Abstract
Objective:
Frailty measures vary widely and the optimal measure for predicting HIV-Associated Neurocognitive Disorders (HAND) is unclear.
Design:
A study was conducted to examine the clinical utility of three widely used frailty measures in identifying HIV-associated neurocognitive disorders.
Methods:
The study involved 284 people with HIV (PWH) ≥50 years enrolled at UC San Diego’s HIV Neurobehavioral Research Program. Frailty measurements included the (1) Fried Phenotype, the (2) Rockwood Frailty Index, and the (3) Veterans Aging Cohort Study (VACS) Index. HAND was diagnosed according to Frascati criteria. ANOVAs examined differences in frailty severity across HAND conditions. ROC analyses evaluated sensitivity and specificity of each measure to detect symptomatic HAND [mild neurocognitive disorder (MND) and HIV-associated dementia (HAD)] from no HAND.
Results:
Across all frailty measures, frailty was found to be higher in HAD compared to no HAND. For Fried and Rockwood (not VACS) frailty was significantly more severe in MND vs. no HAND and in HAD vs. ANI (asymptomatic neurocognitive impairment). For discriminating symptomatic HAND from no HAND, Fried was 37% sensitive and 92% specific, Rockwood was 85% sensitive and 43% specific, and VACS was 58% sensitive and 65% specific.
Conclusions:
These findings demonstrate that Fried and Rockwood outperform VACS in predicting HAND. However, ROC analyses suggest none of the indices had adequate predictive validity in detecting HAND. The results indicate that the combined use of the Rockwood and Fried indices may be an appropriate alternative.
Keywords: Fried, Rockwood, VACS, frailty, AIDS, HAND, Cognition
INTRODUCTION
The physical deterioration associated with aging is an inevitable chapter in the human life cycle. The word frail has been used colloquially to describe a deconditioned older adult. Frail is used ubiquitously in both lay and clinical settings, yet, there is not a universal, agreed-upon definition of frailty nor is there consensus as to how to operationalize this condition using specific criteria [1]. One of the major barriers to operationalizing frailty is a contrasting view as to whether this is (1) an independent medical syndrome, or (2) simply a cumulative collection of discrete symptoms associated with aging and age-related chronic illnesses [2]. The first, most commonly characterized by the Fried Phenotype, follows a biological syndrome paradigm, where frailty is viewed as a constellation of symptoms with a unifying pathophysiology that coalesce into a discrete syndrome [3,4]. The latter, most commonly characterized by the Rockwood Frailty Index, follows an accumulation of deficits paradigm, where frailty is considered as a generalized, accelerated aging process, rather than a distinct pathophysiology [5]. In part, due to the lack of consensus on a universal theoretical definition, there are a plethora of frailty measurements to date [6], thereby impeding the incorporation of frailty into research and clinical care by specialists.
Despite these conflicting theoretical and operational definitions of frailty, there is consistent evidence that increased frailty is associated with worse health outcomes, including cognitive impairment, another condition that increases in prevalence with age. In the general population, frailty is reliably associated with poorer cognitive functioning [7,8], and longitudinal studies suggest a bidirectional relationship between frailty severity and cognition [9,10]. The precise physiologic basis for this bidirectional association in older adults appears to be complex and currently poorly understood, but likely include multifactorial mechanisms. These may involve pathways such as those playing a role in mental health, chronic inflammation, endocrine or energy metabolism imbalances, mitochondrial dysfunction, etc [11–13].
In addition to older adults, individuals with chronic illnesses including HIV also exhibit enhanced risk for frailty and cognitive impairment. In people with HIV (PWH), frailty manifests at higher rates at younger ages [14], and approximately 30–50% of PWH demonstrate some form of HIV-Associated Neurocognitive Disorders (HAND) [15]. We previously demonstrated that when defined by the Fried Phenotype (biological syndrome paradigm), even mild levels of frailty (pre-frailty) predicted worse cognitive outcomes in 2 years among PWH [16], and that frailty and HIV-disease severity synergistically increased risk of HAND [17]. Similarly, we showed that frailty related to worse cognition among PWH [18], when defined with the Rockwood Frailty Index (accumulation of deficits paradigm).
In addition to the Fried Phenotype and Rockwood Frailty Index, the Veterans Aging and Cohort Study (VACS) Index has also been used as a marker of frailty. The VACS Index was primarily designed to predict hospitalization and all-cause mortality specifically in PWH [19], however, it contains a number of components that fit the conceptual framework of frailty (i.e., falls, sarcopenia, etc.) [20]. Like the Rockwood Frailty Index, the VACS Index conceptualizes frailty as an accumulation of deficits and contains general indicators of organ system injury (i.e., hemoglobin and platelet counts), but also considers indicators of HIV disease (i.e., CD4 count, HIV-1 RNA). Prior cross-sectional [21], and longitudinal [22], cognitive studies have also demonstrated the higher VACS Index scores relate to poor cognition in PWH.
While frailty is reliably associated with poorer cognition in PWH, a lack of standard frailty measurement tool makes it difficult to make direct comparisons among studies, which may impede on biological discovery, clinical care, and intervention development. Among PWH, we aim to evaluate the three most widely used measurements of frailty and compare their relationships with cognition. This study compares the Fried Phenotype, Rockwood Frailty Index, and VACS Index in their relation to HAND.
METHODS
Participants
Participants were 284 PWH, aged 50 years or older, enrolled in NIH-funded studies at the UC San Diego’s HIV Neurobehavioral Research Program (HNRP) from 2014 to 2020. Data from participants who completed comprehensive neuromedical and neuropsychological evaluations was included for analysis. Exclusion criteria were: 1) the presence of a severe neurological condition known to impact cognitive functioning (e.g., non-HIV neurological disorder such as Parkinson’s Disease or stroke); 2) a history of head injury with loss of consciousness for longer than 30 minutes, seizure disorder, or central nervous system neoplasm; 3) diagnosis of a psychotic disorder; 4) evidence of a severe learning disorder (e.g., WRAT-4 Reading score <70); and 5) a positive urine toxicology for illicit substances, with the exception of marijuana, on the day of testing. Study participants provided written informed consent and the study was approved by the UC San Diego Institutional Review Board.
