scholar]
- 14.Graul A, Haggerty A, Stickley C, et al. Effect of patient education on palliative care knowledge and acceptability of outpatient palliative care services among gynecologic oncology patients: A randomized controlled trial. Gynecologic Oncology 2020;156:482–487. [DOI] [PubMed] [Google Scholar]
- 15.McLouth LE, Borger T, Bursac V, et al. Palliative care use and utilization determinants among patients treated for advanced stage lung cancer care in the community and academic medical setting. Supportive Care in Cancer 2023;31:190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hay CM, Lefkowits C, Crowley-Matoka M, et al. Strategies for Introducing Outpatient Specialty Palliative Care in Gynecologic Oncology. Journal of Oncology Practice 2017;13:e712–e720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hui D, Heung Y, Bruera E. Timely palliative care: Personalizing the process of referral. Cancers 2022;14:1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Taplin SH, Anhang Price R, Edwards HM, et al. Introduction: Understanding and Influencing Multilevel Factors Across the Cancer Care Continuum. JNCI Monographs 2012;2012:2–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Michie S, Van Stralen MM, West R. The behaviour change wheel: a new method for characterising and designing behaviour change interventions. Implementation science 2011;6:1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kubi B, Enumah ZO, Lee KT, et al. Theory-Based Development of an Implementation Intervention Using Community Health Workers to Increase Palliative Care Use. J Pain Symptom Manage 2020;60:10–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Glasgow RE. What does it mean to be pragmatic? Pragmatic methods, measures, and models to facilitate research translation. Health Educ Behav 2013;40:257–65. [DOI] [PubMed] [Google Scholar]
- 22.Michie S, Johnston M, Abraham C, et al. Making psychological theory useful for implementing evidence based practice: a consensus approach. BMJ Quality & Safety 2005;14:26–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hamilton AB, Finley EP. Qualitative methods in implementation research: An introduction. Psychiatry Research 2019;280:112516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Haun J, Noland-Dodd V, Varnes J, et al. Testing the BRIEF health literacy screening tool. Fed Pract 2009;26:24–31. [Google Scholar]
- 25.Nelson DE, Kreps GL, Hesse BW, et al. The Health Information National Trends Survey (HINTS): development, design, and dissemination. J Health Commun 2004;9:443–60; discussion 81–4. [DOI] [PubMed] [Google Scholar]
- 26.Gale NK, Heath G, Cameron E, Rashid S, Redwood S. Using the framework method for the analysis of qualitative data in multi-disciplinary health research. BMC Medical Research Methodology 2013;13:117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Crabtree BF, Miller WL. Using codes and code manuals: A template organizing style of interpretation. In: Doing Qualitative Research, 2nd ed. Thousand Oaks, CA: Sage Publishing, 1999:163–177. [Google Scholar]
- 28.Weiner BJ, Lewis CC, Stanick C, et al. Psychometric assessment of three newly developed implementation outcome measures. Implementation Science 2017;12:108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.McLouth LE, Nightingale CL, Levine BJ, Burris JL, McDougall JA, Lycan TW Jr, Gabbard J, Ruiz J, Farris M, Blackstock AW Jr, Grant SC, Petty WJ, Weaver KE. Unmet care needs and financial hardship in patients with metastatic non-small-cell lung cancer on immunotherapy or chemoimmunotherapy in clinical practice. JCO Oncol Pract. 2021. Aug;17(8):e1110–e1119. doi: 10.1200/OP.20.00723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Collins A, McLachlan S-A, Philip J. Community knowledge of and attitudes to palliative care: A descriptive study. Palliative Medicine 2019;34:245–252. [DOI] [PubMed] [Google Scholar]
- 31.Parajuli J, Chen ZJ, Walsh A, et al. Knowledge, beliefs, and misconceptions about palliative care among older adults with cancer. Journal of Geriatric Oncology 2023;14:101378. [DOI] [PubMed] [Google Scholar]
- 32.Bruera E, Kuehn N, Miller MJ, Selmser P, Macmillan K. The Edmonton Symptom Assessment System (ESAS): a simple method for the assessment of palliative care patients. Journal of palliative care 1991;7:6–9. [PubMed] [Google Scholar]
- 33.Ancker JS, Edwards A, Nosal S, et al. Effects of workload, work complexity, and repeated alerts on alert fatigue in a clinical decision support system. BMC Medical Informatics and Decision Making 2017;17:36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hui D, Mori M, Meng Y-C, et al. Automatic referral to standardize palliative care access: an international Delphi survey. Supportive Care in Cancer 2018;26:175–180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Center UoKMC. 2021 Kentucky Cancer Needs Assessment. In: 2022.
- 36.Sidorchuk A, Agardh EE, Aremu O, et al. Socioeconomic differences in lung cancer incidence: a systematic review and meta-analysis. Cancer Causes & Control 2009;20:459–471. [DOI] [PubMed] [Google Scholar]
- 37.Griggs JJ. Disparities in Palliative Care in Patients With Cancer. Journal of Clinical Oncology 2020;38:974–979. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Survey data will be made available upon reasonable request to the corresponding author.


