Abstract
Background:
The primary barrier to curing HIV infection is the pool of intact HIV proviruses integrated into host cell DNA throughout the bodies of people living with HIV (PLHIV), called the HIV reservoir. Reservoir size is impacted by the duration of HIV infection, delay in starting antiretroviral therapy (ART), and breakthrough viremia during ART. The leading infectious cause of death worldwide for PLHIV is TB, but we don’t know how TB impacts the HIV reservoir.
Methods:
We designed a case-control study to compare HIV provirus-containing CD4 in PLHIV with vs. without a history of active TB disease. Study participants in the pilot and confirmatory cohort were enrolled at GHESKIO Centers in Port au Prince, Haiti. Intact and non-intact proviral DNA were quantified using droplet digital PCR of PBMC-derived CD4 cells. For a subset, Th1 and Th2 cytokines were assayed in plasma. Kruskal-Wallis tests were used to compare medians with tobit regression for censoring.
Results:
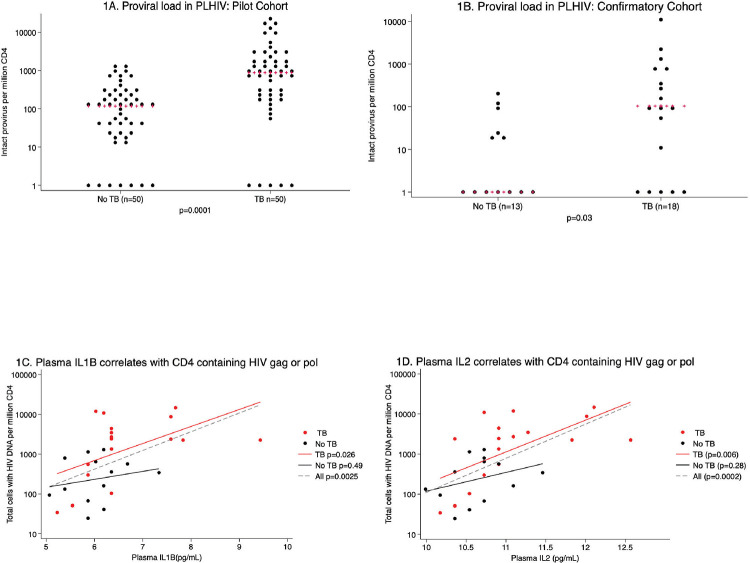
In the pilot cohort, we found that PLHIV with history of active pulmonary TB (n=20) had higher intact provirus than PLHIV without history of active TB (n=47) (794 vs 117 copies per million CD4, respectively; p<0.0001). In the confirmatory cohort, the quantity of intact provirus was higher in the TB group (n=13) compared with the non-TB group (n=18) (median 102 vs. 0 intact provirus per million CD4, respectively p=0.03). Additionally, we found that the frequencies of CD4+ T cells with any detectable proviral fragment was directly proportional to the levels of IL1B (p= 0.0025) and IL2 (p=0.0002).
Conclusions.
This is the first assessment of HIV provirus using IPDA in a clinical cohort from a resource limited setting, and the finding of larger reservoir in PLHIV with history of TB has significant implications for our understanding of TB-HIV coinfection and HIV cure efforts in TB-endemic settings.
The primary barrier to curing HIV infection is the pool of intact HIV proviruses integrated into host cell DNA throughout the bodies of people living with HIV (PLHIV), called the HIV reservoir. Reservoir size is impacted by the duration of HIV infection, delay in starting antiretroviral therapy (ART), and breakthrough viremia during ART. Increased reservoir size has been associated with worse clinical outcomes, including neurocognitive impairment (1). The leading infectious cause of death worldwide for PLHIV is TB, but we don’t know how TB impacts the HIV reservoir. An approximation of reservoir size can be attained by quantitative PCR of HIV DNA in peripheral blood mononuclear cells (PBMC). One group used this approach to study HIV DNA levels in PBMCs from PLHIV with and without active pulmonary TB in Uganda and did not see a statistically significant difference (2). However, in a cohort in China, PLHIV with history of TB had a higher level of circulating HIV DNA than people without TB, but they also had higher viral load pre-ART (3). People with TB-HIV coinfection may have increased risk of virological failure and breakthrough viremias, resulting from or contributing to greater quantities of HIV DNA in circulating CD4+ T cells.