Frailty Assessments
The three most widely used frailty measurements among PWH were utilized. The Fried Phenotype [3], consists of five symptoms of physical functioning: 1) unintentional weight loss (self-reported ≥10 pounds lost in the past year); 2) weakness (<20th percentile of normative sample for grip strength, stratified by sex and body mass index); 3) exhaustion (self-reported via elevations on items 7 and 9 of the Center for Epidemiologic Studies – Depression [CES-D] scale); 4) slow walking speed (<20th percentile of normative sample for total seconds to walk 15 feet, stratified by sex and height); and 5) low physical activity (<383 kcal/week for men, <270 kcal/week for women; self-reported via the International Physical Activity Questionnaire). We examined the continuous total of deficits, summed across the five Fried Phenotype symptoms (possible range = 0 to 5), to facilitate comparisons with the other continuous frailty indices examined in the present study. The Rockwood Frailty Index was constructed using 34 health factors including general health maintenance (e.g., BMI, creatinine, cholesterol) and comorbidities (e.g., Hepatitis C, diabetes) (Table 2a). Participants are evaluated to determine whether they meet “deficit criteria” for each factor. The Rockwood Frailty Index is scored by calculating summing the total deficits, then dividing by 34 such that the Rockwood Index score represents the proportion of total deficits ranging from 0 (no deficits) to 1 (all 34 deficits). These methods were based on previously published guidelines [16,18,23]. The Veterans Aging Cohort Study (VACS) Index was computed using published methods [24]. The VACS Index is a weighted sum of various health factors, including age, HIV disease, and general health maintenance, such that higher scores represent worse overall health (possible range = 0 to 164). VACS Index components and frequencies are presented in Table 2b.
Table 2a.
Variables and Deficit Criteria for the Rockwood Frailty Index
| Variable | Deficit criteria |
|---|---|
| 1. Unemployed status | Positive |
| 2. Low or high BMI | <18 or >25 kg/m2 |
| 3. Abnormal Potassium | <3.5 or >5.3 mEq/L |
| 4. Abnormal Sodium | <135 or >145 mEq/L |
| 5. Abnormal Creatinine | <0.84 or >1.21 mg/dL |
| 6. Abnormal Albumin | <3.5 or >5.4 g/dL |
| 7. Elevated total Bilirubin | >1.1 mg/dL |
| 8. Elevated AST | >31 U/L |
| 9. Elevated ALT | >31 U/L |
| 10. Abnormal Alkaline Phosphatase | <38 or >126 IU/L |
| 11. High total cholesterol | >200 mg/dL |
| 12. Low HDL cholesterol | <40 mg/dL |
| 13. Elevated LDL | >100 mg/dL |
| 14. Elevated Triglycerides | >150 mg/dL |
| 15. Abnormal WBC | <4,000 cells/μL |
| 16. Low HGB | <13.5 g/dL for males; <12 g/dL for females |
| 17. Low HCT | <38.8 g/dL for males; <34.9 g/dL for females |
| 18. Low Platelets | <150 billion/L |
| 19. HCV | Positive |
| 20. Type 2 Diabetes | Positive |
| 21. COPD | Positive |
| 22. Malignancy | Positive |
| 23. Peptic Ulcer | Positive |
| 24. CVA | Positive |
| 25. MI | Positive |
| 26. Renal disease | Positive |
| 27. Congestive heart failure | Positive |
| 28. Metastatic tumor | Positive |
| 29. Moderate to severe liver disease | Positive |
| 30. Rheumatologic disease | Positive |
| 31. Peripheral vascular disease | Positive |
| 32. Hypertension | Positive |
| 33. Hyperlipidemia* | Positive |
| 34. Lifetime Major Depressive Disorder | Positive |
Variables that were not included in the Rockwood Frailty Index due to missing data >5% include phosphorus, CRP, hemi- or paraplegia, current major depressive disorder, current tobacco use, and history of ever using tobacco.
Determined by one or a combination of sources. These include self-report of previous diagnosis by an outside provider, reporting taking a prescribed medication for the condition, and/or in-laboratory blood test values indicative of the condition.
Table 2b.
Components and frequencies of the Veterans Administration Cohort Study (VACS) Index
| VACS component | Pointsa | Frequencies | ||
|---|---|---|---|---|
|
| ||||
| n | % | |||
| Age (n=282) | <50 | 0 | 0 | 0 |
| 50–64 | 12 | 204 | 72 | |
| ≥65 | 27 | 77 | 27 | |
| CD4 count (cells/mm3; n=280) | ≥500 | 0 | 179 | 64 |
| 200–499 | 6 | 83 | 30 | |
| 100–199 | 10 | 11 | 4 | |
| 50–99 | 28 | 5 | 2 | |
| <50 | 29 | 2 | 1 | |
| HIV-1 RNA (copies/mL; n=267) | <500 | 0 | 247 | 93 |
| 500–1 × 105 | 7 | 17 | 6 | |
| ≥1 × 105 | 14 | 3 | 1 | |
| Hemoglobin (g/dL; n=284) | ≥14 | 0 | 157 | 55 |
| 12–13.9 | 10 | 99 | 35 | |
| 10–11.9 | 22 | 22 | 8 | |
| <10 | 38 | 6 | 2 | |
| FIB-4 (n=284) | <1.45 | 0 | 157 | 56 |
| 1.45–3.25 | 6 | 111 | 40 | |
| >3.25 | 25 | 13 | 5 | |
| eGFR (n=281) | ≥ 60 | 0 | 235 | 84 |
| 45–59.9 | 6 | 32 | 11 | |
| 30–44.9 | 8 | 12 | 4 | |
| <30 | 26 | 2 | 1 | |
| HCV Infection (n=284) | Infected | 5 | 46 | 16 |
Point values that are used to generate a VACS Index score are based on Justice et al. (2012); VACS total scores used for frailty comparisons were based on n=280, sample size for each VACS component score indicated; eGFR=estimated glomerular filtration rate (186.3 Χ [serum creatinine −1.154] Χ [age −0.203] Χ [0.742 for women] Χ [1.21 if black]); FIB-4 = fibrosis index-4 ([years of age Χ aspartate aminotransferase]/[platelets in 109/L square root of alanine aminotransferase]); HCV infection established by a positive antibody test or detectable virus.
HAND classification
Participants completed a comprehensive neurocognitive assessment of seven domains often impacted by HIV [15]: verbal fluency, executive functioning, processing speed, learning, delayed recall, working memory/attention, and motor functioning. Raw test scores were adjusted for practice effects and converted to T-scores that applied normative corrections for demographics (age, education, sex, and race/ethnicity where available). In accordance with Frascati criteria for HAND [25], HAND was classified into three conditions in order of increasing severity: asymptomatic neurocognitive impairment (ANI), mild neurocognitive disorder (MND), and HIV-associated dementia (HAD). ANI and MND require mild to moderate impairments in at least two domains (i.e., >1 SD below the mean) without (ANI) or with (MND) perceived changes in functional ability (i.e., symptomatic). HAD requires moderate to severe impairments in at least two domains (i.e., >2 SD below the mean) with marked functional difficulties.
The HAND assessments were conducted by trained staff members with oversight from clinical neuropsychologists (e.g., DJM). Staff completed a rigorous certification process, and all assessments were double scored by a second trained staff member. Frailty assessments were conducted by different trained staff members with expertise in neuromedical evaluations. These staff include bachelors-level trained staff as well as nurses. For the majority of evaluations, the HAND diagnoses and frailty assessments occurred on the same day.
Psychiatric and Neuromedical Assessment
Current depressive symptoms were assessed with the Beck Depression Inventory-Second Edition (BDI-II). The structured Composite International Diagnostic Interview (CIDI) ascertained DSM-IV diagnoses of current and lifetime Major Depressive Disorder (MDD) and lifetime Substance Use Disorder. Participants underwent a standardized neuromedical evaluation consisting of clinical interview, physical examination, and blood draw. HIV seropositivity was confirmed via ELISA and Western blotting. Plasma HIV RNA was measured by rtPCR (Amplicor, Roche Diagnostics, Indianapolis, IN; lower limit of quantitation [LLQ] of 50 copies/ml). Plasma CD4 counts were measured using flow cytometry. Self-reported HIV disease and treatment variables included nadir CD4, AIDS status, duration of HIV disease, and current ART regimen. Medical comorbidities were assessed via self-report and/or medical chart review when available.
Statistical Analyses
To examine differences in demographic and clinical charateristics by HAND group (no HAND, ANI, MND, and HAD), chi-squared tests were used for categorical variables and analysis of variance (ANOVA) or Kruskal-Wallis tests were used for normally or non-normally distributed continuous variables, respectively. Pearson’s correlations were used to compare relationships among the frailty indices. ANOVA models separately examined HAND group differences on each frailty measurement. Pairwise comparisons were conducted using Tukey’s Honest Significant Difference (HSD) or Wilcoxon Signed-rank tests. Cohen’s d and rank correlations (r) are presented as effect size estimates for pairwise comparisons of parametric (Rockwood Frailty Index) and non-parametric data (Fried Phenotype and VACS Index), respectively. Receiver operating characteristic (ROC) analyses evaluated sensitivity and specificity of each frailty measure to detect symptomatic HAND (MND and HAD), as compared to asymptomatic HAND (ANI) or cognitively normal cases (no HAND). An optimal cut-off score was determined by choosing the cut point with the highest Youden’s Index (i.e., 1-specifcity subtracted from sensitivity) [26].
Role of the funding source
The funding source was not involved in the study design, data collection, analysis, interpretation of the data, the writing of the paper, or the decision to submit the paper for publication.
RESULTS
Sample Characteristics
Study sample characteristics are presented in Table 1. Participants were predominantly male (87%) and non-Hispanic White (62%) with an average age of 59.7 years and formal education of 14.3 years. Most participants exhibited evidence of ART-induced immune reconstitution based on higher current CD4 counts (median=587 cells/mm3) compared to nadir CD4 counts (median=128 cells/mm3) and high rates of current ART use (95%) and viral suppression (89%). Of the 284 participants, 134 (47%) did not meet criteria for HAND, 104 (37%) were classified as ANI, 28 (10%) as MND, and 18 (6%) as HAD. Across HAND groups, demographics were comparable (ps > 0.05), apart from race/ethnicity (χ2 = 19.2, p = 0.024). The group without HAND had the highest proportion of Black/African American participants (25%; ANI = 12%; MND = 11%; HAD = 0%), whereas the group with HAD had the highest proportion of Hispanic/Latinx participants (28%; MND = 25%; ANI = 19%; no HAND = 12%). Proportions of non-Hispanic White participants and those who identified as “Other” were similar across HAND groups. BDI-II score also differed significantly across HAND groups (K-W χ2 = 28.8, p < 0.001), such that those with MND had the highest median BDI-II score (19.0, IQR=15.0–22.0), followed by HAD (16.5, IQR=10.5–22.0), ANI (8.0, IQR=3.0–15.0), and no HAND (7.0, IQR=2.0–14.0). All other clinical characteristics were comparable across HAND groups (ps > 0.05).