The emerging gold standard for the measurement of HIV reservoirs is the intact proviral DNA asay (IPDA), which distinguishes intact from defective proviruses, the latter being much more numerous (4). We applied a modified IPDA (5) to a cohort of people with HIV in a resource-limited setting with a TB syndemic to determine how quantities of CD4+ T cells containing HIV provirus vary in people who had had active bacteriologically confirmed pulmonary TB disease concurrent with or after diagnosis of HIV.
Using IPDA (see supplemental methods), we measured proviral loads for 100 PLHIV participating in a TB study at GHESKIO Centers in Port au Prince, Haiti, 50 people with active or history of bacteriologically confirmed pulmonary TB and 50 people with no history of TB (Supplemental Table 1). Within the total cohort, there was a nearly 8-fold difference in intact provirus levels between current or history of TB vs. no TB (median 881 vs 116 copies per million CD4+ T cells, respectively; p=0.0001) (Figure 1A). Tobit regression of detectable intact provirus also showed statistically significant difference in intact provirus (p < 0.0001) between the TB and non-TB groups. There was no statistically significant difference in intact provirus between people with newly diagnosed untreated TB (n=25) vs. history of TB or TB currently being treated (n=25) (median 897 vs. 865 copies per million CD4+ T cells, respectively; p=0.92; Supplemental Figure 1A). In the people with no history of TB, the amount of intact provirus in CD4+ T cells was not statistically significantly different by IGRA status (median 80 for IGRA-positive vs.118 for IGRA-negative per million CD4, p=0.65; Supplemental Figure 1B).
Figure 1.
There was approximately 8-fold higher circulating CD4 T cells with intact HIV provirus in PLHIV with TB or TB history compared with no history of TB (p=0.0001, 1A). This was also found in a confirmatory cohort (p=0.03, 1B). In the confirmatory cohort, plasma IL1B (1C) and IL2 (1D) levels correlated with circulating CD4 T cells containing HIV gag and/or pol (p=0.0025 and p=0.0002, respectively)
To remove potential confounders of the difference in intact provirus observed in the initial groups, we excluded 26 people with HIV diagnosed less than one year before sample collection. We excluded 3 people with no plasma available for p24 testing and 3 people with detectable plasma p24. One person who had TB prior to HIV diagnosis was also excluded. After these adjustments, the difference in intact proviral load between TB and no-TB groups remained statistically significant (median 794 vs 117 copies per million CD4, respectively; p=0.0001) by Kruskal-Wallis test as well as by tobit regression (p<0.0001). This difference remained statistically significant when analyzing women and men independently (p< 0.005).
For the confirmatory study, we began with 34 participants from a study of TB recurrence. One participant’s PBMC did not have enough live cells after thawing to have CD4+ T cells selected and DNA extracted so was excluded from downstream analyses. One DNA sample failed QC with no detectable intact, env fragment, or psi fragment with all tested primer sets and so was excluded. One person had HIV viral load > 1,000 copies/mL as well as detectable p24 after dissociation. This person was excluded as the high-level viremia could have rendered the IPDA quantitation inaccurate. In the final analysis group from the confirmatory cohort (Supplemental Table 2), the quantity of intact provirus was higher in the TB group compared with the non-TB group (median 102 vs. 0 intact provirus per million CD4, respectively p=0.03) (Figure 1B). We also analyzed the intact provirus levels using a tobit regression model with 12 left-censored observations for intact proviral loads less than the lower limit of detection and the difference remained statistically significant (p=0.04). In the participants with no history of TB, there wasn’t a statistically significant difference in intact proviral load with IGRA positivity (median 0 vs 9 intact provirus per million CD4, p=0.96; Supplemental Figure 2A). In participants with history of one or recurrent episodes of TB, there was no statistically significant difference in intact proviral load in CD4+ T cells (median 103 vs 103 intact provirus per million CD4, p=0.55; Supplemental Figure 2B).