Table 1.
Participant characteristics of PWH (n=284)
| Demographics | |
| Age (years), mean (SD) | 59.7 (7.3) |
| Sex (male), N (%) | 247 (87) |
| Race/Ethnicity, N (%) | |
| White | 176 (62) |
| Black/African American | 48 (17) |
| Hispanic/Latinx | 48 (17) |
| Other | 12 (4) |
| Education (years), mean (SD) | 14.3 (2.9) |
| Premorbid verbal IQ estimate, mean (SD)a | 102.4 (14.1) |
|
| |
| Medical Characteristics | |
| Hypertension, N (%) | 166 (58) |
| Hyperlipidemia, N (%) | 172 (61) |
| Diabetes, N (%) | 54 (19) |
| Body mass index, mean (SD) | 26.9 (5.6) |
| Total number of medications, mean (SD) | 4.4 (5.1) |
|
| |
| Psychiatric Characteristics | |
| BDI-II, median [IQR] | 9 [3, 17] |
| Lifetime Substance Use Disorder, N (%) | 187 (67) |
|
| |
| HIV Disease Characteristics | |
| AIDS Diagnosis, N (%) | 189 (67) |
| Estimated years of infection, mean (SD) | 20.5 (9.13) |
| On antiretroviral therapy, N (%) | 268 (95) |
| Nadir CD4 (cells/uL), median [IQR] | 130 [29, 300] |
| Current CD4 (cells/uL), median [IQR] | 589 [418, 798] |
| Plasma HIV RNA (undetectable), N (%) | 238 (89) |
|
| |
| Fried Frailty Phenotype b | |
| Total symptoms, median [IQR] | 1 [0, 3] |
| Frail classification, N (%) | 42 (15) |
| Pre-frail classification, N (%) | 141 (50) |
| Non-frail classification, N (%) | 101 (36) |
| Weight Loss, N (%) | 43 (15) |
| Exhaustion, N (%) | 103 (36) |
| Low Physical Activity, N (%) | 72 (25) |
| Slowness, N (%) | 52 (19) |
| Weakness, N (%) | 71 (25) |
|
| |
| Rockwood Frailty Index | |
| Index score, mean (SD) | .22 (.08) |
|
| |
| VACS Index Score | |
| Index score, median [IQR] | 28 [18, 40] |
|
| |
| Cognition | |
| HAND diagnosis, N (%) | |
| No HAND | 134 (47) |
| ANI | 104 (37) |
| MND | 28 (10) |
| HAD | 18 (6) |
WRAT-4=Wide Range Achievement Test-Fourth Edition
non-frail (0 symptoms), pre-frail (1–2 symptoms), and frail (3 to 5 symptoms); HAND=HIV-associated Neurocognitive Disorders; ANI=Asymptomatic Neurocognitive Impairment; MND=Mild Neurocognitive Disorder; HAD=HIV-associated Dementia
Frailty Characteristics
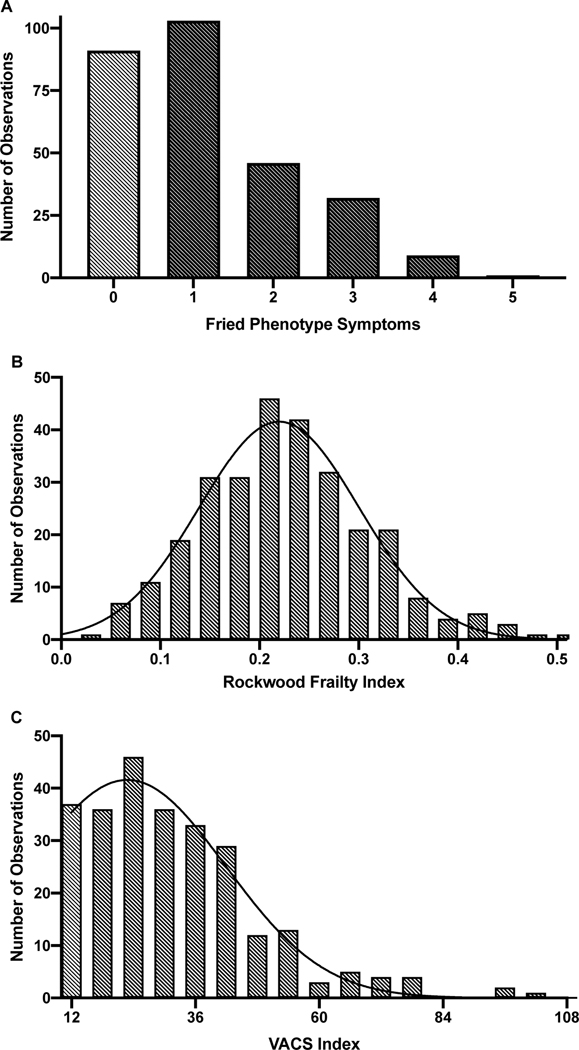
Figure 1 displays distributions for the three frailty indices. On average, participants met criteria for 1.2 (SD=1.3) of the 5 total Fried Phenotype symptoms. Exhaustion was the most common symptom (36%), followed by low physical activity (25%), weakness (25%), slowness (19%), and weight loss (15%). Rockwood Frailty Index scores ranged from .03–.50 (mean=.22, SD=.08) and VACS Index scores ranged from 12–103 (mean=31.2, SD=16.8). The distribution of Rockwood Frailty Index scores (skewness=.40) more closely approximated a normal distribution compared to the positively skewed distribution of the VACS Index (skewness=1.37). The Fried Phenotype was correlated with the Rockwood Frailty Index (r=.23, p<.001) and the VACS Index (r=.19, p=.035); the Rockwood Frailty Index was also correlated with the VACS index (r=.25, p<.001).
Figure 1.
Distribution of frailty observations in HIV sample based on frailty measurement criteria.
HAND Group Differences on Frailty Measures
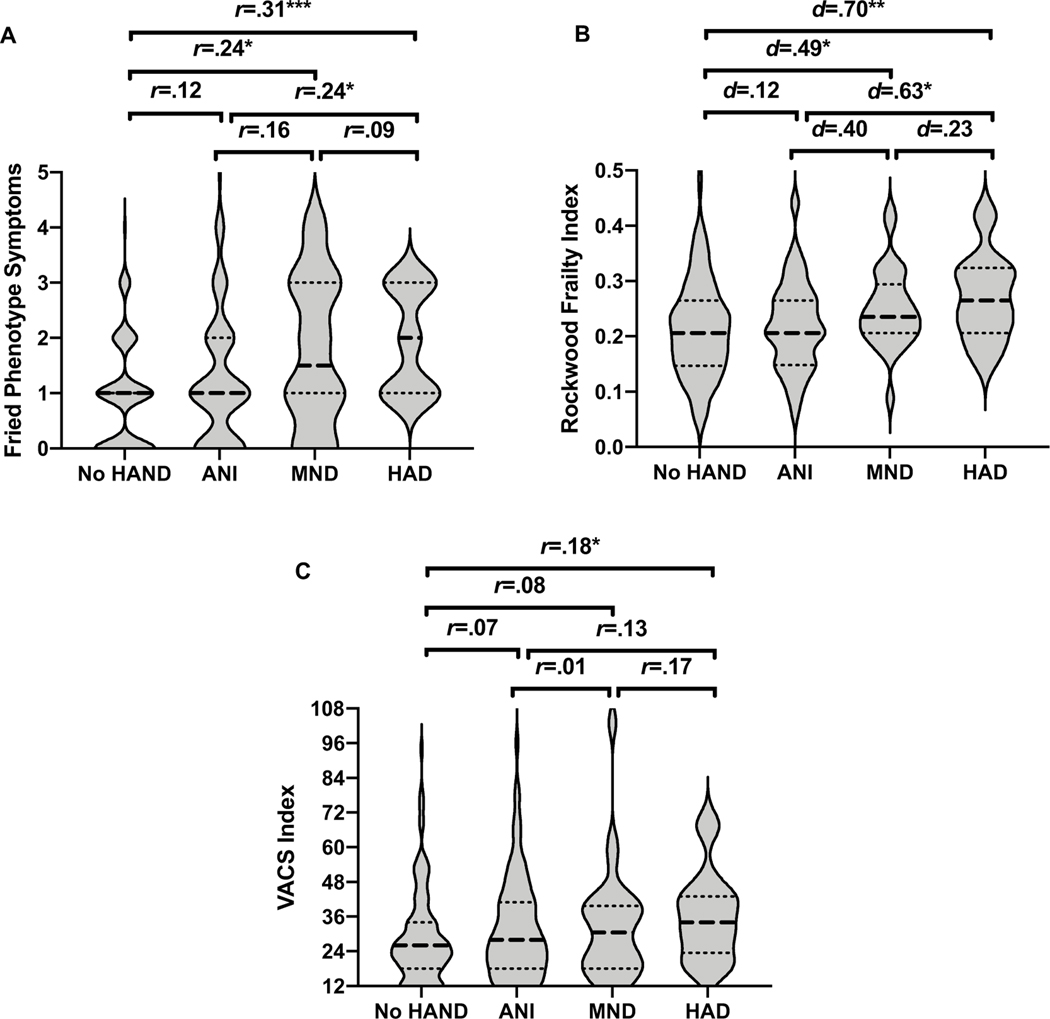
Separate ANOVAs examined HAND group differences on each frailty measure (Figure 2). At the omnibus level, HAND groups significantly differed on Fried Phenotype total symptoms (F3,280 = 5.36, p=.001) and Rockwood Frailty Index scores (F3,280=3.71, p=.012), but not VACS Index scores (F3,280=1.03, p=.38). Pairwise group comparisons indicated significantly higher number of Fried Phenotype symptoms in HAD vs. no HAND (p<.001, r=.31) and vs. ANI (p=.007, r=.24), as well as higher Fried Phenotype symptoms in MND vs. no HAND (p=.003, r=.24). The same pattern of pairwise differences was observed for the Rockwood Frailty Index scores (HAD vs. no HAND: p=.007, d=.70; HAD vs. ANI: p=.02, d=.63; MND vs. no HAND: p=.03, d=.23). Conversely, while VACS Index scores did not differ by HAND status at the omnibus level, non-parametric testing indicated differences between HAD vs. no HAND (p=.04, r=.18).
Figure 2.
Violin plots of frailty measures by HAND condition. Using the Fried Phenotype (panel A) and the Rockwood Frailty Index (panel B), individuals diagnosed with HAD had higher frailty scores than those with no HAND and ANI diagnoses, and those who were diagnosed with MND had higher frailty scores than those with no HAND. Using the VACS Index (panel C), individuals diagnosed with HAD had higher frailty scores than those with no HAND. Violin plots are presented as median frailty symptoms/indices (dashed lines) and quartiles (dotted lines). *p<.05, **p<.01, *** p<.001. Effect sizes are presented as Cohen’s d for parametric data and rank correlation (r) for non-parametric data.
ROC Analyses
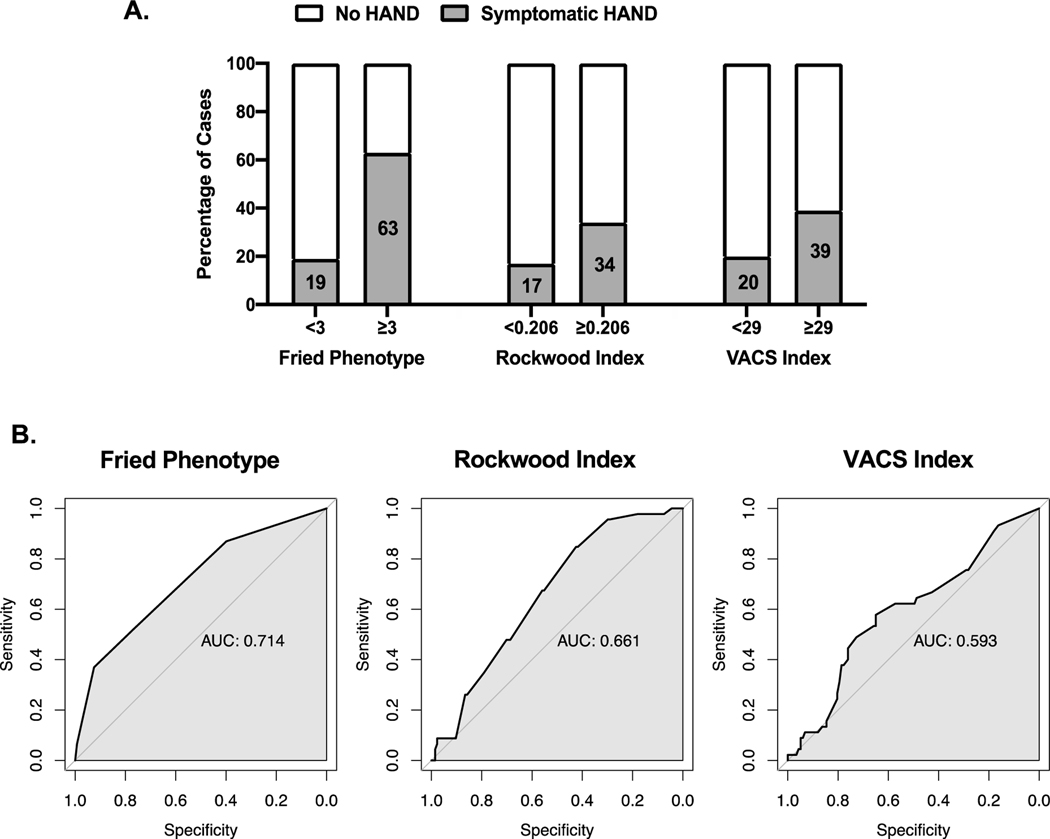
Based on the pairwise associations showing that frailty indices best distinguished symptomatic HAND (i.e., MND and HAD) from those without HAND and asymptomatic HAND (i.e., ANI), separate ROC analyses examined sensitivities and specificities of each measure to detect symptomatic HAND from (1) no HAND and (2) ANI. For discriminating symptomatic HAND from no HAND (see Figure 3), an optimal cutoff of ≥3 was identified for the Fried Phenotype (AUC=.71; Youden’s Index=29) with sensitivity of 37% and specificity of 92%; an optimal cutoff of ≥.206 was identified for the Rockwood Frailty Index (AUC=.66, Youden’s Index=28), with sensitivity of 85% and specificity of 43%; and an optimal cutoff of ≥29 was identified for the VACS Index (AUC=.59, Youden’s Index=23), with sensitivity of 58% and specificity of 65%. To detect symptomatic cases from those with ANI, an optimal cutoff of ≥3 was identified for the Fried Phenotype (AUC=.65; Youden’s Index=22), with sensitivity of 37% and specificity of 85%; an optimal cutoff of ≥.235 was identified for the Rockwood Frailty Index (AUC=.64; Youden’s Index=22), with sensitivity of 67% and specificity of 55%; and an optimal cutoff of ≥34 was identified for the VACS Index (AUC=.54; Youden’s Index=13), with sensitivity of 49% and specificity of 64%.
Figure 3.
Sensitivity and specificity of each frailty measure to detect symptomatic from no HAND cases. A) The Fried Phenotype was 37% sensitive and 92% specific (optimal cutoff ≥3), the Rockwood Frailty index was 85% sensitive and 43% specific (optimal cutoff ≥.206), and the VACS was 58% sensitive and 65% specific (optimal cutoff ≥29). B) The associated area under the curve (AUC) for each frailty measure is also shown. Analyses were also conducted to detect symptomatic from asymptomatic (ANI) cases. A similar pattern of results were observed (results not presented in figure).