Additionally, we found that the frequencies of CD4+ T cells with any detectable proviral fragment was directly proportional to the levels of IL1B (0.0025, Figure 1C), IL2 (p=0.0002, Figure 1D), IL12p70 (p=0.0093), and IL13 (p=0.0067). After Bonferroni correction for the 8 analyzable cytokines, (cutoff padj=0.05/8=0.006), the association between plasma IL-1B and IL2 and CD4 containing HIV gag or pol remained statistically significant. The difference in IL1B and IL2 levels between the TB and no-TB groups was not statistically significant (p=0.08 and p=0.1, respectively), but by observation of the fit lines for the cytokine levels by proviral load, the levels were overall higher for the TB group than for the non-TB group (Figure 1C and 1D).
In our pilot and confirmatory cohorts, we documented significantly higher levels of intact HIV proviruses in circulating CD4+ T cells of PLHIV with histories of bacteriologically confirmed pulmonary TB. Levels of HIV DNA correlated directly with levels of the pro-inflammatory cytokines IL1B and IL2 with a trend to higher levels in people with a history of TB. Strengths of the study include the use of IPDA to discriminate intact versus defective HIV proviruses, translational approach using samples from people who have experienced the true natural history of HIV-TB coinfection, and confirmatory cohort testing. Limitations include incomplete CD4+ T cell count nadir data because of the changing landscape of CD4 testing in HIV care, inclusion of only bacteriologically confirmed pulmonary TB, and HIV proviral load testing at only a single time point. This is the first assessment of HIV provirus using IPDA in a clinical cohort from a resource limited setting, and the finding of 8-fold larger reservoir in PLHIV with history of TB has significant implications for our understanding of TB-HIV coinfection and HIV cure efforts in TB-endemic settings.
PLHIV with TB exhibit systemic inflammation and immune activation. Local inflammation has the propensity to increase HIV replication and viral load at sites where HIV and replicating Mtb exist, even prior to the development of symptomatic TB (6). Mtb cell wall components are able to activate TLR signaling to induce HIV replication without live bacteria and may induce HIV transcription in neighboring cells that are uninfected with Mtb (6). These factors may contribute to the larger HIV reservoir in PLHIV with history of pulmonary TB, and should make TB a serious consideration for all reservoir-targeting HIV cure endeavors.
Supplementary Material
2. Financial Contributions
The authors recognize funding to KD (NIH K23-AI131913 and R01-AI176943, Doris Duke Charitable Foundation Clinical Scientist Development Award) and DWF (NIH U19-AI111143, P30-AI168433, and K24-AI098627). Additionally, this project utilized databases maintained in REDCap, supported by UL1-TR002385 from the National Center for Advancing Translational Sciences of the NIH. We acknowledge the patient care, clinical research, and laboratory teams at GHESKIO Centers in Port au Prince, Haiti.
Footnotes
Conflict of interest statement:
The authors have declared that no conflict of interest exists.
REFERENCES
- 1.de Oliveira MF, Murrell B, Perez-Santiago J, Vargas M, Ellis RJ, Letendre S, et al. Circulating HIV DNA Correlates With Neurocognitive Impairment in Older HIV-infected Adults on Suppressive ART. Sci Rep. 2015;5:17094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Olson A, Ragan EJ, Nakiyingi L, Lin N, Jacobson KR, Ellner JJ, et al. Brief Report: Pulmonary Tuberculosis Is Associated With Persistent Systemic Inflammation and Decreased HIV-1 Reservoir Markers in Coinfected Ugandans. J Acquir Immune Defic Syndr. 2018;79(3):407–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Xun J, Qi T, Zou L, Tang Q, Shen Y, Yang J, et al. Mycobacterium tuberculosis coinfection is associated with increased surrogate marker of the HIV reservoir. AIDS Res Ther. 2020;17(1):63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bruner KM, Wang Z, Simonetti FR, Bender AM, Kwon KJ, Sengupta S, et al. A quantitative approach for measuring the reservoir of latent HIV-1 proviruses. Nature. 2019;566(7742):120–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kinloch NN, Ren Y, Conce Alberto WD, Dong W, Khadka P, Huang SH, et al. HIV-1 diversity considerations in the application of the Intact Proviral DNA Assay (IPDA). Nat Commun. 2021;12(1):165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Waters R, Ndengane M, Abrahams M, Diedrich C, Wilkinson R, Coussens A. The Mtb-HIV syndemic interaction: why treating M. tuberculosis infection may be crucial for HIV-1 eradication. Future Virology. 2020;15(2):101–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.