Given that the Rockwood Frailty Index had highest sensitivity while the Fried Phenotype had highest specificity, posthoc analyses were conducted to test a stepwise approach using sequential indices for HAND risk identification (symptomatic HAND vs. no HAND). As identified above, the Rockwood Frailty Index cutoff of ≥0.206 correctly identifed 85% of participants with HAND, but produced a high rate of false positives (i.e., 57% of those without HAND had an elevated Rockwood Frailty Index). When subsequently applying the Fried Phenotype only to participants with an elevated Rockwood Frailty Index, the Fried Phenotype cutoff of ≥3 continued to have high specificity (91%) equating to a low false positive rate (i.e., 9% of those without HAND were above the Fried Phenotype cutoff). Although sensitivity of the Fried Phenotype within this subset also continued to be low (44% correctly identified as having HAND), this type of sequential approach could be utilized to identify those with the highest risk for HAND.
DISCUSSION
Recent studies consistently link frailty to HAND and suggest that frailty may be a useful clinical indicator for HAND risk [27]. However, given the lack of standard operationalization of frailty, we sought to understand which frailty measure best relates to HAND for suggested use in both clinical and research settings. Findings suggest the Fried Phenotype and Rockwood Frailty Index outperform the VACS Index relative to the association with HAND conditions, particularly the differentiation of symptomatic HAND (i.e., MND and HAD) from ANI and no HAND. However, subsequent ROC analyses suggested that none of the three frailty indices when used alone had adequate associative validity for identifying symptomatic HAND.
Our finding that the Fried and Rockwood indices have stronger associations with HAND compared to that of the VACS Index is somewhat consistent with previous literature examining these frailty measures as distinct predictors of cognition in HIV. Although the metrics for neurocognition also differ across these studies, recent reports have demonstrated medium-to-large-sized associations between Fried Phenotype and HAND [27], medium-sized associations between Rockwood Frailty Index and continuous neurocognitive T-scores [18], and small-to-medium-sized associations between VACS Index and binary neurocognitive impairment status.[21] Similarly to the current study, Marquine and colleagues [21], found that a VACS score of 18 was 60% sensitive and 50% specific to neurocognitive impairment. The lower VACS cutoff compared to our analysis (18 vs. 29) may relate to methodological differences in classification of neurocognitive status (i.e., global deficit score vs. Frascati criteria) and consideration for functional impairment (i.e., performance-based neurocognitive impairment vs. symptomatic HAND).
To our knowledge, no other study has reported sensitivity and specificity for the Fried Phenotype or Rockwood Index in detecting HAND. Although we may hypothesize better associative validity among these measures, which showed stronger associations with HAND diagnoses, results of the ROC analyses revealed this was not consistent. In fact, optimal cutoff scores were difficult to identify for all three frailty indices, as there were no points at which an adequate balance between sensitivity and specificity could be achieved for any index. Notably, the Fried Phenotype consistently showed the highest specificity and the Rockwood Frailty Index consistently showed the highest sensitivity to detect symptomatic HAND. These complementary differences support previous suggestions to integrate different frailty assessments for better identification of adverse outcomes among PWH (e.g., Guaraldi et al., 2017) [28]. We propose that since the Rockwood Index is overly inclusive, it can be clinically useful as a screening tool (high sensitivity). Components of Rockwood are often readily available in medical records and with an appropriate calculator, can be implemented into patient charts. For patients who score high on the Rockwood, a Fried assessment can be completed by a trained clinician, as our data and posthoc analyses suggest this to be a good exclusionary tool (high specificity). This method of combining frailty assessments may be helpful in detecting those with greatest risk for HAND. When both frailty assessments are not available, a formal neuropsychological assessment is still recommended when cognitive impairment is in question. While a comprehensive evaluation is ideal, an abbreviated battery of tests to screen for HAND in PWH have demonstrated good sensitivity and specificity [29].
Our data suggest that HAND status at each level of frailty (regardless of measurement index) is too heterogeneous and it is inappropriate to use frailty as an stand-alone proxy for HAND in research or clinical practice. For example, up to 20% of participants with low frailty (as identified by our data driven cut-points for each measure) were classified as symptomatic HAND, and up to 66% of participants with high frailty did not have symptomatic HAND. Although physical frailty can certainly play a role in the development of neurobehavioral dysfunction among PWH [16,30], there are also many other factors that contribute to HAND risk in this vulnerable population. These factors include but are not limited to HIV disease characteristics, medical comorbidities, biological factors (e.g., chronic inflammation, microbiome activity), psychiatric factors (e.g., depression), lifestyle factors (e.g., physical activity, sleep, employment), substance use, demographic factors (e.g., educational/occupational attainment, SES), and early life exposures (e.g., childhood trauma) [31–35]. Although the frailty metrics examined in the present study incorporate some of these components, most of these factors were not measured in our study (and in many clinical settings), which may prevent frailty from being an accurate stand-alone predictor of HAND status. Similarly, there may be many unmeasured factors that are protective for individuals with high frailty and unimpaired neurobehavioral status (e.g., positive psychological factors) [36].
Several study limitations and their relevance to future clinical research in this area is worthy of discussion. First, the number of individuals who met criteria for MND and HAD was relatively small, as was the number of individuals with 3 or more Fried Phenotype symptoms (i.e., classically defined as the “frail” phenotype). Next, we did not include a comparison sample of people without HIV; however, our goal was to specifically focus on frailty and neurobehavioral impairment in the context of HIV. Finally, a general limitation to the Rockwood Frailty Index is that its components differ across studies, and thus, our Rockwood Frailty Index is not identical to others reported in the literature. This, however, is often also noted as a clinical strength of the index, as it can be comprised of any age-related variable to which a clinic or research setting may have access, and its psychometric properties remain stable as long as it contains at least 30 factors [23].
Future studies could include the examination of the associative value of the three facility indices when measured across a global score on a continuum rather than using the HAND classifications used here, as associative power may be lost when parsing the cognitive continuum into the commonly used HAND categories. Likewise, it may be of value to examine whether these three frailty indices predict different ranges of cognitive domains, and it may be of interest to include MCI and aMCI designations in addition to those studied here. Another potential future direction of study could be to examine how other assessment tools, such as MoCA (Montreal Cognitive Assessment), relate to frailty.
Conclusions
Our findings suggest that none of the examined frailty indices (i.e., Fried Phenotype, Rockwood Frailty Index, and VACS Index) when used alone were accurate predictors of HAND status. Although our results continue to support statistically significant associations between frailty and HAND, findings do not support adequate predictive validity nor the clinical use of a single frailty measure as a stand-alone proxy for HAND risk. The combined use of the Rockwood and Fried frailty indices may be an appropriate alternative, although future longitudinal studies are needed to confirm this proposal. The current study has important implications for the clinical utility of frailty indices in relation to HAND, which is a notable contribution to growing literature about the link between frailty and neurobehavioral dysfunction among the rapidly growing population of older PWH. Given the high risk of HAND among PWH, more effort should be placed on improving access to and resources for neuropsychological testing, as it appears that even factors strongly associated with HAND (e.g., frailty) do not provide adequate prediction of a patient’s neurobehavioral status.
Acknowledgment
David J. Moore, Ph.D.: Conceived of manuscript and study design, obtained funding for study, contributed to writing, statistical analyses, and interpretation
Ni Sun-Suslow, Ph.D.: Wrote initial draft of manuscript, contributed to statistical analyses and interpretation
Ariadne A. Nichol: Wrote sections of manuscript and provided critical revisions
Emily W. Paolillo, Ph.D.: Assisted with statistical analyses, wrote manuscript sections, and provided critical revisions
Rowan Saloner, PhD: Assisted with statistical analyses, wrote manuscript sections, provided critical revisions, and generated figures
Scott L. Letendre, M.D.: Assisted with study conceptualization, writing and revisions
Jennifer Iudicello, Ph.D.: Assisted with study conceptualization, writing and revisions
Erin E. Morgan, Ph.D.: Assisted with study conceptualization, writing and revisions
This research was supported by the National Institute of Mental Health (grant number R01 MH099987, P30 MH062512), the National Institute on Drug Abuse [grant number T32 DA031098 (stipend support to NS) and F31AG064989 (stipend support to RS)] and the National Institute on Alcohol Abuse and Alcoholism [grant number F31 AA027198 (stipend support to EWP)].
The HIV Neurobehavioral Research Center (HNRC) is supported by Center award P30MH062512 from NIMH.
* The San Diego HIV Neurobehavioral Research Center [HNRC] group is affiliated with the University of California, San Diego, the Naval Hospital, San Diego, and the Veterans Affairs San Diego Healthcare System, and includes: Director: Robert K. Heaton, Ph.D., Co-Director: Igor Grant, M.D.; Associate Directors: J. Hampton Atkinson, M.D., Ronald J. Ellis, M.D., Ph.D., and Scott Letendre, M.D.; Center Manager: Jennifer Iudicello, Ph.D.; Donald Franklin, Jr.; Melanie Sherman; NeuroAssessment Core: Ronald J. Ellis, M.D., Ph.D. (P.I.), Scott Letendre, M.D., Thomas D. Marcotte, Ph.D, Christine Fennema-Notestine, Ph.D., Debra Rosario, M.P.H., Matthew Dawson; NeuroBiology Core: Cristian Achim, M.D., Ph.D. (P.I.), Ana Sanchez, Ph.D., Adam Fields, Ph.D.; NeuroGerm Core: Sara Gianella Weibel, M.D. (P.I.), David M. Smith, M.D., Rob Knight, Ph.D., Scott Peterson, Ph.D.; Developmental Core: Scott Letendre, M.D. (P.I.), J. Allen McCutchan; Participant Accrual and Retention Unit: J. Hampton Atkinson, M.D. (P.I.) Susan Little, M.D., Jennifer Marquie-Beck, M.P.H.; Data Management and Information Systems Unit: Lucila Ohno-Machado, Ph.D. (P.I.), Clint Cushman; Statistics Unit: Ian Abramson, Ph.D. (P.I.), Florin Vaida, Ph.D. (Co-PI),, Anya Umlauf, M.S., Bin Tang, M.S.
The views expressed in this article are those of the authors and do not reflect the official policy or position of the Department of the Navy, Department of Defense, nor the United States Government.
Support:
This work was supported by the National Institute of Mental Health (grant number R01 MH099987, P30 MH062512), the National Institute on Drug Abuse [grant number T32 DA031098 (stipend support to NS) and F31AG064989 (stipend support to RS)] and the National Institute on Alcohol Abuse and Alcoholism [grant number F31 AA027198 (stipend support to EWP)]. The HIV Neurobehavioral Research Center (HNRC) is supported by Center award P30MH062512 from NIMH.
Footnotes
Data Sharing Statement
Data from our study will be available upon request. Due to the ethical requirement to maintain patient confidentiality, individual level data cannot be made available. Like in many places in the world, HIV is highly stigmatized in San Diego County. Age 50 years and older is considered an older adult within people with HIV. Since there is a small population of older persons living with HIV in San Diego County, and we report data on several of these older HIV+ persons, protecting persons living with HIV must remain a top priority. The Data Access Committee who imposed these restrictions and to whom data requests should be made is the HNRP Data Resource Committee. This committee may be contacted via email at hnrpresource@ucsd.edu.
Declaration of Interests
The authors do not have a commercial or other association that might pose a conflict of interest.
REFERENCES
- 1.Xue Q-L. The Frailty Syndrome: Definition and Natural History. Clin Geriatr Med 2011; 27:1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.de Vries NM, Staal JB, van Ravensberg CD, Hobbelen JSM, Olde Rikkert MGM, Nijhuis-van der Sanden MWG. Outcome instruments to measure frailty: A systematic review. Ageing Res Rev 2011; 10:104–114. [DOI] [PubMed] [Google Scholar]
- 3.Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, et al. Frailty in Older Adults: Evidence for a Phenotype. Journals Gerontol Ser A Biol Sci Med Sci 2001; 56:M146–M157. [DOI] [PubMed] [Google Scholar]
- 4.Bergman H, Ferrucci L, Guralnik J, Hogan DB, Hummel S, Karunananthan S, et al. Frailty: An Emerging Research and Clinical Paradigm--Issues and Controversies. Journals Gerontol Ser A Biol Sci Med Sci 2007; 62:731–737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rockwood K, Mitnitski A. Frailty in Relation to the Accumulation of Deficits. Journals Gerontol Ser A Biol Sci Med Sci 2007; 62:722–727. [DOI] [PubMed] [Google Scholar]
- 6.Dent E, Kowal P, Hoogendijk EO. Frailty measurement in research and clinical practice: A review. Eur J Intern Med 2016; 31:3–10. [DOI] [PubMed] [Google Scholar]
- 7.Robinson TL, Gogniat MA, Miller LS. Frailty and Cognitive Function in Older Adults: A Systematic Review and Meta-Analysis of Cross-Sectional Studies. Neuropsychol Rev 2022; 32:274–293. [DOI] [PubMed] [Google Scholar]
- 8.Robertson DA, Savva GM, Kenny RA. Frailty and cognitive impairment—A review of the evidence and causal mechanisms. Ageing Res Rev 2013; 12:840–851. [DOI] [PubMed] [Google Scholar]
- 9.Mitnitski A, Fallah N, Rockwood MRH, Rockwood K. Transitions in cognitive status in relation to frailty in older adults: A Comparison of three frailty measures. J Nutr Health Aging 2011; 15:863–867. [DOI] [PubMed] [Google Scholar]
- 10.Buchman AS, Boyle PA, Wilson RS, Tang Y, Bennett DA. Frailty is Associated With Incident Alzheimer’s Disease and Cognitive Decline in the Elderly. Psychosom Med 2007; 69:483–489. [DOI] [PubMed] [Google Scholar]
- 11.de Morais Fabrício D, Chagas MHN, Diniz BS. Frailty and cognitive decline. Transl Res 2020; 221:58–64. [DOI] [PubMed] [Google Scholar]
- 12.Li C, Ge S, Yin Y, Tian C, Mei Y, Han P. Frailty is associated with worse cognitive functioning in older adults. Front Psychiatry 2023; 14:1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ma L, Chan P. Understanding the Physiological Links Between Physical Frailty and Cognitive Decline. Aging Dis 2020; 11:405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Desquilbet L, Jacobson LP, Fried LP, Phair JP, Jamieson BD, Holloway M, et al. HIV-1 Infection Is Associated With an Earlier Occurrence of a Phenotype Related to Frailty. Journals Gerontol Ser A Biol Sci Med Sci 2007; 62:1279–1286. [DOI] [PubMed] [Google Scholar]
- 15.Heaton RK, Clifford DB, Franklin DR, Woods SP, Ake C, Vaida F, et al. HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy: CHARTER Study. Neurology 2010; 75:2087–2096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Paolillo EW, Sun-Suslow N, Pasipanodya EC, Morgan EE, Ellis RJ, Jeste DV., et al. Pre-frailty predicts cognitive decline at 2-year follow-up in persons living with HIV. J Neurovirol 2020; 26:168–180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sun-Suslow N, Paolillo EW, Morgan EE, Letendre S, Iudicello J, Moore DJ. Frailty and HIV Disease Severity Synergistically Increase Risk of HIV-Associated Neurocognitive Disorders. J Acquir Immune Defic Syndr 2020; 84:522–526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Oppenheim H, Paolillo EW, Moore RC, Ellis RJ, Letendre SL, Jeste DV., et al. Neurocognitive functioning predicts frailty index in HIV. Neurology 2018; 91:e162–e170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Justice AC. HIV and Aging: Time for a New Paradigm. Curr HIV/AIDS Rep 2010; 7:69–76. [DOI] [PubMed] [Google Scholar]
- 20.Escota G V, Patel P, Brooks JT, Bush T, Conley L, Baker J, et al. The Veterans Aging Cohort Study Index Is an Effective Tool to Assess Baseline Frailty Status in a Contemporary Cohort of HIV-Infected Persons. AIDS Res Hum Retroviruses 2015; 31:313–317. [DOI] [PubMed] [Google Scholar]
- 21.Marquine MJ, Umlauf A, Rooney AS, Fazeli PL, Gouaux BD, Paul Woods S, et al. The Veterans Aging Cohort Study Index is Associated With Concurrent Risk for Neurocognitive Impairment. JAIDS J Acquir Immune Defic Syndr 2014; 65:190–197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Marquine MJ, Montoya JL, Umlauf A, Fazeli PL, Gouaux B, Heaton RK, et al. The Veterans Aging Cohort Study (VACS) Index and Neurocognitive Change: A Longitudinal Study. Clin Infect Dis 2016; 63:694–702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Searle SD, Mitnitski A, Gahbauer EA, Gill TM, Rockwood K. A standard procedure for creating a frailty index. BMC Geriatr 2008; 8:24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Justice AC, Freiberg MS, Tracy R, Kuller L, Tate JP, Goetz MB, et al. Does an index composed of clinical data reflect effects of inflammation, coagulation, and monocyte activation on mortality among those aging with HIV? Clin Infect Dis 2012; 54:984–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Antinori A, Arendt G, Becker JT, Brew BJ, Byrd DA, Cherner M, et al. Updated research nosology for HIV-associated neurocognitive disorders. Neurology 2007; 69:1789–1799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fluss R, Faraggi D, Reiser B. Estimation of the Youden Index and its Associated Cutoff Point. Biometrical J 2005; 47:458–472. [DOI] [PubMed] [Google Scholar]
- 27.Zamudio-Rodríguez A, Belaunzarán-Zamudio PF, Sierra-Madero JG, Cuellar-Rodríguez J, Crabtree-Ramírez BE, Alcala-Zermeno JL, et al. Association Between Frailty and HIV-Associated Neurodegenerative Disorders Among Older Adults Living with HIV. AIDS Res Hum Retroviruses 2018; 34:449–455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Guaraldi G, Malagoli A, Theou O, Brothers T, Wallace L, Torelli R, et al. Correlates of frailty phenotype and frailty index and their associations with clinical outcomes. HIV Med 2017; 18:764–771. [DOI] [PubMed] [Google Scholar]
- 29.Moore DJ, Roediger MJP, Eberly LE, Blackstone K, Hale B, Weintrob A, et al. Identification of an Abbreviated Test Battery for Detection of HIV-Associated Neurocognitive Impairment in an Early-Managed HIV-Infected Cohort. PLoS One 2012; 7:e47310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Erlandson KM, Perez J, Abdo M, Robertson K, Ellis RJ, Koletar SL, et al. Frailty, Neurocognitive Impairment, or Both in Predicting Poor Health Outcomes Among Adults Living With Human Immunodeficiency Virus. Clin Infect Dis 2019; 68:131–138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Heaton RK, Franklin DR, Ellis RJ, McCutchan JA, Letendre SL, LeBlanc S, et al. HIV-associated neurocognitive disorders before and during the era of combination antiretroviral therapy: differences in rates, nature, and predictors. J Neurovirol 2011; 17:3–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Heaton RK, Franklin DR, Deutsch R, Letendre S, Ellis RJ, Casaletto K, et al. Neurocognitive Change in the Era of HIV Combination Antiretroviral Therapy: The Longitudinal CHARTER Study. Clin Infect Dis 2015; 60:473–480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Valcour VG. HIV, aging, and cognition: emerging issues. Top Antivir Med 2013; 21:119–23. [PMC free article] [PubMed] [Google Scholar]
- 34.Fazeli PL, Marquine MJ, Dufour C, Henry BL, Montoya J, Gouaux B, et al. Physical Activity is Associated with Better Neurocognitive and Everyday Functioning Among Older Adults with HIV Disease. AIDS Behav 2015; 19:1470–1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Spies G, Fennema-Notestine C, Cherner M, Seedat S. Changes in cognitive function in women with HIV infection and early life stress. AIDS Care 2017; 29:14–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Moore DJ, Fazeli PL, Moore RC, Woods SP, Letendre SL, Jeste DV., et al. Positive Psychological Factors are Linked to Successful Cognitive Aging Among Older Persons Living with HIV/AIDS. AIDS Behav 2018; 22:1551–1561. [DOI] [PMC free article] [PubMed] [Google Scholar]